Abstract
The sedimentation behavior of transmissible gastroenteritis coronavirus (TGEV) was analyzed. Upon sucrose gradient centrifugation, the major virus band was found at a density of 1.20 to 1.22 g/cm3. This high density was observed only when TGEV with a functional sialic acid binding activity was analyzed. Mutants of TGEV that lacked sialic acid binding activity due to a point mutation in the sialic acid binding site of the S protein were mainly recovered at a lower-density position on the sucrose gradient (1.18 to 1.19 g/cm3). Neuraminidase treatment of purified virions resulted in a shift of the sedimentation value from the higher to the lower density. These results suggest that binding of sialoglycoproteins to the virion surface is responsible for the sedimentation behavior of TGEV. When purified virions were treated with octylglucoside to solubilize viral glycoproteins, ultracentrifugation resulted in sedimentation of the S protein of TGEV. However, when neuraminidase-treated virions or mutants with a defective sialic acid binding activity were analyzed, the S protein remained in the supernatant rather than in the pellet fraction. These results indicate that the interaction of the surface protein S with sialoglycoconjugates is maintained after solubilization of this viral glycoprotein by detergent treatment.
Transmissible gastroenteritis coronavirus (TGEV) is a prototype enteropathogenic coronavirus that causes diarrhea in pigs of all ages. While adult animals usually recover, newborn piglets generally die from the intestinal infection (14). TGEV is an enveloped virus with three proteins inserted into the viral membrane: S (220 kDa), M (29 to 36 kDa), and E (10 kDa), a minor protein. The S protein plays a key role in the initial stage of infection. It mediates binding of the virus to the cell surface and the subsequent fusion between the viral and cellular membranes. Two binding activities have been assigned to the S protein. Binding to porcine aminopeptidase N, a cellular receptor for TGEV, is a prerequisite for infection of cells (5). A second binding activity enables TGEV to recognize sialic acid residues and attach to sialoglycoconjugates (18). As a consequence of the latter binding activity, TGEV can agglutinate erythrocytes (11, 12). The binding site for aminopeptidase N and the binding site for sialic acid are located on different portions of the S protein (18). Recent studies with mutants of TGEV indicated that a short stretch of amino acids (145 to 209) is important for the recognition of sialic acids (8). Some of the mutants had been selected for resistance to a monoclonal antibody. Interestingly, the point mutations that were responsible for the lack of antibody reactivity also resulted in the concomitant loss of both hemagglutinating activity and enteropathogenicity (8). These results indicated not only that the respective amino acids are located at or close to the sialic acid binding site but also that the sialic acid binding activity is correlated with the enteropathogenicity of TGEV. Other factors may also be required to render TGEV enteropathogenic, but they have not been identified in terms of a molecular interaction. The importance of the sialic acid binding activity for enteropathogenicity is supported by data reported for porcine respiratory coronavirus (PRCoV), which is closely related to TGEV. This virus replicates with high efficiency in the respiratory tract but with very low efficiency in the gut (4). Like the mutants mentioned above, PRCoV has no hemagglutinating activity (18). In the case of PRCoV, the lack of a sialic acid binding activity is explained by a large deletion in the S gene that results in a truncated spike protein (15, 16). The point mutations that resulted in the loss of hemagglutinating activity and enteropathogenicity are located in that portion of the S protein that is present in the TGEV S protein but absent from the PRCoV S protein.
Here we report that the ability of TGEV to attach to sialoglycoconjugates affects the physical state of virus particles. TGEV with a functional sialic acid binding activity was recovered from sucrose gradients at higher densities than PRCoV or mutants of TGEV with a defect in the sialic acid binding site. The difference in sedimentation behavior was also observed with solubilized S protein. It was abolished after neuraminidase treatment of virions, suggesting that bound sialoglycoproteins are responsible for the different sedimentation characteristics.
(Part of this work was done by C.K. in partial fullfilment of the requirements for the Dr. rerum physiologicarum degree at Philipps-Universität Marburg, Marburg, Germany.)
MATERIALS AND METHODS
Virus.
The Purdue strain of TGEV (PUR46-MAD) was used throughout this study. Stock virus was propagated in swine testicular (ST) cells. After incubation for 20 to 24 h at 37°C, the supernatant was harvested, clarified by low-speed centrifugation, and after addition of 1% fetal calf serum (FCS), stored at −80°C.
Cells.
ST cells were grown in Dulbecco's modified Eagle medium supplemented with 10% FCS.
Sucrose gradient centrifugation of TGEV and TGEV mutants.
Confluent monolayers of ST cells were infected with stock virus (PUR46 or HAD mutants) at a multiplicity of infection of 0.1. After incubation for 2 days at 37°C in medium without FCS, the supernatant was clarified by centrifugation for 10 min at 1,400 × g. Virus was sedimented by ultracentrifugation at 140,000 × g for 1 h. The virus pellet was resuspended in phosphate-buffered saline (PBS) and—if indicated—treated with neuraminidase as described below. For purification, the virus was layered onto a 20 to 60% (wt/wt) sucrose gradient (in PBS) and centrifuged for 4 h at 150,000 × g. In some experiments, the gradient was harvested from the bottom of the tube and 10 fractions were collected. The density of the sucrose in each fraction was determined using a refractometer. Each fraction was also analyzed for hemagglutinating activity and for the presence of viral proteins by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). In other experiments, virus bands were harvested and, following dilution with PBS, were sedimented by centrifugation for 1 h at 150,000 × g. Purified virus was analyzed by SDS-PAGE or stored at −20°C.
Sedimentation of virions.
TGEV and the HAD7 mutant were grown in ST cells. After clarification by low-speed centrifugation, aliquots of the supernatant were centrifuged for 1 h at 0 (control), 25,000, 50,000, 75,000, or 100,000 × g. In order to pellet virions that had not been sedimented in the first centrifugation step, the supernatants were subjected to centrifugation for 1 h at 150,000 × g. The pellets of each sample were analyzed by SDS-PAGE. The presence of virus was determined by Western blotting using a monoclonal antibody (6A.C3) directed to the S protein (3).
Neuraminidase treatment of virus.
Virus that had been sedimented was resuspended in 200 μl of PBS per tube and treated with 50 mU of Vibrio cholerae neuraminidase per ml for 30 min at 37°C. Treated virus was purified by sucrose gradient centrifugation as described above.
Octylglucoside treatment of virions.
Purified virions were treated with octylglucoside at a final concentration of 1 or 5%. The sample was incubated for 1 h on ice. For sedimentation of the nucleocapsid, the lysate was centrifuged in a TLA 45 rotor for 1 h at 100,000 × g at 4°C. The supernatants and the pellet fractions were analyzed for the presence of the S protein by Western blotting using a monoclonal antibody as described above.
Electron microscopy.
For negative staining, samples were applied to copper grids, stained with phosphotungstate, and examined with a Zeiss electron microscope 110.
Western blotting.
Viral proteins were separated by SDS-PAGE under nonreducing conditions and blotted to nitrocellulose using a semidry Western blot method, as described by Schultze et al. (17). After blocking of nonspecific binding sites by incubation with 10% nonfat dry milk in PBS, the immobilized TGEV S protein was incubated with different monoclonal antibodies. Bound antibodies were detected with biotinylated sheep anti-mouse immunoglobulin G (Amersham) and a streptavidin-biotinylated horseradish peroxidase complex (Amersham).
For detection of proteins in polyacrylamide gels, a silver stain (Bio-Rad) was used according to the instructions of the manufacturer.
RESULTS
Sucrose gradient centrifugation of TGEV.
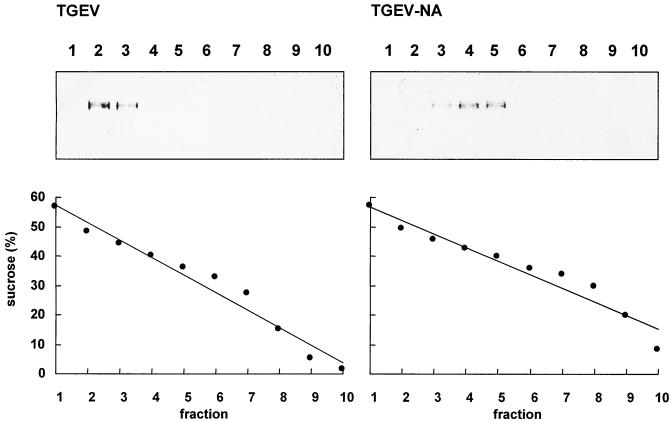
The sialic acid binding activity of TGEV may result in the binding of sialoglycoconjugates to the viral surface. As glyoproteins usually contain more than one oligosaccharide, they have the potential to establish a multivalent interaction with two or more S proteins. Therefore, a single sialoglycoconjugate may even attach to two virions. We wondered whether such interactions affect the sedimentation behavior of TGEV and analyzed a purified virus preparation by sucrose gradient centrifugation. As shown in Fig. 1, the S protein was mainly found in fractions 2 and 3 (upper left panel), corresponding to sucrose concentrations of about 45 to 49% (lower left panel) or a density of 1.20 to 1.22 g/cm3. When the sample was treated with sialidase prior to ultracentrifugation to remove bound sialoglycoconjugates from the virion surface, the majority of the virus particles were found in fractions 4 and 5 (upper right panel), corresponding to sucrose concentrations of about 40 to 43% (lower right panel) or a density of 1.18 to 1.19 g/cm3. This value is close to the value determined for coronaviruses (1.15 to 1.18 g/cm3) by equilibrium sedimentation (2). This result indicates that sialic acid binding activity can affect the sedimentation behavior of TGEV.
FIG. 1.
Sucrose gradient centrifugation of untreated (TGEV) and neuraminidase-treated (TGEV-NA) TGEV. The gradient was fractionated from the bottom. Aliquots of each fraction were used to measure the sucrose concentration (lower panels) and to analyze by SDS-PAGE for the presence of virus (upper panels). The bands were stained with Coomassie brilliant blue. Only the portion of the gel containing the S protein is shown.
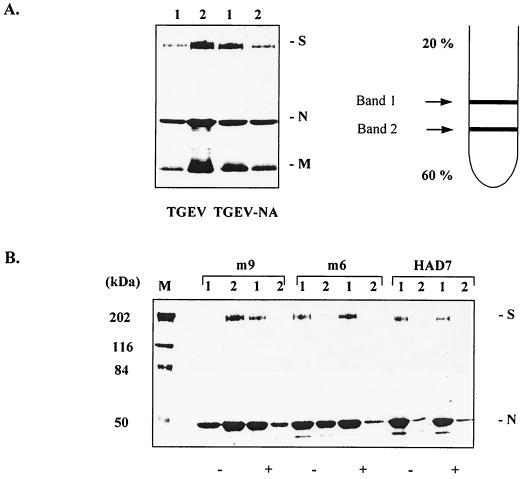
Upon ultracentrifugation of TGEV through sucrose gradients, we usually observed two bands. In light of the above results, these bands may represent populations of virions, the sedimentation of which has been affected or unaffected by sialic acid binding activity. For clarification, untreated and neuraminidase-treated preparations of TGEV were centrifuged through a 20 to 60% sucrose gradient, and the upper and lower bands were harvested and analyzed by SDS-PAGE. As shown in Fig. 2A, the majority of virions in the untreated fraction are present in the lower band (designated 2). This is especially evident in the S protein. Neuraminidase-treated virions are mainly present in the upper band (designated 1). To demonstrate that the positions of the two bands are actually determined by sialic acid binding activity, we also analyzed mutants of TGEV (Fig. 2B). Mutant HAD7 had been selected for its inability to bind to erythrocytes and was shown to lack sialic acid binding activity (9). Virions of this mutant were mainly found in the upper band irrespective of neuraminidase treatment. The same picture was observed with mutant m6. This mutant had been selected for its resistance to a monoclonal antibody (1) and was shown to lack hemagglutinating activity (8). Mutant m9 was selected for resistance to the same monoclonal antibody but has retained its hemagglutinating activity (1, 8). This mutant resembled the parental TGEV; i.e., untreated virions were mainly present in the lower band, whereas neuraminidase-treated virions were mainly detected in the upper band.
FIG. 2.
Sucrose gradient centrifugation of untreated and neuraminidase-treated virions. The upper right panel shows a schematic drawing of the two bands harvested. The upper left panel shows the S, N, and M protein profiles of the upper (1) and lower (2) bands of untreated (TGEV) and neuraminidase-treated (TGEV-NA) TGEV. The lower panel shows the S and N protein profiles of the upper (1) and lower (2) bands of the m9, m6, and HAD7 mutants. Lanes representing untreated virions (−) and neuraminidase-treated virions (+) are indicated. The left lane (M) shows molecular mass markers. The proteins were stained with Coomassie brilliant blue.
Sedimentation of TGEV.
In order to find out whether the difference between hemagglutinating and nonhemagglutinating TGEV is detectable only at the high centrifugation forces applied during sucrose gradient centrifugation, we analyzed the sedimentation behavior under different conditions. Aliquots of the parental TGEV and the HAD7 mutant were sedimented at 25,000, 50,000, 75,000, or 100,000 × g. The pellets were each resuspended in PBS, and both pellet and supernatant fractions were sedimented at 150,000 × g. The pellets were subjected to SDS-PAGE and probed with a monoclonal antibody for the presence of S protein. As shown in Fig. 3, at a centrifugal force as low as 25,000 × g the majority of the virions (PUR46) were found in the pellet fraction; no and only trace amounts of S protein were detectable in the supernatant after centrifugation at 75,000 and 100,000 × g, respectively. With the HAD7 mutants, on the other hand, even after centrifugation at 100,000 × g about half of the virions were still present in the supernatant fraction. This result confirms that the presence or absence of a functional sialic acid binding activity in TGEV may affect the sedimentation behavior of virions.
FIG. 3.
Sedimentation of TGEV and the HAD7 mutant. Purified virions were sedimented for 1 h at 0 (lane 1), 25,000 (lane 2), 50,000 (lane 3), 75,000 (lane 4), and 100,000 × g (lane 5). Virions remaining in the supernatant were sedimented by centrifugation for 1 h at 150,000 × g. The pellet fractions of the first centrifugation (P) and the second centrifugation (S) were analyzed by Western blotting for the presence of S protein.
Lysis of virions by octylglucoside.
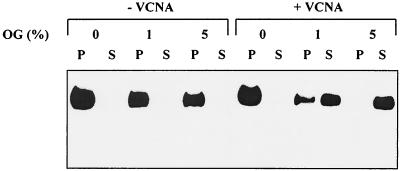
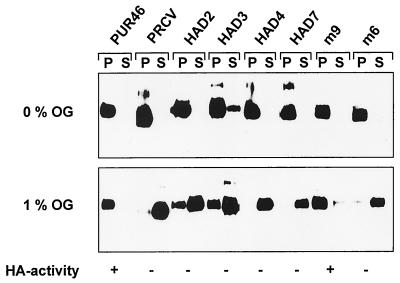
In order to determine whether the sialic acid binding activity also affects sedimentation of the glycoprotein S, virions were solubilized by octylglucoside. The nucleocapsid was sedimented by centrifugation at 100,000 × g. Under these conditions, viral glycoproteins are expected in the supernatant fraction. In order to verify the effectiveness of the solubilization, the supernatant and the pellet fractions were analyzed for the presence of S protein. As shown in Fig. 4, neuraminidase-treated TGE virions showed the characteristics expected for an enveloped virus. In the absence of octylglucoside, the S protein was present in the pellet fraction. After treatment with 1% octylglucoside, more than 50% of the S protein was detected in the supernatant. With virions treated with 5% octylglucoside, all of the S protein was found in the supernatant fraction. A different picture was obtained with purified TGEV that had not been pretreated with neuraminidase. Even at the highest concentration of octylglucoside, no S protein was detectable in the supernatant fraction. To confirm that the difference was due to the sialic acid binding activity, PRCoV, a nonhemagglutinating variant of TGEV, and several mutants of TGEV were analyzed in the same way. As shown in Fig. 5, the viruses that lacked a hemagglutinating activity resembled each other; i.e., after treatment with 1% octylglucoside, the majority of the S protein was detected in the supernatant fraction. In contrast, the hemagglutinating m9 mutant behaved after octylglucoside treatment like the parental virus, with the S protein being present mainly in the pellet fraction.
FIG. 4.
Sedimentation of the S protein after solubilization with octylglucoside (OG). Untreated (−VCNA) and neuraminidase-treated (+VCNA) virions were solubilized with 0, 1, or 5% octylglucoside and sedimented by ultracentrifugation as described in Materials and Methods. Aliquots of the pellet (P) and the supernatant (S) fractions were analyzed by Western blotting for the presence of S protein.
FIG. 5.
Sedimentation of the S proteins of TGEV, PRCoV, and mutants of TGEV (HAD2, HAD3, HAD4, HAD7, m9, and m6) after solubilization with octylglucoside (OG). The purified virus preparations were not pretreated with neuraminidase. Virions were incubated with 0 or 1% octylglucoside and sedimented by ultracentrifugation as described in Materials and Methods. Aliquots of the pellet (P) and the supernatant (S) fractions were analyzed by Western blotting for the presence of S protein. HA, hemagglutinating.
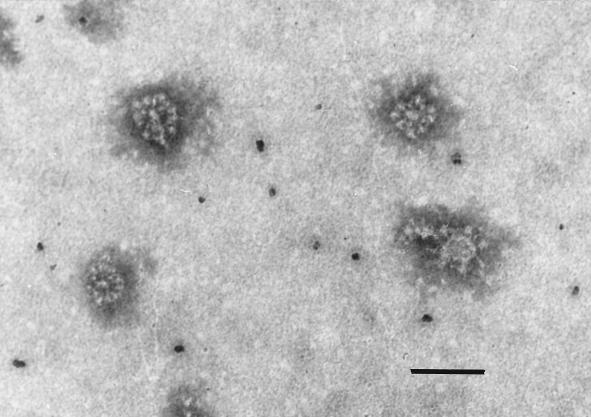
The appearance of the S protein in the pellet fraction might suggest that the virions had resisted detergent treatment. However, no evidence of the presence of intact virions was obtained by analysis with electron microscopy. Rather, the pellet fraction contained rosette-like structures (Fig. 6), as described for other coronavirus glycoproteins after removal of detergent (17). This result indicates that the interactions with sialoglycoconjugates that changed the sedimentation behavior of TGE virions were maintained after detergent lysis of the viral membrane and resulted in sedimentation of the S protein despite the presence of detergent.
FIG. 6.
Electron micrograph of the pellet fraction obtained after centrifugation of octylglucoside-treated virions. Magnification, ×150,000. Bar, 100 nm.
DISCUSSION
Our comparative analysis has demonstrated that TGEV mutants lacking a sialic acid binding activity have the sedimentation characteristics reported for coronaviruses (2). By contrast, the parental virus banded at a higher density of the sucrose gradient. As neuraminidase treatment of virions abolished the difference in sedimentation behavior, we conclude that binding of sialoglycoproteins to the virion surface is responsible for the increased density of untreated TGEV. The binding of sialylated glycoconjugates to TGEV has been suggested before, because virions harvested late in infection are not able to agglutinate erythrocytes (18). The hemagglutinating activity can be restored by neuraminidase treatment. The enzyme treatment obviously removes sialic acid residues that occupy the sialic acid binding site, allowing the S protein to interact with sialic acid residues on the erythrocyte. As the S protein of TGEV is highly glycosylated, one might assume that the sialic acid binding activity of this viral spike protein might interact with a sialic acid residue of another S protein, either on the same virion or on another virus particle. However, the available evidence indicates that mainly sialoglycoconjugates derived from the cell surface are bound to the viral surface. TGEV released into the cell supernatant early in infection is able to agglutinate erythrocytes. The hemagglutinating activity is lost late in infection and can be restored by neuraminidase treatment (18). Rather than treating the virions with enzyme, the hemagglutinating activity can also be restored by treating the confluent cell monolayer with neuraminidase prior to infection (8). This method does not affect the sialylation of the S protein; nevertheless, it is almost as efficient in obtaining hemagglutinating TGEV as enzyme treatment of virions. This finding suggests that sialoglycoconjugates present on the surface of the confluent cell monolayer are the major ligands that bind to the S protein and prevent it from interacting with erythrocytes. These ligands are presumably also responsible for the sedimentation behavior of TGEV described above.
The binding of sialoglycoconjugates to the virion surface does not affect virus infectivity. Whether or not TGEV is treated with neuraminidase, the infectivity titer is the same (18). This may be explained by the findings that sialic acid binding activity is not required for infection of cells and that the binding sites for aminopeptidase and for sialic acid are located at different positions on the S protein. Amino acids involved in the binding to aminopeptidase have been identified between positions 506 and 728 of the mature polypeptide chain (6). Binding to sialic acid was found to require amino acids between positions 145 and 209 (8, 9). Though the three-dimensional structure of the S protein has not yet been elucidated, it appears likely that the two binding sites are located at some distance from each other. Moreover, with virions containing surface-bound sialoglycoconjugates, not every spike protein on the viral surface, but only a fraction of them, is expected to actually interact with a sialylated ligand. This would be sufficient to prevent the virus from agglutinating erythrocytes, because the same type of binding site is involved and because a hemagglutination pattern requires the interaction of a virus particle with two erythrocytes. On the other hand, for the initiation of an infection, a virion has to attach to only a single cell. Even if several spike proteins contain sialylated ligands, there will be a sufficient number of S proteins left to interact with aminopeptidase N on the cell surface.
Influenza viruses or paramyxoviruses that use sialic acid as a receptor determinant for infection of cells contain a neuraminidase. This “receptor-destroying” enzyme is required for efficient spread of virions from the infected cell to other cells. With neuraminidase-deficient mutants or in the presence of neuraminidase inhibitors, aggregates of virions accumulate on the cell surface (10, 13). No such occurrences have been reported for TGEV, though the virus does not have a comparable enzyme. This may be related to the different mode of virus maturation. Influenza viruses and paramyxoviruses are released from the cell by a budding process at the cell surface. Coronaviruses, on the other hand, mature on intracellular membranes, and the virions are transported within membrane vesicles to the cell surface (19). Fusion of the transport vesicles with the plasma membrane results in release of virions into the cell supernatant. This mode of maturation may be more favorable with the interaction of virions with sialoglycoconjugates that are only loosely attached to cells than with membrane-anchored glycoproteins or glycolipids. Furthermore, S proteins not incorporated into virions are transported to the plasma membrane. They may interact with membrane-anchored sialoglycoconjugates and prevent them from binding to virions. Furthermore, the sialic acid binding activity of influenza virus may be stronger than that of TGEV and therefore may require a receptor-destroying enzyme. Recently, mutants of influenza viruses that lacked neuraminidase activity but could nevertheless undergo several replication cycles in cell cultures, eggs, and mice have been described (7). These mutants compensated for the loss of sialidase by a reduced affinity for cellular receptors.
The role of the sialic acid binding activity is not yet known. We reported recently that TGEV virions containing surface-bound sialoglycoconjugates have increased resistance to the action of detergents (9). Neuraminidase-treated TGEV or mutants deficient in sialic acid binding activity were completely inactivated by 0.6% octylglucoside. On the other hand, the infectivity of untreated TGEV was only partially inactivated at this concentration of detergent. Therefore, surface-bound sialoglycoconjugates may be beneficial during passage through the gastrointestinal tract and may protect the virus from the detrimental action of detergent-like substances, such as bile salts. Another possibility is that the sialic acid binding activity contributes to the binding of TGEV to epithelial cells of the intestine. While interaction with aminopeptidase N is sufficient for infection of cultured cells, the initiation of infection in the intestinal environment may require additional receptors to accomplish efficient binding to enterocytes. Attempts to identify sialoglycoconjugates on the surface of the intestinal epithelium that may serve as attachment sites for TGEV are in progress.
ACKNOWLEDGMENTS
The technical assistance of Anja Heiner is gratefully acknowledged. We are grateful to Luis Enjuanes for providing monoclonal antibody 6A.C3 and to Hubert Laude for providing site D mutants.
Financial support was provided by Deutsche Forschungsgemeinschaft (He1168/2-3 and SFB 280).
REFERENCES
- 1.Bernard S, Laude H. Site-specific alteration of transmissible gastroenteritis virus spike protein results in markedly reduced pathogenicity. J Gen Virol. 1995;76:2235–2241. doi: 10.1099/0022-1317-76-9-2235. [DOI] [PubMed] [Google Scholar]
- 2.Cavanagh D, Brian D A, Brinton M A, Enjuanes L, Holmes K V, Horzinek M C, Lai M M C, Laude H, Plagemann P G W, Siddell S G, Span W, Taguchi F, Talbot P J. Revision of the taxonomy of the Coronavirus, Torovirus and Arterivirus genera. Arch Virol. 1994;135:227–237. doi: 10.1007/BF01309782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Correa I, Jimenez G, Sune C, Bullido M J, Enjuanes L. Antigenic structure of the E2 glycoprotein from transmissible gastroenteritis coronavirus. Virus Res. 1988;10:77–93. doi: 10.1016/0168-1702(88)90059-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cox E, Pensaert M B, Callebaut P, van Deun K. Intestinal replication of a porcine respiratory coronavirus closely related antigenically to the enteric transmissible gastroenteritis. Vet Microbiol. 1990;23:237–243. doi: 10.1016/0378-1135(90)90154-N. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Delmas B, Gelfi J, L'Haridon R, Vogel L K, Sjöström H, Noren O, Laude H. Aminopeptidase N is a major receptor for the entero-pathogenic coronavirus TGEV. Nature. 1992;357:417–420. doi: 10.1038/357417a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Godet M, Grosclaude J, Delmas B, Laude H. Major receptor-binding and neutralization determinants are located within the same domain of the transmissible gastroenteritis virus (coronavirus) spike protein. J Virol. 1994;68:8008–8016. doi: 10.1128/jvi.68.12.8008-8016.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hughes M T, Matrosovich M, Rodgers M E, McGregor M, Kawaoka Y. Influenza A viruses lacking sialidase activity can undergo multiple cycles of replication in cell culture, eggs, or mice. J Virol. 2000;74:5206–5212. doi: 10.1128/jvi.74.11.5206-5212.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Krempl C, Schultze B, Laude H, Herrler G. Point mutations in the S protein connect the sialic acid binding activity with the enteropathogenicity of transmissible gastroenteritis coronavirus. J Virol. 1997;71:3285–3287. doi: 10.1128/jvi.71.4.3285-3287.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Krempl C, Ballesteros M L, Zimmer G, Enjuanes L, Klenk H-D, Herrler G. Characterization of the sialic acid binding activity of transmissible gastroenteritis coronavirus by analysis of haemagglutination-deficient mutants. J Gen Virol. 2000;81:489–496. doi: 10.1099/0022-1317-81-2-489. [DOI] [PubMed] [Google Scholar]
- 10.Liu C, Eichelberger M C, Compans R W, Air G M. Influenza type A virus neuraminidase does not play a role in viral entry, replication, assembly, or budding. J Virol. 1995;69:1099–1106. doi: 10.1128/jvi.69.2.1099-1106.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Noda M, Koide F, Asagi M, Inaba Y. Physicochemical properties of transmissible gastroenteritis virus hemagglutinin. Arch Virol. 1988;99:163–172. doi: 10.1007/BF01311067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Noda M, Yamashita H, Koide F, Kadoi K, Omori T, Asagi M, Inaba Y. Hemagglutination with transmissible gastroenteritis virus. Arch Virol. 1987;96:109–115. doi: 10.1007/BF01310994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Palese P, Compans R W. Inhibition of influenza virus replication in tissue culture by 2-deoxy-2,3-dehydro-N-trifluoroacetylneuraminic acid (FANA): mechanism of action. J Gen Virol. 1976;33:159–163. doi: 10.1099/0022-1317-33-1-159. [DOI] [PubMed] [Google Scholar]
- 14.Pensaert M, Callebaut P, Cox E. Enteric coronaviruses of animals. In: Kapikian A Z, editor. Viral infections of the gastrointestinal tract. New York, N.Y: Marcel Dekker; 1993. pp. 627–696. [Google Scholar]
- 15.Rasschaert D, Duarte M, Laude H. Porcine respiratory coronavirus differs from transmissible gastroenteritis virus by a few genomic deletions. J Gen Virol. 1990;71:2599–2607. doi: 10.1099/0022-1317-71-11-2599. [DOI] [PubMed] [Google Scholar]
- 16.Sánchez C M, Gebauer F, Suné C, Mendez A, Dopazo J, Enjuanes L. Genetic evolution and tropism of transmissible gastroenteritis coronaviruses. Virology. 1992;190:92–105. doi: 10.1016/0042-6822(92)91195-Z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schultze B, Gross H-J, Brossmer R, Herrler G. The S protein of bovine coronavirus is a hemagglutinin recognizing 9-O-acetylated sialic acid as a receptor determinant. J Virol. 1991;65:6232–6237. doi: 10.1128/jvi.65.11.6232-6237.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schultze B, Krempl C, Ballesteros M L, Shaw L, Schauer R, Enjuanes L, Herrler G. Transmissible gastroenteritis coronavirus, but not the related porcine respiratory coronavirus, has a sialic acid (N-glycolylneuraminic acid) binding activity. J Virol. 1996;70:5634–5637. doi: 10.1128/jvi.70.8.5634-5637.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tooze J, Tooze S, Warren G. Replication of coronavirus MHV-A59 in sac− cells: determination of the first site of budding of progeny virions. Eur J Cell Biol. 1984;33:281–293. [PubMed] [Google Scholar]