TO THE EDITOR:
The Randomized Trial to Prevent Vascular Events in HIV (REPRIEVE) (Aug. 24 issue)1 was a global trial that assessed pitavastatin calcium as compared with placebo to prevent major adverse cardiovascular events (MACE) among participants with HIV infection who were at low-to-moderate predicted cardiovascular risk. In this trial, MACE was defined as a composite of cardiovascular death, myocardial infarction, hospitalization for unstable angina, stroke, transient ischemic attack, peripheral arterial ischemia, revascularization, or death from an undetermined cause. The trial was designed to detect a hazard ratio of 0.70 with 85% power in the intention-to-treat population on the basis of an estimated 288 primary-outcome events.
In March 2023, the trial was stopped early for efficacy by the data and safety monitoring board during a planned interim analysis that was performed after the occurrence of 225 events (78% of the estimated number) on the basis of follow-up data through December 2022. At that time, the participants in the pitavastatin group had a 35% reduction in MACE as compared with the placebo group, without any unanticipated safety effects. The participants were advised to continue to receive their assigned treatment until a final trial visit between April and August 2023; the trial data were unblinded in November 2023. At the final trial visit, 13 participants (4 in the pitavastatin group and 9 in the placebo group) had started to receive a nonstudy statin since the trial cutoff. This report updates the published analysis with final data regarding efficacy and safety outcomes, including all participant follow-up (Fig. 1). Additional details are provided in Section 1 in the Supplementary Appendix and in the protocol, available with the full text of this letter at NEJM.org.
Figure 1. Treatment Effect of Pitavastatin on Major Adverse Cardiovascular Events.
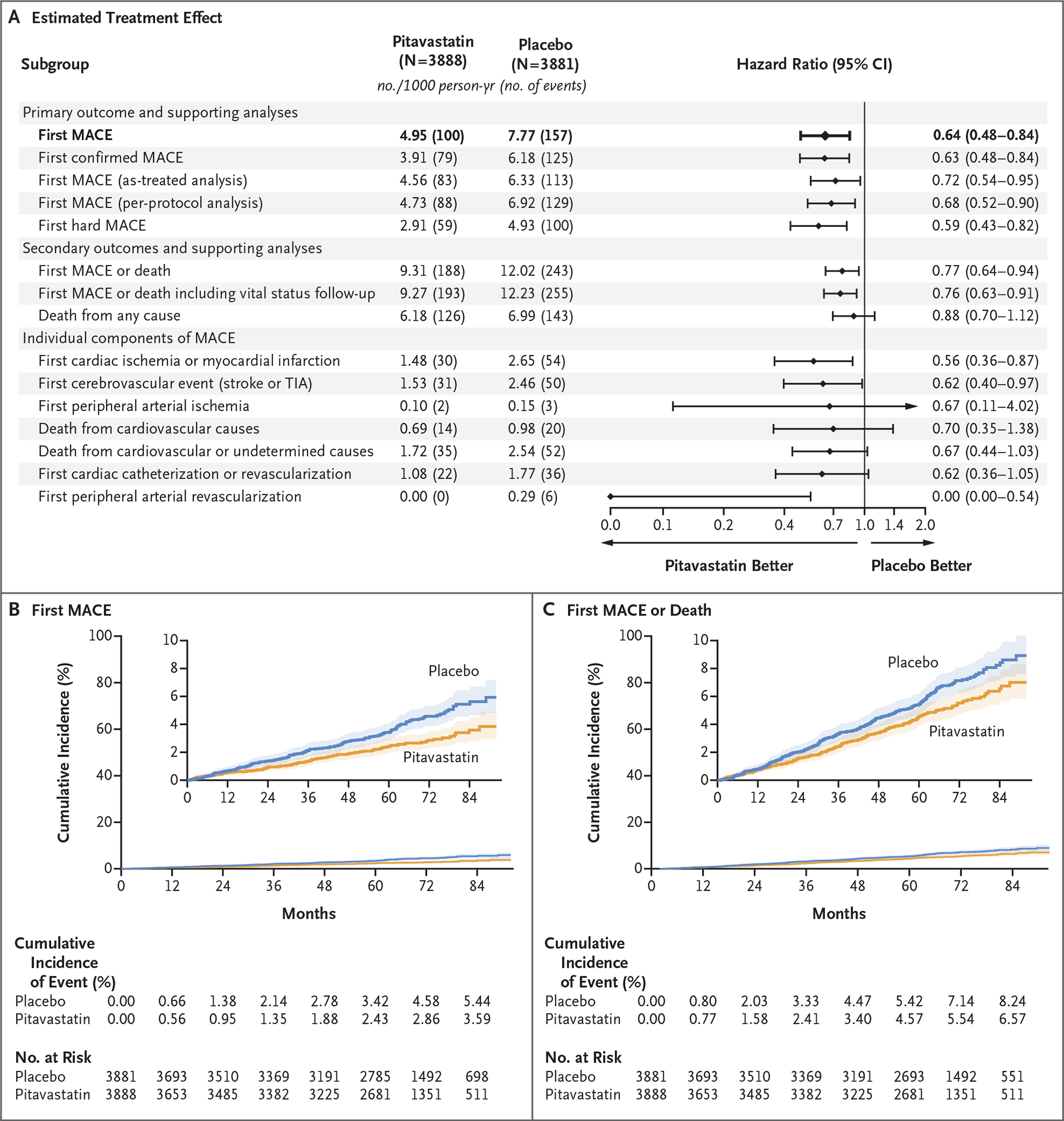
Shown is the incidence of major adverse cardiovascular events (MACE) among trial participants with HIV infection in the pitavastatin group and the placebo group and the estimated treatment effect, according to stratified Cox proportional-hazards analysis (Panel A). The primary outcome of the trial is shown in bold at the top of the graph. In addition to the previously reported outcome measures, a supportive outcome measure that was limited to the hard clinical end points of myocardial infarction, stroke, and cardiovascular death (hard MACE) is presented. Cox proportional-hazards models were stratified according to sex at birth and the CD4 count at screening. Also shown are the cumulative incidence of a primary-outcome event (first MACE) (Panel B) and a key secondary outcome (first MACE or death from any cause) (Panel C). In Panels B and C, the insets show the data on an expanded y axis. Aside from the primary result, the widths of the confidence intervals have not been adjusted for multiplicity and therefore may not be used in place of hypothesis testing. TIA denotes transient ischemic attack.
Of the 7769 participants who were enrolled, 82% completed the trial (Fig. S1 in the Supplementary Appendix). A first MACE had occurred in 257 participants (89% of the estimated number) over a median of 5.6 years of follow-up, totaling 32 more events than had been described in the primary report. All efficacy and safety findings were consistent with those in the primary report (Tables S1 and S2). The estimated rate of the first MACE was 4.95 per 1000 person-years (95% confidence interval [CI], 4.07 to 6.03) in the pitavastatin group and 7.77 per 1000 person-years (95% CI, 6.64 to 9.08) in the placebo group (hazard ratio, 0.64; 95% CI, 0.48 to 0.84) (Fig. 1 and Fig. S3). Nonproportional hazard assumptions were not violated. Effects that were consistent with those in the primary report were seen for secondary outcomes (including the occurrence of a first MACE or death from any cause) and in supportive analyses of the primary outcome and a sensitivity analysis accounting for missing data, as well as for individual MACE components and events within prespecified subgroups (Figs. S3 through S6). In a post hoc analysis, the number of patients who would need to be treated with pitavastatin to prevent one primary-outcome event was 100 (Fig. S10).
Thus, among a group of participants with HIV infection who were at low-to-moderate predicted cardiovascular risk, pitavastatin reduced the incidence of MACE over a follow-up of 5.6 years with an acceptable safety profile. Revised treatment guidelines incorporating data from REPRIEVE that recommend statin therapy in this patient population were recently announced.2
Supplementary Material
| THIS WEEK’S LETTERS | |
|---|---|
| 1626 | Trial Update of Pitavastatin to Prevent Cardiovascular Events in HIV Infection |
| 1628 | CAR T-Cell Therapy in Autoimmune Disease |
| 1632 | A Phase 3 Trial of Resmetirom in NASH with Liver Fibrosis |
| 1633 | Chronic Lymphocytic Leukemia Therapy Guided by Measurable Residual Disease |
| 1635 | Being Well while Doing Well |
Acknowledgments
Supported by the NIH through grants (U01HL123336 and 1UG3HL164285) to the Clinical Coordinating Center for the trial, grants (U01HL123339 and 1U24HL164284) to the Data Coordinating Center for the trial, and a grant (P30DK040561) to the Nutrition Obesity Research Center at Harvard; by Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare; and by grants (UM1 AI068636 and UM1 AI106701) from the NIAID, which supports the AIDS Clinical Trials Group (ACTG) Leadership and Operations Center and the ACTG Laboratory Center.
Footnotes
The views expressed in this letter are those of the authors and do not necessarily represent the views of the National Heart, Lung, and Blood Institute or the National Institute of Allergy and Infectious Diseases (NIAID), the National Institutes of Health (NIH), or the U.S. Department of Health and Human Services.
Disclosure forms provided by the authors are available with the full text of this letter at NEJM.org.
Contributor Information
Steven K. Grinspoon, Massachusetts General Hospital, Boston, MA
Heather J. Ribaudo, Harvard T.H. Chan School of Public Health, Boston, MA
Pamela S. Douglas, Duke Clinical Research Institute, Durham, NC
References
- 1.Grinspoon SK, Fitch KV, Zanni MV, et al. Pitavastatin to prevent cardiovascular disease in HIV infection. N Engl J Med 2023;389:687–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in adults and adolescents with HIV. Rockville, MD: NIH Office of AIDS Research, February 2024. (https://clinicalinfo.hiv.gov/en/guide-lines/adult-and-adolescent-arv). [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


