Abstract
Microglia, the resident immune cells of the central nervous system, play a critical role in maintaining brain homeostasis. However, in neurodegenerative conditions, microglial cells undergo metabolic reprogramming in response to pathological stimuli, including Aβ plaques, Tau tangles, and α-synuclein aggregates. This metabolic shift is characterized by a transition from oxidative phosphorylation (OXPHOS) to glycolysis, increased glucose uptake, enhanced production of lactate, lipids, and succinate, and upregulation of glycolytic enzymes. These metabolic adaptations result in altered microglial functions, such as amplified inflammatory responses and diminished phagocytic capacity, which exacerbate neurodegeneration. This review highlights recent advances in understanding the molecular mechanisms underlying microglial metabolic reprogramming in neurodegenerative diseases and discusses potential therapeutic strategies targeting microglial metabolism to mitigate neuroinflammation and promote brain health.
Graphical Abstract
Microglial Metabolic Reprogramming in Neurodegenerative Diseases
This graphical abstract illustrates the metabolic shift in microglial cells in response to pathological stimuli and highlights potential therapeutic strategies targeting microglial metabolism for improved brain health.
Keywords: Microglia, Metabolic reprogramming, Alzheimer's disease, Parkinson's disease, Multiple sclerosis
Introduction
Microglial cells, the primary immune constituents of the central nervous system (CNS), are crucial in maintaining brain homeostasis and providing immune surveillance. These cells constitute approximately 5–12% of the total glial cell population within the brain and originate from the yolk sac during embryogenesis before migrating to the CNS in early development (Zhang et al. 2022). Microglial cells display remarkable functional diversity and plasticity, dynamically adapting their phenotype and functions in response to environmental cues. In their surveillance state, microglial cells exhibit a ramified morphology, actively scanning the surrounding neuronal environment, participating in synapse pruning, and promoting neuronal health (Cornell et al. 2022). Upon detecting injury, infection, or other pathological stimuli, microglial cells transition to an activated state is characterized by morphological changes, proliferation, migration, and the release of pro-inflammatory cytokines, chemokines, and other mediators (Spiteri et al. 2022). Although they have protective functions, aberrant microglial activation and dysregulation can contribute to the development and progression of various neurodegenerative diseases, such as Alzheimer's disease(AD), Parkinson's disease(PD), and multiple sclerosis(MS) (Kwon and Koh 2020). Growing evidence indicates that microglial cells play a dual role in these conditions, with both neuroprotective and neurotoxic effects, depending on the specific activation state and the molecular mechanisms involved (Geloso et al. 2017; Konishi et al. 2019; Qin et al. 2019; Choudhury et al. 2021). Recent research has emphasized the importance of microglial metabolic reprogramming in modulating their activation and function in the context of neuroinflammation and neurodegeneration (Lauro and Limatola 2020; Bernier et al. 2020b; Wang et al. 2022). This review aims to provide a comprehensive, updated overview of the emerging role of microglial metabolic reprogramming in the pathogenesis of neurodegenerative diseases and discuss its potential as a therapeutic target. We will examine key metabolic pathways involved in microglial activation and polarization, and explore how targeting these pathways may offer novel strategies to modulate microglial functions and ameliorate neurodegenerative processes. Furthermore, we will discuss the current challenges and future directions in understanding the complex interplay between microglial metabolism and neurodegeneration.
Microglial Metabolic Reprogramming
Microglia cells display a constant need for ATP, especially when activated. They express genes involved in the metabolism of the three major energy substrates, including glucose, fatty acid, and glutamine (Kelly and O’Neill 2015). Given the limited glucose availability in the brain, it is reasonable to assume that microglia have the capacity for flexible use of energy substrates. This flexibility may be necessary for microglia to fulfill their beneficial roles in various pathophysiological conditions. Several studies have shown that changes in energy demand can prompt cells to engage different metabolic pathways or utilize alternative substrates. For example, activation of peripheral macrophages induces a metabolic switch from oxidative phosphorylation (OXPHOS) to anaerobic glycolysis to increase ATP production (Kelly and O’Neill 2015). However, little is known about how microglial metabolism is integrated into their functionality. It is conceivable that a metabolic switch in microglia might be accompanied by the generation of metabolites that alter neuronal health. Further research is needed to elucidate the potential metabolic flexibility of microglia and the implications for their function in the brain.
Glucose Metabolism
The brain primarily relies on glucose as its main energy source, with brain glucose concentration at about 20% of that found in blood. Glucose uptake in the brain and peripheral tissues is mediated by 5 main glucose transporters (GLUT 1–5)(Thorens and Mueckler 2010). GLUT1 and 3 are expressed in various tissues, playing a role in regulating basal glucose uptake. Neurons utilize high-affinity GLUT3 transporters to ensure a constant glucose influx even under low glucose conditions (Mergenthaler et al. 2013). Microglia cells express GLUT3 as well, along with GLUT1 and GLUT5 transporters. These transporters facilitate glucose uptake by microglia to meet their energy needs during both resting and activated states (Yenari and Giffard 2001). Microglial activation is implicated in neuronal damage due to low glucose levels. However, when glycolytic glucose utilization is inhibited, neurons seem unaffected, while microglia become necrotic (Vilalta and Brown 2014). Microglia may adjust their glucose metabolic profile in response to changes in their activity state, possibly to meet increased energy demands (Zhang et al. 2014; Lavin et al. 2014). Interestingly, microglia are the only CNS cells expressing GLUT5 (Xu et al. 2016), which has a low affinity for glucose but a high affinity for fructose. Although the precise function of GLUT5 in microglia is unknown, it could play a role in sensing fluctuating glucose concentrations or contribute to fructose-induced metabolic disorders (Zhang et al. 2015; Lin et al. 2016). In vitro studies have revealed that both low and high glucose levels affect microglial survival and activation, respectively (Quan et al. 2011; Vinoth Kumar et al. 2016). However, caution must be exercised when interpreting in vitro data, as they may not accurately reflect in vivo conditions. For instance, diabetic mice show mean blood glucose concentrations of approximately 20 mM, with peak concentrations up to 30 mM, while in vitro microglial cultures are typically exposed to 25 mM glucose conditions (Quan et al. 2011; Gao et al. 2014; Dai et al. 2015; Beins et al. 2016). Furthermore, hyperglycemic db/db mice did not exhibit microglial activation in the hypothalamus (Yang et al. 2021), suggesting that brain glucose concentrations may be maintained at lower levels than previously assumed.
In summary, glucose metabolism plays a critical role in microglial function, and an increased understanding of the complex interactions among glucose, microglia, and neuronal health is needed to further elucidate the underlying mechanisms. The main metabolic pathways involved in glucose metabolism include glycolysis, the pentose phosphate pathway (PPP), and the tricarboxylic acid (TCA) cycle. Each of these pathways plays a specific role in modulating microglial function (Vander Heiden et al. 2009; Tu et al. 2019; Soto-Heredero et al. 2020).
Glycolysis
The significance of glycolysis in microglial activation has been increasingly recognized, with numerous studies emphasizing its role in pro-inflammatory microglial polarization and function. This fundamental metabolic pathway is involved in the breakdown of glucose to generate ATP, serving as a rapid energy source for cells. A series of enzymatic reactions converts glucose into pyruvate, yielding a net production of two ATP and two NADH molecules per glucose molecule (Ghosh et al. 2018). During neuroinflammation, pro-inflammatory microglia display increased glucose uptake and upregulation of glycolytic enzymes, such as hexokinase(HK), phosphofructokinase(PFK), and pyruvate kinase M2(PKM2) (Vander Heiden et al. 2009). The metabolic shift towards glycolysis is essential for microglial status and function, enabling rapid ATP production despite its relative inefficiency (Bolanos et al. 2004). Disruption of pyruvate entry into the TCA cycle during glycolysis leads to the accumulation of nitric oxide(NO) and interleukin(IL)-1β (MacMicking et al. 1997; Klimaszewska-Łata et al. 2015; Zezina et al. 2020). NO irreversibly inhibits the electron transport chain and pyruvate dehydrogenase, which in turn affects cell proliferation. Lipopolysaccharide(LPS)-stimulated microglia cell exhibit elevated expression of GLUT1, HK2, and PFK1, indicating an enhanced glycolytic capacity (Van den Bossche et al. 2016). Blocking GLUT1 or inhibiting HK2 activity reduces pro-inflammatory cytokine production, suggesting the importance of glycolysis in driving the inflammatory response (Masoumi et al. 2020; Wang et al. 2023). 6-phosphofructo-2-kinase/fructose-2, 6-bisphosphatase 3 (PFKFB3), a key regulatory enzymes in glycolysis, has also been implicated in microglial activation (Kim et al. 2012). LPS-stimulated microglia demonstrate elevated PFKFB3 levels and increased production of fructose-2, 6-bisphosphate, which is an allosteric activator of PFK1, leading to enhanced glycolytic flux (Zhang et al. 2021). Genetic or pharmacological inhibition of PFKFB3 diminishes the release of pro-inflammatory cytokines, such as IL-1β, IL-6, and TNF-α (Gao et al. 2012; Xu et al. 2021). The glycolytic enzyme pyruvate kinase M2(PKM2) has also been reported to play a role in regulating pro-inflammatory microglial activation(Li et al. 2021). LPS stimulation induces PKM2 nuclear translocation in primary microglia. Nuclear PKM2 acts as a co-activator for the transcription factor STAT1, promoting the expression of pro-inflammatory genes. Silencing PKM2 or preventing its nuclear translocation by TEPP-46 significantly alleviated the inflammatory response in microglia (Meiser et al. 2016).
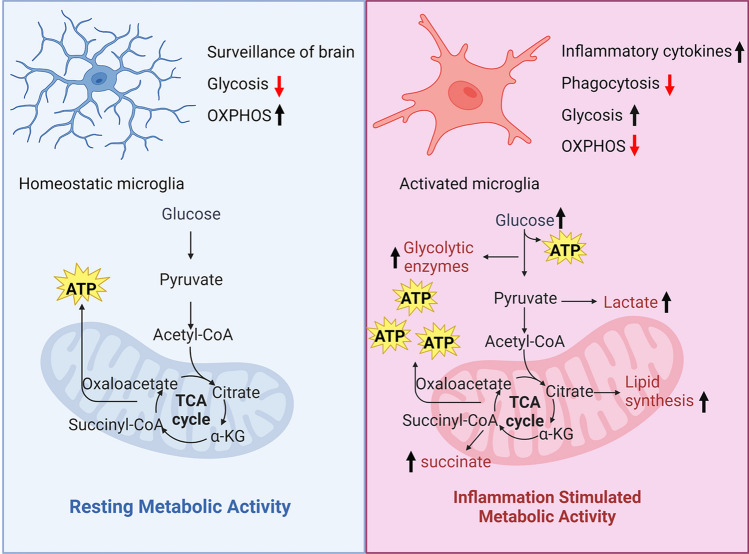
Glycolysis-derived intermediates also contribute to other pathways involved in the pro-inflammatory response. For example, 3-phosphoglycerate(3-PG), a glycolytic intermediate, is converted to serine by the enzyme phosphoglycerate dehydrogenase (PHGDH) (Hannun and Obeid 2018). Serine serves as a precursor for the synthesis of sphingolipids and ceramides, which are implicated in the production of pro-inflammatory cytokines in activated microglia (Zhao and Xu 2022). Additionally, glycolysis-derived pyruvate can be converted to lactate, which has been suggested to modulate microglial activation and neuroinflammation (Orihuela et al. 2016). Increased lactate production by pro-inflammatory microglia can contribute to extracellular acidification, which in turn exacerbate neuroinflammatory processes and neuronal damage. In conclusion, glycolysis is an essential metabolic process in microglial activation and polarization during neuroinflammation. The upregulation of glycolytic enzymes, such as HK, PFK, and PKM2, in pro-inflammatory microglia facilitates rapid energy production and supports the synthesis of pro-inflammatory mediators (Fig. 1). Targeting glycolytic enzymes or modulating glycolytic intermediates may provide novel therapeutic strategies to regulate microglial activation and attenuate neuroinflammation.
Fig. 1.
Microglia metabolic reprogramming during inflammation This infographic illustrates the metabolic reprogramming of microglial cells under normal and inflammatory conditions. In a healthy state, microglial cells primarily rely on oxidative phosphorylation (OXPHOS) for energy production. However, during inflammation, microglia shift their metabolic preference towards glycolysis. This metabolic reprogramming is characterized by increased glucose uptake, lactate production, lipid synthesis, and accumulation of succinate, along with an upregulation of glycolytic enzymes. As a consequence, microglial inflammatory responses are amplified, while phagocytic function is diminished
Tricarboxylic Acid (TCA) Cycle
The TCA cycle, also known as the Krebs cycle or citric acid cycle, is a central metabolic pathway involved in energy production through the oxidation of acetyl-CoA derived from carbohydrates, fats, and proteins. In microglial cells, the TCA cycle is crucial for energy production, redox balance, and the biosynthesis of essential molecules (Wang et al. 2021). Elevated intracellular succinate, an intermediate of the TCA cycle, exerts a protective role in the primary microglia by preventing their conversion into the pro-inflammatory phenotype (Tannahill et al. 2013). The accumulation of succinate led to the stabilization of hypoxia-inducible factor-1α(HIF-1α) and the induction of pro-inflammatory genes (Jha et al. 2015). Moreover, the inhibition of succinate dehydrogenase(SDH), a key enzyme in the TCA cycle, further increased succinate levels and exacerbated the inflammatory response in microglia, indicating the importance of the TCA cycle in regulating microglial activation (Han et al. 2021). Incontrast, anti-inflammatory microglia display an intact TCA cycle and increased OXPHOS activity, suggesting a metabolic shift towards energy production to support their reparative and neuroprotective functions (Bernier et al. 2019). Promoting the TCA cycle and OXPHOS in microglia by activating peroxisome proliferator-activated receptor gamma co-activator 1-alpha(PGC-1α) resulted in a reduced inflammatory response and improved functional recovery in a mouse model of ischemic stroke (Baik et al. 2019). The TCA cycle is also implicated in the regulation of microglial phagocytic function. A study by Baik et al. revealed that defective TCA cycle function in microglia, characterized by the accumulation of α-ketoglutarate, a TCA cycle intermediate, and a decrease in succinate, led to impaired phagocytosis and increased Aβ accumulation in an AD mouse model (Sharkey 2021). Restoration of TCA cycle function by administration of dimethyl α-ketoglutarate improved microglial phagocytic activity and ameliorated Aβ pathology in these mice (Sharkey 2021). This finding highlights the importance of the TCA cycle in maintaining microglial function (Fig. 1) and suggests that targeting the TCA cycle may offer potential therapeutic benefits in neurodegenerative diseases.
Pentose Phosphate Pathway (PPP)
The PPP is another critical metabolic route in microglial cells, playing a significant role in their function and polarization. The PPP branches off from glycolysis and consists of two phases: the oxidative phase and the non-oxidative phase (Stincone et al. 2015). The oxidative phase generates NADPH, which is essential for maintaining cellular redox balance and biosynthetic reactions, while the non-oxidative phase generates ribose-5-phosphate (R5P), a precursor for nucleotide synthesis (Niot et al. 2009). Recent studies have highlighted the importance of the PPP in microglial activation and function. In microglia, LPS stimulation increased the expression of glucose-6-phosphate dehydrogenase(G6PD), the rate-limiting enzyme of the PPP's oxidative phase. Furthermore,inhibition of G6PD suppressed the production of nitric oxide(NO) and pro-inflammatory cytokines, indicating that the PPP plays a role in modulating neuroinflammatory responses (Soto-Heredero et al. 2020). The PPP is also involved in the regulation of microglial polarization. Pro-inflammatory microglia exhibit increased PPP activity, providing NADPH for the production of reactive oxygen species(ROS) and pro-inflammatory cytokines (Wang et al. 2021). In contrast, anti-inflammatory microglia display decreased PPP activity, which contributes to their anti-inflammatory and neuroprotective functions (Wang et al. 2021). Modulating the PPP in microglia may, thus, represent a promising therapeutic approach for neuroinflammatory and neurodegenerative diseases. Apart from its role in the oxidative phase, the PPP’s non-oxidative phase also influences microglial function. Inhibition of the non-oxidative PPP enzymetransketolase(TKT) impaired the phagocytic ability of microglia and promoted the accumulation of amyloid-beta(Aβ) in an AD mouse model (Sharkey 2021). These results suggest that the non-oxidative PPP is crucial for maintaining microglial phagocytic function and may be a potential target for AD therapy. In summary, PPP plays a critical role in microglial activation, polarization, and function. Modulating PPP activity, particularly by targeting key enzymes such as G6PD and TKT, may offer novel therapeutic strategies for mitigating neuroinflammation and neurodegeneration.
Fatty Acid Metabolism
Fatty acids are critical components of cellular metabolism, distinguished by chain length and saturation degree. They play essential roles in energy production, signaling, and microglial activity modulation (Ebert et al. 2003). Fatty acid metabolism, including fatty acid oxidation(FAO), contributes to the brain's energy supply, meeting approximately 20% of total energy demand (Schönfeld and Wojtczak 2008). Microglia have recently been found to express proteins involved in FAO, such as acyl-CoA synthetases(ACSs) and lipoprotein lipase(LPL) (Zhang et al. 2014). Activated microglia exhibit elevated energy demands that may be partly met by utilizing fatty acids (Uttara et al. 2009). However, the increased oxygen consumption required for fatty acid metabolism may pose challenges in the brain’s low-oxygen environment (Uttara et al. 2009). Heightened fatty acid metabolism can also contribute to ROS production and subsequent cellular oxidative stress, which are pathological features of many neurological diseases (Chen et al. 2012b; Carvalho et al. 2017; Nadjar 2018). Fatty acids can function as signaling molecules that modulate microglial activation. Unsaturated fatty acids, such as omega-3 polyunsaturated fatty acids, encourage an anti-inflammatory or phagocytic microglial phenotype (Wang et al. 2012). In contrast, saturated fatty acids induce microglial activation and inflammation through toll-like receptor 4(TLR4) and NF-kB signaling pathways (Duffy et al. 2015, 2017). Short-chain fatty acids(SCFAs), produced exclusively by the gut microbiota, exert mainly anti-inflammatory effects on microglia (Chang et al. 2015; Oishi et al. 2017). SCFAs are vital for proper microglial maturation and function, as demonstrated by the impaired microglial function in germ-free mice, which can be restored by introducing a complex microbiota or supplementing SCFAs in drinking water (Fernandez et al. 2018). The free fatty acid receptor 2(FFAR2) may mediate some of the SCFA effects on microglia, though its precise role remains unclear. While human microglia express FFAR2 (Zhang et al. 2014), murine central nervous system cells, including microglia, reportedly do not(Fernandez et al. 2018). Further investigation is needed to clarify FFAR2’s function in the context of SCFA-mediated microglial modulation. Targeting fatty acid composition and metabolism in microglia may provide a novel therapeutic strategy for CNS diseases. Pro-inflammatory stimuli generally promote fatty acid synthesis and upregulation of saturated fatty acids, while anti-inflammatory phenotypes favor fatty acid oxidation and unsaturated fatty acids (Mashek and Coleman 2006; Hamilton and Brunaldi 2007). Manipulating fatty acid metabolism could influence microglial activation and subsequently impact neurodegenerative disease progression. The balance between saturated and unsaturated fatty acids is crucial for modulating microglial activity. For example, an increase in saturated fatty acids, such as palmitic and stearic acids, contributes to the upregulation of pro-inflammatory cytokines, ROS, and NO (Mitchell and Hatch 2011; Duffy et al. 2015, 2017). In contrast, omega-6 fatty acids like arachidonic acid are upregulated when macrophages are activated by KLA, a LPS substructure that stimulates TLR4 (Xiang et al. 2003). DHA has shown promise in ameliorating LPS-induced damage in microglia by promoting lipid body formation, mitochondrial interaction, and restoring mitochondrial function (Suárez et al. 2002). Acyl-CoA synthetase 6(Acsl6) likely mediates these biological effects, as Acsl6−/− mice exhibit increased microglial activation, altered glutamate metabolism, and reduced DHA-containing lipids in Acsl6−/− tissues (Felipo and Butterworth 2002). Fatty acid transport across the blood–brain barrier(BBB) remains a debated topic, with both passive diffusion (Olney 1969; Pochini et al. 2014) and protein-mediated transport mechanisms (De Vitto et al. 2016) proposed. Further research is required to elucidate the mechanisms governing fatty acid transport into the brain and its implications for microglial function.
In summary, fatty acids play a multifaceted role in modulating microglial activation and function, making them promising targets for therapeutic interventions in neurodegenerative diseases (Fig. 2). The balance between saturated and unsaturated fatty acids is vital for controlling microglial activity, with unsaturated fatty acids generally promoting an anti-inflammatory phenotype, while saturated fatty acids induce inflammation. The effects of SCFAs on microglia are largely anti-inflammatory, and their precise role in appetite regulation and energy homeostasis warrants further investigation. Elucidating the mechanisms behind fatty acid transport across the BBB and their impact on microglial function will be critical in developing novel therapeutic strategies targeting fatty acid metabolism in CNS diseases.
Fig. 2.
Schematic representation of fatty acid metabolism pathways and their influence on microglial activation and phenotypes Fatty Acid Oxidation: Fatty acids are converted to fatty acyl-CoA by acyl-CoA synthetases (ACSs). Fatty acyl-CoA molecules are transported into the mitochondria by carnitine palmitoyltransferase 1 (CPT1) for fatty acid oxidation, generating ATP.Fatty Acid Synthesis: Fatty acids are synthesized in the cytoplasm, with saturated fatty acids (SFAs) produced via fatty acid synthase (FAS) and unsaturated fatty acids (UFAs) generated through the action of stearoyl-CoA desaturase (SCD).Fatty acids modulate microglial activation and phenotypes. Short-chain fatty acids (SCFAs) regulate resting microglia through free fatty acid receptor 2 (FFAR2). Unsaturated fatty acids (UFAs) and omega-3 polyunsaturated fatty acids (omega-3 PUFAs) modulate anti-inflammatory microglia via PPAR-γ signaling. Omega-6 polyunsaturated fatty acids (omega-6 PUFAs), like arachidonic acid, are upregulated during pro-inflammatory activation via TLR4 signaling. Saturated fatty acids (SFAs) also activate pro-inflammatory microglia through TLR4 signaling
Glutamine Metabolism
Glutamine, a highly abundant amino acid in plasma, has been believed not to cross the BBB freely due to multiple transporters (Jin et al. 2015). Interestingly, these transporters not only transport glutamine from the brain to the blood but may also be involved in its uptake into the brain. Astrocytes generate glutamine from excessive glutamate and ammonia, acting as a protective measure against excitotoxicity for neurons (Huang et al. 2011). It’s important to recognize that glutamate and ammonia possess neurotoxic properties (Chen et al. 2012a), and experiments have demonstrated that subcutaneous glutamate injections can lead to obesity (McIntosh et al. 2019). Primarily derived from glutamate and ammonia metabolism, glutamine also serves as an energy source for the brain. Cellular glutamine uptake is mediated by various amino acid transporters, with solute-linked carrier family 1 member A5(SLC1A5) being the primary transporter responsible for this (Yeh et al. 2016). SLC1A5 has a high affinity for glutamine and is upregulated in some high-energy-demanding tumors (Wang et al. 2016). Once inside the cell, glutamine is converted into α-ketoglutarate by glutaminase(GLS) and glutamate dehydrogenase(GLUD1). Notably, GLS, GLUD1, and SLC1A5 are highly expressed in human and mouse microglia. Several studies have highlighted the role of microglial glutamate production in neurotoxicity. A mouse model with increased microglial expression of the glutamine transporter SLC38A1 showed continuous production and secretion of glutamate into the interstitial space, leading to mild neurotoxicity (Wang et al. 2015). In primary human microglial cultures, HIV-1 infection increased extracellular glutamate levels, amplifying neurotoxicity (Jay et al. 2015). Moreover, microglia infected with Japanese encephalitis virus exhibited heightened glutamate release into the medium, causing significant neuronal damage when applied to neuronal cultures (Ulland and Colonna 2018).
Despite these findings, the specific role of hypothalamic microglial glutamine metabolism and its interaction with other energy sources, such as glucose and fatty acids, remains largely unexplored. Further research is needed to determine the extent to which microglia depend on these energy sources and to better understand the implications of these metabolic pathways in brain function and neurological disorders. This knowledge could potentially lead to new therapeutic interventions targeting microglial metabolism in the treatment of various neurodegenerative diseases.
Mitochondrial Dysfunction
Mitochondrial dysfunction in microglial cells plays a crucial role in the pathogenesis of neurodegenerative diseases, and its impact on microglial metabolic reprogramming has recently emerged as a vital area of research. One key aspect of microglial mitochondrial dysfunction is the alteration in the balance of metabolic pathways. In healthy microglia, OXPHOS serves as the primary source of ATP production. However, when mitochondrial dysfunction occurs, microglia cells exhibit a shift towards glycolysis. This metabolic shift is particularly evident in the pro-inflammatory phenotype, which is associated with increased production of pro-inflammatory cytokines and ROS (Orihuela et al. 2016). The anti-inflammatory phenotype, on the other hand, relies more on OXPHOS and is associated with anti-inflammatory and tissue repair functions (Tang and Le 2016).The shift towards glycolysis in pro-inflammatory microglia can be linked to the activation of hypoxia-inducible factor 1-alpha(HIF-1α) (Semenza 2012). Under conditions of mitochondrial dysfunction, increased levels of ROS can stabilize HIF-1α, which in turn upregulates the expression of glycolytic enzymes and glucose transporters (Chandel et al. 1998). This metabolic reprogramming supports the energy demands of activated microglia and perpetuates a pro-inflammatory state that contributes to neurodegenerative processes.
Another critical factor in microglial mitochondrial dysfunction is the dysregulation of calcium homeostasis. Mitochondria play a central role in buffering cytosolic calcium levels, and disruptions in this function can lead to altered microglial activation and inflammatory responses (Pivovarova and Andrews 2010). Specifically, excessive mitochondrial calcium uptake can cause mitochondrial permeability transition pore(mPTP) opening, leading to mitochondrial swelling, the release of pro-apoptotic factors, and cell death(Bernardi and Di Lisa 2015). This process can exacerbate neuroinflammation and neurodegeneration in the context of neurodegenerative diseases.
Furthermore, the impairment of mitophagy, the selective degradation of damaged mitochondria through autophagy, has been implicated in microglial mitochondrial dysfunction. Parkin, an E3 ubiquitin ligase, and PINK1, a mitochondrial kinase, are two key proteins involved in the initiation of mitophagy (Pickrell and Youle 2015). In the context of neurodegenerative diseases, mutations in these genes or dysregulation of their signaling pathways can lead to impaired mitophagy and the accumulation of dysfunctional mitochondria in microglia (Geisler et al. 2010). This results in increased ROS production, inflammation, and neuronal damage, further contributing to the progression of neurodegenerative disorders.
Lastly, the role of mitochondrial dynamics, including fusion and fission processes, in microglial mitochondrial dysfunction should not be overlooked. Dysregulation of mitochondrial dynamics can lead to an imbalance between fused and fragmented mitochondria, ultimately resulting in compromised mitochondrial function (Westermann 2010). Key proteins regulating mitochondrial dynamics, such as mitofusins(MFN1 and MFN2), optic atrophy 1(OPA1), and dynamin-related protein 1(DRP1), have been implicated in the pathogenesis of neurodegenerative diseases (Youle and van der Bliek 2012). Modulating the expression and activity of these proteins in microglia may offer potential therapeutic avenues for alleviating neuroinflammation and neurodegeneration.
In conclusion, mitochondrial dysfunction in microglia is a critical contributor to metabolic reprogramming and the pathogenesis of neurodegenerative diseases. A deeper understanding of the molecular mechanisms involved, including the shift towards glycolysis, dysregulation of calcium homeostasis, impaired mitophagy, and altered mitochondrial dynamics, is essential for the development of novel therapeutic strategies targeting these disorders.
Microglial Metabolic Responses to Acute Injury and Chronic Disease
Microglia cells exhibit diverse and context-dependent responses to different types of insults, including acute injuries and chronic diseases. These responses involve distinct metabolic reprogramming and activation states that can either promote tissue repair and recovery or exacerbate damage and inflammation. Here, we will discuss the differential microglial metabolic responses to acute injuries and chronic diseases.
Acute Injury
In response to acute injuries such as traumatic brain injury(TBI) or ischemic stroke, microglia rapidly activate and undergo metabolic reprogramming to fulfill their roles in the acute phase of injury response (Loane and Kumar 2016). During this phase, microglia adopt a mixed pro-/anti-inflammatory activation phenotype, characterized by the upregulation of both pro-inflammatory cytokines and anti-inflammatory factors (Hu et al. 2015). This mixed activation state facilitates the clearance of damaged cells and debris, as well as the promotion of tissue repair and angiogenesis (Iadecola and Anrather 2011).
Metabolically, acute injury-induced microglial activation is accompanied by a shift towards glycolysis to support the increased energy demands for phagocytosis, cytokine production, and migration (Gimeno-Bayón et al. 2014). Additionally, the activation of the PPP is enhanced to generate NADPH and nucleotide precursors required for cellular proliferation and ROS scavenging (Voloboueva et al. 2013). These metabolic adaptations enable microglia to efficiently perform their protective and reparative functions during the acute injury response.
Chronic Disease
In contrast to acute injury, chronic neurodegenerative diseases are characterized by persistent microglial activation and long-term metabolic reprogramming (Cherry et al. 2014). In these diseases, microglia predominantly exhibit the pro-inflammatory phenotype, which is associated with the production of neurotoxic factors, chronic inflammation, and neuronal damage (Tang and Le 2016).
The chronic activation of microglia in neurodegenerative diseases is driven by a sustained shift towards glycolysis and a reduction in OXPHOS (Orihuela et al. 2016). This metabolic profile supports the energy requirements for the production of pro-inflammatory cytokines and ROS, contributing to the perpetuation of neuroinflammation and neurodegeneration (Bernier et al. 2020a). Moreover, the dysregulation of other metabolic pathways, such as fatty acid oxidation and amino acid metabolism, has been reported in chronically activated microglia, further exacerbating the pathological processes in chronic neurodegenerative diseases (Jha et al. 2019).
In summary, microglial metabolic responses to insults differ between acute injuries and chronic diseases. Acute injuries trigger a transient activation of microglia with mixed pro-/anti-inflammatory phenotypes and metabolic adaptations geared towards tissue repair and recovery. In contrast, chronic neurodegenerative diseases involve sustained microglial activation with a pro-inflammatory phenotype and metabolic reprogramming that perpetuates neuroinflammation and neurodegeneration.
Microglial Metabolic Reprogramming in Neurodegenerative Diseases
The following section provides a comprehensive summary of the role of microglial metabolic reprogramming in the pathogenesis of neurodegenerative diseases, with a specific focus onaging, AD, PD, amyotrophic lateral sclerosis(ALS) and MS. Microglial metabolic changes have been shown to significantly influence the development and progression of these diseases, impacting their phenotypes and functions. In this context, we present a detailed analysis of the metabolic shifts that occur in microglia and how these changes contribute to the underlying pathology of AD, PD, ALS and MS (Table 1). By understanding the mechanisms and consequences of microglial metabolic reprogramming, we aim to shed light on potential therapeutic targets and strategies for the treatment of these devastating neurodegenerative disorders.
Table 1.
Metabolism reprogramming of microglia in neurodegeneration
Role of Microglial During Aging
As the brain ages, it exhibits a heightened state of inflammation, characterized by oxidative stress, an increase in pro-inflammatory mediators, and the development and progression of neurodegenerative conditions (Wendimu and Hooks 2022). This shift in the brain's inflammatory landscape is associated with microglial dysfunction (Hefendehl et al. 2014). Research has shown that during aging, microglial cells in the human brain undergo cellular senescence, impairing their proliferative response to challenges (Hua et al. 2012). This diminished capacity for specific microglial responses results in a preference for the pro-inflammatory phenotype over the anti-inflammatory phenotype (Lee et al. 2013). Therefore, the increasing inflammation in the aging brain and microglial senescence are likely interconnected phenomena. In both humans and animals, senescent and dystrophic microglial cells related to aging have been observed throughout the brain (Streit 2006; Caldeira et al. 2014; Ojo et al. 2015). These dystrophic microglia cells exhibit unique morphological alterations, which are not associated with the classical or alternative phenotypes. Microglia are known to experience a surge in mitotic activity following injury-induced activation, and subsequently undergo cell death to restore cell numbers to a steady state (Gehrmann and Banati 1995). The process of microglial activation places considerable metabolic demands on the cells, making activated microglia more susceptible to death than their non-activated counterparts. Over time, human microglia cells endure metabolic stress from various factors, leading to continuous mitotic cycles and, ultimately, replicative senescence. In vitro research has demonstrated that both rat and human microglia experience senescence due to progressive telomere shortening (Flanary and Streit 2004; Kronenberg et al. 2017). Consequently, the decline in microglial function could play a key role in the emergence of age-related neurodegenerative diseases.
Microglia are proficient at detecting changes in the CNS microenvironment, but their capacity for neuroprotection is believed to diminish with age (Soto et al. 2015). For instance, increased expression of theIL-6 receptor (Hopp et al. 2014) has been linked to cognitive impairment, and aged animals exhibit differences in physiological responses, pathophysiological characteristics, and behavioral outcomes following brain injury compared to their younger counterparts (Sandu et al. 2015). In the aged brain, microglia are polarized towards the pro-inflammatory phenotype, with a persistent impairment of anti-inflammatory responses after ischemic injury (Wynne et al. 2010; Wehrspaun et al. 2015). Furthermore, the ability of microglia to contribute to myelination, support oligodendrocyte differentiation, and facilitate remyelination is significantly impaired in older brains (Poliani et al. 2015). Taken together, these findings support the hypothesis that age-related increases in inflammatory responses may result from alterations in microglial activation and function (Block et al. 2007). Recent studies suggest that inflammation during aging can impact neuron–astrocyte–microglia interactions, which may play a role in the development of neurodegenerative diseases (Lana et al. 2016). Microglia have been implicated in the death of dopaminergic neurons in PD, forebrain neurons in AD, and motor neurons in ALS (Boillée et al. 2006; Mount et al. 2007). In addition, an age-related increase in microglial numbers in the human sub-ependymal zone has been associated with reduced cell proliferation and neuronal differentiation (Weissleder et al. 2016). Considering that PD, AD, and ALS are primarily age-related disorders, microglia play a crucial role in the pathogenesis of these neurodegenerative conditions.
Role of Microglial Metabolic Reprogramming in AD
AD pathogenesis is significantly influenced by microglial metabolic changes, which impact their phenotypes and functions (Sharkey 2021). A key histopathological aspect of AD is the accumulation of Aβ plaques, leading to an upregulation of pro-inflammatory molecules and a shift in microglia metabolism from OXPHOS to aerobic glycolysis (Rong et al. 2023). This alteration is associated with cytokine release and impaired microglial phagocytic ability (Cai et al. 2022). Acute exposure to Aβ boosts glycolysis and phagocytosis in microglia, while chronic exposure diminishes functionality in both areas (Sharkey 2021). Studies have demonstrated a link between glycolytic shift and reduced Aβ phagocytosis (McIntosh et al. 2019). The triggering receptor expressed on myeloid cells 2(TREM2) is essential for microglial Aβ phagocytosis (Lambert et al. 2013; Butovsky et al. 2014). TREM2 deficiency exacerbates Aβ plaque burden in the 5xFAD mouse model (Ulland et al. 2017), but has mixed effects in the APPPS1-21 AD model (Xing et al. 2015). TREM2 is crucial for maintaining microglial function and modulating Aβ-related AD pathology (Piers et al. 2020). Disease-associated microglia(DAM) represent a distinct microglial subset (Bloem et al. 2021), marked by downregulated “homeostatic” microglial genes (Butovsky et al. 2014) and upregulated AD risk factor-associated genes, including TREM2 (Lambert et al. 2013). TREM2 is vital for DAM activation (Bloem et al. 2021). Interestingly, metabolic shifts in microglia link TREM2 deficiency to the DAM profile. TREM2-deficient mice with AD-like pathology show energy metabolism defects, glycolytic defects, reduced ATP levels, impaired anabolic metabolism, and diminished mTOR activation (Harms et al. 2013). These effects might be related to DAP12, DAP10, and the PI3K/AKT-mTOR signaling pathways (Johri et al. 2013).Microglia with the AD-associated TREM2R47H variant have significant metabolic deficits, including reduced mitochondrial respiratory capacity and difficulty switching to glycolysis, leading to ATP production challenges (Dias et al. 2013). Consequently, TREM2-deficient microglia have trouble responding to stressors such as growth factor deprivation and Aβ toxicity, potentially due to metabolic defects (Dias et al. 2013). In addition to the critical role of TREM2 in microglial metabolic reprogramming, the inflammasome, particularly the NLRP3 inflammasome, has been implicated in AD pathology. The NLRP3 inflammasome is an innate immune system sensor that can be activated by various stimuli, including Aβ and oxidative stress (Heneka et al. 2013). Activation of the NLRP3 inflammasome in microglia leads to the release of pro-inflammatory cytokines, such as IL-1β and IL-18, which contribute to neuroinflammation and neuronal damage (Venegas et al. 2017). Metabolic reprogramming in microglia has been linked to the activation of the NLRP3 inflammasome, with glycolytic shifts promoting inflammasome activation (Mills et al. 2016). Inhibition of glycolysis in microglia has been shown to suppress NLRP3 inflammasome activation in response to Aβ, resulting in reduced neuroinflammation and neurodegeneration (Gomez Perdiguero et al. 2015). Moreover, mitochondrial dysfunction in microglia, characterized by increased ROS production, can also trigger the activation of the NLRP3 inflammasome, exacerbating AD-related pathology (Nakahira et al. 2011). Thus, targeting the inflammasome and associated metabolic changes in microglia may represent a potential therapeutic approach for AD.
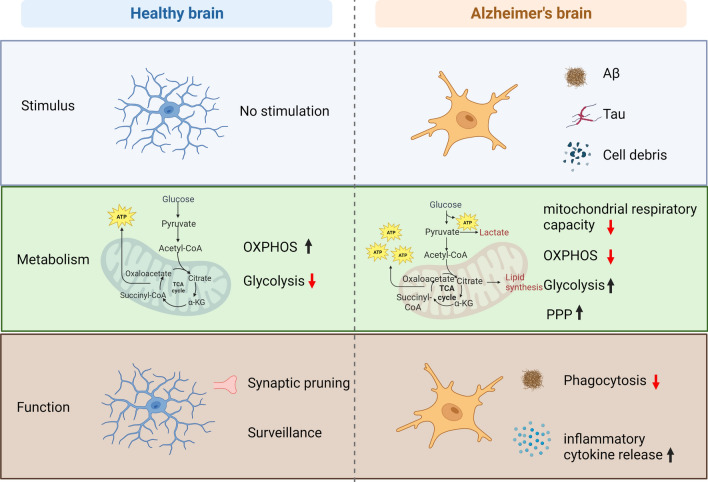
In summary, the metabolic reprogramming of microglia plays a pivotal role in AD pathogenesis, impacting microglial phenotypes and functions (Fig. 3). TREM2, in particular, acts as a nexus between metabolic profiles and AD disease outcomes, involving the activation of DAM. Further research is required to elucidate the mechanisms underlying microglial metabolic reprogramming in AD pathology.
Fig. 3.
Metabolic Reprogramming of Microglia inAD In AD, microglia are exposed to various stimuli, including Aβplaques, Tau tangles, and cellular debris. As a result, microglial cells undergo metabolic reprogramming to adapt to these challenging conditions.The metabolic shift in AD microglia involves changes in energy production pathways, such as a transition from OXPHOS to glycolysis, enhanced PPP and mitochondrial respiratory capacity. These metabolic adaptations in microglia lead to altered cellular functions, including amplified inflammatory responses and diminished phagocytic capacity
Microglial Metabolic Reprogramming in PD
PD is a widespread neurodegenerative disorder marked by the progressive loss of dopaminergic neurons in the substantia nigra pars compacta(SNpc) and the build-up of α-synuclein(α-syn) aggregates (Rostami et al. 2017). Microglial metabolic shifts have been noted in both in vitro and in vivo PD models. In a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced mouse model of PD, microglia displayed increased glycolytic and reduced OXPHOS markers (Zhang et al. 2004). This metabolic shift in microglia is linked to the release of pro-inflammatory substances like cytokines and chemokines, potentially contributing to dopaminergic neuron loss in PD (Huang et al. 2018). By inhibiting glycolysis in microglia, ROS production is reduced, and dopaminergic neurons are protected in an LPS-induced rat PD model (Wang et al. 2021). Another study showed that α-syn-enhanced NADPH oxidase 2(NOX2)-mediated ROS production in microglia led to neurotoxicity in dopaminergic neurons (Glass et al. 2010). Impaired microglial mitochondrial function may also play a role in neuroinflammation and neuronal damage in PD. Johri et al. found that microglial mitochondrial dysfunction caused pro-inflammatory cytokine release and amplified α-syn-induced toxicity in dopaminergic neurons (Volonté et al. 2016). Dysfunctional mitochondria may further contribute to ROS production, intensifying oxidative stress in PD (Philips and Rothstein 2014). The presence of α-syn in microglia has been shown to cause metabolic shifts, resulting in neuroinflammation and the degeneration of dopaminergic neurons. Extracellular α-syn fibrils triggered a glycolytic shift in primary microglia, leading to increased pro-inflammatory cytokines and ROS production (Cassina et al. 2021). Additionally, α-syn activated the NLRP3 inflammasome in microglia, causing the release of pro-inflammatory cytokines and damage to dopaminergic neurons (Perera and Turner 2016). Microglial metabolic shifts may also influence other cell types within the CNS, further promoting PD progression. Astrocytes, for instance, can experience metabolic changes in response to microglial activation, exacerbating neuroinflammation and neurodegeneration (Meissner et al. 2010). Activated microglia induced metabolic changes in astrocytes, resulting in increased pro-inflammatory mediators and disrupted astrocyte-neuron metabolic interactions (Martinez et al. 2011). This evidence suggests that microglial metabolic shifts may indirectly contribute to neuronal damage in PD through interactions with other CNS cell types. As mentioned earlier, α-syn activates the NLRP3 inflammasome in microglia, which contributes to dopaminergic neuron damage in PD (Perera and Turner 2016). The activation of the NLRP3 inflammasome in microglia has been shown to be closely associated with metabolic alterations. A study by Yang et al. demonstrated that α-syn-induced NLRP3 inflammasome activation in microglia is dependent on glycolytic reprogramming, with increased lactate production and glycolytic enzyme expression (Yang et al. 2014). Inhibition of glycolysis in microglia was found to attenuate α-syn-induced NLRP3 inflammasome activation and subsequent neuroinflammation, suggesting that targeting glycolytic reprogramming may be a potential therapeutic strategy for PD (Gordon et al. 2018). Furthermore, mitochondrial dysfunction and oxidative stress in microglia, as a result of metabolic reprogramming, can also activate the NLRP3 inflammasome, further promoting neuroinflammation and dopaminergic neuron damage in PD (Baldwin et al. 2016).
In conclusion, microglial metabolic reprogramming is a critical factor in PD pathogenesis. The transition from OXPHOS to aerobic glycolysis in microglia leads to the release of pro-inflammatory mediators and ROS, contributing to dopaminergic neuron loss. Furthermore, microglial mitochondrial dysfunction exacerbates neuroinflammation and oxidative stress. The presence of α-syn in microglia triggers metabolic reprogramming, resulting in neuroinflammation and dopaminergic neuron degeneration. Moreover, microglial metabolic shifts may affect other CNS cell types, such as astrocytes, contributing to PD progression.
Microglial Metabolic Reprogramming in ALS
Like in PD, microglia in ALS display a metabolic shift from OXPHOS to aerobic glycolysis, resulting in a pro-inflammatory and neurotoxic state (Fendrick et al. 2007). This alteration is believed to contribute to the ongoing motor neuron degeneration in ALS. ALS-related superoxide dismutase 1(SOD1) mutations induce an inflammatory microglial phenotype through metabolic reprogramming, characterized by heightened glycolytic activity and mitochondrial dysfunction (Palazon et al. 2014). Glycolytic enzymes, such as hexokinase-2 and lactate dehydrogenase, are upregulated in mutant SOD1 microglia, exacerbating neuroinflammation and motor neuron death in ALS. Additionally, microglia expressing the ALS-linked C9orf72 mutation show metabolic imbalance, with increased glycolysis and reduced mitochondrial respiration (Saxton and Sabatini 2017). This metabolic reprogramming triggers the release of pro-inflammatory cytokines like IL-1β and TNF-α, resulting in neurotoxicity in ALS. Microglia from the spinal cords of SOD1 mutant mice exhibit enhanced activation of the mammalian target of rapamycin(mTOR) pathway, involved in regulating cellular metabolism (Cantuti-Castelvetri et al. 2018). This mTOR pathway activation in microglia leads to increased glycolysis and reduced oxidative phosphorylation, contributing to the observed pro-inflammatory phenotype and neurotoxicity in ALS.
Moreover, NLRP3 inflammasome activation has been reported in ALS microglia, causing the release of pro-inflammatory cytokines and exacerbating neuroinflammation (Joshi et al. 2019). This activation is connected to microglial metabolic changes, as it relies on glycolytic activity. Microglial metabolic shifts are also implicated in autophagy impairment in ALS. Defective autophagy is an ALS hallmark, and microglial metabolic reprogramming towards glycolysis impairs autophagic flux, leading to toxic protein aggregate accumulation in microglia (Park et al. 2013). Furthermore, the role of astrocytes in metabolic changes and their interaction with microglia in ALS has been explored. Reactive astrocytes in ALS mouse models release factors that stimulate pro-inflammatory and neurotoxic microglial activation, contributing to motor neuron death (Zhong et al. 2019).
In conclusion, microglial metabolic reprogramming in ALS involves a transition from oxidative phosphorylation to aerobic glycolysis, resulting in a pro-inflammatory and neurotoxic state. This metabolic change is linked to various factors, such as SOD1 and C9orf72 mutations, mTOR pathway activation, NLRP3 inflammasome activation, autophagy impairment, and reactive astrocyte influence. Together, these factors contribute to the progressive motor neuron degeneration in ALS and intensify neuroinflammation.
Microglial Metabolic Reprogramming in MS
The glycolytic shift in microglia is marked by the upregulation of essential glycolytic enzymes, such as hexokinase-2(HK2) and lactate dehydrogenase A(LDHA) (Van den Bossche et al. 2016). Recent research indicates that the mTOR/HIF-1α axis activation is a primary contributor to this metabolic change (Park et al. 2015). For example, studies have shown that pharmacological mTOR inhibition using rapamycin leads to decreased HIF-1α stabilization and diminished glycolytic gene expression in microglia (Du et al. 2017). These results emphasize the critical role of the mTOR/HIF-1α signaling pathway in microglial metabolic reprogramming in MS. Regarding lipid metabolism, recent investigations have revealed that elevated cholesterol 25-hydroxylase(CH25H) levels in microglia during demyelination are associated with increased lipid droplet formation and modified cholesterol metabolism (Hansen et al. 2018). Mitochondrial dysfunction is another crucial aspect of microglial metabolic reprogramming in MS. A recent study demonstrated that mitochondrial calcium uniporter(MCU) expression is upregulated in microglia from MS patients and EAE models, resulting in heightened mitochondrial calcium uptake and oxidative stress. Moreover, inhibiting Drp1, a protein involved in mitochondrial fission, improved mitochondrial dysfunction in microglia and reduced neuroinflammation in EAE mice (Subramaniam and Federoff 2017). Additionally, metabolic reprogramming has been shown to influence microglial polarization towards pro-inflammatory or anti-inflammatory phenotypes (Wang et al. 2021). For example, a recent study found that glycolysis inhibition using 2-deoxy-D-glucose (2-DG) led to reduced pro-inflammatory polarization and suppressed pro-inflammatory cytokine production in LPS-stimulated microglia (Gao and Hong 2011). These findings emphasize the intricate relationship between metabolism and microglial polarization in MS.
In conclusion, microglial metabolic reprogramming in MS encompasses a glycolytic shift, changes in lipid metabolism, and mitochondrial dysfunction. Recent studies have provided valuable insights into the molecular mechanisms underlying these processes, implicating the mTOR/HIF-1α axis, PPARγ signaling, and mitochondrial dynamics as crucial factors in microglial reprogramming (Mado et al. 2023). A thorough understanding of these pathways and their functional outcomes may reveal potential therapeutic targets for MS intervention. However, further research is needed to completely clarify their roles in disease pathogenesis.
Benefits of Modulating Microglial Metabolism in Neurodegenerative Diseases
The manipulation of microglial metabolism has gained significant interest as a potential therapeutic strategy for treating neurodegenerative diseases, given its capacity to decrease neuroinflammation, protect neurons, and decelerate disease progression (Bernier et al. 2020b). By targeting crucial metabolic pathways, including glycolysis, PPP, and oxidative phosphorylation, it may be feasible to control microglial activation and polarization, ultimately reducing neuroinflammation and neuronal damage (Miron et al. 2013; Wang et al. 2021).Adjusting microglial metabolism can impact their functional phenotype, potentially transitioning them from pro-inflammatory to anti-inflammatory state. This transformation could foster tissue repair, diminish inflammation, and contribute to resolving neuroinflammation in neurodegenerative diseases (Du et al. 2017). For example, in AD, adjusting microglial metabolism may help decrease Aβ deposition by boosting microglial phagocytic capabilities (Simon et al. 2017). Moreover, modified microglial metabolism might enhance synaptic function and minimize neurotoxicity, potentially slowing cognitive decline in AD patients (Bogie et al. 2014). In PD, targeting microglial metabolism could protect dopaminergic neurons from degeneration, thus, alleviating motor symptoms (Subramaniam and Federoff 2017). Through the modulation of microglial activation and polarization, metabolic interventions could suppress pro-inflammatory cytokine and ROS release (Yamanaka et al. 2012). In ALS, adjusting microglial metabolism could decrease neuroinflammation and prevent motor neuron loss (Heneka et al. 2015). In MS, interventions targeting microglial metabolism might promote remyelination by moving microglia towards an anti-inflammatory phenotype, subsequently supporting oligodendrocyte differentiation and myelin repair (Patil et al. 2014). Importantly, the advantages of modulating microglial metabolism could extend beyond neurodegenerative diseases. In ischemic stroke, for instance, metabolic interventions targeting microglia may reduce inflammation and facilitate brain injury resolution by regulating microglial activation and polarization (Bayliss et al. 2016). In traumatic brain injury (TBI), adjusting microglial metabolism could help minimize secondary injury by decreasing pro-inflammatory mediator production and promoting tissue repair processes (Malagelada et al. 2010).
In summary, adjusting microglial metabolism offers a promising therapeutic approach for treating neurodegenerative diseases, with the potential to decrease neuroinflammation, safeguard neurons, and decelerate disease progression. Nevertheless, further research is required to comprehensively understand the underlying mechanisms, pinpoint the most pertinent metabolic pathways and molecular targets, and optimize intervention timing and duration. With ongoing advancements in the field, targeting microglial metabolism may eventually lead to the development of innovative and effective therapies for various neurodegenerative diseases.
Therapeutic Potential of Targeting Microglial Metabolic Reprogramming
Pharmacological agents targeting microglial metabolism have shown promising results in preclinical models of neurodegenerative diseases. For example, the PPARγ agonist pioglitazone improved cognitive deficits and diminished neuroinflammation in AD and MS animal models (Zhang et al. 2011; Longo and Mattson 2014). Pioglitazone treatment resulted in a 40% reduction in Aβ levels and enhanced spatial learning in an AD mouse model (Halagappa et al. 2007). Metformin, an AMPK activator, has been observed to promote microglial anti-inflammatory polarization and alleviate cognitive deficits in an AD mouse model(APP/PS1 mice) by decreasing amyloid plaque load and pro-inflammatory cytokine production (Kohman et al. 2013). Metformin treatment led to a 50% reduction in Aβ plaques and a 30% decrease in pro-inflammatory cytokines. Furthermore, metformin treatment improved motor function in a rotenone-induced PD mouse model through the activation of autophagy and suppression of mTOR signaling (Speisman et al. 2013). Another promising strategy involves targeting the mTOR pathway, which modulates microglial metabolism and activation. Rapamycin, an mTOR inhibitor, exhibited neuroprotective effects in an MPTP-induced PD mouse model by reducing dopaminergic neuron loss and α-synuclein aggregation (Nichol et al. 2008). Rapamycin treatment in this model led to a 60% reduction in α-synuclein aggregates and a 20% improvement in motor function. In an ALS model, rapamycin treatment extended survival and delayed disease onset in SOD1-G93A mice, potentially by decreasing neuroinflammation and promoting autophagy (O’Neill et al. 2016). Lifestyle modifications, such as dietary interventions and exercise, have also demonstrated potential in modulating microglial metabolism. Caloric restriction and intermittent fasting reduced neuroinflammation, Aβ plaque burden, and α-synuclein aggregation in transgenic mouse models of AD (APP/PS1) and PD(A53T-α-synuclein) (Hammond et al. 2019). Mice subjected to caloric restriction experienced a 45% reduction in pro-inflammatory cytokines and a 30% increase in anti-inflammatory cytokines, along with improved memory performance (Pardridge 2017). Physical exercise has shown benefits in augmenting microglial surveillance and fostering a neuroprotective environment in the aging brain (Colonna and Butovsky 2017). A study found that voluntary wheel running in aged rats led to a 25% increase in microglial surveillance capacity and a 40% reduction in pro-inflammatory markers, suggesting potential benefits of exercise in adjusting microglial metabolism (Erdmann et al. 2016). Additionally, voluntary wheel running enhanced cognitive function and reduced microglial activation in a transgenic mouse model of AD (Tg2576 mice) (Laplante and Sabatini 2012).
In conclusion, these studies highlight the potential advantages of targeting microglial metabolism through pharmacological interventions and lifestyle modifications in neurodegenerative diseases. Promising results from animal models, such as the reduction of Aβ levels and improvement in cognitive and motor functions, underscore the need for further research to translate these findings into clinical applications. Despite the promising results from preclinical models, challenges remain in developing therapies targeting microglial metabolic reprogramming. Understanding the complexity of microglial function, the multiple signaling pathways and molecular targets involved, potential side effects and safety concerns, and patient compliance and accessibility to lifestyle interventions is necessary. Future directions include identifying.
Conclusions
Microglial metabolic reprogramming has emerged as a critical factor in the pathogenesis of neurodegenerative diseases (Hammond et al. 2019). Understanding the molecular and cellular mechanisms underlying microglial metabolic reprogramming is essential for uncovering novel therapeutic targets and strategies. Despite the promising findings, several challenges remain in developing therapies targeting microglial metabolic reprogramming. To accelerate progress in this field, it is essential to adopt innovative perspectives and incorporate personal insights into the design of new strategies. One potential innovative approach is the integration of multi-omics and systems biology techniques to gain a more comprehensive understanding of microglial metabolism in the context of neurodegenerative diseases. This integrative approach could help elucidate the complex interactions between metabolism, inflammation, and neurodegeneration, leading to the development of more specific and effective therapies (Pardridge 2017). The development of targeted drug delivery systems to enhance the specificity and efficacy of therapeutic agents is another innovative perspective (Colonna and Butovsky 2017). By designing nanoparticles or other delivery systems that selectively target microglial populations, researchers could minimize off-target effects and potential side effects associated with systemic administration of drugs. Moreover, these targeted delivery systems could overcome the challenges posed by the BBB, allowing for the direct delivery of therapeutics to the CNS. Drug repurposing, or the identification of new therapeutic applications for existing drugs, could be a promising approach to accelerate the development of therapies targeting microglial metabolic reprogramming (Erdmann et al. 2016). Repurposing such drugs could save time and resources in the drug development process and facilitate the translation of preclinical findings into clinical applications, such as the use of the anti-diabetic drug metformin in AD and PD (Laplante and Sabatini 2012). The development of personalized medicine approaches could help optimize the targeting of microglial metabolic reprogramming in neurodegenerative diseases. By identifying patient-specific molecular signatures and microglial subpopulations, researchers could design-tailored interventions that selectively target the most relevant pathways and cell populations in each patient. Combining pharmacological interventions with lifestyle modifications, such as dietary changes and physical exercise, could offer synergistic effects in modulating microglial metabolism and neuroinflammation (Erdmann et al. 2016). By integrating multiple strategies, researchers could maximize the therapeutic benefits and potentially slow disease progression more effectively than single interventions.
In summary, addressing the challenges in microglial metabolic reprogramming requires a multidisciplinary approach, combining in-depth mechanistic studies, advanced drug delivery systems, drug repurposing, personalized medicine, and combination therapies. By exploring novel approaches and incorporating personal insights, the potential of targeting microglial metabolic reprogramming as a therapeutic strategy for neurodegenerative diseases could be fully realized.
Acknowledgements
Not applicable.
Author Contributions
JM designed the review structure and wrote the main sections of the manuscript. LC, XP, and LL contributed to the literature search, data analysis, and interpretation of the findings. BZ prepared the figures and tables. JL, as the corresponding author, supervised the project and provided critical revisions to the manuscript. All authors reviewed and approved the final version of the manuscript for publication.
Funding
The authors have not disclosed any funding.
Data Availability
Enquiries about data availability should be directed to the authors.
Declarations
Conflict of interest
The authors whose names are listed immediately below certify that they have NO affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Beibei Zhao, Email: zhaobeibei@szbazyy.net.
Jiao Lan, Email: lanjiao@szbazyy.net.
References
- Baik SH, Kang S, Lee W et al (2019) A breakdown in metabolic reprogramming causes microglia dysfunction in Alzheimer’s disease. Cell Metab 30:493–507. 10.1016/j.cmet.2019.06.005 [DOI] [PubMed] [Google Scholar]
- Baldwin AG, Brough D, Freeman S (2016) Inhibiting the inflammasome: a chemical perspective. J Med Chem 59:1691–1710. 10.1021/acs.jmedchem.5b01091 [DOI] [PubMed] [Google Scholar]
- Bayliss JA, Lemus MB, Santos VV et al (2016) Metformin prevents nigrostriatal dopamine degeneration independent of AMPK activation in dopamine neurons. PloS one 11:e0159381. 10.1371/journal.pone.0159381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beins E, Ulas T, Ternes S et al (2016) Characterization of inflammatory markers and transcriptome profiles of differentially activated embryonic stem cell-derived microglia. Glia 64:1007–1020. 10.1002/glia.22979 [DOI] [PubMed] [Google Scholar]
- Bernardi P, Di Lisa F (2015) The mitochondrial permeability transition pore: molecular nature and role as a target in cardioprotection. J Mol Cell Cardiol 78:100–106. 10.1016/j.yjmcc.2014.09.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernier L-P, Bohlen CJ, York EM et al (2019) Nanoscale surveillance of the brain by microglia via cAMP-regulated filopodia. Cell Rep 27:2895-2908.e4. 10.1016/j.celrep.2019.05.010 [DOI] [PubMed] [Google Scholar]
- Bernier L-P, York EM, Kamyabi A et al (2020a) Microglial metabolic flexibility supports immune surveillance of the brain parenchyma. Nat Commun 11:1559. 10.1038/s41467-020-15267-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernier L-P, York EM, MacVicar BA (2020b) Immunometabolism in the brain: How metabolism shapes microglial function. Trends Neurosci 43:854–869. 10.1016/j.tins.2020.08.008 [DOI] [PubMed] [Google Scholar]
- Block ML, Zecca L, Hong J-S (2007) Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci 8:57–69. 10.1038/nrn2038 [DOI] [PubMed] [Google Scholar]
- Bloem BR, Okun MS, Klein C (2021) Parkinson’s disease. Lancet (london, England) 397:2284–2303. 10.1016/S0140-6736(21)00218-X [DOI] [PubMed] [Google Scholar]
- Bogie JFJ, Stinissen P, Hendriks JJA (2014) Macrophage subsets and microglia in multiple sclerosis. Acta Neuropathol 128:191–213. 10.1007/s00401-014-1310-2 [DOI] [PubMed] [Google Scholar]
- Boillée S, Yamanaka K, Lobsiger CS et al (2006) Onset and progression in inherited ALS determined by motor neurons and microglia. Science 312:1389–1392. 10.1126/science.1123511 [DOI] [PubMed] [Google Scholar]
- Bolanos JP, García-Nogales P, Almeida A (2004) Provoking neuroprotection by peroxynitrite. Curr Pharm Des 10:867–877. 10.2174/1381612043452910 [DOI] [PubMed] [Google Scholar]
- Butovsky O, Jedrychowski MP, Moore CS et al (2014) Identification of a unique TGF-β-dependent molecular and functional signature in microglia. Nat Neurosci 17:131–143. 10.1038/nn.3599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai Y, Liu J, Wang B et al (2022) Microglia in the neuroinflammatory pathogenesis of Alzheimer’s disease and related therapeutic targets. Front Immunol 13:856376. 10.3389/fimmu.2022.856376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caldeira C, Oliveira AF, Cunha C et al (2014) Microglia change from a reactive to an age-like phenotype with the time in culture. Front Cell Neurosci 8:152. 10.3389/fncel.2014.00152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantuti-Castelvetri L, Fitzner D, Bosch-Queralt M et al (2018) Defective cholesterol clearance limits remyelination in the aged central nervous system. Science 359:684–688. 10.1126/science.aan4183 [DOI] [PubMed] [Google Scholar]
- Carvalho AN, Firuzi O, Gama MJ et al (2017) Oxidative stress and antioxidants in neurological diseases: is there still hope? Curr Drug Targets 18:705–718. 10.2174/1389450117666160401120514 [DOI] [PubMed] [Google Scholar]
- Cassina P, Miquel E, Martínez-Palma L, Cassina A (2021) Glial metabolic reprogramming in amyotrophic lateral sclerosis. NeuroImmunoModulation 28:204–212. 10.1159/000516926 [DOI] [PubMed] [Google Scholar]
- Chandel NS, Maltepe E, Goldwasser E et al (1998) Mitochondrial reactive oxygen species trigger hypoxia-induced transcription. Proc Natl Acad Sci USA 95:11715–11720. 10.1073/pnas.95.20.11715 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang PK-Y, Khatchadourian A, McKinney RA, Maysinger D (2015) Docosahexaenoic acid (DHA): a modulator of microglia activity and dendritic spine morphology. J Neuroinflammation 12:34. 10.1186/s12974-015-0244-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C-J, Ou Y-C, Chang C-Y et al (2012a) Glutamate released by Japanese encephalitis virus-infected microglia involves TNF-α signaling and contributes to neuronal death. Glia 60:487–501. 10.1002/glia.22282 [DOI] [PubMed] [Google Scholar]
- Chen X, Guo C, Kong J (2012b) Oxidative stress in neurodegenerative diseases. Neural Regen Res 7:376–385. 10.3969/j.issn.1673-5374.2012.05.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherry JD, Olschowka JA, O’Banion MK (2014) Neuroinflammation and M2 microglia: the good, the bad, and the inflamed. J Neuroinflammation 11:98. 10.1186/1742-2094-11-98 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choudhury ME, Kigami Y, Tanaka J (2021) Dual Roles of microglia in the basal ganglia in Parkinson’s disease. Int J Mol Sci. 10.3390/ijms22083907 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colonna M, Butovsky O (2017) Microglia function in the central nervous system during health and neurodegeneration. Annu Rev Immunol 35:441–468. 10.1146/annurev-immunol-051116-052358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornell J, Salinas S, Huang H-Y, Zhou M (2022) Microglia regulation of synaptic plasticity and learning and memory. Neural Regen Res 17:705–716. 10.4103/1673-5374.322423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai X, Li N, Yu L et al (2015) Activation of BV2 microglia by lipopolysaccharide triggers an inflammatory reaction in PC12 cell apoptosis through a toll-like receptor 4-dependent pathway. Cell Stress Chaperones 20:321–331. 10.1007/s12192-014-0552-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Vitto H, Pérez-Valencia J, Radosevich JA (2016) Glutamine at focus: versatile roles in cancer. Tumour Biol J Int Soc Oncodevelopmental Biol Med 37:1541–1558. 10.1007/s13277-015-4671-9 [DOI] [PubMed] [Google Scholar]
- Dias V, Junn E, Mouradian MM (2013) The role of oxidative stress in Parkinson’s disease. J Parkinsons Dis 3:461–491. 10.3233/JPD-130230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du L, Zhang Y, Chen Y et al (2017) Role of microglia in neurological disorders and their potentials as a therapeutic target. Mol Neurobiol 54:7567–7584. 10.1007/s12035-016-0245-0 [DOI] [PubMed] [Google Scholar]
- Duffy CM, Yuan C, Wisdorf LE et al (2015) Role of orexin A signaling in dietary palmitic acid-activated microglial cells. Neurosci Lett 606:140–144. 10.1016/j.neulet.2015.08.033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duffy CM, Xu H, Nixon JP et al (2017) Identification of a fatty acid binding protein4-UCP2 axis regulating microglial mediated neuroinflammation. Mol Cell Neurosci 80:52–57. 10.1016/j.mcn.2017.02.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ebert D, Haller RG, Walton ME (2003) Energy contribution of octanoate to intact rat brain metabolism measured by 13C nuclear magnetic resonance spectroscopy. J Neurosci 23:5928–5935. 10.1523/JNEUROSCI.23-13-05928.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erdmann E, Harding S, Lam H, Perez A (2016) Ten-year observational follow-up of PROactive: a randomized cardiovascular outcomes trial evaluating pioglitazone in type 2 diabetes. Diabetes Obes Metab 18:266–273. 10.1111/dom.12608 [DOI] [PubMed] [Google Scholar]
- Felipo V, Butterworth RF (2002) Neurobiology of ammonia. Prog Neurobiol 67:259–279. 10.1016/s0301-0082(02)00019-9 [DOI] [PubMed] [Google Scholar]
- Fendrick SE, Xue Q-S, Streit WJ (2007) Formation of multinucleated giant cells and microglial degeneration in rats expressing a mutant Cu/Zn superoxide dismutase gene. J Neuroinflammation 4:9. 10.1186/1742-2094-4-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez RF, Kim SQ, Zhao Y et al (2018) Acyl-CoA synthetase 6 enriches the neuroprotective omega-3 fatty acid DHA in the brain. Proc Natl Acad Sci USA 115:12525–12530. 10.1073/pnas.1807958115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flanary BE, Streit WJ (2004) Progressive telomere shortening occurs in cultured rat microglia, but not astrocytes. Glia 45:75–88. 10.1002/glia.10301 [DOI] [PubMed] [Google Scholar]
- Gao H-M, Hong J-S (2011) Gene-environment interactions: key to unraveling the mystery of Parkinson’s disease. Prog Neurobiol 94:1–19. 10.1016/j.pneurobio.2011.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao H-M, Zhou H, Hong J-S (2012) NADPH oxidases: novel therapeutic targets for neurodegenerative diseases. Trends Pharmacol Sci 33:295–303. 10.1016/j.tips.2012.03.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao Y, Ottaway N, Schriever SC et al (2014) Hormones and diet, but not body weight, control hypothalamic microglial activity. Glia 62:17–25. 10.1002/glia.22580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gehrmann J, Banati RB (1995) Microglial turnover in the injured CNS: activated microglia undergo delayed DNA fragmentation following peripheral nerve injury. J Neuropathol Exp Neurol 54:680–688. 10.1097/00005072-199509000-00010 [DOI] [PubMed] [Google Scholar]
- Geisler S, Holmström KM, Skujat D et al (2010) PINK1/Parkin-mediated mitophagy is dependent on VDAC1 and p62/SQSTM1. Nat Cell Biol 12:119–131. 10.1038/ncb2012 [DOI] [PubMed] [Google Scholar]
- Geloso MC, Corvino V, Marchese E et al (2017) The dual role of microglia in ALS: mechanisms and therapeutic approaches. Front Aging Neurosci 9:242. 10.3389/fnagi.2017.00242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh S, Castillo E, Frias ES, Swanson RA (2018) Bioenergetic regulation of microglia. Glia 66:1200–1212. 10.1002/glia.23271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gimeno-Bayón J, López-López A, Rodríguez MJ, Mahy N (2014) Glucose pathways adaptation supports acquisition of activated microglia phenotype. J Neurosci Res 92:723–731. 10.1002/jnr.23356 [DOI] [PubMed] [Google Scholar]
- Glass CK, Saijo K, Winner B et al (2010) Mechanisms underlying inflammation in neurodegeneration. Cell 140:918–934. 10.1016/j.cell.2010.02.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez Perdiguero E, Klapproth K, Schulz C et al (2015) Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature 518:547–551. 10.1038/nature13989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gordon R, Albornoz EA, Christie DC et al (2018) Inflammasome inhibition prevents α-synuclein pathology and dopaminergic neurodegeneration in mice. Sci Transl Med. 10.1126/scitranslmed.aah4066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halagappa VKM, Guo Z, Pearson M et al (2007) Intermittent fasting and caloric restriction ameliorate age-related behavioral deficits in the triple-transgenic mouse model of Alzheimer’s disease. Neurobiol Dis 26:212–220. 10.1016/j.nbd.2006.12.019 [DOI] [PubMed] [Google Scholar]
- Hamilton JA, Brunaldi K (2007) A model for fatty acid transport into the brain. J Mol Neurosci 33:12–17. 10.1007/s12031-007-0050-3 [DOI] [PubMed] [Google Scholar]
- Hammond TR, Dufort C, Dissing-Olesen L et al (2019) Single-cell RNA sequencing of microglia throughout the mouse lifespan and in the injured brain reveals complex cell-state changes. Immunity 50:253-271.e6. 10.1016/j.immuni.2018.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han B, Jiang W, Cui P et al (2021) Microglial PGC-1α protects against ischemic brain injury by suppressing neuroinflammation. Genome Med 13:47. 10.1186/s13073-021-00863-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hannun YA, Obeid LM (2018) Sphingolipids and their metabolism in physiology and disease. Nat Rev Mol Cell Biol 19:175–191. 10.1038/nrm.2017.107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen DV, Hanson JE, Sheng M (2018) Microglia in Alzheimer’s disease. J Cell Biol 217:459–472. 10.1083/jcb.201709069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harms AS, Cao S, Rowse AL et al (2013) MHCII is required for α-synuclein-induced activation of microglia, CD4 T cell proliferation, and dopaminergic neurodegeneration. J Neurosci off J Soc Neurosci 33:9592–9600. 10.1523/JNEUROSCI.5610-12.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hefendehl JK, Neher JJ, Sühs RB et al (2014) Homeostatic and injury-induced microglia behavior in the aging brain. Aging Cell 13:60–69. 10.1111/acel.12149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heneka MT, Kummer MP, Stutz A et al (2013) NLRP3 is activated in Alzheimer’s disease and contributes to pathology in APP/PS1 mice. Nature 493:674–678. 10.1038/nature11729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heneka M, Carson M, El Khoury J, Landreth G (2015) Neuroinflammation in Alzheimer’s disease. Lancet Neurol 14:388–405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hopp SC, D’Angelo HM, Royer SE et al (2014) Differential rescue of spatial memory deficits in aged rats by L-type voltage-dependent calcium channel and ryanodine receptor antagonism. Neuroscience 280:10–18. 10.1016/j.neuroscience.2014.09.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu X, Leak RK, Shi Y et al (2015) Microglial and macrophage polarization—new prospects for brain repair. Nat Rev Neurol 11:56–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hua K, Schindler MK, McQuail JA et al (2012) Regionally distinct responses of microglia and glial progenitor cells to whole brain irradiation in adult and aging rats. PloS one 7:e52728. 10.1371/journal.pone.0052728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang Y, Zhao L, Jia B et al (2011) Glutaminase dysregulation in HIV-1-infected human microglia mediates neurotoxicity: relevant to HIV-1-associated neurocognitive disorders. J Neurosci off J Soc Neurosci 31:15195–15204. 10.1523/JNEUROSCI.2051-11.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang Y, Xu Z, Xiong S et al (2018) Repopulated microglia are solely derived from the proliferation of residual microglia after acute depletion. Nat Neurosci 21:530–540. 10.1038/s41593-018-0090-8 [DOI] [PubMed] [Google Scholar]
- Iadecola C, Anrather J (2011) The immunology of stroke: from mechanisms to translation. Nat Med 17:796–808. 10.1038/nm.2399 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jay TR, Miller CM, Cheng PJ et al (2015) TREM2 deficiency eliminates TREM2+ inflammatory macrophages and ameliorates pathology in Alzheimer’s disease mouse models. J Exp Med 212:287–295. 10.1084/jem.20142322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jha AK, Huang SC-C, Sergushichev A et al (2015) Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization. Immunity 42:419–430. 10.1016/j.immuni.2015.02.005 [DOI] [PubMed] [Google Scholar]
- Jha MK, Jo M, Kim J-H, Suk K (2019) Microglia-astrocyte crosstalk: an intimate molecular conversation. Neurosci a Rev J Bringing Neurobiol Neurol Psychiatry 25:227–240. 10.1177/1073858418783959 [DOI] [PubMed] [Google Scholar]
- Jin L-W, Horiuchi M, Wulff H et al (2015) Dysregulation of glutamine transporter SNAT1 in Rett syndrome microglia: a mechanism for mitochondrial dysfunction and neurotoxicity. J Neurosci off J Soc Neurosci 35:2516–2529. 10.1523/JNEUROSCI.2778-14.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johri A, Chandra A, Flint Beal M (2013) PGC-1α, mitochondrial dysfunction, and Huntington’s disease. Free Radic Biol Med 62:37–46. 10.1016/j.freeradbiomed.2013.04.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joshi AU, Minhas PS, Liddelow SA et al (2019) Fragmented mitochondria released from microglia trigger A1 astrocytic response and propagate inflammatory neurodegeneration. Nat Neurosci 22:1635–1648. 10.1038/s41593-019-0486-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly B, O’Neill LAJ (2015) Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res 25:771–784. 10.1038/cr.2015.68 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim Y-C, Park TY, Baik E, Lee SH (2012) Fructose-1,6-bisphosphate attenuates induction of nitric oxide synthase in microglia stimulated with lipopolysaccharide. Life Sci 90:365–372. 10.1016/j.lfs.2011.12.011 [DOI] [PubMed] [Google Scholar]
- Klimaszewska-Łata J, Gul-Hinc S, Bielarczyk H et al (2015) Differential effects of lipopolysaccharide on energy metabolism in murine microglial N9 and cholinergic SN56 neuronal cells. J Neurochem 133:284–297. 10.1111/jnc.12979 [DOI] [PubMed] [Google Scholar]
- Kohman RA, Bhattacharya TK, Wojcik E, Rhodes JS (2013) Exercise reduces activation of microglia isolated from hippocampus and brain of aged mice. J Neuroinflammation 10:114. 10.1186/1742-2094-10-114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konishi H, Kiyama H, Ueno M (2019) Dual functions of microglia in the formation and refinement of neural circuits during development. Int J Dev Neurosci off J Int Soc Dev Neurosci 77:18–25. 10.1016/j.ijdevneu.2018.09.009 [DOI] [PubMed] [Google Scholar]
- Kronenberg G, Uhlemann R, Schöner J et al (2017) Repression of telomere-associated genes by microglia activation in neuropsychiatric disease. Eur Arch Psychiatry Clin Neurosci 267:473–477. 10.1007/s00406-016-0750-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon HS, Koh S-H (2020) Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes. Transl Neurodegener 9:42. 10.1186/s40035-020-00221-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambert JC, Ibrahim-Verbaas CA, Harold D et al (2013) Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nat Genet 45:1452–1458. 10.1038/ng.2802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lana D, Iovino L, Nosi D et al (2016) The neuron-astrocyte-microglia triad involvement in neuroinflammaging mechanisms in the CA3 hippocampus of memory-impaired aged rats. Exp Gerontol 83:71–88. 10.1016/j.exger.2016.07.011 [DOI] [PubMed] [Google Scholar]
- Laplante M, Sabatini DM (2012) mTOR signaling in growth control and disease. Cell 149:274–293. 10.1016/j.cell.2012.03.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lauro C, Limatola C (2020) Metabolic reprograming of microglia in the regulation of the innate inflammatory response. Front Immunol 11:493. 10.3389/fimmu.2020.00493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavin Y, Winter D, Blecher-Gonen R et al (2014) Tissue-resident macrophage enhancer landscapes are shaped by the local microenvironment. Cell 159:1312–1326. 10.1016/j.cell.2014.11.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JD, Kamaruzaman NA, Fung JNT et al (2013) Dysregulation of the complement cascade in the hSOD1G93A transgenic mouse model of amyotrophic lateral sclerosis. J Neuroinflammation 10:119. 10.1186/1742-2094-10-119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li M, Lu H, Wang X et al (2021) Pyruvate kinase M2 (PKM2) interacts with activating transcription factor 2 (ATF2) to bridge glycolysis and pyroptosis in microglia. Mol Immunol 140:250–266. 10.1016/j.molimm.2021.10.017 [DOI] [PubMed] [Google Scholar]
- Lin W-T, Chan T-F, Huang H-L et al (2016) Fructose-rich beverage intake and central adiposity, uric acid, and pediatric insulin resistance. J Pediatr 171:90–6.e1. 10.1016/j.jpeds.2015.12.061 [DOI] [PubMed] [Google Scholar]
- Loane DJ, Kumar A (2016) Microglia in the TBI brain: the good, the bad, and the dysregulated. Exp Neurol 275(Pt 3):316–327. 10.1016/j.expneurol.2015.08.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longo VD, Mattson MP (2014) Fasting: molecular mechanisms and clinical applications. Cell Metab 19:181–192. 10.1016/j.cmet.2013.12.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacMicking J, Xie QW, Nathan C (1997) Nitric oxide and macrophage function. Annu Rev Immunol 15:323–350. 10.1146/annurev.immunol.15.1.323 [DOI] [PubMed] [Google Scholar]
- Mado H, Adamczyk-Sowa M, Sowa P (2023) Role of microglial cells in the pathophysiology of MS: synergistic or antagonistic? Int J Mol Sci. 10.3390/ijms24031861 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malagelada C, Jin ZH, Jackson-Lewis V et al (2010) Rapamycin protects against neuron death in in vitro and in vivo models of Parkinson’s disease. J Neurosci off J Soc Neurosci 30:1166–1175. 10.1523/JNEUROSCI.3944-09.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez J, Almendinger J, Oberst A et al (2011) Microtubule-associated protein 1 light chain 3 alpha (LC3)-associated phagocytosis is required for the efficient clearance of dead cells. Proc Natl Acad Sci USA 108:17396–17401. 10.1073/pnas.1113421108 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Mashek DG, Coleman RA (2006) Cellular fatty acid uptake: the contribution of metabolism. Curr Opin Lipidol 17:274–278. 10.1097/01.mol.0000226119.20307.2b [DOI] [PubMed] [Google Scholar]
- Masoumi M, Mehrabzadeh M, Mahmoudzehi S et al (2020) Role of glucose metabolism in aggressive phenotype of fibroblast-like synoviocytes: latest evidence and therapeutic approaches in rheumatoid arthritis. Int Immunopharmacol 89:107064. 10.1016/j.intimp.2020.107064 [DOI] [PubMed] [Google Scholar]
- McIntosh A, Mela V, Harty C et al (2019) Iron accumulation in microglia triggers a cascade of events that leads to altered metabolism and compromised function in APP/PS1 mice. Brain Pathol 29:606–621. 10.1111/bpa.12704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meiser J, Krämer L, Sapcariu SC et al (2016) Pro-inflammatory macrophages sustain pyruvate oxidation through pyruvate dehydrogenase for the synthesis of itaconate and to enable cytokine expression. J Biol Chem 291:3932–3946. 10.1074/jbc.M115.676817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meissner F, Molawi K, Zychlinsky A (2010) Mutant superoxide dismutase 1-induced IL-1beta accelerates ALS pathogenesis. Proc Natl Acad Sci USA 107:13046–13050. 10.1073/pnas.1002396107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mergenthaler P, Lindauer U, Dienel GA (2013) Sugar for the brain: the role of glucose in physiological and pathological brain function. Trends Neurosci 36:587–597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mills EL, Kelly B, Logan A et al (2016) Succinate dehydrogenase supports metabolic repurposing of mitochondria to drive inflammatory macrophages. Cell 167:457-470.e13. 10.1016/j.cell.2016.08.064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miron VE, Boyd A, Zhao J-W et al (2013) M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nat Neurosci 16:1211–1218. 10.1038/nn.3469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell RW, Hatch GM (2011) Fatty acid transport into the brain: of fatty acid fables and lipid tails. Prostaglandins Leukot Essent Fatty Acids 85:293–302. 10.1016/j.plefa.2011.04.007 [DOI] [PubMed] [Google Scholar]
- Mount MP, Lira A, Grimes D et al (2007) Involvement of interferon-gamma in microglial-mediated loss of dopaminergic neurons. J Neurosci off J Soc Neurosci 27:3328–3337. 10.1523/JNEUROSCI.5321-06.2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nadjar A (2018) Role of metabolic programming in the modulation of microglia phagocytosis by lipids. Prostaglandins Leukot Essent Fatty Acids 135:63–73. 10.1016/j.plefa.2018.07.006 [DOI] [PubMed] [Google Scholar]
- Nakahira K, Haspel JA, Rathinam VAK et al (2011) Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat Immunol 12:222–230. 10.1038/ni.1980 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nichol KE, Poon WW, Parachikova AI et al (2008) Exercise alters the immune profile in Tg2576 Alzheimer mice toward a response coincident with improved cognitive performance and decreased amyloid. J Neuroinflammation 5:13. 10.1186/1742-2094-5-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niot I, Poirier H, Tran TTT, Besnard P (2009) Intestinal absorption of long-chain fatty acids: evidence and uncertainties. Prog Lipid Res 48:101–115. 10.1016/j.plipres.2009.01.001 [DOI] [PubMed] [Google Scholar]
- O’Neill LAJ, Kishton RJ, Rathmell J (2016) A guide to immunometabolism for immunologists. Nat Rev Immunol 16:553–565. 10.1038/nri.2016.70 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oishi Y, Spann NJ, Link VM et al (2017) SREBP1 contributes to resolution of pro-inflammatory TLR4 signaling by reprogramming fatty acid metabolism. Cell Metab 25:412–427. 10.1016/j.cmet.2016.11.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ojo JO, Rezaie P, Gabbott PL, Stewart MG (2015) Impact of age-related neuroglial cell responses on hippocampal deterioration. Front Aging Neurosci 7:57. 10.3389/fnagi.2015.00057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olney JW (1969) Brain lesions, obesity, and other disturbances in mice treated with monosodium glutamate. Science 164:719–721. 10.1126/science.164.3880.719 [DOI] [PubMed] [Google Scholar]
- Orihuela R, McPherson CA, Harry GJ (2016) Microglial M1/M2 polarization and metabolic states. Br J Pharmacol 173:649–665. 10.1111/bph.13139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palazon A, Goldrath AW, Nizet V, Johnson RS (2014) HIF transcription factors, inflammation, and immunity. Immunity 41:518–528. 10.1016/j.immuni.2014.09.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardridge WM (2017) Delivery of biologics across the blood-brain barrier with molecular trojan horse technology. BioDrugs 31:503–519. 10.1007/s40259-017-0248-z [DOI] [PubMed] [Google Scholar]
- Park J, Choi H, Min J-S et al (2013) Mitochondrial dynamics modulate the expression of pro-inflammatory mediators in microglial cells. J Neurochem 127:221–232. 10.1111/jnc.12361 [DOI] [PubMed] [Google Scholar]
- Park J, Choi H, Min J-S et al (2015) Loss of mitofusin 2 links beta-amyloid-mediated mitochondrial fragmentation and Cdk5-induced oxidative stress in neuron cells. J Neurochem 132:687–702. 10.1111/jnc.12984 [DOI] [PubMed] [Google Scholar]
- Patil SP, Jain PD, Ghumatkar PJ et al (2014) Neuroprotective effect of metformin in MPTP-induced Parkinson’s disease in mice. Neuroscience 277:747–754. 10.1016/j.neuroscience.2014.07.046 [DOI] [PubMed] [Google Scholar]
- Perera ND, Turner BJ (2016) AMPK signalling and defective energy metabolism in amyotrophic lateral sclerosis. Neurochem Res 41:544–553. 10.1007/s11064-015-1665-3 [DOI] [PubMed] [Google Scholar]
- Philips T, Rothstein JD (2014) Glial cells in amyotrophic lateral sclerosis. Exp Neurol 262:111–120. 10.1016/j.expneurol.2014.05.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pickrell AM, Youle RJ (2015) The roles of PINK1, parkin, and mitochondrial fidelity in Parkinson’s disease. Neuron 85:257–273. 10.1016/j.neuron.2014.12.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piers TM, Cosker K, Mallach A et al (2020) A locked immunometabolic switch underlies TREM2 R47H loss of function in human iPSC-derived microglia. FASEB J off Publ Fed Am Soc Exp Biol 34:2436–2450. 10.1096/fj.201902447R [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pivovarova NB, Andrews SB (2010) Calcium-dependent mitochondrial function and dysfunction in neurons. FEBS J 277:3622–3636. 10.1111/j.1742-4658.2010.07754.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pochini L, Scalise M, Galluccio M, Indiveri C (2014) Membrane transporters for the special amino acid glutamine: structure/function relationships and relevance to human health. Front Chem 2:61. 10.3389/fchem.2014.00061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poliani P, Wang Y, Fontana E, Robinette ML (2015) TREM2 sustains microglial expansion during aging and response to demyelination. J Clin Invest 125:2161–2170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin C, Zhou L-Q, Ma X-T et al (2019) Dual functions of microglia in ischemic stroke. Neurosci Bull 35:921–933. 10.1007/s12264-019-00388-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quan Y, Jiang C, Xue B et al (2011) High glucose stimulates TNFα and MCP-1 expression in rat microglia via ROS and NF-κB pathways. Acta Pharmacol Sin 32:188–193. 10.1038/aps.2010.174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rong G, Hongrong W, Qingqi L, Jianfeng Z (2023) Roles of microglia in AD pathology. Curr Alzheimer Res 19:854–869. 10.2174/1567205020666230203112351 [DOI] [PubMed] [Google Scholar]
- Rostami J, Holmqvist S, Lindström V et al (2017) Human astrocytes transfer aggregated alpha-synuclein via tunneling nanotubes. J Neurosci off J Soc Neurosci 37:11835–11853. 10.1523/JNEUROSCI.0983-17.2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandu RE, Uzoni A, Coman C, Popa-Wagner A (2015) Cerebral ischemia in the aged. Limited anti-inflammatory efficacy of the indomethacin treatment. Rom J Morphol Embryol = Rev Roum Morphol Embryol 56:1111–1117 [PubMed] [Google Scholar]
- Saxton RA, Sabatini DM (2017) mTOR signaling in growth, metabolism, and disease. Cell 168:960–976. 10.1016/j.cell.2017.02.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schönfeld P, Wojtczak L (2008) Fatty acids as modulators of the cellular production of reactive oxygen species. Free Radic Biol Med 45:231–241. 10.1016/j.freeradbiomed.2008.04.029 [DOI] [PubMed] [Google Scholar]
- Semenza GL (2012) Hypoxia-inducible factors in physiology and medicine. Cell 148:399–408. 10.1016/j.cell.2012.01.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharkey TD (2021) Pentose phosphate pathway reactions in photosynthesizing cells. Cells. 10.3390/cells10061547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon DW, McGeachy MJ, Bayır H et al (2017) The far-reaching scope of neuroinflammation after traumatic brain injury. Nat Rev Neurol 13:171–191. 10.1038/nrneurol.2017.13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soto I, Graham LC, Richter HJ et al (2015) APOE stabilization by exercise prevents aging neurovascular dysfunction and complement induction. PloS Biol 13:e1002279. 10.1371/journal.pbio.1002279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soto-Heredero G, de Las G, Heras MM, Gabandé-Rodríguez E et al (2020) Glycolysis—a key player in the inflammatory response. FEBS J 287:3350–3369. 10.1111/febs.15327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speisman RB, Kumar A, Rani A et al (2013) Daily exercise improves memory, stimulates hippocampal neurogenesis and modulates immune and neuroimmune cytokines in aging rats. Brain Behav Immun 28:25–43. 10.1016/j.bbi.2012.09.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiteri AG, Wishart CL, Pamphlett R et al (2022) Microglia and monocytes in inflammatory CNS disease: integrating phenotype and function. Acta Neuropathol 143:179–224. 10.1007/s00401-021-02384-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stincone A, Prigione A, Cramer T et al (2015) The return of metabolism: biochemistry and physiology of the pentose phosphate pathway. Biol Rev Camb Philos Soc 90:927–963. 10.1111/brv.12140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Streit WJ (2006) Microglial senescence: does the brain’s immune system have an expiration date? Trends Neurosci 29:506–510. 10.1016/j.tins.2006.07.001 [DOI] [PubMed] [Google Scholar]
- Suárez I, Bodega G, Fernández B (2002) Glutamine synthetase in brain: effect of ammonia. Neurochem Int 41:123–142. 10.1016/s0197-0186(02)00033-5 [DOI] [PubMed] [Google Scholar]
- Subramaniam SR, Federoff HJ (2017) Targeting microglial activation states as a therapeutic avenue in Parkinson’s disease. Front Aging Neurosci 9:176. 10.3389/fnagi.2017.00176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang Y, Le W (2016) Differential roles of M1 and M2 microglia in neurodegenerative diseases. Mol Neurobiol 53:1181–1194. 10.1007/s12035-014-9070-5 [DOI] [PubMed] [Google Scholar]
- Tannahill GM, Curtis AM, Adamik J et al (2013) Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature 496:238–242. 10.1038/nature11986 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorens B, Mueckler M (2010) Glucose transporters in the 21st Century. Am J Physiol Endocrinol Metab 298:E141–E145. 10.1152/ajpendo.00712.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tu D, Gao Y, Yang R et al (2019) The pentose phosphate pathway regulates chronic neuroinflammation and dopaminergic neurodegeneration. J Neuroinflammation 16:255. 10.1186/s12974-019-1659-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulland TK, Colonna M (2018) TREM2—a key player in microglial biology and Alzheimer disease. Nat Rev Neurol 14:667–675. 10.1038/s41582-018-0072-1 [DOI] [PubMed] [Google Scholar]
- Ulland TK, Song WM, Huang SC-C et al (2017) TREM2 maintains microglial metabolic fitness in Alzheimer’s disease. Cell 170:649-663.e13. 10.1016/j.cell.2017.07.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uttara B, Singh AV, Zamboni P, Mahajan RT (2009) Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol 7:65–74. 10.2174/157015909787602823 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van den Bossche J, Baardman J, Otto NA et al (2016) Mitochondrial dysfunction prevents repolarization of inflammatory macrophages. Cell Rep 17:684–696. 10.1016/j.celrep.2016.09.008 [DOI] [PubMed] [Google Scholar]
- Vander Heiden MG, Cantley LC, Thompson CB (2009) Understanding the warburg effect: the metabolic requirements of cell proliferation. Science 324:1029–1033. 10.1126/science.1160809 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venegas C, Kumar S, Franklin BS et al (2017) Microglia-derived ASC specks cross-seed amyloid-β in Alzheimer’s disease. Nature 552:355–361. 10.1038/nature25158 [DOI] [PubMed] [Google Scholar]
- Vilalta A, Brown GC (2014) Deoxyglucose prevents neurodegeneration in culture by eliminating microglia. J Neuroinflammation 11:58. 10.1186/1742-2094-11-58 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vinoth Kumar R, Oh TW, Park Y-K (2016) Anti-inflammatory effects of ginsenoside-Rh2 inhibits LPS-induced activation of microglia and overproduction of inflammatory mediators via modulation of TGF-β1/smad pathway. Neurochem Res 41:951–957. 10.1007/s11064-015-1804-x [DOI] [PubMed] [Google Scholar]
- Voloboueva LA, Emery JF, Sun X, Giffard RG (2013) Inflammatory response of microglial BV-2 cells includes a glycolytic shift and is modulated by mitochondrial glucose-regulated protein 75/mortalin. FEBS Lett 587:756–762. 10.1016/j.febslet.2013.01.067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volonté C, Apolloni S, Parisi C, Amadio S (2016) Purinergic contribution to amyotrophic lateral sclerosis. Neuropharmacology 104:180–193. 10.1016/j.neuropharm.2015.10.026 [DOI] [PubMed] [Google Scholar]
- Wang Z, Liu D, Wang F et al (2012) Saturated fatty acids activate microglia via Toll-like receptor 4/NF-κB signalling. Br J Nutr 107:229–241. 10.1017/S0007114511002868 [DOI] [PubMed] [Google Scholar]
- Wang Y, Cella M, Mallinson K et al (2015) TREM2 lipid sensing sustains the microglial response in an Alzheimer’s disease model. Cell 160:1061–1071. 10.1016/j.cell.2015.01.049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Ulland TK, Ulrich JD et al (2016) TREM2-mediated early microglial response limits diffusion and toxicity of amyloid plaques. J Exp Med 213:667–675. 10.1084/jem.20151948 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L, Zhang Y, Kiprowska M et al (2021) Diethyl succinate modulates microglial polarization and activation by reducing mitochondrial fission and cellular ROS. Metabolites. 10.3390/metabo11120854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q, Lu M, Zhu X et al (2022) The role of microglia immunometabolism in neurodegeneration: focus on molecular determinants and metabolic intermediates of metabolic reprogramming. Biomed Pharmacother 153:113412. 10.1016/j.biopha.2022.113412 [DOI] [PubMed] [Google Scholar]
- Wang Y, Han S, Chen J et al (2023) PFKFB3 knockdown attenuates Amyloid β-Induced microglial activation and retinal pigment epithelium disorders in mice. Int Immunopharmacol 115:109691. 10.1016/j.intimp.2023.109691 [DOI] [PubMed] [Google Scholar]
- Wehrspaun CC, Haerty W, Ponting CP (2015) Microglia recapitulate a hematopoietic master regulator network in the aging human frontal cortex. Neurobiol Aging 36:2443.e9-2443.e20. 10.1016/j.neurobiolaging.2015.04.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weissleder C, Fung SJ, Wong MW et al (2016) Decline in proliferation and immature neuron markers in the human subependymal zone during aging: relationship to EGF- and FGF-related transcripts. Front Aging Neurosci 8:274. 10.3389/fnagi.2016.00274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wendimu MY, Hooks SB (2022) Microglia phenotypes in aging and neurodegenerative diseases. Cells. 10.3390/cells11132091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westermann B (2010) Mitochondrial fusion and fission in cell life and death. Nat Rev Mol Cell Biol 11:872–884. 10.1038/nrm3013 [DOI] [PubMed] [Google Scholar]
- Wynne AM, Henry CJ, Huang Y et al (2010) Protracted downregulation of CX3CR1 on microglia of aged mice after lipopolysaccharide challenge. Brain Behav Immun 24:1190–1201. 10.1016/j.bbi.2010.05.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiang J, Ennis SR, Abdelkarim GE et al (2003) Glutamine transport at the blood-brain and blood-cerebrospinal fluid barriers. Neurochem Int 43:279–288. 10.1016/s0197-0186(03)00013-5 [DOI] [PubMed] [Google Scholar]
- Xing J, Titus AR, Humphrey MB (2015) The TREM2-DAP12 signaling pathway in Nasu-Hakola disease: a molecular genetics perspective. Res Reports Biochem 5:89–100. 10.2147/RRBC.S58057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu M-X, Yu R, Shao L-F et al (2016) Up-regulated fractalkine (FKN) and its receptor CX3CR1 are involved in fructose-induced neuroinflammation: suppression by curcumin. Brain Behav Immun 58:69–81. 10.1016/j.bbi.2016.01.001 [DOI] [PubMed] [Google Scholar]
- Xu J, Wang L, Yang Q et al (2021) Deficiency of myeloid Pfkfb3 protects mice from lung edema and cardiac dysfunction in LPS-induced endotoxemia. Front Cardiovasc Med 8:745810. 10.3389/fcvm.2021.745810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamanaka M, Ishikawa T, Griep A et al (2012) PPARγ/RXRα-induced and CD36-mediated microglial amyloid-β phagocytosis results in cognitive improvement in amyloid precursor protein/presenilin 1 mice. J Neurosci off J Soc Neurosci 32:17321–17331. 10.1523/JNEUROSCI.1569-12.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang F, Wang Z, Wei X et al (2014) NLRP3 deficiency ameliorates neurovascular damage in experimental ischemic stroke. J Cereb Blood Flow Metab off J Int Soc Cereb Blood Flow Metab 34:660–667. 10.1038/jcbfm.2013.242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang S, Qin C, Hu Z-W et al (2021) Microglia reprogram metabolic profiles for phenotype and function changes in central nervous system. Neurobiol Dis 152:105290. 10.1016/j.nbd.2021.105290 [DOI] [PubMed] [Google Scholar]
- Yeh FL, Wang Y, Tom I et al (2016) TREM2 binds to apolipoproteins, including APOE and CLU/APOJ, and thereby facilitates uptake of amyloid-beta by microglia. Neuron 91:328–340. 10.1016/j.neuron.2016.06.015 [DOI] [PubMed] [Google Scholar]
- Yenari MA, Giffard RG (2001) Ischemic vulnerability of primary murine microglial cultures. Neurosci Lett 298:5–8. 10.1016/s0304-3940(00)01724-9 [DOI] [PubMed] [Google Scholar]
- Youle RJ, van der Bliek AM (2012) Mitochondrial fission, fusion, and stress. Science 337:1062–1065. 10.1126/science.1219855 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zezina E, Sercan-Alp O, Herrmann M, Biesemann N (2020) Glucose transporter 1 in rheumatoid arthritis and autoimmunity. Wiley Interdiscip Rev Syst Biol Med 12:e1483. 10.1002/wsbm.1483 [DOI] [PubMed] [Google Scholar]
- Zhang W, Wang T, Qin L et al (2004) Neuroprotective effect of dextromethorphan in the MPTP Parkinson’s disease model: role of NADPH oxidase. FASEB J off Publ Fed Am Soc Exp Biol 18:589–591. 10.1096/fj.03-0983fje [DOI] [PubMed] [Google Scholar]
- Zhang X, Li L, Chen S et al (2011) Rapamycin treatment augments motor neuron degeneration in SOD1(G93A) mouse model of amyotrophic lateral sclerosis. Autophagy 7:412–425. 10.4161/auto.7.4.14541 [DOI] [PubMed] [Google Scholar]
- Zhang Y, Chen K, Sloan SA et al (2014) An RNA-sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex. J Neurosci off J Soc Neurosci 34:11929–11947. 10.1523/JNEUROSCI.1860-14.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Dong H, Zhang S et al (2015) Enhancement of LPS-induced microglial inflammation response via TLR4 under high glucose conditions. Cell Physiol Biochem Int J Exp Cell Physiol Biochem Pharmacol 35:1571–1581. 10.1159/000373972 [DOI] [PubMed] [Google Scholar]
- Zhang Y, Liu W, Wu M et al (2021) PFKFB3 regulates lipopolysaccharide-induced excessive inflammation and cellular dysfunction in HTR-8/Svneo cells: Implications for the role of PFKFB3 in preeclampsia. Placenta 106:67–78. 10.1016/j.placenta.2021.02.014 [DOI] [PubMed] [Google Scholar]
- Zhang L, Cao Y, Zhang X et al (2022) The origin and repopulation of microglia. Dev Neurobiol 82:112–124. 10.1002/dneu.22862 [DOI] [PubMed] [Google Scholar]
- Zhao Y, Xu H (2022) Microglial lactate metabolism as a potential therapeutic target for Alzheimer’s disease. Mol Neurodegener 17:36. 10.1186/s13024-022-00541-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong W-J, Yang H-H, Guan X-X et al (2019) Inhibition of glycolysis alleviates lipopolysaccharide-induced acute lung injury in a mouse model. J Cell Physiol 234:4641–4654. 10.1002/jcp.27261 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Enquiries about data availability should be directed to the authors.