Abstract
Both CD46 and signaling lymphocytic activation molecule (SLAM) have been shown to act as cellular receptors for measles virus (MV). The viruses on throat swabs from nine patients with measles in Japan were titrated on Vero cells stably expressing human SLAM. Samples from all but two patients produced numerous plaques on SLAM-expressing Vero cells, whereas none produced any plaques on Vero cells endogenously expressing CD46. The Edmonston strain of MV, which can use either CD46 or SLAM as a receptor, produced comparable titers on these two types of cells. The results strongly suggest that the viruses in the bodies of measles patients use SLAM but probably not CD46 as a cellular receptor.
Measles virus (MV) is an enveloped negative-strand RNA virus of the Morbillivirus genus in the Paramyxoviridae family (4). Measles remains an important cause of childhood mortality, with approximately one million deaths per year worldwide (2), mainly due to secondary infections caused by MV-induced immunosuppression (4). Human CD46 has been shown to be a cellular receptor for vaccine strains of MV, such as the Edmonston strain (3, 10). However, wild-type MV strains that are commonly isolated in marmoset B-cell line B95a or human B-cell lines usually do not use CD46 as a receptor (5–7, 14, 15, 18), although a study has reported that MV strains isolated from and propagated only in human peripheral blood mononuclear cells (PBMCs) use CD46 as a receptor (9). We have recently demonstrated that signaling lymphocytic activation molecule (SLAM; also known as CDw150) is a cellular receptor for MV, including the Edmonston strain, B95a-isolated strains, and PBMC-isolated strains (20). Thus, some MV strains use SLAM but not CD46 as a receptor, and others, such as the Edmonston strain, use either SLAM or CD46. The type of MV strain obtained depends on the cell types used for virus isolation. In this study, we sought to quantitate the proportions of these two types of MV in measles patients.
Vero cells are susceptible to the Edmonston strain but not to B95a-isolated MV strains (6, 18). In order to titrate B95a-isolated MV strains on Vero cells, we transfected them with the expression plasmid encoding human SLAM (pCAG-hSLAM) (12) and the vector plasmid pCXN2 (11) containing the neomycin resistance (neo) gene; we selected stable clones in the presence of G418. We used the clone expressing the highest level of human SLAM (Vero/hSLAM) in the following experiments.
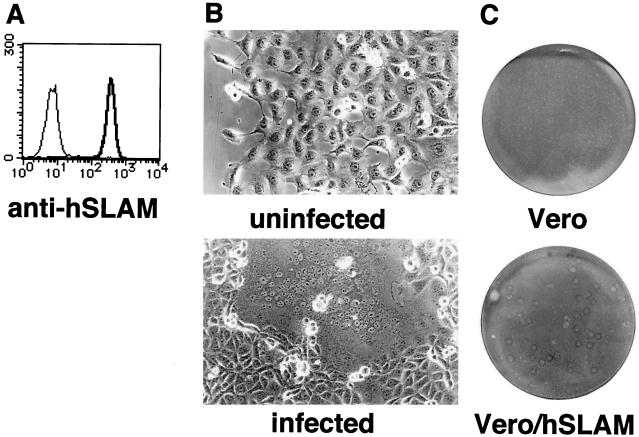
The expression profile of Vero/hSLAM cells stained with anti-human SLAM monoclonal antibody IPO-3 (Kamiya Biomedical) (17) is shown in Fig. 1A. Vero/hSLAM cells were infected with the B95a-isolated KA strain of MV (18–20) at a multiplicity of infection of 0.1. At 24 h after infection, they developed extensive syncytia (Fig. 1B), unlike the parental Vero cells (6, 18). Then, we used Vero and Vero/hSLAM cells for plaque titration of the KA strain. Vero/hSLAM cells developed clear plaques after infection with the KA strain, whereas Vero cells inoculated with the same amount of the virus did not show any plaques (Fig. 1C).
FIG. 1.
Characterization of Vero/hSLAM cells. (A) Vero/hSLAM cells were stained with IPO-3 (thick line) or a mouse immunoglobulin G1 (IgG1) control antibody (thin line), followed by staining with fluorescein isothiocyanate-labeled anti-mouse IgG. The stained cells were analyzed on a FACScan machine (Becton Dickinson). (B) Vero/hSLAM cells were either uninfected or infected with the KA strain of MV and observed under a microscope at 24 h after infection. (C) The diluted stock of the KA strain was titrated on Vero or Vero/hSLAM cells. At 3 days after infection, plates were stained with crystal violet and photographed.
We next compared MV titers on Vero/hSLAM cells with those on Vero cells. B95a cells and Chinese hamster ovary (CHO) cells stably expressing human SLAM (CHO/hSLAM) (20) or human CD46 (CHO/CD46) (19) were also used for the titration. Vero, Vero/hSLAM, CHO, CHO/CD46, and CHO/hSLAM cells (5 × 105 cells per well) were seeded in six-well plates on the day before infection and then inoculated with 0.5 ml of the Edmonston or KA strain of MV appropriately diluted in phosphate-buffered saline containing 5% fetal bovine serum. At 1 h after infection, the cells were overlaid with 3 ml of medium containing 0.5% agarose and then incubated at 37°C in a 5% CO2 incubator. At 3 days after infection, the cells were fixed with formaldehyde and stained with crystal violet. After washing, the numbers of plaques were counted, and PFU per milliliter were calculated. B95a cells were plated in 96-well plates and then infected with the Edmonston or KA strain, and 50% tissue culture infective doses (TCID50) were determined at 3 days after infection. Since B95a cells do not firmly adhere to plates, it is difficult to perform plaque titration accurately using them. Therefore, we determined TCID50 rather than PFU on B95a cells (6, 19).
Table 1 shows the titers obtained on the different types of cells when the same stock of the Edmonston or KA strain was used. The Edmonston strain produced comparable titers on all cell lines except for CHO cells, on which no plaques were produced. The B95a-isolated KA strain produced comparable titers on Vero/hSLAM, CHO/hSLAM, and B95a cells but produced no plaques on Vero, CHO, and CHO/CD46 cells, as expected from its tropism.
TABLE 1.
MV titers determined on different cell lines
| Sample | MV titera on cell line:
|
|||||
|---|---|---|---|---|---|---|
| Vero | Vero/hSLAM | CHO | CHO/CD46 | CHO/hSLAM | B95a | |
| MV strain | ||||||
| Edmonston | 4.0 × 106 | 6.3 × 106 | 0 | 1.8 × 106 | 2.1 × 106 | 6.3 × 106 |
| KA | 0 | 1.8 × 106 | 0 | 0 | 5.0 × 105 | 3.2 × 106 |
| Throat swab from patient | ||||||
| 1 | 0 | 3.6 × 103 | 0 | 0 | 8.0 × 102 | 4.6 × 103 |
| 2 | 0 | 3.1 × 103 | 0 | 0 | 8.4 × 102 | 3.7 × 103 |
| 3 | 0 | 3.3 × 103 | 0 | 0 | 1.5 × 103 | 6.3 × 103 |
| 4 | 0 | 1.9 × 104 | ND | ND | ND | ND |
| 5 | 0 | 5.0 × 102 | ND | ND | ND | ND |
| 6 | 0 | 7.1 × 102 | ND | ND | ND | ND |
| 7 | 0 | 6.3 × 103 | ND | ND | ND | ND |
Reported as PFU per milliliter, except for B95a cells, for which titers are reported as TCID50 per milliliter. ND, not done.
We next determined the titers of viruses on throat swabs from nine patients with measles using Vero and Vero/hSLAM cells. CHO/CD46, CHO/SLAM, and B95a cells were also used for some patients. All of these patients had visited Kitakyushu Municipal Medical Center, Kitakyushu, Japan, in 2000 and were clinically diagnosed as having measles. Throat swabs were collected in 2 ml of phosphate-buffered saline within 3 days after the onset of rash and were stored at −80°C until the titrations were performed. The cells were inoculated with 0.2 ml of throat swabs from individual patients, and titrations were performed as described above. Table 1 shows the titers determined on the different samples. None of the samples produced any plaques on Vero cells. In contrast, samples from seven patients produced between 500 and 19,000 plaques per ml of inoculum on Vero/hSLAM cells (titrations were performed in duplicate). When CHO transfectants and B95a cells were used, the results were consistent with those obtained on Vero and Vero/hSLAM cells. Two samples neither produced any plaques on Vero/hSLAM cells nor caused any cytopathic effects in B95a cells. Considering that the Edmonston strain, which can use either CD46 or SLAM as a receptor, produced comparable titers on Vero and Vero/hSLAM cells or on CHO/CD46 and CHO/hSLAM cells, the proportion of the viruses which use CD46 as a receptor is at least 2 to 4 log units lower than the proportion of viruses which use SLAM only as a receptor.
This study indicated that the viruses in the bodies of measles patients, at least from a single outbreak, use SLAM but not CD46 as a receptor. This result is consistent with previous observations that with B95a cells, one can quickly and efficiently isolate MV from clinical specimens, whereas with Vero cells, the successful isolation rate is low and several blind passages are usually necessary (4, 6). On the other hand, a single amino acid substitution of the hemagglutinin at position 481 to tyrosine (1, 5, 7, 16, 21) or at position 546 to glycine (13, 16) may enable MV strains that use SLAM only as a receptor to interact with CD46. Viruses with such amino acid changes are easily generated during replication in measles patients, and once produced, they should be able to infect almost any cells in the body because CD46 is ubiquitously expressed (8). Although this study did not detect such viruses in the throat, they may exist in other organs or tissues, accounting for the pathogenesis of measles. In fact, Manchester et al. have reported that MV strains which were isolated by coculturing PBMCs of measles patients with human PBMCs and then propagated only in PBMCs use CD46 as a cellular receptor (9). However, we have demonstrated that even these PBMC-isolated strains, which did not efficiently replicate in CHO/CD46 cells (9), caused strong cytopathic effects in CHO/hSLAM cells (20), indicating that SLAM serves better as a receptor for these strains than CD46.
In view of our present results, we propose that the in vivo propagation of viruses that use CD46 as a receptor is suppressed by an unknown mechanism(s). Thus, the usage of CD46 as a receptor by the Edmonston and PBMC-isolated strains may be an in vitro adaptation rather than the in vivo property of the progenitors of those strains. At any rate, the viruses in the throat of a measles patient are transmitted to other susceptible people; therefore, viruses that use SLAM as a receptor are important in the pathogenesis and epidemiology of measles as they first enter the body and transmit the disease.
Acknowledgments
We thank F. Kobune and Y. Murakami for providing reagents and M. B. A. Oldstone for helpful discussions.
This work was supported by grants from the Ministry of Education, Science and Culture of Japan and from the Organization for Drug ADR Relief, R&D Promotion and Product Review of Japan.
REFERENCES
- 1.Bartz R, Brinckmann U, Dunster L M, Rima B, ter Meulen V, Schneider-Schaulies J. Mapping amino acids of the measles virus hemagglutinin responsible for receptor (CD46) downregulation. Virology. 1996;224:334–337. doi: 10.1006/viro.1996.0538. [DOI] [PubMed] [Google Scholar]
- 2.Centers for Disease Control and Prevention. Global measles control and regional elimination, 1998–1999. Morb Mortal Wkly Rep. 1999;48:1124–1130. [PubMed] [Google Scholar]
- 3.Dorig R E, Marcil A, Chopra A, Richardson C D. The human CD46 molecule is a receptor for measles virus (Edmonston strain) Cell. 1993;75:295–305. doi: 10.1016/0092-8674(93)80071-l. [DOI] [PubMed] [Google Scholar]
- 4.Griffin D E, Bellini W J. Measles virus. In: Fields B N, Knipe D M, Howley P M, Chanock R M, Melnick J L, Monath T P, Roizman B, Straus S E, editors. Fields virology. 3rd ed. Philadelphia, Pa: Lippincott-Raven; 1996. pp. 1267–1312. [Google Scholar]
- 5.Hsu E C, Sarangi F, Iorio C, Sidhu M S, Udem S A, Dillehay D L, Xu W, Rota P A, Bellini W J, Richardson C D. A single amino acid change in the hemagglutinin protein of measles virus determines its ability to bind CD46 and reveals another receptor on marmoset B cells. J Virol. 1998;72:2905–2916. doi: 10.1128/jvi.72.4.2905-2916.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kobune F, Sakata H, Sugiura A. Marmoset lymphoblastoid cells as a sensitive host for isolation of measles virus. J Virol. 1990;64:700–705. doi: 10.1128/jvi.64.2.700-705.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lecouturier V, Fayolle J, Caballero M, Carabana J, Celma M L, Fernandez-Munoz R, Wild T F, Buckland R. Identification of two amino acids in the hemagglutinin glycoprotein of measles virus (MV) that govern hemadsorption, HeLa cell fusion, and CD46 downregulation: phenotypic markers that differentiate vaccine and wild-type MV strains. J Virol. 1996;70:4200–4204. doi: 10.1128/jvi.70.7.4200-4204.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liszewski K M, Post T W, Atkinson J P. Membrane cofactor protein (MCP or CD46): newest member of the regulators of complement activation gene cluster. Annu Rev Immunol. 1991;9:431–455. doi: 10.1146/annurev.iy.09.040191.002243. [DOI] [PubMed] [Google Scholar]
- 9.Manchester M, Eto D S, Valsamakis A, Liton P B, Fernandez-Munoz R, Rota P A, Bellini W J, Forthal D N, Oldstone M B A. Clinical isolates of measles virus use CD46 as a cellular receptor. J Virol. 2000;74:3967–3974. doi: 10.1128/jvi.74.9.3967-3974.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Naniche D, Varior-Krishnan G, Cervoni F, Wild T F, Rossi B, Rabourdin-Combe C, Gerlier D. Human membrane cofactor protein (CD46) acts as a cellular receptor for measles virus. J Virol. 1993;67:6025–6032. doi: 10.1128/jvi.67.10.6025-6032.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Niwa H, Yamamura K, Miyazaki J. Efficient selection for high-expression transfectants by a novel eukaryotic vector. Gene. 1991;108:193–200. doi: 10.1016/0378-1119(91)90434-d. [DOI] [PubMed] [Google Scholar]
- 12.Ono N, Tatsuo H, Tanaka K, Minagawa H, Yanagi Y. V domain of human SLAM (CDw150) is essential for its function as a measles virus receptor. J Virol. 2001;75:1594–1600. doi: 10.1128/JVI.75.4.1594-1600.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rima B K, Earle J A P, Baczko K, ter Meulen V, Liebert U G, Carstens C, Carabana J, Caballero M, Celma M L, Fernandez-Munoz R. Sequence divergence of measles virus haemagglutinin during natural evolution and adaptation to cell culture. J Gen Virol. 1997;78:97–106. doi: 10.1099/0022-1317-78-1-97. [DOI] [PubMed] [Google Scholar]
- 14.Schneider-Schaulies J, Dunster L M, Kobune F, Rima B, ter Meulen V. Differential downregulation of CD46 by measles virus strains. J Virol. 1995;69:7257–7259. doi: 10.1128/jvi.69.11.7257-7259.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schneider-Schaulies J, Schnorr J-J, Brinckmann U, Dunster L M, Baczko K, Liebert U G, Schneider-Schaulies S, ter Meulen V. Receptor usage and differential downregulation of CD46 by measles virus wild-type and vaccine strains. Proc Natl Acad Sci USA. 1995;92:3943–3947. doi: 10.1073/pnas.92.9.3943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shibahara K, Hotta H, Katayama Y, Homma M. Increased binding activity of measles virus to monkey red blood cells after long-term passage in Vero cell cultures. J Gen Virol. 1994;75:3511–3516. doi: 10.1099/0022-1317-75-12-3511. [DOI] [PubMed] [Google Scholar]
- 17.Sidorenko S P, Clark E A. Characterization of a cell surface glycoprotein IPO-3, expressed on activated human B and T lymphocytes. J Immunol. 1993;151:4614–4624. [PubMed] [Google Scholar]
- 18.Tanaka K, Xie M, Yanagi Y. The hemagglutinin of recent measles virus isolates induces cell fusion in a marmoset cell line, but not in other CD46-positive human and monkey cell lines, when expressed together with the F protein. Arch Virol. 1998;143:213–225. doi: 10.1007/s007050050281. [DOI] [PubMed] [Google Scholar]
- 19.Tatsuo H, Okuma K, Tanaka K, Ono N, Minagawa H, Takade A, Matsuura Y, Yanagi Y. Virus entry is a major determinant of cell tropism of Edmonston and wild-type strains of measles virus as revealed by vesicular stomatitis virus pseudotypes bearing their envelope proteins. J Virol. 2000;74:4139–4145. doi: 10.1128/jvi.74.9.4139-4145.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tatsuo H, Ono N, Tanaka K, Yanagi Y. SLAM (CDw150) is a cellular receptor for measles virus. Nature. 2000;406:893–897. doi: 10.1038/35022579. [DOI] [PubMed] [Google Scholar]
- 21.Xie M-F, Tanaka K, Ono N, Minagawa H, Yanagi Y. Amino acid substitutions at position 481 differently affect the ability of the measles virus hemagglutinin to induce cell fusion in monkey and marmoset cells co-expressing the fusion protein. Arch Virol. 1999;144:1689–1699. doi: 10.1007/s007050050697. [DOI] [PubMed] [Google Scholar]