Abstract
Damage or microstructural alterations of the white matter can cause dysfunction of the intrinsic neural networks in a condition termed as white matter disease (WMD). Frequently detected on brain computed tomography and magnetic resonance imaging scans, WMD is commonly presented in inflammatory demyelinating diseases like multiple sclerosis (MS) and vascular diseases such as cerebral small vessel disease (CSVD). Prevention of MS and CSVD progression requires early treatments with drastically different medications and approaches, as such, early and accurate diagnosis of WMD, derived from vascular or demyelinating etiologies, is of paramount importance. However, the clinical and imaging similarities between MS, especially during the early stage, and CSVD, pose a significant dilemma in differentiating these two conditions. In this review, we attempt to summarize and contrast the distinguishing features of MS and CSVD for aiding accurate diagnosis to ensure timely corresponding management in the early stages of MS and CSVD.
Keywords: Cerebrovascular Disorders, Magnetic Resonance Imaging, Inflammation
Introduction
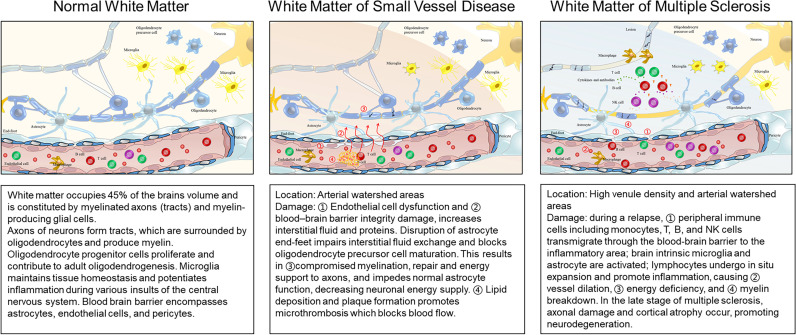
The white matter of the central nervous system (CNS) is primarily constituted of myelinated axons (tracts) and myelin-producing glial cells, connecting and communicating between disparate regions of grey matter,1 which is composed of bodies of neurons. In the human brain, white matter occupies approximately 4×105 mm3, which comprises 45% of the total volume.2 The medullary arteries supplying the cerebral white matter are long-end arteries absent in anastomotic connections and arise perpendicularly from pial arteries. Most of the subcortical arteries penetrate straight through the cortex, but become coiled, looped and/or spiralled on entering the white matter. This coiling architecture loses the ability to react in autoregulation and may serve as a trap for inflamed cells via the bloodstream, contributing to vascular and inflammatory diseases.3 4
Damage or microstructural alterations to the white matter frequently detected by brain CT or MRI scans are designated as white matter diseases (WMD). With the popularisation and application of imaging technology, more and more symptomatic or asymptomatic white matter lesions have been found clinically. The two common causes of WMD are vascular diseases, which are especially common in northern China, and inflammatory demyelinating causes like multiple sclerosis (MS). Differing from acute ischaemic and haemorrhagic strokes, cerebral small vessel disease (CSVD) is relatively mild symptomatically, but its progressive neurological dysfunction has received increasing attention in recent years. The differential diagnosis of MS can be challenging. Early identification of MS can help with the early application of disease-modifying therapy to reduce disease disability. The similarity of image manifestations requires careful screening and early judgement by clinicians, which is conducive to early treatment and long-term management of patients. In this review, we focus on CSVD for its high prevalence and its similar neuroimaging characteristics with MS, which obscures and hinders the differential diagnosis between the two conditions. To this end, we summarise the similarities and differences in the aetiology, clinical features, and imaging characteristics between CSVD and MS to assist accurate diagnoses and treatment.
Pathological basis for white matter lesions in MS and CSVD
Inflammation
Multiple focal areas of myelin loss within the CNS are the main pathologic hallmarks of MS. Demyelination is accompanied by inflammation, gliosis and relative axonal preservation at the early stage of MS. Lesions are disseminated in areas throughout the CNS, with the majority affecting the white matter of the brainstem, cerebellum, juxtacortical and periventricular regions of the brain.5 Evidence of sequential inflammation in subcortical and cortical regions has been shown in patients with MS.6 Formation of plaques in MS is due to a confluence of inflammation, myelin breakdown, astrogliosis, oligodendrocyte injury, neurodegeneration, axonal loss and remyelination5 (figure 1). Acute, chronic active and chronic silent lesions are thought to occur along a continuous timeline, eventually bearing scarred and hardened areas within the CNS. Dysregulation of the blood-brain barrier (BBB) and trans-endothelial migration of activated leukocytes are among the earliest cerebrovascular abnormalities observed in the MS brain and coincide with the release of inflammatory cytokines.7 As MS progresses, inflammation wanes and is followed by neurodegeneration, mostly induced by persistent glial activation, mitochondrial injury, oxidative stress and ionic imbalance.8
Figure 1.
White matter in normal, cerebral small vessel disease or multiple sclerosis. The figure is designed and drawn by the authors.
The role of inflammation in the genesis and progression of CSVD has been proposed recently. Inflammatory cells in the white matter around vessels and in the vicinity of demyelination were found in postmortem samples. Plasma inflammatory cytokines, markers of oxidative stress and vascular inflammation are associated with the white matter hyperintensity volume on MRI.9 In addition, atherosclerosis is an important cause of CSVD. Plenty of evidence supports the involvement of inflammatory responses in atherosclerosis. Inflammation during the genesis of atherosclerosis is triggered by trapping low-density lipoproteins in the intima,10 the accumulation of which exacerbates the stenosis of blood vessels and reduces blood flow leading to ischemia. BBB impairment, chronic inflammatory responses and leucocyte infiltration are classical pathological features of CSVD (figure 1, table 1). Recent studies suggest that components of the coagulation system may trigger inflammation in CSVD.11
Table 1.
Contrast the pathological processes between inflammatory demyelinating white matter disease
| Vascular demyelinating WMD | Inflammatory demyelinating WMD | |
| Causes | Ischaemic or haemorrhagic stroke | MS, NMOSD |
| CSVD including hypertension, diabetes mellitus, migraine, CAA, CADASIL, Susac syndrome and others | Vasculopathy, including SLE, sarcoidosis, Behcet disease, Sjögren syndrome | |
| Pathological process | Various aetiologies affecting the small arteries, arterioles, venules and capillaries of the brain, leading to endothelial dysfunction, BBB damage and secondary destruction of normal myelin | BBB damage and robust inflammatory infiltration combined with demyelination, leading to secondary destruction of normal myelin |
BBB, blood-brain barrier; CAA, cerebral amyloid angiopathy; CADASIL, cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy; CSVD, cerebral small vessel disease; MS, multiple sclerosis; NMOSD, neuromyelitis optica spectrum disorders; SLE, systemic lupus erythematosus.
Cerebral amyloid angiopathy (CAA), characterised by the progressive accumulation of amyloid-β fibrils in the walls of small to medium-sized arteries and arterioles, is another important cause of CSVD.12 Recurrent large lobar haemorrhages, microbleeds, white matter lesions and microinfarcts are main features of CAA.13 Different forms of CAA-associated inflammation have been described: one is a non-vasculitic form called perivascular infiltration, which is characterised by perivascular infiltration of the parenchyma by multinucleated giant cells; the other is a vasculitic form called transmural granulomatous angiitis, which is characterised by inflammation of the vessel wall, with the occasional presence of granulomas.14 Conformational transitions occurring in native soluble amyloid molecules increase their content in β-sheet structures, thereby favouring the formation and deposition of more insoluble oligomeric structures. In turn, these deposits trigger a secondary cascade of events including, among others, the release of inflammatory components, activation of the complement system, oxidative stress, alterations of BBB permeability and cell toxicity.15
Vascular damage
Vascular damage of CSVD encompasses a group of pathological processes with various aetiologies affecting small arteries, arterioles, venules and capillaries of the brain. Disrupted BBB integrity may play a pivotal role in the aetiology of CSVD. A consensus was endorsed on BBB impairment appearing in the MRI of white matter hyperintensity and normal-appearing white matter.16 Pathological findings of CSVD due to arteriolosclerosis present as loss of smooth muscle cells from the tunica media, deposits of fibrohyaline material, narrowing of the lumen and thickening of the vessel wall, which initiate on a damaged endothelium; such blood vessel changes, therefore, occlude the lumen, inducing lacunar infarction.17 Brain damage arising from small-vessel endothelial leakage has been suggested to be a potential cause of CSVD. Further studies are required to test whether BBB failure and perforating artery endothelial leakage might be common pathogenetic mechanisms in lacunar stroke, white matter hyperintensities and dementia. For example, in a stroke-prone spontaneously hypertensive rat model,18 dysfunctional endothelial cells secrete heat shock protein 90α, which blocks oligodendroglia differentiation to contribute to impaired myelination as the first step in the development of SVD.
Acute lesions of MS are characterised by robust inflammatory infiltration combined with demyelination and parenchymal oedema distributed throughout the lesions. Lesions were around vessels forming the specific ‘central vein sign’ on MRI are characteristics of inflammatory demyelination (figure 2). Dysregulation of the BBB and transendothelial migration of activated leukocytes are among the acute phase in MS brains and coincide with the release of inflammatory cytokines.7 Perivascular cuffing is formed by an influx of immune cells centred around vessels, including lymphocytes (predominantly T cells), monocytes and natural killer (NK) cells (figure 1). A subset of the major histocompatibility class II-expressing cells, distributed evenly throughout the lesions, are loaded with lipids (foamy macrophages) and participate in active stripping of myelin from axons.
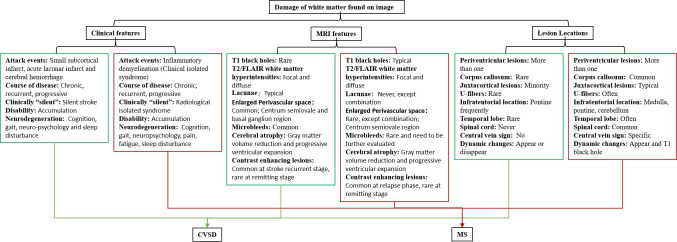
Figure 2.
Key points for differentiating cerebral small vessel disease and multiple sclerosis. FLAIR, fluid-attenuated inversion recovery.
Decreased cerebral perfusion in MS in normal-appearing white matter and grey matter has been demonstrated in MR perfusion imaging. Extracranial arterial studies in MS patients via magnetic resonance angiography demonstrate a significant mid-term decrease in the number and size of the secondary neck vessels.19 Lipoprotein(a) levels are associated with the size of extracranial arteries in MS,20 while the clinical relevance of these findings are currently unknown. The abundant cytokines may be the potential cause for the accelerated narrowing of the cerebral and extracranial arterial vessels in MS (figure 1). The association of neck venous drainage abnormalities with MS is uncertain.21 Optical coherence tomography angiography can investigate the retinal microvascular structures and evaluate the vascular changes in the foveal and peripapillary region in MS. Retinal macular superficial vascular plexus density is reduced in the eyes of MS patients and correlates with disability and visual function.22 This reflects early vascular damage in patients with MS.
Clinical overlap between CSVD and MS
Presenting symptoms and the clinical course of CSVD are variable. Cerebral infarction includes asymptomatic infarcts, episodes of transient cerebral ischemia and neurological deficits manifested as lacunar syndrome. Asymptomatic infarctions are clinically silent and conspicuously termed as ‘silent stroke’, generally occur in hypertensive patients. Lacunar stroke is the most common type of ischaemic stroke, resulting from the occlusion of small penetrating arteries, ranging from 100 µm to 400 µm in diameter, which supplies blood to the brain’s deep structures. Transient cerebral ischaemia may be the only manifestation.23 No matter the acute attack or the asymptomatic lesions, chronic, progressive and accumulated cognitive decline, gait disorder and neuropsychiatric symptoms gradually progress and affect patients’ daily life.
MS undergoes a characteristic relapse-remitting and progressive disease course. Attacks of MS, as well as CSVD, manifest as a focal loss of perception or motor ability during their acute phase. Gait abnormity is a persistent disability affecting the motor system. Non-motor MS symptoms include cognitive decline, depression, pain, fatigue, anxiety, and sleep disorder, all of which overlap CSVD symptoms.6
Therefore, CSVD and MS may both present as acute and as chronic, progressive neurological deficits. Misdiagnoses of MS as an acute stroke or vice versa will result in a vastly different evaluation and treatment. Clinically silent but MRI-positive lesions are another common feature in these two diseases. White matter hyperintensities incidentally found without neurological manifestations are called ‘silent strokes’, with vascular origins and ‘radiologically isolated syndrome’, with inflammatory origins. It is difficult to distinguish the origins of these isolated clinically silent lesions, hindering appropriate diagnosis and prevention of subsequent clinical attacks. Other common presentations include white matter lesion load progression with years, cerebral atrophy and cognition dysfunction. It is prudent for clinicians to consider MS as a differential diagnosis for older individuals or those with vascular risk factors. Furthermore, patients may present both of these risk factors, increasing the difficulty to determine the origin of peri-ventricular lesions. However, CSVD rarely attacks optic nerve and spinal cord, which are commonly damaged in MS patients. Hence, symptoms of optic neuritis or myelitis, such as transverse plane of paraesthesias, dysfunction of the bladder and dysporia, were red alerts for diagnosing CSVD. (figure 2)
Similarities and differences of CSVD and MS in MRI
White matter abnormalities on T2-weighted images are observed in 20%–78% of healthy and demented elderly patients. White matter changes are easily identified in proton density-weighted, T2-weighted and fluid-attenuated inversion recovery (FLAIR) imaging. It is worth noting that incidentally found lesions lead to puzzle of management, especially when these lesions are not relevant to disease presentation or symptoms.24 In patients with CSVD, white matter changes are bilateral, patchy or diffusely distributed hypodensity lesions on CT or hyperintensities on T2-weighted MRI involving the periventricular and centrum semiovale white matter (leukoaraiosis).25 These changes relate to the decline of cognitive and motor performances, and depressive symptoms, urinary disturbances and various neurological abnormalities. In 2013, STandards for ReportIng Vascular changes on nEuroimaging (STRIVE) provided as the standardised imaging features of CSVD.26 27 Correspondingly, European Magnetic Resonance Network in MS (MAGNIMS) and the Consortium of MS Centers recently proposed standardised MRI protocols for the diagnostic process, from prognosis to follow-ups.28 In the modified criteria, symptomatic lesions demonstrating dissemination in space and time include lesions of the periventricular, cortical/juxtacortical, infratentorial, spinal cord and optic nerve. Cortical lesions are considered evidence of dissemination in space, further, when cortical or juxtacortical lesions are present CNS vasculitis and occasionally haemorrhage should be included in the differential diagnosis.6 The 2018 MAGNIMS update in the imaging features differentiates the most common mimics of MS, particularly age-related cerebrovascular diseases and neuromyelitis optica spectrum disorders.29
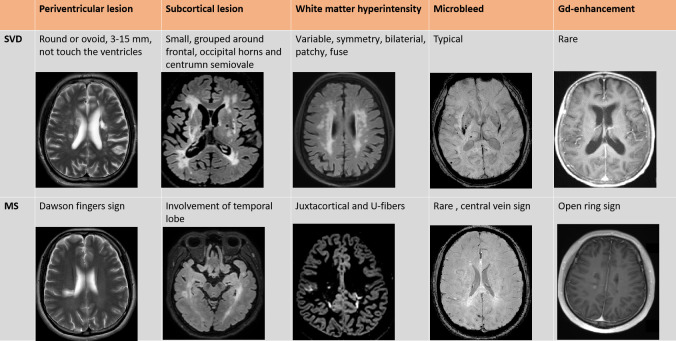
We here summarise the key differences found in MRI to distinguish CVSD and MS (figures 2–4). Multiple, T2-hyperintense white matter lesions on conventional MRI are common features shared by CSVD and MS, mainly presenting as white matter hyperintensity with similar shapes and locations, especially the periventricular lesions. Lacunes of presumed vascular origin are typically white matter lesions seen on MRI in basal ganglia, internal capsule, thalamus and pons, which can occur silently or after an acute small subcortical infarct or haemorrhage,30 which are 3–15 mm cerebrospinal fluid (CSF)-filled cavities. White matter hyperintensities are patchy or confluent depending on the stage of development and severity. A hypointense hole surrounded by a hyperintense rim on FLAIR sequence is distinctive for lacunes. These are usually symmetrically and bilaterally distributed in the white matter, pons, brain stem and deep grey matter in patients with CSVD. But in contrast, Dawson’s finger sign, which is defined as wedge-shaped areas with a broad base to the ventricle, and extensions into adjoining tissue in the form of finger-like processes or ampullae, is specific in MS periventricular lesions. Certain lesions may appear hypointense on T1-weighted images, so-called black holes.31 Similar to a ‘silent stroke’, individuals with T2 hyperintensities on brain MRI strongly suggestive of MS but without neurological presentations or other explanations are distinguished as radiologically isolated syndrome. Radiologically isolated syndromes could provide an early indication and diagnosis of MS and also increases the rate of misdiagnosis and the possibility of adverse effects of immune therapies.32
Figure 3.
Cardinal MRI difference between cerebral small vessel disease and multiple sclerosis. *T2 weighted image; #FLAIR: fluid attenuation inversion recovery; &SWI: susceptibility-weighted imaging; §T1 gadolinium enhancement. The MRI images are collected from the authors’ centres.
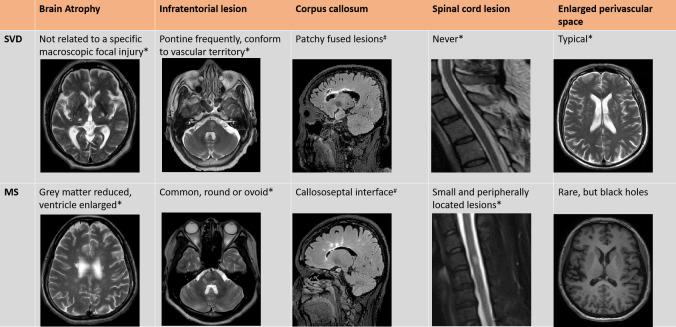
Figure 4.
Additional distinguishing MRI features between cerebral small vessel disease and multiple sclerosis. *T2 weighted image; #FLAIR: fluid attenuation inversion recovery; &SWI: susceptibility-weighted imaging; §T1 gadolinium enhancement. The MRI images are collected from the authors’ centres.
Enlarged perivascular spaces and fluid-filled subarachnoid spaces that surround cerebral microvessels in the brain parenchyma (also called Virchow-Robin spaces) are commonly found as hyperintense on T2 MRI and hypointense on T1 MRI, less than 3 mm, in the basal ganglia, centrum semiovale and brainstem.33 Enlarged perivascular spaces are commonly seen in CSVD and may be associated with cognitive impairment or hypertension. In MS, perivascular spaces are associated with increased inflammatory markers, but enlarged perivascular spaces are rarely visible.
Lesion is common enhanced on T1-weighted imaging after contrast injection in MS patients with acute attack. The nodular or ring-like (closed and open) enhancement correlates with altered BBB permeability in acute perivascular inflammation, thus discriminating active or chronic lesions.34 Gadolinium enhancement varies in size and shape and, usually, lasts a few days to weeks, with an average duration of 3 weeks (97% of lesions enhance in less than 2 months).35 Less than 40% of the lesions transform to black holes over 6 months, correlating pathologically with permanent demyelination and severe axonal loss.31 Black holes in MS are T1 white matter lesions with a hypointense appearance relative to normal-appearing white matter. Persistent black holes may be a marker of axonal loss and tissue destruction, different from the CSF-filled cavities of lacunae.31 Open ring sign can be used as a key point for differentiation, in particular, lesion enhancement is rare in CSVD.
In addition, cerebral microbleeds are small, round, 5–10 mm areas and homogeneous foci of hypointensity found on T2-weighted (gradient echo) (T2*) and susceptibility-weighted imaging, which are sensitive markers indicating BBB dysfunction in CAA or arteriosclerotic CSVD. Microbleeds are rarely found in MS patients. Central vein sign is the imaging manifestation of the perivenular nature of demyelinating plaques. The central vein sign distinguishes MS from inflammatory vasculopathy with a diagnostic accuracy of 100%.36 Ultra-high field MR and T2* sequences are helpful in finding this sign.
Both MS and CSVD undergo a chronically progressed disease course. Cognitive decline and brain atrophy are common findings among MS and CSVD.37 It is often hard to distinguish them solely through clinical manifestation and brain atrophy signs, that is, reduced grey matter volume and enlarged ventricles. However, brain atrophy caused by MS is often associated with lesion load,38 which is not typically related in CSVD.
Utilisation of high-field intensity MRI scanners is conducive to the detection of subtle structural changes in clinical practice and investigation, and perhaps helpful in diagnosing CSVD or MS. With regard to the lesion locations, the brain stem, periventricular and cerebral cortex are commonly attacked by MS and CSVD; lesions in juxtacortical areas, optic nerves and spinal cord are rarely found in CSVD but commonly damaged in MS. At present, 7T MRI scanning can assist to detect CSVD specific microinfarctions (50 µm to a few mm in diameter) in areas appearing normal in 3T MRI scans.39 Similarly, 7T MRI can also detect some microlesions in cortical and juxtacortical areas in MS patients, which are characteristic signs for differentiating from CSVD.
Conclusions and future perspective
Clinical similarities but divergent managements of CSVD and the early phase of MS highlight the need to timely differentiate WMD of these two origins. Patterns and locations of lesions as determined by multimodal MRI are instrumental to reveal the underlying aetiology. Periventricular white matter lesions are common in MS and CSVD. The most characteristic pathological lesions in MS are ‘Dawson fingers’. Age-related vascular white matter lesions mostly show a broad base along the ventricular margin, but occasionally one or more lesions may be found periventricular. Periventricular lesions can increase and fuse over time in both diseases, whereas corpus callosum lesions are usually found in MS but are rare in CSVD. Additionally, oligoclonal band presence in cerebral spinal fluid, spinal cord and optic lesions of MS offer additional clues distinguishing it from CSVD. Juxtacortical white matter is a characteristic location of MS lesions. Cortical lesions and juxtacortical lesions extending into the cortex are typical of MS in histopathological studies; conversely, cortical lesions have been found in CSVD, but juxtacortical lesions are rare.
The similar features of CSVD and MS in brain pathology, MRI findings and clinical presentation cloud the differential diagnosis in real-world busy clinic. New insight into previously unknown pathogenic aspects would offer new knowledge to better understand the white matter changes of these two origins and thus provide additional tools for differential diagnosis. Furthermore, biological changes often occur ahead of pathological damages that can be imaged by MRI, developing new humoral biological markers may provide additional clue for differentiation; for example, neurofilament light chain has recently been identified as a specific marker of MS progression.40 Additionally, regarding treatment options, examining immune-modulatory therapy in CSVD and vascular therapy in MS in preclinical and clinical settings may be justified due to the overlapping roles of vascular damage and inflammation in these two diseases.
Footnotes
Contributors: F-DS, YW, LY, YOL formulated the concept, L-JZ, TD-C and KS searched the literature, all authors drafted the manuscript.
Funding: We thank members of the Jing-Jin Neuroimmunology Team for helpful discussions; Dr. Luc Van Kaer and Elaine Shi for editing the manuscript; Work in the Authors’ Laboratories is supported in part by the National Science Foundation of China (Grants 91642205 and 81830038), funds of Advanced Innovation Center for Human Brain Protection, Capital Medical University, Beijing, China; funded by Tianjin Key Medical Discipline (Specialty) Construction Project (TJYXZDXK-004A).
Competing interests: None declared.
Provenance and peer review: Not commissioned; internally peer reviewed.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
Not applicable.
References
- 1. Matute C, Ransom BR. Roles of white matter in central nervous system pathophysiologies. ASN Neuro 2012;4:e00079. 10.1042/AN20110060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Frahm HD, Stephan H, Stephan M. Comparison of brain structure volumes in insectivora and primates. I. Neocortex. J Hirnforsch 1982;23:375–89. [PubMed] [Google Scholar]
- 3. Nonaka H, Akima M, Hatori T, et al. The Microvasculature of the cerebral white matter: arteries of the subcortical white matter. J Neuropathol Exp Neurol 2003;62:154–61. 10.1093/jnen/62.2.154 [DOI] [PubMed] [Google Scholar]
- 4. Vrselja Z, Brkic H, Curic G. Penetrating arteries of the cerebral white matter: the importance of vascular territories of delivering arteries and completeness of circle of Willis. Int J Stroke 2016;11:36–7. 10.1177/1747493015616639 [DOI] [PubMed] [Google Scholar]
- 5. Frohman EM, Racke MK, Raine CS. Multiple sclerosis--the plaque and its pathogenesis. N Engl J Med 2006;354:942–55. 10.1056/NEJMra052130 [DOI] [PubMed] [Google Scholar]
- 6. Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 2018;17:162–73. 10.1016/S1474-4422(17)30470-2 [DOI] [PubMed] [Google Scholar]
- 7. Ortiz GG, Pacheco-Moisés FP, Macías-Islas MÁ, et al. Role of the blood-brain barrier in multiple sclerosis. Arch Med Res 2014;45:687–97. 10.1016/j.arcmed.2014.11.013 [DOI] [PubMed] [Google Scholar]
- 8. Baecher-Allan C, Kaskow BJ, Weiner HL. Multiple sclerosis: mechanisms and immunotherapy. Neuron 2018;97:742–68. 10.1016/j.neuron.2018.01.021 [DOI] [PubMed] [Google Scholar]
- 9. Brown RB, Traylor M, Burgess S, et al. Do cerebral small vessel disease and multiple sclerosis share common mechanisms of white matter injury? Stroke 2019;50:1968–72. 10.1161/STROKEAHA.118.023649 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Nus M, Mallat Z. Immune-mediated mechanisms of atherosclerosis and implications for the clinic. Expert Rev Clin Immunol 2016;12:1217–37. 10.1080/1744666X.2016.1195686 [DOI] [PubMed] [Google Scholar]
- 11. Wiseman S, Marlborough F, Doubal F, et al. Blood markers of coagulation, fibrinolysis, endothelial dysfunction and inflammation in lacunar stroke versus non-lacunar stroke and non-stroke: systematic review and meta-analysis. Cerebrovasc Dis 2014;37:64–75. 10.1159/000356789 [DOI] [PubMed] [Google Scholar]
- 12. Moussaddy A, Levy A, Strbian D, et al. Inflammatory cerebral amyloid angiopathy, Amyloid-Β-related angiitis, and primary angiitis of the central nervous system: similarities and differences. Stroke 2015;46:e210–3. 10.1161/STROKEAHA.115.010024 [DOI] [PubMed] [Google Scholar]
- 13. Wermer MJH, Greenberg SM. The growing clinical spectrum of cerebral amyloid angiopathy. Curr Opin Neurol 2018;31:28–35. 10.1097/WCO.0000000000000510 [DOI] [PubMed] [Google Scholar]
- 14. Eng JA, Frosch MP, Choi K, et al. Clinical manifestations of cerebral amyloid angiopathy-related inflammation. Ann Neurol 2004;55:250–6. 10.1002/ana.10810 [DOI] [PubMed] [Google Scholar]
- 15. Viswanathan A, Greenberg SM. Cerebral amyloid angiopathy in the elderly. Ann Neurol 2011;70:871–80. 10.1002/ana.22516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Wong SM, Jansen JFA, Zhang CE, et al. Blood-brain barrier impairment and hypoperfusion are linked in cerebral small vessel disease. Neurology 2019;92:e1669–77. 10.1212/WNL.0000000000007263 [DOI] [PubMed] [Google Scholar]
- 17. Wardlaw JM, Sandercock PAG, Dennis MS, et al. Is breakdown of the blood-brain barrier responsible for lacunar stroke, leukoaraiosis, and dementia? Stroke 2003;34:806–12. 10.1161/01.STR.0000058480.77236.B3 [DOI] [PubMed] [Google Scholar]
- 18. Rajani RM, Quick S, Ruigrok SR, et al. Reversal of endothelial dysfunction reduces white matter vulnerability in cerebral small vessel disease in rats. Sci Transl Med 2018;10:eaam9507. 10.1126/scitranslmed.aam9507 [DOI] [PubMed] [Google Scholar]
- 19. Jakimovski D, Topolski M, Kimura K, et al. Decrease in secondary neck vessels in multiple sclerosis: a 5-year longitudinal magnetic resonance angiography study. Curr Neurovasc Res 2019;16:215–23. 10.2174/1567202616666190612111127 [DOI] [PubMed] [Google Scholar]
- 20. Jakimovski D, Zivadinov R, Pelizzari L, et al. Lipoprotein(A) levels are associated with the size of extracranial arteries in multiple sclerosis. J Vasc Res 2020;57:16–23. 10.1159/000502115 [DOI] [PubMed] [Google Scholar]
- 21. Cocozza S, Canna A, Lanzillo R, et al. Lack of correlation between extracranial venous abnormalities and multiple sclerosis: a quantitative MRI study. Br J Radiol 2016;89:20160321. 10.1259/bjr.20160321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Murphy OC, Kwakyi O, Iftikhar M, et al. Alterations in the retinal vasculature occur in multiple sclerosis and exhibit novel correlations with disability and visual function measures. Mult Scler 2020;26:815–28. 10.1177/1352458519845116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Wardlaw JM, Debette S, Jokinen H, et al. ESO guideline on covert cerebral small vessel disease. Eur Stroke J 2021;6:IV. 10.1177/23969873211027002 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 24. Grueter BE, Schulz UG. Age-related cerebral white matter disease (Leukoaraiosis): a review. Postgrad Med J 2012;88:79–87. 10.1136/postgradmedj-2011-130307 [DOI] [PubMed] [Google Scholar]
- 25. Kobari M, Meyer JS, Ichijo M, et al. Leukoaraiosis: correlation of MR and CT findings with blood flow, atrophy, and cognition. AJNR Am J Neuroradiol 1990;11:273–81. [PMC free article] [PubMed] [Google Scholar]
- 26. Wardlaw JM, Smith EE, Biessels GJ, et al. Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol 2013;12:822–38. 10.1016/S1474-4422(13)70124-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Duering M, Biessels GJ, Brodtmann A, et al. Neuroimaging standards for research into small vessel disease-advances since 2013. Lancet Neurol 2023;22:602–18. 10.1016/S1474-4422(23)00131-X [DOI] [PubMed] [Google Scholar]
- 28. Rovira À, Wattjes MP, Tintoré M, et al. Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis-clinical implementation in the diagnostic process. Nat Rev Neurol 2015;11:471–82. 10.1038/nrneurol.2015.106 [DOI] [PubMed] [Google Scholar]
- 29. Geraldes R, Ciccarelli O, Barkhof F, et al. The current role of MRI in differentiating multiple sclerosis from its imaging mimics. Nat Rev Neurol 2018;14:213. 10.1038/nrneurol.2018.39 [DOI] [PubMed] [Google Scholar]
- 30. van der Flier WM, van Straaten ECW, Barkhof F, et al. Small vessel disease and general cognitive function in nondisabled elderly: the LADIS study. Stroke 2005;36:2116–20. 10.1161/01.STR.0000179092.59909.42 [DOI] [PubMed] [Google Scholar]
- 31. Sahraian MA, Radue E-W, Haller S, et al. Black holes in multiple sclerosis: definition, evolution, and clinical correlations: black holes in MS. Acta Neurol Scand 2010;122:1–8. 10.1111/j.1600-0404.2009.01221.x [DOI] [PubMed] [Google Scholar]
- 32. Trojano M, Amato MP. Progress in multiple sclerosis — from diagnosis to therapy. Nat Rev Neurol 2018;14:72–4. 10.1038/nrneurol.2018.3 [DOI] [PubMed] [Google Scholar]
- 33. Bokura H, Kobayashi S, Yamaguchi S. Distinguishing silent lacunar infarction from enlarged Virchow-Robin spaces: a magnetic resonance imaging and pathological study. J Neurol 1998;245:116–22. 10.1007/s004150050189 [DOI] [PubMed] [Google Scholar]
- 34. Cotton F, Weiner HL, Jolesz FA, et al. MRI contrast uptake in new lesions in relapsing-remitting MS followed at weekly intervals. Neurology 2003;60:640–6. 10.1212/01.wnl.0000046587.83503.1e [DOI] [PubMed] [Google Scholar]
- 35. Davis M, Auh S, Riva M, et al. Ring and nodular multiple sclerosis lesions: a retrospective natural history study. Neurology 2010;74:851–6. 10.1212/WNL.0b013e3181d31df5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Maggi P, Absinta M, Grammatico M, et al. Central vein sign differentiates multiple sclerosis from central nervous system inflammatory vasculopathies. Ann Neurol 2018;83:283–94. 10.1002/ana.25146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Eijlers AJC, Dekker I, Steenwijk MD, et al. Cortical atrophy accelerates as cognitive decline worsens in multiple sclerosis. Neurology 2019;93:e1348–59. 10.1212/WNL.0000000000008198 [DOI] [PubMed] [Google Scholar]
- 38. Eshaghi A, Marinescu RV, Young AL, et al. Progression of regional grey matter atrophy in multiple sclerosis. Brain 2018;141:1665–77. 10.1093/brain/awy088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Trattnig S, Springer E, Bogner W, et al. Key clinical benefits of neuroimaging at 7 T. Neuroimage 2018;168:477–89. 10.1016/j.neuroimage.2016.11.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Ziemssen T, Akgün K, Brück W. Molecular biomarkers in multiple sclerosis. J Neuroinflammation 2019;16:272. 10.1186/s12974-019-1674-2 [DOI] [PMC free article] [PubMed] [Google Scholar]