Abstract
Patient: Female, 40-year-old
Final Diagnosis: Hypersplenism
Symptoms: Abdominal pain
Clinical Procedure: —
Specialty: Surgery
Objective:
Unusual clinical course
Background:
Hypersplenism, the rapid and premature destruction of blood cells, encompasses a triad of splenomegaly, cytopenias (anemia, leukopenia, or thrombocytopenia), and compensatory bone marrow proliferation. Secondary hypersplenism results from non-intrinsic splenic diseases, such as hemoglobinopathies. Sickle cell disease consists of a group of genotypes, where hemoglobin sickle C disease (HbSC) is the inheritance of hemoglobin S with hemoglobin C. Most homozygous genotypes undergo complete auto-splenectomy by age 6 years, whereas those with HbSC disease rarely do. We report a rare case of hypersplenism and massive splenomegaly in an adult with sickle cell disease, the HbSC genotype, requiring splenectomy.
Case Report:
A 41-year-old woman with known splenomegaly initially presented to the general surgery clinic for management of abdominal pain. She was found to have anemia, indicating cytopenia likely from hypersplenism. Consequently, she underwent splenic artery embolization, followed by an exploratory laparotomy and splenectomy, with an unremarkable postoperative course.
Conclusions:
Acute splenic sequestration crisis can result from hypersplenism, a potentially fatal complication of sickle hemoglobinemia. The continuous cycle of sickled cell entrapment and stasis causes numerous splenic infarctions, forming splenic parenchymal scar tissue which reduces the spleen’s size and functionality – the process of auto-splenectomy. Adults rarely experience these crises past adolescence, which are secondary to the scarring and atrophy from premature auto-splenectomy. Our patient’s spleen measured 21.1 cm, larger than the average adult’s spleen. In our case, adjunctive preoperative splenic artery embolization likely contributed to decreased intraoperative blood loss during splenectomy, mitigating the need for perioperative transfusions.
Key words: Anemia, Sickle Cell; Embolization, Therapeutic; Hypersplenism; Sickle Cell Trait; Splenectomy
Introduction
Hypersplenism is characterized by rapid and premature destruction of blood cells. It encompasses a triad of splenomegaly, cytopenias, including anemia, leukopenia, and thrombocytopenia, and compensatory bone marrow proliferation [1,2]. Within this triad, splenomegaly refers to a spleen with a weight of greater than 150 g and a craniocaudal length greater than 11 cm. Based on its etiological causes, hypersplenism can be classified as primary or secondary. Primary hypersplenism refers to causes pertaining to intrinsic splenic disorders, whereas secondary hypersplenism is the result of another underlying disease, such as hemolytic anemias, such as sickle cell disease (SCD). Hypersplenism can lead to acute splenic sequestration crisis, which is defined as an acute drop in hemoglobin of 2 g/dL in combination with splenomegaly.
SCD consists of a group of hemoglobinopathies, all caused by the inheritance of defective β-globin alleles, which result in inflexible sickled red blood cells (RBC). Sickle cell anemia, the form more prone to vaso-occlusive crises posing greater risks of microvascular damage, is characterized by the inheritance of 2 hemoglobin S alleles (HbSS). Another genotype of SCD is hemoglobin sickle C disease (HbSC), in which 1 hemoglobin S allele is inherited in association with 1 hemoglobin C allele [3]. In both genotypes, under the deoxygenated state the RBCs can easily polymerize, resulting in sickling of cells causing sludging and impeding appropriate tissue perfusion. The spleen is particularly vulnerable to this process. In sickle cell anemia, hypersplenism is commonly observed in infancy, before repeated episodes of vaso-occlusion and infarction cause progressive splenic atrophy, eventually leading to auto-splenectomy. Most individuals with sickle cell anemia undergo complete auto-splenectomy by 6 years of age, whereas those with HbSC rarely do [2]. Here, we report a rare case of hypersplenism and massive splenomegaly in an adult with SCD, the HbSC genotype, requiring splenectomy.
Case Report
The patient was a 41-year-old woman who initially presented to the general surgery clinic for the management of abdominal pain and known splenomegaly via computed tomography (CT) of the abdomen and pelvis. She had a past medical history significant for pituitary adenoma, gastritis, carpal tunnel syndrome, allergic rhinitis, SCD (HbSC genotype) status after multiple blood transfusions, and various hospitalizations for sickle cell pain crises. Physical examination was remarkable for left upper quadrant abdominal tenderness and splenomegaly. Laboratory workup was consistent with anemia, showing a hemoglobin level of 8.2 g/dL, hematocrit level of 25%, platelet count of 122×103/uL (as low as 109×103/uL), and white blood cell (WBC) count of 6.0×103/uL (as low as 4.7×103/uL). CT of the abdomen and pelvis with contrast showed marked splenomegaly, approximately 21.2 cm in the greatest dimension. During the clinic visit, options for splenectomy were discussed, along with the option for adjunctive splenic artery embolization to reduce spleen size and vascularity, which the patient strongly opted to undergo. However, in the interim of choosing a date for surgery, the patient had a severe sickle cell crisis due to community-acquired pneumonia, which required hospitalization, with multiple blood transfusions leading to the formation of alloantibodies. Upon repeat typing and screening, the patient was found to have crossmatch ABO and D compatible E-, K-, K0, with a frequency of less than 1/1000 compatible donors. During this admission, multiple blood donor facilities were contacted to find a crossmatch for the patient’s rare blood type, which was not found. Subsequently, a decision was made to proceed with the previously planned procedure for splenic artery embolization by interventional radiology, followed by splenectomy for control of hypersplenism and intractable pain. She consequently underwent splenic artery embolization (Figure 1), followed by exploratory laparotomy and splenectomy (Figure 2). This resulted in minimal intraoperative and postoperative blood loss. Microscopic examination of the spleen showed shrunken white pulp and markedly expanded red pulp (Figure 3). The patient’s laboratory workup noted leukocytosis, with the highest WBC at 27.9×103/uL, and thrombocytosis, with highest platelet count at 791×103/uL, in the days following surgery, which normalized over a 2-week period. She was discharged in stable condition with appropriate follow-ups with the departments of General Surgery, Medicine, and Hematology & Oncology. At her 9-month postoperative follow-up, the patient’s complete blood count panel showed an appropriate response, with improvements in hemoglobin and hematocrit levels, 10.0g/dL and 32.8%, respectively, as well as normalized WBC count at 6.7×103/uL and platelet count at 376×103/uL.
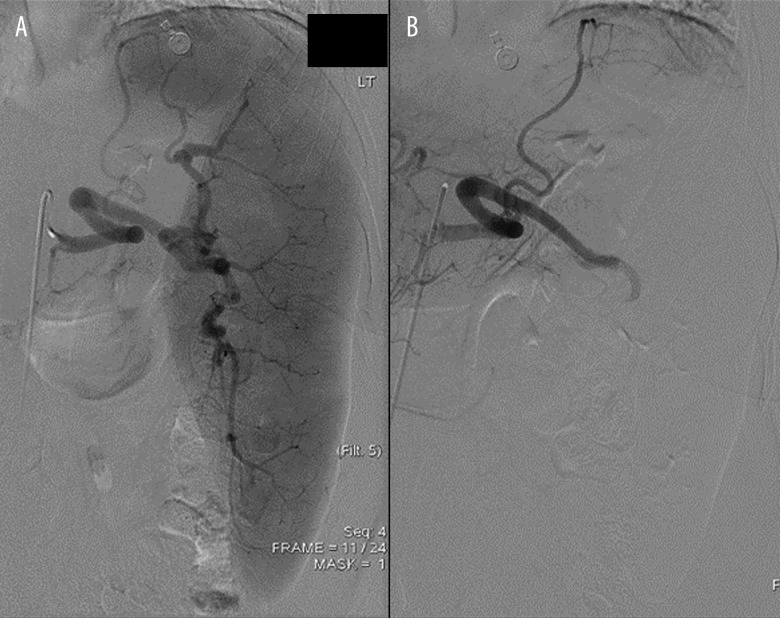
Figure 1.
Splenic angiography via right common femoral artery access. (A) Prior to embolization, splenic angiography demonstrates significant shunting to the splenic artery and splenomegaly. (B) After embolization of splenic artery, splenic angiography is notable for superior branch of splenic artery with blood flow; the remainder of the splenic vessels demonstrate no circulation in response to arterial embolization.
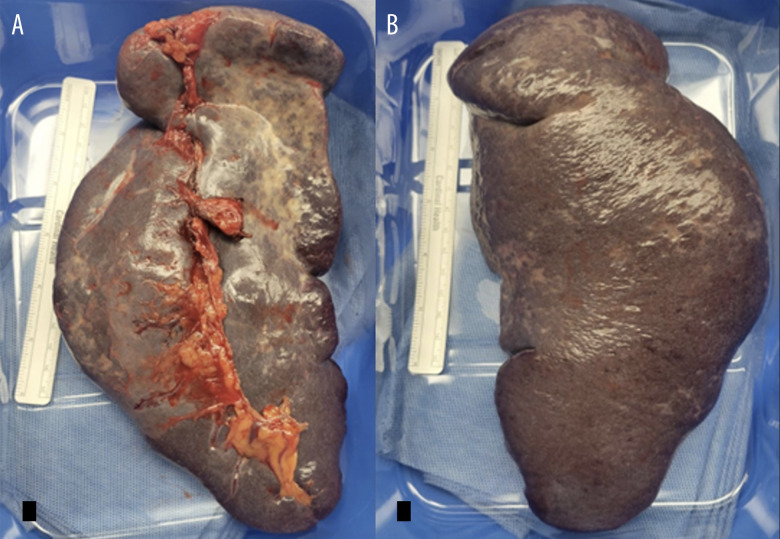
Figure 2.
Encapsulated intact spleen, measuring 18×13×6 cm with a weight of 890 g. The capsule is tan gray-red and smooth. (A) Anterior spleen. (B) Posterior spleen with vascular pedicle.
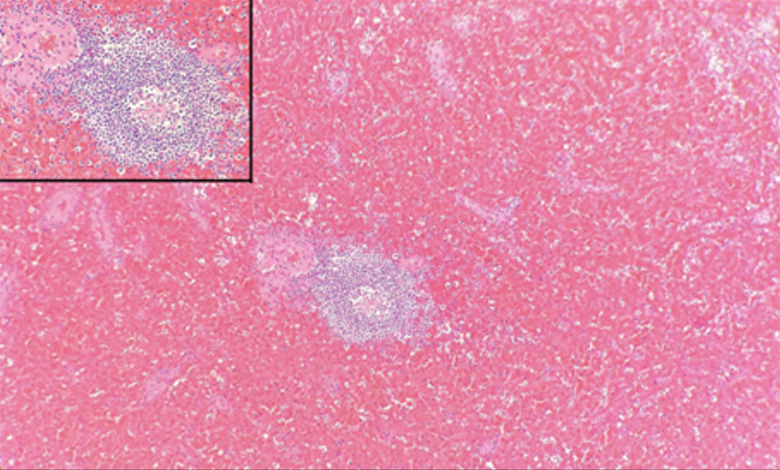
Figure 3.
Microscopic examination of the spleen showing shrunk white pulps and markedly expanded red pulps. Hematoxylin and eosin stain (4×), with insert showing shrunk white pulps (10×).
Discussion
Hemoglobinopathies occur as a result of inheriting a point mutation in the β-globin gene of adult hemoglobin (HbA) and these mutations coexist with the normal HbA in blood [4]. The presence of mutant HbA has numerous sequelae, including induction of extravascular and intravascular hemolysis and coagulopathies. Hemoglobinopathy encompasses a spectrum of disorders that vary immensely in the severity of their clinical presentations and effects on the spleen. SCD is one such disease in which those frequently exposed to a deoxygenated environment leads to hemoglobin polymerization with eventual sickling that drives the transition of plasma and its contents from a dilute solution to a more viscous, gel-like substance [5,6]. The most common genotypes of SCD are HbSS and HbSC. The HbSC genotype is present in approximately 30% of patients with SCD, with clinical signs often being milder than those of HbSS. There is substantial variability in the presentation of illness ascribed to the composition of the HbSC RBCs, which typically contain equal quantities of HbC and HbS [7,8]. Dehydration of RBCs as a result of HbC-induced impaired membrane transport results in increased mean corpuscular hemoglobin concentration, such that HbS at lower concentrations can readily polymerize [9]. In addition, increased blood viscosity, reduced RBC deformability, and increased aggregation have been identified as contributing to the pathophysiology of HbSC disease. Individuals with HbSC commonly have normal-sized spleens into adulthood but can experience hypersplenism with related complications, such as splenic sequestration and splenomegaly. Individuals with HbSC rarely undergo the complete auto-splenectomy that is seen in the homozygous HbSS genotype; rather, these patients often have persistent splenomegaly secondary to recurrent sequestration events [10,11]. Usually, the vaso-occlusive complications of the disease manifest well past the fourth or fifth decades and can become more recurrent as functional asplenia sets in. RBC transfusions have been the standard of treatment for years in patients with HbSC [12]; however, a minority of patients can have a more severe course requiring splenectomy [13].
Hypersplenism is often associated with the clinical presentation of SCD and associated syndromes [4]. It involves the sequestration of blood cells, leading to multiple cytopenias and subsequent splenomegaly. Hypersplenism can lead to acute splenic sequestration crisis, a potentially life-threatening complication characterized by splenomegaly with a drop in hemoglobin by at least 2 g/dL [14,15]. Due to the more acute and severe clinical manifestations of HbS homozygotes beginning at an early age, children with HbSS often undergo recurrent episodes of splenic sequestration crisis until the age of 6 years [14]. The continuous cycle of sickled cell entrapment and stasis leads to numerous splenic infarctions; scar tissue forms within the parenchyma of the spleen, thereby shrinking its size and reducing its functionality – a process referred to as autosplenectomy [11,12]. Splenic sequestration crisis has been shown to occur in nearly 10% to 15% of adults with the HbSC genotype. Adults rarely experience these crises past adolescence, which are secondary to the scarring and atrophy from premature auto-splenectomy [12].
While splenectomy does not cure the manifestations of SCD, it is the preferred treatment for hypersplenism in the setting of splenomegaly and can be a part of managing its long-term complications. Indications for splenectomy include but are not limited to hypersplenism and resulting cytopenia(s), acute splenic sequestration crisis, massive splenic infarction, intractable pain, and infections of the spleen complicated by abscess formation [13]. However, splenectomy is associated with significant complications, with the most notable being intraoperative blood loss necessitating perioperative transfusions [16]. Several studies have found that splenic artery embolization (SAE) used as a preoperative adjunct to splenectomy reduces the risk of intraoperative hemorrhage [17–19]. These investigations focused on hypersplenism caused by portal hypertension. A 2022 meta-analysis including nearly a dozen retrospective studies and randomized trials conjectured that partial splenic artery embolization was as effective as and safer than splenectomy for correcting cytopenias secondary to hypersplenism. This difference was further pronounced when the spleen was larger than 18 cm. Similar findings were reported by Poulin et al, who found that preoperative SAE in spleen sizes greater than 20 cm greatly reduced blood loss [20]. Similarly, other benefits of SAE include reduced size and softening of the spleen, allowing for easier manipulation during laparoscopic splenectomy [7]. However, SAE is associated with an elevated risk of bacteremia and splenic abscess formation. In addition to partial or total splenectomy and embolization procedures to reduce the size of the spleen, hypersplenism can be managed medically pertaining to etiology and external radiofrequency ablation, with both showing symptomatic relief [21].
Our patient’s spleen by comparison measured 21.1 cm, which is in accordance with the previously given values studied in the literature. Additionally, our patient had an unusual blood type for whom a crossmatch could not be obtained. The patient had developed multiple antibodies from the blood transfusions, making it very difficult to find a compatible blood type. Various transfusion centers were contacted, with none being able to provide compatible packed RBCs. As it turned out, our patient did not require any further perioperative blood transfusions. The adjunctive preoperative SAE likely contributed to less intraoperative blood loss during splenectomy and mitigated the need for perioperative transfusions. Previously, there have been reports of adults with splenomegaly, even in those with the HbSS genotype, hypothesized to be lower in severity [22]. Another case study reported a patient with the HbSC genotype with splenomegaly who presented with an acute splenic sequestration crisis, in which the patient was treated with plasma RBC exchange rather than splenectomy and responded well with reduction in spleen size [23]. Additionally, our patient responded appropriately to the adjunctive treatment of preoperative SAE followed by splenectomy, as seen by improvements in all 3 cell lines 9 months after surgery. The present case is unique due to the patient’s presence of the HbSC genotype in combination with hypersplenism, which had not been previously reported.
A limitation of this study is that this was a case report involving 1 patient. This topic would benefit from future studies involving more patients and with data analysis. Further research needs to be conducted on predicting patient populations at greater risk of experiencing hypersplenism-associated complications. Other potential directions of future studies are the exploration of predictive genetic markers of disease and novel management strategies of hypersplenism.
Conclusions
The triad of hypersplenism is often associated with the clinical presentation of SCD and associated syndromes, with severe complications of acute splenic sequestration crisis. Splenectomy, if feasible, should be considered in all symptomatic patients with evidence of splenomegaly, given the recurrence of hyper-splenism and risks of complications with other procedures or medical management. Future research needs to be conducted on identifying risk factors of complications, predictive genetic markers of disease, and strategies suited for the management of hypersplenism.
Footnotes
Publisher’s note: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher
Institute Where the Patient Was Treated
Brooklyn Hospital Center, Brooklyn, NY, USA.
Declaration of Figures’ Authenticity
All figures submitted have been created by the authors who confirm that the images are original with no duplication and have not been previously published in whole or in part.
References:
- 1.Lv Y, Lau WY, Li Y, et al. Hypersplenism: History and current status. Exp Ther Med. 2016;12(4):2377–82. doi: 10.3892/etm.2016.3683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ladu AI, Aiyenigba AO, Adekile A, Bates I. The spectrum of splenic complications in patients with sickle cell disease in Africa: A systematic review. Br J Haematol. 2021;193(1):26–42. doi: 10.1111/bjh.17179. [DOI] [PubMed] [Google Scholar]
- 3.da Guarda CC, Yahouédéhou SCMA, Santiago RP, et al. Sickle cell disease: A distinction of two most frequent genotypes (HbSS and HbSC). PLoS One. 2020;15(1):e0228399. doi: 10.1371/journal.pone.0228399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rivera-Ruiz M, Varon J, Sternbach GL. Acute splenic sequestration in an adult with hemoglobin S-C disease. Am J Emerg Med. 2008;26(9):1064.e5–8. doi: 10.1016/j.ajem.2008.02.031. [DOI] [PubMed] [Google Scholar]
- 5.Old J. Chapter 71 – Hemoglobinopathies and thalassemias. In: Rimoin D, Pyeritz R, Korf B, editors. Emery and Rimoin’s principles and practice of medical genetics (sixth edition) [Internet]. Oxford: Academic Press; 2013. pp. 1–44. Available from: https://www.sciencedirect.com/science/article/pii/B9780123838346000756 [cited 2023 Oct 29] [Google Scholar]
- 6.Jobe SM, Di Paola J. 9 – Congenital and acquired disorders of platelet function and number. In: Kitchens CS, Kessler CM, Konkle BA, Streiff MB, Garcia DA, editors. Consultative hemostasis and thrombosis (fourth edition) [Internet]. Philadelphia: Elsevier; 2019. pp. 145–66. Available from: https://www.sciencedirect.com/science/article/pii/B9780323462020000091 [cited 2023 Oct 29] [Google Scholar]
- 7.Wu Z, Zhou J, Pankaj P, Peng B. Comparative treatment and literature review for laparoscopic splenectomy alone versus preoperative splenic artery embolization splenectomy. Surg Endosc. 2012;26(10):2758–66. doi: 10.1007/s00464-012-2270-z. [DOI] [PubMed] [Google Scholar]
- 8.Sathi BK. Hemoglobin SC Disease: Phenotypic variability and therapeutic options. AJBSR. biomedgrid. 2020;7(5):441. [Google Scholar]
- 9.Lionnet F, Hammoudi N, Stojanovic KS, et al. Hemoglobin sickle cell disease complications: A clinical study of 179 cases. Haematologica. 2012;97(8):1136–41. doi: 10.3324/haematol.2011.055202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Subbannan K, Ustun C, Natarajan K, et al. Acute splenic complications and implications of splenectomy in hemoglobin SC disease. Eur J Haematol. 2009;83(3):258–60. doi: 10.1111/j.1600-0609.2009.01270.x. [DOI] [PubMed] [Google Scholar]
- 11.Al-Salem AH. Splenic complications of sickle cell anemia and the role of splenectomy. ISRN Hematol. 2011;2011:864257. doi: 10.5402/2011/864257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jeker R. [Hypersplenism.] Ther Umsch. 2013;70(3):152–56. doi: 10.1024/0040-5930/a000383. [in German] [DOI] [PubMed] [Google Scholar]
- 13.Wholey MH, Chamorro HA, Rao G, Chapman W. Splenic infarction and spontaneous rupture of the spleen after therapeutic embolization. Cardiovasc Radiol. 1978;1(4):249–53. doi: 10.1007/BF02552051. [DOI] [PubMed] [Google Scholar]
- 14.Lakhani JD, Gill R, Mehta D, Lakhani SJ. Prevalence of splenomegaly and splenic complications in adults with sickle cell disease and its relation to fetal hemoglobin. Int J Hematol Oncol Stem Cell Res. 2022;16(4):198–208. doi: 10.18502/ijhoscr.v16i4.10877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lane PA, O’Connell JL, Lear JL, et al. Functional asplenia in hemoglobin SC disease. Blood. 1995;85(8):2238–44. [PubMed] [Google Scholar]
- 16.Zaitoun MMA, Basha MAA, Raafat A, et al. Splenectomy for hypersplenism with or without preoperative splenic artery embolisation. European Radiology Experimental. 2018;2(1):23. [Google Scholar]
- 17.Tan Y, Wang J, Sun L, Ye Y. Repeated partial splenic artery embolization for hypersplenism improves platelet count. Open Med (Wars) 2022;17(1):808–15. doi: 10.1515/med-2022-0479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.He XH, Gu JJ, Li WT, et al. Comparison of total splenic artery embolization and partial splenic embolization for hypersplenism. World J Gastroenterol. 2012;18(24):3138–44. doi: 10.3748/wjg.v18.i24.3138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Naoum JJ, Silberfein EJ, Zhou W, et al. Concomitant intraoperative splenic artery embolization and laparoscopic splenectomy versus laparoscopic splenectomy: Comparison of treatment outcome. Am J Surg. 2007;193(6):713–18. doi: 10.1016/j.amjsurg.2006.09.043. [DOI] [PubMed] [Google Scholar]
- 20.Poulin E, Thibault C, Mamazza J, et al. Laparoscopic splenectomy: Clinical experience and the role of preoperative splenic artery embolization. Surg Laparosc Endosc. 1993;3(6):445–50. [PubMed] [Google Scholar]
- 21.Feng Kai, Ma KS, Dong JH. [Changes of serum Tuftsin content among hypersplenism patients before and after radiofrequency ablation or splenectomy.] Acta Academiae Medicinae Militaris Tertiae. 2006;28:1983–85. [in Chinese] [Google Scholar]
- 22.Asnani MR, Williams A, Reid M. Splenic enlargement in adults with homozygous sickle cell disease: The Jamaican experience. Hematology. 2013;18(1):46–49. doi: 10.1179/1607845412Y.0000000036. [DOI] [PubMed] [Google Scholar]
- 23.Vijayanarayanan A, Omosule AJ, Saad H, et al. Acute splenic sequestration crisis in hemoglobin SC disease: Efficiency of red cell exchange. Cureus. 2020;12(12):e12224. doi: 10.7759/cureus.12224. [DOI] [PMC free article] [PubMed] [Google Scholar]