Abstract
Bovine viral diarrhea virus (BVDV), a pestivirus of the Flaviviridae family, is an economically important cattle pathogen with a worldwide distribution. Both noncytopathic (ncp) and cytopathic (cp) biotypes of BVDV can be isolated from persistently infected cattle suffering from the lethal mucosal disease. The cp biotype correlates with the production of the NS3 nonstructural protein, which in the corresponding ncp biotype is present in its uncleaved form, NS23. Previously, we have shown that cp but not ncp BVDV induces the formation of α/β interferons in bovine macrophages. In this study, we demonstrate that ncp BVDV inhibits the induction of apoptosis and the expression of interferon α/β by poly(IC), a synthetic double-stranded RNA (dsRNA). Inhibition was observed only in cells which had been infected with ncp BVDV at least 12 h prior to the addition of dsRNA, which indicates that expression of viral proteins is necessary for the ncp virus to inhibit the effects of poly(IC). Additional experiments using transfected poly(IC) showed that ncp BVDV interfered with the intracellular action of dsRNA rather than with its uptake into the cells. Infected cells were not resistant to induction of apoptosis by actinomycin D or staurosporine, which suggests that ncp BVDV may specifically interfere with signaling through dsRNA. Interference with the innate antiviral host responses may explain the successful establishment of persistent infection by ncp BVDV in fetuses early in their development.
To establish a persistent infection, a virus must overcome the defense mechanisms of both the innate and the specific immune responses. Antigenic changes, latency, and replication in immunologically privileged sites permit the viruses to escape the effects of T cells and antibodies (4, 10, 20). Viral mechanisms directed against the innate immune system target, among others, the host's cytokine response and the complement system (4, 51, 68). Apoptosis and the interferon (IFN) response are important antiviral defense mechanisms that act at the level of the host cells. The fact that these mechanisms may be triggered hours to days before the onset of the virus-specific immune response highlights their role as a first line of defense. Moreover, as apoptosis and the IFN response are a characteristic of virtually all cells of metazoan animals, they help the host to fight a broad range of viruses with different cell and tissue tropisms. It is no surprise, therefore, that viruses have evolved a myriad of mechanisms that prevent apoptosis and subvert the IFN response. In the past few years, several viral gene products with antiapoptotic or anti-IFN activities have been identified and described in extensive reviews (15, 18, 51, 57, 62, and references therein). These strategies include modulation of the Bcl-2/Bax pathway, interference with caspases, a group of proteases known to be part of the death effector mechanism of apoptosis, or inhibition of the PKR/RNase L pathway.
Bovine viral diarrhea virus (BVDV), together with classical swine fever and border disease viruses of sheep, belongs to the genus Pestivirus of the Flaviviridae family. BVDV is an economically important cattle pathogen with a worldwide distribution. The virus causes acute (transient) and persistent infections. Cattle infected acutely with BVDV may show either no symptoms or mild diarrhea (caused by “avirulent” BVDV strains), but severe thrombocytopenia and hemorrhages have also been reported (virulent strains) (42, 66). According to their effect in cell cultures, cytopathic (cp) and noncytopathic (ncp) biotypes of BVDV can be isolated. Infection of pregnant animals with the ncp biotype may result in embryo death, resorption, and stillbirth and induce nonfatal teratogenic damage or may lead to the birth of persistently infected calves. Such calves are immunotolerant to the infecting BVDV strain and remain viremic for the rest of their lives. Such persistently infected animals are predisposed to infections with other pathogens and run a high risk of developing the fatal mucosal disease which is characterized by extensive lesions in the gastrointestinal tract (43, 46, 66, and references therein). From animals succumbing to mucosal disease, both an ncp and an antigenically related cp BVDV can be isolated; they are hence referred to as a virus pair (11). The cp biotype may arise in persistently infected animals by genomic rearrangement of the ncp virus, e.g., insertion of cellular sequences or rearrangements in the viral genome (for reviews, see references 42 and 53). In every case studied so far, the genomic changes leading to the cp biotype are paralleled by the production of the nonstructural protein NS3 (p80).
Previously, we reported that cp BVDV induce the synthesis of IFN-α/β (IFN type I) in infected macrophages (1, 50) and kill their host cells by apoptosis (59, 72). By contrast, ncp biotypes of BVDV do not induce the synthesis of IFN, and cells show no signs of viral infection, even though the viral titers produced by cp and ncp virus pairs may be similar. In this paper, we show that cells infected with ncp BVDV resist cell death and do not form IFN in response to the synthetic double-stranded RNA (dsRNA) poly(IC). Resistance to the potent proapoptotic and IFN-inducing effects of dsRNA may be a key factor in the successful invasion of the fetus and lifelong persistence of the virus.
MATERIALS AND METHODS
Reagents.
Fetal calf serum (FCS) and cell culture media were purchased from Seromed (Biochrom, Munich, Germany). FCS was free of BVDV and antibody to BVDV as tested by virus isolation and serum neutralization assays, respectively. Synthetic polyribonucleotides [polyinosinic-polycytidylic acid, poly(IC); polyadenylic-polyuridylic acid, poly(AU); polycytidylic acid, poly(C); and polyinosinic acid, poly(I)] and staurosporine were from Sigma. Actinomycin D was from Alexis Corporation (Läufelfingen, Switzerland). All other chemicals were of the highest purity commercially available.
Cells and viruses.
Primary bovine turbinate (BT) cells were prepared from fetuses obtained from a local abattoir and maintained in minimal essential medium supplemented with 7% FCS (2% FCS after viral infection), penicillin (100 IU/ml), and streptomycin (100 μg/ml) at 37°C in a humidified 5% CO2 atmosphere. MDBK cells were obtained from the American Type Culture Collection (Manassas, Va.) and maintained under the same conditions as the BT cells. Monocyte-derived macrophages (Mφ) were obtained from the blood of Red Holstein cows as described previously (31). All cells were found to be free of BVDV by immunofluorescence testing. The TGAC (54) and SuwaCP cp BVDV strains and the TGAN (54), SD-1, SuwaNCP, R6229/95, R5013/96, and 890 ncp viruses were used (all strains are of BVDV genotype I except for strain 890, which belongs to genotype II). Strains 890, R6229/95, and R5013/96 are virulent strains, whereas all other strains are avirulent. The TGAC and TGAN virus strains were kindly provided by V. Moennig (Hannover, Germany), and strain 890 was kindly provided by J. F. Ridpath (Ames, Iowa), whereas SuwaCP, SuwaNCP, R6229/95, and R5013/96 are virus strains isolated at our institute. SuwaCP and SuwaNCP are a virus pair isolated from an animal with mucosal disease (50), and R6229/95 and R5013/96 are from acutely infected animals showing thrombocytopenia and hemorrhages. Vesicular stomatitis virus (VSV) (strain Indiana) was kindly provided by H. Hengartner, Institute of Experimental Immunology, Zurich, Switzerland. Viruses were passaged and titrated on BT cells as described (2), and the titer of the virus stocks was calculated according to Reed and Muench (52).
Virus infection.
BT cells seeded in microwell plates (96 wells) or six-well plates at a density of 106 (microwell plate) or 1.2 × 106 (six-well plate) cells/plate were infected with the appropriate BVDV strain in a small volume of culture medium without FCS at a multiplicity of infection (MOI) of 1 for 1 h at 37°C. After adsorption of the virus, the inoculum was removed by washing the cells in culture medium without FCS prior to the addition of complete medium with 2% FCS. Using an MOI of 1, expression of NS3 or NS23 was detectable at 6 h postinfection by Western blot or immunohistochemistry, and all cells expressed NS3/NS23 around 12 to 18 h postinfection, as analyzed by immunofluorescence staining using an anti-NS3/NS23 antibody.
DNA fragmentation.
The fragmentation of cellular DNA was analyzed quantitatively by fluorescence-activated cell sorting (FACS) analysis according to Cossarizza et al. (16). Briefly, adherent and detached cells were collected by centrifugation at 250 × g and washed in phosphate-buffered saline (PBS), and the cell pellet (105 cells) was lysed in 250 μl of 0.1 M sodium citrate (pH 6.5)–1% Triton X-100–10 μg of propidium iodide (PI) per ml. Nuclei were analyzed after a 30-min incubation at 4°C in the dark with a FACScan flow cytometer (Becton Dickinson, San José, Calif.), and a minimum of 104 nuclei were analyzed.
Measurement of the cellular redox state.
The cellular redox state was analyzed by determination of the oxidation of dichlorofluorescin to the fluorescent dichlorofluorescein (DCF) and FACS analysis as described (12). Adherent and detached cells were collected as described above and resuspended in 500 μl of PBS. After addition of 10 μM 2′,7′-dichlorodihydrofluorescein diacetate (Kodak, Rochester, N.Y.), cells were incubated for 30 min at 37°C in the dark, followed immediately by FACS analysis. To separate viable and dead cells, PI (final concentration, 10 μg/ml) was added 10 min prior to the end of the incubation. Cell fluorescence was analyzed with a FACScan flow cytometer, with the fluorescence of DCF analyzed in channel 1 (FL1; log scale) and that of PI in channel 2 (FL2; log scale). A minimum of 104 cells per sample were analyzed, and the oxidation state (geometric mean) of the viable cells was calculated by gating for the cells excluding PI, which cannot pass through the intact plasma membrane. For comparison of the different samples, preparations were recorded at single instrument amplification settings.
IFN-α/β activity.
Procedures for measuring biological activity of IFN-α/β in supernatants of virus-infected cells have been described previously. The assay used is based on the reduction of Sendai virus growth by IFN-α/β contained in β-propiolactone-inactivated cell culture supernatants as determined by immunocytochemistry (49). For transfection of cells with poly(IC) to induce IFN-α/β, Lipofectin (Gibco Life-Technologies AG, Basel, Switzerland) at 8 μg/ml was used according to the manufacturer's protocol. Briefly, Lipofectin was incubated at room temperature in serum- and antibiotic-free medium for 45 min before mixing with poly(IC). The Lipofectin-poly(IC) mixture was incubated another 15 min at room temperature before being added to the cells. After incubation for 2 h, the inoculum was replaced by fresh medium containing 2% FCS.
RT-PCR and Southern blot analysis.
Reverse transcription (RT)-PCR and Southern blot analysis were performed with slight modifications according to Sager et al. (58). Briefly, cells were harvested from 25 cm2 culture flasks, and total RNA was isolated with Trizol (Gibco Life Technologies) according to the manufacturer's protocol. RNA (10 μg) was adjusted to 0.1 μg/μl and treated with DNase I (4 U/μg of RNA) (Boehringer, Mannheim, Germany) in the presence of RNasin (8 U/μg of RNA) (Promega, Madison, Wis.) for 2 h at 37°C. After purification of the DNA-free RNA by phenol-chloroform extraction, RT was performed for 2 h at 42°C in the presence of oligo(dT) primers and 10 U of avian myeloblastosis virus reverse transcriptase (AMV-RT) (Promega) in a total volume of 100 μl. To exclude amplification from contaminating IFN-α/β DNA which escaped DNase treatment, a negative control without the addition of AMV-RT was included. Five microliters of the RT product was used in the following PCR. The primers used were specific for subfamilies of IFN (α, β, ω, and γ), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH)-specific primers were included as a loading control. A temperature profile of 1 min at 94°C, 1 min at 55°C, and 1 min at 72°C after “hot start” addition of Taq polymerase was used for 30 cycles. The sequences of the IFN-specific primers used for the PCR have been described (58).
Southern blotting was performed by transferring the amplified material from the agarose gel onto a positively charged nylon membrane (Boehringer Mannheim). The material was cross-linked by heat treatment at 80°C for 2 h. Prehybridization for 4 h and hybridization overnight with digoxigenin (DIG)-labeled RNA probes specific for IFN-α, -β, -ω, and -γ and GAPDH was performed at 50°C. The hybridized blot was washed several times in 2× SSC (1× SSC is 0.15 M NaCl plus 0.015 M sodium citrate, pH 7)–0.1% sodium dodecyl sulfate (SDS) and in 0.1× SSC–0.1% SDS at room temperature and at 68°C, according to the manufacturer's protocol (Boehringer Mannheim). Staining was done with anti-DIG-alkaline phosphatase conjugate (Boehringer Mannheim) and CDP-Star (Tropix, Bedford, Mass.) as substrate. A high-performance autoradiography film (Hyperfilm-MP; Amersham, Buckinghamshire, U.K.) was exposed for 2 to 30 s.
RESULTS
Cell death induced by dsRNA is inhibited by ncp BVDV.
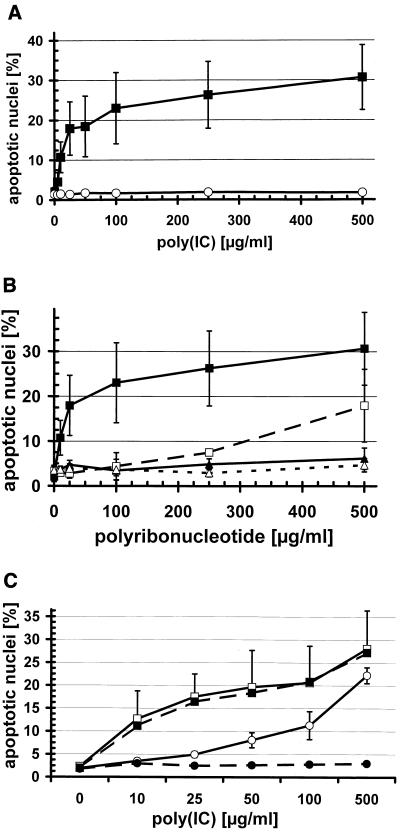
Addition of poly(IC), a synthetic dsRNA mimic, to BT cells resulted in cell death in a concentration-dependent manner, as shown by PI staining of the nuclei and FACS analysis (Fig. 1A). Accordingly, the double-stranded oligonucleotide poly(AU) also induced cell death at 10 to 500 μg/ml, albeit less efficiently than poly(IC) (not shown). By contrast, BT cells which had previously been infected with ncp BVDV were fully protected from cell death induced by poly(IC) (Fig. 1A) or poly(AU) (not shown), even at the highest concentration tested. The single-stranded oligonucleotides poly(I) and poly(C) did not induce cell death in either mock-infected (Fig. 1B) or ncp BVDV-infected BT cells (not shown). However, when poly(I) and the complementary poly(C) were added simultaneously to the cells, they induced cell death, albeit less efficiently than poly(IC) (Fig. 1B), which was again completely inhibited in ncp BVDV-infected cells (not shown). When poly(I) and poly(C) were incubated before addition to BT cells, they induced cell death as efficiently as poly(IC) (not shown), confirming that the dsRNA moiety is important for induction of apoptosis. By contrast, cell death induced by staurosporine, an inhibitor of protein kinase C and a widely used apoptosis inducer, or by actinomycin D, an inhibitor of cellular transcription (70), was not inhibited by infecting BT cells with ncp BVDV (not shown).
FIG. 1.
Ncp BVDV inhibits cell death induced by dsRNA in BT cells. BT cells in six-well plates were either mock infected or infected with the TGAN ncp BVDV strain at an MOI of 1 at 18 h prior to the addition of the oligonucleotides (A and B). DNA fragmentation was analyzed 48 h later by PI staining of nuclei followed by FACS analysis as described in the text. The percentages on the y axis indicate nuclei with subgenomic DNA. (A) Mock- (■) or TGAN-infected (○) BT cells were treated with poly(IC) at the indicated concentrations (mean ± standard deviation [SD], n = 4 to 13). (B) Mock-infected BT cells were treated with poly(IC) (■), poly(I) and poly(C) at equal concentrations (□), poly(I) (▴), or poly(C) (▵) at the indicated concentrations (mean ± SD, n = 2 to 6). (C) BT cells were mock- (■, □) or TGAN- (●, ○) infected 6 h (□, ○) and 12 h (■, ●) prior to the addition of poly(IC), and DNA fragmentation was analyzed 48 h later. For clarity, the mean ± SD (n = 2 to 3) is shown only for cells infected for 6 h. For mock-infected cells, the SD is similar for both time points, and for cells infected by TGAN for 12 h, the SD is smaller than the symbols.
Protein expression is necessary for BVDV to inhibit dsRNA-induced cell death.
To test whether the expression of viral proteins is necessary for protection from dsRNA-induced apoptosis, we infected BT cells with ncp BVDV at 0, 6, 12, or 24 h prior to the addition of poly(IC). Ncp BVDV did not protect cells from apoptosis when added simultaneously with poly(IC) (not shown), whereas infection of BT cells 6 and 12 h prior to poly(IC) addition partially or fully protected BT cells from poly(IC)-induced cell death, respectively (Fig. 1C). This clearly shows that expression of viral or cellular proteins induced by the virus is necessary for the inhibitory activity of ncp BVDV-infected BT cells.
Apoptosis-inhibiting activity is independent of the virus strain used.
Recent analysis identified two major BVDV genotypes, called BVDV-I and BVDV-II (48). Most outbreaks of severe disease after transient infections were associated with BVDV-II, but it is now evident that virulent ncp BVDV strains exist in both genotypes (9). We tested several strains of either BVDV-I or -II and avirulent or virulent ncp BVDV strains to analyze whether the ability to inhibit dsRNA-induced cell death differs between different genotypes or between strains of different virulence. All ncp BVDV strains tested, i.e., the avirulent TGAN, SuwaNCP, and SD-1 and the virulent R6229/95 and R5013/96 strains of genotype I and the 890 BVDV-II strain, completely inhibited cell death induced by poly(IC) at up to 500 μg/ml (not shown).
ROS are not involved in dsRNA-induced cell death.
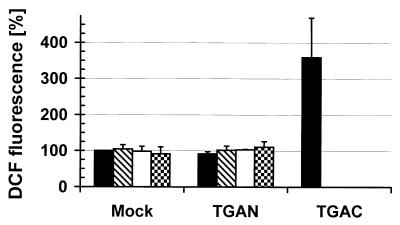
We have previously shown that intracellular oxidative stress is a crucial step in the induction of apoptosis induced by cp BVDV (59). To analyze whether cell death induced by both poly(IC) and cp BVDV requires the production of reactive oxygen species (ROS) and whether ncp BVDV would inhibit such an increase in ROS, we measured the intracellular redox state by quantitating the oxidation of dichlorofluorescin to the fluorescent DCF by FACS analysis (59). In contrast to the TGAC cp BVDV strain, poly(IC) at up to 500 μg/ml did not induce intracellular production of ROS in either mock- or ncp BVDV-infected BT cells (Fig. 2).
FIG. 2.
Poly(IC) does not induce the production of ROS in BT cells. BT cells were either mock infected or infected with the TGAN ncp BVDV strain and incubated for 18 h at 37°C. Thereafter, poly(IC) at 0, 50, 100, or 500 μg/ml (groups of bars from left to right) was added, or BT cells were infected with the TGAC cp BVDV strain (single solid bar). The intracellular redox state was measured 24 h later by quantitation of DCF fluorescence of viable, PI-negative cells by flow cytometry as described in the text. The value (geometric mean) for mock-infected cells in the absence of poly(IC) was set at 100% (mean ± SD, n = 2 to 3).
Inhibition of IFN synthesis.
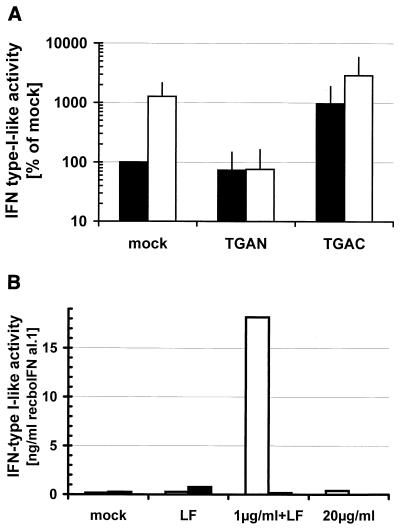
It has been known for a long time that dsRNA, e.g., poly(IC), stimulates the production of IFN-α/β (69). Therefore, we were interested in whether infection by ncp BVDV, besides protecting from cell death, also inhibits the production of IFN induced by dsRNA. As was previously shown (58), uninfected bovine monocyte-derived Mφ produced IFN-α/β in response to poly(IC) (Fig. 3A), as measured by the reduction in Sendai virus replication. By contrast, IFN synthesis induced by poly(IC) was completely inhibited in ncp BVDV-infected Mφ. The production of IFN-α/β in supernatants of cp BVDV-infected Mφ was enhanced by the addition of poly(IC) (Fig. 3A). To exclude the possibility that infection by ncp BVDV merely inhibits the uptake of poly(IC), we transfected poly(IC) into the cells using the liposome Lipofectin (47). Poly(IC) at 1 μg/ml in the presence of Lipofectin strongly induced the production of IFN-α/β from MDBK cells, which was completely inhibited in ncp BVDV-infected cells (Fig. 3B). Neither unstimulated MDBK cells nor the addition of Lipofectin alone or poly(IC) at 20 μg/ml in the absence of Lipofectin induced the production of IFN-α/β in ncp BVDV-infected or uninfected MDBK cells. This strongly suggests that proteins expressed inside ncp BVDV-infected cells were required to inhibit the effects of intracellular dsRNA.
FIG. 3.
Production of IFN-α/β activity is inhibited by ncp BVDV. (A) Bovine monocyte-derived Mφ were either mock infected or infected with the TGAN ncp or TGAC cp BVDV strain and incubated for 18 h at 37°C. Thereafter, the Mφ were incubated for 4 h in medium with (□) or without (■) poly(IC) (50 μg/ml), followed by 18 h in fresh medium in the absence of poly(IC). The IFN-α/β activity in the supernatants was determined as described in the text. The value for mock-infected cells in the absence of poly(IC) was set at 100% (mean ± SD, n = 3). (B) MDBK cells were either mock infected (□, left bars) or infected with the TGAN ncp BVDV strain (■, right bars) and incubated for 18 h at 37°C. Thereafter, poly(IC) (1 μg/ml) plus Lipofectin (LF) (8 μg/ml) or poly(IC) (20 μg/ml) in the absence of Lipofectin was added as described in the text. Medium (mock) or Lipofectin alone was added as a negative control. The IFN-α/β activity in the supernatants was determined after 24 h as described above and expressed as the standard (recboIFNαI.1), that corresponded to the biological activity measured for the sample.
Inhibition of IFN mRNA synthesis.
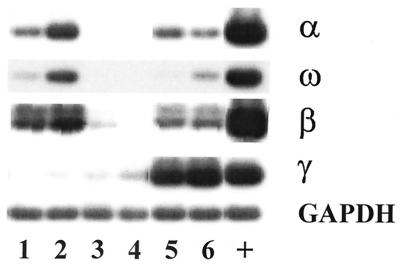
To test whether the inhibition of IFN synthesis occurs at the level of transcription, we analyzed the expression of IFN mRNA by RT-PCR followed by Southern blotting. The primers used were specific for the different bovine IFN families, i.e., IFN-α, -ω, and -β (IFN type I) or IFN-γ (IFN type II), as described in Materials and Methods. Mock-infected monocyte-derived Mφ showed low expression of mRNA for IFN-α, -ω, and -β, but not IFN-γ (Fig. 4, lane 1). The expression of all IFN-α/β but not IFN-γ mRNAs was enhanced by treating the Mφ with poly(IC) (Fig. 4, lane 2). By contrast, ncp BVDV-infected Mφ did not express or only barely expressed mRNA for either IFN-α/β or IFN-γ whether stimulated by poly(IC) or unstimulated (Fig. 4, lanes 3 and 4). Surprisingly, Mφ cultures infected with cp BVDV showed strong induction of IFN-γ mRNA (Fig. 4, lanes 5 and 6), even when the blood-derived monocytes were isolated from cows which were BVDV antibody negative. However, since we never detected IFN-γ mRNA in bone marrow-derived Mφ (3; M. Schweizer, unpublished data), we believe that a cell type different from Mφ, e.g., lymphocytes, present in the cultures may be responsible for the expression of IFN-γ mRNA. PCR products obtained without previous addition of RT showed no bands after hybridization with the specific probes, indicating that no genomic DNA was amplified (not shown).
FIG. 4.
Expression of IFN-α/β mRNA is inhibited by ncp BVDV. Monocyte-derived Mφ were mock infected (lanes 1 and 2) or infected by the TGAN ncp (lanes 3 and 4) or TGAC cp BVDV strain (lanes 5 and 6) 18 h prior to the addition of poly(IC) (50 μg/ml) (lanes 2, 4, and 6). After stimulation for 4 h, Mφ were incubated for 18 h in fresh medium in the absence of poly(IC). Total RNA was isolated, and IFN mRNA was analyzed by RT-PCR and Southern blotting as described in the text. Mφ, which were infected with bovine herpesvirus I were used as positive controls (+) for IFN-α/β (including IFN-ω), and Theileria parva-transformed cells served as positive controls for IFN-γ. GAPDH mRNA was included as a loading control.
Antiviral activity of IFN-α/β is not inhibited.
Since NS5A of hepatitis C virus (HCV) (another member of the Flaviviridae family) has been shown to inhibit the antiviral activity of IFN-α (24), we tested whether intact BVDV inhibits not only the induction but also the activity of IFN-α/β. When VSV was used as the challenge virus, recombinant bovine IFN-α I.1 at between 10 and 100 ng/ml completely inhibited virus replication in mock-infected BT cells and fully blocked the cytopathic effect (CPE) induced by VSV, as measured by FACS analysis. Infection of BT cells by ncp BVDV 18 h prior to the addition of IFN did not inhibit its antiviral activity (not shown). Thus, the replication of VSV and the CPE proceeded as in mock-infected cells, and IFN completely abolished these effects. This shows that ncp BVDV inhibits the induction of IFN-α/β by dsRNA, but it does not inhibit its antiviral activity when IFN is added exogenously.
DISCUSSION
dsRNA is formed during the multiplication of most viruses and is an important trigger for apoptosis and IFN synthesis (14, 30, 32, 35, 37, 69). This study shows that ncp BVDV inhibits apoptosis and IFN mRNA and protein synthesis induced by poly(IC), a synthetic form of dsRNA. This effect is specific to dsRNA, since (i) poly(AU)-induced cell death was also inhibited in ncp BVDV-infected cells, and (ii) cell death induced by staurosporine or by actinomycin D was not inhibited by infection with ncp BVDV. The latter is in contrast to poliovirus, a small positive-strand RNA virus of the genus Enterovirus in the Picornaviridae family, which was reported to inhibit actinomycin D- or cycloheximide-induced apoptosis in HeLa cells (67), or to NIH 3T3 cells expressing a carboxy-terminally truncated NS3 protein of HCV, which were more resistant to apoptosis induced by actinomycin D (22).
A number of reports suggest that triggering and execution of virus-induced apoptosis and of IFN synthesis may have common pathways. Specifically, IFN-α/β were shown to be essential mediators or to potentiate apoptotic cell death in virus-infected cells (6, 64). Evidence is accumulating that PKR, an enzyme activated by dsRNA and involved in the induction of IFN synthesis as well as the in the manifestation of its antiviral activity, may be equally important in triggering apoptosis in virally infected cells (25, 32, 63). Further strengthening the close correlation between the two key mechanisms of antiviral defense is the fact that IFN action and apoptosis are defective in mice devoid of 2′,5′-oligoadenylate-dependent RNase L (73). This enzyme is responsible for degrading single-stranded viral RNA, and its apoptosis-promoting effect may be explained by degradation of cellular mRNA (13, 36).
The observations reported in this paper indicate that ncp BVDV may be added to the growing list of viruses that interfere with apoptosis and IFN induction, two mechanisms that are fundamental to the host defense against viruses at the level of individual cells. Infection with Ebola virus was shown to suppress poly(IC)-induced synthesis of several proteins involved in antiviral defense, e.g., major histocompatibility complex I, 2′,5′-oligoadenylate synthetase, and PKR (28). More directly targeting dsRNA, the NS1 protein of influenza A virus (8, 38), the E3L protein of several poxviruses, and the ς3 protein of reovirus (57 and references therein) were demonstrated to bind to dsRNA and to inhibit the activation of PKR or RNase L.
The precise mechanisms by which BVDV interferes with apoptosis and IFN synthesis remain to be elucidated. The possibility that ncp BVDV may merely inhibit the uptake of poly(IC), e.g., by downregulating its receptor (71), was excluded by transfecting dsRNA using Lipofectin (Figure 3B). The inhibition of apoptosis and IFN production depended on the synthesis of BVDV proteins, a feature different from the particle-bound IFN-suppressing activity of VSV Indiana serotypes (40). The core protein of HCV, a flavivirus closely related to BVDV in its genome structure, has been reported to be immunosuppressive and to inhibit apoptosis, but the results are contradictory (19, 34, and references therein). The HCV E2 protein was reported to inhibit PKR by sequence homology to the PKR and eIF2α phosphorylation sites (65), and NS5A inhibits poly(IC)-induced apoptosis (24) and the antiviral activity of IFN by directly interacting with PKR (23, 33, 60). In the absence of suitable cell culture systems, it is, however, unknown whether these effects, observed by expressing individual viral proteins, are also operative during viral multiplication. Additionally, as suggested by experiments in which all viral proteins were jointly expressed, PKR-independent mechanisms of interference with apoptosis and IFN action may exist (5, 21). However, using intact virus, we showed that infection of BT cells by ncp BVDV prior to the addition of IFN did not inhibit its antiviral activity, i.e., exogenously added IFN-α inhibited the replication and cytopathogenicity of VSV independently of infection by BVDV.
It is noteworthy that the BVDV proteins homologous to those found to have antiapoptotic and anti-IFN properties in HCV are identical in both cp and ncp BVDV. This suggests that if E2 and NS5A are involved in the antiapoptotic and anti-IFN activities of BVDV, their roles would be rather complex. The two biotypes differ in only one nonstructural protein, which is present in ncp BVD V-infected cells in its uncleaved form, NS23, whereas this protein is cleaved into NS2 and NS3 in cells infected with the cp biotype of BVDV (11, 42). Since infectious progeny are formed to similar titers in a given pair of ncp and cp BVDV, the NTPase, RNA helicase, and protease activities of this nonstructural protein (26, 27) appear to be preserved in the cp biotype of BVDV. However, as noted previously, the amount of viral RNA present in cells infected by cp BVDV may vastly exceed that in cells infected with ncp biotypes of BVDV (41; L. Perler and H. P. Stalder, unpublished data). This suggests that cp BVDV may be relatively inefficient at packaging viral RNA compared to its ncp counterpart. Accordingly, RNA replicons of classical swine fever virus lacking the NS2 gene replicated more efficiently and induced CPE, in contrast to replicons expressing the complete NS23 gene (44). Additionally, the replication of flaviviruses, including BVDV, is known to involve replicative intermediates and replicative forms of virus RNA (53). Therefore, dsRNA can be expected to be produced during the replication of cp as well as ncp BVDV. It is tempting to speculate that a large amount of viral RNA may be double-stranded, which would have implications both for the cytopathic and interferon-inducing properties of cp BVDV. This would not necessarily ascribe the evasion by ncp BVDV of apoptosis and IFN induction solely to altered function of the viral helicase after cleavage of NS23 into NS2 and NS3. In fact, while the presence of NS3 (specifically its N-terminal part) clearly correlates with the cytopathic properties of BVDV, the cleavage of NS23 may have implications for the function of other proteins of the replication complex.
It is not only the viral mechanisms for inducing or preventing apoptosis that are different; the biochemical pathways of apoptosis may also vary in virally infected cells (57 and references therein). We have recently demonstrated evidence of oxidative stress in cells infected with cp BVDV. Supporting a causative role for this change, we were able to protect cells from apoptosis by treatment with selected antioxidants (59). The fact that we noted no evidence of oxidative stress in cells undergoing apoptosis in response to poly(IC) (Fig. 2) argues against a simple correlation between cell death induced by dsRNA and by cp BVDV. It does not, however, rule out a role of dsRNA in cp BVDV-induced apoptosis.
The experiments reported in this paper shed new light on observations made some 20 years ago when the capacity to suppress an effect of poly(IC) was used to detect ncp BVDV. Thus, cells were inoculated with serial dilutions of imput ncp BVDV, treated with poly(IC), and subsequently challenged with VSV. Noninfected cells were found to be protected against VSV-induced CPE, whereas ncp BVDV-infected cells were not protected (39). However, the authors suggested a non-IFN-based mechanism for the antiviral activity of poly(IC), since in their experiments, ncp BVDV did not interfere with the production of IFN by poly(IC) (55). The reason for this difference from the findings reported in this paper is not clear. The authors of that study observed that certain cell types were unsuitable for their assay of ncp BVDV (56). However, we used different cell types in our study, i.e., BT cells, MDBK cells, and Mφ, and a variety of different virus strains, which strongly suggests that interference with dsRNA-induced apoptosis and IFN synthesis by ncp BVDV may indeed be a general and important activity of this virus.
As shown by many examples, interference with apoptosis and the IFN response may contribute significantly to the success of viruses in the interaction with their hosts. In some instances, this has been correlated with virulence, i.e., the degree of viral pathogenicity. For example, increased induction of apoptosis has been correlated with attenuation of Sendai virus (29), and wild-type measles virus was shown to suppress the induction of IFN-α/β in phytohemagglutinin-stimulated peripheral blood lymphocytes, which was lost upon attenuation of the virus in cell culture (45). We observed that all ncp BVDV strains tested avoided apoptosis and IFN induction, i.e., virulent strains reported to induce hemorrhages in acutely infected animals as well as avirulent strains were active. This difference from other viruses may explain why transmission to the fetus is apparently correlated with the biotype (ncp versus cp) of BVDV rather than with the capacity to cause clinical signs in pregnant animals. Transmission at an early stage of pregnancy is essential for the establishment of immunological tolerance to BVDV (66). As apoptosis and the IFN response are manifested from the earliest stages of pregnancy (61), evasion of these two key elements of the innate immune system may be crucial not only for transmission to the fetus, but also for the maintenance of immunotolerance (7, 17, and references therein).
ACKNOWLEDGMENTS
This work was supported by the Swiss National Science Foundation, grant 31-50745.97.
We thank C. Dubey for help with the RT-PCR and Southern blot analysis, H. Pfister for performing the assays for IFN-α/β activity, and R. Parham for critically reading the manuscript.
REFERENCES
- 1.Adler B, Adler H, Pfister H, Jungi T W, Peterhans E. Macrophages infected with cytopathic bovine viral diarrhea virus release a factor(s) capable of priming uninfected macrophages for activation-induced apoptosis. J Virol. 1997;71:3255–3258. doi: 10.1128/jvi.71.4.3255-3258.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Adler H, Frech B, Meier P, Jungi T W, Peterhans E. Noncytopathic strains of bovine viral diarrhea virus prime bovine bone marrow-derived macrophages for enhanced generation of nitric oxide. Biochem Biophys Res Commun. 1994;202:1562–1568. doi: 10.1006/bbrc.1994.2109. [DOI] [PubMed] [Google Scholar]
- 3.Adler H, Peterhans E, Jungi T W. Generation and functional characterization of bovine bone marrow-derived macrophages. Vet Immunol Immunopathol. 1994;41:211–227. doi: 10.1016/0165-2427(94)90098-1. [DOI] [PubMed] [Google Scholar]
- 4.Alcami A, Koszinowski U H. Viral mechanisms of immune evasion. Immunol Today. 2000;21:447–455. doi: 10.1016/S0167-5699(00)01699-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Anderson S L, Carton J M, Lou J, Xing L, Rubin B Y. Interferon-induced guanylate binding protein-1 (GBP-1) mediates an antiviral effect against vesicular stomatitis virus and encephalomyocarditis virus. Virology. 1999;256:8–14. doi: 10.1006/viro.1999.9614. [DOI] [PubMed] [Google Scholar]
- 6.Balachandran S, Roberts P C, Kipperman T, Bhalla K N, Compans R W, Archer D R, Barber G N. Alpha/beta interferons potentiate virus-induced apoptosis through activation of the FADD/caspase-8 death signaling pathway. J Virol. 2000;74:1513–1523. doi: 10.1128/jvi.74.3.1513-1523.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bendelac A, Fearon D T. Innate pathways that control acquired immunity. Curr Opin Immunol. 1997;9:1–3. doi: 10.1016/s0952-7915(97)80151-3. [DOI] [PubMed] [Google Scholar]
- 8.Bergmann M, Garcia-Sastre A, Carnero E, Pehamberger H, Wolff K, Palese P, Muster T. Influenza virus NS1 protein counteracts PKR-mediated inhibition of replication. J Virol. 2000;74:6203–6206. doi: 10.1128/jvi.74.13.6203-6206.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bolin S R, Ridpath J F. The clinical significance of genetic variation among bovine viral diarrhea viruses. Vet Med. 1996;91:958–961. [Google Scholar]
- 10.Borrow P, Shaw G M. Cytotoxic T-lymphocyte escape viral variants: how important are they in viral evasion of immune clearance in vivo? Immunol Rev. 1998;164:37–51. doi: 10.1111/j.1600-065X.1998.tb01206.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brownlie J. Pathogenesis of mucosal disease and molecular aspects of bovine virus diarrhoea virus. Vet Microbiol. 1990;23:371–382. doi: 10.1016/0378-1135(90)90169-v. [DOI] [PubMed] [Google Scholar]
- 12.Bustamante J, Tovar A, Montero G, Boveris A. Early redox changes during rat thymocyte apoptosis. Arch Biochem Biophys. 1997;337:121–128. doi: 10.1006/abbi.1996.9754. [DOI] [PubMed] [Google Scholar]
- 13.Castelli J, Wood K A, Youle R J. The 2-5A system in viral infection and apoptosis. Biomed Pharmacother. 1998;52:386–390. doi: 10.1016/s0753-3322(99)80006-7. [DOI] [PubMed] [Google Scholar]
- 14.Castelli J C, Hassel B A, Wood K A, Li X L, Amemiya K, Dalakas M C, Torrence P F, Youle R J. A study of the interferon antiviral mechanism: apoptosis activation by the 2-5A system. J Exp Med. 1997;186:967–972. doi: 10.1084/jem.186.6.967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cebulla C M, Miller D M, Sedmak D D. Viral inhibition of interferon signal transduction. Intervirology. 1999;42:325–330. doi: 10.1159/000053968. [DOI] [PubMed] [Google Scholar]
- 16.Cossarizza A, Franceschi C, Monti D, Salvioli S, Bellesia E, Rivabene R, Biondo L, Rainaldi G, Tinari A, Malorni W. Protective effect of N-acetylcysteine in tumor necrosis factor-α-induced apoptosis in U937 cells: the role of mitochondria. Exp Cell Res. 1995;220:232–240. doi: 10.1006/excr.1995.1311. [DOI] [PubMed] [Google Scholar]
- 17.Cousens L P, Peterson R, Hsu S, Dorner A, Altman J D, Ahmed R, Biron C A. Two roads diverged: interferon alpha/beta- and interleukin 12-mediated pathways in promoting T cell interferon gamma responses during viral infection. J Exp Med. 1999;189:1315–1327. doi: 10.1084/jem.189.8.1315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cuff S, Ruby J. Evasion of apoptosis by DNA viruses. Immunol Cell Biol. 1996;74:527–537. doi: 10.1038/icb.1996.86. [DOI] [PubMed] [Google Scholar]
- 19.Dumoulin F L, Von Dem Bussche A, Söhne J, Sauerbruch T, Spengler U. Hepatitis C virus core protein does not inhibit apoptosis in human hepatoma cells. Eur J Clin Investig. 1999;29:940–946. doi: 10.1046/j.1365-2362.1999.00559.x. [DOI] [PubMed] [Google Scholar]
- 20.Estcourt M J, Ramshaw I A, Ramsay A J. Cytokine responses in virus infections: effects on pathogenesis, recovery and persistence. Curr Opin Microbiol. 1998;1:411–418. doi: 10.1016/s1369-5274(98)80058-1. [DOI] [PubMed] [Google Scholar]
- 21.François C, Duverlie G, Rebouillat D, Khorsi H, Castelain S, Blum H E, Gatignol A, Wychowski C, Moradpour D, Meurs E F. Expression of hepatitis C virus proteins interferes with the antiviral action of interferon independently of PKR-mediated control of protein synthesis. J Virol. 2000;74:5587–5596. doi: 10.1128/jvi.74.12.5587-5596.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fujita T, Ishido S, Muramatsu S, Itoh M, Hotta H. Suppression of actinomycin D-induced apoptosis by the NS3 protein of hepatitis C virus. Biochem Biophys Res Commun. 1996;229:825–831. doi: 10.1006/bbrc.1996.1887. [DOI] [PubMed] [Google Scholar]
- 23.Gale M, Korth M J, Tang N M, Tan S L, Hopkins D A, Dever T E, Polyak S J, Gretch D R, Katze M G. Evidence that hepatitis C virus resistance to interferon is mediated through repression of the PKR protein kinase by the nonstructural 5A protein. Virology. 1997;230:217–227. doi: 10.1006/viro.1997.8493. [DOI] [PubMed] [Google Scholar]
- 24.Gale M, Kwieciszewski B, Dossett M, Nakao H, Katze M G. Antiapoptotic and oncogenic potentials of hepatitis C virus are linked to interferon resistance by viral repression of the PKR protein kinase. J Virol. 1999;73:6506–6516. doi: 10.1128/jvi.73.8.6506-6516.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gil J, Esteban M. Induction of apoptosis by the dsRNA-dependent protein kinase (PKR): mechanism of action. Apoptosis. 2000;5:107–114. doi: 10.1023/a:1009664109241. [DOI] [PubMed] [Google Scholar]
- 26.Grassmann C W, Isken O, Behrens S-E. Assignment of the multifunctional NS3 protein of bovine viral diarrhea virus during RNA replication: an in vivo and in vitro study. J Virol. 1999;73:9196–9205. doi: 10.1128/jvi.73.11.9196-9205.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gu B H, Liu C B, Lin-Goerke J, Maley D R, Gutshall L L, Feltenberger C A, Del Vecchio A M. The RNA helicase and nucleotide triphosphatase activities of the bovine viral diarrhea virus NS3 protein are essential for viral replication. J Virol. 2000;74:1794–1800. doi: 10.1128/jvi.74.4.1794-1800.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Harcourt B H, Sanchez A, Offermann M K. Ebola virus inhibits induction of genes by double-stranded RNA in endothelial cells. Virology. 1998;252:179–188. doi: 10.1006/viro.1998.9446. [DOI] [PubMed] [Google Scholar]
- 29.Itoh M, Hotta H, Homma M. Increased induction of apoptosis by a Sendai virus mutant is associated with attenuation of mouse pathogenicity. J Virol. 1998;72:2927–2934. doi: 10.1128/jvi.72.4.2927-2934.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Janik B, de Clercq E, Sommer R G. Interferon inducing activity of (A)n · (U)n complexes of varying chain length. Biochim Biophys Acta. 1978;517:269–273. doi: 10.1016/0005-2787(78)90055-2. [DOI] [PubMed] [Google Scholar]
- 31.Jungi T W, Pfister H, Sager H, Fatzer R, Vandevelde M, Zurbriggen A. Comparison of inducible nitric oxide synthase expression in the brains of Listeria monocytogenes-infected cattle, sheep, and goats and in macrophages stimulated in vitro. Infect Immun. 1997;65:5279–5288. doi: 10.1128/iai.65.12.5279-5288.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kaufman R J. Double-stranded RNA-activated protein kinase mediates virus-induced apoptosis: a new role for an old actor. Proc Natl Acad Sci USA. 1999;96:11693–11695. doi: 10.1073/pnas.96.21.11693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Korth M J, Katze M G. Evading the interferon response: hepatitis C virus and the interferon-induced protein kinase PKR. In: Hagedorn C H, Rice C M, editors. The hepatitis C viruses. Berlin, Germany: Springer Verlag; 2000. pp. 197–224. [DOI] [PubMed] [Google Scholar]
- 34.Large M K, Kittlesen D J, Hahn Y S. Suppression of host immune response by the core protein of hepatitis C virus: possible implications for hepatitis C virus persistence. J Immunol. 1999;162:931–938. [PubMed] [Google Scholar]
- 35.Lee S B, Esteban M. The interferon-induced double-stranded RNA-activated protein kinase induces apoptosis. Virology. 1994;199:491–496. doi: 10.1006/viro.1994.1151. [DOI] [PubMed] [Google Scholar]
- 36.Li X L, Blackford J A, Hassel B A. RNase L mediates the antiviral effect of interferon through a selective reduction in viral RNA during encephalomyocarditis virus infection. J Virol. 1998;72:2752–2759. doi: 10.1128/jvi.72.4.2752-2759.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Long W F, Burke D C. Interferon production by double-stranded RNA: a comparison of induction by reovirus to that by a synthetic double-stranded polynucleotide. J Gen Virol. 1971;12:1–11. doi: 10.1099/0022-1317-12-1-1. [DOI] [PubMed] [Google Scholar]
- 38.Lu Y, Wambach M, Katze M G, Krug R M. Binding of the influenza virus NS1 protein to double-stranded RNA inhibits the activation of the protein kinase that phosphorylates the eIF-2 translation initiation factor. Virology. 1995;214:222–228. doi: 10.1006/viro.1995.9937. [DOI] [PubMed] [Google Scholar]
- 39.Maisonnave J, Rossi C R. A microtiter test for detecting and titrating noncytopathogenic bovine viral diarrhea virus. Arch Virol. 1982;72:279–287. doi: 10.1007/BF01315224. [DOI] [PubMed] [Google Scholar]
- 40.Marcus P I, Sekellick M J. Interferon induction by viruses. XV. Biological characteristics of interferon induction-suppressing particles of vesicular stomatitis virus. J Interferon Res. 1987;7:269–284. doi: 10.1089/jir.1987.7.269. [DOI] [PubMed] [Google Scholar]
- 41.Mendez E, Ruggli N, Collett M S, Rice C M. Infectious bovine viral diarrhea virus (strain NADL) RNA from stable cDNA clones: a cellular insert determines NS3 production and viral cytopathogenicity. J Virol. 1998;72:4737–4745. doi: 10.1128/jvi.72.6.4737-4745.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Meyers G, Thiel H-J. Molecular characterization of pestiviruses, Adv. Virus Res. 1996;47:53–118. doi: 10.1016/s0065-3527(08)60734-4. [DOI] [PubMed] [Google Scholar]
- 43.Moennig V, Plagemann P G W. The pestiviruses. Adv Virus Res. 1992;41:53–98. doi: 10.1016/s0065-3527(08)60035-4. [DOI] [PubMed] [Google Scholar]
- 44.Moser C, Stettler P, Tratschin J-D, Hofmann M A. Cytopathogenic and noncytopathogenic RNA replicons of classical swine fever virus. J Virol. 1999;73:7787–7794. doi: 10.1128/jvi.73.9.7787-7794.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Naniche D, Yeh A, Eto D, Manchester M, Friedman R M, Oldstone M B A. Evasion of host defenses by measles virus: wild-type measles virus infection interferes with induction of alpha/beta interferon production. J Virol. 2000;74:7478–7484. doi: 10.1128/jvi.74.16.7478-7484.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nettleton P F, Entrican G. Ruminant pestiviruses. Br Vet J. 1995;151:615–642. doi: 10.1016/S0007-1935(95)80145-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Okamoto S, Nakanishi K, Watanabe Y, Yamashita F, Takakura Y, Hashida M. Stimulation side-dependent asymmetrical secretion of poly I:poly C-induced interferon-beta from polarized epithelial cell lines. Biochem Biophys Res Commun. 1999;254:5–9. doi: 10.1006/bbrc.1998.9873. [DOI] [PubMed] [Google Scholar]
- 48.Pellerin C, Van den Hurk J, Lecomte J, Tijssen P. Identification of a new group of bovine viral diarrhea virus strains asssociated with severe outbreaks and high mortalities. Virology. 1994;203:260–268. doi: 10.1006/viro.1994.1483. [DOI] [PubMed] [Google Scholar]
- 49.Perler L, Pfister H, Schweizer M, Peterhans E, Jungi T W. A bioassay for interferon type I based on inhibition of Sendai virus growth. J Immunol Methods. 1999;222:189–196. doi: 10.1016/s0022-1759(98)00198-7. [DOI] [PubMed] [Google Scholar]
- 50.Perler L, Schweizer M, Jungi T W, Peterhans E. Bovine viral diarrhoea virus and bovine herpesvirus-1 prime uninfected macrophages for lipopolysaccharide-triggered apoptosis by interferon-dependent and -independent pathways. J Gen Virol. 2000;81:881–887. doi: 10.1099/0022-1317-81-4-881. [DOI] [PubMed] [Google Scholar]
- 51.Ploegh H L. Viral strategies of immune evasion. Science. 1998;280:248–253. doi: 10.1126/science.280.5361.248. [DOI] [PubMed] [Google Scholar]
- 52.Reed L J, Muench H. A simple method for estimating fifty percent endpoints. Am J Hyg. 1938;27:493–497. [Google Scholar]
- 53.Rice C M. Flaviviridae: the viruses and their replication. In: Fields B N, Knipe D M, Howley P M, editors. Fields virology. 3rd ed. Philadelphia, Pa: Lippincott-Raven Publishers; 1996. pp. 931–959. [Google Scholar]
- 54.Ridpath J F, Lewis T L, Bolin S R, Berry E S. Antigenic and genomic comparison between non-cytopathic and cytopathic bovine viral diarrhoea viruses isolated from cattle that had spontaneous mucosal disease. J Gen Virol. 1991;72:725–729. doi: 10.1099/0022-1317-72-3-725. [DOI] [PubMed] [Google Scholar]
- 55.Rossi C R, Kiesel G K. Factors affecting the production of bovine type I interferon on bovine embryonic lung cells by polyriboinosinic-polyribocytidylic acid. Am J Vet Res. 1980;41:557–560. [PubMed] [Google Scholar]
- 56.Rossi C R, Kiesel G K. Characteristics of the polyriboinosinic acid:polyribocytidylic acid assay for noncytopathogenic bovine viral diarrhea virus. Am J Vet Res. 1983;44:1916–1919. [PubMed] [Google Scholar]
- 57.Roulston A, Marcellus R C, Branton P E. Viruses and apoptosis. Annu Rev Microbiol. 1999;53:577–628. doi: 10.1146/annurev.micro.53.1.577. [DOI] [PubMed] [Google Scholar]
- 58.Sager H, Brunschwiler C, Jungi T W. Interferon production by Theileria annulata-transformed cell lines is restricted to the beta family. Parasite Immunol. 1998;20:175–182. doi: 10.1046/j.1365-3024.1998.00141.x. [DOI] [PubMed] [Google Scholar]
- 59.Schweizer M, Peterhans E. Oxidative stress in cells infected with bovine viral diarrhoea virus: a crucial step in the induction of apoptosis. J Gen Virol. 1999;80:1147–1155. doi: 10.1099/0022-1317-80-5-1147. [DOI] [PubMed] [Google Scholar]
- 60.Song J, Fujii M, Wang F, Itoh M, Hotta H. The NS5A protein of hepatitis C virus partially inhibits the antiviral activity of interferon. J Gen Virol. 1999;80:879–886. doi: 10.1099/0022-1317-80-4-879. [DOI] [PubMed] [Google Scholar]
- 61.Splichal I, Bonneau M, Charley B. Ontogeny of interferon alpha secreting cells in the porcine fetal hematopoietic organs. Immunol Lett. 1994;43:203–208. doi: 10.1016/0165-2478(94)90224-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Stark G R, Kerr I M, Williams B R G, Silverman R H, Schreiber R D. How cells respond to interferons. Annu Rev Biochem. 1998;67:227–264. doi: 10.1146/annurev.biochem.67.1.227. [DOI] [PubMed] [Google Scholar]
- 63.Tan S L, Katze M G. The emerging role of the interferon-induced PKR protein kinase as an apoptotic effector: a new face of death? J Interferon Cytokine Res. 1999;19:543–554. doi: 10.1089/107999099313677. [DOI] [PubMed] [Google Scholar]
- 64.Tanaka N, Sato M, Lamphier M S, Nozawa H, Oda E, Noguchi S, Schreiber R D, Tsujimoto Y, Taniguchi T. Type I interferons are essential mediators of apoptotic death in virally infected cells. Genes Cells. 1998;3:29–37. doi: 10.1046/j.1365-2443.1998.00164.x. [DOI] [PubMed] [Google Scholar]
- 65.Taylor D R, Shi S T, Romano P R, Barber G N, Lai M M C. Inhibition of the interferon-inducible protein kinase PKR by HCV E2 protein. Science. 1999;285:107–110. doi: 10.1126/science.285.5424.107. [DOI] [PubMed] [Google Scholar]
- 66.Thiel H-J, Plagemann P G W, Moennig V. Pestiviruses. In: Fields B N, Knipe D M, Howley P M, editors. Fields virology. 3rd ed. Philadelphia, Pa: Lippincott-Raven Publishers; 1996. pp. 1059–1073. [Google Scholar]
- 67.Tolskaya E A, Romanova L I, Kolesnikova M S, Ivannikova T A, Smirnova E A, Raikhlin N T, Agol V I. Apoptosis-inducing and apoptosis-preventing functions of poliovirus. J Virol. 1995;69:1181–1189. doi: 10.1128/jvi.69.2.1181-1189.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Tortorella D, Gewurz B E, Furman M H, Schust D J, Ploegh H L. Viral subversion of the immune system. Annu Rev Immunol. 2000;18:861–926. doi: 10.1146/annurev.immunol.18.1.861. [DOI] [PubMed] [Google Scholar]
- 69.Vilcek J, Ng M H, Friedman-Kien A E, Krawciw T. Induction of interferon synthesis by synthetic double-stranded polynucleotides. J Virol. 1968;2:648–650. doi: 10.1128/jvi.2.6.648-650.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wertz I E, Hanley M R. Diverse molecular provocation of programmed cell death. Trends Biochem Sci. 1996;21:359–364. [PubMed] [Google Scholar]
- 71.Yoshida I, Azuma M, Kawai H, Fisher H W, Suzutani T. Identification of a cell membrane receptor for interferon induction by poly rI:rC. Acta Virol. 1992;36:347–358. [PubMed] [Google Scholar]
- 72.Zhang G, Aldridge S, Clarke M C, McCauley J W. Cell death induced by cytopathic bovine viral diarrhoea virus is mediated by apoptosis. J Gen Virol. 1996;77:1677–1681. doi: 10.1099/0022-1317-77-8-1677. [DOI] [PubMed] [Google Scholar]
- 73.Zhou A M, Paranjape J, Brown T L, Nie H Q, Naik S, Dong B H, Chang A S, Trapp B, Fairchild R, Colmenares C, Silverman R H. Interferon action and apoptosis are defective in mice devoid of 2′,5′-oligoadenylate-dependent RNase L. EMBO J. 1997;16:6355–6363. doi: 10.1093/emboj/16.21.6355. [DOI] [PMC free article] [PubMed] [Google Scholar]