Abstract
Introduction
The long-acting insulin analogue insulin degludec (IDeg) is increasingly recommended for type two diabetes (T2DM), yet clinical experience in China remains limited. This retrospective study aimed to delineate the initiation strategy for IDeg in Chinese hospitalized patients with T2DM.
Methods
We retrospectively analyzed 217 Chinese hospitalized patients with T2DM who initiated IDeg from December 2018 to June 2020, calculating the initial dose and examining correlations between clinical characteristics and glucose profiles.
Results
The initial IDeg doses ranged from 0.15 to 0.18 IU/kg·d, showing no association with clinical characteristics. During titration, mean blood glucose levels (MEAN) correlated positively with diabetes duration, age, and Glycosylated Hemoglobin (HbA1c), and negatively with body mass index (BMI), triglycerides (TG), and low-density lipoprotein (LDL). The coefficient of variation (CV) in glucose levels correlated positively with HbA1c and negatively with BMI and TG. The mean amplitude of glycemic excursions (MAGE) mirrored these trends, with additional negative correlations to estimated glomerular filtration rate (eGFR) and serum albumin (ALB). Notably, glycemic variability parameters did not correlate with the presence of diabetic ketoacidosis (DKA) at admission. Hypoglycemia was observed in 21 patients, with differences in MEAN and CV during titration being the only significant findings.
Conclusion
The initial IDeg dosing was inadequate and not tailored to clinical features, and there were weak correlations between diabetes duration, age, BMI, eGFR, LDL, and ALB levels and glucose profile post-initiation.
Keywords: T2DM, insulin degludec, glucose variability, insulin initiation
Introduction
Basal insulin is integral to the management of type 2 diabetes mellitus (T2DM), with its long-acting analogs being crucial for achieving glycemic control. The development of second-generation insulin analogs, such as insulin degludec (IDeg) and insulin glargine-U300 (Gla-300), has been driven by the need for agents with smooth pharmacokinetic/pharmacodynamic (PK/PD) profiles, predictable efficacy, and flexible dosing schedules.1,2 These advances address limitations associated with first-generation analogs like Glargine-U100 (Gla-100) and insulin detemir, including a reduced risk of hypoglycemia.3
Despite these benefits, the optimization of basal insulin therapy in real-world settings is often limited by therapeutic inertia and suboptimal titration practices, potentially compromising glycemic control and patient safety.4 Many patients initiate or switch to IDeg during hospitalization in China. Due to inexperience and fear of hypoglycemia, most clinicians tend to be more conservative in the initial dose and titration of IDeg. Moreover, the extra-long half-life results in limited opportunities for dose adjustments during hospitalization. These factors make it challenging to optimize inpatient glycemic control. Our study addresses a significant gap in the literature regarding the initiation of IDeg in Chinese inpatients with T2DM. While existing research has extensively evaluated outpatient cohorts and long-term outcomes,5–7 our focus is distinct: 1) We concentrate on the inpatient setting, which is common in China for initiating basal insulin therapy; 2) We prioritize the initiation strategy including starting dose and subsequent titration; 3) We assess glycemic variability (GV) to refine dosing strategies and mitigate glucose fluctuations and hypoglycemic risks post-IDeg initiation.
This study provides a retrospective analysis of IDeg initiation in a single-center Chinese population, offering insights into dosing, titration, and glucose profiles. The results of this study are expected to inform clinical practice and potentially improve the management of hospitalized patients with T2DM in China and similar populations.
Methods
Study Design and Objectives
This retrospective, single-center study aimed to evaluate the initiation strategy of IDeg in hospitalized Chinese patients with T2DM. Ethical approval was obtained from the Ethics Committee of the First Affiliated Hospital of Xi’an Jiaotong University. As the study did not involve any direct patient contact or intervention that could potentially affect the course of treatment or care, the Ethics Committee granted a waiver for patient consent, emphasizing that the study poses no risk to participants and upholds the principles of the Declaration of Helsinki. All patient information was anonymized prior to analysis, ensuring that no personal identifiers were included in the dataset. This approach safeguards the privacy of the individuals involved and is in strict accordance with the ethical guidelines and standards for research as outlined by the Declaration of Helsinki.
Patients and Stratification
Between December 1, 2018, and June 30, 2020, 330 hospitalized patients with T2DM initiated IDeg at the First Affiliated Hospital of Xi’an Jiaotong University. Comprehensive data, including demographic characteristics, clinical features, laboratory parameters, and IDeg dosing regimens, were collected. After excluding 113 patients due to incomplete data, 217 patients were included in the final analysis. No significant differences were observed in the demographics and clinical features between the original and final patient cohorts (Table 1).
Table 1.
Clinical Characteristics of the Original and Final Patients Settings
| Original (N=330) | Final (N=217) | P | |
|---|---|---|---|
| Gender | 0.916 | ||
| Male | 228 (69.1%) | 149 (68.7%) | |
| Female | 102 (30.9%) | 68 (31.3%) | |
| Duration (months) | 96.00 (24.00, 168.00) | 96.00 (24.00, 168.00) | 0.684 |
| Age (years) | 55.00 (43.00, 65.00) | 56.00 (42.00, 64.00) | 0.817 |
| BMI (kg/m2) | 24.58±3.59 | 24.53±3.42 | 0.870 |
| HbA1c (%) | 10.25 (8.80, 11.83) | 10.30 (8.90, 12.00) | 0.711 |
| TG (mmol/L) | 1.54 (1.04,2.53) | 1.55 (1.08,2.70) | 0.575 |
| LDL (mmol/L) | 2.36 (1.77,3.07) | 2.36 (1.69,3.06) | 0.816 |
| HDL (mmol/L) | 0.88 (0.74,1.08) | 0.86 (0.74,1.04) | 0.454 |
| DPVD | 279 (84.5%) | 187 (86.2%) | 0.600 |
| DR | 77 (23.3%) | 56 (25.8%) | 0.509 |
| DN | 137 (41.5%) | 82 (37.8%) | 0.384 |
| CKD | 61 (18.5%) | 35 (16.1%) | 0.479 |
| DPN | 174 (52.7%) | 114 (52.5%) | 0.965 |
Abbreviations: TG, triglyceride; LDL, low-density lipoprotein; HDL, high-density lipoprotein; DPVN, Diabetic peripheral vascular disease; DR, diabetic retinopathy; DN, diabetic nephropathy; CKD, chronic kidney disease; DPN, diabetic peripheral neuropathy.
The patients were grouped based on the duration of T2DM (≤5 years, 5–10 years, 10–15 years, and >15 years), age (≤40 years old, 40–65 years old, and >65 years old), gender, body mass index (BMI) (≤18 kg/m2, 18–24 kg/m2, 24–28 kg/m2, and >28 kg/m2), glycosylated hemoglobin (HbA1c) (≤7%, 7–9%, and >9%), estimated glomerular filtration rate (eGFR) (eGFR≥90 and <90, ≥60 and <60, ≥30 and <30 mL/(min*1.73m2)), and serum albumin (ALB) level (ALB≥35 g/L and ALB<35 g/L, ALB≥30 g/L and ALB<30 g/L, ALB≥25 g/L and ALB<25 g/L, respectively).
Finger-Prick Blood Glucose Monitoring
Seven-point finger-prick blood glucose monitoring (pre- and 120 min post-meals and at bedtime) was conducted. Additionally, finger-prick blood glucose was measured at 3 AM in patients with suspected nocturnal hypoglycemia, defined as hypoglycemia occurring between 00:00 and 5:59. The glycemic variability indicators, including mean blood glucose level (MEAN), standard deviation of blood glucose (SDBG), coefficient of variation (CV), mean amplitude of glycemic excursions (MAGE), and time in range (TIR), were processed by Shanghai Medpower Tech, referring to the international consensus on continuous glucose monitoring of the American Diabetes Association.8
Statistical Analysis
Statistical analysis was performed using SPSS 22.0 software. Categorical data were presented using the number and proportion. Normally distributed data were expressed as the mean ± standard deviation 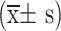 , and comparisons between two groups were conducted using the t-test, while comparisons between multiple groups were performed using one-way analysis of variance (one-way ANOVA). Non-normally distributed data were described using the median and inter-quartile range [M (Q1, Q3)], and non-parametric tests were utilized. Enumeration data were expressed as a percentage and analyzed using Pearson’s χ2 test. Pearson analysis was used for correlation analysis of normally distributed data, whereas Spearman analysis was employed for non-normally distributed data. A P-value of <0.05 was considered statistically significant.
, and comparisons between two groups were conducted using the t-test, while comparisons between multiple groups were performed using one-way analysis of variance (one-way ANOVA). Non-normally distributed data were described using the median and inter-quartile range [M (Q1, Q3)], and non-parametric tests were utilized. Enumeration data were expressed as a percentage and analyzed using Pearson’s χ2 test. Pearson analysis was used for correlation analysis of normally distributed data, whereas Spearman analysis was employed for non-normally distributed data. A P-value of <0.05 was considered statistically significant.
Results
Demographics and Clinical Characteristics of the Patients
As demonstrated in Table 1, for the final patient cohort of 217 patients, the median age was 56 years, with 68.7% being male. The median duration of T2DM was 96 months, and the median HbA1c was 10.30%, slightly higher than reported in previous studies for patients in rural and urban areas in China, which were 10.0% and 9.5%, respectively.9 Diabetic peripheral vascular disease was the most common chronic complication of T2DM (86.2%), followed by diabetic peripheral neuropathy (52.5%). Additionally, 37.8% and 16.1% of the participants had diabetic nephropathy and chronic kidney disease (CKD), respectively.
Initial Dose and Dose at Discharge of IDeg in Different Patient Groups
The patients were grouped by demographics (age and gender, respectively), clinical features (duration of T2DM, BMI, and diabetic ketoacidosis (DKA) status on admission, respectively), and laboratory parameters (HbA1c, eGFR, and serum ALB level, respectively). As depicted in Table 2, the initial daily doses of IDeg (IU/d) ranged from 8 to 14 IU/d, and were higher in patients with higher BMI (P=0.003), higher HbA1c (P=0.021) and significantly lower in patients whose eGFR was below 30% (P=0.026). However, after adjusted with body weight, the initial daily doses of IDeg (IU/kg·d) ranged from 0.15 to 0.18 IU/kg·d in most groups, except for patients with a BMI ≤18 kg/m2, for whom it was 0.21 IU/kg·d. No significant inter-group differences in initial doses were found when patients were grouped according to different clinical features. Similar situation was also observed in the doses at discharge. The doses at discharge ranged from 0.17 to 0.20 IU/kg·d without significant inter-group differences with the exception that female patients had a higher median dose than the male patients (0.20 vs 0.18 IU/kg·d, P=0.029).
Table 2.
Initial Dose of IDeg and Dose at Discharge in Different Groups
| Group | Initial Dose, (IU/d) | Initial Dose, (IU/kg·d) | Dose at Discharge (IU/d) | Dose at Discharge, (IU/kg·d) | |
|---|---|---|---|---|---|
| Duration (years) | ≤5 | 12.00 (10.00, 16.00) | 0.17 (0.14,0.21) | 13.50 (10.00, 16.75) | 0.18 (0.15,0.23) |
| 5–10 | 12.00 (10.00, 14.00) | 0.18 (0.15, 0.22) | 14.00 (10.00, 16.00) | 0.18 (0.14, 0.26) | |
| 10–15 | 10.00 (8.00, 14.00) | 0.15 (0.13, 0.19) | 13.50 (10.00, 16.75) | 0.19 (0.16, 0.25) | |
| >15 | 10.00 (10.00, 15.00) | 0.16 (0.13, 0.21) | 12.00 (8.00, 20.00) | 0.19 (0.13, 0.28) | |
| P | 0.319 | 0.476 | 0.881 | 0.773 | |
| Gender | Male | 12.00 (10.00, 15.00) | 0.17 (0.14, 0.20) | 14.00 (10.00, 17.00) | 0.18 (0.14, 0.23) |
| Female | 11.00 (8.00, 14.00) | 0.17 (0.13, 0.26) | 12.00 (10.00, 16.00) | 0.20 (0.16, 0.28) | |
| P | 0.085 | 0.179 | 0.476 | 0.029* | |
| Age (years) | ≤40 | 12.00 (10.00, 18.00) | 0.16 (0.14, 0.23) | 15.00 (11.00, 18.00) | 0.18 (0.16, 0.26) |
| 40–65 | 12.00 (10.00, 14.00) | 0.17 (0.13, 0.21) | 13.00 (10.00, 16.00) | 0.19 (0.14, 0.25) | |
| >65 | 10.00 (10.00, 14.00) | 0.18 (0.15, 0.22) | 12.00 (8.00, 15.50) | 0.20 (0.13, 0.28) | |
| P | 0.092 | 0.401 | 0.037* | 0.881 | |
| BMI (kg/m2) | ≤18 | 10.00 (7.00, 12.00) | 0.21 (0.14, 0.28) | 14.00 (7.00, 15.00) | 0.27 (0.16, 0.31) |
| 18–24 | 10.00 (10.00, 12.00) | 0.17 (0.14, 0.22) | 12.00 (10.00, 15.00) | 0.20 (0.15, 0.24) | |
| 24–28 | 12.00 (10.00, 16.00) | 0.17 (0.14, 0.21) | 14.00 (10.00, 18.00) | 0.18 (0.14, 0.25) | |
| >28 | 14.00 (10.00, 20.00) | 0.16 (0.10, 0.20) | 16.00 (12.00, 20.00) | 0.17 (0.13, 0.25) | |
| P | 0.003* | 0.397 | 0.013* | 0.305 | |
| DKA | Yes | 12.00 (10.00, 16.00) | 0.17 (0.13, 0.22) | 12.00 (10.00, 16.00) | 0.17 (0.15, 0.24) |
| No | 12.00 (10.00, 14.00) | 0.17 (0.14, 0.21) | 14.00 (10.00, 17.00) | 0.19 (0.14, 0.25) | |
| P | 0.317 | 0.666 | 0.798 | 0.666 | |
| HbA1c (%) | ≤7 | 12.00 (8.00, 18.00) | 0.18 (0.14, 0.24) | 12.00 (8.50, 19.00) | 0.18 (0.13, 0.26) |
| 7–9 | 10.00 (8.00, 12.00) | 0.15 (0.13, 0.19) | 11.50 (9.00, 15.00) | 0.17 (0.12, 0.24) | |
| >9 | 12.00 (10.00, 15.00) | 0.17 (0.14, 0.22) | 14.00 (10.00, 18.00) | 0.19 (0.15, 0.25) | |
| P | 0.021* | 0.058 | 0.031* | 0.151 | |
| eGFR mL/ (min*1.73m2) | ≥90 | 12.00 (10.00, 15.00) | 0.17 (0.14, 0.21) | 14.00 (10.00, 17.00) | 0.19 (0.15, 0.25) |
| <90 | 10.00 (8.00, 14.00) | 0.17 (0.12, 0.21) | 12.00 (8.00, 15.00) | 0.17 (0.13, 0.29) | |
| P | 0.066 | 0.399 | 0.098 | 0.375 | |
| ≥60 | 12.00 (10.00, 14.75) | 0.17 (0.14, 0.21) | 13.50 (10.00, 16.00) | 0.18 (0.14, 0.25) | |
| <60 | 10.00 (7.00, 17.00) | 0.15 (0.12, 0.24) | 13.00 (7.50, 23.00) | 0.19 (0.13, 0.33) | |
| P | 0.284 | 0.596 | 0.664 | 0.906 | |
| ≥30 | 12.00 (10.00, 15.00) | 0.17 (0.14, 0.21) | 13.00 (10.00, 17.00) | 0.19 (0.14, 0.25) | |
| <30 | 8.00 (5.50, 12.00) | 0.14 (0.08, 0.18) | 11.00 (5.50, 17.25) | 0.17 (0.11, 0.26) | |
| P | 0.026* | 0.074 | 0.326 | 0.628 | |
| ALB (g/L) | ≥35 | 12.00 (10.00, 14.00) | 0.17 (0.13, 0.21) | 14.00 (10.00, 16.00) | 0.18 (0.14, 0.24) |
| <35 | 12.00 (10.00, 15.00) | 0.18 (0.14, 0.27) | 12.00 (9.00, 22.00) | 0.19 (0.15, 0.29) | |
| P | 0.798 | 0.149 | 0.917 | 0.318 | |
| ≥30 | 12.00 (10.00, 15.00) | 0.17 (0.14, 0.21) | 14.00 (10.00, 16.00) | 0.19 (0.14, 0.25) | |
| <30 | 12.00 (10.00, 14.00) | 0.17 (0.15, 0.21) | 10.00 (8.00, 23.50) | 0.17 (0.14, 0.29) | |
| P | 0.979 | 0.680 | 0.459 | 0.909 | |
| ≥25 | 12.00 (10.00, 15.00) | 0.17 (0.14, 0.21) | 13.50 (10.00, 16.00) | 0.19 (0.14, 0.25) | |
| <25 | 12.00 (9.00, 13.00) | 0.17 (0.14, 0.18) | 12.00 (10.00, 23.00) | 0.17 (0.16, 0.31) | |
| P | 0.640 | 0.605 | 0.194 | 0.621 | |
Note: * P<0.05.
Abbreviations: IDeg, insulin degludec; Initial dose (IU/kg·d), initial dose of IDeg with adjustment for body weight; Dose at discharge (IU/kg·d), dose at discharge of IDeg with adjustment for body weight; BMI, body mass index; DKA, diabetic ketoacidosis; HbA1c, glycosylated haemoglobin; eGFR, estimated glomerular filtration rate; ALB, albumin.
Glucose Level and Variability After Initiation of IDeg
As an initiation strategy of IDeg for inpatients with T2DM, the focus is on the predictability of the glucose level and the risk of hypoglycemia after its application. Therefore, in this study, we focused on the MEAN and short-term blood glucose fluctuation parameters CV and MAGE during the titration period. The correlations between demographics and clinical characteristics and indicators for GV after initiating IDeg are summarized in Table 3.
Table 3.
Correlations Between Clinical Characteristics and Glucose Features
| Grouping | MEANT (mmol/L) | SDT (mmol/L) | CVT (%) | MAGET (mmol/L) | TIRT (%) | MEAND (mmol/L) | SDD (mmol/L) | CVD (%) | MAGED (mmol/L) | TIRD (%) | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Duration | r | 0.201 | 0.055 | −0.072 | 0.141 | −0.231 | 0.233 | 0.148 | 0.050 | 0.168 | −0.234 |
| P | 0.003* | 0.421 | 0.289 | 0.038* | 0.001* | 0.001* | 0.029* | 0.468 | 0.013* | 0.001* | |
| Gender | r | 0.058 | −0.027 | −0.088 | 0.014 | −0.081 | 0.162 | −0.009 | −0.073 | 0.039 | −0.093 |
| P | 0.392 | 0.688 | 0.197 | 0.833 | 0.232 | 0.017* | 0.898 | 0.283 | 0.572 | 0.174 | |
| Age | r | 0.228 | 0.119 | −0.026 | 0.219 | −0.241 | 0.247 | 0.166 | 0.082 | 0.177 | −0.208 |
| P | 0.001* | 0.081 | 0.705 | 0.001* | 0.000* | 0.000* | 0.014* | 0.231 | 0.009* | 0.002* | |
| BMI | r | −0.138 | −0.243 | −0.194 | −0.331 | 0.132 | −0.224 | −0.308 | −0.270 | −0.255 | 0.219 |
| P | 0.042* | 0.000* | 0.004* | 0.000* | 0.053 | 0.001* | 0.000* | 0.000* | 0.000* | 0.001* | |
| DKA | r | 0.025 | 0.117 | 0.100 | 0.020 | −0.025 | 0.009 | 0.088 | 0.111 | 0.050 | −0.034 |
| P | 0.716 | 0.085 | 0.141 | 0.775 | 0.718 | 0.890 | 0.197 | 0.104 | 0.464 | 0.622 | |
| HbA1c | r | 0.146 | 0.238 | 0.196 | 0.094 | −0.138 | 0.080 | 0.120 | 0.101 | 0.074 | −0.047 |
| P | 0.031* | 0.000* | 0.004* | 0.168 | 0.043* | 0.243 | 0.077 | 0.139 | 0.280 | 0.492 | |
| TG | r | −0.058 | −0.230 | −0.236 | −0.306 | 0.062 | −0.130 | −0.250 | −0.235 | −0.259 | 0.100 |
| P | 0.403 | 0.001* | 0.001* | 0.000* | 0.370 | 0.058 | 0.000* | 0.001* | 0.000* | 0.148 | |
| LDL | r | −0.147 | −0.196 | −0.124 | −0.194 | 0.153 | −0.147 | −0.229 | −0.208 | −0.163 | 0.192 |
| P | 0.032* | 0.004* | 0.070 | 0.005* | 0.025* | 0.032* | 0.001* | 0.002* | 0.017* | 0.005* | |
| HDL | r | −0.114 | −0.059 | 0.028 | 0.006 | 0.088 | 0.004 | −0.004 | −0.010 | −0.021 | 0.061 |
| P | 0.098 | 0.388 | 0.680 | 0.931 | 0.201 | 0.956 | 0.959 | 0.885 | 0.763 | 0.373 | |
| eGFR | r | −0.080 | −0.104 | −0.047 | −0.167 | 0.078 | −0.081 | −0.021 | 0.004 | −0.033 | 0.069 |
| P | 0.243 | 0.128 | 0.488 | 0.014* | 0.252 | 0.235 | 0.757 | 0.951 | 0.631 | 0.312 | |
| ALB | r | −0.098 | −0.113 | −0.054 | −0.151 | 0.088 | −0.149 | −0.144 | −0.096 | −0.167 | 0.121 |
| P | 0.154 | 0.099 | 0.431 | 0.027* | 0.199 | 0.029* | 0.035* | 0.162 | 0.014* | 0.076 |
Notes: T, titration period; D, at discharge; r: 0.00–0.19, very weak correlation; 0.20–0.39, weak correlation; 0.49–0.69, moderate correlated; 0.70–0.89, strong correlation; 0.90–1.00, very strong correlation. * P<0.05.
Abbreviations: BMI, body mass index; DKA, diabetic ketoacidosis; HbA1c, glycosylated haemoglobin; TG, triglyceride; LDL, low-density lipoprotein; HDL, high-density lipoprotein; eGFR, estimated glomerular filtration rate; ALB, albumin; MEAN, mean blood glucose levels; SD, standard deviation; CV, coefficient of variation; MAGE, mean amplitude of glycemic excursions; TIR, time in range.
The MEAN during the titration period showed positive correlations with the duration of T2DM, age, and HbA1c, and negative correlations with BMI and serum low-density lipoprotein (LDL) level. Correspondingly, TIR during the titration period exhibited negative correlations with the duration of T2DM, age, and HbA1c, and positive correlations with serum LDL level. Notably, CV during the titration period displayed positive correlations with the duration of T2DM, and negative correlations with BMI and serum triglycerides (TG) level. MAGE during the titration period displayed positive correlations with the duration of T2DM and age, and negative correlations with BMI, eGFR, and serum ALB level. Similarly, significant correlations were also found between MAGE during the titration period and serum TG and LDL level.
The MEAN at discharge was positively correlated with the duration of T2DM and age, and negatively correlated with BMI, serum ALB and LDL level. Correspondingly, the TIR at discharge was negatively correlated with the duration of T2DM and age, and positively correlated with serum LDL level. The CV at discharge was negatively correlated with BMI, serum TG and LDL levels. The MAGE at discharge was positively correlated with the duration of T2DM and age and was negatively correlated with BMI and serum ALB level. The MAGE at discharge was also negatively correlated with serum TG and LDL level.
Notably, these clinical and laboratory characteristics demonstrated weak correlations with blood glucose levels and variability despite statistical significance. To be noted, there were no correlations between glucose features (level and variability) and DKA status at admission either.
Hypoglycemia After Initiation of IDeg
In total, 21 patients experienced hypoglycemia, with 5 experiencing fasting hypoglycemia and 16 experiencing daytime hypoglycemia (Table 4). Comparing patients with and without hypoglycemia revealed no significant differences in demographics, laboratory parameters, or the prevalence of diabetic complications. There was no significant difference in total daily insulin dose (IU/kg·d) or daily dose of IDeg (IU/kg·d) at initiation for during titration period. The patients with hypoglycemia had a significant lower daily dose of IDeg at discharge (0.16 vs 0.19 IU/kg·d, P=0.038).
Table 4.
Clinical Characteristics of Patients with and without Hypoglycemia After Initiation of IDeg
| Patients without Hypoglycemia, N=196 | Patients with Hypoglycemia, N=21 | P | Patients with Fasting Hypoglycemia, N=5 | P | |
|---|---|---|---|---|---|
| Clinical and laboratory characteristics | |||||
| Gender | 0.482 | 0.986 | |||
| Male | 136 (69.40%) | 13 (61.90%) | 4 (80.0%) | ||
| Female | 60 (30.60%) | 8 (38.10%) | 1 (20.0%) | ||
| Age (years) | 53.00 (39.00, 61.50) | 56.00 (42.00, 64.00) | 0.567 | 52.00 (31.50, 66.00) | 0.455 |
| Duration (months) | 72.00 (16.00, 150.00) | 108.00 (24.00, 168.00) | 0.470 | 74.00 (4.25, 180.00) | 0.660 |
| BMI (kg/m2) | 25.63 (19.26, 27.29) | 24.70 (22.57, 26.70) | 0.713 | 22.14±4.40 | 0.118 |
| HbA1c (%) | 9.70 (8.45, 10.75) | 10.30 (8.93, 12.08) | 0.253 | 9.50 (8.70, 14.00) | 0.549 |
| TG (mmol/L) | 1.32 (0.95, 1.79) | 1.58 (1.12, 2.79) | 0.055 | 1.04 (0.88, 1.98) | 0.195 |
| LDL (mmol/L) | 2.62 (1.54, 3.28) | 2.36 (1.70, 3.04) | 0.547 | 2.56 (1.40, 3.57) | 0.886 |
| HDL (mmol/L) | 0.99 (0.85, 1.20) | 0.85 (0.73, 1.04) | 0.032 | 0.92 (0.81, 1.35) | 0.309 |
| ALB (g/L) | 39.20 (36.40, 42.33) | 39.00 (35.20, 41.40) | 0.606 | 38.40 (31.60, 44.90) | 0.601 |
| Chronic complications of T2DM | |||||
| DPVD | 169 (86.20%) | 18 (85.70%) | 1.000 | 3 (60.0%) | 0.152 |
| DR | 51 (26.00%) | 5 (23.80%) | 0.826 | 2 (40.0%) | 0.852 |
| DN | 72 (36.7%) | 10 (47.6%) | 0.328 | 3 (60.0%) | 0.553 |
| CKD staging | 0.245 | 0.062 | |||
| 1 | 166 (84.7%) | 16 (76.2%) | 3 (60.0%) | ||
| 2 | 19 (9.7%) | 3 (14.3%) | 1 (20.0%) | ||
| 3 | 6 (3.1%) | 1 (4.8%) | 0 (0.0%) | ||
| 4 | 1 (0.5%) | 1 (4.8%) | 1 (20.0%) | ||
| 5 | 4 (2.0%) | 0 (0.0%) | 0 (0.0%) | ||
| DPN | 103 (52.6%) | 11 (52.4%) | 0.988 | 4 (80.0%) | 0.447 |
| Insulin dose | |||||
| TDDT (IU/kg·d) | 0.40 (0.20, 0.49) | 0.41 (0.30, 0.51) | 0.194 | 0.47 (0.20, 0.77) | 0.360 |
| IDegT (IU/kg·d) | 0.17 (0.14, 0.21) | 0.17 (0.13, 0.24) | 0.889 | 0.23 (0.15, 0.36) | 0.171 |
| TDDD (IU/kg·d) | 0.41 (0.20, 0.55) | 0.31 (0.16, 0.47) | 0.148 | 0.26 (0.17, 0.47) | 0.346 |
| IDegD (IU/kg·d) | 0.19 (0.15, 0.25) | 0.16 (0.13, 0.18) | 0.038* | 0.14 (0.13, 0.18) | 0.133 |
| Glucose level and variability | |||||
| FBSA (mmol/L) | 10.40 (8.20, 13.00) | 10.10 (8.21, 12.35) | 0.872 | 13.50±0.71 | 0.123 |
| MEANA (mmol/L) | 12.27 (10.63, 14.82) | 12.39 (10.39, 15.47) | 0.943 | 15.05 (9.93, 16.86) | 0.618 |
| SDA (mmol/L) | 3.15 (2.26, 3.94) | 3.83 (3.08, 5.24) | 0.021* | 4.08 (2.94, 5.00) | 0.152 |
| CVA (%) | 25.00 (19.00, 33.00) | 31.00 (23.00, 38.00) | 0.011* | 30.00 (20.00, 44.00) | 0.290 |
| MAGEA (mmol/L) | 6.46 (4.61, 8.65) | 7.80 (6.35, 9.20) | 0.081 | 8.10 (6.72, 9.53) | 0.167 |
| TIRA (%) | 28.57 (0.00, 42.86) | 28.57 (8.33, 55.00) | 0.545 | 20.00 (0.00, 45.66) | 0.574 |
| MEANT (mmol/L) | 9.94 (8.97, 11.12) | 8.70 (8.15, 10.37) | 0.006* | 10.23 (6.41, 10.75) | 0.409 |
| SDT (mmol/L) | 2.74 (2.23, 3.47) | 2.64 (2.29, 4.35) | 0.226 | 3.83 (2.34, 4.52) | 0.204 |
| CVT (%) | 28.00±7.00 | 35.00±8.00 | 0.000* | 37.00 (35.00, 42.00) | 0.001* |
| MAGET (mmol/L) | 5.12 (4.12, 6.53) | 5.29 (3.94, 9.11) | 0.446 | 9.10 (4.19, 11.58) | 0.128 |
| TIRT (%) | 53.78±22.32 | 60.99±20.45 | 0.158 | 52.29±25.31 | 0.883 |
| FBSD (mmol/L) | 6.65 (5.75, 7.63) | 7.25 (6.00, 8.50) | 0.270 | 5.4±1.56 | 0.102 |
| MEAND (mmol/L) | 8.94 (7.89, 10.08) | 7.86 (7.02, 9.11) | 0.023* | 9.89 (6.36, 11.99) | 0.703 |
| SDD (mmol/L) | 2.04 (1.47, 2.73) | 2.12 (1.49, 2.74) | 0.904 | 2.99 (1.84, 4.87) | 0.091 |
| CVD (%) | 23.00 (18.00, 28.00) | 27.00 (19.00, 35.00) | 0.087 | 37.00 (27.00, 43.00) | 0.021* |
| MAGED (mmol/L) | 4.10 (3.11, 5.99) | 4.70 (3.40, 6.05) | 0.520 | 5.20 (4.45, 10.58) | 0.110 |
| TIRD (%) | 71.43 (50.00, 85.71) | 80.00 (69.05, 89.01) | 0.256 | 36.36 (30.95, 73.21) | 0.102 |
Notes: A- at admission; T -titration period, D- at discharge, * P<0.05.
Abbreviations: IDeg, insulin degludec; BMI, body mass index; HbA1c, glycosylated haemoglobin; TG, triglyceride; LDL, low-density lipoprotein; HDL, high-density lipoprotein; ALB, albumin; DPVD, diabetic peripheral vascular disease; DR, diabetic retinopathy; DN, diabetic nephropathy; CKD, chronic kidney disease; DPN, diabetic peripheral neuropathy; TDD, total daily dose; FBS, fasting blood sugar; MEAN, mean blood glucose levels; SD, standard deviation; CV, coefficient of variation; MAGE, mean amplitude of glycemic excursions; TIR, time in range.
The patients with hypoglycemia also exhibited higher SDBG (3.83 mmol/L vs 3.15 mmol/L, P=0.021) and CV (31.00% vs 25.00%, P=0.011) at admission than those without hypoglycemia, with a similar MEAN (12.39 mmol/L vs 12.27 mmol/L, P=0.943). They also presented with a significantly lower MEAN during the titration period (8.70 mmol/L vs 9.94 mmol/L, P=0.006) and at discharge (7.86 mmol/L vs 8.94 mmol/L, P=0.023), with a remarkably higher CV during the titration period (35.00% vs 28.00%, P=0.000). The TIRs of the two groups were not statistically significant on admission, during titration period or at discharge.
Moreover, the 5 patients experiencing fasting hypoglycemia showed significantly higher CV (P=0.001 and P=0.021 during titration period and at discharge, respectively) but no significant differences in other parameters presented. As shown in Table 5, among the 5 patients with fasting hypoglycemia, one (Patient 1) had comparatively lower HbA1c than the median HbA1c of the whole final patient setting (8.2% vs 10.3%), and two (Patient 4 and 5) had comparatively higher total daily dose (TDD) (0.83 and 0.71 IU/kg·d respectively). Patient 2 was diagnosed with hypopituitarism during hospitalization and was prescribed with hydrocortisone. Patient 3 was with CKD stage 4. Fingertip glucose at 3am was measured in patients with suspected nocturnal hypoglycemia (185 measurements in 69 patients), and no nocturnal hypoglycemia was detected.
Table 5.
Patients with Fasting Hypoglycemia After Initiation of IDeg
| Patient 1 | Patient 2 | Patient 3 | Patient 4 | Patient 5 | |
|---|---|---|---|---|---|
| Gender | Male | Female | Male | Male | Male |
| Duration (months) | 0.5 | 74 | 240 | 8 | 120 |
| Age (years) | 36 | 79 | 52 | 27 | 53 |
| BMI (kg/m2) | 26.12 | 20.05 | 17.26 | 19.82 | 27.43 |
| HbA1c (%) | 8.2 | 9.5 | 9.2 | 10.3 | 9.7 |
| TG (mmol/L) | 1.48 | 1.04 | 0.81 | 2.48 | 0.94 |
| LDL (mmol/L) | 2.56 | 3.27 | 1.29 | 3.87 | 1.50 |
| HDL (mmol/L) | 0.75 | 0.86 | 1.65 | 0.92 | 1.04 |
| DPVD | No | Yes | Yes | No | Yes |
| DR | No | No | Yes | No | Yes |
| DN | No | Yes | Yes | No | No |
| CKD | 1 | 2 | 4 | 1 | 1 |
| DPN | No | Yes | Yes | Yes | Yes |
| Therapy | IDeg+Metformin | IDeg | IDeg+Asp | IDeg+Asp | IDeg+Asp+Metformin |
| Initial dose | |||||
| TDD (IU/d) | 18 | 10 | 22 | 50 | 60 |
| TDD (IU/kg·d) | 0.23 | 0.17 | 0.47 | 0.83 | 0.71 |
| IDeg (IU/d) | 18 | 10 | 6 | 26 | 24 |
| IDeg (IU/kg·d) | 0.23 | 0.17 | 0.13 | 0.43 | 0.29 |
| Dose at discharge | |||||
| TDD (IU/d) | 16 | 8 | 22 | 28 | 22 |
| TDD (IU/kg·d) | 0.20 | 0.13 | 0.47 | 0.47 | 0.26 |
| IDeg (IU/d) | 16 | 8 | 6 | 10 | 12 |
| IDeg (IU/kg·d) | 0.20 | 0.13 | 0.13 | 0.17 | 0.14 |
| TIRA (%) | 61.90 | 0.00 | 29.4 | 20.00 | 0.00 |
| TIRT (%) | 78.90 | 31.90 | 37.50 | 32.30 | 80.90 |
| TIRD (%) | 75.00 | 28.60 | 33.30 | 36.40 | 71.40 |
Notes: A, on admission; T, titration period; D, at discharge.
Abbreviations: IDeg, insulin degludec; BMI, body mass index; HbA1c, glycosylated haemoglobin; TG, triglyceride; LDL, low-density lipoprotein; HDL, high-density lipoprotein; DPVD, diabetic peripheral vascular disease; DR, diabetic retinopathy; DN, diabetic nephropathy; CKD, chronic kidney disease; DPN, diabetic peripheral neuropathy; TDD, total daily dose; TIR, time in range. IDeg, insulin degludec; ASP, insulin aspart.
Discussion
In our retrospective analysis, three pivotal findings emerged: Initially, no statistically significant differences were observed in the initial dosage of IDeg across patients with varying clinical characteristics. Subsequently, during the titration phase, the CV and MAGE were correlated negatively with BMI and TG. Moreover, no notable discrepancies were found in clinical or laboratory parameters between patients who did and did not experience hypoglycemia. However, patients with hypoglycemia displayed a lower MEAN and a higher CV during the titration period.
Several studies have highlighted that the initial dose of second-generation long-acting insulin analogs is often inadequate, aligning with the findings of our retrospective study. The recommended initial dose of IDeg is 0.1–0.3 IU/kg·d, with higher dosages for patients with elevated HbA1c. However, our observations revealed that the initial doses of IDeg and the dosages at discharge for most patients were below 0.2 IU/kg·d, despite the comparatively higher HbA1c levels in our patient population, warranting a theoretically higher initial dose of IDeg. The conservative dosing adjustment observed in our study can be attributed to limited experience with IDeg. Insufficient titration has also been demonstrated hindering the optimized glycemic control. Studies such as the FINE-Asia study and the Achieve study have demonstrated inadequate dose adjustment six months after the initiation of basal insulin.10–12 Similar difficulties were noted in a real-world study of another second-generation basal insulin analog, Gla-U300.4 Given IDeg’s half-life of approximately 25 hours, dose adjustments at intervals of 3 days are recommended.13,14 In clinical practice, weekly adjustments are also proposed.5 However, the limited duration of hospitalization restricts the frequency of dose adjustments. The BEGIN study revealed that IDeg was well tolerated with adjustments ranging from −4 to +6 IU, based on pre-breakfast self-monitoring of blood glucose (SMBG) values.15 Nonetheless, due to concerns about IDeg accumulation and hypoglycemia, as well as inexperience in titrating doses in combination with other antihyperglycemic drugs, particularly novel ones including SGLT-2 (sodium-glucose co-transporter 2) inhibitors and GLP-1RA (glucagon-like peptide-1 receptor agonist), most uptitrations during hospitalization were performed in 2 IU increments and were insufficient. In our study, we noted that the IDeg dosage at discharge was minimally increased relative to the starting dose. Notably, female patients received a higher dose of IDeg adjusted for body weight compared to their male counterparts upon discharge. This clinical finding is potentially associated with the tendency for female patients to demand a longer hospital stay and more stringent glycemic management prior to discharge.
The focus on GV has gained significant attention in diabetes research. Current emphasis is placed on examining the correlation between glycemic fluctuation indicators, particularly TIR, and the development of chronic diabetes complications, as well as the connection to cardiovascular-related deaths and overall mortality rates.16–20 Another area of interest involves employing technologies such as Continuous Glucose Monitoring (CGM) to evaluate the effects of various treatment strategies on GV. There have been comprehensive reviews on the potential influence of IDeg on GV, and comparative studies on the impact of two second-generation long-acting insulin analogs, degludec and Gla-300, on glycemic fluctuations.21 Our research diverges from previous investigations by concentrating on the optimization of initial treatment plans, with a particular emphasis on short-term glycemic variability, primarily gauged by the CV and the MAGE, as a means to reflect the steadiness of its pharmacological influence and the reliability of its therapeutic outcomes. The importance of assessing GV was recognized in the early 1970s with the initial documentation of MAGE and other metrics.22 The SD is extensively utilized in the evaluation of GV due to its straightforward computation. MAGE measures the average extent of glucose oscillations that exceed the SD threshold, thereby accentuating the significance of substantial glycemic swings on overall GV.23 The CV, which normalizes the SD in relation to the MEAN, enables more coherent comparative assessments among diverse datasets. Within the Chinese demographic, a CV exceeding 33% is indicative of an elevated hypoglycemia risk.24 We observed positive correlations between MAGE and both the duration of diabetes and age, while negative correlations were found with BMI, TG, LDL, eGFR, and serum ALB. The positive correlations align with previous research indicating a substantial contribution of β-cell dysfunction to GV.25 The association of age with glucose levels and GV may be due to the extended T2DM in older patients. Interestingly, serum ALB levels and eGFR were inversely related to MAGE, potentially linked to the binding affinity of IDeg for ALB. The extent to which hypoalbuminemia accounts for this relationship requires further investigation. Given the suboptimal dosing in our study’s patient cohort, vigilance is advised regarding the risk of significant glucose fluctuations and hypoglycemia in individuals with pronounced hypoalbuminemia and compromised renal function. Unexpectedly, MAGE showed a negative correlation with BMI, TG, and LDL, which could be related to the shorter diabetes duration in this group and not yet on lipid-lowering therapies. The potential of LDL to predict glucose levels and variability during the dosage titration period should be explored further. Similarly, the CV was positively associated with HbA1c and inversely with BMI and TG. Notably, no correlation was found between glycemic variability parameters and the presence of diabetic ketosis at admission, indicating that dosage determination for IDeg initiation in hospitalized patients does not need to account for DKA at the time of admission.
Hypoglycemia, particularly nocturnal hypoglycemia, is a critical indicator for assessing the safety of basal insulin. Several studies have compared the risks of hypoglycemia of the novel long-acting insulin analogs Gla-U300 and IDeg, yielding inconclusive results. For instance, the BRIGHT study reported a significantly higher risk of hypoglycemia with IDeg during the first 12 weeks, while the CONFIRM study found that IDeg reduced the risk of hypoglycemia by 30% compared with Gla-U300.26,27 A Japanese randomized controlled study identified a higher incidence and longer duration of hypoglycemia in patients with serum ALB levels lower than 3.8 g/dL, possibly related to plasma albumin binding.28,29 In the present study, we observed no significant differences in clinical or laboratory parameters between patients with or without hypoglycemia. Those who experienced hypoglycemia demonstrated a lower MEAN and a higher CV during the titration period. Given our findings that the CV was inversely related to BMI and TG, patients with lower BMI and TG levels should be more closely monitored. Our study also emphasizes the importance of screening for other concomitant diseases in patients who develop fasting hypoglycemia at conservative doses of IDeg, such as adrenal and hypophyseal hypofunction. Nocturnal hypoglycemia was not observed in our study, which may be attributed to the significantly lower dosages of IDeg compared to the BRIGHT and CONFIRM studies, or possibly to the small sample size. Another possibility could be that although fingertip measurement at 3 AM has been widely used to detect nocturnal hypoglycemia since the DCCT study, the glucose nadir of IDeg might not occur at 3 AM.30 The nadir of blood glucose following the injection of second-generation long-acting insulin analogues requires further investigation. Additionally, the potential influence of other antihyperglycemic agents on the glucose profile of IDeg should be addressed in future studies. Our results also underscore the limitations of SMBG in presenting a comprehensive glucose profile. CGM is preferable for future studies.
The present investigation provides a novel perspective on the utilization of IDeg among Chinese inpatients with T2DM. It initially focuses on short-term GV as an indirect measure to evaluate the stability and predictability of IDeg. The research further examines the correlation between blood glucose profiles and clinical attributes during the dosage titration phase, thereby providing a foundation for the personalized refinement of treatment protocols. Additionally, the study discerns that the patients experiencing hypoglycemia demonstrated lower MEAN and elevated CV during the titration period, underscoring the necessity for heightened surveillance in this patient subset. However, it is essential to acknowledge the limitations inherent in our study’s retrospective, single-center design, which may limit the generalizability of our findings to more diverse populations. Additionally, the absence of comparative analysis with other long-acting insulin analogs presents an opportunity for future research to provide a more holistic assessment of therapeutic options in diabetes management.
Conclusion
The initial IDeg dosing was inadequate and not tailored to clinical features, and there were weak correlations between diabetes duration, age, BMI, eGFR, LDL, and ALB levels and glucose profile post-initiation.
Acknowledgments
The authors thank Luan Zhou and Jiao Zhou for the technical help.
Funding Statement
This work was supported by Key Research and Development Program of Shaanxi (Grant No. 2023-ZDLSF-40 and 2021LL-JB-06). No funding body participated in the design of the study and the collection, analysis, and interpretation of data and the writing the manuscript.
Abbreviations
ALB, Albumin; BMI, Body Mass Index; CGM, Continuous Glucose Monitoring; CKD, Chronic Kidney Disease; CV, Coefficient of Variation; DKA, Diabetic Ketoacidosis; eGFR, Estimated Glomerular Filtration Rate; GV, Glucose Variability; HbA1c, Glycosylated Hemoglobin; IDeg, Insulin Degludec; LDL, Low-Density Lipoprotein-Cholesterol; MAGE, Mean Amplitude of Glycemic Excursions; PK/PD, Pharmacokinetics/Pharmacodynamics; SDBG, Standard Deviation of Blood Glucose; SMBG, Self-monitoring of Blood glucoseT2DM, Type 2 Diabetes Mellitus; TG, Triglyceride; TIR, Time in Range.
Data Sharing Statement
All data generated or analyzed during this study are included in this article. Further inquiries can be directed to the corresponding author.
Ethics Approval
Ethical approval was obtained from the Ethics Committee of the First Affiliated Hospital of Xi’an Jiaotong University (No. XJTU1AF2021LSK-182). As the study did not involve any direct patient contact or intervention that could potentially affect the course of treatment or care, the Ethics Committee granted a waiver for patient consent, emphasizing that the study poses no risk to participants and upholds the principles of the Declaration of Helsinki. All patient information was anonymized prior to analysis, ensuring that no personal identifiers were included in the dataset. This approach safeguards the privacy of the individuals involved and is in strict accordance with the ethical guidelines and standards for research as outlined by the Declaration of Helsinki.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors have no conflicts of interest to declare.
References
- 1.Garber AJ. Will the next generation of basal insulins offer clinical advantages?. Diabetes Obes Metab. 2014;16(6):483–491. doi: 10.1111/dom.12219 [DOI] [PubMed] [Google Scholar]
- 2.American Diabetes Association. Pharmacologic Approaches to Glycemic Treatment: standards of Medical Care in Diabetes-2021. Diabetes Care. 2021;44(Suppl 1):S111–S124. doi: 10.2337/dc21-S009 [DOI] [PubMed] [Google Scholar]
- 3.Simon ACR, DeVries JH. The future of basal insulin supplementation. Diabetes Technol Ther. 2011;13 Suppl 1(S1):S103–108. doi: 10.1089/dia.2010.0251 [DOI] [PubMed] [Google Scholar]
- 4.Ragonese M, Larosa M, Angotti S, et al. Clinical Outcomes of Switching to Insulin Glargine 300 U/mL from Other Basal Insulins in People with Type 2 Diabetes in Italy: a Real-World Study. Diabetes Ther. 2020;11(10):2283–2298. doi: 10.1007/s13300-020-00902-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Woo V, Berard L, Roscoe R. Understanding the Clinical Profile of Insulin Degludec, the Latest Basal Insulin Approved for Use in Canada: a Narrative Review. Diabetes Ther. 2020;11(11):2539–2553. doi: 10.1007/s13300-020-00915-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Woo VC. A Review of the Clinical Efficacy and Safety of Insulin Degludec and Glargine 300 U/mL in the Treatment of Diabetes Mellitus. Clin Ther. 2017;39(8S2):S12–S33. doi: 10.1016/j.clinthera.2017.01.007 [DOI] [PubMed] [Google Scholar]
- 7.Dong Z-Y, Feng J-H, Zhang J-F. Efficacy and Tolerability of Insulin Degludec Versus Other Long-acting Basal Insulin Analogues in the Treatment of Type 1 and Type 2 Diabetes Mellitus: a Systematic Review and Meta-analysis. Clin Ther. 2022;44(11):1520–1533. doi: 10.1016/j.clinthera.2022.09.012 [DOI] [PubMed] [Google Scholar]
- 8.Danne T, Nimri R, Battelino T, et al. International Consensus on Use of Continuous Glucose Monitoring. Diabetes Care. 2017;40(12):1631–1640. doi: 10.2337/dc17-1600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ji L, Zhang P, Weng J, et al. Observational Registry of Basal Insulin Treatment (ORBIT) in Patients with Type 2 Diabetes Uncontrolled by Oral Hypoglycemic Agents in China--Study Design and Baseline Characteristics. Diabetes Technol Ther. 2015;17(10):735–744. doi: 10.1089/dia.2015.0054 [DOI] [PubMed] [Google Scholar]
- 10.Tsai S-T, Pathan F, Ji L, et al. First insulinization with basal insulin in patients with Type 2 diabetes in a real-world setting in Asia. J Diabetes. 2011;3(3):208–216. doi: 10.1111/j.1753-0407.2011.00137.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ji L, Tsai S-T, Lin J, et al. National Variations in Comorbidities, Glycosylated Hemoglobin Reduction, and Insulin Dosage in Asian Patients with Type 2 Diabetes: the FINE-Asia Registry. Diabetes Ther. 2015;6(4):519–530. doi: 10.1007/s13300-015-0137-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Home P, Naggar NE, Khamseh M, et al. An observational non-interventional study of people with diabetes beginning or changed to insulin analogue therapy in non-Western countries: the A1chieve study. Diabet Res Clin Pract. 2011;94(3):352–363. doi: 10.1016/j.diabres.2011.10.021 [DOI] [PubMed] [Google Scholar]
- 13.Heise T, Meneghini LF. Insulin stacking versus therapeutic accumulation: understanding the differences. Endocr Pract. 2014;20(1):75–83. doi: 10.4158/EP13090.RA [DOI] [PubMed] [Google Scholar]
- 14.Examples of Insulin Initiation and Titration Regimens in People With Type 2 Diabetes. Canadian Diabetes Association. 2018;42:S317–S318. doi: 10.1016/j.jcjd.2017.11.002 [DOI] [Google Scholar]
- 15.Philis-Tsimikas A, Brod M, Niemeyer M, et al. Insulin degludec once-daily in type 2 diabetes: simple or step-wise titration (BEGIN: once simple use). Adv Ther. 2013;30(6):607–622. doi: 10.1007/s12325-013-0036-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhao W, Lu J, Zhang L, et al. Relationship between time in range and corneal nerve fiber loss in asymptomatic patients with type 2 diabetes. Chin Med J. 2022;135(16):1978–1985. doi: 10.1097/CM9.0000000000002140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang Y, Lu J, Shen Y, et al. Association of time in range with lower extremity atherosclerotic disease in type 2 diabetes mellitus: a prospective cohort study. Endocrine. 2022;76(3):593–600. doi: 10.1007/s12020-022-03038-3 [DOI] [PubMed] [Google Scholar]
- 18.Lu J, Ma X, Shen Y, et al. Time in Range Is Associated with Carotid Intima-Media Thickness in Type 2 Diabetes. Diabetes Technol Ther. 2020;22(2):72–78. doi: 10.1089/dia.2019.0251 [DOI] [PubMed] [Google Scholar]
- 19.Li F, Zhang Y, Li H, et al. TIR generated by continuous glucose monitoring is associated with peripheral nerve function in type 2 diabetes. Diabet Res Clin Pract. 2020;166:108289. doi: 10.1016/j.diabres.2020.108289 [DOI] [PubMed] [Google Scholar]
- 20.Lu J, Wang C, Shen Y, et al. Time in Range in Relation to All-Cause and Cardiovascular Mortality in Patients With Type 2 Diabetes: a Prospective Cohort Study. Diabetes Care. 2021;44(2):549–555. doi: 10.2337/dc20-1862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yang Y, Long C, Li T, et al. Insulin Degludec Versus Insulin Glargine on Glycemic Variability in Diabetic Patients: a Systematic Review and Meta-Analysis of Randomized Controlled Trials. Front Endocrinol. 2022;13:890090. doi: 10.3389/fendo.2022.890090 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rodbard D. The Challenges of Measuring Glycemic Variability. J Diabetes Sci Technol. 2012;6(3):712–715. doi: 10.1177/193229681200600328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Service FJ, Molnar GD, Rosevear JW, et al. Mean Amplitude of Glycemic Excursions, a Measure of Diabetic Instability. Diabetes. 1970;19(9):644–655. doi: 10.2337/diab.19.9.644 [DOI] [PubMed] [Google Scholar]
- 24.Wu J, Lu AD, Zhang LP, et al. Study of clinical outcome and prognosis in pediatric core binding factor-acute myeloid leukemia. Zhonghua Xue Ye Xue Za Zhi. 2019;40(1):52–57. doi: 10.3760/cma.j.issn.0253-2727.2019.01.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kohnert K-D, Augstein P, Zander E, et al. Glycemic variability correlates strongly with postprandial beta-cell dysfunction in a segment of type 2 diabetic patients using oral hypoglycemic agents. Diabetes Care. 2009;32(6):1058–1062. doi: 10.2337/dc08-1956 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rosenstock J, Cheng A, Ritzel R, et al. More Similarities Than Differences Testing Insulin Glargine 300 Units/mL Versus Insulin Degludec 100 Units/mL in Insulin-Naive Type 2 Diabetes: the Randomized Head-to-Head BRIGHT Trial. Diabetes Care. 2018;41(10):2147–2154. doi: 10.2337/dc18-0559 [DOI] [PubMed] [Google Scholar]
- 27.Tibaldi J, Hadley-Brown M, Liebl A, et al. A comparative effectiveness study of degludec and insulin glargine 300 U/mL in insulin-naïve patients with type 2 diabetes. Diabetes Obes Metab. 2019;21(4):1001–1009. doi: 10.1111/dom.13616 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kawaguchi Y, Sawa J, Sakuma N, et al. Efficacy and safety of insulin glargine 300 U/mL vs insulin degludec in patients with type 2 diabetes: a randomized, open-label, cross-over study using continuous glucose monitoring profiles. J Diabetes Investig. 2019;10(2):343–351. doi: 10.1111/jdi.12884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kawaguchi Y, Sawa J, Hamai C, et al. Differential Effect of Hypoalbuminemia on Hypoglycemia on Type 2 Diabetes Patients Treated with Insulin Glargine 300 U/mL and Insulin Degludec. Diabetes Ther. 2019;10(4):1535–1541. doi: 10.1007/s13300-019-0654-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Škrha J, Šoupal J, Škrha J, et al. Glucose variability, HbA1c and microvascular complications. Rev Endocr Metab Disord. 2016;17(1):103–110. doi: 10.1007/s11154-016-9347-2 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this article. Further inquiries can be directed to the corresponding author.


