Abstract
Primary congenital glaucoma (PCG) occurs in children due to developmental abnormalities in the trabecular meshwork and anterior chamber angle. Previous studies have implicated rare variants in CYP1B1, LTBP2, and TEK and their interactions with MYOC, FOXC1, and PRSS56 in the genetic complexity and clinical heterogeneity of PCG. Given that some of the gene-encoded proteins are localized in the centrosomes (MYOC) and perform ciliary functions (TEK), we explored the involvement of a core centrosomal protein, CEP164, which is responsible for ocular development and regulation of intraocular pressure. Deep sequencing of CEP164 in a PCG cohort devoid of homozygous mutations in candidate genes (n = 298) and controls (n = 1757) revealed CEP164 rare pathogenic variants in 16 cases (5.36%). Co-occurrences of heterozygous alleles of CEP164 with other genes were seen in four cases (1.34%), and a physical interaction was noted for CEP164 and CYP1B1 in HEK293 cells. Cases of co-harboring alleles of the CEP164 and other genes had a poor prognosis compared with those with a single copy of the CEP164 allele. We also screened INPP5E, which synergistically interacts with CEP164, and observed a lower frequency of pathogenic variants (0.67%). Our data suggest the potential involvements of CEP164 and INPP5E and the yet unexplored cilia-centrosomal functions in PCG pathogenesis.
Keywords: primary congenital glaucoma, anterior segment, intraocular pressure, centrosome, cilia, gene, CEP164, INPP5E, CYP1B1
1. Introduction
Primary congenital glaucoma (PCG) is a rare autosomal recessive disease in children that occurs due to developmental defects in the trabecular meshwork (TM) and anterior chamber angle with a corresponding rise in intraocular pressure (IOP), optic nerve damage, and loss of vision [1,2]. PCG is clinically and genetically heterogeneous, and its molecular etiology is poorly understood [3]. Primarily, pathogenic variants in cytochrome P450 family 1 subfamily B member 1 (CYP1B1) [4,5,6], latent transforming growth factor beta binding protein 2 (LTBP2) [7,8], and TEK receptor tyrosine kinase (TEK) [9,10] have been implicated in PCG, but collectively they do not explain the entire molecular bases of this disease [3,11]. Additionally, variants in other genes in the anterior segment, cell signaling, extracellular matrix, and so on have been identified through GWAS (genome-wide association study) and deep sequencing approaches, but their specific role(s) in PCG pathogenesis remains elusive [3,12]. Variants in other genes comprising myocilin (MYOC) [13], forkhead box C1 (FOXC1) [14], angiopoietin 1 (ANGPT1) [15], optineurin (OPTN) [16], thrombospondin 1 (THBS1) [17], guanylate cyclase activator 1C (GUCA1C) [18], procollagen-lysine,2-oxoglutarate 5-dioxygenase 2 (PLOD2) [19], collagen type I alpha 1 chain (COL1A1) [20], forkhead box C2 (FOXC2), and paired like homeodomain 2 (PITX2) [21], neurotrophin 4 (NTF4) [22], C3, and PZP like alpha-2-macroglobulin domain containing 8 (CPAMD8) [23], sushi, von Willebrand factor type A, EGF, and pentraxin domain containing 1 (SVEP1) [24] and serine protease 56 (PRSS56) [25] are reported to be infrequent causes of PCG.
Centrosomal and ciliary genes play critical roles in various developmental anomalies in the anterior segment of the eye [26,27,28]. A juvenile open-angle glaucoma (JOAG)-associated gene, MYOC, localizes in the centrosome and the cytoplasmic filaments of TM cells and, to a lesser degree, in the trabecular beams and extracellular matrices in the juxta canalicular region of the TM [29,30]. It also co-localizes with cytochrome c oxidase subunit II of the mitochondria in the TM cells [29]. MYOC is also involved in PCG through its digenic interactions with CYP1B1 [13].
The TM of the eye harbors primary cilia that help in regulating IOP by mechanosensation [31]. Elevated IOP causes the shortening of the cilia along with expressions of pro-inflammatory cytokines [31,32]. Thus, ciliary dysfunction could be an important contributor to the pathophysiology of glaucoma [33,34]. Earlier, we demonstrated the potential role of a ciliary gene TEK in PCG pathogenesis based on its genetic and physical interactions with CYP1B1 [10]. TEK principally localizes to the primary cilia of the surface epithelium of the ovary, bursa, and extra-ovarian rete ducts and to the plasma membranes of ovarian theca and endothelial cells [35]. It has also been localized to the caveolae enriched with various signaling molecules [36] and in ANGPT1 (Angiopoietin 1)-mediated cell–cell junctions in human umbilical vein endothelial cells (HUVECs) [9,10].
We explored the potential involvement of a centrosomal gene (CEP164), which is required for microtubule organization along with maintenance of the primary cilia and genomic stability [37,38] in PCG. Additionally, the Cep164-/- mice have been found to have a lack of connection between cilia and outer segments of the photoreceptors [33]. CEP164 interacts with INPP5E (Inositol Polyphosphate 5-Phosphatase E), which localizes in the cilia and helps in the stability and maintenance of the ciliary structures [39,40,41]. Dysfunction of INPP5E leads to shortened cilia and impaired ciliary function [42].
Mutations in CEP164 and INPP5E have been implicated in retinal ciliopathies, and their functional interactions have already been deciphered [43,44,45,46,47]. Additionally, CEP164 is involved in the regulation of epithelial to mesenchymal transition [48] and INPP5E in embryonic neural development [49]. Based on their potential roles in the centrosome and cilia, along with their implications in ocular development and regulation of IOP, we aimed to understand the involvement of CEP164 and INPP5E in the PCG pathogenesis.
2. Results
2.1. Identification of Rare and Common Variants in CEP164 and INPP5E
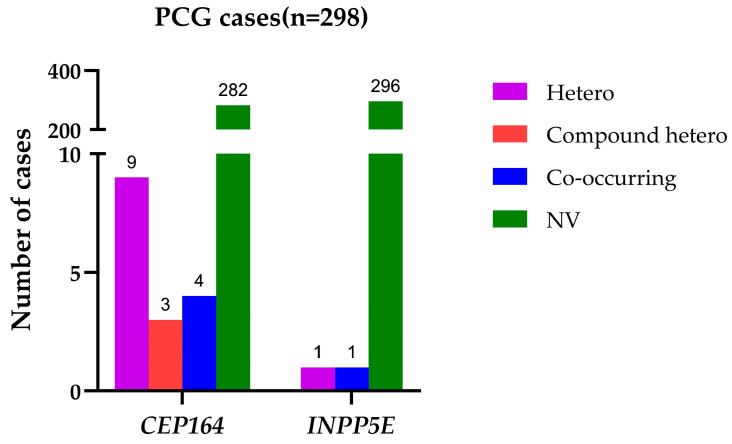
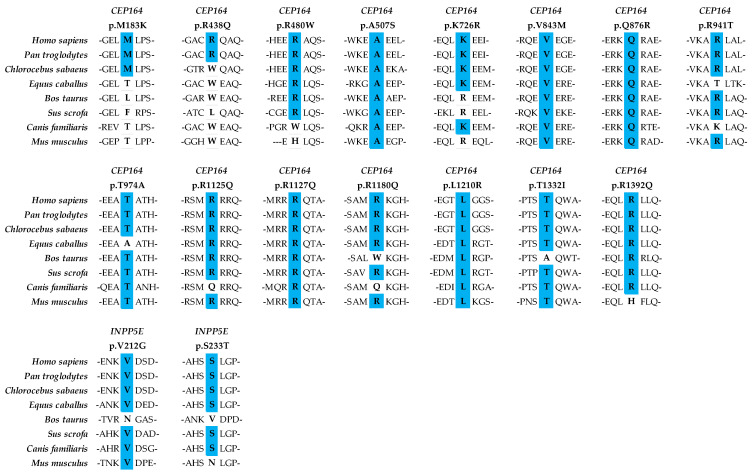
We identified 17 heterozygous missense rare variants across these two genes in our PCG cohort (Table 1). There were relatively more PCG-associated variants in CEP164 compared with the INPP5E gene. There were no homozygous pathogenic changes in either of these genes, and the distributions of overall variants comprised unique heterozygous alleles, compound heterozygous alleles (two heterozygous alleles within the same gene), and co-occurring alleles (co-occurrences of heterozygous alleles of CEP164 and INPP5E along with PCG-associated candidate genes, such as CYP1B1, LTBP2, TEK, and MYOC) (Figure 1). The INPP5E variants exhibited relatively higher REVEL scores compared with the CEP164 variants. All these rare variants were either absent or infrequently present among our ethnically matched control subjects and in the general populations of other global databases (Table 1). Most of these variants were highly conserved across multiple species (Figure 2).
Table 1.
A. Distributions of allele frequencies of rare variants in the CEP164 gene in PCG. B. Distributions of rare variants in the INPP5E gene in PCG.
| Chromosomal Position (GRCh38) | Amino Acid Change | dbSNP ID | REVEL Score | Cases MAF (n = 298) |
Controls MAF (n = 1757) |
p Value | Odds Ratio (95% CI) |
1000 Genomes (n = 2504); gnomAD v4.1 (n = 807,162); All of Us (n = 245,400) |
|---|---|---|---|---|---|---|---|---|
| A | ||||||||
| 11: 117361989T > A | p.M183K | rs144206271 | 0.18 | 0.0016 | 0.0003 | 0.15 | 5.91 (0.37–94.74) | 0.0008; 0.00063; 0.0006 |
| 11: 117375787G > A | p.R438Q | rs137987733 | 0.05 | 0.0016 | 0.0003 | 0.15 | 5.91 (0.37–94.74) | NA; 0.00018; 0.0001 |
| 11: 117381729C > T | p.R480W | rs112209873 | 0.04 | 0.0016 | 0 | - | - | 0.001; 0.00019; 0.00068 |
| 11: 117381810G > T | p.A507S | NA | 0.08 | 0.0016 | 0 | - | - | NA |
| 11: 117391109A > G | p.K726R | rs2044597036 | 0.02 | 0.0016 | 0 | - | - | NA |
| 11: 117393037G > A | p.V843M | rs566117718 | 0.09 | 0.0016 | 0.0008 | 0.55 | 1.96 (0.20–18.98) | 0.0012; 0.00005; 0.000016 |
| 11: 117394360A > G | p.Q876R | rs752659513 | 0.13 | 0.0033 | 0.0003 | 0.01 | 11.86 (1.07–131.20) | NA; 0.000044; 0.000006 |
| 11: 117394981G > C | p.R941T | rs749310077 | 0.03 | 0.0016 | 0 | - | - | NA; 0.0000006; NA |
| 11: 117395553A > G | p.T974A | rs56699807 | 0.04 | 0.0033 | 0.0003 | 0.01 | 11.86 (1.07–131.20) | 0.0002; 0.0003; 0.0003 |
| 11: 117397186G > A | p.R1125Q | rs767918200 | 0.03 | 0.0016 | 0.0008 | 0.55 | 1.96 (0.20–18.98) | NA; 0.000015; 0.000002 |
| 11: 117397192G > A | p.R1127Q | rs753895198 | 0.16 | 0.0016 | 0 | - | - | NA; 0.000009; 0.000016 |
| 11: 117407962G > A | p.R1180Q | rs568896676 | 0.02 | 0.0050 | 0.0006 | 0.004 | 8.92 (1.48–53.61) | NA; 0.00001; 0.000012 |
| 11: 117408909T > G | p.L1210R | rs767571570 | 0.17 | 0.0016 | 0 | - | - | NA; 0.000003; NA |
| 11: 117409864C > T | p.T1332I | rs760788324 | 0.03 | 0.0016 | 0 | - | - | NA; 0.000006; NA |
| 11: 117411806G > A | p.R1392Q | rs772989312 | 0.07 | 0.0016 | 0.0011 | 0.72 | 1.47 (0.16–13.24) | NA; 0.000012; 0.000016 |
| B | ||||||||
| 9: 136438722C > G | p.S233T | rs568767788 | 0.35 | 0.0016 | 0.0003 | 0.15 | 5.91 (0.37–94.74) | 0.00039; 0.000026; 0.000006 |
| 9: 136438785A > C | p.V212G | rs533861933 | 0.54 | 0.0016 | 0.0017 | 0.98 | 0.98 (0.11–8.18) | 0.00019; 0.0000068; 0.00001 |
MAF = Minor allele frequency; NA = Not available.; - = Cannot be determined.
Figure 1.
Distributions of rare variants in CEP164 and INPP5E genes in PCG cases. Hetero = Cases with unique heterozygous alleles of either of these genes; Compound hetero = Cases with two heterozygous alleles within the CEP164 and INPP5E gene, respectively; Co-occurring = Cases with co-occurring alleles of CEP164 and INPP5E, and PCG-associated candidate genes, res; NV = Cases without any variations in the CEP164 and INPP5E genes.
Figure 2.
Conservation of wild-type residues of the rare variants across multiple species in CEP164 and INPP5E. Multiple sequence alignment was done using Clustal W and Clustal X v2.1 on Jalview v2.11 with UniProt database, and a schematic was prepared in Excel. CEP164 protein identifiers: Homo sapiens = Q9UPV0, Pan troglodytes = K7D7G7, Chlorocebus sabaeus = A0A0D9S3C3, Equus caballus = A0A3Q2I1M0, Bos taurus = F1MNI1, Sus scrofa = A0A8W4FCK4, Canis familiaris = A0A8I3MTC7, Mus musculus = Q5DU05. INPP5E protein identifiers: Homo sapiens = Q9NRR6, Pan troglodytes = K7B557, Chlorocebus sabaeus = A0A0D9RTF7, Equus caballus = A0A3Q2HCU7, Bos taurus = E1BAU6, Sus scrofa = A0A287AEE9, Canis familiaris = A0A8I3N0Y9, Mus musculus = Q9JII2.
We observed common variants in CEP164 (n = 17) and INPP5E (n = 12) in our normal controls. The overall profile of allele frequencies of these common variants in our controls to other global databases is provided in Figure S1. Hardy–Weinberg equilibrium (HWE) analysis was performed for all the common variants of CEP164 and INPP5E genes in normal controls, and distributions of their minor allele frequencies (MAFs) are provided in Table S1. Only polymorphic alleles with MAF > 0.05 and in HWE (p > 0.001) were included.
Genetic associations were performed by taking the common variants of CEP164 (n = 10) and INPP5E (n = 9) genes, which were in HWE in our controls. Analysis of genetic associations of the common alleles between PCG cases and controls revealed that the “G” allele of rs73566945 variant (INPP5E) was significantly associated with the risk of PCG (p = 0.003; OR = 2.05; 95% CI, 1.27–3.29), which withstood statistical correction (corrected p value = 0.030) based on 10,000 permutations test (using Haploview). Genetic association for the rs73566945 genotypes was also undertaken. The homozygous genotype (GG) was significantly associated with PCG (p = 0.013; OR = 1.84, 95% CI, 1.13–2.99). The other common variants across these genes did not exhibit any association.
2.2. Haplotype Analysis of CEP164 and INPP5E Genes
The intragenic variants of CEP164 and INPP5E were used to generate linkage disequilibrium (LD) plots (Figure S2), followed by haplotype analysis. There was tight LD across the markers for both genes. The haplotypes ‘C-C’ and ‘G-C’ (rs59763167-rs521099) and ‘A-A-A’ (rs33982662-rs34936112-rs73566945) of CEP164 and INPP5E, respectively, were overrepresented either in the PCG cases or controls and exhibited a possible trend of association (based on uncorrected p value) but did not withstand statistical correction (Table 2). On the other hand, the ‘A-A-G’ (INPP5E) haplotype was significantly associated with the risk of PCG, which withstood statistical correction (based on 10,000 permutations test), which was largely contributed by the risk allele (G) of rs73566945. However, the rare variants across these two genes were not restricted to the risk haplotypes and were present in the background of other haplotypes as well.
Table 2.
Distributions of haplotypes in CEP164 and INPP5E genes.
| Genes | Haplotypes | Overall Haplotype Frequency | Frequency in Cases | Frequency in Controls | Chi Square | Uncorrected p Value | Corrected p Value * |
|---|---|---|---|---|---|---|---|
| CEP164 | C-C | 0.747 | 0.792 | 0.739 | 7.464 | 0.006 | 0.056 |
| CEP164 | C-G | 0.198 | 0.171 | 0.203 | 3.179 | 0.075 | 0.414 |
| CEP164 | G-C | 0.055 | 0.037 | 0.058 | 4.383 | 0.036 | 0.242 |
| INPP5E | C-G-G | 0.844 | 0.834 | 0.846 | 0.613 | 0.434 | 0.954 |
| INPP5E | A-A-G | 0.084 | 0.131 | 0.076 | 20.037 | 7.6 × 10−6 | 0.0005 |
| INPP5E | A-A-A | 0.055 | 0.032 | 0.059 | 7.234 | 0.007 | 0.055 |
Order of haplotypes for CEP164 (rs59763167-rs521099) and INPP5E (rs33982662-rs34936112-rs73566945); * Based on 10,000 permutation test (Haploview v4.2 software). Bold format represents the significant p values.
2.3. Genetic and Physical Interactions between CEP164 and CYP1B1
We observed that the heterozygous rare variants of CEP164 and INPP5E co-occurred with pathogenic variants of PCG-associated candidate genes (CYP1B1, LTBP2, TEK, and MYOC) in 5/298 (1.67%) cases (Figure 1). This indicated a possible multi allelic interactions of these genes in PCG pathogenesis. The distributions of the interacting alleles of these genes are provided in Supplementary Table S2.
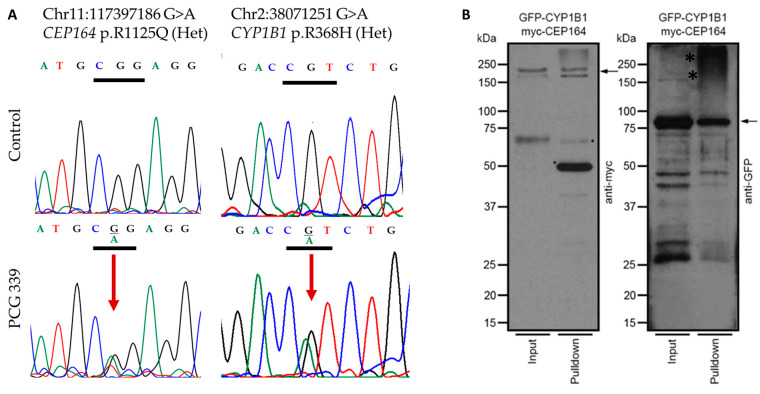
We next assessed potential physical interaction between CYP1B1 with recombinant epitope-tagged versions of these proteins in HEK293 cells. We selected CEP164 for this experiment because of its known localization to centrosomes and a previously reported observation of myocilin, another known glaucoma-related gene in the centrosomes. The HEK293 cells co-transfected with plasmids encoding GFP-CYP1B1 and MYC-CEP164 showed that CEP164 interacts with CYP1B1 (Figure 3).
Figure 3.
(A) Representative chromatograms indicating genetic interactions of CEP164: p.R1125Q and CYP1B1: p.R368H heterozygous rare variants in PCG 339 patient. The upper panel shows the wild-type sequence, and the lower panel indicates the variant marked by a red arrow. (B) HEK293 cells were transiently transfected with plasmids encoding GFP-CYP1B1 and MYC-CEP164. As a negative control, only GFP-encoding plasmid was utilized [10]. The cell extracts were subjected to immunoprecipitation (IP; pull-down) using anti-GFP or anti-myc antibodies, followed by SDS-PAGE and immunoblotting using anti-GFP or anti-myc antibodies, respectively. Arrows indicate the specific and expected protein bands (myc-CEP164 in the left panel and GFP-CYP1B1; kDa: kilo Daltons) detected by the indicated antibodies. Input lanes represent 20% of the protein used for IP. *: non-specific signal; Lower bands are likely the products of degradation. The negative control pull-downs were performed with protein extracts expressing the tags only, while the positive control was CYP1B1-TEK pull-down. These were previously published by our group [10].
2.4. Genotype–Phenotype Correlation
Genotype–phenotype correlation was based on IOP, corneal diameter (CD), cup-to-disc ratios (CDRs), and visual acuity (VA) at presentation and follow-ups at 3 months and one year amongst PCG cases harboring rare variants in the CEP164 gene. These cases were further classified based on their allelic configurations pertaining to the presence of either one heterozygous allele of a gene or combinations of multiple alleles based on co-occurring alleles of other candidate genes (Table 3). We could not perform a similar analysis for INPP5E due to a lack of PCG cases harboring rare variants in this gene (Table 1).
Table 3.
Genotype–phenotype correlation based on the presenting and follow-up IOP, corneal diameter, cup-to-disc ratio, and visual acuity in patients harboring various combinations of CEP164 variants.
| Genotype Combinations in PCG Cases (Number of Cases) |
Intraocular Pressure (mmHg) | Cup-to-Disc Ratio | Corneal Diameter (mm) | Visual Acuity (logMAR) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| At Presen-tation | After 3 Months | After 1 Year | At Presen-tation | After 3 Months | After 1 Year | At Presen-tation | After 3 Months | After 1 Year | At Presen-tation | After 3 Months | After 1 Year | |
| Cases with unique heterozygous CEP164 alleles only (n = 9) |
25.75 ± 6.63 | 12.86 ± 3.8 | 11.63 ± 1.69 | 0.48 ± 0.13 | 0.54 ± 0.22 | 0.46 ± 0.2 | 13.06 ± 0.98 | 12.71 ± 1.19 | 13.1 ± 0.74 | NA | NA | 1.19 ± 0.47 |
| Cases with compound heterozygous CEP164 alleles (n = 3) |
28 ± 6.93 |
15.33 ± 5.03 | 10.33 ± 2.52 | NA | 0.47 ± 0.23 | 0.5 ± 0.26 | 13.67 ± 0.58 | 13.33 ± 0.58 | 13.67 ± 0.29 | NA | NA | |
| Cases with co-occurring CEP164 alleles along with heterozygous alleles of other genes (n = 4) | 24.25 ± 6.65 | NA | 17.67 ± 5.86 | 0.83 ± 0.06 | NA | 0.63 ± 0.31 | 15 ± 2.29 |
NA | NA | NA | NA | 2.34 ± 0.75 |
| * p value (Unique heterozygous CEP164 alleles versus co-occurring CEP164 and alleles of other genes) | 0.72 | NA | 0.019 | 0.004 | NA | 0.284 | 0.056 | NA | NA | NA | NA | 0.053 |
| * p value (Compound heterozygous CEP164 alleles versus co-occurring CEP164 and alleles of other genes) |
0.5 | NA | 0.117 | NA | NA | 0.598 | 0.384 | NA | NA | NA | NA | NA |
* p value is based on Student’s t test; NA: Data are not available. Bold format represents the significant p values.
PCG cases harboring heterozygous alleles of CEP164, along with other candidate genes, had a relatively poor prognosis compared with those who harbored only a single copy of the CEP164 allele. The mean IOP was uncontrolled and significantly raised in these cases at one-year follow-up (p = 0.019). Similarly, the cup-to-disc ratios were significantly higher in these cases at presentation (p = 0.004). However, visual acuity (VA) could not be graded at presentation or immediate follow-up in cases harboring CEP164 variants (Table 3).
3. Discussion
PCG is a complex disease and is collectively attributed to multiple gene variants with varying magnitudes of effects. The PCG-associated candidate genes (CYP1B1, LTBP2, TEK, MYOC, and FOXC1) do not contribute to our complete understanding of the underlying molecular bases of this disease [3,10,11]. The discovery of novel genes and their functional interactions have provided new insights into biological pathways that may be implicated in PCG pathogenesis. Our earlier efforts in this direction have led to the identification of genic interactions of CYP1B1 with MYOC [13], FOXC1 [14], and PRSS56 [25] in PCG. Additionally, we also demonstrated physical interactions of CYP1B1 with TEK (a ciliary gene) due to their digenic involvement in PCG [10]. We have now extended our efforts toward understanding the possible involvements of centrosomal (CEP164) and its interacting ciliary (INPP5E) gene in PCG cases. Based on its genetic profile, functional interaction, and genotype–phenotype correlation, our study suggested that the CEP164 gene could be a novel candidate in PCG pathogenesis.
We identified multiple novel rare variants in the highly conserved regions of CEP164 and INPP5E in PCG cases devoid of homozygous mutations in the known PCG-associated candidate genes (Table 1; Figure 2). It is intriguing that we did not observe any homozygous pathogenic alleles in these genes, unlike in most other PCG-associated genes. The overall allelic contributions of CEP164 (2.51%) and INPP5E (0.33%) in PCG were very low compared with the major candidate gene, CYP1B1 (38.07%). However, their frequencies were comparable to other genes, comprising LTBP2, TEK, MYOC, and FOXC1, which ranged from 0.31 to 3.55% in our PCG cohort.
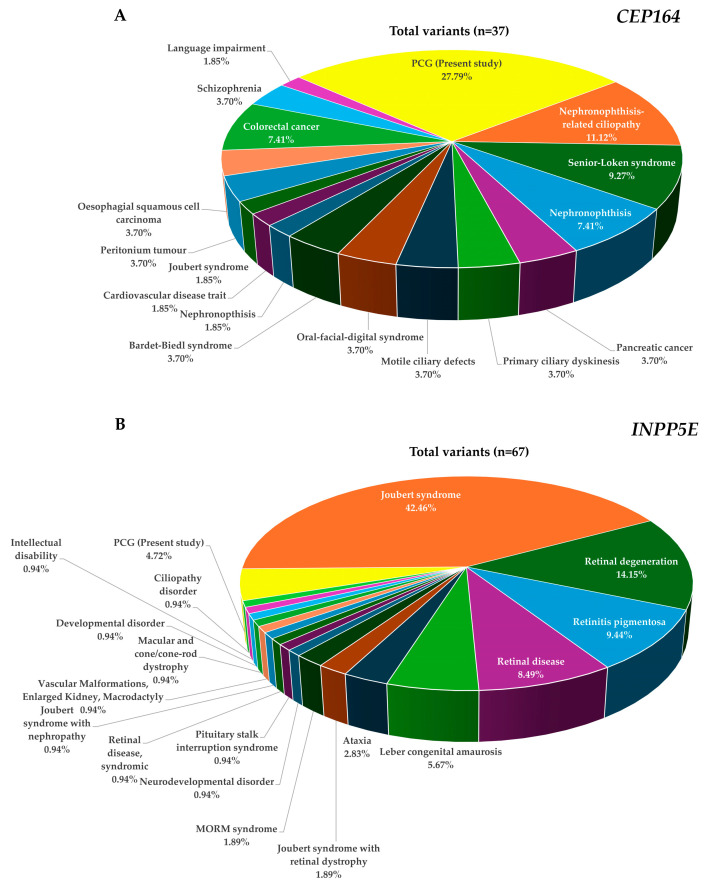
CEP164 is involved in microtubule organization and maintenance for the formation of primary cilia, which are essential for the proper functioning of the TM and the anterior chamber angle [37,38]. Additionally, it is also involved in DNA damage response and chromosome segregation, which are critical processes in maintaining genomic stability. Mutations in CEP164 have been associated with severe ciliopathy phenotypes (Figure 4A), such as nephronophthisis, occipital encephalocele, and liver fibrosis, and milder phenotypes, like nephronophthisis with Leber congenital amaurosis [33,44,45,50,51]. The exact mechanisms by which CEP164 would contribute to PCG are not fully understood, but mutations in this gene may possibly interfere with cell cycle progression, apoptosis, and epithelial-to-mesenchymal transition during developmental stages [48].
Figure 4.
Pie charts depicting the proportions of observed pathogenic variants in CEP164 (A) and INPP5E (B) across different phenotypes and the present study (PCG). The data show the frequencies of the classified disease mutations as per the HGMD database (Tables S3 and S4).
We also observed co-occurrences of heterozygous pathogenic alleles of CEP164 (n = 4) that genetically interact with heterozygous alleles of other PCG-associated genes (Figure 1). Interestingly, the genetic interaction of CEP164 (p.R1125Q) with CYP1B1 (p.R368H) was supported by a corresponding physical interaction in HEK293 cells (Figure 3), like our earlier observations on TEK and CYP1B1 [10]. We speculate that co-occurring mutations in CEP164 and CYP1B1 (Figure 3) may perturb this interaction and disrupt ciliary functions and regulation of IOP. However, this needs to be confirmed with additional functional validations.
We have also demonstrated that another JOAG-associated gene, MYOC, localizes in the centrosome and genetically interacts with CYP1B1 through a digenic mechanism in PCG [13]. While we did not observe any genetic interactions of MYOC and CEP164 alleles in our present cohort, the presence of pathogenic variants of MYOC in PCG in our earlier study [13] and in other populations [22,52,53,54,55,56,57,58,59] suggest the potential involvement of centrosomal proteins in PCG, which need to be functionally characterized.
Interestingly, PCG patients harboring heterozygous alleles of CEP164, along with heterozygous alleles of other PCG-associated genes had a relatively poor prognosis in terms of their IOP control and cup-to-disc ratios (Table 3). This has been consistently observed with other multi-allelic scenarios involving MYOC [13] and TEK [10] with CYP1B1 in our PCG cohort.
The INPP5E, on the other hand, is a widely expressed ciliary gene that plays a critical role in controlling ciliary function by regulating the length of the cilia [42]. Mutations in INPP5E have been associated with various ciliopathies, including Joubert syndrome, which can present with glaucoma as one of the clinical features (Figure 4B). Our data revealed relatively smaller numbers of rare variants in INPP5E compared with CEP164 (Table 1). There was only one co-occurring allele of INPP5E with another gene (MYOC), and genotype–phenotype correlation was inconclusive considering the number of PCG patients harboring rare variants in this gene (Table 3). Although there was no evidence of any genetic interactions between CEP164 and INPP5E genes, it could be speculated that defects in INPP5E may lead to shortened cilia and impaired ciliary function, which may further contribute to the development of PCG [40,42].
Overall, CEP164 and INPP5E have revealed a total of 22 and 65 pathogenic variants, respectively, across multiple phenotypes as per the HGMD (Human Gene Mutation database) [60]. The discovery of rare variants in these genes in PCG has expanded the mutation spectra of CEP164 (n = 37) and INPP5E (n = 67) genes (Figure 4A,B; Tables S3 and S4). We observed a relatively larger number of rare variants in CEP164 compared with INPP5E in PCG (Figure 4). The observation of a functional interaction and poor prognosis in cases with co-occurring alleles of CEP164 and other genes, along with a strong pathogenic potential of INPP5E, might be indicative of their underlying role(s) in PCG that need to be elucidated further in additional cohorts.
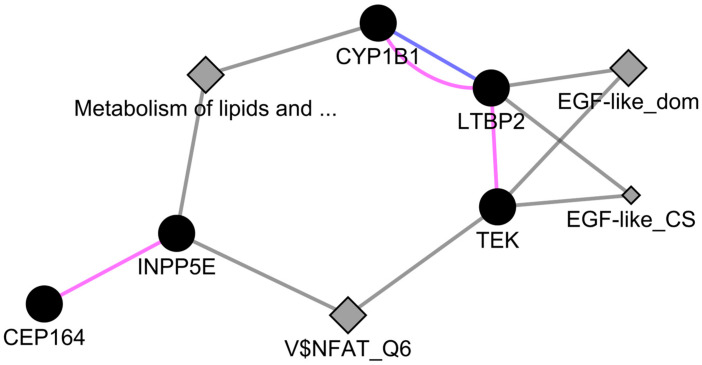
Network analysis of CEP164 and INPP5E, along with PCG-associated candidate genes, indicated their interactions through pathways, which is further suggestive of their involvement with PCG (Figure 5). Further, metabolic interactions of lipid and lipo-proteins were noted between CYP1B1 and INPP5E (that further interacts with CEP164). On the other hand, TEK and INPP5E shared a common transcription factor target. The involvement of these genes in ciliopathies and various retinal functions indicated a possible mechanism in PCG pathogenesis [44,45,46,47].
Figure 5.
Network analysis of CEP164 and INPP5E with PCG-associated candidate genes. The network was generated using GeneMania of Cytoscape v3.5.3 software. Solid nodes represent genes. Diamond nodes represent the consolidated pathways, domains and transcription factor targets. Color scheme: Pink = Co-expression, Blue = Co-localization, Grey = Shared attribute. “V$NFAT_Q6” = Transcription factor target motif, “EGF-like_dom” = EGF-like domain, “EGF-like_CS” = EGF-like conserved site, “Metabolism of lipids and …” = Metabolism of lipids and lipoproteins.
CEP164 is also involved in retinal photoreceptor layer development [33] and INPP5E in embryonic neural development [49]. Both these genes synergistically interact with each other in the formation of a functional network involving the primary cilia [43]. Based on the involvement of these genes in our cohort, we speculate their involvement in retinal damage in PCG. Reduction in retinal nerve fiber layer thickness (RNFL) and its correlation with IOP among PCG patients [61,62] have already indicated the involvement of retinal-associated genes in disease pathogenesis. This is further supported by the fact that the primary PCG-associated gene, CYP1B1, is expressed in retinal ganglion cells (RGCs) and promotes its survival [63], but the possible mechanisms leading to retinal damage in PCG remains elusive.
In summary, CEP164 and INPP5E genes may play critical roles in PCG considering their involvements in centrosomal, ciliary, and retinal functions and the regulation of IOP. Further research would be needed to fully understand their underlying molecular mechanisms through which these genes contribute to PCG pathogenesis.
4. Materials and Methods
4.1. Study Approval
The study was approved by the Institutional Review Board of L V Prasad Eye Institute (LEC 09-18-141) and adhered to the tenets of the Declaration of Helsinki. A written informed consent was obtained from all the study subjects and guardians of minors.
4.2. Enrolment of Cases and Controls
The study comprised 2055 subjects, including PCG cases (n = 298) and ethnically matched normal controls (n = 1757). Detailed inclusion and exclusion criteria have been described earlier [10,64]. Briefly, all study participants underwent a comprehensive ocular examination. Each case was independently diagnosed by at least two glaucoma specialists, and a good inter-observer agreement was seen based on kappa statistics (κ = 0.94). Cases that had discordant diagnoses amongst the clinicians were excluded. Demographic details of subjects, including their gender, history of consanguinity, and age at disease onset and intervention, were recorded. Quantitative data of clinical variables such as intraocular pressure (IOP), corneal diameter (CD), cup-to-disc ratio (CDR), and visual acuity (VA) were collected from all the PCG cases at presentation and further follow-ups following surgery.
4.3. Targeted Sequencing, Cell Culture, and Pull-Down Assay
A customized, targeted gene panel (Applied Biosystems, Foster City, CA, USA) comprising CEP164 and INPP5E genes, along with other PCG-associated genes, was used for screening. Library preparation, amplification, enrichments, and deep sequencings were performed as per the manufacturer’s guidelines (Applied Biosystems, Foster City, CA, USA). The quality control measures for data cleaning, data analysis pipelines, and interpretations have been described earlier [10]. The observed variants were validated through Sanger sequencing using the BigDye Terminator (v3.1) chemistry on a 3130xl Genetic Analyzer (Applied Biosystems, Foster City, CA, USA). The cell culture experiments with HEK293 cells and GFP pull-down assay were as described earlier [10].
4.4. Allele and Haplotype Analysis
The Ensembl canonical transcripts of CEP164: ENST00000278935.8 and INPP5E: ENST00000371712.4 were considered for analysis. Allele frequencies were calculated using the gene counting method along with their odds ratios and 95% confidence intervals. p values were based on the Chi-square test. These frequencies were compared with the global allele frequencies reported in the 1000 Genomes [65], gnomAD [66], and All of Us [67] databases.
Pathogenicity predictions were based on REVEL scores [68], which provide a score combining 13 different tools (SIFT, PolyPhen2, Mutation Taster, Mutation Assessor, FATHMM v2.3, MutPred, VEST 3.0, PROVEAN, LRT, phyloP, SiPhy, GERP++, and phastCons).
Hardy–Weinberg equilibrium (HWE) and haplotypes analysis were conducted using the HaploView (version 4.2) software [69]. The HWE cut-off p value was 0.001 based on the default parameter of this software. The minimum genotype cut-off percent was set at 100%, and the minimum minor allele frequency (MAF) was >0.05. Only haplotypes with frequencies above 5% were considered. Phenotypes of genes and disease-causing mutations were extracted from the HGMD database (accessed on 19 July 2024) [60].
4.5. Conservation of Amino Acids
Multiple sequence alignment was performed using Clustal W and Clustal X v2.1 [70] on the Jalview v2.11 platform [71]. With three residues on either side for the target residue, the remaining intermediary amino acids were removed from the sequence.
4.6. Network Analysis
Network analysis was conducted using the GeneMania v3.5.3 app on the Cytoscape software platform [72]. Gene list as an input, co-expression, co-localization, genetic interactions, pathways, physical interactions, predicted interactions along with attributes including consolidated pathways, drug interactions, InterPro, miRNA-target-predictions, and transcriptional factor targets were analyzed. The top twenty related genes and, at most, twenty attributes were used with automatic weighting.
5. Conclusions
The potential functional involvement and genotype–phenotype correlation exhibited by CEP164 variants, along with a strong pathogenic potential of INPP5E variant, suggest these genes as a potential candidate(s) in PCG and a yet unexplored involvement of cilia-centrosomal functions in disease pathogenesis.
Acknowledgments
We thank all the patients and the normal volunteers for their participation in this study. We also acknowledge the clinical support of Ravi Thomas, Rajul Parikh, Harsha Rao and Sirisha Senthil for their help with clinical diagnosis of some patients. Goutham Pyatla was supported by a fellowship (09/815(0023)/2017-EMR-I) from the Council of Scientific and Industrial Research (CSIR); Samir Bera (45/04/2022/HUM/BMS) and Ashish Mishra (3/1/3/JRF-2020/HRD/134416/33) are currently supported with fellowships from the Indian Council of Medical Research (ICMR), Government of India.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/ijms251810028/s1.
Author Contributions
Conceptualization, S.C.; methodology, G.P., M.K., W.Z., S.R., A.M., S.P., S.B. (Samir Bera), S.B. (Seema Banerjee), A.A.A., R.D. and K.S.; software, S.C., G.P., M.K., W.Z. and S.R.; validation, G.P., M.K., W.Z., S.R., S.B., A.A.A., A.M., S.M. and I.K.; formal analysis, S.C., G.P., M.K., W.Z. and S.R.; investigation, S.C., G.P., M.K., W.Z., S.R. and A.K.M.; resources, S.C. and R.C.K.; data curation, G.P., M.K., W.Z., S.R., S.B. (Seema Banerjee), K.S. and S.M.; writing—original draft preparation, S.C., G.P., S.B. and A.M.; writing—review and editing, S.C., A.K.M., I.K. and R.C.K.; visualization, G.P., M.K., W.Z. and S.R.; supervision, S.C., R.C.K., I.K. and S.M.; project administration, S.C., I.K. and R.C.K.; funding acquisition, S.C. and R.C.K. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of L V Prasad Eye Institute (LEC 09-18-141) in September 2018.
Informed Consent Statement
A written informed consent was obtained from all the study subjects and guardians of minors.
Data Availability Statement
All data are provided in the text and Supplementary Materials of this manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding Statement
The study was supported in parts by extramural research grants from the Department of Biotechnology (BT/PR32404/MED/30/2136/2019), Indian Council of Medical Research (2021-14979/Genomics/Adhoc-BMS), Science Engineering and Research Board (SERB; EMR/2016/006979), Government of India, and the Hyderabad Eye Research Foundation (HERF), Hyderabad, India.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.de Luise V.P., Anderson D.R. Primary infantile glaucoma (congenital glaucoma) Surv. Ophthalmol. 1983;28:1–19. doi: 10.1016/0039-6257(83)90174-1. [DOI] [PubMed] [Google Scholar]
- 2.Karaconji T., Zagora S., Grigg J.R. Approach to childhood glaucoma: A review. Clin. Exp. Ophthalmol. 2022;50:232–246. doi: 10.1111/ceo.14039. [DOI] [PubMed] [Google Scholar]
- 3.Pan Y., Iwata T. Exploring the genetic landscape of childhood glaucoma. Children. 2024;11:454. doi: 10.3390/children11040454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stoilov I., Akarsu A.N., Sarfarazi M. Identification of three different truncating mutations in cytochrome P4501B1 (CYP1B1) as the principal cause of primary congenital glaucoma (Buphthalmos) in families linked to the GLC3A locus on chromosome 2p21. Hum. Mol. Genet. 1997;6:641–647. doi: 10.1093/hmg/6.4.641. [DOI] [PubMed] [Google Scholar]
- 5.Li N., Zhou Y., Du L., Wei M., Chen X. Overview of Cytochrome P450 1B1 gene mutations in patients with primary congenital glaucoma. Exp. Eye Res. 2011;93:572–579. doi: 10.1016/j.exer.2011.07.009. [DOI] [PubMed] [Google Scholar]
- 6.Chouiter L., Nadifi S. Analysis of CYP1B1 Gene Mutations in Patients with Primary Congenital Glaucoma. J. Pediatr. Genet. 2017;6:205–214. doi: 10.1055/s-0037-1602695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ali M., McKibbin M., Booth A., Parry D.A., Jain P., Riazuddin S.A., Hejtmancik J.F., Khan S.N., Firasat S., Shires M., et al. Null mutations in LTBP2 cause primary congenital glaucoma. Am. J. Hum. Genet. 2009;84:664–671. doi: 10.1016/j.ajhg.2009.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Narooie-Nejad M., Paylakhi S.H., Shojaee S., Fazlali Z., Rezaei Kanavi M., Nilforushan N., Yazdani S., Babrzadeh F., Suri F., Ronaghi M., et al. Loss of function mutations in the gene encoding latent transforming growth factor beta binding protein 2, LTBP2, cause primary congenital glaucoma. Hum. Mol. Genet. 2009;18:3969–3977. doi: 10.1093/hmg/ddp338. [DOI] [PubMed] [Google Scholar]
- 9.Souma T., Tompson S.W., Thomson B.R., Siggs O.M., Kizhatil K., Yamaguchi S., Feng L., Limviphuvadh V., Whisenhunt K.N., Maurer-Stroh S., et al. Angiopoietin receptor TEK mutations underlie primary congenital glaucoma with variable expressivity. J. Clin. Invest. 2016;126:2575–2587. doi: 10.1172/JCI85830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kabra M., Zhang W., Rathi S., Mandal A.K., Senthil S., Pyatla G., Ramappa M., Banerjee S., Shekhar K., Marmamula S., et al. Angiopoietin receptor TEK interacts with CYP1B1 in primary congenital glaucoma. Hum. Genet. 2017;136:941–949. doi: 10.1007/s00439-017-1823-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ling C., Zhang D., Zhang J., Sun H., Du Q., Li X. Updates on the molecular genetics of primary congenital glaucoma (Review) Exp. Ther. Med. 2020;20:968–977. doi: 10.3892/etm.2020.8767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Han X., Gharahkhani P., Hamel A.R., Ong J.S., Rentería M.E., Mehta P., Dong X., Pasutto F., Hammond C., Young T.L., et al. Large-scale multitrait genome-wide association analyses identify hundreds of glaucoma risk loci. Nat. Genet. 2023;55:1116–1125. doi: 10.1038/s41588-023-01428-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kaur K., Reddy A.B., Mukhopadhyay A., Mandal A.K., Hasnain S.E., Ray K., Thomas R., Balasubramanian D., Chakrabarti S. Myocilin gene implicated in primary congenital glaucoma. Clin. Genet. 2005;67:335–340. doi: 10.1111/j.1399-0004.2005.00411.x. [DOI] [PubMed] [Google Scholar]
- 14.Chakrabarti S., Kaur K., Rao K.N., Mandal A.K., Kaur I., Parikh R.S., Thomas R. The transcription factor gene FOXC1 exhibits a limited role in primary congenital glaucoma. Invest. Ophthalmol. Vis. Sci. 2009;50:75–83. doi: 10.1167/iovs.08-2253. [DOI] [PubMed] [Google Scholar]
- 15.Makhoul N.J., Wehbi Z., El Hadi D., Noureddine B., Boustany R.M., Al-Haddad C. Whole-exome screening for primary congenital glaucoma in Lebanon. Ophthalmic Genet. 2023;44:234–245. doi: 10.1080/13816810.2023.2189949. [DOI] [PubMed] [Google Scholar]
- 16.Yadav M., Yadav A., Bhardwaj A., Dhull C.S., Sachdeva S., Yadav R., Tanwar M. A rare optineurin mutation in an Indian family with coexistence of JOAG and PCG. Indian J. Ophthalmol. 2023;71:3016–3023. doi: 10.4103/IJO.IJO_3383_22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fu H., Siggs O.M., Knight L.S., Staffieri S.E., Ruddle J.B., Birsner A.E., Collantes E.R., Craig J.E., Wiggs J.L., D’Amato R.J. Thrombospondin 1 missense alleles induce extracellular matrix protein aggregation and TM dysfunction in congenital glaucoma. J. Clin. Invest. 2022;132:e156967. doi: 10.1172/JCI156967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Morales-Cámara S., Alexandre-Moreno S., Bonet-Fernández J.M., Atienzar-Aroca R., Aroca-Aguilar J.D., Ferre-Fernández J.J., Méndez C.D., Morales L., Fernández-Sánchez L., Cuenca N., et al. Role of GUCA1C in Primary Congenital Glaucoma and in the Retina: Functional Evaluation in Zebrafish. Genes. 2020;11:550. doi: 10.3390/genes11050550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gupta V., Somarajan B.I., Kaur G., Gupta S., Singh R., Pradhan D., Singh H., Kaur P., Sharma A., Chawla B., et al. Exome sequencing identifies procollagen-lysine 2-oxoglutarate 5-dioxygenase 2 mutations in primary congenital and juvenile glaucoma. Indian J. Ophthalmol. 2021;69:2710–2716. doi: 10.4103/ijo.IJO_1750_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mauri L., Uebe S., Sticht H., Vossmerbaeumer U., Weisschuh N., Manfredini E., Maselli E., Patrosso M., Weinreb R.N., Penco S., et al. Expanding the clinical spectrum of COL1A1 mutations in different forms of glaucoma. Orphanet. J. Rare Dis. 2016;11:108. doi: 10.1186/s13023-016-0495-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Medina-Trillo C., Aroca-Aguilar J.D., Ferre-Fernandez J.J., Alexandre-Moreno S., Morales L., Mendez-Hernandez C.D., Garcia-Feijoo J., Escribano J. Role of FOXC2 and PITX2 rare variants associated with mild functional alterations as modifier factors in congenital glaucoma. PLoS ONE. 2019;14:e0211029. doi: 10.1371/journal.pone.0211029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hadrami M., Bonnet C., Zeitz C., Veten F., Biya M., Hamed C.T., Condroyer C., Wang P., Sidi M.M., Cheikh S., et al. Mutation profile of glaucoma candidate genes in Mauritanian families with primary congenital glaucoma. Mol. Vis. 2019;25:373–381. [PMC free article] [PubMed] [Google Scholar]
- 23.Bonet-Fernández J.M., Aroca-Aguilar J.D., Corton M., Ramírez A.I., Alexandre-Moreno S., García-Antón M.T., Salazar J.J., Ferre-Fernández J.J., Atienzar-Aroca R., Villaverde C., et al. CPAMD8 loss-of-function underlies non-dominant congenital glaucoma with variable anterior segment dysgenesis and abnormal extracellular matrix. Hum. Genet. 2020;139:1209–1231. doi: 10.1007/s00439-020-02164-0. [DOI] [PubMed] [Google Scholar]
- 24.Young T.L., Whisenhunt K.N., Jin J., LaMartina S.M., Martin S.M., Souma T., Limviphuvadh V., Suri F., Souzeau E., Zhang X., et al. SVEP1 as a Genetic Modifier of TEK-Related Primary Congenital Glaucoma. Invest. Ophthalmol. Vis. Sci. 2020;61:6. doi: 10.1167/iovs.61.12.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Labelle-Dumais C., Pyatla G., Paylakhi S., Tolman N.G., Hameed S., Seymens Y., Dang E., Mandal A.K., Senthil S., Khanna R.C., et al. Loss of PRSS56 function leads to ocular angle defects and increased susceptibility to high intraocular pressure. Dis. Model. Mech. 2020;13:dmm042853. doi: 10.1242/dmm.042853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shivanna M., Anand M., Chakrabarti S., Khanna H. Ocular Ciliopathies: Genetic and Mechanistic Insights into Developing Therapies. Curr. Med. Chem. 2019;26:3120–3131. doi: 10.2174/0929867325666180917102557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Seo S., Sonn S.K., Kweon H.Y., Jin J., Kume T., Ko J.Y., Park J.H., Oh G.T. Primary Cilium in Neural Crest Cells Crucial for Anterior Segment Development and Corneal Avascularity. Invest. Ophthalmol. Vis. Sci. 2024;65:30. doi: 10.1167/iovs.65.3.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Portal C., Rompolas P., Lwigale P., Iomini C. Primary cilia deficiency in neural crest cells models anterior segment dysgenesis in mouse. Elife. 2019;8:e52423. doi: 10.7554/eLife.52423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ueda J., Wentz–Hunter K.K., Cheng E.L., Fukuchi T., Abe H., Yue B.Y. Ultrastructural localization of myocilin in human trabecular meshwork cells and tissues. J. Histochem. Cytochem. 2000;48:1321–1330. doi: 10.1177/002215540004801003. [DOI] [PubMed] [Google Scholar]
- 30.Noda S., Mashima Y., Obazawa M., Kubota R., Oguchi Y., Kudoh J., Minoshima S., Shimizu N. Myocilin expression in the astrocytes of the optic nerve head. Biochem. Biophys. Res. Commun. 2000;276:1129–1135. doi: 10.1006/bbrc.2000.3591. [DOI] [PubMed] [Google Scholar]
- 31.Luo N., Conwell M.D., Chen X., Kettenhofen C.I., Westlake C.J., Cantor L.B., Wells C.D., Weinreb R.N., Corson T.W., Spandau D.F., et al. Primary cilia signaling mediates intraocular pressure sensation. Proc. Natl. Acad. Sci. USA. 2014;111:12871–12876. doi: 10.1073/pnas.1323292111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhou P., Zhou J. The Primary Cilium as a Therapeutic Target in Ocular Diseases. Front. Pharmacol. 2020;11:977. doi: 10.3389/fphar.2020.00977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Reed M., Takemaru K.I., Ying G., Frederick J.M., Baehr W. Deletion of CEP164 in mouse photoreceptors post-ciliogenesis interrupts ciliary intraflagellar transport (IFT) PLoS Genet. 2022;18:e1010154. doi: 10.1371/journal.pgen.1010154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Li B., Xie T., Nawy S., Shen Y. The development and the genetic diseases of the ciliary body. Cell Insight. 2024;3:100162. doi: 10.1016/j.cellin.2024.100162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Teilmann S.C., Christensen S.T. Localization of the angiopoietin receptors Tie-1 and Tie-2 on the primary cilia in the female reproductive organs. Cell Biol. Int. 2005;29:340–346. doi: 10.1016/j.cellbi.2005.03.006. [DOI] [PubMed] [Google Scholar]
- 36.Yoon M.J., Cho C.H., Lee C.S., Jang I.H., Ryu S.H., Koh G.Y. Localization of Tie2 and phospholipase D in endothelial caveolae is involved in angiopoietin-1-induced MEK/ERK phosphorylation and migration in endothelial cells. Biochem. Biophys. Res. Commun. 2003;308:101–105. doi: 10.1016/S0006-291X(03)01341-X. [DOI] [PubMed] [Google Scholar]
- 37.Graser S., Stierhof Y.-D., Lavoie S.B., Gassner O.S., Lamla S., Le Clech M., Nigg E.A. Cep164, a novel centriole appendage protein required for primary cilium formation. J. Cell Biol. 2007;179:321–330. doi: 10.1083/jcb.200707181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sivasubramaniam S., Sun X., Pan Y.R., Wang S., Eva Y.H.L. Cep164 is a mediator protein required for the maintenance of genomic stability through modulation of MDC1, RPA, and CHK1. Genes Dev. 2008;22:587–600. doi: 10.1101/gad.1627708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Luo N., Lu J., Sun Y. Evidence of a role of inositol polyphosphate 5-phosphatase INPP5E in cilia formation in zebrafish. Vision Res. 2012;75:98–107. doi: 10.1016/j.visres.2012.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang R., Tang J., Li T., Zhou J., Pan W. INPP5E and Coordination of Signaling Networks in Cilia. Front. Mol. Biosci. 2022;9:885592. doi: 10.3389/fmolb.2022.885592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Plotnikova O.V., Seo S., Cottle D.L., Conduit S., Hakim S., Dyson J.M., Mitchell C.A., Smyth I.M. INPP5E interacts with AURKA, linking phosphoinositide signaling to primary cilium stability. J. Cell Sci. 2015;128:364–372. doi: 10.1242/jcs.161323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yue H., Li S., Qin J., Gao T., Lyu J., Liu Y., Wang X., Guan Z., Zhu Z., Niu B., et al. Down-Regulation of Inpp5e Associated With Abnormal Ciliogenesis During Embryonic Neurodevelopment Under Inositol Deficiency. Front. Neurol. 2021;12:579998. doi: 10.3389/fneur.2021.579998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Humbert M.C., Weihbrecht K., Searby C.C., Li Y., Pope R.M., Sheffield V.C., Seo S. ARL13B, PDE6D, and CEP164 form a functional network for INPP5E ciliary targeting. Proc. Natl. Acad. Sci. USA. 2012;109:19691–19696. doi: 10.1073/pnas.1210916109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Maria M., Lamers I.J., Schmidts M., Ajmal M., Jaffar S., Ullah E., Mustafa B., Ahmad S., Nazmutdinova K., Hoskins B., et al. Genetic and clinical characterization of Pakistani families with Bardet-Biedl syndrome extends the genetic and phenotypic spectrum. Sci. Rep. 2016;6:34764. doi: 10.1038/srep34764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Vilboux T., Doherty D.A., Glass I.A., Parisi M.A., Phelps I.G., Cullinane A.R., Zein W., Brooks B.P., Heller T., Soldatos A., et al. Molecular genetic findings and clinical correlations in 100 patients with Joubert syndrome and related disorders prospectively evaluated at a single center. Genet. Med. 2017;19:875–882. doi: 10.1038/gim.2016.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jacoby M., Cox J.J., Gayral S., Hampshire D.J., Ayub M., Blockmans M., Pernot E., Kisseleva M.V., Compere P., Schiffmann S.N., et al. INPP5E mutations cause primary cilium signaling defects, ciliary instability and ciliopathies in human and mouse. Nat. Genet. 2009;41:1027–1031. doi: 10.1038/ng.427. [DOI] [PubMed] [Google Scholar]
- 47.Travaglini L., Brancati F., Silhavy J., Iannicelli M., Nickerson E., Elkhartoufi N., Scott E., Spencer E., Gabriel S., Thomas S., et al. Phenotypic spectrum and prevalence of INPP5E mutations in Joubert syndrome and related disorders. Eur. J. Hum. Genet. 2013;21:1074–1078. doi: 10.1038/ejhg.2012.305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Slaats G.G., Ghosh A.K., Falke L.L., Le Corre S., Shaltiel I.A., van de Hoek G., Klasson T.D., Stokman M.F., Logister I., Verhaar M.C. Nephronophthisis-associated CEP164 regulates cell cycle progression, apoptosis and epithelial-to-mesenchymal transition. PLoS Genet. 2014;10:e1004594. doi: 10.1371/journal.pgen.1004594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yue H., Zhu X., Li S., Wang F., Wang X., Guan Z., Zhu Z., Niu B., Zhang T., Guo J., et al. Relationship Between INPP5E Gene Expression and Embryonic Neural Development in a Mouse Model of Neural Tube Defect. Med. Sci. Monit. 2018;24:2053–2059. doi: 10.12659/MSM.906095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Chaki M., Airik R., Ghosh A.K., Giles R.H., Chen R., Slaats G.G., Wang H., Hurd T.W., Zhou W., Cluckey A., et al. Exome capture reveals ZNF423 and CEP164 mutations, linking renal ciliopathies to DNA damage response signaling. Cell. 2012;150:533–548. doi: 10.1016/j.cell.2012.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Devlin L.A., Ramsbottom S.A., Overman L.M., Lisgo S.N., Clowry G., Molinari E., Powell L., Miles C.G., Sayer J.A. Embryonic and foetal expression patterns of the ciliopathy gene CEP164. PLoS ONE. 2020;15:e0221914. doi: 10.1371/journal.pone.0221914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kim H.-J., Suh W., Park S.C., Kim C.Y., Park K.H., Kook M.S., Kim Y.Y., Kim C.-S., Park C.K., Ki C.-S., et al. Mutation spectrum of CYP1B1 and MYOC genes in Korean patients with primary congenital glaucoma. Mol. Vis. 2011;17:2093–2101. [PMC free article] [PubMed] [Google Scholar]
- 53.Chen Y., Jiang D., Yu L., Katz B., Zhang K., Wan B., Sun X. CYP1B1 and MYOC mutations in 116 Chinese patients with primary congenital glaucoma. Arch. Ophthalmol. 2008;126:1443–1447. doi: 10.1001/archopht.126.10.1443. [DOI] [PubMed] [Google Scholar]
- 54.Geyer O., Wolf A., Levinger E., Harari-Shacham A., Walton D.S., Shochat C., Korem S., Bercovich D. Genotype/phenotype correlation in primary congenital glaucoma patients from different ethnic groups of the Israeli population. Am. J. Ophthalmol. 2011;151:263–271. doi: 10.1016/j.ajo.2010.08.038. [DOI] [PubMed] [Google Scholar]
- 55.Suh W., Kee C. A clinical and molecular genetics study of primary congenital glaucoma in South Korea. Br. J. Ophthalmol. 2012;96:1372–1377. doi: 10.1136/bjophthalmol-2012-301517. [DOI] [PubMed] [Google Scholar]
- 56.Hilal L., Boutayeb S., Serrou A., Refass-Buret L., Shisseh H., Bencherifa F., El Mzibri M., Benazzouz B., Berraho A. Screening of CYP1B1 and MYOC in Moroccan families with primary congenital glaucoma: Three novel mutations in CYP1B1. Mol. Vis. 2010;16:1215–1226. [PMC free article] [PubMed] [Google Scholar]
- 57.Fassad M.R., Amin A.K., Morsy H.A., Issa N.M., Bayoumi N.H., El Shafei S.A., Kholeif S.F. CYP1B1 and myocilin gene mutations in Egyptian patients with primary congenital glaucoma. Egypt J. Med. Hum. Genet. 2017;18:219–224. doi: 10.1016/j.ejmhg.2016.07.003. [DOI] [Google Scholar]
- 58.Akbas A.C., Erdem E., Bozdogan S.T., Harbiyeli I.I., Yagmur M. CYP1B1 and MYOC Gene Analysis of Patients with Primary Congenital Glaucoma in the Cukurova Region of Turkey. J. Pediatr. Genet. 2023 doi: 10.1055/s-0043-1774294. [DOI] [Google Scholar]
- 59.Vincent A.L., Billingsley G., Buys Y., Levin A.V., Priston M., Trope G., Williams-Lyn D., Héon E. Digenic inheritance of early-onset glaucoma: CYP1B1, a potential modifier gene. Am. J. Hum. Genet. 2002;70:448–460. doi: 10.1086/338709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Stenson P.D., Ball E.V., Mort M., Phillips A.D., Shiel J.A., Thomas N.S., Abeysinghe S., Krawczak M., Cooper D.N. Human Gene Mutation Database (HGMD): 2003 update. Hum. Mutat. 2003;21:577–581. doi: 10.1002/humu.10212. [DOI] [PubMed] [Google Scholar]
- 61.Naik A., Sihota R., Mahalingam K., Angmo D., Dada T., Kumar A., Kumar A., Gupta A. Evaluation of visual field changes with retinal nerve fiber layer thickness in primary congenital glaucoma. Indian J. Ophthalmol. 2022;70:3556–3561. doi: 10.4103/ijo.IJO_396_22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Alvarez-Falcón S., Glaser T., Go M.S., Kelly M.P., Chen X., Freedman S.F., El-Dairi M. Retinal injury identified by overhead-mounted optical coherence tomography in two young children with infantile-onset glaucoma. J. AAPOS. 2023;27:28.e21–28.e26. doi: 10.1016/j.jaapos.2022.11.010. [DOI] [PubMed] [Google Scholar]
- 63.Sen J., Harpavat S., Peters M.A., Cepko C.L. Retinoic acid regulates the expression of dorsoventral topographic guidance molecules in the chick retina. Development. 2005;132:5147–5159. doi: 10.1242/dev.02100. [DOI] [PubMed] [Google Scholar]
- 64.Chakrabarti S., Kaur K., Kaur I., Mandal A.K., Parikh R.S., Thomas R., Majumder P.P. Globally, CYP1B1 Mutations in Primary Congenital Glaucoma Are Strongly Structured by Geographic and Haplotype Backgrounds. Invest. Opthalmol. Vis. Sci. 2006;47:43–47. doi: 10.1167/iovs.05-0912. [DOI] [PubMed] [Google Scholar]
- 65.Gibbs R.A., Boerwinkle E., Doddapaneni H., Han Y., Korchina V., Kovar C., Lee S., Muzny D., Reid J.G., Zhu Y., et al. A global references for human genetic variation. Nature. 2015;526:68–74. doi: 10.1038/nature15393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chen S., Francioli L.C., Goodrich J.K., Collins R.L., Kanai M., Wang Q., Alföldi J., Watts N.A., Vittal C., Gauthier L.D., et al. A genomic mutational constraint map using variation in 76,156 human genomes. Nature. 2024;625:92–100. doi: 10.1038/s41586-023-06045-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.All of Us Research Program I. Denny J.C., Rutter J.L., Goldstein D.B., Philippakis A., Smoller J.W., Jenkins G., Dishman E. The “All of Us” Research Program. N. Engl. J. Med. 2019;381:668–676. doi: 10.1056/NEJMsr1809937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.IIoannidis N.M., Rothstein J.H., Pejaver V., Middha S., McDonnell S.K., Baheti S., Musolf A., Li Q., Holzinger E., Karyadi D., et al. REVEL: An Ensemble Method for Predicting the Pathogenicity of Rare Missense Variants. Am. J. Hum. Genet. 2016;99:877–885. doi: 10.1016/j.ajhg.2016.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Barrett J.C., Fry B., Maller J.D.M.J., Daly M.J. Haploview: Analysis and visualization of LD and haplotype maps. Bioinformatics. 2005;21:263–265. doi: 10.1093/bioinformatics/bth457. [DOI] [PubMed] [Google Scholar]
- 70.Larkin M.A., Blackshields G., Brown N.P., Chenna R., McGettigan P.A., McWilliam H., Valentin F., Wallace I.M., Wilm A., Lopez R., et al. Clustal W and Clustal X version 2.0. Bioinformatics. 2007;23:2947–2948. doi: 10.1093/bioinformatics/btm404. [DOI] [PubMed] [Google Scholar]
- 71.Waterhouse A.M., Procter J.B., Martin D.M., Clamp M., Barton G.J. Jalview Version 2--a multiple sequence alignment editor and analysis workbench. Bioinformatics. 2009;25:1189–1191. doi: 10.1093/bioinformatics/btp033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Shannon P., Markiel A., Ozier O., Baliga N.S., Wang J.T., Ramage D., Amin N., Schwikowski B., Ideker T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003;13:2498–2504. doi: 10.1101/gr.1239303. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data are provided in the text and Supplementary Materials of this manuscript.