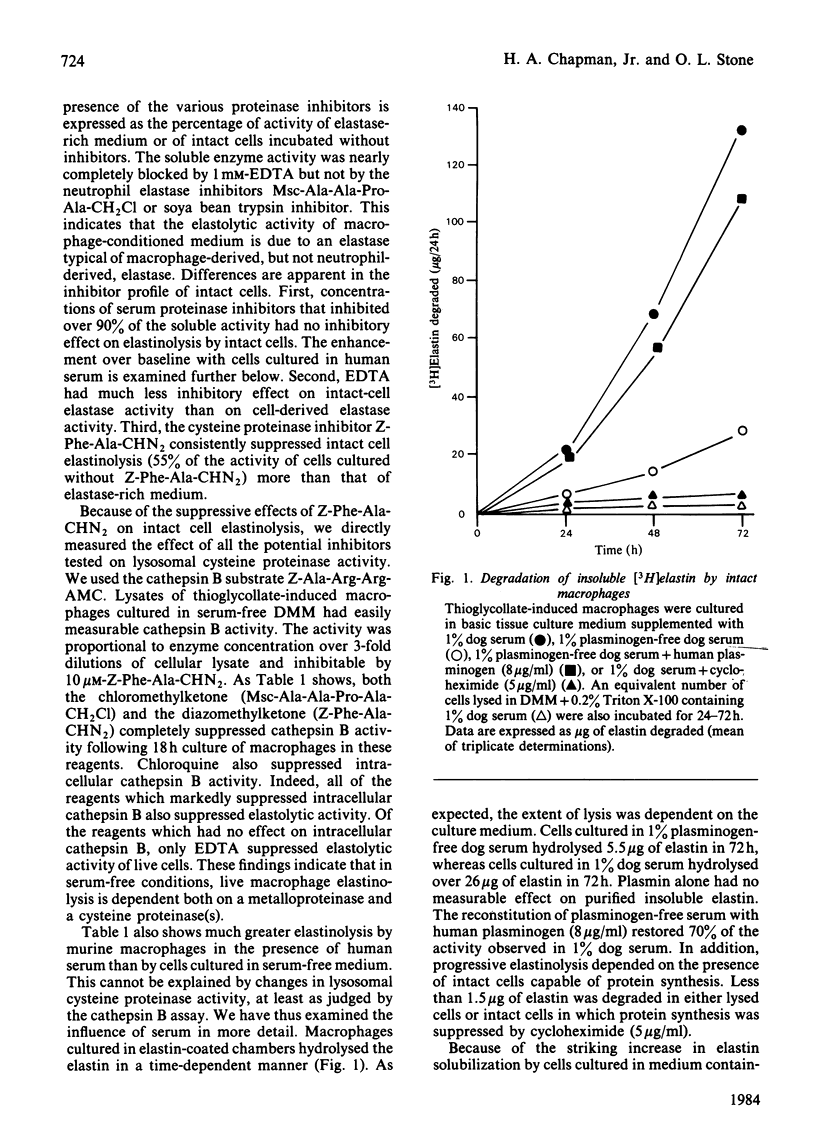
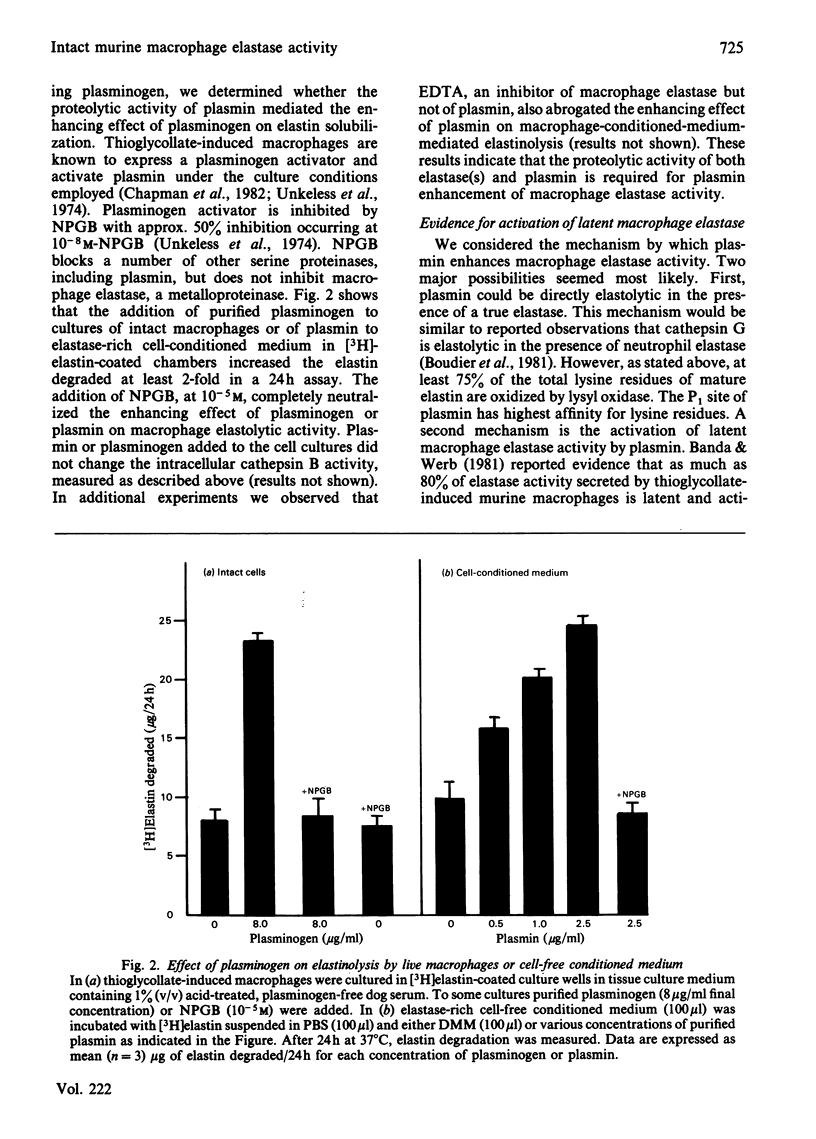
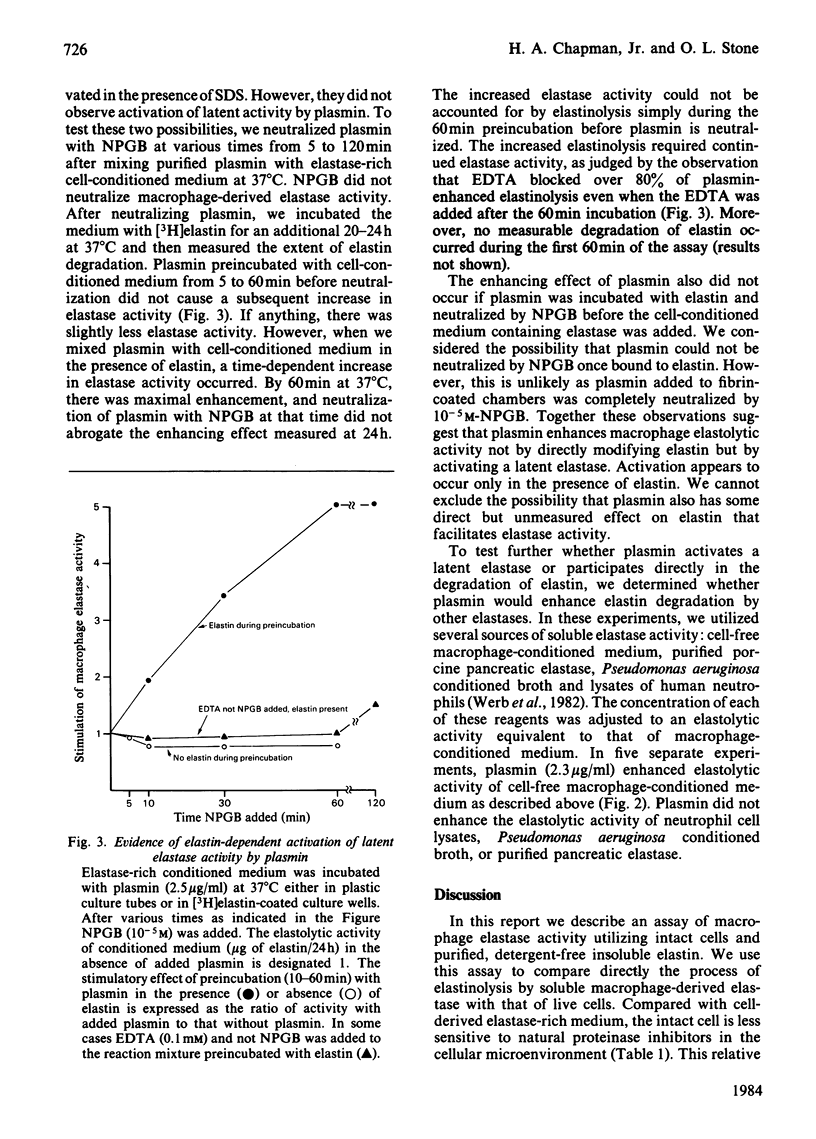
Abstract
Intact, thioglycollate-stimulated murine macrophages cultured on an insoluble [3H]-elastin substratum progressively hydrolysed the elastin. Cell lysates had little activity. We compared the effect of various proteinase inhibitors on elastinolysis by either live cells or cell-free, elastase-rich conditioned medium. Only known inhibitors of macrophage elastase blocked the activity of elastase-rich cell-conditioned medium whereas inhibitors of cathepsin B also suppressed intact cell activity. Serum proteinase inhibitors blocked cell-derived soluble elastase activity but not intact cell elastolytic activity. We also observed that plasminogen added to the cell cultures markedly increased elastinolysis by live macrophages or cell-free elastase-rich medium. Purified plasmin alone had no measurable effect on native elastin. Additional experiments indicated that the plasmin enhancement was due to elastin-dependent activation of latent macrophage elastase. These results indicate that live macrophage elastinolysis is a co-operative process involving multiple proteinases, especially a cysteine proteinase(s) and elastase. Plasmin may be a physiological activator of latent macrophage elastase.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Banda M. J., Werb Z. Mouse macrophage elastase. Purification and characterization as a metalloproteinase. Biochem J. 1981 Feb 1;193(2):589–605. doi: 10.1042/bj1930589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrett A. J., Kirschke H. Cathepsin B, Cathepsin H, and cathepsin L. Methods Enzymol. 1981;80(Pt 100):535–561. doi: 10.1016/s0076-6879(81)80043-2. [DOI] [PubMed] [Google Scholar]
- Boudier C., Holle C., Bieth J. G. Stimulation of the elastolytic activity of leukocyte elastase by leukocyte cathepsin G. J Biol Chem. 1981 Oct 25;256(20):10256–10258. [PubMed] [Google Scholar]
- Brown C. R., Hartree E. F. Effects of acrosin inhibitors on the soluble and membrane-bound forms of ram acrosin, and a reappraisal of the role of the enzyme in fertilization. Hoppe Seylers Z Physiol Chem. 1976 Jan;357(1):57–65. doi: 10.1515/bchm2.1976.357.1.57. [DOI] [PubMed] [Google Scholar]
- Chapman H. A., Jr, Stone O. L., Vavrin Z. Degradation of fibrin and elastin by intact human alveolar macrophages in vitro. Characterization of a plasminogen activator and its role in matrix degradation. J Clin Invest. 1984 Mar;73(3):806–815. doi: 10.1172/JCI111275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman H. A., Jr, Vavrin Z., Hibbs J. B., Jr Macrophage fibrinolytic activity: identification of two pathways of plasmin formation by intact cells and of a plasminogen activator inhibitor. Cell. 1982 Mar;28(3):653–662. doi: 10.1016/0092-8674(82)90220-3. [DOI] [PubMed] [Google Scholar]
- Eeckhout Y., Vaes G. Further studies on the activation of procollagenase, the latent precursor of bone collagenase. Effects of lysosomal cathepsin B, plasmin and kallikrein, and spontaneous activation. Biochem J. 1977 Jul 15;166(1):21–31. doi: 10.1042/bj1660021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones P. A., Scott-Burden T. Activated macrophages digest the extracellular matrix proteins produced by cultured cells. Biochem Biophys Res Commun. 1979 Jan 15;86(1):71–77. doi: 10.1016/0006-291x(79)90383-8. [DOI] [PubMed] [Google Scholar]
- Jones P. A., Werb Z. Degradation of connective tissue matrices by macrophages. II. Influence of matrix composition on proteolysis of glycoproteins, elastin, and collagen by macrophages in culture. J Exp Med. 1980 Dec 1;152(6):1527–1536. doi: 10.1084/jem.152.6.1527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones P. A., Werb Z. Degradation of connective tissue matrices by macrophages. II. Influence of matrix composition on proteolysis of glycoproteins, elastin, and collagen by macrophages in culture. J Exp Med. 1980 Dec 1;152(6):1527–1536. doi: 10.1084/jem.152.6.1527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kagan H. M., Crombie G. D., Jordan R. E., Lewis W., Franzblau C. Proteolysis of elastin-ligand complexes. Stimulation of elastase digestion of insoluble elastin by sodium dodecyl sulfate. Biochemistry. 1972 Aug 29;11(18):3412–3418. doi: 10.1021/bi00768a014. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MOSESSON M. W. The preparation of human fibrinogen free of plasminogen. Biochim Biophys Acta. 1962 Feb 26;57:204–213. doi: 10.1016/0006-3002(62)91112-5. [DOI] [PubMed] [Google Scholar]
- Reilly C. F., Travis J. The degradation of human lung elastin by neutrophil proteinases. Biochim Biophys Acta. 1980 Jan 24;621(1):147–157. doi: 10.1016/0005-2795(80)90070-7. [DOI] [PubMed] [Google Scholar]
- Riches D. W., Morris C. J., Stanworth D. R. Induction of selective acid hydrolase release from mouse macrophages during exposure to chloroquine and quinine. Biochem Pharmacol. 1981 Mar 15;30(6):629–634. doi: 10.1016/0006-2952(81)90136-2. [DOI] [PubMed] [Google Scholar]
- Shaw E., Dean R. T. The inhibition of macrophage protein turnover by a selective inhibitor of thiol proteinases. Biochem J. 1980 Feb 15;186(2):385–390. doi: 10.1042/bj1860385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi S., Seifter S., Yang F. C. A new radioactive assay for enzymes with elastolytic activity using reduced tritiated elastin. The effect of sodium dodecyl sulfate on elastolysis. Biochim Biophys Acta. 1973 Nov 15;327(1):138–145. doi: 10.1016/0005-2744(73)90111-3. [DOI] [PubMed] [Google Scholar]
- Unkeless J. C., Gordon S., Reich E. Secretion of plasminogen activator by stimulated macrophages. J Exp Med. 1974 Apr 1;139(4):834–850. doi: 10.1084/jem.139.4.834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walsh P. N. Platelets and coagulation proteins. Fed Proc. 1981 May 15;40(7):2086–2091. [PubMed] [Google Scholar]
- Werb Z., Bainton D. F., Jones P. A. Degradation of connective tissue matrices by macrophages. III. Morphological and biochemical studies on extracellular, pericellular, and intracellular events in matrix proteolysis by macrophages in culture. J Exp Med. 1980 Dec 1;152(6):1537–1553. doi: 10.1084/jem.152.6.1537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werb Z., Gordon S. Elastase secretion by stimulated macrophages. Characterization and regulation. J Exp Med. 1975 Aug 1;142(2):361–377. doi: 10.1084/jem.142.2.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werb Z., Mainardi C. L., Vater C. A., Harris E. D., Jr Endogenous activiation of latent collagenase by rheumatoid synovial cells. Evidence for a role of plasminogen activator. N Engl J Med. 1977 May 5;296(18):1017–1023. doi: 10.1056/NEJM197705052961801. [DOI] [PubMed] [Google Scholar]
- Wiman B., Collen D. Molecular mechanism of physiological fibrinolysis. Nature. 1978 Apr 6;272(5653):549–550. doi: 10.1038/272549a0. [DOI] [PubMed] [Google Scholar]


