Abstract
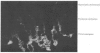
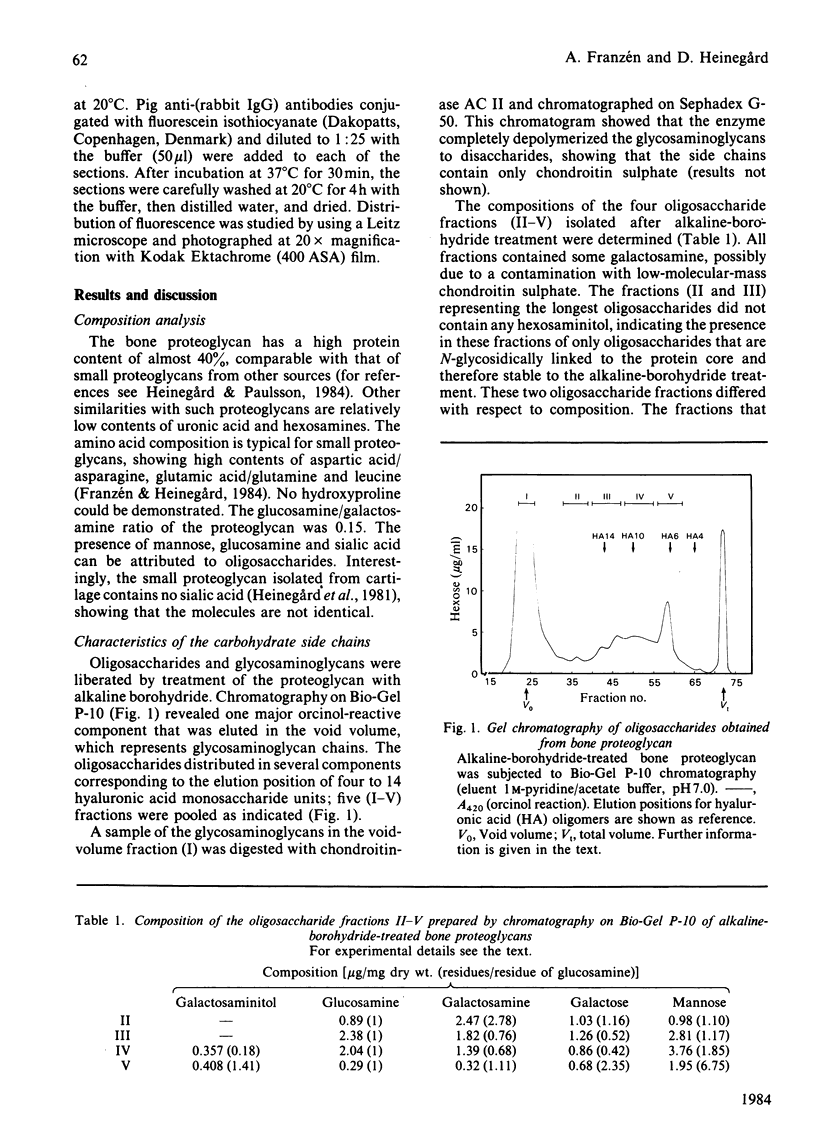
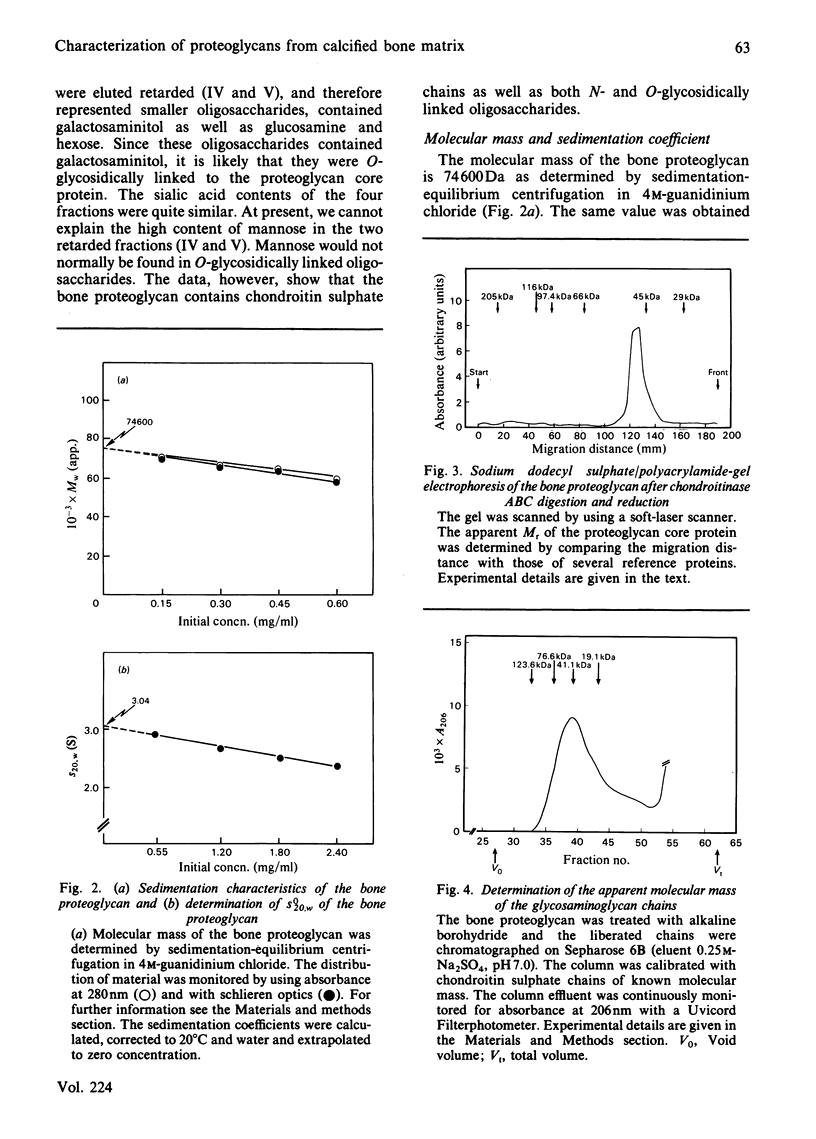
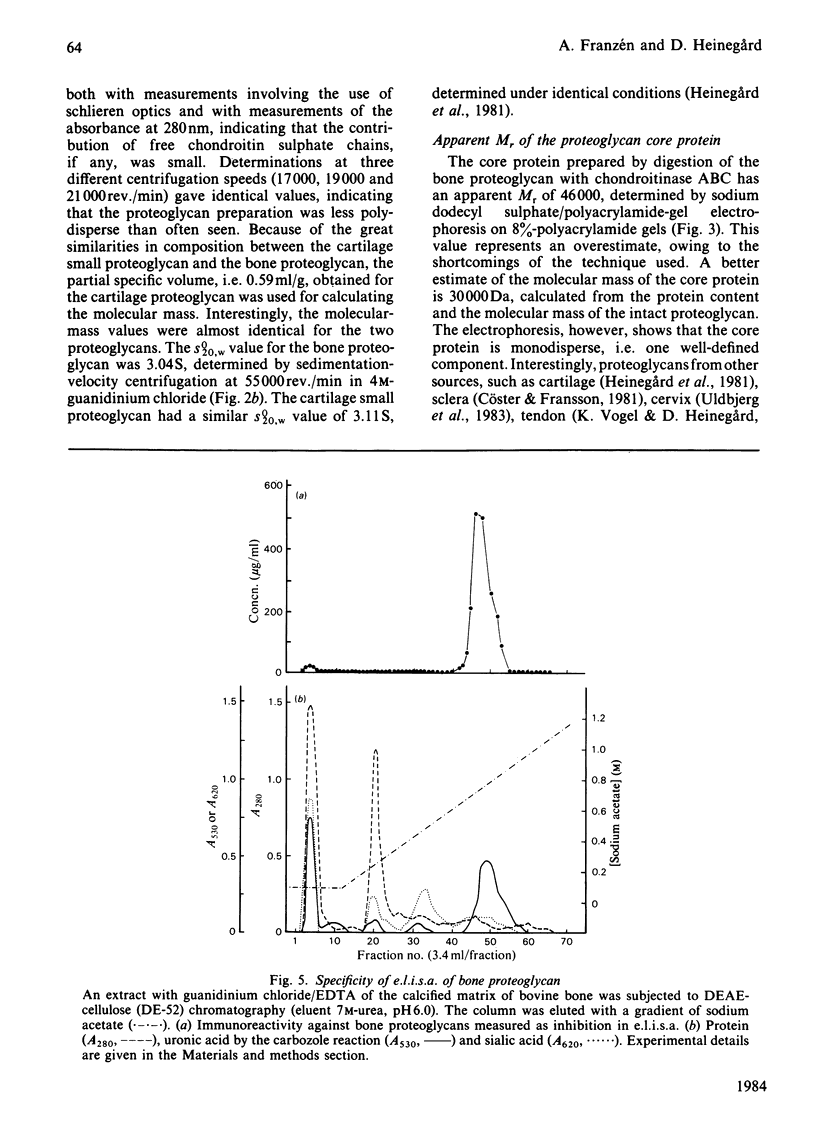
The proteoglycans characterized were those isolated from the calcified matrix of mature bovine bone [Franzén & Heinegård (1984) Biochem. J. 224, 47-58]. The average molecular mass of the bone proteoglycan is 74 600 Da, determined by sedimentation-equilibrium centrifugation in 4M-guanidinium chloride. Its sedimentation coefficient (s0(20),w) is 3.04 S. The apparent Mr of its core protein is 46 000, estimated by sodium dodecyl sulphate/polyacrylamide-gel electrophoresis of the chondroitinase ABC-digested proteoglycan. A more likely molecular mass of the core protein is 30 000 Da, as calculated from the molecular mass and the protein content (40%) of the proteoglycan. The bone proteoglycan contains one or probably two chondroitin sulphate chains each with a molecular mass (weight-average) of 33 700 Da and several oligosaccharides both of the N-glycosidically and the O-glycosidically linked type. Antibodies against the homogeneous bone proteoglycans were raised in rabbits. An e.l.i.s.a. (enzyme-linked immunosorbent assay) method was developed that allowed specific quantification of bone proteoglycans at nanogram levels. The specificity of the antibodies was tested by using the e.l.i.s.a. method. The bone proteoglycan showed partial cross-reactivity with the small proteoglycan of cartilage. The antibodies were used to localize immunoreactivity of bone proteoglycans by indirect immunofluorescence in frozen sections of foetal bovine epiphysial growth plate. The fluorescence was entirely found in the primary spongiosa, and no fluorescence was found among the hypertrophied chondrocytes or in the region of provisional calcification.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrews A. T., Herring G. M., Kent P. W. Some studies on the composition of bovine cortical-bone sialoprotein. Biochem J. 1967 Sep;104(3):705–715. doi: 10.1042/bj1040705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Axelsson I., Heinegård D. Characterization of the keratan sulphate proteoglycans from bovine corneal stroma. Biochem J. 1978 Mar 1;169(3):517–530. doi: 10.1042/bj1690517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlson D. M. Structures and immunochemical properties of oligosaccharides isolated from pig submaxillary mucins. J Biol Chem. 1968 Feb 10;243(3):616–626. [PubMed] [Google Scholar]
- Franzén A., Heinegård D. Extraction and purification of proteoglycans from mature bovine bone. Biochem J. 1984 Nov 15;224(1):47–58. doi: 10.1042/bj2240047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hascall V. C., Heinegård D. Aggregation of cartilage proteoglycans. II. Oligosaccharide competitors of the proteoglycan-hyaluronic acid interaction. J Biol Chem. 1974 Jul 10;249(13):4242–4249. [PubMed] [Google Scholar]
- Heinegård D., Paulsson M., Inerot S., Carlström C. A novel low-molecular weight chondroitin sulphate proteoglycan isolated from cartilage. Biochem J. 1981 Aug 1;197(2):355–366. doi: 10.1042/bj1970355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inerot S., Heinegård D. Bovine tracheal cartilage proteoglycans. Variations in structure and composition with age. Coll Relat Res. 1983 May;3(3):245–262. doi: 10.1016/s0174-173x(83)80007-7. [DOI] [PubMed] [Google Scholar]
- Jourdian G. W., Dean L., Roseman S. The sialic acids. XI. A periodate-resorcinol method for the quantitative estimation of free sialic acids and their glycosides. J Biol Chem. 1971 Jan 25;246(2):430–435. [PubMed] [Google Scholar]
- Lohmander L. S., De Luca S., Nilsson B., Hascall V. C., Caputo C. B., Kimura J. H., Heinegard D. Oligosaccharides on proteoglycans from the swarm rat chondrosarcoma. J Biol Chem. 1980 Jul 10;255(13):6084–6091. [PubMed] [Google Scholar]
- Neville D. M., Jr Molecular weight determination of protein-dodecyl sulfate complexes by gel electrophoresis in a discontinuous buffer system. J Biol Chem. 1971 Oct 25;246(20):6328–6334. [PubMed] [Google Scholar]
- Stegemann H., Stalder K. Determination of hydroxyproline. Clin Chim Acta. 1967 Nov;18(2):267–273. doi: 10.1016/0009-8981(67)90167-2. [DOI] [PubMed] [Google Scholar]
- Uldbjerg N., Malmström A., Ekman G., Sheehan J., Ulmsten U., Wingerup L. Isolation and characterization of dermatan sulphate proteoglycan from human uterine cervix. Biochem J. 1983 Feb 1;209(2):497–503. doi: 10.1042/bj2090497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wasteson A. A method for the determination of the molecular weight and molecular-weight distribution of chondroitin sulphate. J Chromatogr. 1971 Jul 8;59(1):87–97. doi: 10.1016/s0021-9673(01)80009-1. [DOI] [PubMed] [Google Scholar]
- YPHANTIS D. A. EQUILIBRIUM ULTRACENTRIFUGATION OF DILUTE SOLUTIONS. Biochemistry. 1964 Mar;3:297–317. doi: 10.1021/bi00891a003. [DOI] [PubMed] [Google Scholar]