Abstract
Objective:
Abdominal aortic aneurysm (AAA) is an aneurysm-like dilated and highly fatal cardiovascular disease. CD8+ T cells have been shown to be critical for vascular pathological processes, but the contribution of these lymphocytes to vascular diseases remains elusive.
Methods and results:
Eight-week-old male wildtype (CD8+/+) and Cd8a knockout (CD8−/−) mice were used in a calcium chloride2 (CaCl2)-induced experimental AAA model. At 6 weeks after surgery, CD8+ T-cell deletion prevented the formation of AAA, accompanied by reductions of the levels of inflammatory (interferon-γ [IFN-γ], interleukin-1β, monocyte chemoattractant protein-1, intracellular adhesion molecule-1, vascular cell adhesion molecule-1, NOD-like receptor protein 3, caspase-1), oxidative stress [NADPH oxidase and gp91phox], and proteolysis (cathepsin S, cathepsin K, matrix metalloproteinase-2 [MMP-2] and MMP-9) proteins and/or genes in plasma and/or AAA tissues. Immunoreactivities of MMP-2 and MMP-9 were observed in macrophages. An injection of IFN-γ and adoptive transfer of CD8+ T cells of IFN-γ+/+ mice diminished CD8−/−-mediated vasculoprotective actions in the AAA mice. In vitro, IFN-γ enhanced MMP-2 and MMP-9 gelatinolytic activities in macrophage and/or vascular smooth muscle cells.
Conclusion:
The vasculoprotective effects of CD8+ T-cell deletion in a mouse CaCl2-induced AAA model were likely attributable to, at least in part, the attenuation of IFN-γ-dependent inflammation action, oxidative stress production, and proteolysis, suggesting a novel therapeutic target for AAA formation by regulating CD8+ T-cell-derived IFN-γ secretion.
Keywords: abdominal aortic aneurysm, CD8+ T-cell, inflammasome, interferon-γ, macrophage
INTRODUCTION
An abdominal aortic aneurysm (AAA) is a commonly permanent vasodilatory disease that is associated with high mortality [1,2]. The histological features of AAAs include vascular smooth muscle cell (SMC) apoptosis and elastin disruption, as well as chronic adventitial and medial inflammatory cell infiltration [3]. For the treatment of AAA, the aneurysm is resected and a synthetic graft replaces the affected vessel [4]. Despite the progress achieved in surgical treatments, AAA still presents a high mortality rate, and more nonsurgical solutions for the treatment of AAA are desired.
CD8+ T cells, also known as cytotoxic T lymphocytes, are generated in the bone marrow and are dependent on a specialized thymus microenvironment for their development and maturation, where they undergo a series of differentiation and development steps to become mature T cells, which subsequently migrate to peripheral lymphoid tissues and organs and proliferate and differentiate into effector T cells upon stimulation with appropriate antigens [5]. CD8+ T cells directly or indirectly kill target cells by secreting cytotoxic granules, fas-ligand, and cytokines such as tumor necrosis factor-α (TNF-α) and interferon-γ (IFN-γ), which induce apoptosis and inflammation [6,7]. CD8+ T cells have been shown to play an important role not only in various autoimmune diseases, including type 1 diabetes, multiple sclerosis, systemic lupus erythematosus, and aplastic anemia; they are also closely related to diseases of the cardiovascular system such as atherosclerosis, hypertension, and AAA [6–9]. Clinical study showed that AAA patients had increased levels of CD8+ T cells as well as serum TNF-α and interleukin-6 (IL-6) levels as compared to control group, whereas the levels of CD4+ T cells and CD4+/CD8+ were decreased in AAA patients [10]. Zhou et al. demonstrated that CD8+ T cells potentially contribute to elastase-induced murine AAA formation. However, the precise role(s) of CD8+ T cell in the initiation and progression of AAA largely uncertain.
Macrophages are one of the most important types of inflammatory cells in the process of AAA formation. Macrophages of two different phenotypes, that is, the M1 phenotype which is pro-inflammation and the M2 phenotype, which is antiinflammation, accumulate in the adventitia of injured arteries and regulate aneurysm formation, influencing the secretion of proinflammatory and anti-inflammatory cytokines by regulating the ratio of M1 to M2 macrophages [11–13]. An investigation conducted in 2020 revealed that macrophages can release several proteinases (including matrix metalloproteinases [MMPs] and cathepsins), leading to the promotion of aortic elastic breakage and AAA formation [13]. In mice, a CD43-mediated production of IFN-γ by CD8+ T cells promoted the formation of AAA [14]. There is limited information regarding how the interaction of macrophages and CD8+ T cells contributes to aortic extracellular metabolism and AAA formation, even in mice.
We conducted the present study to investigate the role(s) of CD8+ T cells in the development of calcium chloride (CaCl2)-induced AAAs in CD8−/− mice. In one experiment, CD8−/− mice that had been subjected to CaCl2-induced AAA surgery were injected with IFN-γ prior to morphological and biological evaluations. In an in-vitro experiment, a mouse macrophage cell line (RAW 264.7 cells) was treated with different concentrations of IFN-γ for 24 h and then subjected to biological assays.
MATERIALS AND METHODS
Animals
Six-week-old male C57BL/6 mice were purchased from the Experimental Animal Research Center of Yanbian University (Yanji, Jilin PR, China) and were used as wild-type controls (CD8+/+: wild-type [WT]). Male CD8−/− (Cd8a knockout [KO]) and IFN-γ−/− (IFN-γ knockout [KO]) mice were purchased from Shanghai Biomodel Organism Science & Technology Development Co. (Shanghai, China; Protocol no. 2021-W5–2174) and were bred for the preparation of male CD8−/− mice and male IFN-γ−/− of the same age. The mice of both genotypes (20–25 g weight) were provided a standard diet and tap water ad libitum and maintained in a 22°C room with a 12-h light/dark cycle for 2 weeks prior to the experiments. All of the animal protocols were approved by the Animal Care Committee of the Ethics Committee on Animal Research at Yanbian University (protocol nos.: YD20-1128006 and YD20240044) and conducted in accord with the care guidelines of those committees. The mice were monitored throughout the experimental period, and all of experimental protocols involving the mice were performed by trained research staff.
Mouse abdominal aortic aneurysm model
All the procedures were performed in mice anaesthetized with isoflurane (3–5% in an induction chamber and between 0.5 and 2% for maintenance, depending on the procedure and its duration). Eight-week-old male CD8−/− and CD8+/+ mice were placed under anesthesia; the abdominal aorta between the renal and iliac arteries was exposed and isolated from the surrounding peritoneal structures as described [15]. A small piece of gauze soaked in 0.5 mol/l CaCl2 was applied perivascularly for 20 min. A piece of gauze with NaCl (0.9%) was used for the sham operation in the control CD8−/− and CD8+/+ mice. In some of the experiments, CD8−/− mice were intraperitoneally (i.p.) administered recombinant murine IFN-γ (rmIFN-γ) (50 000 U/mouse, P1015; APExBIO Technology, Houston, Texas, USA) or the same volume of PBS 2 × /week beginning on the day of aneurysm induction for 3 weeks (total six times). In addition, CD8−/−mice were also tail intravenously injected with 1 × 107 CD8+ T cells isolated from IFN-γ+/+ or IFN-γ−/− mice via the tail vein at day 3 and 7 after AAA induction (see adoptive transfer of CD8+ T cells’ method). In all of the experiments, the mice were sacrificed at 6 weeks after the surgery for the collection of blood samples and the aortas for subsequent analyses.
Histology and microscopy
The abdominal aorta of each mouse was fixed in 4% paraformaldehyde and embedded in Tissue-Tek O.C.T. Compound (Sakura, Finetechnical, Tokyo), and the samples were prepared into 5-μm sections for the following observations. The frozen sections were stained with a hematoxylin and eosin (H&E) kit, a Masson trichrome kit (Solarbio Life Sciences, Beijing, China), and an Elastica van Gieson (EVG) kit (Abcam, Cambridge, Massachusetts, USA) for the evaluations of morphological changes and the contents of collagen and elastin. The grades for elastin degradation were evaluated as described [16]. No elastin degradation or mild elastin degradation, 1 point; moderate, 2 points; moderate to severe, 3 points; and severe elastin degradation, 4 points as described [17]. For the quantification of the elastin disruption degree, we took four to five images of each vessel (100 × magnification), and we counted the elastin score and collagen deposition area and averaged the value for each mouse.
Immunofluorescence
After undergoing blocking with 5% goat serum to reduce nonspecific reactions, frozen 5-μm thick sections of abdominal arteries were incubated with PE mouse anti-CD8a (1 : 100, 553032, BD Pharmingen, San Diego, California, USA), anti-MMP-2 (1 : 100, sc-13594, Santa Cruz Biotechnology, Santa Cruz, California, USA), anti-MMP-9 (1 : 100, sc-393859, Santa Cruz) and rabbit anti-IFN-γ (1 : 100, cat. no. 8455, CST, Cell Signaling Technology Beverly, Massachusetts, USA), anti-CD68 (1 : 100, bs-0649R, Bioss, Woburn, Massachusetts, USA), respectively, followed by incubation with secondary antibody against mouse IgG (1;200, Abcam) or rabbit (1 : 200, APExBIO Technology), respectively. A drop of antifluorescence quenching sealing solution with DAPI (Beyotime, Shanghai, China) was added for the visualization of the cell nuclei. Signals were observed using an Evos microscope (Invitrogen, Carlsbad, California) and analyzed using Image J software. For the quantification of positive cell staining, we took five images of each section using a 40 × objective, and we counted the numbers of CD68+ cells and averaged the numbers for each mouse.
Real-time quantitative gene assay
Total RNA was isolated from the lysates of the aortic tissues or cells following the Trizol protocol (Invitrogen) and then reverse-transcribed to cDNA with a reverse transcriptase kit (ZOMANBIO, Beijing, China). The resulting cDNA was used for a real-time quantitative PCR (qPCR) using an ABI 7300 real-time PCR system (Applied Biosystems, Foster City, California, USA) with primers specific for MMP-2, MMP-9, cathepsin K (CatK), CatS, gp91phox, monocyte chemotaxis protein-1 (MCP-1), intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), Collagen I, Collagen III, interleukin-1β (IL-1β), and NOD-like receptor protein 3 (NLRP3). The quantitative comparison of mRNA levels was normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA levels. The primer sequences are listed in Supplemental Table S1.
Western blot analysis
Total proteins were extracted from the aortic tissues from the different mouse groups, and the cultured macrophages were lysed in RIPA buffer including protease inhibitor and phosphatase inhibitor as described [18]. Protein concentrations were determined by a BCA protein quantification kit (Solarbio Life Sciences). A total of 40 μg of protein was loaded and separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene fluoride (PVDF) membranes. The membranes were blocked with 5% skimmed milk or 5% bovine serum albumin (BSA) for 1 h, then incubated overnight with primary antibodies against the following: NLRP3 (cat. no. 15101), caspase-1 (cat. no. 24232), and β-actin (cat. no. 4970) (CST, MA; 1 : 1000); gp91phox (cat. no. 611415, BD Transduction Laboratories, San Jose, California, USA; 1 : 1000), collagen I (cat. no. ab260043), and collagen III (cat. no. ab184993) (Abcam; 1 : 1000). The membranes were then treated with the related secondary antibodies at a 1 : 5000 to 1 : 10 000 dilution for 2 h at room temperature. The protein bands were visualized using chemiluminescent substrates (Merck Millipore, Darmstadt, Germany). Protein levels quantitated from western blots were normalized by loading internal controls.
Gelatin zymography
Protein extracts from abdominal aortic and conditional media from macrophages and aortic SMCs treated with or without IFN-γ were added to SDS sample buffer with nonreducing agent and loaded onto a 10% SDS-polyacrylamide gel containing 1 mg/ml gelatin, as described [19]. After electrophoresis, the gels were washed twice with 2.5% Triton X-100 and then incubated in incubation buffer (1% Triton X-100, 50 mmol/l Tris-HCl, and 5 mol/l CaCl2, 1 μmol/l ZnCl2 pH 7.5) at 37°C overnight. The gel was stained with Coomassie Brilliant Blue for 30–60 min and then incubated with destaining solution until bands could clearly be seen.
ELISA and nitric oxide release measurement
The levels of serum and cell supernatant IL-1β and (or) IFN-γ were examined with the use of ELISA kits (MLBIO, Shanghai, China) according to the manufacturer's instruction. The release of nitric oxide from macrophages was analyzed using a nitric oxide detection kit (Beyotime).
NADPH oxidase dependent superoxide production assay
Fresh abdominal aortas were lysed in lysis buffer (20 mmol/l Tris-HCl, 150 mmol/l NaCl, 1 mmol EDTA, 1 mmol/l EGTA, and 1% Triton X-100; pH 7.5). Using a lucigenin-based enhanced chemiluminescence assay, we measured the superoxide (O2−) production of the obtained homogenates of aorta, as described [20]. In brief, 1 ml of prepared homogenate protein was transferred to an assay tube, and NADPH and dark-adapted lucigenin were added to final concentrations of 100 and 5 μmol/l, respectively, immediately before the measurement of chemiluminescence. The chemiluminescence signal was sampled every 60 s for 12 min with a tube luminometer (20/20; Tuner Designs, Sunnyvale, California, USA), and the respective background counts were subtracted from the experimental values.
Isolation and infusion of CD8+ T cells
CD8+ T cells were isolated from the spleens of male IFN-γ+/+ and IFN-γ−/− mice by CD8a+ T Cell Isolation Kits (#130-104-075, Miltenyi Biotech, Bergisch Gladbach, Germany) according to the manufacturer's instructions. Cell purity was consistently more than 95%. Following resuspending of CD8a+ T Cells in 100 μl PBS, the CD8−/−mice were received tail intravenous injection (1 × 107 cells/mouse) at day 3 and 7 after AAA surgery.
Cell culture and IFN-γ treatment
The mouse aortic SMC line and the macrophage cell line (RAW 264.7) were purchased from Fuheng Biotechnology Co. (Shanghai, China) and cultured and propagated in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum (FBS). SMCs were plated on six-well plates at 2 × 105 cells/well. After growing to 80% confluence, the cells were serum-starved for 24 h and then treated with or without rmIFN-γ at 0, 1, or 10 ng/ml in DMEM. Two hours after seeding at 4 × 105 cells/six-well plates, macrophages were rinsed to remove nonadherent cells. The cells were then treated with or without rmIFN-γ at 0, 1, or 10 ng/ml in DMEM. After 24 h of incubation, all conditioned media were collected and centrifuged to eliminate dead cells and debris for the gelatin zymography assay. The macrophages were subjected to western blotting for the examination of related proteins and the levels of macrophages supernatant IL-1β were examined for ELISA.
Statistical analyses
All data are expressed as the mean ± standard error of the mean (SEM). The data were subjected to the statistical analyses after the distribution status of the test data was determined. We performed a one-way analysis of variance (ANOVA) for comparisons of multiple groups, followed by Tukey's posthoc test or Student's t-test for comparisons of two independent sample groups (with GraphPad Prism Version 8.0.2). The AAA morphological analyses were evaluated by two observers in a blind manner, and the values they obtained were averaged. A P value less than 0.05 was considered significant.
RESULTS
Characteristics of the CaCl2-induced abdominal aortic aneurysms in the mice
The CaCl2 model has been widely used to study AAA mechanisms and drug screening. In this study, to investigate the role(s) of CD8+ T cells in the development of AAA, we used CaCl2 to induce AAAs in CD8+/+ and CD8−/− mice (Suppl. Figure S1A). We observed that the peri-aortic application of CaCl2 resulted in an increased external aortic diameter of the abdominal aorta compared to the control and led to the formation of AAA in the CD8+/+ mice (Suppl. Figure S1B–D). The AAA CD8+/+ mice had increased levels of serum IFN-γ (Suppl. Figure S1E). The qPCR data showed that the CaCl2 induction also resulted in increases in the levels of the inflammation- (MCP-1, ICAM-1, VCAM-1, NLRP3, IL-1β), oxidative stress (gp91phox), proteolysis- (CatS, CatK, MMP-2, and MMP-9), and extracellular matrix (ECM) protein (Collagen I and Collagen III)-related genes compared to the control non-AAA CD8+/+ mice (Suppl. Figure S2).
CD8+ T-cell deletion mitigated CaCl2-induced abdominal aortic aneurysm formation in vivo
As shown in Fig. 1a,b, CD8+ T-cell deletion decreased the external aortic diameter of AAA mice compared to the AAA CD8+/+ mice, whereas there was no difference in AAA size between the two genotypes of mice treated with NaCl. CD8+ T-cell deletion also lowered the levels of the targeted proteins (NLRP3, caspase-1, gp91phox, collagen I, and collagen III) (Fig. 1c–f). As anticipated, the ELISA data yielded the same conclusions regarding the levels of IFN-γ and IL-1β (Fig. 1g,h). At 6 weeks after the CaCl2 induction, H&E staining showed less histopathological damage in the AAA CD8−/− mice compared to the CD8+/+ mice (Fig. 2a). The quantitative data of the Masson and EVG staining analyses revealed that the AAA lesions from the CD8−/− mice had not only significantly lower aortic wall elastin fragmentation grades but also lower collagen degradable areas compared to those from the CD8+/+ mice (Fig. 2b,c). We have observed that the abundance of IFN-γ+/CD8a+ T cells in the adventitia of the AAA CD8+/+ aortas but not in AAA CD8−/− aortas (Fig. 2d and Suppl. Figure S3D). Immunoreactivities of MMP-2 and MMP-9 were observed in macrophages (Fig. 2e,f). CD8 T-cell deletion reduced MMP-2+/CD68+ and MMP-9+/CD68+ macrophage infiltration in the adventitia of the mouse AAA aortas (Fig. 2e,f and Suppl. Figure S3E,F). Consistently, CD8+ T-cell deficiency inhibited the aortic tissue NADPH oxidase activity of the AAA mice (Suppl. Figure S3A). The gelatin zymography revealed that CD8 T-cell deletion had mitigated the gelatinolytic activities of MMP-2 and MMP-9 (Suppl. Figure S3B,C). Moreover, the qPCR data demonstrated that CD8−/− resulted in a reduction in the levels of MCP-1, ICAM-1, VCAM-1, NLRP3, IL-1β, gp91phox, CatS, CatK, MMP-2, MMP-9, Collagen I, and Collagen III in the aortic tissues of the AAA mice (Suppl. Figure S4).
FIGURE 1.
CD8+ T-cell deletion mitigated the CaCl2-induced AAA formation and the investigated protein levels in aortic tissues. A,B: Representative photographs and data of the maximal external aortic diameter in CD8+/+ and CD8−/− mice treated with NaCl or CaCl2, respectively (n = 8/group). C–F: Representative western blotting images and quantitative data for the levels of NLRP3, caspase-1, gp91phox, collagen I, and collagen III proteins in four experimental groups (n = 4/group). G,H: The ELISA results showing the serum levels of IFN-γ and IL-1β in the four groups (n = 6/group).
FIGURE 2.
CD8+ T-cell deficiency alleviated the histopathological changes and recruitment of inflammatory cells. A: The histopathological analysis (H&E, EVG, and Masson trichrome staining) of abdominal aortas of CD8+/+ and CD8−/− mice treated with NaCl or CaCl2, respectively. Scale bar: upper layer 100 μm, lower layer 50 μm. B: Quantification of the collagen deposition area of the abdominal aortas (n = 5/group). C: Quantification of the elastin degradation score of the abdominal aortas (n = 5/group). D-F: Representative images of IFN-γ+/CD8a+ T cell and MMP-2+/ and MMP-9+/CD68+ macrophage in the adventitia of CD8+/+ and CD8−/− AAA mice (n = 5/group). Scale bar: 50 μm. L, Lumen.
Interferon-γ loading accelerated CaCl2-induced abdominal aortic aneurysm formation in CD8−/− mice
To understand the effect of IFN-γ on AAAs, we injected rmIFN-γ (50 000 U/mouse, 2 × /week, total six times) into AAAs that had been induced in CD8−/− mice. On operative day 42 after AAA induction, we observed that the IFN-γ loading resulted an increase in the external aortic diameter compared to the control CD8−/− mice treated with PBS (Fig. 3a,b). As anticipated, the levels of NLRP3, caspase-1, gp91phox, collagen I, and collagen III proteins were also markedly higher in the AAA CD8−/−-IFN-γ mice compared to the AAA CD8−/−-PBS mice (Fig. 3c,d). Likewise, the levels of IL-1β protein and NADPH oxidase activity were higher in the IFN-γ treatment group than in the PBS treatment group (Fig. 3e, Suppl. Figure S5A). The representative images and quantitative data of the H&E, Masson, and EVG staining demonstrated that the IFN-γ loading exerted harmful effects on the aortic damage, elastin degradation, and collagen deposition (Fig. 4a–c). Figure 4d showed that there was no IFN-γ+/CD8+ T cell staining signal in both experimental groups. We have observed that IFN-γ-loading increased the infiltration of MMP-2+/CD68+ and MMP-9+/CD68+ macrophages in the injured outer aortic wall of the AAA CD8−/− mice (Fig. 4d-f and Suppl. Figure S5D,E). The gelatin zymography revealed that the IFN-γ treatment had accelerated the gelatinolytic activities of MMP-2 and MMP-9 (Suppl. Figure S5B,C).
FIGURE 3.
IFN-γ loading accelerated the CaCl2-induced AAA formation in the CD8−/− mice. A,B: Representative photographs and quantitative data of the maximal external aortic diameter in the PBS-injected (Vehicle) or IFN-γ-injected CD8−/− mice after CaCl2 aneurysm induction (n = 8/group). C,D: Representative immunoblotting images and quantitative data for the levels of NLRP3, caspase-1, gp91phox, collagen I, and collagen III proteins in the two groups (n = 3/group). E: The ELISA results showing the serum levels of IL-1β in both groups (n = 6/group).
FIGURE 4.
IFN-γ accelerated the histopathological changes and the recruitment of inflammatory cells in the CaCl2-induced AAAs in the CD8−/− mice. A: The histopathological analysis (H&E, EVG, and Masson trichrome staining) of abdominal aortas of PBS-injected (Vehicle) and IFN-γ-injected CD8−/− mice after CaCl2 aneurysm induction. Scale bar: upper layer 100 μm, lower layer 50 μm. B: Quantification of the collagen deposition area of the abdominal aortas (n = 5/group). C: Quantification of the elastin degradation score of the abdominal aortas (n = 6/group). D-F: Representative images of IFN-γ+/CD8a+ T cell and MMP-2+/ and MMP-9+/CD68+ macrophage in the adventitia of CD8−/− AAA mice treated with PBS and IFN-γ (n = 5/group). Scale bar: 50 μm. L, lumen.
Defective interferon-γ production of CD8+ T cells prevented CD8−/− mice from CaCl2-induced abdominal aortic aneurysm formation
To further explore CD8+ T cells-derived IFN-γ plays a unique role in AAA formation, CD8−/− mice received a tail intravenously injected CD8+ T cells isolated from IFN-γ+/+ wildtype or IFN-γ−/− mice were underwent AAA induction. As compared with CD8−/− mice treated with CD8+ T cells of IFN-γ+/+ mice, CD8+ T cells of IFN-γ−/− mice markedly lowered the external aortic diameter and collagen I and III protein expressions as well as histopathological damage (Suppl. Figure S6).
Interferon-γ increased inflammasome activation and MMP-2 and MMP-9 activities in macrophages and/or smooth muscle cells
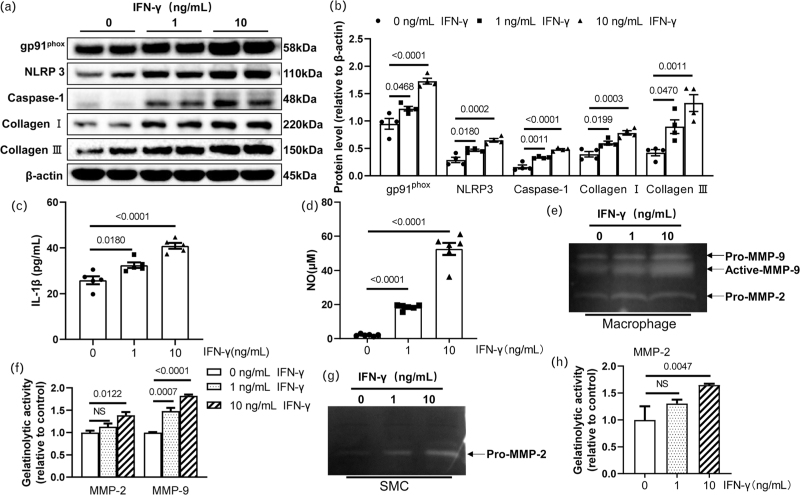
In the in vitro experiment, RAW264.7 cells were stimulated by 0, 1, of 10 ng/ml of IFN-γ for 24 h. As shown in Fig. 5a,b, the IFN-γ treatment increased the levels of NLRP3, caspase-1, gp91phox, collagen I, and collagen III proteins. IFN-γ increased the levels of IL-1β protein and release of nitric oxide in the macrophages (Fig. 5c,d). The IFN-γ treatment also increased the gelatinolytic activities of MMP-2 and MMP-9 in the macrophages (Fig. 5e,f) and promoted the gelatinolytic activities of MMP-2 in the SMCs (Fig. 5g,h).
FIGURE 5.
IFN-γ enhanced the inflammasome-related protein expressions and gelatinase activities in the macrophages and smooth muscle cells (SMCs). The macrophages and SMCs were treated with 0, 1, or 10 ng/mL of IFN-γ for 24 hr, and the lysates were then subjected to western blotting; the conditioned media were collected for the gelatin zymography assays. A,B: Representative western blotting images and quantitative data for NLRP3, caspase-1, gp91phox, collagen I, and collagen III (n = 4/group). C,D: IFN-γ increased IL-1β levels and nitric oxide (NO) release (n = 5–6/group). E-H: Representative images and the quantitative data of the gelatin zymography assays of MMP-2 and/or MMP-9 in the condition medium of macrophages and SMCs (n = 3/group).
DISCUSSION
We focused on novel role(s) of CD8+ T cells and IFN-γ in the development of AAAs. Our results demonstrated that CD8+ T-cell deficiency was resistant to CaCl2-induced AAA formation. At the molecular and cellular levels, CD8+ T-cell deletion lessened the harmful increases in the following: the levels of pro-inflammatory cytokine proteins and genes (IFN-γ, IL-1β, MCP-1, ICAM-1, and VCAM-1) in serum and/or abdominal aortic tissues; NADPH oxidase-mediated oxidative stress production; and aortic lesion macrophages-derived proteolysis enzyme (MMP-2 and MMP-9) activities and genes. These beneficial changes were diminished by IFN-γ treatment and adoptive transfer of CD8+ T cells of IFN-γ+/+ mice in the AAA CD8−/− mice. The in vitro results indicated that IFN-γ enhanced the protein levels of the macrophage-derived inflammasome, providing evidence of the involvement of CD8+ T cells in the development of AAAs.
A transmural infiltration of inflammatory cells is an important cause of AAA formation [21]. Macrophages, T cells, neutrophils, and mast cells are common cell types that cause an extensive inflammation action at the lesion site of AAAs [22,23]. On week 6 after AAA induction in the present study, we observed that CaCl2 treatment can lead to AAA formation and an increase in serum IFN-γ levels. CD8+ T-cell deletion ameliorated the abdominal aortic elastin disruption and collagen deposition as well as IFN-γ+ CD8a+ T cell and MMP-2/9+ CD68+ infiltrations in the adventitia of AAA aortas in mice. At the same time, we also discovered that the levels of serum IFN-γ protein and aortic inflammation-related chemokine genes (MCP-1, ICAM-1, and VCAM-1) and inflammasome-related proteins (NLRP3, caspase-1) were markedly reduced in the CD8−/−-AAA mice. As compared with control group, AAA patients had increased levels of CD8+ T and decreased CD4+ T cells and CD4+/CD8+ ratios [10]. CD43-mediated IFN-γ production by CD8+ T cells promoted elastase-induced AAA in mice [14]. Because CD8+ T cells are one of the main IFN-γ secretion pro-inflammatory cell types [24], we propose that CD8+ T cell-derived IFN-γ functions as an important inflammatory mediator of CaCl2-induced AAA formation.
This hypothesis was further supported by the results of the IFN-γ treatment, that is, that IFN-γ loading diminished the CD8+ T-cell deficiency-mediated vasculoprotective effects on the medial elastin disruption, collagen deposition, and AAA formation that are associated with the increases in the levels of serum IL-1β protein and aortic NLRP3, caspase-1, MCP-1, ICAM-1, and VCAM-1 proteins and/or genes as well as the numbers of AAA lesion macrophages in CD8−/−-AAA mice. The M1 macrophage phenotype is activated through the classical pathway, and these cells accumulate at sites of inflammation and produce pro-inflammatory factors such as TNF-α,IL-1β, IL-6, IL-12, inducible nitric oxide synthase, and nitric oxide [11,12]. In our present in-vitro experiments, IFN-γ increased the levels of NLRP3, caspase-1, gp91phox, collagen I, and collagen III proteins and promoted nitric oxide release in macrophages. Zhou et al.[14] demonstrated in an elastase-perfusion model of AAA, that CD8−/− mice are resistant to AAA induction, and that adoptive transfer of wild type CD8+ T cells into CD8−/− mice can rescue these effects and restore AAA outcome in an IFN-γ-dependent manner. Taken together, our results suggest that in mice, CD8+ T cells promote both the formation of AAA induced by CaCl2 through these cells ability to activate inflammatory macrophage activation and responses that are mediated by IFN-γ. This was further supported by our data of the adaptive transfers of IFN-γ+/+ and IFN-γ−/− CD8+ T cells that defective IFN-γ production of CD8+ T cells prevented CD8−/− mice from CaCl2-induced AAA formation. It should be noted that CD8a−/− mice lack not only CD8a+ T cells but also CD8a+ dendritic cells. It was reported that CD8a expressed on dendritic cell may not only be a lineage/maturation marker but also contribute to dendritic cell function, suggesting that dendritic cell function might be impaired in CD8a−/− mice [25]. Importantly, although a major function of the CD8a+ dendritic cell subset is cross-presentation of antigen to CD8+ T cells, the subset is also a significant producer of IL-12 upon their activation, and as such the subset can potentially prime CD4+ T cell responses, potentially promoting Th1 differentiation which results in much production of IFN-γ [26]. CD8a+ dendritic cells have been shown to aggravate atherosclerosis, likely by inducing Th1 cell response, which promoted CCL5 expression in macrophages and increased infiltration of leukocytes and lesion inflammation [27]. Furthermore, recent study showed that Th1 participated in cardiovascular disease processes including AAA [28]. Collectively, these findings suggested that CD8a+ dendritic cells might be involved in AAA formation via the IL-12-mediated Th1 differentiation and its IFN-γ production in mice under our experimental conditions.
Accumulating evidence indicates that reactive oxygen species (ROS) contribute to AAA formation, progression, and rupture [29]. NADPH oxidase is the main source of ROS and is present in phagocytes, neuroepithelial cells, vascular SMCs, and endothelial cells [30]. gp91phox is an integral component of the NADPH oxidase complex, which is involved in ROS production. Pharmacological interventions targeting NADPH oxidase have been shown to reverse AAA progression and rupture [31]. We observed that NADPH oxidase activity and oxidative stress (gp91phox)-related genes were decreased in the CD8−/− mice after the CaCl2 application. Compared to the CD8−/− mice that were not injected with IFN-γ, the IFN-γ supplementation in CD8−/− mice injected with IFN-γ not only exerted a promotive effect on aneurysm generation but also increased the NADPH oxidase activity and the expression of oxidative stress (gp91phox)-related genes in the abdominal aorta, suggesting that CD8+ T cells mediate oxidative stress via regulation by IFN-γ and thus impact the formation of AAAs.
The ECM is important not only for maintaining the structural and mechanical properties required for vascular function; it also provides instructional signals that induce, define, and stabilize the vascular cell phenotype [32]. Collagen is a crucial structural protein in the ECM of arteries [33]. A compensatory increase in collagen synthesis is observed during the onset and early development of an AAA [34]. However, it was also considered that CaCl2-induced AAAs may increase collagen degradation, resulting in a decreased collagen content. As the most thoroughly investigated ECM-degrading proteinases, many MMPs (especially MMP-2 and MMP-9) have been reported to aggravate AAA formation [35,36]. It was reported that that MMP activity within the aorta of AAA animals is impacted by deletion of CD43 and/or IFN-γ on CD8+ T cells in the elastase-induced AAA mice, and that this could be restored by administration of either IFN-γ+/+ producing CD8+ T cells or recombinant IFN-γ [14]. Other investigations have demonstrated that CatS and CatK, which are members of cathepsin family, play crucial roles in AAA formation by enhancing elastin degradation and regulating the expression and activity of vascular cell proteases [37,38]. Our present results showed that CaCl2-induced vascular injury increased the expressions of proteolytic enzyme genes (CatS, CatK, MMP-2, and MMP-9) and collagen genes (Collagen I, Collagen III) in aortas, and CD8+ T-cell deficiency was resistant to this alteration. In another set of experiments in which IFN-γ was administered to CaCl2-treated CD8−/− mice, IFN-γ supplementation increased the expression of the above genes and proteins. Our data of the immunofluorescence revealed that CD8 deletion reduced MMP-2+/CD68+ and MMP-9+/CD68+ macrophage infiltration in the adventitia of the mouse AAA aortas and IFN-γ-loading increased both cell infiltrations in the adventitia of CD8−/− mice. In our in vitro cell experiments, SMCs treated with IFN-γ showed enhanced expressions of MMPs. Macrophages treated with IFN-γ had enhanced MMP expressions and enhanced collagen I and collagen III gene and protein levels. Our findings thus indicate that IFN-γ might play a role in the formation of AAAs, at least in part depending on the increase of the expressions of CatS, CatK, MMP-2, and MMP-9, exerting a promoting effect on aneurysm generation. We observed that macrophages produced collagen I and collagen III in response to IFN-γ stimulation, which suggests that the collagen in the aneurysmal tissue, at least in part, was derived from macrophages. CD8+ T cells and IFN-γ-regulated collagen accumulation are involved in aneurysm formation, possibly increasing the degree of fibrosis, leading to the loss of arterial flexibility, which together with the massive destruction of elastin of media enhances aneurysms.
Study limitations should be considered. First, we could no explore whether CD8a+ dendritic cells can modulate AAA formation via the IL-12-mediated Th1 differentiation and IFN-γ production in mice under our experimental conditions. Second, we could not provide the direct evidence of the vascular and inflammatory cellular crosstalk of IFN-γ in AAA formation. Second, we could not provide the direct evidence of the vascular and inflammatory cellular crosstalk of IFN-γ in AAA formation. Finally, we also did not evaluate the cell source of IL-1β in vivo. Further study will be needed to investigate these issues.
In summary, our present findings demonstrated that CD8+ T-cell deficiency lessened the formation of CaCl2-induced AAAs by decreasing the secretion of IFN-γ, leading to decreased macrophage recruitment, which reduces inflammation, oxidative stress, and ECM degradation, providing a potential treatment target against AAA.
ACKNOWLEDGEMENTS
This work was supported in part by grants from the National Natural Science Foundation of China (nos. 81560240, 81770485, and 82370424 to XWC; no. 82160087 to Y.L.).
Z. Lin conceived the project, designed and performed experiments, implemented and analyzed the data, interpreted data, prepared figures, and drafted and wrote the article. M. Zhao, X. Yue, X. Zheng, and M. Zhang participated in the mouse handling and immunoblotting and performed the blinded data analysis of immunostained and H&E-stained images. X. Zhang taught to create mouse aneurysm model. J. Piao performed the mouse breeding and genotyping. G.P. Shi, R. Cui, and X. Yue discussed the results and revised and critically reviewed the article. X.W. Cheng conceived the project, designed and performed experiments, interpreted data, and wrote, revised and critically reviewed the article. All authors read and approved the article.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships or conflicts of interests that could have appeared to influence the work reported in this article.
Supplementary Material
Footnotes
Abbreviations: AAA, bdominal aortic aneurysm; ANOVA, analysis of variance; CaCl2, calcium chloride2; CatK, cathepsin K; CatS, cathepsin S; DC, dendritic cell; DMEM, Dulbecco's modified Eagle's medium; ECM, extracellular matrix; EVG, Elastica van Gieson; FBS, fetal bovine serum; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; H&E, hematoxylin and eosin; ICAM-1, intercellular adhesion molecule-1; IFN-γ, interferon-gama; IL-1β, interleukin-1beta; MCP-1, monocyte chemoattractant protein-1; MMP, metalloproteinase; MMP-2, matrix metalloproteinase-2; MMP-9, metalloproteinase-9; NADPH, nicotinamide adenine dinucleotide phosphate; NLRP3, NOD-like receptor protein 3; ROS, reactive oxygen species; RT-PCR, real-time PCR; SMC, smooth muscle cell; TNF-α, tumor necrosis factor-alpha; VCAM-1, vascular cell adhesion molecule-1
Supplemental digital content is available for this article.
REFERENCES
- 1.Lu Y, Ma Q, Tan H, Li X, Zhang X, Tie Y. Specific inhibition of SHP2 suppressed abdominal aortic aneurysm formation in mice by augmenting the immunosuppressive function of MDSCs. Life Sci 2021; 265:118751. [DOI] [PubMed] [Google Scholar]
- 2.Xu B, Li G, Guo J, Ikezoe T, Kasirajan K, Zhao S, et al. Angiotensin-converting enzyme 2, coronavirus disease 2019, and abdominal aortic aneurysms. J Vasc Surg 2021; 74:1740–1751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sakalihasan N, Michel JB, Katsargyris A, Kuivaniemi H, Defraigne JO, Nchimi A, et al. Abdominal aortic aneurysms. Nat Rev Dis Primers 2018; 4:34. [DOI] [PubMed] [Google Scholar]
- 4.Jiang H, Sasaki T, Jin E, Kuzuya M, Cheng XW. Inflammatory cells and proteases in abdominal aortic aneurysm and its complications. Curr Drug Targets 2018; 19:1289–1296. [DOI] [PubMed] [Google Scholar]
- 5.Koh CH, Lee S, Kwak M, Kim BS, Chung Y. CD8 T-cell subsets: heterogeneity, functions, and therapeutic potential. Exp Mol Med 2023; 55:2287–2299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schäfer S, Zernecke A. CD8(+) T cells in atherosclerosis. Cells 2020; 10:37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.de Araújo-Souza PS, Hanschke SC, Viola JP. Epigenetic control of interferon-gamma expression in CD8 T cells. J Immunol Res 2015; 2015:849573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Benson LN, Liu Y, Wang X, Xiong Y, Rhee SW, Guo Y, et al. The IFNγ-PDL1 pathway enhances CD8T-DCT interaction to promote hypertension. Circ Res 2022; 130:1550–1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Deng Q, Luo Y, Chang C, Wu H, Ding Y, Xiao R. The emerging epigenetic role of CD8+T cells in autoimmune diseases: a systematic review. Front Immunol 2019; 10:856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Xie X, Song Z, Wang L, Qi M. Expression and significance of T lymphocyte subsets, RANTES and inflammatory factors levels in serum of patients with abdominal aortic aneurysm. Am J Transl Res 2021; 13:11987–11992. [PMC free article] [PubMed] [Google Scholar]
- 11.Cai D, Sun C, Murashita T, Que X, Chen SY. ADAR1 non-editing function in macrophage activation and abdominal aortic aneurysm. Circ Res 2023; 132:e78–e93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yuan Z, Lu Y, Wei J, Wu J, Yang J, Cai Z. Abdominal aortic aneurysm: roles of inflammatory cells. Front Immunol 2020; 11:609161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ishida Y, Kuninaka Y, Nosaka M, Kimura A, Taruya A, Furuta M, et al. Prevention of CaCl(2)-induced aortic inflammation and subsequent aneurysm formation by the CCL3-CCR5 axis. Nat Commun 2020; 11:5994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhou HF, Yan H, Cannon JL, Springer LE, Green JM, Pham CT. CD43-mediated IFN-γ production by CD8+ T cells promotes abdominal aortic aneurysm in mice. J Immunol 2013; 190:5078–5085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu CL, Ren J, Wang Y, Zhang X, Sukhova GK, Liao M, et al. Adipocytes promote interleukin-18 binding to its receptors during abdominal aortic aneurysm formation in mice. Eur Heart J 2020; 41:2456–2468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sukhova GK, Zhang Y, Pan JH, Wada Y, Yamamoto T, Naito M, et al. Deficiency of cathepsin S reduces atherosclerosis in LDL receptor-deficient mice. J Clin Invest 2003; 111:897–906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Song H, Yang Y, Sun Y, Wei G, Zheng H, Chen Y, et al. Circular RNA Cdyl promotes abdominal aortic aneurysm formation by inducing M1 macrophage polarization and M1-type inflammation. Mol Ther 2022; 30:915–931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hu L, Huang Z, Ishii H, Wu H, Suzuki S, Inoue A, et al. PLF-1 (Proliferin-1) modulates smooth muscle cell proliferation and development of experimental intimal hyperplasia. J Am Heart Assoc 2019; 8:e005886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Qian HS, Gu JM, Liu P, Kauser K, Halks-Miller M, Vergona R, et al. Overexpression of PAI-1 prevents the development of abdominal aortic aneurysm in mice. Gene Ther 2008; 15:224–232. [DOI] [PubMed] [Google Scholar]
- 20.Inoue A, Piao L, Yue X, Huang Z, Hu L, Wu H, et al. Young bone marrow transplantation prevents aging-related muscle atrophy in a senescence-accelerated mouse prone 10 model. J Cachexia Sarcopenia Muscle 2022; 13:3078–3090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Varona S, Puertas L, Galán M, Orriols M, Cañes L, Aguiló S, et al. Rolipram prevents the formation of abdominal aortic aneurysm (AAA) in mice: PDE4B as a target in AAA. Antioxidants (Basel) 2021; 10:460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Qin Y, Cao X, Guo J, Zhang Y, Pan L, Zhang H, et al. Deficiency of cathepsin S attenuates angiotensin II-induced abdominal aortic aneurysm formation in apolipoprotein E-deficient mice. Cardiovasc Res 2012; 96:401–410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Liu CL, Liu X, Zhang Y, Liu J, Yang C, Luo S, et al. Eosinophils protect mice from angiotensin-II perfusion-induced abdominal aortic aneurysm. Circ Res 2021; 128:188–202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ikeda T, Sato K, Kawaguchi SI, Izawa J, Takayama N, Hayakawa H, et al. Forodesine enhances immune responses through guanosine-mediated TLR7 activation while preventing graft-versus-host disease. J Immunol 2023. [DOI] [PubMed] [Google Scholar]
- 25.Hong L, Webb TJ, Wilkes DS. Dendritic cell-T cell interactions: CD8 alpha alpha expressed on dendritic cells regulates T cell proliferation. Immunol Lett 2007; 108:174–178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Maldonado-López R, De Smedt T, Pajak B, Heirman C, Thielemans K, Leo O, et al. Role of CD8alpha+ and CD8alpha- dendritic cells in the induction of primary immune responses in vivo. J Leukoc Biol 1999; 66:242–246. [DOI] [PubMed] [Google Scholar]
- 27.Li Y, Liu X, Duan W, Tian H, Zhu G, He H, et al. Batf3-dependent CD8α(+) dendritic cells aggravates atherosclerosis via Th1 cell induction and enhanced CCL5 expression in plaque macrophages. EBioMedicine 2017; 18:188–198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Aria H, Kalani M, Hodjati H, Doroudchi M. Different cytokine patterns induced by Helicobacter pylori and Lactobacillus acidophilus extracts in PBMCs of patients with abdominal aortic aneurysm. Comp Immunol Microbiol Infect Dis 2020; 70:101449. [DOI] [PubMed] [Google Scholar]
- 29.Sun W, Pang Y, Liu Z, Sun L, Liu B, Xu M, et al. Macrophage inflammasome mediates hyperhomocysteinemia-aggravated abdominal aortic aneurysm. J Mol Cell Cardiol 2015; 81:96–106. [DOI] [PubMed] [Google Scholar]
- 30.Salmon M. NADPH oxidases in aortic aneurysms. Antioxidants (Basel) 2022; 11:1830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Green SP, Cairns B, Rae J, Errett-Baroncini C, Hongo JA, Erickson RW, et al. Induction of gp91-phox, a component of the phagocyte NADPH oxidase, in microglial cells during central nervous system inflammation. J Cereb Blood Flow Metab 2001; 21:374–384. [DOI] [PubMed] [Google Scholar]
- 32.Wagenseil JE, Mecham RP. Vascular extracellular matrix and arterial mechanics. Physiol Rev 2009; 89:957–989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.de Oliveira Neves VG, Blascke de Mello MM, Rodrigues D, Pernomian L, de Oliveira IS, Parente JM, et al. Type I collagen proteolysis by matrix metalloproteinase-2 contributes to focal adhesion kinase activation and vascular smooth muscle cell proliferation in the aorta in early hypertension. Vasc Pharmacol 2023; 152:107211. [DOI] [PubMed] [Google Scholar]
- 34.Brangsch J, Reimann C, Collettini F, Buchert R, Botnar RM, Makowski MR. Molecular imaging of abdominal aortic aneurysms. Trends Mol Med 2017; 23:150–164. [DOI] [PubMed] [Google Scholar]
- 35.Li Z, Cong X, Kong W. Matricellular proteins: potential biomarkers and mechanistic factors in aortic aneurysms. J Mol Cell Cardiol 2022; 169:41–56. [DOI] [PubMed] [Google Scholar]
- 36.Cai D, Sun C, Zhang G, Que X, Fujise K, Weintraub NL, et al. A novel mechanism underlying inflammatory smooth muscle phenotype in abdominal aortic aneurysm. Circ Res 2021; 129:e202–e214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wan Y, Piao L, Xu S, Meng X, Huang Z, Inoue A, et al. Cathepsin S activity controls chronic stress-induced muscle atrophy and dysfunction in mice. Cell Mol Life Sci 2023; 80:254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sun J, Sukhova GK, Zhang J, Chen H, Sjöberg S, Libby P, et al. Cathepsin K deficiency reduces elastase perfusion-induced abdominal aortic aneurysms in mice. Arterioscler Thromb Vasc Biol 2012; 32:15–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.