Abstract
Breast cancer remains a significant public health issue, often resulting in severe side effects such as neutropenia, highlighting the need for reliable predictors of clinical outcomes. This study aimed to evaluate the predictive value of body composition measures for mortality, recurrence, and chemotherapy-induced neutropenia in patients with breast cancer following surgery and chemotherapy. We retrospectively analyzed 85 breast cancer patients who underwent surgery and chemotherapy between 2006 and 2016. Body composition was assessed using computed tomography (CT) or positron emission tomography (PET) at diagnosis and three years and five years post-diagnosis. Metrics included skeletal muscle area (SMA), skeletal muscle index (SMI), subcutaneous adipose tissue area (SAT), and visceral adipose tissue area (VAT). Longitudinal analysis revealed a decrease in muscle mass (P < 0.001 for both SMA and SMI) and nonsignificant changes in fat mass (P = 0.449 for SAT and P = 0.798 for VAT). A lower SMI at diagnosis was significantly associated with increased mortality (P = 0.019) and a higher incidence of grade 4 neutropenia (P = 0.008). There was no significant association between SMI at diagnosis and recurrence (P = 0.691). No associations were found between body composition measurements during the follow-up period and the clinical outcomes. Lower skeletal muscle mass at diagnosis is strongly associated with higher mortality and chemotherapy-induced complications in patients with breast cancer, highlighting the potential of readily available imaging techniques as valuable predictors of clinical outcomes.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-024-74060-w.
Keywords: Breast cancer, Body composition, Sarcopenia, Chemotherapy-induced neutropenia, Mortality, Prognosis
Subject terms: Breast cancer, Prognostic markers, Oncology, Risk factors
Introduction
Breast cancer, which accounts for a significant proportion of cancer diagnoses among women globally, remains a prominent public health concern owing to its prevalence and impact on mortality rates, according to data from Global Cancer Statistics1. By 2023, it is estimated that there will be approximately 297,790 new cases of breast cancer and 43,170 breast cancer-related deaths in the United States, representing 15.2% and 7.1% of all new cancer cases and cancer-related deaths, respectively2.
Breast cancer treatment involves a multidisciplinary approach encompassing locoregional interventions, such as surgery and radiotherapy, as well as chemotherapy tailored to the molecular subtype. Despite its widespread use, chemotherapy frequently results in various adverse effects, including hair loss, nausea, vomiting, diarrhea, and neutropenia. The frequent occurrence of neutropenia following chemotherapy is crucial because it puts patients at risk of life-threatening complications, such as febrile neutropenia and neutropenic enterocolitis, thereby amplifying both the morbidity and mortality associated with cancer treatment, as well as the overall cost of care for cancer patients3,4.
The search for reliable predictors of clinical outcomes and treatment-related toxicities in patients with breast cancer is ongoing. The body mass index (BMI) has traditionally served as a common metric for evaluating the nutritional status of patients. Several studies have explored the association between obesity and decreased survival rates in breast cancer patients5–7. However, other studies reported no significant correlation8,9. Owing to its low representability, recent research has focused on body composition parameters such as skeletal muscle area (SMA), subcutaneous adipose tissue area (SAT), and visceral adipose tissue area (VAT), which are discernible through imaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI). Well-established reference values for skeletal muscle index (SMI) and VAT exist in the literature10–12, and these cutoffs have been widely used in studies of non-breast cancer populations. However, discrepancies in how sarcopenia is defined, especially in breast cancer cohorts, have led to the need for cancer-specific cutoffs. Furthermore, some authors suggest other metrics, such as the VAT/SAT ratio, which has shown potential prognostic relevance in certain breast cancer populations but still requires further investigation to clarify its broader impact on outcomes13.
Several studies have shown a relationship between sarcopenia and poor cancer prognoses14–22. However, these studies used varying definitions of sarcopenia that diverge from the generally accepted threshold, defined as absolute muscularity below the 5th percentile10,11, leading to confusion. Some studies, for instance, have applied cutoffs derived from cohorts of non-breast cancer patients, such as those with respiratory or gastrointestinal tumors, which may not be appropriate for breast cancer patients23,24. This inconsistency highlights the need for breast cancer-specific definitions of sarcopenia, particularly when assessing skeletal muscle depletion using metrics like SMI. Research on adipose tissue, encompassing both subcutaneous and visceral types, is sparse and presents contradictory findings in breast cancer patients13,16,21,22,25–28, and unlike skeletal muscle with established metrics, such as SMI, fat lacks universally accepted indices.
There is a growing need for new prognostic parameters to predict the clinical outcomes in patients with breast cancer, and body composition measures have garnered increasing attention. However, these studies were controversial, and the parameters lacked standardization. In this study, we present optimal cutoff values for body composition measures that best predict clinical outcomes such as mortality, recurrence, and chemotherapy-induced neutropenia in patients with breast cancer who underwent surgery and chemotherapy.
Methods
Study population and treatment
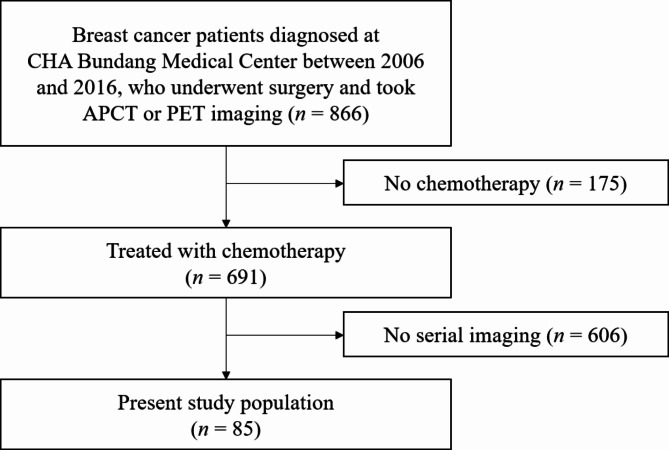
This study was a retrospective analysis of patients diagnosed with stage I–III primary breast cancer who underwent surgery at the CHA Bundang Medical Center, Seongnam, South Korea, between 2006 and 2016. Among patients who had undergone either abdominopelvic CT (APCT) or positron emission tomography (PET), those treated with chemotherapy were eligible. Patients also had to undergo serial image follow-up, defined as APCT or PET images taken at least three times during a five-year follow-up period: at the initial diagnosis (± 1 month), three years (± 6 months) after diagnosis, and five years (± 6 months) after diagnosis. A detailed flowchart of the study is shown in Fig. 1. All participants underwent standard therapeutic interventions, including surgery, neoadjuvant or adjuvant chemotherapy, hormonal therapy, targeted therapy, and radiotherapy, according to the National Comprehensive Cancer Network (NCCN) guidelines29. Details regarding the follow-up duration and time intervals between imaging sessions were documented.
Fig. 1.
Flowchart of the study population. Serial imaging refers to a minimum of three APCT or PET scans obtained at the time of initial breast cancer diagnosis, within three years (± 6 months) and five years (± 6 months) after diagnosis. APCT abdominopelvic computed tomography, PET positron emission tomography.
This study was approved by the Institutional Review Board (IRB) of CHA Bundang Medical Center, Seongnam, South Korea (IRB No. 2022-12-025). The IRB also approved a waiver of written informed consent due to the retrospective nature of the study and the use of anonymous clinical data. All methods were performed in accordance with the relevant guidelines and regulations.
Data collection and analyses
Using electronic medical records, we extracted a comprehensive set of data comprising general patient characteristics (age, sex, and BMI), disease status (histological grade, molecular subtypes, and pathological stage), treatment (surgical procedure, neoadjuvant chemotherapy, adjuvant chemotherapy, hormonal therapy, and radiotherapy), and clinical outcomes (complications, mortality, and recurrence).
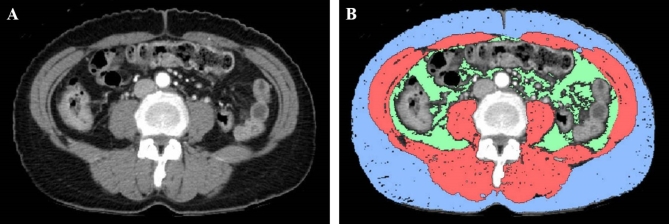
Body composition measures, including SMA, SAT, and VAT, were calculated from the aforementioned sequential APCT and PET images using CT Analyser version 1.17.7.2+. The analysis was conducted on a single axial slice positioned at the L3 transverse process level, a method frequently used in previous studies because of its optimal representation of the overall body composition30,31. The abdominal adipose tissue and muscle area were semiautomatically measured using a dedicated workstation (CT Analyser) (Fig. 2). The preestablished Hounsfield Unit (HU) thresholds of − 29 to + 150 were applied to identify skeletal muscle, and thresholds of − 300 to − 50 were used for adipose tissue14,16,28. One of our medical experts manually distinguished the tissues based on anatomical features in the CT images. Initially, the regional SAT from the skin surface to the outer surface of the abdominal muscles was manually traced and measured using a workstation. Subsequently, VAT and SMA were measured using a process that included some manual measurements. SMI was defined as the SMA divided by the square of the height in meters.
Fig. 2.
Measurement of skeletal muscle and fat at the umbilical (L3) level. (A) CT image pre-analysis. (B) CT image post-analysis. The axial section of the CT image is segmented into SMA (red), SAT (blue), and VAT (green). CT computed tomography, SMA skeletal muscle area, SAT subcutaneous adipose tissue area, VAT visceral adipose tissue area.
The endpoints were overall survival (OS) and disease-free survival (DFS). OS was defined as the duration from the date of diagnosis to the date of death from any cause or the date of the most recent follow-up. DFS was defined as the duration from the date of diagnosis to the first recurrence (local or distant) of the disease, date of death from any cause, or date of the most recent follow-up. We also confirmed the occurrence of neutropenia as a representative side effect of chemotherapy. Neutropenia was defined according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE), version 5.032, and granulocyte colony-stimulating factor (G-CSF) was administered in cases of grade 4 neutropenia (< 500/mm³ absolute neutrophil count). The final follow-up date was July 25, 2022.
Statistical analysis
Categorical data are expressed as absolute numbers and corresponding percentages, whereas continuous data are presented as means with standard deviation or medians with standard deviation, as appropriate. To compare body composition measurements over time, the Kruskal–Wallis test was conducted, which was selected after verifying that the data did not follow a normal distribution using the Shapiro–Wilk test. To determine the optimal cutoff value for body composition measures to distinguish clinical outcomes, receiver operating characteristic (ROC) curve analysis was utilized. To build the binary classification model, individuals were segregated based on the cutoff values of body composition measures (SMI, SAT, and VAT) at the time of diagnosis. Consequently, dichotomous participant groups, determined by the cutoff values of body composition measures at diagnosis, served as input variables, whereas mortality served as the target variable for the binary classification model. Youden’s J-statistic was used to identify the optimal cutoff threshold that maximized the performance of the model. Group differences were assessed using the chi-squared test. OS and DFS were estimated using the Kaplan–Meier method and compared using the log-rank test. Variables with a significance of P < 0.10 in the univariable analysis for a given endpoint were included in the multivariate Cox or logistic regression model for that endpoint, along with SMI, SAT, and VAT, regardless of their P values in the univariable analysis. A significance level of P < 0.05 was considered statistically meaningful. All statistical analyses were performed using the R Statistical Software version 3.6.3 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient characteristics and outcomes
A total of 85 eligible patients were included in the final cohort (Fig. 1). The baseline patient characteristics and clinical outcomes are described in Table 1. The median age was 49.8 ± 8.0 years (range, 31–70). At the time of diagnosis, the study cohort’s mean BMI was 23.7 ± 3.3 kg/m2. Breast cancer stage distribution in the study cohort revealed that 35 patients (41.1%) were classified as stage I, 32 (37.6%) as stage II, and 18 (21.2%) as stage III. Among the patients, 46 (54.1%) underwent total mastectomy, and 39 (45.9%) underwent partial mastectomy. Neoadjuvant chemotherapy was administered to nine patients (10.6%), and adjuvant chemotherapy was administered to 76 patients (89.4%). 65 patients (76.5%) received hormonal therapy, and 53 (62.4%) underwent radiotherapy. The median follow-up period was 103.4 ± 30.5 months. Throughout the follow-up period, 6 cases (7.1%) of mortality, 10 (11.8%) of recurrence, and 20 (23.5%) of grade 4 neutropenia were recorded. The median time to death was 91.6 ± 32.2 months, while the median time to recurrence was 47.9 ± 18.9 months.
Table 1.
Patients characteristics and clinical outcomes.
| Characteristics | Results |
|---|---|
| Age (years) | 49.8 ± 8.0 |
| BMI (kg/m2) | 23.7 ± 3.3 |
| AJCC stage | |
| I | 35 (41.1) |
| II | 32 (37.6) |
| III | 18 (21.2) |
| T stage | |
| 1 | 40 (47.1) |
| 2 | 35 (41.2) |
| 3 | 10 (11.8) |
| N stage | |
| 0 | 55 (64.7) |
| 1 | 15 (17.6) |
| 2 | 8 (9.4) |
| 3 | 7 (8.2) |
| Histologic type | |
| Invasive ductal carcinoma | 77 (90.6) |
| Others | 8 (9.4) |
| Histologic grade | |
| 1 | 16 (18.8) |
| 2 | 38 (44.7) |
| 3 | 29 (34.1) |
| Unknown | 2 (2.4) |
| Hormone receptor status | |
| ER positive | 58 (68.2) |
| PR positive | 48 (56.5) |
| HER2 positive | 27 (31.8) |
| Surgery type | |
| Total mastectomy | 46 (54.1) |
| Partial mastectomy | 39 (45.9) |
| Treatment type | |
| Chemotherapy | |
| Neoadjuvant chemotherapy | 9 (10.6) |
| Adjuvant chemotherapy | 76 (89.4) |
| Hormonal therapy | 65 (76.5) |
| Radiotherapy | 53 (62.4) |
| Clinical outcomes | |
| Death | 6 (7.1) |
| Recurrence | 10 (11.8) |
| Grade 4 neutropenia | 20 (23.5) |
Data, excluding clinical outcomes, are gathered at the time of diagnosis. Data are expressed as the number of patients with percentages in parentheses or mean ± standard deviation. Temporal data are expressed as median ± standard deviation.
BMI body mass index, AJCC American Joint Committee on Cancer, ER estrogen receptor, PR progesterone receptor, HER2 human epidermal growth factor 2.
Body composition measures
Changes in body composition over time are shown in Table 2. The mean values for SMA, SMI, SAT, and VAT at diagnosis were 121.9 ± 21.0 cm2, 48.9 ± 8.3 cm2/m2, 192.9 ± 80.2 cm2, and 80.6 ± 44.3 cm2, respectively. The muscle-related indices showed a decrease, with most of the decrease within three years after diagnosis and a tendency to maintain the decrease within five years after diagnosis (P < 0.001 for SMA, P < 0.001 for SMI). Fat-related indices showed an increasing trend but were not statistically significant. SAT increased three years after diagnosis and decreased five years after diagnosis, whereas VAT increased five years after diagnosis (P = 0.449 for SAT, P = 0.798 for VAT).
Table 2.
Body composition measures over time.
| At diagnosis | After 3 years | After 5 years | P | |
|---|---|---|---|---|
| SMA (cm2) | 121.9 ± 21.0 | 106.0 ± 25.8 | 106.0 ± 27.9 | < 0.001 |
| SMI (cm2/m2) | 48.9 ± 8.3 | 42.5 ± 10.4 | 42.5 ± 11.1 | < 0.001 |
| SAT (cm2) | 192.9 ± 80.2 | 205.9 ± 72.4 | 200.1 ± 77.5 | 0.449 |
| VAT (cm2) | 80.6 ± 44.3 | 84.7 ± 49.4 | 85.0 ± 46.2 | 0.798 |
Data are expressed as mean ± standard deviation.
SMA skeletal muscle area, SMI skeletal muscle index, SAT subcutaneous adipose tissue area, VAT visceral adipose tissue area.
Associations between body composition measures and clinical outcomes
The optimal cutoff value for SMI at the time of diagnosis, which provided the best discrimination for patient mortality, was determined to be 48.6 cm2/m2 (AUC = 0.63). The optimal cutoff values for SAT and VAT at diagnosis were identified in the same manner, resulting in 226.6 cm2 (AUC = 0.53) and 45.5 cm2 (AUC = 0.57), respectively (Supporting Information Fig. 1). Patients were categorized into two groups based on the optimal cutoff values of body composition measures at diagnosis, followed by evaluation of clinical outcomes (Table 3).
Table 3.
Associations between body composition measures and clinical outcomes.
| At diagnosis | After 3 years | After 5 years | |||||||
|---|---|---|---|---|---|---|---|---|---|
| SMI < 48.6 cm2/m2 (n = 46) | SMI ≥ 48.6 cm2/m2 (n = 39) | P | SMI < 48.6 cm2/m2 (n = 65) | SMI ≥ 48.6 cm2/m2 (n = 20) | P | SMI < 48.6 cm2/m2 (n = 62) | SMI ≥ 48.6 cm2/m2 (n = 23) | P | |
| Death | 6 (13.0) | 0 (0.0) | 0.019 | 5 (7.7) | 1 (5.0) | 0.681 | 3 (4.8) | 3 (13.0) | 0.190 |
| Recurrence | 6 (13.0) | 4 (10.3) | 0.691 | 7 (10.8) | 3 (15.0) | 0.608 | 7 (11.3) | 3 (13.0) | 0.824 |
| Grade 4 Neutropenia | 16 (34.8) | 4 (10.3) | 0.008 | 18 (27.7) | 2 (10.0) | 0.103 | 17 (27.4) | 3 (13.0) | 0.165 |
| SAT < 226.6 cm2 (n = 54) | SAT ≥ 226.6 cm2 (n = 31) | P | SAT < 226.6 cm2 (n = 51) | SAT ≥ 226.6 cm2 (n = 34) | P | SAT < 226.6 cm2 (n = 56) | SAT ≥ 226.6 cm2 (n = 29) | P | |
|---|---|---|---|---|---|---|---|---|---|
| Death | 6 (11.1) | 0 (0.0) | 0.054 | 4 (7.8) | 2 (5.9) | 0.730 | 5 (8.9) | 1 (3.4) | 0.349 |
| Recurrence | 8 (14.8) | 2 (6.5) | 0.249 | 7 (13.7) | 3 (8.8) | 0.492 | 9 (16.1) | 1 (3.4) | 0.087 |
| Grade 4 Neutropenia | 13 (24.1) | 7 (22.6) | 0.876 | 11 (21.6) | 9 (26.5) | 0.602 | 12 (21.4) | 8 (27.6) | 0.526 |
| VAT < 45.5 cm2 (n = 19) | VAT ≥ 45.5 cm2 (n = 66) | P | VAT < 45.5 cm2 (n = 20) | VAT ≥ 45.5 cm2 (n = 65) | P | VAT < 45.5 cm2 (n = 18) | VAT ≥ 45.5 cm2 (n = 67) | P | |
|---|---|---|---|---|---|---|---|---|---|
| Death | 3 (15.8) | 3 (4.5) | 0.092 | 2 (10.0) | 4 (6.2) | 0.557 | 2 (11.1) | 4 (6.0) | 0.450 |
| Recurrence | 2 (10.5) | 8 (12.1) | 0.849 | 3 (15.0) | 7 (10.8) | 0.608 | 4 (22.2) | 6 (9.0) | 0.121 |
| Grade 4 Neutropenia | 5 (26.3) | 15 (22.7) | 0.745 | 5 (25.0) | 15 (23.1) | 0.859 | 4 (22.2) | 16 (23.9) | 0.883 |
Data are expressed as the number of patients with percentages in parentheses.
SMI skeletal muscle index, SAT subcutaneous adipose tissue area, VAT visceral adipose tissue area.
A lower SMI at diagnosis was significantly associated with mortality, as indicated by the occurrence of all six mortalities within the subgroup characterized by a lower SMI at diagnosis (P = 0.019). In addition, this subgroup had a notably higher incidence of grade 4 neutropenia (P = 0.008). However, there was no discernible association between SMI at diagnosis and recurrence (P = 0.691). Patients with a lower SAT at diagnosis showed a trend towards higher mortality (P = 0.054), although statistical significance was not reached. Neither recurrence nor incidence of grade 4 neutropenia was associated with SAT at diagnosis (P = 0.249 for recurrence, P = 0.876 for grade 4 neutropenia). VAT was not associated with any clinical outcome (P = 0.092 for death, P = 0.849 for recurrence, P = 0.745 for grade 4 neutropenia).
To investigate the potential association between body composition measures after diagnosis and clinical outcomes, patients were regrouped based on follow-up body composition measures at three and five years. However, none of the groups displayed a significant association with any clinical outcomes.
Body composition as a predictor of survival and neutropenia
Survival analyses were conducted to investigate the association between body composition and survival. Consistent with the aforementioned findings, patient subgroups with a lower SMI at diagnosis demonstrated a significantly lower OS than those with a higher SMI at diagnosis (P = 0.027, Fig. 3A). To examine the impact of a low SMI after three or five years, analyses were conducted on patients with a lower SMI at diagnosis and after three or five years, compared with the remaining patients. The impact of a lower SMI at diagnosis diminished at three and five years post-diagnosis, exhibiting no statistical significance (P = 0.07 for after three years, P = 0.92 for after five years, Fig. 3B,C). Fat levels at any time point were not associated with OS (Supporting Information Fig. 2). None of the body composition measurements exhibited a statistically significant relationship with DFS (Supporting Information Fig. 3).
Fig. 3.
OS of patient subgroups by SMI. (A) OS of patients with lower SMI than the cutoff at diagnosis versus others. (B) OS of patients with lower SMI than the cutoff at diagnosis and after 3 years versus others. (C) OS of patients with lower SMI than the cutoff at diagnosis and after 5 years versus others. OS overall survival, SMI skeletal muscle index.
To further assess the impact of muscle mass reduction during follow-up, patients were divided into two groups based on changes in SMI. Kaplan-Meier analysis revealed that patients who experienced at least one reduction in SMI during follow-up timepoints did not demonstrate a significant difference in either OS (P = 0.24) or DFS (P = 0.86) (Supporting Information Fig. 4).
The predictive capability of body composition for survival and grade 4 neutropenia was assessed using Cox regression and logistic regression analyses, respectively. In both univariate and multivariate Cox regression analyses for OS and DFS, none of the body composition measurements reached statistical significance (Supporting Information Tables 1 and 2). However, in the univariate and multivariate logistic regression analyses for grade 4 neutropenia, a higher-than-cutoff SMI was independently associated with a reduced occurrence of grade 4 neutropenia (OR 0.221, 95% CI 0.052–0.765, P = 0.025) (Table 4).
Table 4.
Univariate and multivariate logistic regression analyses for risk factors associated with grade 4 neutropenia.
| Variable | Univariate | Multivariate (SMI) | Multivariate (SAT) | Multivariate (VAT) | ||||
|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | |
| SMI | ||||||||
| < 48.6 cm2/m2 | 1 | 1 | ||||||
| ≥ 48.6 cm2/m2 | 0.214 (0.057–0.658) | 0.012 | 0.221 (0.052–0.765) | 0.025 | ||||
| SAT | ||||||||
| < 226.6 cm2 | 1 | 1 | ||||||
| ≥ 226.6 cm2 | 0.920 (0.309–2.575) | 0.876 | 1.557 (0.412–6.024) | 0.512 | ||||
| VAT | ||||||||
| < 45.5 cm2 | 1 | 1 | ||||||
| ≥ 45.5 cm2 | 0.824 (0.265–2.877) | 0.745 | 1.437 (0.395–6.197) | 0.600 | ||||
| Age (years) | 0.956 (0.891–1.020) | 0.186 | ||||||
| BMI (kg/m2) | 0.863 (0.717–1.017) | 0.095 | 0.925 (0.746–1.131) | 0.460 | 0.835 (0.663–1.019) | 0.099 | 0.855 (0.696–1.022) | 0.106 |
| AJCC stage | 2.486 (1.270–5.159) | 0.010 | 2.267 (0.299–17.248) | 0.422 | 1.905 (0.253–13.973) | 0.524 | 2.422 (0.317–18.503) | 0.387 |
| T stage | 2.668 (1.276–5.940) | 0.011 | 1.619 (0.409–6.590) | 0.488 | 1.955 (0.469–8.684) | 0.360 | 1.662 (0.419–6.899) | 0.469 |
| N stage | 1.542 (0.943–2.519) | 0.079 | 0.867 (0.286–2.670) | 0.799 | 0.805 (0.281–2.319) | 0.682 | 0.758 (0.259–2.245) | 0.610 |
| Histologic grade | 1.229 (0.646–2.446) | 0.539 | ||||||
| HR status | ||||||||
| ER positive | 1.114 (0.386–3.515) | 0.846 | ||||||
| PR positive | 0.545 (0.194–1.496) | 0.240 | ||||||
| HER2 positive | 0.457 (0.120–1.420) | 0.203 | ||||||
| Surgery type | ||||||||
| Total mastectomy | 1 | |||||||
| Partial mastectomy | 0.416 (0.133–1.173) | 0.109 | ||||||
| Hormonal therapy | 0.641 (0.212–2.079) | 0.437 | ||||||
| Radiotherapy | 1.556 (0.547–4.868) | 0.422 | ||||||
| Targeted therapy | 2.118 (0.402–9.555) | 0.336 | ||||||
Multivariate logistic regression included variables with a P < 0.10 from the univariable analysis, as well as SMI, SAT, and VAT regardless of their P in the univariable analysis.
SMI skeletal muscle index, SAT subcutaneous adipose tissue, VAT visceral adipose tissue, BMI body mass index, AJCC American Joint Committee on Cancer, HR hormone receptor, ER estrogen receptor, PR progesterone receptor, HER2 human epidermal growth factor 2.
Discussion
This study investigated body composition as a predictor of clinical outcomes in patients with breast cancer and suggested that anthropometric parameters may have significant implications for treatment decisions. According to our analysis, muscle-related indices exhibited a significant decrease after diagnosis, whereas fat showed an increasing tendency without statistically significant differences. We established novel optimal cutoff values for body composition measures that discriminated mortality most optimally within our retrospective cohort. Specifically, SMI < 48.6 cm2/m2 at diagnosis was significantly associated with increased mortality and severe chemotherapy-induced neutropenia risk. Notably, all deaths (N = 6) occurred in patients with an SMI lower than the cutoff. Our study found that adipose tissue was not significantly associated with long-term clinical outcomes.
There was a significant reduction in skeletal muscle mass, while fat mass remained unchanged at three and five years after diagnosis. One study on patients with breast cancer observed a notable decline in muscle mass by 5.0 ± 2.5 cm2 per year after treatment, while no significant variances were noted in subcutaneous and visceral fat33. This finding aligns with our study, including the comparable decrease in SMA observed annually in our cohort three years after diagnosis and treatment. Considering that physiological muscle mass loss typically ranges from 1 to 2% per year34, the observed decline in skeletal muscle mass in these patients may be interpreted as a pathological consequence. However, debate persists on whether this decrease is induced by the disease or by the treatment. Research on other types of cancer has yielded conflicting findings; for example, Kim et al. observed a significant decrease in skeletal muscle and visceral fat following treatment in patients with epithelial ovarian cancer, with no variance noted in subcutaneous fat35, whereas Arora et al. documented a decrease in skeletal muscle but an increase in both subcutaneous and visceral fat after treatment in testicular cancer36. These findings emphasize the importance of an individualized approach for different cancers. Further studies should focus on various types of cancers to understand changes in anthropometric parameters before and after treatment.
Our study’s finding that a lower SMI at diagnosis is associated with higher mortality and increased risk of severe neutropenia in patients with breast cancer aligns with a growing body of research. Studies have shown that sarcopenia, or the loss of skeletal muscle, is associated with poor outcomes in patients with cancer, including increased mortality15–22 and treatment-related complications14–16. These findings could be explained by the fact that skeletal muscle is a critical protein source and energy reserve, especially during catabolic phases37,38. However, a unified approach is lacking, as demonstrated by the varied definitions of sarcopenia based on the different SMI cutoffs in the aforementioned studies. For instance, Prado et al. characterized sarcopenia as SMI ≤ 38.5 cm2/m214, Caan et al. as SMI ≤ 40 cm2/m222, and Deluche et al. as SMI ≤ 41 cm2/m221. These criteria for sarcopenia stem from the work of Baumgartner et al., who established a cutoff value based on data from healthy individuals39. What is considered normal in healthy individuals might not be applicable to cancer patients; for instance, the normal range for hemoglobin levels could be lower in cancer patients than in the general population, emphasizing the need for tailored parameter definitions for cancer patients40. To the best of our knowledge, this is the first study to redefine sarcopenia in patients with breast cancer, with a focus on its predictive capacity for clinical outcomes. The newly established SMI cutoff, which is higher than conventional definitions, highlights the critical role of baseline muscle mass in cancer treatment and survivorship, emphasizing the necessity for further targeted research and clinical strategies focused on understanding the impact of muscle mass at diagnosis on treatment outcomes in cancer patients.
The higher SMI cutoff observed in our study may be attributed to several unique characteristics of breast cancer. Breast cancer is often hormonally driven, with estrogen playing a significant role in body composition, including muscle mass retention41. Reed et al. demonstrated that postmenopausal breast cancer patients have significantly higher plasma estrogen levels compared to healthy postmenopausal women42. These hormonal differences may necessitate a higher threshold for defining muscle depletion to accurately distinguish clinical outcomes in breast cancer patients. Additionally, breast cancer patients generally have higher survival rates compared to those with other cancers43,44. This extended disease course suggests that even small differences in muscle mass could significantly affect long-term outcomes, further justifying the need for a higher SMI threshold in this population. Further long-term follow-up studies are needed to explore these hypotheses in greater detail.
Surprisingly, body composition measures at the three- and five-year follow-up periods were not associated with the clinical outcomes. However, these results should not be interpreted as implying that proper muscle and fat composition at the time of diagnosis should be the only focus in those with breast cancer. Muscle building and fat reduction contribute to overall health45–47, suggesting that efforts to maintain a healthy body composition may have ongoing benefits beyond the initial diagnosis. This outcome could be attributed to the small number of patients included in this study. To our knowledge, no previous study has examined the changes in body composition over extended follow-up periods, as in our study. Should other studies with longer follow-up periods be published, the results could be compared and discussed accordingly. Meanwhile, patients with SAT < 226.6 cm2 at diagnosis tended to exhibit a lower OS (P = 0.09, Supporting Information Fig. 2). VAT was not associated with OS at any time point. Bradshaw et al. demonstrated that SAT (HR 1.13; 95% CI, 1.02–1.26) was a risk factor for breast cancer-related death but not VAT (HR 1.02; 95% CI, 0.91–1.14)26. However, Kim et al. revealed that visceral obesity increased the risk of breast cancer in post-menopausal women by HR 1.50 (95% CI, 0.75–2.98) but not in pre-menopausal women27. We did not conduct subanalyses based on menopausal status, cancer subtype, or treatment. Further subanalyses with a larger number of patients are necessary to obtain more reliable interpretations.
This study has several strengths that contribute to its scientific significance. First, the optimization of the cutoff value for body composition measures enhanced the specificity and applicability of our findings to breast cancer prognosis. This optimization process, rooted in our serially collected data, offers a robust approach for tailoring interventions and predicting patient outcomes more accurately. Second, the longitudinal nature of our data collection provided a comprehensive view of body composition changes over time, adding depth to the existing literature on sarcopenia in patients with breast cancer. To the best of our knowledge, this study is the first to conduct a longitudinal follow-up of body composition in patients with breast cancer and associate it with clinical outcomes over time.
However, our study has some limitations. The small sample size (85 patients) limits the generalizability of our findings and may contribute to a potential bias. This was attributed to the stringent inclusion criteria in our study design, such as selecting breast cancer patients who underwent surgery and chemotherapy, had a follow-up period exceeding five years, and provided satisfactory serial images during follow-up. Future studies with larger cohorts are essential to confirm the prognostic value of body composition measures in breast cancer. Moreover, unlike SMI, which is widely used as an index representing muscle, the lack of reliable indices for abdominal fat is a significant limitation, given the increasing recognition of the role of adiposity in cancer outcomes. This underscores the need for more refined body composition assessments to capture the complex interplay between the different body compartments and cancer progression. Another limitation is the variability in chemotherapy regimens in the study population. Different chemotherapy regimens and their respective effects on body composition may not have been fully accounted for, which could have influenced the generalizability of the results. Because of variations in the pharmacokinetics and toxicity profiles of chemotherapy protocols, we advocate future investigations to distinguish between these regimens individually. While our study primarily focuses on skeletal muscle parameters derived from CT and PET imaging, including muscle area, it is important to recognize additional variables identified in previous studies, such as those from Bauckneht et al.48. These include muscle density (HU) and metabolic activity (standardized uptake values, SUV), both of which are associated with cancer prognosis. Unfortunately, due to the retrospective design of our study, we could not include these metabolic measures. In addition, the current definition of sarcopenia encompasses not only the quantification of muscle mass but also the evaluation of muscle strength and performance49. As our study focuses primarily on muscle area, without assessments of strength or physical performance, our definition of sarcopenia has inherent limitations. Future prospective studies should incorporate these advanced imaging parameters to gain a more comprehensive understanding of the relationship between skeletal muscle function and clinical outcomes in cancer patients.
Conclusion
Among diverse body composition measures, muscle mass at diagnosis correlates with mortality and the risk of chemotherapy-induced neutropenia, whereas alterations in body composition over long follow-up periods showed no significant associations with clinical outcomes. Our distinct definition of sarcopenia in patients with breast cancer suggests that conventional standards of anthropometric measures may need to be tailored specifically for patients with cancer. Using existing imaging technology and readily available software, skeletal muscle mass assessment can be seamlessly integrated into clinical practice for patients with breast cancer, allowing simplified and standardized image analysis in both academic and community settings.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (Nos. 2022R1A2C2005916, RS-2023-00245268) and by a grant of the Research Driven Hospital R&D project, funded by CHA Bundang Medical Center (Grant Number: BDCHA R&D 2023-301).
Author contributions
H.K.: Conceptualization, data curation, formal analysis, investigation, methodology, writing—original draft. I.K.: Conceptualization, data curation, investigation, methodology, writing—original draft, Writing—review and editing. H.P.: Data curation, resources. W.A.: Data curation, formal analysis, resources. S.K.K.: Data curation, resources. S.L.: Conceptualization, funding acquisition, project administration, supervision, writing—review and editing. All authors read and approved the final version of the manuscript for submission.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Declarations
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These two authors contributed equally to this work
References
- 1.Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin.71(3), 209–249. 10.3322/caac.21660 (2021). [DOI] [PubMed] [Google Scholar]
- 2.National Cancer Institute. Surveillance Epidemiology and End Results (SEER) Program. Cancer Stat Facts: Female Breast Cancer. https://seer.cancer.gov/statfacts/html/breast.html. (2023). Accessed 18 March 2024.
- 3.Lustberg, M. B. Management of neutropenia in cancer patients. Clin. Adv. Hematol. Oncol.10(12), 825–826 (2012). [PMC free article] [PubMed] [Google Scholar]
- 4.Lyman, G. H. Risks and consequences of chemotherapy-induced neutropenia. Clin. Cornerstone8(Suppl 5), S12–S18. 10.1016/s1098-3597(06)80054-2 (2006). [DOI] [PubMed] [Google Scholar]
- 5.Arce-Salinas, C. et al. Overweight and obesity as poor prognostic factors in locally advanced breast cancer patients. Breast Cancer Res. Treat.146(1), 183–188. 10.1007/s10549-014-2977-8 (2014). [DOI] [PubMed] [Google Scholar]
- 6.Herlevic, V. C. et al. Breast cancer outcomes in a population with high prevalence of obesity. J. Surg. Res.198(2), 371–376. 10.1016/j.jss.2015.03.088 (2015). [DOI] [PubMed] [Google Scholar]
- 7.Ligibel, J. A. & Wollins, D. American Society of Clinical Oncology obesity initiative: Rationale, progress, and future directions. J. Clin. Oncol.34(35), 4256–4260. 10.1200/JCO.2016.67.4051 (2016). [DOI] [PubMed] [Google Scholar]
- 8.Cecchini, R. S. et al. Body mass index at diagnosis and breast cancer survival prognosis in clinical trial populations from NRG oncology/NSABP B-30, B-31, B-34, and B-38. Cancer Epidemiol. Biomark. Prev.25(1), 51–59. 10.1158/1055-9965.EPI-15-0334-T (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gennari, A. et al. Impact of body mass index (BMI) on the prognosis of high-risk early breast cancer (EBC) patients treated with adjuvant chemotherapy. Breast Cancer Res. Treat.159(1), 79–86. 10.1007/s10549-016-3923-8 (2016). [DOI] [PubMed] [Google Scholar]
- 10.Baracos, V. & Kazemi-Bajestani, S. M. Clinical outcomes related to muscle mass in humans with cancer and catabolic illnesses. Int. J. Biochem. Cell. Biol.45(10), 2302–2308. 10.1016/j.biocel.2013.06.016 (2013). [DOI] [PubMed] [Google Scholar]
- 11.Fearon, K. et al. Definition and classification of cancer cachexia: An international consensus. Lancet Oncol.12(5), 489–495. 10.1016/S1470-2045(10)70218-7 (2011). [DOI] [PubMed] [Google Scholar]
- 12.Murray, T. É., Williams, D. & Lee, M. J. Osteoporosis, obesity, and sarcopenia on abdominal CT: A review of epidemiology, diagnostic criteria, and management strategies for the reporting radiologist. Abdom. Radiol. (N.Y.)42(9), 2376–2386. 10.1007/s00261-017-1124-5 (2017). Erratum in: Abdom. Radiol. (N.Y.) 2018;43(5):1281. 10.1007/s00261-017-1274-5. [DOI] [PubMed]
- 13.Iwase, T. et al. The prognostic impact of body composition for locally advanced breast cancer patients who received neoadjuvant chemotherapy. Cancers (Basel)13(4), 608. 10.3390/cancers13040608 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Prado, C. M. et al. Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin. Cancer Res.15(8), 2920–2926. 10.1158/1078-0432.CCR-08-2242 (2009). [DOI] [PubMed] [Google Scholar]
- 15.Shachar, S. S. et al. Skeletal muscle measures as predictors of toxicity, hospitalization, and survival in patients with metastatic breast cancer receiving taxane-based chemotherapy. Clin. Cancer Res.23(3), 658–665. 10.1158/1078-0432.CCR-16-0940 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kazemi-Bajestani, S. M., Mazurak, V. C. & Baracos, V. Computed tomography-defined muscle and fat wasting are associated with cancer clinical outcomes. Semin. Cell Dev. Biol.54, 2–10. 10.1016/j.semcdb.2015.09.001 (2016). [DOI] [PubMed] [Google Scholar]
- 17.Shachar, S. S., Williams, G. R., Muss, H. B. & Nishijima, T. F. Prognostic value of sarcopenia in adults with solid tumours: A meta-analysis and systematic review. Eur. J. Cancer57, 58–67. 10.1016/j.ejca.2015.12.030 (2016). [DOI] [PubMed] [Google Scholar]
- 18.Villaseñor, A. et al. Prevalence and prognostic effect of sarcopenia in breast cancer survivors: the HEAL Study. J. Cancer Surv.6(4), 398–406. 10.1007/s11764-012-0234-x (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rier, H. N. et al. Low muscle attenuation is a prognostic factor for survival in metastatic breast cancer patients treated with first line palliative chemotherapy. Breast31, 9–15. 10.1016/j.breast.2016.10.014 (2017). [DOI] [PubMed] [Google Scholar]
- 20.Aleixo, G. F. P., Williams, G. R., Nyrop, K. A., Muss, H. B. & Shachar, S. S. Muscle composition and outcomes in patients with breast cancer: Meta-analysis and systematic review. Breast Cancer Res. Treat.177(3), 569–579. 10.1007/s10549-019-05352-3 (2019). [DOI] [PubMed] [Google Scholar]
- 21.Deluche, E. et al. Impact of body composition on outcome in patients with early breast cancer. Support Care Cancer26(3), 861–868. 10.1007/s00520-017-3902-6 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Caan, B. J. et al. Association of muscle and adiposity measured by computed tomography with survival in patients with nonmetastatic breast cancer. JAMA Oncol.4(6), 798–804. 10.1001/jamaoncol.2018.0137 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Prado, C. M. et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: A population-based study. Lancet Oncol.9(7), 629–635. 10.1016/S1470-2045(08)70153-0 (2008). [DOI] [PubMed] [Google Scholar]
- 24.Martin, L. et al. Cancer cachexia in the age of obesity: Skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J. Clin. Oncol.31(12), 1539–1547. 10.1200/JCO.2012.45.2722 (2013). [DOI] [PubMed] [Google Scholar]
- 25.Battisti, S. et al. Modification of abdominal fat distribution after aromatase inhibitor therapy in breast cancer patients visualized using 3-D computed tomography volumetry. Clin. Breast Cancer14(5), 365–370. 10.1016/j.clbc.2014.02.003 (2014). [DOI] [PubMed] [Google Scholar]
- 26.Bradshaw, P. T. et al. Adipose tissue distribution and survival among women with nonmetastatic breast cancer. Obesity (Silver Spring)27(6), 997–1004. 10.1002/oby.22458 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kim, M. S., Choi, Y. J. & Lee, Y. H. Visceral fat measured by computed tomography and the risk of breast cancer. Transl. Cancer Res.8(5), 199–199. 10.21037/tcr.2019.09.16 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kwon, M. R. et al. Impact of skeletal muscle loss and visceral obesity measured using serial CT on the prognosis of operable breast cancers in Asian Patients. Korean J. Radiol.23(2), 159–171. 10.3348/kjr.2020.1475 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gradishar, W. J. et al. Breast cancer, version 3.2022, NCCN clinical practice guidelines in oncology. J. Natl. Compr. Cancer Netw.20(6), 691–722. 10.6004/jnccn.2022.0030 (2022). [DOI] [PubMed] [Google Scholar]
- 30.Schweitzer, L. et al. What is the best reference site for a single MRI slice to assess whole-body skeletal muscle and adipose tissue volumes in healthy adults?. Am. J. Clin. Nutr.102(1), 58–65. 10.3945/ajcn.115.111203 (2015). [DOI] [PubMed] [Google Scholar]
- 31.Dabiri, S. et al. Muscle segmentation in axial computed tomography (CT) images at the lumbar (L3) and thoracic (T4) levels for body composition analysis. Comput. Med. Imaging Graph.75, 47–55. 10.1016/j.compmedimag.2019.04.007 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (2017) version 5.0. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_5x7.pdf. Accessed 20 Jan 2023.
- 33.Solomayer, E. F. et al. Muscle mass loss in patients with metastatic breast cancer. Arch. Gynecol. Obstet.300(1), 201–206. 10.1007/s00404-019-05168-y (2019). [DOI] [PubMed] [Google Scholar]
- 34.Rolland, Y. et al. Sarcopenia: Its assessment, etiology, pathogenesis, consequences and future perspectives. J. Nutr. Health Aging12(7), 433–450. 10.1007/BF02982704 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kim, S. I. et al. Prognostic implications of body composition change during primary treatment in patients with ovarian cancer: A retrospective study using an artificial intelligence-based volumetric technique. Gynecol. Oncol.162(1), 72–79. 10.1016/j.ygyno.2021.05.004 (2021). [DOI] [PubMed] [Google Scholar]
- 36.Arora, K. K. et al. Increasing adipose burden in young men with metastatic testicular cancer following front-line cytotoxic chemotherapy. J. Clin. Oncol.37(7_suppl), 525–525. 10.1200/jco.2019.37.7_suppl.525 (2019).30592639 [Google Scholar]
- 37.Tsai, S. Importance of lean body mass in the oncologic patient. Nutr. Clin. Pract.27(5), 593–598. 10.1177/0884533612457949 (2012). [DOI] [PubMed] [Google Scholar]
- 38.Williams, G. R., Rier, H. N., McDonald, A. & Shachar, S. S. Sarcopenia and aging in cancer. J. Geriatr. Oncol.10(3), 374–377. 10.1016/j.jgo.2018.10.009 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Baumgartner, R. N. et al. Epidemiology of sarcopenia among the elderly in New Mexico. Am. J. Epidemiol.147(8), 755–763. 10.1093/oxfordjournals.aje.a009520 (1998). Erratum in: Am J Epidemiol 1999 Jun 15;149(12):1161. [DOI] [PubMed]
- 40.Knight, K., Wade, S. & Balducci, L. Prevalence and outcomes of anemia in cancer: A systematic review of the literature. Am. J. Med.5(116 Suppl 7A), 11S-26S. 10.1016/j.amjmed.2003.12.008 (2004). [DOI] [PubMed] [Google Scholar]
- 41.Tiidus, P. M., Lowe, D. A. & Brown, M. Estrogen replacement and skeletal muscle: Mechanisms and population health. J. Appl. Physiol.115(5), 569–578. 10.1152/japplphysiol.00629.2013 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Reed, M. J., Cheng, R. W., Noel, C. T., Dudley, H. A. & James, V. H. Plasma levels of estrone, estrone sulfate, and estradiol and the percentage of unbound estradiol in postmenopausal women with and without breast disease. Cancer Res.43(8), 3940–3943 (1983). [PubMed] [Google Scholar]
- 43.Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin.74(1), 12–49. 10.3322/caac.21820 (2024). Erratum in: CA Cancer J Clin. 2024 Mar-Apr;74(2):203. 10.3322/caac.21830. [DOI] [PubMed]
- 44.Allemani, C. et al. Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): Analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet391(10125), 1023–1075. 10.1016/S0140-6736(17)33326-3 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tyrovolas, S. et al. The role of muscle mass and body fat on disability among older adults: A cross-national analysis. Exp. Gerontol.69, 27–35. 10.1016/j.exger.2015.06.002 (2015). [DOI] [PubMed] [Google Scholar]
- 46.Beavers, K. M. et al. Effect of an 18-month physical activity and weight loss intervention on body composition in overweight and obese older adults. Obesity (Silver Spring)22(2), 325–331. 10.1002/oby.20607 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Srikanthan, P. & Karlamangla, A. S. Muscle mass index as a predictor of longevity in older adults. Am. J. Med.127(6), 547–553. 10.1016/j.amjmed.2014.02.007 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bauckneht, M. et al. Opportunistic skeletal muscle metrics as prognostic tools in metastatic castration-resistant prostate cancer patients candidates to receive Radium-223. Ann. Nucl. Med.36(4), 373–383. 10.1007/s12149-022-01716-w (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cruz-Jentoft, A. J. et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing48(1), 16–31. 10.1093/ageing/afy169 (2019). Erratum in: Age Ageing. 2019 Jul 1;48(4):601. 10.1093/ageing/afz046. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.