Abstract
The role of contrast-enhanced ultrasound (CEUS) imaging is being widely explored by various groups for its use in the pediatric population. Clinical implementation of new diagnostic or therapeutic techniques requires extensive and meticulous preclinical testing and evaluation. The impact of CEUS will be determined in part by the extent to which studies are oriented specifically toward a pediatric population. Rather than simply applying principles and techniques used in the adult population, these studies are expected to advance and augment preexisting knowledge with pediatric-specific information. To further develop this imaging modality for use in children, pediatric-focused preclinical research is essential. In this paper we describe the development and implementation of the pediatric-specific preclinical animal and phantom models that are being used to evaluate CEUS with the goal of clinical translation to children.
Keywords: Children, Contrast-enhanced ultrasound, Fetus, Neonates, Phantom, Translational research, Ultrasound, Ultrasound contrast agents
Introduction
In March 2016, the United States Food and Drug Administration (FDA) approved the intravenous administration of the ultrasound contrast agent (UCA) Lumason (Bracco Diagnostics Inc., Monroe Township, NJ) for characterization of focal liver lesions in both adult and pediatric patients. The following December, the FDA approved intravesicular use of this same UCA for ultrasound (US) of the urinary tract in children, this time for the purpose of evaluating suspected or known vesicoureteral reflux. By using the newly approved UCA, clinicians could avoid radiation exposure in these children who would otherwise have undergone fluoroscopic voiding cystourethrography [1, 2]. CEUS is increasingly used in children for a variety of applications, propelled by the FDA approvals in 2016. The most common of the intravenous applications is the indication for evaluating focal liver lesions, though CEUS is also being used for assessing blunt abdominal trauma in children, monitoring liver transplants and assessing bowel wall in the case of Crohn disease [3-10]. More recently, CEUS has been implemented in the evaluation of hypoxic–ischemic brain injury in neonates and infants, showing promise for characterizing this type of injury [11, 12].
Contrast-enhanced US’s popularity as an imaging modality for children continues to grow. Rapid advancements in US technology and development of pediatric-focused US transducers are broadening its potential applications. For example, some of the larger manufacturers of US equipment are producing pediatric-specific high-frequency and small-footprint transducers with CEUS capabilities that are already in clinical use, thus increasing opportunities to employ CEUS to image neonatal diseases. This impetus for innovation and research is fueled by CEUS’s distinct advantages for pediatric applications. Its excellent safety profile and lack of ionizing radiation, as well as its bypassing of sedation and utility for bedside examinations, make CEUS optimal for imaging children, particularly the sickest and most vulnerable patients. Here we provide an overview of the pediatric-focused CEUS translational research that is driving the exploration of new applications in this patient population.
Pediatric tumor models
Because angiogenesis (the generation of new blood vessels) is necessary for tumor development, growth and metastasis, the assessment of tumor blood flow is an important area of investigation in oncology [13, 14]. Modern CEUS software systems are able to quantify tumor blood flow, allowing an objective assessment at diagnosis and a potential method of monitoring response during neoadjuvant therapy. The fact that UCAs are completely intravascular makes them most suitable for this purpose. Quantification of CEUS, based either on the measurement of the UCA signal intensity at a specific time point in a study or on a continuous measurement over the duration of a period of time beginning immediately after the UCA injection, is used to extrapolate perfusion parameters based on intensity (i.e. peak enhancement or enhancement at a specific time point) and time (i.e. time-to-peak, wash-in rate, washout rate, arrival time, washout time). These parameters can aid in identification and quantification of lesion vascularity; they are especially valuable in children treated with antiangiogenic agents, which can restrict tumor vasculature without causing tumors to decrease in size [15]. In these cases, the standard method of evaluating tumor response to therapy (i.e. measurement of tumor size) might not give a complete picture of the impact of treatment. If CEUS could be used to depict decreased tumor vascularity following pharmacotherapy, clinicians could use this information to assess treatment efficacy regardless of the effect on lesion size, allowing for more timely adjustments in treatment regimens.
In a preclinical trial of CEUS in a murine model of neuroblastoma treated with several antiangiogenic agents, peak enhancement and rate of enhancement decreased significantly in size-matched treated tumors when compared to control tumors. Additionally, the rate of enhancement was positively correlated with histological measurements of vessel density in both treated and control groups [16]. In another preclinical trial using a murine model of alveolar rhabdomyosarcoma, peak enhancement in tumors treated with interferon-β (IFN-β) was significantly higher than in controls, suggesting that IFN-β, at least transiently, increases tumor vascularity [17]. These results support the feasibility of using CEUS as a biomarker of tumor blood flow in pediatric tumors, which might allow for improved monitoring of treatment response, independent of lesion size.
However, direct translation of quantitative CEUS from small animal models or adult applications into clinical pediatric oncology trials has some obstacles. For example, differences in patient body habitus and tumor location make intersubject comparisons difficult. Often in small animal tumor imaging studies (in mice and rats), tumor growth happens in a controlled fashion and therefore provides an ideal tumor environment for imaging studies, which is not the case in humans. Direct translation of these types of tumor imaging studies is not necessarily a linear process.
Thus, efforts to accurately and reproducibly measure tumor blood flow with CEUS and to standardize the procedure of quantitative CEUS in clinical trials are an ongoing initiative of the Radiological Society of North America (RSNA) Quantitative Imaging Biomarkers Alliance Ultrasound Committee [18].
Neonatal brain models
Hypoxic–ischemic neonatal brain injury is a relatively common cause of morbidity and mortality in newborns. Even term infants who recover from acute hypoxic events can develop hypoxic–ischemic encephalopathy (HIE), which occurs in 2.5 of 1,000 live births, and many have detrimental long-term neurologic sequelae including cognitive disabilities, seizures and cerebral palsy [19]. Early detection is crucial for timely neuroprotective interventions. MRI is considered the primary imaging modality to evaluate the brain; however, mobilizing critically ill neonates to MRI can be difficult or impossible. Given the lack of sensitivity of gray-scale US in evaluating for ischemic changes, CEUS is an emerging application that is receiving increasing attention in evaluating the neonatal brain. The unique pathophysiology of neonatal diseases can be better characterized with CEUS than with other imaging modalities. Thanks to the small size of the microbubbles (at 2–3 μm, they are smaller than red blood cells), UCA can provide real-time imaging of microvessels in neonates, suggesting potential applications in evaluating cerebral perfusion.
To fully utilize CEUS qualitatively and quantitatively in the neonatal brain setting, it is crucial that animal or phantom models be used to validate CEUS-based perfusion parameters by establishing normative values and expected variations that reflect alterations in physiology. Numerical values of extrapolated parameters — such as peak enhancement, time-to-peak and wash-in and washout characteristics — might vary among subjects and organ/tissue types, or even depending on depth of imaging. Repeated measures within subjects to assess for interval changes in these parameters might provide useful data to create a reference standard of perfusion values for CEUS. To that end, a neonatal porcine model has been developed that takes advantage of the anatomical, physiological and functional similarities between neonatal piglet and human neonatal brains [20-23]. The porcine animal model is more accessible and affordable than the non-human primate model [24]. Compared to small animal models such as mice or rats, piglets possess marked similarities to human neonatal brain morphology and maturational stages. As in humans, the major brain growth in pigs occurs from the late prenatal to the postnatal period [23]. The brain size, gyrencephalic morphology and gray/white matter ratio of piglets are comparable with that of human neonates. Moreover, the physical size of the piglets enables direct application of the same imaging technique and therapeutic instruments that are used in humans, facilitating a smooth clinical translation.
Research using piglet models builds a crucial translational bridge to discovering important mechanisms and therapeutic targets in human neonates. The earliest of these studies demonstrated lack of heat damage when using UCAs, thus providing safety data for future investigations and establishing a foundation for human research [25]. Currently, the neonatal porcine model is being used to assess brain perfusion characteristics at baseline compared with those parameters obtained after a brain injury [26]. In a recent study, an acoustic window was created in the skull of a neonatal pig by using a burr hole to mimic a fontanelle (piglets lack the anterior fontanelle observed in human neonates). Then, CEUS imaging was performed using an intravenous injection of microbubbles, similarly to how it is done in humans [26]. UCA was delivered by both bolus injection and infusion methods to evaluate the ability of each technique to produce quantifiable perfusion information. Continuous hemodynamic monitoring was also performed during the course of the study to identify any possible effect of the administration of a UCA on the hemodynamic stability of these piglets. The results from this ongoing study are expected to provide crucial insight into the perfusion alterations in the brain in the setting of hypoxic–ischemic or traumatic injury.
The piglet model has also been useful in showing ventricular patency following intraventricular UCA administration, thus providing valuable information about the diagnosis and treatment efficacy in the setting of neonatal hydrocephalus [27]. These studies provide important information regarding application of CEUS in evaluation of the normal neonatal brain as well as pathological aberrations. If these applications are translated to human neonates, they could potentially improve disease characterization and management.
Fetal models
Premature birth is associated with high morbidity and mortality owing to immaturity of critical organs. In addition to being susceptible to cerebral hypoxic injury, poor outcomes in premature infants are associated with the underdevelopment of the fetal lungs. While the previously discussed porcine models offer similar characteristics when compared to humans for investigation of neonatal CEUS applications in the brain, the ovine model provides the most similar correlate in regard to fetal brain and lung development [28-30]. In the hopes of mitigating the detrimental effects of an early delivery there has been longstanding exploration of an artificial environment or “artificial placenta” to support the preterm infant; already this has been developed in ovine animal models, and translation to humans is anticipated in the near future [31-33].
The unique support equipment used in these extrauterine environments requires that any imaging evaluation be performed by a single imaging modality: US. But CEUS can be used in these same studies to augment gray-scale US and provide both anatomical and functional information about fetal organs, particularly their development and maturity. CEUS can provide physiological assessment in these extrauterine environments and potentially inform delivery planning. Preliminary studies have demonstrated excellent safety outcomes in one system of extracorporeal support, with attention given to interaction of the UCA with the oxygenator membrane and to CEUS’s influence on animal hemodynamic parameters [34]. Additional initial CEUS examinations have established the optimal UCA dose for visualizing the fetal sheep brain and quantifying perfusion; given these results, researchers anticipate a wide application of CEUS to better understand fetal physiology and development (Fig. 1). While early in investigation, the expectation is that soon these animal models will be translated to human fetal imaging, potentially both in utero and in children being maintained on extrauterine support.
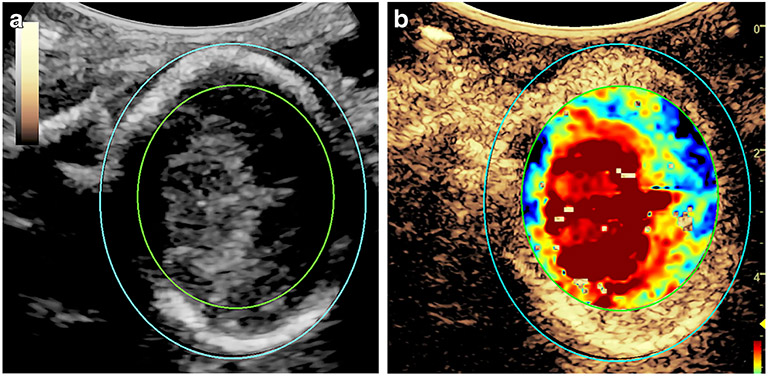
Fig. 1.
Dual-screen transcranial US image (coronal plane) through the fetal lamb (108 days of gestation) brain using VueBox (Bracco, Milan, Italy) analysis software. a Gray-scale US image for localization. b Contrast-enhanced US image with color map represents peak enhancement in the region of interest included in the green circle
Testicular models
Acute scrotal pain is a common symptom, developing in approximately 1 in 160 males by the age of 25 years. Testicular torsion, the most serious cause of acute scrotal pain, has an incidence of about 1 in 4,000 [35]. Testicular torsion occurs most often in children, with a peak incidence in neonates and adolescents [36]. Clinical examination of the scrotum is often unreliable in children because of poor cooperation, small testicular size, and the frequent presence of a reactive hydrocele. Because testicular infarction can occur within hours of the onset of pain, diagnosis must be rapid and accurate to identify patients requiring urgent surgical intervention [37]. The traditional imaging examination of choice for the evaluation of these cases of acute scrotal pain is color Doppler US [38-41]. However, in children its use might be limited by the difficulty of detecting blood flow in small-volume testicles [42-46]. CEUS can improve sensitivity to flow in small blood vessels, and several publications describe its successful use in the evaluation of acute scrotal symptoms in adults [47-54]. To date, there have been no studies of CEUS in a pediatric population with suspected testicular torsion.
In the hope of improving diagnostic accuracy in testicles of small volume, a number of experimental models of testicular torsion and ischemia have been developed. These studies evaluated both qualitative and quantitative CEUS methods of assessing testicular blood flow. In an initial investigation, CEUS was used to quantify testicular blood flow in a rabbit model. In this model, progressive degrees of unilateral spermatic cord constriction were experimentally induced, ranging from mild to moderate and severe vascular occlusion [55]. Doppler US and qualitative CEUS were used to evaluate and compare perfusion of the testicle with spermatic cord occlusion and the normal contralateral testicle (Fig. 2). In addition, the perfusion ratios between the experimentally occluded and control testicles were calculated using CEUS parenchymal enhancement data at baseline and at different degrees of vascular occlusion. These ratios were compared with the radiolabeled-microsphere technique, which is the reference standard for testicular perfusion. The study showed that CEUS could detect different degrees of testicular perfusion more accurately than Doppler US, and that quantitative CEUS was superior to qualitative visual assessment of CEUS images. The perfusion ratios derived from quantitative CEUS analysis were comparable to the reference radiolabeled-microsphere technique. These initial experimental imaging findings support the use of CEUS for accurate identification of testicular vascular compromise. In this study, a single two-dimensional (2-D) plane was used for CEUS image acquisition; however, this might not be representative of volumetric testicular perfusion.
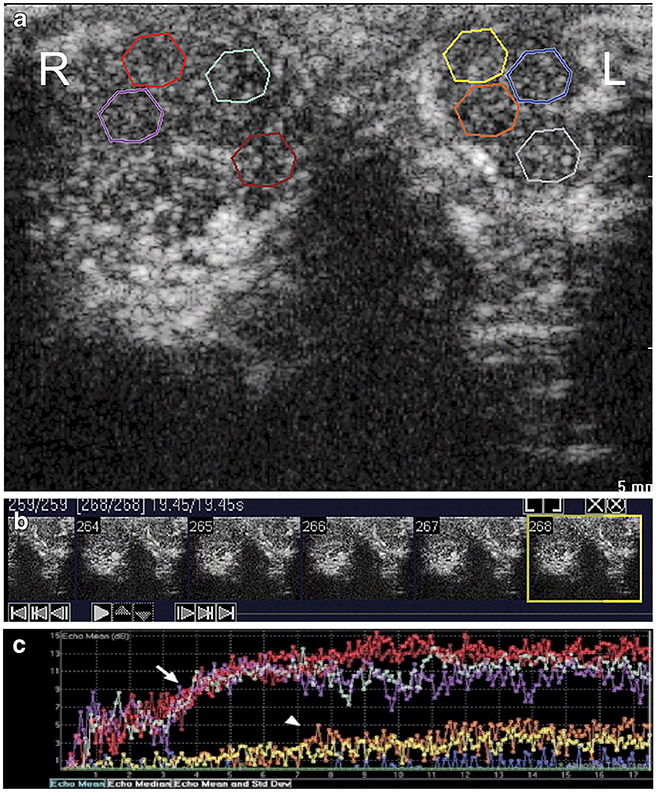
Fig. 2.
Severe left spermatic cord occlusion. a Final frame from contrast-enhanced ultrasound (CEUS) imaging sequence shows four regions of interest within the right (R) and left (L) testicles in a rabbit model of testicular ischemia. The experimentally occluded left testicle appears swollen and hypoechoic compared with the contralateral normal right testicle and is surrounded by a small hydrocele. b Individual image frames are shown, with the final frame corresponding to (a), outlined with yellow box. c Corresponding testicular replenishment curves. There is a significant delay in the rate of rise of the left testicular replenishment curves (arrowhead) compared with the right testicular curves (arrow). A decrease in the amplitude of the plateau levels of the left testicle, as compared with the right testicle, is also seen. Reprinted with permission from [55]
In an attempt to better quantify blood flow through a larger portion of the testicle, a follow-up study was performed by the same group using a multi-planar CEUS technique to estimate volumetric testicular perfusion in torsed and normal rabbit testicles. As in the previous study, CEUS imaging findings were compared with the reference standard radiolabeled-microsphere technique [56]. Standard linear US transducers were used to sample blood flow measurements from five tissue planes per testicle, three transverse and two longitudinal. Measurements were obtained at baseline and at specific time intervals during an 8-h period following surgically induced testicular torsion. Testicular perfusion ratios between the torsed and normal testicles derived from quantitative CEUS were averaged from the transverse, longitudinal and combined transverse/longitudinal planes and compared to the radiolabeled-microsphere blood flow ratios (Fig. 3). The results from this study showed a high correlation of the testicular perfusion between the multiplanar CEUS and radiolabeled-microsphere techniques. These findings further supported the use of multiplanar CEUS for more accurate evaluation of testicular blood flow.
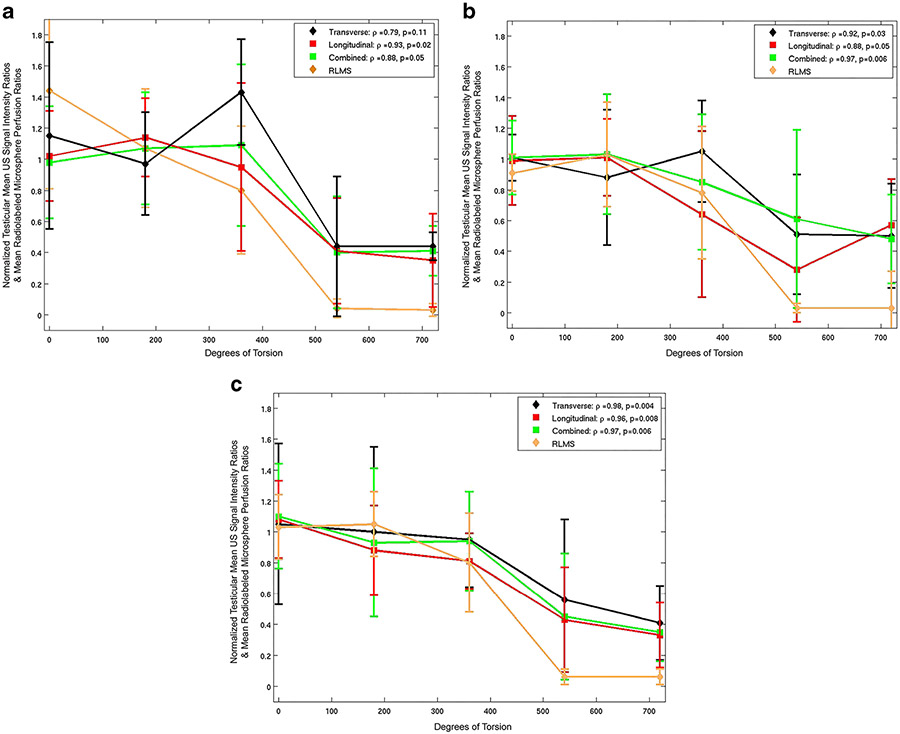
Fig. 3.
Testicular mean US pixel intensity ratios (y-axis) in transverse (black), longitudinal (pink) and combined transverse and longitudinal (green) planes at different degrees of surgically induced testicular torsion (x-axis) follow a trend resembling that of the reference standard mean radiolabeled microsphere (RLMS) blood flow ratios (orange) at each degree of torsion. a–c Measurements obtained immediately (a) and 4 h (b) and 8 h (c) after surgery. There was no consistent improvement in correlation when data from the transverse and longitudinal planes were combined over those obtained from separate analysis of data from the averaged transverse planes and averaged longitudinal planes, although the weakest correlation was seen with US data derived from the averaged transverse planes alone in the immediate postoperative period. Reprinted with permission from [56]
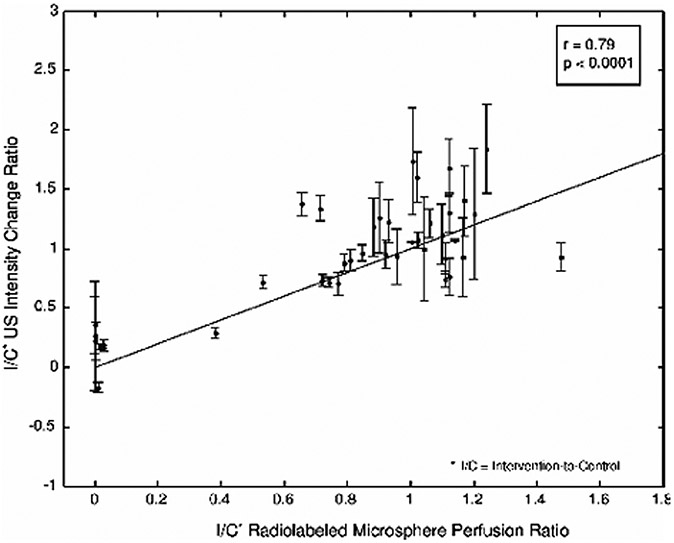
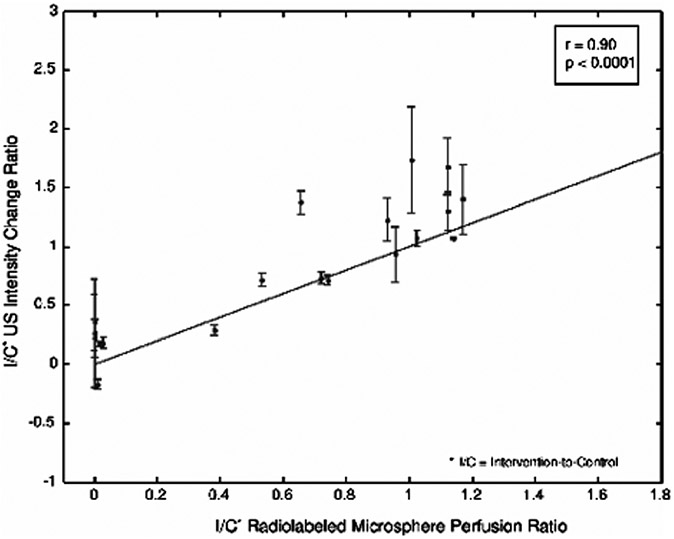
The relatively recent innovation of matrix-phased-array US transducers for three-dimensional (3-D) imaging with the capability of quantification analysis permits real-time interrogation of a tissue volume. This is expected to provide even better evaluation of organs such as the testicle and overcome the limitations of the quantitative multiplanar sampling technique used in the earlier experimental studies. Therefore, a third experimental study from the same group in the field of testicular perfusion expanded on their earlier experience with 2-D and multiplanar CEUS quantification of blood flow within torsed and normal rabbit testicles [57]. The hypothesis of this study was that because slight spatial variations in blood flow might occur within the volume of an organ, 3-D assessment of perfusion over time might provide a potential method for achieving four-dimensional (4-D) perfusion information. In this study, a 2-D matrix-phased-array US transducer was employed and UCA was administered over 2 min using a continuous infusion technique both at baseline and after surgically induced torsion [57]. Volumetric data were acquired at 1-s sampling intervals over the 2-min period during the wash-in phase of contrast. Results from this study showed a strong correlation of 3-D CEUS testicular parenchymal signal intensity measurements with tissue blood flow measurements obtained with radiolabeled microspheres (Figs. 4, 5 and 6). The series of these experimental studies provide ample supportive evidence of CEUS’s capabilities in the evaluation of testicular perfusion.
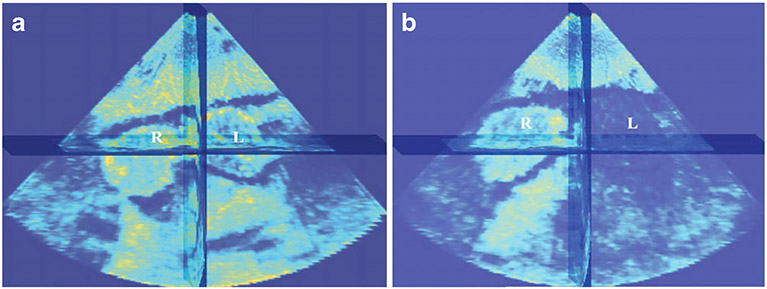
Fig. 4.
Real-time three-dimensional (3-D) assessment of blood flow from a tissue volume obtained using a 2-D matrix-phased-array US transducer in a rabbit model of unilateral testicular torsion. a, b The cumulative change in signal intensity for the right (R) and left (L) testicles is depicted at baseline (a) and after 720° of left-side torsion (b), where there is a visually obvious decrease in cumulative signal intensity in the left testicle compared to the normal right testicle. The brightest signal intensity in yellow corresponds to the highest amount of US contrast agent signal and the lowest is represented by the blue areas. Reprinted with permission from [57]
Fig. 5.
Baseline values for testicular perfusion. Scatter plot shows the contrast-enhanced US intensity change ratios (y-axis) compared to the reference standard radiolabeled microsphere testicular perfusion ratios (x-axis) between the intervention and control testicles at baseline and postoperatively for different degrees of torsion. I/C intervention (testis)/control (testis). Reprinted with permission [57]
Fig. 6.
Postoperative values for testicular perfusion. Scatter plot shows the contrast-enhanced US intensity change ratios (y-axis) compared to the reference standard radiolabeled microsphere testicular perfusion ratios (x-axis) between the intervention and control testicles restricted to postoperative values at different degrees of torsion. I/C intervention (testicle)/control (testicle). Reprinted with permission [57]
Pediatric contrast-enhanced voiding urosonography teaching phantoms
Vesicoureteral reflux (VUR), the most frequently detected problem of the pediatric urinary tract, is diagnosed in 25% to 40% of children and 20% of neonates presenting with a urinary tract infection [58, 59]. VUR is graded according to the degree of dilation and morphology of the renal collecting system, from grade 1 (mild) to grade 5 (severe) [60]. The diagnostic imaging methods available to evaluate for VUR in children include voiding cystourethrography (VCUG), radionuclide cystography and, more recently, contrast-enhanced VUS. With the advent of contrast-enhanced VUS, it is possible to diagnose VUR in children using a highly sensitive radiation-free examination. The intravesicular administration of UCA required by contrast-enhanced VUS has been shown to provide high concordance with VCUG in VUR detection and grading [1, 61-63]. Since the FDA approved the intravesicular use of UCAs for contrast-enhanced VUS in children, there has been a rising interest among physicians and US technologists in learning how to perform the contrast-enhanced VUS examination, which raises the question of how to appropriately educate sonographers and providers how to perform it.
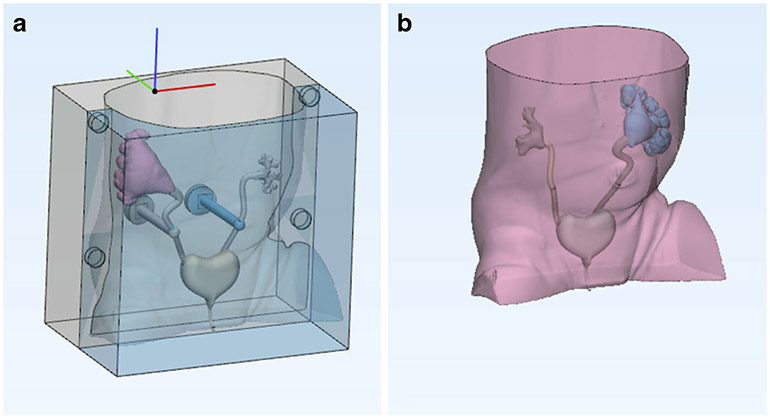
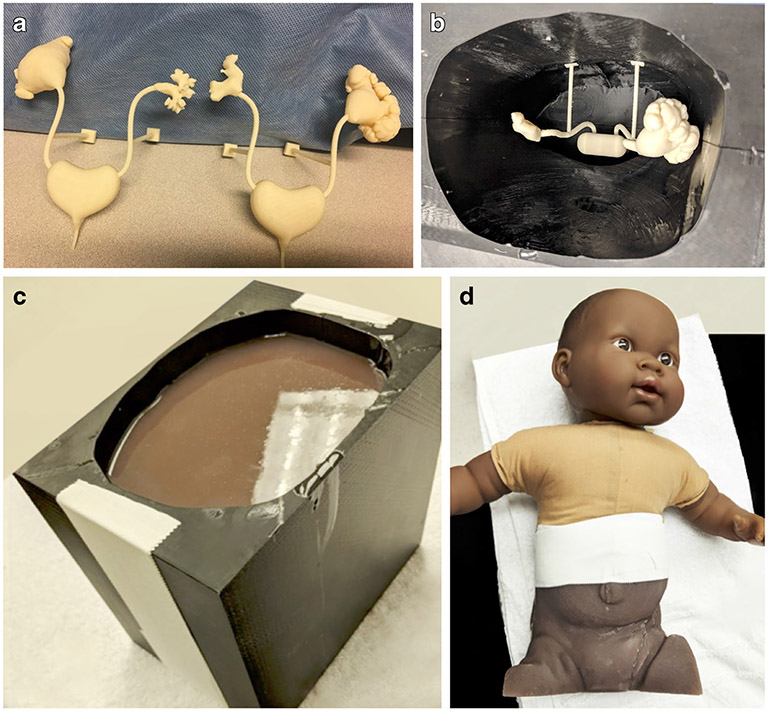
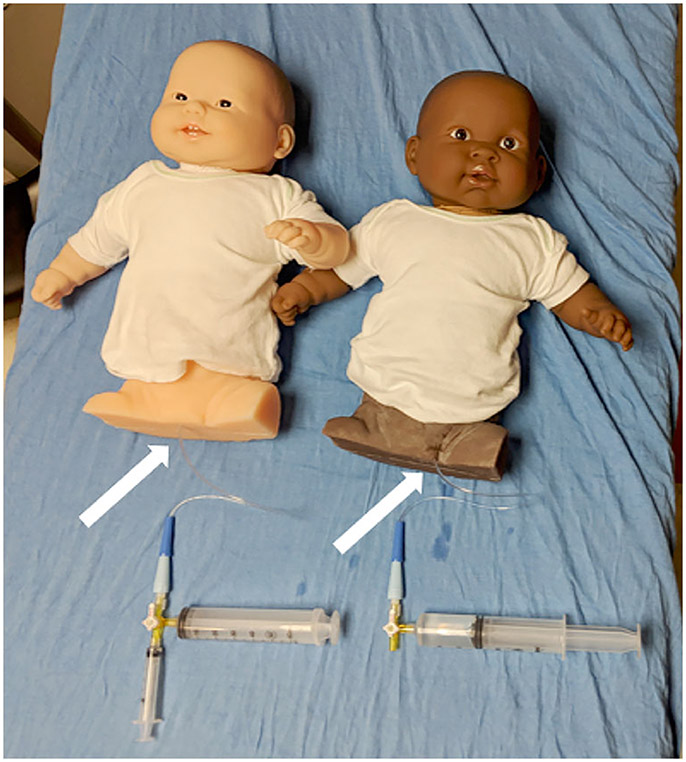
One solution to the problem of contrast-enhanced VUS training could come from US phantoms that depict VUR. Although these phantoms that aid in education and training are not commercially available, 3-D-printed phantoms are being used as educational tools. At Children’s Hospital of Philadelphia (Philadelphia, PA), where these phantoms were developed with an internal 3-D printing lab, they to simulate the grades of VUR during a contrast-enhanced VUS examination [64]. To achieve this goal, two sets of kidneys were created simulating the five grades of reflux that involve the upper urinary tract (Fig. 7). The two renal models were printed in polyvinyl alcohol and an external mold was printed in acrylonitrile butadiene styrene (Fig. 8). Silicone (Smooth-on; Ecoflex 30, Macungie, PA) was poured into the acrylonitrile butadiene styrene mold of an infantile abdomen and pelvis, embedding the polyvinyl alcohol parts (Fig. 8). The polyvinyl alcohol parts were then dissolved or broken away, leaving behind cavities of the renal anatomy in the silicone. The upper torso and head of a baby doll were attached to the silicone abdomen and pelvis to provide a holistic infant simulation, and a catheter was placed in the urethra to enable filling (Figs. 8 and 9). US imaging confirmed visualization of the kidney, ureters and bladder in a single image, resembling the results of clinical examinations. At this point, UCA and saline were administered via catheter for successful visualization of the simulated reflux (Fig. 10). The phantoms that were created are reusable and can continue to serve as a versatile teaching tool to demonstrate and practice the contrast-enhanced VUS procedure, as well as depict the appearance of different grades of VUR in children. Such an accessible educational channel might be expanded to include training for sonographers and providers in other applications of CEUS in children.
Fig. 7.
Kidney models. Three-dimensional model of two contrast-enhanced voiding urosonography phantoms on 3-matic software (Materialise NV, Leuven, Belgium). a Kidney model simulates vesicoureteral reflux (VUR) grades 4 (right) and 2 (left). b Kidneymodel simulates VUR grades 3 (right) and 5 (left)
Fig. 8.
Vesicoureteral reflux (VUR) models. a Three-dimensional (3-D)-printed kidney polyvinyl alcohol models depict VUR grades 4 and 2 (left) and VUR grades 3 and 5 (right). b 3-D-printed external acrylonitrile butadiene styrene mold of an infantile abdomen and pelvis (black), and kidney model (white) embedded inside the mold. c Silicone poured into the acrylonitrile butadiene styrene mold with the polyvinyl alcohol parts embedded. d Contrast-enhanced voiding urosonography phantom with the upper torso and head of a baby doll attached to the silicone abdomen and pelvis
Fig. 9.
Voiding urosonography phantoms. Catheters are placed in the urethra (arrows) of the two contrast-enhanced voiding urosonography phantoms to enable filling
Fig. 10.
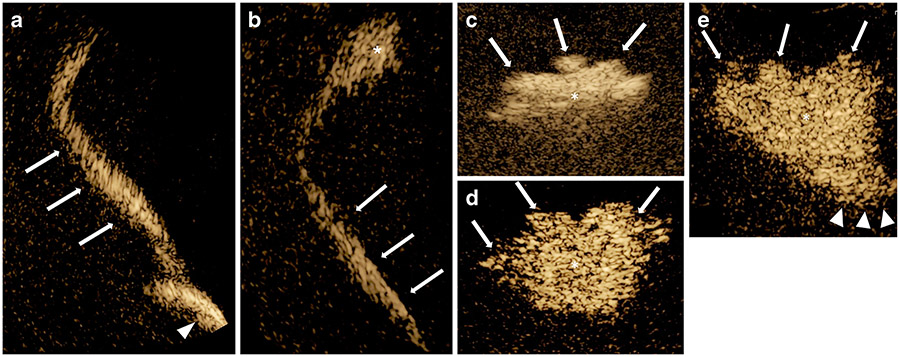
Contrast-specific US images of contrast-enhanced voiding urosonography (VUS) phantoms depict the five grades of vesicoureteral reflux (VUR) after contrast-saline solution injection. a VUR grade 1, coronal scan from the flank. Microbubbles are visible in the bladder (arrowhead) and in the ureter (arrows). b VUR grade 2. Microbubbles are visible within the ureter (arrows) and the renal pelvis (asterisk), with no significant pelvic dilation. c VUR grade 3. Microbubbles are in the pelvicalyces (asterisk), with significant pelvic dilation and moderate calyceal dilation (arrows). d VUR grade 4. Microbubbles are in the pelvicalyces (asterisk), with significant pelvic and severe calyceal dilation (arrows). e VUR grade 5. Microbubbles appear in the pelvicalyces (asterisk), with significant pelvicalyceal dilation, loss of pelvicalyceal contour (arrows) and dilated proximal tortuous ureter (arrowheads)
Future direction of pediatric contrast-enhanced ultrasound research
Classic development of new imaging and therapeutic agents used in medicine involves an initial exploration with in vitro and animal models. Typically, after demonstrating safety and efficacy in pre-clinical models, researchers introduce new experimental treatments/technologies in adults and then slowly introduce them in the pediatric population. UCAs are no exception to this rule and have followed this pathway of evolution. At present, two primary trends in the development of UCAs can be identified: one method is focused on extending the diagnostic imaging capabilities of the microbubbles, and the second method seeks to evaluate CEUS’s therapeutic applications.
Novel CEUS imaging techniques that could improve diagnostic imaging capabilities rely on UCA design and software development. Manufacturers are attempting to increase the endurance of the encapsulating shell of the microbubble to prolong its lifetime and improve its scattering properties, as well as to produce uniform-size microbubble populations [65, 66]. After these improvements, it is now possible to implement super-harmonic imaging techniques that can produce high-resolution images of microvascular structures as small as 150 μm in animal studies [67]. These methods use low frequencies to excite the microbubbles and high frequencies to detect their signal [67, 68]. The new developments offer a promising approach for evaluating smaller structures, particularly for applications in pediatric patients.
The development of US and UCAs for molecular imaging for both diagnostic and therapeutic purposes is more advanced and requires modifications to the physical and chemical properties of microbubbles. Essentially, the microbubbles are combined with targeting ligands that have an affinity toward specific vascular biomarkers of inflammation or neovascularization. This method allows the signal from the molecularly targeted microbubbles to be selectively localized, and it also makes it possible to quantify the molecular target concentration [69]. Applications of this type of US molecular imaging have been explored in atherosclerosis, inflammatory bowel disease and cancer (ovary, breast and prostate) [70-72]. However, this ongoing area of research is relatively new, and future studies are expected to shed light on its potential applications in children.
Another field of research in its infancy is investigating how microbubble transport/drug-loading capabilities can be utilized for therapeutic applications. Treatment agents can be directly attached to the external surface of the microbubbles, or they can be associated with liposomes or nanoparticles on the microbubble surface [73]. If the properties of microbubbles are properly utilized, they can selectively deliver drugs or genes to a target site with relatively high efficacy and without potential injury or intoxication of healthy cells.
Finally, another therapeutic application in development is exploring how drug uptake and biodistribution into target tissues can be enhanced by the sonoporation phenomenon. Specific US waves produced by the transducer can lead to the volumetric oscillations of microbubbles and their inertial cavitation [74]. By this mechanism, microbubbles expand larger than their critical size and then collapse into smaller microbubbles [74, 75]. This collapse generates shock waves and microjets (a jet of liquid produced during a cavitation event), inducing sonoporation of the cell membrane and making the blood vessels permeable [76]. In a previous study, paclitaxel-liposome-microbubble complexes with a core of perfluoropropane (C3F8) were used for US-triggered targeted chemotherapy against breast cancer; the treatment was considered effective, with a 70% reduction in tumor volume [77]. More recently, a large number of pre-clinical studies have demonstrated the utility of CEUS as an effective tool for drug delivery in atherosclerosis, myocardial ischemia, inflammatory bowel disease, cardiovascular disease and cancer (liver, pancreatic, renal, ovarian) [71, 78-81]. It is expected that as these studies continue to be evaluated and improved, their results will eventually be translated to the clinical space for application in adults and subsequently in children.
Conclusion
The many successful applications of CEUS in pediatric patients have established a propitious foundation for the future of pediatric imaging. While several avenues of technological development are relatively new, the potential capabilities of CEUS are vast and will continue to grow as technology expands and as research findings are translated into clinical practice. Already, preclinical models have explored uses of CEUS in imaging pediatric tumors, neonatal brains, fetal physiology and testicular perfusion. Three-dimensional printing provides an alternative model for training and educational purposes, particularly in pediatric contrast-enhanced VUS studies. Continued investigation of microbubbles as both imaging and therapeutic delivery agents is expected to increase the potential uses of UCA and, along with them, the opportunities for diagnosis and management support in pediatric illnesses and disease processes. Ultimately, translational research models are expected to move CEUS from the theoretical stage to effective implementation in both adults and children.
Acknowledgments
Dr. Maciej Piskunowicz was supported by the Kosciuszko Foundation.
Footnotes
Conflicts of interest Shelby Kutty is a consultant for GE Healthcare.
References
- 1.Darge K (2010) Voiding urosonography with US contrast agent for the diagnosis of vesicoureteric reflux in children: an update. Pediatr Radiol 40:956–962 [DOI] [PubMed] [Google Scholar]
- 2.Darge K, Moeller RT, Trusen A et al. (2005) Diagnosis of vesicoureteric reflux with low-dose contrast-enhanced harmonic ultrasound imaging. Pediatr Radiol 35:73–78 [DOI] [PubMed] [Google Scholar]
- 3.Piskunowicz M, Kosiak W, Batko T et al. (2015) Safety of intravenous application of second-generation ultrasound contrast agent in children: prospective analysis. Ultrasound Med Biol 41:1095–1099 [DOI] [PubMed] [Google Scholar]
- 4.Stenzel M (2013) Intravenous contrast-enhanced sonography in children and adolescents — a single center experience. J Ultrason 13:133–144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jacob J, Deganello A, Sellars ME et al. (2013) Contrast enhanced ultrasound (CEUS) characterization of grey-scale sonographic indeterminate focal liver lesions in pediatric practice. Ultraschall Med 34:529–540 [DOI] [PubMed] [Google Scholar]
- 6.Bonini G, Pezzotta G, Morzenti C et al. (2007) Contrast-enhanced ultrasound with SonoVue in the evaluation of postoperative complications in pediatric liver transplant recipients. J Ultrasound 10:99–106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ntoulia A, Anupindi SA, Darge K, Back SJ (2018) Applications of contrast-enhanced ultrasound in the pediatric abdomen. Abdom Radiol 43:948–959 [DOI] [PubMed] [Google Scholar]
- 8.Armstrong LB, Mooney DP, Paltiel H et al. (2018) Contrast enhanced ultrasound for the evaluation of blunt pediatric abdominal trauma. J Pediatr Surg 53:548–552 [DOI] [PubMed] [Google Scholar]
- 9.Menichini G, Sessa B, Trinci M et al. (2015) Accuracy of contrast-enhanced ultrasound (CEUS) in the identification and characterization of traumatic solid organ lesions in children: a retrospective comparison with baseline US and CE-MDCT. Radiol Med 120:989–1001 [DOI] [PubMed] [Google Scholar]
- 10.Kljucevsek D, Vidmar D, Urlep D, Dezman R (2016) Dynamic contrast-enhanced ultrasound of the bowel wall with quantitative assessment of Crohn’s disease activity in childhood. Radiol Oncol 50:347–354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hwang M, De Jong RM, Herman S et al. (2017) Novel contrast-enhanced ultrasound evaluation in neonatal hypoxic ischemic injury: clinical application and future directions. J Ultrasound Med 36:2379–2386 [DOI] [PubMed] [Google Scholar]
- 12.Hwang M, Sridharan A, Darge K et al. (2019) Novel quantitative contrast-enhanced ultrasound detection of hypoxic ischemic injury in neonates and infants: pilot study 1. J Ultrasound Med 38:2025–2038 [DOI] [PubMed] [Google Scholar]
- 13.Maj E, Papiernik D, Wietrzyk J (2016) Antiangiogenic cancer treatment: the great discovery and greater complexity. Int J Oncol 49:1773–1784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hendry SA, Farnsworth RH, Solomon B et al. (2016) The role of the tumor vasculature in the host immune response: implications for therapeutic strategies targeting the tumor microenvironment. Front Immunol 7:621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hlatky L, Hahnfeldt P, Folkman J (2002) Clinical application of antiangiogenic therapy: microvessel density, what it does and doesn’t tell us. J Natl Cancer Inst 94:883–893 [DOI] [PubMed] [Google Scholar]
- 16.McCarville MB, Streck CJ, Dickson PV et al. (2006) Angiogenesis inhibitors in a murine neuroblastoma model: quantitative assessment of intratumoral blood flow with contrast-enhanced gray-scale US. Radiology 240:73–81 [DOI] [PubMed] [Google Scholar]
- 17.Sims TL, McGee M, Williams RF et al. (2010) IFN-β restricts tumor growth and sensitizes alveolar rhabdomyosarcoma to ionizing radiation. Mol Cancer Ther 9:761–771 [DOI] [PubMed] [Google Scholar]
- 18.Carson P (2012) MO-D-218-01: overview of methodology and standards (QIBA, IEC, AIUM and AAPM). Med Phys 39:3869–3870 [DOI] [PubMed] [Google Scholar]
- 19.Graham EM, Ruis KA, Hartman AL et al. (2008) A systematic review of the role of intrapartum hypoxia-ischemia in the causation of neonatal encephalopathy. Am J Obstet Gynecol 199:587–595 [DOI] [PubMed] [Google Scholar]
- 20.Radlowski EC, Conrad MS, Lezmi S et al. (2014) A neonatal piglet model for investigating brain and cognitive development in small for gestational age human infants. PLoS One 9:e91951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Thibault KL, Margulies SS (1998) Age-dependent material properties of the porcine cerebrum: effect on pediatric inertial head injury criteria. J Biomech 31:1119–1126 [DOI] [PubMed] [Google Scholar]
- 22.Dickerson JW, Dobbing J (1967) Prenatal and postnatal growth and development of the central nervous system of the pig. Proc R Soc Lond B Biol Sci 166:384–395 [DOI] [PubMed] [Google Scholar]
- 23.Dobbing J, Sands J (1979) Comparative aspects of the brain growth spurt. Early Hum Dev 3:79–83 [DOI] [PubMed] [Google Scholar]
- 24.Eiby YA, Wright LL, Kalanjati VP et al. (2013) A pig model of the preterm neonate: anthropometric and physiological characteristics. PLoS One 8:e68763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Taylor GA, Barnewolt CE, Dunning PS (1998) Neonatal pig brain: lack of heating during Doppler US. Radiology 207:525–528 [DOI] [PubMed] [Google Scholar]
- 26.Sridharan A, Poznick L, Roberts AL et al. (2019) Contrast-enhanced ultrasound (CEUS) in pediatric swine as a pediatric preclinical model for brain imaging. In: Radiological Society of North America 2019 scientific assembly and annual meeting, pp SSQ17–01 [Google Scholar]
- 27.Taylor GA, Soul JS, Dunning PS (1998) Sonographic ventriculography: a new potential use for sonographic contrast agents in neonatal hydrocephalus. AJNR Am J Neuroradiol 19:1931–1934 [PMC free article] [PubMed] [Google Scholar]
- 28.Morrison JL, Berry MJ, Botting KJ et al. (2018) Improving pregnancy outcomes in humans through studies in sheep. Am J Physiol Regul Integr Comp Physiol 315:R1123–R1153 [DOI] [PubMed] [Google Scholar]
- 29.Back SA, Riddle A, Hohimer AR (2006) Role of instrumented fetal sheep preparations in defining the pathogenesis of human periventricular white-matter injury. J Child Neurol 21:582–589 [DOI] [PubMed] [Google Scholar]
- 30.Pringle KC (1986) Human fetal lung development and related animal models. Clin Obstet Gynecol 29:502–513 [PubMed] [Google Scholar]
- 31.Partridge EA, Davey MG, Hornick MA et al. (2017) An extrauterine system to physiologically support the extreme premature lamb. Nat Commun 8:15112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bryner BS, Mychaliska GB (2014) ECLS for preemies: the artificial placenta. Semin Perinatol 38:122–129 [DOI] [PubMed] [Google Scholar]
- 33.Bryner B, Gray B, Perkins E et al. (2015) An extracorporeal artificial placenta supports extremely premature lambs for 1 week. J Pediatr Surg 50:44–49 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Didier RA, Sridharan A, Lawrence K et al. (2019) Contrast-enhanced ultrasound in extracorporeal support: in vitro studies and initial experience and safety data in the extreme premature fetal lamb maintained by the extrauterine environment for neonatal development. J Ultrasound Med 38:1971–1978 [DOI] [PubMed] [Google Scholar]
- 35.Williamson RC (1976) Torsion of the testis and allied conditions. Br J Surg 63:465–476 [DOI] [PubMed] [Google Scholar]
- 36.Osumah TS, Jimbo M, Granberg CF, Gargollo PC (2018) Frontiers in pediatric testicular torsion: an integrated review of prevailing trends and management outcomes. J Pediatr Urol 14:394–401 [DOI] [PubMed] [Google Scholar]
- 37.Nason GJ, Tareen F, McLoughlin D et al. (2013) Scrotal exploration for acute scrotal pain: a 10-year experience in two tertiary referral paediatric units. Scand J Urol 47:418–422 [DOI] [PubMed] [Google Scholar]
- 38.Rebik K, Wagner JM, Middleton W (2019) Scrotal ultrasound. Radiol Clin N Am 57:635–648 [DOI] [PubMed] [Google Scholar]
- 39.Pepe P, Panella P, Pennisi M, Aragona F (2006) Does color Doppler sonography improve the clinical assessment of patients with acute scrotum? Eur J Radiol 60:120–124 [DOI] [PubMed] [Google Scholar]
- 40.Burks DD, Markey BJ, Burkhard TK et al. (1990) Suspected testicular torsion and ischemia: evaluation with color Doppler sonography. Radiology 175:815–821 [DOI] [PubMed] [Google Scholar]
- 41.Lerner RM, Mevorach RA, Hulbert WC, Rabinowitz R (1990) Color Doppler US in the evaluation of acute scrotal disease. Radiology 176:355–358 [DOI] [PubMed] [Google Scholar]
- 42.Sung EK, Setty BN, Castro-Aragon I (2012) Sonography of the pediatric scrotum: emphasis on the Ts — torsion, trauma, and tumors. AJR Am J Roentgenol 198:996–1003 [DOI] [PubMed] [Google Scholar]
- 43.Kalfa N, Veyrac C, Baud C et al. (2004) Ultrasonography of the spermatic cord in children with testicular torsion: impact on the surgical strategy. J Urol 172:1692–1695 [DOI] [PubMed] [Google Scholar]
- 44.Blask ARN, Bulas D, Shalaby-Rana E et al. (2002) Color Doppler sonography and scintigraphy of the testis: a prospective, comparative analysis in children with acute scrotal pain. Pediatr Emerg Care 18:67–71 [DOI] [PubMed] [Google Scholar]
- 45.Paltiel HJ, Connolly LP, Atala A et al. (1998) Acute scrotal symptoms in boys with an indeterminate clinical presentation: comparison of color Doppler sonography and scintigraphy. Radiology 207:223–232 [DOI] [PubMed] [Google Scholar]
- 46.Paltiel HJ, Rupich RC, Babcock DS (1994) Maturational changes in arterial impedance of the normal testis in boys: Doppler sonographic study. AJR Am J Roentgenol 163:1189–1193 [DOI] [PubMed] [Google Scholar]
- 47.Wilson SR, Burns PN (2010) Microbubble-enhanced US in body imaging: what role? Radiology 257:24–39 [DOI] [PubMed] [Google Scholar]
- 48.Sidhu PS, Cantisani V, Dietrich CF et al. (2018) The EFSUMB guidelines and recommendations for the clinical practice of contrast-enhanced ultrasound (CEUS) in non-hepatic applications: update 2017 (long version). Ultraschall Med 39:e2–e44 [DOI] [PubMed] [Google Scholar]
- 49.Claudon M, Dietrich CF, Choi BI et al. (2013) Guidelines and good clinical practice recommendations for contrast enhanced ultrasound (CEUS) in the liver — update 2012. Ultraschall Med 34:11–29 [DOI] [PubMed] [Google Scholar]
- 50.Liu X, Jang HJ, Khalili K et al. (2018) Successful integration of contrast-enhanced US into routine abdominal imaging. Radiographics 38:1454–1477 [DOI] [PubMed] [Google Scholar]
- 51.Huang DY, Yusuf GT, Daneshi M et al. (2017) Contrast-enhanced US-guided interventions: improving success rate and avoiding complications using US contrast agents. Radiographics 37:652–664 [DOI] [PubMed] [Google Scholar]
- 52.Lu C, Merrill C, Medellin A et al. (2019) Bowel ultrasound state of the art: grayscale and Doppler ultrasound, contrast enhancement, and elastography in Crohn disease. J Ultrasound Med 38:271–288 [DOI] [PubMed] [Google Scholar]
- 53.Valentino M, Bertolotto M, Derchi L et al. (2011) Role of contrast enhanced ultrasound in acute scrotal diseases. Eur Radiol 21:1831–1840 [DOI] [PubMed] [Google Scholar]
- 54.Catalano O, Lobianco R, Sandomenico F et al. (2004) Real-time, contrast-enhanced sonographic imaging in emergency radiology. Radiol Med 108:454–469 [PubMed] [Google Scholar]
- 55.Paltiel HJ, Kalish LA, Susaeta RA et al. (2006) Pulse-inversion US imaging of testicular ischemia: quantitative and qualitative analyses in a rabbit model. Radiology 239:718–729 [DOI] [PubMed] [Google Scholar]
- 56.Paltiel HJ, Estrada CR, Alomari AI et al. (2014) Multi-planar dynamic contrast-enhanced ultrasound assessment of blood flow in a rabbit model of testicular torsion. Ultrasound Med Biol 40:361–370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Paltiel HJ, Padua HM, Gargollo PC et al. (2011) Contrast-enhanced, real-time volumetric ultrasound imaging of tissue perfusion: preliminary results in a rabbit model of testicular torsion. Phys Med Biol 56:2183–2197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Tse KS, Wong LS, Lau HY et al. (2014) Paediatric vesicoureteric reflux imaging: where are we? Novel ultrasound-based voiding urosonography. Hong Kong Med J 20:437–443 [DOI] [PubMed] [Google Scholar]
- 59.Wong LS, Tse KS, Fan TW et al. (2014) Voiding urosonography with second-generation ultrasound contrast versus micturating cystourethrography in the diagnosis of vesicoureteric reflux. Eur J Pediatr 173:1095–1101 [DOI] [PubMed] [Google Scholar]
- 60.Lebowitz RL, Olbing H, Parkkulainen KV et al. (1985) International system of radiographic grading of vesicoureteric reflux. Pediatr Radiol 15:105–109 [DOI] [PubMed] [Google Scholar]
- 61.Darge K (2008) Voiding urosonography with ultrasound contrast agents for the diagnosis of vesicoureteric reflux in children: I. procedure. Pediatr Radiol 38:40–53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Darge K (2008) Voiding urosonography with US contrast agents for the diagnosis of vesicoureteric reflux in children: II. Comparison with radiological examinations. Pediatr Radiol 38:54–63 [DOI] [PubMed] [Google Scholar]
- 63.Ntoulia A, Back SJ, Shellikeri S et al. (2018) Contrast-enhanced voiding urosonography (ceVUS) with the intravesical administration of the ultrasound contrast agent Optison™ for vesicoureteral reflux detection in children: a prospective clinical trial. Pediatr Radiol 48:216–226 [DOI] [PubMed] [Google Scholar]
- 64.Shellikeri S, Silvestro E, Poznick L et al. (2019) 3D printed anatomic contrast enhanced voiding urosonography (ceVUS) teaching phantoms: bringing pediatric vesicoureteral reflux (VUR) to life. Pediatr Radiol 49:S81 [Google Scholar]
- 65.Gong Y, Cabodi M, Porter TM (2014) Acoustic investigation of pressure-dependent resonance and shell elasticity of lipid-coated monodisperse microbubbles. Appl Phys Lett 104:074103 [Google Scholar]
- 66.Helfield BL, Leung BYC, Huo X, Goertz DE (2014) Scaling of the viscoelastic shell properties of phospholipid encapsulated microbubbles with ultrasound frequency. Ultrasonics 54:1419–1424 [DOI] [PubMed] [Google Scholar]
- 67.Gessner R, Lukacs M, Lee M et al. (2010) High-resolution, high-contrast ultrasound imaging using a prototype dual-frequency transducer: in vitro and in vivo studies. IEEE Trans Ultrason Ferroelectr Freq Control 57:1772–1781 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bouakaz A, Frigstad S, Ten Cate FJ, de Jong N (2002) Super harmonic imaging: a new imaging technique for improved contrast detection. Ultrasound Med Biol 28:59–68 [DOI] [PubMed] [Google Scholar]
- 69.Wang S, Hossack JA, Klibanov AL (2018) Targeting of microbubbles: contrast agents for ultrasound molecular imaging. J Drug Target 26:420–434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Pochon S, Tardy I, Bussat P et al. (2010) BR55: a lipopeptide-based VEGFR2-targeted ultrasound contrast agent for molecular imaging of angiogenesis. Investig Radiol 45:89–95 [DOI] [PubMed] [Google Scholar]
- 71.Abou-Elkacem L, Bachawal SV, Willmann JK (2015) Ultrasound molecular imaging: moving toward clinical translation. Eur J Radiol 84:1685–1693 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Willmann JK, Bonomo L, Testa AC et al. (2017) Ultrasound molecular imaging with BR55 in patients with breast & ovarian lesions: first-in-human results. J Clin Oncol 35:2133–2140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Hernot S, Klibanov AL (2008) Microbubbles in ultrasound-triggered drug and gene delivery. Adv Drug Deliv Rev 60:1153–1166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Stride E (2015) Physical principles of microbubbles for ultrasound imaging and therapy. Front Neurol Neurosci 36:11–22 [DOI] [PubMed] [Google Scholar]
- 75.Kinoshita M, Hynynen K (2007) Key factors that affect sonoporation efficiency in in vitro settings: the importance of standing wave in sonoporation. Biochem Biophys Res Commun 359:860–865 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Phenix CP, Togtema M, Pichardo S et al. (2014) High intensity focused ultrasound technology, its scope and applications in therapy and drug delivery. J Pharm Pharm Sci 17:136–153 [DOI] [PubMed] [Google Scholar]
- 77.Yan F, Li L, Deng Z et al. (2013) Paclitaxel-liposome-microbubble complexes as ultrasound-triggered therapeutic drug delivery carriers. J Control Release 166:246–255 [DOI] [PubMed] [Google Scholar]
- 78.Nittayacharn P, Nasongkla N (2017) Development of self-forming doxorubicin-loaded polymeric depots as an injectable drug delivery system for liver cancer chemotherapy. J Mater Sci Mater Med 28:101. [DOI] [PubMed] [Google Scholar]
- 79.Nesbitt H, Sheng Y, Kamila S et al. (2018) Gemcitabine loaded microbubbles for targeted chemo-sonodynamic therapy of pancreatic cancer. J Control Release 279:8–16 [DOI] [PubMed] [Google Scholar]
- 80.Li F, Jin L, Wang H et al. (2014) The dual effect of ultrasound-targeted microbubble destruction in mediating recombinant adenoassociated virus delivery in renal cell carcinoma: transfection enhancement and tumor inhibition. J Gene Med 16:28–39 [DOI] [PubMed] [Google Scholar]
- 81.Liu H, Chang S, Sun J et al. (2014) Ultrasound-mediated destruction of LHRHa-targeted and paclitaxel-loaded lipid microbubbles induces proliferation inhibition and apoptosis in ovarian cancer cells. Mol Pharm 11:40–48 [DOI] [PMC free article] [PubMed] [Google Scholar]