Abstract
The collection of big data on skin and appendage phenotypes has revolutionized the field of personalized diagnosis and treatment by enabling the evaluation of individual characteristics and early detection of abnormalities. To establish a standardized system for collecting and measuring big data on phenotypes, a systematic categorization of measurement entries has been undertaken, accompanied by recommendations on measurement entries, environmental equipment requirements, and collection processes, tailored to the needs of different usage scenarios. Specific collection sites have also been recommended based on different index characteristics. A multi-center, multi-regional collaboration has been initiated to collect big date on phenotypes of healthy and diseased skin in the Chinese population. This data will be correlated with patient disease information, exploring the factors influencing skin phenotype, analyzing the phenotypic data features that can predict prognosis, and ultimately promoting the exploration of the pathophysiology and pathogenesis of skin diseases and therapeutic approaches. Non-invasive skin measurement robots are also in development. This consensus aims to provide a reference for the study of phenomics and the standardization of phenotypic measurements of skin and appendages in China.
Keywords: Skin and appendages, Disease phenotype, Big data, Standardized system
Introduction
As the largest and most superficial organ of the human body, skin is mainly composed of the epidermis, dermis, and subcutaneous tissues, as well as skin appendages such as nails, hairs, hair follicles, sebaceous glands, and sweat glands (Uitto and Christiano 1992). Additionally, the skin is rich in varies components, including blood vessels, lymphatic vessels, nerves, and muscles.
Phenotypes refer to the physical and biological characteristics of living organisms, and they are shaped by a combination of genetic and the environmental factors (Xue and Leibler 2018). The complexity of the skin and appendages’ morphology, structure, and function is influenced by numerous factors, including age, gender, genetics, skin type, pigmentation, skin thickness changes, the presence of hair, and environmental exposure, which give rise to phenotypic variations. Therefore, the acquisition of large data on skin and appendage phenotypes can help in the assessment of individual skin appendage traits, early detection of abnormalities, and providing personalized skin assessments, guidance, and treatment.
Recognizing and quantifying the phenotype of the skin offers substantial benefits owing to its prominent visual characteristics. It is crucial to emphasize that phenotype is intricately connected with various diseases. First, various skin diseases exhibit evident phenotypic changes. For instance, psoriasis typically presents as locally thickened erythema with scales, an increase in local skin temperature, elevated skin pH (Yazdanparast et al. 2018), altered dermatoglyphs (Dogramaci et al. 2010), heightened transepidermal water loss (TEWL) value, and changes in stratum corneum hydration (Montero-Vilchez et al. 2021). Similar changes are observed in conditions such as atopic dermatitis, lichen planus, etc. (Yazdanparast et al. 2018). Furthermore, facial recognition technology proves valuable in diagnosing select genetic diseases due to unique facial features exhibited by affected individuals. Facial phenotype serves as a critical diagnostic indicator for numerous genetic syndromes (Dingemans et al. 2023; Hallgrímsson et al. 2020). Genome scans targeting diverse cohorts have also revealed hundreds of genetic loci associated with various aspects of facial variation within the normal range, shedding light on the intricate genetic architecture of facial morphology variation (White et al. 2021). Other easily accessible skin phenotypic indicators, such as dermatoglyphs, have been reported to bear relevance to genetic diseases, enabling early screening and aiding in the auxiliary diagnosis of certain skeletal system diseases, non-congenital limb deformities, and other conditions.
To evaluate the health and characteristics of the skin and appendages, various indicators have been devised. These include physiological markers such as skin color, pH balance, and hydration levels, as well as mechanical and physical markers like skin elasticity and hardness. Other crucial indicators are functional markers, which imply skin barrier and metabolic functions, and the immune system response. Microanatomical indicators are also significant in evaluating skin health, where in vitro techniques such as histological sampling and in vivo imaging techniques like reflectance confocal microscopy (RCM) and optical coherence tomography (OCT) provide microanatomical parameters such as data on epidermal thickness, melanocyte density, and hair follicle density (Xu et al. 2017). Skin imaging parameters, including dermoscopy and infrared thermography, and valid constants and scales such as physical skin constant and psoriasis area and severity index (PASI) scores (Li et al. 2021a) are also considered valuable methods for measuring and evaluating skin and its attributes.
The lack of standardized criteria for skin and appendages phenotypic data has led to arbitrary and fragmented choices in their selection of measurements. The absence of uniform standards hampers the objective assessment of morphological characteristics and deprives early detection of skin abnormalities, the basis for personalized diagnosis and treatment, and the efficacy and safety evaluation of products intended for skin use. To tackle such problems, the Tumor Research Centre of Chinese Society of Dermatology and the Skin Oncology Group of China Dermatologist Association, together with Human Phenome Institute, Fudan University, issued expert consensus. It is aimed to provide a reference basis for the study of phenomics and establish a standardized approach to measure human skin and appendage phenotypes in China. Such an effort is deemed essential for clinical research and to advance the study of skin pathophysiological mechanisms.
Systematic Grouping of Skin and Appendage Phenotypic Measurement Entries
By systematically grouping skin and appendage phenotypic measurements, it becomes easier to categorize and analyze the tremendous amount of data produced (as shown in Fig. 1, Table 1). This process not only recommends collection sites based on the characteristics of these indicators, but also takes into account phenotypic entry volume, environmental equipment requirements, and collection procedures. Such grouping and analysis optimize data collection and analysis procedures, making accurate phenotypic measurements crucial in clinical research and improving the diagnosis and treatment of skin-related health conditions.
Fig. 1.
Systematic grouping of skin and appendage phenotypic measurement entries
Table 1.
Summary of normal values of basic physiological indexes of skin
| Indicators | Equipments | Methods | Normal value | Influence factors | |
|---|---|---|---|---|---|
| Skin temperature | Contact or infrared instrument | Compare and interpret the international standard ISO 9886 | 0–2 years old: 36.4–38.0 °C; 3–10 years old: 36.1–37.8 °C; 11–65 years old: 35.9–37.6 °C; > 65 years old: 35.8–37.5 °C | Environmental temperature; Blood heat; Blood flow; Effects of metabolism of epidermis and related appendages | |
| Skin pH | Skin pH tester | N/A |
Whole body: 4.2–6.1 (except the former), showing a Gaussian distribution trend; Newborns: higher than adults 1 day after birth, reaching adult status 4 days after birth; Sweat: 4.0–6.8 Oral mucosa: about 7, the highest in hard palate Armpit, perineum, interdigital (toe) area: about 7 |
Skin cleaning method; Sweat; Degree of occlusion; Presence of special microorganisms; Circadian rhythm | |
| Skin-type evaluation | Fitzpatrick's skin type | Color difference tester | Quantify skin color based on three primary color stimulation values (L *, a *, b *) |
> 55°: very shallow 55° to 41°: shallow 41° to 28°: medium 28° to 10°: tanning 10° to − 30°: brown < − 30°: black |
Sunlight exposure |
| Chinese skin type | N/A | N/A |
Neutral: the water content of the stratum corneum is normal (10–20%), the sebum secretion is moderate, the skin is tight, elastic, and the surface is smooth, moist and delicate Dryness: the moisture content of the dermis is less than 10%, the sebum secretion is low, the skin is dry, desquamate, delicate and lusterless, the skin color is dark, and it is prone to small wrinkles and pigmentation Oiliness: the water content of the dermis is normal or reduced, the sebum secretion is vigorous, the skin surface is oily and shiny, the pores are thick, and acne and folliculitis are prone to occur |
Moisture content of skin dermis; Oil secretion | |
| Complexion evaluation | Skin erythema | Narrow-wave skin spectrophotometer, Spectrophotometer, Mexameter®, Dermacatch (Colorix) | Measure hemoglobin and express erythema as hemoglobin index EI | Relative value, there is no relevant report on the value range. The smaller the value is, the thinner and more sensitive the skin is | Ambient and local temperature (affecting the expansion and contraction of blood vessels); Ambient light |
| Skin melanin | Measurement of melanin index MI | 0–999, no unit, the higher the value, the higher the melanin content in the skin | |||
| Skin pigmentation | N/A | N/A | Chloasma, freckles, ADM, PIH | N/A | |
| Daylight tolerance | Minimum erythema dose | Solar ultraviolet simulator | The emission light source takes the UVB (290—320 nm) as the main peak, the power is 500W, the irradiation site is the skin of the unexposed area (back, chest or forearm flexor side, etc.), the shortest exposure time is 15 s, and the observation is conducted in sufficient natural light for 24 h | Take the data of normal people as a control | Individual differences; Skin color; Irradiation site; season; Spectrum and intensity of light source |
| Minimum persistent pigment darkening | Solar ultraviolet simulation system, ultraviolet phototherapy instrument, solar protection index tester | UVA and UVB are dominant (290—400 nm), and the output rate of light sources below 290 nm and above 400 nm is less than 2% | Take the data of normal people as a control | ||
| Epidermal evaluation | Degree of stratum corneum exfoliation | Glue coating plate, different sizes of D-Squame tapes, etc | Semi-passive collection method, standardized tape sticking method, mechanical forced collection method and forced desquamation method | N/A | N/A |
| Epidermal friction | Three-axis quartz force plate, skin friction instrument | N/A | Any unit (au) | Material Science; Temperature; Positive force; Contact surface area; Age; Body parts; Stratum corneum water content | |
| Skin glossiness | Skin glossmeter, GonioLux (Orion Concept) and Translucymeter (TLS 850, Diagron Ltd) | N/A | Usually difficult to quantify | Skin color; Light reflection; Skin surface texture | |
| Surface texture, roughness | Optical profile measurement technology | Usually difficult to quantify | |||
| Skin barrier functions | Transepidermal water loss | Skin moisture loss tester (Tewameter TM210) and Evaporimeter | Subjects wait in a constant test environment | Fluctuate greatly | External humidity; Temperature; Atmospheric pressure |
| Stratum corneum water content | GPSkin Barrier | N/A | N/A | N/A | |
| Dermal evaluation | Microcirculation system | RCM, OCT, MSOT | Detected by immunohistochemistry, specific lymphoid markers include hyaluronic acid receptor-1, Prox-1, podoplanin, VEGFR-3, etc |
The first form: subcutaneous vascular network consists of papillary ring and horizontal vascular plexus in the superficial dermis; The second form: the vascular network around the hair follicle that nourishes the sebaceous gland hair follicle; The third form: periglandular vascular network |
N/A |
| Lymphatic vascular microcirculation | N/A | ||||
| Skin elasticity |
Dermal Torque Meter; Skin tester—Cutter SEM575 |
Use the torsion parallel to the skin surface as the stress; Use the suction (vacuum) perpendicular to the skin surface as the stress | N/A | Content of skin collagen, elastin and natural fat | |
| Dermatoglyphs | Finger / toe print, palm print | Impression method; Chalk—carbon ink—transparent tape method; Impression method for patients with hand and foot deformities, ninhydrin-monosodium glutamate method for occult dermatoglyphic display, etc |
Finger / Toe print: bow pattern, dustpan pattern and bucket pattern Palm print: three areas of the palm (greater thenar, lesser thenar, interdigital area), trigeminal, seven trigeminal and palm fold |
N/A | Related to individual vulnerability |
| Skin appendages | Sweat glands | Chromatograph, etc | Direct weighing method, water evaporation quantitative method, sweat staining visualization method, special organic sweat compound staining method, modeling method and skin capacitance mapping imaging method | N/A | Related to temperature, emotion and taste stress |
| Hairs | Clinical commonly used photographic equipment, hair mirror, etc | Full scalp photography technology, contrast-enhanced hair image analysis technology, etc |
Hair quantity: about 80–100 hairs/cm2 at the top of the head, about 120–180 hairs/cm2 at the back of the pillow, and 15–20 hairs/cm2 at the back of the hand The shape of the hairline of the forehead: L, M, C and U, and the two special types are F and V |
N/A | |
| Surface sebum | Sebumeter | Spectrophotometric detection | N/A | N/A | |
| Nails | Thickness | Calipers, 20 or 25 MHz ultrasonic imaging | N/A | N/A | N/A |
| Roughness | Impression | Computer imaging analysis or optical profilometry | N/A | N/A | |
| Color | Spectrophotometer, wood lamp | N/A | N/A | N/A | |
| Nail fold | Capillarioscope | Check the structure of superficial microvessels |
Physiological state: the capillary loop is hairpin, with two parallel longitudinal vessels; Pathological changes: branching vessels, increased length of vascular loop, huge capillaries, parallel structure disorder and microaneurysm |
N/A | |
| Nail bed | RCM, OCT, MRI | Perspective, etc | N/A | N/A | |
EI Erythema index; MI Melanin index; ADM Acquired dermal melanosis; PIH Post-inflammatory pigmentation; UVA Long-wave ultraviolet; UVB Medium-wave ultraviolet; RCM Reflectance confocal microscopy; OCT Optical coherence tomography; MSOT Multispectral photoacoustic imaging; Prox-1 Rat homeobox gene transcription factor 1; VEGFR-3 Vascular endothelial growth factor receptor 3; MRI Magnetic resonance imaging
Basic Skin Physiological Indicators
Skin Temperature
The human body maintains its temperature within a certain range, typically around 37 °C. However, skin temperature, which represents peripheral temperatures, can fluctuate from a few degrees to 44 °C. The skin serves as an interface between the environment and the body's core, and its surface temperature is primarily determined by the environmental temperature, blood heat, blood flow (Wa and Maibach 2010), epidermis, and related appendage metabolism (Wa and Maibach 2010). The palmar and plantar surfaces, which represent glabrous skin, are typically the primary sites for heat exchange in humans and other mammals (Caldwell et al. 2014). This is likely due to the abundance of arteriovenous anastomosis (AVA) shunt vessels in these areas, allowing for a significantly greater heat transfer capacity per unit surface area compared to nonglabrous skin (Caldwell et al. 2014).
To measure skin temperature, local temperature is first measured through a contact thermometer or infrared measurement, and overall skin temperature is calculated by the international standard ISO 9886 (Agache 2004). For instance, normal skin temperature for those aged 0–2 ranges between 36.4 and 38.0 °C. For people aged 3–10, the range is 36.1–37.8 °C, for those aged 11–65, it is 35.9 °C–37.6 °C, and for those over 65, it is between 35.8 and 37.5 °C (Wa and Maibach 2010). Changes in skin temperature are observed in a variety of skin conditions (Civan and Kurama 2023). Recent research has indicated that reducing skin temperature can help mitigate the risk of mechanical skin damage (Valenza et al. 2023).
pH
The skin pH level is crucial in maintaining skin barrier integrity and microecological balance. Skin acidity arises from free fatty acids in the surface ecosystem, and cleaning behaviors, even using soap with a higher acidity than the skin, increase the skin pH. Skin pH can be measured by a skin pH meter, with the average range being 4.2 and 6.1, excluding the forehead area (Mehta et al. 2018). Newborns have higher skin pH levels, but within the fourth days following birth, their skin pH reached adult levels (Giusti et al. 2001). Sweat affects surface pH but not baseline level. Oral mucosa typically has a pH of around seven, with the hard palate having the highest value. Areas with physiological pores, like axillary, perineal, and interdigital toe regions, have skin pH levels similar to seven. Local pH differences help promote the survival of specific microorganisms. Circadian rhythm also affects the pH of human skin (Li et al. 2021a). An increase in skin pH is typically indicative of a change in skin barrier function (Yazdanparast et al. 2018), which can be observed in various skin conditions (Cork et al. 2009; Wat et al. 2018). Additionally, there are studies suggesting that elevated skin pH and TEWL may potentially serve as predictive indicators for the development of atopic dermatitis (Hülpüsch et al. 2020; Marini et al. 2023). In a study conducted on Asians, it was observed that an elevated skin acidity level was associated with a notable decrease in wrinkle formation (Jung et al. 2013).
Skin-Type Evaluation
Skin type is predominantly determined by an individual’s genetic makeup. Different ethnic groups exhibit varying genes traits and skin types, which can be further influenced by environmental factors and exposure to light. In clinical settings, skin type plays a pivotal role in determining the optimal dosage for photoelectrotherapy, evaluating the individual’s sensitivity to ultraviolet (UV) light, predicting the response and outcomes of photoelectrotherapy, and assessing the efficacy of cosmetics (Jo et al. 2022).
Fitzpatrick’s Skin Type
In 1972, Thomas B. Fitzpatrick (1988) introduced a skin typing system that categorizes skin color into six types based on its response to sun exposure. The system, ranging from types I to VI, defines skin characteristics such as tendency to burn or tan. For instance, Type I skin always burns and never tans, while type VI skin never burns but always tans.
Skin types are classified into six groups based on melanin content in the basal layer. Types I and II are typically European and American skin types that have low melanin content. Skin types III and IV are common to the yellow skin of Southeast Asia, while the brown–black skin of Africa falls under types V and VI, which have high melanin content (Fitzpatrick 1988). The current traditional skin typing evaluation method is subjective, influences quantitative evaluation, and has limitations, particularly for Asians (Fajuyigbe and Young 2016). In response, more objective methods have been proposed, such as Individual Typology Angle (ITA) skin technique. This method employs primary color stimulation values (L*, a*, b*) and the ITA° values to quantify skin color in six skin-tone groups, ranging from very light to dark skin (equation: ITA = [arctan (L*-50)/b*)] *180/π, where L* represents luminance ranging from black (0) to white (100) and b* ranging from yellow to blue). The larger the ITA value, the lighter the skin color. The smaller the ITA value, the deeper the skin color. Refers as below: Very Light (> 55), Light (41 to < 55), Intermediate (28 to < 41), Tan (10 to < 28), Brown (− 30 to < 10), and Dark (< − 30) (Li et al. 2021a). This method’s strengths are its superior precision and quantitative evaluation using a color difference tester. ITA classification is linked to pigmentation composition (Del Bino and Bernerd 2013), and some studies propose that DNA damage in melanocytes is influenced by ITA, which may explain the heightened risk of skin cancer, including melanoma, in individuals with lighter skin types (Del Bino and Bernerd 2013). While this subtype has been identified as being linked to the risk of skin cancer, it is important to note that it cannot provide a quantitative measure of that risk. Individuals with Fitzpatrick skin types III–VI are more susceptible to acne and may experience excessive post-inflammatory pigmentation (PIH) as a result (Arsiwala and Desai 2019).
Chinese Skin Type
The Beauty Sub-Professional Committee of the Dermatologist Branch of the Chinese Medical Association complied the "Chinese facial skin classification and skin care guidelines" in 2009 (Li and Li 2009), categorizing skin based on water content and oil secretion of stratum corneum. The guidelines delineate three classifications: neutral skin, dry skin, and oily skin based on criteria such as skin water content, sebum secretion, skin tightness, elasticity, surface smoothness, and delicacy. Mixed skin implies a mix of criteria in facial T areas and cheeks, categorized accordingly. While age-dependent additional skin problems may emerge, the above classification remains the most frequently used in the Chinese population. Meta-analysis findings suggest that skin type, particularly oily skin, can impact the severity of acne (Heng and Chew 2020).
Recent studies have also noted two stable community composition patterns of skin microorganisms at the population level of the Chinese population, namely Moraxella osmosis-dominated M-cutotype and Cutibacterium acnes-dominated C-cutotype (Li et al. 2021b), potentially used for skin typing. This classification system helps dermatologists and the public evaluate skin quality and provide personalized skin care guidance based on physiological features of each skin type.
Complexion Evaluation
The complexion is determined by the conciseness of melanin content (Bajpai et al. 2023), oxygenated and deoxygenated hemoglobin content (Stamatas et al. 2008), and endogenous or exogenous pigmentation (Martin et al. 2017).
Skin Erythema and Melanin
Erythema is a common skin condition associated with inflammation and blood vessels that are often indicative of underlying conditions such as acne (Eichenfield et al. 2021), inflammation (Yazdanparast et al. 2018), rosacea (van Zuuren 2017), hemangioma (Requena and Sangueza 1997), or spider nevus (Requena and Sangueza 1997). The red value occurs due to the presence of blood vessels and hemoglobin is present in the dermal papillae of the skin (Requena and Sangueza 1997). Hemoglobin levels, expressed as an index known as the erythema index (EI), can be measured non-invasively using narrow-wave skin spectrophotometers, such as Mexameter®. The device compares the intensity of light reflected (Ired) between red and green wavelength (Igreen), with lower EI values indicating thinner and more sensitive skin that is prone to capillary dilation.
In contrast, melanocytes produce melanin, which is the primary determinant of skin complexion. Differences in melanin content and distribution contribute to the variety of skin tones among individuals and races (Del Bino and Bernerd 2013). The melanin index (MI) represents the melanin content of the skin and ranges from 0 to 999, with lower values associated with a lighter complexion and lower melanin content. Narrow-wave skin spectrophotometers non-invasively measure MI through the skin’s spectral absorption of three wavelengths of light (green, red, infrared, etc.) emitted by the device’s probe and measured by a receiver.
Peripheral and local temperature affects skin color by inducing vasodilation or contraction of blood vessels. Spectrophotometer and Mexameter measurements are useful, but accuracy can compromised by environmental light interference, requiring measurement in a dark room (Li et al. 2021a). While a spectrophotometer probe has minimal effect on results, Mexameter requires optimal probe pressure and placement angle. Dermacatch (Colorix) uses full-spectrum light-emitting diode (LED) for more sensitive and precise readings (Baquié and Kasraee 2014), independent of ambient light. However, small objects may still be difficult to measure accurately.
Skin Pigmentation
Hyperpigmentation refers to the darkening of the skin due to an increase in melanin production (Kundu et al. 2023). The severity of pigmentation varies depending on the degree of uneven complexion on the face. As different types and degrees of uneven skin tone exist, hyperpigmentation can manifest as several distinct conditions. Specifically, hyperpigmentation can be categorized into four conditions: melasma (Kundu et al. 2023), freckles (Kovacs et al. 2010), acquired dermal melanocytosis (Jimenez et al. 1994), and post-inflammatory hyperpigmentation (Silpa-Archa et al. 2017), which frequently occur.
Daylight Tolerance
Minimum Erythema Dose
Minimum erythema dose (MED) is used to determine skin tolerance to sun exposure for patients with UV-sensitive skin conditions, enabling them to select the appropriate sunscreen and sun protection factor (SPF). The use of a sunlight UV simulator, emitting medium-wave UV (290–320 nm) with 500 W, is required. The irradiation site should be in the non-exposed area like the back or forearm. The minimum erythema amount is defined as the irradiation time or dose that can cause the weakest erythema with consistent size, clear contours, and uniform color, which can be observed under natural light for 24 h after irradiation (Palmer and Friedmann 2004).
It should be noted that MED varies more with individual differences, complexion, irradiation site, season, the spectrum and intensity of the light source, etc. Clinical data of normal people should be used as a control (Palmer and Friedmann 2004).
Minimum Persistent Pigment Darkening
Minimum persistent pigment darkening (MPPD) is the smallest dose of long-wave UV radiation needed to produce visible skin darkening. To stimulate actual skin conditions, a daylight UV simulation system, UV phototherapy, or a daylight protection index meter should be used. Long-wave ultraviolet (UVA) and medium-wave ultraviolet (UVB) light sources are recommended, with an output wavelength range of 290–400 nm and an output rate < 2% for wavelength below 290 nm and above 400 nm. The measurement site should be on both sides of the spine with participants randomly selected for irradiation (Ravnbak et al. 2010). However, it’s worth noting that differences in measurements and judging criteria across centers can create controversy around MPPD and MED. Clinicians should be cautions in deciding on the applicability of MPPD and MED measurements for diagnosis and treatment (Ravnbak et al. 2010).
Epidermal Evaluation
Degree of Stratum Corneum Desquamation
The epidermal stratum corneum maintains its function through a balance continuous accumulation and shedding (Simpson et al. 2011). The epidermal turnover time, the duration required for self-renewal from the basal layer to the skin surface, is typically around 28 days under normal conditions. Shedding of the stratum corneum begins with a transition from a dense to a loose layer (Simpson et al. 2011). However, dehydration can cause keratinocyte cohesion to impede shedding, resulting in epidermal proliferation, thickening, scaling, and damage to the skin barrier (Walter 2022). Various methods can assess dermal cohesion and exfoliation, including the semi-passive collection technique (Piérard-Franchimont et al. 2011), standardized adhesive tape method (Keurentjes et al. 2021), mechanical forced collection method, and forced desquamation method. The semi-passive collection is typically used to collect dandruff from the scalp (Piérard-Franchimont et al. 2011). The standardized adhesive tape method is ideal for evaluating topical drug bioavailability. This involves using different sizes of D-Squame tapes to collect cells from the stratum corneum for confocal imaging using software such as VivaScan Version 7 White VivaScope 1500 (Lucid Inc, Rochester, NY, USA), followed by image analysis with software such as ImageJ for quantitative analysis of the stratum corneum thickness (Keurentjes et al. 2021).
Epidermal Friction
Epidermal friction represents the skin's resistance to the movement of objects on its surface, and the skin friction coefficient is a measurement of this ability (Comaish and Bottoms 1971; Veijgen et al. 2013). A three-axis quartz force plate and a skin friction meter (Aca-Derm Inc, California, USA), which operate on similar detection principles. However, several factors, such as the materials (Vilhena et al. 2023), temperature, positive force, contact surface area, age, body part, and stratum corneum water content, can affect the measurement. There are notable variations in hair properties among different skin types, with regions devoid of hair, such as the pads of the index finger, exhibiting higher friction coefficients. Proper environmental and instrument stability is crucial to ensure accurate and interpretation of findings. As such, researchers should pay attention to these factors when conducting their experiments. Overall, understanding epidermal friction can help determine the most effective ways to maintain healthy skin and manage related medical conditions.
Skin Glossiness and Surface Texture, Roughness
Skin glossiness and surface texture, including roughness, are important factors in evaluating skin health and attractiveness (Lee et al. 2023). Evenness of skin surface texture and glossiness are reflected when light shines on the skin surface. About 5% of the light is directly reflected, creating skin glow. An irregular skin surface texture hinders light absorption, leading to loss of skin glossiness.
Several factors determine skin glossiness, including skin color, light reflection, and skin surface texture (Lee et al. 2023). Higher glossiness is generally perceived as a sign of healthy, attractive skin. Devices like skin glucometers (e.g., GonioLux by Orion Concept) and translucymeters (e.g., TLS 850 by Diastron Ltd) (Li et al. 2021a) can assess reflective ability. However, glossiness can be challenging to quantify.
Skin texture and roughness are significant factors in assessing skin aging and wrinkles (Zhang et al. 2022). Skin texture shape and volume can indicate texture characteristics (Zhang et al. 2022). Optical profilometry using in vitro inverted film or in vivo fringe projection can evaluate skin texture. Data collection must be standardized to minimize variability in results due to minor alterations in the subject's posture, attitude, and expression (Zhang et al. 2022).
Skin Barrier Functions
The stratum corneum is a vital component in maintaining the skin’s barrier function. This layer consists of keratinocytes and intercellular lipids, where cholesteryl esters, cholesterol, long-chain saturated fatty acids, and ceramides serves as the key constituents of the lipid barrier. This "brick wall structure" provides stability to the stratum corneum (Zouboulis 2004). To assess skin barrier function non-invasively, in vitro assays are employed, with TEWL and stratum corneum water content serving as representative indicators.
Transepidermal Water Loss
TEWL is an essential metric for evaluating insensible water loss through percutaneous evaporation (Alexander et al. 2018; Schuler et al. 2023). High and low values indicate impaired and restored skin barrier function, respectively (Piérard-Franchimont et al. 2011). TEWL is a commonly employed non-invasive method for assessing the effectiveness of cosmetics or drugs in improving skin barrier function (Alexander et al. 2018). The Tewameter TM210 and skin water loss meter are widely used devices for measuring TEWL. It is crucial to note, however, that external factors such as humidity, temperature, and atmospheric pressure can affect the measured values. Therefore, it is imperative to ensure that subjects are maintained in a constant testing environment to obtain accurate and dependable measurements (Alexander et al. 2018; Schuler et al. 2023). It is also worth acknowledging that this technique can be time-consuming and may result in significant fluctuations in measured values. The measurement of TEWL can be a valuable tool for guiding the prevention of atopic dermatitis in clinical practice (Alexander et al. 2018; Marini et al. 2023), while continuous monitoring of TEWL may serve as a useful method for predicting allergic reactions (Schuler et al. 2023).
Stratum Corneum Water Content
The moisture content in the stratum corneum and the state of skin moisturization have a significant impact on skin health and appearance. Measuring the ability of stratum corneum to absorb and retain water is important for evaluating the effectiveness of moisturizing products (Crowther et al. 2008). Various techniques are available, but they can be challenging for non-specialists and are affected by environment factors. The GPSkin Barrier is a handheld instrument that can conveniently measure skin barrier function, including TEWL rate, moisture content, epidermal temperature, and epidermal humidity. It can also be used remotely to acquire data. The reduction in hydration and impairment of the skin's barrier function have been associated with certain skin diseases, such as atopic dermatitis (Sator et al. 2003) and ichthyosis (Ohman and Vahlquist 1998).
Dermal Evaluation
Microcirculation or Microvascular System
The skin’s nutrient vessels are a vital component of its anatomy and physiology. There are three types of vascular networks in the skin: subcutaneous, perifollicular, and periglandular. The subcutaneous network consists of a visible capillary ring and a horizontal plexus of vessels in the superficial dermis (Yousef et al. 2023), enabling the exchange of substances between the intravascular and extravascular compartments.
The perifollicular network nourishes the sebaceous follicles and contains a well-developed papillary ring and vascular network surrounding the outer epithelium of the follicle. The periglandular vascular network is densely distributed around each sweat gland and duct, extending into the epidermis. The development of vascular networks around hair follicles and sweat glands is most advanced in the middle and deep dermis. Dermal blood vessels act solely as transmitting vessels rather than exchanging vessels, and the subcutaneous circulation is sustained by the gradual branching of small arteries in the middle and upper dermis. The deep dermal vascular network is derived from a vast network of fascial arteries supplied by the cutaneous arteries, which then directly feed into the progressive network of small arteries and adnexal vessels (Mauroux et al. 2023).
Varies lymphatic markers, such as hyaluronic acid receptor-1, rat homeobox gene transcription factor 1 (Prox-1), podoplanin, and vascular endothelial growth factor receptor 3 (VEGFR-3), can be qualified for diagnostic purposes. In addition, imaging techniques such as RCM, OCT, and multispectral photoacoustic imaging (MSOT) can capture and evaluate various features, including angiography information. These image features can be further correlated with melanin and hemoglobin content in MSOT and histopathological findings (Li et al. 2022b).
Lymphatic Vascular Microcirculation
The lymphatic system maintains body fluid balance by reabsorbing extracellular fluid and transporting lymph back into circulation. Lymph fluid originates from intercellular fluid and flows through lymphatic capillaries, merging into the collecting lymphatic vessels and ultimately returning into the bloodstream. Lymphatic vessels are present in the dermis but not in the epidermis. The lymphatic system has roles in immune function and could increase the metastatic potential of primary skin tumors (Smile et al. 2022). Langerhans cells travel through lymphatic channels to the lymph nodes. The lymphatic system develops from embryonic veins alongside the cardiovascular system (Fredman et al. 2021).
To confirm the presence of lymphatic vessels in skin, immunohistochemical staining uses specific lymphatic markers (Lee et al. 2021).
Skin Elasticity
Skin elasticity depends on collagen, elastin, and fat content, which determine the skin’s ability to stretch and return to its original shape. Resistance of collagen bundles to deformation from external pressure significantly affects skin elasticity. Skin's biomechanical properties reflect complex interactions between the epidermis, dermis, and subcutaneous tissue, affecting its overall elasticity, viscosity, and plasticity (Wahlsten et al. 2023).
Two types of methods exist for measuring the elasticity of the stratum corneum. These commonly used methods can be categorized into two groups: the skin torque meter and the skin tester. The Dermal Torque Meter (DTM) developed by Dia-Stron Ltd uses a torque applied parallel to the skin surface as a stress; while, the Cutometer SEM575 developed by Courage Khazaka applies a suction force (vacuum) perpendicular to the skin surface as a stress.
The decline in skin elasticity is a key characteristic of human aging (Rebehn et al. 2023), as well as a primary feature of diseases caused by mutations in the elastin gene, commonly referred to as cutis laxa (Schmelzer and Duca 2022).
Dermatoglyphs
Dermatoglyphs mainly refers to the distinctive physiological patterns present on the surfaces of the palms and soles, with the fingertips and toes being the areas of highest concentration (Glover et al. 2023; Wang 2013). These patterns can be classified into two types: convex patterns formed by elevated elevations that persist throughout an individual's lifetime, and concave patterns formed by skin furrows that can change over time (Glover et al. 2023).
Finger or Toe Print
There are three primary types of fingerprints: arch, loop, and whorl. In the arch fingerprint pattern, the ridge line runs from one side of the finger to the other side, creating a curved shape. The loop pattern begins on one side of the finger, passes through the center, form a semicircle, and then returns to the starting point. The whorl pattern is more complex than the arch and loop patterns, and includes several different sub-patterns, such as a central pattern resembling a sac, a ring-shaped pattern with concentric circles, a spiral or screw-shaped pattern, and a central pattern consisting of a ruler and a radial loop (Glover et al. 2023; Wang 2013).
Palm Print
The palm can be divided into four main areas: the thenar area, the hypothenar area, the interphalangeal area, and the digital triradius. Additionally, there are seven trigeminal areas and several palmar creases. The majority of people have three primary folds on their palms, referred to as the first, second, and third flexor creases. Together, these creases form an overall pattern, which can be described as either claw shaped or Chuan shaped (Wang 2013). However, in some individuals, the wrinkle pattern on the palm can vary and may appear bridged, forked, of medium strength, or continuous.
Methods for Collecting Dermatoglyphs Samples
The most common method at present is the impression clay method, which utilizes uncomplicated equipment. This method is cost effective, allows for large-scale data collection, and facilitates long-term preservation (Li et al. 2021a). The chalk–carbon ink–transparent tape method is primarily used in phalangeal configuration studies, while specialized methods exist for hand–foot deformities and recessive dermatoglyphics manifestation. Dermatoglyphics are polygenic traits that are associated with individual susceptibility. Li et al (2022a) identified multiple genetic loci and confirmed the polygenic structure of dermatoglyphs through a genome-wide association study conducted on Han Chinese cohorts. Subsequent studies revealed distinct developmental patterns between fingerprints and hair (Glover et al. 2023). Dermatoglyphics exhibit unique individual differences and remain constant throughout a person's life. As a result, dermatoglyphics, particularly fingerprints, can be utilized for individual identification (Yoon and Jain 2015). Cohort studies have indicated a potential link between ADAMTS9-AS2 and fingerprint formation (Ho et al. 2016), highlighting the importance of epigenetic regulation guided by long non-coding RNA (lncRNA) during early human development (Walsh et al. 2016). Furthermore, changes in dermatoglyphics can be utilized for the early diagnosis of specific genetic disorders affecting the skeletal system, particularly for estimating the timing of certain congenital limb deformities because dermatoglyphic patterns are formed before the 14th week of gestation in the fetus and generally remain stable thereafter (Glover et al. 2023). Additionally, dermatoglyphic changes can be employed as a screening tool for non-congenital limb deformities including psoriasis (Dogramaci et al. 2010), pseudohypoparathyroidism (Forbes 1964), Down syndrome (Achs et al. 1966), schizophrenia (Shamir et al. 2013), etc. However, it is important to note that these changes are not entirely specific, thereby serving as a supplementary diagnostic approach or initial screening method for such conditions. And in China, fingerprints offer valuable insights into Chinese medicine diagnosis and treatment.
Skin Appendages
Sweat Glands
The eccrine and apocrine sweat glands have different functions. Eccrine sweat gland secretions respond to temperature, emotion, and gustatory stress, regulated by cholinergic sympathetic nerve stimulation with acetylcholine. Apocrine sweat glands, found in specific areas and activated during adolescence, produce pheromones, make zone, signals warnings, and producing odor (Li et al. 2021a). Of particular interest are hyperhidrosis (Nawrocki and Cha 2019) and bromhidrosis (Malik et al. 2023), which can significantly impact social interactions.
Methods for examining eccrine gland function include direct weighing, water evaporation quantification, sweat staining visualization, staining of specific organic sweat compounds, mold making, and skin capacitance mapping, with the latter being more commonly used (Li et al. 2021a; Yang et al. 2023). Osmidrosis assessment requires limiting armpit hair removal, use of cleansing products, deodorant, perfume, and armpit use frequently. Chromatographic analysis provides indirect assessment but does not permit quantitative measurements (Li et al. 2021a; Yang et al. 2023). Furthermore, sweat glands can also be utilized as diagnostic biological fluids. Recent advancements in engineering have made it possible to utilize microfluidic systems at the skin interface to analyze endocrine sweat, providing valuable insights into an individual's health status and various diseases (Yang et al. 2023).
Hairs
As an important appendage of the human body, hair has gained increasing attention due to its health and esthetic benefits, as well as its role in regulating temperature and protecting against UV radiation. In modern society, having thick and healthy hair and minimal body hair is highly desirable. Hair follicles undergo a dynamic growth cycle that includes an anagen phase lasting for two–six years, a regression phase lasting for two–three weeks, and a resting phase for lasting two–three months. An estimated 90% of hairs are in the anagen phase, while only about 1% are in the regressive phase (Brown and Krishnamurthy 2023).
Clinical evaluation of hair disorders involves assessing baldness patterns, spot baldness, hair diameter, and hair density. Typically, hair is the densest on the head, with approximately 80–100 hairs/cm2 on the top of the head, and about 120–180 hairs/cm2 on the occipital area. However, hair density is much lower on the other areas of the body, with only 15–20 hairs/cm2 on the opisthenar and four–six times that density on the forehead and cheeks to the trunk and limbs.
Hair loss, particularly pattern hair loss (PHL), also known as androgenetic alopecia, is a significant concern. PHL is classified based on the shape of the frontal hairline and hair density on the top and front of the head, with types ranging from basic (L, M, C, U) to special (F and V). Hair loss is commonly accompanied by a receding hairline and thinning on the top of the head (Griggs et al. 2021; Workman and Piliang 2023). Technical detection of hairs and hair follicles involves various dimensions, briefly described below.
Hair loss is typically assessed through clinical symptoms like itching, desquamation, and scalp oil, and a comparison between hair loss and a healthy range of strands lost per day (Griggs et al. 2021; Workman and Piliang 2023). The hair pull test can be done to determine hair loss, with fewer than six hairs pulled indicating a negative result. In complex or specialized cases, hair pathology evaluation may be used (Valdebran et al. 2020). Full scalp photography technology analyzes and evaluates hair treatment by comparing the scalp before and after treatment for changes, while the contrast-enhanced phototrichogram (CT-PTG) analyzed transitions of hair follicles (Piérard-Franchimont et al. 2000).
Hair microscopy is a dermatology tool widely used to examine the scalp and hair. Through hair microscopy, hair structure elements such as shed hair (presence of hair papillae), hair shaft, follicular opening, perifollicular epidermis, and vascularity are analyzed to identify characteristic manifestations for different types of alopecia diseases and scalp-related skin lesions (Wallace and de Berker 2010). For example, alopecia areata presents with yellow dot, broken hair, black dot, increased vellus hair, and exclamation point hair. Alopecia androgenetica, on the other hand, is characterized by uneven hair shaft thickness and a more than 20% variation in diameter with a slightly depressed brown halo around the follicular orifice in early-stage lesions, which is known as perifollicular sign. Hair microscopy is an important complementary diagnostic method used to evaluate hair and scalp diseases. It can also be used for quantitative assessment of hair loss and treatment efficacy (Wallace and de Berker 2010).
Surface Sebum
Surface sebum is a complex mixture of nonpolar and epidermal lipids that can vary significantly among individuals and at different ages and body sites (Pappas 2009). Sebum levels are highest in areas with the most sebaceous glands, such as the face, back, and chest, and can be a crucial aspect of dermatology, cosmetics, and pharmaceuticals. The accuracy of measurement of sebum levels depends on the sampling and detection methods, with the sebumeter and cassette sebumeter being widely used clinical tools (Firooz et al. 2012; Man et al. 2009; Shi et al. 2015). The sebumeter measures sebum levels by placing a film on the skin for 30 s followed by spectrophotometric detection, while the cassette sebumeter uses oil spot photometry to determine sebum content by measuring transparency and transmittance of light. These tools are essential for determining the level and rate of sebum secretion on the skin surface, objectively classifying skin types, evaluating the effectiveness of skincare and hair-care products, and assessing sebaceous gland activity in various diseases to support drug design, development, and evaluation.
Nails
The measurement of nails can be divided into three areas: nail plate, nail fold, and nail bed, each evaluated with different testing tools to assess physiological and pathological conditions.
(1) Nail Plate Length: Nail growing from the nail matrix can be measured for length, mass, or thickness at both the histological and macroscopic levels using dermatoscopy with an integral scale. During testing, subjects should not push the nail cuticle toward the proximal end.
-
(2) Nail Plate Thickness, Roughness, and Color:
a) Nail Plate Thickness: Calipers are the easiest way to measure free and nail thickness. Electronic micrometer provides greater accuracy up to about 2 μm, while ultrasound graphs at 20 or 25 MHz are commonly used for measurement in other body locations.
b) Roughness of the Nail Plate: In vivo or impression-based measurements evaluate surface undulation. Impression-based measurements can eliminate chattering artifacts, while computer imaging analysis or optical profilometry can provide quantitative data for morphological changes such as nail hypertrophy, nail surface depressions, and grooves, indicating accelerated nail growth.
c) Nail Plate Color: Nail color change can occur due to varied reasons. A spectrophotometer effectively tests for nail pigmentation origin, including melanin, while Wood's lamp can identify drug-induced color changes. These diagnostic methods are valuable in assessing the causes of nail discoloration in dermatology.
(3) Nail Fold: Capillaroscopy can be used to examine the structure of superficial capillaries. The capillary loop in the nail fold in the physiological state is hairpin-shaped with two parallel longitudinal vessels. Pathological changes are manifested by branching vessels, increased length of vascular collaterals, giant capillaries, disorganized parallel structures, and microaneurysms (Sathe and Saleh 2023).
(4) Nail Bed: Nail bed evaluation methods include fluoroscopy, OCT, RCM, dual-channel flow cytometry, and Magnetic Resonance Imaging (MRI). Fluoroscopy is a cost-effective technique, useful for determining the cystic nature of the lesion, especially in atypical or deep lesions, and for diagnosing subungual glomus tumor, which has a different color. OCT measures infrared reflectance of tissues and serves as a diagnostic tool for non-melanoma skin tumors and nail fungal disease. RCM may be applied as an auxiliary diagnostic tool to observe the capillary loop and deeper layers of the nail bed. Dual-channel flow cytometry is used to distinguish free fungus and fungal hyphae bound with monoclonal antibodies, showing fungal fluorescence. MRI is an effective for localizing paronychia masses, such as subungual glomus tumor, and compares tumor density, fluid, and fat content of surrounding tissue (Li et al. 2021a).
Measurement Tools Commonly Used in Clinical Diagnosis and Treatment Work
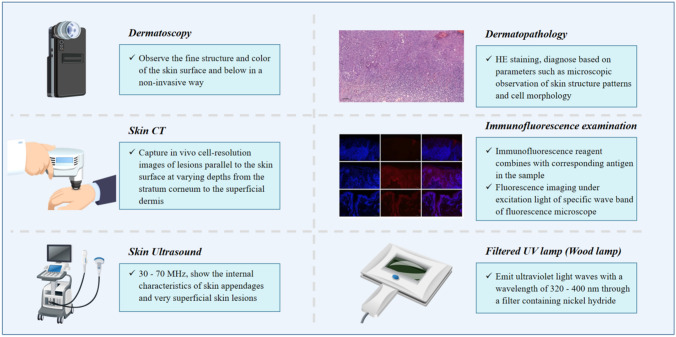
Measurement tools commonly used in clinical practice include dermoscopy and skin computed tomography (CT), which are utilized to evaluate the phenotype of a normal population, assess the physiology of the skin and appendages, or detect underlying pathology (as shown in Fig. 2).
Fig. 2.
Measurement tools commonly used in clinical diagnosis and treatment work
Dermatology Clinical Imaging Data Acquisition and Specification
In dermatology, the ability of a dermatologist to visually inspect the skin is crucial for the diagnosis of skin disease. Therefore, clinical images play a critical role in dermatological clinical imaging data. With the emergence of teledermatology, standardized skin clinical images of high quality are of significant importance for the diagnosis and treatment of skin diseases, as well as scientific research. Standardized skin images improve the comparability of images taken from different environments, photographers, and times. This consensus proposes several suggestions for optimizing and standardizing skin images, including image acquisition scene, lighting, equipment, and direction (as shown in Fig. 3).
(1) Scene: Indoor imaging is recommended for skin imaging as it simulates real clinical practice.
(2) Lighting: Lighting is a crucial factor in skin imaging, as it can significantly affect the color and contrast of skin lesions in images. Direct or flash photography can cause images to appear lighter than actual skin and reduce contrast, while darker light is also detrimental to image acquisition. Although natural light is the most recommended light source, it is not always practical in clinical practice and scientific research. Therefore, we suggest using wide-spectrum illumination and positioning the light source to the side of the area being photographed. Adding local light sources or using flash photography is not recommended (Finnane et al. 2017).
(3) Background: Reflected light from the background may change the color and appearance of the area we are photographing, so it is best to use a green background for all skin tones, with intermediate skin tones (yellow to brown) working best (Zhou et al. 2022). It has been suggested that black backgrounds are suitable for lighter skin tones while blue background is suitable for darker skin tones (Finnane et al. 2017). It is important to remove any potential interference around the lesion, such as jewelry or hats.
(4) Device: Ideal device for skin lesion imaging should accurately reflect the condition of the skin lesion with high resolution, without fuzziness in skin texture or hair, and without altering the color of the skin in images. Furthermore, future imaging devices should be easily accessible, operated, and promoted to avoid any bias in different dermatological centers. Previous studies have shown that cameras, phones, and tablets are commonly used devices for skin imaging, but none have been proven to be significantly superior to others (Maier et al. 2015; Panetta et al. 2023).
(5) Approach: The standardized shooting angles and viewing ranges can make the images comparable throughout the different courses of the disease. For lesions in the head or face, it is recommended to take at least three angles: one from the front, one from the left 45° side, and one from the right 45° side. For lesions on the trunk, one angle from the front and one angle from the left or right 90° side are recommended. For lesions on the extremity, it is recommended to include the two joints adjacent to the lesion in the photos to retain the anatomical location of the lesion in subsequent application. The shooting equipment should be kept perpendicular to the surface of the skin during image collection, and macro, close-up, and medium views are all needed to comprehensively understand the lesion for future use. It is recommended to capture three photos from each angle to facilitate subsequent analysis steps (Huang et al. 2023).
Fig. 3.
Dermatology clinical imaging data acquisition scene
The suggestions regarding skin image collection are essential because most skin diseases are chronic disorders, the follow-up care for patients with skin diseases is often required over a long period. Continuous and normative management of these patients is critical for their effective treatment.
Dermoscopy
Dermoscopy is a non-invasive device that uses optical magnification to observe the color and microstructure of the skin surface and below. It can improve clinicians’ sensitivity and specificity in distinguishing between benign and malignant skin lesions and aid in the differential diagnosis of pigmented nevi and malignant melanoma (Longo et al. 2023). Dermoscopy also enhances diagnostic accuracy for some pigmented and vascular lesions (Navarrete-Dechent et al. 2023) and non-pigmented lesions (Haenssle et al. 2020). Additionally, it can be used for trichoscopy to evaluate hair and diagnose scalp diseases.
Several methods are commonly for systematic differential diagnosis of pigmented skin lesions, including (Li et al. 2021a):
(1) Pattern Analysis: This method requires experience to identify melanocytic vs non-melanocytic lesions and evaluate specificity criteria.
(2) ABCD Rule: This semi-quantitative method scores skin lesions based on asymmetry (A), borders (B), color (C), and diameter (D).
(3) Menzies Scoring: This method scores eleven different criteria to diagnose melanoma.
(4) Seven-Point Evaluation: this newer method scores major and minor criteria in skin lesions with a total score of three or more indicating a 95% sensitivity for diagnosing melanoma.
(5) Three-Point Evaluation Method: This method simplifies the dermoscopic pattern analysis method to just three criteria: asymmetry, atypical pigment network, and blue–white structure. This method provides comparable diagnostic results in sensitivity and specificity to other complex algorithms without requiring extensive training.
Each method has its own advantages and limitations, and the choice of method depends on the clinical context and available resources.
Skin CT
Skin confocal microscopy, also referred to as RCM, is a non-invasive imaging tool used for visualizing skin. It operates based on the principle of optical confocal, whereby a laser beam passes through the illumination pinhole, reflects off the objective lens through a beam splitter, focuses on the skin, and scans each point on the inner focal plane of the skin. After scanning, the reflected light is directed back along the original incident light path, detected by the photomultiplier tube, and displayed as a black-and-white image on a display. Skin CT can capture cell-resolution images of lesions in vivo, at varying depths from the stratum corneum to the superficial dermis (Fredman et al. 2021). It has been widely researched to identify skin tumors, thereby reducing the need for biopsy of benign lesions. The scope of its applications has broadened to include preoperative tumor margin localization, monitoring tumor recurrence, non-invasive treatments monitoring, and inflammatory diseases classification.
Skin Ultrasound
Skin ultrasound is a modern alternative to traditional ultrasound for diagnostic purposes. Traditional ultrasound’s frequency range of 2–20 MHz lacks the resolution required to accurately observe the epidermis, making it seldom used for skin diseases diagnosis. However, technological advancements have produced high-resolution ultrasound with a resolution of 0.1 mm that’s capable of displaying skin and subcutaneous tissue layers. High-frequency skin ultrasound typically operates between 20 and 75 MHz and contains very high frequency between 20 and 30 MHz and ultra-high frequency between 30 and 75 MHz. Very-high-frequency (VHF) ultrasound is effective in displaying skin hierarchy and structure, while ultra-high-frequency ultrasound is ideal for displaying internal characteristics of skin appendages and superficial skin lesions (El Gammal et al. 1999). Skin ultrasound is valuable in differentiating between malignancies and benign skin tumors, measuring size and thickness, and determining invasiveness that helps develop an optimal surgical strategy. Skin ultrasounds also aid in auxiliary diagnosis and treatment of inflammatory, infectious, infectious, and physical skin diseases (Superficial Organs and Vasculology Group et al. 2022).
Dermatopathology
Pathology is responsible for diagnosing skin diseases by analyzing various parameters such as microscopic observation of skin structure patterns and cell morphology. The standard method for skin pathology diagnosis is hematoxylin–eosin (HE) staining, which is able to accurately diagnose many skin diseases. However, some diseases require special staining techniques such as periodic acid–Schiff (PAS) or hexamine silver staining to identify infectious substances such as fungi. Special stains like Giemsa or toluidine blue can also help to identify infiltrating cell types such as mast cells. A thorough skin histopathological examination is crucial for accurately diagnosing and differentiating skin diseases and determining their etiology.
Immunofluorescence Examination
Pathogen immunofluorescence technology identifies pathogenic microorganisms with fluorescent imaging through an immunofluorescence reagent. There are two methods: direct and indirect examination. Direct examination requires biopsy specimens to directly the antigen, while indirect examination requires blood and serum. This knowledge provides an important diagnosis tool for detecting microorganism infections in patients.
Filtered UV Lamp (Wood Lamp)
The wood lamp, equipped with a nickel hydride filter, emits UV light waves of 320–400 nm. The skin absorbs this radiation, resulting in local energy loss and visible range radiation with longer wavelengths. Conducted in darkness, the examination involves direct irradiation of the affected area under the Wood lamp to observe the type of fluorescence. Wood lamps are frequently employed to aid in diagnosing various skin conditions, including pigmented skin diseases (such as vitiligo, pityriasis albicans), skin bacterial infections (such as Pseudomonas aeruginosa, Corynebacterium microbes), skin fungal infections (including chromosporum rust, Microsporum canis, Malassezia Bacteria, etc.), and metabolic skin diseases (such as porphyria).
Skin and Appendage Phenotype Data Collection Sites
Physiological Indicators
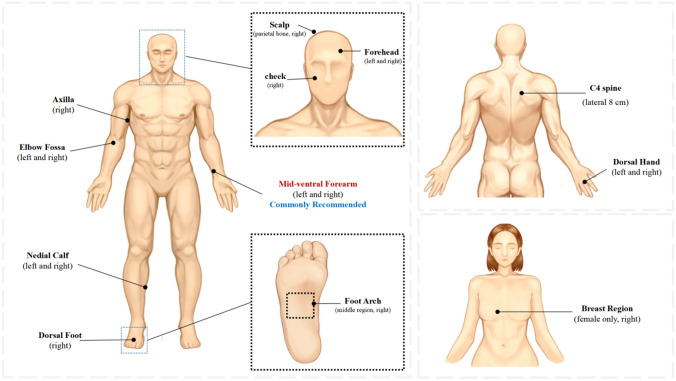
The forearm's palmar region is the traditional collection site for phenotypic skin and appendage measures (Van der Mei et al. 2003). However, anatomical sites vary in structure, composition, and exposure. For more comprehensive data, studies often extend collection sites to various body parts such as the mid-ventral forearm, dorsal and ventral hands, elbow fossa, scalp, forehead, axilla, spine, foot, calf (Firooz et al. 2012; Lintzeri et al. 2022), and breast (for females) (as shown in Fig. 4) (Kleesz et al. 2012). Collection sites can be adjusted depending on study goals. Further division into regions such as parieto-occipital, frontal, and cheek is necessary for studies limited to facial evaluation due to phenotypic variations and biophysical parameters (Firooz et al. 2012; Lee et al. 2013).
Fig. 4.
Skin and appendage phenotype data collection sites—physiological indicators
Skin temperatures varied across different body locations with the arch of the foot (27.6 °C) and the axilla (33.1 °C) recording the lowest and highest temperature, respectively. Other areas with relatively low temperatures were the dorsum of the foot, calf, and breast; while, the highest temperatures were recorded in the forehead, C4 spine, and axilla (Kleesz et al. 2012). Factors such as skin microcirculation, subcutaneous fat, and abdominal fat distribution significantly influence skin surface temperature (Wa and Maibach 2010). The study also found that skin temperature was higher on the right side of the dorsal hand, ventral hand, and forearm (Lee et al. 2013). Furthermore, the pH is lowest in the forehead and higher in the axilla and both right-hand sides with no significant differences between the right and left upper limbs.
Differences in skin color, erythema, pigmentation, temperature, and sun tolerance are factors influencing susceptibility to UV exposure (Kleesz et al. 2012). These differences can be explained by variations in microvasculature, skin vasodilation, and thermoregulatory sweating. UV exposure affects the cheek and forehead more due to a denser vascular network resulting in higher erythema and melanin indices. Lower legs have lower associated indices due to less sun exposure. In the assessment of the MI, hair can interfere, thus requiring separate consideration for the scalp and axilla. Areas with the lowest MI include the spine, arch of the foot, and ventral side of the hand where hyperpigmentation under UV exposure is uncommon.
TEWL is the predominant reference indicator for evaluating epidermal permeability barrier function. Sweat gland activity closely relates to TEWL and varies with anatomical site. Palmar side of the hand, inner arch of the foot, and the axilla show the highest TEWL values (Pinnagoda et al. 1990). Differences in epidermal density, thickness, degree of stratum corneum exfoliation, and corneal water content can be traced back to variations in morphology, thickness, and fat content of the stratum corneum across different anatomical regions. Corneal water content measurements are lowest on the scalp and highest in C4 region of spine, elbow fossa, forehead, cheeks, and axillae. Sebum level is significantly higher above the nasolabial fold than other areas. Forehead is the standard area for measuring sebaceous gland activity using a sebum meter (Kendall et al. 2018; Kleesz et al. 2012).
Skin elasticity is generally related to skin collagen content, elastic fiber network distribution, age, hormone levels, etc. Skin elasticity is generally higher in the neck and chest and lower in the legs (Firooz et al. 2012).
Disease-Related Collection Sites
Numerous studies have demonstrated that skin phenotypes undergo considerable modifications as a result of skin ailments such as atopic dermatitis (Jungersted et al. 2010). Skin samples for analysis are usually sourced from three areas: the lesion, uninvolved perilesional (approximately one centimeter away), and uninvolved symmetrical skin, all collected at the same time (Guttman-Yassky et al. 2019a, 2019b; Yazdanparast et al. 2018).
Scenarios and Requirements for Phenotypic Measurement of Skin and Appendages
Phenotypic Measurements in Natural Populations
The term natural population refers to the entire population, and phenotypic measurements aim to comprehensively obtain data on human skin and appendageal phenotypic measurements, including all detectable indicators. Hence, it is essential to comprehensively detect various regions and judiciously incorporate subjective scoring scales, such as the skin type self-test. This scenario requires skilled testing personnel, standardization of the process, and sufficient time for accurate results. Data collection occurs in a sealed room with constant temperature and humidity, typically found in research institutes or hospitals. Participants are required to register basic information, refrain from using cosmetics and cleaning products 24 h prior to the examination, and avoid water contact and skin care products on the test day. Measurements are taken in sequence and may include questionnaires, scans, and sampling. If the procedure causes discomfort, it should be stopped immediately.
Phenotypic Measurements for Epidemiological Studies
Epidemiological studies usually focus on specific cohorts or subpopulations within the general population, tailored to different research purposes. Measurement items are then simplified based on the requirements mentioned above, and the collection sites are appropriately reduced to align with the research objectives. The data collection takes place at a fixed point with multiple follow-up visits and repeated measurements, if necessary. The collection process involves at least two steps of diagnosis and evaluation. In the first step, dermatologists or graduate students with standardized training conduct an inquiry and whole-body skin examination, confirm and fill in the skin phenotypic indicators, and record them. In the second step, an experienced dermatologist confirms the positive indicators and assesses the situation. If possible, lesion image data can also be collected for review and diagnosis. Alternatively, artificial intelligence (AI) technology can be used to aid in the diagnosis.
Skin Health Physical Examination
Tailored for individuals requiring skin health screenings, this scenario is suitable for, including but not limited to skin health consultations, skincare guidance, dermatology, or medical beauty assessment before and after treatment. Items will be simplified according to the above requirements, with an emphasis on collecting samples from exposed areas and focusing on assessing skin and appendageal health, aging conditions, and aesthetic evaluation. The collection takes place mainly in major hospitals but may also occur in social organizations. Professional medical staff conducts the visual examination, starting with bright light, preferably natural light, and then fluorescent light. For more extensive skin diseases, the entire body’s skin is examined, including hair, finger or toe nails, and mucous membranes. After the visual examination, palpation is performed to assess skin hardness, the relationship with surrounding tissues, temperature, accessory lymph nodes, and other indicators. Finally, other phenotypic measurements are taken. After the examination, the study issues targeted skin physical examination reports and provides skin care guidance and esthetic suggestions.
Conclusions
As acousto-optic and AI technologies continue to develop at an unprecedented pace, compelling advancements in the area of skin and appendage phenotypic data measurements are being made. From bulk data to microstructural data and from invasive skin biopsy to non-invasive skin testing techniques, the field of dermatology is witnessing a remarkable shift. This nascent paradigm offers a remarkable opportunity for clinicians to evaluate skin disease conditions innovatively and with great efficiency. In particular, with the advent of deep learning and neural networks, computer-aided detection or diagnosis systems have made significant advancements in image extraction, detection, classification, and other aspects compared to traditional methods. Various algorithms leverage suitable preprocessing techniques to handle images, utilizing multi-channel networks to integrate and extract features for analysis. Currently, the primary sources of data for these systems are dermoscopic images or photographs of skin lesions. A comparative study conducted in 2017 between AI and 21 dermatology experts for the classification of skin cancer demonstrated that AI possesses a comparable ability to distinguish between benign and malignant tumors (Esteva et al. 2017). Similar capabilities have also been observed in areas such as the classification of pigmented skin lesions (Tschandl et al. 2019), diagnosis of inflammatory skin diseases (Raj et al. 2021), and diagnosis of autoimmune diseases (Wu et al. 2021). AI tools have also accelerated the objective quantification of phenotype data, aiding in the analysis of genetic facial features (Dingemans et al. 2023; Hallgrímsson et al. 2020) and dermatoglyphic characteristics (Wojtowicz and Wajs 2012), thus contributing to the interpretation of genetic variations. Deep learning-based models are increasingly utilized to enhance the accuracy of various biometric recognition systems (Minaee et al. 2023). Among commonly used biometric technologies, palmprints have the advantages of stable and rich line features, minimal distortion, and easy self-positioning (Fan et al. 2022; Fei et al. 2022; Shao and Zhong 2021; Wei et al. 2017). The extraction of phenotype features based on the main lines in palmprints facilitates large-scale population studies (Fan et al. 2022).
However, there remains a dearth of research conducted on Chinese population cohorts in this regard. Therefore, it is imperative to engage in a collaborative approach to capture phenotypic data from healthy skin and unhealthy skin, affected by various disease under different environmental conditions and age groups throughout the Chinese population, including physiological indicators, barrier function, dermatoglyphs, etc. These data serve as a reference point for developing a comprehensive skin multimodal database. It also helps ensure progressive and objective measurements to facilitate accurate diagnosis and treatment of diseases with greater precision. By gathering phenotypic data for different diseases and incorporating information regarding the patient's individual circumstances, disease presentation, physical signs, prognosis, and more, we strive to delve into the genetic and environmental factors influencing skin phenotypes. This data analysis also aids us in identifying phenotypic features that can predict patient outcomes and advances our understanding of the pathophysiology, etiology, and treatment approaches for skin diseases.
Additionally, we seek to leverage AI tools to develop cutting-edge measurement robots capable of conducting rapid, accurate and non-invasive skin and appendage phenotype measurements. This technology offers a valuable tool to assist primary care physicians, who often work in highly unbalanced doctor-patient ratio context. Moving forward, we aim to investigate the synergistic integration of AI and emerging technologies like extremely weak magnetism and hyperspectral imaging. Incorporating these novel techniques will enhance the objectivity and standardization of phenotype data measurement and collection. By streaming the measurement process with sophisticated and integrated tools, we stand to benefit significantly from a more vibrant, automated and comprehensive strategy in skin phenotypic data collection and analysis.
Abbreviations
- TEWL
Transepidermal water loss
- RCM
Reflectance confocal microscopy
- OCT
Optical coherence tomography
- PASI
Psoriasis area and severity index
- UV
Ultraviolet
- AVA
Arteriovenous anastomosis
- ITA
Individual typology angle
- PIH
Post-inflammatory pigmentation
- EI
Erythema index
- MI
Melanin index
- LED
Light-emitting diode
- MED
Minimum erythema dose
- SPF
Sun protection factor
- MPPD
Minimum persistent pigment darkening
- UVA
Long-wave ultraviolet
- UVB
Medium-wave ultraviolet
- Prox-1
Rat homeobox gene transcription factor 1
- VEGFR-3
Vascular endothelial growth factor receptor 3
- MSOT
Multispectral photoacoustic imaging
- DTM
Dermal Torque Meter
- lncRNA
Long non-coding RNA
- PHL
Pattern hair loss
- CT-PTG
Contrast-enhanced phototrichogram
- MRI
Magnetic resonance imaging
- CT
Computed tomography
- ABCD
Asymmetry, borders, color, and diameter
- VHF
Very high frequency
- HE
Hematoxylin–eosin
- PAS
Periodic acid–Schiff
- AI
Artificial intelligence
Authors’ Contributions
SZ, ZLL, and YW: Design, drafting, revision and approval of final manuscript. XHG, JT, and YC: Concept and design, data interpretation and analysis, drafting, revision and approval of final manuscript. AJC, DXC, YD, HG, JYG, CJ, XJK, QJL, CZL, ML, WL (Wei Li), WL (Wei Liu), XL, YZL, XYM, JJQ, LDS, YLS, WYW, JXX, RX, BY, YHK, and MZ: Design, drafting, revision and approval of final manuscript. ZYC, JYF, JK, and MHY: Data collection, data interpretation and analysis, revision and approval of final manuscript. XC, XJZ, and JS: Concept and design, data interpretation and analysis, revision and approval of final manuscript.
Funding
This study was partially sponsored by the Project of Intelligent Management Software for Multimodal Medical Big Data for New Generation Information Technology, Ministry of Industry and Information Technology of People’s Republic of China (TC210804V), and the Research on the Precision Prevention and Treatment System for Skin Malignant Tumors, National Key Research and Development Program of China (2022YFC2504701, 2022YFC2504705).
Availability of Data and Materials
Not applicable.
Declarations
Conflict of interest
On behalf of all authors, the corresponding authors state that there is no conflict of interest.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Footnotes
Shuang Zhao, Zhongling Luo and Ying Wang are co-first authors contributing to the manuscript equally.
Contributor Information
Juan Su, Email: sujuanderm@csu.edu.cn.
Xuejun Zhang, Email: ayzxj@vip.sina.com.
Xiang Chen, Email: chenxiangck@126.com.
References
- Achs R, Harper RG, Harrick NJ (1966) Unusual dermatolyphics associated with major congenital malformations. N Engl J Med 275(23):1273–1278. 10.1056/nejm196612082752303 [DOI] [PubMed] [Google Scholar]
- Agache P (2004) Thermometry and remote thermography. In: Handbook of Measuring the skin 1st edn. Berlin: Springer, pp 354–362
- Alexander H, Brown S, Danby S et al (2018) Research Techniques Made Simple: Transepidermal Water Loss Measurement as a Research Tool. J Invest Dermatol 138(11):2295-2300.e2291. 10.1016/j.jid.2018.09.001 [DOI] [PubMed] [Google Scholar]
- Arsiwala SZ, Desai SR (2019) Fractional Carbon Dioxide Laser: Optimizing Treatment Outcomes for Pigmented Atrophic Acne Scars in Skin of Color. J Cutan Aesthet Surg 12(2):85–94. 10.4103/jcas.Jcas_171_18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bajpai VK, Swigut T, Mohammed J et al (2023) A genome-wide genetic screen uncovers determinants of human pigmentation. Science (new York, NY) 381(6658):eade6289. 10.1126/science.ade6289 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baquié M, Kasraee B (2014) Discrimination between cutaneous pigmentation and erythema: comparison of the skin colorimeters Dermacatch and Mexameter. Skin research and technology : official journal of International Society for Bioengineering and the Skin (ISBS) [and] International Society for Digital Imaging of Skin (ISDIS) [and] International Society for Skin Imaging (ISSI) 20(2):218–227. 10.1111/srt.12109 [DOI] [PubMed]
- Brown TM, Krishnamurthy K (2023) Histology, hair and follicle. In: StatPearls. StatPearls Publishing, Treasure Island (FL). https://www.ncbi.nlm.nih.gov/books/NBK532929/ [PubMed]
- Caldwell JN, Matsuda-Nakamura M, Taylor NA (2014) Three-dimensional interactions of mean body and local skin temperatures in the control of hand and foot blood flows. Eur J Appl Physiol 114(8):1679–1689. 10.1007/s00421-014-2894-x [DOI] [PubMed] [Google Scholar]
- Civan L, Kurama S (2023) Preparation and characterization of intelligent thermochromic fabric coatings for the detection of fever diseases. Mater Chem Phys 305:127977. 10.1016/j.matchemphys.2023.127977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Comaish S, Bottoms E (1971) The skin and friction: deviations from Amonton’s laws, and the effects of hydration and lubrication. Br J Dermatol 84(1):37–43. 10.1111/j.1365-2133.1971.tb14194.x [DOI] [PubMed] [Google Scholar]
- Cork MJ, Danby SG, Vasilopoulos Y et al (2009) Epidermal barrier dysfunction in atopic dermatitis. J Invest Dermatol 129(8):1892–1908. 10.1038/jid.2009.133 [DOI] [PubMed] [Google Scholar]
- Crowther JM, Sieg A, Blenkiron P et al (2008) Measuring the effects of topical moisturizers on changes in stratum corneum thickness, water gradients and hydration in vivo. Br J Dermatol 159(3):567–577. 10.1111/j.1365-2133.2008.08703.x [DOI] [PubMed] [Google Scholar]
- Del Bino S, Bernerd F (2013) Variations in skin colour and the biological consequences of ultraviolet radiation exposure. Br J Dermatol 169(Suppl 3):33–40. 10.1111/bjd.12529 [DOI] [PubMed] [Google Scholar]
- Dingemans AJM, Hinne M, Truijen KMG et al (2023) PhenoScore quantifies phenotypic variation for rare genetic diseases by combining facial analysis with other clinical features using a machine-learning framework. Nat Genet 55(9):1598–1607. 10.1038/s41588-023-01469-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dogramaci AC, Yenin JZ, Bagriacik MA et al (2010) Dermatoglyphs in patients with psoriasis. J Eur Acad Dermatol Venereol 24(1):88–89. 10.1111/j.1468-3083.2009.03294.x [DOI] [PubMed] [Google Scholar]
- Eichenfield DZ, Sprague J, Eichenfield LF (2021) Management of Acne Vulgaris: A Review. JAMA 326(20):2055–2067. 10.1001/jama.2021.17633 [DOI] [PubMed] [Google Scholar]
- El Gammal S, El Gammal C, Kaspar K et al (1999) Sonography of the skin at 100 MHz enables in vivo visualization of stratum corneum and viable epidermis in palmar skin and psoriatic plaques. J Invest Dermatol 113(5):821–829. 10.1046/j.1523-1747.1999.00795.x [DOI] [PubMed] [Google Scholar]
- Esteva A, Kuprel B, Novoa RA et al (2017) Dermatologist-level classification of skin cancer with deep neural networks. Nature 542(7639):115–118. 10.1038/nature21056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fajuyigbe D, Young AR (2016) The impact of skin colour on human photobiological responses. Pigment Cell Melanoma Res 29(6):607–618. 10.1111/pcmr.12511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan Y, Li J, Song S, Zhang H et al (2022) Palmprint Phenotype Feature Extraction and Classification Based on Deep Learning. Phenomics (cham, Switzerland) 2(4):219–229. 10.1007/s43657-022-00063-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fei L, Zhao S, Jia W et al (2022) Toward Efficient Palmprint Feature Extraction by Learning a Single-Layer Convolution Network. IEEE Transact Neural Netw Learn Syst. 10.1109/tnnls.2022.3160597 [DOI] [PubMed] [Google Scholar]
- Finnane A, Curiel-Lewandrowski C, Wimberley G et al (2017) Proposed Technical Guidelines for the Acquisition of Clinical Images of Skin-Related Conditions. JAMA Dermatol 153(5):453–457. 10.1001/jamadermatol.2016.6214 [DOI] [PubMed] [Google Scholar]
- Firooz A, Sadr B, Babakoohi S et al (2012) Variation of biophysical parameters of the skin with age, gender, and body region. Sci World J 2012:386936. 10.1100/2012/386936 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fitzpatrick TB (1988) The validity and practicality of sun-reactive skin types I through VI. Arch Dermatol 124(6):869–871. 10.1001/archderm.124.6.869 [DOI] [PubMed] [Google Scholar]
- Forbes AP (1964) Fingerprints and palm prints (dermatoglyphics) and palmar-flexion creases in gonadal dysgenesis, pseudohypoparathyroidism and Klinefelter’s syndrome. N Engl J Med 270:1268–1277. 10.1056/nejm196406112702402 [DOI] [PubMed] [Google Scholar]
- Fredman G, Qiu Y, Ardigò M et al (2021) Skin tags imaged by reflectance confocal microscopy, optical coherence tomography and multispectral optoacoustic tomography at the bedside. Skin research and technology : official journal of International Society for Bioengineering and the Skin (ISBS) [and] International Society for Digital Imaging of Skin (ISDIS) [and] International Society for Skin Imaging (ISSI) 27(3):324–331. 10.1111/srt.12943 [DOI] [PubMed]
- Giusti F, Martella A, Bertoni L et al (2001) Skin barrier, hydration, and pH of the skin of infants under 2 years of age. Pediatr Dermatol 18(2):93–96. 10.1046/j.1525-1470.2001.018002093.x [DOI] [PubMed] [Google Scholar]
- Glover JD, Sudderick ZR, Shih BB et al (2023) The developmental basis of fingerprint pattern formation and variation. Cell 186(5):940-956.e920. 10.1016/j.cell.2023.01.015 [DOI] [PubMed] [Google Scholar]
- Griggs J, Burroway B, Tosti A (2021) Pediatric androgenetic alopecia: A review. J Am Acad Dermatol 85(5):1267–1273. 10.1016/j.jaad.2019.08.018 [DOI] [PubMed] [Google Scholar]
- Guttman-Yassky E, Bissonnette R, Ungar B et al (2019a) Dupilumab progressively improves systemic and cutaneous abnormalities in patients with atopic dermatitis. J Allergy Clin Immunol 143(1):155–172. 10.1016/j.jaci.2018.08.022 [DOI] [PubMed] [Google Scholar]
- Guttman-Yassky E, Diaz A, Pavel AB et al (2019b) Use of Tape Strips to Detect Immune and Barrier Abnormalities in the Skin of Children With Early-Onset Atopic Dermatitis. JAMA Dermatol 155(12):1358–1370. 10.1001/jamadermatol.2019.2983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haenssle HA, Fink C, Toberer F et al (2020) Man against machine reloaded: performance of a market-approved convolutional neural network in classifying a broad spectrum of skin lesions in comparison with 96 dermatologists working under less artificial conditions. Ann Oncol off J Eur Soc Med Oncol 31(1):137–143. 10.1016/j.annonc.2019.10.013 [DOI] [PubMed] [Google Scholar]
- Hallgrímsson B, Aponte JD, Katz DC et al (2020) Automated syndrome diagnosis by three-dimensional facial imaging. Genet Med off J Am Coll Med Genet 22(10):1682–1693. 10.1038/s41436-020-0845-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heng AHS, Chew FT (2020) Systematic review of the epidemiology of acne vulgaris. Sci Rep 10(1):5754. 10.1038/s41598-020-62715-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho YYW, Evans DM, Montgomery GW et al (2016) Common Genetic Variants Influence Whorls in Fingerprint Patterns. J Invest Dermatol 136(4):859–862. 10.1016/j.jid.2015.10.062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang K, Wu X, Li Y et al (2023) Artificial Intelligence-Based Psoriasis Severity Assessment: Real-world Study and Application. J Med Internet Res 25:e44932. 10.2196/44932 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hülpüsch C, Tremmel K, Hammel G et al (2020) Skin pH-dependent Staphylococcus aureus abundance as predictor for increasing atopic dermatitis severity. Allergy 75(11):2888–2898. 10.1111/all.14461 [DOI] [PubMed] [Google Scholar]
- Jimenez E, Valle P, Villegas C et al (1994) Unusual acquired dermal melanocytosis. J Am Acad Dermatol 30(2 Pt 1):277–278. 10.1016/s0190-9622(08)81929-9 [DOI] [PubMed] [Google Scholar]
- Jo DJ, Shin JY, Na SJ (2022) Evaluation of changes for sebum, skin pore, texture, and redness before and after sleep in oily and nonoily skin. Skin Res Technol 28(6):851–855. 10.1111/srt.13224 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung YC, Kim EJ, Cho JC et al (2013) Effect of skin pH for wrinkle formation on Asian: Korean, Vietnamese and Singaporean. J Eur Acad Dermatol Venereol 27(3):e328-332. 10.1111/j.1468-3083.2012.04660.x [DOI] [PubMed] [Google Scholar]
- Jungersted JM, Scheer H, Mempel M et al (2010) Stratum corneum lipids, skin barrier function and filaggrin mutations in patients with atopic eczema. Allergy 65(7):911–918. 10.1111/j.1398-9995.2010.02326.x [DOI] [PubMed] [Google Scholar]
- Kendall AC, Koszyczarek MM, Jones EA et al (2018) Lipidomics for translational skin research: A primer for the uninitiated. Exp Dermatol 27(7):721–728. 10.1111/exd.13558 [DOI] [PubMed] [Google Scholar]
- Keurentjes AJ, Jakasa I, Kezic S (2021) Research Techniques Made Simple: Stratum Corneum Tape Stripping. J Invest Dermatol 141(5):1129-1133.e1121. 10.1016/j.jid.2021.01.004 [DOI] [PubMed] [Google Scholar]
- Kleesz P, Darlenski R, Fluhr JW (2012) Full-body skin mapping for six biophysical parameters: baseline values at 16 anatomical sites in 125 human subjects. Skin Pharmacol Physiol 25(1):25–33. 10.1159/000330721 [DOI] [PubMed] [Google Scholar]
- Kovacs D, Cardinali G, Aspite N et al (2010) Role of fibroblast-derived growth factors in regulating hyperpigmentation of solar lentigo. Br J Dermatol 163(5):1020–1027. 10.1111/j.1365-2133.2010.09946.x [DOI] [PubMed] [Google Scholar]
- Kundu RV, Aderibigbe O, Riley JM (2023) Managing facial hyperpigmentation. JAMA Dermatol 159(7):778–779. 10.1001/jamadermatol.2023.1414 [DOI] [PubMed] [Google Scholar]
- Lee M, Jung Y, Kim E (2023) Study for skin gloss perception using computer modulation and frequency separation on surface reflection. Int J Cosmet Sci 45(4):500–511. 10.1111/ics.12859 [DOI] [PubMed] [Google Scholar]
- Lee MR, Nam GW, Jung YC et al (2013) Comparison of the skin biophysical parameters of Southeast Asia females: forehead-cheek and ethnic groups. J Eur Acad Dermatol Venereol 27(12):1521–1526. 10.1111/jdv.12042 [DOI] [PubMed] [Google Scholar]
- Lee SJ, Im ST, Wu J et al (2021) Corneal lymphangiogenesis in dry eye disease is regulated by substance P/neurokinin-1 receptor system through controlling expression of vascular endothelial growth factor receptor 3. Ocul Surf 22:72–79. 10.1016/j.jtos.2021.07.003 [DOI] [PubMed] [Google Scholar]
- Li H, Li L (2009) Chinese facial skin classification and skincare guidelines. J Dermatol Venerol 31(4):14–15. 10.3969/j.issn.1002-1310.2009.04.007 [Google Scholar]
- Li J, Glover JD, Zhang H et al (2022a) Limb development genes underlie variation in human fingerprint patterns. Cell 185(1):95-112.e118. 10.1016/j.cell.2021.12.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li L, Li H, Wei LMt (2021a) Agache's Measuring the Skin eng (2). (France)Humbert, Philippe
- Li X, Moothanchery M, Kwa CY et al (2022b) Multispectral raster-scanning optoacoustic mesoscopy differentiate lesional from non-lesional atopic dermatitis skin using structural and functional imaging markers. Photoacoustics 28:100399. 10.1016/j.pacs.2022.100399 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z, Xia J, Jiang L et al (2021b) Characterization of the human skin resistome and identification of two microbiota cutotypes. Microbiome 9(1):47. 10.1186/s40168-020-00995-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lintzeri DA, Karimian N, Blume-Peytavi U et al (2022) Epidermal thickness in healthy humans: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol 36(8):1191–1200. 10.1111/jdv.18123 [DOI] [PubMed] [Google Scholar]
- Longo C, Pampena R, Moscarella E et al (2023) Dermoscopy of melanoma according to different body sites: Head and neck, trunk, limbs, nail, mucosal and acral. J Eur Acad Dermatol Venereol 37(9):1718–1730. 10.1111/jdv.19221 [DOI] [PubMed] [Google Scholar]
- Maier T, Kulichova D, Schotten K et al (2015) Accuracy of a smartphone application using fractal image analysis of pigmented moles compared to clinical diagnosis and histological result. J Eur Acad Dermatol Venereol 29(4):663–667. 10.1111/jdv.12648 [DOI] [PubMed] [Google Scholar]
- Malik AS, Porter CL, Feldman SR (2023) Bromhidrosis treatment modalities: A literature review. J Am Acad Dermatol 89(1):81–89. 10.1016/j.jaad.2021.01.030 [DOI] [PubMed] [Google Scholar]
- Man MQ, Xin SJ, Song SP et al (2009) Variation of skin surface pH, sebum content and stratum corneum hydration with age and gender in a large Chinese population. Skin Pharmacol Physiol 22(4):190–199. 10.1159/000231524 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin AR, Lin M, Granka JM et al (2017) An Unexpectedly Complex Architecture for Skin Pigmentation in Africans. Cell 171(6):1340-1353.e1314. 10.1016/j.cell.2017.11.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marini A, Aue N, Demessant AL et al (2023) Increased skin pH and transepidermal water loss may serve as predictor for atopic dermatitis flares. J Eur Acad Dermatol Venereol 37(2):e252–e254. 10.1111/jdv.18558 [DOI] [PubMed] [Google Scholar]
- Mauroux A, Joncour P, Brassard-Jollive N et al (2023) Papillary and reticular fibroblasts generate distinct microenvironments that differentially impact angiogenesis. Acta Biomater 168:210–222. 10.1016/j.actbio.2023.06.040 [DOI] [PubMed] [Google Scholar]
- Mehta HH, Nikam VV, Jaiswal CR et al (2018) A cross-sectional study of variations in the biophysical parameters of skin among healthy volunteers. Indian J Dermatol Venereol Leprol 84(4):521. 10.4103/ijdvl.IJDVL_1151_15 [DOI] [PubMed] [Google Scholar]
- Minaee S, Abdolrashidi A, Su H et al (2023) Biometrics recognition using deep learning: a survey. Artif Intell Rev 56(8):8647–8695. 10.1007/s10462-022-10237-x [Google Scholar]
- Montero-Vilchez T, Segura-Fernández-Nogueras MV, Pérez-Rodríguez I et al (2021) Skin Barrier Function in Psoriasis and Atopic Dermatitis: Transepidermal Water Loss and Temperature as Useful Tools to Assess Disease Severity. J Clin Med. 10.3390/jcm10020359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Navarrete-Dechent C, Jaimes N, Dusza SW et al (2023) Perifollicular linear projections: A dermoscopic criterion for the diagnosis of lentigo maligna on the face. J Am Acad Dermatol. 10.1016/j.jaad.2023.07.1036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nawrocki S, Cha J (2019) The etiology, diagnosis, and management of hyperhidrosis: A comprehensive review: Etiology and clinical work-up. J Am Acad Dermatol 81(3):657–666. 10.1016/j.jaad.2018.12.071 [DOI] [PubMed] [Google Scholar]
- Ohman H, Vahlquist A (1998) The pH gradient over the stratum corneum differs in X-linked recessive and autosomal dominant ichthyosis: a clue to the molecular origin of the “acid skin mantle”? J Invest Dermatol 111(4):674–677. 10.1046/j.1523-1747.1998.00356.x [DOI] [PubMed] [Google Scholar]
- Palmer RA, Friedmann PS (2004) Ultraviolet radiation causes less immunosuppression in patients with polymorphic light eruption than in controls. J Invest Dermatol 122(2):291–294. 10.1046/j.0022-202X.2004.22213.x [DOI] [PubMed] [Google Scholar]
- Panetta K, K MS, Rao SP et al (2023) Deep Perceptual Image Enhancement Network for Exposure Restoration. IEEE Transact Cybern 53(7):4718-4731. 10.1109/tcyb.2021.3140202 [DOI] [PubMed]
- Pappas A (2009) Epidermal Surface Lipids. Dermato-Endocrinology 1(2):72–76. 10.4161/derm.1.2.7811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piérard-Franchimont C, Henry F, Piérard GE (2000) The SACD method and the XLRS squamometry tests revisited. Int J Cosmet Sci 22(6):437–446 [PubMed] [Google Scholar]
- Piérard-Franchimont C, Quatresooz P, Piérard GE (2011) Specular light reflectance of flakes in seborrhoeic dermatitis of the scalp: a pilot study. Clin Exp Dermatol 36(7):793–796. 10.1111/j.1365-2230.2011.04120.x [DOI] [PubMed] [Google Scholar]
- Pinnagoda J, Tupker RA, Agner T et al (1990) Guidelines for transepidermal water loss (TEWL) measurement. A report from the Standardization Group of the European Society of Contact Dermatitis. Contact Dermatitis 22(3):164–178. 10.1111/j.1600-0536.1990.tb01553.x [DOI] [PubMed] [Google Scholar]
- Raj R, Londhe ND, Sonawane R (2021) Automated psoriasis lesion segmentation from unconstrained environment using residual U-Net with transfer learning. Comput Methods Programs Biomed 206:106123. 10.1016/j.cmpb.2021.106123 [DOI] [PubMed] [Google Scholar]
- Ravnbak MH, Philipsen PA, Wulf HC (2010) The minimal melanogenesis dose/minimal erythema dose ratio declines with increasing skin pigmentation using solar simulator and narrowband ultraviolet B exposure. Photodermatol Photoimmunol Photomed 26(3):133–137. 10.1111/j.1600-0781.2010.00508.x [DOI] [PubMed] [Google Scholar]
- Rebehn L, Khalaji S, KleinJan F et al (2023) The weakness of senescent dermal fibroblasts. Proc Natl Acad Sci USA 120(34):e2301880120. 10.1073/pnas.2301880120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Requena L, Sangueza OP (1997) Cutaneous vascular anomalies Part I Hamartomas, malformations, and dilation of preexisting vessels. J Am Acad Dermatol 37(4):523–549. 10.1016/s0190-9622(97)70169-5. (quiz 549-552) [DOI] [PubMed] [Google Scholar]
- Sathe NC, Saleh HM (2023) Onychoscopy. In: StatPearls. StatPearls Publishing, Treasure Island (FL). https://www.ncbi.nlm.nih.gov/books/NBK585099/ [Google Scholar]
- Sator PG, Schmidt JB, Hönigsmann H (2003) Comparison of epidermal hydration and skin surface lipids in healthy individuals and in patients with atopic dermatitis. J Am Acad Dermatol 48(3):352–358. 10.1067/mjd.2003.105 [DOI] [PubMed] [Google Scholar]
- Schmelzer CEH, Duca L (2022) Elastic fibers: formation, function, and fate during aging and disease. FEBS J 289(13):3704–3730. 10.1111/febs.15899 [DOI] [PubMed] [Google Scholar]
- Schuler CFt, O'Shea KM, Troost JP et al (2023) Transepidermal water loss rises before food anaphylaxis and predicts food challenge outcomes. J Clin Investig 133(16). 10.1172/jci168965 [DOI] [PMC free article] [PubMed]
- Shamir EZ, Cassan SM, Levy A et al (2013) Biometric parameters of the hand as an index of schizophrenia–a preliminary study. Psychiatry Res 210(3):716–720. 10.1016/j.psychres.2013.08.026 [DOI] [PubMed] [Google Scholar]
- Shao H, Zhong D (2021) Towards Cross-Dataset Palmprint Recognition Via Joint Pixel and Feature Alignment. IEEE Transact Image Process Publication IEEE Signal Proces Soc 30:3764–3777. 10.1109/tip.2021.3065220 [DOI] [PubMed] [Google Scholar]
- Shi VY, Leo M, Hassoun L et al (2015) Role of sebaceous glands in inflammatory dermatoses. J Am Acad Dermatol 73(5):856–863. 10.1016/j.jaad.2015.08.015 [DOI] [PubMed] [Google Scholar]
- Silpa-Archa N, Kohli I, Chaowattanapanit S et al (2017) Postinflammatory hyperpigmentation: A comprehensive overview: Epidemiology, pathogenesis, clinical presentation, and noninvasive assessment technique. J Am Acad Dermatol 77(4):591–605. 10.1016/j.jaad.2017.01.035 [DOI] [PubMed] [Google Scholar]
- Simpson CL, Patel DM, Green KJ (2011) Deconstructing the skin: cytoarchitectural determinants of epidermal morphogenesis. Nat Rev Mol Cell Biol 12(9):565–580. 10.1038/nrm3175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smile TD, Ruiz ES, Kus KJB et al (2022) Implications of Satellitosis or In-transit Metastasis in Cutaneous Squamous Cell Carcinoma: A Prognostic Omission in Cancer Staging Systems. JAMA Dermatol 158(4):390–394. 10.1001/jamadermatol.2022.0001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stamatas GN, Zmudzka BZ, Kollias N et al (2008) In vivo measurement of skin erythema and pigmentation: new means of implementation of diffuse reflectance spectroscopy with a commercial instrument. Br J Dermatol 159(3):683–690. 10.1111/j.1365-2133.2008.08642.x [DOI] [PubMed] [Google Scholar]
- Superficial Organs and Vasculology Group USotCMA, Chinese Society of Integrative Medicine SaVDSC, Centre SUDEaTR, Remote and Mobile Ultrasound Professional Committee UETB et al (2022) Guidelines on ultrasound examination of skin diseases (2022 Edition). Chinese Journal of Ultrasonography 31(7):553–578. 10.3760/cma.j.cn131148-20220408-00245
- Tschandl P, Codella N, Akay BN et al (2019) Comparison of the accuracy of human readers versus machine-learning algorithms for pigmented skin lesion classification: an open, web-based, international, diagnostic study. Lancet Oncol 20(7):938–947. 10.1016/s1470-2045(19)30333-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uitto J, Christiano AM (1992) Molecular genetics of the cutaneous basement membrane zone. Perspectives on epidermolysis bullosa and other blistering skin diseases. J Clin Investig 90(3):687–692. 10.1172/jci115938 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valdebran M, Mo J, Elston DM et al (2020) Pattern hair loss: Assessment of inflammation and fibrosis on histologic sections. J Am Acad Dermatol 82(3):757–758. 10.1016/j.jaad.2019.09.013 [DOI] [PubMed] [Google Scholar]
- Valenza A, Rykaczewski K, Martinez DM et al (2023) Thermal modulation of skin friction at the finger pad. J Mech Behav Biomed Mater 146:106072. 10.1016/j.jmbbm.2023.106072 [DOI] [PubMed] [Google Scholar]
- Van der Mei IA, Ponsonby AL, Dwyer T et al (2003) Past exposure to sun, skin phenotype, and risk of multiple sclerosis: case-control study. BMJ (clin Res Ed) 327(7410):316. 10.1136/bmj.327.7410.316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Zuuren EJ (2017) Rosacea. N Engl J Med 377(18):1754–1764. 10.1056/NEJMcp1506630 [DOI] [PubMed] [Google Scholar]
- Veijgen NK, Masen MA, van der Heide E (2013) Variables influencing the frictional behaviour of in vivo human skin. J Mech Behav Biomed Mater 28:448–461. 10.1016/j.jmbbm.2013.02.009 [DOI] [PubMed] [Google Scholar]
- Vilhena L, Afonso L, Ramalho A (2023) Skin Friction: Mechanical and Tribological Characterization of Different Papers Used in Everyday Life. Materials (basel, Switz). 10.3390/ma16165724 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wa CV, Maibach HI (2010) Mapping the human face: biophysical properties. Skin research and technology : official journal of International Society for Bioengineering and the Skin (ISBS) [and] International Society for Digital Imaging of Skin (ISDIS) [and] International Society for Skin Imaging (ISSI) 16(1):38–54. 10.1111/j.1600-0846.2009.00400.x [DOI] [PubMed]
- Wahlsten A, Stracuzzi A, Lüchtefeld I et al (2023) Multiscale mechanical analysis of the elastic modulus of skin. Acta Biomater. 10.1016/j.actbio.2023.08.030 [DOI] [PubMed] [Google Scholar]
- Wallace MP, de Berker DA (2010) Hair diagnoses and signs: the use of dermatoscopy. Clin Exp Dermatol 35(1):41–46. 10.1111/j.1365-2230.2009.03383.x [DOI] [PubMed] [Google Scholar]
- Walsh S, Pośpiech E, Branicki W (2016) Hot on the Trail of Genes that Shape Our Fingerprints. J Invest Dermatol 136(4):740–742. 10.1016/j.jid.2015.12.044 [DOI] [PubMed] [Google Scholar]
- Walter K (2022) Psoriasis. JAMA 327(19):1936. 10.1001/jama.2022.5270 [DOI] [PubMed] [Google Scholar]
- Wang X (2013) The Fundamentals of Medical Genetics. Beijing, China
- Wat M, Olicker A, Meyerson H et al (2018) Topical Hypochlorite and Skin Acidification Improves Erythroderma of Omenn Syndrome. Pediatrics 141(5):S408-s411. 10.1542/peds.2017-1249 [DOI] [PubMed] [Google Scholar]
- Wei J, Zhang B, Jingting L et al (2017) Palmprint Recognition Based on Complete Direction Representation. IEEE Transact Image Proces Publication IEEE Signal Proces Soc 26(9):4483–4498. 10.1109/tip.2017.2705424 [DOI] [PubMed] [Google Scholar]
- White JD, Indencleef K, Naqvi S et al (2021) Insights into the genetic architecture of the human face. Nat Genet 53(1):45–53. 10.1038/s41588-020-00741-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wojtowicz H, Wajs W (2012) Medical Decision Support System for Assessment of Dermatoglyphic Indices and Diagnosis of Down’s Syndrome. In. Springer Berlin Heidelberg, Berlin, Heidelberg, pp 69–78
- Workman K, Piliang M (2023) Approach to the patient with hair loss. J Am Acad Dermatol 89(2s):S3-s8. 10.1016/j.jaad.2023.05.040 [DOI] [PubMed] [Google Scholar]
- Wu H, Yin H, Chen H et al (2021) A deep learning-based smartphone platform for cutaneous lupus erythematosus classification assistance: Simplifying the diagnosis of complicated diseases. J Am Acad Dermatol 85(3):792–793. 10.1016/j.jaad.2021.02.043 [DOI] [PubMed] [Google Scholar]
- Xu H, Fonseca M, Wolner Z et al (2017) Reference values for skin microanatomy: A systematic review and meta-analysis of ex vivo studies. J Am Acad Dermatol 77(6):1133-1144.e1134. 10.1016/j.jaad.2017.06.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue B, Leibler S (2018) Benefits of phenotypic plasticity for population growth in varying environments. Proc Natl Acad Sci USA 115(50):12745–12750. 10.1073/pnas.1813447115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang DS, Ghaffari R, Rogers JA (2023) Sweat as a diagnostic biofluid. Science (new York, NY) 379(6634):760–761. 10.1126/science.abq5916 [DOI] [PubMed] [Google Scholar]
- Yazdanparast T, Yazdani K, Humbert P et al (2018) Comparison of biophysical, biomechanical and ultrasonographic properties of skin in chronic dermatitis, psoriasis and lichen planus. Med J Islamic Republic of Iran 32:108. 10.1419/mjiri.32.108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon S, Jain AK (2015) Longitudinal study of fingerprint recognition. Proc Natl Acad Sci USA 112(28):8555–8560. 10.1073/pnas.1410272112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yousef H, Alhajj M, Sharma S (2023) Anatomy, skin (integument), epidermis. In: StatPearls. StatPearls Publishing, Treasure Island (FL). https://www.ncbi.nlm.nih.gov/books/NBK470464/ [PubMed]
- Zhang Y, Liu X, Wang J et al (2022) Analysis of Multi-Part Phenotypic Changes in Skin to Characterize the Trajectory of Skin Aging in Chinese Women. Clin Cosmet Investig Dermatol 15:631–642. 10.2147/ccid.S349401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou J, Wu Z, Jiang Z et al (2022) Background selection schema on deep learning-based classification of dermatological disease. Comput Biol Med 149:105966. 10.1016/j.compbiomed.2022.105966 [DOI] [PubMed] [Google Scholar]
- Zouboulis CC (2004) Acne and sebaceous gland function. Clin Dermatol 22(5):360–366. 10.1016/j.clindermatol.2004.03.004 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.