Abstract
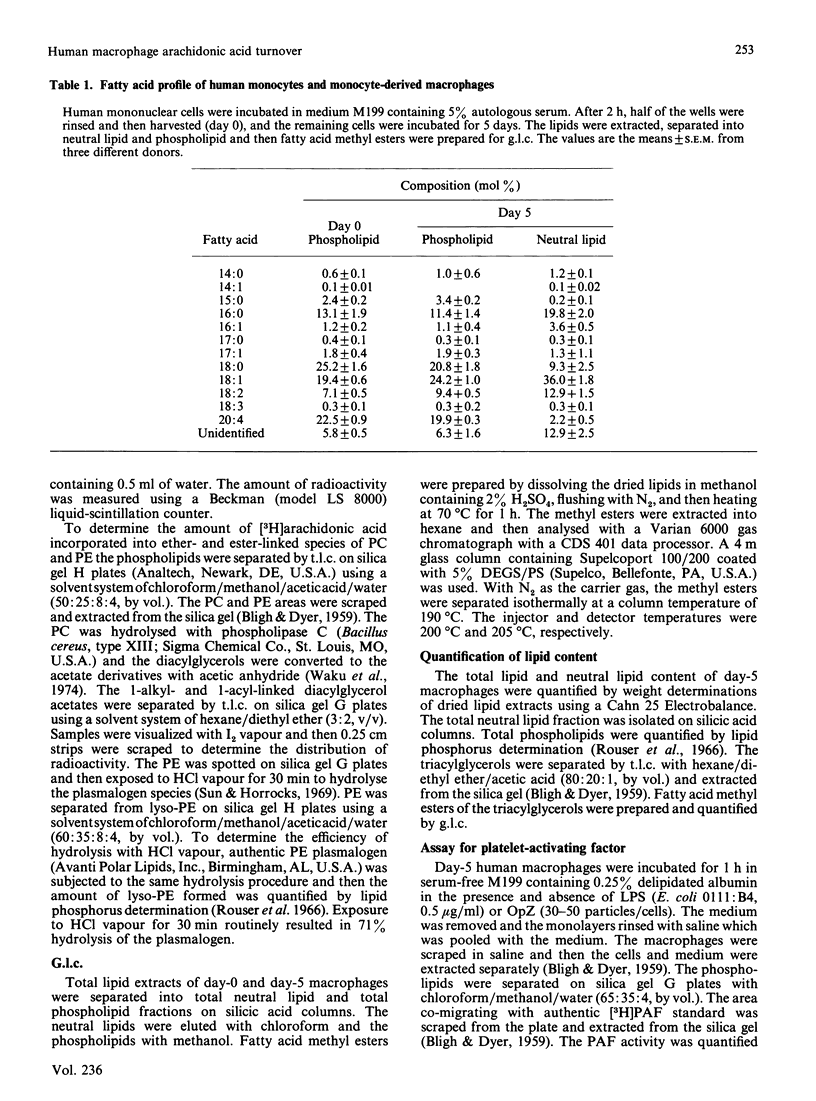
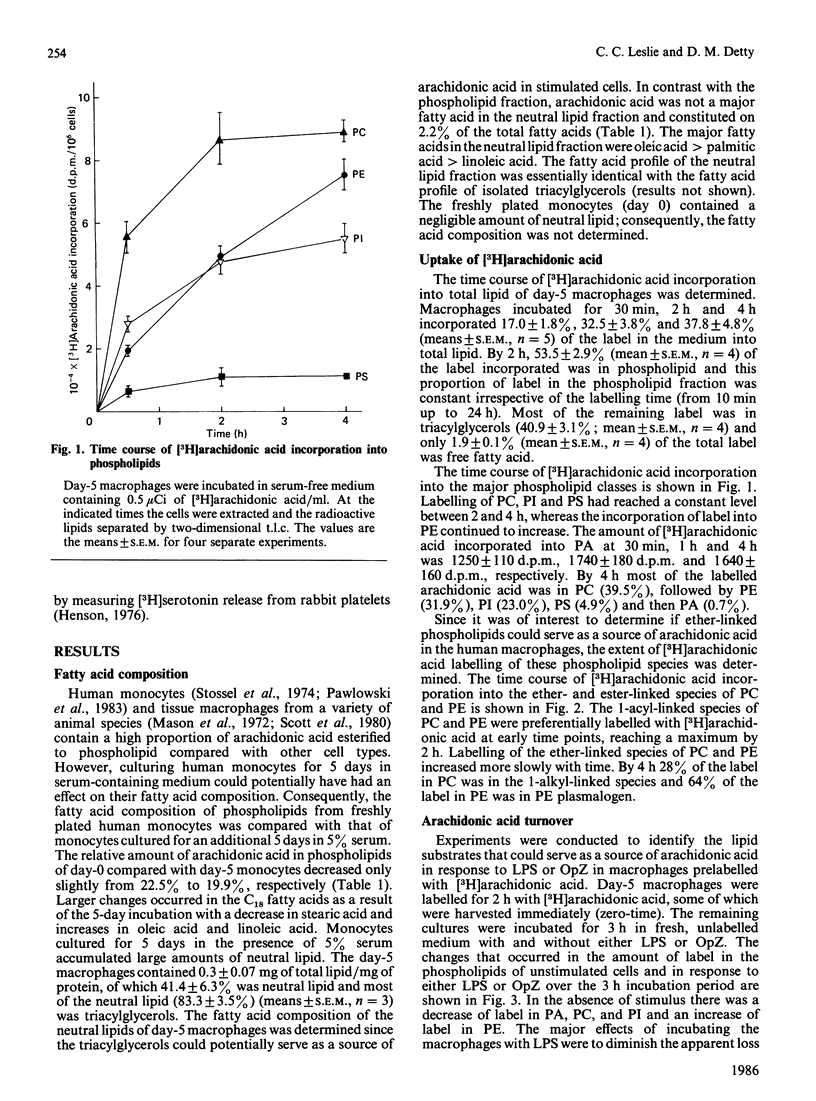
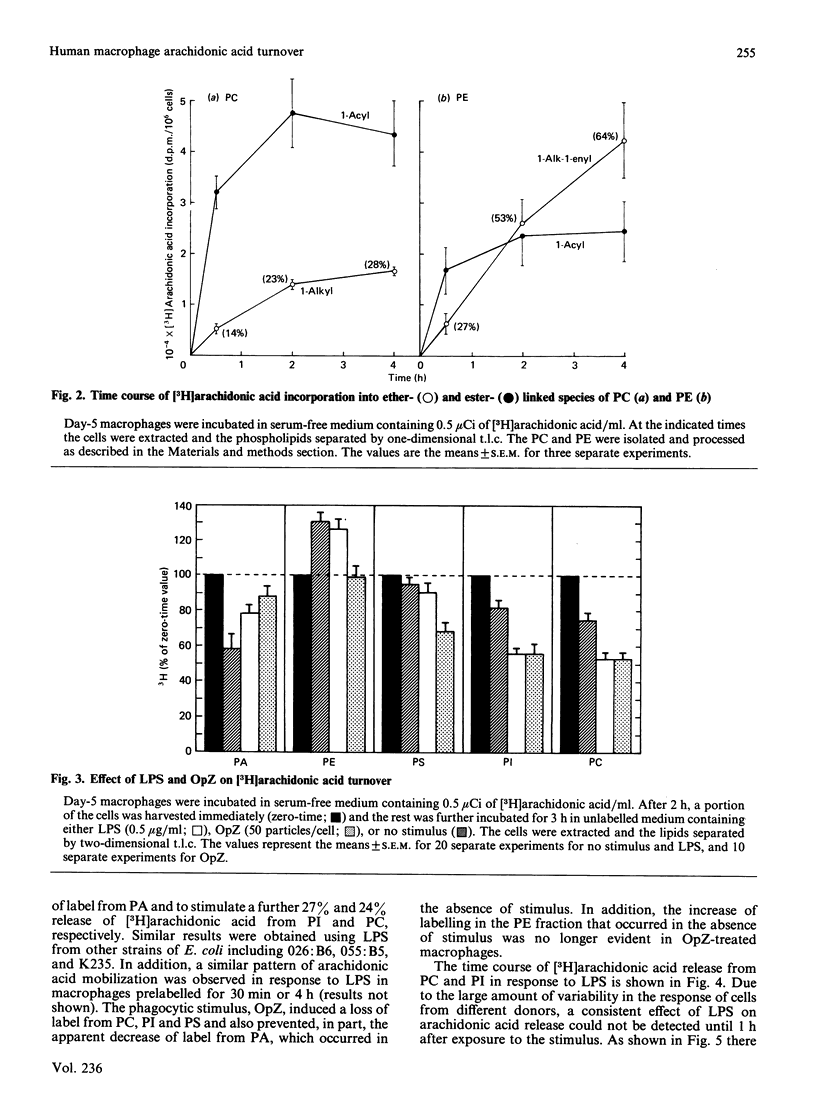
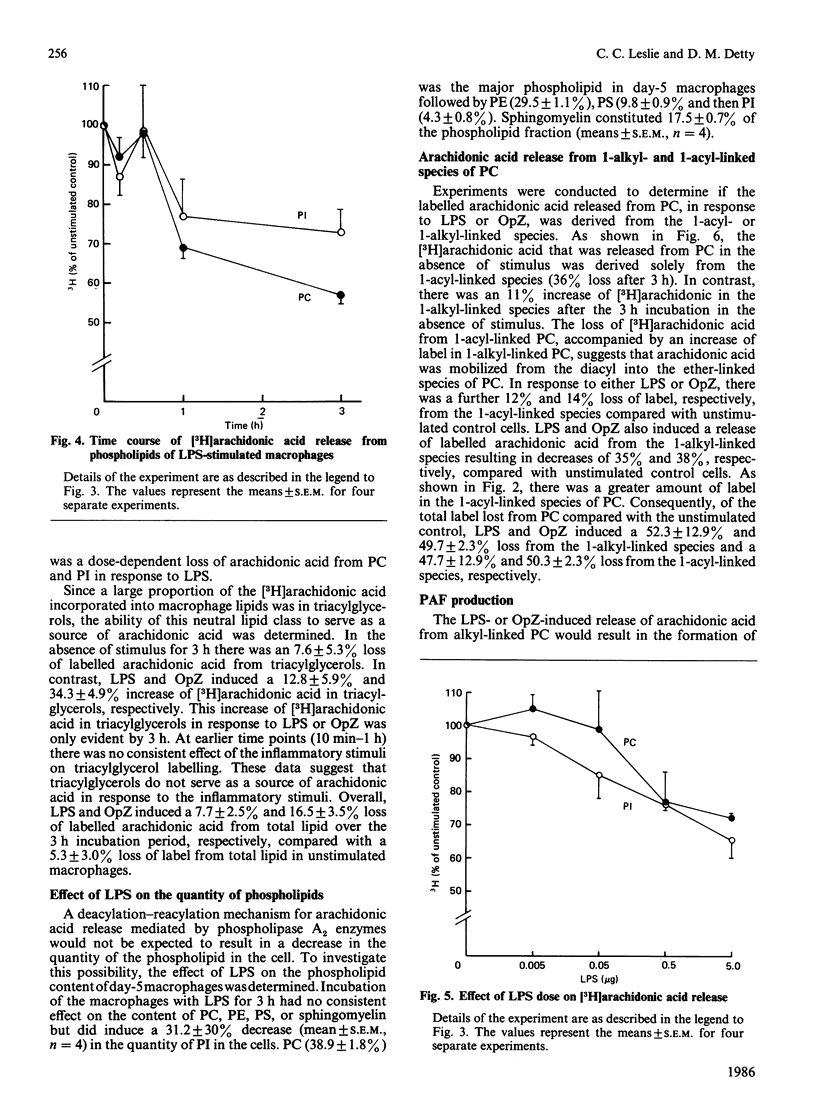
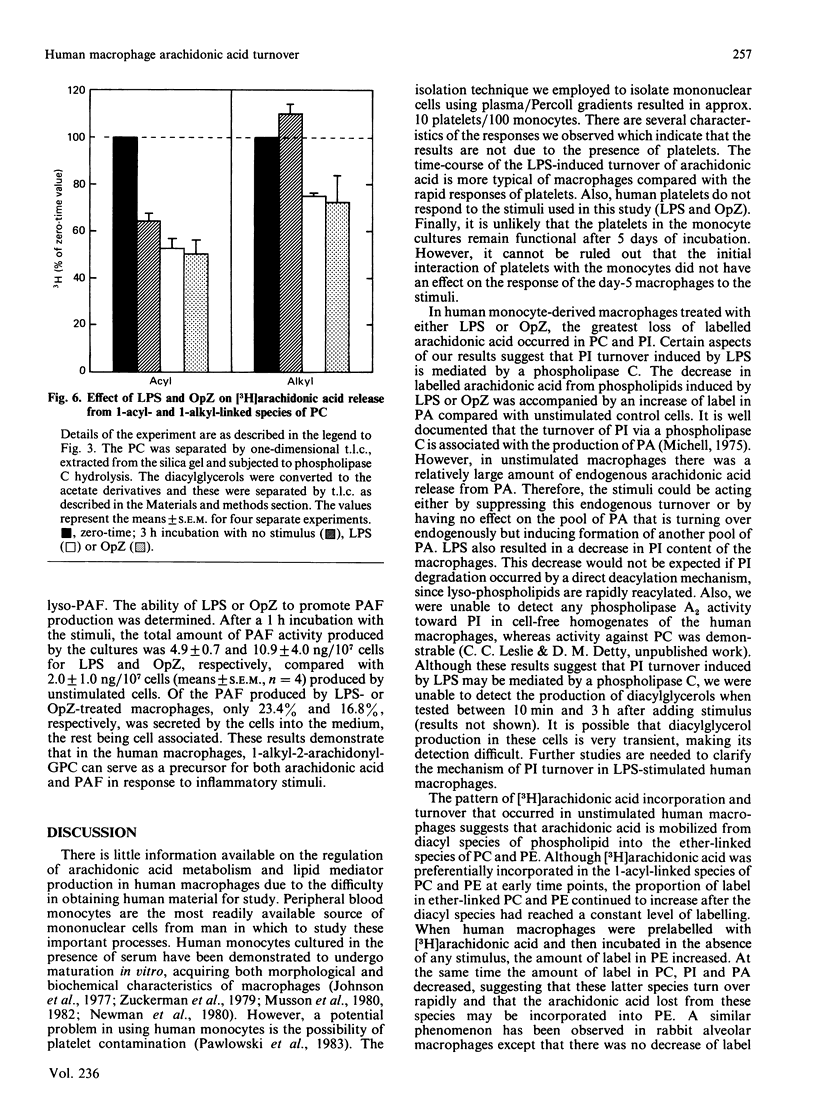
Macrophages are an important source of the lipid mediators, arachidonic acid metabolites and platelet-activating factor (PAF), produced during inflammation. Studies were undertaken to identify the phospholipid substrates that can serve as a source of arachidonic acid in human monocyte-derived macrophages exposed to the inflammatory stimuli bacterial lipopolysaccharide (LPS) and opsonized zymosan (OpZ). Since PAF is derived from 1-alkyl-2-acyl-glycerophosphocholine, it was of interest to determine if this phospholipid precursor could also serve as a source of arachidonic acid. The day-5 macrophages incorporated 38% of the available [3H]arachidonic acid into lipid by 4 h, 54% of which was in phospholipid [phosphatidylcholine (PC) greater than phosphatidylethanolamine (PE) greater than phosphatidylinositol (PI)]. The proportion of label incorporated into ether-linked PC and PE increased with time. After prelabelling with [3H]arachidonic acid, the effect of stimuli on the redistribution of label within phospholipids was followed. Without stimulus there was a loss of label from PC, PI and phosphatidic acid by 3 h, but an increase of label in PE. The [3H]arachidonic acid that was lost from PC in the absence of stimulus was derived solely from the 1-acyl-linked species of PC, whereas an increase in label occurred in the 1-alkyl-linked species of PC. By contrast, LPS stimulation resulted in a preferential, dose-dependent loss of label from PC and PI, which was maximal between 1 and 3 h after adding the LPS. In addition, LPS induced a 35% decrease in the molar quantity of PI in the macrophages but had no effect on the quantity of PC, PE or phosphatidylserine. Stimulation with OpZ also resulted in a loss of label, mainly from PC and PI. Of the total label lost from PC in response to LPS or OpZ, approx. 50% was derived from the 1-alkyl-linked species. The results suggest that phospholipase C- and phospholipase A2-mediated mechanisms for arachidonic acid release are activated in human macrophages exposed to the inflammatory stimuli LPS and OpZ. In addition, 1-alkyl-linked PC can serve as a source of arachidonic acid and as a precursor for PAF production in the stimulated macrophages.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Albert D. H., Snyder F. Biosynthesis of 1-alkyl-2-acetyl-sn-glycero-3-phosphocholine (platelet-activating factor) from 1-alkyl-2-acyl-sn-glycero-3-phosphocholine by rat alveolar macrophages. Phospholipase A2 and acetyltransferase activities during phagocytosis and ionophore stimulation. J Biol Chem. 1983 Jan 10;258(1):97–102. [PubMed] [Google Scholar]
- Albert D. H., Snyder F. Release of arachidonic acid from 1-alkyl-2-acyl-sn-glycero-3-phosphocholine, a precursor of platelet-activating factor, in rat alveolar macrophages. Biochim Biophys Acta. 1984 Oct 24;796(1):92–101. doi: 10.1016/0005-2760(84)90242-x. [DOI] [PubMed] [Google Scholar]
- BLIGH E. G., DYER W. J. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959 Aug;37(8):911–917. doi: 10.1139/o59-099. [DOI] [PubMed] [Google Scholar]
- Berridge M. J. Inositol trisphosphate and diacylglycerol as second messengers. Biochem J. 1984 Jun 1;220(2):345–360. doi: 10.1042/bj2200345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonney R. J., Wightman P. D., Davies P., Sadowski S. J., Kuehl F. A., Jr, Humes J. L. Regulation of prostaglandin synthesis and of the selective release of lysosomal hydrolases by mouse peritoneal macrophages. Biochem J. 1978 Nov 15;176(2):433–442. doi: 10.1042/bj1760433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cham B. E., Knowles B. R. A solvent system for delipidation of plasma or serum without protein precipitation. J Lipid Res. 1976 Mar;17(2):176–181. [PubMed] [Google Scholar]
- Chensue S. W., Kunkel S. L. Arachidonic acid metabolism and macrophage activation. Clin Lab Med. 1983 Dec;3(4):677–694. [PubMed] [Google Scholar]
- Chilton F. H., Ellis J. M., Olson S. C., Wykle R. L. 1-O-alkyl-2-arachidonoyl-sn-glycero-3-phosphocholine. A common source of platelet-activating factor and arachidonate in human polymorphonuclear leukocytes. J Biol Chem. 1984 Oct 10;259(19):12014–12019. [PubMed] [Google Scholar]
- Chilton F. H., O'Flaherty J. T., Ellis J. M., Swendsen C. L., Wykle R. L. Selective acylation of lyso platelet activating factor by arachidonate in human neutrophils. J Biol Chem. 1983 Jun 25;258(12):7268–7271. [PubMed] [Google Scholar]
- Demopoulos C. A., Pinckard R. N., Hanahan D. J. Platelet-activating factor. Evidence for 1-O-alkyl-2-acetyl-sn-glyceryl-3-phosphorylcholine as the active component (a new class of lipid chemical mediators). J Biol Chem. 1979 Oct 10;254(19):9355–9358. [PubMed] [Google Scholar]
- Emilsson A., Sundler R. Differential activation of phosphatidylinositol deacylation and a pathway via diphosphoinositide in macrophages responding to zymosan and ionophore A23187. J Biol Chem. 1984 Mar 10;259(5):3111–3116. [PubMed] [Google Scholar]
- Gemsa D., Leser H. G., Seitz M., Deimann W., Bärlin E. Membrane perturbation and stimulation of arachidonic acid metabolism. Mol Immunol. 1982 Oct;19(10):1287–1296. doi: 10.1016/0161-5890(82)90295-4. [DOI] [PubMed] [Google Scholar]
- Haslett C., Guthrie L. A., Kopaniak M. M., Johnston R. B., Jr, Henson P. M. Modulation of multiple neutrophil functions by preparative methods or trace concentrations of bacterial lipopolysaccharide. Am J Pathol. 1985 Apr;119(1):101–110. [PMC free article] [PubMed] [Google Scholar]
- Henson P. M. Activation and desensitization of platelets by platelet-activating factor (PAF) derived from IgE-sensitized basophils. I. Characteristics of the secretory response. J Exp Med. 1976 Apr 1;143(4):937–952. doi: 10.1084/jem.143.4.937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Homma Y., Onozaki K., Hashimoto T., Nagai Y., Takenawa T. Differential activation of phospholipids metabolism by formylated peptide and ionophore A23187 in guinea pig peritoneal macrophages. J Immunol. 1982 Oct;129(4):1619–1626. [PubMed] [Google Scholar]
- Hsueh W. Prostaglandin biosynthesis in pulmonary macrophages. Am J Pathol. 1979 Oct;97(1):137–148. [PMC free article] [PubMed] [Google Scholar]
- Hsueh W., Sun F. F. Leukotriene B4 biosynthesis by alveolar macrophages. Biochem Biophys Res Commun. 1982 Jun 30;106(4):1085–1091. doi: 10.1016/0006-291x(82)91223-2. [DOI] [PubMed] [Google Scholar]
- Johnson W. D., Jr, Mei B., Cohn Z. A. The separation, long-term cultivation, and maturation of the human monocyte. J Exp Med. 1977 Dec 1;146(6):1613–1626. doi: 10.1084/jem.146.6.1613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurland J. I., Bockman R. Prostaglandin E production by human blood monocytes and mouse peritoneal macrophages. J Exp Med. 1978 Mar 1;147(3):952–957. doi: 10.1084/jem.147.3.952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason R. J., Stossel T. P., Vaughan M. Lipids of alveolar macrophages, polymorphonuclear leukocytes, and their phagocytic vesicles. J Clin Invest. 1972 Sep;51(9):2399–2407. doi: 10.1172/JCI107052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michell R. H. Inositol phospholipids and cell surface receptor function. Biochim Biophys Acta. 1975 Mar 25;415(1):81–47. doi: 10.1016/0304-4157(75)90017-9. [DOI] [PubMed] [Google Scholar]
- Mueller H. W., O'Flaherty J. T., Wykle R. L. Biosynthesis of platelet activating factor in rabbit polymorphonuclear neutrophils. J Biol Chem. 1983 May 25;258(10):6213–6218. [PubMed] [Google Scholar]
- Musson R. A., McPhail L. C., Shafran H., Johnston R. B., Jr Differences in the ability of human peripheral blood monocytes and in vitro monocyte-derived macrophages to produce superoxide anion: studies with cells from normals and patients with chronic granulomatous disease. J Reticuloendothel Soc. 1982 Mar;31(3):261–266. [PubMed] [Google Scholar]
- Musson R. A., Shafran H., Henson P. M. Intracellular levels and stimulated release of lysosomal enzymes from human peripheral blood monocytes and monocyte-derived macrophages. J Reticuloendothel Soc. 1980 Sep;28(3):249–264. [PubMed] [Google Scholar]
- Newman S. L., Musson R. A., Henson P. M. Development of functional complement receptors during in vitro maturation of human monocytes into macrophages. J Immunol. 1980 Nov;125(5):2236–2244. [PubMed] [Google Scholar]
- Ogmundsdóttir H. M., Weir D. M. Stimulation of phosphatidylinositol turnover in the macrophage plasma membrane: a possible mechanism for signal transmission. Immunology. 1979 Jul;37(3):689–696. [PMC free article] [PubMed] [Google Scholar]
- Pawlowski N. A., Kaplan G., Hamill A. L., Cohn Z. A., Scott W. A. Arachidonic acid metabolism by human monocytes. Studies with platelet-depleted cultures. J Exp Med. 1983 Aug 1;158(2):393–412. doi: 10.1084/jem.158.2.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rittenhouse-Simmons S., Russell F. A., Deykin D. Mobilization of arachidonic acid in human platelets. Kinetics and Ca2+ dependency. Biochim Biophys Acta. 1977 Sep 28;488(3):370–380. doi: 10.1016/0005-2760(77)90196-5. [DOI] [PubMed] [Google Scholar]
- Rouser G., Siakotos A. N., Fleischer S. Quantitative analysis of phospholipids by thin-layer chromatography and phosphorus analysis of spots. Lipids. 1966 Jan;1(1):85–86. doi: 10.1007/BF02668129. [DOI] [PubMed] [Google Scholar]
- Rouzer C. A., Scott W. A., Cohn Z. A., Blackburn P., Manning J. M. Mouse peritoneal macrophages release leukotriene C in response to a phagocytic stimulus. Proc Natl Acad Sci U S A. 1980 Aug;77(8):4928–4932. doi: 10.1073/pnas.77.8.4928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott W. A., Zrike J. M., Hamill A. L., Kempe J., Cohn Z. A. Regulation of arachidonic acid metabolites in macrophages. J Exp Med. 1980 Aug 1;152(2):324–335. doi: 10.1084/jem.152.2.324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stossel T. P., Mason R. J., Smith A. L. Lipid peroxidation by human blood phagocytes. J Clin Invest. 1974 Sep;54(3):638–645. doi: 10.1172/JCI107801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugiura T., Katayama O., Fukui J., Nakagawa Y., Waku K. Mobilization of arachidonic acid between diacyl and ether phospholipids in rabbit alveolar macrophages. FEBS Lett. 1984 Jan 9;165(2):273–276. doi: 10.1016/0014-5793(84)80184-2. [DOI] [PubMed] [Google Scholar]
- Sugiura T., Waku K. CoA-independent transfer of arachidonic acid from 1,2-diacyl-sn-glycero-3-phosphocholine to 1-O-alkyl-sn-glycero-3-phosphocholine (lyso platelet-activating factor) by macrophage microsomes. Biochem Biophys Res Commun. 1985 Feb 28;127(1):384–390. doi: 10.1016/s0006-291x(85)80171-6. [DOI] [PubMed] [Google Scholar]
- Sun G. Y., Horrocks L. A. Acyl and alk-1-enyl group compositions of the alk-1'-enyl acyl and the diacyl glycerophosphoryl ethanolamines of mouse and ox brain. J Lipid Res. 1969 Mar;10(2):153–157. [PubMed] [Google Scholar]
- Swendsen C. L., Ellis J. M., Chilton F. H., 3rd, O'Flaherty J. T., Wykle R. L. 1-O-alkyl-2-acyl-sn-glycero-3-phosphocholine: a novel source of arachidonic acid in neutrophils stimulated by the calcium ionophore A23187. Biochem Biophys Res Commun. 1983 May 31;113(1):72–79. doi: 10.1016/0006-291x(83)90433-3. [DOI] [PubMed] [Google Scholar]
- Waku K., Ito H., Bito T., Nakazawa Y. Fatty chains of acyl, alkenyl, and alkyl phosphoglycerides of rabbit sarcoplasmic reticulum. The metabolic relationship considered on the basis of structural analyses. J Biochem. 1974 Jun;75(6):1307–1312. doi: 10.1093/oxfordjournals.jbchem.a130515. [DOI] [PubMed] [Google Scholar]
- Zuckerman S. H., Ackerman S. K., Douglas S. D. Long-term human peripheral blood monocyte cultures: establishment, metabolism and morphology of primary human monocyte-macrophage cell cultures. Immunology. 1979 Oct;38(2):401–411. [PMC free article] [PubMed] [Google Scholar]



