Abstract
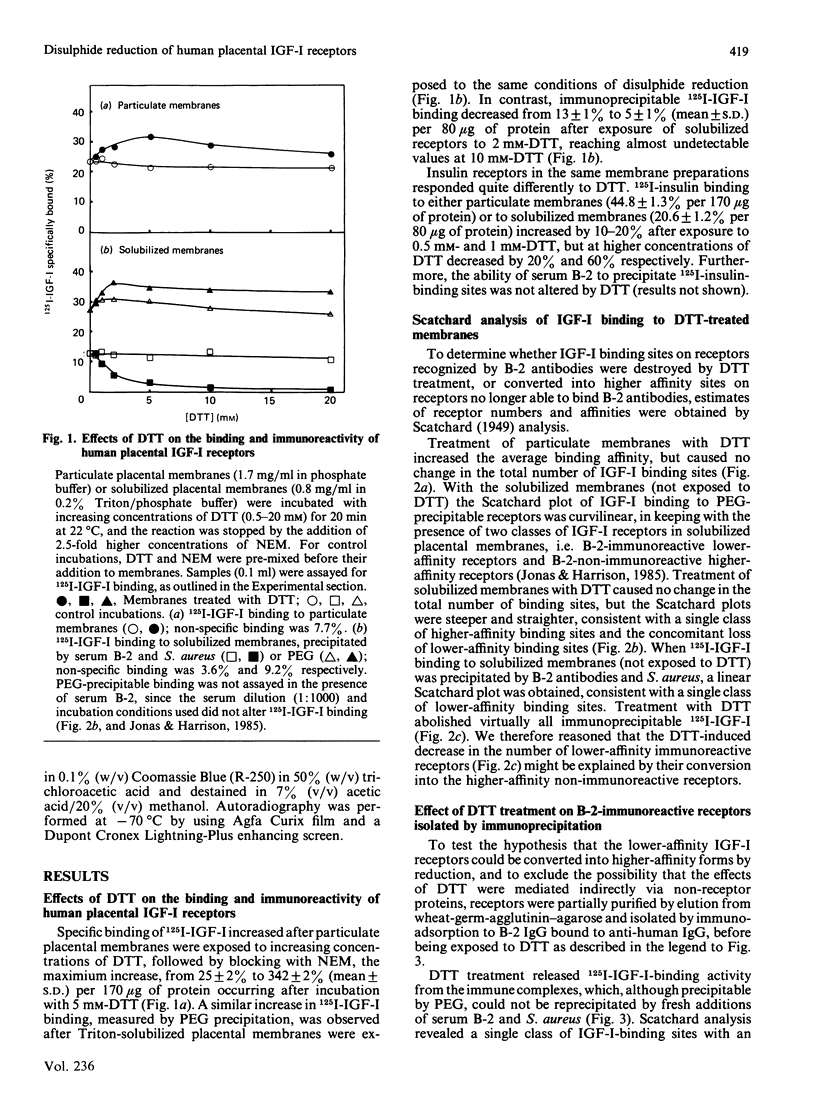
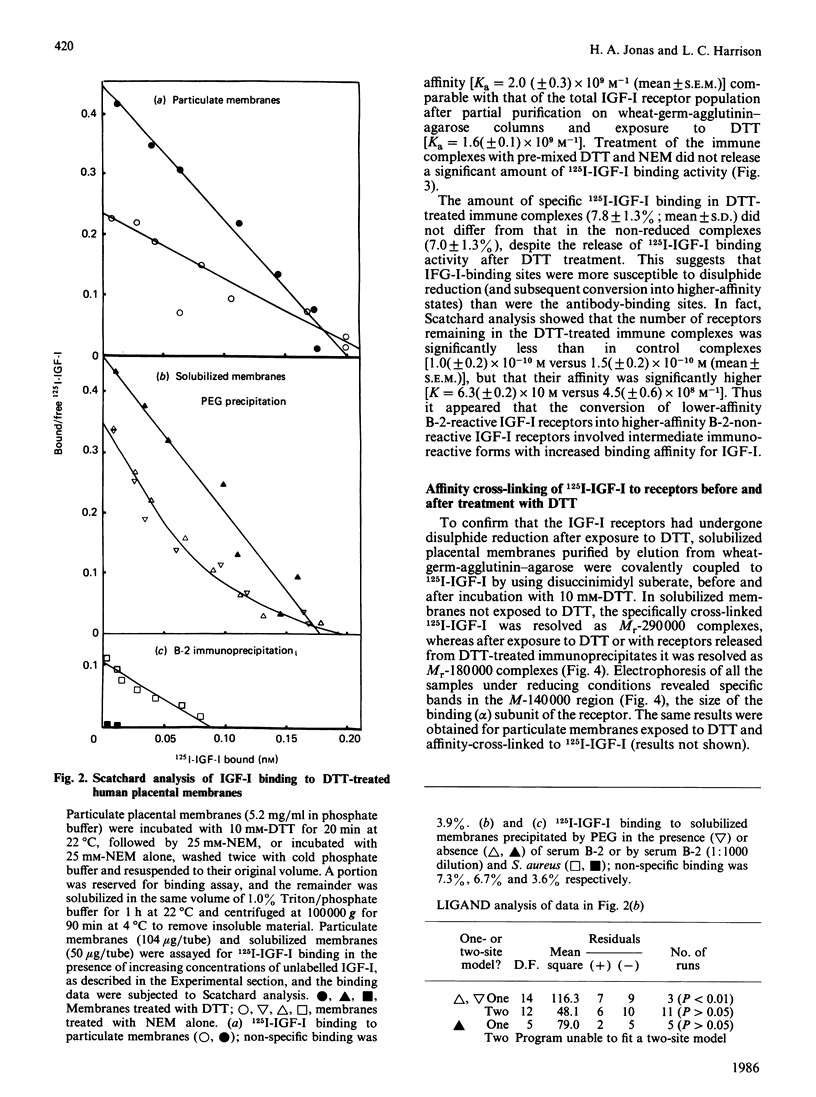
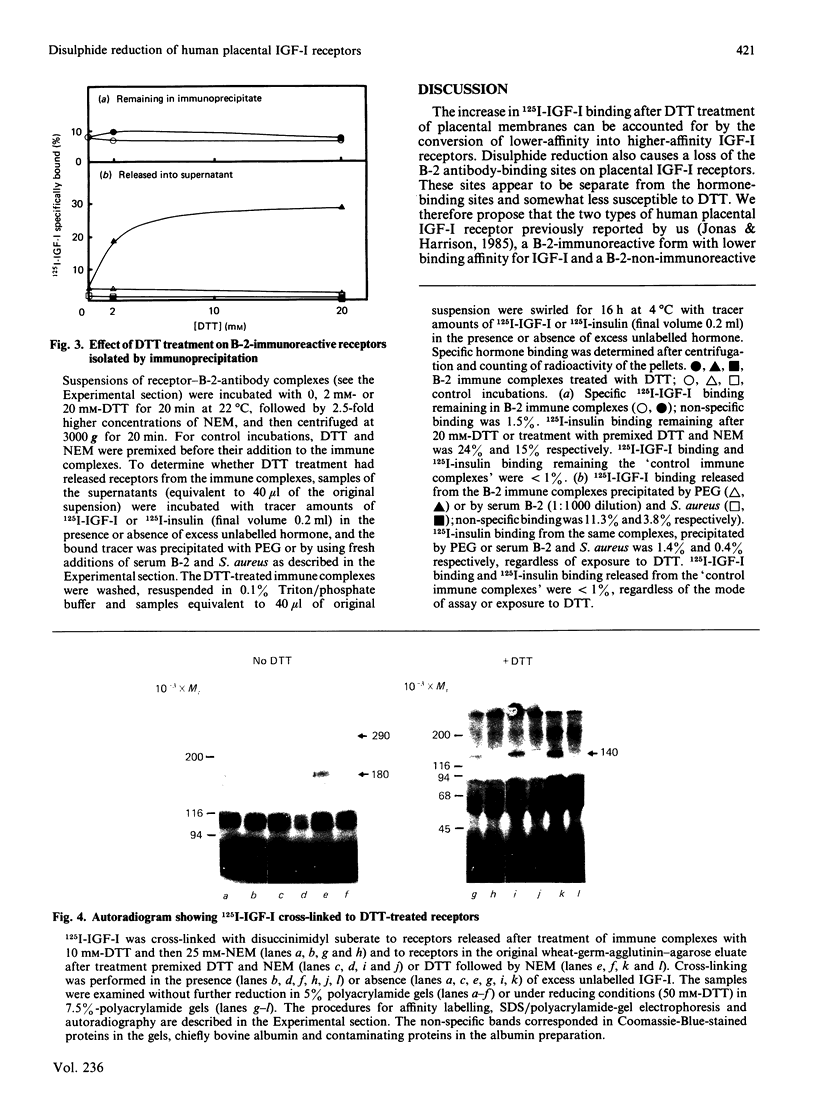
We previously identified two forms of the insulin-like growth-factor-I (IGF-I) receptor in human placenta: a lower-affinity form reactive with an autoantiserum (B-2) to the insulin receptor and a higher-affinity non-immunoreactive form [Jonas & Harrison (1985) J. Biol. Chem. 260, 2288-2294]. Evidence is now presented that the lower-affinity immunoreactive forms are convertible into higher-affinity non-immunoreactive forms via reduction of receptor disulphide bonds. Treatment of placental membranes with increasing concentrations of dithiothreitol (DTT): (1) converted native Mr-290 000 heterotetrameric IGF-I receptors into Mr-180 000 dimers (determined by chemical cross-linking of 125I-IGF-I with disuccinimidyl suberate); (2) increased 125I-IGF-I binding, owing to an increase in receptor affinity; and (3) abolished the reactivity of Triton-solubilized IGF-I receptors with antiserum B-2 and transformed the curvilinear plot of IGF-I binding to a linear form. In isolated complexes between receptor and B-2 antibody, DTT increased 125I-IGF-I binding and released a single class of higher affinity IGF-I receptors of Mr 180,000. Thus DTT-treated IGF-I receptors have similar properties to the higher-affinity non-immunoreactive forms of the native receptor, except that reduced dimeric forms are not detected by cross-linking of 125I-IGF-I to native membranes. Cleavage of the inter-dimeric disulphide bonds is therefore not a prerequisite for higher-affinity binding or loss of immunoreactivity. These observations suggest that the thiol redox state of the IGF-I receptor in vivo is an important determinant of receptor conformation and therefore of the biological responses to IGF-I.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aiyer R. A. Structural characterization of insulin receptors. I. Hydrodynamic properties of receptors from turkey erythrocytes. J Biol Chem. 1983 Dec 25;258(24):14992–14999. [PubMed] [Google Scholar]
- Avruch J., Nemenoff R. A., Blackshear P. J., Pierce M. W., Osathanondh R. Insulin-stimulated tyrosine phosphorylation of the insulin receptor in detergent extracts of human placental membranes. Comparison to epidermal growth factor-stimulated phosphorylation. J Biol Chem. 1982 Dec 25;257(24):15162–15166. [PubMed] [Google Scholar]
- Baxter R. C., Brown A. S. Purification of tracer for somatomedin C radioimmunoassay by hydrophobic interaction chromatography. Clin Chem. 1982 Mar;28(3):485–487. [PubMed] [Google Scholar]
- Bhaumick B., Bala R. M., Hollenberg M. D. Somatomedin receptor of human placenta: solubilization, photolabeling, partial purification, and comparison with insulin receptor. Proc Natl Acad Sci U S A. 1981 Jul;78(7):4279–4283. doi: 10.1073/pnas.78.7.4279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blackshear P. J., Nemenoff R. A., Avruch J. Characteristics of insulin and epidermal growth factor stimulation of receptor autophosphorylation in detergent extracts of rat liver and transplantable rat hepatomas. Endocrinology. 1984 Jan;114(1):141–152. doi: 10.1210/endo-114-1-141. [DOI] [PubMed] [Google Scholar]
- Chernausek S. D., Jacobs S., Van Wyk J. J. Structural similarities between human receptors for somatomedin C and insulin: analysis by affinity labeling. Biochemistry. 1981 Dec 22;20(26):7345–7350. doi: 10.1021/bi00529a004. [DOI] [PubMed] [Google Scholar]
- Clark S., Harrison L. C. Disulfide exchange between insulin and its receptor. A possible post-binding step in insulin action. J Biol Chem. 1983 Oct 10;258(19):11434–11437. [PubMed] [Google Scholar]
- Clark S., Harrison L. C. Insulin binding leads to the formation of covalent (-S-S-) hormone receptor complexes. J Biol Chem. 1982 Oct 25;257(20):12239–12244. [PubMed] [Google Scholar]
- Fujita-Yamaguchi Y., Kathuria S. The monomeric alpha beta form of the insulin receptor exhibits much higher insulin-dependent tyrosine-specific protein kinase activity than the intact alpha 2 beta 2 form of the receptor. Proc Natl Acad Sci U S A. 1985 Sep;82(18):6095–6099. doi: 10.1073/pnas.82.18.6095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harmon J. T., Hedo J. A., Kahn C. R. Characterization of a membrane regulator of insulin receptor affinity. J Biol Chem. 1983 Jun 10;258(11):6875–6881. [PubMed] [Google Scholar]
- Harrison L. C., Billington T., East I. J., Nichols R. J., Clark S. The effect of solubilization on the properties of the insulin receptor of human placental membranes. Endocrinology. 1978 May;102(5):1485–1495. doi: 10.1210/endo-102-5-1485. [DOI] [PubMed] [Google Scholar]
- Jacobs S., Cuatrecasas P. Disulfide reduction converts the insulin receptor of human placenta to a low affinity form. J Clin Invest. 1980 Dec;66(6):1424–1427. doi: 10.1172/JCI109996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobs S., Kull F. C., Jr, Earp H. S., Svoboda M. E., Van Wyk J. J., Cuatrecasas P. Somatomedin-C stimulates the phosphorylation of the beta-subunit of its own receptor. J Biol Chem. 1983 Aug 25;258(16):9581–9584. [PubMed] [Google Scholar]
- Jonas H. A., Baxter R. C., Harrison L. C. Structural differences between insulin and somatomedin-C/insulin-like growth factor-1 receptors revealed by autoantibodies to the insulin receptor. Biochem Biophys Res Commun. 1982 Nov 30;109(2):463–470. doi: 10.1016/0006-291x(82)91744-2. [DOI] [PubMed] [Google Scholar]
- Jonas H. A., Harrison L. C. The human placenta contains two distinct binding and immunoreactive species of insulin-like growth factor-I receptors. J Biol Chem. 1985 Feb 25;260(4):2288–2294. [PubMed] [Google Scholar]
- Kasuga M., Zick Y., Blithe D. L., Crettaz M., Kahn C. R. Insulin stimulates tyrosine phosphorylation of the insulin receptor in a cell-free system. Nature. 1982 Aug 12;298(5875):667–669. doi: 10.1038/298667a0. [DOI] [PubMed] [Google Scholar]
- Kull F. C., Jr, Jacobs S., Su Y. F., Svoboda M. E., Van Wyk J. J., Cuatrecasas P. Monoclonal antibodies to receptors for insulin and somatomedin-C. J Biol Chem. 1983 May 25;258(10):6561–6566. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Massague J., Pilch P. F., Czech M. P. A unique proteolytic cleavage site on the beta subunit of the insulin receptor. J Biol Chem. 1981 Apr 10;256(7):3182–3190. [PubMed] [Google Scholar]
- Massagué J., Czech M. P. Role of disulfides in the subunit structure of the insulin receptor. Reduction of class I disulfides does not impair transmembrane signalling. J Biol Chem. 1982 Jun 25;257(12):6729–6738. [PubMed] [Google Scholar]
- Massagué J., Czech M. P. The subunit structures of two distinct receptors for insulin-like growth factors I and II and their relationship to the insulin receptor. J Biol Chem. 1982 May 10;257(9):5038–5045. [PubMed] [Google Scholar]
- Maturo J. M., 3rd, Hollenberg M. D., Aglio L. S. Insulin receptor: insulin-modulated interconversion between distinct molecular forms involving disulfide-sulfhydryl exchange. Biochemistry. 1983 May 10;22(10):2579–2586. doi: 10.1021/bi00279a040. [DOI] [PubMed] [Google Scholar]
- Munson P. J., Rodbard D. Ligand: a versatile computerized approach for characterization of ligand-binding systems. Anal Biochem. 1980 Sep 1;107(1):220–239. doi: 10.1016/0003-2697(80)90515-1. [DOI] [PubMed] [Google Scholar]
- Petruzzelli L., Herrera R., Rosen O. M. Insulin receptor is an insulin-dependent tyrosine protein kinase: copurification of insulin-binding activity and protein kinase activity to homogeneity from human placenta. Proc Natl Acad Sci U S A. 1984 Jun;81(11):3327–3331. doi: 10.1073/pnas.81.11.3327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosen O. M., Herrera R., Olowe Y., Petruzzelli L. M., Cobb M. H. Phosphorylation activates the insulin receptor tyrosine protein kinase. Proc Natl Acad Sci U S A. 1983 Jun;80(11):3237–3240. doi: 10.1073/pnas.80.11.3237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth J. Methods for assessing immunologic and biologic properties of iodinated peptide hormones. Methods Enzymol. 1975;37:223–233. doi: 10.1016/s0076-6879(75)37018-3. [DOI] [PubMed] [Google Scholar]
- Rubin J. B., Shia M. A., Pilch P. F. Stimulation of tyrosine-specific phosphorylation in vitro by insulin-like growth factor I. 1983 Sep 29-Oct 5Nature. 305(5933):438–440. doi: 10.1038/305438a0. [DOI] [PubMed] [Google Scholar]
- Schweitzer J. B., Smith R. M., Jarett L. Differences in organizational structure of insulin receptor on rat adipocyte and liver plasma membranes: role of disulfide bonds. Proc Natl Acad Sci U S A. 1980 Aug;77(8):4692–4696. doi: 10.1073/pnas.77.8.4692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shia M. A., Rubin J. B., Pilch P. F. The insulin receptor protein kinase. Physicochemical requirements for activity. J Biol Chem. 1983 Dec 10;258(23):14450–14455. [PubMed] [Google Scholar]
- Yip C. C., Moule M. L. Structure of the insulin receptor of rat adipocytes. The three interconvertible redox forms. Diabetes. 1983 Aug;32(8):760–767. doi: 10.2337/diab.32.8.760. [DOI] [PubMed] [Google Scholar]
- Yu K. T., Czech M. P. Tyrosine phosphorylation of the insulin receptor beta subunit activates the receptor-associated tyrosine kinase activity. J Biol Chem. 1984 Apr 25;259(8):5277–5286. [PubMed] [Google Scholar]
- Zapf J., Rinderknecht E., Humbel R. E., Froesch E. R. Nonsuppressible insulin-like activity (NSILA) from human serum: recent accomplishments and their physiologic implications. Metabolism. 1978 Dec;27(12):1803–1828. doi: 10.1016/0026-0495(78)90267-6. [DOI] [PubMed] [Google Scholar]



