Abstract
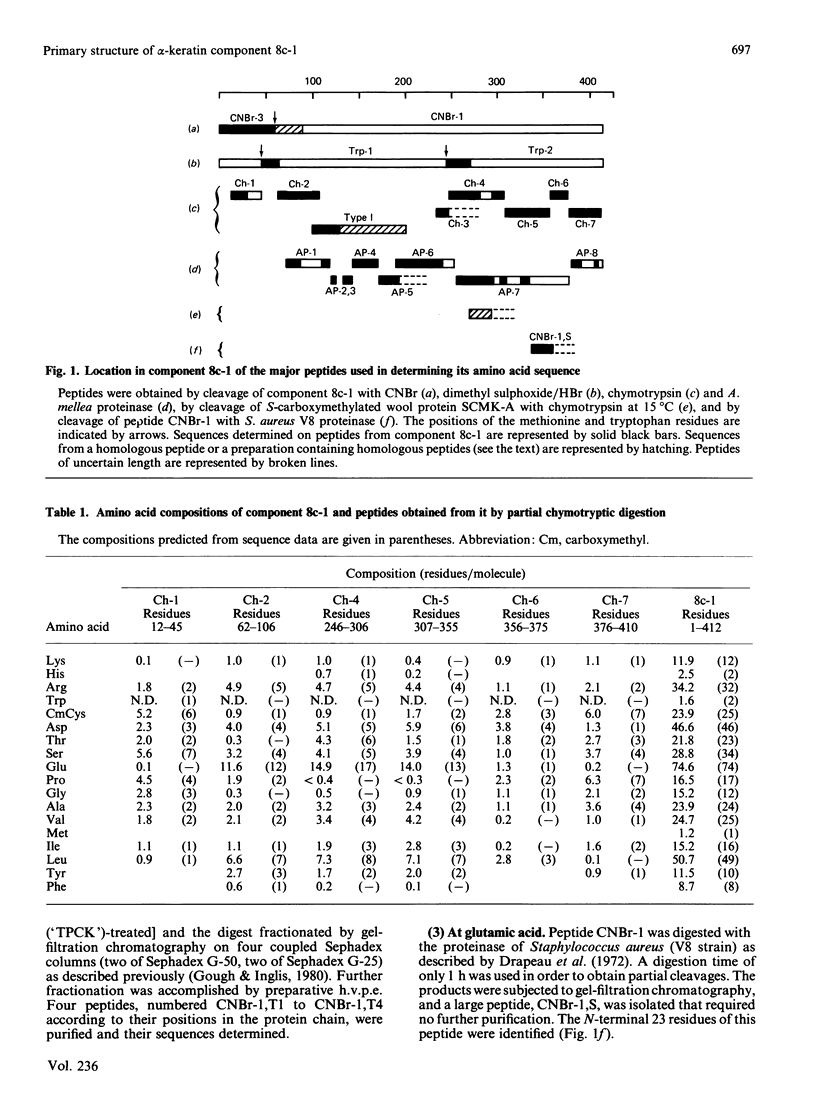
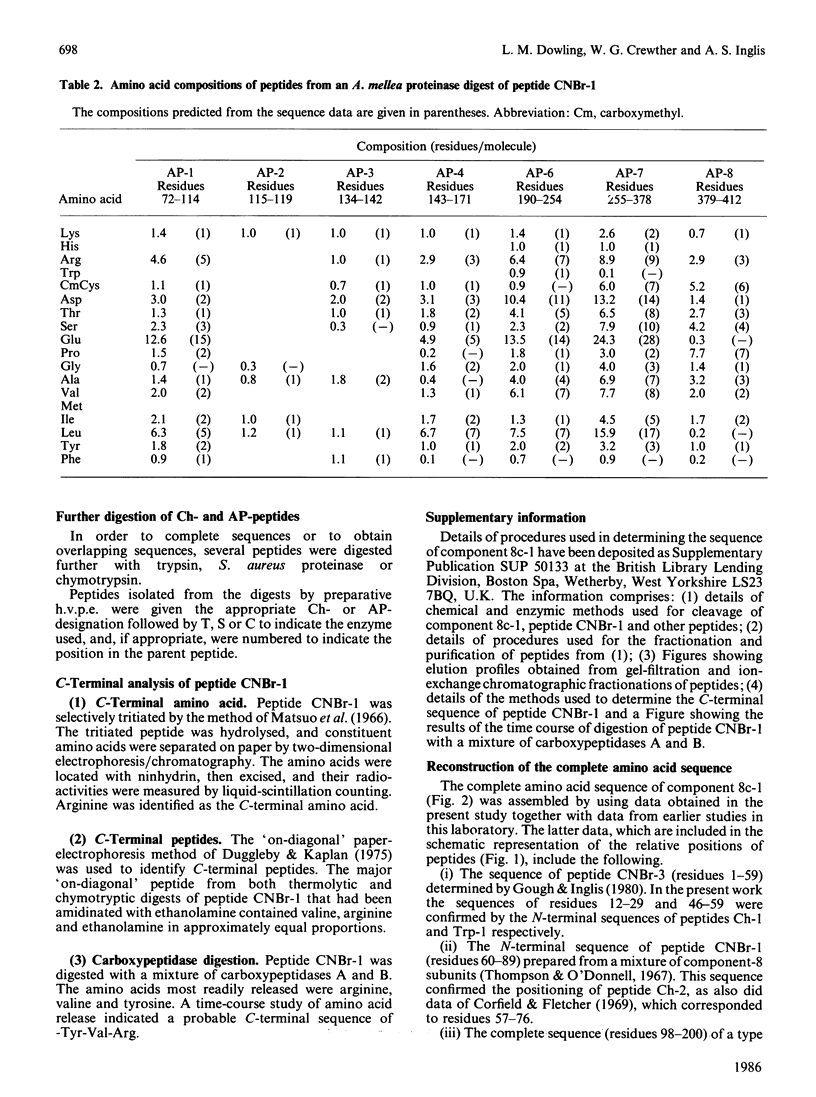
Component 8c-1, one of four highly homologous component-8 subunit proteins present in the microfibrils of wool, was isolated as its S-carboxymethyl derivative and its amino acid sequence was determined. Large peptides were isolated after cleaving the protein chemically or enzymically and the sequence of each was determined with an automatic Sequenator. The peptides were ordered by sequence overlaps and, in some instances, by homology with known sequences from other component-8 subunits. The C-terminal residues were identified by three procedures. Full details of the various procedures used have been deposited as Supplementary Publication SUP 50133 (4 pp.) at the British Library Lending Division, Boston Spa, Wetherby, West Yorkshire LS23 7BQ, U.K., from whom copies can be obtained on the terms indicated in Biochem. J. (1986) 233, 5. The result showed that the protein comprises 412 residues and has an Mr, including the N-terminal acetyl group, of 48,300. The sequence of residues 98-200 of component 8c-1 was found to correspond to the partial or complete sequences of four homologous type I helical segments previously isolated from helical fragments recovered from chymotryptic digests of microfibrillar proteins of wool [Crewther & Dowling (1971) Appl. Polym. Symp. 18, 1-20; Crewther, Gough, Inglis & McKern (1978) Text. Res. J. 48, 160-162; Gough, Inglis & Crewther (1978) Biochem. J. 173, 385]. Considered in relation to amino acid sequences of other intermediate-filament proteins, the sequence is in accord with the view that keratin filament proteins are of two types [Hanukoglu & Fuchs (1983) Cell (Cambridge, Mass.) 33, 915-924]. Filament proteins from non-keratinous tissues, such as desmin, vimentin, neurofilament proteins and the glial fibrillary acidic protein, which form monocomponent filaments, constitute a third type. It is suggested that as a whole the proteins from intermediate filaments be classed as filamentins, the three types at present identified forming subgroups of this class. The significant homologies between types I, II and III occur almost exclusively in segments of the chain that have been identified as having a coiled-coil structure together with the relatively short sections connecting these segments. The non-coiled-coil segments at the C- and N-termini show no significant homology between types, nor is homology in these segments apparent in all members of one type. Component 8c-1 does not show homology in its terminal segments with the known sequence of any other filamentin.(ABSTRACT TRUNCATED AT 400 WORDS)
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Corfield M. C., Fletcher J. C. Amino acid sequences of peptides from a chymotryptic digest of a urea-soluble protein fraction (U.S.3) from oxidized wool. Biochem J. 1969 Nov;115(2):323–334. doi: 10.1042/bj1150323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corfield M. C., Fletcher J. C., Robson A. Amino acid sequences of peptides from a tryptic digest of a urea-soluble protein fraction (U.S.3) from oxidized wool. Biochem J. 1967 Mar;102(3):801–814. doi: 10.1042/bj1020801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crewther W. G., Inglis A. S., McKern N. M. Amino acid sequences of alpha-helical segments from S-carboxymethylkerateine-A. Complete sequence of a type-II segment. Biochem J. 1978 Aug 1;173(2):365–371. doi: 10.1042/bj1730365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doonan S., Doonan H. J., Hanford R., Vernon C. A., Walker J. M., da Airold L. P., Bossa F., Barra D., Carloni M., Fasella P. The primary structure of aspartate aminotransferase from pig heart muscle. Digestion with a proteinase having specificity for lysine residues. Biochem J. 1975 Sep;149(3):497–506. doi: 10.1042/bj1490497d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dopheide T. A. The primary structure of a protein, component 0.62, rich in glycine and aromatic residues, obtained from wool keratin. Eur J Biochem. 1973 Apr 2;34(1):120–124. doi: 10.1111/j.1432-1033.1973.tb02737.x. [DOI] [PubMed] [Google Scholar]
- Dowling L. M., Crewther W. G. Isolation of components from the low-sulphur proteins of wool by fractional precipitation. Prep Biochem. 1974;4(3):203–226. doi: 10.1080/00327487408068199. [DOI] [PubMed] [Google Scholar]
- Dowling L. M., Crewther W. G., Parry D. A. Secondary structure of component 8c-1 of alpha-keratin. An analysis of the amino acid sequence. Biochem J. 1986 Jun 15;236(3):705–712. doi: 10.1042/bj2360705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dowling L. M., Parry D. A., Sparrow L. G. Structural homology between hard alpha-keratin and the intermediate filament proteins desmin and vimentin. Biosci Rep. 1983 Jan;3(1):73–78. doi: 10.1007/BF01121573. [DOI] [PubMed] [Google Scholar]
- Drapeau G. R., Boily Y., Houmard J. Purification and properties of an extracellular protease of Staphylococcus aureus. J Biol Chem. 1972 Oct 25;247(20):6720–6726. [PubMed] [Google Scholar]
- Duggleby R. G., Kaplan H. A general method for the determination of the carboxyl-terminal sequence of proteins. Anal Biochem. 1975 May 12;65(1-2):346–354. doi: 10.1016/0003-2697(75)90518-7. [DOI] [PubMed] [Google Scholar]
- Elleman T. C., Crewther W. G., Van Der Touw J. Amino acid sequences of alpha-helical segments from S-carboxymethylkerateine-A. Statistical analysis. Biochem J. 1978 Aug 1;173(2):387–391. doi: 10.1042/bj1730387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geisler N., Kaufmann E., Fischer S., Plessmann U., Weber K. Neurofilament architecture combines structural principles of intermediate filaments with carboxy-terminal extensions increasing in size between triplet proteins. EMBO J. 1983;2(8):1295–1302. doi: 10.1002/j.1460-2075.1983.tb01584.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geisler N., Plessmann U., Weber K. Related amino acid sequences in neurofilaments and non-neural intermediate filaments. Nature. 1982 Apr 1;296(5856):448–450. doi: 10.1038/296448a0. [DOI] [PubMed] [Google Scholar]
- Geisler N., Weber K. Amino acid sequence data on glial fibrillary acidic protein (GFA); implications for the subdivision of intermediate filaments into epithelial and non-epithelial members. EMBO J. 1983;2(11):2059–2063. doi: 10.1002/j.1460-2075.1983.tb01700.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geisler N., Weber K. Comparison of the proteins of two immunologically distinct intermediate-sized filaments by amino acid sequence analysis: desmin and vimentin. Proc Natl Acad Sci U S A. 1981 Jul;78(7):4120–4123. doi: 10.1073/pnas.78.7.4120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geisler N., Weber K. The amino acid sequence of chicken muscle desmin provides a common structural model for intermediate filament proteins. EMBO J. 1982;1(12):1649–1656. doi: 10.1002/j.1460-2075.1982.tb01368.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gough K. H., Inglis A. S., Crewther W. G. Amino acid sequences of alpha-helical segments from S-carbosymethylkerateine-A. Complete sequence of a type-I segment. Biochem J. 1978 Aug 1;173(2):373–385. doi: 10.1042/bj1730373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanukoglu I., Fuchs E. The cDNA sequence of a Type II cytoskeletal keratin reveals constant and variable structural domains among keratins. Cell. 1983 Jul;33(3):915–924. doi: 10.1016/0092-8674(83)90034-x. [DOI] [PubMed] [Google Scholar]
- Hanukoglu I., Fuchs E. The cDNA sequence of a human epidermal keratin: divergence of sequence but conservation of structure among intermediate filament proteins. Cell. 1982 Nov;31(1):243–252. doi: 10.1016/0092-8674(82)90424-x. [DOI] [PubMed] [Google Scholar]
- Inglis A. S. Modified extraction procedure in spinning-cup sequenators. Methods Enzymol. 1983;91:443–450. doi: 10.1016/s0076-6879(83)91041-8. [DOI] [PubMed] [Google Scholar]
- Inglis A. S., Rubira M. R., Strike P. M. The protein sequenator: a new extraction principle and a metal reaction cup. J Biochem Biophys Methods. 1981 Jun;4(5-6):279–285. doi: 10.1016/0165-022x(81)90068-3. [DOI] [PubMed] [Google Scholar]
- Inglis M. S., Wheatley D. N. Discharge of intracellular amino acid pools. Cell Biol Int Rep. 1980 Mar;4(3):295–301. doi: 10.1016/0309-1651(80)90062-4. [DOI] [PubMed] [Google Scholar]
- Jones L. N. Studies on microfibrils from alpha-keratin. Biochim Biophys Acta. 1976 Oct 28;446(2):515–524. doi: 10.1016/0005-2795(76)90018-0. [DOI] [PubMed] [Google Scholar]
- Krieg T. M., Schafer M. P., Cheng C. K., Filpula D., Flaherty P., Steinert P. M., Roop D. R. Organization of a type I keratin gene. Evidence for evolution of intermediate filaments from a common ancestral gene. J Biol Chem. 1985 May 25;260(10):5867–5870. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lewis S. A., Balcarek J. M., Krek V., Shelanski M., Cowan N. J. Sequence of a cDNA clone encoding mouse glial fibrillary acidic protein: structural conservation of intermediate filaments. Proc Natl Acad Sci U S A. 1984 May;81(9):2743–2746. doi: 10.1073/pnas.81.9.2743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuo H., Fujimoto Y., Tatsuno T. A novel method for the determination of C-terminal amino acid in polypeptides by selective tritium labelling. Biochem Biophys Res Commun. 1966 Jan 4;22(1):69–74. doi: 10.1016/0006-291x(66)90604-8. [DOI] [PubMed] [Google Scholar]
- Quax-Jeuken Y. E., Quax W. J., Bloemendal H. Primary and secondary structure of hamster vimentin predicted from the nucleotide sequence. Proc Natl Acad Sci U S A. 1983 Jun;80(12):3548–3552. doi: 10.1073/pnas.80.12.3548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savige W. E., Fontana A. Cleavage of the tryptophanyl peptide bond by dimethyl sulfoxide-hydrobromic acid. Methods Enzymol. 1977;47:459–469. doi: 10.1016/0076-6879(77)47046-0. [DOI] [PubMed] [Google Scholar]
- Simpson R. J., Neuberger M. R., Liu T. Y. Complete amino acid analysis of proteins from a single hydrolysate. J Biol Chem. 1976 Apr 10;251(7):1936–1940. [PubMed] [Google Scholar]
- Steinert P. M., Parry D. A., Racoosin E. L., Idler W. W., Steven A. C., Trus B. L., Roop D. R. The complete cDNA and deduced amino acid sequence of a type II mouse epidermal keratin of 60,000 Da: analysis of sequence differences between type I and type II keratins. Proc Natl Acad Sci U S A. 1984 Sep;81(18):5709–5713. doi: 10.1073/pnas.81.18.5709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinert P. M., Rice R. H., Roop D. R., Trus B. L., Steven A. C. Complete amino acid sequence of a mouse epidermal keratin subunit and implications for the structure of intermediate filaments. Nature. 1983 Apr 28;302(5911):794–800. doi: 10.1038/302794a0. [DOI] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]



