Abstract
Background
Previous research introduced V-PFCRC as an effective spot urinary dilution adjustment method for various metal analytes, including the major environmental toxin arsenic. V-PFCRC normalizes analytes to 1 g/L creatinine (CRN) by adopting more advanced power-functional corrective equations accounting for variation in exposure level. This study expands on previous work by examining the impacts of age and sex on corrective functions.
Methods
Literature review of the effects of sex and age on urinary dilution and the excretion of CRN and arsenic. Data analysis included a Data Set 1 of 5,752 urine samples and a partly overlapping Data Set 2 of 1,154 combined EDTA blood and urine samples. Both sets were classified into age bands, and the means, medians, and interquartile ranges for CRN and Total Weight urinary Arsenic (TWuAs) in uncorrected (UC), conventionally CRN-corrected (CCRC), simple power-functional (S-PFCRC), sex-aggregated (V-PFCRC SA), and sex-differentiated V-PFCRC SD modes were compared. Correlation analyses assessed residual relationships between CRN, TWuAs, and age. V-PFCRC functions were compared across three numerically similar age groups and both sexes. The efficacy of systemic dilution adjustment error compensation was evaluated through power-functional regression analysis of residual CRN and the association between arsenic in blood and all tested urinary result modes.
Results
Significant sex differences in UC and blood were neutralized by CCRC and reduced by V-PFCRC. Age showed a positive association with blood arsenic and TWuAs in all result modes, indicating factual increments in exposure. Sex-differentiated V-PFCRC best matched the sex-age kinetics of blood arsenic. V-PFCRC formulas varied by sex and age and appeared to reflect urinary osmolality sex-age-kinetics reported in previous research.
V-PFCRC minimized residual biases of CRN on TWuAs across all age groups and sexes, demonstrating improved standardization efficacy compared to UC and CCRC arsenic.
Interpretation
Sex differences in UC and CCRC arsenic are primarily attributable to urinary dilution and are effectively compensated by V-PFCRC. While the sex and age influence on V-PFCRC formulas align with sex- and age-specific urinary osmolality and assumed baseline vasopressor activities, their impact on correction validity for entire collectives is minimal.
Conclusion
The V-PFCRC method offers a robust correction for urinary arsenic dilution, significantly reducing systemic dilution adjustment errors. Its application in various demographic contexts enhances the accuracy of urinary biomarker assessments, benefiting clinical and epidemiological research. V-PFCRC effectively compensates for sex differences in urinary arsenic. Age-related increases in TWuAs are exposure-related and should be additionally accounted for by algebraic normalization, covariate models, or standard range adjustments.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12882-024-03758-w.
Keywords: Non-linear dilution adjustment, Spot urine, Biomarkers, Arsenic, Sex-age differences, Exposure studies
Introduction
Variable Power Functional Creatinine Correction (V-PFCRC)
Urinary biomarkers are essential for medical, epidemiological, and environmental research. However, variations in hydration and urine dilution can significantly affect these biomarkers. Traditional dilution adjustment methods that relate biomarker concentrations to creatinine (CCRC), osmolality, or specific gravity have limitations due to confounders such as age, sex, and muscle mass, as well as complex biochemical interactions [1–3].
One significant limitation of the creatinine (CRN) method is that the excretion of creatinine varies with diuresis in a manner different from most analytes. This discrepancy can lead to misinterpretations, particularly at the extremes of dilution, necessitating restrictions to the accepted dilution range [2, 3].
Previous studies on dilution adjustment of arsenic and various metals demonstrated substantial neutralization of these errors by power functional modifications of the traditional ratio formations between analytes and their correctors [2]. This led to the development of the Variable Power-Functional Creatinine Correction (V-PFCRC) method, which, in contrast to previous power-functional adjustments, further accounts for variations in corrective exponents with analyte levels [3]. It standardizes urinary analytes to 1 g/L CRN by combining uncorrected analyte concentrations and two analyte-specific coefficients c and d according to the general V-PFCRC formula:
uAN = normalized analyte to 1 g/L CRN.
uAUC = uncorrected analyte c and d: numerically derived analyte-specific coefficients.
The innovative V-PFCRC formula has shown promising spot urinary adjustment results for the top priority environmental toxin arsenic, four other metals (cesium, molybdenum, strontium, zinc), and iodine. It effectively neutralized residual bias and improved correlations between blood and urine findings, indicating its potential for medical, forensic, and environmental applications.
Beyond the mathematically tangible systemic dilution adjustment error caused by differential diuresis dependencies, there are further non-linear, exposure-differentiated skews addressed by V-PFCRC. These include complex metabolic and excretion-related interactions between analytes and CRN. For instance, arsenic may limit CRN synthesis, and both may interact through the methyl donor S-Adenosyl Methionine (SAMe) [4]. Additionally, common renal tubular transport mechanisms for CRN and arsenic, such as Multidrug and Toxin Extrusion protein 1 (MATE1) or P-glycoprotein 1/Multidrug Resistance Protein 1 (PGP1/ MDR1), add to the complexity, underscoring the need for innovative approaches to address these challenges [3–6].
While the V-PFCRC method can minimize these non-linear errors, other confounders like age, sex, and lean muscle mass, which V-PFCRC does not compensate, remain and can still influence concentrations of correctors and analytes [2, 3]. The main goal of this study was to assess whether and how far additional adjustments based on age and sex are necessary for V-PFCRC formulas or reference ranges for urinary arsenic. To accomplish this objective, the following steps were undertaken:
Literature Review: Existing literature was reviewed to understand the extent of age- and sex-specific variability in total weight arsenic urine samples and their physiological, pathological, and hormonal backgrounds.
Influence of Age and Sex: The influence of age and sex on various result modes of arsenic spot urine samples was investigated on a large dataset (Set 1) comprising 5,725 samples previously utilized to develop V-PFCRC formulas.
Total Weight Arsenic Analysis in Blood and Urine: The effects of sex and age on total weight arsenic in 1,154 urine and parallel blood samples (Set 2), substantially overlapping with Set 1, were studied.
Quartile Comparison: The blood quartile limits (1–3) and different urinary result modes were compared to assess the alignment of gender ratios and age-related changes relative to younger individuals.
V-PFCRC Formula Assessment: The age- and sex-specific influences on the coefficients of the V-PFCRC formulas were explored to evaluate the need for potential modifications.
Validation: The study further validated the variable power-functional CRN dilution correction determined in previous work by comparing residual CRN-biases of different result modes within age- and sex-specific subgroups. This rigorous validation process ensures the reliability and accuracy of the novel V-PFCRC method.
Aging and sex influence fluid balance and the metabolism of creatinine, toxins, and their respective urinary biomarkers through interconnected structural and functional changes in the kidneys and other organs. Understanding these factors is crucial for accurately adjusting spot urine samples for dilution, ensuring reliable and meaningful exposure estimates.
Sex Differences in Osmoregulation, Urinary Dilution, and Creatinine
Sex differences in urinary dilution and their hormonal-physiological backgrounds are summarized and referenced in Supplements 1–7. Women tend to produce higher weight-adjusted 24-h urine volumes and display higher water turnover and natriuresis (sodium excretion) [7–9]. They typically have lower urinary concentration, osmolality (uOsm), total excreted osmotic load, and creatinine (CRN) levels than men [7, 10–12].
Sex hormones influence CRN in several ways. Androgens (male hormones) increase lean muscle mass and energy metabolism, producing nitrogenous waste products like urea, uric acid, and CRN [13]. Men compensate for the excess CRN production mainly through increased glomerular filtration, proportional to plasma concentrations of freely filtrable CRN and renal plasma flow. Additionally, androgens enhance tubular secretion of CRN by upregulating its primary transporters, Organic Cation Transporter 2 (OCT2), for its basolateral uptake and MATE1 for its apical excretion, while estrogens inhibit these transporters [5, 10, 14–16]. Male hormones also enhance the actions of the Renin–Angiotensin–Aldosterone System (RAAS) and Arginine-Vasopressin (AVP), both crucial for osmoregulation [7, 8, 10, 17–25]. Androgens further increase the sensitivity of AVP-immunoreactive neurons and the number of renal AVP-binding sites [26, 27]. This promotes water and sodium reabsorption, resulting in higher general urinary concentration and CRN levels [7, 10]. Conversely, female hormones inhibit these effects, either directly on the hypothalamic-pituitary axis and kidneys or through the stimulation of renal prostaglandins and NT-proBNP, which act as natural antagonists to the renal fluid-retaining actions of AVP [20, 25, 28, 29]. This explains the higher water turnovers [8], natriureses [30, 31], urinary dilutions [7, 14], and lower extracellular volumes of women [19, 20, 32, 33].
Sex Differences in Renal Arsenic Excretion
Several studies have underscored the superior methylation capacity in women, a critical factor in the metabolic biotransformation of arsenic [34–36]. This finding suggests that sex differences in arsenic metabolism may contribute to the generally lower female arsenic toxicity. The process involves a complex interplay of hormonal, enzymatic, and renal tubular transport factors. Women tend to dispose of higher levels of S-adenosylmethionine (SAMe), the essential methyl donor in the oxidative methylation of arsenic. This may be partially due to estrogen-dependent upregulation of choline synthesis, a critical cofactor in SAMe production [35], besides the lower female lean muscle mass implicating reduced SAMe consumption for creatinine synthesis. Furthermore, women exhibit higher expression of antioxidative protective enzymes such as Endothelial Nitric Oxide Synthase (eNOS), Metallothionein, Manganese Superoxide Dismutase (MnSOD), and Carbonic Anhydrase, which may further enhance the oxidative methylation of inorganic arsenic [10, 37, 38].
Consistently, several sex-comparative studies reported significantly higher urinary proportions of methylated dimethylarsinic acid (%DMA) with lower inorganic arsenic (%iAs) and monomethylarsonic acid (%MMA) in women [34, 36, 39–43]. In contrast to the evident sex-differentiate proportions of iAs, MMA, and DMA [44], no significant differences in covariate dilution-adjusted total weight arsenic and non-toxic arsenobetaine (‘fish-arsenic’) between males and females were found in a sizeable recent Korean study [34].
The complexity of sex differences in arsenic excretion is highlighted by the male-dominant baseline expression of sodium-coupled phosphate cotransporter II (NaPi-IIa). This transporter primarily facilitates sodium reabsorption in the proximal tubule using phosphate (PO4 −) as the primary co-transported anion, with inorganic arsenate (iAsV) able to substitute for PO4 − [34, 45]. However, the processing of these anions in renal tubular cells differs. Plasma PO4 − levels increase with NaPi-IIa upregulation, indicating excess PO4 −reabsorption [46]. In contrast, upon its absorption, arsenate is partly converted to arsenite (iAsIII) within the tubule cells. Inorganic arsenic in tubular cells is either excreted in the urine, reabsorbed into the blood, or stored in the tubular cell, with all options leading to further metabolization of significant parts of this tubular inorganic arsenic [47, 48]. Trivalent arsenite undergoes more efficient methylation than pentavalent arsenate in liver and kidney cells, and the increased uptake of arsenate and its subsequent conversion to arsenite may lead to enhanced net methylation of excreted arsenic [47, 49–52]. This is supported by the observation of higher urinary %DMA levels in postmenopausal women, older individuals, and people with chronic kidney disease, all of whom show increased NaPi-IIa activity due to reduced expression of the proteohormone transmembrane Klotho (m-Kl) [4, 39, 53–61]. Estrogen inhibits NaPi-IIa transporters via upregulation of m-Kl, thus enhancing the excretion of PO4 −and iAsV [62, 63]. However, the expected sex difference in NaPi-IIa activity does not reflect in lower female serum PO4 −levels in younger adults [53, 54]. Only after menopausal estrogen decline does serum PO4 − become female-dominant. Testosterone and other factors could play more age-constant roles in inhibiting PO4 −reabsorption in males or counterbalancing the estrogen effects in females [64]. Postmenopausal changes, including a sharper estrogen decline in women compared to testosterone in men in addition to the above-mentioned female methylation advantages, may explain higher serum phosphate and %DMA, along with lower %iAs in women [53, 54].
Influence of Age on Renal Function and Fluid Balance
Aging induces complex changes in renal function and fluid balance due to an interrelated combination of normal organ aging, toxic build-up, and degenerative diseases like diabetes, hypertension, atherosclerosis, and congestive heart disease [65–68]. The combination of these factors induces specific changes in the kidney, known as nephrosclerosis. Arteriosclerotic fibro-intimal hyperplasia of afferent arterioles is the expected initial pathogenetic step of nephrosclerosis, a condition encompassing four primary aspects:
1. Arteriosclerosis, 2. Glomerulosclerosis, 3. Tubular Atrophy, and 4. Interstitial Fibrosis. Nephrosclerosis is linked to a reduction in kidney mass, renal plasma flow (RPF), number of functioning nephrons, filtration membrane area, and glomerular capillary permeability. These factors contribute to the typical age-related declines in glomerular filtration rate (GFR) [66–69].
Glomerular Hemodynamics in Aging
In the early stages of kidney damage, reduced vascular resistance in the afferent arterioles and increased responsiveness of the efferent arterioles to vasoconstrictive signals lead to elevated glomerular capillary hydrostatic pressure (GCHP) [66]. Despite the increased GCHP, the reactive reduction of glomerular capillary ultrafiltration coefficients prevents a corresponding increase in filtration efficiency and glomerular filtration rate (GFR). Initially, hyperfiltration may occur, but over time, the reduction in ultrafiltration coefficients and increased vasoconstriction of afferent arterioles also impair capillary filtration efficiency and GFR [66, 70].
Age- and Sex-Related Decline of the Glomerular Filtration Rate (GFR)
The GFR is low at birth and increases to adult levels by the age of two years. It remains stable until around 40 and gradually decreases by about 5%–10% per decade [68]. This decline is mainly due to the progressive loss of functioning nephrons. Women typically have lower GFR than men until around the sixth decade, when sex- disparities diminish and may reverse due to a faster decline in male GFR (1.20 vs. 0.96 ml/min per 1.73 m2 per year) [71]. This phenomenon is attributed to higher blood pressure and earlier arteriosclerotic cardiovascular disease in men [68, 71–76]. Hormonal factors additionally contribute to the gender difference in GFR decline as estrogens exert renoprotective effects by inhibiting the RAAS and facilitating advantages in methylation and antioxidation [10, 67, 68, 77, 78].
By contrast, testosterone tends to increase potentially harmful RAAS activity, and androgen receptors mediate cellular aging by telomere shortening and induction of tubular cell apoptosis (Supplements 3 and 6) [10, 77, 78]. Consistently, experimental estrogen therapy and androgen deprivation could be demonstrated to mitigate the progression of chronic kidney disease (CKD) [66].
In advanced stages, the decline in GFR is accompanied by increased permeability of the glomerular basement membrane to macromolecules, facilitating the urinary excretion of significant amounts of albumin and other proteins [66, 79].
Tubular System Changes
Aging also affects the tubular system, leading to tubulointerstitial fibrosis and impairing the secretion and reabsorption of various urinary components. The loop of Henle gradually loses control over salt reabsorption, and the decreased sensitivity of distal tubules and collecting ducts to aldosterone and arginine vasopressin (AVP) results in increased urinary dilution and further reductions in urinary creatinine (CRN) [23, 80, 81].
Impact of Aging on Renal Vascular Function
As individuals age, renal vascular function progressively declines due to atherosclerosis and altered hormonal vasoregulation. Enhanced responses to vasoconstrictors and reduced reactions to vasodilators in various resistance vessels increase total renal vascular resistance and reduce renal plasma flow (RPF), especially in hypertension and albuminuria [82]. These changes result from increased vasoconstrictive activities of AVP and the renin–angiotensin–aldosterone system (RAAS) besides decreased nitric oxide (NO) activity, amplifying renal vasoconstriction, sodium retention, matrix production, and mesangial fibrosis [66, 68, 82–86].
Creatinine Clearance and Creatinine in Serum and Urine
The decline in GFR with age leads to decreased creatinine clearance and lower 24-h urinary CRN. However, spot urine measurements of CRN are unreliable estimates for creatinine clearance due to their strong dependence on hydration status. Similarly, serum creatinine is not a sensitive marker of kidney function in older adults. This is because their reduced muscle mass results in a lower creatinine (CRN) load to be excreted by the kidneys, and the increased secretion of creatinine in the tubules prevents a rise in serum CRN levels in the early stages of chronic kidney disease (CKD), despite reduced glomerular filtration rates (GFR) [50, 66, 87].
Influence of Age on Renal Arsenic Excretion
Numerous studies have investigated how age affects the renal excretion of arsenic. These studies have focused on measuring total weight urinary arsenic (TWuAs), including arsenobetaine, and the sum of inorganic arsenic and its primary metabolites, MMA and DMA (total urinary arsenic, TuAs), excluding arsenobetaine. Additionally, they have examined the concentration of individual arsenic species in urine in absolute terms and relative to total weight arsenic [34, 39, 55, 88–90]. A significant challenge in interpreting these studies is the issue of adequate dilution adjustment. Older adults generally exhibit lower CRN, urinary osmolality, and specific gravity due to reduced renal urine concentration capacity and muscle mass [91]. This can lead to inaccuracies, with uncorrected urine values being lower due to higher dilution, while classically corrected values may be inadequately high due to the smaller corrective denominators [2, 3].
In a Korean study, arsenic urine levels of 1,507 participants were standardized using a covariate-adjusted method that accounted for the influences of age, sex, and BMI on CRN [1, 34, 44]. This advanced approach revealed significant positive associations between age and TWuAs or arsenobetaine in adults over 18. Positive trends for iAs, MMA, and DMA were observed in adults over 65 but did not reach statistical significance. Another study of 904 participants in Nevada, which included CRN as an independent covariate in a multiple linear regression model, found that age significantly negatively influenced iAs and %iAs while showing positive but non-significant trends for MMA, DMA, %DMA, and TuAs [55]. Age was positively associated with methylation capacity, significantly affecting the primary (PMI = MMA/iAs) and marginally affecting the secondary methylation index (SMI = DMA/MMA). These findings align with previous studies showing increased methylation of urinary arsenic in adults, with lower %iAs and higher %DMA [39, 88].
The reduction in %iAs with age may also be influenced by declines in the proteohormone transmembrane Klotho (m-Kl) associated with aging or chronic kidney disease (CKD) [56, 92–94]. As an essential cofactor of fibroblast growth factor 23 (FGF23), m-Kl assists the urinary excretion of iAsVby inactivating the apical tubular sodium-phosphate cotransporters NaPi-IIa (and NaPi-IIc) mentioned above [95–97]. Lower activity of m-Kl in CKD increases the reabsorption of iAsV and its cellular conversion into iAsIII and subsequent methylation. Dehydration, common in older adults, exacerbates this effect by activating AVP and ATII, which further reduces the excretion of PO4 − and iAsV [98, 99]. Serum levels of m-Kl are higher in females throughout life and are positively correlated with estrogen, testosterone, and SHBG in men [57, 58].
Contrary results were observed in Taiwan's hyperendemic arseniasis area [42], where older individuals exhibited increased %iAs and decreased %DMA, suggesting regional differences in age-dependent urinary arsenic composition due to diverse exposure sources or genetic differences in methylation efficiency [40]. A study in Bangladesh involving children found lower primary (PMI) and higher secondary methylation indices (SMI) in children than adults, further supporting age-dependent differences in methylation efficiency and potentially explaining the absence of arsenic-induced skin lesions in children [89].
In individuals over 90, there may be a reversal in age-related inorganic arsenic (iAs) reabsorption, inferred from observed drops in serum phosphate (PO4 –) levels in this age group, which tend to increase from the third decade of life [53]. Age-related declines in renal concentrating ability and calcitriol (vitamin D3) formation lead to higher renal losses and reduced intestinal absorption of calcium [57, 58]. This increases plasma parathyroid hormone (PTH), potentially facilitating renal hyperexcretion of PO4 − and iAsvvia downregulating NaPi-IIa transporters [56, 92–94]. While in younger seniors, these effects are overcompensated by the age-related decline in membrane-Klotho (m-KL), in the very old, these processes may prevail and lead to increased PO4 − and iAsvexcretion. However, the biotransformation and renal handling of arsenic in this age group remains to be studied in more detail [53].
Influence of Kidney Function on Arsenic Excretion
Chronic kidney disease (CKD) has a well-established association with urinary arsenic levels [4, 59, 62]. Several studies have shown significant negative correlations between the estimated glomerular filtration rate (eGFR) and total urinary arsenic (TuAs), including inorganic arsenic and its primary metabolites, MMA and DMA [4, 64, 100]. Additionally, there have been positive associations between TuAs and serum cystatin C [101], albuminuria, or proteinuria [62]. Moreover, there are positive correlations between TuAs and urinary markers for tubular damage, such as β2-microglobulin and N-acetyl-β-D-glucosaminidase (NAG) [4, 100], and in various populations, CKD mortality rates consistently increase with rising TuAs levels [4, 102, 103].
Studies investigating the influence of renal function on arsenic excretion, akin to those on age (See Influence of Age on Renal Function and Fluid Balance), indicate a decrease in the percentage of inorganic arsenic (%iAs) alongside concurrent elevations of the percentage of dimethylarsinic acid (%DMA) as glomerular filtration declines [60, 61]. Furthermore, there is evidence for impaired excretion of inorganic arsenic from blood to urine in dysfunctional kidneys, as the corresponding elevation of urinary %iAs is diminished with reduced GFR for a given increase in blood %iAs [60].
These findings support the interpretation as 'reverse causation,' where reduced GFR is understood as a causal factor for impaired renal excretion of inorganic arsenic [61]. Prolonged retention of inorganic arsenic in the blood may facilitate its further methylation, leading to eventual excretion as dimethylarsinic acid. Biochemical evidence supports this notion. Specifically, the downregulation of membrane-Klotho (m-KL) results in higher reabsorption and lower excretion of phosphate and inorganic arsenic in chronic kidney disease (CKD) (See Influence of Age on Renal Function and Fluid Balance and Supplement 3) [104]. In CKD, increased renal angiotensin II (ATII), due to renin release in response to reduced renal plasma flow, inhibits m-KL. The pharmacological blockade of ATII consistently upregulates m-KL expression [105], thereby counteracting the inhibitory influence of m-KL depletion on renal inorganic arsenic excretion [106].
In advanced chronic kidney disease, the effects of m-KL reduction on total urinary arsenic (TuAs) composition may be augmented by concurrent tubular dysfunction. This may result in further shifts from inorganic arsenic (%iAs) to dimethylarsinic acid (%DMA) due to disproportionate alterations in the net excretion of various total urinary arsenic constituents [3, 4].
However, the concept of reverse causation in the observed associations between glomerular filtration rate and urinary arsenic and its methylated forms is challenged. A higher percentage of dimethylarsinic acid (%DMA) and a lower percentage of inorganic arsenic (%iAs) in CKD could potentially induce kidney damage rather than result from it [4]. This is because the oxidative methylation of inorganic arsenic to dimethylarsinic acid generates more toxic intermediates, such as MMAIII or DMAIII, which have been inconsistently found to negatively correlate with the estimated glomerular filtration rate (eGFR) [4, 107–110].
This study utilized a comprehensive dataset from diverse populations to elucidate the intricate influence of sex- and age-related variability of the metabolism of arsenic and CRN, kidney function, and fluid balance on Total Weight arsenic levels in both blood and urine across various dilution adjustment modes.
Methodology
Study Population
The study was based on anonymized data of n = 5,752 spot urine samples (Set 1) collected between January 2014 and August 2022 from the Institute for Medical Diagnostics Berlin-Potsdam, Germany (IMD), as used in previous research for V-PFRC formula generation [3]. The data included information on sex, age, CRN, and total weight of urinary arsenic, which was part of multi-element metal screening panels in the urine of diverse populations in Germany, Switzerland, and Austria. These panels were conducted as routine preventive and diagnostic medical screening procedures, ensuring a comprehensive and generally demographically representative dataset. Apart from sex and age, no further health-related clinical information was available from the Institute for Medical Diagnostics.
A further dataset (Set 2) of 1,170 samples was investigated to compare urinary results with blood, where total weight arsenic was detected in parallel in EDTA whole blood and urine. The samples of Set 2 were obtained and analyzed by IMD between April 2014 and March 2024. The original 1,175 samples were trimmed for CRN < 3.5 g/L and age ≤ 85. Of the remaining 1,154 samples, 70.4% were part of the first dataset, while 29.6% were collected later. The key statistics of Set 1 and Set 2 are summarized in Tables 1 and 2. Histograms comparing data distributions of both sets are depicted in Supplements 8–13. It can be assumed that the samples represent the combined background exposure of inorganic and organic arsenic, as there was no specific suspicion of arsenic contamination in drinking water leading to the multi-element metal screening. The recommended WHO limit of 10 µg/L arsenic in drinking water is enforced and generally met in the sample collection areas [111–114]. Any significant elevations of total weight arsenic levels in these regions are most commonly attributable to arsenobetaine, mainly from marine food, considering the quantitative predominance of this organic source [49, 115].
Table 1.
Basic statistics of Set 1. Sex-aggregated and -differentiated data are given for age, CRN, and urinary arsenic of the five result modes detailed in Table 3: Uncorrected (UC), Conventional CRN-correction (CCRC), Sex-differentiated simple power functional CRN correction (S-PFCRC SD), Sex-aggregated (V-PFCRC SA) and Sex-differentiated variable power functional CRN correction (V-PFCRC SD)
| Set 1 All (F + M), n = 5752 |
Age (years) |
CRN (g/L) |
UC (µg/L) |
CCRC (µg/g CRN) |
S-PFCRC SD (µg/1 g CRN) |
V-PFCRC SA (µg/1 g CRN) |
V-PFCRC SD (µg/1 g CRN) |
|---|---|---|---|---|---|---|---|
| Mean | 51 | 0.98 | 23.8 | 26.1 | 24.5 | 25.3 | 23.8 |
| Std. Deviation | 15.7 | 0.71 | 99.1 | 89.0 | 87.8 | 88.4 | 81.2 |
| Minimum | 0 | 0.03 | 0.49 | 0.67 | 0.71 | 0.73 | 0.64 |
| Maximum | 94 | 5.84 | 5763 | 5100 | 5170 | 5073 | 4879 |
| Quartile 1 | 42 | 0.42 | 4.0 | 5.6 | 5.4 | 5.5 | 5.5 |
| Quartile 2 (Median) | 52 | 0.81 | 8.5 | 10.8 | 10.0 | 10.1 | 10.1 |
| Quartile 3 | 61 | 1.37 | 20.3 | 24.9 | 22.8 | 23.7 | 22.5 |
| Interquartile Range | 19 | 0.95 | 16.3 | 19.2 | 17.5 | 18.2 | 17.0 |
| Females, n = 3086 | Age | CRN | UC | CCRC | S-PFCRC SD | V-PFCRC SA | V-PFCRC SD |
| Mean | 51.4 | 0.8 | 18.0 | 25.6 | 22.7 | 24.2 | 21.4 |
| Std. Deviation | 14.6 | 0.62 | 43.6 | 60.2 | 53.8 | 59.5 | 45.6 |
| Minimum | 0 | 0.04 | 0.49 | 0.67 | 0.71 | 0.73 | 0.64 |
| Maximum | 91 | 5.84 | 1219 | 2102 | 1875 | 2121 | 1397 |
| Quartile 1 | 42 | 0.33 | 3.2 | 5.9 | 5.4 | 5.5 | 5.6 |
| Quartile 2 (Median) | 53 | 0.62 | 7.0 | 11.0 | 9.7 | 10.0 | 9.8 |
| Quartile 3 | 61 | 1.1 | 16.2 | 24.0 | 21.0 | 22.2 | 20.3 |
| Interquartile Range | 19 | 0.77 | 13.0 | 18.1 | 15.7 | 16.7 | 14.7 |
| Males, n = 2666 | Age | CRN | UC | CCRC | S-PFCRC SD | V-PFCRC SA | V-PFCRC SD |
| Mean | 50.6 | 1.19 | 30.5 | 26.7 | 26.6 | 26.5 | 26.6 |
| Std. Deviation | 16.7 | 0.75 | 137.5 | 113.5 | 115.3 | 112.9 | 108.6 |
| Minimum | 0 | 0.03 | 0.51 | 0.81 | 0.85 | 0.89 | 0.92 |
| Maximum | 94 | 5.52 | 5763 | 5100 | 5170 | 5073 | 4879 |
| Quartile 1 | 41 | 0.62 | 5 | 5.4 | 5.4 | 5.4 | 5.4 |
| Quartile 2 (Median) | 52 | 1.08 | 10.6 | 10.6 | 10.4 | 10.5 | 10.4 |
| Quartile 3 | 62 | 1.59 | 26.4 | 25.4 | 25.1 | 25.1 | 25.2 |
| Interquartile Range | 21 | 0.97 | 21.4 | 20.0 | 19.7 | 19.7 | 19.8 |
Table 2.
Basic statistics of Set 2. Sex-aggregated and -differentiated data are given for age, CRN, and arsenic in EDTA blood and urine in the five result modes detailed in Table 3
| Set 2 All (F + M), n = 1170 |
Age (years) |
CRN (g/L) |
UC (µg/L) |
CCRC (µg/g CRN) |
S-PFCRC SD (µg/1 g CRN) |
V-PFCRC SA (µg/1 g CRN) |
V-PFCRC SD (µg/1 g CRN) |
EDTA Blood (µg/L) |
|---|---|---|---|---|---|---|---|---|
| Mean | 51.6 | 1.12 | 33.2 | 29.8 | 29.1 | 29.2 | 28.5 | 1.7 |
| Std. Deviation | 14.21 | 0.77 | 112.0 | 79.8 | 81.4 | 79.1 | 74.9 | 3 |
| Minimum | 5.6 | 0.04 | 0.4 | 0.8 | 0.8 | 0.9 | 0.9 | 0.08 |
| Maximum | 87.9 | 5.84 | 3176 | 2146 | 2241 | 2133 | 1917 | 44 |
| Quartile 1 | 42.3 | 0.54 | 4.3 | 5.3 | 5.2 | 5.2 | 5.3 | 0.4 |
| Quartile 2 (Median) | 52.2 | 0.96 | 10.2 | 10.8 | 10.6 | 10.6 | 10.5 | 0.8 |
| Quartile 3 | 60.4 | 1.53 | 26.4 | 29.5 | 27.9 | 28.8 | 26.9 | 1.8 |
| Interquartile Range | 18.1 | 0.99 | 22.1 | 24.2 | 22.7 | 23.6 | 21.6 | 1.4 |
| Females, n = 596 | Age | CRN | UC | CCRC | S-PFCRC SD | V-PFCRC SA | V-PFCRC SD | EDTA Blood |
| Mean | 51.5 | 0.95 | 22.7 | 26.3 | 24.5 | 25.0 | 24.0 | 1.4 |
| Std. Deviation | 14.2 | 0.72 | 52.3 | 51.6 | 49.9 | 50.3 | 49.4 | 2.9 |
| Minimum | 5.7 | 0.04 | 0.4 | 1.1 | 1.2 | 1.1 | 1.0 | 0.10 |
| Maximum | 84.8 | 5.84 | 778 | 778 | 778 | 778 | 778 | 44 |
| Quartile 1 | 42.4 | 0.4 | 3.3 | 5.2 | 4.8 | 4.9 | 5.0 | 0.3 |
| Quartile 2 (Median) | 51.8 | 0.78 | 7.7 | 9.6 | 9.1 | 9.4 | 9.1 | 0.7 |
| Quartile 3 | 59.7 | 1.3 | 19.3 | 26.4 | 22.7 | 23.1 | 21.9 | 1.5 |
| Interquartile Range | 17.3 | 0.9 | 16.0 | 21.2 | 18.0 | 18.2 | 16.9 | 1.2 |
| Males, n = 574 | Age | CRN | UC | CCRC | S-PFCRC SD | V-PFCRC SA | V-PFCRC SD | EDTA Blood |
| Mean | 51.8 | 1.29 | 44.2 | 33.4 | 33.8 | 33.6 | 33.1 | 2.0 |
| Std. Deviation | 14.3 | 0.78 | 150.0 | 101.0 | 104.4 | 100.6 | 94.1 | 3.1 |
| Minimum | 5.6 | 0.12 | 0.6 | 0.8 | 0.8 | 0.9 | 0.9 | 0.08 |
| Maximum | 87.9 | 4.48 | 3176 | 2146 | 2241 | 2132 | 1917 | 29 |
| Quartile 1 | 42.2 | 0.67 | 5.5 | 5.6 | 5.7 | 5.8 | 5.7 | 0.5 |
| Quartile 2 (Median) | 53.1 | 1.19 | 13.0 | 12.1 | 11.9 | 11.9 | 11.9 | 1.0 |
| Quartile 3 | 60.8 | 1.73 | 37.6 | 33.5 | 32.9 | 32.9 | 33.3 | 2.3 |
| Interquartile Range | 18.6 | 1.06 | 32.1 | 27.9 | 27.2 | 27.1 | 27.6 | 1.8 |
Treatment of Arsenic Values Below the Detection Limit
The 116 female and 32 male samples displaying uncorrected urinary arsenic levels below the ICP-MS detection limit of 1 µg/L were assumed to range between 0.5 and 0.99 µg/L due to prevalent arsenic exposure rendering values 0.5µg/L improbable. Values below the detection limit were estimated considering the apparent strong positive association of uncorrected arsenic and CRN using the formulas:
Females: estimated uAsUC = 0.92 + 0.17 × ln (CRN g/L)
Males: estimated uAsUC = 0.99 + 0.20 × ln (CRN g/L)
This method distributed samples within the 0.5–0.99 µg/L range, averaging 0.62 µg/L for females and 0.72 µg/L for males. This approach provided more plausible distributions than omitting values or using the usual fixed substitutions like half the detection limit (0.5 µg/L) or the limit divided by √2 (0.71 µg/L). For the 69 blood samples below the 0.2 µg/L detection limit, values were estimated based on the strong association of CRN-corrected urinary with blood arsenic using the unisex formula:
Estimated blood Arsenic = 0.082 + 0.028 × ln (V-PFCRC SD Arsenic)
This approach distributed blood values below the detection limit between 0.08 and 0.19 µg/L, averaging 0.12 µg/L, which corresponds to 60% of the detection limit.
Sample collection, preservation, transportation, and storage procedures
Urine
Urine samples were collected in neutral monovettes (Sarstedt) and shipped to the laboratory at room temperature within 24 h for immediate CRN measurement. They were then stored at 4°C for 1–4 days before ICP-MS measurement.
Blood
Venous whole blood was collected in tubes with EDTA for anticoagulation (Sarstedt) and transported to the laboratory overnight or by surface mail. The stability of elements in these specimens was validated over 14 days at room temperature.
Sample preparation and analysis procedures
Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) Measurements of arsenic
The total weight of arsenic in anticoagulated whole blood and urine was determined using ICP-MS. Arsenic species were not analyzed. For ICP-MS analysis, urine samples were diluted 1:10 in 1% HNO3 (Suprapur, Supelco), and serum and anticoagulated whole blood samples were diluted 1:20 in 0.1% NH3 (Suprapur, Supelco) and 0.02% Lutrol F88 (AppliChem). Metals were analyzed in collision/reaction cell mode using the ICapQ ICP-MS system (Thermo Fisher), with external and internal standard calibration (Elemental Scientific). Results are the means of three measurements.
Creatinine Measurement
Creatinine analysis was performed enzymatically using the Alinity assay (Abbott Laboratories).
Normalization of arsenic to urinary concentrations of 1 g/L CRN
The standardization of CRN-corrected urine measurements to 1 g/L CRN and the process of V-PFCRC formula generation are explained in detail in the Supplementary File (V-PFCRC Formula Generation) and in previous work [3]. Two general forms of power-functional correction can be distinguished:
The simple power functional CRN-correction (S-PFCRC) employing a fixed exponent b in the corrective formula:
uAsN = urinary arsenic normalized to 1 g/L CRN.
uAsUC = uncorrected urinary arsenic (µg/L).
Uncorrected and conventionally CRN-corrected (CCRC) results can be seen as particular cases of S-PFCRC, with the uncorrected results taking the exponent b as 0 and the CCRC as 1.
-
2.
The variable power functional correction (V-PFCRC) using two analyte specific coefficients, c and d, allows the calculation of a variable exponent b depending on the concentrations of the analyte and CRN:
Both the simple and variable power functional correction formulas were adopted from the previous study [3]. The simple formula was tested in a sex-differentiated mode, using separate exponents b for males and females. The variable formula was tested in sex-differentiated and sex-aggregated modes, the latter using identical c and d values in both sexes. For all power-functional modes tested in this paper, the asymmetrical distribution of CRN values was compensated as described in the Supplementary File, as asymmetry-compensation improved the accuracy of V-PFCRC [3]. Table 3 summarizes the exponents b (for S-PFCRC) and the coefficients c and d (for V-PFCRC) used.
Table 3.
Exponents and coefficients used in corrective formulas [3]
| Result Mode | Sex- differentiate or aggregate SD / SA |
Exponent b (S-PFCRC) |
Coefficient c (V-PFCRC) |
Coefficient d (V-PFCRC) |
|||
|---|---|---|---|---|---|---|---|
| Female | Male | Female | Male | Female | Male | ||
| Uncorrected | – | 0 | – | ||||
| CCRC | – | 1 | – | ||||
| V-PFCRC | SD | – | – 0.11 | 0.079 | 1.05 | 0.69 | |
| SA | – | 0.03 | 0.78 | ||||
| S-PFCRC | SD | 0.79 | 0.89 | – | |||
Statistical analysis
Data were analyzed using Microsoft Excel and Datatab.net software. Results were expressed as mean ± standard error, medians, and quartile limits (Q1 and Q3). The normality of data distribution was evaluated with Q-Q plots and the Kolmogorov–Smirnov tests. For normally or near-normally distributed data, 2-sided Student's t-tests and Pearson correlation analysis were applied. For non-normally distributed data, Spearman rank correlations were calculated, and Mann–Whitney U-tests were used to test significance. A p-value of < 0.05 was considered statistically significant.
Analysis of age- and sex-effects on arsenic in blood and urine
Urine results from 5,752 samples (Set 1) and 1,154 urine and blood samples (Set 2) were analyzed for age-related effects on arsenic and creatinine levels using Spearman correlation. In Set 1, samples were grouped into nine ten-year age bands, excluding the top and bottom 5% of SD V-PFCRC arsenic values in each band to avoid outliers. Basic statistics (number, mean, standard deviation, standard error, minimum, maximum, and quartiles 1–3) were calculated for the remaining 5,166 samples (2,773 female and 2,393 male) across age, creatinine, and all urinary outcome modes listed in Table 3. Set 2 was similarly analyzed but divided into four 15-year age bands due to the smaller sample size. Detailed statistics for both sets are provided in the Supplementary Excel file.
To better illustrate the effects of age on arsenic concentrations in blood and urine result modes (UC, CCRC, and V-PFCRC-SD), the percentage deviations of quartiles 1–3 for the three older age groups from the values of the youngest age group in Set 2 were calculated and the results presented in bar charts.
Determination of age and sex-specific v-pfcrc-formulas
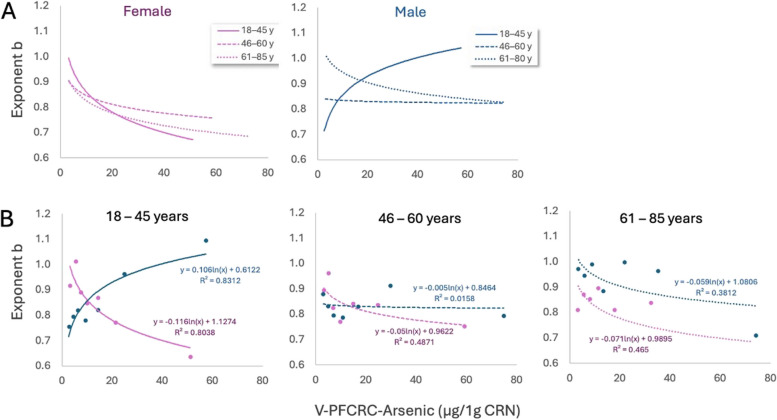
To investigate the impact of age and sex on the formulas, the sample population was divided into three age groups: 18–45, 46–60, and 61–85. Vital statistics for these groups are summarized in the Supplementary Excel file. For each age group, V-PFCRC correction formulas were determined for both sexes, as detailed in the Supplementary File (V-PFCRC Formula Generation). The six age- and sex-specific log-linear regression curves between V-PFCRC-adjusted arsenic and exponent b, defining the analyte-specific coefficients c and d, were compared within each sex (Fig. 6a) and between both sexes (Fig. 6b).
Fig. 6.
Age- and Sex-differentiated log-linear relationships between Exponents b and V-PFCRC normalized arsenic to 1 g/L CRN. Curves are sorted by either sex (A) or age group (B)
Validation of standardization efficacy in separate age groups
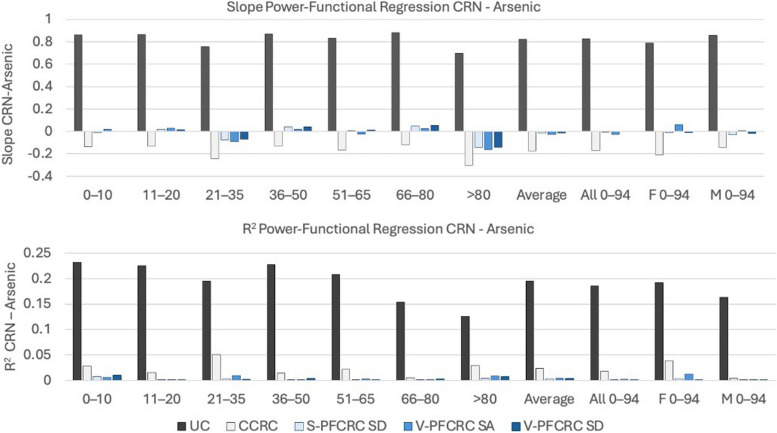
The V-PFCRC and S-PFCRC formulas were initially developed for a broad age range (14–82 years). To evaluate the effectiveness of these general standardization formulas across different ages, Set 1 was divided into seven age bands. A power regression analysis (Arsenic = a x CRN^b) was conducted for each band to examine the relationship between creatinine (CRN) and urinary arsenic. Lower slope (b) and R [2] values indicated better CRN independence, reflecting higher standardization efficacy. Results were compared using bar charts for each age group and the entire Set 1, with additional analysis for both sexes.
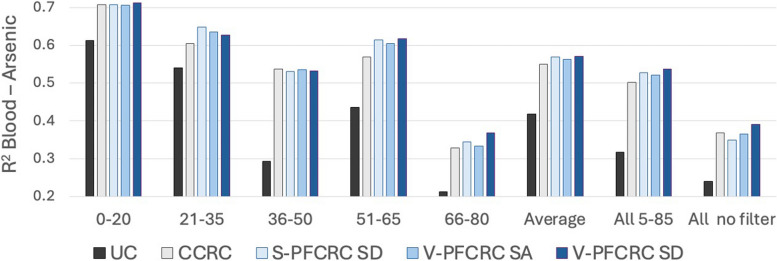
Similarly, Set 2 was analyzed to evaluate the agreement between blood and urine results. Coefficients of determination (R2) values were calculated for five age groups (0–20, 21–35, 36–50, 51–65, and 66–80) to assess the correlation between arsenic levels in blood and urine, indicating effective dilution adjustment. A patient with implausibly low urine and high blood arsenic values was excluded. Detailed statistics for age groups are provided in the Supplementary Excel file.
Results
Effects of age and sex on CRN and urinary arsenic
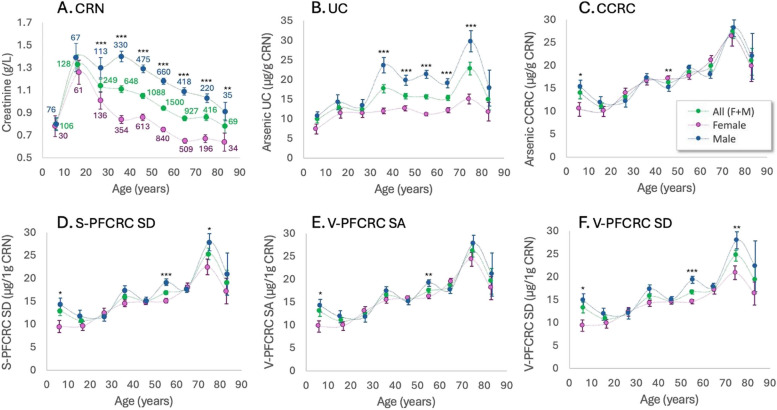
Figures 1a-f illustrate how age and sex influence CRN and urinary arsenic levels across five result modes in Set 1. Creatinine levels increase steadily in children and adolescents due to growing lean muscle mass, peak in early adulthood, and gradually diminish with age. This age decline is more pronounced in males (r = -0.14) than in females (r = -0.1), starting later in males between the second and third decades. For females, the decrease begins shortly after puberty, influenced by generally lower lean muscle mass and the sex hormonal influence on fluid metabolism outlined in the introduction (Sex differences in Osmoregulation, Urinary Dilution, and Creatinine).
Fig. 1.
Sex-Differentiated Influence of Age on CRN and Urinary Arsenic across five result modes in Set 1. Means ± standard errors are plotted for nine 10-year age bands. Outliers above the 95% and below the 5% percentile of each age band were not analyzed. Post-outlier exclusion sample sizes are listed in Graph A. Urinary result modes are detailed in Table 3. Group statistics are provided in the Supplementary Excel file. Significance levels of sex differences (2-sided t-test): * 0.05 ≥ p > 0.025, ** 0.025 ≥ p > 0.01, *** p < 0.001
In contrast, arsenic levels show an age-related rise for both sexes across all urine result modes, indicating actual age-related exposure increments beyond the effects of declining urinary CRN. The correlation with age for CRN-corrected arsenic levels is weak but significant (p < 0.001): overall r = 0.11–0.12, females r = 0.09–0.11, and males r = 0.13–0.14. Linear regression analysis indicates an average annual increase in V-PFCRC arsenic from puberty to the mid-seventies of 0.23 µg/g CRN for males and 0.16 µg/g CRN for females.
In CCRC urine, the lower CRN levels in females computationally amplify the apparent increase in arsenic more than in males, effectively narrowing the gender gap observed in uncorrected urinary arsenic (see Fig. 1b and c). The effect is less pronounced in the power-functional CRN corrections, particularly in the sex-differentiated V-PFCRC SD, which retains a significant male bias in urinary arsenic. This residual gender difference in simple and variable PFCRC appears to reflect a more accurate representation of actual exposure, as this gap is also present in the EDTA blood of Set 2 (Fig. 2b). Consequently, the evidence supports the use of V-PFCRC SD and, to a lesser extent, the other two power-functional correction modes.
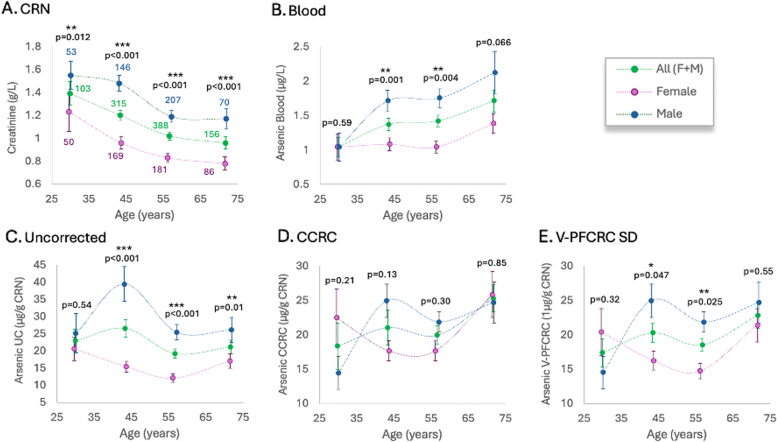
Fig. 2.
Sex-Differentiated Influence of Age on CRN, Urine, and Blood Arsenic in Set 2. Analogous representation to Fig. 1 for Data Set 2, including arsenic values in EDTA blood determined in parallel with urine of three result modes in four 15-year-age bands (21–35, 36–50, 51–65, and 66–80 years). Means ± standard errors for the remaining 486 female and 476 male samples are plotted. Significance level of sex-difference (2-sided t-tests): * 0.05 > p > 0.025, ** 0.025 ≥ p > 0.01, *** p < 0.001
Males consistently exhibit higher creatinine levels across all adult age groups, a disparity not seen in younger children and adolescents. Corresponding sex differences in uncorrected arsenic (uAsUC) lag those of CRN and are partly confounded by the generally higher urine concentration of males explained above. The sex differences in uAsUC emerge in young adulthood and peak in the mid-forties due to steeper increases in males generating significantly higher uAsUC across all adult age bands except the third decade. Consistently low but significant positive correlations between uAsUC and age are observed in the combined and male data but not in females (overall r = 0.05, males r = 0.08).
Notably, both sexes experience steep increases in arsenic levels between the seventh and eighth decades. This late-life urinary arsenic flush, in the absence of corresponding drops in CRN, suggests actual overexposure to arsenic in this age group. This finding is supported by the highest arsenic levels in EDTA blood in both sexes at older ages, likely due to the redistribution of stored arsenic from internal sources.
Comparison of the effects of age and sex on urinary and blood arsenic
The relationship between age and arsenic levels in blood and urine was analyzed using a smaller dataset (Set 2), which included limited samples from patients under 20 and over 80. Therefore, the age range of 20 to 80 was divided into four 15-year intervals. In line with previous research [116, 117], average arsenic levels in blood were substantially lower than in urine, with V-PFCRC urine concentrations being 17.5 times higher in women and 16.6 times higher in men compared to EDTA whole blood. For women, blood arsenic levels remained stable across the first three age groups, increasing significantly only in the oldest group. Conversely, men showed a more continuous increase in blood arsenic levels from the youngest to the oldest age group, leading to significant male-dominated gender differences in the three older groups.
Both uncorrected and V-PFCRC results in Set 2 widely reflect the gender disparity and age dependence seen in Set 1. At the same time, CCRC adjustment consistently reduced this gender disparity to insignificance in both sets. Unlike the age-increasing kinetic of Set 1, women in Set 2 exhibited a trough-shaped trend in urinary arsenic levels, with an initial decrease in the younger age bands followed by an increase in the sixth decade of life. Men displayed an initial increase between the first and second groups, a decrease between the second and third groups, and a subsequent rise towards the oldest group. These discontinuous trends resulted in positive correlations between age and urinary arsenic, though less pronounced than those in Set 1 or those between age and blood arsenic, as detailed in Tables 4 and 5.
Table 4.
Correlations between Age, CRN, and all urinary arsenic result modes of Set 1. Spearman coefficients r and significances p are given for non-normally distributed data
| Set 1 / 21–94 Years | ||||||||
|---|---|---|---|---|---|---|---|---|
| All (F + M) | n = 5488 | Age | CRN | UC | CCRC | S-PFCRC SD | V-PFCRC SA | V-PFCRC SD |
| Age | Correlation | -0.11 | 0.04 | 0.11 | 0.11 | 0.1 | 0.1 | |
| p | < .001 | 0.004 | < .001 | < .001 | < .001 | < .001 | ||
| CRN (g/L) | Correlation | -0.11 | 0.53 | -0.12 | 0.01 | -0.01 | 0.01 | |
| p | < .001 | < .001 | < .001 | 0.591 | 0.625 | 0.522 | ||
| Female | n = 2983 | Age | CRN | UC | CCRC | S-PFCRC SD | V-PFCRC SA | V-PFCRC SD |
| Age | Correlation | -0.1 | 0.02 | 0.09 | 0.08 | 0.08 | 0.08 | |
| p | < .001 | 0.405 | < .001 | < .001 | < .001 | < .001 | ||
| CRN (g/L) | Correlation | -0.1 | 0.56 | -0.13 | 0.03 | -0.01 | 0.03 | |
| p | < .001 | < .001 | < .001 | 0.08 | 0.713 | 0.154 | ||
| Male | n = 2505 | Age | CRN | UC | CCRC | S-PFCRC SD | V-PFCRC SA | V-PFCRC SD |
| Age | Correlation | -0.14 | 0.06 | 0.14 | 0.13 | 0.13 | 0.13 | |
| p | < .001 | 0.002 | < .001 | < .001 | < .001 | < .001 | ||
| CRN (g/L) | Correlation | -0.14 | 0.46 | -0.11 | -0.04 | -0.01 | -0.03 | |
| p | < .001 | < .001 | < .001 | 0.065 | 0.516 | 0.185 | ||
Table 5.
Spearman Correlations and significances between age and arsenic in all urinary arsenic result modes and blood of Set 2
| Set 2 / 21–88 Years | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| All (F + M) | n = 1144 | Age | CRN | UC | CCRC | S-PFCRC SD | V-PFCRC SA | V-PFCRC SD | Blood |
| Age | Correlation | -0.15 | -001 | 0.08 | 0.06 | 0.07 | 0.06 | 0.1 | |
| p | < .001 | 0.724 | 0.009 | 0.033 | 0.027 | 0.03 | < .001 | ||
| CRN (g/L) | Correlation | -0.15 | 0.5 | -0.09 | 0.03 | 0.01 | 0.02 | -0.03 | |
| p | < .001 | < .001 | 0.003 | 0.374 | 0.706 | 0.449 | 0.373 | ||
| Blood | Correlation | 0.1 | -0.03 | 0.67 | 0.81 | 0.81 | 0.81 | 0.81 | |
| p | < .001 | 0.373 | < .001 | < .001 | < .001 | < .001 | < .001 | ||
| Female | n = 582 | Age | CRN | UC | CCRC | S-PFCRC SD | V-PFCRC SA | V-PFCRC SD | Blood |
| Age | Correlation | -0.13 | -0.02 | 0.08 | 0.06 | 0.06 | 0.06 | 0.1 | |
| p | 0.002 | 0.662 | 0.064 | 0.156 | 0.125 | 0.152 | 0.013 | ||
| CRN (g/L) | Correlation | -0.13 | 0.52 | -0.16 | 0 | -0.05 | -0.02 | -0.12 | |
| p | 0.002 | < .001 | < .001 | 0.954 | 0.271 | 0.65 | 0.003 | ||
| Blood | Correlation | 0.1 | -0.12 |
0.58 < .001 |
0.79 | 0.77 | 0.78 | 0.78 | |
| p | 0.013 | 0.003 | < .001 | < .001 | < .001 | < .001 | |||
| Male | n = 562 | Age | CRN | UC | CCRC | S-PFCRC SD | V-PFCRC SA | V-PFCRC SD | Blood |
| Age | Correlation | -0.19 | -0.02 | 0.07 | 0.06 | 0.06 | 0.06 | 0.1 | |
| p | < .001 | 0.71 | 0.093 | 0.147 | 0.155 | 0.128 | 0.022 | ||
| CRN (g/L) | Correlation | -0.19 | 0.44 | -0.05 | 0.01 | 0.03 | 0.02 | -0.02 | |
| p | < .001 | < .001 | 0.273 | 0.764 | 0.463 | 0.701 | 0.598 | ||
| Blood | Correlation | 0.1 | -0.02 |
0.73 < .001 |
0.84 | 0.84 | 0.83 | 0.83 | |
| p | 0.022 | 0.598 | < .001 | < .001 | < .001 | < .001 | |||
Figure 2 shows that the V-PFCRC age-sex dynamics align more closely with blood arsenic levels than the CCRC.
Differential effects of aging on arsenic in blood and urine
As shown in the previous section, mean arsenic concentrations in blood and urine increase with age in both sexes, though the kinetics vary depending on sex, test medium, and result mode. Figure 3 illustrates the relative age changes of three older groups compared to the youngest group (21–35 years). Differentiation according to quartile limits allows for investigating the influence of arsenic exposure on age-related changes in arsenic levels. In blood, the oldest age group shows the most significant increase in arsenic concentrations for both sexes and across all quartiles compared to the younger reference group. This increase in older age is higher than the blood arsenic increases observed in the two middle-aged groups and proportionally more substantial than the corresponding increase in all three urine modes of the oldest group.
Fig. 3.
Comparison of Age Effects on Arsenic Concentrations in Blood and Urine. Percentage deviations of medians and quartile (Q1 and Q3) limits of the three older age groups of Set 2 (36–50,51–65, and 66–80) from the youngest age group (21–35 years) are plotted for blood and urine in UC, CCRC, and V-PFCRC mode
Age-related changes in uncorrected urine in both sexes show the most significant discrepancies from blood. Arsenic in CCRC and V-PFCRC mode exhibit comparatively more robust agreement with blood values across all quartiles except for the female Q1. The alignment between urine and blood age kinetics, being generally more robust in men, improves in both sexes from quartile 1 through the median to quartile 3. Women show consistent age-related increases or parity in quartile 1 across all groups. In contrast, the median and Q3 values of the two middle-aged groups even show a reduction in arsenic levels compared to the youngest group. This suggests that gender differences in arsenic metabolism and excretion may be more pronounced at higher arsenic exposures. These findings highlight the complex interplay between age, sex, and arsenic metabolism, underscoring the importance of considering these factors in assessing arsenic exposure.
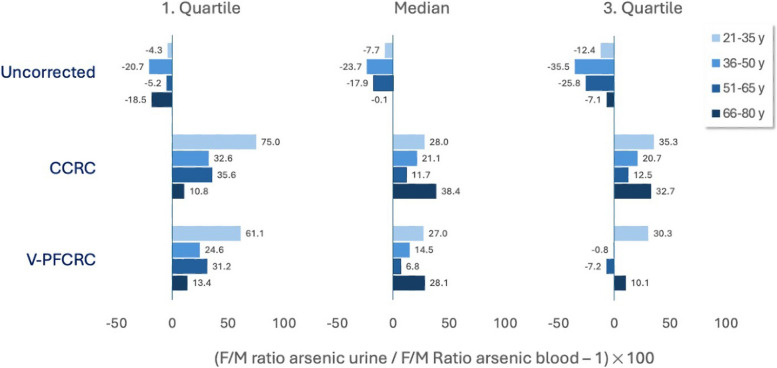
Age-specific sex ratios of quartile limits 1–3
To investigate the extent to which the sex ratios in the individual urine result modes correspond to the blood ratios, the sex ratios of arsenic quartile limits 1–3 for blood and the three urine modes of Set 2 (UC, CCRC, and V-PFCRC SD) were calculated and compared. Figure 4 illustrates the higher arsenic levels in males across most age groups and parameters represented by Female/Male ratios predominantly taking values < 1. Significant exceptions are observed in the group of 21–35-year-olds, where female CCRC and V-PFCRC urine values for medians and quartile 3 are significantly higher than those of males, and sex disparities in quartile 1 are broadly neutralized. The robustness of this inversion of sex ratios is supported by its consistent presence in the 20 to 30-year-old group (but not in the 31–40-year-olds) of the more extensive Set 1, as illustrated in Fig. 1 and the detailed group statistics provided in the Supplementary Excel File. These urine findings correspond to the narrow female dominance in the median, gender neutrality in quartile 3, and converse male dominance in quartile 1 of blood arsenic in this age group. This suggests a gender paradoxical handling of arsenic limited to younger age groups and more pronounced in medium to higher arsenic exposures. Since hormonal differences are most apparent in this age group, a potential connection with endocrine factors is evident.
Fig. 4.
Sex-Ratios of Quartile Limits for CRN, Blood- and Urine Arsenic
Figure 5 illustrates the percentage deviations of urinary from blood sex ratios of arsenic in Quartiles 1–3. The most minor deviations between blood and urine are found in uncorrected urine, while the most considerable deviations occur in CCRC. Compared to CCRC, the analysis indicates that V-PFCRC provides a better match for all quartiles and age groups, except for the 1st quartile in the 66–80 age group, where CCRC has a slight advantage. The most pronounced deviations are seen in the youngest group's 1st quartile of CCRC and V-PFCRC, where urinary female/male ratios are significantly higher than those in blood. This suggests that young women may excrete arsenic more effectively in urine than men, possibly due to their greater efficiency of arsenic methylation related to estrogen.
Fig. 5.
Percentual deviations of urinary from blood sex ratios of Arsenic Quartiles 1–3. The Female/Male ratios of arsenic quartiles 1–3 for urine in UC, CCRC, and V-PFCRC mode were divided by those for blood. The percentual deviation was calculated as shown in the abscissa labeling. Values less than 0 indicate a higher female/male blood ratio than urine and vice versa. The smaller the deviation from zero, the higher the agreement between blood and urine
It is important to note that while these ratios can indicate the reliability of the dilution adjustment method, differences between blood and urine measurements may not necessarily stem from errors in the measurement process. Instead, they may reflect genuine physiological differences in how males and females metabolize and process arsenic. Potential spurious influences can blur these physiological differences and create a misleadingly better match between blood and urine. This is particularly relevant for uncorrected urine, which generally shows good agreement with blood/sex ratios. This agreement, however, is strongly affected by the gender discrepancies of urinary dilution, artificially widening the gap between female and male arsenic in uncorrected urine and spuriously narrowing it in CCRC mode, as shown in Fig. 4. This counteracts any physiological deviations in sex ratios due to more effective female urinary arsenic methylation and excretion. As a result, aligning uncorrected urine with blood may be achieved without accurately reflecting the true efficacy of dilution adjustment.
Effects of age and sex on V-PFCRC equations
To explore the effects of age and sex on the normalization equation, samples were categorized into three age groups. For each group, the log relationship between V-PFCRC normalized arsenic and exponent b was calculated separately for both sexes.
Figure 6 shows the most pronounced differences in curve characteristics observed in the 18–45 age group. Younger men exhibit positive slopes in the log-linear curves, with increasing exponents b for higher arsenic exposures. At the same time, women show substantially negative slopes, with exponents b declining as arsenic levels rise.
In the middle age group, male b-values experience a significant drop and a flattening of the logarithmic component, marked by a now minimal, negative slope c. In contrast, the female curves for the younger and middle-aged groups differ only slightly, resulting in a broad alignment of the curves for both sexes in middle age.
At older ages, the female curve level decreases while the male curve level rises, both entailing a renewed age difference. However, this difference is much less noticeable than in the youngest group. In addition, the curves for both sexes now run mostly parallel due to similar negative slopes, as illustrated in Fig. 6b.
Validation of V-PFCRC by Age- and Sex-Stratified Power Functional Regression Analysis
To evaluate the relevance of apparent sex- and age-related differences in V-PFCRC functions for the validity of uniform normalization across various sex- and age-specific subgroups, samples from Set 1 were subdivided into six sex-aggregated age groups and comprehensive sex-aggregated and sex-differentiated cross-age groups. The residual bias between CRN and arsenic was determined for each group using power-functional regression analysis. Arsenic levels in all groups were standardized using the CCRC, V-PFCRC, and S-PFCRC methods outlined in the Methodology section, with identical coefficients and exponents b as detailed in Table 3.
Figure 7 summarizes the slopes and coefficient of determination (R2) for each group's five urinary result modes. In all subgroups, residual dependence was highest in uncorrected urine, followed by CCRC. All three power-functional result modes performed substantially better than the first two, with only minor differences in both slopes and R2 among them. This indicates that applying the sex-differentiated CRN-asymmetry compensated V-PFCRC SD formulas, determined for sexes between 14 and 82 years [3], has also significantly mitigated the systemic dilution adjustment error (SDAE) in all studied subgroups.
Fig. 7.
Sex- and age-differentiated efficacy of standardization determined by residual dependence of Arsenic on CRN in various age and sex groups of Set 1. Residual dependence was analyzed based on slopes and R2 of power-functional regression analysis of each subset
Similar power-functional regression analyses were performed for the association of the five urinary arsenic modes with blood arsenic in Set 2. In all subgroups of Fig. 8, the correlation between blood and urinary arsenic improves with CRN correction, whether conventional, simple-, or variable power functional. Following the correlation results in entire data sets of previous work [3], also the subsets analyzed here display only minor differences in R2 values across the various CRN corrections and vary between individual subgroups. Like the earlier study, this age-stratified analysis shows a lead for the sex-differentiated V-PFCRC SD mode. The optimized alignment of quartile 3 blood-urine sex ratios shown in Fig. 5, along with the promising Spearman correlation results of previous work on trimmed datasets excluding values below the median, demonstrates the significant advantages of V-PFCRC SD over other dilution adjustments of urinary arsenic. These advantages become more apparent at higher and more reliable blood levels since lower blood arsenic results are compromised by weak resolution [3].
Fig. 8.
Sex- and age-differentiated associations between blood and urine arsenic in Set 2 subgroups. Dependence was analyzed based on R2 of power-functional regression analysis of each subset
Discussion and conclusion
Sex- and age-related differences in arsenic excretion and V-PFCRC formulas
The results of this study strongly suggest that V-PFCRC represents a significant advancement in accurately correcting urinary biomarkers for dilution effects. The method addresses non-linear interactions and biases associated with differential diuresis dependencies through variable power-functional adjustments. The implications of the novel V-PFCRC method extend to various medical and research applications, including the diagnosis and monitoring of diseases, environmental exposure assessments, and forensic investigations. The improved accuracy of urinary biomarker correction can lead to better patient outcomes and more informed public health decisions.
Apart from the significant biases addressed by V-PFCRC, various factors affecting urinary creatinine and arsenic still need to be considered in assessing exposure, even after adjusting for dilution by V-PFCRC. The most critical interconnected factors are lean muscle mass, sex, and age [50, 118, 119]. While no direct data was available for lean muscle mass, this study systematically investigates the complex interplay of physiological and biochemical factors influencing urinary arsenic levels, considering age and sex as critical variables. Using an extensive cohort of urinary total weight arsenic samples (Set 1) and a smaller cohort of total weight EDTA blood samples analyzed in parallel (Set 2), the effects of age and sex on arsenic levels across various traditional and power-functional dilution adjustment modes were compared.
The findings reveal significant differences in arsenic excretion patterns in all investigated urinary result modes, blood levels, and V-PFCRC formulas related to age and sex. Estimating the need for further adjustments to these formulas or reference ranges will depend on a deeper understanding of the underlying causes of these differences and their epidemiological significance. Variations in urinary arsenic due to age and sex may result from a range of interconnected factors, summarized in Supplements 1–7. A crucial question is whether the observed sex- and age-related variations in urinary arsenic reflect actual changes in internal or external exposure or if they are driven by age- and sex-related factors affecting the metabolism of arsenic and CRN, fluid regulation, or urine production. After summarizing the key findings of this study, the following section will delve into their intricate biochemical and physiological underpinnings, providing a foundation for further refinement of the V-PFCRC method.
Summary of Key Findings
Impact of age and sex on creatinine levels
In agreement with existing literature, both sexes exhibited an age-dependent rise in creatinine (CRN) levels, peaking in early adulthood and then continuously declining [50]. Males generally maintained higher CRN levels than females across most age groups, aligning with the expected physiological differences influenced by muscle mass and hormonal factors [50, 51].
Impact of age and sex on urine arsenic levels
Increased urinary arsenic levels were observed with age for both sexes, though the rise in uncorrected arsenic values was more pronounced in males. While CCRC diminished or even reversed this sex difference, it is still noticeable in S-PFCRC and V-PFCRC results and blood arsenic.
Comparison of urinary and blood arsenic levels
EDTA blood arsenic levels were consistently lower than urinary arsenic levels across all ages, and blood arsenic exhibits marked age increases, especially in males. Age increases in arsenic were more pronounced in blood than in urine, suggesting differential metabolic and excretion dynamics that require further exploration. Sex-differentiated V-PFCRC-SD arsenic concentrations and sex-age dynamics showed more substantial alignment with blood arsenic than conventional CCRC, emphasizing the robustness of V-PFCRC in reflecting accurate arsenic exposure.
Age and sex-specific V-PFCRC equations
Age and sex significantly influenced the log-linear relationships between normalized arsenic and the exponent b, being fundamental to corrective V-PFCRC equations. Younger males exhibited increasing exponents with higher arsenic exposures, while females showed declining exponents. Due to a substantial drop in the male curve level, these differences became less pronounced in middle-aged groups, where both sexes showed more aligned curves. Sex differences re-emerged to a lesser extent in older age groups.
Further validation of V-PFCRC
The uniform application of the sex-differentiated V-PFCRC formulas, determined initially for a wide age band from 14 to 82 years [3], also consistently reduced the residual dependence of arsenic levels on CRN across all age and sex subgroups, demonstrating superior standardization efficacy than conventional correction methods even without further adjustment to sex and age.
Regression analyses of urinary arsenic modes with blood arsenic provided additional evidence of V-PFCRC presenting the most accurate reflection of actual arsenic exposure, supporting its use in diverse demographic settings.
In summary, S-PFCRC and V-PFCRC significantly reduce the substantial CRN bias found in uncorrected urine and the considerable residual CRN bias in CCRC urine. This is demonstrated in Tables 4 and 5 for sets 1 and 2 by the insignificantly low Spearman correlations between S-PFCRC- or V-PFCRC arsenic and CRN).
Although the age factor considerably impacts corrective V-PFCRC formulas, it only has a minor influence on bias removal by V-PFCRC, as clearly shown in the validation results of Fig. 7. Broad bias reduction is a universal effect of V-PFCRC that applies to each age group. The differences among age groups are minimal compared to the significant improvements in CRN-bias removal by V-PFCRC from UC or CCRC arsenic.
Physiological, hormonal, and toxicokinetic considerations
Sex differences in urine and blood
The general male increases in urinary concentration in this study indicate a near-synchronous male dominance of CRN and uncorrected arsenic, suggesting extensive commonalities between these two analytes concerning the hydration-dependency of their renal excretory mechanisms. The generally higher male urinary concentration is primarily due to the more substantial impacts of the Renin–Angiotensin–Aldosterone System and Vasopressin (AVP), along with the weaker influence of AVP-attenuating renal prostaglandins and NT-proBNP in men, as summarized in Supplements 1 and 2. Additionally, joint tubular transporters, such as MATE-1, may further synchronize the excretion of CRN and arsenic [10, 14–16, 120].
The sex difference in uncorrected urinary arsenic, however, does not seem to be solely caused by general sex differences in urinary dilution, as the analysis of Set 2 revealed a significant age-related increase in male dominance of blood arsenic levels. While CCRC adjustments neutralized the sex difference in arsenic to insignificance in Sets 1 and 2,
V-PFCRC in both sets maintained a partly significant male bias, aligning better with the concurrent higher blood arsenic levels in males in Set 2. The lower levels of arsenic found in the blood and urine of females in the older three age groups of Set 2 may indicate reduced exposure to arsenic, possibly due to consuming fewer arsenic-containing foods or absorbing less through the intestines. Alternatively, arsenic exposure and daily weight-adjusted net excretion through urine might be less gender-dissimilar or even female-dominant, with lower urinary arsenic levels in females resulting from their higher daily urine per weight production [7, 9, 121].
The gender inverted higher female arsenic medians and upper quartiles in blood and urine among 21–35-year-olds in Set 2 (Fig. 4) and 21–30-year-olds in Set 1 (Supplementary Excel File), suggest an influence of sex hormones, which are most abundant in this age group. By contrast, lower arsenic values (quartile 1) do not show this inversion, implying that the effect might be limited to moderate and higher arsenic exposure rather than background levels. As explained in the introduction (Sex Differences in Renal Arsenic Excretion), estrogens inhibit the reabsorption of inorganic arsenate in the proximal tubule by downregulating the Na-iPT-II transporter. Additionally, estrogens promote the methylation of inorganic arsenic to DMA, enhancing its renal excretion [34, 35]. The enhanced methylation in females facilitates the clearance of arsenic from the liver and other organs into the bloodstream, thereby enhancing the renal excretion of total weight urinary arsenic (TWuAs) [122, 123]. Thus, arsenic tissue storage in younger women is expected to be lower than in men, with blood levels comparable or even slightly elevated. With estrogen declining during menopause, these effects weaken, resulting in a male bias in blood and urine arsenic levels in middle and old age, as shown in Fig. 4. In this context, the less pronounced late-life urinary arsenic flush of older women in Figs. 1 and 2 may reflect a smaller female total body burden due to the mentioned methylation advantages, especially at younger, more estrogenic ages.
Adopting sex-differentiate V-PFCRC SD formulas improves dilution adjustments compared to sex-aggregated V-PFCRC SA. These advantages are substantiated by the significant age differences of V-PFCRC formulas illustrated in Fig. 6, better alignment of sex ratios of urinary to blood arsenic (Figs. 1 and 2), and the closer agreement to blood in sex-differentiated V-PFCRC SD than sex-aggregated V-PFCRC SA results in previous research [3].
Influence of age
Variations in arsenic levels in urine and blood
In both data sets and across sexes, a strong positive correlation between CRN and uncorrected arsenic is evident, as shown in Tables 4 and 5). However, age appears to affect these parameters in opposing ways, as illustrated in Fig. 1. CRN levels decrease steadily from peak levels in young adulthood, whereas urinary arsenic in Set 1 exhibits a consistent upward trend with age across all result modes and sexes. Set 2 confirms this trend in males, while females show a late-life rise preceded by declines in urinary arsenic. Due to the smaller sample size of Set 2, these observations might be less representative than those in Set 1. Notably, in both sets the age increase of urinary and blood arsenic is consistently more pronounced in men (Figs. 1 and 2). This might be due to higher retention of arsenic during younger years and subsequent increased internal exposure from decaying storage tissues in older age.
The systemic computational error from dividing by smaller CRN denominators due to age contributes to the apparent age increments in the CRN corrected results. However, this appears to be only one of the factors since these increases are also evident in uncorrected urine and blood. A plausible explanation for elevated blood and urine arsenic in older adults could be higher external and internal exposure. Age-related bone degradation, a long-term storage site for arsenic, may lead to more arsenic being released into circulation [115, 124–126], while increased intestinal permeability in older adults due to factors such as dysbiosis, medication, and nutrient deficiencies may raise the absorption rate of external arsenic [127].
As depicted in Fig. 3, blood exhibits a markedly more pronounced increase of arsenic concentrations with age than urine. This discrepancy is particularly notable in the oldest age groups (66–80). Alongside the assumed higher internal and external exposure, progressive renal insufficiency in older adults will limit the transfer of arsenic from blood to urine, thereby accentuating age-increases in blood arsenic relative to urine [4, 124–127].
Also, V-PFCRC formulas significantly vary with age (Fig. 6), with the unusually high curves of young men being particularly noteworthy. This suggests that the V-PFCRC method could be enhanced by incorporating age-stratified correction formulas. Generating and implementing these formulas would be straightforward, provided sufficiently large data sets are available. While adopting age-stratified V-PFCRC formulas could yield even more accurate exposure estimates, especially in intra- and interindividual comparisons, bias in larger cohorts is not expected to be reduced significantly. Despite the substantial age deviations of V-PFCRC formulas shown in Fig. 6, the use of cross-age sex-differentiated V-PFCRC formulas generated from data on 14–82-year-olds still substantially mitigated residual biases across the various age groups tested separately (Fig. 7).
Role of common tubular transport for opposing age trends of CRN and arsenic
The inverse age-dependent trends of CRN and urinary arsenic may appear contradictory to a joint transport mechanism initially, but they may support it [4]. As age advances, lean muscle mass and metabolic turnover decline, reducing the production of nitrogenous waste products like CRN, which are excreted by the kidneys. This reduction in CRN excretion may lead to enhanced tubular secretion of arsenic in middle-aged and older individuals. This is because common tubular transporters, such as MATE1, may become more available for this purpose, provided the glomerular filtration rate is not significantly impaired and the tubular excretion of creatinine is not overly strained. The increased methylation capacity with age may further enhance the excretion of total weight arsenic, resulting in higher proportions of more soluble and easily excreted arsenic species [34, 88, 128, 129]. Increased methylation at higher ages could also be biochemically linked to the declining biosynthesis of creatine and phosphatidylcholine attributable to lower lean muscle mass. Together, these two compounds require about 80% of the available SAMe, the essential methyl donor in arsenic metabolism [4].
Variation of Urinary Species Proportions with Age
Previous studies have reported significant age-related increases in total arsenic, arsenobetaine, and, to a lesser extent, MMA and DMA, with tendential reductions in inorganic arsenic [34, 39, 55, 88]. In this study, only total arsenic was measured. However, based on previous findings, it is expected that methylated arsenic components, more abundant in urine, are the main contributors to the age-related rise in arsenic [39, 42, 43, 88]. A mechanistic link could be the age-increased baseline actions of Arginine Vasopressin (AVP) on the V1-Receptor and the Renin–Angiotensin–Aldosterone-System, both of which inhibit m-KL and enhance tubular reabsorption of PO4 −and arsenate [130–133]. Additionally, both hormonal factors limit renal plasma flow and, to a lesser degree, glomerular filtration rate, possibly contributing to increased urinary total weight arsenic in older adults by the mechanisms proposed in V-PFCRC formulas related to age and sex.
Need for further age-adjustment in assessment of arsenic exposure
Age proved to be a significant confounding factor for urinary arsenic, independent of the dilution adjustment method adopted. Unlike the dilution-based differences between sexes, which sex-differentiated V-PFCRC formulas can better adjust, these methods do not compensate for the impact of age.
The epidemiological parameters, including total body burden or exposure periods represented by urine concentrations of metals and other biomarkers, can vary considerably with age [115]. While in older individuals, blood and urine levels may broadly reflect an accumulated metal body burden over many years due to redistribution from degraded bone, the levels of the identical biomarker in younger individuals might indicate rather acute exposure fluctuations [115, 124–126]. An interesting example is the age-specific interpretation of urinary cadmium levels. In healthy, younger kidneys, urinary cadmium reflects the total body cadmium burden due to the constant renal cumulation and excretion proportional to renal loads. Impaired kidneys yet excrete higher amounts as cadmium passes from deteriorating tubular cells into the urine. Similar mechanisms may also apply to arsenic stored in bones and kidneys, explaining its age-related increase [115].
Given these shifts, accurately assessing acute arsenic exposure necessitates age-related adjustments. This applies to both inter-/intra-individual comparisons and cohort studies with asymmetric age distributions. Since the age increases in urine and blood arsenic reflect real exposure differences, adjustments are essential for all dilution modes, regardless of the evident age variability in V-PFCRC formulas. This can be achieved by integrating age into multilinear covariate models, using age-adjusted reference ranges, or normalizing values to a specific age [1, 34, 44]. By contrast, adopting age-adjusted V-PFCRC formulas will not adjust for age variation in internal or external exposure.
Variations of V-PFCRC formulas related to age and sex
The sex differences in V-PFCRC standardization formulas illustrated in Fig. 6 are most pronounced in young individuals. They diminish in middle and older age groups, primarily due to changes in male characteristics, whereas females display rather age-constant V-PFCRC formulas. These variations result from the compound influences of multiple sex—and age-specific renal fluid regulatory factors, as detailed in Supplements 1–7.
Pivotal role of osmoregulation in age- and sex-variations of V-PFCRC functions
Evidence in the literature suggests that differences in osmoregulation based on age and sex may be linked to the abovementioned variations. Previous studies have shown that the age-sex dynamics of urinary osmolality exhibit characteristics similar to the relations between V-PFCRC arsenic and exponent b, illustrated in Fig. 6 [7]. Young men exhibited a higher estimated urinary osmolality (uOsm) than young women, with these differences diminishing in middle age due to a decrease in male uOsm, as shown in Supplement 14 [7]. This male-dominant difference in uOsm re-emerges in older age matching the reopening gender gap of V-PFCRC curves in that age group.The hypothesized sex and age modifications in osmolality set points and their primary actuators, RAAS (ATII) and AVP (Supplements 1 and 2), likely result in similar alterations in the relationship between V-PFCRC arsenic and exponent b. This concept aligns with mitigating age-related arsenic increases observed following intravenous fluid administration, suppressing AVP/ATII before sample collection (data to be published).
There are parallels between the slopes and exponents b in Fig. 6and the age- and sex-variable renal actions of ATII/AVP summarized in Supplements 1 and 2. The actions of these hormones are generally more pronounced in males, as they are stimulated by androgens and attenuated by estrogen [7, 8, 10, 17–29]. This corresponds to the higher sex discrepancy in b-values at younger ages. The decline in sex hormone activity towards middle age, more significant in menopausal women, results in gender alignment of AVP/RAAS activities and uOsm. In older age groups, a minor inconsistency between b-values and uOsm for males consists of a tendential drop in uOsm, which does not correspond to the rising trend of b-values.
Conversely, there is good alignment in women as both uOsm and the exponents decrease. This inconsistency in men could be due to different age group distributions in the studies or intrinsic renal aging mechanisms, which are more pronounced in men and cause fluid homeostasis inelasticity, such as declining renal prostaglandin levels and tubular dysfunction [134–139]. While ATII/AVP activities increase with age, the paradoxical urinary concentrating ability deficit in seniors is likely due to these intrinsic renal causes [135, 140].
In conclusion, the variations in the exponent b specific to age and sex consistently reflect the established baseline renal actions of ATII/AVP. These variations are primarily influenced by sex hormones in young individuals and by renodegenerative changes in elderly individuals. This relationship appears to remain valid despite the age-related decline in renal concentrating capacity.
Mechanistic explanation of ATII/AVP-Mediated effects on the exponent b
As exponent b approaches 1, the mass ratios between arsenic and CRN remain constant across varying hydration/urinary flow rates. Arsenic exponent b-values are typically < 1, indicating reduced arsenic/CRN mass ratios at higher urinary concentrations and enhanced ATII/AVP activities [3]. Therefore, enhanced actions of ATII will either decrease net TWuAs excretion, increase net CRN excretion, or exert any summative effect on both analytes, resulting in relatively higher excretion of CRN than TWuAs in dehydrated states.
Increases in b approximate the exponent to 1, indicating a better balance of arsenic and CRN secretions across the urinary dilution range. Creatinine and most blood arsenic components are freely glomerularly filtered, so their hydration-dependent excretion discrepancy will be predominantly due to their differential tubular handling [141–144]. Any relative restriction of tubular function about glomerular filtration rate might thus align the excretion behaviors of CRN and TWuAs, translating into an approximation of b to 1. Age-related tubular renal damage progresses faster in males than in females [136–139], which could conclusively explain the reemerging male-dominant age gap in exponents b at higher ages, as illustrated in Fig. 6.
Apart from kidney damage, higher baseline ATII/AVP activities in males and at higher ages can limit tubular relative to glomerular function [139, 145, 146]. Angiotensin II (ATII) preferentially constricts efferent arterioles, increasing glomerular filtration pressure but reducing renal and peritubular capillary flow, thus impeding tubular function. Arginine Vasopressin (AVP), primarily involved in water reabsorption in the collecting ducts, also contributes to renal vasoconstriction. Together, these hormones can exacerbate tubular hypoxia and injury, enhancing glomerular at the expense of tubular function [70, 147, 148].
The effects of ATII and AVP on CRN excretion are twofold. In prolonged or excessive ATII/AVP activity, such as during advanced volume loss or dehydration, GFR decreases, leading to increased tubular CRN secretion [111, 146]. Conversely, moderate ATII/AVP activity, which is more common, primarily increases the glomerular capillary hydrostatic pressure (GCHP) and enhances GFR, potentially reducing the tubular contribution to urinary CRN [70, 149, 150]. This could explain the generally increased exponent b in men, as their higher baseline ATII/AVP activities may enhance GFR, reduce the relative contribution of the tubules to CRN excretion, and thus counterbalance discrepancies in the tubular handling of arsenic and CRN.
The tubular excretion of arsenic species also depends on urinary flow rate and corresponding ATII/AVP activity yet differs from CRN in this regard. Although there is a significant knowledge gap concerning the tubular handling of different arsenic species, it is known that at least the reabsorption of inorganic arsenate (iAsV) is favored by slower urinary flow [4, 151]. More dehydrated states, associated with higher ATII and AVP activities, result in higher tubular reabsorption and lower net secretion of arsenate.
In such states, the increased excretion of creatinine (CRN) leads to a shift in the mass ratio between CRN and arsenic in favor of CRN. This shift in more dehydrated states with elevated ATII/AVP activities can plausibly explain why the exponents of arsenic generally have values smaller than 1. Contradicting this explanation is the association of higher inorganic arsenate (iAsV) reabsorption with higher methylation and total weight arsenic, which is the relevant parameter for this study [34, 38, 39, 55]. The methylation process of arsenate and its increasing effect on TWuAs may yet lag the reabsorption of arsenate and, therefore, not immediately affect urinary TWuAs.
The restraining effects of prolonged ATII/AVP elevation on membrane protein Klotho (m-KL) expression further complicate these mechanisms. Lower m-KL levels increase arsenate (iAsV) reabsorption and favor its methylation, leading to higher proportions of easily excreted methylated TWuAs components and increased TWuAs, as outlined in the introduction. There is empirical evidence of opposing ATII/AVP effects on net tubular excretion of iAs (inhibitory) and TWuAs (stimulatory), which can explain lower %iAs and higher TWuAs in seniors and gender-identical TWuAs excretion in males despite female methylation advantages [4, 34]. Males could thus compensate for methylation drawbacks with enhanced TWuAs excretion-promoting effects of ATII/AVP.
In summary, prolonged states of lower m-KL expression associated with higher baseline ATII/AVP activities, such as in males and older age, generally seem to increase exponents b and thus reduce the systemic dilution correction error.
The increasing exponents b in young men with rising arsenic levels, contrasted by opposite trends in all other groups, require additional explanation. This might be due to the high CRN production in young men, which may cause faster saturations of tubular CRN transporters in dehydrated states, shifting TWuAs/CRN mass ratios in dehydration towards arsenic, entailing higher exponents b. Higher arsenic exposure could intensify this effect through additional competition and impairment of common tubular transporters, explaining the positive slope of coefficient c as detailed in the introductory section 'Variable Power Functional Creatinine Correction (V-PFCRC)' and in preliminary work [3].
Additionally, the sex-differentiated methylation capacity of arsenic could play a role here. Interestingly, at lower arsenic levels, which in the present study population generally can be assumed to have higher proportions of inorganic arsenic, the exponent b of young women is higher than that of men and much closer to 1. The female methylation advantage could be noticeable here, as higher proportions of methylated arsenic can be better excreted in urine and behave more similarly to CRN in the tubule. This effect diminishes at higher arsenic levels, where mostly organic loads with predominantly methylated arsenic are present. At higher arsenic exposures, the advantage of female tubular CRN secretion over arsenobetaine in the dehydrated state may become more significant, causing a relatively steep negative slope (coefficient c) in young women.
These theoretical considerations rationalize sex-age-specific differences in the corrective formulas (Fig. 6). Yet further research is needed to validate these findings and to elucidate the tubular handling of the individual arsenic species [4, 151].
Evaluation of V-PFCRC
The role of V-PFCRC in compensation of confounders of spot-urinary results
When estimating the benefits and drawbacks of V-PFCRC, it is essential to understand the types of errors it compensates for. As outlined previously, three categories of biases essentially limit the validity of spot urinary biomarker results: [3]
-
Factors influencing the production, metabolism, and renal excretion of either analyte (here arsenic) or dilution corrector (here CRN) unidirectionally:
These factors tend to either increase or decrease spot urinary results. In addition to sex and age, which are the only essential determinants examined in this work, other important variables influence urinary CRN and arsenic. In the case of CRN, these are primarily related to lean body mass and muscle metabolism. Still, renal function, pregnancy, diet, exercise, and pharmacological interventions play a significant role here [3, 152]. Concerning arsenic, kidney function and the methylation status of the metalloid are essential confounders. On the one hand, arsenic methylation depends on the ingested chemical forms (i.e., methylated, more easily renally eliminated arsenobetaine or more toxic unmethylated inorganic arsenic) and, on the other hand, the individual oxidative methylation capacity, determined by genetic variability and availability of essential cofactors like the methyl donor SAME and reduced glutathione [3].
These factors tend to shift urine results in one direction, either increasing or decreasing them, and neither V-PFCRC nor S-PFCRC can compensate for these shifts. In more extreme cases, particularly when multiple unidirectional factors with the same effect combine, a substantial distortion may result in misinterpretation of exposure [152]. Partial compensation is possible through adjusted reference ranges or within multiple linear regression or covariate models, assuming accurate linearity and independence of the various confounders. Due to the large number and variety of influencing factors in this category, they generally tend to moderate each other.
Unlike this first category of errors, V-PFCRC effectively compensates for the following two significant error categories: [3]
-
Confounding and biochemical interactions:
These complex interactions between analytes and CRN can cause unpredictable fluctuations in results. An example is the relationship between CRN and arsenic, where biases arise from their shared dependence on S-adenosyl-methionine (SAME) [4, 60].
-
Bidirectional shifts due to distinct renal handling of CRN and analyte:
This error arises from the incorrect assumption of constant mass ratios between analytes and CRN across different hydration levels, leading to divergences in opposite directions at extreme diuresis levels [2, 3]. Unaccounted discrepant diuresis dependencies of renal handling of CRN and analytes contribute to this error category.
Adopting sex-differentiated V-PFCRC formulas will effectively compensate for the systemic error categories II and III. Alone by more adequately assuming power-functional rather than linear relationships between uncorrected arsenic and CRN, a significant source of error in dilution adjustment in more extensive cohort studies could be neutralized despite remaining biases of the first error category unaccounted for by power-functional adjustments. In previous research, compared to uncorrected and CCRC values, even simple power-functional corrections (S-PFCRC) have significantly improved correlations of standardized total urinary arsenic (TuAs) with drinking water exposures [2].
As the present study demonstrates, age adjustments in addition to V-PFCRC in reference range modifications or covariate models are sensible due to the age-related increase in internal exposure. They will further enhance the assessment of external arsenic exposure. By contrast, the expected benefit of age-adjusting V-PFCRC formulas appears limited. Even though they are beneficial in intra- and interindividual comparisons, the apparent age and gender differences in the V-PFCRC formulas (Fig. 6) appear to only marginally compromise the standardization quality within the individual subgroups, as shown in Figs. 7 and 8. This corroborates the significantly improved precision and comparability of V-PFCRC results across all age groups and sexes compared to uncorrected and CCRC arsenic. Also, the improvements of sex-differentiated V-PFCRC SD over alternative power functional corrections tested in this study (sex aggregated V-PFCRC SA, sex-differentiated simple S-PFCRC) were relatively narrow.
While this study did not have additional data on the potentially influential unidirectional confounders of the first category beyond sex and age, future research may find it challenging to investigate their impact on V-PFCRC formulas.
Practical applications of the V-PFCRC method
As the V-PFCRC method compensates for the non-linear biases in hydration adjustment by normalizing levels to the expected value at 1g/L CRN, it delivers more reliable raw data in individual measurements. V-PFCRC advantages are expected to be most evident in intra- and inter-individual comparisons on fewer participants. In more extensive cohort studies, where dilution errors will average out with increasing sample sizes, the advantages of V-PFCRC may be less apparent. However, even in such studies, achieving the most accurate representation of exposure in urine is beneficial, as it reduces the number of samples needed to sufficiently mitigate dilution correction errors, leading to more conclusive results. The following scenarios may serve as examples for a better assessment of the method:
-
Reduction of intra-individual hydration-dependent fluctuations in results:
Consider a scenario from Set 1, illustrated in Fig. 9: A person consumes herring, reaching the 55th percentile of arsenic exposure in their reference collective. The next day, after high fluid intake, their urinary CRN drops to 0.4 g/L, and uncorrected arsenic reads 7 µg/L. With reduced fluid intake and a CRN of 2.7 g/L, uncorrected arsenic would rise to 29 µg/L—a 4.1 × increase. Conventional CRN adjustments (CCRC) overcorrect, yielding 17 µg/g CRN at 0.4 g/L and 10 µg/g CRN at 2.7 g/L, with the added limitation of WHO's valid CRN range of 0.3–3 g/L. In contrast, V-PFCRC normalizes the value to 13.5 µg/1 g CRN across all CRN levels, ensuring consistent intra-individual results. The benefits of the V-PFCRC method are particularly valuable in clinical and forensic exposure studies, significantly enhancing reliability in fields such as toxin exposure, detoxification efficacy, nephrology, pharmacology, endocrinology, and nutritional research.
-
Public health study on genetically similar collectives displaying distinct hydration:
Arsenic exposures in village A, facing water scarcity, are compared to arsenic exposure in village B, disposing of an abundant drinking water supply and well-hydrated inhabitants. Average daily arsenic uptakes were about the same in both villages. Uncorrected mean values in village B will be consistently lower, whilst CCRC values will be higher than in village A due to systemic overcorrection. Like in the first example, V-PFCRC will effectively abolish this bias. In the absence of other systematic confounders, the mean urine arsenic values will continue to converge as the number of participants increases.
-
Arsenic exposure study on carnivores and vegans within a village of genetically and metabolically similar inhabitants:
In this scenario, higher CRN intake in carnivores leads to greater CRN excretion, resulting in consistently lower arsenic/CRN ratios than in vegans. This discrepancy causes higher classical corrected (CCRC) arsenic values for vegans with no bias in uncorrected values. The urinary flow rate (UFR) for vegans with 1 g/L CRN is lower than for carnivores, leading also to higher corrected V-PFCRC values across the whole dilution range. While V-PFCRC will reduce non-linear hydration biases, a systemic CRN bias will persist in this example, causing downward shifts in carnivore CCRC and V-PFCRC arsenic values. To address this, a multi-linear model including meat intake as a covariate or alternative correctors like specific gravity or osmolality could be used. Even uncorrected values could be compared if sample sizes were sufficient and there were no systemic differences concerning hydration status.
Fig. 9.
Dilution-dependent result fluctuations of arsenic in uncorrected, conventional, and variable power-functionally corrected result mode
These three examples underscore the V-PFCRC method’s versatility and its specific advantages across various applications. They highlight the importance of carefully selecting the most appropriate urine dilution adjustment method based on the study’s objectives and conditions. Given the regional differences in diet and genetics affecting arsenic detoxification, regional V-PFCRC formulas may be beneficial for clinical and forensic purposes. However, for larger epidemiological studies, the use of general cross-regional,-cultural, and -ethical formulas should generally suffice, as minor variations in the V-PFCRC coefficients generally do not significantly impact residual bias elimination (Figs. 7 and 8).
V-PFCRC and multivariate regression models
While the advantages of V-PFCRC over CCRC and S-PFCRC are evident, how does it compare with more elaborated covariate-adjusted standardizations, which reportedly improved results over classical models? In general, the V-PFCRC method corrects a substantial bias brought about by erroneously assumed linearity in factually non-linear physiological relations. In covariate-adjusted models, this notable misconception is either symptomatically addressed by restricting CRN ranges or ignored within accepted limits [1, 3, 34, 152]. This confers on V-PFCRC the advantage of widely dispensing with CRN range restrictions and associated sample rejects. At the same time, more reliable, dilution-independent standardized values are achieved in intraindividual and, with minor limitations, interindividual comparisons. Fewer sample rejects imply resource savings and the disposal of more valid samples, increasing the power of any epidemiological study involving urinary biomarkers.
Despite the mathematical adjustment of this significant bias by V-PFCRC, residual inaccuracies may result from the unaccounted impact of other biasing factors on CRN and the analyte, primarily age and lean muscle mass (BMI) and, to a lesser extent, comorbidities, ethnicity, exercise, nutrition, and the time of sample collection [1, 34, 152]. The combined spurious influence of these biases on CRN production, secretion, and any CRN correction will average out in comparable cohorts with sufficient subject numbers. Various factors can also influence CRN in opposite directions, thus moderating the summative distorting effect on corrected results, particularly in larger cohorts. The results in this study on arsenic and iodine in the previous study demonstrated better correlations of all conventional and power-functional CRN-corrected arsenic and iodine modes with EDTA blood (arsenic) or serum (iodine) [3]. There is evidence that cumulative biases on CRN production might be less relevant for accuracy than the systemic dilution adjustment error addressed in this paper and previous work [2]. They may still cause considerable misinterpretation in exposure analyses of smaller groups and comparative studies of dissimilar individuals or collectives concerning covariates influencing creatine formation and CRN. In pursuing optimized precision, it can be worthwhile to accommodate these factors additionally.
While a sex-differentiated, age-adjusted variable power-functional correction (V-PFCRC) or a similar adjustment by SG or uOsm is expected to yield sufficiently reliable results for most clinical, forensic, and epidemiological scenarios, thereby reducing sample rejections, it may be worthwhile integrating V-PFCRC into more complex covariate-adjusted models, depending on the study design [1, 34]. Ideally, three sequential standardization steps should be adopted in a more sophisticated correction procedure:
-
I.
Standardization of CRN (SG, or uOsm) for age, BMI, sex, and other factors by multiple regression covariate adjustment.
-
II.
Calculating covariate-CRN adjusted V-PFCRC values for analytes (here, all TWuAs species separately) using V-PFCRC formulas determined with covariate-adjusted CRN.
-
III.
Standardization of the obtained V-PFCRC values for age (e.g., 50 years).
Limitations of this study
Population specificity
This study is based on a cohort from Central Europe, which may limit the generalizability of the findings. Genetic, environmental, and lifestyle differences across populations might influence the V-PFCRC formulas and the effects of age and sex on urinary arsenic excretion. To ensure broader applicability, future studies should aim to validate the V-PFCRC method in diverse populations.
Cross-sectional design
The cross-sectional data in this study covers a period from January 2014 to August 2022 but does not account for potential temporal changes in urinary arsenic excretion. Future longitudinal studies could validate the V-PFCRC method over time and under varying climatic or physiological conditions. However, since V-PFCRC effectively addresses the fundamental biomathematical misperception inherent in classical CRN adjustment, any changes to the corrective formulas due to common temporal variations are expected to be of comparatively minor relevance.
Potential for overfitting
Developing age- and sex-specific adjustment formulas introduce a potential risk of overfitting to the specific dataset used in this study. The large urine sample dataset (Set 1) forming the basis for V-PFCRC formula development, along with the exclusion of extreme values, helped mitigate this concern. The validation process showed improvement in reducing residual bias despite differences in age-specific V-PFCRC formulas. However, overfitting may be more concerning when interpreting blood and urine data from the smaller dataset (Set 2), which showed some deviation from the urine data of Set 1. Independent validation with external datasets will be crucial to confirm the method's robustness and reliability in future research.
Unmeasured confounders
Although this study focuses on age and sex, other potential confounders like diet, environmental arsenic exposure, medical conditions, and genetic predispositions were not explicitly controlled for. These factors might influence urinary CRN and arsenic levels, potentially introducing bias into the V-PFCRC formulas. Future studies could explore the influence of these variables on V-PFCRC adjustment formulas and the mathematical model presented here could serve as a basis for further refinement using machine learning algorithms.
Limited biomarker scope
While the V-PFCRC method was initially developed for arsenic, it has been successfully generalized to other urinary biomarkers, including cesium, molybdenum, strontium, zinc, and iodine [3]. The analysis of an extensive dataset of over 58,000 iodine samples robustly confirmed the log-linear relationship between normalized results and the exponent b in both sexes, validating the mathematical foundation of V-PFCRC. Additionally, V-PFCRC improved blood-urine iodine associations in both sexes. However, further exploration is needed to assess V-PFCRC's applicability to more urinary biomarkers, and the sex-age kinetics of other elements have yet to be systematically studied.
Complexity of the V-PFCRC method
While the V-PFCRC method offers a more accurate yet sophisticated mathematical approach than conventional creatinine correction (CCRC), it does not necessarily add complexity to routine laboratory settings. The main challenges lie in the initial development rather than its practical use. Once the coefficients c and d are established, the V-PFCRC method can seamlessly integrate into existing laboratory software, much like calculating simple ratios in conventional CRN correction. This integration allows for accurate and efficient application without additional operational burdens, making it feasible and beneficial even in resource-limited settings.
Clinical relevance
While the study successfully reduces systemic bias in arsenic measurement, it also demonstrates improved biomarker accuracy and reliability, as highlighted in Evaluation of V-PFCRC. However, the clinical significance of these adjustments across various spot urinary biomarkers requires further exploration. Establishing its impact on clinical decision-making and patient outcomes across multiple biomarkers would strengthen the case for adopting the V-PFCRC method into routine clinical practice.
Conclusions
The V-PFCRC method significantly advances accurately correcting urinary biomarkers for dilution effects. This method effectively addresses non-linear interactions and biases associated with differential diuresis dependencies by incorporating variable power-functional adjustments. In clinical and epidemiological contexts, adopting V-PFCRC can lead to more reliable assessments of urinary biomarkers, ultimately improving the diagnosis, monitoring, and research of various medical conditions and environmental exposures.
The findings confirm the significant improvements of the novel V-PFCRC method over uncorrected and conventionally CRN-corrected urine in rigorous age- and sex-stratified analysis and, in comparison with blood arsenic detected in parallel.
Based on age-sex-stratified formula generation and literature review, sex-age-kinetics of P-VFCRC corrective formulas were shown to align well with urinary osmolality, predominantly determined by the activities of the Renin–Angiotensin–Aldosterone System (RAAS) and Arginine Vasopressin (AVP).
While the novel sex-differentiated V-PFCRC method largely accounted for sex biases, the presented results underscore the potential for further refinement of V-PFCRC formulas according to age, as it proved to significantly affect both arsenic exposure and the most common default dilution correctors, such as CRN, osmolality, and specific gravity [2, 3].
Future research should expand on the physiological and biochemical underpinnings of these differences, mainly focusing on hormonal influences and metabolic pathways. Moreover, validation across broader populations and with additional biomarkers will enhance the accuracy, generalizability, and applicability of the V-PFCRC method.
Supplementary Information
Acknowledgements
Dr. Katrin Huesker, department manager of Special Immunology, IMD Berlin, is thanked for providing anonymized spot urine data.
Abbreviations
- ATII
Angiotensin II
- AVP
Arginine Vasopressin (Adiuretin, ADH)
- CCRC
Conventional Creatinine Correction
- CKD
Chronic Kidney Disease
- CRN
Creatinine
- DMA
Dimethylarsinic acid
- E
Estrogen
- FGF23
Fibroblast Growth Factor 23
- GCHP
Glomerular Capillary Hydrostatic Pressure
- GFR
Glomerular Filtration Rate
- iAs
Inorganic Arsenic (iAsIII + iAsV)
- %iAs
Percentage of iAs in Total weight arsenic
- iAsIII
Trivalent Arsenite
- iAsV
Pentavalent Arsenate
- ICP-MS
Inductively Coupled Plasma Mass Spectrometry
- MATE1
Multidrug and Toxin Extrusion Protein 1
- m-KL
Proteohormone transmembrane Klotho
- MMA
Monomethylarsonic acid
- NaPi-IIa
Sodium-coupled PO4 − Co-Transporters
- NT-proBNP
N-terminal pro B-type Natriuretic Peptide
- oAs
Organic Arsenic (Sum of DMA + MMA, Arsenobetaine, Arseno–sugars, –lipids, and –choline)
- OCT2
Organic Cation Transporter 2
- uOsm
Urinary Osmolality
- P
Progesterone
- PO4−
Phosphate
- RAAS
Renin-Angiotensin-Aldosterone System
- RPF
Renal Plasma Flow
- SAMe
S-Adenosyl-Methionine
- S-PFCRC
Simple Power Functional Creatinine Correction using fixed exponents
- T
Testosterone
- TuAs
Sum of iAs and methylated primary metabolites of iAs (iAs + DMA + MMA)
- TWuAs
Total Weight Urinary Arsenic = all ICP-MS detectable forms of inorganic and organic arsenic (measured in this study)
- AsUC
Uncorrected TWuAs in µg/L
- uAsC
Conventionally Creatinine-corrected TWuAs
- uAsN
Urinary Total Weight Arsenic, normalized to 1 g/L CRN by V-PFCRC SD
- UC
Uncorrected urinary result mode (in µg/L for Arsenic)
- uOsm
Urinary Osmolality
- UFR
Urinary Flow Rate (e.g. ml/min)
- V-PFCRC
Variable Power Functional Creatinine Correction (all forms)
- V-PFCRC SA
Sex-Aggregated Variable Power Functional Creatinine Correction using identical analyte-specific coefficients c and d for both sexes
- V-PFCRC SD
Sex-Differentiated Variable Power Functional Creatinine Correction using sex-specific coefficients c and d
Authors’ contribution
The author was responsible for designing the study, collecting data, reviewing the literature, conducting statistical analysis, and writing the manuscript.
Funding
The study was not funded.
Availability of data and materials
The supplementary Excel file includes the datasets used and/or analyzed during the current study (Supplementary Material 15). Further information is available from the corresponding author upon reasonable request.
Declarations
Ethics approval and consent to participate
The study was exempt from ethical approval by the Ethics Committee of Northwest and Central Switzerland (EKNZ), 4056 Basel, based on retrospective analysis of anonymized data without clinical information other than urinary creatinine/TWuAs, age in years, sex, and exam date. The purpose was methodological quality improvement only. There was no pharmacological or other medical intervention, risk for physical or psychological harm, identifiability, or breach of confidentiality for any subject involved.
Conflict of Interest
The authors declare no competing interests.
Footnotes
The original version of this article was revised: the authors identified paragraph and punctuatuation errors. Also, Fig. 8 has been updated and the coefficient c for Males in Table 3 has been corrected.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
11/28/2024
A Correction to this paper has been published: 10.1186/s12882-024-03834-1
References
- 1.O'Brien KM, Upson K, Cook NR, Weinberg CR. Environmental Chemicals in Urine and Blood: Improving Methods for Creatinine and Lipid Adjustment. Environ Health Perspect. 2016 Feb;124(2):220–7. 10.1289/ehp.1509693. Epub 2015 Jul 24. PMID: 26219104; PMCID: PMC4749084. Covariate-adjusted standardization method available in: http://www.niehs.nih.gov/research/resources/ software/biostatistics/covariate/index.cfm [DOI] [PMC free article] [PubMed]
- 2.Middleton DRS, Watts MJ, Polya DA. A comparative assessment of dilution correction methods for spot urinary analyte concentrations in a UK population exposed to arsenic in drinking water. Environ Int. 2019 Sep; 130: 104721. 10.1016/j.envint.2019.03.069. Epub 2019 Jun 14. PMID: 31207477; PMCID: PMC6686075. [DOI] [PMC free article] [PubMed]
- 3.Carmine TC. Variable Power Functional Dilution Adjustment of Spot Urine. medRxiv [Preprint]. 2024. 10.1101/2023.02.09.23285582.
- 4.Peters BA, Hall MN, Liu X, Neugut YD, Pilsner JR, Levy D, et al. Creatinine, Arsenic Metabolism, and Renal Function in an Arsenic-Exposed Population in Bangladesh. PLoS ONE. 2014;9(12): e113760. 10.1371/journal.pone.0113760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Liu J, Chen H, Miller DS, Saavedra JE, Keefer LK, Johnson DR, Klaassen CD, Waalkes MP. Overexpression of glutathione S-transferase II and multidrug resistance transport proteins is associated with acquired tolerance to inorganic arsenic. Mol Pharmacol. 2001;60(2):302–9. 10.1124/mol.60.2.302. PMID: 11455017. [DOI] [PubMed] [Google Scholar]
- 6.Nishizawa K, Yoda N, Morokado F, Komori H, Nakanishi T, Tamai I. Changes of drug pharmacokinetics mediated by downregulation of kidney organic cation transporters Mate1 and Oct2 in a rat model of hyperuricemia. PLoS ONE. 2019;14(4): e0214862. 10.1371/journal.pone.0214862.PMID:30951542;PMCID:PMC6450621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Perucca J, Bouby N, Valeix P, Bankir L. Sex difference in urine concentration across differing ages, sodium intake, and level of kidney disease. Am J Physiol Regul Integr Comp Physiol. 2007;292(2):R700–5. 10.1152/ajpregu.00500.2006. Epub 2006 Sep 21 PMID: 16990487. [DOI] [PubMed] [Google Scholar]
- 8.Claybaugh JR, Sato AK, Crosswhite LK, Hassell LH. Effects of time of day, gender, and menstrual cycle phase on the human response to a water load. Am J Physiol Regul Integr Comp Physiol. 2000;279(3):R966–73. 10.1152/ajpregu.2000.279.3.R966. PMID: 10956255. [DOI] [PubMed] [Google Scholar]
- 9.Schoen T, Blum J, Paccaud F, Burnier M, Bochud M, Conen D; Swiss Survey on Salt investigators. Factors associated with 24-hour urinary volume: the Swiss salt survey. BMC Nephrol. 2013 Nov 7;14:246. 10.1186/1471-2369-14-246. PMID: 24199987; PMCID: PMC4226251. [DOI] [PMC free article] [PubMed]
- 10.Sabolić I, Asif AR, Budach WE, Wanke C, Bahn A, Burckhardt G. Gender differences in kidney function. Pflugers Arch. 2007;455(3):397–429. 10.1007/s00424-007-0308-1. Epub 2007 Jul 19 PMID: 17638010. [DOI] [PubMed] [Google Scholar]
- 11.Ozdemir S, Sears CG, Harrington JM, Poulsen AH, Buckley J, Howe CJ, James KA, Tjonneland A, Wellenius GA, Raaschou-Nielsen O, Meliker J. Relationship between Urine Creatinine and Urine Osmolality in Spot Samples among Men and Women in the Danish Diet Cancer and Health Cohort. Toxics. 2021;9(11):282. 10.3390/toxics9110282.PMID:34822673;PMCID:PMC8625939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bankir L, Perucca J, Weinberger MH. Ethnic differences in urine concentration: possible relationship to blood pressure. Clin J Am Soc Nephrol. 2007;2(2):304–12. 10.2215/CJN.03401006. Epub 2007 Jan 10 PMID: 17699429. [DOI] [PubMed] [Google Scholar]
- 13.Brosnan JT, da Silva RP, Brosnan ME. The metabolic burden of creatine synthesis. Amino Acids. 2011;40(5):1325–31. 10.1007/s00726-011-0853-y. Epub 2011 Mar 9 PMID: 21387089. [DOI] [PubMed] [Google Scholar]
- 14.Ligen A, Dandan Z, Wolin H, Junxi L, Junling T, Huijuan L, Taishan Z, Fang L. Gonadal hormone regulates the expression MATE1 and OCT2 in type 2 diabetes mice. Endocrine Abstracts (2015) 37 EP376 | 10.1530/endoabs.37.EP376.
- 15.He R, Ai L, Zhang D, Wan L, Zheng T, Yin J, Lu H, Lu J, Lu F, Liu F, Jia W. Different effect of testosterone and oestrogen on urinary excretion of metformin via regulating OCTs and MATEs expression in the kidney of mice. J Cell Mol Med. 2016 Dec;20(12):2309–2317. 10.1111/jcmm.12922. Epub 2016 Jul 29. PMID: 27469532; PMCID: PMC5134372. [DOI] [PMC free article] [PubMed]
- 16.Lepist EI, Zhang X, Hao J, Huang J, Kosaka A, Birkus G, Murray BP, Bannister R, Cihlar T, Huang Y, Ray AS. Contribution of the organic anion transporter OAT2 to the renal active tubular secretion of creatinine and mechanism for serum creatinine elevations caused by cobicistat. Kidney Int. 2014 Aug;86(2):350–7. 10.1038/ki.2014.66. Epub 2014 Mar 19. PMID: 24646860; PMCID: PMC4120670. [DOI] [PMC free article] [PubMed]
- 17.Medina D, Mehay D, Arnold AC. Sex differences in cardiovascular actions of the renin-angiotensin system. Clin Auton Res. 2020 Oct;30(5):393–408. 10.1007/s10286-020-00720-2. Epub 2020 Aug 29. PMID: 32860555; PMCID: PMC7572792. [DOI] [PMC free article] [PubMed]
- 18.Komukai K, Mochizuki S, Yoshimura M. Gender and the renin-angiotensin-aldosterone system. Fundam Clin Pharmacol. 2010;24(6):687–98. 10.1111/j.1472-8206.2010.00854.x. PMID: 20608988. [DOI] [PubMed] [Google Scholar]
- 19.Toering TJ, Gant CM, Visser FW, van der Graaf AM, Laverman GD, Danser AHJ, Faas MM, Navis G, Lely AT. Sex differences in renin-angiotensin-aldosterone system affect extracellular volume in healthy subjects. Am J Physiol Renal Physiol. 2018;314(5):F873–8. 10.1152/ajprenal.00109.2017. Epub 2017 Jun 7 PMID: 28592435. [DOI] [PubMed] [Google Scholar]
- 20.Stachenfeld NS. Sex hormone effects on body fluid regulation. Exerc Sport Sci Rev. 2008;36(3):152–9. 10.1097/JES.0b013e31817be928.PMID:18580296;PMCID:PMC2849969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Stachenfeld NS, Splenser AE, Calzone WL, Taylor MP, Keefe DL. Sex differences in osmotic regulation of AVP and renal sodium handling. J Appl Physiol (1985). 2001 Oct; 91(4):1893–901. 10.1152/jappl.2001.91.4.1893. PMID: 11568177. [DOI] [PubMed]
- 22.Grikiniene J, Volbekas V, Stakisaitis D. Gender differences of sodium metabolism and hyponatremia as an adverse drug effect. Medicina (Kaunas). 2004;40(10):935–42 PMID: 15516815. [PubMed] [Google Scholar]
- 23.De Vries GJ, Wang Z, Bullock NA, Numan S. Sex differences in the effects of testosterone and its metabolites on vasopressin messenger RNA levels in the bed nucleus of the stria terminalis of rats. J Neurosci. 1994;14(3 Pt 2):1789–94. 10.1523/JNEUROSCI.14-03-01789.1994.PMID:8126571;PMCID:PMC6577591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stachenfeld NS, DiPietro L, Palter SF, Nadel ER. Estrogen influences osmotic secretion of AVP and body water balance in postmenopausal women. Am J Physiol. 1998;274(1):R187–95. 10.1152/ajpregu.1998.274.1.R187. PMID: 9458917. [DOI] [PubMed] [Google Scholar]
- 25.Nishimura K, Yoshino K, Sanada K, Beppu H, Akiyama Y, Nishimura H, Tanaka K, Sonoda S, Ueno H, Yoshimura M, Maruyama T, Ozawa H, Ueta Y. Effect of oestrogen-dependent vasopressin on HPA axis in the median eminence of female rats. Sci Rep. 2019;9(1):5153. 10.1038/s41598-019-41714-z.PMID:30914732;PMCID:PMC6435644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Herzberg NH, Goudsmit E, Kruisbrink J, Boer GJ. Testosterone treatment restores reduced vasopressin-binding sites in the kidney of the ageing rat. J Endocrinol. 1989;123(1):59–63. 10.1677/joe.0.1230059. PMID: 2809490. [DOI] [PubMed] [Google Scholar]
- 27.Pávó I, Varga C, Szücs M, László F, Szécsi M, Gardi J, László FA. Effects of testosterone on the rat renal medullary vasopressin receptor concentration and the antidiuretic response. Life Sci. 1995;56(14):1215–22. 10.1016/0024-3205(95)00061-a. PMID: 7475899. [DOI] [PubMed] [Google Scholar]
- 28.Chang AY, Abdullah SM, Jain T, Stanek HG, Das SR, McGuire DK, Auchus RJ, de Lemos JA. Associations among androgens, estrogens, and natriuretic peptides in young women: observations from the Dallas Heart Study. J Am Coll Cardiol. 2007;49(1):109–16. 10.1016/j.jacc.2006.10.040. Epub 2006 Nov 13 PMID: 17207730. [DOI] [PubMed] [Google Scholar]
- 29.Glisic M, Rojas LZ, Asllanaj E, Vargas KG, Kavousi M, Ikram MA, Fauser BCJM, Laven JSE, Muka T, Franco OH. Sex steroids, sex hormone-binding globulin and N-terminal pro-brain natriureticin postmenopausal women. Int J Cardiol. 2018;15(261):189–95. 10.1016/j.ijcard.2018.03.008. Epub 2018 Mar 6 PMID: 29551255. [DOI] [PubMed] [Google Scholar]
- 30.Oparil S, Ehrlich EN, Lindheimer MD. Effect of progesterone on renal sodium handling in man: relation to aldosterone excretion and plasma renin activity. Clin Sci Mol Med. 1975;49(2):139–47. 10.1042/cs0490139. PMID: 1149405. [DOI] [PubMed] [Google Scholar]
- 31.Franz S, Skopp G, Musshoff F. The effect of creatine ingestion on urinary creatinine concentration: Does supplementation mask a heavy dilution? Drug Test Anal. 2022;14(1):162–8. 10.1002/dta.3165. Epub 2021 Oct 18 PMID: 34559477. [DOI] [PubMed] [Google Scholar]
- 32.Stachenfeld NS, Taylor HS. Effects of estrogen and progesterone administration on extracellular fluid. J Appl Physiol (1985). 2004 Mar;96(3):1011–8. 10.1152/japplphysiol.01032.2003. Epub 2003 Dec 5. PMID: 14660504. [DOI] [PubMed]
- 33.Johannsson G, Gibney J, Wolthers T, Leung KC, Ho KK. Independent and combined effects of testosterone and growth hormone on extracellular water in hypopituitary men. J Clin Endocrinol Metab. 2005;90(7):3989–94. 10.1210/jc.2005-0553. Epub 2005 Apr 12 PMID: 15827107. [DOI] [PubMed] [Google Scholar]
- 34.Choi JW, Song YC, Cheong NY, Lee K, Kim S, Lee KM, Ji K, Shin MY, Kim S. Concentrations of blood and urinary arsenic species and their characteristics in general Korean population. Environ Res. 2022 Nov;214(Pt 2):113846. 10.1016/j.envres.2022.113846. Epub 2022 Jul 9. PMID: 35820651. [DOI] [PubMed]
- 35.Fischer LM, da Costa KA, Kwock L, Galanko J, Zeisel SH. Dietary choline requirements of women: effects of estrogen and genetic variation. Am J Clin Nutr. 2010 Nov;92(5):1113–9. 10.3945/ajcn.2010.30064. Epub 2010 Sep 22. PMID: 20861172; PMCID: PMC2954445. [DOI] [PMC free article] [PubMed]
- 36.Muhetaer M, Yang M, Xia R, Lai Y, Wu J. Gender difference in arsenic biotransformation is an important metabolic basis for arsenic toxicity. BMC Pharmacol Toxicol. 2022;23(1):15. 10.1186/s40360-022-00554-w.PMID:35227329;PMCID:PMC8883647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dubey RK, Jackson EK. Estrogen-induced cardiorenal protection: potential cellular, biochemical, and molecular mechanisms. Am J Physiol Renal Physiol. 2001;280(3):365–88. 10.1152/ajprenal.2001.280.3.F365. PMID: 11181399. [DOI] [PubMed] [Google Scholar]
- 38.Monti DM, De Simone G, Langella E, Supuran CT, Di Fiore A, Monti SM. Insights into the role of reactive sulfhydryl groups of Carbonic Anhydrase III and VII during oxidative damage. J Enzyme Inhib Med Chem. 2017 Dec;32(1):5–12. 10.1080/14756366.2016.1225046. Epub 2016 Oct 21. PMID: 27766895; PMCID: PMC6010095. [DOI] [PMC free article] [PubMed]
- 39.Hopenhayn-Rich C, Biggs ML, Smith AH, Kalman DA, Moore LE. Methylation study of a population environmentally exposed to arsenic in drinking water. Environ Health Perspect. 1996;104(6):620–8. 10.1289/ehp.96104620.PMID:8793350;PMCID:PMC1469390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.García-Alvarado FJ, Neri-Meléndez H, Pérez Armendáriz L, Rivera Guillen M. Polimorfismos del gen Arsénico 3 Metiltransferasa (As3MT) y la eficiencia urinaria del metabolismo del arsénico en una población del norte de México [Polymorphisms of the Arsenite Methyltransferase (As3MT) gene and urinary efficiency of arsenic metabolism in a population in northern Mexico]. Rev Peru Med Exp Salud Publica. Jan-Mar;35(1):72–76. Spanish. 2018. 10.17843/rpmesp.2018.351.3565. PMID: 29924282. [DOI] [PubMed] [Google Scholar]
- 41.Torres-Sánchez L, López-Carrillo L, Rosado JL, Rodriguez VM, Vera-Aguilar E, Kordas K, García-Vargas GG, Cebrian ME. Sex differences in the reduction of arsenic methylation capacity as a function of urinary total and inorganic arsenic in Mexican children. Environ Res. 2016;151:38–43. 10.1016/j.envres.2016.07.020. Epub 2016 Jul 21 PMID: 27450997. [DOI] [PubMed] [Google Scholar]
- 42.Hsueh YM, Huang YL, Huang CC, Wu WL, Chen HM, Yang MH, Lue LC, Chen CJ. Urinary levels of inorganic and organic arsenic metabolites among residents in an arseniasis-hyperendemic area in Taiwan. J Toxicol Environ Health A. 1998;54(6):431–44. 10.1080/009841098158728. PMID: 9661909. [DOI] [PubMed] [Google Scholar]
- 43.Loffredo CA, Aposhian HV, Cebrian ME, Yamauchi H, Silbergeld EK. Variability in human metabolism of arsenic. Environ Res. 2003;92(2):85–91. 10.1016/s0013-9351(02)00081-6. PMID: 12854687. [DOI] [PubMed] [Google Scholar]
- 44.Bulka CM, Mabila SL, Lash JP, Turyk ME, Argos M. Arsenic and Obesity: A Comparison of Urine Dilution Adjustment Methods. Environ Health Perspect. 2017;125(8): 087020. 10.1289/EHP1202.PMID:28858828;PMCID:PMC5783631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Veiras LC, Girardi ACC, Curry J, Pei L, Ralph DL, et.al. Sexual Dimorphic Pattern of Renal Transporters and Electrolyte Homeostasis. J Am Soc Nephrol. 2017 Dec;28(12):3504–3517. 10.1681/ASN.2017030295. Epub 2017 Aug 3. PMID: 28774999; PMCID: PMC5698077. [DOI] [PMC free article] [PubMed]
- 46.Prasad N, Bhadauria D. Renal phosphate handling: Physiology. Indian J Endocrinol Metab. 2013;17(4):620–7. 10.4103/2230-8210.113752.PMID:23961477;PMCID:PMC3743361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hughes MF. Arsenic toxicity and potential mechanisms of action. Toxicol Lett. 2002;133(1):1–16. 10.1016/s0378-4274(02)00084-x. PMID: 12076506. [DOI] [PubMed] [Google Scholar]
- 48.Rosen BP, Liu Z. Transport pathways for arsenic and selenium: a minireview. Environ Int. 2009 Apr;35(3):512–5. 10.1016/j.envint.2008.07.023. Epub 2008 Sep 11. PMID: 18789529; PMCID: PMC2719050. [DOI] [PMC free article] [PubMed]
- 49.Roswall N, Hvidtfeldt UA, Harrington J, Levine KE, Sørensen M, Tjønneland A, Meliker JR, Raaschou-Nielsen O. Predictors of Urinary Arsenic Levels among Postmenopausal Danish Women. Int J Environ Res Public Health. 2018;15(7):1340. 10.3390/ijerph15071340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sumien N, Shetty RA, Gonzales EB. Creatine, creatine kinase, and aging. In: Harris J, Korolchuk V, editors. Biochemistry and cell biology of aging: Part I biomedical science. Subcell Biochem. 2018;90:145–168. 10.1007/978-981-13-2835-0_6. [DOI] [PubMed]
- 51.James GD, Sealey JE, Alderman M, Ljungman S, Mueller FB, Pecker MS, Laragh JH. A longitudinal study of urinary creatinine and creatinine clearance in normal subjects. Race, sex, and age differences. Am J Hypertens. 1988 Apr;1(2):124–31. 10.1093/ajh/1.2.124. PMID: 3401350. [DOI] [PubMed]
- 52.Ehlert AM, Wilson PB. The associations between body mass index, estimated lean body mass, and urinary hydration markers at the population level. Meas Phys Educ Exerc Sci. 2020;25(2):163–70. 10.1080/1091367X.2020.1858300. [Google Scholar]
- 53.Koek WNH, Campos-Obando N, van der Eerden BCJ, et al. Age-dependent sex differences in calcium and phosphate homeostasis. Endocr Connect. 2021;10(3):273–82. 10.1530/EC-20-0509.PMID:33543729;PMCID:PMC8052581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Cirillo M, Bilancio G, Marcarelli F. Ageing and changes in phosphate transport: clinical implications. J Nephrol. 2010 Nov-Dec;23 Suppl 16:S152–7. PMID: 21170873. [PubMed]
- 55.Hudgens EE, Drobna Z, He B, Le XC, Styblo M, Rogers J, Thomas DJ. Biological and behavioral factors modify urinary arsenic metabolic profiles in a U.S. population. Environ Health. 2016 May 26;15(1):62. 10.1186/s12940-016-0144-x. PMID: 27230915; PMCID: PMC4880853. [DOI] [PMC free article] [PubMed]
- 56.Buchanan S, Combet E, Stenvinkel P, Shiels PG. Klotho, Aging, and the Failing Kidney. Front Endocrinol (Lausanne). 2020;27(11):560. 10.3389/fendo.2020.00560.PMID:32982966;PMCID:PMC7481361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Charbonneau A, Leclerc M, Brunette MG. Effect of angiotensin II on calcium reabsorption by the luminal membranes of the nephron. Am J Physiol Endocrinol Metab. 2001;280(6):E928–36. 10.1152/ajpendo.2001.280.6.E928. PMID: 11350774. [DOI] [PubMed] [Google Scholar]
- 58.Hanouna G, Haymann JP, Baud L, Letavernier E. Vasopressin regulates renal calcium excretion in humans. Physiol Rep. 2015;3(11): e12562. 10.14814/phy2.12562.PMID:26620256;PMCID:PMC4673622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Orr SE, Bridges CC. Chronic Kidney Disease and Exposure to Nephrotoxic Metals. Int J Mol Sci. 2017;18(5):1039. 10.3390/ijms18051039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Peters BA, Hall MN, Liu X, Slavkovich V, Ilievski V, Alam S, Siddique AB, Islam T, Graziano JH, Gamble MV. Renal function is associated with indicators of arsenic methylation capacity in Bangladeshi adults. Environ Res. 2015 Nov;143(Pt A):123–30. 10.1016/j.envres.2015.10.001. Epub 2015 Oct 19. PMID: 26476787; PMCID: PMC4740972. [DOI] [PMC free article] [PubMed]
- 61.Zheng LY, Umans JG, Yeh F, Francesconi KA, Goessler W, Silbergeld EK, Bandeen-Roche K, Guallar E, Howard BV, Weaver VM, Navas-Acien A. The association of urine arsenic with prevalent and incident chronic kidney disease: evidence from the Strong Heart Study. Epidemiology. 2015;26(4):601–12. 10.1097/EDE.0000000000000313.PMID:25929811;PMCID:PMC4844343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zheng L, Kuo CC, Fadrowski J, Agnew J, Weaver VM, Navas-Acien A. Arsenic and Chronic Kidney Disease: A Systematic Review. Curr Environ Health Rep. 2014;1(3):192–207. 10.1007/s40572-014-0024-x.PMID:25221743;PMCID:PMC4159179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hsueh YM, Chung CJ, Shiue HS, et al. Urinary arsenic species and CKD in a Taiwanese population: a case-control study. Am J Kidney Dis. 2009;54(5):859–70. 10.1053/j.ajkd.2009.06.016. Epub 2009 Aug 14 PMID: 19682779. [DOI] [PubMed] [Google Scholar]
- 64.Meng J, Ohlsson C, Laughlin GA, Chonchol M, Wassel CL, Ljunggren O, Karlsson MK, Mellstrom D, Orwoll ES, Barrett-Connor E, Ix JH; Osteoporotic Fractures in Men (MrOs) Study Group. Associations of estradiol and testosterone with serum phosphorus in older men: the Osteoporotic Fractures in Men study. Kidney Int. 2010 Aug;78(4):415–22. 10.1038/ki.2010.161. Epub 2010 Jun 9. PMID: 20531458; PMCID: PMC3787687. [DOI] [PMC free article] [PubMed]
- 65.Rowe JW, Andres R, Tobin JD, Norris AH, Shock NW. The effect of age on creatinine clearance in men: a cross-sectional and longitudinal study. J Gerontol. 1976;31(2):155–63. 10.1093/geronj/31.2.155. PMID: 1249404. [DOI] [PubMed] [Google Scholar]
- 66.Weinstein JR, Anderson S. The aging kidney: physiological changes. Adv Chronic Kidney Dis. 2010;17(4):302–7. 10.1053/j.ackd.2010.05.002.PMID:20610357;PMCID:PMC2901622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Schmitt R, Melk A. Molecular mechanisms of renal aging. Kidney Int. 2017;92(3):569–79. 10.1016/j.kint.2017.02.036. Epub 2017 Jul 18 PMID: 28729036. [DOI] [PubMed] [Google Scholar]
- 68.Glassock RJ, Rule AD. The implications of anatomical and functional changes of the aging kidney: with an emphasis on the glomeruli. Kidney Int. 2012 Aug;82(3):270–7. 10.1038/ki.2012.65. Epub 2012 Mar 21. PMID: 22437416; PMCID: PMC3513938. [DOI] [PMC free article] [PubMed]
- 69.Martin JE, Sheaff MT. Renal ageing. J Pathol. 2007;211(2):198–205. 10.1002/path.2111. PMID: 17200944. [DOI] [PubMed] [Google Scholar]
- 70.Torres VE. Vasopressin in chronic kidney disease: an elephant in the room? Kidney Int. 2009;76(9):925–8. 10.1038/ki.2009.325.PMID:19829311;PMCID:PMC2925670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Melsom T, Norvik JV, Enoksen IT, Stefansson V, Mathisen UD, Fuskevåg OM, Jenssen TG, Solbu MD, Eriksen BO. Sex Differences in Age-Related Loss of Kidney Function. J Am Soc Nephrol. 2022 Oct;33(10):1891–1902. 10.1681/ASN.2022030323. Epub 2022 Aug 17. PMID: 35977806; PMCID: PMC9528336. [DOI] [PMC free article] [PubMed]
- 72.Hoang K, Tan JC, Derby G, Blouch KL, Masek M, Ma I, Lemley KV, Myers BD. Determinants of glomerular hypofiltration in aging humans. Kidney Int. 2003;64(4):1417–24. 10.1046/j.1523-1755.2003.00207.x. PMID: 12969161. [DOI] [PubMed] [Google Scholar]
- 73.Maas AH, Appelman YE. Gender differences in coronary heart disease. Neth Heart J. 2010;18(12):598–602. 10.1007/s12471-010-0841-y.PMID:21301622;PMCID:PMC3018605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Towfighi A, Zheng L, Ovbiagele B. Sex-specific trends in midlife coronary heart disease risk and prevalence. Arch Intern Med. 2009;169(19):1762–6. 10.1001/archinternmed.2009.318. PMID: 19858433. [DOI] [PubMed] [Google Scholar]
- 75.Man JJ, Beckman JA, Jaffe IZ. Sex as a Biological Variable in Atherosclerosis. Circ Res. 2020 Apr 24;126(9):1297–1319. 10.1161/CIRCRESAHA.120.315930. Epub 2020 Apr 23. PMID: 32324497; PMCID: PMC7185045. [DOI] [PMC free article] [PubMed]
- 76.Vakhtangadze T, Singh Tak R, Singh U, Baig MS, Bezsonov E. Gender Differences in Atherosclerotic Vascular Disease: From Lipids to Clinical Outcomes. Front Cardiovasc Med. 2021;28(8): 707889. 10.3389/fcvm.2021.707889.PMID:34262956;PMCID:PMC8273377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Berg UB. Differences in decline in GFR with age between males and females. Reference data on clearances of inulin and PAH in potential kidney donors. Nephrol Dial Transplant. 2006 Sep;21(9):2577–82. 10.1093/ndt/gfl227. Epub 2006 May 23. PMID: 16720595. [DOI] [PubMed]
- 78.Melsom T, Norvik JV, Enoksen IT, Stefansson V, Mathisen UD, Fuskevåg OM, Jenssen TG, Solbu MD, Eriksen BO. Sex Differences in Age-Related Loss of Kidney Function. J Am Soc Nephrol. 2022 Oct;33(10):1891–1902. 10.1681/ASN.2022030323. Epub 2022 Aug 17. PMID: 35977806; PMCID: PMC9528336. [DOI] [PMC free article] [PubMed]
- 79.Wiggins JE, Goyal M, Sanden SK, Wharram BL, Shedden KA, Misek DE, Kuick RD, Wiggins RC. Podocyte hypertrophy, “adaptation,” and “decompensation” associated with glomerular enlargement and glomerulosclerosis in the aging rat: prevention by calorie restriction. J Am Soc Nephrol. 2005;16(10):2953–66. 10.1681/ASN.2005050488. Epub 2005 Aug 24 PMID: 16120818. [DOI] [PubMed] [Google Scholar]
- 80.Musso CG, Macías-Núñez JF. Dysfunction of the thick loop of Henle and senescence: from molecular biology to clinical geriatrics. Int Urol Nephrol. 2011;43(1):249–52. 10.1007/s11255-010-9783-y. Epub 2010 Nov 12 PMID: 21072593. [DOI] [PubMed] [Google Scholar]
- 81.O’Sullivan ED, Hughes J, Ferenbach DA. Renal Aging: Causes and Consequences. J Am Soc Nephrol. 2017;28(2):407–20. 10.1681/ASN.2015121308.Epub2016Nov15.PMID:28143966;PMCID:PMC5280008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Chade AR. Renal vascular structure and rarefaction. Compr Physiol. 2013;3(2):817–31. 10.1002/cphy.c120012.PMID:23720331;PMCID:PMC3936257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Usberti M, Federico S, Di Minno G, Ungaro B, Ardillo G, Pecoraro C, Cianciaruso B, Cerbone AM, Cirillo F, Pannain M, et al. Effects of angiotensin II on plasma ADH, prostaglandin synthesis, and water excretion in normal humans. Am J Physiol. 1985;248(2 Pt 2):254–9. 10.1152/ajprenal.1985.248.2.F254. PMID: 3881995. [DOI] [PubMed] [Google Scholar]
- 84.Clark CM, Monahan KD, Drew RC. Aging augments renal vasoconstrictor response to orthostatic stress in humans. Am J Physiol Regul Integr Comp Physiol. 2015 Dec 15;309(12):R1474–8. 10.1152/ajpregu.00291.2015. Epub 2015 Oct 14. PMID: 26468259; PMCID: PMC4698412. [DOI] [PMC free article] [PubMed]
- 85.Patel HM, Mast JL, Sinoway LI, Muller MD. Effect of healthy aging on renal vascular responses to local cooling and apnea. J Appl Physiol (1985). 2013 Jul 1;115(1):90–6. 10.1152/japplphysiol.00089.2013. Epub 2013 May 2. PMID: 23640587; PMCID: PMC3727011. [DOI] [PMC free article] [PubMed]
- 86.Baylis C. Sexual dimorphism in the aging kidney: differences in the nitric oxide system. Nat Rev Nephrol. 2009;5(7):384–96. 10.1038/nrneph.2009.90. Epub 2009 Jun 2 PMID: 19488070. [DOI] [PubMed] [Google Scholar]
- 87.Rule AD, Bailey KR, Lieske JC, Peyser PA, Turner ST. Estimating the glomerular filtration rate from serum creatinine is better than from cystatin C for evaluating risk factors associated with chronic kidney disease. Kidney Int. 2013 Jun;83(6):1169–76. 10.1038/ki.2013.7. Epub 2013 Feb 20. Erratum in: Kidney Int. 2013 Aug;84(2):419. PMID: 23423253; PMCID: PMC3661736. [DOI] [PMC free article] [PubMed]
- 88.Kile ML, Hoffman E, Hsueh Y-M, Afroz S, Quamruzzaman Q, Rahman M, Mahiuddin G, Ryan LM, Christiani DC. Variability in Biomarkers of Arsenic Exposure and Metabolism in Adults over Time. Environ Health Perspect. 2009;117:455–60. 10.1289/ehp.11251. PMID: 15577230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Chowdhury UK, Rahman MM, Sengupta MK, Lodh D, Chanda CR, Roy S, Quamruzzaman Q, Tokunaga H, Ando M, Chakraborti D. Pattern of excretion of arsenic compounds [arsenite, arsenate, MMA(V), DMA(V)] in urine of children compared to adults from an arsenic exposed area in Bangladesh. J Environ Sci Health A Tox Hazard Subst Environ Eng. 2003;38(1):87–113. 10.1081/ese-120016883. PMID: 12635821. [DOI] [PubMed] [Google Scholar]
- 90.Bae HS, Ryu DY, Choi BS, Park JD. Urinary Arsenic Concentrations and their Associated Factors in Korean Adults. Toxicol Res. 2013;29(2):137–42. 10.5487/TR.2013.29.2.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Moriguchi J, Ezaki T, Tsukahara T, Furuki K, Fukui Y, Okamoto S, Ukai H, Sakurai H, Shimbo S, Ikeda M. Comparative evaluation of four urinary tubular dysfunction markers, with special references to the effects of aging and correction for creatinine concentration. Toxicol Lett. 2003;143(3):279–90. 10.1016/s0378-4274(03)00181-4. PMID: 12849688. [DOI] [PubMed] [Google Scholar]
- 92.Tan SJ, Smith ER, Hewitson TD, Holt SG, Toussaint ND. The importance of klotho in phosphate metabolism and kidney disease. Nephrology (Carlton). 2014;19(8):439–49. 10.1111/nep.12268. PMID: 24750549. [DOI] [PubMed] [Google Scholar]
- 93.Torres PA, De Brauwere DP. Three feedback loops precisely regulating serum phosphate concentration. Kidney Int. 2011;80(5):443–5. 10.1038/ki.2011.146. PMID: 21841832. [DOI] [PubMed] [Google Scholar]
- 94.Behringer V, Stevens JMG, Deschner T, Sonnweber R, Hohmann G. Aging and sex affect soluble alpha klotho levels in bonobos and chimpanzees. Front Zool. 2018;19(15):35. 10.1186/s12983-018-0282-9.PMID:30250491;PMCID:PMC6146871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Pedersen L, Pedersen SM, Brasen CL, Rasmussen LM. Soluble serum Klotho levels in healthy subjects. Comparison of two different immunoassays. Clin Biochem. 2013 Aug;46(12):1079–1083. 10.1016/j.clinbiochem.2013.05.046. Epub 2013 May 23. PMID: 23707222. [DOI] [PubMed]
- 96.Gkentzi D, Efthymiadou A, Kritikou D, Chrysis D. Fibroblast growth factor 23 and Klotho serum levels in healthy children. Bone. 2014;66:8–14. 10.1016/j.bone.2014.05.012. Epub 2014 May 29 PMID: 24880094. [DOI] [PubMed] [Google Scholar]
- 97.Zhang Z, Qiu S, Huang X, Jin K, Zhou X, Lin T, Zou X, Yang Q, Yang L, Wei Q. Association between testosterone and serum soluble α-klotho in U.S. males: a cross-sectional study. BMC Geriatr. 2022 Jul 11;22(1):570. 10.1186/s12877-022-03265-3. PMID: 35820842; PMCID: PMC9275159. [DOI] [PMC free article] [PubMed]
- 98.Ix JH, Chonchol M, Laughlin GA, Shlipak MG, Whooley MA. Relation of sex and estrogen therapy to serum fibroblast growth factor 23, serum phosphorus, and urine phosphorus: the Heart and Soul Study. Am J Kidney Dis. 2011 Nov;58(5):737–45. 10.1053/j.ajkd.2011.06.011. Epub 2011 Aug 19. PMID: 21855188; PMCID: PMC3199317. [DOI] [PMC free article] [PubMed]
- 99.Chen K, Sun Z. Estrogen inhibits renal Na-Pi Co-transporters and improves klotho deficiency-induced acute heart failure. Redox Biol. 2021 Nov;47:102173. 10.1016/j.redox.2021.102173. Epub 2021 Oct 18. PMID: 34678656; PMCID: PMC8577443. [DOI] [PMC free article] [PubMed]
- 100.Chen JW, Chen HY, Li WF, Liou SH, Chen CJ, Wu JH, Wang SL. The association between total urinary arsenic concentration and renal dysfunction in a community-based population from central Taiwan. Chemosphere. 2011;84(1):17–24. 10.1016/j.chemosphere.2011.02.091. Epub 2011 Apr 1 PMID: 21458841. [DOI] [PubMed] [Google Scholar]
- 101.Poręba R, Gać P, Poręba M, Antonowicz-Juchniewicz J, Andrzejak R. Relation between occupational exposure to lead, cadmium, arsenic and concentration of cystatin C. Toxicology. 2011;283(2–3):88–95. 10.1016/j.tox.2011.02.008. Epub 2011 Feb 26 PMID: 21356263. [DOI] [PubMed] [Google Scholar]
- 102.Meliker JR, Wahl RL, Cameron LL, Nriagu JO. Arsenic in drinking water and cerebrovascular disease, diabetes mellitus, and kidney disease in Michigan: a standardized mortality ratio analysis. Environ Health. 2007;2(6):4. 10.1186/1476-069X-6-4.PMID:17274811;PMCID:PMC1797014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Tsai SM, Wang TN, Ko YC. Mortality for certain diseases in areas with high levels of arsenic in drinking water. Arch Environ Health. 1999 May-Jun;54(3):186–93. 10.1080/00039899909602258. PMID: 10444040. [DOI] [PubMed]
- 104.Zou D, Wu W, He Y, Ma S, Gao J. The role of klotho in chronic kidney disease. BMC Nephrol. 2018;19(1):285. 10.1186/s12882-018-1094-z.PMID:30348110;PMCID:PMC6198535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Yoon HE, Ghee JY, Piao S, Song JH, Han DH, Kim S, Ohashi N, Kobori H, Kuro-o M, Yang CW. Angiotensin II blockade upregulates the expression of Klotho, the anti-ageing gene, in an experimental model of chronic cyclosporine nephropathy. Nephrol Dial Transplant. 2011 Mar;26(3):800–13. 10.1093/ndt/gfq537. Epub 2010 Sep 2. PMID: 20813770; PMCID: PMC3108350. [DOI] [PMC free article] [PubMed]
- 106.Laffer CL, Elijovich F, Sahinoz M, Pitzer A, Kirabo A. New Insights Into the Renin-Angiotensin System in Chronic Kidney Disease. Circ Res. 2020 Aug 14;127(5):607–609. 10.1161/CIRCRESAHA.120.317624. Epub 2020 Aug 13. PMID: 32790525; PMCID: PMC7430039. [DOI] [PMC free article] [PubMed]
- 107.Petrick JS, Ayala-Fierro F, Cullen WR, Carter DE, Vasken AH. Monomethylarsonous acid (MMA(III)) is more toxic than arsenite in Chang human hepatocytes. Toxicol Appl Pharmacol. 2000;163(2):203–7. 10.1006/taap.1999.8872. PMID: 10698679. [DOI] [PubMed] [Google Scholar]
- 108.Vahter M, Concha G. Role of Metabolism in Arsenic Toxicity. Pharmacol Toxicol. 2001;89:1–5. 10.1111/j.1600-0773.2001.890101.x. [DOI] [PubMed] [Google Scholar]
- 109.Stýblo M, Drobná Z, Jaspers I, Lin S, Thomas DJ. The role of biomethylation in toxicity and carcinogenicity of arsenic: a research update. Environ Health Perspect. 2002 Oct;110 Suppl 5(Suppl 5):767–71. 10.1289/ehp.110-1241242. PMID: 12426129; PMCID: PMC1241242. [DOI] [PMC free article] [PubMed]
- 110.Steinmaus C, Yuan Y, Kalman D, Rey OA, Skibola CF, Dauphine D, Basu A, Porter KE, Hubbard A, Bates MN, Smith MT, Smith AH. Individual differences in arsenic metabolism and lung cancer in a case-control study in Cordoba, Argentina. Toxicol Appl Pharmacol. 2010 Sep 1;247(2):138–45. 10.1016/j.taap.2010.06.006. Epub 2010 Jun 17. PMID: 20600216; PMCID: PMC3849353. [DOI] [PMC free article] [PubMed]
- 111.Drinking Water (Trinkwasser) from the website of Swiss Federal Food Safety and Veterinary Office (Bundesamt für Lebensmittelsicherheit und Veterinärwesen) https://www.blv.admin.ch/blv/de/home/lebensmittel-undernaehrung/Lebensmittelsicherheit/verantwortlichkeiten/sicheres-trinkwasser.html
- 112.Metals in groundwater in Austria– Maps and explanations (Metalle im Grundwasser in Österreich, Karten und Erläuterungen) from the website of the Austrian Ministry of Life (Lebensministerium.at) https://info.bml.gv.at/dam/jcr:24b11c8f-fa52-4629-beac-f042da4db709/Metalle_im_Grundwasser_Karten_und_Erl%C3%A4uterungen.pdf
- 113.Ordinance on the quality of water intended for human consumption, Drinking Water Ordinance - TrinkwV (Verordnung über die Qualität von Wasser für den menschlichen Gebrauch, Trinkwasserverordnung - TrinkwV) from the website of the German Federal Office of Justice (Bundesamt für Justiz), https://www.gesetze-im-internet.de/trinkwv_2023/anlage_2.html
- 114.National Research Council (US) Subcommittee on Arsenic in Drinking Water. Arsenic in Drinking Water. Washington (DC): National Academies Press (US); 1999. 6, Biomarkers of Arsenic Exposure. Available from: https://www.ncbi.nlm.nih.gov/books/NBK230898/ [PubMed]
- 115.GF Nordberg BA Fowler M Nordberg Eds 2022 Handbook on the Toxicology of Metals 2 5 Academic Press Burlington, MA 9780128229460. 10.1016/B978-0-12-822946-0.00037-4
- 116.Ellingsen DG, Weinbruch S, Sallsten G, Berlinger B, Barregard L. The variability of arsenic in blood and urine of humans. J Trace Elem Med Biol. 2023;78 127179 10.1016/j.jtemb.2023.127179 Epub 2023 May 3 PMID: 37148695. [DOI] [PubMed]
- 117.National Research Council (US) Subcommittee on Arsenic in Drinking Water. Arsenic in Drinking Water. Washington (DC): National Academies Press (US); 1999. 6, Biomarkers of Arsenic Exposure. Available from: https://www.ncbi.nlm.nih.gov/books/NBK230898/ [PubMed]
- 118.Lee DH, Keum N, Hu FB, Orav EJ, Rimm EB, Sun Q, Willett WC, Giovannucci EL. Development and validation of anthropometric prediction equations for lean body mass, fat mass and percent fat in adults using the National Health and Nutrition Examination Survey (NHANES) 1999–2006. Br J Nutr. 2017;118(10):858–66. 10.1017/S0007114517002665. Epub 2017 Nov 7 PMID: 29110742. [DOI] [PubMed] [Google Scholar]
- 119.Forni Ogna V, Ogna A, Vuistiner P, Pruijm M, Ponte B, Ackermann D, Gabutti L, Vakilzadeh N, Mohaupt M, Martin PY, Guessous I, Péchère-Bertschi A, Paccaud F, Bochud M, Burnier M; Swiss Survey on Salt Group. New anthropometry-based age- and sex-specific reference values for urinary 24-hour creatinine excretion based on the adult Swiss population. BMC Med. 2015 Feb 27;13:40. 10.1186/s12916-015-0275-x. PMID: 25858764; PMCID: PMC4354997. [DOI] [PMC free article] [PubMed]
- 120.Orr SE, Bridges CC. Chronic Kidney Disease and Exposure to Nephrotoxic Metals. Int J Mol Sci. 2017;18(5):1039. 10.3390/ijms18051039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Schäppi J, Stringhini S, Guessous I, Staub K, Matthes KL. Body height in adult women and men in a cross-sectional population-based survey in Geneva: temporal trends, association with general health status and height loss after age 50. BMJ Open. 2022;12(7): e059568. 10.1136/bmjopen-2021-059568.PMID:35803618;PMCID:PMC9272122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Shen H, Niu Q, Xu M, Rui D, Xu S, Feng G, Ding Y, Li S, Jing M. Factors Affecting Arsenic Methylation in Arsenic-Exposed Humans: A Systematic Review and Meta-Analysis. Int J Environ Res Public Health. 2016;13(2):205. 10.3390/ijerph13020205.PMID:26861378;PMCID:PMC4772225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Pandey R, Garg A, Gupta K, Shukla P, Mandrah K, Roy S, Chattopadhyay N, Bandyopadhyay S. Arsenic Induces Differential Neurotoxicity in Male, Female, and E2-Deficient Females: Comparative Effects on Hippocampal Neurons and Cognition in Adult Rats. Mol Neurobiol. 2022;59(5):2729–44. 10.1007/s12035-022-02770-1. Epub 2022 Feb 17 PMID: 35175559. [DOI] [PubMed] [Google Scholar]
- 124.Chang LZ, Sheng S, Zhe Z, Xiaoxiao S, Qing J. Study on the relationship between age and the concentrations of heavy metal elements in human bone. Ann Transl Med. 2018;6(16):320. 10.21037/atm.2018.08.09.PMCID:PMC6186975. PMID: 30363972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Akbal A, Yılmaz H, Tutkun E. Arsenic exposure associated with decreased bone mineralization in male. Aging Male. 2014;17(4):256–8. 10.3109/13685538.2013.819326. [DOI] [PubMed] [Google Scholar]
- 126.Chang LZ, Sheng S, Zhe Z, Xiaoxiao S, Qing J. Study on the relationship between age and the concentrations of heavy metal elements in human bone. Ann Transl Med. 2018;6(16):320. 10.21037/atm.2018.08.09.PMCID:PMC6186975. PMID: 30363972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Qi Y, Goel R, Kim S, Richards EM, Carter CS, Pepine CJ, Raizada MK, Buford TW. Intestinal Permeability Biomarker Zonulin is Elevated in Healthy Aging. J Am Med Dir Assoc. 2017 Sep 1;18(9):810.e1–810.e4. 10.1016/j.jamda.2017.05.018. Epub 2017 Jul 1. PMID: 28676292; PMCID: PMC5581307. [DOI] [PMC free article] [PubMed]
- 128.Jansen RJ, Argos M, Tong L, Li J, Rakibuz-Zaman M, Islam MT, Slavkovich V, Ahmed A, Navas-Acien A, Parvez F, Chen Y, Gamble MV, Graziano JH, Pierce BL, Ahsan H. Determinants and Consequences of Arsenic Metabolism Efficiency among 4,794 Individuals: Demographics, Lifestyle, Genetics, and Toxicity. Cancer Epidemiol Biomarkers Prev. 2016 Feb;25(2):381–90. 10.1158/1055-9965.EPI-15-0718. Epub 2015 Dec 16. PMID: 26677206; PMCID: PMC4767610. [DOI] [PMC free article] [PubMed]
- 129.Tseng CH. A review on environmental factors regulating arsenic methylation in humans. Toxicol Appl Pharmacol. 2009;235(3):338–50. 10.1016/j.taap.2008.12.016. Epub 2008 Dec 30 PMID: 19168087. [DOI] [PubMed] [Google Scholar]
- 130.Mavani GP, DeVita MV, Michelis MF. A review of the nonpressor and nonantidiuretic actions of the hormone vasopressin. Front Med (Lausanne). 2015;24(2):19. 10.3389/fmed.2015.00019.PMID:25853137;PMCID:PMC4371647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Tang C, Pathare G, Michael D, Fajol A, Eichenmüller M, Lang F. Downregulation of klotho expression by dehydration. Am J Physiol Renal Physiol. 2011;301(4):F745–50. 10.1152/ajprenal.00037.2011. [DOI] [PubMed] [Google Scholar]
- 132.Kale A, Sankrityayan H, Anders HJ, Gaikwad AB. Klotho in kidney diseases: a crosstalk between the renin-angiotensin system and endoplasmic reticulum stress. Nephrol Dial Transplant. 2023;38(4):819–25. 10.1093/ndt/gfab340. PMID: 34850136. [DOI] [PubMed] [Google Scholar]
- 133.Johnson AG, Crawford GA, Kelly D, Nguyen TV, Gyory AZ. Arginine vasopressin and osmolality in the elderly. J Am Geriatr Soc. 1994;42(4):399–404. 10.1111/j.1532-5415.1994.tb07488.x. PMID: 8144825. [DOI] [PubMed] [Google Scholar]
- 134.Yoon HE, Choi BS. The renin-angiotensin system and aging in the kidney. Korean J Intern Med. 2014 May;29(3):291–5. 10.3904/kjim.2014.29.3.291. Epub 2014 Apr 29. PMID: 24851061; PMCID: PMC4028516. [DOI] [PMC free article] [PubMed]
- 135.Phillips PA, Rolls BJ, Ledingham JG, Forsling ML, Morton JJ, Crowe MJ, Wollner L. Reduced thirst after water deprivation in healthy elderly men. N Engl J Med. 1984;311(12):753–9. 10.1056/NEJM198409203111202. PMID: 6472364. [DOI] [PubMed] [Google Scholar]
- 136.Ansari A, Walton SL, Denton KM. Sex- and age-related differences in renal and cardiac injury and senescence in stroke-prone spontaneously hypertensive rats. Biol Sex Differ. 2023;14(1):33. 10.1186/s13293-023-00519-6.PMID:37217968;PMCID:PMC10201739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Menendez-Castro C, Cordasic N, Fahlbusch FB, Woelfle J, Hilgers KF, Hartner A. Sex differences in long-term kidney fibrosis following neonatal nephron loss during ongoing nephrogenesis. Mol Cell Pediatr. 2023;10(1):8. 10.1186/s40348-023-00164-4.PMID:37624430;PMCID:PMC10457250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Garate-Carrillo A, Gonzalez J, Ceballos G, Ramirez-Sanchez I, Villarreal F. Sex related differences in the pathogenesis of organ fibrosis. Transl Res. 2020 Aug;222:41–55. 10.1016/j.trsl.2020.03.008. Epub 2020 Mar 16. PMID: 32289256; PMCID: PMC7721117. [DOI] [PMC free article] [PubMed]
- 139.Bajwa S, Luebbe A, Vo NDN, Piskor EM, Kosan C, Wolf G, Loeffler I. RAGE is a critical factor of sex-based differences in age-induced kidney damage. Front Physiol. 2023;29(14):1154551. 10.3389/fphys.2023.1154551.PMID:37064891;PMCID:PMC10090518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Birder LA, Wolf-Johnston AS, Jackson EK, Wein AJ, Dmochowski R. Aging increases the expression of vasopressin receptors in both the kidney and urinary bladder. Neurourol Urodyn. 2019 Jan;38(1):393–397. 10.1002/nau.23830. Epub 2018 Oct 12. PMID: 30311671; PMCID: PMC9839378. [DOI] [PMC free article] [PubMed]
- 141.Smith HW, Finkelstein RP. The renal clearances of substances contained in blood. J Biol Chem. 1940;133:1–16. [Google Scholar]
- 142.Kaise T, Fukui S. The chemical form and acute toxicity of arsenic compounds in marine organisms. Appl Organomet Chem. 1992;6(2):155–60. 10.1002/aoc.590060209. [Google Scholar]
- 143.Tchounwou PB, Centeno JA, Patlolla AK. Arsenic toxicity, mutagenesis, and carcinogenesis-a health risk assessment and management approach. Mol Cell Biochem. 2004;255(1–2):47–55. 10.1023/b:mcbi.0000007260.32981.b9. PMID: 14971645. [DOI] [PubMed] [Google Scholar]
- 144.Le XC, Cullen WR, Reimer KJ. Human urinary arsenic excretion after one-time ingestion of seaweed, crab, and shrimp. Clin Chem. 1994;40(4):617–24 PMID: 8149620. [PubMed] [Google Scholar]
- 145.Sica DA. Sodium and water retention in heart failure and diuretic therapy: basic mechanisms. Cleve Clin J Med. 2006 Jun;73 Suppl 2:S2–7; discussion S30–3. 10.3949/ccjm.73.suppl_2.s2. PMID: 16786906. [DOI] [PubMed]
- 146.Wang P, Ouyang J, Jia Z, Zhang A, Yang Y. Roles of DNA damage in renal tubular epithelial cells injury. Front Physiol. 2023;6(14):1162546. 10.3389/fphys.2023.1162546.PMID:37089416;PMCID:PMC10117683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Dalal R, Bruss ZS, Sehdev JS. Physiology, Renal Blood Flow and Filtration. [Updated 2022 Jul 25]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482248/. [PubMed]
- 148.Denton KM, Fennessy PA, Alcorn D, Anderson WP. Morphometric analysis of the actions of angiotensin II on renal arterioles and glomeruli. Am J Physiol. 1992;262(3 Pt 2):F367–72. 10.1152/ajprenal.1992.262.3.F367. PMID: 1558155. [DOI] [PubMed] [Google Scholar]
- 149.Shemesh O, Golbetz H, Kriss JP, Myers BD. Limitations of creatinine as a filtration marker in glomerulopathic patients. Kidney Int. 1985;28(5):830–8. 10.1038/ki.1985.205. PMID: 2418254. [DOI] [PubMed] [Google Scholar]
- 150.Tsuda A, Ishimura E, Machiba Y, Uedono H, Nakatani S, Mori K, Uchida J, Emoto M. Increased Glomerular Hydrostatic Pressure is Associated with Tubular Creatinine Reabsorption in Healthy Subjects. Kidney Blood Press Res. 2020;45:996–1008. 10.1159/000510838. [DOI] [PubMed] [Google Scholar]
- 151.Ginsburg JM. The renal mechanism for excretion and transformation of arsenic in the dog. Am J Physiol. 1965;208:832–40. 10.1152/ajplegacy.1965.208.5.832. PMID: 14286849. [DOI] [PubMed] [Google Scholar]
- 152.Barr DB, Wilder LC, Caudill SP, Gonzalez AJ, Needham LL, Pirkle JL. Urinary creatinine concentrations in the U.S. population: implications for urinary biologic monitoring measurements. Environmental health perspectives. 2005;113:192–200. 10.1289/ehp.7337. [DOI] [PMC free article] [PubMed]
- 153.Worcester EM, Bergsland KJ, Gillen DL, Coe FL. Mechanism for higher urine pH in normal women compared with men. Am J Physiol Renal Physiol. 2018 Apr 1;314(4):F623-F629. 10.1152/ajprenal.00494.2017. Epub 2017 Dec 20. PMID: 29357436; PMCID: PMC5966764. [DOI] [PMC free article] [PubMed]
- 154.Menezes CJ, Worcester EM, Coe FL, Asplin J, Bergsland KJ, Ko B. Mechanisms for falling urine pH with age in stone formers. Am J Physiol Renal Physiol. 2019 Jul 1;317(7):F65-F72. 10.1152/ajprenal.00066.2019. Epub 2019 Apr 24. PMID: 31017011; PMCID: PMC6692718. [DOI] [PMC free article] [PubMed]
- 155.Suzuki S, Yoshida J, Takahashi T. Effect of testosterone on carbonic anhydrase and MG(2+)-dependent HCO3-stimulated ATPase activities in rat kidney: comparison with estradiol effect. Comp Biochem Physiol C Pharmacol Toxicol Endocrinol. 1996;114(2):105–12. 10.1016/0742-8413(96)00021-7. PMID: 8760605. [DOI] [PubMed] [Google Scholar]
- 156.Toering TJ, van der Graaf AM, Visser FW, Buikema H, Navis G, Faas MM, Lely AT. Gender differences in response to acute and chronic angiotensin II infusion: a translational approach. Physiol Rep. 2015;3(7): e12434. 10.14814/phy2.12434.PMID:26149279;PMCID:PMC4552520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Eloy L, Grünfeld JP, Bayle F, Bankir L, Ramos-Frendo B, Trinh-Trang-Tan MM. Papillary plasma flow in rats. II Hormonal control Pflugers Arch. 1983;398(3):253–8. 10.1007/BF00657161. PMID: 6356035. [DOI] [PubMed] [Google Scholar]
- 158.Lote CJ. Renal prostaglandins and sodium excretion. Q J Exp Physiol. 1982;67(3):377–85. 10.1113/expphysiol.1982.sp002652. PMID: 7051084. [DOI] [PubMed] [Google Scholar]
- 159.Parekh N, Zou AP, Jüngling I, Endlich K, Sadowski J, Steinhausen M. Sex differences in control of renal outer medullary circulation in rats: role of prostaglandins. Am J Physiol. 1993;264(4 Pt 2):F629–36. 10.1152/ajprenal.1993.264.4.F629. PMID: 8476076. [DOI] [PubMed] [Google Scholar]
- 160.Ho LL, Kench JG, Handelsman DJ, Scheffer GL, Stricker PD, Grygiel JG, Sutherland RL, Henshall SM, Allen JD, Horvath LG. Androgen regulation of multidrug resistance-associated protein 4 (MRP4/ABCC4) in prostate cancer. Prostate. 2008;68(13):1421–9. 10.1002/pros.20809. PMID: 18615486. [DOI] [PubMed] [Google Scholar]
- 161.Huang YT, Zhou J, Shi S, Xu HY, Qu F, Zhang D, Chen YD, Yang J, Huang HF, Sheng JZ. Identification of Estrogen Response Element in Aquaporin-3 Gene that Mediates Estrogen-induced Cell Migration and Invasion in Estrogen Receptor-positive Breast Cancer. Sci Rep. 2015;29(5):12484. 10.1038/srep12484.PMID:26219409;PMCID:PMC4518221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Dibas AI, Mia AJ, Yorio T. Aquaporins (water channels): role in vasopressin-activated water transport. Proc Soc Exp Biol Med. 1998;219(3):183–99. 10.3181/00379727-219-44332. PMID: 9824541. [DOI] [PubMed] [Google Scholar]
- 163.Hilliard LM, Nematbakhsh M, Kett MM, Teichman E, Sampson AK, Widdop RE, Evans RG, Denton KM. Gender differences in pressure-natriuresis and renal autoregulation: role of the Angiotensin type 2 receptor. Hypertension. 2011;57(2):275–82. 10.1161/HYPERTENSIONAHA.110.166827. Epub 2010 Dec 28 PMID: 21189402. [DOI] [PubMed] [Google Scholar]
- 164.Oelkers WKH, Effects of estrogens and progestogens on the renin-aldosterone system and blood pressure, Steroids,Volume 61, Issue 4,1996, Pages 166–171, ISSN 0039–128X, 10.1016/0039-128X(96)00007-4. [DOI] [PubMed]
- 165.Mishra JS, More AS, Gopalakrishnan K, Kumar S. Testosterone plays a permissive role in angiotensin II-induced hypertension and cardiac hypertrophy in male rats. Biol Reprod. 2019;100(1):139–48. 10.1093/biolre/ioy179.PMID:30102356;PMCID:PMC6335213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Zhang D, Maalouf NM, Adams-Huet B, Moe OW, Sakhaee K. Effects of sex and postmenopausal estrogen use on serum phosphorus levels: a cross-sectional study of the National Health and Nutrition Examination Survey (NHANES) 2003–2006. Am J Kidney Dis. 2014;63(2):198–205. 10.1053/j.ajkd.2013.07.012. Epub 2013 Sep 17 PMID: 24051078. [DOI] [PubMed] [Google Scholar]
- 167.Hamm LL, Simon EE. Roles and mechanisms of urinary buffer excretion. Am J Physiol. 1987;253(4 Pt 2):F595–605. 10.1152/ajprenal.1987.253.4.F595. PMID: 3310662. [DOI] [PubMed] [Google Scholar]
- 168.Elalouf JM, Roinel N, de Rouffignac C. Effects of antidiuretic hormone on electrolyte reabsorption and secretion in distal tubules of rat kidney. Pflugers Arch. 1984;401(2):167–73. 10.1007/BF00583877. PMID: 6473069. [DOI] [PubMed] [Google Scholar]
- 169.Faroqui S, Levi M, Soleimani M, Amlal H. Estrogen downregulates the proximal tubule type IIa sodium phosphate cotransporter causing phosphate wasting and hypophosphatemia. Kidney Int. 2008 May;73(10):1141–50. 10.1038/ki.2008.33. Epub 2008 Feb 27. PMID: 18305465; PMCID: PMC2738940. [DOI] [PMC free article] [PubMed]
- 170.Everitt AV. The Urinary Excretion of Chloride, Phosphate, and Pigment in Ageing Male Rats. Gerontologia. 1958;2:123–35. 10.1159/000210732. [DOI] [PubMed] [Google Scholar]
- 171.de Borst MH, Vervloet MG, ter Wee PM, Navis G. Cross talk between the renin-angiotensin-aldosterone system and vitamin D-FGF-23-klotho in chronic kidney disease. J Am Soc Nephrol. 2011 Sep;22(9):1603–9. 10.1681/ASN.2010121251. Epub 2011 Aug 18. PMID: 21852584; PMCID: PMC3171931. [DOI] [PMC free article] [PubMed]
- 172.Macova M, Armando I, Zhou J, Baiardi G, Tyurmin D, Larrayoz-Roldan IM, Saavedra JM. Estrogen reduces aldosterone, upregulates adrenal angiotensin II AT2 receptors and normalizes adrenomedullary Fra-2 in ovariectomized rats. Neuroendocrinology. 2008;88(4):276–86. 10.1159/000150977. Epub 2008 Aug 4. PMID: 18679017; PMCID: PMC2677380. [DOI] [PMC free article] [PubMed]
- 173.Usberti M, Federico S, Di Minno G, et al. Effects of angiotensin II on plasma ADH, prostaglandin synthesis, and water excretion in normal humans. Am J Physiol. 1985;248(2 Pt 2):F254–9. 10.1152/ajprenal.1985.248.2.F254. PMID: 3881995. [DOI] [PubMed] [Google Scholar]
- 174.Maranduca MA, Clim A, Pinzariu AC, Statescu C, Sascau RA, Tanase DM, Serban DN, Branisteanu DC, Branisteanu DE, Huzum B, Serban IL. Role of arterial hypertension and angiotensin II in chronic kidney disease (Review). Exp Ther Med. 2023;25(4):153. 10.3892/etm.2023.11852.PMID:36911375;PMCID:PMC9996360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Qadri F, Culman J, Veltmar A, Maas K, Rascher W, Unger T. Angiotensin II-induced vasopressin release is mediated through alpha-1 adrenoceptors and angiotensin II AT1 receptors in the supraoptic nucleus. J Pharmacol Exp Ther. 1993;267(2):567–74 PMID: 8246129. [PubMed] [Google Scholar]
- 176.Navar LG. Intrarenal renin-angiotensin system in regulation of glomerular function. Curr Opin Nephrol Hypertens. 2014;23(1):38–45. 10.1097/01.mnh.0000436544.86508.f1.PMID:24275770;PMCID:PMC3982859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Torres-Pinzon DL, Ralph DL, Veiras LC, McDonough AA. Sex-specific adaptations to high-salt diet preserve electrolyte homeostasis with distinct sodium transporter profiles. Am J Physiol Cell Physiol. 2021 Nov 1;321(5):C897-C909. 10.1152/ajpcell.00282.2021. Epub 2021 Oct 6. PMID: 34613843; PMCID: PMC8616593. [DOI] [PMC free article] [PubMed]
- 178.Pan PP, Zhan QT, Le F, Zheng YM, Jin F. Angiotensin-converting enzymes play a dominant role in fertility. Int J Mol Sci. 2013;14(10):21071–86. 10.3390/ijms141021071.PMID:24152441;PMCID:PMC3821659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Wang K, Kestenbaum B. Proximal Tubular Secretory Clearance: A Neglected Partner of Kidney Function. Clin J Am Soc Nephrol. 2018 Aug 7;13(8):1291–1296. 10.2215/CJN.12001017. Epub 2018 Feb 28. PMID: 29490976; PMCID: PMC6086711. [DOI] [PMC free article] [PubMed]
- 180.Cowen LE, Hodak SP, Verbalis JG. Age-associated abnormalities of water homeostasis. Endocrinol Metab Clin North Am. 2013 Jun;42(2):349–70. 10.1016/j.ecl.2013.02.005. Epub 2013 Apr 17. PMID: 23702406; PMCID: PMC3682932. [DOI] [PMC free article] [PubMed]
- 181.Kuro-O M. Phosphate and Klotho. Kidney Int Suppl. 2011 Apr;79(121):S20–3. 10.1038/ki.2011.26. Epub 2011 Feb 23. PMID: 21346722; PMCID: PMC3260960. [DOI] [PMC free article] [PubMed]
- 182.Bachmann KN, Huang S, Lee H, Dichtel LE, Gupta DK, Burnett JC Jr, Miller KK, Wang TJ, Finkelstein JS. Effect of Testosterone on Natriuretic Peptide Levels. J Am Coll Cardiol. 2019;73(11):1288–96. 10.1016/j.jacc.2018.12.062.PMID:30898204;PMCID:PMC6588352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Carrivick SJ, Walsh JP, Brown SJ, Wardrop R, Hadlow NC. Brief report: Does PTH increase with age, independent of 25-hydroxyvitamin D, phosphate, renal function, and ionized calcium? J Clin Endocrinol Metab. 2015;100(5):2131–4. 10.1210/jc.2014-4370. Epub 2015 Mar 9 PMID: 25751107. [DOI] [PubMed] [Google Scholar]
- 184.Need AG, O’Loughlin PD, Morris HA, Horowitz M, Nordin BEC. The Effects of Age and Other Variables on Serum Parathyroid Hormone in Postmenopausal Women Attending an Osteoporosis Center. J Clin Endocrinol Metab. 2004;89(4):1646–9. 10.1210/jc.2003-031539. [DOI] [PubMed] [Google Scholar]
- 185.Sharma S, Katz R, Dubin RF, Drew DA, Gutierrez OM, Shlipak MG, Sarnak MJ, Ix JH. FGF23 and Cause-Specific Mortality in Community-Living Individuals-The Health, Aging, and Body Composition Study. J Am Geriatr Soc. 2021 Mar;69(3):711–717. 10.1111/jgs.16910. Epub 2020 Nov 10. PMID: 33170519; PMCID: PMC8175094. [DOI] [PMC free article] [PubMed]
- 186.Laffer CL, Elijovich F, Sahinoz M, Pitzer A, Kirabo A. New Insights into the Renin-Angiotensin System in Chronic Kidney Disease. Circ Res. 2020 Aug 14;127(5):607–609. 10.1161/CIRCRESAHA.120.317624. Epub 2020 Aug 13. PMID: 32790525; PMCID: PMC7430039. [DOI] [PMC free article] [PubMed]
- 187.Tank JE, Vora JP, Houghton DC, Anderson S. Altered renal vascular responses in the aging rat kidney. Am J Physiol. 1994 Jun;266(6 Pt2):F942–8. 10.1152/ajprenal.1994.266.6.F942. PMID: 8023973. [DOI] [PubMed]
- 188.Perucca J, Bichet DG, Bardoux P, Bouby N, Bankir L. Sodium excretion in response to vasopressin and selective vasopressin receptor antagonists. J Am Soc Nephrol. 2008 Sep;19(9):1721–31. 10.1681/ASN.2008010021. Epub 2008 Jul 2. PMID: 18596120; PMCID: PMC2518442. [DOI] [PMC free article] [PubMed]
- 189.Arruda JA, Stipanuk S, Walter R, Kurtzman NA. Effect of vasopressin administration on sodium excretion and plasma phosphate concentration. Proc Soc Exp Biol Med. 1977;155(3):308–13. 10.3181/00379727-155-39796. PMID: 877126. [DOI] [PubMed] [Google Scholar]
- 190.Tozuka K, Hara Y, Moriguchi H, Kikuchi T, Tokue A. [The role of urine pH in the occurrence of phosphaturia]. Hinyokika Kiyo. 1989 Sep;35(9):1475–8. Japanese. PMID: 2816612. [PubMed]
- 191.Sachse A, Wolf G. Angiotensin II-induced reactive oxygen species and the kidney. J Am Soc Nephrol. 2007;18(9):2439–46. 10.1681/ASN.2007020149. Epub 2007 Aug 8 PMID: 17687073. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The supplementary Excel file includes the datasets used and/or analyzed during the current study (Supplementary Material 15). Further information is available from the corresponding author upon reasonable request.