Abstract
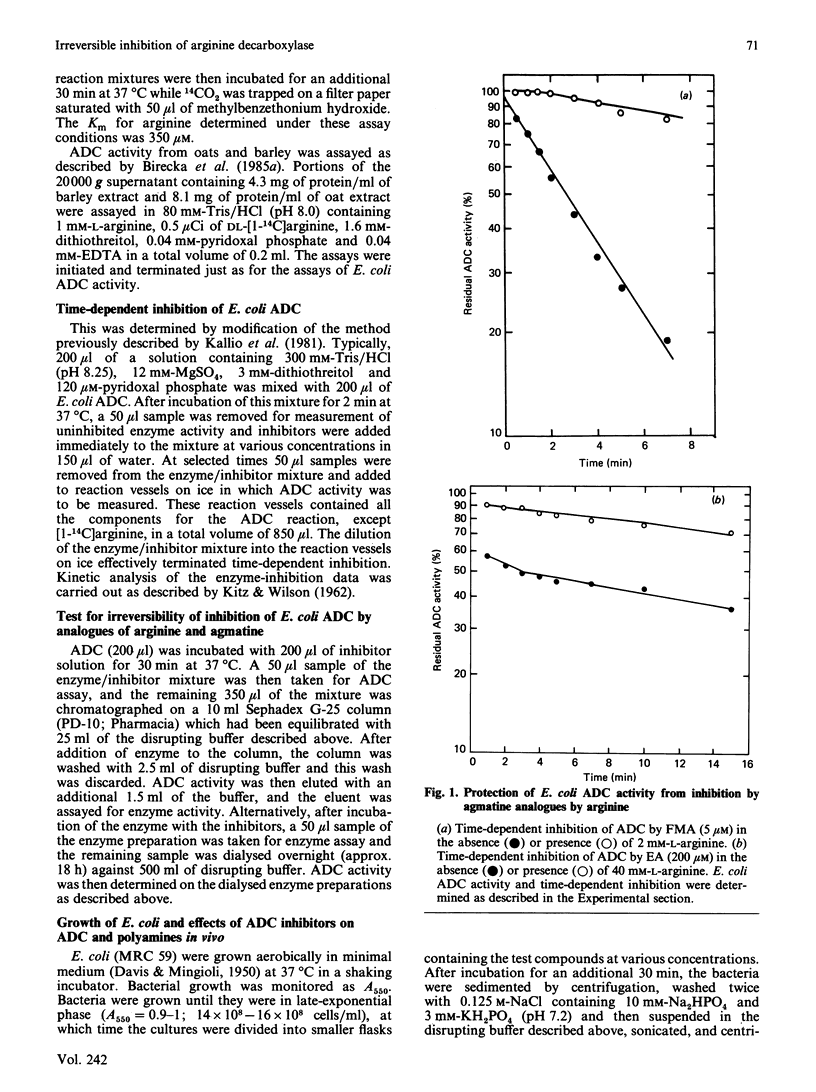
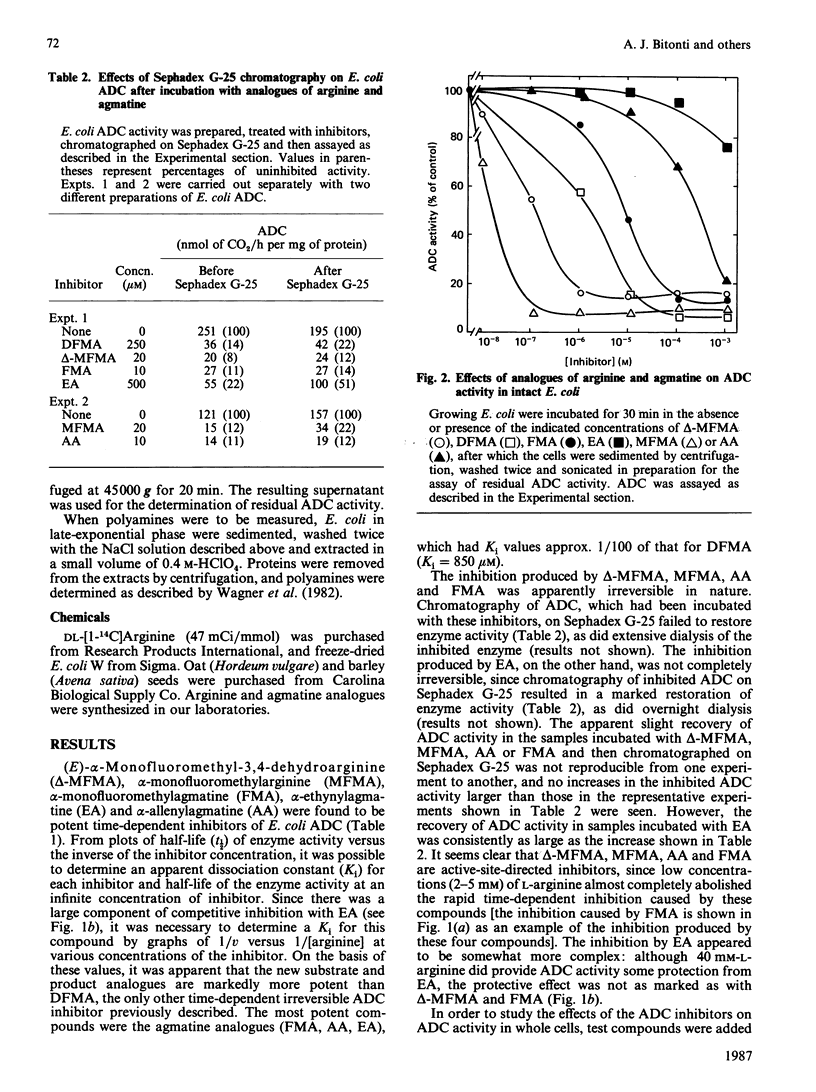
Arginine decarboxylase (ADC) activity from Escherichia coli and two plant species (oats and barley) was inhibited by five new substrate (arginine) and product (agmatine) analogues. The five compounds, (E)-alpha-monofluoromethyldehydroarginine (delta-MFMA), alpha-monofluoromethylarginine (MFMA), alpha-monofluoromethylagatine (FMA), alpha-ethynylagmatine (EA) and alpha-allenylagmatine (AA), were all more potent inhibitors of ADC activity than was alpha-difluoromethylarginine (DFMA), the only irreversible inhibitor of this enzyme described previously. The inhibition caused by the five compounds was apparently enzyme-activated and irreversible, since the loss of enzyme activity followed pseudo-first-order kinetics, was time-dependent, the natural substrate of ADC (arginine) blocked the effects of the inhibitors, and the inhibition remained after chromatography of inhibited ADC on Sephadex G-25 or on overnight dialysis of the enzyme. DFMA, FMA, delta-MFMA and MFMA were effective at very low concentrations (10 nM-10 microM) at inhibiting ADC activity in growing E. coli. FMA was also shown to deplete putrescine effectively in E. coli, particularly when combined with an inhibitor of ornithine decarboxylase, alpha-monofluoromethyl-putrescine. The potential uses of the compounds for the study of the role of polyamine biosynthesis in bacteria and plants is discussed.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Birecka H., Bitonti A. J., McCann P. P. Activities of arginine and ornithine decarboxylases in various plant species. Plant Physiol. 1985 Oct;79(2):515–519. doi: 10.1104/pp.79.2.515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birecka H., Bitonti A. J., McCann P. P. Assaying ornithine and arginine decarboxylases in some plant species. Plant Physiol. 1985 Oct;79(2):509–514. doi: 10.1104/pp.79.2.509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bitonti A. J., Kelly S. E., McCann P. P. Regulation of growth and macromolecular synthesis by putrescine and spermidine in Pseudomonas aeruginosa. Life Sci. 1984 Apr 16;34(16):1513–1520. doi: 10.1016/0024-3205(84)90605-2. [DOI] [PubMed] [Google Scholar]
- Bitonti A. J., McCann P. P., Sjoerdsma A. Restriction of bacterial growth by inhibition of polyamine biosynthesis by using monofluoromethylornithine, difluoromethylarginine and dicyclohexylammonium sulphate. Biochem J. 1982 Nov 15;208(2):435–441. doi: 10.1042/bj2080435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAVIS B. D., MINGIOLI E. S. Mutants of Escherichia coli requiring methionine or vitamin B12. J Bacteriol. 1950 Jul;60(1):17–28. doi: 10.1128/jb.60.1.17-28.1950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai Y. R., Kaur-Sawhney R., Galston A. W. Promotion by gibberellic Acid of polyamine biosynthesis in internodes of light-grown dwarf peas. Plant Physiol. 1982 Jan;69(1):103–106. doi: 10.1104/pp.69.1.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feirer R. P., Mignon G., Litvay J. D. Arginine decarboxylase and polyamines required for embryogenesis in the wild carrot. Science. 1984 Mar 30;223(4643):1433–1435. doi: 10.1126/science.223.4643.1433. [DOI] [PubMed] [Google Scholar]
- Flores H. E., Galston A. W. Polyamines and plant stress: activation of putrescine biosynthesis by osmotic shock. Science. 1982 Sep 24;217(4566):1259–1261. doi: 10.1126/science.217.4566.1259. [DOI] [PubMed] [Google Scholar]
- Gale E. F. The production of amines by bacteria: The decarboxylation of amino-acids by strains of Bacterium coli. Biochem J. 1940 Mar;34(3):392–413. doi: 10.1042/bj0340392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KITZ R., WILSON I. B. Esters of methanesulfonic acid as irreversible inhibitors of acetylcholinesterase. J Biol Chem. 1962 Oct;237:3245–3249. [PubMed] [Google Scholar]
- Kallio A., McCann P. P., Bey P. DL-a-Monofluoromethylputrescine is a potent irreversible inhibitor of Escherichia coli ornithine decarboxylase. Biochem J. 1982 Jun 15;204(3):771–775. doi: 10.1042/bj2040771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kallio A., McCann P. P., Bey P. DL-alpha-(Difluoromethyl)arginine: a potent enzyme-activated irreversible inhibitor of bacterial decarboxylases. Biochemistry. 1981 May 26;20(11):3163–3168. doi: 10.1021/bi00514a027. [DOI] [PubMed] [Google Scholar]
- Kallio A., McCann P. P. Difluoromethylornithine irreversibly inactivates ornithine decarboxylase of Pseudomonas aeruginosa, but does not inhibit the enzymes of Escherichia coli. Biochem J. 1981 Oct 15;200(1):69–75. doi: 10.1042/bj2000069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Morris D. R., Pardee A. B. Multiple pathways of putrescine biosynthesis in Escherichia coli. J Biol Chem. 1966 Jul 10;241(13):3129–3135. [PubMed] [Google Scholar]
- Pegg A. E., McCann P. P. Polyamine metabolism and function. Am J Physiol. 1982 Nov;243(5):C212–C221. doi: 10.1152/ajpcell.1982.243.5.C212. [DOI] [PubMed] [Google Scholar]
- Slocum R. D., Kaur-Sawhney R., Galston A. W. The physiology and biochemistry of polyamines in plants. Arch Biochem Biophys. 1984 Dec;235(2):283–303. doi: 10.1016/0003-9861(84)90201-7. [DOI] [PubMed] [Google Scholar]
- Tabor C. W., Tabor H. Polyamines in microorganisms. Microbiol Rev. 1985 Mar;49(1):81–99. doi: 10.1128/mr.49.1.81-99.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner J., Danzin C., Mamont P. Reversed-phase ion-pair liquid chromatographic procedure for the simultaneous analysis of S-adenosylmethionine, its metabolites and the natural polyamines. J Chromatogr. 1982 Feb 12;227(2):349–368. doi: 10.1016/s0378-4347(00)80389-8. [DOI] [PubMed] [Google Scholar]
- Wu W. H., Morris D. R. Biosynthetic arginine decarboxylase from Escherichia coli. Purification and properties. J Biol Chem. 1973 Mar 10;248(5):1687–1695. [PubMed] [Google Scholar]


