Abstract
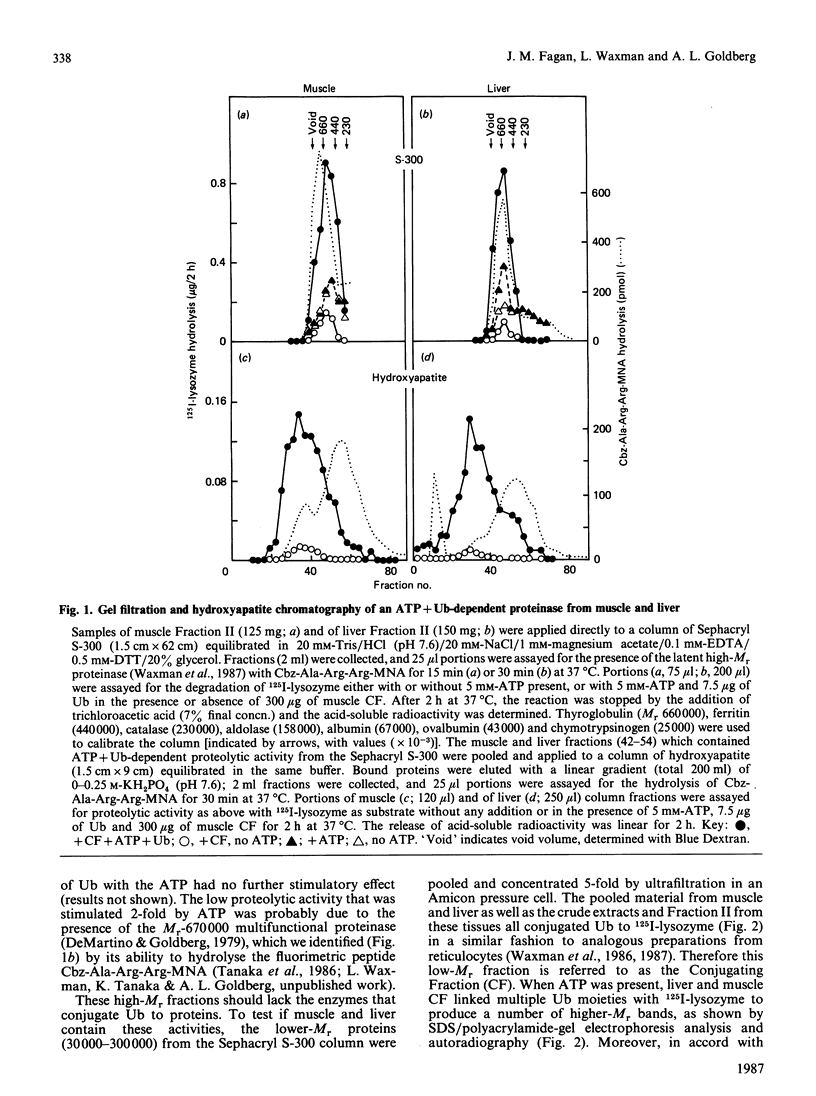
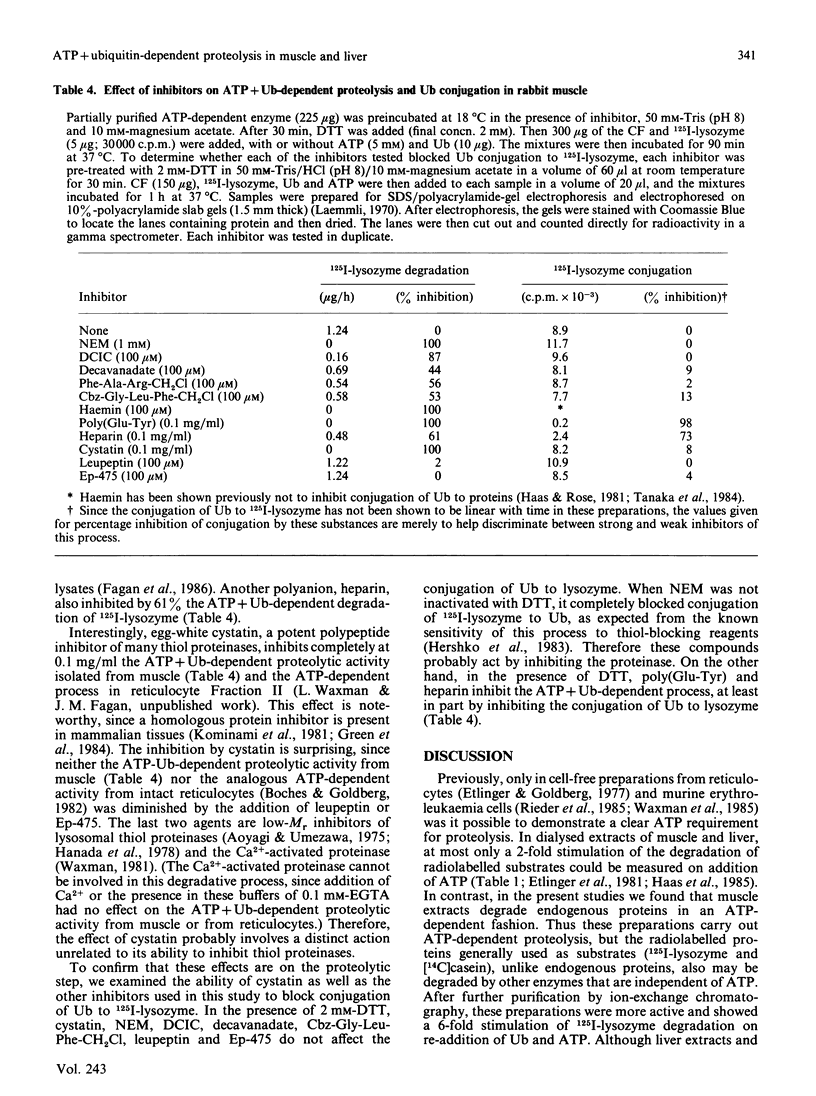
Although protein breakdown in most cells seems to require metabolic energy, it has only been possible to establish a soluble ATP-dependent proteolytic system in extracts of reticulocytes and erythroleukemia cells. We have now succeeded in demonstrating in soluble extracts and more purified preparations from rabbit skeletal muscle a 12-fold stimulation by ATP of breakdown of endogenous proteins and a 6-fold stimulation of 125I-lysozyme degradation. However, it has still not been possible to demonstrate such large effects of ATP in similar preparations from liver. Nevertheless, after fractionation by DEAE-chromatography and gel filtration, we found that extracts from liver as well as muscle contain both the enzymes which conjugate ubiquitin to 125I-lysozyme and an enzyme which specifically degrades the ubiquitin-protein conjugates. When this proteolytic activity was recombined with the conjugating enzymes, ATP + ubiquitin-dependent degradation of many proteins was observed. This proteinase is unusually large, approx. 1500 kDa, requires ATP hydrolysis for activity and resembles the ubiquitin-protein-conjugate degrading activity isolated from reticulocytes. Thus the ATP + ubiquitin-dependent pathway is likely to be present in all mammalian cells, although certain tissues may contain inhibitory factors.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Boches F. S., Goldberg A. L. Role for the adenosine triphosphate-dependent proteolytic pathway in reticulocyte maturation. Science. 1982 Feb 19;215(4535):978–980. doi: 10.1126/science.7156977. [DOI] [PubMed] [Google Scholar]
- Charette M. F., Henderson G. W., Markovitz A. ATP hydrolysis-dependent protease activity of the lon (capR) protein of Escherichia coli K-12. Proc Natl Acad Sci U S A. 1981 Aug;78(8):4728–4732. doi: 10.1073/pnas.78.8.4728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chin D. T., Carlson N., Kuehl L., Rechsteiner M. The degradation of guanidinated lysozyme in reticulocyte lysate. J Biol Chem. 1986 Mar 15;261(8):3883–3890. [PubMed] [Google Scholar]
- Choate G., Mansour T. E. Studies on heart phosphofructokinase. Decavanadate as a potent allosteric inhibitor at alkaline and acidic pH. J Biol Chem. 1979 Nov 25;254(22):11457–11462. [PubMed] [Google Scholar]
- Chung C. H., Goldberg A. L. The product of the lon (capR) gene in Escherichia coli is the ATP-dependent protease, protease La. Proc Natl Acad Sci U S A. 1981 Aug;78(8):4931–4935. doi: 10.1073/pnas.78.8.4931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciechanover A., Elias S., Heller H., Hershko A. "Covalent affinity" purification of ubiquitin-activating enzyme. J Biol Chem. 1982 Mar 10;257(5):2537–2542. [PubMed] [Google Scholar]
- Ciechanover A., Finley D., Varshavsky A. Ubiquitin dependence of selective protein degradation demonstrated in the mammalian cell cycle mutant ts85. Cell. 1984 May;37(1):57–66. doi: 10.1016/0092-8674(84)90300-3. [DOI] [PubMed] [Google Scholar]
- Ciehanover A., Hod Y., Hershko A. A heat-stable polypeptide component of an ATP-dependent proteolytic system from reticulocytes. Biochem Biophys Res Commun. 1978 Apr 28;81(4):1100–1105. doi: 10.1016/0006-291x(78)91249-4. [DOI] [PubMed] [Google Scholar]
- Dahlmann B., Kuehn L., Reinauer H. Identification of three high molecular mass cysteine proteinases from rat skeletal muscle. FEBS Lett. 1983 Aug 22;160(1-2):243–247. doi: 10.1016/0014-5793(83)80975-2. [DOI] [PubMed] [Google Scholar]
- Dahlmann B., Kuehn L., Rutschmann M., Reinauer H. Purification and characterization of a multicatalytic high-molecular-mass proteinase from rat skeletal muscle. Biochem J. 1985 May 15;228(1):161–170. doi: 10.1042/bj2280161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeMartino G. N., Goldberg A. L. Identification and partial purification of an ATP-stimulated alkaline protease in rat liver. J Biol Chem. 1979 May 25;254(10):3712–3715. [PubMed] [Google Scholar]
- DeMartino G. N. Identification of a high molecular weight alkaline protease in rat heart. J Mol Cell Cardiol. 1983 Jan;15(1):17–29. doi: 10.1016/0022-2828(83)90304-8. [DOI] [PubMed] [Google Scholar]
- DeMaster E. G., Mitchell A. A comparison of arsenate and vanadate as inhibitors or uncouplers of mitochondrial and glycolytic energy metabolism. Biochemistry. 1973 Sep 11;12(19):3616–3621. doi: 10.1021/bi00743a007. [DOI] [PubMed] [Google Scholar]
- Desautels M., Goldberg A. L. Liver mitochondria contain an ATP-dependent, vanadate-sensitive pathway for the degradation of proteins. Proc Natl Acad Sci U S A. 1982 Mar;79(6):1869–1873. doi: 10.1073/pnas.79.6.1869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Etlinger J. D., Goldberg A. L. A soluble ATP-dependent proteolytic system responsible for the degradation of abnormal proteins in reticulocytes. Proc Natl Acad Sci U S A. 1977 Jan;74(1):54–58. doi: 10.1073/pnas.74.1.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Etlinger J. D., Goldberg A. L. Control of protein degradation in reticulocytes and reticulocyte extracts by hemin. J Biol Chem. 1980 May 25;255(10):4563–4568. [PubMed] [Google Scholar]
- Etlinger J. D., Speiser S., Wajnberg E., Glucksman M. J. ATP-dependent proteolysis in erythroid and muscle cells. Acta Biol Med Ger. 1981;40(10-11):1285–1291. [PubMed] [Google Scholar]
- Fagan J. M., Waxman L., Goldberg A. L. Red blood cells contain a pathway for the degradation of oxidant-damaged hemoglobin that does not require ATP or ubiquitin. J Biol Chem. 1986 May 5;261(13):5705–5713. [PubMed] [Google Scholar]
- Finley D., Ciechanover A., Varshavsky A. Thermolability of ubiquitin-activating enzyme from the mammalian cell cycle mutant ts85. Cell. 1984 May;37(1):43–55. doi: 10.1016/0092-8674(84)90299-x. [DOI] [PubMed] [Google Scholar]
- Fulks R. M., Li J. B., Goldberg A. L. Effects of insulin, glucose, and amino acids on protein turnover in rat diaphragm. J Biol Chem. 1975 Jan 10;250(1):290–298. [PubMed] [Google Scholar]
- Goldberg A. L., St John A. C. Intracellular protein degradation in mammalian and bacterial cells: Part 2. Annu Rev Biochem. 1976;45:747–803. doi: 10.1146/annurev.bi.45.070176.003531. [DOI] [PubMed] [Google Scholar]
- Green G. D., Kembhavi A. A., Davies M. E., Barrett A. J. Cystatin-like cysteine proteinase inhibitors from human liver. Biochem J. 1984 Mar 15;218(3):939–946. doi: 10.1042/bj2180939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gronostajski R. M., Pardee A. B., Goldberg A. L. The ATP dependence of the degradation of short- and long-lived proteins in growing fibroblasts. J Biol Chem. 1985 Mar 25;260(6):3344–3349. [PubMed] [Google Scholar]
- Guidotti G. Studies on the chemistry of hemoglobin. I. The reactive sulfhydryl groups. J Biol Chem. 1967 Aug 25;242(16):3673–3684. [PubMed] [Google Scholar]
- Haas A. L., Murphy K. E., Bright P. M. The inactivation of ubiquitin accounts for the inability to demonstrate ATP, ubiquitin-dependent proteolysis in liver extracts. J Biol Chem. 1985 Apr 25;260(8):4694–4703. [PubMed] [Google Scholar]
- Haas A. L., Rose I. A. Hemin inhibits ATP-dependent ubiquitin-dependent proteolysis: role of hemin in regulating ubiquitin conjugate degradation. Proc Natl Acad Sci U S A. 1981 Nov;78(11):6845–6848. doi: 10.1073/pnas.78.11.6845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harper J. W., Hemmi K., Powers J. C. Reaction of serine proteases with substituted isocoumarins: discovery of 3,4-dichloroisocoumarin, a new general mechanism based serine protease inhibitor. Biochemistry. 1985 Apr 9;24(8):1831–1841. doi: 10.1021/bi00329a005. [DOI] [PubMed] [Google Scholar]
- Hershko A., Ciechanover A., Heller H., Haas A. L., Rose I. A. Proposed role of ATP in protein breakdown: conjugation of protein with multiple chains of the polypeptide of ATP-dependent proteolysis. Proc Natl Acad Sci U S A. 1980 Apr;77(4):1783–1786. doi: 10.1073/pnas.77.4.1783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershko A., Ciechanover A. Mechanisms of intracellular protein breakdown. Annu Rev Biochem. 1982;51:335–364. doi: 10.1146/annurev.bi.51.070182.002003. [DOI] [PubMed] [Google Scholar]
- Hershko A., Heller H., Elias S., Ciechanover A. Components of ubiquitin-protein ligase system. Resolution, affinity purification, and role in protein breakdown. J Biol Chem. 1983 Jul 10;258(13):8206–8214. [PubMed] [Google Scholar]
- Hershko A., Leshinsky E., Ganoth D., Heller H. ATP-dependent degradation of ubiquitin-protein conjugates. Proc Natl Acad Sci U S A. 1984 Mar;81(6):1619–1623. doi: 10.1073/pnas.81.6.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershko A., Tomkins G. M. Studies on the degradation of tyrosine aminotransferase in hepatoma cells in culture. Influence of the composition of the medium and adenosine triphosphate dependence. J Biol Chem. 1971 Feb 10;246(3):710–714. [PubMed] [Google Scholar]
- Hershko A. Ubiquitin: roles in protein modification and breakdown. Cell. 1983 Aug;34(1):11–12. doi: 10.1016/0092-8674(83)90131-9. [DOI] [PubMed] [Google Scholar]
- Hough R., Pratt G., Rechsteiner M. Ubiquitin-lysozyme conjugates. Identification and characterization of an ATP-dependent protease from rabbit reticulocyte lysates. J Biol Chem. 1986 Feb 15;261(5):2400–2408. [PubMed] [Google Scholar]
- Hough R., Rechsteiner M. Ubiquitin-lysozyme conjugates. Purification and susceptibility to proteolysis. J Biol Chem. 1986 Feb 15;261(5):2391–2399. [PubMed] [Google Scholar]
- Katznelson R., Kulka R. G. Effects of denaturation and methylation on the degradation of proteins in cultured hepatoma cells and in reticulocyte cell-free systems. Eur J Biochem. 1985 Jan 15;146(2):437–442. doi: 10.1111/j.1432-1033.1985.tb08670.x. [DOI] [PubMed] [Google Scholar]
- Kettner C., Shaw E. Inactivation of trypsin-like enzymes with peptides of arginine chloromethyl ketone. Methods Enzymol. 1981;80(Pt 100):826–842. doi: 10.1016/s0076-6879(81)80065-1. [DOI] [PubMed] [Google Scholar]
- Kominami E., Wakamatsu N., Katunuma N. Endogenous thiol protease inhibitor from rat liver. Biochem Biophys Res Commun. 1981 Mar 31;99(2):568–575. doi: 10.1016/0006-291x(81)91783-6. [DOI] [PubMed] [Google Scholar]
- Kulka R. G., Cohen H. Regulation of glutamine synthetase activity of hepatoma tissue culture cells by glutamine and dexamethasone. J Biol Chem. 1973 Oct 10;248(19):6738–6743. [PubMed] [Google Scholar]
- Kurachi K., Powers J. C., Wilcox P. E. Kinetics of the reaction of chymotrypsin A with peptide chloromethyl ketones in relation to its subsite specificity. Biochemistry. 1973 Feb;12(4):771–777. doi: 10.1021/bi00728a029. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Larimore F. S., Waxman L., Goldberg A. L. Studies of the ATP-dependent proteolytic enzyme, protease La, from Escherichia coli. J Biol Chem. 1982 Apr 25;257(8):4187–4195. [PubMed] [Google Scholar]
- Magnani M., Stocchi V., Chiarantini L., Serafini G., Dachà M., Fornaini G. Rabbit red blood cell hexokinase. Decay mechanism during reticulocyte maturation. J Biol Chem. 1986 Jun 25;261(18):8327–8333. [PubMed] [Google Scholar]
- Milman G., Portnoff L. S., Tiemeier D. C. Immunochemical evidence for glutamine-mediated degradation of glutamine synthetase in cultured Chinese hamster cells. J Biol Chem. 1975 Feb 25;250(4):1393–1399. [PubMed] [Google Scholar]
- Moore A. T., Williams K. E., Lloyd J. B. The effect of chemical treatments of albumin and orosomucoid on rate of clearance from the rat bloodstream and rate of pinocytic capture of rat yolk sac cultured in vitro. Biochem J. 1977 Jun 15;164(3):607–616. doi: 10.1042/bj1640607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller M., Dubiel W., Rathmann J., Rapoport S. Determination and characteristics of energy-dependent proteolysis in rabbit reticulocytes. Eur J Biochem. 1980 Aug;109(2):405–410. doi: 10.1111/j.1432-1033.1980.tb04808.x. [DOI] [PubMed] [Google Scholar]
- Rice R. H., Means G. E. Radioactive labeling of proteins in vitro. J Biol Chem. 1971 Feb 10;246(3):831–832. [PubMed] [Google Scholar]
- Rieder R. F., Ibrahim A., Etlinger J. D. A particle-associated ATP-dependent proteolytic activity in erythroleukemia cells. J Biol Chem. 1985 Feb 25;260(4):2015–2018. [PubMed] [Google Scholar]
- Rose I. A., Warms J. V., Hershko A. A high molecular weight protease in liver cytosol. J Biol Chem. 1979 Sep 10;254(17):8135–8138. [PubMed] [Google Scholar]
- SIMPSON M. V. The release of labeled amino acids from the proteins of rat liver slices. J Biol Chem. 1953 Mar;201(1):143–154. [PubMed] [Google Scholar]
- Speiser S., Etlinger J. D. Loss of ATP-dependent proteolysis with maturation of reticulocytes and erythrocytes. J Biol Chem. 1982 Dec 10;257(23):14122–14127. [PubMed] [Google Scholar]
- Tanaka K., Ii K., Ichihara A., Waxman L., Goldberg A. L. A high molecular weight protease in the cytosol of rat liver. I. Purification, enzymological properties, and tissue distribution. J Biol Chem. 1986 Nov 15;261(32):15197–15203. [PubMed] [Google Scholar]
- Tanaka K., Waxman L., Goldberg A. L. ATP serves two distinct roles in protein degradation in reticulocytes, one requiring and one independent of ubiquitin. J Cell Biol. 1983 Jun;96(6):1580–1585. doi: 10.1083/jcb.96.6.1580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka K., Waxman L., Goldberg A. L. Vanadate inhibits the ATP-dependent degradation of proteins in reticulocytes without affecting ubiquitin conjugation. J Biol Chem. 1984 Mar 10;259(5):2803–2809. [PubMed] [Google Scholar]
- Turk V., Brzin J., Lenarcic B., Locnikar P., Popović T., Ritonja A., Babnik J., Bode W., Machleidt W. Structure and function of lysosomal cysteine proteinases and their protein inhibitors. Prog Clin Biol Res. 1985;180:91–103. [PubMed] [Google Scholar]
- Vijaya S., Crane F. L., Ramasarma T. A vanadate-stimulated NADH oxidase in erythrocyte membrane generates hydrogen peroxide. Mol Cell Biochem. 1984 Jun;62(2):175–185. doi: 10.1007/BF00223308. [DOI] [PubMed] [Google Scholar]
- Watabe S., Kimura T. ATP-dependent protease in bovine adrenal cortex. Tissue specificity, subcellular localization, and partial characterization. J Biol Chem. 1985 May 10;260(9):5511–5517. [PubMed] [Google Scholar]
- Waxman L. Calcium-activated proteases in mammalian tissues. Methods Enzymol. 1981;80(Pt 100):664–680. doi: 10.1016/s0076-6879(81)80051-1. [DOI] [PubMed] [Google Scholar]
- Waxman L., Fagan J. M., Tanaka K., Goldberg A. L. A soluble ATP-dependent system for protein degradation from murine erythroleukemia cells. Evidence for a protease which requires ATP hydrolysis but not ubiquitin. J Biol Chem. 1985 Oct 5;260(22):11994–12000. [PubMed] [Google Scholar]
- Waxman L., Goldberg A. L. Protease La from Escherichia coli hydrolyzes ATP and proteins in a linked fashion. Proc Natl Acad Sci U S A. 1982 Aug;79(16):4883–4887. doi: 10.1073/pnas.79.16.4883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waxman L., Goldberg A. L. Protease La, the lon gene product, cleaves specific fluorogenic peptides in an ATP-dependent reaction. J Biol Chem. 1985 Oct 5;260(22):12022–12028. [PubMed] [Google Scholar]
- Wilk S., Orlowski M. Cation-sensitive neutral endopeptidase: isolation and specificity of the bovine pituitary enzyme. J Neurochem. 1980 Nov;35(5):1172–1182. doi: 10.1111/j.1471-4159.1980.tb07873.x. [DOI] [PubMed] [Google Scholar]
- Wilk S., Orlowski M. Evidence that pituitary cation-sensitive neutral endopeptidase is a multicatalytic protease complex. J Neurochem. 1983 Mar;40(3):842–849. doi: 10.1111/j.1471-4159.1983.tb08056.x. [DOI] [PubMed] [Google Scholar]
- Wilkinson K. D., Urban M. K., Haas A. L. Ubiquitin is the ATP-dependent proteolysis factor I of rabbit reticulocytes. J Biol Chem. 1980 Aug 25;255(16):7529–7532. [PubMed] [Google Scholar]



