Abstract
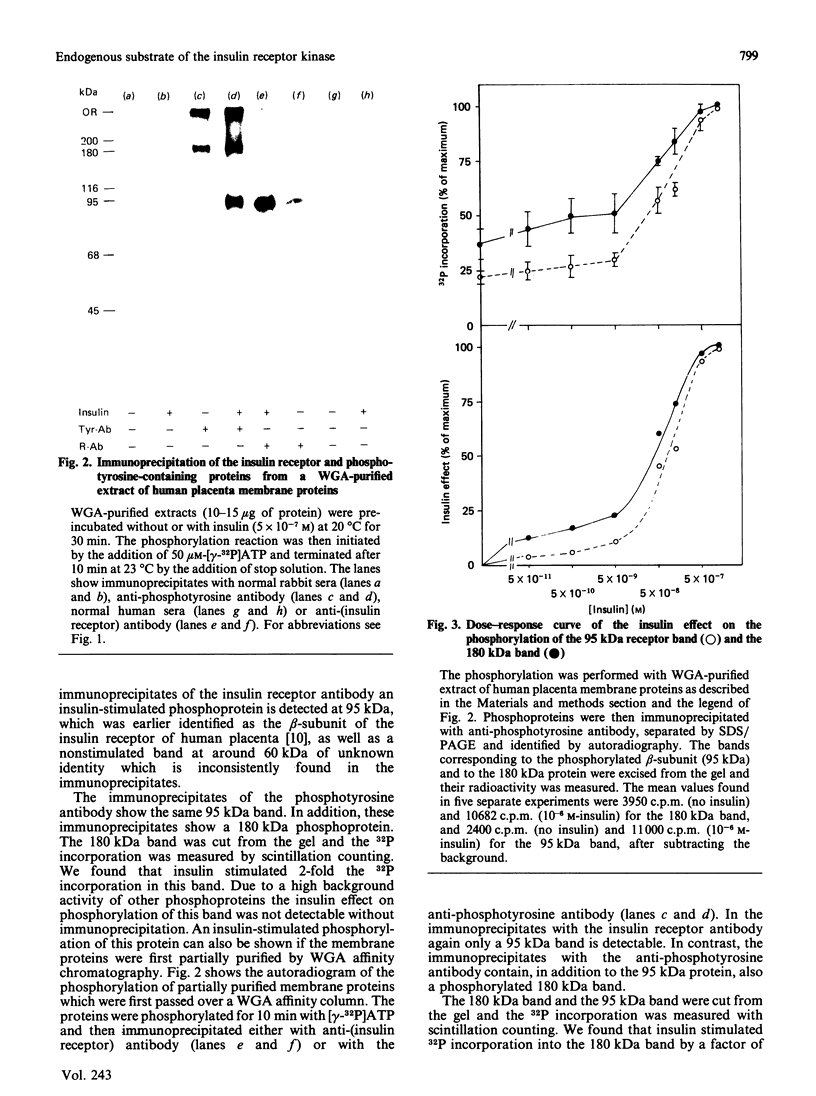
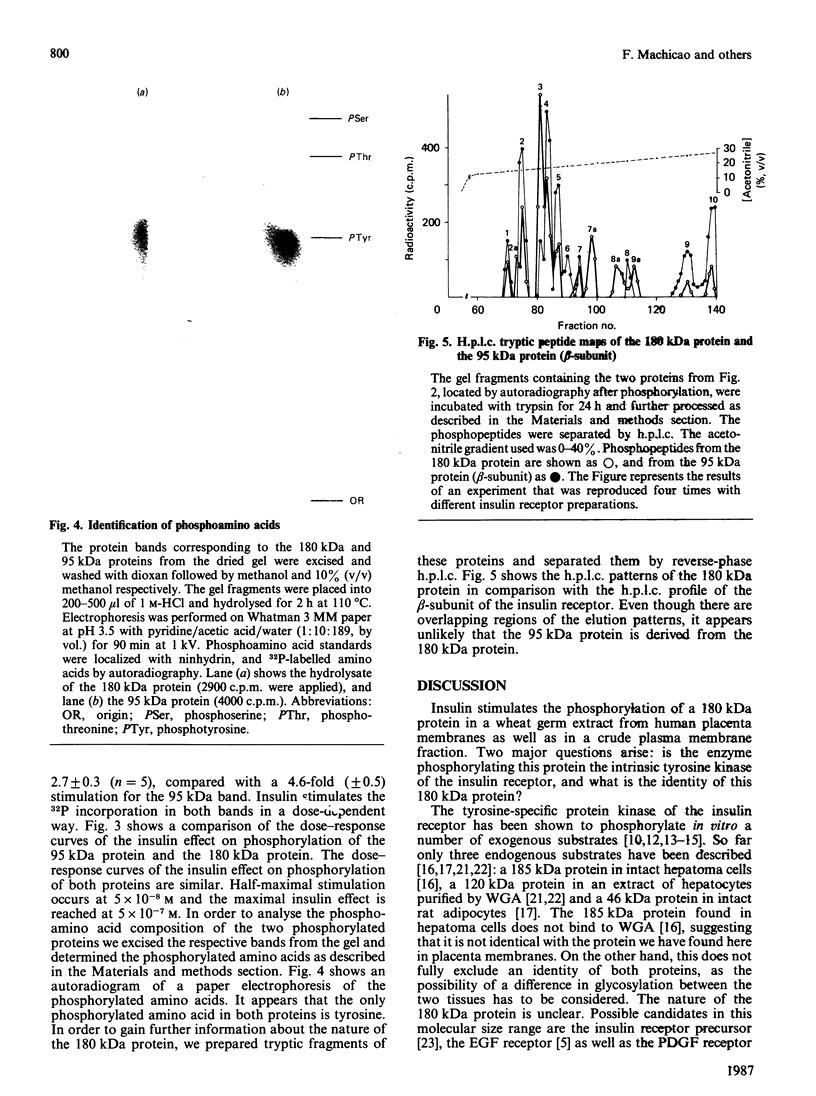
The beta-subunit of the insulin receptor contains a tyrosine-specific protein kinase. Insulin binding activates this kinase and causes phosphorylation of the beta-subunit of the insulin receptor. It is believed that phosphorylation of other proteins might transmit the insulin signal from the receptor to the cell. In the present study we used a polyclonal anti-phosphotyrosine antibody to detect other proteins that become tyrosine phosphorylated upon insulin stimulation. Glycoproteins from human placenta membranes were enriched by wheat germ agglutinin chromatography and phosphorylation was studied with [gamma-32P]ATP and insulin in vitro. Phosphorylated proteins were immunoprecipitated by antibodies against the insulin receptor and by serum containing the anti-phosphotyrosine antibody. Beside the insulin-stimulated phosphorylation of the 95 kDa beta-subunit of the insulin receptor, an insulin-stimulated phosphorylation of a 180 kDa protein was found. The phosphorylation of both proteins occurred only on tyrosine residues. Insulin increased 32P incorporation into the 180 kDa band 2.7-fold (S.E.M. +/- 0.3, n = 5). The 180 kDa protein was not precipitated by antibodies against the insulin receptor. H.p.l.c. chromatograms of tryptic fragments of the phosphorylated 180 kDa protein and of the beta-subunit of the insulin receptor revealed different patterns for both proteins. Insulin-stimulated phosphorylation of the 180 kDa protein was also detectable in unfractionated detergent-solubilized membranes. The phosphorylation of the 180 kDa protein was stimulated by insulin with the same dose-response curve as the phosphorylation of the beta-subunit, suggesting that this protein might be another endogenous substrate of the insulin receptor kinase.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Avruch J., Nemenoff R. A., Blackshear P. J., Pierce M. W., Osathanondh R. Insulin-stimulated tyrosine phosphorylation of the insulin receptor in detergent extracts of human placental membranes. Comparison to epidermal growth factor-stimulated phosphorylation. J Biol Chem. 1982 Dec 25;257(24):15162–15166. [PubMed] [Google Scholar]
- Cooper J. A., Hunter T. Regulation of cell growth and transformation by tyrosine-specific protein kinases: the search for important cellular substrate proteins. Curr Top Microbiol Immunol. 1983;107:125–161. doi: 10.1007/978-3-642-69075-4_4. [DOI] [PubMed] [Google Scholar]
- Ek B., Westermark B., Wasteson A., Heldin C. H. Stimulation of tyrosine-specific phosphorylation by platelet-derived growth factor. Nature. 1982 Feb 4;295(5848):419–420. doi: 10.1038/295419a0. [DOI] [PubMed] [Google Scholar]
- Hedo J. A., Gorden P. Biosynthesis of the insulin receptor. Horm Metab Res. 1985 Oct;17(10):487–490. doi: 10.1055/s-2007-1013586. [DOI] [PubMed] [Google Scholar]
- Hunter T., Sefton B. M. Transforming gene product of Rous sarcoma virus phosphorylates tyrosine. Proc Natl Acad Sci U S A. 1980 Mar;77(3):1311–1315. doi: 10.1073/pnas.77.3.1311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Häring H. U., Kasuga M., Kahn C. R. Insulin receptor phosphorylation in intact adipocytes and in a cell-free system. Biochem Biophys Res Commun. 1982 Oct 29;108(4):1538–1545. doi: 10.1016/s0006-291x(82)80082-x. [DOI] [PubMed] [Google Scholar]
- Häring H. U., White M. F., Machicao F., Ermel B., Schleicher E., Obermaier B. Insulin rapidly stimulates phosphorylation of a 46-kDa membrane protein on tyrosine residues as well as phosphorylation of several soluble proteins in intact fat cells. Proc Natl Acad Sci U S A. 1987 Jan;84(1):113–117. doi: 10.1073/pnas.84.1.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasuga M., Fujita-Yamaguchi Y., Blithe D. L., Kahn C. R. Tyrosine-specific protein kinase activity is associated with the purified insulin receptor. Proc Natl Acad Sci U S A. 1983 Apr;80(8):2137–2141. doi: 10.1073/pnas.80.8.2137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasuga M., Karlsson F. A., Kahn C. R. Insulin stimulates the phosphorylation of the 95,000-dalton subunit of its own receptor. Science. 1982 Jan 8;215(4529):185–187. doi: 10.1126/science.7031900. [DOI] [PubMed] [Google Scholar]
- Kasuga M., Zick Y., Blith D. L., Karlsson F. A., Häring H. U., Kahn C. R. Insulin stimulation of phosphorylation of the beta subunit of the insulin receptor. Formation of both phosphoserine and phosphotyrosine. J Biol Chem. 1982 Sep 10;257(17):9891–9894. [PubMed] [Google Scholar]
- Machicao F., Urumow T., Wieland O. H. Evidence for phosphorylation of actin by the insulin receptor-associated protein kinase from human placenta. FEBS Lett. 1983 Oct 31;163(1):76–80. doi: 10.1016/0014-5793(83)81167-3. [DOI] [PubMed] [Google Scholar]
- Machicao F., Urumow T., Wieland O. H. Phosphorylation--dephosphorylation of purified insulin receptor from human placenta. Effect of insulin. FEBS Lett. 1982 Nov 22;149(1):96–100. doi: 10.1016/0014-5793(82)81079-x. [DOI] [PubMed] [Google Scholar]
- Machicao F., Wieland O. H. Insulin-stimulated phosphorylation of actin by human placental insulin receptor preparations. Curr Top Cell Regul. 1985;27:95–105. doi: 10.1016/b978-0-12-152827-0.50015-3. [DOI] [PubMed] [Google Scholar]
- Pang D. T., Sharma B. R., Shafer J. A. Purification of the catalytically active phosphorylated form of insulin receptor kinase by affinity chromatography with O-phosphotyrosyl-binding antibodies. Arch Biochem Biophys. 1985 Oct;242(1):176–186. doi: 10.1016/0003-9861(85)90491-6. [DOI] [PubMed] [Google Scholar]
- Petruzzelli L. M., Ganguly S., Smith C. J., Cobb M. H., Rubin C. S., Rosen O. M. Insulin activates a tyrosine-specific protein kinase in extracts of 3T3-L1 adipocytes and human placenta. Proc Natl Acad Sci U S A. 1982 Nov;79(22):6792–6796. doi: 10.1073/pnas.79.22.6792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rees-Jones R. W., Taylor S. I. An endogenous substrate for the insulin receptor-associated tyrosine kinase. J Biol Chem. 1985 Apr 10;260(7):4461–4467. [PubMed] [Google Scholar]
- Sadoul J. L., Peyron J. F., Ballotti R., Debant A., Fehlmann M., Van Obberghen E. Identification of a cellular 110 000-Da protein substrate for the insulin-receptor kinase. Biochem J. 1985 May 1;227(3):887–892. doi: 10.1042/bj2270887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ushiro H., Cohen S. Identification of phosphotyrosine as a product of epidermal growth factor-activated protein kinase in A-431 cell membranes. J Biol Chem. 1980 Sep 25;255(18):8363–8365. [PubMed] [Google Scholar]
- Van Obberghen E., Rossi B., Kowalski A., Gazzano H., Ponzio G. Receptor-mediated phosphorylation of the hepatic insulin receptor: evidence that the Mr 95,000 receptor subunit is its own kinase. Proc Natl Acad Sci U S A. 1983 Feb;80(4):945–949. doi: 10.1073/pnas.80.4.945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White M. F., Maron R., Kahn C. R. Insulin rapidly stimulates tyrosine phosphorylation of a Mr-185,000 protein in intact cells. Nature. 1985 Nov 14;318(6042):183–186. doi: 10.1038/318183a0. [DOI] [PubMed] [Google Scholar]
- Witte O. N., Dasgupta A., Baltimore D. Abelson murine leukaemia virus protein is phosphorylated in vitro to form phosphotyrosine. Nature. 1980 Feb 28;283(5750):826–831. doi: 10.1038/283826a0. [DOI] [PubMed] [Google Scholar]
- Yu K. T., Czech M. P. Tyrosine phosphorylation of the insulin receptor beta subunit activates the receptor-associated tyrosine kinase activity. J Biol Chem. 1984 Apr 25;259(8):5277–5286. [PubMed] [Google Scholar]