Abstract
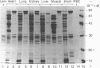
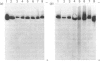
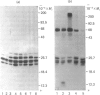
Lens neutral endopeptidase (EC 3.4.24.5) was previously thought to be unique to the eye lens. We report here the finding of a neutral endopeptidase, in a variety of bovine and human tissues, which is very similar both biochemically and immunologically to the lens endopeptidase. SDS/polyacrylamide-gel electrophoresis of partially purified enzyme fractions from various bovine tissues shows the characteristic pattern of at least eight bands with Mr values ranging from 24,000 to 32,000 which was described for the bovine-lens neutral endopeptidase. The relative activity of the enzyme varies from tissue to tissue with lung having the highest activity. Partially purified enzyme fractions from these tissues cross-react with antiserum raised in rabbit against bovine lens endopeptidase showing apparent identity when examined side by side in Ouchterlony double-diffusion tests. The human enzyme also cross-reacts with the antiserum but when tested by double-diffusion against the bovine enzyme the precipitin lines show spurring at the joining edges indicating a structural difference between the human and the bovine enzymes. It was also found by Western blot experiments, after denaturing polyacrylamide-gel electrophoresis of the enzyme, that the polypeptide components of the human and bovine enzymes show somewhat different banding patterns.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blow A. M., Heyningen R. V., Barrett A. J. Metal-dependent proteinase of the lens. Assay, purification and properties of the bovine enzyme. Biochem J. 1975 Mar;145(3):591–599. doi: 10.1042/bj1450591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Dahlmann B., Kuehn L., Rutschmann M., Reinauer H. Purification and characterization of a multicatalytic high-molecular-mass proteinase from rat skeletal muscle. Biochem J. 1985 May 15;228(1):161–170. doi: 10.1042/bj2280161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dahlmann B., Rutschmann M., Kuehn L., Reinauer H. Activation of the multicatalytic proteinase from rat skeletal muscle by fatty acids or sodium dodecyl sulphate. Biochem J. 1985 May 15;228(1):171–177. doi: 10.1042/bj2280171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GOLDBARG J. A., RUTENBURG A. M. The colorimetric determination of leucine aminopeptidase in urine and serum of normal subjects and patients with cancer and other diseases. Cancer. 1958 Mar-Apr;11(2):283–291. doi: 10.1002/1097-0142(195803/04)11:2<283::aid-cncr2820110209>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- Garland P. B., Randle P. J. Regulation of glucose uptake by muscles. 10. Effects of alloxan-diabetes, starvation, hypophysectomy and adrenalectomy, and of fatty acids, ketone bodies and pyruvate, on the glycerol output and concentrations of free fatty acids, long-chain fatty acyl-coenzyme A, glycerol phosphate and citrate-cycle intermediates in rat heart and diaphragm muscles. Biochem J. 1964 Dec;93(3):678–687. doi: 10.1042/bj0930678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- McGuire M. J., DeMartino G. N. Purification and characterization of a high molecular weight proteinase (macropain) from human erythrocytes. Biochim Biophys Acta. 1986 Sep 26;873(2):279–289. doi: 10.1016/0167-4838(86)90055-5. [DOI] [PubMed] [Google Scholar]
- Ray K., Harris H. Comparative studies on lens neutral endopeptidase and pituitary neutral proteinase: two closely similar enzymes. FEBS Lett. 1986 Jan 1;194(1):91–95. doi: 10.1016/0014-5793(86)80057-6. [DOI] [PubMed] [Google Scholar]
- Ray K., Harris H. Purification of neutral lens endopeptidase: close similarity to a neutral proteinase in pituitary. Proc Natl Acad Sci U S A. 1985 Nov;82(22):7545–7549. doi: 10.1073/pnas.82.22.7545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivett A. J. Purification of a liver alkaline protease which degrades oxidatively modified glutamine synthetase. Characterization as a high molecular weight cysteine proteinase. J Biol Chem. 1985 Oct 15;260(23):12600–12606. [PubMed] [Google Scholar]
- Tanaka K., Ii K., Ichihara A., Waxman L., Goldberg A. L. A high molecular weight protease in the cytosol of rat liver. I. Purification, enzymological properties, and tissue distribution. J Biol Chem. 1986 Nov 15;261(32):15197–15203. [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trayhurn P., van Heyningen R. Neutral proteinase activity in the human lens. Exp Eye Res. 1976 Mar;22(3):251–257. doi: 10.1016/0014-4835(76)90052-x. [DOI] [PubMed] [Google Scholar]
- Wilk S., Orlowski M. Cation-sensitive neutral endopeptidase: isolation and specificity of the bovine pituitary enzyme. J Neurochem. 1980 Nov;35(5):1172–1182. doi: 10.1111/j.1471-4159.1980.tb07873.x. [DOI] [PubMed] [Google Scholar]
- Wilk S., Orlowski M. Evidence that pituitary cation-sensitive neutral endopeptidase is a multicatalytic protease complex. J Neurochem. 1983 Mar;40(3):842–849. doi: 10.1111/j.1471-4159.1983.tb08056.x. [DOI] [PubMed] [Google Scholar]
- Zolfaghari R., Baker C. R., Jr, Canizaro P. C., Amirgholami A., Behal F. J. A high-molecular-mass neutral endopeptidase-24.5 from human lung. Biochem J. 1987 Jan 1;241(1):129–135. doi: 10.1042/bj2410129. [DOI] [PMC free article] [PubMed] [Google Scholar]