Abstract
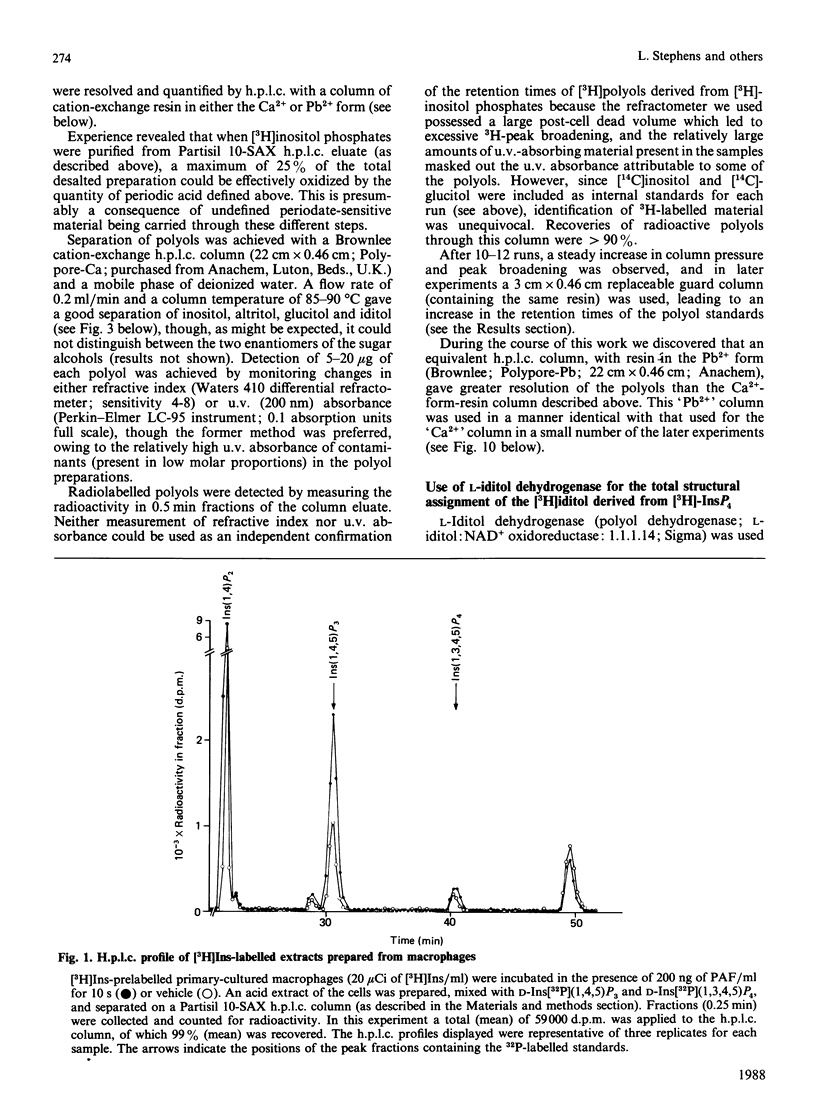
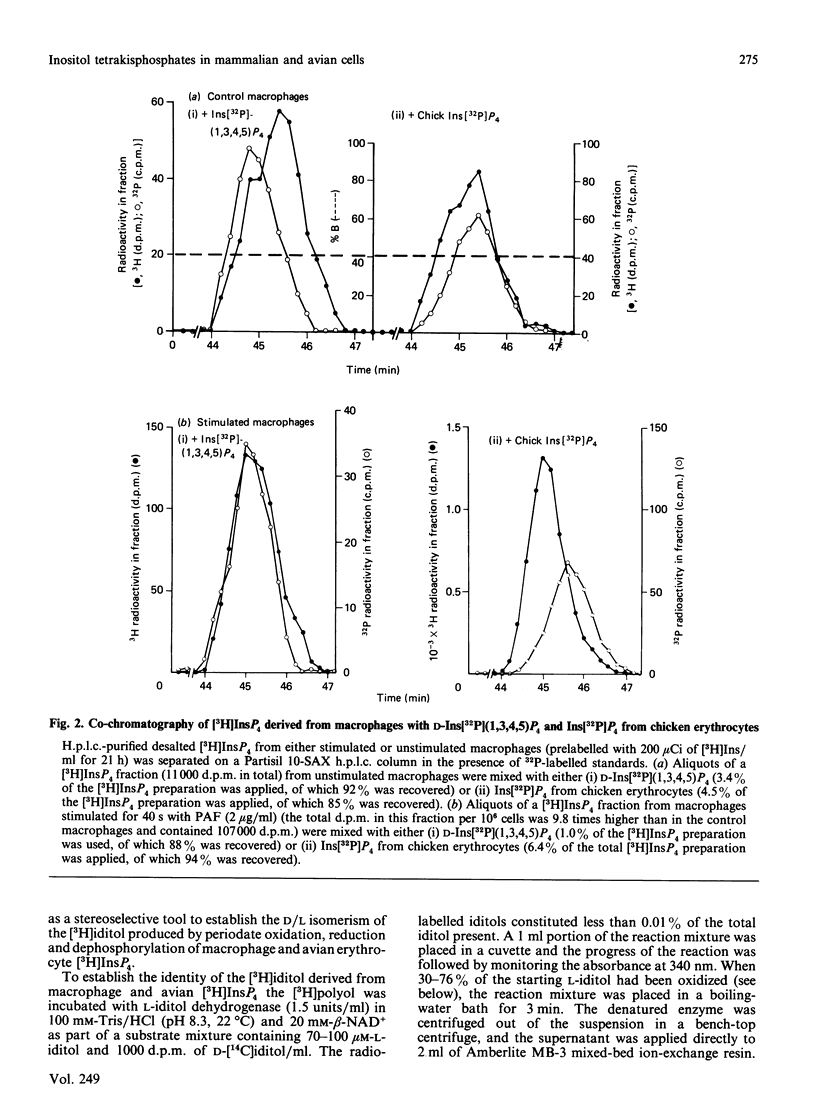
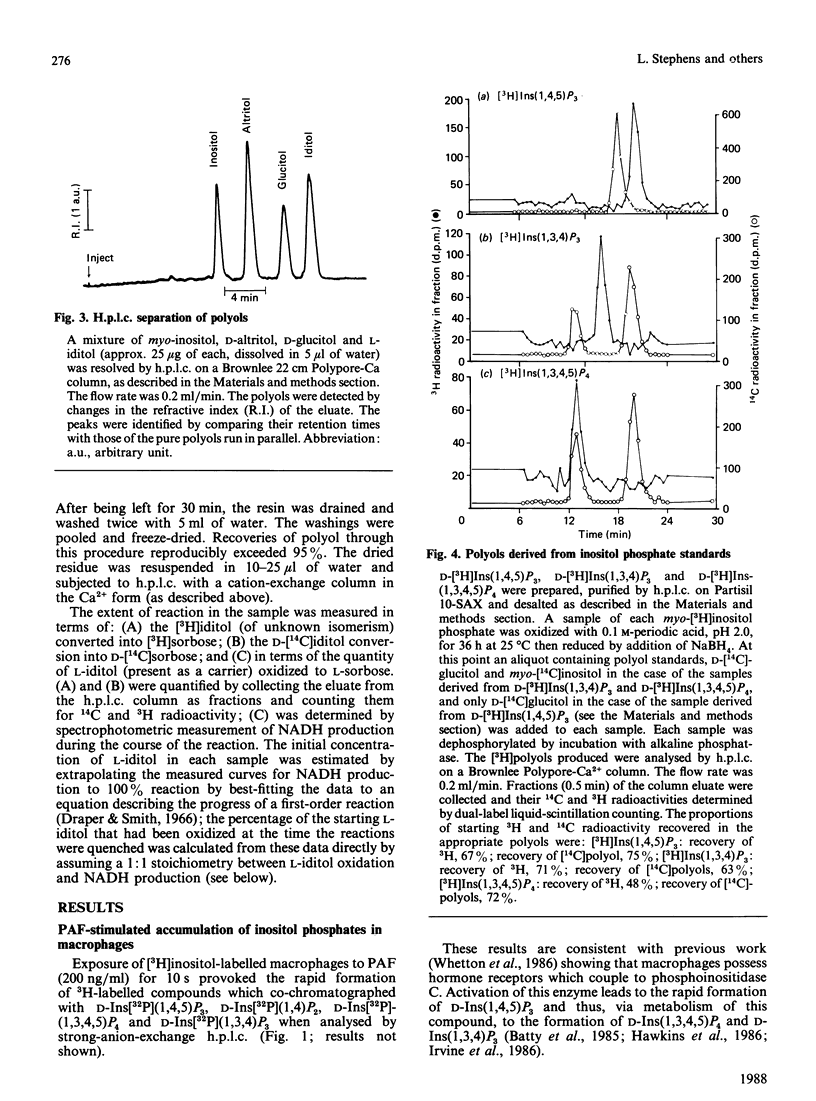
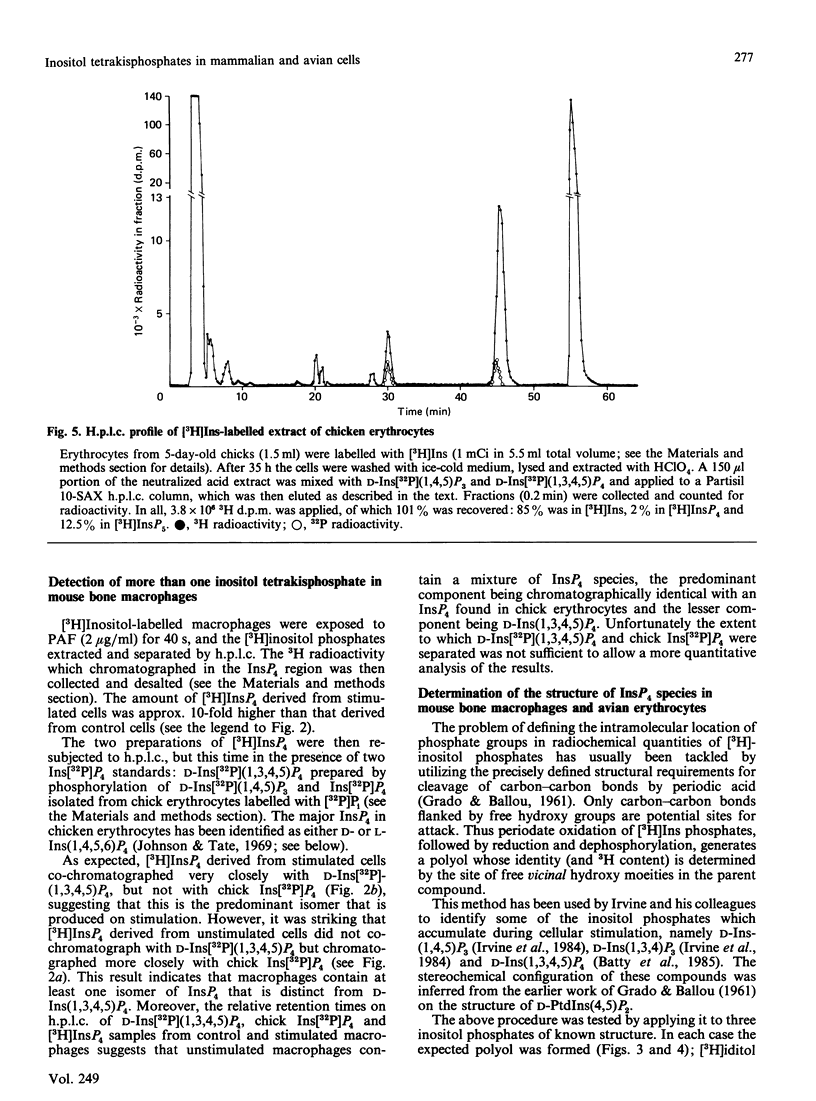
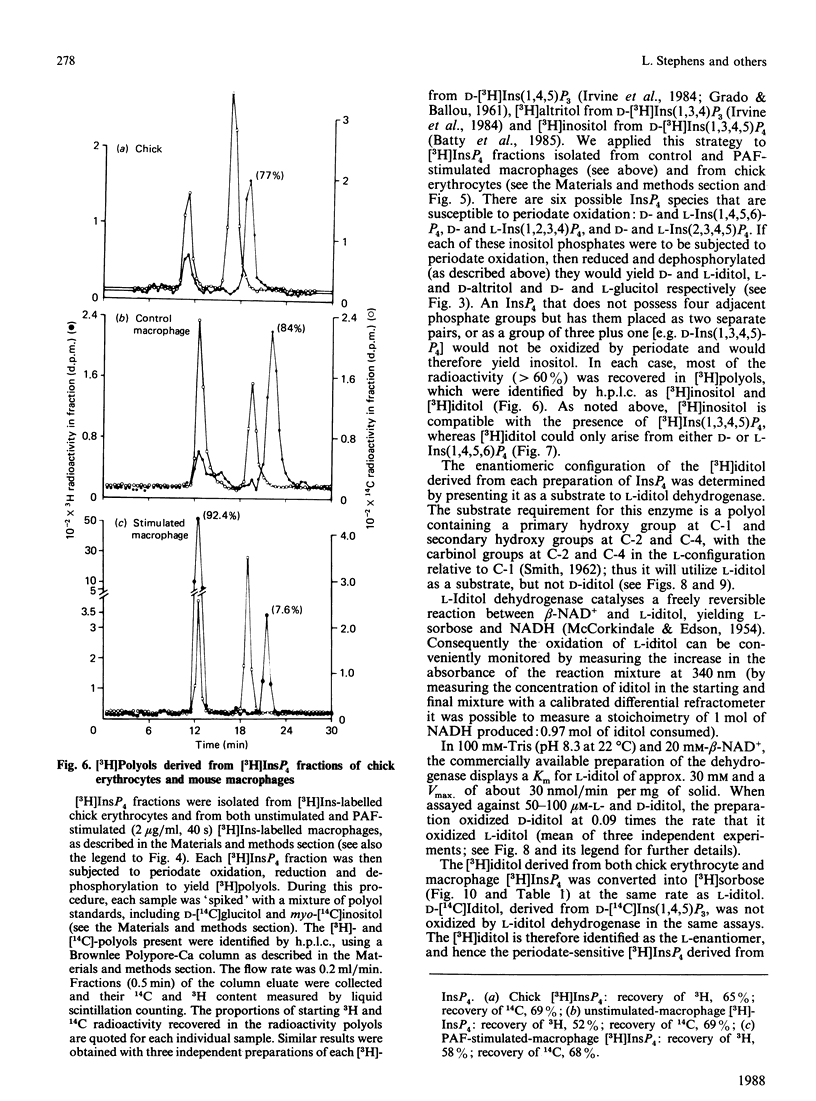
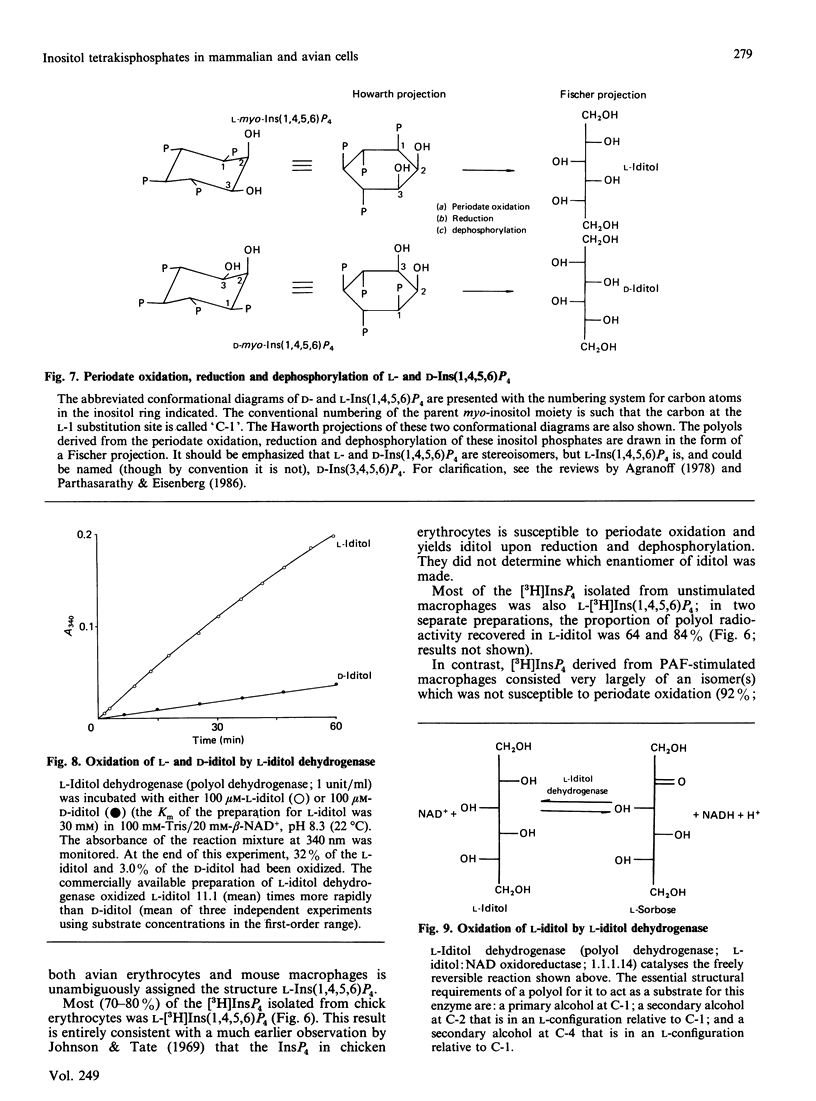
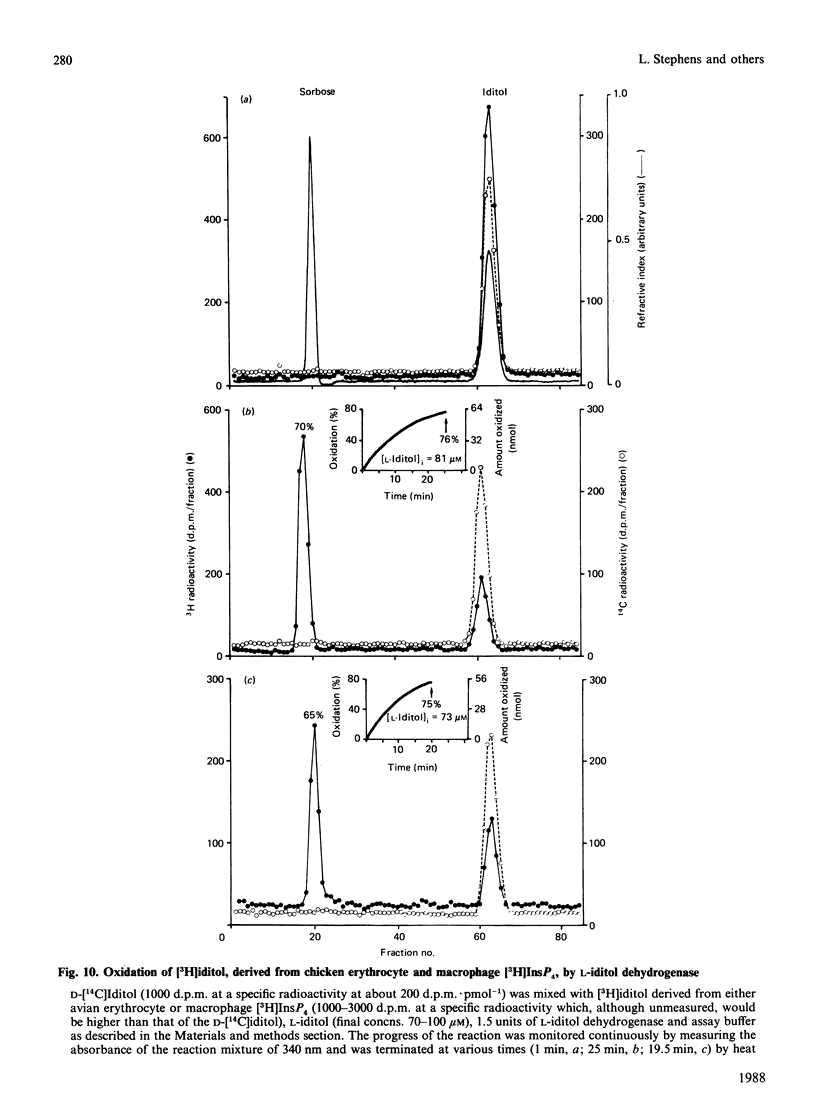
When myo-[3H]inositol-prelabelled primary-cultured murine bone-marrow-derived macrophages were challenged with platelet-activating factor (PAF; 200 ng/ml), there was a rapid (2.5-fold at 10 s) rise in the intracellular concentration of D-myo-[3H]inositol 1,4,5-trisphosphate, followed by a rise in myo-[3H]inositol tetrakisphosphate. myo-[3H]Inositol tetrakisphosphate fractions were isolated by high-performance anion-exchange chromatography from myo-[3H]inositol-prelabelled chick erythrocytes and primary-cultured macrophages. In both cases [3H]iditol and [3H]inositol were the only significant products (greater than 90% of recovered radioactivity) after oxidation to completion with periodic acid, reduction with NaBH4 and dephosphorylation with alkaline phosphatase. The presence of [3H]inositol after this procedure is consistent with the occurrence of [3H]inositol 1,3,4,5-tetrakisphosphate in the cell extracts, whereas [3H]iditol could only be derived from D- or L-inositol 1,4,5,6-tetrakisphosphate. When [3H]inositol tetrakisphosphate fractions obtained from (A) unstimulated macrophages, (B) macrophages that had been stimulated with PAF for 40s or (C) chick erythrocytes were subjected to the above procedure, radioactivity was recovered in these polyols in the following proportions: A, 60-90% in iditol, with 10-40% in inositol; B, total radioactivity increased by a factor of 9.8, 94% being recovered in inositol and 8% in iditol; C, 70-80% in iditol and 20-30% in inositol. [3H]Iditol derived from myo-[3H]inositol tetrakisphosphate fractions from macrophages and chick erythrocytes was oxidized to sorbose by L-iditol dehydrogenase (L-iditol:NAD+2-oxidoreductase, 1.1.1.14) at the same rate as authentic L-iditol. D-[14C]Iditol, derived from D-myo-inositol 1,4,5-trisphosphate, was not oxidized by L-iditol dehydrogenase. This result indicates that the [3H]iditol was derived from L-myo-inositol inositol 1,4,5,6-tetrakisphosphate. The data are consistent with rapid PAF-sensitive synthesis of D-myo-[3H]inositol 1,3,4,5-tetrakisphosphate in macrophages, and demonstrate that L-myo-inositol 1,4,5,6-tetrakisphosphate is synthesized in both mammalian and avian cells. The levels of L-myo-[3H]inositol 1,4,5,6-tetrakisphosphate in primary-cultured macrophages are not acutely sensitive to PAF.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Batty I. R., Nahorski S. R., Irvine R. F. Rapid formation of inositol 1,3,4,5-tetrakisphosphate following muscarinic receptor stimulation of rat cerebral cortical slices. Biochem J. 1985 Nov 15;232(1):211–215. doi: 10.1042/bj2320211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown D. M., Stewart J. C. The structure of triphosphoinositide from beef brain. Biochim Biophys Acta. 1966 Dec 7;125(3):413–421. doi: 10.1016/0005-2760(66)90029-4. [DOI] [PubMed] [Google Scholar]
- Clarke N. G., Dawson R. M. Alkaline O leads to N-transacylation. A new method for the quantitative deacylation of phospholipids. Biochem J. 1981 Apr 1;195(1):301–306. doi: 10.1042/bj1950301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Creba J. A., Downes C. P., Hawkins P. T., Brewster G., Michell R. H., Kirk C. J. Rapid breakdown of phosphatidylinositol 4-phosphate and phosphatidylinositol 4,5-bisphosphate in rat hepatocytes stimulated by vasopressin and other Ca2+-mobilizing hormones. Biochem J. 1983 Jun 15;212(3):733–747. doi: 10.1042/bj2120733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Downes C. P., Hawkins P. T., Irvine R. F. Inositol 1,3,4,5-tetrakisphosphate and not phosphatidylinositol 3,4-bisphosphate is the probable precursor of inositol 1,3,4-trisphosphate in agonist-stimulated parotid gland. Biochem J. 1986 Sep 1;238(2):501–506. doi: 10.1042/bj2380501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Downes C. P., Michell R. H. The polyphosphoinositide phosphodiesterase of erythrocyte membranes. Biochem J. 1981 Jul 15;198(1):133–140. doi: 10.1042/bj1980133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GRADO C., BALLOU C. E. Myo-inositol phosphates obtained by alkaline hydrolysis of beef brain phosphoinositide. J Biol Chem. 1961 Jan;236:54–60. [PubMed] [Google Scholar]
- Hawkins P. T., Stephens L., Downes C. P. Rapid formation of inositol 1,3,4,5-tetrakisphosphate and inositol 1,3,4-trisphosphate in rat parotid glands may both result indirectly from receptor-stimulated release of inositol 1,4,5-trisphosphate from phosphatidylinositol 4,5-bisphosphate. Biochem J. 1986 Sep 1;238(2):507–516. doi: 10.1042/bj2380507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irvine R. F., Letcher A. J., Heslop J. P., Berridge M. J. The inositol tris/tetrakisphosphate pathway--demonstration of Ins(1,4,5)P3 3-kinase activity in animal tissues. Nature. 1986 Apr 17;320(6063):631–634. doi: 10.1038/320631a0. [DOI] [PubMed] [Google Scholar]
- Irvine R. F., Letcher A. J., Lander D. J., Downes C. P. Inositol trisphosphates in carbachol-stimulated rat parotid glands. Biochem J. 1984 Oct 1;223(1):237–243. doi: 10.1042/bj2230237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irvine R. F., Moor R. M. Micro-injection of inositol 1,3,4,5-tetrakisphosphate activates sea urchin eggs by a mechanism dependent on external Ca2+. Biochem J. 1986 Dec 15;240(3):917–920. doi: 10.1042/bj2400917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayr G. W., Dietrich W. The only inositol tetrakisphosphate detectable in avian erythrocytes is the isomer lacking phosphate at position 3: a NMR study. FEBS Lett. 1987 Mar 23;213(2):278–282. doi: 10.1016/0014-5793(87)81505-3. [DOI] [PubMed] [Google Scholar]
- McCORKINDALE J., EDSON N. L. Polyol dehydrogenases. I. The specificity of rat-liver polyol dehydrogenase. Biochem J. 1954 Jul;57(3):518–523. doi: 10.1042/bj0570518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parthasarathy R., Eisenberg F., Jr The inositol phospholipids: a stereochemical view of biological activity. Biochem J. 1986 Apr 15;235(2):313–322. doi: 10.1042/bj2350313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SMITH M. G. Polyol dehydrogenases. 4. Crystallization of the L-iditol dehydrogenase of sheep liver. Biochem J. 1962 Apr;83:135–144. doi: 10.1042/bj0830135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharps E. S., McCarl R. L. A high-performance liquid chromatographic method to measure 32P incorporation into phosphorylated metabolites in cultured cells. Anal Biochem. 1982 Aug;124(2):421–424. doi: 10.1016/0003-2697(82)90059-8. [DOI] [PubMed] [Google Scholar]
- Stephens L. R., Hawkins P. T., Morris A. J., Downes P. C. L-myo-inositol 1,4,5,6-tetrakisphosphate (3-hydroxy)kinase. Biochem J. 1988 Jan 1;249(1):283–292. doi: 10.1042/bj2490283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whetton A. D., Monk P. N., Consalvey S. D., Downes C. P. The haemopoietic growth factors interleukin 3 and colony stimulating factor-1 stimulate proliferation but do not induce inositol lipid breakdown in murine bone-marrow-derived macrophages. EMBO J. 1986 Dec 1;5(12):3281–3286. doi: 10.1002/j.1460-2075.1986.tb04640.x. [DOI] [PMC free article] [PubMed] [Google Scholar]


