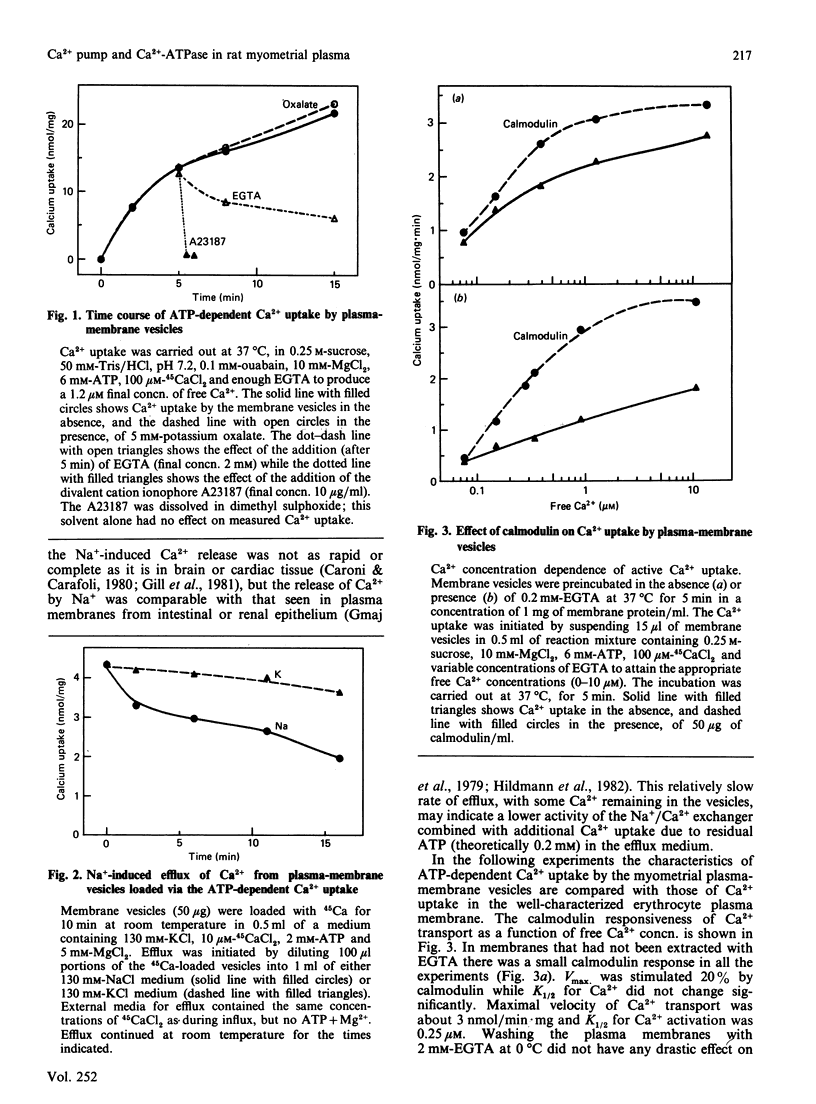
Abstract
A plasma membrane-enriched fraction from rat myometrium shows ATP-Mg2+-dependent active calcium uptake which is independent of the presence of oxalate and is abolished by the Ca2+ ionophore A23187. Ca2+ loaded into vesicles via the ATP-dependent Ca2+ uptake was released by extravesicular Na+. This showed that the Na+/Ca2+ exchange and the Ca2+ uptake were both occurring in plasma membrane vesicles. In a medium containing KCl, vanadate readily inhibited the Ca2+ uptake (K1/2 5 microM); when sucrose replaced KCl, 400 microM-vanadate was required for half inhibition. Only a slight stimulation of the calcium pump by calmodulin was observed in untreated membrane vesicles. Extraction of endogenous calmodulin from the membranes by EGTA decreased the activity and Ca2+ affinity of the calcium pump; both activity and affinity were fully restored by adding back calmodulin or by limited proteolysis. A monoclonal antibody (JA3) directed against the human erythrocyte Ca2+ pump reacted with the 140 kDa Ca2+-pump protein of the myometrial plasma membrane. The Ca2+-ATPase activity of these membranes is not specific for ATP, and is not inhibited by mercurial agents, whereas Ca2+ uptake has the opposite properties. Ca2+-ATPase activity is also over 100 times that of calcium transport; it appears that the ATPase responsible for transport is largely masked by the presence of another Ca2+-ATPase of unknown function. Measurements of total Ca2+-ATPase activity are, therefore, probably not directly relevant to the question of intracellular Ca2+ control.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akerman K. E., Wikström M. K. (Ca2+ + Mg2+)-stimulated ATPase activity of rabbit myometrium plasma membrane is blocked by oxytocin. FEBS Lett. 1979 Jan 15;97(2):283–287. doi: 10.1016/0014-5793(79)80103-9. [DOI] [PubMed] [Google Scholar]
- Barrabin H., Garrahan P. J., Rega A. F. Vanadate inhibition of the Ca2+-ATPase from human red cell membranes. Biochim Biophys Acta. 1980 Aug 14;600(3):796–804. doi: 10.1016/0005-2736(80)90482-4. [DOI] [PubMed] [Google Scholar]
- Borke J. L., Minami J., Verma A., Penniston J. T., Kumar R. Monoclonal antibodies to human erythrocyte membrane Ca++-Mg++ adenosine triphosphatase pump recognize an epitope in the basolateral membrane of human kidney distal tubule cells. J Clin Invest. 1987 Nov;80(5):1225–1231. doi: 10.1172/JCI113196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Caroni P., Carafoli E. An ATP-dependent Ca2+-pumping system in dog heart sarcolemma. Nature. 1980 Feb 21;283(5749):765–767. doi: 10.1038/283765a0. [DOI] [PubMed] [Google Scholar]
- ERNSTER L., LINDBERG O. Determination of organic phosphorus compounds by phosphate analysis. Methods Biochem Anal. 1956;3:1–22. doi: 10.1002/9780470110195.ch1. [DOI] [PubMed] [Google Scholar]
- Gill D. L., Grollman E. F., Kohn L. D. Calcium transport mechanisms in membrane vesicles from guinea pig brain synaptosomes. J Biol Chem. 1981 Jan 10;256(1):184–192. [PubMed] [Google Scholar]
- Gmaj P., Murer H., Kinne R. Calcium ion transport across plasma membranes isolated from rat kidney cortex. Biochem J. 1979 Mar 15;178(3):549–557. doi: 10.1042/bj1780549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grover A. K., Kwan C. Y., Daniel E. E. Ca2+ dependence of calcium uptake by rat myometrium plasma membrane-enriched fraction. Am J Physiol. 1982 May;242(5):C278–C282. doi: 10.1152/ajpcell.1982.242.5.C278. [DOI] [PubMed] [Google Scholar]
- Grover A. K., Kwan C. Y., Daniel E. E. Na--Ca exchange in rat myometrium membrane vesicles highly enriched in plasma membranes. Am J Physiol. 1981 May;240(5):C175–C182. doi: 10.1152/ajpcell.1981.240.5.C175. [DOI] [PubMed] [Google Scholar]
- Grover A. K., Kwan C. Y., Rangachari P. K., Daniel E. E. Na-Ca exchange in a smooth muscle plasma membrane-enriched fraction. Am J Physiol. 1983 Mar;244(3):C158–C165. doi: 10.1152/ajpcell.1983.244.3.C158. [DOI] [PubMed] [Google Scholar]
- Hildmann B., Schmidt A., Murer H. Ca++-transport across basal-lateral plasma membranes from rat small intestinal epithelial cells. J Membr Biol. 1982;65(1-2):55–62. doi: 10.1007/BF01870469. [DOI] [PubMed] [Google Scholar]
- Kwan C. Y., Kostka P., Grover A. K., Law J. S., Daniel E. E. Calmodulin stimulation of plasmalemmal Ca2+-pump of canine aortic smooth muscle. Blood Vessels. 1986;23(1):22–33. doi: 10.1159/000158622. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lin S. H. Novel ATP-dependent calcium transport component from rat liver plasma membranes. The transporter and the previously reported (Ca2+-Mg2+)-ATPase are different proteins. J Biol Chem. 1985 Jul 5;260(13):7850–7856. [PubMed] [Google Scholar]
- Lin S. H. The rat liver plasma membrane high affinity (Ca2+-Mg2+)-ATPase is not a calcium pump. Comparison with ATP-dependent calcium transporter. J Biol Chem. 1985 Sep 15;260(20):10976–10980. [PubMed] [Google Scholar]
- Minami J., Penniston J. T. Ca2+ uptake by corpus-luteum plasma membranes. Evidence for the presence of both a Ca2+-pumping ATPase and a Ca2+-dependent nucleoside triphosphatase. Biochem J. 1987 Mar 15;242(3):889–894. doi: 10.1042/bj2420889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niggli V., Adunyah E. S., Penniston J. T., Carafoli E. Purified (Ca2+-Mg2+)-ATPase of the erythrocyte membrane. Reconstitution and effect of calmodulin and phospholipids. J Biol Chem. 1981 Jan 10;256(1):395–401. [PubMed] [Google Scholar]
- Popescu L. M., Nutu O., Panoiu C. Oxytocin contracts the human uterus at term by inhibiting the myometrial Ca2+-extrusion pump. Biosci Rep. 1985 Jan;5(1):21–28. doi: 10.1007/BF01117437. [DOI] [PubMed] [Google Scholar]
- Raeymaekers L., Wuytack F., Casteels R. Subcellular fractionation of pig stomach smooth muscle. A study of the distribution of the (Ca2+ + Mg2+)-ATPase activity in plasmalemma and endoplasmic reticulum. Biochim Biophys Acta. 1985 May 28;815(3):441–454. doi: 10.1016/0005-2736(85)90372-4. [DOI] [PubMed] [Google Scholar]
- Raeymaekers L., Wuytack F., Eggermont J., De Schutter G., Casteels R. Isolation of a plasma-membrane fraction from gastric smooth muscle. Comparison of the calcium uptake with that in endoplasmic reticulum. Biochem J. 1983 Feb 15;210(2):315–322. doi: 10.1042/bj2100315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soloff M. S., Sweet P. Oxytocin inhibition of (Ca2+ + Mg2+)-ATPase activity in rat myometrial plasma membranes. J Biol Chem. 1982 Sep 25;257(18):10687–10693. [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verma A. K., Penniston J. T. A high affinity Ca2+-stimulated and Mg2+-dependent ATPase in rat corpus luteum plasma membrane fractions. J Biol Chem. 1981 Feb 10;256(3):1269–1275. [PubMed] [Google Scholar]
- Wuytack F., De Schutter G., Casteels R. Purification of (Ca2+ + Mg2+)-ATPase from smooth muscle by calmodulin affinity chromatography. FEBS Lett. 1981 Jul 6;129(2):297–300. doi: 10.1016/0014-5793(81)80187-1. [DOI] [PubMed] [Google Scholar]
- Wuytack F., De Schutter G., Casteels R. The effect of calmodulin on the active calcium-ion transport and (Ca2+ + Mg2+)-dependent ATPase in microsomal fractions of smooth muscle compared with that in erythrocytes and cardiac muscle. Biochem J. 1980 Sep 15;190(3):827–831. doi: 10.1042/bj1900827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wuytack F., De Schutter G., Verbist J., Casteels R. Antibodies to the calmodulin-binding Ca2+-transport ATPase from smooth muscle. FEBS Lett. 1983 Apr 5;154(1):191–195. doi: 10.1016/0014-5793(83)80901-6. [DOI] [PubMed] [Google Scholar]
- Wuytack F., Raeymaekers L., De Schutter G., Casteels R. Demonstration of the phosphorylated intermediates of the Ca2+-transport ATPase in a microsomal fraction and in a (Ca2+ + Mg2+)-ATPase purified from smooth muscle by means of calmodulin affinity chromatography. Biochim Biophys Acta. 1982 Dec 8;693(1):45–52. doi: 10.1016/0005-2736(82)90469-2. [DOI] [PubMed] [Google Scholar]
- Wuytack F., Raeymaekers L., Verbist J., De Smedt H., Casteels R. Evidence for the presence in smooth muscle of two types of Ca2+-transport ATPase. Biochem J. 1984 Dec 1;224(2):445–451. doi: 10.1042/bj2240445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zurini M., Krebs J., Penniston J. T., Carafoli E. Controlled proteolysis of the purified Ca2+-ATPase of the erythrocyte membrane. A correlation between the structure and the function of the enzyme. J Biol Chem. 1984 Jan 10;259(1):618–627. [PubMed] [Google Scholar]



