Abstract
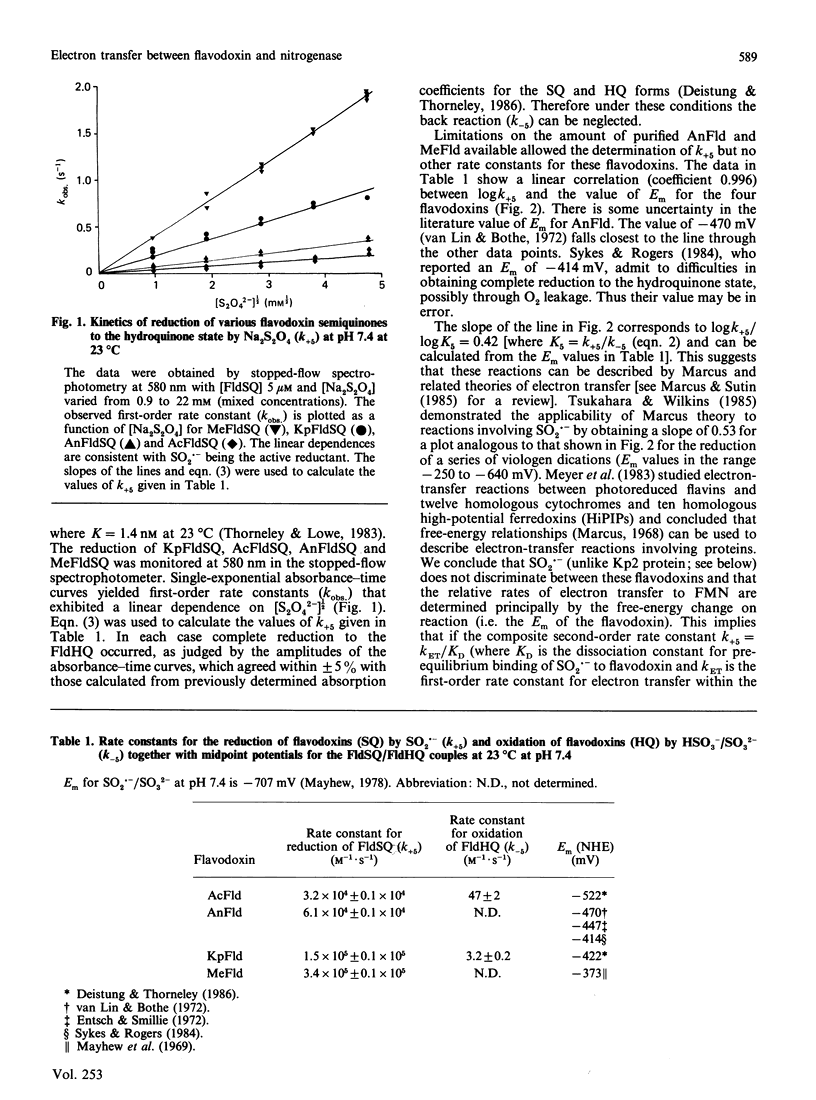
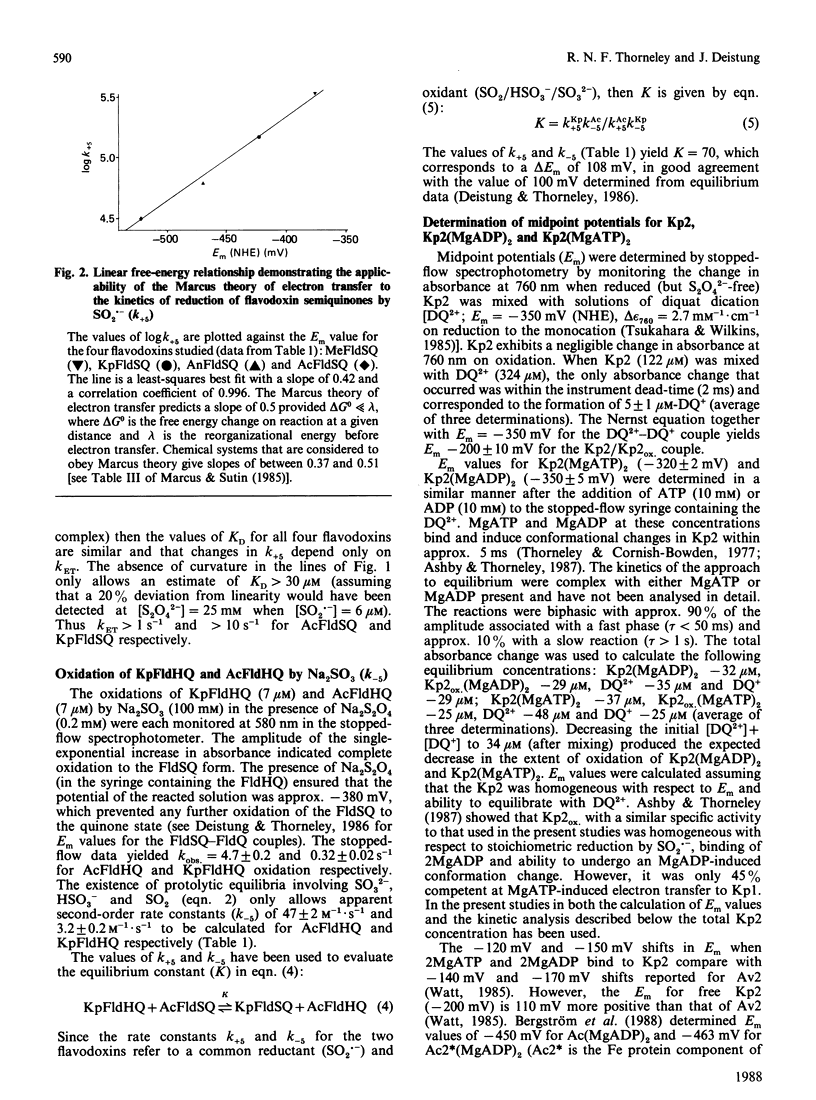
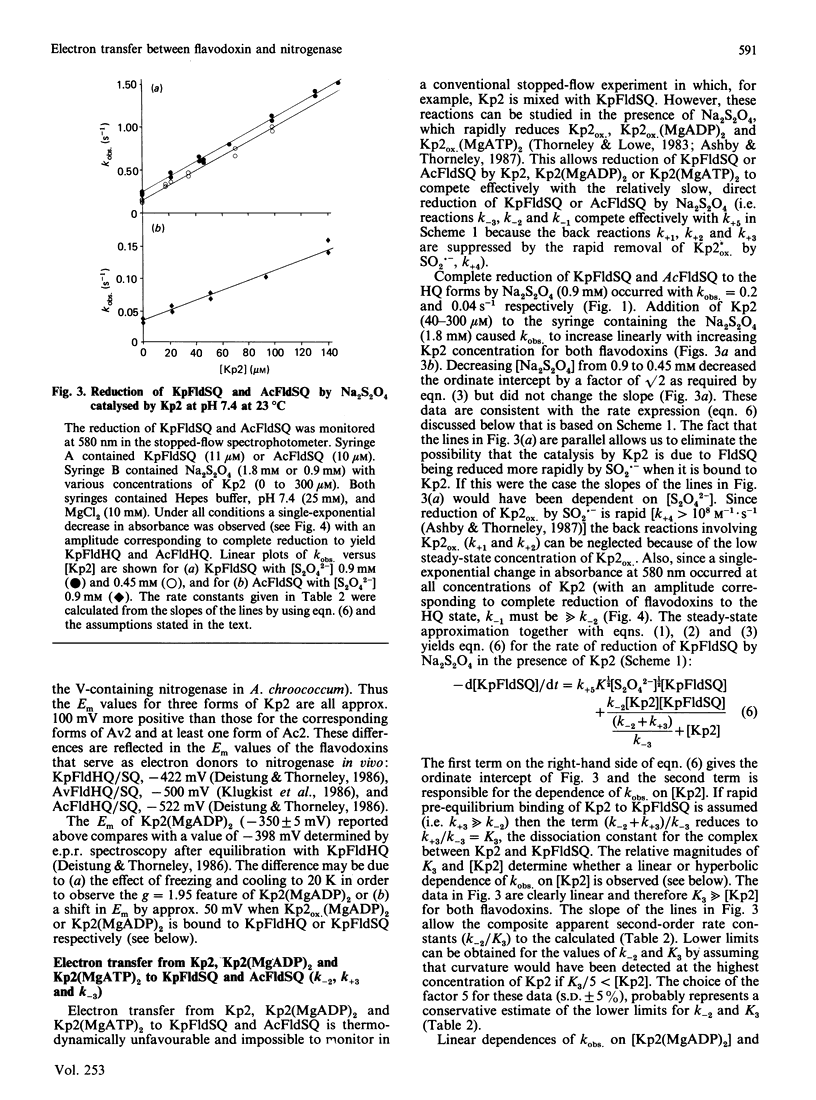
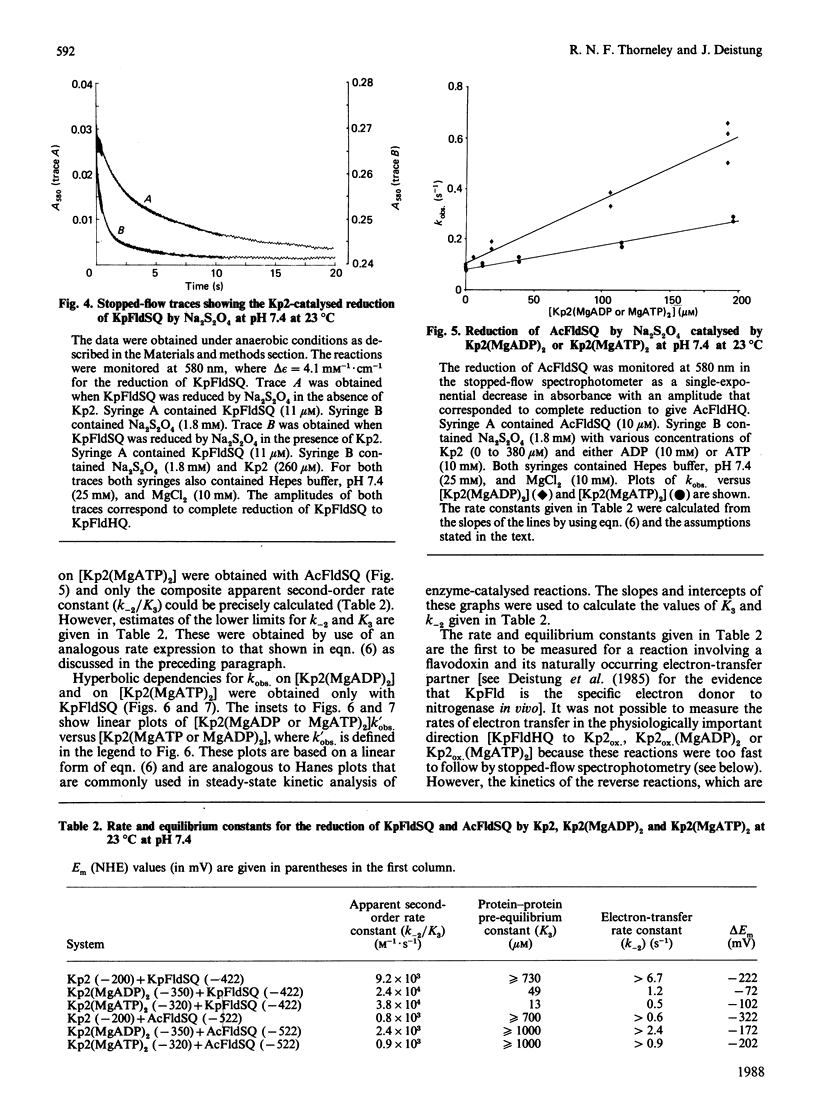
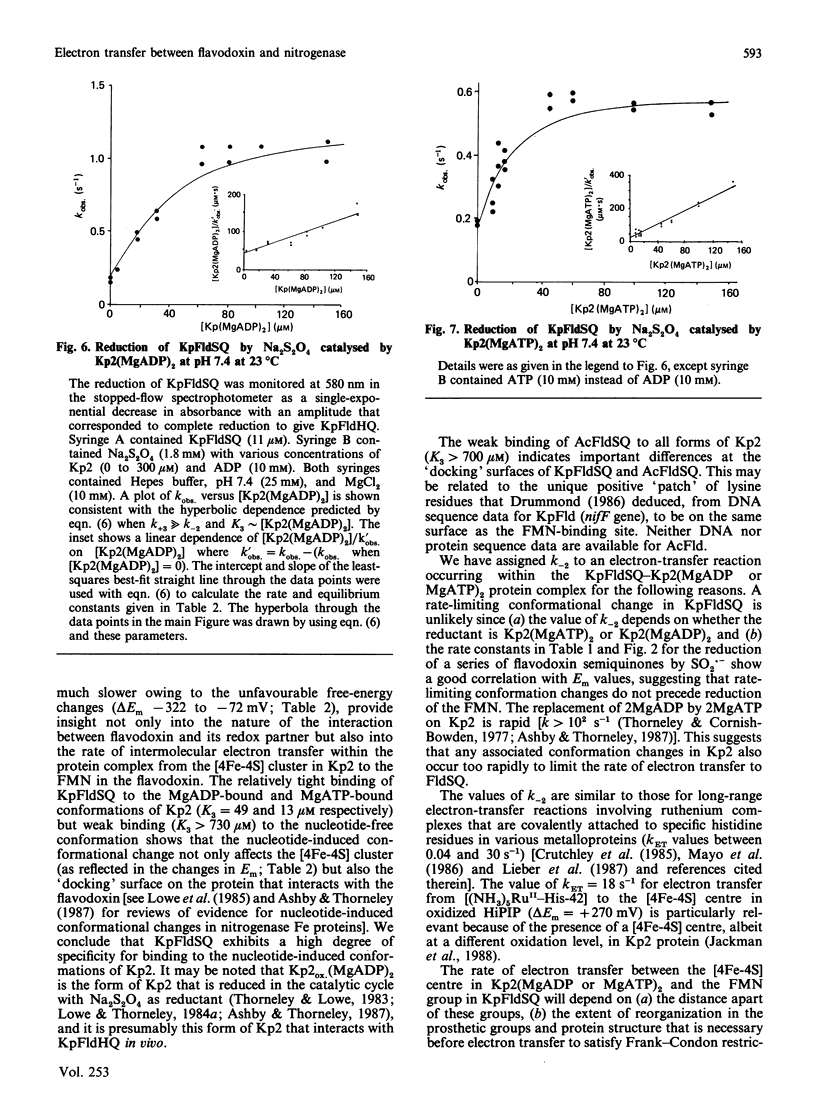
The kinetics of electron-transfer reactions involving flavodoxins from Klebsiella pneumoniae (KpFld), Azotobacter chroococcum (AcFld), Anacystis nidulans (AnFld) and Megasphaera elsdenii (MeFld), the free, MgADP-bound and MgATP-bound forms of the Fe protein component of nitrogenase from K. pneumoniae [Kp2, Kp2(MgADP)2 and Kp2(MgATP)2] and Na2S2O4 were studied by stopped-flow spectrophotometry. Kinetic evidence was obtained for the formation of binary protein complexes involving KpFldSQ (semiquinone) with either Kp2(MgADP)2 (KD = 49 microM) or Kp2(MgATP)2 (KD = 13 microM) but not with Kp2 (KD greater than 730 microM). The binding of 2MgATP or 2MgADP to Kp2 therefore not only shifts the midpoint potential (Em) of the [4Fe-4S] centre from -200 mV to -320 mV or -350 mV respectively but also changes the affinity of Kp2 for KpFldSQ. Thermodynamically unfavourable electron from Kp2(MgADP)2 and Kp2(MgATP)2 to KpFldSQ occurs within the protein complexes with k = 1.2 s-1 (delta E = -72 mV) and 0.5 s-1 (delta E = -120 mV) respectively. Although AcFldSQ is reduced by Kp2, Kp2(MgADP)2 and Kp2(MgATP)2 (k = 8 x 10(3), 2.4 x 10(3) and 9 x 10(2) M-1.s-1 respectively), protein-complex formation is weak in each case (KD greater than 700 microM). Electron transfer in the physiologically important and thermodynamically favourable direction from Kp2FldHQ (hydroquinone) and AcFldHQ to Kp2ox.(MgADP)2 (the state of Kp2 that accepts electrons from FldHQ in the catalytic cycle of nitrogenase) is rapid (k greater than 10(6) M-1.s-1). The second-order rate constants for the reduction of KpFldSQ, AcFldSQ, AnFldSQ and MeFldSQ by SO2.- (active reductant formed by the predissociation of S2O4(2-) ion) exhibited the linear free-energy relationship predicted by the Marcus theory of electron transfer.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ashby G. A., Thorneley R. N. Nitrogenase of Klebsiella pneumoniae. Kinetic studies on the Fe protein involving reduction by sodium dithionite, the binding of MgADP and a conformation change that alters the reactivity of the 4Fe-4S centre. Biochem J. 1987 Sep 1;246(2):455–465. doi: 10.1042/bj2460455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benemann J. R., Yoch D. C., Valentine R. C., Arnon D. I. The electron transport system in nitrogen fixation by Azotobacter. I. Azotoflavin as an electron carrier. Proc Natl Acad Sci U S A. 1969 Nov;64(3):1079–1086. doi: 10.1073/pnas.64.3.1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benemann J. R., Yoch D. C., Valentine R. C., Arnon D. I. The electron transport system in nitrogen fixation by azotobacter. 3. Requirements for NADPH-supported nitrogenase activity. Biochim Biophys Acta. 1971 Mar 2;226(2):205–212. doi: 10.1016/0005-2728(71)90087-9. [DOI] [PubMed] [Google Scholar]
- Bergström J., Eady R. R., Thorneley R. N. The vanadium- and molybdenum-containing nitrogenases of Azotobacter chroococcum. Comparison of mid-point potentials and kinetics of reduction by sodium dithionite of the iron proteins with bound magnesium adenosine 5'-diphosphate. Biochem J. 1988 Apr 1;251(1):165–169. doi: 10.1042/bj2510165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Creutz C., Sutin N. Reduction of ferricytochrome c by dithionite ion: electron transfer by parallel adjacent and remote pathways. Proc Natl Acad Sci U S A. 1973 Jun;70(6):1701–1703. doi: 10.1073/pnas.70.6.1701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deistung J., Cannon F. C., Cannon M. C., Hill S., Thorneley R. N. Electron transfer to nitrogenase in Klebsiella pneumoniae. nifF gene cloned and the gene product, a flavodoxin, purified. Biochem J. 1985 Nov 1;231(3):743–753. doi: 10.1042/bj2310743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deistung J., Thorneley R. N. Electron transfer to nitrogenase. Characterization of flavodoxin from Azotobacter chroococcum and comparison of its redox potentials with those of flavodoxins from Azotobacter vinelandii and Klebsiella pneumoniae (nifF-gene product). Biochem J. 1986 Oct 1;239(1):69–75. doi: 10.1042/bj2390069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drummond M. H. Structure predictions and surface charge of nitrogenase flavodoxins from Klebsiella pneumoniae and Azotobacter vinelandii. Eur J Biochem. 1986 Sep 15;159(3):549–553. doi: 10.1111/j.1432-1033.1986.tb09921.x. [DOI] [PubMed] [Google Scholar]
- Eady R. R., Lowe D. J., Thorneley R. N. Nitrogenase of Klebsiella pneumoniae: a pre-steady state burst of ATP hydrolysis is coupled to electron transfer between the component proteins. FEBS Lett. 1978 Nov 15;95(2):211–213. doi: 10.1016/0014-5793(78)80995-8. [DOI] [PubMed] [Google Scholar]
- Entsch B., Smillie R. M. Oxidation--reduction properties of phytoflavin, a flavoprotein from blue-green algae. Arch Biochem Biophys. 1972 Aug;151(2):378–386. doi: 10.1016/0003-9861(72)90512-7. [DOI] [PubMed] [Google Scholar]
- Hageman R. V., Burris R. H. Kinetic studies on electron transfer and interaction between nitrogenase components from Azotobacter vinelandii. Biochemistry. 1978 Oct 3;17(20):4117–4124. doi: 10.1021/bi00613a002. [DOI] [PubMed] [Google Scholar]
- Hageman R. V., Orme-Johnson W. H., Burris R. H. Role of magnesium adenosine 5'-triphosphate in the hydrogen evolution reaction catalyzed by nitrogenase from Azotobacter vinelandii. Biochemistry. 1980 May 27;19(11):2333–2342. doi: 10.1021/bi00552a009. [DOI] [PubMed] [Google Scholar]
- Hill S., Kavanagh E. P. Roles of nifF and nifJ gene products in electron transport to nitrogenase in Klebsiella pneumoniae. J Bacteriol. 1980 Feb;141(2):470–475. doi: 10.1128/jb.141.2.470-475.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klugkist J., Haaker H., Wassink H., Veeger C. The catalytic activity of nitrogenase in intact Azotobacter vinelandii cells. Eur J Biochem. 1985 Feb 1;146(3):509–515. doi: 10.1111/j.1432-1033.1985.tb08681.x. [DOI] [PubMed] [Google Scholar]
- Klugkist J., Voorberg J., Haaker H., Veeger C. Characterization of three different flavodoxins from Azotobacter vinelandii. Eur J Biochem. 1986 Feb 17;155(1):33–40. doi: 10.1111/j.1432-1033.1986.tb09455.x. [DOI] [PubMed] [Google Scholar]
- Lambeth D. O., Palmer G. The kinetics and mechanism of reduction of electron transfer proteins and other compounds of biological interest by dithionite. J Biol Chem. 1973 Sep 10;248(17):6095–6103. [PubMed] [Google Scholar]
- Lowe D. J., Thorneley R. N. The mechanism of Klebsiella pneumoniae nitrogenase action. Pre-steady-state kinetics of H2 formation. Biochem J. 1984 Dec 15;224(3):877–886. doi: 10.1042/bj2240877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowe D. J., Thorneley R. N. The mechanism of Klebsiella pneumoniae nitrogenase action. The determination of rate constants required for the simulation of the kinetics of N2 reduction and H2 evolution. Biochem J. 1984 Dec 15;224(3):895–901. doi: 10.1042/bj2240895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayhew S. G., Foust G. P., Massey V. Oxidation-reduction properties of flavodoxin from Peptostreptococcus elsdenii. J Biol Chem. 1969 Feb 10;244(3):803–810. [PubMed] [Google Scholar]
- Mayhew S. G. The redox potential of dithionite and SO-2 from equilibrium reactions with flavodoxins, methyl viologen and hydrogen plus hydrogenase. Eur J Biochem. 1978 Apr 17;85(2):535–547. doi: 10.1111/j.1432-1033.1978.tb12269.x. [DOI] [PubMed] [Google Scholar]
- Mayo S. L., Ellis W. R., Jr, Crutchley R. J., Gray H. B. Long-range electron transfer in heme proteins. Science. 1986 Aug 29;233(4767):948–952. doi: 10.1126/science.3016897. [DOI] [PubMed] [Google Scholar]
- Meyer T. E., Przysiecki C. T., Watkins J. A., Bhattacharyya A., Simondsen R. P., Cusanovich M. A., Tollin G. Correlation between rate constant for reduction and redox potential as a basis for systematic investigation of reaction mechanisms of electron transfer proteins. Proc Natl Acad Sci U S A. 1983 Nov;80(22):6740–6744. doi: 10.1073/pnas.80.22.6740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieva-Gómez D., Roberts G. P., Klevickis S., Brill W. J. Electron transport to nitrogenase in Klebsiella pneumoniae. Proc Natl Acad Sci U S A. 1980 May;77(5):2555–2558. doi: 10.1073/pnas.77.5.2555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah V. K., Stacey G., Brill W. J. Electron transport to nitrogenase. Purification and characterization of pyruvate:flavodoxin oxidoreductase. The nifJ gene product. J Biol Chem. 1983 Oct 10;258(19):12064–12068. [PubMed] [Google Scholar]
- Sykes G. A., Rogers L. J. Redox potentials of algal and cyanobacterial flavodoxins. Biochem J. 1984 Feb 1;217(3):845–850. doi: 10.1042/bj2170845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorneley R. N., Cornish-Bowden A. Kinetics of nitrogenase of Klebsiella pneumoniae. Heterotropic interactions between magnesium-adenosine 5'-diphosphate and magnesium-adenosine 5'-triphosphate. Biochem J. 1977 Aug 1;165(2):255–262. doi: 10.1042/bj1650255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorneley R. N., Lowe D. J. Nitrogenase of Klebsiella pneumoniae. Kinetics of the dissociation of oxidized iron protein from molybdenum-iron protein: identification of the rate-limiting step for substrate reduction. Biochem J. 1983 Nov 1;215(2):393–403. doi: 10.1042/bj2150393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorneley R. N., Lowe D. J. The mechanism of Klebsiella pneumoniae nitrogenase action. Pre-steady-state kinetics of an enzyme-bound intermediate in N2 reduction and of NH3 formation. Biochem J. 1984 Dec 15;224(3):887–894. doi: 10.1042/bj2240887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorneley R. N., Lowe D. J. The mechanism of Klebsiella pneumoniae nitrogenase action. Simulation of the dependences of H2-evolution rate on component-protein concentration and ratio and sodium dithionite concentration. Biochem J. 1984 Dec 15;224(3):903–909. doi: 10.1042/bj2240903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Lin B., Bothe H. Flavodoxin from Azotobacter vinelandii. Arch Mikrobiol. 1972;82(2):155–172. doi: 10.1007/BF01890407. [DOI] [PubMed] [Google Scholar]
- Yates M. G. Electron transport to nitrogenase in Azotobacter chroococcum: Azotobacter flavodoxin hydroquinone as an electron donor. FEBS Lett. 1972 Oct 15;27(1):63–67. doi: 10.1016/0014-5793(72)80410-1. [DOI] [PubMed] [Google Scholar]


