Abstract
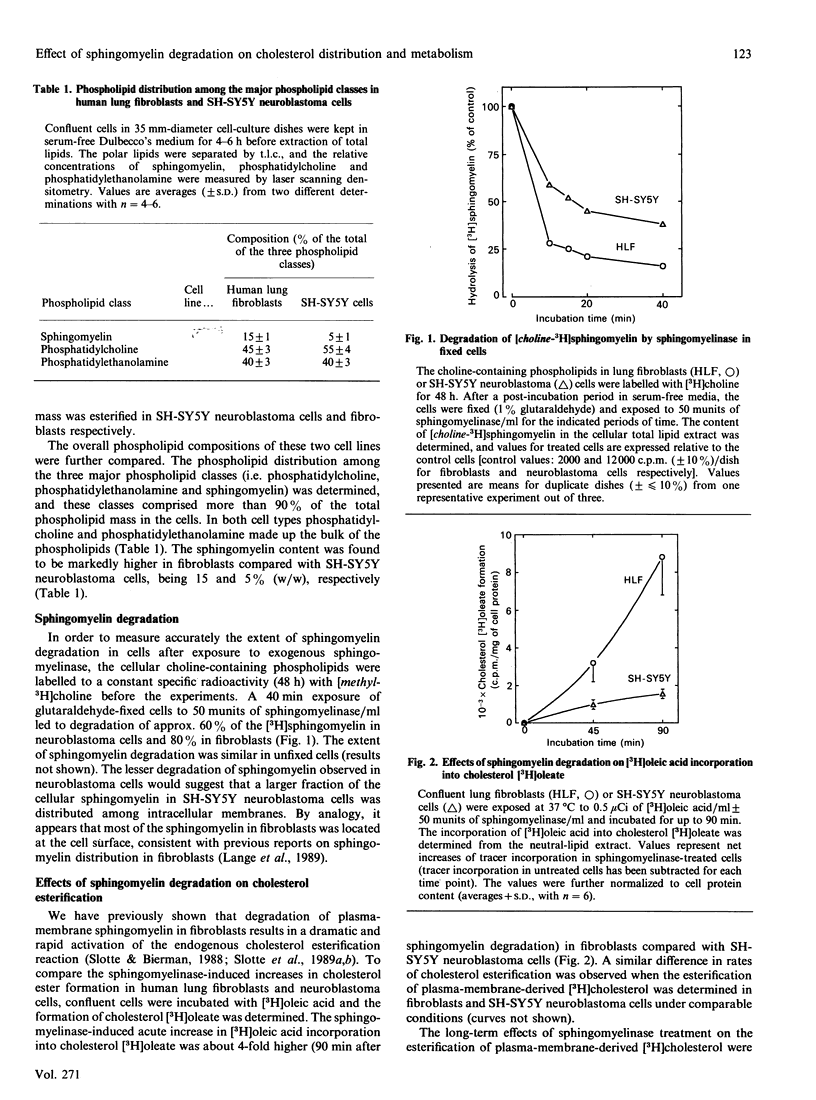
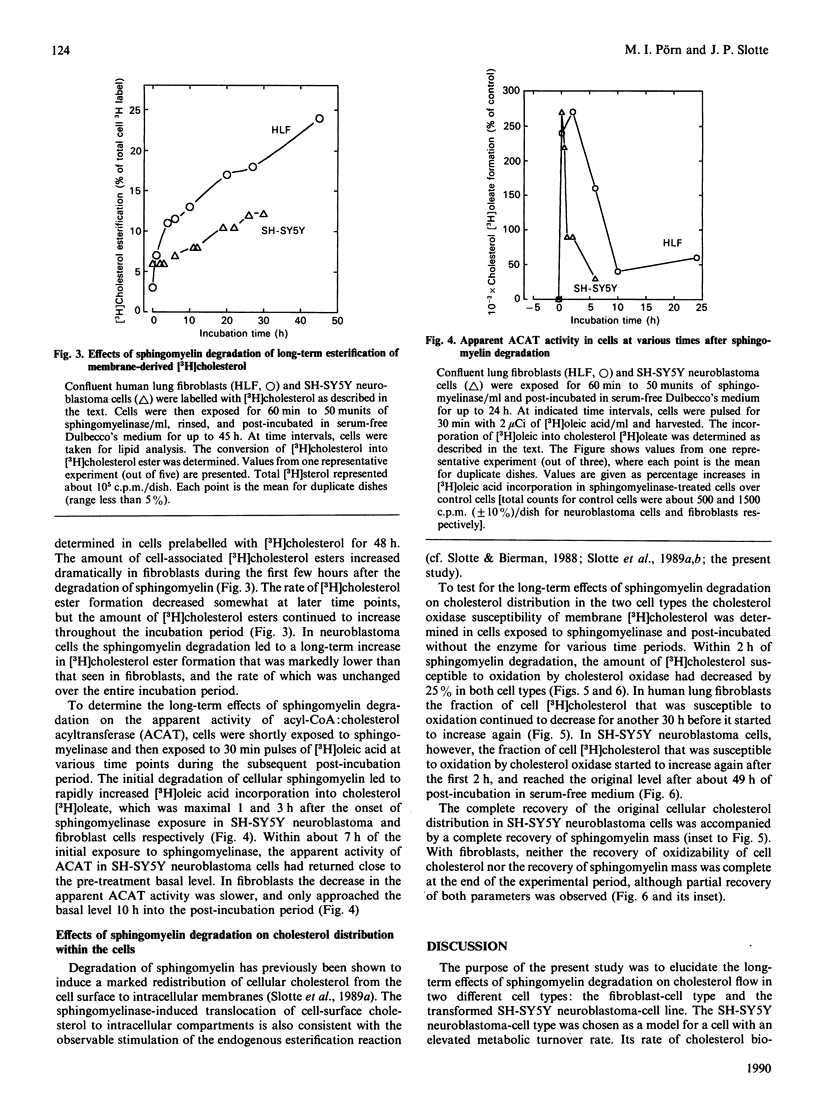
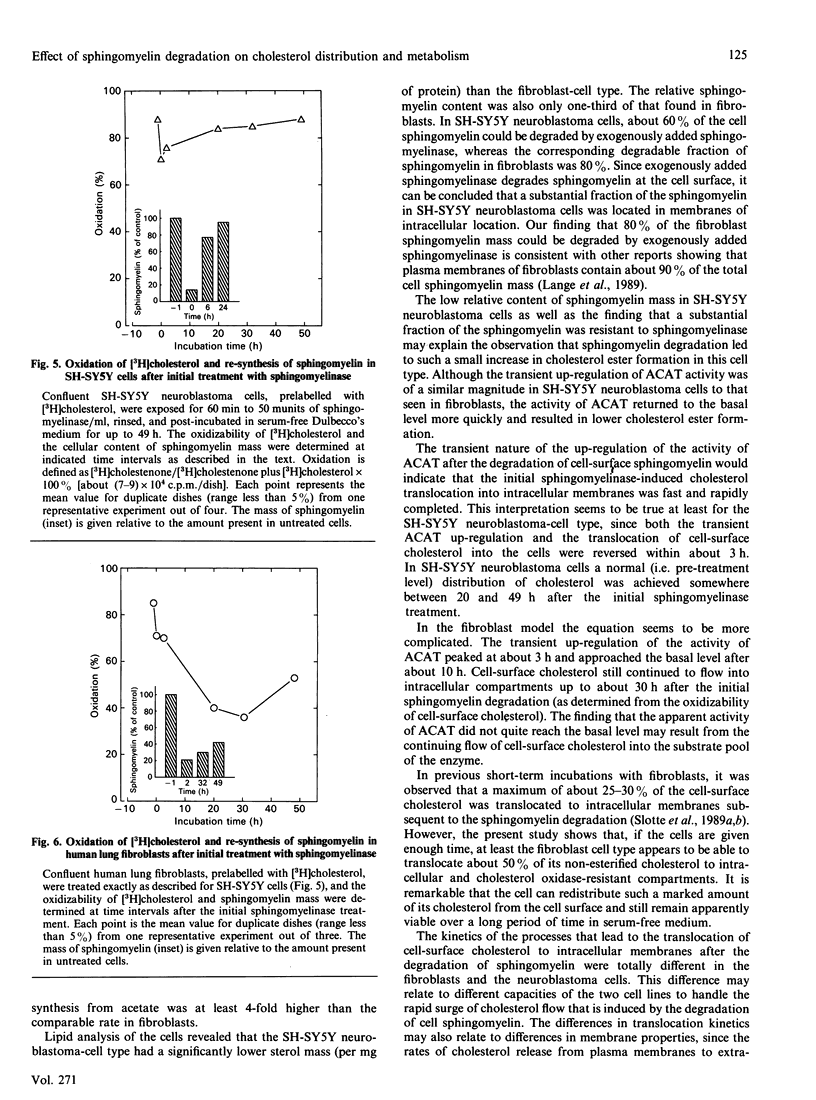
Plasma-membrane sphingomyelin appears to be one of the major determinants of the preferential allocation of cell cholesterol into the plasma-membrane compartment, since removal of sphingomyelin leads to a dramatic redistribution of cholesterol within the cell [Slotte & Bierman (1988) Biochem. J. 250, 653-658]. In the present study we examined the long-term effects of sphingomyelin degradation on cholesterol redistribution in cells and determined the reversibility of the process. In a human lung fibroblast-cell line, removal of 80% of the sphingomyelin led to a rapid and transient up-regulation (3-fold) of acyl-CoA:cholesterol acyltransferase (ACAT) activity, and also, within 30 h, to the translocation of about 50% of the cell non-esterified cholesterol from a cholesterol oxidase-susceptible compartment (i.e. the cell surface) to oxidase-resistant compartments. At 49 h after the initial sphingomyelin degradation, the cell sphingomyelin level was back to 45% of the control level, and the direction of cell cholesterol flow was toward the cell surface, although the original distribution was not achieved. In a transformed neuroblastoma cell line (SH-SY5Y), the depletion of sphingomyelin led to a similarly rapid and transient up-regulation of ACAT activity, and to the translocation of about 25% of cell-surface cholesterol into internal membranes (within 3 h). The flow of cholesterol back to the cholesterol oxidase-susceptible pool was rapid, and a pretreatment cholesterol distribution was reached within 20-49 h. Also, the resynthesis of sphingomyelin was faster in SH-SY5Y neuroblastoma cells and reached control levels within 24 h. The findings of the present study show that the cellular redistribution of cholesterol, as induced by sphingomyelin degradation, is reversible and suggest that the normalization of cellular cholesterol distribution is linked to the re-synthesis of sphingomyelin.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bellini F., Phillips M. C., Pickell C., Rothblat G. H. Role of the plasma membrane in the mechanism of cholesterol efflux from cells. Biochim Biophys Acta. 1984 Nov 7;777(2):209–215. doi: 10.1016/0005-2736(84)90422-x. [DOI] [PubMed] [Google Scholar]
- Calhoun W. I., Shipley G. G. Sphingomyelin--lecithin bilayers and their interaction with cholesterol. Biochemistry. 1979 May 1;18(9):1717–1722. doi: 10.1021/bi00576a013. [DOI] [PubMed] [Google Scholar]
- Chen H. W., Kandutsch A. A., Heiniger H. J. The role of cholesterol in malignancy. Prog Exp Tumor Res. 1978;22:275–316. doi: 10.1159/000401203. [DOI] [PubMed] [Google Scholar]
- Clejan S., Bittman R. Decreases in rates of lipid exchange between Mycoplasma gallisepticum cells and unilamellar vesicles by incorporation of sphingomyelin. J Biol Chem. 1984 Sep 10;259(17):10823–10826. [PubMed] [Google Scholar]
- Colbeau A., Nachbaur J., Vignais P. M. Enzymic characterization and lipid composition of rat liver subcellular membranes. Biochim Biophys Acta. 1971 Dec 3;249(2):462–492. doi: 10.1016/0005-2736(71)90123-4. [DOI] [PubMed] [Google Scholar]
- Damen J., Regts J., Scherphof G. Transfer and exchange of phospholipid between small unilamellar liposomes and rat plasma high density lipoproteins. Dependence on cholesterol content and phospholipid composition. Biochim Biophys Acta. 1981 Sep 24;665(3):538–545. doi: 10.1016/0005-2760(81)90268-x. [DOI] [PubMed] [Google Scholar]
- Fugler L., Clejan S., Bittman R. Movement of cholesterol between vesicles prepared with different phospholipids or sizes. J Biol Chem. 1985 Apr 10;260(7):4098–4102. [PubMed] [Google Scholar]
- Gatt S., Bierman E. L. Sphingomyelin suppresses the binding and utilization of low density lipoproteins by skin fibroblasts. J Biol Chem. 1980 Apr 25;255(8):3371–3376. [PubMed] [Google Scholar]
- Heider J. G., Boyett R. L. The picomole determination of free and total cholesterol in cells in culture. J Lipid Res. 1978 May;19(4):514–518. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lange Y., Ramos B. V. Analysis of the distribution of cholesterol in the intact cell. J Biol Chem. 1983 Dec 25;258(24):15130–15134. [PubMed] [Google Scholar]
- Lange Y., Swaisgood M. H., Ramos B. V., Steck T. L. Plasma membranes contain half the phospholipid and 90% of the cholesterol and sphingomyelin in cultured human fibroblasts. J Biol Chem. 1989 Mar 5;264(7):3786–3793. [PubMed] [Google Scholar]
- Lund-Katz S., Laboda H. M., McLean L. R., Phillips M. C. Influence of molecular packing and phospholipid type on rates of cholesterol exchange. Biochemistry. 1988 May 3;27(9):3416–3423. doi: 10.1021/bi00409a044. [DOI] [PubMed] [Google Scholar]
- Schroeder F., Nemecz G. Interaction of sphingomyelins and phosphatidylcholines with fluorescent dehydroergosterol. Biochemistry. 1989 Jul 11;28(14):5992–6000. doi: 10.1021/bi00440a041. [DOI] [PubMed] [Google Scholar]
- Skipski V. P., Peterson R. F., Barclay M. Quantitative analysis of phospholipids by thin-layer chromatography. Biochem J. 1964 Feb;90(2):374–378. doi: 10.1042/bj0900374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slotte J. P., Bierman E. L. Depletion of plasma-membrane sphingomyelin rapidly alters the distribution of cholesterol between plasma membranes and intracellular cholesterol pools in cultured fibroblasts. Biochem J. 1988 Mar 15;250(3):653–658. doi: 10.1042/bj2500653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slotte J. P., Hedström G., Bierman E. L. Intracellular transport of cholesterol in type C Niemann-Pick fibroblasts. Biochim Biophys Acta. 1989 Oct 17;1005(3):303–309. doi: 10.1016/0005-2760(89)90053-2. [DOI] [PubMed] [Google Scholar]
- Slotte J. P., Hedström G., Rannström S., Ekman S. Effects of sphingomyelin degradation on cell cholesterol oxidizability and steady-state distribution between the cell surface and the cell interior. Biochim Biophys Acta. 1989 Oct 2;985(1):90–96. doi: 10.1016/0005-2736(89)90108-9. [DOI] [PubMed] [Google Scholar]
- Wattenberg B. W., Silbert D. F. Sterol partitioning among intracellular membranes. Testing a model for cellular sterol distribution. J Biol Chem. 1983 Feb 25;258(4):2284–2289. [PubMed] [Google Scholar]
- Yeagle P. L. Cholesterol and the cell membrane. Biochim Biophys Acta. 1985 Dec 9;822(3-4):267–287. doi: 10.1016/0304-4157(85)90011-5. [DOI] [PubMed] [Google Scholar]
- Yeagle P. L., Young J. E. Factors contributing to the distribution of cholesterol among phospholipid vesicles. J Biol Chem. 1986 Jun 25;261(18):8175–8181. [PubMed] [Google Scholar]


