Abstract
Left ventricular (LV) global longitudinal strain (GLS) has recently garnered attention as a reliable and objective method for evaluating LV systolic function. One of the key advantages of GLS is its ability to detect subtle abnormalities even when the ejection fraction (EF) appears to be preserved. However, it is important to note that GLS, much like LVEF, is significantly influenced by load conditions. In recent years, researchers and clinicians have been exploring noninvasive myocardial work (MW) quantification as an innovative tool for assessing myocardial function. This method integrates measurements of strain and LV pressure, providing a comprehensive evaluation of the heart’s performance. Notably, MW offers an advantage over GLS and LVEF because it provides a load-independent assessment of myocardial performance. The implementation of commercial echocardiographic software that facilitates the noninvasive calculation of MW has significantly broadened the scope of its application. This advanced technology is now being utilized in multiple clinical settings, including ischemic heart disease, valvular diseases, cardiomyopathies, cardio-oncology, and hypertension. One of the fundamental aspects of MW is its correlation with myocardial oxygen consumption, which allows for the assessment of work efficiency. Understanding this relationship is crucial for diagnosing and managing various cardiac conditions. The aim of this review is to provide an overview of the noninvasive assessment of myocardial by echocardiography, from basic principles and methodology to current clinical applications.
Keywords: Cardiomyopathies, echocardiography, global longitudinal strain, heart failure, myocardial work, speckle-tracking echocardiography, valvular diseases
INTRODUCTION
Cardiac imaging is pivotal in evaluating left ventricular (LV) function and measuring the cardiovascular response to treatment. In the past, physicians relied on invasive ventriculograms to assess LV function.[1] Pressure–volume loops (PVLs), obtained through invasive procedures in the cardiac catheterization laboratory, significantly improved the understanding of cardiac function by measuring myocardial work (MW), correlating it with myocardial oxygen consumption (VO2), and revealing crucial relationships among preload, afterload, and myocardial contractility.[2] However, the impracticality of invasive PVL in routine clinical practice prompted the development of noninvasive techniques.
Transthoracic echocardiography emerged as the go-to method for assessing LV function with the measurement of LV ejection fraction (LVEF). LVEF is widely used in cardiac diseases as a surrogate marker for LV function due to its broad accessibility, ease of estimation, and extensive application in clinical trials and guidelines.[3] However, LVEF has significant limitations, including reliance on geometric assumptions and high sensitivity to external factors like load, leading to reproducibility challenges. Changes in heart geometry and limited sensitivity in detecting declining ventricular function further hinder its accuracy.[3]
These limitations have prompted the exploration of new indices for assessing myocardial function, with LV global longitudinal strain (GLS) emerging as a promising alternative for evaluating LV systolic function in the last decade.[4] Utilizing speckle-tracking echocardiography (STE), GLS offers a semi-automated approach with reduced variability among observers during myocardial contraction analysis.[5] By quantifying peak GLS, STE can identify subclinical LV dysfunction in various cardiovascular conditions such as cardiotoxicity,[6] valvular heart disease,[7] and cardiac amyloidosis (CA).[8] However, several studies have indicated that GLS, like LVEF, is significantly influenced by load dependency, making it susceptible to changes in conditions of elevated pre- or afterload.[9]
In 2018, a novel technique known as MW revolutionized LV function assessment by integrating LV pressure measurement into strain calculations during echocardiography.[10] This innovative approach was validated and made commercially available, providing a more load-independent measure of LV performance compared to GLS. MW incorporates afterload determination through cuff blood pressure, offering a more accurate assessment of LV function.[11]
The transition from invasive ventriculograms to noninvasive echocardiography, the evolution from LVEF to GLS measurement, and the introduction of MW reflect the continuous advancements in cardiac imaging for evaluating LV function. These progressions have enhanced the precision and sensitivity of assessing LV function, particularly in detecting subtle dysfunction across a range of cardiovascular conditions.[12]
The primary objective of this narrative review is to provide an overview of the noninvasive assessment of MW by echocardiography, from basic principles and methodology to current clinical applications.
MYOCARDIAL WORK: CORE PRINCIPLES AND CONCEPTS
Since Otto Frank’s original description in 1895, PVLs have played a crucial role in understanding LV hemodynamics.[13] Frank’s foundational work illustrated the relationship between preload, afterload, and contractility within the LV myocardium through the elegant representation of the PVL. This graphical representation, derived from measurements in the cardiac catheterization laboratory, captures the phases of the cardiac cycle – isovolumic contraction, systolic ejection, isovolumic relaxation, and diastolic filling – in a rectangular form.[14]
The analysis of PVL enables the measurement of the energy imparted to the blood, as the loop area represents stroke work.[15] Suga et al. subsequently showed in an experimental study that pressure–volume area predicts cardiac VO2;[2] this result was further confirmed by Takaoka et al. in a clinical setting.[16] This same principle has been applied to pressure–dimension loops to estimate regional LV function and segmental work. This application has proven particularly useful for studying the synchrony or dyssynchrony of contraction.[17] However, the requirement for invasive measures to estimate pressure–dimension loop area has limited the widespread use of this index.
Recently, Russell et al. introduced a noninvasive method for assessing regional MW through the analysis of LV pressure–strain loops (PSLs).[18] This innovative method derives the LV pressure curve from noninvasively acquired brachial artery cuff pressure. The LV pressure profile is then adjusted according to the duration of the isovolumic and ejection phases, which are defined by the timing of aortic and mitral valve events as recorded by echocardiography. Thus, LV PSL integrates the estimated LV pressure curve with strain measurements obtained through STE.
In their experimental study, Russell et al. demonstrated a good agreement between the proposed noninvasive method and the PVL area obtained via invasive measures.[18] Moreover, they found a strong correlation between noninvasive PSL area and regional glucose metabolism using 18-fluorodeoxyglucose-positron emission tomography.[18,19] These findings support the use of this noninvasive method as a reliable index of regional MW, encouraging its inclusion in clinical practice.
HOW TO PERFORM MYOCARDIAL WORK ANALYSIS THROUGH ECHOCARDIOGRAPHY
MW assessment utilizes a vendor-specific algorithm from GE Healthcare (Pewaukee, WI).[20] The initial step involves obtaining transthoracic views for GLS analysis. Three standard apical views are captured at a frame rate >40 frames/s, ensuring image quality adequate to visualize myocardial borders. STE is employed for GLS analysis, and valvular event timings can either be automatically assessed by the machine or manually adjusted through visual assessment from the apical long-axis view. Automated functional imaging calculates GLS from the apical two-, three-, and four-chamber long-axis views, with manual adjustments possible to align with the myocardium. This analysis results in a bull’s-eye GLS plot displaying the GLS values.[21]
Subsequently, noninvasive systolic blood pressure measurement is performed using a sphygmomanometer on the arm. The peak LV systolic pressure (LVSP) is estimated from the noninvasive systolic cuff pressure and should be recorded at the time of image acquisition. This pressure reading is then input into the echocardiography machine for MW analysis.
MW enhances automated functional imaging by incorporating dynamic LV pressures. Once the blood pressure is entered into the software, a MW bull’s-eye plot is generated, similar to GLS. Four key values are calculated: global work index (GWI), global constructive work (GCW), global wasted work (GWW), and global work efficiency (GWE) [Figure 1].[11] The explanation of the main MW parameters is listed below:
Figure 1.

The left ventricular (LV) global longitudinal strain (GLS) data, measured using the R-wave onset in the electrocardiogram as a common time reference (left panel), are combined with peak arterial pressure, measured using a cuff manometer (central panel). The right panel displays a representative trace showing the LV pressure–strain loop and a 17-segment bull’s-eye representation of myocardial work index (top), along with 17-segment bull’s-eye representations of GLS and myocardial work efficiency (bottom)
GWI represents the average work performed by the myocardium derived from the PSL
GCW indicates positive work performed during systole and negative work (segment lengthening) during isovolumic relaxation
GWW reflects negative work (segment lengthening) during systole and positive work (segment shortening) during isovolumic relaxation
GWE is calculated as GCW/(GCW + GWW).
Similar to GLS, color-coding visually distinguishes high, normal, and reduced MW. Each segment can be individually analyzed for its MW indices. PSLs can be generated globally and for individual segments. Normal ranges in healthy individuals were assessed in the European Association of Cardiovascular Imaging Normal Reference Ranges for Echocardiography Study[22] and in a larger and more recent study conducted by Olsen et al. on 1827 participants, included as part of the fifth study visit of the Copenhagen City Heart Study.[23]
EXPLORING TECHNICAL LIMITATIONS IN MYOCARDIAL WORK ANALYSIS
Because MW analysis is derived from GLS analysis, the limitations of GLS acquisition also apply to MW. Poor image quality and low frame rates hinder speckle-tracking, making it impossible to assess either GLS or MW. Without quality imaging, speckle-tracking and MW analysis are not feasible. A precise and timely brachial cuff pressure reading is also necessary for the analysis. Atrial fibrillation and other abnormal rhythms can impede GLS acquisition, especially with significant heart rate variability.[21] Intra- and inter-observer variability of MW is similar to GLS analysis. Beyond GLS analysis and the input of blood pressure, the values generated are automatically produced by an algorithm. Additionally, accurate noninvasive cuff pressure measurements are crucial for MW acquisition.[12] MW analysis cannot be performed in clinical situations where LVSP does not equal noninvasive systolic blood pressure, such as in patients with severe aortic stenosis (AS) or fixed LV outflow tract obstruction; these patients were excluded from the original validation studies.[18]
Blood pressure should be measured in the arm, using a sphygmomanometer, while the patient is in the same position as the one used during image acquisition to ensure it accurately reflects the LV’s afterload at the time images are acquired. Strain analysis may use different algorithms depending on the vendor platform.
Currently, there is only one vendor platform with a noninvasive MW algorithm, making MW dependent solely on this available software.
In addition to technical limitations, other constraints can arise from patient-specific factors or their underlying pathology. The accuracy of measuring MW may be compromised in patients with significant LV remodeling.[24] PSLs do not account for the direction of blood flow, which is particularly important in conditions like severe mitral regurgitation. Furthermore, this analysis neglects critical factors such as wall stress, thickness, and curvature, all of which contribute to afterload. The construction of PSL also fails to incorporate variations in diastolic pressure. Research by Lembo et al. has shown that cohorts with elevated diastolic blood pressure exhibit altered indices of MW, underscoring the need for further investigation.[25]
HYPERTENSION
Arterial hypertension (HTN) serves as an excellent model to examine myocardial deformation and performance changes due to pressure overload and the development of LV concentric geometry. Arterial pressure is a significant independent predictor of accelerated decline in GLS during follow-up.[26,27] Investigating LV function in hypertensive (HT) patients through MW can account for acute loading during the cardiac cycle, isolating the influence of blood pressure at the time of observation from chronic remodeling’s impact on regional deformation. Chan et al. identified increased GWI and GCW values in a small group with varying HTN degrees, especially in those with uncontrolled HTN. These values reflect the enhanced contractility of the LV, necessitated by pumping blood against elevated pressures.[28] Tadic et al. confirmed these findings and showed that type 2 diabetes mellitus further affects GCW in HT patients.[29]
An analysis of 170 HT patients revealed an apex-to-base gradient in MW distribution, indicating basal segment impairment compensated by the apical region, particularly in patients with basal septal hypertrophy.[30]
Huang et al. conducted a study on segmental and global MW in HT patients with various LV geometries.[31] They divided 125 HT patients into four groups: normal geometry, concentric remodeling, concentric hypertrophy (CH), and eccentric hypertrophy (EH). Utilizing echocardiography, they measured indices such as GWI, GCW, GWW, and GWE. They observed a gradual decrease in GLS across the groups (P < 0.001). Despite no changes in GWI and GCW, GWW increased and GWE decreased in CH and EH groups (P < 0.001). Segmental analysis showed a reduction in longitudinal strain in basal and middle regions across all HT subgroups, with apical reduction only in CH and EH groups (P < 0.001). Significant findings included the association between left atrial volume index (LAVI), GLS, and GWE with LV hypertrophy. This study demonstrates that apical MW increases initially as a compensatory mechanism in LV remodeling, maintaining normal GWI.[31] Thus, MW appears more sensitive in the context of HT patients. Larger scale studies are needed to establish MW indices’ clinical utility and prognostic impact on cardiovascular outcomes in HT patients. An example of MW analysis application in the context of an HT patient is shown in Figure 2.
Figure 2.

Hypertension. Blood pressure at the time of the echocardiogram was 185/90 mmHg. Left ventricular ejection fraction (63%) and global longitudinal strain (-19%) are both preserved (a), global work index is elevated (2393 mmH%) with multiple, red-shaded segments, especially in the apex (b), pressure-strain loop is normal (c) and global work efficiency is relatively unaffected (92%), as demonstrated by all green-shaded segments, except for the basal septum that is yellow-shaded which indicates reduced efficiency (d). GWE = Global work efficiency, GWW = Global wasted work, GCW = Global constructive work, GWI = Global work index, GLS = Global longitudinal strain, BP = Blood pressure
CORONARY ARTERY DISEASE
Detecting early ischemia in patients with significant coronary artery disease (CAD) and normal resting systolic function remains challenging and continues to be investigated.[32] Many questions also remain in the context of myocardial infarction with nonobstructive coronary arteries.[33] Previous studies have shown that GLS is a strong predictor of stable ischemic cardiopathy, even in the absence of wall motion abnormalities.[34,35] However, there is currently a lack of agreement on the most effective GLS diagnostic cutoff value, as it can vary greatly among studies as a result of clinical characteristics, dependence on afterload, or differences between vendors. Furthermore, the contractile patterns of ischemic myocardium are strongly affected by loading conditions, often resulting in rapid shifts from hypokinesis to dyskinesis following a sudden increase in afterload.[36] MW estimation has shown to overcome these limitations and provide valuable diagnostic and prognostic information in both chronic and acute settings.[37]
Edwards et al. demonstrated that in patients with suspected CAD and normal systolic function, in the presence of obstructive coronary disease, GWI, GCW, and GWE are significantly lower, while GWW is slightly increased. They reported that a GWI value of 1810 mmHg% could detect CAD with a 95% positive predictive value. Notably, GWI outperformed GLS in predicting significant CAD, with an area under the curve (AUC) of 0.786 for GWI compared to 0.693 for GLS.[38]
In non-ST-segment acute coronary syndrome, the regional MW index has outperformed other echocardiographic parameters (such as GLS and LVEF) in identifying acute coronary artery occlusion.[39] The presence of four adjacent segments with an MW index <1700 mmHg% showed 81% sensitivity and 82% specificity in detecting coronary occlusion, with a negative predictive value of 94%. This surpasses the functional risk area measured by strain, which has 78% sensitivity, 65% specificity, and a 91% negative predictive value.[39]
The superiority of GWI in identifying patients with acute coronary occlusion is evident when compared to LVEF, showing 70% sensitivity versus 63%, 82% specificity versus 62%, and a negative predictive value of 91% versus 86%.[40] In a study involving 93 patients with anterior ST-elevation myocardial infarction, all MW indices were reduced in the left descending coronary artery territory. However, a significant improvement was observed at a 3-month follow-up in patients who experienced LV recovery. Among the indices, GCW demonstrated an independent and incremental value in predicting both segmental and global LV recovery, outperforming standard parameters like LVEF and advanced parameters like GLS. GCW also predicted inhospital complications such as heart failure (HF) or LV apical thrombus.[41]
Conversely, STEMI patients who experienced LV ischemic remodeling at the 3-month follow-up exhibited lower levels of GWI, GCW, and GWE but higher levels of GWW.[42] Supporting these findings, El Mahdiui et al. showed that GWE is lower in patients who have undergone primary percutaneous coronary intervention for STEMI compared to healthy controls and those with cardiovascular risk factors and becomes even more impaired in the presence of HF with reduced ejection fraction (HFrEF).[43] These results suggest that MW impairment reflects altered (persistent anaerobic) energy metabolism occurring in remodeled myocardium. An example is provided in Figure 3.
Figure 3.

Coronary artery disease. A patient presenting with an anterior ST elevation myocardial infarction caused by occlusion at the mid-segment of the left anterior descending artery. Left ventricular ejection fraction is mildly reduced (48%). (a) The global longitudinal strain (GLS) bull’s eye shows impaired longitudinal deformation of apical and mid-anteroseptal segments with coronary distribution and reduced GLS value (−7%), (b) Global work index bull’s eye shows the regional impairment of myocardial work index in the same segments that show reduced longitudinal deformation, (c) The pressure–strain loop shows a reduced area under the curve, and it is shifted toward right, which means a significant impairment of myocardial work, (d) Global work efficiency (GWE) bull’s eye shows impairment of myocardial work efficiency especially in apical segment, that are red shaded, with a GWE significantly reduced (72%). GWE = Global work efficiency, GWW = Global wasted work, GCW = Global constructive work, GWI = Global work index, GLS = Global longitudinal strain, BP = Blood pressure
Stress echocardiography using noninvasive MW estimation is valuable for addressing loading changes during the test. Halabi et al. found MW feasible during stress echocardiography, with peak stress GWI correlated to functional capacity.[44] In the context of CAD, MW analysis has shown promising results. Lin et al. studied 85 angina pectoris patients without prior CAD history, who underwent treadmill exercise stress echocardiography and coronary angiography.[45] Global MW, derived from STE, was analyzed using logistic regression to assess CAD presence. Significant coronary stenosis was found in 41 patients, with 25 showing positive stress echocardiograms. Higher GWW and lower GWE were observed in significant CAD cases (P < 0.05). Peak GWE and recovery GWW effectively predicted CAD, with combined parameters improving identification accuracy. Global MW with exercise stress provides accurate noninvasive CAD screening.[45]
HEART FAILURE AND CARDIAC RESYNCHRONIZATION THERAPY
The first promising use of segmental MW is predicting therapeutic outcomes in HF patients undergoing cardiac resynchronization therapy (CRT). CRT is essential for treating symptomatic HF patients with LVEF ≤35% and a wide QRS complex.[46] However, about 30% of patients do not experience significant benefits from CRT. These patients necessitate more frequent clinical reassessments, both in person and via telemedicine, leading to increased healthcare costs.[47]
Efforts to identify echocardiographic parameters to predict CRT response have not yielded promising results. A multicenter trial with 498 patients found that none of the 12 conventional and tissue Doppler-based echocardiographic dyssynchrony indices reliably predicted CRT response. Although longitudinal, circumferential, and radial strain and strain rate have shown good accuracy in identifying CRT-beneficial patients, prospective randomized trials using STE are still needed.[48,49,50]
Some authors have proposed that assessing residual myocardial contractility in patients with dyssynchrony might predict LV functional recovery after CRT. In this context, MW demonstrates the potential for identifying patients likely to experience myocardial improvement postresynchronization. A recent study found that GWW and septum average MW were higher in CRT responders than nonresponders. Post-CRT implantation, both indices significantly reduced to near-normal heart values.[51]
Moreover, combining septal wasted work (WW) with the LV wall motion score, used to identify transmural myocardial scar, improved CRT response prediction. The AUC for combined parameters was 0.86, compared to 0.80 for septal WW and 0.63 for the wall motion score alone. This suggests that a combined approach is beneficial.[51]
A larger prospective multicenter study, with 200 CRT recipients, demonstrated that the work difference between septum and lateral wall is a strong predictor of CRT response, yielding an AUC of 0.77.[52] Combining this work difference with septal viability, evaluated via cardiac magnetic resonance, increased the AUC to 0.88, with 86% sensitivity and 84% specificity. This combination proved superior to other electrocardiographic and echocardiographic parameters in predicting CRT response. Further, MW redistribution across the septal and lateral walls after CRT is a useful index for predicting long-term reverse remodeling.[53]
In a cohort of 97 patients, Galli et al. showed that GCW is an independent predictor of CRT response at 6 months and is significantly associated with myocardial remodeling extent in both ischemic and nonischemic patients.[54] Despite the promising results, comparisons of AUCs did not reveal a statistically significant difference, emphasizing the necessity for further research to confirm these findings and potentially refine predictive models for CRT responses.
The available evidence on these new echocardiographic parameters is insufficient to guide treatment decisions independently. However, an integrative approach combining clinical, electrocardiographic, and advanced echocardiographic parameters may assist in identifying suitable candidates for CRT.
In patients with HFrEF, treatment with sacubitril/valsartan has demonstrated a significant increase in GCW after a 6-month follow-up period. Moreover, improvements in GWE were observed at the 12-month follow-up compared to baseline, suggesting a reduction in LV wall stress and an increase in myocardial metabolism induced by this medication.[55] Conversely, a baseline GCW value below 910 mmHg% is indicative of high cardiovascular risk in patients.[55] Furthermore, in patients with HF with preserved ejection fraction (HFpEF), an increase in GCW during exercise has been associated with improved 6-month follow-up exercise capacity in response to spironolactone treatment.[56] Nonetheless, the prognostic significance of MW indices in HF patients remains inconclusive, warranting further research to determine the utility of MW in clinical practice for this patient population.
HYPERTROPHIC CARDIOMYOPATHY AND ATHLETE’S HEART
Hypertrophic cardiomyopathy (HCM) and athlete’s heart exhibit similar features on echocardiography, making it challenging to differentiate between adaptive hypertrophy and cardiomyopathy.[57]
Cardiac magnetic resonance imaging (CMRI) enables an optimal depiction of hypertrophic phenocopies through detailed tissue characterization. However, it has limitations, including being time-consuming and often impractical for patients with conditions such as claustrophobia.[58]
Various studies have examined GLS, radial strain, LV twist, and atrial strain[59] as potential methods of distinguishing HCM from other hypertrophic phenocopies, such as athlete’s heart and amyloidosis; however, there is no consensus on the effectiveness of strain in this particular population.[60,61,62,63,64] Reduced GLS is frequently observed in patients with HCM and is often correlated with fibrosis detected on CMRI.[65]
Recently, there has been interest in MW as a valuable tool for assessing LV systolic function in individuals with HCM. Galli et al. demonstrated that GCW is significantly decreased in patients with nonobstructive HCM when compared to healthy controls.[54] Their research also revealed a noteworthy association between GCW and substantial fibrosis on CMRI. Similarly, Gonçalves et al. found that a GCW cutoff of ≤1550 mmHg% was linked with ≥15% late gadolinium enhancement on CMRI, with high sensitivity and specificity.[66] Hiemstra et al. showed that a reduction in MW parameters in patients with nonobstructive HCM was significantly linked to poorer long-term outcomes, with patients exhibiting a GCW >1730 mmHg% showing better event-free survival.[67]
Regarding differential diagnosis in the context of hypertrophic phenocopies, de Gregorio et al. assessed MW in transthyretin amyloid cardiomyopathy (ATTR-CM) and nonobstructive HCM versus well-controlled HTN patients.[68] The study involved 83 participants: 32 with ATTR, 29 with HCM, and 22 HTN controls. ATTR patients had higher LV mass and lower LVEF than HCM patients and HTN controls. ATTR was further divided into ATTR1 with LVEF ≤50% and ATTR2 with LVEF >50%. GWI and GCW were notably lower in ATTR, especially in ATTR1 group, and GWI effectively distinguished ATTR2 from HCM better than GLS and LVEF.[68] An example of MW analysis application to HCM is provided in Figure 4.
Figure 4.
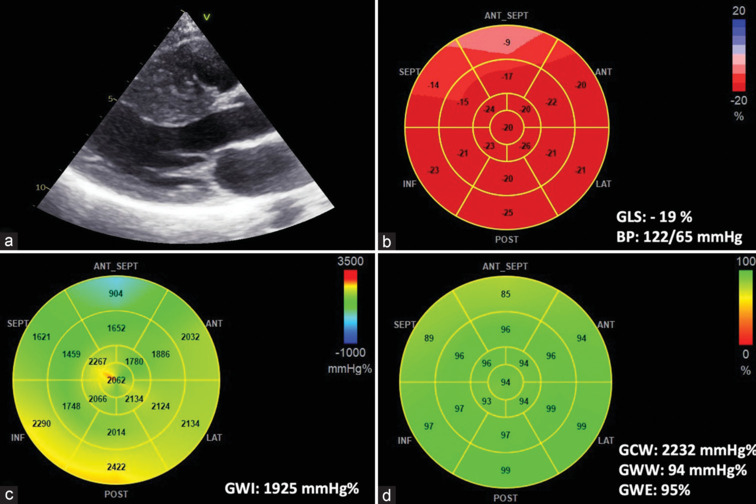
Hypertrophic cardiomyopathy. (a) Parasternal long axis view showing severe hypertrophy of the interventricular septum in a patient affected by sarcomeric hypertrophic cardiomyopathy, (b) Global longitudinal strain’s bull’s eye showing impairment of longitudinal deformation in anterior-septum segments, interested by hypertrophy, (c) Myocardial work index bull’s eye showing a decreased amount of myocardial work performed by basal anteroseptal segment, light-blue shaded, (d) Myocardial work efficiency’s bull’s eye showing preserved global work efficiency of 95% and decreased regional work efficiency in segments interested by hypertrophy. GWE = Global work efficiency, GWW = Global wasted work, GCW = Global constructive work, GWI = Global work index, GLS = Global longitudinal strain, BP = Blood pressure
Unlike HCM, LV hypertrophy in athletes is considered to be an adaptative and reversible response to training. Athletes typically show increased end-diastolic volume with normal or slightly reduced LVEF, which significantly increases during exertion. MW, taking into account afterload – often decreased in endurance athletes – may offer a more precise measurement of LV function. In a study conducted by D’Andrea et al. comparing STE strain and MW in 350 endurance athletes with 150 healthy controls, it was found that while resting LV GLS was lower in endurance athletes compared to controls (−18.4 ± 2.6 vs. −22.4 ± 3.3%; P < 0.01), GWE showed no significant difference.[69] Furthermore, GWE at rest was closely linked to maximal watts, peak VO2, and LV E/e’ during peak exertion. Regular exercise training was associated with decreased GLS but increased GWI at rest, with GWI positively correlated with VO2/kg.[70] These findings suggest that GWI at rest may serve as a more precise predictor of an athlete’s heart performance compared to other indicators of LV systolic function. In a preliminary study, Junianto et al. highlighted that in elite air force soldiers, higher intensity of physical training was associated with greater LV GLS, GWE, GCW, and GWI and a lower GWW. This suggests more significant physiological adaptations compared to lower-intensity training.[71]
Even among elite power athletes, D’Andrea et al. demonstrated that GWE at rest was the strongest predictor of maximum watts (P < 0.0001), peak VO2 (P < 0.0001), increase in pulmonary artery systolic pressure (P < 0.001), and the number of B-lines (P < 0.001), all measured at peak effort.[72]
The application of MW analysis in stress echocardiography to evaluate myocardial performance in athletes, both endurance and power athletes, has highlighted in the study conducted by Borzì et al. that at peak exertion, both groups exhibit a significant increase in GWI compared to controls (nonathletes) and reduced GWE values in power athletes compared to endurance athletes.[73] This underscores that endurance athletes demonstrate better cardiovascular performance, but further studies are needed to confirm these results.
AMYLOIDOSIS
Advancements in cardiac imaging have led to more accurate and timely diagnosis of CA in the echocardiography laboratory.[8,63,74] STE has proven to be a reliable tool in identifying cardiac amyloid, particularly through the observation of relative apical sparing which serves as a sensitive and specific marker of the disease.[59,63,75,76] Studies have also explored MW in patients with CA, with findings indicating significantly reduced GWI and GWE in these individuals compared to controls.[68,77,78] While MW demonstrated lower accuracy in diagnosing ATTR-CM compared to parameters such as relative wall thickness or relative apical sparing measured by GLS, it showed a significant correlation with NT-proBNP and troponin levels.[79] Research further revealed correlations between GWI and NT-proBNP, mortality, and peak VO2 in ATTR-CM patients, although performance was not superior to GLS.[80] On the contrary, GWI was found to be predictive of major adverse cardiac events and all-cause mortality by Clemmensen et al.[81] In the context of hypertrophic phenocopies, de Gregorio et al.[82] assessed systemic vascular resistance (SVR) and GWI in HFpEF patients. Sixty outpatients, divided equally between those with nonobstructive HCM and wild-type ATTR, were examined. Participants were classified into four functional classes using SVR and GWI thresholds from earlier studies.[81,83,84] Results showed 85% had high SVR, with 100% ATTR and 70% HCM affected. Furthermore, 43% of HCM and 93% of ATTR patients had impaired SVR and GWI, suggesting that this classification may provide deeper insights into the pathophysiological and clinical characteristics of these hypertrophic phenotypes.[82]
The utility of MW indices in the context of CA warrants additional investigation to determine their specific role in clinical management. The description of a case with CA in Figure 5 depicts increased LV wall thickness alongside the characteristic apical sparing GLS pattern indicative of amyloidosis, with evidence of normal work at the apex and negative work at the base shown through GWI. A case of ATTR is shown in Figure 6.
Figure 5.

Dilatative cardiomyopathy. (a) Four-chamber view demonstrates severely dilated left ventricle in a patient with nonischemic dilatative cardiomyopathy and left ventricular ejection fraction equal to 30%, (b) A bull’s-eye plot for the same patient correlates with a severely reduced global longitudinal strain (−4%), (c) A bull’s-eye plot demonstrates severely decreased global work index (397 mmHg%), (d) Myocardial efficiency bull’s eye showing diffuse impairment of work efficiency with global work efficiency severely impaired and equal to 66%. GWE = Global work efficiency, GWW = Global wasted work, GCW = Global constructive work, GWI = Global work index, GLS = Global longitudinal strain, BP = Blood pressure
Figure 6.

Transthyretin cardiac amyloidosis. (a) Four-chamber view demonstrates severely thickened left ventricular walls and granular sparkling aspect of the interventricular septum, (b) A bull’s-eye plot of strain from the same patient demonstrates the typical apical sparing seen in amyloid heart disease, (c) A bull’s-eye plot of myocardial work index in the same patient shows decreased global work index (global work index; 824 mmHg%) with normal work index at the apex and negative work at the base, (d) Work efficiency bull’s eye showing preserved myocardial work efficiency at the apical segments. GWE = Global work efficiency, GWW = Global wasted work, GCW = Global constructive work, GWI = Global work index, GLS = Global longitudinal strain, BP = Blood pressure
DILATED CARDIOMYOPATHY
Accurately characterizing LV function is crucial for patients with dilated cardiomyopathy (DCM) to determine prognosis, adjust treatment plans, and monitor for any deterioration. GLS has been proven to be a more precise predictor of prognosis in HF patients than LVEF.[85,86,87] The utilization of PVL in assessing DCM is a relatively recent development. Both invasive and CMRI assessments of PVL have shown diminished MW in patients with DCM compared to healthy individuals.[88]
Recently, noninvasive echocardiographic evaluation of MW has been investigated as a method to assess LV performance in DCM patients, revealing reduced GWI, increased GWW, and reduced GWE compared to controls.[28] In previous studies, researchers found that reduced GWI, GCW, and GWE were all better indicators of fibrosis on CMRI compared to GLS.[89] Furthermore, GWI and GCW have been shown to predict a combined endpoint of all-cause mortality, LV assist device implantation, or heart transplantation in patients with advanced HF.[90] One study even demonstrated that treatment with sacubitril/valsartan led to improved GCW and GWE.[55] More research is needed to comprehensively evaluate MW parameters in patients with DCM to better understand the potential of this technique in characterizing myocardial performance and tracking changes over time in response to the natural progression of the disease and various treatments.
Detailed in Figure 5 is a case of a patient with nonischemic DCM, showcasing severely reduced GWI and an abnormal PSL indicative of severe LV dysfunction.
VALVULAR HEART DISEASE
Aortic stenosis
AS is a disease that affects both the valve and the LV myocardium, as it tries to compensate for the fixed obstruction. While LVEF may be preserved initially, GLS is often found to be abnormal in severe AS. This indicates subclinical myocardial dysfunction even when LVEF appears normal. Studies have linked abnormal GLS to poor clinical outcomes in severe AS patients, with GLS abnormalities persisting in some individuals even after AS correction.[91] This suggests that abnormal GLS can identify subclinical LV dysfunction, possibly indicating a later stage of the disease where compensatory mechanisms of the LV have already failed.
In this context, MW has shown promise in assessing AS, since both GLS and LVEF are dependent on afterload. Initial validations excluded patients with severe AS because systolic blood pressure was unsuitable as a surrogate for LVSP due to fixed obstruction. Jain et al. studied transcatheter aortic valve replacement (TAVR) patients in the cardiac catheterization laboratory, directly measuring LVSP, brachial cuff pressure, left radial arterial pressure, and echocardiographic parameters.[92] Their findings showed that a noninvasive method for assessing LVSP in severe AS (systolic blood pressure + mean aortic gradient) closely matched invasively measured LVSP (r = 0.92).[92] Fortuni et al. confirmed this correlation in 120 severe AS patients undergoing TAVR (r = 0.846; P < 0.001).[93]
MW can help distinguish between two severe AS profiles: patients with abnormal GLS and high GWI and those with abnormal GLS and decreased GWI; the latter group may indicate more persistent LV dysfunction.[92] De Rosa et al. observed decreased GWI and GCW postintervention, similar to Jain et al.’s findings.[92] Jain et al. documented reductions in GWI and GCW from pre-TAVR to post-TAVR. De Rosa et al. also noted that low-flow, low-gradient AS patients did not experience improvement in GWI post-TAVR.[94] Lastly, Ilardi et al. recently investigated the prognostic significance of MW in AS patients. They observed higher GWI, GCW, and GWW in AS patients compared to controls and found that decreased GWI (hazard ratio [HR]: 0.998, 95% confidence interval [CI]: 0.997–1.000, P = 0.034) and GCW (HR: 0.998, 95% CI: 0.997–0.999, P = 0.003) measurements after follow-up were significantly linked to increased mortality.[95] Further research is needed to assess the effectiveness of MW indices in severe AS for risk assessment and postintervention clinical outcomes.
Aortic regurgitation
Chronic aortic regurgitation (AR) patients may be asymptomatic for an extended period before experiencing symptoms. The evaluation of LV dysfunction is crucial in decision-making for asymptomatic AR patients. In this context, Meucci et al.[96] examined 57 patients with chronic severe AR and preserved LVEF who underwent surgical aortic valve replacement. Their findings indicated that baseline LVEF was normal along with normal to increased GWI and GCW (LVEF: 59.7% ±4.4%, GWI: 2084 ± 483 mmHg%, and GCW: 2482 ± 525 mmHg%). GWI and GCW were positively correlated with markers of AR severity, effective regurgitant orifice area, and regurgitant volume.[96] Moreover, the surgical intervention resulted in a reduction in GWI, GCW, and GWE. It was observed that impaired GWI following the surgery was linked to unfavorable LV reverse remodeling, with a threshold of 1285 mmHg% for GWI identified as a predictor (AUC: 0.790, 95% CI: 0.651–0.929, P = 0.001). Another study involving 151 patients with severe, asymptomatic AR conducted by D’Andrea et al.[97] revealed that baseline GLS and GWE were significantly lower in individuals with normal LVEF compared to healthy controls. The use of MW may offer valuable insights into determining the optimal timing for intervening in AR to prevent postoperative myocardial dysfunction.
Mitral valve disease
Functional mitral regurgitation (FMR) results from LV dysfunction impacting mitral valve function.[98] MW offers additional insights into LV performance in FMR patients and has been linked to outcomes and survival.[99]
Verbeke et al. assessed MW in 181 HF and FMR patients, finding GWI and GLS to be independently associated with cardiovascular mortality and HF hospitalization (GWI: HR 0.927, P = 0.034; GLS: HR 0.884, P = 0.015). However, GWI did not significantly surpass GLS (delta-2 log-likelihood = 0.8, P = 0.37).[99]
Yedidya et al. studied 373 FMR patients and found that GWI, GCW, and GWW were significantly lower, while GWE was higher in severe FMR cases compared to mild and moderate ones. These parameters correlated with worse long-term survival: GWI ≤500 mmHg%, GCW ≤750 mmHg%, and GWW <300 mmHg%.[100]
In FMR patients treated with percutaneous edge-to-edge mitral valve repair, MW was also evaluated.
Papadopoulos et al. found a significant increase in GWI and GCW postintervention in 86 patients. Baseline LVEF, GLS, GWI, and GCW were linked to reduced LV end-diastolic volume 1 year postintervention, while baseline GCW was associated with a reduction in LV end-systolic volume (P < 0.05 for all).[101]
At 6-month follow-up, Hubert et al. observed in 56 patients a significant postintervention improvement in GCW (977 ± 366 vs. 1101 ± 404 mmHg%, P = 0.003), while LVEF and GLS remained unchanged. For patients with LVEF < 35%, GWI predicted poor outcomes (AUC 0.882, P = 0.009).[102]
Yedidya et al. studied 70 FMR patients, categorizing them into improvers (≥20% increase in forward stroke volume [SV] index) and nonimprovers (<20%). Nonimprovers exhibited worsened GWI and GCW (P = 0.005, P = 0.004), while improvers showed no change (P = 0.093, P = 0.112). GWE correlated with SV improvement postintervention, independent of LV systolic function.[103]
Regarding severe rheumatic mitral stenosis, Rudiktyo et al. evaluated 33 patients with isolated mitral stenosis and demonstrated a direct and positive correlation between GWE and the patient’s functional capacity, objectively measured through exercise testing.[104]
RIGHT VENTRICULAR MYOCARDIAL WORK
Assessing right ventricular (RV) function remains clinically challenging. Various echocardiographic parameters such as tricuspid annular plane systolic excursion, fractional area change, and S’ are used, each with technical limitations. Limited studies have explored RV MW using a standard algorithm.
In a recent study conducted by Landra et al. on 23 patients who underwent LVAD implantation, it was demonstrated that, among MW indices, the performance for early right HF prediction was greatest for RV GWE. The AUC for RV GWE was 0.92, with a cutoff of 77%, yielding 100% sensitivity and 82% specificity. During long-term follow-up, death occurred in 4 of 14 patients (28.6%) in the group with RVGWE >77%, compared to 6 of 9 patients (66.7%) in the group with RVGWE <77%, resulting in an HR of 0.25 (95% CI: 0.07–0.90, P = 0.033).[105]
Butcher et al. found RV GCW moderately correlates with invasively measured SV and SV index (r = 0.63, P = 0.002 and r = 0.59, P = 0.004) in 22 HF patients with reduced ejection fraction (EF).[106] A follow-up study on 51 pulmonary HTN patients and 21 patients without structural heart disease demonstrated that RV GCW (HR: 1.42 per 100 mmHg% <900 mmHg%, 95% CI: 1.12–1.81, P = 0.004) and RV GWI (HR: 1.46 per 100 mmHg% <650 mmHg%, 95% CI: 1.09–1.94, P = 0.010) correlate with all-cause mortality, unlike conventional RV function parameters.[107]
In the context of atrial septal defects (ASDs), Wu et al. analyzed 29 patients with ASD, comparing them to an equal number of age- and sex-matched controls without cardiovascular diseases.[108] The ASD patients underwent right heart catheterization (RHC).[108] The authors demonstrated that the RV GWI, RV GCW, and RV GWW were significantly higher in ASD patients than in controls, while no significant difference was found in RV GWE. RV GWI, RV GCW, and RV GWW showed significant correlations with RHC-derived SV and SV index, outperforming RV GLS as predictors of ASD.[108]
These studies utilized software initially designed for LV MW, emphasizing the need for dedicated RV MW software and validation.
ROLE OF MYOCARDIAL WORK ANALYSIS IN CARDIO-ONCOLOGY
The field of oncology is continuously expanding, with new therapeutic agents being developed on a daily basis, leading to the complexity of cardio-oncology as a specialty. The GLS has proven more effective than LVEF in identifying and managing cancer patients with cancer therapeutics-related cardiac dysfunction (CTRCD). Consequently, it has been included in the recent European Society of Cardiology guidelines on cardio-oncology.[109] It is standard practice to regularly assess both GLS and LVEF; however, the reliability of GLS can be affected by changes in systolic blood pressure, making serial comparisons challenging. A recent review on subclinical cardiotoxicity and the potential role of MW in these cases suggested that MW could offer promising insights for cancer patients at a higher risk of cardiac issues.[110] While studies on MW in breast cancer patients have yielded mixed results, a study conducted by Guan et al.[111] compared MW and GLS in breast cancer patients undergoing different treatments. The study found that while GLS was still a sensitive indicator of cardiac dysfunction, changes in MW indices were observed after the sixth cycle of treatment.[111] Additionally, Calvillo-Argüelles et al. studied 136 HER2 + breast cancer patients undergoing anthracycline and trastuzumab therapy and determined that MW indices did not provide any meaningful advantages over GLS and established clinical risk factors in predicting CTRCD at subsequent appointments.
Li et al.[113] conducted a study on HER2 + breast cancer patients undergoing pertuzumab and trastuzumab therapy. Following four cycles of treatment, GLS, GWI, GCW, and GWE were significantly reduced, detecting toxicity more promptly and sensitively than LVEF. However, it remains uncertain if the changes in myocardial parameters correspond to occurrences of myocardial toxic events.[113]
Kosmala et al.[114] explored the ability of MW indices to differentiate changes in GLS specifically caused by chemotherapy versus changes in afterload. Patients were categorized into four groups based on their CTRCD status and blood pressure levels. The findings revealed that patients who were positive for CTRCD but did not experience a rise in blood pressure exhibited considerably larger decreases in GWI and GCW. The researchers noted that the decline in GLS alongside an increase in GWI and GCW indicated the impact of heightened afterload on LV performance in the absence of actual myocardial dysfunction.[114] Vaz Ferreira et al.[115] analyzed MW before and after treatment in breast cancer patients receiving anthracycline and/or anti-HER therapy. All MW indices showed impairment at 4–6 months posttreatment, with a tendency to return to baseline levels at 12–14 months. Patients with CTRCD exhibited a notable decrease in GWI and GWE at 4–6 months compared to those without CTRCD.
The feasibility of MW indices compared to traditional echocardiographic parameters was also explored in a population of children and young adults (598 patients) undergoing anthracycline therapy.[116] Zhan et al. observed that MW indices offered an earlier and more sensitive indicator of progression toward CTRCD than conventional measures such as LVEF and GLS.[116] Further research is necessary to ascertain the clinical significance of MW in cardio-oncology and CTRCD and its added value compared to GLS alone. Additionally, it remains unclear whether a MW-oriented approach can decrease the long-term risk of HF and enhance clinical outcomes. In Table 1, the main clinical applications of Myocardial Work Analysis are summarized.[112]
Table 1:
Current applications of myocardial work in clinical practice
| Cardiac disease | MW parameters |
|---|---|
| CAD | Regional MW is superior to LVEF and GLS for detecting acute coronary occlusion in NSTEMI patients[39] Regional MW decreases during stress echocardiography in areas supplied by stenosed arteries, along with decreased GWE[117] A GWI value of ≤1810 mmHg% has a 95% positive predictive value for detecting significant CAD[38] |
| Hypertension | GWI and GCW increase with higher afterload (HTN), while GLS and LVEF remain unchanged[28,29] GWI increases progressively with each stage of HTN[118] |
| Dyssynchrony | Patients with LBBB exhibit significantly higher WW, which improves with CRT[119] WW in the septum combined with the wall motion score index strongly predicts CRT response[51] |
| Amyloidosis | GWI has been found to be superior to GLS in predicting major adverse cardiac events and all-cause mortality[81] |
| HCM | Patients with HCM have significantly reduced GCW[120] A GCW greater than 1730 mmHg% is linked to better event-free survival[67] |
| Valvular heart disease | In AS patients, GWI, GCW, and GWW are increased than normal reference values. Reduced GWI and GCW at follow-up are linked to higher mortality[95] Reduced GWI, GCW, and GWW are associated with worse long-term survival in patients with severe functional mitral regurgitation. GCW improves after transcatheter edge-to-edge repair, while GLS and LVEF do not[102] GWI and GCW positively correlate with the severity of AR in patients with normal LVEF. A GWI threshold of 1285 mmHg% predicts LV reverse remodeling[96] |
| Cradioncology | In a small subset of patients with a systolic blood pressure reduction >21 mmHg and a GLS change of <3.3%, MW indices are more strongly associated with concurrent CTRCD than GLS[112] |
WW=Wasted work, CRT=Cardiac resynchronization therapy, CTRCD=Cancer therapeutics-related cardiac dysfunction, GCW=Global constructive work, GLS=Global longitudinal strain, GWE=Global work efficiency, GWI=Global work index, GWW=Global WW, HTN=Hypertension, LBBB=Left bundle branch block, LV=Left ventricular, LVEF=LV ejection fraction, MRI=Magnetic resonance imaging, NSTEMI=Non-ST-segment-elevation myocardial infarction, MW=Myocardial work, AS=Aortic stenosis, HCM=Hypertrophic cardiomyopathy, CAD=Coronary artery disease, AR=Aortic regurgitation
CONCLUSIONS
Validation studies have proven that the noninvasive assessment of MW indices using LV PSL is highly correlated with invasive measurements of stroke work and cardiac metabolism. This strong correlation has led to the widespread utilization of MW measurement in a variety of clinical settings.
The key benefit of this advanced diagnostic tool is its ability to combine strain measurement with pressure, resulting in a more objective assessment of ventricular function that considers loading conditions and overcomes the limitations of EF and GLS. As a result, MW evaluation is particularly valuable in determining whether reduced contraction is due to increased afterload, as seen in arterial HTN, or decreased contractility, as seen in ischemic cardiomyopathy.
Furthermore, MW indices provide comprehensive insights into both segmental and global myocardial energetics, opening up new possibilities for studying cardiomyopathies and improving the prediction of therapeutic responses. Increasing evidence supports the feasibility and reliability of MW measurements in various clinical applications.
However, to fully establish the added value of this technique, well-designed multicenter studies involving larger and diverse populations are necessary. Such studies would confirm the significance of MW indices and potentially integrate them into routine echocardiographic assessments.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Suga H. Total mechanical energy of a ventricle model and cardiac oxygen consumption. Am J Physiol. 1979;236:H498–505. doi: 10.1152/ajpheart.1979.236.3.H498. [DOI] [PubMed] [Google Scholar]
- 2.Suga H, Hayashi T, Shirahata M. Ventricular systolic pressure-volume area as predictor of cardiac oxygen consumption. Am J Physiol. 1981;240:H39–44. doi: 10.1152/ajpheart.1981.240.1.H39. [DOI] [PubMed] [Google Scholar]
- 3.Marwick TH. Ejection fraction pros and cons: JACC state-of-the-art review. J Am Coll Cardiol. 2018;72:2360–79. doi: 10.1016/j.jacc.2018.08.2162. [DOI] [PubMed] [Google Scholar]
- 4.Zito C, Longobardo L, Citro R, Galderisi M, Oreto L, Carerj ML, et al. Ten years of 2D longitudinal strain for early myocardial dysfunction detection: A clinical overview. Biomed Res Int. 2018;2018:8979407.. doi: 10.1155/2018/8979407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cameli M, Mandoli GE, Sciaccaluga C, Mondillo S. More than 10 years of speckle tracking echocardiography: Still a novel technique or a definite tool for clinical practice? Echocardiography. 2019;36:958–70. doi: 10.1111/echo.14339. [DOI] [PubMed] [Google Scholar]
- 6.Zito C, Manganaro R, Cusmà Piccione M, Madonna R, Monte I, Novo G, et al. Anthracyclines and regional myocardial damage in breast cancer patients. A multicentre study from the Working Group on Drug Cardiotoxicity and Cardioprotection, Italian Society of Cardiology (SIC) Eur Heart J Cardiovasc Imaging. 2021;22:406–15. doi: 10.1093/ehjci/jeaa339. [DOI] [PubMed] [Google Scholar]
- 7.Fava AM, Meredith D, Desai MY. Clinical applications of echo strain imaging: A current appraisal. Curr Treat Options Cardiovasc Med. 2019;21:50.. doi: 10.1007/s11936-019-0761-0. [DOI] [PubMed] [Google Scholar]
- 8.Di Bella G, Minutoli F, Piaggi P, Casale M, Mazzeo A, Zito C, et al. Usefulness of combining electrocardiographic and echocardiographic findings and brain natriuretic peptide in early detection of cardiac amyloidosis in subjects with transthyretin gene mutation. Am J Cardiol. 2015;116:1122–7. doi: 10.1016/j.amjcard.2015.07.008. [DOI] [PubMed] [Google Scholar]
- 9.Yingchoncharoen T, Agarwal S, Popović ZB, Marwick TH. Normal ranges of left ventricular strain: A meta-analysis. J Am Soc Echocardiogr. 2013;26:185–91. doi: 10.1016/j.echo.2012.10.008. [DOI] [PubMed] [Google Scholar]
- 10.Boe E, Skulstad H, Smiseth OA. Myocardial work by echocardiography: A novel method ready for clinical testing. Eur Heart J Cardiovasc Imaging. 2019;20:18–20. doi: 10.1093/ehjci/jey156. [DOI] [PubMed] [Google Scholar]
- 11.Manganaro R, Marchetta S, Dulgheru R, Sugimoto T, Tsugu T, Ilardi F, et al. Correlation between non-invasive myocardial work indices and main parameters of systolic and diastolic function: Results from the EACVI NORRE study. Eur Heart J Cardiovasc Imaging. 2020;21:533–41. doi: 10.1093/ehjci/jez203. [DOI] [PubMed] [Google Scholar]
- 12.Roemer S, Jaglan A, Santos D, Umland M, Jain R, Tajik AJ, et al. The Utility of Myocardial Work in Clinical Practice. J Am Soc Echocardiogr. 2021;34:807–18. doi: 10.1016/j.echo.2021.04.013. [DOI] [PubMed] [Google Scholar]
- 13.Kuhtz-Buschbeck JP, Drake-Holland A, Noble MI, Lohff B, Schaefer J. Rediscovery of Otto Frank’s contribution to science. J Mol Cell Cardiol. 2018;119:96–103. doi: 10.1016/j.yjmcc.2018.04.017. [DOI] [PubMed] [Google Scholar]
- 14.Bastos MB, Burkhoff D, Maly J, Daemen J, den Uil CA, Ameloot K, et al. Invasive left ventricle pressure-volume analysis: Overview and practical clinical implications. Eur Heart J. 2020;41:1286–97. doi: 10.1093/eurheartj/ehz552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dini FL, Buralli S, Gallina S, Galderisi M, Mele D, Mondillo S, et al. Principles of cardiovascular dynamics in the perspective of echocardiography. G Ital Cardiol (Rome) 2008;9:536–44. [PubMed] [Google Scholar]
- 16.Takaoka H, Takeuchi M, Odake M, Yokoyama M. Assessment of myocardial oxygen consumption (Vo2) and systolic pressure-volume area (PVA) in human hearts. Eur Heart J. 1992;13(Suppl E):85–90. doi: 10.1093/eurheartj/13.suppl_e.85. [DOI] [PubMed] [Google Scholar]
- 17.Hisano R, Cooper G., 4th Correlation of force-length area with oxygen consumption in ferret papillary muscle. Circ Res. 1987;61:318–28. doi: 10.1161/01.res.61.3.318. [DOI] [PubMed] [Google Scholar]
- 18.Russell K, Eriksen M, Aaberge L, Wilhelmsen N, Skulstad H, Remme EW, et al. A novel clinical method for quantification of regional left ventricular pressure-strain loop area: A non-invasive index of myocardial work. Eur Heart J. 2012;33:724–33. doi: 10.1093/eurheartj/ehs016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hubert A, Le Rolle V, Leclercq C, Galli E, Samset E, Casset C, et al. Estimation of myocardial work from pressure-strain loops analysis: An experimental evaluation. Eur Heart J Cardiovasc Imaging. 2018;19:1372–9. doi: 10.1093/ehjci/jey024. [DOI] [PubMed] [Google Scholar]
- 20.Marzlin N, Hays AG, Peters M, Kaminski A, Roemer S, O’Leary P, et al. Myocardial work in echocardiography. Circ Cardiovasc Imaging. 2023;16:e014419.. doi: 10.1161/CIRCIMAGING.122.014419. [DOI] [PubMed] [Google Scholar]
- 21.Moya A, Buytaert D, Penicka M, Bartunek J, Vanderheyden M. State-of-the-Art: Noninvasive assessment of left ventricular function through myocardial work. J Am Soc Echocardiogr. 2023;36:1027–42. doi: 10.1016/j.echo.2023.07.002. [DOI] [PubMed] [Google Scholar]
- 22.Manganaro R, Marchetta S, Dulgheru R, Ilardi F, Sugimoto T, Robinet S, et al. Echocardiographic reference ranges for normal non-invasive myocardial work indices: Results from the EACVI NORRE study. Eur Heart J Cardiovasc Imaging. 2019;20:582–90. doi: 10.1093/ehjci/jey188. [DOI] [PubMed] [Google Scholar]
- 23.Olsen FJ, Skaarup KG, Lassen MC, Johansen ND, Sengeløv M, Jensen GB, et al. Normal values for myocardial work indices derived from pressure-strain loop analyses: From the CCHS. Circ Cardiovasc Imaging. 2022;15:e013712.. doi: 10.1161/CIRCIMAGING.121.013712. [DOI] [PubMed] [Google Scholar]
- 24.Sahiti F, Morbach C, Cejka V, Albert J, Eichner FA, Gelbrich G, et al. Left ventricular remodeling and myocardial work: Results from the population-based STAAB cohort study. Front Cardiovasc Med. 2021;8:669335.. doi: 10.3389/fcvm.2021.669335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lembo M, Santoro C, Casciano O, Capone V, Fedele T, Luciano F, et al. Impact of diastolic blood pressure on speckle tracking derived myocardial work components in a population of normotensive and untreated hypertensive patients. Eur Heart J. 2020;41(Suppl 2):ehaa946.2700. [Google Scholar]
- 26.Skaarup KG, Lassen MC, Marott JL, Biering-Sørensen SR, Jørgensen PG, Appleyard M, et al. The impact of cardiovascular risk factors on global longitudinal strain over a decade in the general population: The Copenhagen city heart study. Int J Cardiovasc Imaging. 2020;36:1907–16. doi: 10.1007/s10554-020-01906-5. [DOI] [PubMed] [Google Scholar]
- 27.Tadic M, Sala C, Carugo S, Mancia G, Grassi G, Cuspidi C. Myocardial strain in hypertension: A meta-analysis of two-dimensional speckle tracking echocardiographic studies. J Hypertens. 2021;39:2103–12. doi: 10.1097/HJH.0000000000002898. [DOI] [PubMed] [Google Scholar]
- 28.Chan J, Edwards NF, Khandheria BK, Shiino K, Sabapathy S, Anderson B, et al. A new approach to assess myocardial work by non-invasive left ventricular pressure-strain relations in hypertension and dilated cardiomyopathy. Eur Heart J Cardiovasc Imaging. 2019;20:31–9. doi: 10.1093/ehjci/jey131. [DOI] [PubMed] [Google Scholar]
- 29.Tadic M, Cuspidi C, Pencic B, Grassi G, Celic V. Myocardial work in hypertensive patients with and without diabetes: An echocardiographic study. J Clin Hypertens (Greenwich) 2020;22:2121–7. doi: 10.1111/jch.14053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Loncaric F, Marciniak M, Nunno L, Mimbrero M, Fernandes JF, Fabijanovic D, et al. Distribution of myocardial work in arterial hypertension: Insights from non-invasive left ventricular pressure-strain relations. Int J Cardiovasc Imaging. 2021;37:145–54. doi: 10.1007/s10554-020-01969-4. [DOI] [PubMed] [Google Scholar]
- 31.Huang H, Fu L, Ruan Q, You Z, Yan L. Segmental and global myocardial work in hypertensive patients with different left ventricular geometry. Cardiovasc Ultrasound. 2023;21:11.. doi: 10.1186/s12947-023-00310-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kohsaka S, Ejiri K, Takagi H, Watanabe I, Gatate Y, Fukushima K, et al. Diagnostic and therapeutic strategies for stable coronary artery disease following the ISCHEMIA trial. JACC Asia. 2023;3:15–30. doi: 10.1016/j.jacasi.2022.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Trimarchi G, Teresi L, Licordari R, Pingitore A, Pizzino F, Grimaldi P, et al. Transient left ventricular dysfunction from cardiomyopathies to myocardial viability: When and why cardiac function recovers. Biomedicines. 2024;12:1051.. doi: 10.3390/biomedicines12051051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Choi JO, Kim EY, Lee GY, Lee SC, Park SW, Kim DK, et al. Predictors of left ventricular reverse remodeling and subsequent outcome in nonischemic dilated cardiomyopathy. Circ J. 2013;77:462–9. doi: 10.1253/circj.cj-12-0507. [DOI] [PubMed] [Google Scholar]
- 35.Licordari R, Casale M, Correale M, Imbalzano E, Crea P, Santo Signorelli S, et al. Prognostic value of two-dimensional strain in early ischemic heart disease: A 5-year follow-up study. Echocardiography. 2022;39:768–75. doi: 10.1111/echo.15360. [DOI] [PubMed] [Google Scholar]
- 36.Lu S, Hu X, Zhang J, Zhu Y, Zhou W, Liu Y, et al. Post-systolic shortening is superior to global longitudinal strain in predicting adverse events in patients with stable coronary artery disease and preserved systolic function. Insights Imaging. 2022;13:35.. doi: 10.1186/s13244-022-01174-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Frișan AC, Mornoș C, Lazăr MA, Șoșdean R, Crișan S, Ionac I, et al. Echocardiographic myocardial work: A novel method to assess left ventricular function in patients with coronary artery disease and diabetes mellitus. Medicina (Kaunas) 2024;60:199.. doi: 10.3390/medicina60020199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Edwards NF, Scalia GM, Shiino K, Sabapathy S, Anderson B, Chamberlain R, et al. Global myocardial work is superior to global longitudinal strain to predict significant coronary artery disease in patients with normal left ventricular function and wall motion. J Am Soc Echocardiogr. 2019;32:947–57. doi: 10.1016/j.echo.2019.02.014. [DOI] [PubMed] [Google Scholar]
- 39.Boe E, Russell K, Eek C, Eriksen M, Remme EW, Smiseth OA, et al. Non-invasive myocardial work index identifies acute coronary occlusion in patients with non-ST-segment elevation-acute coronary syndrome. Eur Heart J Cardiovasc Imaging. 2015;16:1247–55. doi: 10.1093/ehjci/jev078. [DOI] [PubMed] [Google Scholar]
- 40.Gheorghiu A, Arnautu SF, Slovenski M, Malița CD, Tomescu MC, Arnautu DA. Myocardial work evaluation-a useful non-invasive method to predict coronary artery sub-occlusion in a patient with unstable angina and multiple myocardial revascularization interventions. Diagnostics (Basel) 2023;13:1459.. doi: 10.3390/diagnostics13081459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Meimoun P, Abdani S, Stracchi V, Elmkies F, Boulanger J, Botoro T, et al. Usefulness of noninvasive myocardial work to predict left ventricular recovery and acute complications after acute anterior myocardial infarction treated by percutaneous coronary intervention. J Am Soc Echocardiogr. 2020;33:1180–90. doi: 10.1016/j.echo.2020.07.008. [DOI] [PubMed] [Google Scholar]
- 42.Lustosa RP, van der Bijl P, El Mahdiui M, Montero-Cabezas JM, Kostyukevich MV, Ajmone Marsan N, et al. Noninvasive myocardial work indices 3 months after ST-segment elevation myocardial infarction: Prevalence and characteristics of patients with postinfarction cardiac remodeling. J Am Soc Echocardiogr. 2020;33:1172–9. doi: 10.1016/j.echo.2020.05.001. [DOI] [PubMed] [Google Scholar]
- 43.El Mahdiui M, van der Bijl P, Abou R, Ajmone Marsan N, Delgado V, Bax JJ. Global left ventricular myocardial work efficiency in healthy individuals and patients with cardiovascular disease. J Am Soc Echocardiogr. 2019;32:1120–7. doi: 10.1016/j.echo.2019.05.002. [DOI] [PubMed] [Google Scholar]
- 44.Halabi A, Wright L, Marwick TH. Measurement of global myocardial work with exercise testing. J Am Soc Echocardiogr. 2020;33:631–2. doi: 10.1016/j.echo.2020.01.021. [DOI] [PubMed] [Google Scholar]
- 45.Lin J, Wu W, Gao L, He J, Zhu Z, Pang K, et al. Global myocardial work combined with treadmill exercise stress to detect significant coronary artery disease. J Am Soc Echocardiogr. 2022;35:247–57. doi: 10.1016/j.echo.2021.10.009. [DOI] [PubMed] [Google Scholar]
- 46.Brignole M, Auricchio A, Baron-Esquivias G, Bordachar P, Boriani G, Breithardt OA, et al. 2013 ESC guidelines on cardiac pacing and cardiac resynchronization therapy: The task force on cardiac pacing and resynchronization therapy of the European Society of Cardiology (ESC). Developed in collaboration with the European Heart Rhythm Association (EHRA) Eur Heart J. 2013;34:2281–329. doi: 10.1093/eurheartj/eht150. [DOI] [PubMed] [Google Scholar]
- 47.Tedeschi A, Palazzini M, Trimarchi G, Conti N, Di Spigno F, Gentile P, et al. Heart failure management through telehealth: Expanding care and connecting hearts. J Clin Med. 2024;13:2592.. doi: 10.3390/jcm13092592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lim P, Donal E, Lafitte S, Derumeaux G, Habib G, Réant P, et al. Multicentre study using strain delay index for predicting response to cardiac resynchronization therapy (MUSIC study) Eur J Heart Fail. 2011;13:984–91. doi: 10.1093/eurjhf/hfr073. [DOI] [PubMed] [Google Scholar]
- 49.Delgado-Montero A, Tayal B, Goda A, Ryo K, Marek JJ, Sugahara M, et al. Additive prognostic value of echocardiographic global longitudinal and global circumferential strain to electrocardiographic criteria in patients with heart failure undergoing cardiac resynchronization therapy. Circ Cardiovasc Imaging. 2016;9:e004241.. doi: 10.1161/CIRCIMAGING.115.004241. [DOI] [PubMed] [Google Scholar]
- 50.Iwano H, Yamada S, Watanabe M, Mitsuyama H, Mizukami K, Nishino H, et al. Strain rate dispersion index can predict changes in left ventricular volume and adverse cardiac events following cardiac resynchronization therapy. Circ J. 2013;77:2757–65. doi: 10.1253/circj.cj-13-0483. [DOI] [PubMed] [Google Scholar]
- 51.Vecera J, Penicka M, Eriksen M, Russell K, Bartunek J, Vanderheyden M, et al. Wasted septal work in left ventricular dyssynchrony: A novel principle to predict response to cardiac resynchronization therapy. Eur Heart J Cardiovasc Imaging. 2016;17:624–32. doi: 10.1093/ehjci/jew019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Aalen JM, Donal E, Larsen CK, Duchenne J, Lederlin M, Cvijic M, et al. Imaging predictors of response to cardiac resynchronization therapy: Left ventricular work asymmetry by echocardiography and septal viability by cardiac magnetic resonance. Eur Heart J. 2020;41:3813–23. doi: 10.1093/eurheartj/ehaa603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Duchenne J, Aalen JM, Cvijic M, Larsen CK, Galli E, Bézy S, et al. Acute redistribution of regional left ventricular work by cardiac resynchronization therapy determines long-term remodelling. Eur Heart J Cardiovasc Imaging. 2020;21:619–28. doi: 10.1093/ehjci/jeaa003. [DOI] [PubMed] [Google Scholar]
- 54.Galli E, Leclercq C, Hubert A, Bernard A, Smiseth OA, Mabo P, et al. Role of myocardial constructive work in the identification of responders to CRT. Eur Heart J Cardiovasc Imaging. 2018;19:1010–8. doi: 10.1093/ehjci/jex191. [DOI] [PubMed] [Google Scholar]
- 55.Bouali Y, Donal E, Gallard A, Laurin C, Hubert A, Bidaut A, et al. Prognostic usefulness of myocardial work in patients with heart failure and reduced ejection fraction treated by sacubitril/valsartan. Am J Cardiol. 2020;125:1856–62. doi: 10.1016/j.amjcard.2020.03.031. [DOI] [PubMed] [Google Scholar]
- 56.Przewlocka-Kosmala M, Marwick TH, Mysiak A, Kosowski W, Kosmala W. Usefulness of myocardial work measurement in the assessment of left ventricular systolic reserve response to spironolactone in heart failure with preserved ejection fraction. Eur Heart J Cardiovasc Imaging. 2019;20:1138–46. doi: 10.1093/ehjci/jez027. [DOI] [PubMed] [Google Scholar]
- 57.King G, Wood MJ. The heart of the endurance athlete assessed by echocardiography and its modalities: “Embracing the delicate balance”. Curr Cardiol Rep. 2013;15:383.. doi: 10.1007/s11886-013-0383-1. [DOI] [PubMed] [Google Scholar]
- 58.Licordari R, Trimarchi G, Teresi L, Restelli D, Lofrumento F, Perna A, et al. Cardiac magnetic resonance in HCM phenocopies: From diagnosis to risk stratification and therapeutic management. J Clin Med. 2023;12:3481.. doi: 10.3390/jcm12103481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Monte IP, Faro DC, Trimarchi G, de Gaetano F, Campisi M, Losi V, et al. Left atrial strain imaging by speckle tracking echocardiography: The supportive diagnostic value in cardiac amyloidosis and hypertrophic cardiomyopathy. J Cardiovasc Dev Dis. 2023;10:261.. doi: 10.3390/jcdd10060261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Saghir M, Areces M, Makan M. Strain rate imaging differentiates hypertensive cardiac hypertrophy from physiologic cardiac hypertrophy (athlete’s heart) J Am Soc Echocardiogr. 2007;20:151–7. doi: 10.1016/j.echo.2006.08.006. [DOI] [PubMed] [Google Scholar]
- 61.Galderisi M, Cardim N, D’Andrea A, Bruder O, Cosyns B, Davin L, et al. The multi-modality cardiac imaging approach to the Athlete’s heart: An expert consensus of the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2015;16:353.. doi: 10.1093/ehjci/jeu323. [DOI] [PubMed] [Google Scholar]
- 62.Galderisi M, Lomoriello VS, Santoro A, Esposito R, Olibet M, Raia R, et al. Differences of myocardial systolic deformation and correlates of diastolic function in competitive rowers and young hypertensives: A speckle-tracking echocardiography study. J Am Soc Echocardiogr. 2010;23:1190–8. doi: 10.1016/j.echo.2010.07.010. [DOI] [PubMed] [Google Scholar]
- 63.Di Bella G, Minutoli F, Pingitore A, Zito C, Mazzeo A, Aquaro GD, et al. Endocardial and epicardial deformations in cardiac amyloidosis and hypertrophic cardiomyopathy. Circ J. 2011;75:1200–8. doi: 10.1253/circj.cj-10-0844. [DOI] [PubMed] [Google Scholar]
- 64.Richand V, Lafitte S, Reant P, Serri K, Lafitte M, Brette S, et al. An ultrasound speckle tracking (two-dimensional strain) analysis of myocardial deformation in professional soccer players compared with healthy subjects and hypertrophic cardiomyopathy. Am J Cardiol. 2007;100:128–32. doi: 10.1016/j.amjcard.2007.02.063. [DOI] [PubMed] [Google Scholar]
- 65.de Gregorio C, Di Bella G. Merging strain-echo measurements with late gadolinium enhancement at cardiac MRI: An upcoming chance for advanced functional assessment of fibrosis in hypertrophic cardiomyopathy? Int J Cardiol. 2016;203:632–4. doi: 10.1016/j.ijcard.2015.10.245. [DOI] [PubMed] [Google Scholar]
- 66.Gonçalves AV, Rosa SA, Branco L, Galrinho A, Fiarresga A, Lopes LR, et al. Myocardial work is associated with significant left ventricular myocardial fibrosis in patients with hypertrophic cardiomyopathy. Int J Cardiovasc Imaging. 2021;37:2237–44. doi: 10.1007/s10554-021-02186-3. [DOI] [PubMed] [Google Scholar]
- 67.Hiemstra YL, van der Bijl P, El Mahdiui M, Bax JJ, Delgado V, Marsan NA. Myocardial work in nonobstructive hypertrophic cardiomyopathy: Implications for outcome. J Am Soc Echocardiogr. 2020;33:1201–8. doi: 10.1016/j.echo.2020.05.010. [DOI] [PubMed] [Google Scholar]
- 68.de Gregorio C, Trimarchi G, Faro DC, De Gaetano F, Campisi M, Losi V, et al. Myocardial work appraisal in transthyretin cardiac amyloidosis and nonobstructive hypertrophic cardiomyopathy. Am J Cardiol. 2023;208:173–9. doi: 10.1016/j.amjcard.2023.09.055. [DOI] [PubMed] [Google Scholar]
- 69.D’Andrea A, Radmilovic J, Carbone A, Mandoli GE, Santoro C, Evola V, et al. Speckle tracking evaluation in endurance athletes: The “optimal” myocardial work. Int J Cardiovasc Imaging. 2020;36:1679–88. doi: 10.1007/s10554-020-01871-z. [DOI] [PubMed] [Google Scholar]
- 70.Tokodi M, Oláh A, Fábián A, Lakatos BK, Hizoh I, Ruppert M, et al. Novel insights into the athlete’s heart: Is myocardial work the new champion of systolic function? Eur Heart J Cardiovasc Imaging. 2022;23:188–97. doi: 10.1093/ehjci/jeab162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Junianto I, Soesanto AM, Radi B, Yonas E, Kuncoro AS, Atmadikoesoemah CA, et al. The influence of special military training on left ventricular adaptation to exercise in elite air force soldiers. J Cardiovasc Echogr. 2023;33:69–75. doi: 10.4103/jcecho.jcecho_67_22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.D’Andrea A, Carbone A, Radmilovic J, Russo V, Fabiani D, Maio MD, et al. Myocardial work efficiency in physiologic left ventricular hypertrophy of power athletes. J Cardiovasc Echogr. 2022;32:154–9. doi: 10.4103/jcecho.jcecho_11_22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Borzì DD, Saladino S, Losi V, Faro DC, Monte IP. Strain and myocardial work index during echo exercise to evaluate myocardial function in athletes. J Cardiovasc Echogr. 2022;32:82–8. doi: 10.4103/jcecho.jcecho_1_22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Di Bella G, Cappelli F, Licordari R, Piaggi P, Campisi M, Bellavia D, et al. Prevalence and diagnostic value of extra-left ventricle echocardiographic findings in transthyretin-related cardiac amyloidosis. Amyloid. 2022;29:197–204. doi: 10.1080/13506129.2022.2064739. [DOI] [PubMed] [Google Scholar]
- 75.Di Nunzio D, Recupero A, de Gregorio C, Zito C, Carerj S, Di Bella G. Echocardiographic findings in cardiac amyloidosis: Inside two-dimensional, doppler, and strain imaging. Curr Cardiol Rep. 2019;21:7.. doi: 10.1007/s11886-019-1094-z. [DOI] [PubMed] [Google Scholar]
- 76.de Gregorio C, Dattilo G, Casale M, Terrizzi A, Donato R, Di Bella G. Left atrial morphology, size and function in patients with transthyretin cardiac amyloidosis and primary hypertrophic cardiomyopathy – Comparative strain imaging study. Circ J. 2016;80:1830–7. doi: 10.1253/circj.CJ-16-0364. [DOI] [PubMed] [Google Scholar]
- 77.Clemmensen TS, Eiskjær H, Mikkelsen F, Granstam SO, Flachskampf FA, Sørensen J, et al. Left ventricular pressure-strain-derived myocardial work at rest and during exercise in patients with cardiac amyloidosis. J Am Soc Echocardiogr. 2020;33:573–82. doi: 10.1016/j.echo.2019.11.018. [DOI] [PubMed] [Google Scholar]
- 78.Palmiero G, Rubino M, Monda E, Caiazza M, D’Urso L, Carlomagno G, et al. Global left ventricular myocardial work efficiency in heart failure patients with cardiac amyloidosis: Pathophysiological implications and role in differential diagnosis. J Cardiovasc Echogr. 2021;31:157–64. doi: 10.4103/jcecho.jcecho_16_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Henein MY, Lindqvist P. Myocardial work does not have additional diagnostic value in the assessment of ATTR cardiac amyloidosis. J Clin Med. 2021;10:4555.. doi: 10.3390/jcm10194555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Roger-Rollé A, Cariou E, Rguez K, Fournier P, Lavie-Badie Y, Blanchard V, et al. Can myocardial work indices contribute to the exploration of patients with cardiac amyloidosis? Open Heart. 2020;7:e001346.. doi: 10.1136/openhrt-2020-001346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Clemmensen TS, Eiskjær H, Ladefoged B, Mikkelsen F, Sørensen J, Granstam SO, et al. Prognostic implications of left ventricular myocardial work indices in cardiac amyloidosis. Eur Heart J Cardiovasc Imaging. 2021;22:695–704. doi: 10.1093/ehjci/jeaa097. [DOI] [PubMed] [Google Scholar]
- 82.de Gregorio C, Trimarchi G, Faro DC, Poleggi C, Teresi L, De Gaetano F, et al. Systemic vascular resistance and myocardial work analysis in hypertrophic cardiomyopathy and transthyretin cardiac amyloidosis with preserved left ventricular ejection fraction. J Clin Med. 2024;13:1671.. doi: 10.3390/jcm13061671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Naderi N. Hemodynamic study. In: Maleki M, Alizadehasl A, Haghjoo M, editors. Practical Cardiology. 2nd. Amsterdam, Netherlands: Elsevier; 2022. pp. 201–16. Ch. 11. Available from: https://www.sciencedirect.com/science/article/pii/B9780323809153000132 . [Last accessed on 2024 Jan 24] [Google Scholar]
- 84.Ikonomidis I, Aboyans V, Blacher J, Brodmann M, Brutsaert DL, Chirinos JA, et al. The role of ventricular-arterial coupling in cardiac disease and heart failure: Assessment, clinical implications and therapeutic interventions. A consensus document of the European Society of Cardiology Working Group on Aorta and Peripheral Vascular Diseases, European Association of Cardiovascular Imaging, and Heart Failure Association. Eur J Heart Fail. 2019;21:402–24. doi: 10.1002/ejhf.1436. [DOI] [PubMed] [Google Scholar]
- 85.Cho GY, Marwick TH, Kim HS, Kim MK, Hong KS, Oh DJ. Global 2-dimensional strain as a new prognosticator in patients with heart failure. J Am Coll Cardiol. 2009;54:618–24. doi: 10.1016/j.jacc.2009.04.061. [DOI] [PubMed] [Google Scholar]
- 86.Sengeløv M, Jørgensen PG, Jensen JS, Bruun NE, Olsen FJ, Fritz-Hansen T, et al. Global longitudinal strain is a superior predictor of all-cause mortality in heart failure with reduced ejection fraction. JACC Cardiovasc Imaging. 2015;8:1351–9. doi: 10.1016/j.jcmg.2015.07.013. [DOI] [PubMed] [Google Scholar]
- 87.Raafs AG, Boscutti A, Henkens MT, van den Broek WW, Verdonschot JA, Weerts J, et al. Global longitudinal strain is incremental to left ventricular ejection fraction for the prediction of outcome in optimally treated dilated cardiomyopathy patients. J Am Heart Assoc. 2022;11:e024505.. doi: 10.1161/JAHA.121.024505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Alter P, Rupp H, Rominger MB, Klose KJ, Maisch B. A new methodological approach to assess cardiac work by pressure-volume and stress-length relations in patients with aortic valve stenosis and dilated cardiomyopathy. Pflugers Arch. 2008;455:627–36. doi: 10.1007/s00424-007-0323-2. [DOI] [PubMed] [Google Scholar]
- 89.Cui C, Liu L, Li Y, Liu Y, Huang D, Hu Y, et al. Left ventricular pressure-strain loop-based quantitative examination of the global and regional myocardial work of patients with dilated cardiomyopathy. Ultrasound Med Biol. 2020;46:2834–45. doi: 10.1016/j.ultrasmedbio.2020.06.008. [DOI] [PubMed] [Google Scholar]
- 90.Hedwig F, Nemchyna O, Stein J, Knosalla C, Merke N, Knebel F, et al. Myocardial work assessment for the prediction of prognosis in advanced heart failure. Front Cardiovasc Med. 2021;8:691611.. doi: 10.3389/fcvm.2021.691611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Dahl JS, Magne J, Pellikka PA, Donal E, Marwick TH. Assessment of subclinical left ventricular dysfunction in aortic stenosis. JACC Cardiovasc Imaging. 2019;12:163–71. doi: 10.1016/j.jcmg.2018.08.040. [DOI] [PubMed] [Google Scholar]
- 92.Jain R, Bajwa T, Roemer S, Huisheree H, Allaqaband SQ, Kroboth S, et al. Myocardial work assessment in severe aortic stenosis undergoing transcatheter aortic valve replacement. Eur Heart J Cardiovasc Imaging. 2021;22:715–21. doi: 10.1093/ehjci/jeaa257. [DOI] [PubMed] [Google Scholar]
- 93.Fortuni F, Butcher SC, van der Kley F, Lustosa RP, Karalis I, de Weger A, et al. Left ventricular myocardial work in patients with severe aortic stenosis. J Am Soc Echocardiogr. 2021;34:257–66. doi: 10.1016/j.echo.2020.10.014. [DOI] [PubMed] [Google Scholar]
- 94.De Rosa S, Sabatino J, Strangio A, Leo I, Romano LR, Spaccarotella CA, et al. Non-invasive myocardial work in patients with severe aortic stenosis. J Clin Med. 2022;11:747.. doi: 10.3390/jcm11030747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ilardi F, Postolache A, Dulgheru R, Trung MN, de Marneffe N, Sugimoto T, et al. Prognostic value of non-invasive global myocardial work in asymptomatic aortic stenosis. J Clin Med. 2022;11:1555.. doi: 10.3390/jcm11061555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Meucci MC, Butcher SC, Galloo X, van der Velde ET, Marsan NA, Bax JJ, et al. Noninvasive left ventricular myocardial work in patients with chronic aortic regurgitation and preserved left ventricular ejection fraction. J Am Soc Echocardiogr. 2022;35:703–11.e3. doi: 10.1016/j.echo.2022.01.008. [DOI] [PubMed] [Google Scholar]
- 97.D’Andrea A, Sperlongano S, Formisano T, Tocci G, Cameli M, Tusa M, et al. Stress echocardiography and strain in aortic regurgitation (SESAR protocol): Left ventricular contractile reserve and myocardial work in asymptomatic patients with severe aortic regurgitation. Echocardiography. 2020;37:1213–21. doi: 10.1111/echo.14804. [DOI] [PubMed] [Google Scholar]
- 98.Riccardi M, Cikes M, Adamo M, Pagnesi M, Lombardi CM, Solomon SD, et al. Functional mitral regurgitation and heart failure with preserved ejection fraction: Clinical implications and management. J Card Fail. 2024;30:929–39. doi: 10.1016/j.cardfail.2024.02.024. [DOI] [PubMed] [Google Scholar]
- 99.Verbeke J, Calle S, Kamoen V, De Buyzere M, Timmermans F. Prognostic value of myocardial work and global longitudinal strain in patients with heart failure and functional mitral regurgitation. Int J Cardiovasc Imaging. 2022;38:803–12. doi: 10.1007/s10554-021-02474-y. [DOI] [PubMed] [Google Scholar]
- 100.Yedidya I, Lustosa RP, Fortuni F, van der Bijl P, Namazi F, Vo NM, et al. Prognostic implications of left ventricular myocardial work indices in patients with secondary mitral regurgitation. Circ Cardiovasc Imaging. 2021;14:e012142.. doi: 10.1161/CIRCIMAGING.120.012142. [DOI] [PubMed] [Google Scholar]
- 101.Papadopoulos K, Ikonomidis I, Chrissoheris M, Chalapas A, Kourkoveli P, Parissis J, et al. MitraClip and left ventricular reverse remodelling: A strain imaging study. ESC Heart Fail. 2020;7:1409–18. doi: 10.1002/ehf2.12750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hubert A, Galli E, Leurent G, Corbineau H, Auriane B, Guillaume L, et al. Left ventricular function after correction of mitral regurgitation: Impact of the clipping approach. Echocardiography. 2019;36:2010–8. doi: 10.1111/echo.14523. [DOI] [PubMed] [Google Scholar]
- 103.Yedidya I, Stassen J, Butcher SC, Pio SM, Lustosa RP, van der Bijl P, et al. Relation of Myocardial work indexes and forward flow reserve in patients with significant secondary mitral regurgitation undergoing transcatheter mitral valve repair. Am J Cardiol. 2022;178:106–11. doi: 10.1016/j.amjcard.2022.05.013. [DOI] [PubMed] [Google Scholar]
- 104.Rudiktyo E, Cramer MJ, Yonas E, Teske AJ, Siswanto BB, Doevendans PA, et al. Left ventricle myocardial work correlated with functional capacity in severe rheumatic mitral stenosis with preserved left ventricular ejection fraction. J Cardiovasc Echogr. 2024;34:57–62. doi: 10.4103/jcecho.jcecho_14_24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Landra F, Sciaccaluga C, Pastore MC, Gallone G, Barilli M, Fusi C, et al. Right ventricular myocardial work for the prediction of early right heart failure and long-term mortality after left ventricular assist device implant. Eur Heart J Cardiovasc Imaging. 2023;25:105–15. doi: 10.1093/ehjci/jead193. [DOI] [PubMed] [Google Scholar]
- 106.Butcher SC, Fortuni F, Montero-Cabezas JM, Abou R, El Mahdiui M, van der Bijl P, et al. Right ventricular myocardial work: Proof-of-concept for non-invasive assessment of right ventricular function. Eur Heart J Cardiovasc Imaging. 2021;22:142–52. doi: 10.1093/ehjci/jeaa261. [DOI] [PubMed] [Google Scholar]
- 107.Butcher SC, Feloukidis C, Kamperidis V, Yedidya I, Stassen J, Fortuni F, et al. Right ventricular myocardial work characterization in patients with pulmonary hypertension and relation to invasive hemodynamic parameters and outcomes. Am J Cardiol. 2022;177:151–61. doi: 10.1016/j.amjcard.2022.04.058. [DOI] [PubMed] [Google Scholar]
- 108.Wu J, Huang X, Chen W, Tang Y, Chen X, Wang X, et al. Noninvasive right ventricular work in patients with atrial septal defects: A proof-of-concept study. Cardiovasc Ultrasound. 2023;21:10.. doi: 10.1186/s12947-023-00306-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Lyon AR, López-Fernández T, Couch LS, Asteggiano R, Aznar MC, Bergler-Klein J, et al. 2022 ESC guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS) Eur Heart J. 2022;43:4229–361. doi: 10.1093/eurheartj/ehac244. [DOI] [PubMed] [Google Scholar]
- 110.Di Lisi D, Manno G, Novo G. Subclinical cardiotoxicity: The emerging role of myocardial work and other imaging techniques. Curr Probl Cardiol. 2021;46:100818.. doi: 10.1016/j.cpcardiol.2021.100818. [DOI] [PubMed] [Google Scholar]
- 111.Guan J, Bao W, Xu Y, Yang W, Li M, Xu M, et al. Assessment of myocardial work in cancer therapy-related cardiac dysfunction and analysis of CTRCD prediction by echocardiography. Front Pharmacol. 2021;12:770580.. doi: 10.3389/fphar.2021.770580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Calvillo-Argüelles O, Thampinathan B, Somerset E, Shalmon T, Amir E, Steve Fan CP, et al. Diagnostic and prognostic value of myocardial work indices for identification of cancer therapy-related cardiotoxicity. JACC Cardiovasc Imaging. 2022;15:1361–76. doi: 10.1016/j.jcmg.2022.02.027. [DOI] [PubMed] [Google Scholar]
- 113.Li Y, Hu Z, Wang J, Zhang Y, Chen Z, Zhang H. Endovascular chimney technique for aortic arch pathologies treatment: A systematic review and meta-analysis. Ann Vasc Surg. 2018;47:305–15. doi: 10.1016/j.avsg.2017.09.006. [DOI] [PubMed] [Google Scholar]
- 114.Kosmala W, Negishi T, Thavendiranathan P, Penicka M, De Blois J, Murbræch K, et al. Incremental value of myocardial work over global longitudinal strain in the surveillance for cancer-treatment-related cardiac dysfunction: A case-control study. J Clin Med. 2022;11:912.. doi: 10.3390/jcm11040912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Vaz Ferreira V, Mano TB, Cardoso I, Coutinho Cruz M, Moura Branco L, Almeida-Morais L, et al. Myocardial work brings new insights into left ventricular remodelling in cardio-oncology patients. Int J Environ Res Public Health. 2022;19:2826.. doi: 10.3390/ijerph19052826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Zhan J, Van den Eynde J, Cordrey K, Long R, Danford DA, Hays AG, et al. Deterioration in myocardial work indices precedes changes in global longitudinal strain following anthracycline chemotherapy. Int J Cardiol. 2022;363:171–8. doi: 10.1016/j.ijcard.2022.06.067. [DOI] [PubMed] [Google Scholar]
- 117.Borrie A, Goggin C, Ershad S, Robinson W, Sasse A. Noninvasive myocardial work index: Characterizing the normal and ischemic response to exercise. J Am Soc Echocardiogr. 2020;33:1191–200. doi: 10.1016/j.echo.2020.05.003. [DOI] [PubMed] [Google Scholar]
- 118.Jaglan A, Roemer S, Perez Moreno AC, Khandheria BK. Myocardial work in stage 1 and 2 hypertensive patients. Eur Heart J Cardiovasc Imaging. 2021;22:744–50. doi: 10.1093/ehjci/jeab043. [DOI] [PubMed] [Google Scholar]
- 119.Russell K, Eriksen M, Aaberge L, Wilhelmsen N, Skulstad H, Gjesdal O, et al. Assessment of wasted myocardial work: A novel method to quantify energy loss due to uncoordinated left ventricular contractions. Am J Physiol Heart Circ Physiol. 2013;305:H996–1003. doi: 10.1152/ajpheart.00191.2013. [DOI] [PubMed] [Google Scholar]
- 120.Galli E, Vitel E, Schnell F, Le Rolle V, Hubert A, Lederlin M, et al. Myocardial constructive work is impaired in hypertrophic cardiomyopathy and predicts left ventricular fibrosis. Echocardiography. 2019;36:74–82. doi: 10.1111/echo.14210. [DOI] [PubMed] [Google Scholar]


