Abstract
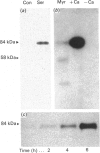
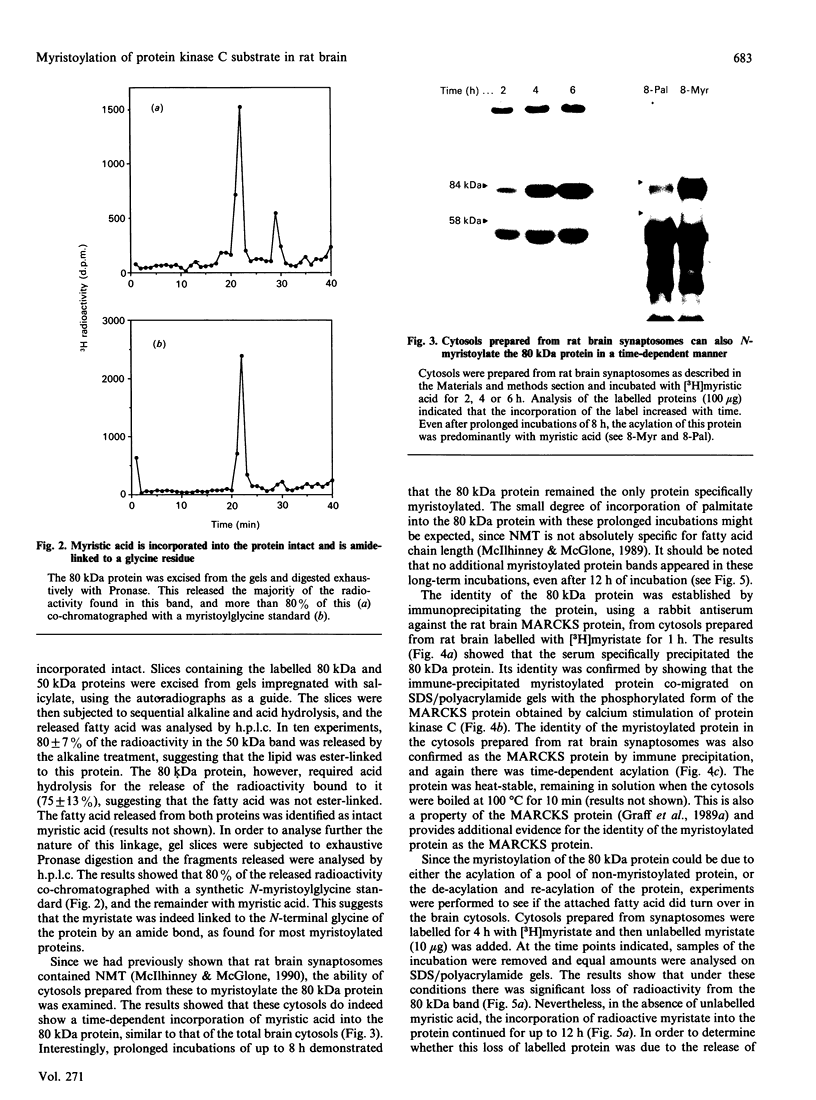
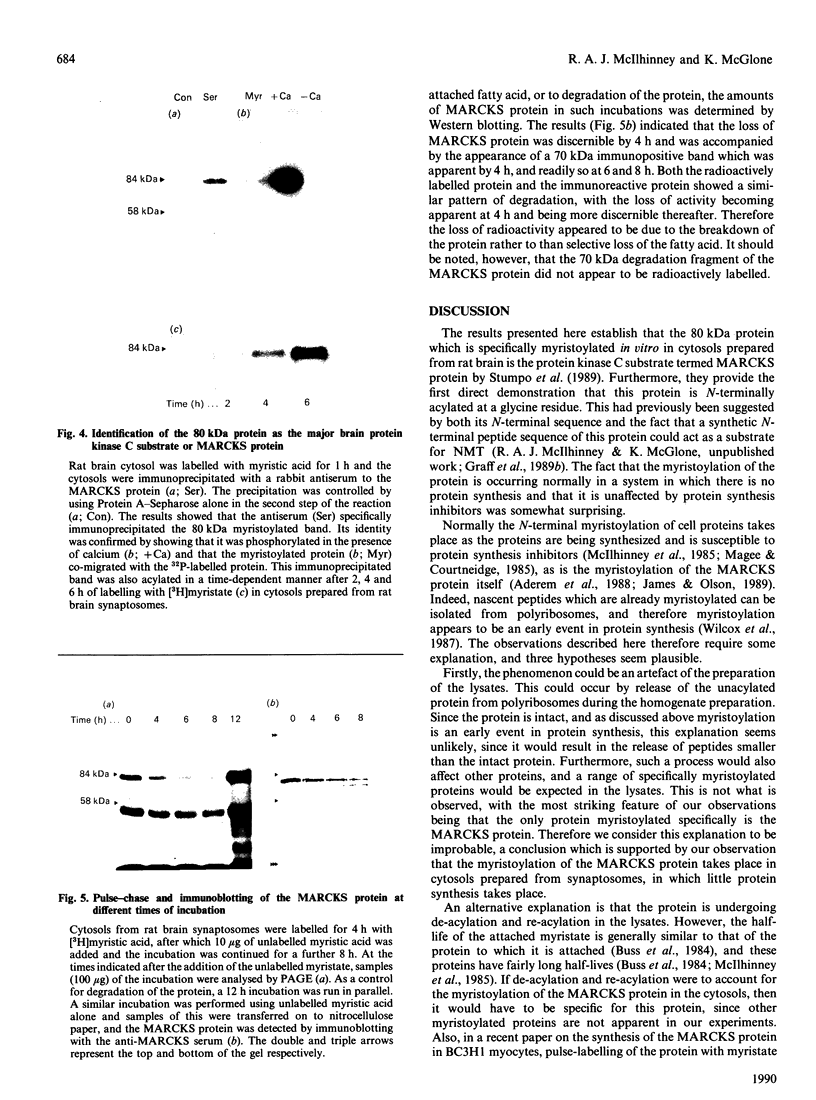
A protein of 80 kDa apparent molecular mass was found to be specifically myristolylated in rat brain cytosols derived from either whole brain or synaptosomes. The attachment of the fatty acid took place in the absence of protein synthesis, since the cytosols did not incorporate [14C]lysine into protein, nor did cycloheximide affect the incorporation of the myristic acid into the protein. The fatty acid was incorporated into the protein via an acid-labile/alkali-resistant band, and Pronase digestion of the labelled protein showed that the lipid was covalently linked to a glycine residue. Together, these data suggested that the myristic acid was amide-linked to the N-terminal residue of the protein. The protein was identified as one of the major protein kinase C substrates, the MARCKS (myristoylated alanine-rich C kinase substrate) protein, by showing that Ca2+ stimulated its phosphorylation, by its heat stability and by immune precipitation (using an antiserum to the MARCKS protein). Incorporation of myristic acid into intact protein continued for up to 12 h, despite the fact that over this period some degradation of the protein could be demonstrated. In pulse-chase experiments, the pattern of loss of the incorporated fatty acid was similar to that of the protein itself, and therefore the loss of radioactivity probably reflects protein degradation rather than specific de-acylation of the protein. Together, these results suggest that there is a pool of unacylated MARCKS protein in the rat brain.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aderem A. A., Albert K. A., Keum M. M., Wang J. K., Greengard P., Cohn Z. A. Stimulus-dependent myristoylation of a major substrate for protein kinase C. Nature. 1988 Mar 24;332(6162):362–364. doi: 10.1038/332362a0. [DOI] [PubMed] [Google Scholar]
- Agrawal H. C., Randle C. L., Agrawal D. In vivo acylation of rat brain myelin proteolipid protein. J Biol Chem. 1982 Apr 25;257(8):4588–4592. [PubMed] [Google Scholar]
- Aitken A., Cohen P., Santikarn S., Williams D. H., Calder A. G., Smith A., Klee C. B. Identification of the NH2-terminal blocking group of calcineurin B as myristic acid. FEBS Lett. 1982 Dec 27;150(2):314–318. doi: 10.1016/0014-5793(82)80759-x. [DOI] [PubMed] [Google Scholar]
- Albert K. A., Nairn A. C., Greengard P. The 87-kDa protein, a major specific substrate for protein kinase C: purification from bovine brain and characterization. Proc Natl Acad Sci U S A. 1987 Oct;84(20):7046–7050. doi: 10.1073/pnas.84.20.7046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albert K. A., Walaas S. I., Wang J. K., Greengard P. Widespread occurrence of "87 kDa," a major specific substrate for protein kinase C. Proc Natl Acad Sci U S A. 1986 May;83(9):2822–2826. doi: 10.1073/pnas.83.9.2822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Buss J. E., Kamps M. P., Sefton B. M. Myristic acid is attached to the transforming protein of Rous sarcoma virus during or immediately after synthesis and is present in both soluble and membrane-bound forms of the protein. Mol Cell Biol. 1984 Dec;4(12):2697–2704. doi: 10.1128/mcb.4.12.2697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buss J. E., Mumby S. M., Casey P. J., Gilman A. G., Sefton B. M. Myristoylated alpha subunits of guanine nucleotide-binding regulatory proteins. Proc Natl Acad Sci U S A. 1987 Nov;84(21):7493–7497. doi: 10.1073/pnas.84.21.7493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chamberlain J. P. Fluorographic detection of radioactivity in polyacrylamide gels with the water-soluble fluor, sodium salicylate. Anal Biochem. 1979 Sep 15;98(1):132–135. doi: 10.1016/0003-2697(79)90716-4. [DOI] [PubMed] [Google Scholar]
- FOLCH J., LEES M. Proteolipides, a new type of tissue lipoproteins; their isolation from brain. J Biol Chem. 1951 Aug;191(2):807–817. [PubMed] [Google Scholar]
- GRAY E. G., WHITTAKER V. P. The isolation of nerve endings from brain: an electron-microscopic study of cell fragments derived by homogenization and centrifugation. J Anat. 1962 Jan;96:79–88. [PMC free article] [PubMed] [Google Scholar]
- Glover C. J., Goddard C., Felsted R. L. N-myristoylation of p60src. Identification of a myristoyl-CoA:glycylpeptide N-myristoyltransferase in rat tissues. Biochem J. 1988 Mar 1;250(2):485–491. doi: 10.1042/bj2500485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graff J. M., Gordon J. I., Blackshear P. J. Myristoylated and nonmyristoylated forms of a protein are phosphorylated by protein kinase C. Science. 1989 Oct 27;246(4929):503–506. doi: 10.1126/science.2814478. [DOI] [PubMed] [Google Scholar]
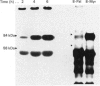
- Graff J. M., Stumpo D. J., Blackshear P. J. Characterization of the phosphorylation sites in the chicken and bovine myristoylated alanine-rich C kinase substrate protein, a prominent cellular substrate for protein kinase C. J Biol Chem. 1989 Jul 15;264(20):11912–11919. [PubMed] [Google Scholar]
- Grand R. J. Acylation of viral and eukaryotic proteins. Biochem J. 1989 Mar 15;258(3):625–638. doi: 10.1042/bj2580625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- James G., Olson E. N. Myristoylation, phosphorylation, and subcellular distribution of the 80-kDa protein kinase C substrate in BC3H1 myocytes. J Biol Chem. 1989 Dec 15;264(35):20928–20933. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Magee A. I., Courtneidge S. A. Two classes of fatty acid acylated proteins exist in eukaryotic cells. EMBO J. 1985 May;4(5):1137–1144. doi: 10.1002/j.1460-2075.1985.tb03751.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McIlhinney R. A., McGlone K. A simplified assay for the enzyme responsible for the attachment of myristic acid to the N-terminal glycine residue of proteins, myristoyl-CoA: glycylpeptide N-myristoyltransferase. Biochem J. 1989 Oct 15;263(2):387–391. doi: 10.1042/bj2630387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McIlhinney R. A., McGlone K. Characterisation of a myristoyl CoA:glycylpeptide N-myristoyl transferase activity in rat brain: subcellular and regional distribution. J Neurochem. 1990 Jan;54(1):110–117. doi: 10.1111/j.1471-4159.1990.tb13289.x. [DOI] [PubMed] [Google Scholar]
- McIlhinney R. A., Pelly S. J., Chadwick J. K., Cowley G. P. Studies on the attachment of myristic and palmitic acid to cell proteins in human squamous carcinoma cell lines: evidence for two pathways. EMBO J. 1985 May;4(5):1145–1152. doi: 10.1002/j.1460-2075.1985.tb03752.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt J. W., Catterall W. A. Palmitylation, sulfation, and glycosylation of the alpha subunit of the sodium channel. Role of post-translational modifications in channel assembly. J Biol Chem. 1987 Oct 5;262(28):13713–13723. [PubMed] [Google Scholar]
- Schmidt M. F. Fatty acylation of proteins. Biochim Biophys Acta. 1989 Dec 6;988(3):411–426. doi: 10.1016/0304-4157(89)90013-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultz A. M., Tsai S. C., Kung H. F., Oroszlan S., Moss J., Vaughan M. Hydroxylamine-stable covalent linkage of myristic acid in G0 alpha, a guanine nucleotide-binding protein of bovine brain. Biochem Biophys Res Commun. 1987 Aug 14;146(3):1234–1239. doi: 10.1016/0006-291x(87)90780-7. [DOI] [PubMed] [Google Scholar]
- Slomiany A., Jozwiak Z., Takagi A., Slomiany B. L. The role of covalently bound fatty acids in the degradation of human gastric mucus glycoprotein. Arch Biochem Biophys. 1984 Mar;229(2):560–567. doi: 10.1016/0003-9861(84)90188-7. [DOI] [PubMed] [Google Scholar]
- Slomiany B. L., Murty V. L., Sarosiek J., Piotrowski J., Slomiany A. Role of associated and covalently bound lipids in salivary mucin hydrophobicity: effect of proteolysis and disulfide bridge reduction. Biochem Biophys Res Commun. 1988 Mar 30;151(3):1046–1053. doi: 10.1016/s0006-291x(88)80471-6. [DOI] [PubMed] [Google Scholar]
- Stumpo D. J., Graff J. M., Albert K. A., Greengard P., Blackshear P. J. Molecular cloning, characterization, and expression of a cDNA encoding the "80- to 87-kDa" myristoylated alanine-rich C kinase substrate: a major cellular substrate for protein kinase C. Proc Natl Acad Sci U S A. 1989 Jun;86(11):4012–4016. doi: 10.1073/pnas.86.11.4012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towler D. A., Adams S. P., Eubanks S. R., Towery D. S., Jackson-Machelski E., Glaser L., Gordon J. I. Myristoyl CoA:protein N-myristoyltransferase activities from rat liver and yeast possess overlapping yet distinct peptide substrate specificities. J Biol Chem. 1988 Feb 5;263(4):1784–1790. [PubMed] [Google Scholar]
- Towler D. A., Gordon J. I., Adams S. P., Glaser L. The biology and enzymology of eukaryotic protein acylation. Annu Rev Biochem. 1988;57:69–99. doi: 10.1146/annurev.bi.57.070188.000441. [DOI] [PubMed] [Google Scholar]
- Wilcox C., Hu J. S., Olson E. N. Acylation of proteins with myristic acid occurs cotranslationally. Science. 1987 Nov 27;238(4831):1275–1278. doi: 10.1126/science.3685978. [DOI] [PubMed] [Google Scholar]