Abstract
Background
This study assessed the association between a novel inflammatory marker, uric acid (UA)-to-albumin ratio (UAR), and preprocedural intracoronary artery thrombus (ICAT) in ST elevation myocardial infarction (STEMI).
Material/Methods
A total of 171 STEMI patients treated by primary percutaneous coronary intervention between February and December 2023 were evaluated prospectively in this cross-sectional study. The patients were stratified into 2 groups as low (grades 1 to 3) and high-(ICAT) groups (grades 4 and 5). To determine the independent predictors of lower and higher ICAT, multivariate regression analysis was performed.
Results
C-reactive protein (CRP), UA, and UAR were significantly higher in the high ICAT group (1.11 (0.3–2.8) vs 0.80 (0.10–2.8), P=0.037; 5.4 (3.5–7.2) vs 4.9 (3.4–5.6), P<0.001; 1.78 (0.82–3) vs 1.48 (0.77–2.57), P<0.001, respectively). However, albumin levels were similar between groups (3.1 (2.1–4.4) vs 3.3 (2.1–4.4), P=0.243). Higher UAR (OR: 3.95% CI: 1.23–12.7, P=0.021), lower left ventricular ejection fraction (LVEF) (OR=0.802; 95% CI 0.7537–0.872; P<0.001), longer pain-wire crossing time (OR=1; 95% CI: 1–1.02; P<0.001), and diabetes mellitus (OR=0.181; 95% CI 0.46–0.7; P<0.001) were independent predictors of ICAT.
Conclusions
UAR, a marker of inflammation, is an independent predictor of ICAT in patients with STEMI.
Keywords: Albumins, Coronary Artery Disease, Inflammation
Introduction
Despite advancements in management, acute ST elevation myocardial infarction (STEMI) continues to be an important disease. Use of primary percutaneous coronary intervention (PCI) has caused decrease in mortality, but proper management during and after diagnosis is still quite important [1,2]. Defining the patient’s risk at admission can help set appropriate medical and interventional therapies to define discharge time and time for follow-up and prevent adverse outcomes [2,3].
The role of inflammation in atherosclerosis and STEMI has been shown in several studies [4–6]. Uric acid (UA), which is the final product of purine metabolism, possesses antioxidant properties under normal conditions [7]. However, UA may act as a pro-oxidant molecule and accelerate atherosclerosis in coronary plaques and was reported to be related to coronary artery disease (CAD) and mortality [8,9]. Serum albumin (SA) has antioxidant and anticoagulant functions and performs many vital functions in the body [10]. During inflammation, SA levels decrease because of synthesis-degradation imbalance in favor of degradation [11]. Several inflammatory parameters, including uric acid (UA) to SA ratio (UAR), have been proposed to predict cardiovascular adverse outcomes in patients with STEMI [5,12]. In patients with acute coronary syndrome (ACS), angiographic preprocedural intracoronary thrombus was reported to be present in nearly one-fifth of patients. In addition, high preprocedural intracoronary artery thrombus (ICAT) was shown to be significantly related to increased thrombotic complications and death [13,14]. High ICAT has been demonstrated to be associated with compromised microvascular perfusion resulting from distal embolization, even after complete reopening of the infarct-related artery [14]. Several studies have shown the relationship between inflammation and ICAT [13,15,16]. In this study, our objective was to explore the relationship between UAR and ICAT in STEMI patients who underwent primary PCI treatment.
Material and Methods
Study Population and Design
The patients and their relatives were informed about the study and provided written consent. This article was reviewed by the Recep Tayyip Erdoğan University Non-Interventional Clinical Research Ethics Committee at its meeting held on 11.01.2024, in accordance with the ethics committee directive, and was found to be scientifically and ethically appropriate (approval number 2024/09).
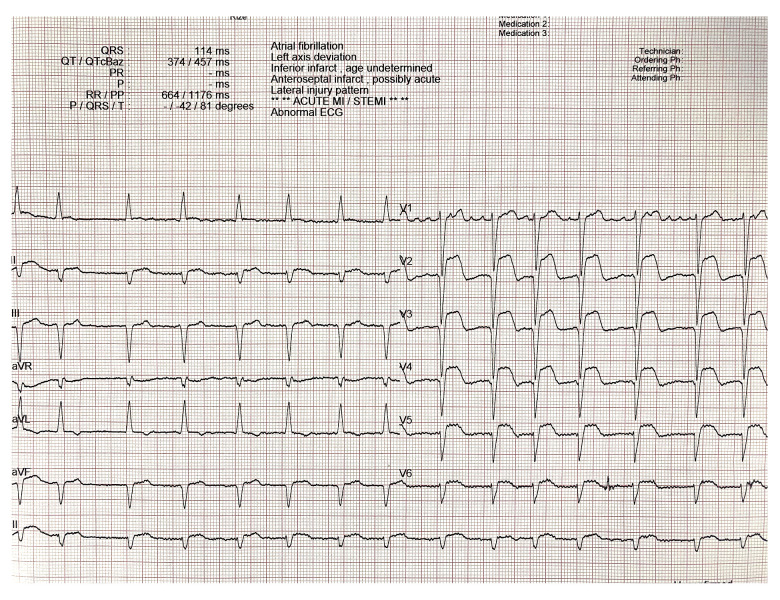
A total of 171 STEMI patients admitted to our emergency service that were treated by primary PCI between February 2023 and December 2023 were evaluated prospectively in this cross-sectional study. The clinical and laboratory parameters were documented upon admission. A comprehensive physical examination was conducted, collecting data on smoking status, history of coronary artery disease (CAD), hypertension (defined as systolic blood pressure [SBP] >140 mmHg and/or diastolic blood pressure [DBP] >90 mmHg in at least 2 measurements or the use of antihypertensive therapy), diabetes mellitus (DM), family history of CAD, hyperlipidemia (HL), and extra-cardiac conditions such as active or chronic infections, cancer, chronic obstructive pulmonary disease, chronic autoimmune and systemic inflammatory diseases, and chronic renal or hepatic diseases. The current use of antiplatelets, beta blockers (BB), statins, and angiotensin-converting enzyme inhibitors (ACEIs) was documented. Patients’ heart rate, systolic blood pressure (SBP), and diastolic blood pressure (DBP) were recorded. To diagnose STEMI, a standard 12-lead ECG was performed using an electrocardiography (ECG) device (Nihon Kohden, Tokyo, Japan). The electrocardiographic and clinical diagnosis of STEMI was established according to the fourth universal definition of myocardial infarction (MI) criteria. STEMI diagnosis was based on the following criteria according to the current guidelines: in the presence of ischemic chest pain, ≥1 mm new ST-segment elevation or more at the J point in 2 or more contiguous leads in at least 2 contiguous anterior chest leads on electrocardiography (ECG) [17] (Figure 1). The MI patterns were defined as anterior and non-anterior, including inferior, high lateral, and true posterior MI. The pain to wire crossing (PWC) time was determined according to the latest STEMI guidelines as the time in seconds between the onset of ischemic chest pain and to wire crossing that provides reperfusion in culprit distal coronary artery [2,18,19]. Emergency transthoracic echocardiography at admission with measurement of left ventricular ejection fraction (LVEF) was performed in all patients (GE VividTM 8 Ultrasound Machine; GE Healthcare, Piscataway, NJ, USA) according to the current clinical practice guidelines [20]. Body mass index (BMI) was computed as weight in kilograms divided by the square of height in meters. After the initial assessment, each patient was transferred to the coronary catheterization unit. We excluded patients with rheumatologic/connective tissue diseases and gout, those who underwent fibrinolytic therapy and required emergency coronary bypass surgery, individuals with end-stage kidney disease and malignancy, and those with culprit lesions in the left main coronary artery and myocardial infarction with non-obstructive coronary arteries.
Figure 1.
STEMI diagnosis based on ECG.
Blood Samples
Blood samples were drawn from the antecubital vein. Admission hemoglobin (Hgb), troponin, C-reactive protein (CRP), albumin concentration (Abbott Architect c1600 Clinical Chemistry; Abbott Core Laboratory, Germany, normal range: 3.5–5.5 g/dL), and serum UA level (Roche Cobas C analyzer with colorimetric uricase method [Roche Diagnostics, Indianapolis, IN, normal range: 1.5–7 mg/dL]) were measured with standard methods from the obtained blood specimens. UA, creatinine, SA, and serum lipids, including total cholesterol (TC), low-density lipoprotein (LDL), high-density lipoprotein (HDL), and triglycerides (TG), were measured using an ARCHITECT c16000 clinical chemistry analyzer (Abbott Laboratories, Abbott Park, IL, USA). Complete blood count, including white blood cells (WBC) and platelet counts (PLT), was assessed using a CELL-DYN Ruby hematology analyzer (Abbott Laboratories). The UA- to-albumin ratio was determined by dividing the UA level by the SA level. The glomerular filtration rate (GFR) was calculated using the Cockroft-Gault formula.
Coronary Angiographic Definitions and Primary PCI
Coronary angiography data were collected by reviewing the saved images. The coronary angiography was performed through either femoral or radial artery with a C-arm angiography system (Siemens, Erlangen, Germany), and ≥50% stenosis in a major coronary artery was accepted as significant obstruction. The appropriate anticoagulant and antiplatelet treatments were started at admission with 300 mg chewable aspirin and a loading dose of 600 mg clopidogrel given. A standard heparin bolus of 70 U/kg was given before the procedure. Intravenous tirofiban infusion was started at the discretion of the physician with a bolus of 10 mg/kg and 0.15 mg/kg/min maintenance dose. Percutaneous coronary intervention (PCI) for the target artery and complete revascularization, when required, were carried out by following current revascularization guidelines [22]. To navigate through the coronary blockage, 0.014-inch guidewires were employed. The angiograms were reviewed by 2 experienced interventional cardiologists who were blinded to the patients’ clinical data. In addition, all patients were treated with the same medication and invasive procedure, and none of patients received thrombolytic therapy during the follow-up.
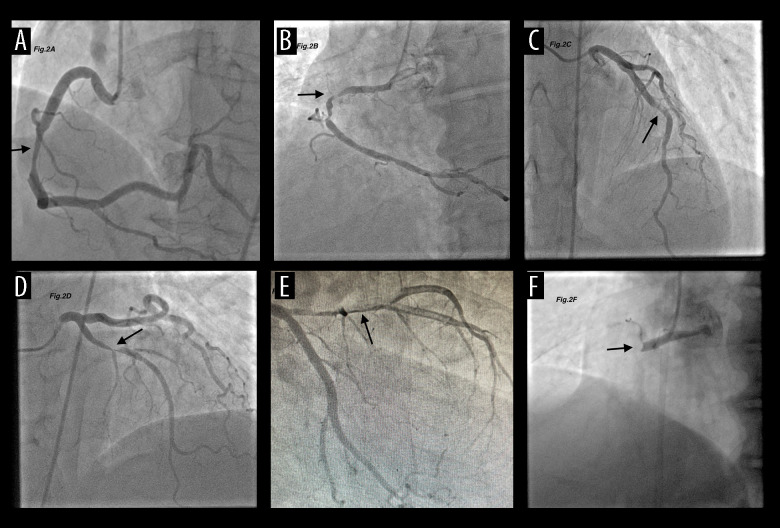
Definition of High-Grade ICAT
Angiographic ICAT burden was classified as the following TIMI grades: Grade 0: no thrombus, Grade 1: Possible thrombus, Grade 2: the greatest dimension of thrombus is <1/2 of the vessel diameter, Grade 3: The greatest dimension of thrombus is between >1/2 and <2 of vessel diameters, Grade 4: The greatest dimension of thrombus is >2 of vessel diameters, and Grade 5: total vessel occlusion due to the thrombus. Grade ≥4 was considered to be a high-grade ICT burden [23] (Figure 2). ICAT assessment was done immediately after anterograde flow was restored by guidewire passage or minor balloon inflation. Patients were categorized into low-ICAT (grades 1 to 3) and high-ICAT groups (grades 4 and 5) based on the final thrombus score. To evaluate coronary no-reflow, the thrombolysis in myocardial infarction (TIMI) flow of the culprit artery was assessed. This was classified as TIMI 0 for complete obstruction after the lesion, TIMI 1 if contrast reached the obstruction without distal contrast penetration, TIMI 2 if contrast filled the entire artery but with slow flow, and TIMI 3 if normal perfusion was achieved. Coronary no-reflow was defined as TIMI 0–1 flow in the culprit artery after balloon angioplasty and stenting [24].
Figure 2.
Thrombus burden classification. (A) No thrombus, (B) Possible thrombus, (C) thrombus ≤1/2 reference vessel diameter (RDV), (D) thrombus >1/2 RDV <2RDV, (E) thrombus ≥2RDV, (F) total occlusion. The Thrombolysis In the Myocardial Infarction (TIMI) thrombus classification consists of 6 grades from grade 0 (no thrombus) to grade 5 (total occlusion).
Statistical Analyses
The statistical analyses were performed with SPSS 18 (SPSS, Inc, Chicago, Illinois). A P value of <0.05 was assumed to be significant. Shapiro-Wilk test was used to determine the distribution of normality of continuous variables. The continuous and categorical variables are presented as mean±standard deviation (±SD) and percentages (%), respectively. Median and interquartile range (IQR) were used for variables with non-normal distribution. The categorical variables are presented as percentages and numbers, and the chi-square test was used to compare the groups. We used the t test or Mann-Whitney U test to compare continuous variables between the groups, depending on normal or non-normal distribution of these variables. The relationship between ICAT groups and other parameters was evaluated by logistic regression analysis. The variables with P value less than 0.05 in univariate analysis were included in multivariate logistic regression analysis. To determine the independent predictors of lower and higher ICAT, multivariate logistic regression analysis was performed by using the variables that were determined in univariate analysis. According to the parameters that showed co-variability, we formed 2 distinct models: UAR (model 1), and UA, albumin, and CRP (model 2). The predictive validities were quantified as the area under the ROC curve (c statistics) to estimate sensitivity and specificity of the variables which had P value <0.05 in univariate analysis [25].
Results
Our study included a total of 171 consecutive patients diagnosed with STEMI. Table 1 summarizes the basic clinical, demographic, and laboratory features of the study groups and their comparisons. The patients were allocated into 2 groups according to their ICAT category: 114 patients had high ICAT and 57 had low ICAT. The mean age of the patients was 59.9 years and 42.1% were female. The demographic characteristics, including age and sex, were similar between groups (P=0.753 and P=0.105, respectively). The family history and number of hypertensives were also similar between groups (P=0.203 and P=0.884, respectively). The number of diabetic patients were higher in high ICAT group, without reaching statistical significance (22.1% vs 10.7%, P=0.052). The systolic blood pressures at admission were significantly lower in the high ICAT group (135±11 vs 141±120, P=0.046). The MI localizations were similar between groups (55.3% anterior vs 44.7% non-anterior MI in the high ICAT group, P=0.109). The proportion of patients with no-reflow was higher in the high ICAT group, without reaching statistical significance (5.3% vs 1.8%, P=0.275). PWC time was significantly longer in the high ICAT group (341±96 vs 265.7±127, P<0.001). The troponin levels were significantly higher in the high ICAT group [645 (100–7000) vs 480 (55–4000), P=0.003]. The serum levels of LDL (150±15 vs 146±14, P=0.176), HDL (30±13 vs 30±10, P=0.935), triglycerides (205±71 vs 192±69, P=0.702) and GFR (60.8±18 vs 63±17, P=0.460) were similar between groups. CRP, UA, and UAR were significantly higher in the high ICAT group (1.11 (0.3–2.8) vs 0.80 (0.10–2.8), P=0.037; 5.4 (3.5–7.2) vs 4.9 (3.4–5.6), P<0.001; 1.78 (0.82–3) vs 1.48 (0.77–2.57), P<0.001, respectively). However, albumin level was similar between groups (3.1 (2.1–4.4) vs 3.3 (2.1–4.4), P=0.243). In univariate logistic regression analysis, age, diabetes, CRP, and troponin levels did not reach statistical significance. However, UA, UAR, LVEF, and PWC time had significant association. Table 2 presents 2 distinct models in our multivariate analysis. Model 1 showed that higher UAR (odds ratio (OR): 3.95% CI: 1.23–12.7, P=0.021), lower LVEF (OR=0.802; 95% CI 0.7537–0.872; P<0.001), longer PWC time (OR=1; 95% CI: 1–1.02; P<0.001), and diabetes (OR=0.181; 95% CI 0.46–0.7; P<0.001) had significant association with ICAT. Model 2 showed significant association between higher UA and high ICAT (OR=2.42; 95% CI: 1.36–4.28; P=0.002), while a nonsignificant association was found between SA and high ICAT (OR=0.912; 95% CI: 0.38–2.17; P=0.836). The cutoff values of UAR, LVEF, and PWC time in predicting high ICAT were 1.75 with a sensitivity of 51.8% and a specificity of 82.5% (area under curve (AUC), 0.680; 95% CI, 0.604–0.749; P<0.001), 44% with a sensitivity of 63.2% and a specificity of 84.2% (AUC, 0.806; 95% CI, 0.739–0.862; P<0.001), and 280 with a sensitivity of 94.7% and a specificity of 63.2% (AUC, 0.846; 95% CI, 0.783–0.896; P<0.001), respectively, in the ROC curve analysis (Figure 3).
Table 1.
Baseline clinical and laboratory characteristics according to ICAT.
| Variable | All patients (n=171) | Low TB (n=57) | High TB (n=114) | P value |
|---|---|---|---|---|
| Age, years | 59.9±10.8 | 59.5±10.8 | 60.2±10.8 | 0.703 |
| Sex, male % | 42.1 | 43.5 | 49.5 | 0.105 |
| Diabetes, % | 18.3 | 10.7 | 22.1 | 0.052 |
| Hypertension, % | 38.6 | 33.3 | 41.6 | 0.203 |
| Smoking, % | 21.6 | 19.3 | 22.8 | 0.376 |
| Dyslipidemia, % | 41.9 | 41.8 | 42.0 | 0.560 |
| Previous history of CAD, % | 18.3 | 15.8 | 16.7 | 0.536 |
| Family history of CAD | 32.4 | 15.8 | 16.7 | 0.884 |
| SBP, mmHg | 137±18 | 141±120 | 135±11 | 0.046 |
| CRP, mg/dL | 1 (0.1–2.80) | 0.80 (0.10–2.8) | 1.11 (0.3–2.8) | 0.037 |
| Uric acid mg/dL | 5.2 (3.4–7.2) | 4.9 (3.4–5.6) | 5.4 (3.5–7.2) | <0.001 |
| Albumin, g/dL | 3.2 (2.7–4.4) | 3.3 (2.1–4.4) | 3.1 (2.1–4.4) | 0.243 |
| UAR | 1.61 (0.77–3) | 1.48 (0.77–2.57) | 1.78 (0.82–3) | <0.001 |
| Hemoglobin, g/dL | 13.4±2 | 14.8±1.6 | 14.5±1.7 | 0.430 |
| WBC count×103/μL | 7.9±2.5 | 8.3±2.6 | 7.8±2.4 | 0.198 |
| Baseline troponin I, Ng/L* | 555 (55–7000) | 480 (55–4000) | 605 (100–7000) | 0.003 |
| LDL cholesterol, mg/dL | 148±14 | 150±15 | 146±14 | 0.176 |
| HDL cholesterol, mg/dL | 30±11 | 30±13 | 30±10 | 0.935 |
| Triglyceride, mg/dL | 196±70 | 205±71 | 192±69 | 0.702 |
| GFR | 61±17 | 60.8±18 | 63±17 | 0.460 |
| BMI | 24 (16–36) | 23 (16–31) | 24 (16.9–36) | 0.206 |
| LVEF, % | 46.2±9.2 | 50.5±6.3 | 42.3±6.7 | <0.001 |
| No-reflow % | 4.1 | 1.8 | 5.3 | 0.275 |
| PWC time, min | 315±87 | 265.7±127 | 341±96 | <0.001 |
| Previous medications, % | ||||
| Aspirin | 20.5 | 19.3 | 21.1 | 0.479 |
| Statin | 17.5 | 19.3 | 16.7 | 0.674 |
| ACE inhibitors/ARB | 24.0 | 21.1 | 25.4 | 0.574 |
| β-blocker | 12.9 | 14.0 | 12.3 | 0.810 |
ACE – angiotensin converting enzyme; ARB – angiotensin receptor blocker; BMI – body mass index; CAD – coronary artery disease; CCB – calcium channel blocker; CRP – C-reactive protein; LVEF – left ventricular ejection fraction; GFR – glomerular filtration rate; HDL – high-density lipoprotein; ICAT – intracoronary arterial thrombus; LDL – low-density lipoprotein; PWC – pain to wire crossing time; SBP – systolic blood pressure; UA – uric acid; UAR – uric acid albumin ratio; WBC – white blood cell.
The normal cut-off value of troponin in our laboratory <100 Ng/L.
Table 2.
Independent predictors of preprocedural intracoronary artery thrombus (ICAT) with multivariate P value, OR with 95% CI.
| Variable | Univariate | Multivariate | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Model 1 (UAR) | Model 2 (UA, albumin, CRP) | ||||||||
| OR | 95% CI | P value | OR | 95% CI | P value | OR | 95% CI | P value | |
| Age | 1 | 0.977–1036 | 0.701 | ||||||
| Diabetes | 0.422 | 0.160–1.09 | 0.77 | 0.181 | 0.46–0.7 | 0.014 | 0.13 | 0.032–0.59 | 0.008 |
| UA | 2.57 | 1.70–3.89 | <0.001 | 2.42 | 1.36–4.28 | 0.002 | |||
| Albumin | 0.70 | 0.40–1.22 | 0.212 | 0.912 | 0.38–2.17 | 0.836 | |||
| UAR | 5 | 2.19–11.4 | <0.001 | 3.95 | 1.23–12.7 | 0.021 | |||
| CRP | 1.88 | 1.02–3.4 | 0.40 | 1.2 | 0.51–2.88 | 0.650 | 1 | 0.39–2.50 | 0.1 |
| Troponin | 1 | 1–1 | 0.55 | 1 | 1–1.00 | 0.44 | 1 | 1–1.00 | 0.101 |
| LVEF | 0.64 | 0.55–0.74 | <0.001 | 0.802 | 0.7537–0.872 | <0.001 | 0.85 | 0.73–0.87 | <0.001 |
| PWC time | 1.01 | 1.01–1.02 | <0.001 | 1 | 1–1.02 | <0.001 | 1.01 | 1–1.02 | <0.001 |
CRP – C-reactive protein; LVEF – left ventricular ejection fraction; ICAT – intracoronary arterial thrombus; PWC – pain to wire crossing; UA – uric acid; OR – odds ratio; UAR – uric acid albumin ratio.
Figure 3.
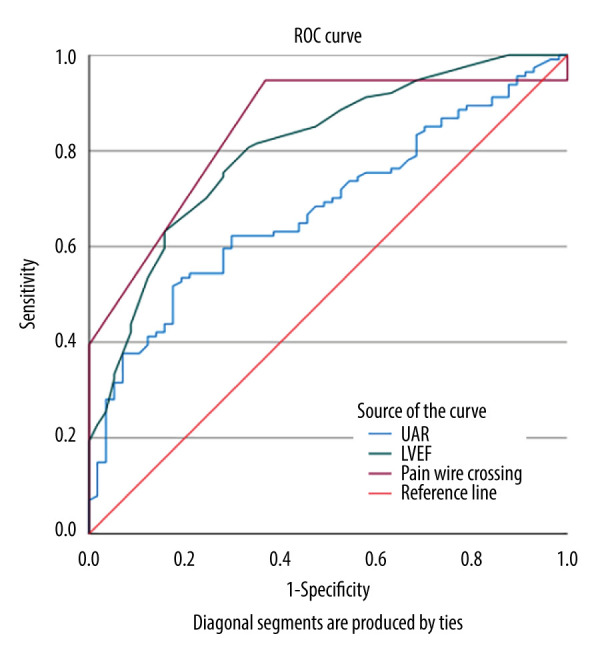
The receiver operating characteristic (ROC) curve comparison of uric acid-to-albumin ratio (UAR), pain to wire crossing time and left ventricular ejection fraction (LVEF) in the prediction of low–high preprocedural intracoronary artery thrombus. (SPSS Statistics version 29.0.2.0.(20) IBM SPSS Statistics software).
Discussion
The main finding in this study was that higher ICAT was significantly related to CRP, UA, and UAR in patients with acute STEMI. Higher UAR and UA, lower LVEF, longer PWC time, and diabetes mellitus are independent predictors of ICAT.
STEMI, one of the most devastating presentations of acute coronary syndromes, was reported to have an annual incidence of 750 000, including new and recurrent admissions [26]. Besides medical therapy, prompt coronary intervention and recognition and treatment of complications remains the mainstay of the management [26].
High ICAT in STEMI was reported to be related to larger infarct size with increased risk of distal embolization, coronary no-reflow, and death [27,28]. Inflammation and thrombosis are closely related with a conjoint association, in which inflammation is both the cause and result [13]. After acute artery occlusion, inflammatory molecules are released from endothelium and ischemic myocardial tissue [13,29]. Inflammation has been extensively studied in atherosclerosis and acute STEMI [30]. Several inflammatory parameters play a role as surrogate markers of atherosclerotic disease severity and help to discriminate more extensive disease and individuals with higher risk [5,13,15,16,25]. These may help clinicians to decide the aggressiveness of medical and interventional therapies and discharge time, as well as closer follow-up [28].
The prevalence of traditional risk factors, including male predominance, older age, presence of diabetes mellitus (DM), hypertension (HT), smoking, dyslipidemia, and previous history of coronary artery disease (CAD), were similar between our low and high thrombosis burden groups. However, most of these risk factors were present in the high ICAT group [31,32]. This finding may be an advantage in comparison of our inflammation parameters between groups. The number of diabetics was slightly higher in the high ICAT group, without reaching statistical significance. Diabetes mellitus is an important risk factor and was reported to be related to high thrombogenicity with increased recurrent ischemia risk even after treatment by PCI and guideline-directed medical therapy in acute coronary syndrome patients as compared to individuals without DM [25,33]. This finding led us to include DM in our multivariate analysis, even though it was not significantly correlated in our univariate analysis. In both multivariate analysis models, DM was an independent predictor of high ICAT. The admission SBPs were lower in the high ICAT group, which may be explained by including more higher-risk individuals in this group. In some studies, patients with higher admission SBP were reported to have better outcomes in terms of major adverse cardiovascular events and in-hospital mortality independent of ICAT [34,35]. Admission CRP levels were higher in our high ICAT group, which may indicate higher rates of inflammation and more thrombotic risk in this group. In the study of Kaplangoray et al, CRP-to-albumin ratio was reported to be associated with high ICAT in patients with newly diagnosed STEMI [36]. CRP was also previously demonstrated to be related to high ICAT in another study [37]. Significantly higher troponin level in the high ICAT group is another finding similar to previous research [38]. Although they were found to be similar between groups, moderately elevated LDL and TG levels and lower HDL levels in our patients may reflect the fact that dyslipidemia is a traditional risk factor for STEMI [1]. Our findings showed that LVEF was significantly lower in the high ICAT group and was an independent predictor of higher ICAT. LVEF, as a prognostic marker in STEMI, is related to adverse cardiovascular events, including heart failure and death [1]. A very recent meta-analysis of randomized trials reported that thrombus aspiration had favorable effects on myocardial remodeling and left ventricular function in STEMI [38]. The data regarding the relationship between LVEF and ICAT presented in our paper shows the important effect of ICAT on left ventricular functions in STEMI. PWC time is crucial for salvage of myocardium under risk [2,3,39]. The direct association of ICAT and PWC time has not been reported previously. In this study, we demonstrated significantly higher PWC times in the high ICAT group. Additionally, PWC time was observed to be an independent predictor of ICAT, with a cutoff value of 280 seconds to discriminate low and high ICAT, with sensitivity of 95% and 63% specificity. In our model-2 multivariate analysis, UAR and UA were also found to be independent predictors of high ICAT. This finding is remarkable since the role of UA may be more important than albumin in prediction of high ICAT. Although the albumin level was lower in the high ICAT group, the difference was not statistically significant and did not predict higher ICAT. This may be due to the similarity of the ICAT groups in terms of traditional risk factors and GFR. However, a different relationship was reported in our previous study [13] in which we found significant relationship between the CRP–albumin ratio and high ICAT. We also reported a similar significant relationship between lower albumin levels and high ICAT and found a significant difference in albumin levels between our study groups [13]. Since albumin is also a nutritional marker, its levels may have been affected by some other uncontrollable parameters in our study [10,11]. Nevertheless, our findings regarding UAR and UA are comparable with previous studies [5,8,9,25]. The association of several parameters such as risk of development of new onset atrial fibrillation, mortality, and no-reflow phenomenon with UAR in STEMI patients were studied recently [5,40,41]. UAR is a relatively novel surrogate parameter that can be used to predict high ICAT in STEMI. Along with clinical and angiographic findings, it can help to select the optimal management in this patient cohort. To the best of our knowledge, this is the first report of a relationship between UAR and high ICAT in patients with STEMI. Further studies are needed to elucidate its validity in STEMI.
Our study has several limitations. The primary limitation is that it was a single-center study with a relatively small number of patients. Another limitation is the lack of patient follow-up due to the cross-sectional nature of our study. Additionally, a control group was not included in this study.
Conclusions
As an inflammatory marker, UAR, together with PWC time and admission LVEF, may be related to the preprocedural high ICAT in patients presenting with acute STEMI. These surrogate variables may be helpful to identify patients at higher risk and determine which patients need more aggressive management.
Acknowledgements
All authors would like to thank the management at Recep Tayyip Erdoğan University.
Footnotes
Conflict of interest: None declared
Publisher’s note: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher
Declaration of Figures’ Authenticity: All figures submitted have been created by the authors, who confirm that the images are original with no duplication and have not been previously published in whole or in part.
Financial support: None declared
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
References
- 1.Elendu C, Amaechi DC, Elendu TC, et al. Comprehensive review of ST-segment elevation myocardial infarction: Understanding pathophysiology, diagnostic strategies, and current treatment approaches. Medicine (Baltimore) 2023;102(43):e35687. doi: 10.1097/MD.0000000000035687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ibanez B, James S, Agewall S, et al. ESC Scientific Document Group. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC) Eur Heart J. 2018;39(2):119–77. doi: 10.1093/eurheartj/ehx393. [DOI] [PubMed] [Google Scholar]
- 3.Thune JJ, Hoefsten DE, Lindholm MG, et al. Simple risk stratification at admission to identify patients with reduced mortality from primary angioplasty. Circulation. 2005;112:2017–21. doi: 10.1161/CIRCULATIONAHA.105.558676. [DOI] [PubMed] [Google Scholar]
- 4.Raggi P, Genest J, Giles JT, et al. Role of inflammation in the pathogenesis of atherosclerosis and therapeutic interventions. Atherosclerosis. 2018;276:98–108. doi: 10.1016/j.atherosclerosis.2018.07.014. [DOI] [PubMed] [Google Scholar]
- 5.Kalkan S, Cagan Efe S, Karagöz A, et al. A new predictor of mortality in ST-elevation myocardial infarction: The uric acid albumin ratio. Angiology. 2022;73(5):461–69. doi: 10.1177/00033197211066362. [DOI] [PubMed] [Google Scholar]
- 6.Ipek E, Yolcu M, Yildirim E, et al. A novel marker of inflammation: Azurocidin in patients with ST Segment Elevation Myocardial Infarction. Int J Mol Sci. 2018;19(12):3797. doi: 10.3390/ijms19123797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Glantzounis GK, Tsimoyiannis EC, Kappas AM, Galaris DA. Uric acid and oxidative stress. Curr Pharm Des. 2005;11(32):4145–51. doi: 10.2174/138161205774913255. [DOI] [PubMed] [Google Scholar]
- 8.Feig DI, Kang DH, Johnson RJ. Uric acid and cardiovascular risk. N Engl J Med. 2008;359:1811–21. doi: 10.1056/NEJMra0800885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ndrepepa G. Uric acid and cardiovascular disease. Clin Chim Acta. 2018;484:150–63. doi: 10.1016/j.cca.2018.05.046. [DOI] [PubMed] [Google Scholar]
- 10.Cheng CW, Lee CW, Chien SC, et al. Serum albumin was associated with a long term cardiovascular mortality among elderly patients with stable coronary artery disease. Acta Cardiol Sin. 2024;40(1):87–96. doi: 10.6515/ACS.202401_40(1).20230825A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Don BR, Kaysen G. Serum albumin: Relationship to inflammation and nutrition. Semin Dial. 2004;17:432–37. doi: 10.1111/j.0894-0959.2004.17603.x. [DOI] [PubMed] [Google Scholar]
- 12.Liu Y, Liu J, Liu L, et al. Association of systemic inflammatory response index and pan-immune-inflammation-value with long-term adverse cardiovascular events in ST-segment elevation myocardial infarction patients after primary percutaneous coronary intervention. J Inflamm Res. 2023;16:3437–54. doi: 10.2147/JIR.S421491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Duman H, Çinier G, Bakırcı EM, et al. Relationship between C-reactive protein to albumin ratio and thrombus burden in patients with acute coronary syndrome. Clin Appl Thromb Hemost. 2019;25:1076029618824418. doi: 10.1177/1076029618824418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Goto K, Lansky AJ, Nikolsky E, et al. Prognostic significance of coronary thrombus in patients undergoing percutaneous coronary intervention for acute coronary syndromes: A subanalysis of the ACUITY (Acute Catheterization and Urgent Intervention Triage strategY) trial. JACC Cardiovasc Interv. 2011;4(7):769–77. doi: 10.1016/j.jcin.2011.02.019. [DOI] [PubMed] [Google Scholar]
- 15.Dolu AK, Karayiğit O, Ozkan C, et al. Relationship between intracoronary thrombus burden and systemic immune-inflammation index in patients with ST-segment elevation myocardial infarction. Acta Cardiol. 2023;78(1):72–79. doi: 10.1080/00015385.2022.2035082. [DOI] [PubMed] [Google Scholar]
- 16.Özkan U, Gürdoğan M, Öztürk C, et al. Systemic immune-inflammation index: A novel predictor of coronary thrombus burden in patients with non-ST acute coronary syndrome. Medicina (Kaunas) 2022;58(2):143. doi: 10.3390/medicina58020143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Thygesen K, Alpert JS, Jaffe AS, et al. Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction. Fourth universal definition of myocardial infarction (2018) Circulation. 2018;138(20):e618–e51. doi: 10.1161/CIR.0000000000000617. [DOI] [PubMed] [Google Scholar]
- 18.O’Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;61:e78–140. doi: 10.1016/j.jacc.2012.11.019. [DOI] [PubMed] [Google Scholar]
- 19.Gulati M, Levy P, Amsterdam E, et al. 2021 AHA/ACC/ASE/CHEST/SAEM/SCCT/SCMR Guideline for the Evaluation and Diagnosis of Chest Pain: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2021;78(22):e187–285. doi: 10.1016/j.jacc.2021.07.053. [DOI] [PubMed] [Google Scholar]
- 20.Mitchell C, Rahko PS, Blauwet LA, et al. Guidelines for performing a comprehensive transthoracic echocardiographic examination in adults: Recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr. 2019;32(1):1–64. doi: 10.1016/j.echo.2018.06.004. [DOI] [PubMed] [Google Scholar]
- 21.Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16(1):31–41. doi: 10.1159/000180580. [DOI] [PubMed] [Google Scholar]
- 22.Writing Committee Members. Lawton JS, Tamis-Holland JE, Bangalore S, et al. 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79(2):197–215. doi: 10.1016/j.jacc.2021.09.005. [DOI] [PubMed] [Google Scholar]
- 23.Sianos G, Papafaklis MI, Serruys PW. Angiographic thrombus burden classification in patients with ST-segment elevation myocardial infarction treated with percutaneous coronary intervention. J Invasive Cardiol. 2010;22(10 Suppl B):6B–14B. [PubMed] [Google Scholar]
- 24.Braunwald E, Sabatine MS. The Thrombolysis in myocardial ınfarction (TIMI) study Group experience. J Thorac Cardiovasc Surg. 2012;144(4):762–70. doi: 10.1016/j.jtcvs.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 25.Çakmak EÖ, Bayam E, Çelik M, et al. Uric acid-to-albumin ratio: A novel marker for the extent of coronary artery disease in patients with non-ST-elevated myocardial infarction. Pulse (Basel) 2021;8(3–4):99–107. doi: 10.1159/000514533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Akbar H, Foth C, Kahloon RA, et al. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Acute ST-elevation myocardial infarction. [Updated 2023 Jul 31] Available from: https://www.ncbi.nlm.nih.gov/books/NBK532281/ [PubMed] [Google Scholar]
- 27.Napodano M, Dariol G, Al Mamary AH, et al. Thrombus burden and myocardial damage during primary percutaneous coronary intervention. Am J Cardiol. 2014;113:1449–56. doi: 10.1016/j.amjcard.2014.01.423. [DOI] [PubMed] [Google Scholar]
- 28.Zhang Z, Sheng Z, Che W, et al. Design and rationale of the ATTRACTIVE trial: A randomised trial of intrAThrombus Thrombolysis versus aspiRAtion thrombeCTomy during prImary percutaneous coronary interVEntion in ST-segment elevation myocardial infarction patients with high thrombus burden. BMJ Open. 2023;13(11):e076476. doi: 10.1136/bmjopen-2023-076476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kottoor SJ, Arora RR. The utility of anti-inflammatory agents in cardiovascular disease: A novel perspective on the treatment of atherosclerosis. J Cardiovasc Pharmacol Ther. 2018;23(6):483–93. doi: 10.1177/1074248418778548. [DOI] [PubMed] [Google Scholar]
- 30.Schelemei P, Wagner E, Picard FSR, Winkels H. Macrophage mediators and mechanisms in cardiovascular disease. FASEB J. 2024;38(2):e23424. doi: 10.1096/fj.202302001R. [DOI] [PubMed] [Google Scholar]
- 31.Shamaki GR, Safiriyu I, Kesiena O, et al. Prevalence and outcomes in STEMI patients without standard modifiable cardiovascular risk factors: A national ınpatient sample analysis. Curr Probl Cardiol. 2022;47(11):101343. doi: 10.1016/j.cpcardiol.2022.101343. [DOI] [PubMed] [Google Scholar]
- 32.Shrestha B, Shrestha DB, Sedhai YR, et al. Differences in treatment and outcomes among patients with ST-segment elevation myocardial infarction with and without standard modifiable risk factors: A systematic review and meta-analysis. Ann Med Surg (Lond) 2023;85(6):2916–23. doi: 10.1097/MS9.0000000000000738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Balasubramaniam K, Viswanathan GN, Marshall SM, Zaman AG. Increased atherothrombotic burden in patients with diabetes mellitus and acute coronary syndrome: A review of antiplatelet therapy. Cardiol Res Pract. 2012;2012:909154. doi: 10.1155/2012/909154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pei J, Wang X, Xing Z, et al. Association between admission systolic blood pressure and major adverse cardiovascular events in patients with acute myocardial infarction. PLoS One. 2020;15(6):e0234935. doi: 10.1371/journal.pone.0234935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Park JS, Cha KS, Shin D, et al. Prognostic significance of presenting blood pressure in patients with ST-elevation myocardial infarction undergoing percutaneous coronary intervention. Am J Hypertens. 2015;28(6):797–805. doi: 10.1093/ajh/hpu230. [DOI] [PubMed] [Google Scholar]
- 36.Kaplangoray M, Toprak K, Aslan R, et al. High CRP-albumin ratio is associated high thrombus burden in patients with newly diagnosed STEMI. Medicine (Baltimore) 2023;102(41):e35363. doi: 10.1097/MD.0000000000035363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Niccoli G, Menozzi A, Capodanno D, et al. Relationship between serum inflammatory biomarkers and thrombus characteristics in patients with ST segment elevation myocardial infarction. Cardiology. 2017;137(1):27–35. doi: 10.1159/000452705. [DOI] [PubMed] [Google Scholar]
- 38.Bianchini E, Lombardi M, Buonpane A, et al. Impact of thrombus aspiration on left ventricular remodeling and function in patients with ST-segment elevation myocardial infarction: A meta-analysis of randomized controlled trials. Int J Cardiol. 2024;397:131590. doi: 10.1016/j.ijcard.2023.131590. [DOI] [PubMed] [Google Scholar]
- 39.Wei TF, Zhao B, Liu PL, et al. [Impact of symptom onset to first medical contact time on the prognosis of patients with acute ST-segment elevation myocardial infarction]. Zhonghua Xin Xue Guan Bing Za Zhi. 2017;45(5):393–98. doi: 10.3760/cma.j.issn.0253-3758.2017.05.006. [in Chinese] [DOI] [PubMed] [Google Scholar]
- 40.Selçuk M, Çınar T, Şaylık F, et al. Predictive value of uric acid/albumin ratio for the prediction of new-onset atrial fibrillation in patients with ST-Elevation myocardial infarction. Rev Invest Clin. 2022;74(3):156–64. doi: 10.24875/RIC.22000072. [DOI] [PubMed] [Google Scholar]
- 41.Çınar T, Şaylık F, Hayıroğlu Mİ, et al. the association of serum uric acid/albumin ratio with no-reflow in patients with ST elevation myocardial infarction. Angiology. 2023;74(4):381–86. doi: 10.1177/00033197221110700. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.