Abstract
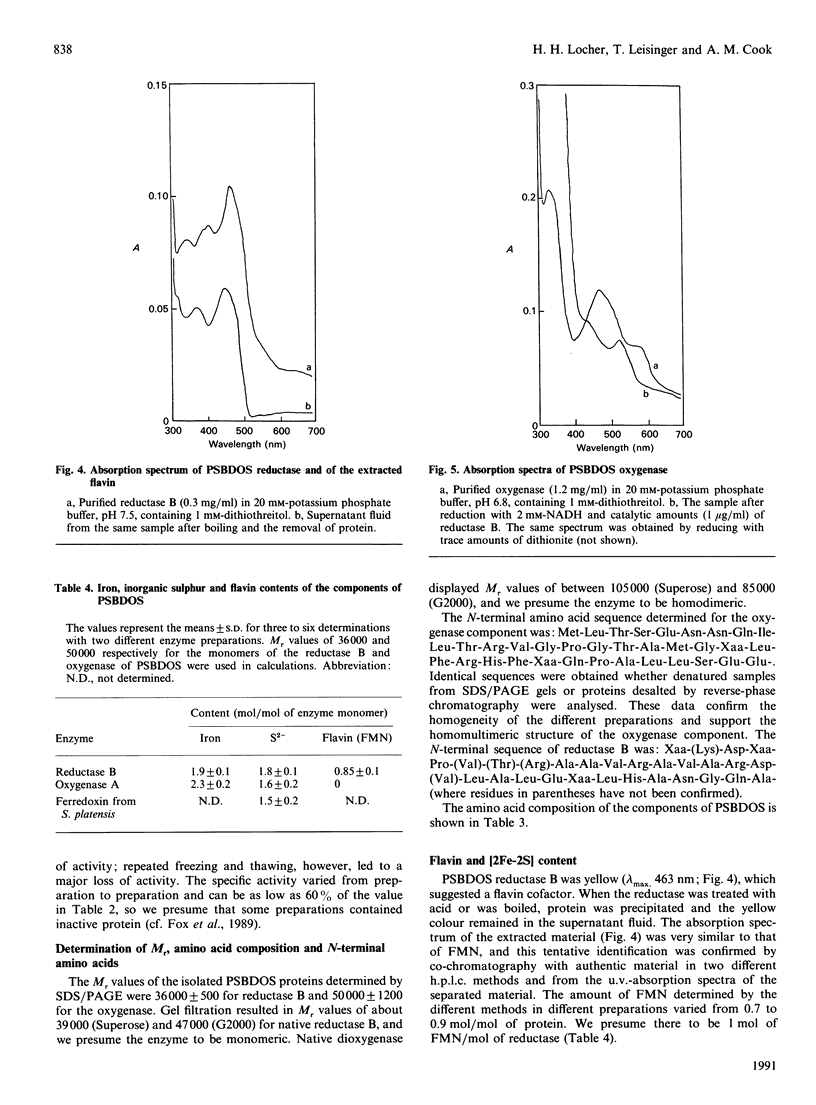
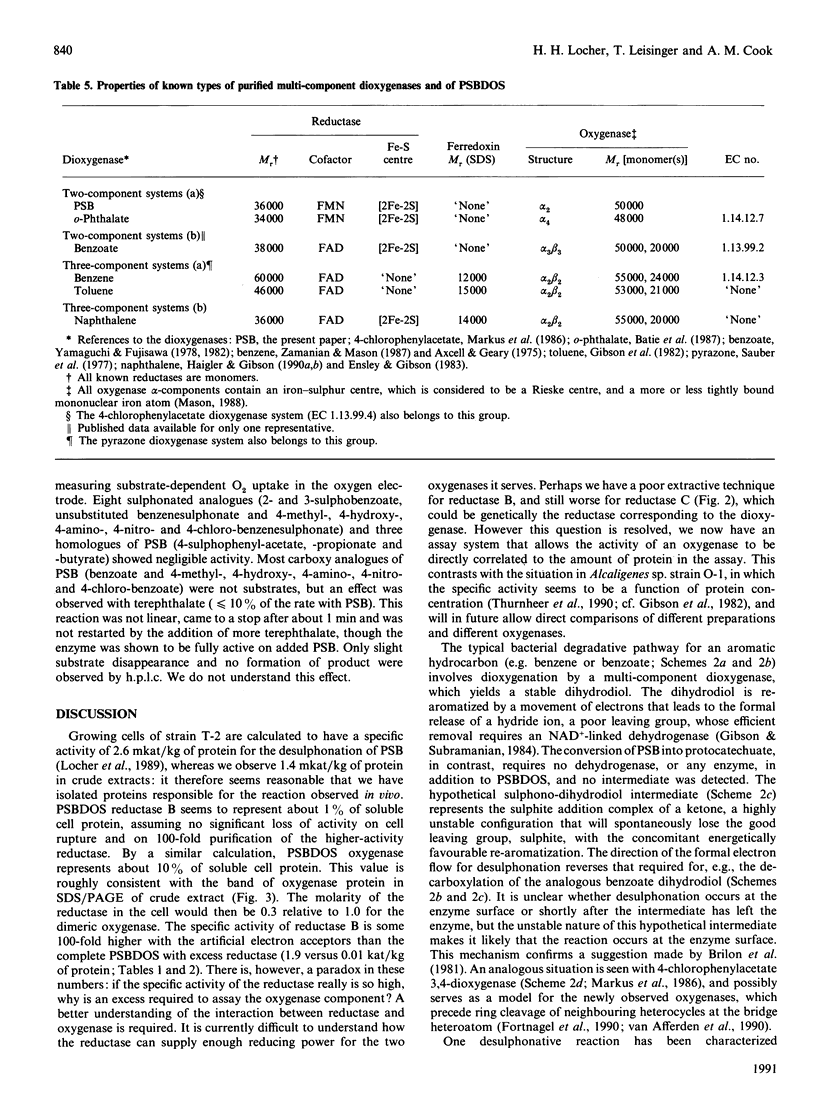
Cell-free extracts of Comamonas testosteroni T-2 grown in toluene-p-sulphonate/salts medium catalyse the conversion of p-sulphobenzoate (PSB) into protocatechuate and sulphite by an NADH-requiring and Fe2(+)-activated dioxygenase. Anion-exchange chromatography of extracts yielded red (A) and yellow (B) protein fractions, both of which were necessary for dioxygenative activity. Further purification of each fraction by hydrophobic interaction chromatography and gel filtration led to two homogeneous protein components (A and B), which together converted 1 mol each of PSB, O2 and NADH into 1 mol each of protocatechuate, sulphite and, presumably, NAD+. The system was named 4-sulphobenzoate 3,4-dioxygenase (PSB dioxygenase system). Monomeric component B (Mr 36,000) was determined to be a reductase that contained 1 mol of FMN and about 2 mol each of iron and inorganic sulphur per mol. This component transferred electrons from NADH to the oxygenase component (A) or to, e.g., cytochrome c. Homodimeric component A (subunit Mr 50,000) of the PSB dioxygenase system contained one [2Fe-2S] centre per subunit and its u.v.-visible-absorption spectrum corresponded to a Rieske-type iron-sulphur centre. The requirement for activation by iron was interpreted as partial loss of mononuclear iron during purification of component A. Component A could be reduced by dithionite or by NADH plus catalytic amounts of component B. The PSB dioxygenase system displayed a narrow substrate range: none of 18 sulphonated or non-sulphonated analogues of PSB showed significant substrate-dependent O2 uptake. The physical properties of the PSB dioxygenase system resemble those of other bacterial multi-component dioxygenase, especially phthalate dioxygenase. However, it differs from most characterized systems in its overall reaction; the product is a vicinal diphenol, and not a dihydrodiol.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Axcell B. C., Geary P. J. Purification and some properties of a soluble benzene-oxidizing system from a strain of Pseudomonas. Biochem J. 1975 Jan;146(1):173–183. doi: 10.1042/bj1460173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Batie C. J., LaHaie E., Ballou D. P. Purification and characterization of phthalate oxygenase and phthalate oxygenase reductase from Pseudomonas cepacia. J Biol Chem. 1987 Feb 5;262(4):1510–1518. [PubMed] [Google Scholar]
- Beinert H. Semi-micro methods for analysis of labile sulfide and of labile sulfide plus sulfane sulfur in unusually stable iron-sulfur proteins. Anal Biochem. 1983 Jun;131(2):373–378. doi: 10.1016/0003-2697(83)90186-0. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Brilon C., Beckmann W., Knackmuss H. J. Catabolism of Naphthalenesulfonic Acids by Pseudomonas sp. A3 and Pseudomonas sp. C22. Appl Environ Microbiol. 1981 Jul;42(1):44–55. doi: 10.1128/aem.42.1.44-55.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buder R., Fuchs G. 2-Aminobenzoyl-CoA monooxygenase/reductase, a novel type of flavoenzyme. Purification and some properties of the enzyme. Eur J Biochem. 1989 Nov 20;185(3):629–635. doi: 10.1111/j.1432-1033.1989.tb15159.x. [DOI] [PubMed] [Google Scholar]
- Cain R. B., Farr D. R. Metabolism of arylsulphonates by micro-organisms. Biochem J. 1968 Feb;106(4):859–877. doi: 10.1042/bj1060859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Endo K., Kondo H., Ishimoto M. Degradation of benzenesulfonate to sulfite in bacterial extract. J Biochem. 1977 Nov;82(5):1397–1402. doi: 10.1093/oxfordjournals.jbchem.a131827. [DOI] [PubMed] [Google Scholar]
- Ensley B. D., Gibson D. T., Laborde A. L. Oxidation of naphthalene by a multicomponent enzyme system from Pseudomonas sp. strain NCIB 9816. J Bacteriol. 1982 Mar;149(3):948–954. doi: 10.1128/jb.149.3.948-954.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ensley B. D., Gibson D. T. Naphthalene dioxygenase: purification and properties of a terminal oxygenase component. J Bacteriol. 1983 Aug;155(2):505–511. doi: 10.1128/jb.155.2.505-511.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fortnagel P., Harms H., Wittich R. M., Krohn S., Meyer H., Sinnwell V., Wilkes H., Francke W. Metabolism of Dibenzofuran by Pseudomonas sp. Strain HH69 and the Mixed Culture HH27. Appl Environ Microbiol. 1990 Apr;56(4):1148–1156. doi: 10.1128/aem.56.4.1148-1156.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox B. G., Froland W. A., Dege J. E., Lipscomb J. D. Methane monooxygenase from Methylosinus trichosporium OB3b. Purification and properties of a three-component system with high specific activity from a type II methanotroph. J Biol Chem. 1989 Jun 15;264(17):10023–10033. [PubMed] [Google Scholar]
- Haigler B. E., Gibson D. T. Purification and properties of NADH-ferredoxinNAP reductase, a component of naphthalene dioxygenase from Pseudomonas sp. strain NCIB 9816. J Bacteriol. 1990 Jan;172(1):457–464. doi: 10.1128/jb.172.1.457-464.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haigler B. E., Gibson D. T. Purification and properties of ferredoxinNAP, a component of naphthalene dioxygenase from Pseudomonas sp. strain NCIB 9816. J Bacteriol. 1990 Jan;172(1):465–468. doi: 10.1128/jb.172.1.465-468.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kondo H., Ishimoto M. Enzymatic formation of sulfite and acetate from sulfoacetaldehyde, a degradation product of taurine. J Biochem. 1972 Aug;72(2):487–489. doi: 10.1093/oxfordjournals.jbchem.a129926. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Locher H. H., Leisinger T., Cook A. M. Degradation of p-toluenesulphonic acid via sidechain oxidation, desulphonation and meta ring cleavage in Pseudomonas (Comamonas) testosteroni T-2. J Gen Microbiol. 1989 Jul;135(7):1969–1978. doi: 10.1099/00221287-135-7-1969. [DOI] [PubMed] [Google Scholar]
- Markus A., Krekel D., Lingens F. Purification and some properties of component A of the 4-chlorophenylacetate 3,4-dioxygenase from Pseudomonas species strain CBS. J Biol Chem. 1986 Sep 25;261(27):12883–12888. [PubMed] [Google Scholar]
- Merril C. R., Goldman D., Van Keuren M. L. Silver staining methods for polyacrylamide gel electrophoresis. Methods Enzymol. 1983;96:230–239. doi: 10.1016/s0076-6879(83)96021-4. [DOI] [PubMed] [Google Scholar]
- Reineke W., Knackmuss H. J. Microbial degradation of haloaromatics. Annu Rev Microbiol. 1988;42:263–287. doi: 10.1146/annurev.mi.42.100188.001403. [DOI] [PubMed] [Google Scholar]
- Sauber K., Fröhner C., Rosenberg G., Eberspächer J., Lingens F. Purification and properties of pyrazon dioxygenase from pyrazon-degrading bacteria. Eur J Biochem. 1977 Mar 15;74(1):89–97. doi: 10.1111/j.1432-1033.1977.tb11370.x. [DOI] [PubMed] [Google Scholar]
- Scholtz R., Leisinger T., Suter F., Cook A. M. Characterization of 1-chlorohexane halidohydrolase, a dehalogenase of wide substrate range from an Arthrobacter sp. J Bacteriol. 1987 Nov;169(11):5016–5021. doi: 10.1128/jb.169.11.5016-5021.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimamoto G., Berk R. S. Catabolism of taurine in Pseudomonas aeruginosa. Biochim Biophys Acta. 1979 Aug 15;569(2):287–292. doi: 10.1016/0005-2744(79)90064-0. [DOI] [PubMed] [Google Scholar]
- Shimamoto G., Berk R. S. Taurine catabolism. II. biochemical and genetic evidence for sulfoacetaldehyde sulfo-lyase involvement. Biochim Biophys Acta. 1980 Sep 17;632(1):121–130. doi: 10.1016/0304-4165(80)90255-x. [DOI] [PubMed] [Google Scholar]
- Suhara K., Takemori S., Katagiri M., Wada K., Kobayashi H. Estimation of labile sulfide in iron-sulfur proteins. Anal Biochem. 1975 Oct;68(2):632–636. doi: 10.1016/0003-2697(75)90659-4. [DOI] [PubMed] [Google Scholar]
- Thurnheer T., Zürrer D., Höglinger O., Leisinger T., Cook A. M. Initial steps in the degradation of benzene sulfonic acid, 4-toluene sulfonic acids, and orthanilic acid in Alcaligenes sp. strain O-1. Biodegradation. 1990;1(1):55–64. doi: 10.1007/BF00117051. [DOI] [PubMed] [Google Scholar]
- Thysse G. J., Wanders T. H. Initial steps in the degradation of n-alkane-1-sulphonates by Pseudomonas. Antonie Van Leeuwenhoek. 1974;40(1):25–37. doi: 10.1007/BF00394550. [DOI] [PubMed] [Google Scholar]
- Yamaguchi M., Fujisawa H. Characterization of NADH-cytochrome c reductase, a component of benzoate 1,2-dioxygenase system from Pseudomonas arvilla c-1. J Biol Chem. 1978 Dec 25;253(24):8848–8853. [PubMed] [Google Scholar]
- Yamaguchi M., Fujisawa H. Subunit structure of oxygenase component in benzoate-1,2-dioxygenase system from Pseudomonas arvilla C-1. J Biol Chem. 1982 Nov 10;257(21):12497–12502. [PubMed] [Google Scholar]
- Zamanian M., Mason J. R. Benzene dioxygenase in Pseudomonas putida. Subunit composition and immuno-cross-reactivity with other aromatic dioxygenases. Biochem J. 1987 Jun 15;244(3):611–616. doi: 10.1042/bj2440611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zylstra G. J., Gibson D. T. Toluene degradation by Pseudomonas putida F1. Nucleotide sequence of the todC1C2BADE genes and their expression in Escherichia coli. J Biol Chem. 1989 Sep 5;264(25):14940–14946. [PubMed] [Google Scholar]
- Zürrer D., Cook A. M., Leisinger T. Microbial desulfonation of substituted naphthalenesulfonic acids and benzenesulfonic acids. Appl Environ Microbiol. 1987 Jul;53(7):1459–1463. doi: 10.1128/aem.53.7.1459-1463.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]