Abstract
The high-Mr alkali-stable form of cathepsin B was purified from purulent human sputum. It was shown to solubilize proteoglycan monomer entrapped in polyacrylamide at a rate comparable with that of human lysosomal cathepsin B. Like the enzyme from lysosomes, sputum cathepsin B was bound by human alpha 2-macroglobulin, which inhibited its action on proteoglycan. Cystatin C in purulent sputum was shown to be the N-terminally truncated form generated by neutrophil elastase cleavage, and sputum cathepsin B was only weakly inhibited by recombinant cystatin C that had been cleaved by neutrophil elastase in vitro. Addition of neutrophil elastase to mucoid sputum led to a 5-fold increase in cathepsin B activity concomitant with a lowering in Mr of the cysteine proteinase from 40,000 to 37,000, i.e. the size of the active enzyme purified from purulent sputum. It is concluded that the high-Mr form of cathepsin B present in purulent sputum is a functional proteinase, unlike similar forms of the enzyme secreted by mammary gland in organ culture. The activity of cathepsin B in sputum is modulated by neutrophil elastase, by a combination of inhibitor inactivation and zymogen activation.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abrahamson M., Barrett A. J., Salvesen G., Grubb A. Isolation of six cysteine proteinase inhibitors from human urine. Their physicochemical and enzyme kinetic properties and concentrations in biological fluids. J Biol Chem. 1986 Aug 25;261(24):11282–11289. [PubMed] [Google Scholar]
- Abrahamson M., Dalbøge H., Olafsson I., Carlsen S., Grubb A. Efficient production of native, biologically active human cystatin C by Escherichia coli. FEBS Lett. 1988 Aug 15;236(1):14–18. doi: 10.1016/0014-5793(88)80276-x. [DOI] [PubMed] [Google Scholar]
- Abrahamson M., Mason R. W., Hansson H., Buttle D. J., Grubb A., Ohlsson K. Human cystatin C. role of the N-terminal segment in the inhibition of human cysteine proteinases and in its inactivation by leucocyte elastase. Biochem J. 1991 Feb 1;273(Pt 3):621–626. doi: 10.1042/bj2730621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anastasi A., Brown M. A., Kembhavi A. A., Nicklin M. J., Sayers C. A., Sunter D. C., Barrett A. J. Cystatin, a protein inhibitor of cysteine proteinases. Improved purification from egg white, characterization, and detection in chicken serum. Biochem J. 1983 Apr 1;211(1):129–138. doi: 10.1042/bj2110129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrett A. J., Brown M. A., Sayers C. A. The electrophoretically 'slow' and 'fast' forms of the alpha 2-macroglobulin molecule. Biochem J. 1979 Aug 1;181(2):401–418. doi: 10.1042/bj1810401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrett A. J., Kirschke H. Cathepsin B, Cathepsin H, and cathepsin L. Methods Enzymol. 1981;80(Pt 100):535–561. doi: 10.1016/s0076-6879(81)80043-2. [DOI] [PubMed] [Google Scholar]
- Barrett A. J., Starkey P. M. The interaction of alpha 2-macroglobulin with proteinases. Characteristics and specificity of the reaction, and a hypothesis concerning its molecular mechanism. Biochem J. 1973 Aug;133(4):709–724. doi: 10.1042/bj1330709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnett D., Crocker J., Stockley R. A. Cathepsin B-like cysteine proteinase activity in sputum and immunohistologic identification of cathepsin B in alveolar macrophages. Am Rev Respir Dis. 1983 Nov;128(5):915–919. doi: 10.1164/arrd.1983.128.5.915. [DOI] [PubMed] [Google Scholar]
- Buttle D. J., Bonner B. C., Burnett D., Barrett A. J. A catalytically active high-Mr form of human cathepsin B from sputum. Biochem J. 1988 Sep 15;254(3):693–699. doi: 10.1042/bj2540693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buttle D. J., Burnett D., Abrahamson M. Levels of neutrophil elastase and cathepsin B activities, and cystatins in human sputum: relationship to inflammation. Scand J Clin Lab Invest. 1990 Sep;50(5):509–516. doi: 10.1080/00365519009089165. [DOI] [PubMed] [Google Scholar]
- Farndale R. W., Buttle D. J., Barrett A. J. Improved quantitation and discrimination of sulphated glycosaminoglycans by use of dimethylmethylene blue. Biochim Biophys Acta. 1986 Sep 4;883(2):173–177. doi: 10.1016/0304-4165(86)90306-5. [DOI] [PubMed] [Google Scholar]
- Gauthier F., Fryksmark U., Ohlsson K., Bieth J. G. Kinetics of the inhibition of leukocyte elastase by the bronchial inhibitor. Biochim Biophys Acta. 1982 Jan 18;700(2):178–183. doi: 10.1016/0167-4838(82)90095-4. [DOI] [PubMed] [Google Scholar]
- Grubb A., Löfberg H. Human gamma-trace, a basic microprotein: amino acid sequence and presence in the adenohypophysis. Proc Natl Acad Sci U S A. 1982 May;79(9):3024–3027. doi: 10.1073/pnas.79.9.3024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeppsson J. O., Laurell C. B., Franzén B. Agarose gel electrophoresis. Clin Chem. 1979 Apr;25(4):629–638. [PubMed] [Google Scholar]
- Kurecki T., Kress L. F., Laskowski M., Sr Purification of human plasma alpha 2 macroglobulin and alpha 1 proteinase inhibitor using zinc chelate chromatography. Anal Biochem. 1979 Nov 1;99(2):415–420. doi: 10.1016/s0003-2697(79)80026-3. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Linden M., Håkansson L., Ohlsson K., Sjödin K., Tegner H., Tunek A., Venge P. Glutathione in bronchoalveolar lavage fluid from smokers is related to humoral markers of inflammatory cell activity. Inflammation. 1989 Dec;13(6):651–658. doi: 10.1007/BF00914309. [DOI] [PubMed] [Google Scholar]
- Martodam R. R., Baugh R. J., Twumasi D. Y., Liener I. E. A rapid procedure for the large scale purification of elastase and cathepsin G from human sputum. Prep Biochem. 1979;9(1):15–31. doi: 10.1080/00327487908061669. [DOI] [PubMed] [Google Scholar]
- Mason R. W. Interaction of lysosomal cysteine proteinases with alpha 2-macroglobulin: conclusive evidence for the endopeptidase activities of cathepsins B and H. Arch Biochem Biophys. 1989 Sep;273(2):367–374. doi: 10.1016/0003-9861(89)90495-5. [DOI] [PubMed] [Google Scholar]
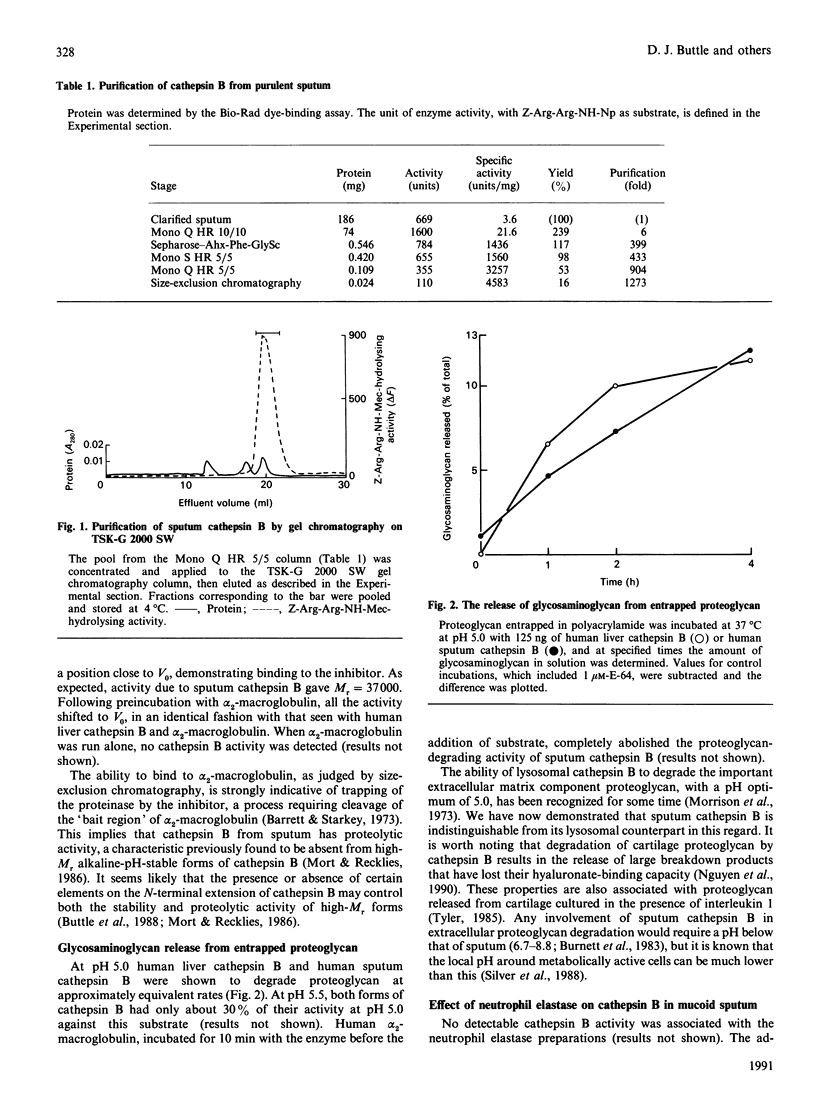
- Morrison R. I., Barrett A. J., Dingle J. T., Prior D. Cathepsins BI and D. Action on human cartilage proteoglycans. Biochim Biophys Acta. 1973 Apr 12;302(2):411–419. doi: 10.1016/0005-2744(73)90170-8. [DOI] [PubMed] [Google Scholar]
- Mort J. S., Recklies A. D. Interrelationship of active and latent secreted human cathepsin B precursors. Biochem J. 1986 Jan 1;233(1):57–63. doi: 10.1042/bj2330057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mort J. S., Recklies A. D., Poole A. R. Characterization of a thiol proteinase secreted by malignant human breast tumours. Biochim Biophys Acta. 1980 Jul 10;614(1):134–143. doi: 10.1016/0005-2744(80)90174-6. [DOI] [PubMed] [Google Scholar]
- Nagase H., Woessner J. F., Jr An improved assay for proteases and polysaccharidases employing a cartilage proteoglycan substrate entrapped in polyacrylamide particles. Anal Biochem. 1980 Sep 15;107(2):385–392. doi: 10.1016/0003-2697(80)90400-5. [DOI] [PubMed] [Google Scholar]
- Nguyen Q., Mort J. S., Roughley P. J. Cartilage proteoglycan aggregate is degraded more extensively by cathepsin L than by cathepsin B. Biochem J. 1990 Mar 1;266(2):569–573. [PMC free article] [PubMed] [Google Scholar]
- Olafsson I., Gudmundsson G., Abrahamson M., Jensson O., Grubb A. The amino terminal portion of cerebrospinal fluid cystatin C in hereditary cystatin C amyloid angiopathy is not truncated: direct sequence analysis from agarose gel electropherograms. Scand J Clin Lab Invest. 1990 Feb;50(1):85–93. doi: 10.1080/00365519009091569. [DOI] [PubMed] [Google Scholar]
- Recklies A. D., Mort J. S. Characterization of a cysteine proteinase secreted by mouse mammary gland. Cancer Res. 1985 May;45(5):2302–2307. [PubMed] [Google Scholar]
- Recklies A. D., Mort J. S. Rat mammary gland in culture secretes a stable high molecular weight form of cathepsin L. Biochem Biophys Res Commun. 1985 Aug 30;131(1):402–407. doi: 10.1016/0006-291x(85)91816-9. [DOI] [PubMed] [Google Scholar]
- Rich D. H., Brown M. A., Barrett A. J. Purification of cathepsin B by a new form of affinity chromatography. Biochem J. 1986 May 1;235(3):731–734. doi: 10.1042/bj2350731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SCHEIDEGGER J. J. Une micro-méthode de l'immuno-electrophorèse. Int Arch Allergy Appl Immunol. 1955;7(2):103–110. [PubMed] [Google Scholar]
- Silver I. A., Murrills R. J., Etherington D. J. Microelectrode studies on the acid microenvironment beneath adherent macrophages and osteoclasts. Exp Cell Res. 1988 Apr;175(2):266–276. doi: 10.1016/0014-4827(88)90191-7. [DOI] [PubMed] [Google Scholar]
- Tyler J. A. Chondrocyte-mediated depletion of articular cartilage proteoglycans in vitro. Biochem J. 1985 Jan 15;225(2):493–507. doi: 10.1042/bj2250493. [DOI] [PMC free article] [PubMed] [Google Scholar]