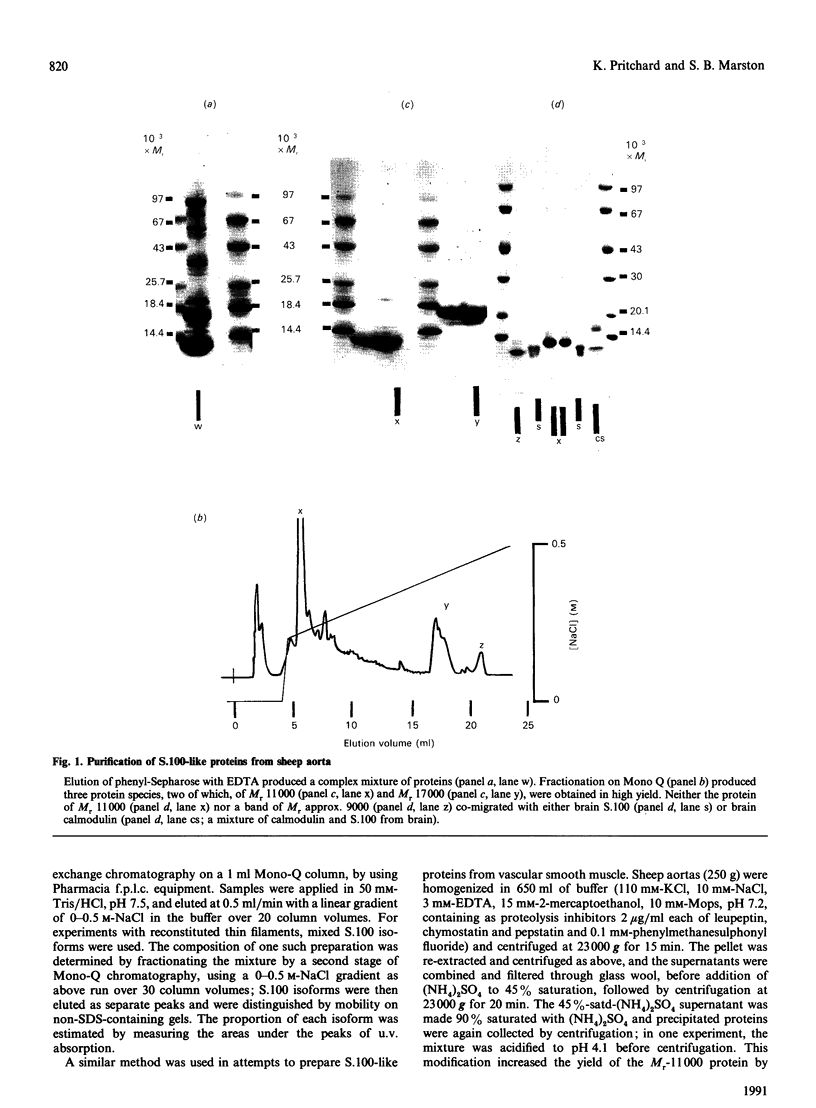
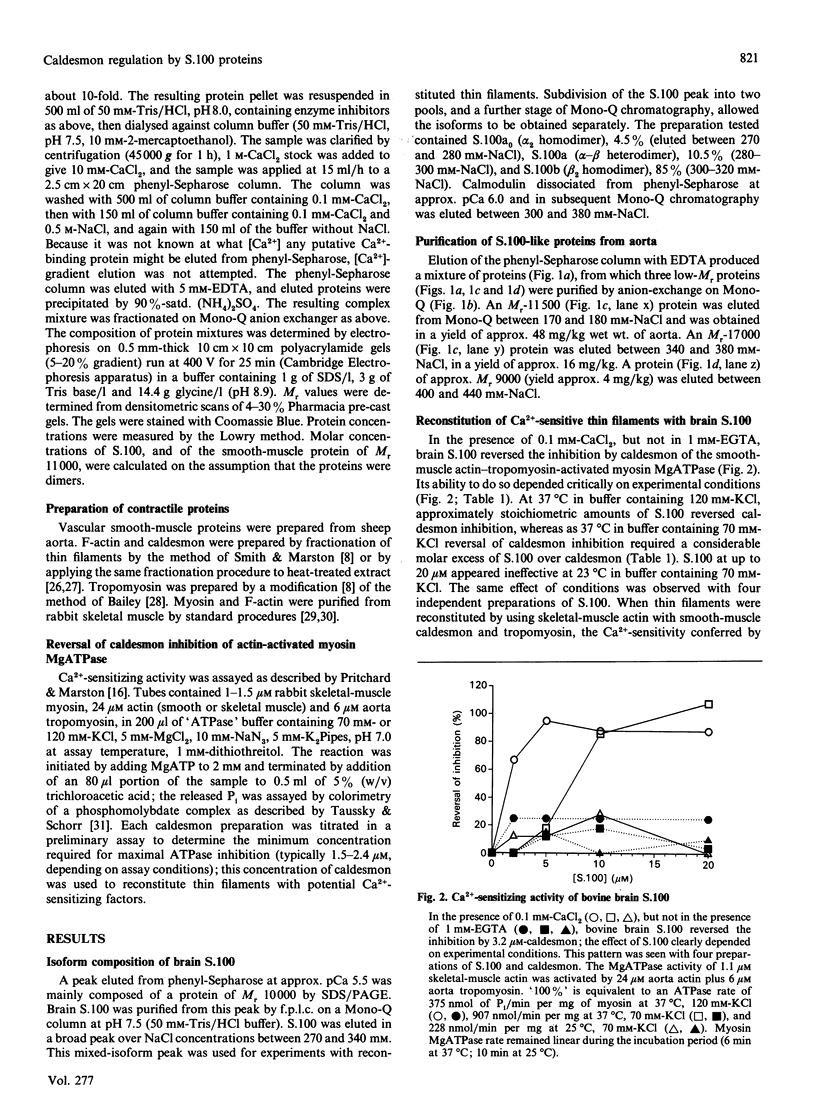
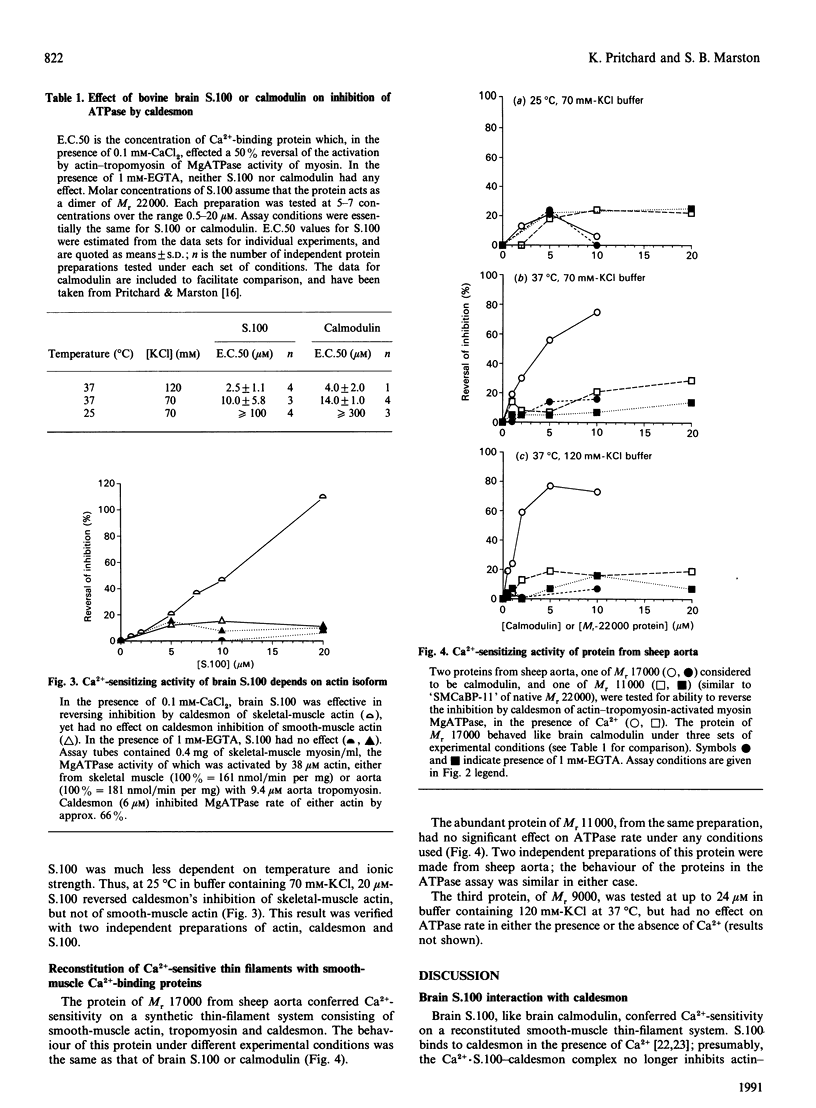
Abstract
1. We have investigated the ability of bovine brain S.100, and of three related proteins from sheep aorta smooth muscle, to confer Ca(2+)-sensitivity on thin filaments reconstituted from smooth-muscle actin, tropomyosin and caldesmon. 2. At 37 degrees C in pH 7.0 buffer containing 120 mM-KCl, approximately stoichiometric amounts of S.100 reversed caldesmon's inhibition of the activation of myosin MgATPase by smooth-muscle actin-tropomyosin. The [S.100] which reversed by 50% the inhibition by caldesmon (the E.C.50) was 2.5 microM when [caldesmon] = 2-3 microM in the assay mixture. When [KCl] was decreased to 70 mM, E.C.50 = 11.5 microM; at 25 degrees C in 70 mM-KCl, up to 20 microM-S.100 had no effect. When skeletal-muscle actin rather than smooth-muscle actin was used to reconstitute thin filaments, 20 microM-S.100 did reverse inhibition by caldesmon, at 25 degrees C in buffer containing 70 mM-KCl. This dependence on conditions is also characteristic of the calmodulin-caldesmon interaction. 3. These results suggested that S.100 or a related protein might interact with caldesmon in smooth muscle. We therefore attempted to prepare such a protein from sheep aorta. Three proteins were purified: an Mr-17,000 protein (yield 16 mg/kg), an abundant Mr-11,000 protein (yield 48 mg/kg), and an Mr-9000 protein (yield 4 mg/kg). Neither of the last two low-Mr proteins had any effect on activation of myosin MgATPase by reconstituted thin filaments. The protein of Mr 17,000 had Ca(2+)-sensitizing activity, and behaved exactly like brain calmodulin in the assay system.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barraclough R., Savin J., Dube S. K., Rudland P. S. Molecular cloning and sequence of the gene for p9Ka. A cultured myoepithelial cell protein with strong homology to S-100, a calcium-binding protein. J Mol Biol. 1987 Nov 5;198(1):13–20. doi: 10.1016/0022-2836(87)90453-0. [DOI] [PubMed] [Google Scholar]
- Bretscher A. Smooth muscle caldesmon. Rapid purification and F-actin cross-linking properties. J Biol Chem. 1984 Oct 25;259(20):12873–12880. [PubMed] [Google Scholar]
- Dabrowska R., Aromatorio D., Sherry J. M., Hartshorne D. J. Composition of the myosin light chain kinase from chicken gizzard. Biochem Biophys Res Commun. 1977 Oct 24;78(4):1263–1272. doi: 10.1016/0006-291x(77)91429-2. [DOI] [PubMed] [Google Scholar]
- Dabrowska R., Goch A., Gałazkiewicz B., Osińska H. The influence of caldesmon on ATPase activity of the skeletal muscle actomyosin and bundling of actin filaments. Biochim Biophys Acta. 1985 Sep 27;842(1):70–75. doi: 10.1016/0304-4165(85)90295-8. [DOI] [PubMed] [Google Scholar]
- Donato R. S-100 proteins. Cell Calcium. 1986 Jun;7(3):123–145. doi: 10.1016/0143-4160(86)90017-5. [DOI] [PubMed] [Google Scholar]
- Ebashi S. The Croonian lecture, 1979: Regulation of muscle contraction. Proc R Soc Lond B Biol Sci. 1980 Mar 21;207(1168):259–286. doi: 10.1098/rspb.1980.0024. [DOI] [PubMed] [Google Scholar]
- Fujii T., Machino K., Andoh H., Satoh T., Kondo Y. Calcium-dependent control of caldesmon-actin interaction by S100 protein. J Biochem. 1990 Jan;107(1):133–137. doi: 10.1093/oxfordjournals.jbchem.a122996. [DOI] [PubMed] [Google Scholar]
- Gopalakrishna R., Anderson W. B. Ca2+-induced hydrophobic site on calmodulin: application for purification of calmodulin by phenyl-Sepharose affinity chromatography. Biochem Biophys Res Commun. 1982 Jan 29;104(2):830–836. doi: 10.1016/0006-291x(82)90712-4. [DOI] [PubMed] [Google Scholar]
- Grand R. J., Perry S. V., Weeks R. A. Troponin C-like proteins (calmodulins) from mammalian smooth muscle and other tissues. Biochem J. 1979 Feb 1;177(2):521–529. doi: 10.1042/bj1770521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutteridge J. M., Winyard P. G., Blake D. R., Lunec J., Brailsford S., Halliwell B. The behaviour of caeruloplasmin in stored human extracellular fluids in relation to ferroxidase II activity, lipid peroxidation and phenanthroline-detectable copper. Biochem J. 1985 Sep 1;230(2):517–523. doi: 10.1042/bj2300517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kallfelz F. A., Taylor A. N., Wasserman R. H. Vitamin D-induced calcium binding factor in rat intestinal mucosa. Proc Soc Exp Biol Med. 1967 May;125(1):54–58. doi: 10.3181/00379727-125-32011. [DOI] [PubMed] [Google Scholar]
- Kligman D., Hilt D. C. The S100 protein family. Trends Biochem Sci. 1988 Nov;13(11):437–443. doi: 10.1016/0968-0004(88)90218-6. [DOI] [PubMed] [Google Scholar]
- Kuźnicki J., Filipek A., Hunziker P. E., Huber S., Heizmann C. W. Calcium-binding protein from mouse Ehrlich ascites-tumour cells is homologous to human calcyclin. Biochem J. 1989 Nov 1;263(3):951–956. doi: 10.1042/bj2630951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehman W., Szent-Györgyi A. G. Regulation of muscular contraction. Distribution of actin control and myosin control in the animal kingdom. J Gen Physiol. 1975 Jul;66(1):1–30. doi: 10.1085/jgp.66.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacManus J. P., Watson D. C., Yaguchi M. The complete amino acid sequence of oncomodulin--a parvalbumin-like calcium-binding protein from Morris hepatoma 5123tc. Eur J Biochem. 1983 Oct 17;136(1):9–17. doi: 10.1111/j.1432-1033.1983.tb07698.x. [DOI] [PubMed] [Google Scholar]
- Mani R. S., Kay C. M. Isolation and characterization of a novel molecular weight 11,000 Ca2(+)-binding protein from smooth muscle. Biochemistry. 1990 Feb 13;29(6):1398–1404. doi: 10.1021/bi00458a009. [DOI] [PubMed] [Google Scholar]
- Marston S. B., Lehman W. Caldesmon is a Ca2+-regulatory component of native smooth-muscle thin filaments. Biochem J. 1985 Nov 1;231(3):517–522. doi: 10.1042/bj2310517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marston S. B., Redwood C. S., Lehman W. Reversal of caldesmon function by anti-caldesmon antibodies confirms its role in the calcium regulation of vascular smooth muscle thin filaments. Biochem Biophys Res Commun. 1988 Aug 30;155(1):197–202. doi: 10.1016/s0006-291x(88)81068-4. [DOI] [PubMed] [Google Scholar]
- Marston S. B., Smith C. W. The thin filaments of smooth muscles. J Muscle Res Cell Motil. 1985 Dec;6(6):669–708. doi: 10.1007/BF00712237. [DOI] [PubMed] [Google Scholar]
- Marston S. B., Trevett R. M., Walters M. Calcium ion-regulated thin filaments from vascular smooth muscle. Biochem J. 1980 Feb 1;185(2):355–365. doi: 10.1042/bj1850355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marston S., Pritchard K., Redwood C., Taggart M. Ca2+ regulation of the thin filaments: biochemical mechanism and physiological role. Biochem Soc Trans. 1988 Aug;16(4):494–497. doi: 10.1042/bst0160494. [DOI] [PubMed] [Google Scholar]
- Marston S. Stoichiometry and stability of caldesmon in native thin filaments from sheep aorta smooth muscle. Biochem J. 1990 Dec 1;272(2):305–310. doi: 10.1042/bj2720305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masure H. R., Head J. F., Tice H. M. Studies on the alpha-subunit of bovine brain S-100 protein. Biochem J. 1984 Mar 15;218(3):691–696. doi: 10.1042/bj2180691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore P. B., Kraus-Friedmann N., Dedman J. R. Unique calcium-dependent hydrophobic binding proteins: possible independent mediators of intracellular calcium distinct from calmodulin. J Cell Sci. 1984 Dec;72:121–133. doi: 10.1242/jcs.72.1.121. [DOI] [PubMed] [Google Scholar]
- Pritchard K., Marston S. B. Ca2+-calmodulin binding to caldesmon and the caldesmon-actin-tropomyosin complex. Its role in Ca2+ regulation of the activity of synthetic smooth-muscle thin filaments. Biochem J. 1989 Feb 1;257(3):839–843. doi: 10.1042/bj2570839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rüegg J. C., Pfitzer G., Zimmer M., Hofmann F. The calmodulin fraction responsible for contraction in an intestinal smooth muscle. FEBS Lett. 1984 May 21;170(2):383–386. doi: 10.1016/0014-5793(84)81349-6. [DOI] [PubMed] [Google Scholar]
- Shirinsky V. P., Bushueva T. L., Frolova S. I. Caldesmon-calmodulin interaction. Study by the method of protein intrinsic tryptophan fluorescence. Biochem J. 1988 Oct 1;255(1):203–208. [PMC free article] [PubMed] [Google Scholar]
- Skripnikova E. V., Gusev N. B. Interaction of smooth muscle caldesmon with S-100 protein. FEBS Lett. 1989 Nov 6;257(2):380–382. doi: 10.1016/0014-5793(89)81577-7. [DOI] [PubMed] [Google Scholar]
- Smith C. W., Marston S. B. Disassembly and reconstitution of the Ca2+-sensitive thin filaments of vascular smooth muscle. FEBS Lett. 1985 May 6;184(1):115–119. doi: 10.1016/0014-5793(85)80665-7. [DOI] [PubMed] [Google Scholar]
- Smith C. W., Pritchard K., Marston S. B. The mechanism of Ca2+ regulation of vascular smooth muscle thin filaments by caldesmon and calmodulin. J Biol Chem. 1987 Jan 5;262(1):116–122. [PubMed] [Google Scholar]
- Sobue K., Muramoto Y., Fujita M., Kakiuchi S. Purification of a calmodulin-binding protein from chicken gizzard that interacts with F-actin. Proc Natl Acad Sci U S A. 1981 Sep;78(9):5652–5655. doi: 10.1073/pnas.78.9.5652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spudich J. A., Watt S. The regulation of rabbit skeletal muscle contraction. I. Biochemical studies of the interaction of the tropomyosin-troponin complex with actin and the proteolytic fragments of myosin. J Biol Chem. 1971 Aug 10;246(15):4866–4871. [PubMed] [Google Scholar]
- TAUSSKY H. H., SHORR E. A microcolorimetric method for the determination of inorganic phosphorus. J Biol Chem. 1953 Jun;202(2):675–685. [PubMed] [Google Scholar]
- Taggart M. J., Marston S. B. The effects of vascular smooth muscle caldesmon on force production by 'desensitised' skeletal muscle fibres. FEBS Lett. 1988 Dec 19;242(1):171–174. doi: 10.1016/0014-5793(88)81009-3. [DOI] [PubMed] [Google Scholar]
- Zimmer D. B., Van Eldik L. J. Tissue distribution of rat S100 alpha and S100 beta and S100-binding proteins. Am J Physiol. 1987 Mar;252(3 Pt 1):C285–C289. doi: 10.1152/ajpcell.1987.252.3.C285. [DOI] [PubMed] [Google Scholar]