Abstract
Repeated low-dose exposure to carbofuran exerts its neurotoxic effects by non-cholinergic mechanisms. Emerging evidence indicates that oxidative stress plays an important role in carbofuran neurotoxicity after sub-chronic exposure. The purpose of the present study is to evaluate the role of mitochondrial oxidative stress and dysfunction as a primary event responsible for neurotoxic effects observed after sub-chronic carbofuran exposure. Carbofuran was administered to rats at a dose of 1 mg/kg orally for a period of 28 days. There was a significant inhibition in the activity of acetylcholinesterase (66.6%) in brain samples after 28 days of carbofuran exposure. Mitochondrial respiratory chain functions were assessed in terms of MTT (3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide) reduction and activity of succinate dehydrogenase in isolated mitochondria. It was observed that carbofuran exposure significantly inhibited MTT reduction (31%) and succinate dehydrogenase activity (57%). This was accompanied by decrease in low-molecular weight thiols (66.6%) and total thiols (37.4%) and an increase in lipid peroxidation (43.7%) in the mitochondria isolated from carbofuran-exposed rat brain. The changes in mitochondrial oxidative stress and functions were associated with impaired cognitive and motor functions in the animals exposed to carbofuran as compared to the control animals. Based on these results, it is clear that carbofuran exerts its neurotoxicity by impairing mitochondrial functions leading to oxidative stress and neurobehavioral deficits.
Keywords: Cerebral cortex, Carbofuran, Oxidative stress, Mitochondrial dysfunction, Behavior
Introduction
Carbofuran (2,3-Dihydro-2,2-dimethyl-7-benzofuranol N-methylcarbamate) is a highly toxic carbamate pesticide that kills insects, mites, and nematodes on contact and after ingestion. The acute neurobehavioral effects of carbamate insecticides are primarily due to overstimulation of the cholinergic system as a result of central and peripheral cholinesterase (ChE) inhibition (Moretto 1998). Sub-chronic carbofuran exposure produces many biochemical/neurochemical effects, which are unrelated to its ability to inhibit acetylcholinesterase (Gupta 2004). Carbofuran has been reported to inhibit serine-containing esterases, alter activity of enzymes like creatine kinase and dehydrogenases, deplete high-energy phosphates (Gupta et al. 1991), deplete intracellular glutathione (GSH) levels (Barlow et al. 2005), and increased generation of oxygen and nitrogen free radicals (Gupta and Goad 2000). In addition, carbofuran administration has been shown to cause significant changes in neurotransmitter concentration viz. gamma-amino butyric acid (GABA), epinephrine, norepinephrine, and dopamine (Farage-Elawar and Blaker 1992).
Increasing evidence points to the central role of oxidative stress in pathophysiology of neurodegenerative diseases (Halliwell 2006). Carbofuran has been reported to disturb pro-oxidant/antioxidant balance in brain leading to oxidative stress (Gupta et al. 2007; Kamboj et al. 2006; Rai and Sharma 2007). Mitochondria are the major cellular sources of reactive oxygen species (ROS) and key contributors to neurodegenerative disorders (Leuner et al. 2007). Almost all intracellular ATP is known to be generated in the mitochondria and about one-third of the cellular adenine nucleotides are located in this organelle (Papa 1996); therefore, chemicals causing mitochondrial dysfunction may deplete ATP, leading to excessive generation of ROS (Tsujimoto 1997). Liu et al. (2003) have reported that accumulation of oxidative damage to mitochondria in brain may lead to neuronal and cognitive dysfunction. Recent studies have shown that mitochondrial-formed oxidants are mediator of molecular signaling and have implicated in mitochondrial-dependent apoptosis (Gogvadze and Zhivotovsky 2007). In the present investigation, we analyzed the role of mitochondrial dysfunction and oxidative stress in causing carbofuran neurotoxicity following sub-chronic exposure and to correlate the changes at the biochemical level with the behavioral changes.
Materials and Methods
Animals and their Treatment
Male Wistar rats weighing between 100 and 120 g were procured from the Central Animal House of Panjab University, Chandigarh. The rats were acclimatized for a week before initiating the experiments. They were housed in polypropylene cages under hygienic conditions and were fed standard pellet diet and water ad libitum. The protocols used in the study were strictly according to the guidelines on the human use and care of laboratory animals and were approved by the Institutional Ethics Committee. The animals were administered carbofuran (1 mg/kg/body weight) dissolved in 0.2 ml sunflower oil orally for 28 days. Control animals received an equivalent amount of vehicle (sunflower oil). Carbofuran (technical grade, having a minimum of 97% carbofuran) used was a gift from the International Testing Center, Panchkula.
Biochemical Estimations
Isolation of Brain Mitochondria
The animals were euthanized under light ether anesthesia after 28 days of treatment. Their brains were removed, rinsed in ice-cold isotonic saline (0.9% w/v NaCl) and the cerebral cortex was dissected. Mitochondria were isolated by the method of Stahl et al. (1963). A 10% (w/v) tissue homogenate of cerebral cortex was prepared in extraction buffer (0.32 M Sucrose, 20 mM Tris, 5 mM EDTA, pH 8.0) using mechanically driven Teflon-fitted Potter-Elvehjam type homogenizer. The homogenates were centrifuged at 1500 × g for 15 min to remove nuclei and unbroken cells. Pellets were discarded and the supernatant was again centrifuged at 8000 × g for 15 min to form primary mitochondrial pellet. The supernatant fluid was decanted and the pellet was gently resuspended in 5 ml of 0.1 M Tris–HCl buffer (pH 7.4) for washing. The pellet was recentrifuged and the supernatant was decanted and the final mitochondrial pellet was suspended in 5 ml of 0.1 M Tris–HCl buffer (pH 7.4).
Acetylcholinesterase
Acetylcholinesterase (AChE) activity was assayed in the homogenate by the method of Ellman et al. (1961) using 5,5′-dithio-bis-2-nitrobenzoate (DTNB). The assay mixture consisted of 2.8 ml of phosphate buffer (0.1 M, pH 8.0 containing 0.013% Triton-X 100), 0.1 ml of Ellman reagent (10 mM), 0.1 ml of acetylthiocholine iodide (14.9 mM), and brain homogenate (40–50 μg protein). The activity of enzyme was measured by monitoring the increase in absorbance at 412 nm. AChE activity was calculated using molar extinction coefficient of 5-mercapto-2-nitrobenzoate (13.6 × 103 M−1 cm−1) and the results were expressed as nmoles of substrate hydrolyzed/min/mg protein.
Lipid Peroxidation
The quantitative measurement of lipid peroxidation was performed in the mitochondria according to the method of Wills (1966). The amount of malondialdehyde (MDA) formed was measured by the reaction with thiobarbituric acid at 532 nm. The results were expressed as nmoles of MDA/mg protein using molar extinction coefficient of MDA–thiobarbituric chromophore (1.56 × 105 M−1 cm−1).
Low-Molecular Weight Thiols
Low-molecular weight thiol (LMW-SH) content was estimated in the mitochondria by the method of Ellman (1959). The 0.75 ml of mitochondrial extract was treated with 0.75 ml of 4% (w/v) sulphosalicylic acid, centrifuged at 1,200 × g for 5 min to obtain supernatant which was then mixed with Ellman’s reagent (0.1 mM DTNB in 0.1 M sodium phosphate buffer, pH 8.0). DTNB was reduced by free –SH groups of LMW-SH to form 5-mercapto-2-nitrobenzoate and its absorbance was measured at 412 nm. Results were expressed as nmoles of LMW-SH/mg protein.
Total Thiols
Total thiol groups (TSH) were quantified in the mitochondria according to the method of Sedlak and Lindsay (1968). 0.4 ml of Tris–HCl buffer (0.2 M, pH 8.2 containing 0.02 M EDTA) was added to 0.4 ml of mitochondrial preparation followed by the addition of 0.1 ml of DTNB (0.01 M) in capped tubes. This was followed by addition of 3 ml of absolute methanol and incubation at room temperature for 15 min. The contents were filtered through Whatman No. 1 filter paper and the absorbance was read at 412 nm. Results were expressed as nmoles of TSH/mg protein.
Protein Thiols
Protein thiols were calculated from the difference between the values of total thiols and LMW-SHs.
Succinate Dehydrogenase
Succinate dehydrogenase activity in the mitochondrial fraction was estimated according to the method of King et al. (1976). The reaction mixture consists of 1.5 ml of phosphate buffer (0.2 M, pH 7.8), 0.2 ml of succinic acid (0.6 M, pH 7.8), 0.3 ml of BSA (1% w/v), 0.1 ml of 0.03 M ferricyanide and the reaction was started by addition of mitochondrial preparation (50 μg protein) and the decerase in absorbance was recorded at 420 nm for 3 min. Results were expressed as μmoles of succinate oxidized/min/mg protein.
Mitochondrial Respiratory Chain
Reduction of MTT was used to assess the activity of the mitochondrial respiratory chain in mitochondria by the method of Liu et al. (1997). The reaction mixture containing mitochondrial preparation (50 μg protein) and MTT (0.1 mg/ml) was incubated at 37°C for 30 mins and then centrifuged to form the formazan pellet and dissolved in 1 ml of absolute ethanol and the mixture was recentrifuged and the absorbance of the supernatant was measured at 595 nm. Results were expressed as μg formazan formed/min/mg protein by using blue-formazan as standard.
Estimation of Protein
The protein content was estimated according to the method of Lowry et al. (1951).
Neurobehavioral Studies
Rota-rod Treadmill
Rota-rod treadmill test was performed to measure the muscle strength and coordinated movements of the animals (Cartmell et al. 1991). The rotating rod apparatus (Accelerating Model, Hicon Enterprises, New Delhi, India) was used to evaluate motor function. The rats were placed on the rotating rod for two trials each on day 0 and 28 of the dose regimen. The rats were initially trained to maintain themselves on the rotating rod for more than 3 min. Each trial lasted a maximum of 8 min, during which the rotating rod underwent linear acceleration from 4 to 30 rpm over the first 5 min of the trial and then remained at maximum speed for the remaining 3 min. Animals were scored for their latency to fall (in seconds) in each trial.
Active Avoidance Task
Active avoidance apparatus consisted of a shuttle box with two adjacent chambers connected through a passage with a guillotine door (Fabricated by Sophisticated Analytical Instrumentation Facility of the University). Each of the rats was acclimatized for 3 min. A 5-s unavoidable scrambled foot shock (0.8 mA) was immediately delivered to the animal through the stainless steel grid floor in one of the chambers. Both the chambers were uniformly lit and an electric buzzer fixed in one chamber served as the source of conditioned stimulus. Each trial consisted of a conditioned stimulus for 5 s, followed by a buzzer plus foot shock (0.8 mA) for 5 s at an interval of 1 min. Each animal was then subjected to 10 trials per day. Animals that achieved eight to ten avoidances were included in the study. Total number of avoidances per trial was recorded for each animal.
Statistical Analysis
All values were expressed as mean ± S.D. of six animals per group. The data was analyzed by Student’s t-test and the level of significance is indicated at P < 0.05.
Results and Disscusion
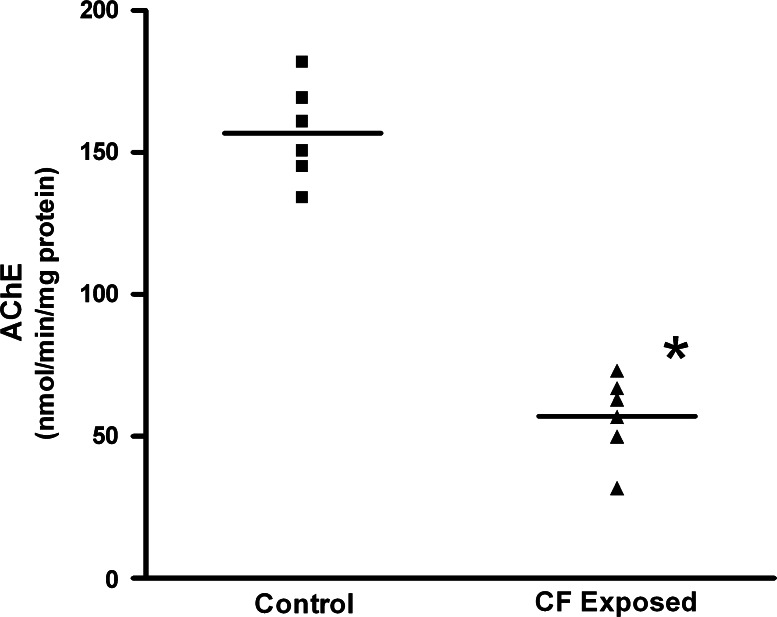
AChE is the primary target of carbofuran and involves carbomylation of the active serine residue of the enzyme resulting in the accumulation of acetylcholine at the nerve terminals, which is responsible for over-stimulation of muscarinic and nicotinic acetylcholine receptors (Fukuto 1990). Effect of in vivo administration of carbofuran on the activity of AChE from brain samples is presented in Fig. 1. The activity of AChE significantly decreased in brain (66.6%) of carbofuran-exposed animals as compared to the control animals. These results are in accordance with our previous findings wherein a decrease in AChE activity was observed in liver after both acute and chronic carbofuran exposure (Kaur and Sandhir 2006). The activity of AChE is often used in monitoring the pesticide exposure (Lessenger and Reese 1999). The inhibition in the activity of AChE suggests significant exposure to carbofuran at the doses studied. The dosage of carbofuran administered to the animals in the present study corresponds to the occupational exposure to carbofuran observed in humans (Hussain et al. 1990).
Fig. 1.
Scatter plots showing the acetylcholinesterase activity in control and carbofuran-exposed animals (n = 6). Group mean values are indicated by lines. Acetylcholinesterase activity was determined in the homogenates prepared from cerebral cortex of control and carbofuran-exposed animals using acetylthiochloline as substrate. Values are expressed as nmol of acetylthiocholine hydrolyzed/min/mg protein. Data was analyzed by Student’s unpaired t-test and values having P < 0.05 were considered significant. * Significantly different from control group
Mitochondrial respiratory chain functions were assessed in terms of reduction of MTT to blue formazan and activity of succinate dehydrogenase. MTT reduction by isolated mitochondria was significantly inhibited in carbofuran-treated animals (31%) as compared to the control animals. In addition, there was a concomitant decrease in the mitochondrial succinate dehydrogenase activity (57%) following carbofuran treatment as compared to the animals from the control group (Table 1). Nakagawa et al. (2004) have shown that the toxicity of chlorpropham, a carbamate, is associated with a rapid depletion of ATP via impairment of mitochondrial function. Another carbamate, carbaryl, has been shown to inhibit succinate dehydrogenase activity and mitochondrial bioenergetics (Moreno et al. 2007). Domico et al. (2006) have shown that acute exposure to high doses of carbamates produce equipotent toxic effects in both dopamine and GABA neurons that may be associated with perturbations in mitochondrial respiration. Owing to the carbomylation of –SH groups by carbofuran, it might interfere with biological processes necessitating the essential –SH group leading to reduction in the activities of succinate dehydrogenase and cytochrome c oxidase, two critical enzymes in the electron transport chain, ultimately leading to a decrease in ATP production and enhanced generation of free radicals. Swerdlow (2007) has also reported that changes in mitochondrial function are primary event associated with various neurodegenerative diseases.
Table 1.
Effect of chronic carbofuran exposure on activity of succinate dehydrogenase and mitochondrial respiration in mitochondria isolated from rat brain
| Groups | Succinate dehydrogenase (μmol succinate oxidized/min/mg protein) | MTT reduction (μg formazan formed/min/ mg protein) |
|---|---|---|
| Control | 24.2 ± 1.51 | 2.03 ± 0.16 |
| Carbofuran exposed | 10.4 ± 1.50* | 1.40 ± 0.19* |
Mitochondria were isolated from cerebral cortex of control and carbofuran-exposed animals. Succinate dehydrogenase activity and mitochondrial respiration was ascertained by following the reduction of ferricyanide to ferrocyanide by succinate or reduction of MTT to blue formazan, respectively, by the isolated mitochondrial preparation. Values are expressed as mean ± S.D; n = 6. Data was analyzed by Student’s unpaired t-test and values having P < 0.05 were considered significant
* Significantly different from control group
GSH is an endogenous thiol antioxidant that functions as a physiological reducing agent to maintain an adequate intracellular redox status (Meister and Anderson 1983). As a reductant, GSH maintains intracellular sulfhydryl-containing proteins in the reduced and active form by either the reduction of potentially toxic peroxides or by the action of thiol-disulfide exchange reactions (Meister and Anderson 1983). Mitochondrion is a subcellular organelle whose function is closely linked to maintenance of redox balance (Jocelyn and Dickson 1980). The redox status of GSH and other thiols has long been known to be critical for proper mitochondrial function (Lash 2006). Alterations in GSH concentration and redox status have been associated with oxidative stress induced by peroxides and other oxidants in mitochondria of the brain (Ravindranath and Reed 1990). Mitochondrial LMW-SH, which mainly consists of GSH, showed a drastic reduction (66.6%) after sub-chronic exposure as compared to the control (Table 2). The mechanism involved in LMW-SH depletion after carbamate exposure involves carbomylation of –SH groups (Ningaraj et al. 1998). More recently, changes in mitochondrial –SH status has also been associated with activation of signaling pathways and expression of genes that regulate apoptosis (Davis et al. 2001). The decreased mitochondrial LMW-SH content was also accompanied by a decrease in mitochondrial TSH content (37.4%), on carbofuran exposure which might be contributed by reduction in GSH levels and/or could be due to decrease in protein –SH groups as observed in the present study (Table 2). Thus the decreased –SH content might affect –SH/–S–S– ratio in proteins resulting in impaired functioning of essential mitochondrial enzymes ensuing mitochondrial dysfunction.
Table 2.
Effect of chronic carbofuran exposure on levels of total, low-molecular weight (LMW), and protein thiols in mitochondria isolated from rat brain
| Groups | Total thiols (nmol/mg protein) | LMW thiols (nmol/mg protein) | Protein thiols (nmol/mg protein) |
|---|---|---|---|
| Control | 56.1 ± 2.45 | 21.4 ± 2.33 | 35.3 ± 4.06 |
| Carbofuran exposed | 35.1 ± 2.28* | 7.14 ± 0.49* | 29.6 ± 2.06* |
Mitochondria were isolated from cerebral cortex of control and carbofuran-exposed animals. LMW and total thiol content of the isolated mitochondrial preparation was estimated by reaction with DTNB. Protein thiols were calculated from the difference between total and LMW thiols. Values are expressed as Mean ± SD; n = 6. Data was analyzed by Student’s unpaired t-test and values having P < 0.05 were considered significant
* Significantly different from control group
The mitochondrial respiratory chain is one of the major sources of damaging free radicals (Navarro and Boveris 2007). The impaired mitochondrial electron transfer favors the mitochondrial generation of free radicals. Mitochondrial lipid peroxidation measured in terms of malondialdehyde formed was observed to accentuate after sub-chronic carbofuran exposure. There was a 43.7% increase in lipid peroxidation in mitochondria from sub-chronic carbofuran-exposed animals as compared to control animals (Fig. 2). A significant correlation was observed between the activity of succinate dehydrogenase and mitochondrial respiration with lipid peroxidation levels in the mitochondria (Figs. 3a and 4a). We have earlier reported an increase in lipid peroxidation in brain after long-term carbofuran exposure (Kamboj et al. 2006). This increase in oxidative stress seem to arise from either the depletion of GSH or the generation of free radicals and ROS because of the impaired mitochondrial electron transport chain. Gupta et al. (2001) have demonstrated that carbofuran treatment resulted in enhanced generation of reactive oxygen and nitrogen species and depleted ATP levels that involve in the inhibition of cytochrome c oxidase activity. The sequence of biochemical and cellular events that might be involved in the carbofuran-induced damage to brain may involve mitochondria-dependent apoptosis. It has been reported by Bustamante et al. (2004) that increased mitochondrial generation of oxidants or the decrease in the antioxidants could lead to oxidation of cytochrome c, and consequently to mitochondria-dependent apoptosis.
Fig. 2.
Scatter plots for lipid peroxidation in isolated mitochondria isolated from control and carbofuran-exposed animals (n = 6). Group mean values are indicated by lines. Mitochondria were isolated from cerebral cortex of control and carbofuran-exposed animals. Lipid peroxidation was estimated by measuring the amount of malondialdehyde (MDA) using thiobarbituric acid. Values are expressed as nmol MDA/mg protein. Data was analyzed by Student’s unpaired t-test and values having P < 0.05 were considered significant. * Significantly different from control group
Fig. 3.
Correlation between rate of mitochondrial respiration and lipid peroxidation (a), rota rod treadmill (b), and active avoidance performance (c). Data was analyzed by linear regression and values having P < 0.05 were considered significant. * Significantly different from control group
Fig. 4.
Correlation between succinate dehydrogenase activity and lipid peroxidation (a), rota rod treadmill (b), and active avoidance performance (c). Data was analyzed by linear regression and values having P < 0.05 were considered significant. * Significantly different from control group
Neurobehavioral functions were assessed as an in vivo functional correlate for the biochemical and pathological effects observed following carbofuran exposure. Rota-rod treadmill test revealed a marked impairment in the muscle strength and motor coordination of the carbofuran-treated animals in terms of reduction in the retention time of the carbofuran-treated animals on the rotating rod (Fig. 5). None of the animals exposed to carbofuran could maintain themselves on the rotating rod for 180 s. Cognitive functions were assessed using active avoidance task in control and carbofuran-exposed animals. Active avoidance is basically a step through inhibitory avoidance test, in which, following an auditory warning signal, the satisfactorily trained and conditioned rat jumps to a shock-free chamber to avoid the foot shock delivered through the grid floor of the cage. The results of active avoidance task have been presented in Fig. 5. Cognitive behavior was assessed by the number of times animal avoids the foot shock in the given set of ten trials. Initially, rats in all the groups made errors, with the animals jumping to the shock-free chamber only after receiving the foot shock. But after a few training trials, there was a marked improvement in the performance of the control animals, which required one to two trials to make the first avoidance. Carbofuran-treated rats revealed significantly reduced number of avoidances per ten trials as compared to the control animals. A strong correlation was observed between the inhibition in the mitochondrial functions (succinate dehydrogenase activity and mitochondrial respiration) with impaired motor and memory functions (Figs. 3b, c and 4b, c). Evidence in the literature suggests that there is a functional association between mitochondrial dysfunction and cognitive impairment (Roubertoux et al. 2003). Carbamates, like other anticholinesterases, have been shown to interfere with the process of learning and memory (Farage-Elawar 1989). The results suggest that impaired mitochondrial functions might influence neurobehavioral deficits, observed following carbofuran exposure.
Fig. 5.
Scatter plots for rota-rod treadmill (a) and active avoidance (b) performance of control and carbofuran-exposed animals (n = 6). Group mean values are indicated by lines. Rota rod treadmill performance of the control and carbofuran-treated animals was assessed by their ability of the animals to maintain on accelerating rotating rod (4–30 rpm) for 3 min. Values were expressed as retention time (s). Active avoidance of control and carbofuran exposed rats was analyzed by subjecting to conditioning by pairing tone with foot shock. Each session consisted of ten trials. Values are expressed as number of avoidances. Data was analyzed by Student’s unpaired t-test and values having P < 0.05 were considered significant. * Significantly different from control group
In summary, results reported here indicate that carbofuran exposure impedes mitochondrial respiratory chain functions along with the decrease in GSH levels resulting in accentuation in mitochondrial oxidative stress, which eventually might be involved in impaired neuronal functioning thus leading to neurobehavioral deficits.
References
- Barlow BK, Lee DW, Cory-Slechta DA, Opanashuk LA (2005) Modulation of antioxidant defense systems by the environmental pesticide maneb in dopaminergic cells. NeuroToxicology 26:63–75 [DOI] [PubMed] [Google Scholar]
- Bustamante J, Di Libero E, Fernandez-Cobo M, Monti N, Cadenas E, Boveris A (2004) Kinetic analysis of thapsigargin-induced thymocyte apoptosis. Free Radic Biol Med 37:1490–1498 [DOI] [PubMed] [Google Scholar]
- Cartmell SM, Gelgor L, Mitchell D (1991) A revised rotarod procedure for measuring the effect of antinociceptive drugs on motor function in the rat. J Pharmacol Methods 26:149–159 [DOI] [PubMed] [Google Scholar]
- Davis W Jr, Ronai Z, Tew KD (2001) Cellular thiols and reactive oxygen species in drug-induced apoptosis. J Pharmacol Exp Ther 296:1–6 [PubMed] [Google Scholar]
- Domico LM, Zeevalk GD, Bernard LP, Cooper KR (2006) Acute neurotoxic effects of mancozeb and maneb in mesencephalic neuronal cultures are associated with mitochondrial dysfunction. NeuroToxicology 27:816–825 [DOI] [PubMed] [Google Scholar]
- Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77 [DOI] [PubMed] [Google Scholar]
- Ellman GL, Courtney KD, Andres V Jr, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95 [DOI] [PubMed] [Google Scholar]
- Farage-Elawar M (1989) Enzyme and behavioral changes in young chicks as a result of carbaryl treatment. J Toxicol Environ Health 26:119–131 [DOI] [PubMed] [Google Scholar]
- Farage-Elawar M, Blaker WD (1992) Chick embryo exposure to carbamates alters neurochemical parameters and behavior. J Appl Toxicol 12:421–426 [DOI] [PubMed] [Google Scholar]
- Fukuto TR (1990) Mechanism of action of organophosphorus and carbamate insecticides. Environ Health Perspect 87:245–254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gogvadze V, Zhivotovsky B (2007) Alteration of mitochondrial function and cell sensitization to death. J Bioenerg Biomembr 39:23–30 [DOI] [PubMed] [Google Scholar]
- Gupta RC (2004) Brain regional heterogeneity and toxicological mechanisms of organophosphates and carbamates. Toxicol Mech Methods 14:103–143 [DOI] [PubMed] [Google Scholar]
- Gupta RC, Goad JT (2000) Role of high-energy phosphates and their metabolites in protection of carbofuran-induced biochemical changes in diaphragm muscle by memantine. Arch Toxicol 74:13–20 [DOI] [PubMed] [Google Scholar]
- Gupta RC, Goad JT, Kadel WL (1991) Carbofuran-induced alterations (in vivo) in high-energy phosphates, creatine kinase (CK) and CK isoenzymes. Arch Toxicol 65:304–310 [DOI] [PubMed] [Google Scholar]
- Gupta RC, Milatovic D, Dettbarn WD (2001) Nitric oxide modulates high-energy phosphates in brain regions of rats intoxicated with diisopropylphosphorofluoridate or carbofuran: prevention by N-tert-butyl-alpha-phenylnitrone or vitamin E. Arch Toxicol 75:346–356 [DOI] [PubMed] [Google Scholar]
- Gupta RC, Milatovic S, Dettbarn WD, Aschner M, Milatovic D (2007) Neuronal oxidative injury and dendritic damage induced by carbofuran: protection by memantine. Toxicol Appl Pharmacol 219:97–105 [DOI] [PubMed] [Google Scholar]
- Halliwell B (2006) Oxidative stress and neurodegeneration: where are we now? J Neurochem 97:1634–1658 [DOI] [PubMed] [Google Scholar]
- Hussain M, Yoshida K, Atiemo M, Johnston D (1990) Occupational exposure of grain farmers to carbofuran. Arch Environ Contam Toxicol 19:197–204 [DOI] [PubMed] [Google Scholar]
- Jocelyn PC, Dickson J (1980) Glutathione and the mitochondrial reduction of hydroperoxides. Biochim Biophys Acta Bioenerg 590:1–12 [DOI] [PubMed] [Google Scholar]
- Kamboj A, Kiran R, Sandhir R (2006) Carbofuran-induced neurochemical and neurobehavioral alterations in rats: attenuation by N-acetylcysteine. Exp Brain Res 170:567–575 [DOI] [PubMed] [Google Scholar]
- Kaur M, Sandhir R (2006) Comparative effects of acute and chronic carbofuran exposure on oxidative stress and drug-metabolizing enzymes in liver. Drug Chem Toxicol 29:415–421 [DOI] [PubMed] [Google Scholar]
- King TE, Ohnishi T, Winter DB, Wu JT (1976) Biochemical and EPR probes for structure-function studies of iron sulfur centers of succinate dehydrogenase. Adv Exp Med Biol 74:182–227 [DOI] [PubMed] [Google Scholar]
- Lash LH (2006) Mitochondrial glutathione transport: physiological, pathological and toxicological implications. Chem Biol Interact 163:54–67 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lessenger JE, Reese BE (1999) Rational use of cholinesterase activity testing in pesticide poisoning. J Am Board Fam Pract 12:307–314 [DOI] [PubMed] [Google Scholar]
- Leuner K, Hauptmann S, Abdel-Kader R, Scherping I, Keil U, Strosznajder JB, Eckert A, Muller WE (2007) Mitochondrial dysfunction: the first domino in brain aging and Alzheimer’s disease? Antioxid Redox Signal 9:1659–1675 [DOI] [PubMed] [Google Scholar]
- Liu Y, Peterson DA, Kimura H, Schubert D (1997) Mechanism of cellular 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) reduction. J Neurochem 69:581–593 [DOI] [PubMed] [Google Scholar]
- Liu R, Liu IY, Bi X, Thompson RF, Doctrow SR, Malfroy B, Baudry M (2003) Reversal of age-related learning deficits and brain oxidative stress in mice with superoxide dismutase/catalase mimetics. Proc Natl Acad Sci USA 100:8526–8531 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951). Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275 [PubMed] [Google Scholar]
- Meister A, Anderson ME (1983). Glutathione. Annu Rev Biochem 52:711–760 [DOI] [PubMed] [Google Scholar]
- Moreno AJM, Serafim TL, Oliveira PJ, Madeira VMC (2007) Inhibition of mitochondrial bioenergetics by carbaryl is only evident for higher concentrations – relevance for carbaryl toxicity mechanisms. Chemosphere 66:404–411 [DOI] [PubMed] [Google Scholar]
- Moretto A (1998) Experimental and clinical toxicology of anticholinesterase agents. Toxicol Lett 102–103:509–513 [DOI] [PubMed] [Google Scholar]
- Nakagawa Y, Nakajima K, Suzuki T (2004) Chlorpropham induces mitochondrial dysfunction in rat hepatocytes. Toxicology 200:123–133 [DOI] [PubMed] [Google Scholar]
- Navarro A, Boveris A (2007) The mitochondrial energy transduction system and the aging process. Am J Physiol Cell Physiol 292:C670–686 [DOI] [PubMed] [Google Scholar]
- Ningaraj NS, Schloss JV, Williams TD, Faiman MD (1998) Glutathione carbamoylation with S-methyl N,N-diethylthiolcarbamate sulfoxide and sulfone. Mitochondrial low Km aldehyde dehydrogenase inhibition and implications for its alcohol-deterrent action. Biochem Pharmacol 55:749–756 [DOI] [PubMed] [Google Scholar]
- Papa S (1996) Mitochondrial oxidative phosphorylation changes in the life span. Molecular aspects and physiopathological implications. Biochim Biophys Acta Bioenerg 1276:87–105 [DOI] [PubMed] [Google Scholar]
- Rai DK, Sharma B (2007) Carbofuran-induced oxidative stress in mammalian brain. Mol Biotechnol 37:66–71 [DOI] [PubMed] [Google Scholar]
- Ravindranath V, Reed DJ (1990) Glutathione depletion and formation of glutathione-protein mixed disulfide following exposure of brain mitochondria to oxidative stress. Biochem Biophys Res Commun 169:1075–1079 [DOI] [PubMed] [Google Scholar]
- Roubertoux PL, Sluyter F, Carlier M, Marcet B, Maarouf-Veray F, Cherif C, Marican C, Arrechi P, Godin F, Jamon M, Verrier B, Cohen-Salmon C (2003) Mitochondrial DNA modifies cognition in interaction with the nuclear genome and age in mice. Nat Genet 35:65–69 [DOI] [PubMed] [Google Scholar]
- Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205 [DOI] [PubMed] [Google Scholar]
- Stahl WL, Smith JC, Napolitano LM, Basford RE (1963) Brain mitochondria. I. Isolation of bovine brain mitochondria. J Cell Biol 19:293–307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swerdlow RH (2007) Treating neurodegeneration by modifying mitochondria:potential solutions to a “complex” problem. Antioxid Redox Signal 9:1591–1603 [DOI] [PubMed] [Google Scholar]
- Tsujimoto Y (1997) Apoptosis and necrosis: intracellular ATP level as a determinant for cell death modes. Cell Death Differ 4:429–434 [DOI] [PubMed] [Google Scholar]
- Wills ED (1966) Mechanisms of lipid peroxide formation in animal tissues. Biochem J 99:667–676 [DOI] [PMC free article] [PubMed] [Google Scholar]