Abstract
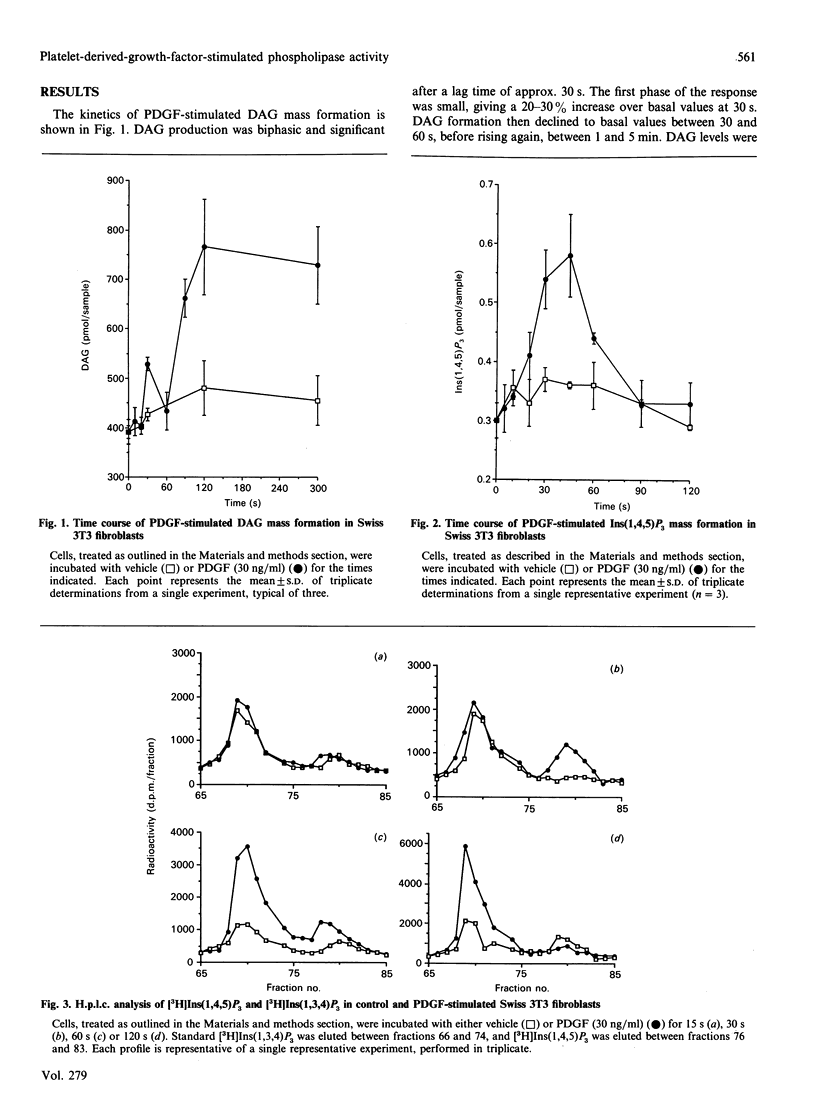
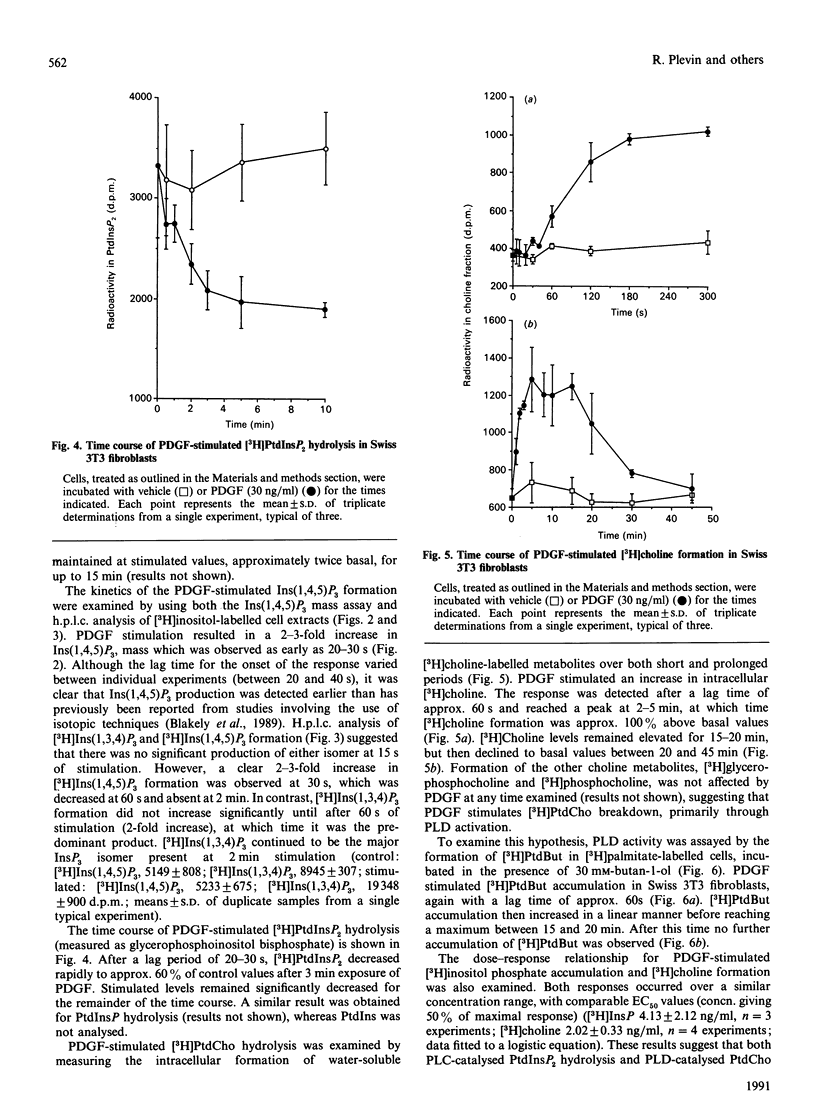
Platelet-derived growth factor (PDGF) stimulated sn-1,2-diacylglycerol (DAG) mass formation in Swiss 3T3 fibroblasts with a lag time of some 30 s. The response was biphasic, with the second phase being sustained over time. PDGF also stimulated the formation of Ins(1,4,5)P3 with a similar lag time to the DAG response, suggesting that DAG is derived from PtdIns(4,5)P2 hydrolysis at this time point. PDGF-stimulated phosphatidylcholine (PtdCho) hydrolysis in Swiss 3T3 fibroblasts, as measured by the formation of water-soluble choline metabolites and phosphatidylbutanol (PtdBut) accumulation, was by a phospholipase D (PLD)-catalysed pathway which was kinetically downstream of initial PtdIns(4,5)P2 hydrolysis. Accumulation of PtdBut increased up to 15 min, suggesting that PLD activity is not rapidly densitized in response to PDGF. The kinetics of PtdCho hydrolysis closely paralleled the second phase of DAG formation, strongly suggesting that during prolonged stimulation periods PtdCho is a major source of DAG in these cells. However, since PtdIns(4,5)P2 breakdown was also prolonged, PDGF-stimulated DAG may be derived from both phospholipids. Down-regulation of protein kinase C (PKC), by pre-treatment with phorbol 12-myristate 13-acetate, abolished both [3H]choline and [3H]PtdBut formation, suggesting that PLD-catalysed PtdCho hydrolysis may be dependent on PKC activation, supporting its dependence on prior PtdIns(4,5)P2 hydrolysis.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berridge M. J., Heslop J. P., Irvine R. F., Brown K. D. Inositol trisphosphate formation and calcium mobilization in Swiss 3T3 cells in response to platelet-derived growth factor. Biochem J. 1984 Aug 15;222(1):195–201. doi: 10.1042/bj2220195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berridge M. J., Irvine R. F. Inositol trisphosphate, a novel second messenger in cellular signal transduction. Nature. 1984 Nov 22;312(5992):315–321. doi: 10.1038/312315a0. [DOI] [PubMed] [Google Scholar]
- Besterman J. M., Duronio V., Cuatrecasas P. Rapid formation of diacylglycerol from phosphatidylcholine: a pathway for generation of a second messenger. Proc Natl Acad Sci U S A. 1986 Sep;83(18):6785–6789. doi: 10.1073/pnas.83.18.6785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Billah M. M., Anthes J. C. The regulation and cellular functions of phosphatidylcholine hydrolysis. Biochem J. 1990 Jul 15;269(2):281–291. doi: 10.1042/bj2690281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Billah M. M., Pai J. K., Mullmann T. J., Egan R. W., Siegel M. I. Regulation of phospholipase D in HL-60 granulocytes. Activation by phorbol esters, diglyceride, and calcium ionophore via protein kinase- independent mechanisms. J Biol Chem. 1989 May 25;264(15):9069–9076. [PubMed] [Google Scholar]
- Black F. M., Wakelam M. J. Activation of inositol phospholipid breakdown by prostaglandin F2 alpha without any stimulation of proliferation in quiescent NIH-3T3 fibroblasts. Biochem J. 1990 Mar 15;266(3):661–667. doi: 10.1042/bj2660661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blakeley D. M., Corps A. N., Brown K. D. Bombesin and platelet-derived growth factor stimulate formation of inositol phosphates and Ca2+ mobilization in Swiss 3T3 cells by different mechanisms. Biochem J. 1989 Feb 15;258(1):177–185. doi: 10.1042/bj2580177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown K. D., Littlewood C. J., Blakeley D. M. Differential potentiation of mitogen-stimulated phosphoinositide hydrolysis in protein kinase C-depleted Swiss 3T3 cells. Biochem J. 1990 Sep 1;270(2):557–560. doi: 10.1042/bj2700557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook S. J., Palmer S., Plevin R., Wakelam M. J. Mass measurement of inositol 1,4,5-trisphosphate and sn-1,2-diacylglycerol in bombesin-stimulated Swiss 3T3 mouse fibroblasts. Biochem J. 1990 Jan 15;265(2):617–620. doi: 10.1042/bj2650617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook S. J., Wakelam M. J. Analysis of the water-soluble products of phosphatidylcholine breakdown by ion-exchange chromatography. Bombesin and TPA (12-O-tetradecanoylphorbol 13-acetate) stimulate choline generation in Swiss 3T3 cells by a common mechanism. Biochem J. 1989 Oct 15;263(2):581–587. doi: 10.1042/bj2630581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook S. J., Wakelam M. J. Hydrolysis of phosphatidylcholine by phospholipase D is a common response to mitogens which stimulate inositol lipid hydrolysis in Swiss 3T3 fibroblasts. Biochim Biophys Acta. 1991 Apr 17;1092(2):265–272. doi: 10.1016/0167-4889(91)90166-u. [DOI] [PubMed] [Google Scholar]
- Creba J. A., Downes C. P., Hawkins P. T., Brewster G., Michell R. H., Kirk C. J. Rapid breakdown of phosphatidylinositol 4-phosphate and phosphatidylinositol 4,5-bisphosphate in rat hepatocytes stimulated by vasopressin and other Ca2+-mobilizing hormones. Biochem J. 1983 Jun 15;212(3):733–747. doi: 10.1042/bj2120733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eldar H., Zisman Y., Ullrich A., Livneh E. Overexpression of protein kinase C alpha-subtype in Swiss/3T3 fibroblasts causes loss of both high and low affinity receptor numbers for epidermal growth factor. J Biol Chem. 1990 Aug 5;265(22):13290–13296. [PubMed] [Google Scholar]
- Escobedo J. A., Williams L. T. A PDGF receptor domain essential for mitogenesis but not for many other responses to PDGF. Nature. 1988 Sep 1;335(6185):85–87. doi: 10.1038/335085a0. [DOI] [PubMed] [Google Scholar]
- Exton J. H. Signaling through phosphatidylcholine breakdown. J Biol Chem. 1990 Jan 5;265(1):1–4. [PubMed] [Google Scholar]
- Fukami K., Takenawa T. Quantitative changes in polyphosphoinositides 1,2-diacylglycerol and inositol 1,4,5-trisphosphate by platelet-derived growth factor and prostaglandin F2 alpha. J Biol Chem. 1989 Sep 5;264(25):14985–14989. [PubMed] [Google Scholar]
- Griendling K. K., Rittenhouse S. E., Brock T. A., Ekstein L. S., Gimbrone M. A., Jr, Alexander R. W. Sustained diacylglycerol formation from inositol phospholipids in angiotensin II-stimulated vascular smooth muscle cells. J Biol Chem. 1986 May 5;261(13):5901–5906. [PubMed] [Google Scholar]
- Hasegawa-Sasaki H. Early changes in inositol lipids and their metabolites induced by platelet-derived growth factor in quiescent Swiss mouse 3T3 cells. Biochem J. 1985 Nov 15;232(1):99–109. doi: 10.1042/bj2320099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kazlauskas A., Cooper J. A. Autophosphorylation of the PDGF receptor in the kinase insert region regulates interactions with cell proteins. Cell. 1989 Sep 22;58(6):1121–1133. doi: 10.1016/0092-8674(89)90510-2. [DOI] [PubMed] [Google Scholar]
- Krauss R. S., Housey G. M., Johnson M. D., Weinstein I. B. Disturbances in growth control and gene expression in a C3H/10T1/2 cell line that stably overproduces protein kinase C. Oncogene. 1989 Aug;4(8):991–998. [PubMed] [Google Scholar]
- Lacal J. C., Fleming T. P., Warren B. S., Blumberg P. M., Aaronson S. A. Involvement of functional protein kinase C in the mitogenic response to the H-ras oncogene product. Mol Cell Biol. 1987 Nov;7(11):4146–4149. doi: 10.1128/mcb.7.11.4146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larrodera P., Cornet M. E., Diaz-Meco M. T., Lopez-Barahona M., Diaz-Laviada I., Guddal P. H., Johansen T., Moscat J. Phospholipase C-mediated hydrolysis of phosphatidylcholine is an important step in PDGF-stimulated DNA synthesis. Cell. 1990 Jun 15;61(6):1113–1120. doi: 10.1016/0092-8674(90)90074-o. [DOI] [PubMed] [Google Scholar]
- Lavie Y., Liscovitch M. Activation of phospholipase D by sphingoid bases in NG108-15 neural-derived cells. J Biol Chem. 1990 Mar 5;265(7):3868–3872. [PubMed] [Google Scholar]
- Löffelholz K. Receptor regulation of choline phospholipid hydrolysis. A novel source of diacylglycerol and phosphatidic acid. Biochem Pharmacol. 1989 May 15;38(10):1543–1549. doi: 10.1016/0006-2952(89)90299-2. [DOI] [PubMed] [Google Scholar]
- MacNulty E. E., Plevin R., Wakelam M. J. Stimulation of the hydrolysis of phosphatidylinositol 4,5-bisphosphate and phosphatidylcholine by endothelin, a complete mitogen for Rat-1 fibroblasts. Biochem J. 1990 Dec 15;272(3):761–766. doi: 10.1042/bj2720761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin T. W., Michaelis K. P2-purinergic agonists stimulate phosphodiesteratic cleavage of phosphatidylcholine in endothelial cells. Evidence for activation of phospholipase D. J Biol Chem. 1989 May 25;264(15):8847–8856. [PubMed] [Google Scholar]
- Martinson E. A., Trilivas I., Brown J. H. Rapid protein kinase C-dependent activation of phospholipase D leads to delayed 1,2-diglyceride accumulation. J Biol Chem. 1990 Dec 25;265(36):22282–22287. [PubMed] [Google Scholar]
- Meisenhelder J., Suh P. G., Rhee S. G., Hunter T. Phospholipase C-gamma is a substrate for the PDGF and EGF receptor protein-tyrosine kinases in vivo and in vitro. Cell. 1989 Jun 30;57(7):1109–1122. doi: 10.1016/0092-8674(89)90048-2. [DOI] [PubMed] [Google Scholar]
- Nishizuka Y. The role of protein kinase C in cell surface signal transduction and tumour promotion. Nature. 1984 Apr 19;308(5961):693–698. doi: 10.1038/308693a0. [DOI] [PubMed] [Google Scholar]
- Nånberg E., Rozengurt E. Temporal relationship between inositol polyphosphate formation and increases in cytosolic Ca2+ in quiescent 3T3 cells stimulated by platelet-derived growth factor, bombesin and vasopressin. EMBO J. 1988 Sep;7(9):2741–2747. doi: 10.1002/j.1460-2075.1988.tb03128.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmer S., Hughes K. T., Lee D. Y., Wakelam M. J. Development of a novel, Ins(1,4,5)P3-specific binding assay. Its use to determine the intracellular concentration of Ins(1,4,5)P3 in unstimulated and vasopressin-stimulated rat hepatocytes. Cell Signal. 1989;1(2):147–156. doi: 10.1016/0898-6568(89)90004-1. [DOI] [PubMed] [Google Scholar]
- Pessin M. S., Baldassare J. J., Raben D. M. Molecular species analysis of mitogen-stimulated 1,2-diglycerides in fibroblasts. Comparison of alpha-thrombin, epidermal growth factor, and platelet-derived growth factor. J Biol Chem. 1990 May 15;265(14):7959–7966. [PubMed] [Google Scholar]
- Pessin M. S., Raben D. M. Molecular species analysis of 1,2-diglycerides stimulated by alpha-thrombin in cultured fibroblasts. J Biol Chem. 1989 May 25;264(15):8729–8738. [PubMed] [Google Scholar]
- Price B. D., Morris J. D., Hall A. Stimulation of phosphatidylcholine breakdown and diacylglycerol production by growth factors in Swiss-3T3 cells. Biochem J. 1989 Dec 1;264(2):509–515. doi: 10.1042/bj2640509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Pena A., Rozengurt E. Disappearance of Ca2+-sensitive, phospholipid-dependent protein kinase activity in phorbol ester-treated 3T3 cells. Biochem Biophys Res Commun. 1984 May 16;120(3):1053–1059. doi: 10.1016/s0006-291x(84)80213-2. [DOI] [PubMed] [Google Scholar]
- Ross R., Raines E. W., Bowen-Pope D. F. The biology of platelet-derived growth factor. Cell. 1986 Jul 18;46(2):155–169. doi: 10.1016/0092-8674(86)90733-6. [DOI] [PubMed] [Google Scholar]
- Sachinidis A., Locher R., Vetter W., Tatje D., Hoppe J. Different effects of platelet-derived growth factor isoforms on rat vascular smooth muscle cells. J Biol Chem. 1990 Jun 25;265(18):10238–10243. [PubMed] [Google Scholar]
- Slivka S. R., Meier K. E., Insel P. A. Alpha 1-adrenergic receptors promote phosphatidylcholine hydrolysis in MDCK-D1 cells. A mechanism for rapid activation of protein kinase C. J Biol Chem. 1988 Sep 5;263(25):12242–12246. [PubMed] [Google Scholar]
- Suzuki-Sekimori R., Matuoka K., Nagai Y., Takenawa T. Diacylglycerol, but not inositol 1,4,5-trisphosphate, accounts for platelet-derived growth factor-stimulated proliferation of BALB 3T3 cells. J Cell Physiol. 1989 Sep;140(3):432–438. doi: 10.1002/jcp.1041400305. [DOI] [PubMed] [Google Scholar]
- Ullrich A., Schlessinger J. Signal transduction by receptors with tyrosine kinase activity. Cell. 1990 Apr 20;61(2):203–212. doi: 10.1016/0092-8674(90)90801-k. [DOI] [PubMed] [Google Scholar]
- Wahl M. I., Olashaw N. E., Nishibe S., Rhee S. G., Pledger W. J., Carpenter G. Platelet-derived growth factor induces rapid and sustained tyrosine phosphorylation of phospholipase C-gamma in quiescent BALB/c 3T3 cells. Mol Cell Biol. 1989 Jul;9(7):2934–2943. doi: 10.1128/mcb.9.7.2934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wakelam M. J., Murphy G. J., Hruby V. J., Houslay M. D. Activation of two signal-transduction systems in hepatocytes by glucagon. Nature. 1986 Sep 4;323(6083):68–71. doi: 10.1038/323068a0. [DOI] [PubMed] [Google Scholar]
- Warden C. H., Friedkin M. Regulation of choline kinase activity and phosphatidylcholine biosynthesis by mitogenic growth factors in 3T3 fibroblasts. J Biol Chem. 1985 May 25;260(10):6006–6011. [PubMed] [Google Scholar]
- Westermark B., Siegbahn A., Heldin C. H., Claesson-Welsh L. B-type receptor for platelet-derived growth factor mediates a chemotactic response by means of ligand-induced activation of the receptor protein-tyrosine kinase. Proc Natl Acad Sci U S A. 1990 Jan;87(1):128–132. doi: 10.1073/pnas.87.1.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whetton A. D., Monk P. N., Consalvey S. D., Huang S. J., Dexter T. M., Downes C. P. Interleukin 3 stimulates proliferation via protein kinase C activation without increasing inositol lipid turnover. Proc Natl Acad Sci U S A. 1988 May;85(10):3284–3288. doi: 10.1073/pnas.85.10.3284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright T. M., Shin H. S., Raben D. M. Sustained increase in 1,2-diacylglycerol precedes DNA synthesis in epidermal-growth-factor-stimulated fibroblasts. Evidence for stimulated phosphatidylcholine hydrolysis. Biochem J. 1990 Apr 15;267(2):501–507. doi: 10.1042/bj2670501. [DOI] [PMC free article] [PubMed] [Google Scholar]


