Abstract
For clinical and epidemiological purposes, it is necessary to be able to classify serological responses during dengue virus infection. Thus, it is important to develop a test that can distinguish between primary and secondary serological responses. The hemagglutination inhibition (HI) test, which is currently recommended by the World Health Organization, is complicated to perform. We developed an enzyme-linked immunosorbent assay based on changes in the avidity of immunoglobulin G during the infectious episode. This test can discriminate between primary and secondary infections by using a single serum sample collected during the acute phase of infection. We took 1,140 avidity measurements with 118 pairs of serum samples or sequential samples taken from patients classified as having primary or secondary infection according to World Health Organization laboratory criteria. The mean percent avidity was significantly lower during primary infection (25.9%) than during secondary infection (66.3%) (Student t test, P < 0.001). The test had a sensitivity of 82.7% (95% confidence interval [CI] = 79.0 to 86.6) and a specificity of 77.5% (95% CI = 73.3 to 81.7). Based on analysis of only blood samples collected between the third and seventh days of the illness, during which most clinical complications occur, the sensitivity and specificity of the test were 95.1% (95% CI = 92.6 to 97.7) and 80.0% (95% CI = 75.3 to 84.7), respectively. This rapid and simple test appears to be an excellent alternative to the HI test for discriminating between primary and secondary dengue virus infections during the acute phase of dengue.
Dengue fever (DF) is an acute infectious disease that occurs in subtropical and tropical areas, and one of the most severe arthropod-borne viral diseases in terms of human mortality and morbidity. Dengue virus belongs to the Flaviviridae family, genus Flavivirus. Four distinct serotypes (DEN-1, DEN-2, DEN-3, and DEN-4) have been identified. Infection with any of these viruses may be asymptomatic or cause a self-limiting febrile illness known as DF. In a small percentage of cases, it may result in hepatitis (18), encephalitis (16, 21), or a life-threatening syndrome called dengue hemorrhagic fever (DHF)/dengue shock syndrome.
Infection confers lasting immunity to the serotype involved. Until recently, the most commonly used serological techniques for the routine diagnosis of dengue virus were the hemagglutination inhibition (HI) test (6) and capture immunoglobulin M (IgM) by an enzyme-linked immunosorbent assay (ELISA) (3). The HI test is the reference test recommended by the World Health Organization (WHO) to discriminate between primary and secondary dengue virus infection (27). Secondary infections are characterized by the presence of HI antibodies in acute-phase samples and by high titers of HI antibodies (>1:1,280) in serum from convalescence phase serum samples.
The pathogenesis of DHF is still unclear (10, 11), although since the 1970s secondary infection with a different serotype has been suspected to be a risk factor for DHF (12, 13). If this antibody-dependent enhancement (ADE) hypothesis is verified, it could be useful for the treating clinician to determine the immunological status of each hospitalized patient with dengue infection.
Many investigators have questioned the general applicability of using HI titers to distinguish primary from secondary infections in regions where two or more flaviviruses are cocirculating because IgG antibodies measured are broadly flavivirus reactive. This reference test also requires paired serum samples and cannot give an early diagnosis. Innis et al. developed an ELISA for the diagnosis of dengue infection (17). This technique, based on the IgM/IgG ratio, can distinguish between primary and secondary infections. However, IgM can persist for more than 8 months and can be produced in a nonspecific manner (4, 5). This can make it difficult to determine the immunological status of the patient. These observations suggest that an alternative method is required to discriminate primary from secondary dengue virus infections.
The method of choice seems to be the measurement of the antigen-binding avidity of specific IgG: a first study showed the usefulness of IgG avidity to discriminate between primary and secondary dengue infection (8). The avidity assay is based on the fact that the first antibodies synthesized after an antigenic challenge or primary infection have a lower affinity for the antigen than those produced later on. It has been shown to be useful for distinguishing primary from chronic or recurrent infections of other viruses, such as rubella virus (14), hantavirus (15), or human herpesviruses (9, 26). Therefore, we designed an avidity test requiring a single serum sample collected during the acute phase of infection to determine the immunological status of dengue patients.
MATERIALS AND METHODS
Reference sera.
Sera from the collection of the Centre National de Reference des Arbovirus from the Institut Pasteur de la Guyane were used for this study. We considered that the first day of fever was day 1 of the disease. For each pair of serum samples, the first sample was collected during the acute phase (days 1 to 5), and dengue virus was isolated from an Aedes pseudoscutellaris cell culture (AP61) or detected by reverse transcription-PCR with the methods routinely used in our laboratory (19, 23). The second sample was collected during the convalescent phase, 7 days or more after the onset of fever.
WHO laboratory criteria were used to classify the sera as primary or secondary dengue infection according to the results of the HI tests (27). These tests were previously performed for each serum samples analyzed with our IgG avidity test described below. Three groups of serum samples were defined. (i) Group A consisted of 30 pairs of sera from patients infected with the Den-3 serotype: 16 primary infections and 14 secondary infections. This group was used to show that the avidity index was statistically different between the primary and secondary dengue infection serum samples. (ii) Group B consisted of group A plus 43 additional pairs of sera and 21 sequential serum samples from seven dengue patients. Among all of the patients unique to group B, 34 had a primary infection and 16 had a secondary infection. Using a discriminant analysis, this group was used to build a classification function from the avidity index and the day of fever to distinguish between primary and secondary dengue infection. (iii) Group C consisted of 25 pairs of sera and 55 sequential serum samples from 13 patients infected with dengue virus. Among all of these patients, 23 had a primary infection and 15 had a secondary infection. This group was used to assess the sensitivity and the specificity of the test. Patients from groups B and C were infected by serotype DEN-1, DEN-2, or DEN-3.
Antigens.
The dengue antigens (DEN-2 and DEN-3) used for the present study were prepared by the sucrose acetone method (6) and were obtained from the reference collection of the Centre National de Reference des Arbovirus. Normal mouse brain antigen was used as a control in all tests.
Development of the IgG avidity test.
The best dilutions of the different reagents for the IgG ELISA were determined in preliminary tests with positive and negative reference sera. We used as a negative control sera from patients vaccinated against yellow fever without any history of dengue infection. Some of the wells of two flat-bottom microplates (Polysorp; Nunc) were coated with 100 μl of dengue antigen diluted 1:2,000 in phosphate-buffered saline (PBS). Normal mouse brain antigen was diluted in an identical manner and used to coat other adjacent wells. After overnight incubation at 4°C, the microplates were washed three times with PBS containing 0.1% Tween 20 (PBS-T; both reagents from Sigma Laboratories, l'Isle d'Abeau Chesnes, France). The sera were diluted (1:100) in PBS containing 0.1% Tween 20 and 5% nonfat dried milk (PBS-T-NDM) and added to each well of two separate microplates (100 μl/well). Six negative and two positive reference sera were included in each plate as controls. The negative controls consisted of sera from patients with other febrile illnesses but negative for dengue infection. The plates were then incubated for 2 h at 37°C. One of the plates was washed three times with PBS-T, whereas 300 μl of PBS-T containing 8 M urea (Sigma Laboratories) was added to each well of the second plate, followed by incubation at 37°C for 5 min, and then washed three times with PBS-T. Each serum was tested at least in duplicate or in six wells on each plate. Bound IgG was detected by the addition of 100 μl per well of conjugated goat anti-human IgG horseradish peroxidase (Jackson Immunoresearch Laboratories, West Grove, PA) diluted 1:2,000 in PBS-T-NDM. After 1 h at 37°C, the plates were washed with PBS-T. Bound antibodies were visualized by adding tetramethylbenzidine (Sigma Laboratories) at 100 μl per well.
After 15 min at room temperature, the absorbance at 650 nm (A650) was read with a microplate reader, and the ΔA650 was calculated (ΔA650 = A650 Den Ag − A650 control Ag). To validate the test, the ΔA650 of the positive control serum had to be ≥0.5 and the ΔA650 of the negative control serum had to be ≤0.2. The mean ± the standard deviation A650 values for the negative controls were then determined. A result was considered negative when the A650 values were less than the mean value for the negative control plus two standard deviations, indeterminate when the A650 values were between two and three standard deviations, and positive when the A650 values were more than three standard deviations. An avidity index or percentage of avidity was then calculated for each sample in which dengue IgG were detected: (A650 assay with urea/A650 assay without urea) × 100.
Statistical analysis.
We used BMDP statistical software for the statistical analysis (BMDP statistical software 1992). We used the W statistic of the Wilk's test for normality to test the normal distribution of the avidity level and the Student t test to compare the mean avidity levels according to immune status.
First, we checked that the avidity level was significantly different between primary and secondary dengue infection. For this, we used a covariance analysis. This method takes into account the repeated measures for each serum, the inter-individual variations, and the temporal trends in avidity levels during the disease. It can also compare trends over time between primary and secondary dengue infection. Using covariance analysis we assessed the adjustment of the model, testing the level of significance of the model (P value) with or without the variables included in the model (20, 22). When P value is <0.05 for a given variable, we concluded that this variable has a significant role in the level of avidity of the sera. If the immune status, i.e., primary or secondary dengue infection, has a significant level, then it was possible to conclude that avidity levels are significantly different between primary and secondary dengue infection cases.
Second, we used a discriminant analysis to classify the category of serum samples according to WHO laboratory criteria for identification of dengue infections as primary or secondary. This discriminant analysis, based on the level of avidity and the day of fever when the sample had been collected, finds the combination of the variables that best predicts the category (primary or secondary) to which a serum sample belongs. The combination of predictor variables is called a classification function. It is possible to assess the performances of the classification function (sensitivity and specificity) by using the Jack Knife method. However, these results are calculated based on the sample that has been also used for building the classification function. Thus, in the third step, in order to assess the real performance of the method, we classified cases that were not used in the calculations in the second step.
RESULTS
Validation of avidity test.
To validate the test biologically, serum samples from group A were tested with the DEN-2 antigen, i.e., a heterologous antigen-antibody reaction. These samples were also tested with the homologous antigen DEN-3. The covariance analysis made with the homologous DEN-3 antigen showed that the immunity level (P = 0.004) and the day of blood sample (P < 0.001) play a significant part in explaining the level of avidity. Hence, in all subsequent experiments, the ELISAs were carried out with the DEN-2 antigen regardless of the viral serotype initially detected in the serum.
Among the 1,140 avidity measurements taken in the present study, avidity ranged from 0 to 108, with a mean value of 40.1, a standard deviation of 35.8, and a median value of 42. The avidity level, followed a normal distribution (W statistic = 0.98, P > 0.05). The mean avidity was significantly lower among the 738 primary dengue infection sera (25.9%) than among the 402 secondary dengue infection sera (66.3%) (Student t test, P < 0.001) (Table 1).
TABLE 1.
Comparison of the mean IgG avidity values for primary and secondary dengue infectionsa
| Days after onset of DF | Primary infection
|
Secondary infection
|
||
|---|---|---|---|---|
| Avidity measure | Mean AI (SD) | Avidity measure | Mean AI (SD) | |
| 1 | 34 | 0.0 (0.00) | 12 | 52.2 (29.87) |
| 2 | 144 | 5.7 (16.81) | 60 | 58.1 (36.33) |
| 3 | 74 | 1.2 (6.85) | 66 | 36.3 (35.17) |
| 4 | 60 | 3.7 (9.86) | 44 | 67.0 (19.33) |
| 5 | 88 | 18.0 (24.22) | 26 | 70.7 (9.25) |
| 6 | 20 | 23.4 (18.11) | 12 | 66.7 (18.56) |
| 7-9 | 86 | 45.4 (22.29) | 18 | 72.9 (21.67) |
| 10-19 | 126 | 52.1 (29.16) | 62 | 88.9 (12.09) |
| 20-29 | 92 | 50.1 (22.50) | 88 | 75.5 (23.83) |
| >30 | 14 | 62.4 (23.67) | 14 | 77.1 (24.85) |
| Total | 738 | 25.9 (29.98) | 402 | 66.3 (30.47) |
Based on WHO criteria. AI, avidity index.
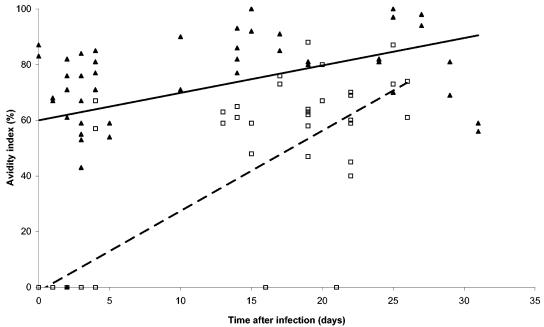
Using the 154 avidity measures taken between days 1 and 31 in the 16 primary and 14 secondary sera from group A, we found that the mean avidity level was significantly lower among the 76 measures provided by primary dengue infection sera (26.3%) than among the 78 measures from secondary dengue infection sera (68.8%) (Student t test, P < 0.001). The way in which the dengue avidity of IgG from group A sera changed over time is shown in Fig. 1.
FIG. 1.
Temporal changes in IgG antibody avidity in sera from patients (group A) with primary (□) and secondary (▴) dengue infections according to WHO criteria. Trends for a primary (- -) and a secondary infection (—) are indicated.
We carried out linear regression analysis to examine the correlation between the avidity level of the sera and the day of blood collection, as well as the immune status.
The level of avidity of each serum sample was significantly related to the day of blood collection (P < 0.001), to the immune status (P = 0.03), and to the interaction between day of blood collection and immune status (P < 0.001). Whatever the immune status was, the level of avidity increased with the day of blood collection, but the avidity level of the primary sera was always lower than that of the secondary sera. The equations of the linear regressions for the primary and secondary dengue infection cases were, respectively, avidity index = 2.5 × day of blood collection − 1.38 and avidity index = 1.3 × day of blood collection + 12.
Classification function.
After checking that the immune status was significantly related to the level of avidity, we carried out discriminant analysis with 764 measures of avidity: 500 measures were obtained from primary dengue infection sera and the others from secondary dengue infection sera (group B). The variables used in the discriminant analysis were the day of blood collection (from day 1 of disease to day 142), the level of avidity of the sera, and the interaction between day of collection and level of avidity. For each serum, the classification function was equal to: (0.11 × day) − (0.046 × avidity) − (0.00088 × day × avidity) + 1.6. When the result was positive the serum was classified as a primary dengue infection, and when negative the serum was classified as a secondary dengue infection. The sensitivity and the specificity values calculated by the Jack Knife method were, respectively, 75.0% (95% confidence interval [CI] = 71.9 to 78.1) and 76.5% (95% CI = 73.5 to 79.5). In other words, the classification function allowed us to classify 375 of 500 (75%) primary sera as primary dengue infections and 202 of 264 (76.5%) secondary sera as secondary dengue infections on the basis of their avidity indexes (Table 2).
TABLE 2.
Performance of IgG avidity test with sera from patients with primary or secondary dengue infections as determined by using the HI test as a reference testa
| Group | Infection | IgG avidity test
|
|||
|---|---|---|---|---|---|
| Avidity measure | Primary infection | Secondary infection | % Agreement | ||
| B | Primary | 500 | 375 | 125 | 75.0 |
| Secondary | 264 | 62 | 202 | 76.5 | |
| C | Primary | 238 | 197 | 41 | 82.7 |
| Secondary | 138 | 31 | 107 | 77.5 | |
Based on WHO criteria.
Sensitivity and specificity of avidity test.
After determining the classification function, we assessed the sensitivity and the specificity of the test by using the avidity level measured from 376 avidity values obtained from group C sera, among which 238 were collected from patients with primary dengue infection, and the others were from patients with secondary dengue infection: 197 of 238 (82.7%, 95% CI = 79.0 to 86.6) and 107 of 138 (77.5%, 95% CI = 73.3 to 81.7), respectively, were classified in agreement with the results of the HI test. The sensitivity and the specificity of the method were 82.7 and 77.5%, respectively (Table 2). Between days 3 and 7 of disease, the sensitivity and specificity of the avidity test increased to 95.1% (95% CI = 92.6 to 97.7) and 80.0% (95% CI = 75.3 to 84.7), respectively.
DISCUSSION
According to the ADE hypothesis, secondary dengue virus infections are risk factors for DHF/dengue shock syndrome (13). There are thus two reasons to find a simple test to distinguish between primary and secondary infection with early serum samples. The first reason is to be able to carry out an epidemiological study to check whether the incidence of severe cases of dengue is significantly higher among secondary infections than among primary infections (7, 24). The second reason is to know the immunological status of patients to allow clinicians to take it into account in the progression of the disease until the ADE hypothesis has been confirmed or disproved.
The HI test is still recommended by the WHO as a help for distinguishing primary from secondary infection (27). The technical difficulties of this test mean that it cannot be used routinely, especially during epidemics. Furthermore, it has a couple of disadvantages: the need to take two samples at an appropriate time interval and a strong cross-reaction with other flavivirus-specific IgG antibodies (1). In the absence of a second sample, it is not possible to determine the immunological status of patients or to know whether some primary infections are misclassified as secondary infections because they present very high levels of antibodies even in the first days of infection (2, 24).
Other techniques have been proposed, including an ELISA based on the IgM/IgG ratio that allows rapid differential diagnosis (17, 25). However, the persistence of IgM for more than 8 months after infection can result in interpretation errors.
The level of avidity of serum samples, based on the strength of the affinity of antibodies for the pathogen, appears to be an excellent alternative. The studies carried out by De Souza et al. have shown for the first time that, by using a commercial kit for serological dengue diagnosis, it was possible to discriminate between a case of primary dengue infection and a case of secondary dengue infection by detecting avid IgG (8). Our results, obtained with a serological test developed in our laboratory, confirm those obtained by De Souza. Indeed, during the infectious episode, we observed a mean avidity of 25.9% during a primary infection and 66.3% during a secondary infection. The sensitivity and specificity of the test were 82.7 and 77.5%, respectively, with a single sample. If we focused on serum samples collected between days 3 and 7 of infection, during which most clinical complications of the disease occur, the sensitivity and specificity of the test increased to 95.1 and 80.0%, respectively. Moreover, it is likely that the performances of this test are underestimated. Indeed, since we had no alternative to the HI test, it is highly probable that some of the 138 measures of avidity obtained with sera defined as “secondary infection” used to evaluate the sensitivity and specificity of the method were in fact primary infections.
The avidity test, which is easier to carry out than the HI test, may become a useful reference technique for clinicians and epidemiologists because it can discriminate between primary and secondary infections with just one acute-phase serum sample. To definitively evaluate the performances of this test, it is necessary to obtain samples from patients for whom the diagnosis of primary or secondary infection is not based solely on the HI test. For example, for patients with second dengue infection, complementary informations, such as whether the patient previously tested positive for the virus, would be more informative. This method will eliminate the incertitude linked to the HI test interpretation criteria.
Acknowledgments
We thank V. Lacoste and J. M. Heraud, Institut Pasteur de la Guyane, Cayenne, French Guiana, for helpful discussions.
REFERENCES
- 1.Allwinn, R., H. W. Doerr, P. Emmerich, H. Schmitz, and W. Preiser. 2002. Cross-reactivity in flavivirus serology: new implications of an old finding? Med. Microbiol. Immunol. 190:199-202. [DOI] [PubMed] [Google Scholar]
- 2.Barnes, W. J., and L. Rosen. 1974. Fatal hemorrhagic disease and shock associated with primary dengue infection on a Pacific island. Am. J. Trop. Med. Hyg. 23:495-506. [DOI] [PubMed] [Google Scholar]
- 3.Bundo, K., and A. Igarashi. 1985. Antibody-capture ELISA for detection of immunoglobulin M antibodies in sera from Japanese encephalitis and dengue hemorrhagic fever patients. J. Virol. Methods 11:15-22. [DOI] [PubMed] [Google Scholar]
- 4.Chen, W. J., K. P. Hwang, and A. H. Fang. 1991. Detection of IgM antibodies from cerebrospinal fluid and sera of dengue fever patients. Southeast Asian. J. Trop. Med. Public Health 22:659-663. [PubMed] [Google Scholar]
- 5.Chow, L., and S. T. Hsu. 1989. MAC-ELISA for the detection of IgM antibodies to dengue type I virus. Chin. J. Microbiol. Immunol. 22:278-285. [PubMed] [Google Scholar]
- 6.Clarke, D. H., and J. Casals. 1958. Techniques for hemagglutination and hemagglutination-inhibition with arthropod-borne viruses. Am. J. Trop. Med. Hyg. 7:561-573. [DOI] [PubMed] [Google Scholar]
- 7.Deparis, X., C. Roche, B. Murgue, and E. Chungue. 1998. Possible dengue sequential infection: dengue spread in a neighborhood during the 1996/97 dengue-2 epidemic in French Polynesia. Trop. Med. Int. Health. 3:866-871. [DOI] [PubMed] [Google Scholar]
- 8.De Souza, V. A., S. Fernandes, E. S. Araujo, A. F. Tateno, O. M. Oliveira, R. R. Oliveira, and C. S. Pannuti. 2004. Use of an immunoglobulin G avidity test to discriminate between primary and secondary dengue virus infections. J. Clin. Microbiol. 42:1782-1784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Grangeot-Keros, L., M. J. Mayaux, P. Lebon, F. Freymuth, G. Eugene, R. Stricker, and E. Dussaix. 1997. Value of cytomegalovirus (CMV) IgG avidity index for the diagnosis of primary CMV infection in pregnant women. J. Infect. Dis. 175:944-946. [DOI] [PubMed] [Google Scholar]
- 10.Gubler, D. J. 1998. Dengue and dengue hemorrhagic fever. Clin. Microbiol. Rev. 11:480-496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Guzman, M. G., and G. Kouri. 2002. Dengue: an update. Lancet Infect. Dis. 2:33-42. [DOI] [PubMed] [Google Scholar]
- 12.Halstead, S. B. 1988. Pathogenesis of dengue: challenges to molecular biology. Science 239:476-481. [DOI] [PubMed] [Google Scholar]
- 13.Halstead, S. B., and E. J. O'Rourke. 1977. Dengue viruses and mononuclear phagocytes. I. Infection enhancement by non-neutralizing antibody. J. Exp. Med. 146:201-217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hedman, K., and S. A. Rousseau. 1989. Measurement of avidity of specific IgG for verification of recent primary rubella. J. Med. Virol. 27:288-292. [DOI] [PubMed] [Google Scholar]
- 15.Hedman, K., A. Vaheri, and M. Brummer-Korvenkontio. 1991. Rapid diagnosis of hantavirus disease with an IgG-avidity assay. Lancet 338:1353-1356. [DOI] [PubMed] [Google Scholar]
- 16.Hommel, D., A. Talarmin, V. Deubel, J. M. Reynes, M. T. Drouet, J. L. Sarthou, and A. Hulin. 1998. Dengue encephalitis in French Guiana. Res. Virol. 149:235-238. [DOI] [PubMed] [Google Scholar]
- 17.Innis, B. L., A. Nisalak, S. Nimmannitya, S. Kusalerdchariya, V. Chongswasdi, S. Suntayakorn, P. Puttisri, and C. H. Hoke. 1989. An enzyme-linked immunosorbent assay to characterize dengue infections where dengue and Japanese encephalitis co-circulate. Am. J. Trop. Med. Hyg. 40:418-427. [DOI] [PubMed] [Google Scholar]
- 18.Kalayanarooj, S., D. W. Vaughn, S. Nimmannitya, S. Green, S. Suntayakorn, N. Kunentrasai, W. Viramitrachai, S. Ratanachu-eke, S. Kiatpolpoj, B. L. Innis, A. L. Rothman, A. Nisalak, and F. A. Ennis. 1997. Early clinical and laboratory indicators of acute dengue illness. J. Infect. Dis. 176:313-321. [DOI] [PubMed] [Google Scholar]
- 19.Lanciotti, R. S., C. H. Calisher, D. J. Gubler, G. J. Chang, and A. V. Vorndam. 1992. Rapid detection and typing of dengue viruses from clinical samples by using reverse transcriptase-polymerase chain reaction. J. Clin. Microbiol. 30:545-551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Laur, F., B. Murgue, X. Deparis, C. Roche, O. Cassar, and E. Chungue. 1998. Plasma levels of tumour necrosis factor alpha and transforming growth factor beta-1 in children with dengue 2 virus infection in French Polynesia. Trans. R. Soc. Trop. Med. Hyg. 92:654-656. [DOI] [PubMed] [Google Scholar]
- 21.Murgue, B., X. Deparis, E. Chungue, O. Cassar, and C. Roche. 1999. Dengue: an evaluation of dengue severity in French Polynesia based on an analysis of 403 laboratory-confirmed cases. Trop. Med. Int. Health. 4:765-773. [DOI] [PubMed] [Google Scholar]
- 22.Murgue, B., C. Roche, E. Chungue, and X. Deparis. 2000. Prospective study of the duration and magnitude of viraemia in children hospitalized during the 1996-1997 dengue-2 outbreak in French Polynesia. J. Med. Virol. 60:432-438. [DOI] [PubMed] [Google Scholar]
- 23.Reynes, J. M., A. Laurent, V. Deubel, E. Telliam, and J. P. Moreau. 1994. The first epidemic of dengue hemorrhagic fever in French Guiana. Am. J. Trop. Med. Hyg. 51:545-553. [PubMed] [Google Scholar]
- 24.Rosen, L. 1977. The Emperor's New Clothes revisited, or reflections on the pathogenesis of dengue hemorrhagic fever. Am. J. Trop. Med. Hyg. 26:337-343. [DOI] [PubMed] [Google Scholar]
- 25.Vaughn, D. W., A. Nisalak, T. Solomon, S. Kalayanarooj, M. D. Nguyen, R. Kneen, A. Cuzzubbo, and P. L. Devine. 1999. Rapid serologic diagnosis of dengue virus infection using a commercial capture ELISA that distinguishes primary and secondary infections. Am. J. Trop. Med. Hyg. 60:693-698. [DOI] [PubMed] [Google Scholar]
- 26.Ward, K. N., J. J. Gray, M. E. Joslin, and M. J. Sheldon. 1993. Avidity of IgG antibodies to human herpesvirus-6 distinguishes primary from recurrent infection in organ transplant recipients and excludes cross-reactivity with other herpesviruses. J. Med. Virol. 39:44-49. [DOI] [PubMed] [Google Scholar]
- 27.World Health Organization. 1997. Dengue haemorrhagic fever: diagnosis; treatment and control. World Health Organization, Geneva, Switzerland.