Abstract
Sodium bicarbonate (SB) supplementation is an ergogenic strategy for athletes competing in high-intensity exercise, but the efficacy of SB for accelerating recovery from exercise and thus improving performance during repeated bouts of exercise is not fully understood. In a similar fashion to using SB as a pre-exercise buffer, it is possible accelerated restoration of blood pH and bicarbonate following an exercise bout mechanistically underpins the use of SB as a recovery aid. Physiological mechanisms contributing to beneficial effects for SB during repeated bout exercise could be more far-reaching however, as alterations in strong ion difference (SID) and attenuated cellular stress response might also contribute to accelerated recovery from exercise. From inspection of existing literature, ingestion of 0.3 g kg−1 body mass SB ~60–90 min pre-exercise seems to be the most common dosage strategy, but there is evidence emerging for the potential application of post-exercise supplementation timing, gradual SB doses throughout a competition day, or even ingestion during exercise. Based on this review of literature, an SB ingestion recovery framework is proposed to guide athletes and practitioners on the use of SB to enhance performance for multiple bouts of exercise.
Keywords: Acid–base balance, Recovery, Repeated bout exercise, Supplements
Introduction
Sodium bicarbonate (SB) supplementation prior to exercise is an ergogenic strategy for improving athletic performance during single and repeated bouts of high-intensity exercise (Carr et al. 2011a; de Oliveira et al. 2022). Unlike many commercially available sport supplements, there is empirical evidence supporting the efficacy of SB, and thus, it was recommended for use by athletes in the 2018 International Olympics Committee (IOC) consensus statement (Maughan et al. 2018). Ergogenic benefits following pre-exercise SB ingestion have traditionally been linked to the assumption that accumulation of hydrogen cations (H+) in the cytosol of muscle fibres, which by definition computes to a decrease in intracellular pH, may contribute towards skeletal muscle fatigue (Fitts 2016). Whilst the deleterious effects of H+ accumulation on skeletal muscle function have been debated (Westerblad 2016), it is proposed that offsetting declining intramuscular pH during intense exercise prevents the inhibition of metabolic processes required to generate adenosine triphosphate (ATP) (Spriet et al. 1989; Messonnier et al. 2007) and sustain rates of contractile cycling (Debold et al. 2008).
Ingesting SB augments the body’s extracellular buffering capacity by increasing blood bicarbonate (HCO3−) concentration, subsequently allowing for greater efflux of H+ from contracting muscles and thus protecting against biochemical disturbances to intramuscular acid–base balance (Hollidge-Horvat et al. 2000; Bishop et al. 2004). SB ingestion has also been shown to elicit changes in intra- and extracellular distribution of ions [e.g., sodium (Na+), potassium (K+), calcium (Ca2+), and chloride (Cl−)] that may preserve muscle excitability during intense exercise (Cairns and Lindinger 2008; Kent-Braun et al. 2012). Since there is likely no singular mechanism underpinning the performance enhancing effects of SB ingestion, it is important researchers adopt a multifaceted perspective when studying physiological systems that may contribute.
Most independent placebo-controlled studies and review articles have focused on physiological responses and ergogenic benefits for SB from its context as a pre-exercise extracellular buffer. A brief inspection of scientific literature reveals that a considerable number of research studies (~200) have been conducted investigating the effect of SB ingestion on exercise performance. Work dates back as far as 1930 when Dennig et al. (1931) first reported pre-exercise SB improved accumulated oxygen debt during 15 min steady-state running. Throughout the past decade, numerous meta-analyses have shown pre-exercise SB ingestion to elicit moderate performance benefits, with the greatest improvements thought to exist during exercise tasks lasting between 45 s and 10 min (Carr et al. 2011a; Peart et al. 2012; Hadzic et al. 2019; de Oliveira et al. 2022). Compared to this widely accepted use of SB ingestion as a pre-exercise ergogenic aid, less focus has been given to the potential application of SB for improving recovery between two or more exercise bouts. From our experience of talking to members of the scientific community and applied practitioners, it is evident that SB is still viewed as a pre-exercise extracellular buffering aid. As scientific literature becomes increasingly overwhelmed by research studies examining the effect of SB on exercise performance, it is believed that shifting the narrative more towards studies examining whether SB is able to accelerate recovery from exercise will provide researchers with a fresh perspective and maximise practical implications for athletes.
One challenge, however, is precisely defining what “recovery from exercise” means from a physiological viewpoint. In short, it can be termed as the amount of time between finishing an initial exercise bout and the subsequent restoration of physiological systems towards baseline levels (Luttrell and Halliwill 2015). That being said, the temporal definition of “recovery” varies depending on the physiological system or pathway being studied. In light of these different interpretations of “recovery from exercise”, it is important to provide a clear explanation of how SB may accelerate recovery between two or more bouts of exercise. In the context of SB ingestion and recovery, numerous studies have showed SB ingestion prior to an initial exercise bout elevates blood pH and [HCO3−] during recovery periods lasting 15–40 min (Siegler et al. 2008; Pruscino et al. 2008; Gough et al. 2018). Considering the potential role of extracellular buffering capacity for protecting against declining intramuscular pH and offsetting skeletal muscle fatigue (Fitts 2016), accelerating restoration of blood acid–base balance prior to subsequent exercise bouts might be crucial to maximising performance. In a similar fashion, there is a small body of literature suggesting that pre-exercise SB ingestion may accelerate the rate at which strong ion difference (SID) recovers to baseline levels after exercise (Sostaric et al. 2006; Gough et al. 2019a, b). These ionic changes are thought to have important implications for preserving muscle excitability (Cairns and Lindinger 2008), which may contribute to force generating capacity. From a longer term sense on a “recovery” continuum, a series of studies investigated whether SB was able to attenuate cellular stress responses and oxidative stress up to 24 h following exercise (Peart et al. 2011, 2013a, 2016). The authors reported that SB ingestion alleviates production of reactive oxygen species (ROS) (Peart et al. 2011), and this might reduce oxidative stress and improve skeletal muscle function (Powers and Jackson 2008).
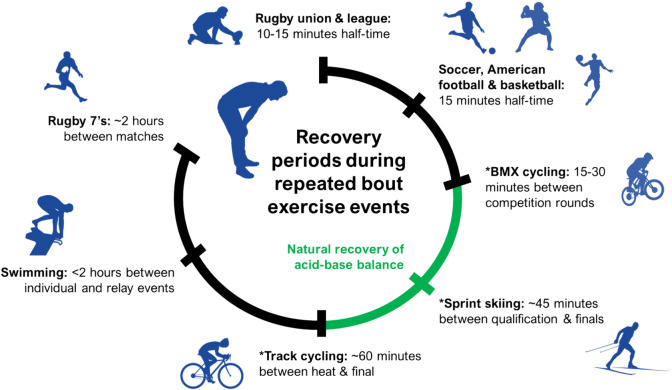
Although further research is required to better understand how SB ingestion influences these physiological mechanisms and consequently accelerates “recovery from exercise”, there is a strong ‘real-world’ rationale for the use of SB as a recovery aid. Assuming that SB restores homeostasis of physiological pathways that contribute to skeletal muscle fatigue during an initial exercise bout, then logically SB ingestion would be an ergogenic strategy for athletes required to perform multiple exercise bouts on the same day (Fig. 1). For instance, sporting disciplines such as swimming and BMX/track cycling that see athletes take part in heat/qualification rounds and finals throughout a single competition day (Mero et al. 2013; Peinado et al. 2019). There might also be scope for using SB as a recovery aid during sports such as rugby sevens and cross-country skiing, with both characterised by excessive H+ accumulation during exercise and featuring short recovery periods between repeated exercise bouts (Ross et al. 2014; Losnegard 2019). Application of SB during these sports is yet to receive scientific interest, but it is hoped that this review will educate readers on the potential beneficial effects of SB ingestion as a recovery aid and help conceptualise future scientific work across a variety of sporting disciplines.
Fig. 1.
Schematic representation of sporting disciplines that may benefit from using SB as a recovery aid. Approximate timescales between repeated bouts of exercise presented as a chronological sequence. Green section denotes estimated duration required for blood acid–base balance status to recover to baseline following high-intensity exercise. * Denotes the timeframe that a single SB dose has improved recovery and performance in studies to date
In light of the potential benefits of SB ingestion for accelerating “recovery from exercise”, this review will first discuss how SB ingestion could influence recovery of three pertinent physiological systems/pathways: (1) acid–base balance, (2) SID, and (3) cellular stress responses and oxidative stress. From an applied perspective, it is crucial to produce clear recommendations of how to use SB for athletes competing in multiple exercise bouts on the same day. As such, second, we will outline the significance of dosage, ingestion strategy and timing for optimising blood buffering capacity and repeated bout exercise performance. Additionally, this review will provide a comprehensive evaluation of findings from placebo-controlled research studies investigating the effect of SB ingestion on repeated bout exercise performance. For the purpose of this review, we are defining repeated bout exercise performance as “two or more exercise events performed on the same day separated by at least 10 min recovery”. This temporal pattern of “recovery” was based off previous studies examining the effect of SB on acid–base balance recovery, whereby ~10 min following an initial exercise bout is when blood pH and [HCO3−] started their recovery towards baseline (Verbitsky et al. 1997; Siegler et al. 2008). Whilst outside the scope of this review, potential ergogenic benefits of SB ingestion during repeated and intermittent exercise comprising shorter work/recovery ratios have been discussed elsewhere (Lopes-Silva et al. 2019; Grgic 2022). Finally, based on the existing literature, an SB ingestion framework will be presented for athletes and coaches that wish to incorporate the supplement into their nutritional regimes.
Proposed mechanisms for accelerating recovery from exercise
In a similar fashion to studies examining physiological mechanisms underpinning the use of SB as a pre-exercise buffer (Siegler et al. 2016), greatest attention has been given to effect of SB ingestion on the recovery of blood acid–base balance after exercise (Pruscino et al. 2008; Gough et al. 2019a, b). Early work by Costill et al. (1984) observed that compared to a placebo, ingesting SB prior to 5 × 1 min cycling bouts increased blood pH [~0.05 arbitrary units (au)] and [HCO3−] (~4 mmol L−1) after 30 min recovery (both p<0.05). In agreement with these findings, studies have shown SB ingested pre-exercise to elevate blood pH and [HCO3−] during recovery periods ranging between 15 and 40 min post-exercise (Pruscino et al. 2008; Siegler et al. 2008; Gough et al. 2018). Additionally, higher blood pH and [HCO3−] have been reported following 75–90 min recovery when SB was ingested after an initial exercise bout (Gough et al. 2017a, 2019a, b).
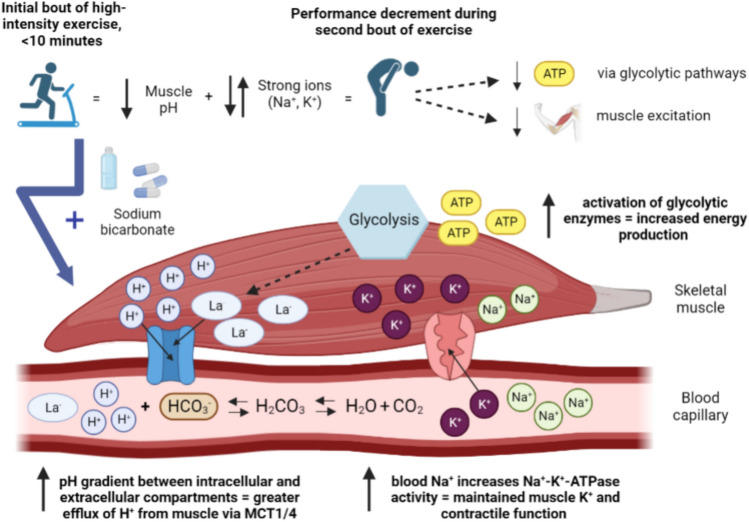
It should be noted that the aforementioned studies all focused on changes occurring in extracellular compartments, but evidence from early animal model work suggests that similar responses may occur within intramuscular compartments following alkalosis (Spriet et al. 1985, 1986; Bishop et al. 2004). As such, is it reasonable to assume that SB ingestion would also have raised intramuscular acid–base balance prior to subsequent exercise, which could protect against the inhibition of key glycolytic enzymes (i.e., glycogen phosphorylase) during subsequent exercise, thus increasing the rate of glycolytic flux and ATP production (Fig. 2) (Messonnier et al. 2007; Fitts 2016). This could be particularly true when only a limited recovery window is available and athletes without SB ingestion would commence the second bout in a fatigued state and their acid–base balance would be below baseline levels.
Fig. 2.
Schematic outlining a focused viewpoint of how changes in acid–base balance and strong ions mechanistically underpin performance benefits for repeated bouts of high-intensity exercise following SB ingestion. Top section: exercise-induced disruptions to acid–base balance and strong ion difference. Bottom section: physiological changes (shown in bold) following SB ingestion that contribute towards improvements in performance. (Schematic created in BioRender.com)
Whilst current findings seem to suggest that ingestion of SB is an effective strategy for elevating blood acid–base balance status between repeated bouts of exercise, it might be misleading to base conclusions regarding its efficacy from studies with only a few blood sampling points during recovery. Robergs et al. (2005) comprehensively examined short- (i.e., 2 min sampling points for the first 20 min) and long-phase (i.e., 4 min sampling points for the final 40 min) blood pH and HCO3− recovery pharmacokinetics throughout a 60 min recovery period after pre-exercise SB ingestion. These researchers demonstrated that SB increased blood pH and [HCO3−] up to 60 min after time-to-exhaustion (TTE) cycling compared to a placebo. Interestingly, they also reported that blood pH recovery on monoexponential slopes (~0.6) and half-time constants (t0.5 = ~12-min) were not different between SB and placebo. In short, these results from Robergs et al. (2005) suggest that the higher blood pH and [HCO3−] reported following SB ingestion during recovery from intense exercise might not be related to accelerated blood acid–base balance recovery pharmacokinetics per se, but instead to reduced acid–base balance perturbations during the initial exercise bout (Bishop et al. 2004).
Since most studies report significantly higher blood pH and [HCO3−] immediately post-exercise after SB supplementation, it is possible that dampened metabolic perturbation during the initial exercise bout contributes to acid–base balance recovering back to baseline levels faster (i.e., recovery starts from a higher level). However, studies that have adopted a post-exercise ingestion strategy to ensure SB supplementation occurred from a state of metabolic acidosis consistent with placebo suggest SB does accelerate recovery (Gough et al. 2019a, b; Gurton et al. 2021a). Post-exercise ingestion of SB elevated blood acid–base balance prior to and/or after a second exercise bout compared to a placebo, but the lack of blood sampling points during recovery (i.e., only measured at the start and end) meant that it was difficult to evaluate the efficacy of SB for accelerating pH and HCO3− recovery pharmacokinetics. Future studies should replicate the methodology used by Robergs et al. (2005) to examine the effect of post-exercise SB on acid–base balance recovery pharmacokinetics (i.e., monoexponential slopes, half-time constants) to unpick the mechanism further.
Another mechanism that may underpin the application of SB ingestion as a recovery aid are alterations to SID (Sostaric et al. 2006; Gough et al. 2019a, b). Disturbances to the SID within skeletal muscle and surrounding interstitium during intense exercise contribute towards muscle fatigue by depressing maximal Na+,K+-ATPase activity, subsequently impairing cell membrane excitability (Fraser et al. 2002; Sostaric et al. 2006). SB ingestion may increase influx of K+ into muscles and attenuate exercise-induced release of K+ back into extracellular mediums (Siegler and Gleadall-Siddall 2010). Combined with increased muscular Cl− uptake and plasma [Na+] following SB (Fig. 2) (Siegler and Hirscher 2009; Gough et al. 2017b), it is possible that SB upregulates Na+/K+-ATPase and Na+/K+/2Cl−-ATPase activity to limit depolarization and preserve excitation–contraction coupling (Allen et al. 2008). In vitro experimental models have demonstrated that stimulating Na+–K+-ATPase activity delays muscle fatigability and accelerates subsequent recovery (Clausen and Everts 1991; Clausen et al. 1993), and therefore, SB mediated changes in SID during initial fatiguing exercise tasks could also be beneficial to repeated exercise performance. Indeed, Sostaric et al. (2006) examined the effect of SB on SID during exhaustive dynamic forearm exercise and found that plasma [Na+] and muscle Cl− uptake were higher for SB compared with a control. Somewhat unexpectedly, SB increased release of K+ from muscles into plasma at fatigue (~49% higher compared with control; p<0.05). This finding could partly be attributed to greater work completed during the exhaustive exercise task (25% greater than placebo; p<0.05). On the other hand, Sostaric et al. (2006) also observed that SB ingestion elevated uptake of K+ into the muscles compared with a placebo throughout a 10-min recovery (~17% higher), which the authors attributed to the higher K+ release at fatigue following SB ingestion and the opening of some K+ channels owing to augmented Na+, K+-ATPase (and possibly Na+/K+/2Cl−-ATPase) activity. However, as Sostaric et al. (2006) investigated a finger flexion exercise task, it remained unclear for years whether similar changes would be observed for whole-body repeated bout exercise.
More recently, in a series of investigations, Gough et al. (2018, 2019a, b) explored the effect of SB on SID during ecologically valid exercise protocols [i.e., 4-km time trials (TT), boxing simulation]. Studying SID instead of individual changes in SID allows the collective assessment of the balance of fully dissociated cations and anions in intra- and extracellular fluid (Stewart 1983). In two similar studies, Gough et al. (2018, 2019a) observed that SB accelerated restoration of arterialised (finger) blood SID during 40-min recovery periods after 4-km cycling TT in normobaric hypoxic conditions (~5% greater than placebo; p<0.05). Expanding upon these findings, Gough et al. (2019b) demonstrated higher blood SID (~10% greater than placebo; p<0.001) after 75 min recovery following a TTE running/boxing simulation exercise task. These researchers attributed accelerated recovery of SID to elevated blood [Na+] concomitant with reductions in blood [K+] and [Cl−] for SB compared to the placebo, consequently inferring increased uptake of K+ and Cl− into the muscles to preserve excitation–contraction coupling and action potentials (Fig. 2). Whilst both studies employed a second bout of exercise and reported performance benefits for SB, only Gough et al. (2019b) showed SID to remain elevated for SB compared to placebo, in turn raising questions about whether collective changes in SID significantly influence repeated bout exercise performance. Interestingly, Gough et al. (2018) observed that blood [K+] was higher for SB after their second 4-km cycling TT, reinforcing previous research that changes in [K+] may play the most important role of the strong ions for sustaining muscle excitability (Cairns and Lindinger 2008).
Since muscle force is substantially depressed by reduced intracellular-to-extracellular [K+] ratio (Cairns et al. 1995; Nielsen et al. 1998), it is likely lower plasma [K+] after SB ingestion contributes to maintaining a high intracellular-to-extracellular [K+] ratio (Sostaric et al. 2006). Of note, however, is that the results from Gough et al. (2018, 2019a, b) only reveal changes occurring in extracellular compartments. This does not directly confirm SB protected against ionic disturbances within contracting muscle, leaving it as an avenue for future research to explore.
Finally, there is a small body of scientific literature examining the effect of pre-exercise SB on physiological stress responses during recovery from high-intensity exercise (Peart et al. 2011, 2013a, b, 2016). Researchers have focused on the relationship between pre-exercise alkalosis and cellular stress responses [i.e., intracellular heat shock protein 72 (HSP72), oxidative stress]. Briefly, expression of intramuscular HSP72 increases after high-intensity exercise to protect against stress-induced apoptosis (Mosser et al. 1997) due to production of reactive oxygen species (ROS) via cellular respiration (Taylor et al. 2011). Since in vitro experimental models have demonstrated that HSP72 response is also linked to an intramuscular acidosis (Gapen and Moseley 1995), it was theorized that greater efflux of H+ from cells following SB may dampen HSP72 expression after intense exercise. Peart et al. (2011) demonstrated that SB decreased HSP72 response after “all-out” anaerobic cycling (~40% lower than placebo; p=0.013). They also reported reduced post-exercise lipid peroxidation [via plasma thiobarbituric acid reactive substances (TBARS)] following SB. Attenuated HSP72 for SB could be attributed to reduced oxidative stress, as greater efflux of H+ from intramuscular compartments alleviates production of ROS during cellular respiration that would otherwise impair skeletal muscle function (Powers and Jackson 2008). Peart et al. (2011) acknowledged that their results should be interpreted with caution however, since TBARS is not exclusively related to lipid peroxidation, meaning that it might overestimate oxidative stress (Koster et al. 1985; Powers et al. 2010). Across two follow-up studies, Peart et al. (2013a, b) examined the effect of SB ingestion on exercise-specific markers of oxidative stress, including total antioxidant capacity, total glutathione (TGSH), and oxidised glutathione (GSSG). SB attenuated HSP72 expression and TBARS concentration after repeated sprint cycling, although no differences were reported between treatments for either total antioxidant capacity or the ratio of GSSG/TSGH. In light of these results, Peart et al. (2013a, b) concluded that the maintenance of intracellular acid–base balance following SB ingestion is likely the primary factor contributing to reductions in HSP72 expression, as opposed to changes in ROS generation.
Interestingly, recent findings from Thomas et al. (2023) demonstrated that SB decreased oxidative stress (24 h post-exercise protein carbonylation ~30% lower than placebo; p<0.05) after performing 3 × 30 s Wingate cycling tests. Additional research is required to further understand the effect of SB ingestion on post-exercise oxidative stress, but regardless of the underlying physiological mechanisms, dampening HSP72 responses after an initial exercise task could have practical implications for recovery as theoretically athletes would experience less physiological stress during subsequent exercise bouts.
Importantly, depending on the specific situation, the prior results suggest that SB could be used to reduce exercise stress (i.e., during competition) or enhance physiological adaptations from training. This hypothesis was studied during work by Peart et al. (2016) that investigated whether SB mediated changes in post-exercise HSP72 expression influenced cellular stress responses following a second bout of high-intensity exercise (bout 1: 10 × 15 s sprints; bout 2:90 min intermittent sprint cycling). HSP72 expression was reduced for SB following the first exercise bout (~25% lower than placebo; p=0.05), but was similar between treatments after the second bout. Whilst these results suggest that improvements in intramuscular acid–base balance do not reduce cellular stress responses during repeated exercise bouts, it is possible that the magnitude of HSP72 expression following their initial exercise task was not high enough to influence physiological stress responses during subsequent exercise (Taylor et al. 2011). Additional studies are required to examine the effect of SB on post-exercise oxidative stress, including ingestion during heavy workloads (i.e., especially in competition) that induce substantial HSP72 expression.
Dosage, ingestion strategy, and timing
Researchers examining the effect of SB on recovery from exercise have typically chosen 0.2–0.3 g kg−1 BM doses (Costill et al. 1984; Pierce et al. 1992; Gough et al. 2017a). Similarities likely originate from early work by McNaughton (1992) that proposed 0.3 g kg−1 BM SB ingested pre-exercise induced a sufficient blood alkalosis ([HCO3−]: ~31 mmol L−1), without causing severe gastrointestinal (GI) discomfort unlike their 0.4–0.5 g kg−1 BM doses. Since more contemporary research has suggested that 0.2 g kg−1 BM SB elicits substantial changes in [HCO3−] and alleviates GI discomfort compared to 0.3 g kg−1 BM (Gough et al. 2018; Gurton et al. 2020), it could be an attractive dose for athletes known to suffer from side effects (Gough et al. 2018; Gurton et al. 2020). The composition of SB is ~27% Na+ (assuming 1 mol SB is 84 g mol−1), meaning that a 70 kg athlete ingesting 0.4 g kg−1 BM SB would consume ~7500 mg of Na+, which exceeds the recommended upper limit of 2300 mg for daily intake (Bibbins-Domingo 2014). As such, athletes regularly consuming SB could be at higher risk of developing cardiovascular complications (i.e., hypertension) linked to excessive dietary Na+ intake (Strazzullo et al. 2009). Whilst outside the scope of this review, future studies should examine the impact of chronic SB supplementation on long-term health outcomes.
Traditionally, most placebo-controlled studies have opted to administer SB in solution or capsules (gelatine/vegetarian/enteric-coated) (Gurton et al. 2023a), but within recent years, topically applied lotion (Gurton et al. 2023b) and minitabs (Gough and Sparks 2024) have also emerged within the literature. Regardless of personal preferences for dose and supplement timing, solution and capsule administration approaches are considered best practice (de Oliveira et al. 2022). A key determinant of the ingestion method used by researchers and/or practitioners is the resulting GI discomfort. In a study examining eight SB supplementation approaches, Carr et al. (2011b) reported that solution ingestion protocols cause more severe side effects compared to capsules. These authors concluded that coingestion of SB alongside a carbohydrate meal (~1.5 g kg−1 BM) further reduced the occurrence of GI discomfort, although does not guarantee side effects will be eliminated completely (Carr et al. 2011b). It has also been suggested that splitting SB into multiple doses prior to exercise can alleviate GI discomfort, whilst still inducing a sufficient pre-exercise alkalosis (HCO3− ~32 mmol L−1; pH ~7.50 au; Saunders et al. 2014). One drawback, however, is that a split-dosage approach may not be effective if ingestion of SB is needed after an initial exercise bout, as there would likely be insufficient time available to see elevated acid base balance recovery pharmacokinetics.
Recently, two novel forms of SB administration, topically applied lotion (PR Lotion; Momentous) and minitabs with a hydrogel carbohydrate solution (Maurten, Sweden) have emerged within the literature. To date, equivocal findings exist for topical SB (McKay et al. 2020; Gibson et al. 2023), but Gurton et al. (2023b) observed improvements in 8 × 25 m repeated running sprint times for topical SB (~2%; p=0.036) without any GI discomfort. On the other hand, Gough and Sparks (2024) reported that aggregated GI discomfort (i.e., sum of all GI symptoms) was reduced for a hydrogel minitab SB delivery approach versus traditional vegetarian capsules (9±9 vs. 85±63 au; p=0.003; ES=1.62). These novel approaches have not yet been applied to a recovery context; therefore, further research studies are needed before conclusions can be made regarding their efficacy.
Researchers examining the effect of SB ingestion on repeated bout exercise performance have traditionally opted to give the supplement as a single dose ~60–90 min prior to an initial exercise bout, irrespective of whether they use solution or capsule SB administration approaches (Pierce et al. 1992; Zabala et al. 2008, 2011; Mero et al. 2013). Similarities in supplement timing can be traced back to early work examining the effect of SB on blood pH and [HCO3−] recovery (Costill et al. 1984; Katz et al. 1984). Interestingly though, studies profiling interindividual differences in blood pH and [HCO3−] responses after 0.3 g kg−1 BM SB suggest that peak changes occur ~60–90 min when adopting a solution approach (Gough et al. 2017b; Deb et al. 2018) and between 120 and 240 min when SB is consumed via capsules (Jones et al. 2016; de Oliveira et al. 2020). Given this large degree of variability for HCO3− absorption, aligning SB ingestion with pre-determined individualised time-to-peak [HCO3−] has been recommended (Boegman et al. 2020; Gurton et al. 2021b; Gough et al. 2021). In a series of investigations, Gough et al. (2018, 2019a) studied the effect of individualised SB ingestion on blood buffering capacity during cycling TT’s. Their preliminary study showed that SB increased blood pH and [HCO3−] compared with a placebo after 40-min recovery from a 4-km cycling TT in hypoxia (~0.08 au and 7 mmol L−1, respectively; p<0.05). In their follow-up study, Gough et al. (2019a) demonstrated that SB accelerated the restoration of [HCO3−] between 2 × 4-km cycling TT’s (6.1 mmol L−1 higher than placebo; p<0.001) separated by 40 min, with elevated blood buffering capacity likely contributing to improved performance during a second 4-km TT. Despite these promising findings, it must be considered that not all athletes have access to blood gas analysis (i.e., financial and/or logistical restraints), and therefore, alternative supplementation timing approaches potentially able to maximise blood buffering capacity during repeated bout exercise events should also be investigated.
SB ingestion has also been administered using a split-dose strategy, including early research by Pruscino et al. (2008) reporting higher [HCO3−] compared to the placebo at the end of a 30-min recovery period between repeated 200-m swimming TTs (~7 mmol L−1; p<0.05). In a study examining the effect of SB on repeated “all-out” cycling performance across a single day, Dalle et al. (2019) divided SB supplementation into three identical 3 h cycles: a 6.3 g bolus at the start, followed by 2.1 g doses at +1 h and +2 h, which equated to 0.4 g kg−1 BM SB being consumed over 9 h. Importantly, these authors observed elevated blood pH and [HCO3−] at the end of a 9-h competition day (pH: ~7.47 au; HCO3−: ~31 mmol L−1). The concept of “topping up” with smaller SB doses raised by Dalle et al. (2019) could have important practical implications for athletes performing multiple exercise events separated by shorter recovery durations (e.g., ~60–90 min between track cycling heats and final). It is logical to assume that ~0.2–0.3 g kg−1 BM SB ingested pre-exercise combined with a “top up” dose of ~0.1 g kg−1 BM SB immediately at the start of recovery will lead to more pronounced changes in blood pH and [HCO3−] prior to subsequent exercise (compared with only a single SB dose consumed before the initial exercise bout). On the other hand, athletes could consume these small “top up” doses during exercise to replenish [HCO3−] throughout a competition event (Dalle et al. 2021). Dose–response blood acid–base balance pharmacokinetics data indicate that ~2–3 mmol L−1 changes in [HCO3−] occur after ~40 min for 0.1 g kg−1 BM SB (Jones et al. 2016), which suggests that the use of “top up” SB doses during a competition event would likely be limited to endurance exercise (e.g., Grand Tour cycling stages, ~3–4 h) as sufficient time is needed for changes in [HCO3−] to occur.
Another timing strategy that has emerged within scientific literature is post-exercise SB ingestion (Gough et al. 2017a, 2019a, b; Gurton et al. 2021a). This theoretically ensures that enhanced HCO3− buffering is present throughout recovery, as opposed to the initial exercise bout, thus maximising any benefits during subsequent exercise bouts. From a practical perspective, post-exercise SB timing could be of interest to athletes performing multiple events/training sessions on the same competition day, for instance when a heat and/or qualification round is followed by a final of greater importance. During their initial study, Gough et al. (2017a) showed that post-exercise SB elevated blood pH and [HCO3−] (~0.10 au, 7.0 mmol L−1; p<0.001) compared with placebo at the end of a 90 min recovery period between two TTE cycling tasks. However, as the authors waited until 30 min post-exercise to administer SB, blood pH and [HCO3−] had recovered close to baseline prior to supplementation for both conditions, meaning that it was difficult to draw conclusions for the efficacy of post-exercise SB at accelerating blood acid–base balance recovery. In a follow-up study, Gough et al. (2019b) reported elevated [HCO3−] (8.0±2.1 mmol L−1 higher than placebo) after 75-min recovery when SB was ingested 10 min post-exercise, at which point blood [HCO3−] was still considerably below baseline levels. Importantly, both studies (Gough et al. 2017a; 2019a, b) showed ergogenic effects in the subsequent bout of exercise (100% TTE and a boxing simulation, respectively), suggesting that post-exercise SB ingestion might be an effective timing strategy when sufficient recovery is available. In contrast, post-exercise SB ingestion may not be warranted when only a short recovery period separates exercise bouts. Gurton et al. (2021a) found that SB ingested immediately following TTE running at a velocity corresponding to maximal oxygen consumption (100% v-VO2max) did not increase blood pH or [HCO3−] at 35-min recovery. These authors concluded that insufficient time was available between consuming SB and commencing the second bout of exercise for enough HCO3− to be absorbed that would elicit substantial changes in blood acid–base balance, with significant elevations in [HCO3−] only reported after the second TTE bout (~2.5 mmol L−1 higher than placebo). Based on the existing evidence, it seems that when ~75 min recovery is available, post-exercise SB ingestion might be an effective strategy for improving subsequent exercise performance.
Repeated bout exercise performance
This section of the review focuses on studies that have investigated the effect of SB on performance outcomes during two or more bouts of high-intensity exercise performed on the same day interspersed by ≥10 min. Recent meta-analyses and systematic reviews discuss ergogenic benefits of SB during repeated and intermittent sprint exercise comprising shorter work/rest ratios (Lopes-Silva et al. 2019; Grgic 2022). An overview of methodological design (i.e., dosage, ingestion strategy, and timing) and performance outcomes from studies examining the effect of SB on repeated bout exercise is displayed in Table 1.
Table 1.
Effect of SB supplementation on repeated bout exercise performance
| Study | Participants | Experimental design | Supplementation (dosage, timing & ingestion strategy) | Exercise protocol & recovery duration | Performance outcomes |
|---|---|---|---|---|---|
| Pierce et al. (1992) | University male swimmers (n = 7, age = 19.3 ± 0.42 years, BM = 71.6 ± 1.8 kg) | Randomised, double-blind, placebo-controlled, crossover; 3 day washout | SB: 0.2 g kg−1 BM; PLA: 1 g NaCl; CON: no drink; single dose, 60 min pre-exercise, 400 ml water | 2 × 200-yard swim (style of their choice) separated by 20 min |
100 yard time: ↔ 2 × 200 yards: ↔ |
| Pruscino et al. (2008) | Elite male freestyle swimmers (n = 6) | Randomised, double-blind, placebo-controlled, crossover; ≥3 day washout | SB: 0.3 g kg−1 BM; PLA: glucose; split into 7 doses, 120–30 min pre-exercise, given in capsules with 20 ml kg−1 BM water | 2 × 200 m swim freestyle TT separated by 30 min |
TT1: ↔ TT2 vs. TT1: ↑ |
| Zabala et al. (2008) | Elite male BMX athletes (n = 9, age = 19.4 ± 2.3 years, BM = 73.8 ± 9.9 kg) | Randomised, double-blind, placebo-controlled, crossover; 4 day washout | SB: 0.3 g kg−1 BM; PLA: 0.05 g kg−1 NaCl; single dose, 90 min pre-exercise, 1000 ml flavoured water | 3 × 30 s WT separated by 30 min; CMJ after WT |
Jump height: ↔ WT peak power, mean power and fatigue index: ↔ |
| Zabala et al. (2011) | Elite male BMX athletes (n = 10, age = 20.7 ± 1.4 years, BM = 77.9 ± 2.1 kg) | Randomised, double-blind, placebo-controlled, counterbalanced crossover; 4 day washout | SB: 0.3 g kg−1 BM; PLA: matched capsules; single dose, 90 min pre-exercise, given in capsules with water provided ad libitum | 3 × 30 s WT separated by 15 min; CMJ after WT |
Jump height: ↔ WT peak power, mean power and fatigue index: ↔ |
| Mero et al. (2013) | Elite male swimmers (n = 13, age = 20.5 ± 1.4 years, BM = 80.1 ± 8.1 kg) | Randomised, double-blind, placebo-controlled, crossover; 7 day washout | SB: 0.3 g kg−1 BM; PLA: calcium carbonate; single dose, 60 min pre-exercise, given in capsules | 2 × 100 m swim freestyle TT separated by 12 min |
TT1: ↔ TT2 vs. TT1: ↑ |
| Stöggl et al. (2014) | Endurance trained males (n = 12, age = 32.8 ± 3.8 years, BM = 74 ± 6 kg) | Randomised, double-blind, placebo-controlled, counterbalanced crossover; 5–8 day washout | SB: 0.3 g kg−1 BM; PLA: artificial sweetener and NaCl; single dose, 90 min pre-exercise, 6 ml kg−1 BM water | 3 × TTE at 19 km h−1 (5%) separated by 25 min |
TTE1: ↔ TTE2: ↑ Decline from TTE1 to TTE2 and TTE3: ↑ |
| Gough et al. (2017a, b) | Recreationally active males (n = 9, age = 23 ± 2 years, BM = 74 ± 9 kg) | Double-blind, placebo-controlled, crossover; 3–10 day washout | SB: 0.3 g kg−1 BM; PLA: 0.1 g kg−1 NaCl; single dose, 30 min post-exercise, 5 ml kg−1 BM water/squash | 2 × TTE cycling at 100% Wpeak separated by 90 min |
TTE1: ↔ TTE2: ↑ |
| Dalle et al. (2019) | Recreationally active males (n = 12, age = 21 ± 1 years, BM = 74.4 ± 9.9 kg) | Randomised, double-blind, placebo-controlled, crossover; 7 day washout | SB: 31.5 g; PLA: matched NaCl; split over 9 h, every 3 h: 6.3 g + 2 × 2.1 g, given in capsules with 250 ml water | 4 × 2 min “all-out” sprints separated by 180 min |
Sprint 1, 2 & 4: ↔ Sprint 3: ↑ Average mean power output: ↑ |
| Gough et al. (2018) | Trained male cyclists (n = 10, age = 27 ± 8 years, BM = 82 ± 9 kg) | Randomised, double-blind, placebo-controlled, crossover; 7 day washout | SB: 0.2 g kg−1 and 0.3 g kg−1 BM; PLA: 0.07 g kg−1 NaCl; single dose, time-to-peak bicarbonate pre-exercise, dissolved in 450 ml water/squash | 2 × 4 km cycling TT separated by 40 min in hypoxia (FiO2 14.5%) |
TT1 and TT2: ↑ Decline from TT1 to TT2: ↔ |
| Gough et al. (2019a, b) | Elite male boxers (n = 7, age = 27.1 ± 5.1 years, BM = 72.2 ± 10.3 kg) | Randomised, double-blind, placebo-controlled, crossover; 7 day washout | SB: 0.3 g kg−1 BM; PLA: 0.1 g kg−1 NaCl; single dose, 10 min post-exercise, dissolved in 5 ml kg−1 BM water/squash | 4 × 30 s bouts of 90% and 75% v-VO2max running, boxing specific drills, 2 × TTE runs at 90% v-VO2max separated by 75 min |
TTE1: ↔ TTE2: ↑ Change from TTE1 to TTE2: ↑ |
| Peinado et al. (2019) | Elite male BMX athletes (n = 12, age = 19.2 ± 3.4 years, BM = 72.4 ± 8.4 kg) | Randomised, double-blind, placebo-controlled, counterbalanced crossover; 4 day washout | SB: 0.3 g kg−1 BM; PLA: 0.045 g kg−1 NaCl; single dose, 90 min pre-exercise, given in capsules with water provided ad libitum | 3 × BMX races on an outdoor track (400 m length) | Time, peak velocity and time-to-peak velocity: ↔ |
| Gurton et al. (2021a) | Recreational male runners (n = 11, age = 31.0 ± 9.7 years, BM = 74.4 ± 6.5 kg) | Randomised, single-blind, placebo-controlled, crossover; 5–7 day washout | SB: 0.3 g kg−1 BM; PLA: 0.03 g kg−1 NaCl; single dose, 5 min post-exercise, dissolved in 500 ml water/squash | 2 × TTE runs at v-VO2max (1%) separated by 40 min |
TTE1: ↔ TTE2: ↔ 6/11 improved above test re-test: ↑ |
| Thomas et al. (2022) | Elite track cyclists (men, n = 6, age = 19.8 ± 1.5 years, BM = 83 ± 6 kg; women, n = 2 age = 21.5 ± 2.1 years, BM = 60 ± 0 kg) | Randomised, double-blind, placebo-controlled, crossover; 7 day washout | SB: 0.3 g kg−1 BM; PLA: 0.2 g kg−1 BM calcium carbonate; single dose, 90 min pre-exercise, given in capsules with water provided ad libitum |
4 × 1000 m constant power efforts separated by 20 min 3 × 500 m “all-out” sprints separated by 20 min, squat jumps pre- and post-sprints |
4 × 1000 m constant power efforts: ↔ Average velocity during “all-out” sprints: ↔ Pre-exercise jump height: ↔ Post-exercise jump height: ↑ |
| Thomas et al. (2023) | Active men (n = 8, age = 22 ± 4 years, BM = 80.3 ± 13.0 kg) | Randomised, double-blind, placebo-controlled, crossover; 7 day washout | SB: 0.3 g kg−1 BM; placebo: 0.2 g kg−1 BM calcium carbonate; single dose, 90 min pre-exercise, given in capsules with water provided ad libitum | 3 × 30 s WT separated by 20 min | WT peak power, mean power and fatigue index: ↔ |
BM body mass, SB sodium bicarbonate, PLA placebo, NaCl sodium chloride, WU warm up, WT Wingate test, CMJ countermovement jump, TT time-trial, TTE time-to-exhaustion, Wpeak peak aerobic power output, v-VO2max maximal aerobic velocity, VO2max maximal oxygen consumption
↑ Significant improvement (p < 0.05), ↔ no significant change (p > 0.05)
Some studies have examined the effect of SB on repeated swimming TT performance separated by <30-min recovery (Pierce et al. 1992; Pruscino et al. 2008; Mero et al. 2013). Since athletes competing in elite swimming events sometimes have to perform multiple races for different disciplines during a single competition day (Capelli et al. 1998; Mero et al. 2013), it is possible that if SB can accelerate recovery after an initial event and improve repeated bout exercise performance, then it could be the difference between winning and finishing outside of medal positions. Pierce et al. (1992) reported that SB ingestion had no effect on performance during 2 × 200-yard swimming TTs separated by 20 min recovery. Whilst these authors were unable to measure blood acid–base balance, it is reasonable to suggest that their 0.2 g kg−1 BM SB dose administered at a standardised time frame did not adequately increase [HCO3−] to improve repeated swim performance. In a study examining 2 × 200-m freestyle TTs separated by 30 min, Pruscino et al. (2008) demonstrated a beneficial effect of SB on changes in completion times from TT1 to TT2 (0.7% less drop-off compared with placebo, p=0.05). Mero et al. (2013) also reported that SB reduced the decline in performance during 2 × 100-m freestyle swimming TTs separated by 12 min (1.5 s less than placebo, p<0.05). Discrepancies in these results for the efficacy of SB during repeated bout swimming TTs could relate to factors, such as participant training status, exercise duration, and/or SB ingestion timing. In particular, ergogenic effects of SB might be greater during middle-distance compared with short-distance swimming events (Grgic and Mikulic 2021), and therefore, future research should explore the efficacy of individualised SB supplementation for improving performance during repeated 200-m or 400-m swimming TT’s.
Accelerating recovery following an initial exercise bout could also have important applied benefits to competitive track cyclists, where repeated high-intensity efforts are a common requirement of training and/or competition (Monedero and Donne 2000; Al-Nawaiseh et al. 2016). For instance, the first round and final of the men’s team pursuit at the Rio 2016 Olympics were separated by ~60 min, meaning that athletes needed to be able to recover quickly between bouts. Two studies have investigated the effect of SB ingestion on repeated cycling TT performance (Gough et al. 2018; Thomas et al. 2022). Firstly, Gough et al. (2018) demonstrated that individualised SB in doses of both 0.2 g kg−1 and 0.3 kg−1 BM improved 2 × 4-km cycling TT performance in hypoxic conditions compared to a placebo (40-min recovery; TT1/TT2, both ~1.5% faster; p<0.05), with minimal differences between doses. More recently, Thomas et al. (2022) reported that SB did not improve performance during 4 × 1-km constant power TT’s separated by 20 min. They did, however, observe lower RPE after the 4 × 1-km cycling TT’s for SB compared to placebo (Borg-CR10: ~2 au less; p<0.05). A body of literature exists linking attenuated RPE after SB ingestion to centrally mediated mechanisms (Swank and Robertson 1989; Robertson et al. 1992). This has been underpinned by the assumption that excessive accumulation of H+ within intracellular compartments negatively influences force generating capabilities of muscles (Fitts 2016), which is characterised by localized pain sensations (Robertson et al. 1992). SB ingestion leads to peripheral alterations (i.e. fewer intracellular H+) that modulate activation of group III and IV muscle afferents, in turn reducing negative feedback from muscles and sustaining drive to motor neurons that lowers RPE (Amann et al. 2015). Importantly, from a practical sense, reducing RPE throughout near maximal intensity events could have notable real-world implications for athletes competing in multiple competitions on the same day.
A number of studies have examined the effect of SB during repeated “all-out” cycling exercise, most likely as BMX events require athletes to repeatedly perform high-intensity efforts during qualification phases compromising at least three rounds (~30 min recovery between each) (Peinado et al. 2019). In a series of investigations from Zabala et al. (2008, 2011) and Thomas et al. (2023), it was found that SB did not improve performance during 3 × 30-s Wingate cycling tests interspersed by 15–30-min recovery periods. Peinado et al. (2019) observed no ergogenic benefits of SB during three competitive BMX races (~30 s duration) each separated by 15 min. Thomas et al. (2022) also reported no improvements in average velocity for SB during 3 × 500 m “all-out” cycling sprints. These type of exercise tests comprising “all-out” efforts might not be sensitive enough to observe performance benefits after SB (Grgic 2022). Improvements in performance are most likely to be shown when the rate of decrease in intramuscular pH is relatively slow, typically during exercise lasting 4–8 min (Matson and Tran 1993). Rapid rate of decline in pH during “all-out” cycling tests results in the monocarboxylate transporter 1/4 becoming oversaturated with H+, as accumulation in intracellular compartments overwhelms clearance rates (Messonnier et al. 2007). This means that the beneficial effects of enhanced circulating HCO3− on restoring intracellular acid–base balance are diminished, subsequently limiting the ergogenic capacity of SB (Higgins et al. 2013).
Dalle et al. (2019) showed SB to increase mean power during 4 × 2 min “all-out” cycling tests separated by 180 min throughout an endurance cycling protocol (1.4% higher than a placebo; p=0.035). As alluded to, their longer exercise duration is one explanation for why performance benefits were observed for SB. Interestingly, Thomas et al. (2022) reported greater squat jump height for SB after their “all-out” cycling sprints (~2.5% compared with placebo; p<0.05). Inconsistencies between the effect of SB ingestion on vertical jump height and “all-out” cycling could be attributed to their differing energy system requirements (Thomas et al. 2022). Short-duration plyometric exercises are predominately influenced by changes in phosphocreatine (PCr) hydrolysis opposed to glycolytic flux (Hultman et al. 1967), which suggests that they might be more sensitive to post-exercise kinetics of PCr recovery. Further mechanistical work is required, but since PCr recovery following high-intensity exercise is somewhat associated with muscle acidity (Lodi et al. 1997), it is possible that elevated acid–base balance after SB ingestion contributes to improvements in post-exercise explosive plyometric movement tasks such as vertical jumps that are reliant on the ATP-PCr system.
Finally, there is a growing body of scientific literature investigating the ergogenic capacity of SB ingestion during repeated TTE bouts (Stöggl et al. 2014; Gough et al. 2017a, 2019b; Gurton et al. 2021a), with equivocal findings to date. Indeed, Stöggl et al. (2014) reported benefits during 3 × 19 km h−1 bouts (25 min between each, TTE2: +15% higher for SB than control, p=0.048), but Gurton et al. (2021a) observed no differences between two bouts of exhaustive running (100% v-VO2max) separated by 40 min. The inconsistency in findings might be due to the timing of SB ingestion, as Gurton et al. (2021a) adopted a post-exercise SB ingestion strategy that may have allowed sufficient time to observe substantial changes in [HCO3−] that would lead to ergogenic benefits during subsequent exercise. In support of this idea, Gough et al. (2017a) reported that administering SB 30 min after an initial TTE cycling bout improved performance during a second TTE cycling test after a longer, 90-min recovery (+16.6% greater than placebo; p=0.007). However, as blood pH and [HCO3−] prior to the second TTE task were reflective of studies opting for pre-exercise SB ingestion, it should be noted that their results do not directly suggest enhanced recovery per se, but instead perhaps show the known ergogenic effects of pre-exercise SB. In their follow-up study, Gough et al. (2019b) demonstrated that change in performance from TTE1 to TTE2 (separated by 75 min) was greater when SB was administered 10 min after the initial exercise (91 s longer TTE than placebo; p=0.02). Based on this evidence, the effects of SB ingestion on repeated TTE performance are promising, although as athletes would rarely complete two exhaustive tasks to exhaustion, the practical application of this is unclear.
Practical implications and the sodium bicarbonate ingestion recovery framework
Based on the findings of this narrative review Table 2 depicts an SB ingestion recovery framework. It is envisaged that this SB ingestion recovery framework will act as a tool for practitioners and athletes wishing to easily identify where ingestion may aid exercise performance across repeated bouts of exercise. In short, it is proposed that scenarios where only a recovery duration of up to 30 min exists between bouts of exercise, SB ingestion is unlikely to provide ergogenic benefits to performance. On the other hand, between 30 and 90 min of recovery seems to be the optimal for eliciting ergogenic benefits for SB, as there is sufficient acid base balance recovery between each bout, which then can contribute to improvements during the subsequent bout of exercise. To date, no research has explored the effect of a single SB dose on performance in two or more exercise bouts separated by more than 90 min, and thus, further studies examining this time frame are required to refine practical recommendations. Finally, the concept of ‘top-up’ SB doses is a novel area that has shown promise and could be particularly beneficial for athletes competing in endurance exercise.
Table 2.
Practical framework for the ingestion of sodium bicarbonate during repeated bout effort sporting events
| Recovery time frame between exercise bouts | Recommendation | Recommended scenarios for use | Sports that could benefit | References | Future research directions |
|---|---|---|---|---|---|
| 0–30 min |
SB unlikely to be effective as acid base balance cannot recover in time, although some benefits have been observed with three bouts of exercise (*) |
During half-time to improve second half performance Qualification rounds separated by a short rest period |
Team sports (football/soccer, rugby) BMX cycling |
Pruscino et al. (2008), Mero et al. (2013), Pierce et al. (1992), Thomas et al. (2022), Zabala et al. (2008), Peinado et al. (2019), Thomas et al. (2023) | Individualised time-to-peak alkalosis ingestion strategy may provide more consistent benefits and a stronger recovery profile of acid–base balance |
| 30–90 min |
0.2–0.3 g kg−1 BM SB either prior to or post the initial bout of exercise standardised or individual time-to-peak ingestion timing (***) |
Heat and final events whereby only a short recovery is available Training whereby repeated bouts are completed within a short time frame |
Cycling (primarily track), swimming, sprint skiing | Gough et al. (2018), Gough et al. (2017a, b), Gough et al. (2019a), Gough et al. (2019b), Gurton et al. (2021a, b) |
Pre- versus post-exercise ingestion of SB Investigations into ecologically valid sporting scenarios |
| 90+ min |
Promise for SB to work, but research lacking (*) |
Heat and final events whereby a longer recovery is available Training whereby repeated bouts are completed on the same day |
Some swimming events, rugby 7’s | N/A | Studies examining longer time frames of recovery are required as none to date have gone past 90 min recovery for a single SB dose |
| During exercise/a competition event |
Initial and “top-up” doses may be effective for endurance-based exercise, with SB ingestion also prior to exercise (**) |
Grand Tour stage races (3–4 h). Use a pre-exercise dose and then top-up halfway through | Endurance cycling or running events | Dalle et al. (2019), Dalle et al. (2021) | Due to minimal scientific evidence, further studies investigating a range of sports are required |
The table has been constructed based on the evidence in the area and the expertise within the authorship group in both research and applied experiences with an end goal to be used a practical point of reference for practitioners and athletes within their given sports
BM body mass; SB sodium bicarbonate
Symbols denote level of empirical evidence for the efficacy of SB: * little-to-none despite extensive research, or more research required before conclusions can be made, ** might be beneficial in specific circumstances, *** high likelihood of performance benefits
Future research directions
Since existing literature has mainly focused on the pre-exercise effects of SB ingestion, it is recommended that greater attention is given to the application of SB as a recovery aid in the future (using either pre- or post-exercise ingestion). First, from a mechanistical viewpoint, further research is needed examining the influence of SB ingestion on recovery of the three pertinent physiological systems/pathways discussed in this review: (1) acid–base balance, (2) SID, and (3) cellular stress responses and oxidative stress. Particular focus should be given to whether SB mediated changes in cellular stress responses (e.g., HSP72, oxidative stress) lead to noticeable improvements in recovery from exercise, repeated bout performance, and training adaptations. Future mechanistical work could also examine the role of SB on inorganic phosphate accumulation and calcium signalling during repeated bout exercise, as these contribute to skeletal muscle contractile function (Allen and Trajanovska 2012). Second, it would be worthwhile to examine the importance of SB timing by comparing the effect of pre- and post-exercise SB ingestion on blood pH, [HCO3−] and strong ions across different recovery durations. In a similar fashion, exploration into the concept of “top up” SB doses would be beneficial based on promising findings of Dalle et al. (2019) during endurance exercise. The use of SB is not typically associated with endurance exercise, yet these “top up” doses could be useful for athletes competing in longer duration sports (e.g., Grand Tour cyclists). Additionally, research is warranted comparing factors such as sex-specific or training status differences that may influence the efficacy of SB as a recovery aid, as this remains an underexplored area that could have important real-world implications for practitioners and athletes.
Finally, since studies discussed in this review have primarily concentrated on cycling and swimming, it is important for future research to investigate other exercise modalities and events that could potentially benefit from accelerated indices of recovery between repeated bouts of exercise. One example could be during 15–20 min half-time periods in team sports (e.g., rugby, soccer, and basketball), as existing evidence suggests that SB ingestion increases blood pH, [HCO3−] and SID during similar recovery durations (Verbitsky et al. 1997; Siegler et al. 2008). Other sporting disciplines that could be of interest to the application of SB as a recovery aid are rugby sevens and cross-country skiing, as they feature excessive H+ accumulation and short recovery period between exercise bouts (Ross et al. 2014; Losnegard 2019).
Conclusion
Based on the literature, it is reasonable to suggest that SB ingestion could play a key role in improving recovery of acid–base balance and thus performance, given the correct ingestion strategy and recovery period between two bouts of exercise. In light of this, the ingestion framework presented within this review may serve as a tool for practitioners to use with athletes. Further research is needed to determine the optimal SB ingestion approach for athletes in multiple sports to further explore the role of this supplement on sports performance.
Abbreviations
- ATP
Adenosine triphosphate
- au
Arbitrary units
- BM
Body mass
- Cl−
Chloride ion
- GI
Gastrointestinal
- GSSG
Oxidised glutathione
- H+
Hydrogen ion
- HCO3−
Blood bicarbonate
- HSP72
Heat shock protein 72
- K+
Potassium ion
- Na+
Sodium ion
- PCr
Phosphocreatine
- ROS
Reactive oxygen species
- SB
Sodium bicarbonate
- SID
Strong ion difference
- TBARS
Thiobarbituric acid reactive substances
- TGSH
Total glutathione
- TT
Time-trial
- TTE
Time-to-exhaustion
- v-VO2max
Running velocity at maximal oxygen consumption
Author contributions
WHG and LAG conceptualised and designed the research. WG and LAG performed the literature search. WG and LAG wrote the manuscript with contributions from JCS. All other authors offered critical comments and approved the manuscript.
Data availability
Data collected from literature search is available upon reasonable request.
Declarations
Conflict of interest
The authors declare no conflicts of interest relating to the findings of this manuscript. No funds, grants, or other support was received.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Allen DG, Trajanovska S (2012) The multiple roles of phosphate in muscle fatigue. Front Physiol 3:36880. 10.3389/fphys.2012.00463 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen DG, Lamb GD, Westerblad H (2008) Skeletal muscle fatigue: cellular mechanisms. Physiol Rev 88(1):287–332 [DOI] [PubMed] [Google Scholar]
- Al-Nawaiseh AM, Pritchett RC, Bishop PA (2016) Enhancing short-term recovery after high-intensity anaerobic exercise. J Strength Cond Res 30:320. 10.1519/JSC.0000000000001060 [DOI] [PubMed] [Google Scholar]
- Amann M, Sidhu SK, Weavil JC, Mangum TS, Venturelli M (2015) Autonomic responses to exercise: group III/IV muscle afferents and fatigue. Auton Neurosci 188:19–23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bibbins-Domingo K (2014) The institute of medicine report sodium intake in populations: assessment of evidence: summary of primary findings and implications for clinicians. JAMA Intern Med 174:136–137. 10.1001/jamainternmed.2013.11818 [DOI] [PubMed] [Google Scholar]
- Bishop D, Edge J, Davis C, Goodman C (2004) Induced metabolic alkalosis affects muscle metabolism and repeated-sprint ability. Med Sci Sports Exerc 36:807–813. 10.1249/01.MSS.0000126392.20025.17 [DOI] [PubMed] [Google Scholar]
- Boegman S, Stellingwerff T, Shaw G et al (2020) The impact of individualizing sodium bicarbonate supplementation strategies on world-class rowing performance. Front Nutr 7:138. 10.3389/fnut.2020.00138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cairns SP, Lindinger MI (2008) Do multiple ionic interactions contribute to skeletal muscle fatigue? J Physiol 586:4039–4054. 10.1113/jphysiol.2008.155424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cairns SP, Flatman JA, Clausen T (1995) Relation between extracellular [K+], membrane potential and contraction in rat soleus muscle: modulation by the Na+–K+ pump. Pflugers Arch 430:909–915. 10.1007/BF01837404 [DOI] [PubMed] [Google Scholar]
- Capelli C, Pendergast DR, Termin B (1998) Energetics of swimming at maximal speeds in humans. Eur J Appl Physiol 78:385–393. 10.1007/s004210050435 [DOI] [PubMed] [Google Scholar]
- Carr AJ, Hopkins WG, Gore CJ (2011a) Effects of acute alkalosis and acidosis on performance. Sports Med 41:801–814. 10.2165/11591440-000000000-00000 [DOI] [PubMed] [Google Scholar]
- Carr AJ, Slater GJ, Gore CJ, Dawson B, Burke LM (2011b) Effect of sodium bicarbonate on [HCO3−], pH, and gastrointestinal symptoms. Int J Sport Nutr Exerc Metab 21:189–194. 10.1123/ijsnem.21.3.189 [DOI] [PubMed] [Google Scholar]
- Clausen T, Everts ME (1991) K(+)-induced inhibition of contractile force in rat skeletal muscle: role of active Na(+)–K+ transport. Am J Physiol 261:C799–C807. 10.1152/ajpcell.1991.261.5.C799 [DOI] [PubMed] [Google Scholar]
- Clausen T, Andersen SL, Flatman JA (1993) Na(+)–K+ pump stimulation elicits recovery of contractility in K(+)-paralysed rat muscle. J Physiol 472:521–536. 10.1113/jphysiol.1993.sp019960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costill DL, Verstappen F, Kuipers H et al (1984) Acid-base balance during repeated bouts of exercise: influence of HCO3. Int J Sports Med 05:228–231. 10.1055/s-2008-1025910 [DOI] [PubMed] [Google Scholar]
- Dalle S, Smet SD, Geuns W et al (2019) Effect of stacked sodium bicarbonate loading on repeated all-out exercise. Int J Sports Med 40:711–716. 10.1055/a-0978-5139 [DOI] [PubMed] [Google Scholar]
- Dalle S, Koppo K, Hespel P (2021) Sodium bicarbonate improves sprint performance in endurance cycling. J Sci Med Sport 24:301–306. 10.1016/j.jsams.2020.09.011 [DOI] [PubMed] [Google Scholar]
- de Oliveira L, Saunders B, Yamaguchi G et al (2020) Is individualization of sodium bicarbonate ingestion based on time to peak necessary? Med Sci Sports Exerc 52:1801–1808. 10.1249/MSS.0000000000002313 [DOI] [PubMed] [Google Scholar]
- de Oliveira LF, Dolan E, Swinton PA et al (2022) Extracellular buffering supplements to improve exercise capacity and performance: a comprehensive systematic review and meta-analysis. Sport Med 1:1–22. 10.1007/s40279-021-01575-x [DOI] [PubMed] [Google Scholar]
- Deb SK, Gough LA, Sparks SA, McNaughton LR (2018) Sodium bicarbonate supplementation improves severe-intensity intermittent exercise under moderate acute hypoxic conditions. Eur J Appl Physiol 118:607–615. 10.1007/s00421-018-3801-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debold EP, Beck SE, Warshaw DM (2008) Effect of low pH on single skeletal muscle myosin mechanics and kinetics. Am J Physiol 295:C173–C179. 10.1152/ajpcell.00172.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dennig H, Talbott JH, Edwards HT, Dill DB (1931) Effect of acidosis and alkalosis upon capacity for work. J Clin Med 9:601–613. 10.1172/JCI100324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fitts R (2016) The role of acidosis in fatigue: pro perspective. Med Sci Sports Exerc 48:2335–2338. 10.1249/MSS.0000000000001043 [DOI] [PubMed] [Google Scholar]
- Fraser SF, Li JL, Carey MF et al (2002) Fatigue depresses maximal in vitro skeletal muscle Na+–K+-ATPase activity in untrained and trained individuals. J Appl Physiol 93:1650–1659. 10.1152/japplphysiol.01247.2001 [DOI] [PubMed] [Google Scholar]
- Gapen CJ, Moseley PL (1995) Acidosis alters the hyperthermic cytotoxicity and the cellular stress response. J Therm Biol 20:321–325. 10.1016/0306-4565(94)00063-O [Google Scholar]
- Gibson BM, Needham KW, Kaiser BW et al (2023) Transcutaneous delivery of sodium bicarbonate increases intramuscular pH. Front Physiol 14:1142567. 10.3389/fphys.2023.1142567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gough LA, Sparks SA (2024) The effects of a carbohydrate hydrogel system for the delivery of bicarbonate mini-tablets on acid–base buffering and gastrointestinal symptoms in resting well-trained male cyclists. Sports Med Open 10:1–9. 10.1186/s40798-024-00684-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gough LA, Deb SK, Sparks AS, McNaughton LR (2017a) The reproducibility of blood acid base responses in male collegiate athletes following individualised doses of sodium bicarbonate: a randomised controlled crossover study. Sports Med 47:2117–2127. 10.1007/s40279-017-0699-x [DOI] [PubMed] [Google Scholar]
- Gough L, Rimmer S, Osler C, Higgins M (2017b) Ingestion of sodium bicarbonate (NaHCO3) following a fatiguing bout of exercise accelerates post-exercise acid–base balance recovery and improves subsequent high-intensity cycling time to exhaustion. Int J Sport Nutr Exerc Metab 27:1–25. 10.1123/ijsnem.2017-0065 [DOI] [PubMed] [Google Scholar]
- Gough LA, Brown D, Deb SK et al (2018) The influence of alkalosis on repeated high-intensity exercise performance and acid–base balance recovery in acute moderate hypoxic conditions. Eur J Appl Physiol 118:2489–2498. 10.1007/s00421-018-3975-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gough LA, Deb SK, Brown D et al (2019a) The effects of sodium bicarbonate ingestion on cycling performance and acid base balance recovery in acute normobaric hypoxia. J Sport Sci 37:1464–1471. 10.1080/02640414.2019.1568173 [DOI] [PubMed] [Google Scholar]
- Gough LA, Rimmer S, Sparks SA et al (2019b) Post-exercise supplementation of sodium bicarbonate improves acid base balance recovery and subsequent high-intensity boxing specific performance. Front Nutr 6:155. 10.3389/fnut.2019.00155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gough LA, Williams JJ, Newbury JW, Gurton WH (2021) The effects of sodium bicarbonate supplementation at individual time-to-peak blood bicarbonate on 4-km cycling time trial performance in the heat. Eur J Sport Sci 22:1856–1864. 10.1080/17461391.2021.1998644 [DOI] [PubMed] [Google Scholar]
- Grgic J (2022) Effects of sodium bicarbonate ingestion on measures of Wingate test performance: a meta-analysis. J Am Nutr Assoc 41:1–10. 10.1080/07315724.2020.1850370 [DOI] [PubMed] [Google Scholar]
- Grgic J, Pedisic Z, Saunders B et al (2021) International society of sports nutrition position stand: sodium bicarbonate and exercise performance. J Int Soc Sports Nutr 18:61. 10.1186/s12970-021-00458-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurton WH, Gough LA, Sparks SA et al (2020) Sodium bicarbonate ingestion improves time-to-exhaustion cycling performance and alters estimated energy system contribution: a dose-response investigation. Front Nutr 7:154. 10.3389/fnut.2020.00154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurton WH, Faulkner SH, James RM (2021a) Effect of warm-up and sodium bicarbonate ingestion on 4-km cycling time-trial performance. Int J Sports Physiol Perform 16:1573–1579. 10.1123/ijspp.2020-0743 [DOI] [PubMed] [Google Scholar]
- Gurton W, Macrae H, Gough L, King DG (2021b) Effects of post-exercise sodium bicarbonate ingestion on acid–base balance recovery and time-to-exhaustion running performance: a randomised crossover trial in recreational athletes. Appl Physiol Nutr Metab 46:1111–1118. 10.1139/apnm-2020-1120 [DOI] [PubMed] [Google Scholar]
- Gurton WH, Greally J, Chudzikiewicz K et al (2023a) Beneficial effects of oral and topical sodium bicarbonate during a battery of team sport-specific exercise tests in recreationally trained male athletes. J Int Soc Sports Nutr 20:2216678. 10.1080/15502783.2023.2216678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurton WH, Matta GG, Gough LA et al (2023b) Sodium bicarbonate and time-to-exhaustion cycling performance: a retrospective analysis exploring the mediating role of expectation. Sports Med Open 9:65. 10.1186/s40798-023-00612-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadzic M, Eckstein ML, Schugardt M (2019) The impact of sodium bicarbonate on performance in response to exercise duration in athletes: a systematic review. J Sports Sci Med 18:271–281 [PMC free article] [PubMed] [Google Scholar]
- Higgins MF, James RS, Price MJ (2013) The effects of sodium bicarbonate (NaHCO3) ingestion on high intensity cycling capacity. J Sports Sci 31:972–981. 10.1080/02640414.2012.758868 [DOI] [PubMed] [Google Scholar]
- Hollidge-Horvat MG, Parolin ML, Wong D et al (2000) Effect of induced metabolic alkalosis on human skeletal muscle metabolism during exercise. Am J Physiol Endocrinol Metab 278:E316–E329. 10.1152/ajpendo.2000.278.2.E316 [DOI] [PubMed] [Google Scholar]
- Hultman E, Bergström J, Anderson NM (1967) Breakdown and resynthesis of phosphorylcreatine and adenosine triphosphate in connection with muscular work in man. Scand J Clin Lab Invest 19:56–66. 10.3109/00365516709093481 [DOI] [PubMed] [Google Scholar]
- Jones RL, Stellingwerff T, Artioli GG et al (2016) Dose-response of sodium bicarbonate ingestion highlights individuality in time course of blood analyte responses. Int J Sport Nutr Exerc Metab 26:445–453. 10.1123/ijsnem.2015-0286 [DOI] [PubMed] [Google Scholar]
- Katz A, Costill DL, King DS et al (1984) Maximal exercise tolerance after induced alkalosis. Int J Sports Med 5:107–110. 10.1055/s-2008-1025890 [DOI] [PubMed] [Google Scholar]
- Kent-Braun JA, Fitts RH, Christie A (2012) Skeletal muscle fatigue. Compr Physiol 2:997–1044. 10.1002/cphy.c110029 [DOI] [PubMed] [Google Scholar]
- Koster JF, Slee RG, Essed CE, Stam H (1985) Studies on cumene hydroperoxide-induced lipid peroxidation in the isolated perfused rat heart. J Mol Cell Cardiol 17:701–708. 10.1016/S0022-2828(85)80069-9 [DOI] [PubMed] [Google Scholar]
- Lodi R, Muntoni F, Taylor J et al (1997) Correlative MR imaging and 31P-MR spectroscopy study in sarcoglycan deficient limb girdle muscular dystrophy. Neuromuscul Disord 7:505–511. 10.1016/s0960-8966(97)00108-9 [DOI] [PubMed] [Google Scholar]
- Lopes-Silva JP, Reale R, Franchini E (2019) Acute and chronic effect of sodium bicarbonate ingestion on Wingate test performance: a systematic review and meta-analysis. J Sports Sci 37:762–771. 10.1080/02640414.2018.1524739 [DOI] [PubMed] [Google Scholar]
- Losnegard T (2019) Energy system contribution during competitive cross-country skiing. Eur J Appl Physiol 119:1675–1690. 10.1007/s00421-019-04158-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luttrell M, Halliwill J (2015) Recovery from exercise: vulnerable state, window of opportunity, or crystal ball? Front Physiol 6:204. 10.3389/fphys.2015.00204 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matson LG, Tran ZV (1993) Effects of sodium bicarbonate ingestion on anaerobic performance: a meta-analytic review. Int J Sport Nutr 3:2–28. 10.1123/ijsn.3.1.2 [DOI] [PubMed] [Google Scholar]
- Maughan RJ, Burke LM, Dvorak J, Larson-Meyer DE, Peeling P, Phillips SM, Engebretsen L (2018) IOC consensus statement: dietary supplements and the high-performance athlete. Int J Sport Nutr Exerc Metab 28(2):104–125 [DOI] [PubMed] [Google Scholar]
- McKay AK, Peeling P, Binnie MJ et al (2020) Topical sodium bicarbonate: no improvement in blood buffering capacity or exercise performance. Int J Sports Physiol Perform 15:1005–1011. 10.1123/ijspp.2019-0345 [DOI] [PubMed] [Google Scholar]
- McNaughton LR (1992) Bicarbonate ingestion: effects of dosage on 60 s cycle ergometry. J Sports Sci 10:415–423. 10.1080/02640419208729940 [DOI] [PubMed] [Google Scholar]
- Mero AA, Hirvonen P, Saarela J et al (2013) Effect of sodium bicarbonate and beta-alanine supplementation on maximal sprint swimming. J Int Soc Sports Nutr 10:52. 10.1186/1550-2783-10-52 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messonnier L, Kristensen M, Juel C, Denis C (2007) Importance of pH regulation and lactate/H+ transport capacity for work production during supramaximal exercise in humans. J Appl Physiol 102:1936–1944. 10.1152/japplphysiol.00691.2006 [DOI] [PubMed] [Google Scholar]
- Monedero J, Donne B (2000) Effect of recovery interventions on lactate removal and subsequent performance. Int J Sports Med 21:593–597. 10.1055/s-2000-8488 [DOI] [PubMed] [Google Scholar]
- Mosser DD, Caron AW, Bourget L et al (1997) Role of the human heat shock protein hsp70 in protection against stress-induced apoptosis. Mol Cell Biol 17:5317–5327. 10.1128/MCB.17.9.5317 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen OB, Hilsted L, Clausen T (1998) Excitation-induced force recovery in potassium-inhibited rat soleus muscle. J Physiol 512(Pt 3):819–829. 10.1111/j.1469-7793.1998.819bd.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peart DJ, McNaughton LR, Midgley AW et al (2011) Pre-exercise alkalosis attenuates the heat shock protein 72 response to a single-bout of anaerobic exercise. J Sci Med Sport 14:435–440. 10.1016/j.jsams.2011.03.006 [DOI] [PubMed] [Google Scholar]
- Peart DJ, Siegler JC, Vince RV (2012) Practical recommendations for coaches and athletes: a meta-analysis of sodium bicarbonate use for athletic performance. J Strength Conditioning Res 26(7): 1975–1983 [DOI] [PubMed] [Google Scholar]
- Peart DJ, Kirk RJ, Hillman AR et al (2013a) The physiological stress response to high-intensity sprint exercise following the ingestion of sodium bicarbonate. Eur J Appl Physiol 113:127–134. 10.1007/s00421-012-2419-4 [DOI] [PubMed] [Google Scholar]
- Peart DJ, Kirk RJ, Madden LA et al (2013b) The influence of exogenous carbohydrate provision and pre-exercise alkalosis on the heat shock protein response to prolonged interval cycling. Amino Acids 44:903–910. 10.1007/s00726-012-1419-3 [DOI] [PubMed] [Google Scholar]
- Peart DJ, Kirk RJ, Madden LA, Vince RV (2016) Implications of a pre-exercise alkalosis-mediated attenuation of HSP72 on its response to a subsequent bout of exercise. Amino Acids 48:499–504. 10.1007/s00726-015-2103-1 [DOI] [PubMed] [Google Scholar]
- Peinado AB, Holgado D, Luque-Casado A et al (2019) Effect of induced alkalosis on performance during a field-simulated BMX cycling competition. J Sci Med Sport 22:335–341. 10.1016/j.jsams.2018.08.010 [DOI] [PubMed] [Google Scholar]
- Pierce EF, Eastman NW, Hammer WH, Lynn TD (1992) Effect of induced alkalosis on swimming time trials. J Sports Sci 10:255–259. 10.1080/02640419208729924 [DOI] [PubMed] [Google Scholar]
- Powers SK, Jackson MJ (2008) Exercise-induced oxidative stress: cellular mechanisms and impact on muscle force production. Physiol Rev 88:1243–1276. 10.1152/physrev.00031.2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powers S, Smuder A, Kavazis A, Hudson M (2010) Experimental guidelines for studies designed to investigate the impact of antioxidant supplementation on exercise performance. Int J Sport Nutr Exerc Metab 20:2–14. 10.1123/ijsnem.20.1.2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pruscino CL, Ross MLR, Gregory JR et al (2008) Effects of sodium bicarbonate, caffeine, and their combination on repeated 200-m freestyle performance. Int J Sport Nutr Exerc Metab 18:116–130. 10.1123/ijsnem.18.2.116 [DOI] [PubMed] [Google Scholar]
- Robergs R, Hutchinson K, Hendee S et al (2005) Influence of pre-exercise acidosis and alkalosis on the kinetics of acid–base recovery following intense exercise. Int J Sport Nutr Exerc Metab 15:59–74. 10.1123/ijsnem.15.1.59 [DOI] [PubMed] [Google Scholar]
- Robertson RJ, Nixon PA, Caspersen CJ, Metz KF, Abbott RA, Goss FL (1992) Abatement of exertional perceptions following dynamic exercise: physiological mediators. Med Sci Sports Exerc 24(3):346–353 [PubMed] [Google Scholar]
- Ross A, Gill N, Cronin J (2014) Match analysis and player characteristics in rugby sevens. Sports Med 44:357–367. 10.1007/s40279-013-0123-0 [DOI] [PubMed] [Google Scholar]
- Saunders B, Sale C, Harris RC, Sunderland C (2014) Sodium bicarbonate and high-intensity-cycling capacity: variability in responses. Int J Sports Physiol Perform 9:627–632. 10.1123/ijspp.2013-0295 [DOI] [PubMed] [Google Scholar]
- Siegler JC, Gleadall-Siddall DO (2010) Sodium bicarbonate ingestion and repeated swim sprint performance. J Strength Cond Res 24:3105–3111. 10.1519/JSC.0b013e3181f55eb1 [DOI] [PubMed] [Google Scholar]
- Siegler J, Hirscher K (2009) Sodium bicarbonate ingestion and boxing performance. J Strength Cond Res 24:103–108. 10.1519/JSC.0b013e3181a392b2 [DOI] [PubMed] [Google Scholar]
- Siegler JC, Keatley S, Midgiey AW et al (2008) Pre-exercise alkalosis and acid–base recovery. Int J Sports Med 29:545–551. 10.1055/s-2007-989261 [DOI] [PubMed] [Google Scholar]
- Siegler JC, Marshall PW, Bishop D, Shaw G, Green S (2016) Mechanistic insights into the efficacy of sodium bicarbonate supplementation to improve athletic performance. Sports Med Open 2:1–13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sostaric SM, Skinner SL, Brown MJ et al (2006) Alkalosis increases muscle K+ release, but lowers plasma [K+] and delays fatigue during dynamic forearm exercise. J Physiol 570:185–205. 10.1113/jphysiol.2005.094615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spriet LL, Matsos CG, Peters SJ, Heigenhauser GJ, Jones NL (1985) Effects of acidosis on rat muscle metabolism and performance during heavy exercise. Am J Physiol Cell Physiol 248:C337–C347. 10.1152/ajpcell.1985.248.3.C337 [DOI] [PubMed] [Google Scholar]
- Spriet LL, Lindinger MI, Heigenhauser GJ, Jones NL (1986) Effects of alkalosis on skeletal muscle metabolism and performance during exercise. Am J Physiol Regul Integr Comp Physiol 251:R833–R839. 10.1152/ajpregu.1986.251.5.R833 [DOI] [PubMed] [Google Scholar]
- Spriet LL, Lindinger MI, McKelvie RS et al (1989) Muscle glycogenolysis and H+ concentration during maximal intermittent cycling. J Appl Physiol 66:8–13. 10.1152/jappl.1989.66.1.8 [DOI] [PubMed] [Google Scholar]
- Stewart PA (1983) Modern quantitative acid–base chemistry. Can J Physiol Pharmacol 61:1444–1461. 10.1139/y83-207 [DOI] [PubMed] [Google Scholar]
- Stöggl T, Torres-Peralta R, Cetin E, Nagasaki M (2014) Repeated high intensity bouts with long recovery: are bicarbonate or carbohydrate supplements an option? Sci World J 2014:145747. 10.1155/2014/145747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strazzullo P, D’Elia L, Kandala N-B, Cappuccio FP (2009) Salt intake, stroke, and cardiovascular disease: meta-analysis of prospective studies. BMJ 339:b4567–b4567. 10.1136/bmj.b4567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swank A, Robertson RJ (1989) Effect of induced alkalosis on perception of exertion during intermittent exercise. J Appl Physiol 67:1862–1867. 10.1152/jappl.1989.67.5.1862 [DOI] [PubMed] [Google Scholar]
- Taylor L, Midgley AW, Chrismas B et al (2011) Daily hypoxia increases basal monocyte HSP72 expression in healthy human subjects. Amino Acids 40:393–401. 10.1007/s00726-010-0644-x [DOI] [PubMed] [Google Scholar]
- Thomas C, Delfour-Peyrethon R, Dorel S, Hanon C (2022) Positive effects of pre-exercise metabolic alkalosis on perceived exertion and post-exercise squat jump performance in world-class cyclists. J Strength Cond Res 36:2602–2609. 10.1519/jsc.0000000000003855 [DOI] [PubMed] [Google Scholar]
- Thomas C, Delfour-Peyrethon R, Lambert K et al (2023) The effect of pre-exercise alkalosis on lactate/pH regulation and mitochondrial respiration following sprint-interval exercise in humans. Front Physiol 14:1073407. 10.3389/fphys.2023.1073407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verbitsky O, Mizrahi J, Levin M, Isakov E (1997) Effect of ingested sodium bicarbonate on muscle force, fatigue, and recovery. J Appl Physiol 83:333–337. 10.1152/jappl.1997.83.2.333 [DOI] [PubMed] [Google Scholar]
- Westerblad H (2016) Acidosis is not a significant cause of skeletal muscle fatigue. Med Sci Sports Exerc 48(11):2339–2342 [DOI] [PubMed] [Google Scholar]
- Zabala M, Requena B, Sánchez-Muñoz C et al (2008) Effects of sodium bicarbonate ingestion on performance and perceptual responses in a laboratory-simulated BMX cycling qualification series. J Strength Cond Res 22:1645–1653. 10.1519/JSC.0b013e318181febe [DOI] [PubMed] [Google Scholar]
- Zabala M, Peinado AB, Calderón FJ et al (2011) Bicarbonate ingestion has no ergogenic effect on consecutive all out sprint tests in BMX elite cyclists. Eur J Appl Physiol 111:3127–3134. 10.1007/s00421-011-1938-8 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data collected from literature search is available upon reasonable request.