Abstract
Staphylococcus aureus isolates (n = 70) from 65 patients (36 canine, 18 equine, 7 bovine, 2 avian, and 2 feline) at seven veterinary teaching hospitals in the United States were studied. The majority of patients (83%) with an S. aureus infection were canine and equine, but this may have reflected a sample bias based on clinic case loads and diagnostic lab submissions at the participating institutions. Fourteen percent of patients with an S. aureus infection were infected with a methicillin-resistant S. aureus (MRSA) isolate. Six of seven institutions had at least one MRSA infection during the study. Pulsed-field gel electrophoresis on 63 of the 70 isolates yielded 58 unique strains of S. aureus. None of the strain types of the MRSA isolates matched each other or the type of any other S. aureus isolate. The proportions of patients infected with an MRSA isolate were not significantly different between institutions or animal species (P ≥ 0.222). Methicillin-resistant S. aureus isolates in this study seemed to be community acquired rather than hospital acquired.
Antimicrobial resistance is a major concern to physicians and veterinarians worldwide. Methicillin-resistant Staphylococcus aureus (MRSA) nosocomial infections are well documented in human hospitals (5). Methicillin-resistant S. aureus is of particular concern because very few antimicrobials are effective in treating infections caused by this pathogen. Data regarding the prevalence of MRSA in the veterinary field are limited, but a report from Michigan State University illustrates the emergence and increasing significance of MRSA in veterinary medicine (11). The isolates found in the Michigan outbreak came from equine postoperative wound infections, were genotypically indistinguishable, and were suspected to be of human origin. Equine postoperative wound infection with MRSA has been reported at other veterinary teaching hospitals (6). A Japanese study demonstrated that 14 of 15 MRSA isolates from mares with metritis and a stallion with dermatitis had identical pulsed-field gel electrophoretic patterns, suggesting a common origin of infection (12). However, unlike in other reports, the equine MRSA isolates characterized in Japan did not appear to be of human origin. Methicillin-resistant S. aureus has also been reported in the bovine mastitis literature (2, 3) and in small animal patients (9, 13).
Two reports suggest that animals may serve as reservoirs for infection of humans (1, 10). In the first report, a dog was implicated as a reservoir for the reinfection of two nurses who carried MRSA and infected their human patients. In the second, a cat was implicated as the source of MRSA that infected nurses who became MRSA carriers in a geriatric nursing facility. In the latter case, removal of the cat from the ward resolved the MRSA outbreak. A recent comprehensive review regarding MRSA in dogs and cats concluded that the risk to human health appears to be small but that a survey of MRSA in animals was required (4).
The significance of MRSA nosocomial infections in veterinary hospitals in the United States has not been documented. While surveying all veterinary hospitals in the United States was unrealistic, we felt that a survey of veterinary teaching hospitals via their associated diagnostic laboratories would provide some baseline data on the importance of MRSA in veterinary hospitals. Hence, the goal of the present study was to determine the proportion of S. aureus isolates from veterinary teaching hospital diagnostic laboratory submissions that were methicillin resistant and to determine whether S. aureus isolates were likely to originate from a common source. Based on previous studies, we hypothesized that MRSA would be most commonly isolated from equine surgical patients and would be acquired from a common source or sources within the hospital.
Representatives from the bacteriology section of the diagnostic laboratories associated with each of the 27 veterinary teaching hospitals in the United States were contacted by e-mail and telephone. Each was given a description of the study and asked to participate in a 6-month surveillance study between 1 October 2001 and 31 March 2002.
Bacterial isolates identified as S. aureus by the participating laboratories were sent to the principal investigator (PI) (J. R. Middleton) in storage media (brain heart infusion slants; Becton Dickinson & Co., Sparks, MD), and stored at −80°C until processed. Medical history and patient signalment, when available, were collected with each S. aureus isolate. All S. aureus isolates submitted to the PI were streaked onto Columbia blood agar containing 5% sheep blood (Remel, Lenexa, KS) and incubated at 37°C for 24 h. Staphylococcus aureus isolates were identified based on colonial morphology, hemolytic patterns, and the tube coagulase test (Remel). All isolates were confirmed to be S. aureus using a commercial biochemical typing system (apiSTAPH; bioMerieux, St. Louis, MO).
All isolates confirmed to be S. aureus were tested for methicillin resistance by determining the MIC of oxacillin for each using the broth microdilution technique (Sensititre; TREK Diagnostic Systems Inc., Cleveland, OH) in accordance with National Committee for Clinical Laboratory Standards (NCCLS) guidelines (8). Staphylococcus aureus isolates with an MIC of ≥4 μg/ml for oxacillin were considered to be MRSA. Staphylococcus aureus isolates with an MIC of ≤2 μg/ml for oxacillin were regarded as susceptible to methicillin. The S. aureus quality control strain used in the MIC determinations was ATCC 29213 (American Type Culture Collection, Manassas, VA). MICs for ampicillin, ceftiofur, cephalothin, erythromycin, penicillin, penicillin-novobiocin, pirlimycin, sulfadimethoxine, and tetracycline were also determined, as these were present on the 96-well plate used to determine oxacillin resistance.
All S. aureus isolates were strain typed using the technique described by Middleton et al. (7). Briefly, SmaI digests (Invitrogen, Carlsbad, CA) of staphylococcal DNA were separated by pulsed-field gel electrophoresis (PFGE) in a 1% agarose gel (pulsed-field certified agarose; Bio-Rad, Hercules, CA) immersed in 0.5% Tris-boric acid-EDTA buffer at 14°C for 20 h at 6 V/cm with a 5- to 50-s pulse time on a CHEF DRIII PFGE machine (Bio-Rad, Hercules, CA). A lambda ladder molecular weight size standard (Bio-Rad, Hercules, CA) was run in the first and last lanes of each gel. Isolate banding patterns were compared visually, and isolates with identical PFGE banding patterns were regarded as the same strain type. All isolates suspected of being different strains based on initial visual examination of each gel were run side-by-side on a subsequent gel for confirmation.
Fisher's exact test (Sigma Stat 2.03; Sigma, San Rafael, CA) was used to compare methicillin-resistant proportions of S. aureus isolates between institutions and animal species (α = 0.05, where α is the probability of a type I error). Isolates were categorized based on the site of isolation and animal species from which they were isolated.
Twenty-two diagnostic laboratories agreed to participate by verbal or electronic communication at the beginning of the study. At the conclusion of the study, six laboratories submitted S. aureus isolates to the PI for further examination (Table 1), and one further laboratory reported that no S. aureus had been isolated from teaching hospital submissions during the study period. The seven participating laboratories submitted 70 S. aureus isolates from 65 patients to the PI. The mean number of patients with an S. aureus infection ± the standard deviation was 9 ± 7 patients per institution. Confirmatory testing in the PI's laboratory showed that each isolate was correctly identified as S. aureus by the contributing laboratory. Patient description and available medical history were provided for all but two isolates from two patients.
TABLE 1.
Cumulative data for veterinary patients with Staphylococcus aureus infections categorized by site of infection, species affected, and infections that were acquired after a surgical procedure
| Site of isolation | No. of animal patients from which S. aureus (MRSA) was isolated
|
||||||
|---|---|---|---|---|---|---|---|
| Avian | Bovine | Canine | Equine | Feline | All species | Postoperative infectiona | |
| Blood | 2 | 2 | |||||
| Feces | 1 | 1 | |||||
| Genitourinary tract | 10 | 3 | 13 | ||||
| Integument | 1 | 8 (2) | 7 (2) | 16 (4) | 4 | ||
| Milk | 7 | 7 | |||||
| Musculoskeletal system | 1 | 6 (1) | 5 (1) | 1 | 13 (2) | 5 (1) | |
| Eyes | 2 | 1 (1) | 3 (1) | ||||
| Ears | 3 | 3 | 1 | ||||
| Respiratory tract | 3 (1) | 1 | 1 (1) | 5 (2) | |||
| Unknown | 1 | 1 | 2 | ||||
| All S. aureus | 2 | 7 | 36 | 18 | 2 | 65 | 10 |
| All MRSA | 4 | 4 | 1 | 9 | 1 | ||
| % MRSA | 0 | 0 | 11 | 22 | 50 | 14 | 10 |
Ten of 65 patients had a postoperative infection. Nine of 10 patients with postoperative infections were dogs, and one was a cat. The MRSA isolate was from a canine orthopedic pin tract infection.
The 70 S. aureus isolates were grouped by body system/site of isolation as follows: blood (n = 2), feces (n = 1), genitourinary tract (n = 13), integument (n = 18), milk (n = 7), musculoskeletal system (n = 15), ocular (n = 3), otic (n = 3), respiratory tract (n = 6), and unknown (n = 2). The 65 patients were grouped by species as follows: avian (n = 2), bovine (n = 7), canine (n = 36), equine (n = 18), and feline (n = 2). Institution A submitted two isolates from each of four patients (one avian and three canine), and laboratory F submitted two isolates from a single canine patient. All other patients were represented by a single S. aureus isolate. Cumulative data for all institutions are shown in Table 1.
The majority (83%; 54 of 65) of patients with S. aureus infections were canine and equine (n = 36 and 18 patients, respectively). Overall, 9 of 65 (14%) patients had an MRSA infection and 9 of 70 (13%) isolates were MRSA. Methicillin-resistant S. aureus infections were most prevalent (89%; eight of nine) among canine and equine patients (four each). One of two feline patients, none of two avian patients (three isolates), and none of seven bovine patients had an MRSA infection. The proportions of patients infected with an MRSA strain were not significantly different between institutions (P ≥ 0.342). Likewise, when the cumulative data from all institutions were analyzed, the proportions of MRSA-infected patients were not significantly different between species (P ≥ 0.222). In addition to isolates from veterinary patients, a single MRSA isolate from a human which was collected as part of a hand-washing study from a person working in the small-animal area of their veterinary teaching hospital was submitted by institution F. All MRSA isolates were multidrug resistant, with isolates being resistant to 5 to 8 of 10 antibiotics tested. All MRSA isolates were resistant to ampicillin, ceftiofur, cephalothin, oxacillin, and penicillin. Seven of nine were resistant to erythromycin, two of nine were resistant to pirlimycin, five of nine were resistant to sulfadimethoxine, and four of nine were resistant to tetracycline. Within individual institutions none of the isolates exhibited identical antimicrobial resistance patterns. The human MRSA isolate was resistant to 8 of 10 antibiotics tested.
Sixty-seven isolates were available for strain typing by PFGE (three isolates yielded no growth from storage media). Four of 67 isolates did not generate an interpretable PFGE banding pattern and were excluded from this portion of the data analysis. Pulsed-field gel electrophoresis banding patterns were compared between isolates within each institution. Overall, 58 different S. aureus strain types were identified among the 63 isolates that were successfully typed. The number of S. aureus strains per institution ranged from 4 to 18 (Table 2). All isolates from institutions C, D, and E had unique PFGE banding patterns. Institution A had two isolates from a single canine patient, two isolates from a single avian patient, and two isolates from two different equine patients that shared a PFGE banding pattern. Institution B had one isolate from a bovine patient and one isolate from an equine patient that shared the same strain type. Finally, institution F had two isolates from a single canine patient that shared the same strain type (Fig. 1). The PFGE banding patterns of none of the MRSA isolates matched each other or those of other S. aureus isolates from patients at a given institution. The human MRSA isolate from institution F had a PFGE banding pattern different from those of other S. aureus isolates from institution F and from those of the other nine MRSA isolates.
TABLE 2.
Numbers of Staphylococcus aureus isolates, isolates typed by PFGE, and strain types identified by PFGE at each institution
| Institution | No. of S. aureus isolates
|
No. of S. aureus strains identified by PFGE | |
|---|---|---|---|
| Overall | Typed by PFGE | ||
| A | 23 | 21 | 18 |
| B | 10 | 10 | 9 |
| C | 8 | 5 | 5 |
| D | 6 | 6 | 6 |
| E | 17 | 16 | 16 |
| F | 6 | 5 | 4 |
| G | 0 | 0 | 0 |
| Total | 70 | 63 | 57 |
FIG. 1.
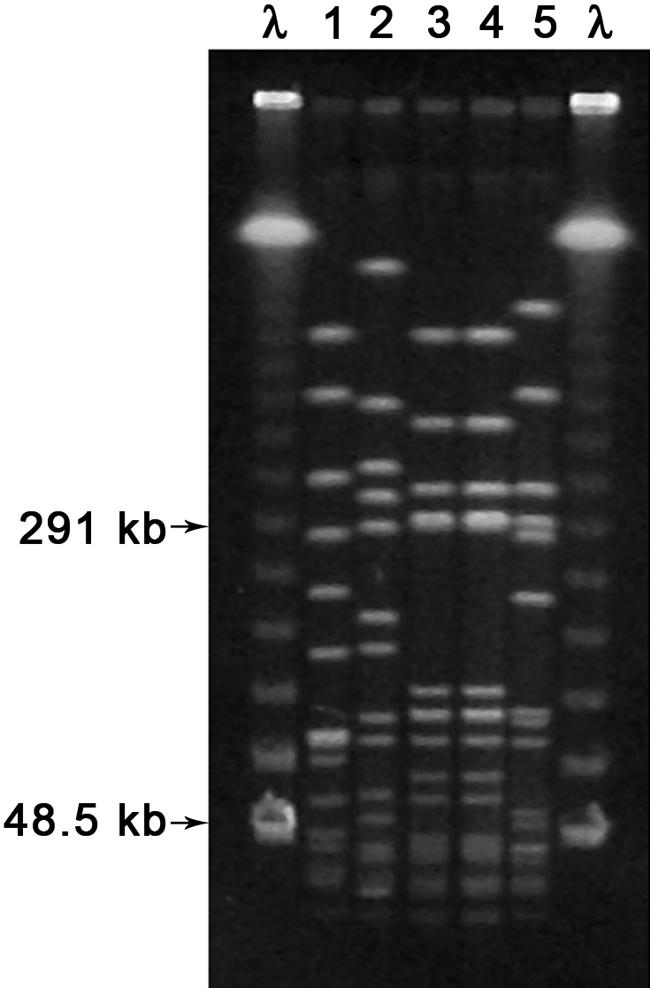
Photograph of the results of PFGE of SmaI digests of five S. aureus isolates from institution F. The first and last lanes represent lambda (λ) ladder size standards. Lanes 1 and 2 represent S. aureus isolates from each of two different equine patients, lanes 3 and 4 represent two S. aureus isolates from a single canine patient, and lane 5 represents a single MRSA isolate from a second canine patient. Lanes with identical banding patterns were considered to indicate the same strain.
Based on patient histories, isolates were identified as being from postoperative or nonpostoperative infections (Table 1). Postoperative infections were defined as those which occurred while the patient was at the veterinary teaching hospital and in each case followed a surgical intervention. Institutions B, C, D, and G did not have any postoperative S. aureus infections. Overall, 15% (10 of 65) of patients had a postoperative S. aureus infection, with only 1 of 10 infections being caused by an MRSA strain. The only postoperative MRSA isolate was from a canine patient with an orthopedic pin tract infection.
While the number of S. aureus isolations at the seven institutions studied seems small, the true prevalence of S. aureus strains among veterinary diagnostic laboratory submissions could not be determined because we did not have data on each teaching hospital's case load or the number of bacteriology submissions from each veterinary teaching hospital. Staphylococcus aureus strains were most commonly isolated from canine and equine patients, but our results may reflect a sampling bias due to case loads being skewed towards canine and equine patients. Staphylococcus aureus strains are commonly isolated from bovine milk, whereas the most common coagulase-positive staphylococcus isolated from small animals tends to be Staphylococcus intermedius (4, 7). Only seven bovine isolates (all from milk) were collected, suggesting that the participating diagnostic laboratories did not routinely perform milk cultures. In addition, because only 7 of 22 institutions provided data on S. aureus infections, there may have been a nonresponse bias in that institutions with a high prevalence of S. aureus and/or MRSA infections may have opted not to submit data or isolates.
The diversity of S. aureus strains identified in the present study suggests that most S. aureus infections at the institutions studied were community acquired rather than hospital acquired. Only two institutions (A and B) had an S. aureus strain that was shared by more than one patient. Institution A had one S. aureus strain that was shared by two different equine patients, each with a leg wound, suggesting a possible common source of infection. Institution B isolated the same strain of S. aureus from a bovine milk sample and an equine wound infection, but a common fomite or individual shared between these two cases was not evident, although the submissions were made to the diagnostic lab within 7 days of each other.
While only 9 of the 70 S. aureus isolates were found to be MRSA, MRSA strains were isolated from at least one patient in all but one of the veterinary teaching hospitals. There were no detectable differences in the proportions of S. aureus isolates that were MRSA between animal species or institutions (P ≥ 0.222), but this may be due to the lack of statistical power associated with the low numbers of S. aureus and MRSA isolates collected at each institution. Based on PFGE fingerprinting, the MRSA infections appeared to be community acquired rather than hospital acquired. The single human isolate had a different PFGE banding pattern from all other S. aureus isolates at the same institution and was unique compared with the other nine MRSA isolates. In addition, while 10 postoperative S. aureus infections were identified, only one was caused by an MRSA. These results are in agreement with a recent report which evaluated 311 staphylococcal isolates from animals in The Netherlands and found only two MRSA isolates, each with a unique PFGE banding pattern (14). Data regarding human MRSA infections suggest that community-acquired MRSA infections may be at least as important as hospital-acquired MRSA infections, particularly in groups of patients without predisposing risk factors for infection (4).
Since the data for this study were collected, institution D reported a small outbreak of MRSA infections in horses (R. D. Welsh, personal communication). Similarly, institution C reported that between January 2003 and July 2004, 59 S. aureus strains were isolated from dogs, with 27% of these isolates being MRSA (J. M. Kinyon, personal communication). Several other institutions which did not participate in our study verbally reported to the PI that they had experienced MRSA outbreaks in equine patients in recent years. A recent report from Canada concurs with earlier reports that equine populations seem to be at risk for hospital-acquired MRSA infections (15). The same report provides evidence that MRSA may be harbored in the environment in veterinary teaching hospitals, which is in contrast to traditional dogma that MRSA infections are harbored by hospital personnel. Our study evaluated only isolates from veterinary patients and a single human patient, so we cannot draw any conclusions about environmental sources of MRSA in the seven hospitals studied.
In summary, based on the data presented here it seems prudent to monitor veterinary hospitals and hospital patients for MRSA. While previous reports in the veterinary literature detail outbreaks of MRSA associated with nosocomial infection, the data presented here demonstrate that community-acquired MRSA infections occur with some frequency. The recent review by Duquette and Nuttall (4) concluded that the risk to human health from animal MRSA infections is small but that monitoring for MRSA in therapy animals and pets of immunocompromised individuals is indicated. Transfer of resistance genes between animal and human staphylococci cannot be ignored, and therefore, in addition to MRSA surveillance as part of a veterinary hospital biosafety program, responsible use of antimicrobials in veterinary patients should be practiced.
Acknowledgments
This research was funded by a USDA Animal Health Formula Fund grant (project no. CD000142).
We thank Julie Holle, Marion Butcher, Ryan Bader, Antonio Lombeida, and Irene Ganjam for technical assistance.
REFERENCES
- 1.Cefai, C., S. Ashurst, and C. Owens. 1994. Human carriage of methicillin resistant Staphylococcus aureus linked with pet dog. Lancet 344:539-540. [DOI] [PubMed] [Google Scholar]
- 2.Devriese, L. A., and J. Hommez. 1975. Epidemiology of methicillin-resistant Staphylococcus aureus in dairy herds. Res. Vet. Sci. 19:23-27. [PubMed] [Google Scholar]
- 3.Devriese, L. A., L. R. Van Damme, and L. Fameree. 1972. Methicillin (cloxacillin)-resistant Staphylococcus aureus strains isolated from bovine mastitis cases. Zentbl. Vetmed. Reihe B 19:598-605. [DOI] [PubMed] [Google Scholar]
- 4.Duquette, R. A., and T. J. Nuttall. 2004. Methicillin-resistant Staphylococcus aureus in dogs and cats: an emerging problem? J. Small Anim. Pract. 45:591-597. [DOI] [PubMed] [Google Scholar]
- 5.Emori, T. G., and R. P. Gaynes. 1993. An overview of nosocomial infections, including the role of the microbiology laboratory. Clin. Microbiol. Rev. 6:428-442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hartmann, F. A., S. S. Trostle, and A. A. O. Klohen. 1997. Isolation of methicillin-resistant Staphylococcus aureus from a postoperative wound infection in a horse. J. Am. Vet. Med. Assoc. 211:590-592. [PubMed] [Google Scholar]
- 7.Middleton, J. R., L. K. Fox, J. M. Gay, J. W. Tyler, and T. E. Besser. 2002. Use of pulsed-field gel electrophoresis for detecting differences in Staphylococcus aureus strain populations between dairy herds with different cattle importation practices. Epidemiol. Infect. 129:387-395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.National Committee for Clinical Laboratory Standards. 2003. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard (document no. M07-A6), 6th ed. NCCLS, Wayne, Pa.
- 9.Pak, S. I., H. R. Han, and A. Shimizu. 1999. Characterization of methicillin-resistant Staphylococcus aureus isolated from dogs in Korea. J. Vet. Med. Sci. 61:1013-1018. [DOI] [PubMed] [Google Scholar]
- 10.Scott, G. M., R. Thomson, J. Malone-Lee, and G. L. Ridgway. 1988. Cross-infection between animals and man: possible feline transmission of Staphylococcus aureus infection in humans? J. Hosp. Infect. 12:29-34. [DOI] [PubMed] [Google Scholar]
- 11.Seguin, J. C., R. D. Walker, J. P. Caron, W. E. Kloos, C. G. George, R. J. Hollis, R. N. Jones, and M. A. Pfaller. 1999. Methicillin-resistant Staphylococcus aureus outbreak in a veterinary teaching hospital: potential human-to-animal transmission. J. Clin. Microbiol. 37:1459-1463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shimizu, A., J. Kawano, C. Yamamoto, O. Kakutani, T. Anzai, and M. Kamada. 1997. Genetic analysis of equine methicillin-resistant Staphylococcus aureus by pulsed-field gel electrophoresis. J. Vet. Med. Sci. 59:935-937. [DOI] [PubMed] [Google Scholar]
- 13.Tomlin, J., M. J. Pead, D. H. Lloyd, S. Howell, F. Hartmann, H. A. Jackson, and P. Muir. 1999. Methicillin-resistant Staphylococcus aureus infections in 11 dogs. Vet. Rec. 144:60-64. [DOI] [PubMed] [Google Scholar]
- 14.van Duijkeren, E., A. T. A. Box, M. E. O. C. Heck, W. J. B. Wannet, and A. C. Fluit. 2004. Methicillin-resistant staphylococci isolated from animals. Vet. Microbiol. 103:91-97. [DOI] [PubMed] [Google Scholar]
- 15.Weese, J. S., T. DaCosta, L. Button, K. Goth, M. Ethier, and K. Boehnke. 2004. Isolation of methicillin-resistant Staphylococcus aureus from the environment in a veterinary teaching hospital. J. Vet. Intern. Med. 18:468-470. [DOI] [PubMed] [Google Scholar]


