Abstract
Introduction
Patients with anaplastic lymphoma kinase (ALK)-rearranged non-small cell lung cancer (NSCLC) are at increased risk of central nervous system (CNS) metastasis at initial diagnosis and throughout treatment. In a phase 3 trial, lorlatinib, a third-generation ALK tyrosine kinase inhibitor, significantly improved progression-free survival. In further analysis, lorlatinib revealed superior intracranial efficacy and prolonged time to intracranial progression compared with crizotinib.
Case Presentation
Herein, we report a case of ALK-positive NSCLC with leptomeningeal metastasis that was successfully treated with lorlatinib after progression to brigatinib and alectinib. This case demonstrates the potential of lorlatinib in managing leptomeningeal metastasis in ALK-positive NSCLC.
Conclusion
The case suggests a paradigm shift in therapeutic approaches for CNS metastasis, including brain and leptomeningeal metastases.
Keywords: Anaplastic lymphoma kinase fusion, Leptomeningeal metastasis, Lorlatinib, Non-small cell lung cancer
Introduction
The discovery of driver mutations in advanced non-small cell lung cancer (NSCLC) has considerably influenced treatment approaches based on the molecular characteristics of the tumor [1, 2]. Molecular targeted agents (MTAs) demonstrate high efficacy and durability; however, most patients eventually develop resistance [3]. The main types of resistance to tyrosine kinase inhibitors (TKIs) include acquired mutations in the kinase domain of cancer cells, activation of bypass signaling pathways, and progression in sanctuary sites, such as the central nervous system (CNS), where TKI penetration is limited.
Anaplastic lymphoma kinase (ALK)-rearranged NSCLC accounts for 2%–5% of lung adenocarcinomas. These patients have a higher risk of brain metastasis at initial diagnosis and throughout treatment than those with non-ALK NSCLC [4–6]. As of 2024, five ALK-TKIs, namely crizotinib, alectinib, ceritinib, lorlatinib, and brigatinib, have been approved for the treatment of advanced ALK-positive NSCLC. As brain and CNS metastases significantly lead to a decreased quality of life and poor survival, treatment is performed with these concerns [7]. Lorlatinib, a third-generation ALK-TKI, has been engineered to overcome many ALK resistance mutations and enhance CNS penetration [8].
In the CROWN trial, lorlatinib significantly improved progression-free survival [9]. Further analysis revealed superior intracranial efficacy and prolonged time to intracranial progression compared with crizotinib, even in patients with and without preexisting brain metastases [9–11]. Although patients with leptomeningeal metastasis were excluded from the CROWN trial, this case report discusses a patient with ALK-positive + NSCLC who experienced a durable response to lorlatinib after developing leptomeningeal metastases. Written informed consent was obtained from the patient for publication of this case report and any accompanying images. The CARE Checklist has been completed by the authors for this case report, attached as online supplementary material (for all online suppl. material, see https://doi.org/10.1159/000540445).
Case Presentation
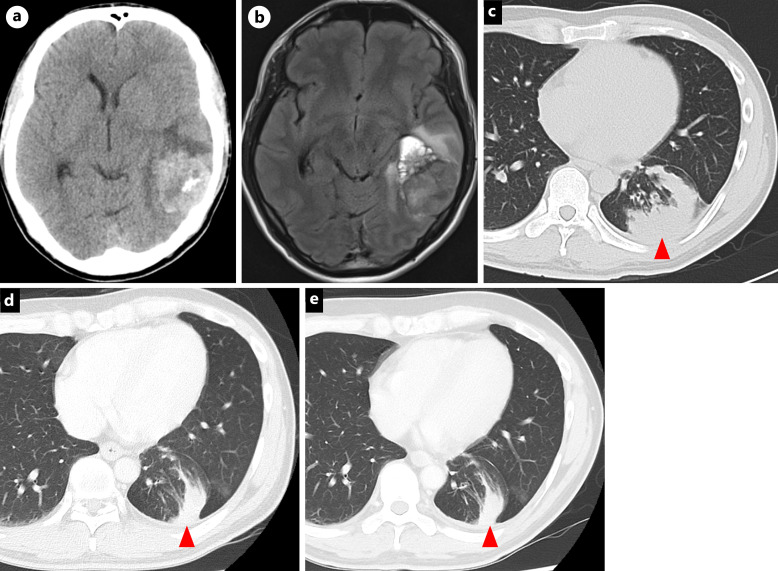
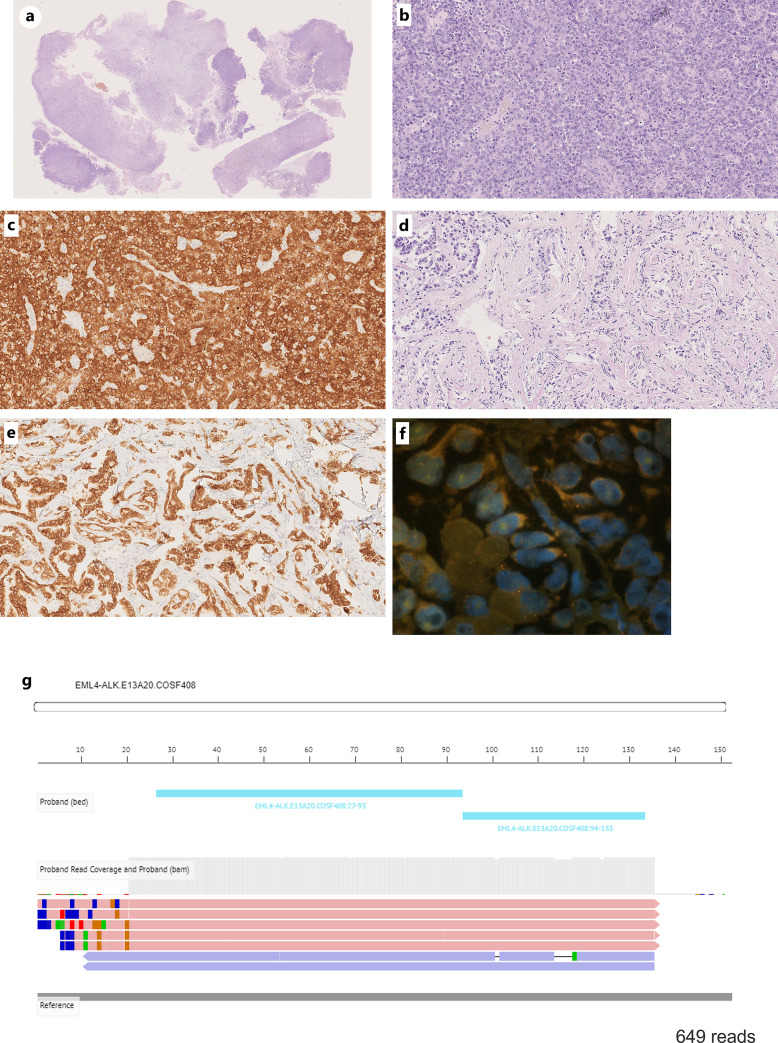
A 35-year-old man with a 17-pack-year smoking history presented with headache and diplopia prompting an emergency room visit in January 20XX. Computed tomography (CT) and brain magnetic resonance imaging showed a 40 mm long solitary brain tumor in the left temporal lobe causing a midline shift (Fig. 1a, b). CT revealed a primary lung cancer in the left lower lobe (Fig. 1c). After brain tumor surgery, a metastatic brain tumor from NSCLC was pathologically diagnosed, and the EML4-ALK fusion was identified through fluorescence in situ hybridization and immunohistochemistry testing (Fig. 2a–f). We initially diagnosed the NSCLC as stage IVA (cT2bN2M1b in the TNM 8th edition).
Fig. 1.
Brain CT (a), MRI (b), and chest CT (c) at initial diagnosis. Chest CT at the onset of leptomeningeal metastases (d) and 2 years after lorlatinib (e). Red arrow: primary lung lesion. CT, computed tomography; MRI, magnetic resonance imaging.
Fig. 2.
Histopathological findings of brain metastasis in low power field (a). Hematoxylin and eosin (HE) stained shows a predominant solid adenocarcinoma (b) with mixed spindle and acinar components (c). ALK-IHC staining (5A4, Abcam) was positive in both solid adenocarcinoma (d) and acinar components (e). FISH testing detected ALK rearrangement (f). Next generation panel sequence detected EML4-ALK variant 1 (E13:A20) (g). FISH, fluorescence in situ hybridization; IHC, immunohistochemistry.
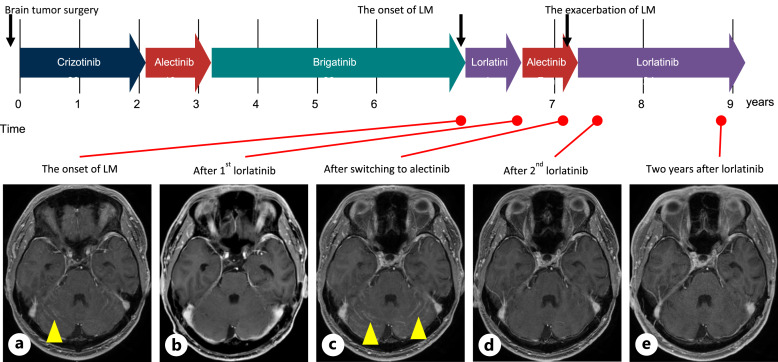
In March 20XX, he started receiving crizotinib for 2 years, followed by alectinib for 1 year and brigatinib for 3 years due to disease progression. In September 20XX + 6, he developed leptomeningeal metastases with diplopia, as confirmed by magnetic resonance imaging (Fig. 1d, 3; online suppl. Fig. 1A) and started to receive lorlatinib. However, lorlatinib at 100 mg/day caused adverse psychological events, including dementia and mood alterations, including depression and suicidal ideation. He hoped to switch back to alectinib, but leptomeningeal metastases progressed 3 months after re-administration of alectinib (Fig. 3; online suppl. Fig. 1B, C). A carefully shared decision-making process led to the resumption of lorlatinib at a reduced dose of 75 mg/day from June 20XX + 7. He developed dyslipidemia; however, psychological events and mood alterations did not recur. He continued lorlatinib treatment, with a sustained complete response to the leptomeningeal metastases after 2 years (Fig. 1e, 3; online suppl. Fig. 1D, E).
Fig. 3.
Timeline of the patient’s treatment history and representative brain MRI. Brain MRI at the onset of LM (a), after 1st lorlatinib (b), after switching to alectinib (c), after 2nd lorlatinib (d), and at present (e). Yellow arrow: leptomeningeal metastases. LM, leptomeningeal metastases; MRI, magnetic resonance imaging.
Discussion
This report highlights the potential efficacy of lorlatinib in treating leptomeningeal metastases in patients with ALK-positive NSCLC, even when brigatinib and alectinib failed to treat leptomeningeal metastases. Adjusting the lorlatinib dose effectively managed the adverse psychological effects, emphasizing the importance of personalized treatment strategies to balance efficacy and quality of life.
Pathological findings of the brain tumor revealed a predominantly solid adenocarcinoma with mixed spindle and acinar components, all of which were positive for ALK-immunohistochemistry staining. The EML4-ALK variant 1 (E13:A20) using Ion Torrent S5 Sequencer™ (Thermo Fisher Scientific, Wilmington, DE, USA) was confirmed in this patient (Fig. 2g). EML4-ALK fusion accounts for approximately 85% of the ALK fusion variants with many of the EML4-ALK variants depending on the fusion breakpoint of EML4 to the exon 20 of the ALK gene. EML4-ALK variant 1 (E13:A20) is one of the most common EML4-ALK variants, along with variant 2 (E20:A20) and variant 3 (E6:A20) [12]. ALK-TKIs are generally longer progression-free survival in patients with EML4-ALK variant 1 than in those with EML4-ALK variant 3 [13–15]. The patient had a highly sensitive variant 1 to ALK-TKIs, which may have contributed to his long-term survival.
It has previously been noted that the cerebrospinal/plasma concentration ratio observed with lorlatinib (range: 0.61–0.96) was much higher than with crizotinib (range: 0.0006–0.026) and alectinib (range: 0.002–0.005), although alectinib also had antitumor activity in patients with CNS metastasis [16, 17]. The superior CNS penetration of lorlatinib was attributed to the complete and reproducible response on leptomeningeal metastasis in this patient, even when leptomeningeal metastasis was progressed with brigatinib and alectinib. Highly potent ALK inhibition of lorlatinib may avoid the need for whole-brain irradiation in such cases [18, 19].
Several studies have discussed the mechanism of ALK-TKI resistance [20–24]. Gainor et al. [20] reported that lorlatinib retained its potency against G1202R, G1202del, and D1203N+E1210K mutations, whereas other ALK-TKIs were inactive. This patient might have a mutation that was resistant to crizotinib, alectinib, and brigatinib but sensitive to lorlatinib. Because the progressive site was only the leptomeningeal metastasis, no tissue specimens were available after progression to these previous ALK-TKIs. This is a limitation of this case report.
Conclusion
This case demonstrates the potential of lorlatinib in managing leptomeningeal metastasis in ALK-positive NSCLC, suggesting a paradigm shift in therapeutic approaches for CNS metastasis.
Acknowledgments
The authors greatly appreciate the patient and his family and the assistance of the staff of the Department of Thoracic Oncology, Aichi Cancer Center. We would like to thank Editage (www.editage.com) for the English language editing.
Statement of Ethics
This case report was approved by the Ethics Committee of Aichi Cancer Center, Nagoya, Japan (permit no. T06004). Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Conflict of Interest Statement
Y.F. reports receiving personal fees for honoraria for lectures from AstraZeneca, Amgen, Bristol Myers Squibb, Chugai, Daiichi-Sankyo, Eli Lilly, Merck Biopharma, Merck Sharp & Dohme, Novartis, Novocure, Ono Pharmaceutical, Pfizer, Takeda, and Taiho Pharmaceutical, outside the submitted work; personal fees for being on the advisory board from AstraZeneca, Chiome Bioscience, Daiichi-Sankyo, Micron, Otsuka Pharmaceutical, and Ono Pharmaceutical, outside the submitted work; and research grants from AbbVie, Amgen, AnHeart, AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Chugai, Eli Lilly, Incyte, Merck KGaA, Merck Sharp & Dohme, and Taiho Pharmaceutical, outside the submitted work. R.M. reports receiving personal fees for honoraria for lectures from AstraZeneca, Boehringer Ingelheim, Chugai, Eli Lilly, Merck Sharp & Dohme, Ono Pharmaceutical, Pfizer, and Taiho Pharmaceutical, outside the submitted work. T.Y. reports receiving personal fees for honoraria for lectures from AstraZeneca, Bristol Myers Squibb, Boehringer Ingelheim, Eli Lilly, Merck Biopharma, Merck Sharp & Dohme, Ono Pharmaceutical, and Taiho Pharmaceutical, outside the submitted work, and reports receiving personal fees for being on the advisory board from Daiichi-Sankyo. J.S. reports receiving personal fees for honoraria for lectures from AstraZeneca, Amgen, Chugai, Merck Biopharma, Merck Sharp & Dohme, Novartis, Pfizer, Taiho Pharmaceutical, and Takeda Pharmaceutical, outside the submitted work. The other authors have no conflicts of interest to declare.
Funding Sources
This study was not supported by any sponsor or funding.
Author Contributions
Y.F. was responsible for the clinical management of the patient and contributed to writing this manuscript. K.M. and E.S. interpreted the histological data and contributed to the histological analysis. R.M., T.Y., N.W., J.S., and Y.H. wrote, reviewed, and edited the manuscript. All the authors have read and approved the final manuscript.
Funding Statement
This study was not supported by any sponsor or funding.
Data Availability Statement
All data generated or analyzed in this study have been included in the published article. Further inquiries can be directed to the corresponding authors.
Supplementary Material.
Supplementary Material.
Supplementary Material.
Supplementary Material.
Supplementary Material.
Supplementary Material.
References
- 1. Jordan EJ, Kim HR, Arcila ME, Barron D, Chakravarty D, Gao J, et al. Prospective comprehensive molecular characterization of lung adenocarcinomas for efficient patient matching to approved and emerging therapies. Cancer Discov. 2017;7(6):596–609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Kalemkerian GP, Narula N, Kennedy EB, Biermann WA, Donington J, Leighl NB, et al. Molecular testing guideline for the selection of patients with lung cancer for treatment with targeted tyrosine kinase inhibitors: American society of clinical Oncology endorsement of the college of American pathologists/international association for the study of lung cancer/association for molecular pathology clinical practice guideline update. J Clin Oncol. 2018;36(9):911–9. [DOI] [PubMed] [Google Scholar]
- 3. Kris MG, Johnson BE, Berry LD, Kwiatkowski DJ, Iafrate AJ, Wistuba II, et al. Using multiplexed assays of oncogenic drivers in lung cancers to select targeted drugs. JAMA. 2014;311(19):1998–2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Gainor JF, Tseng D, Yoda S, Dagogo-Jack I, Friboulet L, Lin JJ, et al. Patterns of metastatic spread and mechanisms of resistance to crizotinib in ROS1-positive non-small-cell lung cancer. JCO Precis Oncol. 2017;2017(1):1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Drilon A, Lin JJ, Filleron T, Ni A, Milia J, Bergagnini I, et al. Frequency of brain metastases and multikinase inhibitor outcomes in patients with RET-rearranged lung cancers. J Thorac Oncol. 2018;13(10):1595–601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Patil T, Smith DE, Bunn PA, Aisner DL, Le AT, Hancock M, et al. The incidence of brain metastases in stage IV ROS1-rearranged non-small cell lung cancer and rate of central nervous system progression on crizotinib. J Thorac Oncol. 2018;13(11):1717–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Peters S, Bexelius C, Munk V, Leighl N. The impact of brain metastasis on quality of life, resource utilization and survival in patients with non-small-cell lung cancer. Cancer Treat Rev. 2016;45:139–62. [DOI] [PubMed] [Google Scholar]
- 8. Johnson TW, Richardson PF, Bailey S, Brooun A, Burke BJ, Collins MR, et al. Discovery of (10R)-7-amino-12-fluoro-2,10,16-trimethyl-15-oxo-10,15,16,17-tetrahydro-2H-8,4-(metheno)pyrazolo[4,3-h] [2,5,11]-benzoxadiazacyclotetradecine-3-carbonitrile (PF-06463922), a macrocyclic inhibitor of Anaplastic Lymphoma Kinase (ALK) and c-ros oncogene 1 (ROS1) with preclinical brain exposure and broad-spectrum potency against ALK-resistant mutations. J Med Chem. 2014;57(11):4720–44. [DOI] [PubMed] [Google Scholar]
- 9. Shaw AT, Bauer TM, de Marinis F, Felip E, Goto Y, Liu G, et al. First-Line lorlatinib or crizotinib in advanced ALK-positive lung cancer. N Engl J Med. 2020;383(21):2018–29. [DOI] [PubMed] [Google Scholar]
- 10. Solomon BJ, Bauer TM, Ignatius Ou SH, Liu G, Hayashi H, Bearz A, et al. Post hoc analysis of lorlatinib intracranial efficacy and safety in patients with ALK-positive advanced non-small-cell lung cancer from the phase III CROWN study. J Clin Oncol. 2022;40(31):3593–602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Solomon BJ, Bauer TM, Mok TSK, Liu G, Mazieres J, de Marinis F, et al. Efficacy and safety of first-line lorlatinib versus crizotinib in patients with advanced, ALK-positive non-small-cell lung cancer: updated analysis of data from the phase 3, randomised, open-label CROWN study. Lancet Respir Med. 2023;11(4):354–66. [DOI] [PubMed] [Google Scholar]
- 12. Zhang SS, Nagasaka M, Zhu VW, Ou SHI. Going beneath the tip of the iceberg. Identifying and understanding EML4-ALK variants and TP53 mutations to optimize treatment of ALK fusion positive (ALK+) NSCLC. Lung Cancer. 2021;158:126–36. [DOI] [PubMed] [Google Scholar]
- 13. Lin JJ, Zhu VW, Yoda S, Yeap BY, Schrock AB, Dagogo-Jack I, et al. Impact of EML4-ALK variant on resistance mechanisms and clinical outcomes in ALK-positive lung cancer. J Clin Oncol. 2018;36(12):1199–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Camidge DR, Kim HR, Ahn MJ, Yang JCH, Han JY, Hochmair MJ, et al. Brigatinib versus crizotinib in ALK inhibitor-naive advanced ALK-positive NSCLC: final results of phase 3 ALTA-1L trial. J Thorac Oncol. 2021;16(12):2091–108. [DOI] [PubMed] [Google Scholar]
- 15. Bearz A, Martini JF, Jassem J, Kim SW, Chang GC, Shaw AT, et al. Efficacy of lorlatinib in treatment-naive patients with ALK-positive advanced NSCLC in relation to EML4::ALK variant type and ALK with or without TP53 mutations. J Thorac Oncol. 2023;18(11):1581–93. [DOI] [PubMed] [Google Scholar]
- 16. Gadgeel SM, Gandhi L, Riely GJ, Chiappori AA, West HL, Azada MC, et al. Safety and activity of alectinib against systemic disease and brain metastases in patients with crizotinib-resistant ALK-rearranged non-small-cell lung cancer (AF-002JG): results from the dose-finding portion of a phase 1/2 study. Lancet Oncol. 2014;15(10):1119–28. [DOI] [PubMed] [Google Scholar]
- 17. Sun S, Pithavala YK, Martini JF, Chen J. Evaluation of lorlatinib cerebrospinal fluid concentrations in relation to target concentrations for Anaplastic Lymphoma Kinase (ALK) inhibition. J Clin Pharmacol. 2022;62(9):1170–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Vogelbaum MA, Brown PD, Messersmith H, Brastianos PK, Burri S, Cahill D, et al. Treatment for brain metastases: ASCO-SNO-ASTRO guideline. J Clin Oncol. 2022;40(5):492–516. [DOI] [PubMed] [Google Scholar]
- 19. Meng Y, Zhu M, Yang J, Wang X, Liang Y, Yu M, et al. Treatment advances in lung cancer with leptomeningeal metastasis. Curr Cancer Drug Targets. 2024;24(9):910–9. [DOI] [PubMed] [Google Scholar]
- 20. Gainor JF, Dardaei L, Yoda S, Friboulet L, Leshchiner I, Katayama R, et al. Molecular mechanisms of resistance to first- and second-generation ALK inhibitors in ALK-rearranged lung cancer. Cancer Discov. 2016;6(10):1118–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Sabari JK, Santini FC, Schram AM, Bergagnini I, Chen R, Mrad C, et al. The activity, safety, and evolving role of brigatinib in patients with ALK-rearranged non-small cell lung cancers. Onco Targets Ther. 2017;10:1983–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Yoda S, Lin JJ, Lawrence MS, Burke BJ, Friboulet L, Langenbucher A, et al. Sequential ALK inhibitors can select for lorlatinib-resistant compound ALK mutations in ALK-positive lung cancer. Cancer Discov. 2018;8(6):714–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Yanagitani N, Uchibori K, Koike S, Tsukahara M, Kitazono S, Yoshizawa T, et al. Drug resistance mechanisms in Japanese anaplastic lymphoma kinase-positive non-small cell lung cancer and the clinical responses based on the resistant mechanisms. Cancer Sci. 2020;111(3):932–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Mizuta H, Okada K, Araki M, Adachi J, Takemoto A, Kutkowska J, et al. Gilteritinib overcomes lorlatinib resistance in ALK-rearranged cancer. Nat Commun. 2021;12(1):1261. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated or analyzed in this study have been included in the published article. Further inquiries can be directed to the corresponding authors.