Abstract

Phosphinopyridyl ligands are used to synthesize a class of Ni(II) bis(chelate) complexes, which have been comprehensively characterized in both solid and solution phases. The structures display a square–planar configuration within the primary coordination sphere, with axially positioned labile binding sites. Their electrochemical data reveal two redox couples during the reduction process, suggesting the possibility of accessing two–electron reduction states. Significantly, these complexes serve as robust catalysts for homogeneous photocatalytic H2 evolution. In a system utilizing an organic photosensitizer and a sacrificial electron donor, an optimal turnover number of 27,100 is achieved in an alcohol–containing aqueous solution. A series of photophysical and electrochemical measurements were conducted to elucidate the reaction mechanism of photocatalytic hydrogen generation. Density function theory calculations propose a catalytic pathway involving two successive one–electron reduction steps, followed by two proton discharges. The sustained photocatalytic activity of these complexes stems from their distinct ligand system, which includes phosphine and pyridine donors that aid in stabilizing the low oxidation states of the Ni center.
Keywords: nickel complex, hydrogen evolution, photolytic catalysis, phosphinopyridyl ligand, density functional theory
Artificial photosynthesis (AP) offers a sustainable avenue to convert solar energy into fuels.1 For example, producing hydrogen directly from benign water through an AP system is highly desirable.2,3 An artificial photosynthetic system for hydrogen production comprises two essential components: a photosensitizer (PS) and a catalyst.4 Developing these components using earth-abundant elements is crucial for achieving sustainability in an AP system.5 As a result, significant efforts have been made to use first-row transition metal complexes, such as Fe, Co, and Ni, as hydrogen–evolving catalysts (HECs) and metal-free photosensitizers in AP systems.5−12 However, most of these systems suffer from catalytic instability and only reach a turnover number (TON) of hundreds to thousands. Among reported examples of HECs with commonly used organic and inorganic synthetic dyes, a cobalt complex with a softer polypyridyl ligand, [Co(Py3Me-Bpy)(OH2)](PF6)2, has a high TON (15,000) by using Ru(bpy)32+ as a PS.13 Chalcogenide quantum dots have significantly enhanced catalytic performance when replacing organic and inorganic dyes as PSs;14−16 however, they introduce disadvantages such as elemental scarcity, high cost, and toxicity.
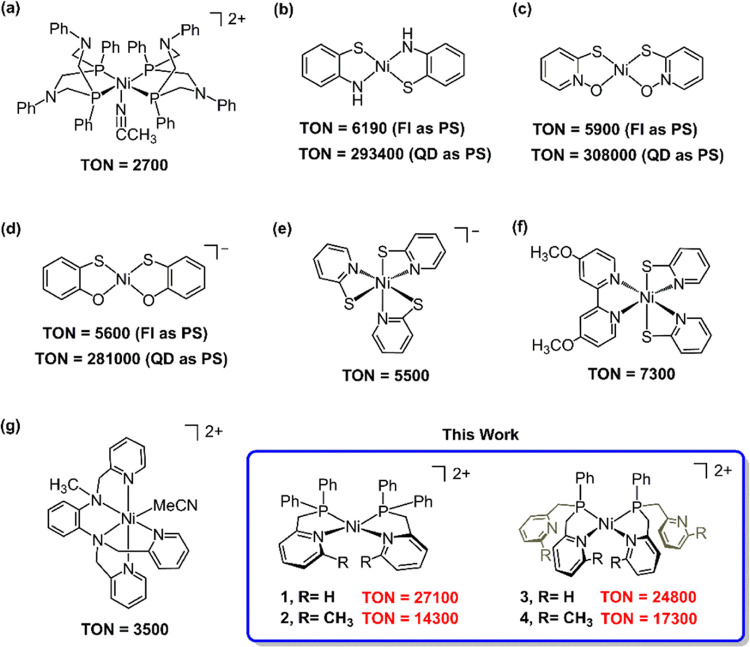
Nickel catalysts are of significant interest for hydrogen evolution due to their relevance to [NiFe]—hydrogenases,17 the abundance of nickel on earth, and their lower cost in comparison to noble metals. Additionally, Ni compounds are accessible at diverse oxidation states, thereby demonstrating versatile reactivity and efficient catalytic performance in a variety of organic syntheses.18,19 Eisenberg, Holland, and their co–workers first investigated Ni(II) bis(diphosphine) complexes that were initially reported as electrocatalysts for H2 generation by DuBois and co–workers, yielding a TON of 2700 (Scheme 1a).20−22 Later, the same group further reported other Ni(II) bis(chelate) catalysts with various bidentate ligands, including benzenedithiolate, 2–aminobenzenethiolate, 2–mercaptophenolate, and mercaptopyridyl–N–oxide (Scheme 1b–d).16 These systems exhibited a TON of 5600–6190 when an organic dye, fluorescein (Fl) was used as the PS and a TON of over 280,000 when CdSe quantum dots were substituted for the dye. Ni(II) tris(pyridinethiolate) and bis(pyridinethiolate)(bipyridine) complexes were also evaluated for their activity by the same group, with a TON of 7300 (Scheme 1e–f).23,24 Additionally, Verani and co–workers reported a Ni catalyst with a polypyridyl ligand, giving a TON of 3500 (Scheme 1g).25
Scheme 1. Ni Complexes for Photocatalytic H2 Evolution Reported in the Literature and This Work.
The search for a robust nickel catalyst that can produce hydrogen economically from inexpensive organic dyes is what inspired our study. Optimizing the properties of a supporting ligand is critical for maintaining the stability of metal catalysts throughout the catalytic cycle. For this aim, we specifically synthesized and characterized a class of Ni(II) bis(chelate) complexes that feature phosphinopyridyl ligand derivatives, PN1, PN1Me, PN2, and PN2Me (Scheme 1). The underlying hypothesis is that both phosphine and pyridine donors, with their unique π–back–donating characteristics, can enhance the accessibility of low–valent oxidation states at the Ni center. The addition of a 6–methyl substituent to pyridine donor ligands (PN1Me and PN2Me) allows for the introduction of steric effects and likely further tuning of the electronic properties in nickel complexes, as inspired by iron chemistry involving tris(pyridyl)amine (TPA) derivatives.26 While previous research has explored a similar ligand motif, 6–((diphenylphosphino)–methyl)pyridin–2–amine of Ni complexes, for electrocatalytic hydrogen production,27,28 their photocatalytic activity for hydrogen evolution reaction (HER) was not investigated yet. To the best of our knowledge, our system outperforms other first-row transition–metal complexes in terms of durability in hydrogen evolution while utilizing organic dyes.8
Results and Discussion
Syntheses and Characterization
The phosphinopyridyl ligand derivatives (PN1, PN1Me, PN2 and PN2Me) were synthesized in accordance with the procedures described in the literature.30−35 The reactions of Ni(ClO4)2 in CH3CN with the ligand derivatives resulted in nickel(II) bis(chelate) complexes. These complexes were designated as [Ni(PN1)2]2+ (1), [Ni(PN1Me)2]2+ (2), [Ni(PN2)2]2+ (3), and [Ni(PN2Me)2]2+ (4), respectively. When ether was layered into the reaction, brown crystalline solids of 1–4 with two ClO4– anions were produced in high yields. The X–ray crystallographic structures have a slightly distorted square–planar geometry, with the Ni(II) centers binding to two phosphino and two pyridyl groups in a cis configuration (Figures 1 and S1–S4).36 As shown in Tables S1–S8, all complexes have relatively comparable bond distances and angles. The incorporation of a 6–methyl substituent into pyridine donor ligands (PN1 vs PN1Me and PN2 vs PN2Me) does not induce structural differences within the first coordination sphere. Our density function theory (DFT) calculations indicate that even in the solvent the cis forms of complexes 1 and 2 are more stable than the corresponding trans forms by 0.13 eV in both cases, which is consistent with the observations of X-ray crystal structures. The pendant pyridyl groups of the ligands weakly interact with the Ni center in axial positions in complex 3, as indicated by a Ni–N2 distance of 3.32 Å (Figure S3). In solution, this interaction becomes more pronounced, resulting in the formation of a six–coordinate Ni complex, as supported by spectroscopic studies (vide infra). In contrast, the pendant pyridines in complex 4 remain uncoordinated, possibly as depicted in the space–filling diagrams (Figure S5), due to the steric hindrance caused by the 6–methyl substituent, which prevents axial interaction.
Figure 1.
ORTEP diagrams of (A) [1][(ClO4)2], (B) [2][(ClO4)2], (C) [3][(ClO4)2] and (D) [4][(ClO4)2]·2.5CH3CN with a 35% probability. The anions ClO4–, H atoms and solvated molecules are omitted for clarity. The nickel ion is located in a crystallographic inversion center in [3][(ClO4)2]. Two Ni complexes are contained in an asymmetry unit with similar structural parameters in [4][(ClO4)2]·2.5CH3CN. Only one is shown, while the other is omitted.
The electronic spectra of 1, 2, and 4 are similar, with weak absorption bands at 424 nm (ε = 4.6 × 102 M–1 cm–1), 428 nm (ε = 4.0 × 102 M–1 cm–1) and 443 nm (ε = 3.2 × 102 M–1 cm–1), respectively (Figure S6). The features with small molar extinction coefficients are assigned as d–d transitions, 1A1g → 1Eg.37 Complex 3, on the other hand, has a distinct spectrum that includes a band at 475 nm (ε = 3.2 × 102 M–1 cm–1) and a broad shoulder at 700–800 nm. These two absorptions are most likely attributed to d–d transitions in an octahedral geometry, 3A2g → 3T1g and 3A2g → 3T2g, indicating that two weakly binding pyridyl groups shown in the crystal structure also have interaction with Ni(II) ion in the solution state, resulting in a six–coordinate Ni(II) center.38
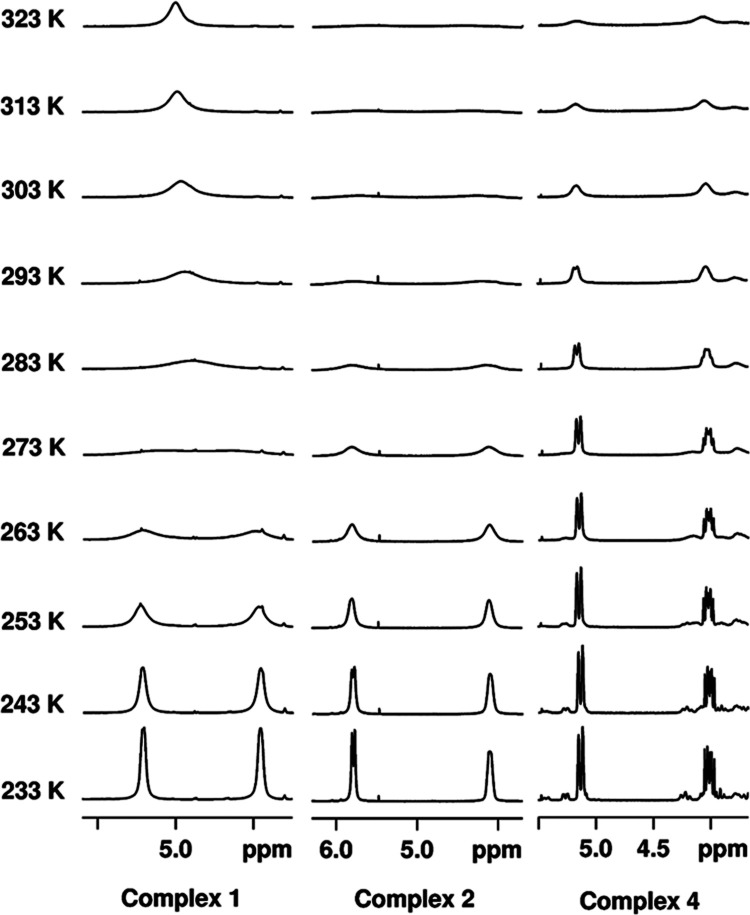
The variable–temperature (233 to 323 K) NMR spectroscopic investigations of 1–4 in CD3CN provided valuable insights into their dynamic and magnetic behavior in solution. At 233 K, the 1H NMR spectra of 1, 2 and 4 revealed well–resolved signals within the diamagnetic range (0–10 ppm), consistent with a low–spin square–planar d8 configuration (Figures 2 and S7–S9). Further confirmation of peak assignments was made through two-dimensional (2D) correlation experiments, as detailed in the Supporting Information and Figures S10–S18. In addition, the 31P NMR spectra of these three complexes exhibited peaks at 44.4, 54.6, and 50.4 ppm at 233 K, respectively (Figure S19). However, upon reaching room temperature, the signals gradually weakened, indicating the possible interconversion of different conformers induced by chelate ring dynamics. In contrast, the 1H NMR spectrum of 3 at 233 K displayed clear paramagnetic features in the range of 65 to −51 ppm, consistent with the identification of high–spin Ni(II) species in the electronic spectrum (Figure S20). Furthermore, the paramagnetic nature of 3 was evident in the significantly broader 31P NMR peak at 40.5 ppm at 233 K compared to those of 1, 2, and 4 (Figure S21). Utilizing the Evans method, the magnetic susceptibility of 3 in the solution state was determined to be 2.6 μB, implying a ground state with S = 1.
Figure 2.
Variable–temperature 1H NMR spectra of complexes 1, 2 and 4 at 3.5 to 6.3 ppm.
The variable–temperature NMR spectra further revealed the fluxional nature of 1, 2, and 4 in solution. At low temperatures, all displayed an AB pattern for the diastereotopic methylene protons of the ligands (Figure 2). However, as the temperature increased, the interconversion of different conformers, facilitated by chelate rings, became fast on the NMR time scale. The coalescence temperature for 1 was identified at 283 K, with a free energy of activation (ΔG‡) of 47.9 kJ/mol.39−41 Complexes 2 and 4 exhibited higher coalescence temperatures than 323 K, due to the methyl substituents on the pyridyl groups, which elevates ΔG‡ of the interconversion process. Similarly, the phenyl protons of the phosphine donors demonstrated comparable dynamic behavior.
Electrochemical Behavior
The electrochemical properties of 1–4 were investigated by cyclic voltammetry (CV) (Figure 3 and Table 1). During the reduction process, the voltammograms revealed the presence of two redox couples with E1/2 values ranging from −0.66 to −0.96 V and −1.48 to −1.63 V (vs Fc+/Fc). These results were further corroborated by differential pulse voltammetry measurements (Figure S22). These redox peaks are probably associated with the metal–centered redox couples, Ni(II/I) and Ni(I/0), as suggested by the absence of notable waves in this specific region in the CV data of the PN1 ligand (Figure S23). The former waves involving Ni(II/I) for 1 and 3 are quasi–reversible, whereas those for 2 and 4 are irreversible with larger ΔE values. The waves corresponding to the Ni(I/0) redox couple for all four complexes are irreversible, exhibiting significant peak-to-peak separations. The linear correlation between the square root of the scan rate and the reduction current (ip) suggests that the electrochemical process is governed by diffusion-controlled behavior (Figures S24–S27).
Figure 3.
Cyclic voltammograms of 1–4 (A–D) in CH3CN. Conditions: NBu4PF6, Pt, and Ag/AgNO3 as the supporting electrolyte, working electrode, and reference electrode, respectively. Scan rate: 100 mV/s.
Table 1. Cyclic Voltammetry Data for 1–4 in CH3CN (V vs Fc+/Fc)a.
| complexes | E1/2first | ΔEfirst | E1/2second | ΔEsecond |
|---|---|---|---|---|
| 1 | –0.844 | 0.078 | –1.573 | 0.149 |
| 2 | –0.666 | 0.128 | –1.486 | 0.216 |
| 3 | –0.995 | 0.086 | –1.628 | 0.155 |
| 4 | –0.750 | 0.222 | –1.629 | 0.157 |
Conditions: NBu4PF6, Pt, and Ag/AgNO3 as the supporting electrolyte, working electrode, and reference electrode, respectively. Scan rate: 100 mV/s.
The incorporation of a 6–methyl substituent into pyridine donor ligands is most likely responsible for the irreversibility of Ni(II/I) in 4. Density function theory (DFT) calculations suggest that this substitution destabilizes the Ni(I) state, potentially leading to ligand dissociation (vide infra). In contrast, the significant peak separation observed for Ni(I/0) couples in all complexes is most likely due to the complex reorganizing from a square planar to a tetrahedral geometry during reduction, as indicated by DFT calculations (vide infra). Moreover, introducing methyl substituents to the ligands leads to a more positive redox potential (1 vs 2 and 3 vs 4), contrary to the expected electronic donation effect. Iron and dimanganese complexes with TPA ligand derivatives show similar behaviors in redox potential upon the addition of a 6–methyl substituent.26,42 Pyridine–containing ligands with 6–methyl substituents have a preference for a lower oxidation–state metal center with a larger ionic radius, resulting in a more positive redox potential. Consequently, steric hindrance from the methyl substituent likely plays a crucial role in facilitating the attainment of lower Ni oxidation states. Moreover, the higher coordination number observed in 3, as supported by electronic spectroscopic data, leads to a more negative potential in comparison to 1. The redox potentials of square–planar Ni(II) complexes reported in the literature vary depending on the donor sets.16,28,43 Complexes 1–4 share similarities with [NiL2]2+ (where L = 6–((diphenylphosphino)–methyl)pyridin–2–amine) described by Masuda et al. in terms of their redox properties.28
Photocatalytic Hydrogen Evolution Reaction
Photocatalytic activity experiments of 1–4 for HER were conducted to determine optimal conditions by varying PS (Table S9), sacrificial electron donor (SED) (Table S10), solvent system (Table S11), pH value (Table S12) and catalyst concentration (Table S13). The quantification of hydrogen production was carried out using gas chromatography with thermal conductivity detector (GC–TCD). The presence of all three components (catalyst, PS, and SED) was necessary for the system to produce hydrogen. The catalytic reactions performed in an alcohol–mixed aqueous solution at pH = 10.45 with Fl as the PS and triethanolamine (TEOA) as the SED yielded the best TON (Figure 4 and Table S14). The production of H2 increased linearly for all complexes over the initial 4 days, with an average turnover frequency (TOF) ranging from 2000 to 2500 d–1. However, 2 and 4 exhibited a gradual decline in activity after 4 days, resulting in respective TONs of 14,300 and 17,300. In contrast, 1 and 3 showed a steady increase in H2 production until the eighth or ninth day, after which they began to decrease, resulting in TONs of 27,100 and 24,800, respectively. The apparent quantum yield is approximately 0.56%. Adding an additional equivalent of catalyst to the 14–day reaction catalyzed by 1 partially restored its activity, leading to a three–day average TOF of 1400 d–1 (Figure S28). This observation suggests that the catalyst undergoes decomposition during the catalytic cycle, possibly due to irradiation or the instability of Ni complexes at lower oxidation states (+1 or 0). The HER catalyzed by 1 was subjected to a mercury poisoning test. The results showed no significant difference between reactions with and without the addition of Hg (Figure S29), indicating that catalytic activity of 1 functions as a molecular catalyst rather than the formation of nanoparticles under photocatalytic conditions.44
Figure 4.

Photocatalytic hydrogen production from systems containing 1–4 (4.4 μM) as catalysts, Fl (18.6 mM) and TEOA (0.42 M). Reactions were carried out in MeOH/H2O (1:1) (for 1 and 2) and EtOH/H2O (1:1) (for 3 and 4) at pH 10.45 at room temperature upon irradiation (λ = 415–420 nm, 300 mW, LED).
Despite the fact that 2 and 4 have more positive reduction potentials than 1 and 3 by introducing 6–methyl substituents to the ligand, indicating easier access to the lower oxidation state of the Ni center, the initial catalytic performances of all complexes were comparable. However, 1 and 3 exhibited greater stability than 2 and 4, resulting in greater TONs for the former. The differences in robustness observed among the catalysts may stem from various factors, including the stability and solubility of catalytic intermediates. According to electrochemical data, the reduction of Ni(II) to Ni(I) is less reversible in 2 and 4 compared to 1 and 3; this suggests that the latter have more stable reduced states. Furthermore, the inclusion of 6–methyl substituents may lead to steric effects that result in instability in Ni(I) states, thereby diminishing the overall catalytic performance. DFT calculations also revealed the less stable Ni(I) state caused by the 6–methyl substituent (vide infra). Overall, 1 and 3 are the most robust molecular catalysts among non–noble metal complexes for photolytic HERs containing organic or inorganic dyes as PS.
Mechanistic Studies and DFT Analysis
A series of photophysical and electrochemical measurements were conducted to elucidate the reaction mechanism of photocatalytic hydrogen generation. Initially, the quenching mechanism between Fl and complex 1, as well as between Fl and TEOA, was investigated using Stern–Volmer quenching experiments. As shown in Figure S30, the fluorescence intensity of Fl diminished with increasing TEOA concentration in the solution. The bimolecular quenching constant kq(TEOA) for TEOA is 1.16 × 109 M–1 s–1. In contrast, the quenching constant in the presence of 1 was significantly higher at 4.18 × 1013 M–1 s–1, exceeding the diffusion–controlled limit (Figure S31). This implies that the interaction between Fl and complex 1 occurs in the ground state. Further titration experiments monitored by ultraviolet–visible (UV–vis) spectroscopy revealed that a complex formed between 1 and Fl as the concentration of 1 in the solution increased (Figure S32). No change in UV–vis spectra in the presence of TEOA was observed (Figure S33). In addition, the high concentration of 1 in the photocatalytic system drastically reduced the TOF value (Table S13). These observations indicate that Fl undergoes a dynamic quenching process with TEOA, whereas the interaction with 1 results in static non–fluorescence quenching. Moreover, in our photocatalytic system, the TEOA concentration is at least 4 orders of magnitude higher than that of complex 1. These results suggest that the reductive quenching pathway is dominant (Figure 5). First, the photosensitizer Fl undergoes reductive quenching, where its excited state is quenched by TEOA to form the reduced Fl– species. This Fl– species subsequently reduces the Ni complex, facilitating the hydrogen evolution reaction. The reduction potential of Fl/Fl– species was reported to be −1.53 V,45 which is more negative than the Ni(II/I) redox potentials of 1, but comparable to that of the Ni(I/0) couple in 1.
Figure 5.
(A) Proposed photocatalytic mechanism for hydrogen evolution and (B) energy levels of relevant species in the photocatalytic reaction.
To determine whether the HER mechanism mediated by complex 1 follows an ECEC or EECC pathway, electrochemical experiments were performed. Electrocatalytic voltammograms of 1 in the presence of acetic acid revealed the onset of electrocatalytic current around −1.5 V (Figure S34) compared to the redox couple of Ni(II)/Ni(I) at −0.84 V and Ni(I)/Ni(0) at −1.53 V. Combined with the observation of 91% Faraday efficiency in bulk electrolysis experiments at −2.1 V (vs Fc/Fc+), it indicates HER reaction catalyzed by 1 proceeds via an EECC mechanism. The low driving force between Fl/Fl– and the Ni(I/0) redox couple from 1 could account for the low turnover frequency observed in the photocatalytic system. Based on these mechanistic studies, the overall photocatalytic reaction mechanism is summarized in Figure 5.
To evaluate the stability of the Ni(II) catalysts in an alcohol-containing aqueous solution, the ultraviolet–visible–near infrared (UV–vis–NIR) spectrum of complex 1 was recorded in a mixed solvent of CH3CN/CH3OH/H2O in a 1:2:2 ratio (with CH3CN added to ensure solubility). The spectral pattern remained unchanged compared to that recorded in pure CH3CN (Figure S35). To elucidate the catalytic intermediates in Ni(I) and Ni(0) states, the chemical reduction of complex 1 was employed. The reaction with one equivalent of decamethylcobaltocene (CoCp*2) caused a clear shift in the UV–vis–NIR spectrum, which showed that the Ni(I) state was accessible (Figure S36). However, the spectral change in the presence of two equivalents was less pronounced. In situ UV–vis–NIR spectroelectrochemical measurements further facilitated the identification of the Ni(I) state. At a potential of −1.05 V (vs Fc/Fc+), the feature at 400–500 nm increased within 10 s, indicating a Ni center oxidation state change (Figure S37). The spectral change showed minimal deviation from the setup at −1.05 V when the potential was set at −1.77 V (vs Fc/Fc+), consistent with observations using chemical reduction. The X-band electron paramagnetic resonance (EPR) spectrum of complex 1, after applying a potential of −1.3 V (vs Fc/Fc+) for 0.5 h, exhibits a g ≈ 2 signal, consistent with the formation of a Ni(I) species. In contrast, the solution subjected to a potential of −1.9 V (vs Fc/Fc+) is EPR-silent, likely indicating the generation of a Ni(0) state (Figure S38). To further investigate a Ni(0) state within this ligand system, bis(1,5–cyclooctadiene)nickel(0) (Ni(COD)2) was reacted with two equivalents of the PN1 ligand in CD3CN. The resulting brown solution was EPR silent (Figure S39) and exhibited a relatively sharp 31P NMR signal, suggesting the formation of a diamagnetic species (Figure S40). Also, the resonance at 27.4 ppm, which is less shielded than the free ligand’s resonance at −9.6 ppm,31 shows that the ligand binds to the Ni center. The 31P NMR peak is more shielded than that of 1 (44.4 ppm), consistent with a lower oxidation state than Ni(II) in 1. Collectively, these results suggest that both Ni(I) and Ni(0) states are likely accessible within this ligand system.
DFT calculations (BP86/def2–TZVP//BP86/def2–SVP, see the SI) were conducted to examine the d-orbital occupancies of [Ni(II)]2+, [Ni(I)]+, and [Ni(0)]0. It is evident that during both reduction processes, electrons are sequentially added to the vacant dx2–y2 orbital, which is the lowest unoccupied molecular orbital (LUMO) of [Ni(II)]2+ (Figure S41). When [Ni(II)]2+ is reduced to [Ni(I)]+ through a single electron transfer, the Ni center retains its square planar structure. However, an additional electron transfers to [Ni(0)]0 leads to a double occupancy of the dx2–y2 orbital, inducing a shift from the square–planar to a tetrahedral configuration.
DFT calculations were also performed to provide further insights into the mechanism of HERs catalyzed by 1 and 2. Due to its paramagnetism, discussions of DFT calculations for 3 are included (Scheme S1). Based on our experiments, the initial step involves the one–electron reduction of the Ni complexes from Ni(II) to Ni(I). Our DFT calculations predicted a reduction potential of −0.89 V vs Fc+/Fc for 1 in acetonitrile, which aligns with our experimental measurement of −0.84 V vs Fc+/Fc.
To align with the experimental setup for H2 production in the aqueous phase, we computed Gibbs free energy surfaces under the condition of the applied potential (U) of −1.53 V vs Fc+/Fc, owing to the incorporation of Fl as the PS,45 and a pH of 10.45. Initially, [Ni(II)]2+ is reduced by one electron to form [Ni(I)]+, exhibiting a ΔG value of −0.56 eV (Scheme 2). Following this, [Ni(I)]+ has two potential pathways: further reduction to [Ni(0)]0 or protonation to form [Ni–NH]2+. Our DFT analysis indicates a thermodynamic preference for the reduction route (ΔG = −0.04 eV) over the protonation (ΔG = 1.10 eV). This reduction route is followed by protonation on Ni to form [Ni–H]+ with a ΔG of 0.12 eV.
Scheme 2. Free Energy Profile for the HER of 1 in an Aqueous Medium, Calculated at U = −1.53 V vs Fc+/Fc and pH 10.45.
Beginning with the [Ni–H]+ complex, H2 can be produced through two distinct pathways. The first involves binding the second proton to the nitrogen of the dissociated pyridine moiety, with the subsequent coupling of this proton and a hydrogen on Ni to form H2. However, our DFT calculations show for 1 this pathway is less likely, as adding a proton to the nitrogen of the dissociated pyridine moiety incurs a free energy cost of 0.61 eV, and the free energy barrier for the H–H coupling is 0.77 eV, leading to a total free energy barrier of 1.38 eV.
An alternative route for H2 formation involves the coupling of a Ni–bound hydrogen with a proton from the solvent. Utilizing H3O+ as the proton source in our calculations, we determined the free energy barrier for H2 formation via this mechanism is 0.96 eV. This value is notably 0.42 eV lower than the barrier of the first pathway, thereby indicating this route as the predominant pathway for H2 formation. However, it is important to note that this barrier, being 0.21 eV higher than the 0.75 eV kinetic threshold necessary for rapid electrochemical reactions,46 implies that H2 formation is a slow process. This result aligns with what we have observed experimentally.
Complex 2 follows the same reaction pathway and has a kinetic barrier (0.95 eV, Scheme S2) for H–H coupling similar to that of complex 1. This result is also consistent with our experimental observation that complexes 1 and 2 exhibit similar reactivity toward H2 formation in the early stage of the catalysis. We also computed the reaction mechanism for complex 3 and found that it follows the same pathway as complexes 1 and 2, even though it has a pendant pyridine to host a proton (Scheme S3). Our DFT calculations reveal that the reaction free energies (ΔG) for the one–electron reductions of Ni(I) to Ni(0) in complex 1 and complex 3 using Fl– are quite similar, with values of −0.04 and −0.07 eV, respectively (Schemes 2 and S2), differing by only 0.03 eV. This similarity aligns with the comparable HER performance observed for both complexes. Additionally, our experiments demonstrate that the reduction of Ni(I) to Ni(0) in 1 and 3 occurs at similar applied voltages. In summary, our DFT results support an EECC reaction pathway, encompassing two-electron reduction followed by two chemical processes. The mechanism is different from ECEC or CECE mechanisms; the former is proposed for Ni bis(diphosphine) systems (Scheme 1a)47 and Ni(II) bis(chelate) catalysts (Scheme 1b–d).16 The latter is suggested for Ni pyridylthiolate catalysts (Scheme 1e,f).24
Finally, we attempt to use DFT calculations to rationalize how the substituents on the phosphinopyridine ligand influence the TON of hydrogen production. The stability of photosensitizers and metal catalysts is crucial for the durability of a hydrogen evolution photocatalytic system. Photosensitizers (e.g., fluorescein) may degrade during reductive quenching, and photocatalysts can lose activity due to ligand dissociation, exchange, or hydrogenation.24,49,50 Since the same photosensitizer is used, the difference in the TON is likely to relate to the stability of the homogeneous catalysts, which could be deactivated due to the dissociation of the supporting ligands.
We thus compared the ligand dissociation energies (ΔGdiss) for 1, 2, and 3 in their [Ni(II)]2+, [Ni(I)]+, and [Ni(0)]0 states. It was observed that ΔGdiss is notably smaller at the [Ni(I)]+ state compared to the other two states, indicating a higher likelihood of phosphinopyridine ligand dissociation in this state (Figure S42). Importantly, at the [Ni(I)]+ state, 1 shows a larger ΔGdiss than 2 (1.02 vs 0.82 eV), suggesting greater stability of 1 against ligand dissociation. This aligns with the higher TON observed for 1 compared to 2. Additionally, when comparing ΔGdiss between 1 and 3 at the [Ni(I)]+ state, 1 also exhibits a slightly higher ΔGdiss than 3 (1.02 vs 1.01 eV). This indicates that 1 is marginally more stable against ligand dissociation than 3, which correlates with the slightly higher TON for 1 as compared to 3.
Conclusions
A series of Ni(II) complexes featuring phosphinopyridyl ligands has been successfully synthesized and thoroughly characterized in both solid and solution states. While complexes 1, 2, and 4 exhibit square–planar Ni(II) centers in both states, complex 3, displaying paramagnetic behavior, reveals a six–coordinate Ni(II) center by interacting with pendant pyridyl groups axially. Electrochemical analyses reveal two distinct redox couples during the reduction process, indicating accessibility to Ni(I) and Ni(0) states. Incorporating 6–methyl substituents into the pyridine donors of the ligands is hypothesized to facilitate access to lower metal oxidation states, as evidenced by a significant increase in the redox potential of complexes 2 and 4 compared to complexes 1 and 3, respectively. However, this modification may compromise the stability of Ni(I) states, as indicated by DFT calculations, potentially affecting catalytic durability. Nonetheless, these complexes demonstrate robust performance in catalytic systems, yielding high TONs surpassing those reported for first–row transition–metal molecular catalysts utilizing organic or inorganic dyes as PSs. Photophysical investigations suggest that the reductive quenching pathway is dominant. Electrochemical studies, combined with DFT calculations, propose an EECC mechanism involving two electrons added to the dx2–y2 orbital in the initial reaction stage, followed by two protonation processes. The exceptional catalytic stability of these complexes can be attributed to their unique ligand system, which comprises both phosphine and pyridine donors with π–back–donating properties. This characteristic likely stabilizes Ni centers in lower oxidation states (+1 and 0) and catalytic intermediates, thereby enhancing catalyst durability. In addition, a square–planar nickel center within the primary coordination sphere, with axially located labile binding sites, is essential for substrate accessibility. Furthermore, when the oxidation state changes from Ni(II) to Ni(0), bis(chelate) coordination allows for a geometric transformation from square-planar to tetrahedron. This study offers valuable insights for the development of robust catalysts for homogeneous hydrogen evolution.
Methods
Materials and General Methods
All experimental procedures were conducted under a nitrogen atmosphere using standard Schlenk techniques or within a glovebox under dinitrogen. Solvents were dried and distilled according to established protocols. All starting materials were obtained from commercial suppliers and used without further purification. The ligands PN1, PN1Me and PN2 were synthesized following previously reported methods.30−35
Spectroscopic, Electrochemical, and Structural Characterization of Complexes
Electronic absorption spectra were recorded using a Hewlett-Packard 8453 spectrophotometer. NMR spectra were acquired on Bruker AVNEO 500 spectrometers. The 1H NMR spectra were referenced to the residual proton signals of the solvents, and the 31P NMR spectra were referenced to the external standard O=P(OPh)3 at −17.7 ppm. The magnetic susceptibility of [3][(ClO4)2] was measured by Evans method using tBuOH as an internal standard.51 Elemental analyses were performed on an Elementar Vario EL III instrument. Electrochemical measurements were conducted using a CHI Model 421 electroanalyzer, with complexes dissolved in an CH3CN solution (0.1 mM) containing NBu4PF6 (0.1 M) as the supporting electrolyte. A platinum working electrode and Ag/AgNO3 reference electrode were employed, and potentials were referenced to the ferrocenium/ferrocene (Fc+/Fc) couple using ferrocene (Fc) as an external standard. Differential pulse voltammetry measurements were obtained using a Metrohm Autolab PGSTAT IMP system. X-ray crystallographic data were collected on a Bruker D8 Venture 3.0 diffractometer with Mo Kα radiation and a CCD detector. Least-squares refinement of positional and anisotropic thermal parameters was performed for all non-hydrogen atoms, while hydrogen atoms were placed in fixed positions. The refinements were based on F2 data, and an SADABS absorption correction was applied.52 The SHELXTL program was used for structure refinement, with anisotropic displacement factors refined for all non-hydrogen atoms, and hydrogen atoms treated using the riding model.53
Photocatalytic Hydrogen Evolution
Photocatalytic H2 evolution experiments were conducted in a sealed quartz cell under visible-light LED illumination (415–420 nm, 300 mW). The reaction mixture, comprising the nickel complex catalyst, photosensitizer, and sacrificial electron donor, was prepared in a hybrid solvent system (ethanol/water or methanol/water). Before irradiation, the mixture was purged with N2 for 5 min and degassed with an Ar/CH4 (9:1 v/v) mixture, using CH4 as an internal standard for quantitative analysis. After irradiation, the headspace gases were collected with a gastight syringe and analyzed for H2 content using a GC-TCD (Agilent Technologies 6890N). The amount of H2 generated was quantified daily, with the reaction degassed and reilluminated between measurements. Daily H2 production was added to the cumulative total. Turnover numbers (TONs) were calculated as the ratio of moles of H2 produced to moles of nickel catalyst used, over the course of the reaction.
In Situ Spectroelectrochemical Measurement
Electrochemical measurements were recorded using an Autolab PGSTAT302N potentiostat. Spectroelectrochemical experiments were performed in a thin-layer quartz cuvette (optical path length: 1 mm) equipped with a platinum gauze as the working electrode, a platinum wire as the counter electrode, and an Ag/Ag+ reference electrode. UV–vis spectra were acquired using an SR2 UV–vis spectrometer (Ocean Insight).
Calculation of Quantum Yield for Photocatalytic Hydrogen Evolution
Quantum yield calculations were performed using systems containing 4.4 μM [1][(ClO4)2] (22 nmol), 18.6 mM Fl (93 μmol), and 0.42 M TEOA (2.1 mmol). The light power (P) was measured with a PM100D power meter (Thorlabs) equipped with an S314C detector (Thorlabs). The quantum yield (Φ) was calculated after 1 day of irradiation according to reported methods.54
Fluorescence Quenching Measurement
Fluorescence detection
was performed under monochromatic light at 420 nm, generated using
a 0.3 W blue LED light source and measured with a spectrophotometer
(Hitachi F7000 or Hong-Ming Technology Co., Ltd. QME, PS060). Stern–Volmer
quenching studies were conducted by measuring the emission intensity
of Fl using a fluorescence spectrophotometer. Samples were prepared
in sealed cuvettes and degassed with N2 prior to measurements.
The quenching ratio  and
the Stern–Volmer constant (KSV)
were calculated using the equation
and
the Stern–Volmer constant (KSV)
were calculated using the equation  , where I0f represents
the emission intensity
of the photosensitizer in the absence of the quencher, and [Q] denotes
the concentration of the quencher. The quenching rate constant (kq) was determined using the formula
, where I0f represents
the emission intensity
of the photosensitizer in the absence of the quencher, and [Q] denotes
the concentration of the quencher. The quenching rate constant (kq) was determined using the formula  , where
τ0 is the lifetime
of the photosensitizer.
, where
τ0 is the lifetime
of the photosensitizer.
Faradaic Efficiency Measurement
Electrochemical measurements were recorded using an Autolab PGSTAT IMP potentiostat. Bulk electrolysis was conducted in an H-cell divided into two compartments by a glass frit, each containing 10 mL of acetonitrile with 0.1 M NBu4PF6. [1][(ClO4)2] (1 mM) and acetic acid (100 mM) were added to both compartments. Two glassy carbon plates (1.5 × 1.0 cm2 submerged area) served as the working and counter electrodes. The reference electrode was a silver wire immersed in acetonitrile containing 0.01 M silver nitrate and 0.1 M NBu4PF6. Electrolysis was performed at −2.1 V (vs Fc+/Fc) for 5 min, with continuous stirring. After electrolysis, a 300 μL sample of the headspace gas was collected using a gastight syringe. The H2 concentration was analyzed by a Shimadzu Nexis GC-2010 gas chromatograph, equipped with a Micropacked ST column (2.0 m × 1.0 mm ID) and using helium (99.9999% purity) as the carrier gas at a flow rate of 300 kPa. A barrier discharge ionization detector was used for product analysis
EPR Measurement Following Bulk Electrolysis
Bulk electrolysis was conducted in an H-cell divided into two compartments by a glass frit. Each compartment contained 10 mL of acetonitrile with 0.1 M NBu4PF6, and [1][(ClO4)2] (1.5 mM) was added to both compartments. Two glassy carbon plates (1.5 × 1.0 cm2 submerged area) were used as the working and counter electrodes. The reference electrode was a silver wire immersed in acetonitrile containing 0.01 M silver nitrate and 0.1 M NBu4PF6. Electrolysis was performed at −1.3 and −1.9 V (vs Fc+/Fc) for 30 min, with continuous stirring. Postelectrolysis, the solutions were collected for EPR measurements using the BEMXnano BENCH-TOP system
Synthesis
Bis[(6–methyl–2–pyridyl)methyl]phenylphosphine (PN2Me)
2,6-Lutidine (8.7 mL, 75 mmol) was dissolved in 100 mL of THF and cooled to −78 °C. nBuLi (75 mmol, 1.6 M in hexane) was added dropwise over the course of 1 h with continuous stirring. Trimethylsilyl chloride (8.2 g, 75 mmol) was then added dropwise to the solution at −78 °C. The reaction mixture was allowed to warm to room temperature and stirred overnight. The solvent was removed under vacuum, and the residue was extracted with pentane. The solid was removed by filtration. After evaporating the pentane under vacuum, the oily residue was distilled to afford 6-methyl-2-[(trimethylsilyl)methyl]pyridine. This product (12.2 g, 68 mmol) was dissolved in 100 mL of THF, and PPhCl2 (5.2 mL, 34 mmol) was added at −78 °C. The mixture was stirred overnight, and the THF was removed under vacuum to yield the final product. Yield: 95%. 1H NMR (500 MHz, CD3CN): δ ABX spin system (A = B = H, X = P) 2.40 (6H, s, methyl H), 3.26 (2H, d, 1JAB = 13.31 Hz), 3.27 (2H, dd, 1JAB = 13.31 Hz, 2JXB = 1.48 Hz), 6.84 (2H, d, 2J = 7.70 Hz, pyridyl H), 6.95 (2H, d, 2J = 7.65 Hz, pyridyl H), 7.31–7.34 (3H, m, aromatic H), 7.40 (2H, t, 2J = 7.70 Hz, pyridyl H), 7.47–7.50 (2H, m, aromatic H); 13C{1H} NMR (125.8 MHz, CD3CN): δ 23.46 (s, CH3 of pyridyl), 37.23 (d, 1JPC = 18.20 Hz, P–CH2), 120.16 (s, CH of pyridyl), 120.42 (d, 3JPC = 4.91 Hz, CH of pyridyl), 128.15 (d, 3JPC = 6.83 Hz, m–CH of phenyl), 129.05 (s, p–CH of phenyl), 132.78 (d, 2JPC = 20.04 Hz, o–CH of phenyl), 136.37 (s, CH of pyridyl), 137.54 (d, 1JPC = 19.35 Hz, P–C of phenyl), 157.66 (s, CH of pyridyl), 157.74 (d, 2JPC = 5.87 Hz, N–C–CH2 of pyridyl); 31P{1H} NMR (202.5 MHz, CD3CN): δ – 13.9.
[Ni(PN1)2][(ClO4)2] ([1][(ClO4)2])
PN1 (0.100 g, 0.36 mmol) and Ni(ClO4)2·6H2O (0.066 g, 0.18 mmol) were dissolved in CH3CN, forming a brown solution. The reaction mixture was allowed to stand at room temperature and was subsequently layered with ether. After 2 to 4 days, brown crystalline solids of [1][(ClO4)2] were obtained. Yield: 66%. Anal. Calcd for C36H32Cl2N2NiO8P2: C, 53.24; N, 3.45; H,3.97. Found: C, 53.73; N, 3.44; H, 4.14. 1H NMR (500 MHz, CD3CN, 233 K): δ 3.93 (2H, s, CH2), 5.44 (2H, s, CH2), 7.12 (4H, s, o–H of phenyl), 7.30 (4H, s, m–H of phenyl), 7.38 (4H, s, m–H of phenyl), 7.42 (2H, s, H of pyridyl), 7.56 (4H, s, p–H of phenyl), 7.64 (6H, s, o–H of phenyl and H of pyridyl), 8.03 (2H, t, 1J = 7.3 Hz, H of pyridyl), 8.17 (2H, s, H of pyridyl); 13C{1H} NMR (125.8 MHz, CD3CN, 233 K): δ 41.65 (s, N–C–CH2), 122.32 (s, P–C of phenyl), 124.13 (s, P–C of phenyl), 124.54 (s, CH of pyridyl), 125.09 (s, CH of pyridyl), 129.30 (s, m–CH of phenyl), 132.96 (s, o–CH of phenyl), 133.49 (s, p–CH of phenyl), 134.31 (s, o–CH of phenyl), 141.83 (s, CH of pyridyl), 152.93 (s, CH of pyridyl), 158.29 (s, N–C–CH2); 31P{1H} NMR (202.5 MHz, CD3CN, 233 K): δ 44.4. UV–vis–NIR in CH3CN (λ, nm; ε, M–1 cm–1): 424 (4.6 × 102).
[Ni(PN1Me)2][(ClO4)2] ([2][(ClO4)2])
PN1Me (0.100 g, 0.34 mmol) and Ni(ClO4)2·6H2O (0.063 g, 0.17 mmol) were dissolved in CH3CN, forming a brown solution. The reaction mixture was layered with acetone and ether at room temperature. After 5 to 7 days, brown crystalline solids of [2][(ClO4)2] were obtained. Yield: 46%. Anal. Calcd for C38H36Cl2N2NiO8P2: C, 54.32; N, 3.33; H,4.32. Found: C, 54.73; N, 3.61; H, 4.17. 1H NMR (500 MHz, CD3CN, 233 K): δ 2.31 (6H, s, CH3), 4.10 (2H, s, CH2), 5.79 (2H, d, 1J = 16.90 Hz, CH2), 7.10 (4H, s, o–H of phenyl), 7.22 (4H, t, m–H of phenyl), 7.26 (2H, d, 1J = 7.83 Hz, H of pyridyl), 7.47 (2H, d, 1J = 7.76 Hz, H of pyridyl), 7.51 (6H, m, m–H and p–H of phenyl), 7.63 (2H, t, 1J = 6.73 Hz, p–H of phenyl), 7.86 (2H, t, 1J = 7.80 Hz, H of pyridyl), 7.94 (4H, s, o–H of phenyl); 13C{1H} NMR (125.8 MHz, CD3CN, 233 K): δ 24.61 (s, CH3), 39.92 (s, N–C–CH2), 120.81 (s, P–C of phenyl), 123.22 (s, CH of pyridyl), 123.68 (s, P–C of phenyl), 126.45 (s, CH of pyridyl), 129.45 (s, m– or p–CH of phenyl), 129.70 (s, m–CH of phenyl), 132.37 (s, o–CH of phenyl), 133.65 (s, m– or p–CH of phenyl), 134.21 (s, p–CH of phenyl), 135.14 (s, o–CH of phenyl), 142.12 (s, CH of pyridyl), 156.06 (s, N–C–CH2), 161.47 (s, N–C–CH3); 31P{1H} NMR (202.5 MHz, CD3CN, 233 K): δ 54.6. UV–vis–NIR in CH3CN (λ, nm; ε, M–1 cm–1): 428 (4.0 × 102).
[Ni(PN2)2][(ClO4)2] ([3][(ClO4)2])
PN2 (0.100 g, 0.33 mmol) and Ni(ClO4)2·6H2O (0.063 g, 0.17 mmol) were dissolved in CH3CN, forming a brown solution. The reaction mixture was then layered with ether at room temperature. After 1 week, brown crystalline solids of [3][(ClO4)2] were obtained. Yield: 90%. Anal. Calcd for C36H34Cl2N4NiO8P2: C, 51.30; N, 6.60; H, 4.00. Found: C, 51.27; N, 6.67; H, 4.12. 31P{1H} NMR (202.5 MHz, CD3CN): δ 40.5. UV–vis–NIR in CH3CN (λ, nm; ε, M–1 cm–1): 475(3.2 × 102).
[Ni(PN2Me)2][(ClO4)2]·2.5CH3CN ([4][(ClO4)2]·2.5CH3CN)
PN2Me (0.100 g, 0.27 mmol) and Ni(ClO4)2·6H2O (0.063 g, 0.17 mmol) were dissolved in CH3CN, forming a brown solution. The reaction mixture was layered with ether at room temperature. After 1 week, brown crystalline solids of [4][(ClO4)2]·2.5CH3CN were obtained. Yield: 33%. Anal. Calcd for C40H42Cl2N4NiO8P2: C, 53.47; N, 6.23; H, 4.68. Found: C, 52.68; N, 6.22; H, 4.83. 1H NMR (500 MHz, CD3CN, 233 K): δ 1.64 (6H, s, CH3), 2.57 (6H, s, CH3), 2.87 (2H, m, CH2), 3.32 (2H, m, CH2), 4.01 (2H, m, CH2), 5.13 (2H, d, 1J = 17.62 Hz, CH2), 6.70 (2H, d, 2J = 7.52 Hz, H of pyridyl), 6.83 (2H, d, 2J = 7.75 Hz, H of pyridyl), 6.85 (2H, d, 2J = 7.77 Hz, H of pyridyl), 7.19 (2H, dd, 2J = 7.52 Hz, 2J = 7.77 Hz, H of pyridyl), 7.40 (2H, d, 2J = 7.73 Hz, H of pyridyl), 7.66 (4H, t, 2J = 7.68 Hz, m–H of phenyl), 7.70 (2H, t, 2J = 7.81 Hz, H of pyridyl), 7.82 (2H, t, 2J = 7.48 Hz, p–H of phenyl), 7.97 (4H, m, o–H of phenyl); 13C{1H} NMR (125.8 MHz, CD3CN, 233 K): δ 24.31 (s, CH3), 24.91 (s, CH3), 30.54 (t, CH2), 39.63 (t, CH2), 121.44 (s, CH of pyridyl), 122.41 (s, CH of pyridyl), 122.70 (s, CH of pyridyl), 123.63 (s, P–C of phenyl), 125.88 (s, CH of pyridyl), 129.80 (s, m–CH phenyl), 132.82 (s, o–CH of phenyl), 134.48 (s, p–CH of phenyl), 137.93 (s, CH of pyridyl), 141.39 (s, CH of pyridyl), 150.63 (s, N–C–CH2), 156.60 (s, N–C–CH2), 159.24 (s, N–C–CH3); 160.67 (s, N–C–CH3), 31P{1H} NMR (202.5 MHz, CD3CN, 233 K): δ 50.4. UV–vis–NIR in CH3CN (λ, nm; ε, M–1 cm–1): 443(3.2 × 102).
Computational Details
DFT calculations utilized the BP8655 functional unless otherwise specified. For all calculations, CPCM56,57 implicit solvation model was employed. For the calculation of redox potential from Ni(II) to Ni(I) and the HER free energy profile, acetonitrile and water were selected as solvent, respectively. Geometry optimization and vibrational frequency calculations were performed using the double−ξ quality basis set, def2–SVP.58 To rectify inaccuracies in vibrational entropies that arise from low–frequency modes, we have modified frequencies lower than 50 cm–1 to be exactly 50 cm–1 when performing thermal corrections.59,60 To attain a higher level of accuracy in electronic energy, a single point calculation was executed using the triple−ξ quality basis set, def2–TZVP.58 In our study, we address the common issue of overestimating molecular entropy when treating molecules as ideal gases, especially in solvent environments. To mitigate this, we have implemented the correction approach initially introduced by Wertz,61 which was subsequently applied in the work of Cooper and Ziegler.62
The Gibbs free energies were determined by the following equation
where Eelec, ZPE, Hvib and denote electronic energy, zero–point energy, and enthalpy contribution from vibration, respectively. The 4RT term encompasses translational thermal correction, rotational thermal correction, and PV work. The temperature, T, was set to 298.15 K. The Ss term represents the entropy in the solvent phase.
To determine the Gibbs free energy of a proton in water, we combine the gas–phase Gibbs free energy of a proton with the experimentally measured solvation free energy of a proton in water, which is −264.0 kcal/mol.63 In the definition of standard hydrogen electrode (SHE)
at pH = 0 and U = 0.0 VSHE.Thus, by calculating G(H2(g)), the G(e–) is determined to be −4.324 eV. Lastly, the impacts of pH and applied potential are accounted for using the following equation
at arbitrary pH and U.
Acknowledgments
The authors thank Prof. Kuei-Feng Hsu and Prof. Hsin-Lun Wu at the Department of Chemistry of National Cheng Kung University for the discussion. The authors acknowledge the use of EPR, X-ray crystallography, and elemental analysis equipment belonging to the Core Facility Centers of National Sun Yat-sen University, National Chung Hsing University, National Taiwan University, and National Cheng Kung University.
Glossary
Abbreviations
- Py3Me-Bpy
6-[6-(1,1-dipyridin-2-yl-ethyl)-pyridin-2-ylmethyl]-[2,2′]bipyridinyl
- PN1
(2-pyridyl)methyldiphenylphosphine
- PN1Me
[(6-methyl-2-pyridyl)methyl]diphenylphosphine
- PN2
bis[(2-pyridyl)methyl]phenylphosphine
- PN2Me
bis[(6-methyl-2- pyridyl)methyl]phenylphosphine
- TPA
tris(2-pyridylmethyl)amine
Supporting Information Available
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/jacsau.4c00714.
Characterization; additional experimental data and computational details (PDF)
Author Contributions
∥ C.-T.W. and H.-R.P. contributed equally to this work.
This work was supported by National Science and Technology Council in Taiwan. (NSTC 112–2113-M-006–007 for H.-F.H.; NSTC 111–2113-M-006–010-MY3 for M.-J.C.; NSTC 113–2636-M-110–004 for V.W.)
The authors declare no competing financial interest.
Supplementary Material
References
- Nocera D. G. Solar Fuels and Solar Chemicals Industry. Acc. Chem. Res. 2017, 50 (3), 616–619. 10.1021/acs.accounts.6b00615. [DOI] [PubMed] [Google Scholar]
- Lewis N. S.; Nocera D. G. Powering the planet: Chemical challenges in solar energy utilization. Proc. Natl. Acad. Sci. U.S.A. 2006, 103 (43), 15729–15735. 10.1073/pnas.0603395103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberg R. Rethinking water splitting. Science 2009, 324 (5923), 44–45. 10.1126/science.1172247. [DOI] [PubMed] [Google Scholar]
- Han Z.; Eisenberg R. Fuel from Water: The Photochemical Generation of Hydrogen from Water. Acc. Chem. Res. 2014, 47, 2537–2544. 10.1021/ar5001605. [DOI] [PubMed] [Google Scholar]
- Bullock R. M.; Chen J. G.; Gagliardi L.; Chirik P. J.; Farha O. K.; Hendon C. H.; Jones C. W.; Keith J. A.; Klosin J.; Minteer S. D.; Morris R. H.; Radosevich A. T.; Rauchfuss T. B.; Strotman N. A.; Vojvodic A.; Ward T. R.; Yang J. Y.; Surendranath Y. Using nature’s blueprint to expand catalysis with Earth-abundant metals. Science 2020, 369 (6505), eabc3183 10.1126/science.abc3183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalle K. E.; Warnan J.; Leung J. J.; Reuillard B.; Karmel I. S.; Reisner E. Electro- and Solar-Driven Fuel Synthesis with First Row Transition Metal Complexes. Chem. Rev. 2019, 119 (4), 2752–2875. 10.1021/acs.chemrev.8b00392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Droghetti F.; Lucarini F.; Molinari A.; Ruggi A.; Natali M. Recent findings and future directions in photosynthetic hydrogen evolution using polypyridine cobalt complexes. Dalton Trans. 2022, 51 (28), 10658–10673. 10.1039/D2DT00476C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazzeo A.; Santalla S.; Gaviglio C.; Doctorovich F.; Pellegrino J. Recent progress in homogeneous light-driven hydrogen evolution using first-row transition metal catalysts. Inorg. Chim. Acta 2021, 517, 119950 10.1016/j.ica.2020.119950. [DOI] [Google Scholar]
- Eckenhoff W. T. Molecular catalysts of Co, Ni, Fe, and Mo for hydrogen generation in artificial photosynthetic systems. Coord. Chem. Rev. 2018, 373, 295–316. 10.1016/j.ccr.2017.11.002. [DOI] [Google Scholar]
- Gupta R.; Sahni P.; Sharma S.; Roy A.; Pal A. K. Structurally modified T-metal complexes and organic dyes as photosensitizers and earth-abundant T-metal catalysts for photo-driven hydrogen evolution in artificial photosynthetic systems. Sustainable Energy Fuels 2022, 6 (8), 1891–1922. 10.1039/D2SE00177B. [DOI] [Google Scholar]
- Zhang B.; Sun L. Artificial photosynthesis: opportunities and challenges of molecular catalysts. Chem. Soc. Rev. 2019, 48 (7), 2216–2264. 10.1039/C8CS00897C. [DOI] [PubMed] [Google Scholar]
- Hartley C. L.; DiRisio R. J.; Screen M. E.; Mayer K. J.; McNamara W. R. Iron Polypyridyl Complexes for Photocatalytic Hydrogen Generation. Inorg. Chem. 2016, 55 (17), 8865–8870. 10.1021/acs.inorgchem.6b01413. [DOI] [PubMed] [Google Scholar]
- Wang P.; Liang G.; Smith N.; Hill K.; Donnadieu B.; Webster C. E.; Zhao X. Enhanced Hydrogen Evolution in Neutral Water Catalyzed by a Cobalt Complex with a Softer Polypyridyl Ligand. Angew. Chem., Int. Ed. 2020, 59 (31), 12694–12697. 10.1002/anie.202002640. [DOI] [PubMed] [Google Scholar]
- Han Z.; Qiu F.; Eisenberg R.; Holland P. L.; Krauss T. D. Robust Photogeneration of H2 in Water Using Semiconductor Nanocrystals and a Nickel Catalyst. Science 2012, 338 (6112), 1321–1324. 10.1126/science.1227775. [DOI] [PubMed] [Google Scholar]
- Han K.; Wang M.; Zhang S.; Wu S.; Yang Y.; Sun L. Photochemical hydrogen production from water catalyzed by CdTe quantum dots/molecular cobalt catalyst hybrid systems. Chem. Commun. 2015, 51, 7008–7011. 10.1039/C5CC00536A. [DOI] [PubMed] [Google Scholar]
- Das A.; Han Z.; Brennessel W. W.; Holland P. L.; Eisenberg R. Nickel complexes for robust light-driven and electrocatalytic hydrogen production from water. ACS Catal. 2015, 5 (3), 1397–1406. 10.1021/acscatal.5b00045. [DOI] [Google Scholar]
- Lubitz W.; Ogata H.; Rüdiger O.; Reijerse E. Hydrogenases. Chem. Rev. 2014, 114, 4081–4148. 10.1021/cr4005814. [DOI] [PubMed] [Google Scholar]
- Ananikov V. P. Nickel: The “Spirited Horse” of Transition Metal Catalysis. ACS Catal. 2015, 5 (3), 1964–1971. 10.1021/acscatal.5b00072. [DOI] [Google Scholar]
- Chernyshev V. M.; Ananikov V. P. Nickel and Palladium Catalysis: Stronger Demand than Ever. ACS Catal. 2022, 12 (2), 1180–1200. 10.1021/acscatal.1c04705. [DOI] [Google Scholar]
- Helm M. L.; Stewart M. P.; Bullock R. M.; DuBois M. R.; DuBois D. L. A Synthetic Nickel Electrocatalyst with a Turnover Frequency Above 100,000 s–1 for H2 Production. Science 2011, 333 (6044), 863–866. 10.1126/science.1205864. [DOI] [PubMed] [Google Scholar]
- Kilgore U. J.; Roberts J. A. S.; Pool D. H.; Appel A. M.; Stewart M. P.; DuBois M. R.; Dougherty W. G.; Kassel W. S.; Bullock R. M.; Daniel D. L. [Ni(P2PhN2C6H4X)2]2+ Complexes as Electrocatalysts for H2 Production: Effect of Substituents, Acids, and Water on Catalytic Rates. J. Am. Chem. Soc. 2011, 133, 5861–5872. 10.1021/ja109755f. [DOI] [PubMed] [Google Scholar]
- McLaughlin M. P.; McCormick T. M.; Eisenberg R.; Holland P. L. A stable molecular nickel catalyst for the homogeneous photogeneration of hydrogen in aqueous solution. Chem. Commun. 2011, 47 (28), 7989–7991. 10.1039/c1cc12347e. [DOI] [PubMed] [Google Scholar]
- Han Z.; McNamara W. R.; Eum M.-S.; Holland P. L.; Eisenberg R. A Nickel Thiolate Catalyst for the Long-Lived Photocatalytic Production of Hydrogen in a Noble-Metal-Free System. Angew. Chem., Int. Ed. 2012, 51 (7), 1667–1670. 10.1002/anie.201107329. [DOI] [PubMed] [Google Scholar]
- Han Z.; Shen L.; Brennessel W. W.; Holland P. L.; Eisenberg R. Nickel Pyridinethiolate Complexes as Catalysts for the Light-Driven Production of Hydrogen from Aqueous Solutions in Noble-Metal-Free Systems. J. Am. Chem. Soc. 2013, 135 (39), 14659–14669. 10.1021/ja405257s. [DOI] [PubMed] [Google Scholar]
- Kankanamalage P. H. A.; Mazumder S.; Tiwari V.; Kpogo K. K.; Schlegel H. B.; Verani C. N. Efficient electro/photocatalytic water reduction using a [NiII(N2Py3)]2+ complex. Chem. Commun. 2016, 52 (91), 13357–13360. 10.1039/C6CC06629A. [DOI] [PubMed] [Google Scholar]
- Zang Y.; Kim J.; Dong Y.; Wilkinson E. C.; Appelman E. H.; Que L. Models for Nonheme Iron Intermediates: Structural Basis for Tuning the Spin States of Fe(TPA) Complexes. J. Am. Chem. Soc. 1997, 119 (18), 4197–4205. 10.1021/ja9638521. [DOI] [Google Scholar]
- Tatematsu R.; Inomata T.; Ozawa T.; Masuda H. Electrocatalytic Hydrogen Production by a Nickel(II) Complex with a Phosphinopyridyl Ligand. Angew. Chem., Int. Ed. 2016, 55 (17), 5247–5250. 10.1002/anie.201511621. [DOI] [PubMed] [Google Scholar]
- Kato T.; Tatematsu R.; Nakao K.; Inomata T.; Ozawa T.; Masuda H. Effect of Counteranions in Electrocatalytic Hydrogen Generation Promoted by Bis(phosphinopyridyl) Ni(II) Complexes. Inorg. Chem. 2021, 60 (11), 7670–7679. 10.1021/acs.inorgchem.0c03657. [DOI] [PubMed] [Google Scholar]
- Kermagoret A.; Tomicki F.; Braunstein P. Nickel and iron complexes with N,P,N-type ligands: synthesis, structure and catalytic oligomerization of ethylene. Dalton Trans. 2008, (22), 2945–2955. 10.1039/b802009d. [DOI] [PubMed] [Google Scholar]
- Flapper J.; Kooijman H.; Lutz M.; Spek A. L.; van Leeuwen P. W. N. M.; Elsevier C. J.; Kamer P. C. J. Nickel and Palladium Complexes of Pyridine–Phosphine Ligands as Ethene Oligomerization Catalysts. Organometallics 2009, 28 (4), 1180–1192. 10.1021/om800903n. [DOI] [Google Scholar]
- Jaafar H.; Li H.; Misal Castro L. C.; Zheng J.; Roisnel T.; Dorcet V.; Sortais J.-B.; Darcel C. Phosphane-Pyridine Iron Complexes: Synthesis, Characterization and Application in Reductive Amination through the Hydrosilylation Reaction. Eur. J. Inorg. Chem. 2012, 2012 (22), 3546–3550. 10.1002/ejic.201200550. [DOI] [Google Scholar]
- Mothes E.; Sentets S.; Luquin M. A.; Mathieu R.; Lugan N.; Lavigne G. New Insight into the Reactivity of Pyridine-Functionalized Phosphine Complexes of Ruthenium(II) with Respect to Olefin Metathesis and Transfer Hydrogenation. Organometallics 2008, 27 (6), 1193–1206. 10.1021/om7012106. [DOI] [Google Scholar]
- Kinoshita E.; Arashiba K.; Kuriyama S.; Miyake Y.; Shimazaki R.; Nakanishi H.; Nishibayashi Y. Synthesis and Catalytic Activity of Molybdenum–Dinitrogen Complexes Bearing Unsymmetric PNP-Type Pincer Ligands. Organometallics 2012, 31 (23), 8437–8443. 10.1021/om301046t. [DOI] [Google Scholar]
- Hettstedt C.; Unglert M.; Mayer R. J.; Frank A.; Karaghiosoff K. Methoxyphenyl Substituted Bis(picolyl)phosphines and Phosphine Oxides. Eur. J. Inorg. Chem. 2016, 2016 (9), 1405–1414. 10.1002/ejic.201600032. [DOI] [Google Scholar]
- τ4= 0.15, 0.17, 0.18 and 0.15 (0.17) for complexes 1–4, respectively.; Yang L.; Powell D. R.; Houser R. P. Structural variation in copper(I) complexes with pyridylmethylamide ligands: structural analysis with a new four-coordinate geometry index, τ4. Dalton Trans. 2007, (9), 955–964. 10.1039/B617136B. [DOI] [PubMed] [Google Scholar]
- Gray H. B.; Preer J. R. Electronic structures of low-spin, square-pyramidal complexes of nickel(II). J. Am. Chem. Soc. 1970, 92 (25), 7306–7312. 10.1021/ja00728a013. [DOI] [Google Scholar]
- Reimann C. W. The Single Crystal Spectra of Dichlorotetrapyrazolenickel(II), Dibromotetrapyrazolenickel(II), and Hexapyrazolenickel(II) Nitrate. J. Phys. Chem. A 1970, 74 (3), 561–568. 10.1021/j100698a014. [DOI] [Google Scholar]
- ΔG‡ = 2.303RTc(10.319 + logTc – log kc); kc = πΔν/√2 s–1; R = 8.314 JK–1mol–1.
- Fischer P.; Fettig A. Chelate-type intramolecular hydrogen bridging in 2,4-diaryl-6-(2-hydroxy-4-methoxyphenyl)-1,3,5-triazines. A dynamic 1H/13C NMR study. Magn. Reson. Chem. 1997, 35 (12), 839–844. 10.1002/(SICI)1097-458X(199712)35:12<839::AID-OMR182>3.0.CO;2-G. [DOI] [Google Scholar]
- Bonnot C.; Chambron J.-C.; Espinosa E.; Graff R. The in, out Asymmetric Pseudo-Triple Helical Form of a D3h Diaza-Macropentacycle. J. Org. Chem. 2008, 73 (3), 868–881. 10.1021/jo701984z. [DOI] [PubMed] [Google Scholar]
- Goodson P. A.; Oki A. R.; Glerup J.; Hodgson D. J. Design, synthesis, and characterization of bis(μ-oxo)dimanganese(III,III) complexes. Steric and electronic control of redox potentials. J. Am. Chem. Soc. 1990, 112 (17), 6248–6254. 10.1021/ja00173a011. [DOI] [Google Scholar]
- Wilson A. D.; Newell R. H.; McNevin M. J.; Muckerman J. T.; DuBois M. R.; DuBois D. L. Hydrogen Oxidation and Production Using Nickel-Based Molecular Catalysts with Positioned Proton Relays. J. Am. Chem. Soc. 2006, 128 (1), 358–366. 10.1021/ja056442y. [DOI] [PubMed] [Google Scholar]
- Artero V.; Fontecave M. Solar fuels generation and molecular systems: is it homogeneous or heterogeneous catalysis?. Chem. Soc. Rev. 2013, 42 (6), 2338–2356. 10.1039/C2CS35334B. [DOI] [PubMed] [Google Scholar]
- Lisovskaya A.; Kanjana K.; Bartels D. M. One-electron redox kinetics of aqueous transition metal couples Zn2+/+, Co2+/+, and Ni2+/+ using pulse radiolysis. Phys. Chem. Chem. Phys. 2020, 22 (34), 19046–19058. 10.1039/D0CP03214J. [DOI] [PubMed] [Google Scholar]
- Shi C.; Chan K.; Yoo J. S.; Nørskov J. K. Barriers of Electrochemical CO2 Reduction on Transition Metals. Org. Process Res. Dev. 2016, 20 (8), 1424–1430. 10.1021/acs.oprd.6b00103. [DOI] [Google Scholar]
- Stewart M. P.; Ho M.-H.; Wiese S.; Lindstrom M. L.; Thogerson C. E.; Raugei S.; Bullock R. M.; Helm M. L. High Catalytic Rates for Hydrogen Production Using Nickel Electrocatalysts with Seven-Membered Cyclic Diphosphine Ligands Containing One Pendant Amine. J. Am. Chem. Soc. 2013, 135 (16), 6033–6046. 10.1021/ja400181a. [DOI] [PubMed] [Google Scholar]
- Lawrence M. A. W.; Celestine M. J.; Artis E. T.; Joseph L. S.; Esquivel D. L.; Ledbetter A. J.; Cropek D. M.; Jarrett W. L.; Bayse C. A.; Brewer M. I.; Holder A. A. Computational, electrochemical, and spectroscopic studies of two mononuclear cobaloximes: the influence of an axial pyridine and solvent on the redox behaviour and evidence for pyridine coordination to cobalt(i) and cobalt(ii) metal centres. Dalton Trans. 2016, 45 (25), 10326–10342. 10.1039/C6DT01583B. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ledbetter K.; Larsen C. B.; Lim H.; Zoric M. R.; Koroidov S.; Pemmaraju C. D.; Gaffney K. J.; Cordones A. A. Dissociation of Pyridinethiolate Ligands during Hydrogen Evolution Reactions of Ni-Based Catalysts: Evidence from X-ray Absorption Spectroscopy. Inorg. Chem. 2022, 61 (26), 9868–9876. 10.1021/acs.inorgchem.2c00167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans D. F. The determination of the paramagnetic susceptibility of substances in solution by nuclear magnetic resonance. J. Chem. Soc. 1959, 2003–2005. 10.1039/jr9590002003. [DOI] [Google Scholar]
- Sheldrick G. M.Simens Area Detector Absorption Correction Program.; University of Göttingen: Göttingen, Germany, 1996.
- Sheldrick G. M.Program for Crystal Structure Determination.; Siemens Analytical X-ray Instruments Inc.: MAdison, WI, 1994.
- Boutin E.; Merakeb L.; Ma B.; Boudy B.; Wang M.; Bonin J.; Anxolabéhère-Mallart E.; Robert M. Molecular catalysis of CO2 reduction: recent advances and perspectives in electrochemical and light-driven processes with selected Fe, Ni and Co aza macrocyclic and polypyridine complexes. Chem. Soc. Rev. 2020, 49 (16), 5772–5809. 10.1039/D0CS00218F. [DOI] [PubMed] [Google Scholar]
- Becke A. D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38 (6), 3098–3100. 10.1103/PhysRevA.38.3098. [DOI] [PubMed] [Google Scholar]
- Barone V.; Cossi M. Quantum Calculation of Molecular Energies and Energy Gradients in Solution by a Conductor Solvent Model. J. Phys. Chem. A 1998, 102 (11), 1995–2001. 10.1021/jp9716997. [DOI] [Google Scholar]
- Cossi M.; Rega N.; Scalmani G.; Barone V. Energies, structures, and electronic properties of molecules in solution with the C-PCM solvation model. J. Comput. Chem. 2003, 24 (6), 669–681. 10.1002/jcc.10189. [DOI] [PubMed] [Google Scholar]
- Weigend F.; Ahlrichs R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7 (18), 3297–3305. 10.1039/b508541a. [DOI] [PubMed] [Google Scholar]
- Keith J. A.; Nielsen R. J.; Oxgaard J.; Goddard W. A. Unraveling the Wacker Oxidation Mechanisms. J. Am. Chem. Soc. 2007, 129 (41), 12342–12343. 10.1021/ja072400t. [DOI] [PubMed] [Google Scholar]
- Cramer C. J.Essentials of Computational Chemistry: Theories and Models.; John Wiley & Sons: 2013. [Google Scholar]
- Wertz D. H. Relationship between the gas-phase entropies of molecules and their entropies of solvation in water and 1-octanol. J. Am. Chem. Soc. 1980, 102 (16), 5316–5322. 10.1021/ja00536a033. [DOI] [Google Scholar]
- Cooper J.; Ziegler T. A Density Functional Study of SN2 Substitution at Square-Planar Platinum(II) Complexes. Inorg. Chem. 2002, 41 (25), 6614–6622. 10.1021/ic020294k. [DOI] [PubMed] [Google Scholar]
- Tissandier M. D.; Cowen K. A.; Feng W. Y.; Gundlach E.; Cohen M. H.; Earhart A. D.; Coe J. V.; Tuttle T. R. The Proton’s Absolute Aqueous Enthalpy and Gibbs Free Energy of Solvation from Cluster-Ion Solvation Data. J. Phys. Chem. A 1998, 102 (40), 7787–7794. 10.1021/jp982638r. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.