Abstract
Acute respiratory distress syndrome (ARDS) is a severe lung disease characterized by significant hypoxemia, which impairs the oxygen supply necessary for optimal lung function. This study aimed to investigate the effects of sodium propionate (SP), the primary end product of intestinal flora fermentation of dietary fiber, on lipopolysaccharide (LPS)-induced ARDS in rats. The rats were treated with SP, after which the lung wet/dry ratio, arterial partial oxygen pressure (PaO2), levels of pro- and anti-inflammatory cytokines, tight junction proteins ZO-1 and Occludin, as well as LC3 and phosphorylated PI3K (p-PI3K)/p-AKT/p-mTOR protein levels, were measured. Additionally, histopathological analysis was conducted. The results indicated that SP effectively alleviated arterial hypoxemia in rats and mitigated the pathological damage to both intestinal and lung tissues caused by LPS. Notably, SP significantly reduced the levels of inflammatory factors TNF-α and IL-6 in the blood and bronchoalveolar lavage fluid (BALF) of ARDS rats, while increasing the concentration of the anti-inflammatory factor IL-10. Furthermore, SP inhibited the activation of the PI3K/AKT/mTOR signaling pathway and enhanced the LC3II/LC3I ratio in lung tissue. Therefore, SP may improve LPS-induced ARDS in rats by inhibiting the activation of the PI3K/AKT/mTOR signaling pathway, promoting autophagy, decreasing the production and release of inflammatory markers, and reducing alveolar epithelial damage.
Keywords: Acute respiratory distress syndrome, Short-chain fatty acids, Sodium propionate, PI3K/AKT/mTOR signaling pathway
Introduction
Acute respiratory distress syndrome (ARDS) is a serious and life-threatening condition associated with significant morbidity and mortality (Bellani et al. 2016). Exposure to pathogenic factors such as infection, trauma, and shock triggers an inflammatory response in the lungs, causing damage to the alveolar epithelial and endothelial cells of the capillaries. This damage results in alveolar hemorrhage, pulmonary edema, and hyaline membrane formation, which further impair gas exchange. Clinically, ARDS is characterized by respiratory distress and refractory hypoxemia. However, treatment options for ARDS are currently limited to general supportive care, leading to minimal improvement in patient mortality rates. Our previous research has demonstrated that fecal microbiota transplantation (FMT) can effectively regulate disrupted intestinal microbiota, resulting in an improvement in ARDS. This is achieved through a reduction in the secretion of pro-inflammatory factors and the inhibition of oxidative stress mechanisms (Li et al. 2020). Additionally, research has confirmed the existence of a communication network between the gut microbiota and the lung microbiota (Chakradhar 2017). Short-chain fatty acids (SCFAs), metabolic products generated by the gut microbiota, play a vital role in maintaining an anti-inflammatory/pro-inflammatory balance. Consequently, SCFAs are recognized as the primary mediators of communication between the gut microbiota and immune inflammation. Studies have shown that SCFAs exert not only local effects in the gut but also possess immunomodulatory effects on multiple organs and systems within the body (Samuelson et al. 2015). One specific SCFA, sodium propionate (SP), plays a crucial role in maintaining gut and immune homeostasis. Furthermore, SP has been demonstrated to possess anti-tumor and anti-inflammatory properties, lower cholesterol levels, and reduce fat accumulation (Li et al. 2023). Studies indicate that SP can regulate lung immune responses in vitro and in vivo, thereby influencing inflammatory reactions (Tian et al. 2019). Previous studies have demonstrated that FMT can improve ARDS by modulating the phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT)/nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling pathway. The PI3K/AKT pathway is involved in various cellular processes, including cell proliferation, apoptosis, angiogenesis, immune function, and inflammatory responses. Furthermore, the mammalian target of rapamycin (mTOR) pathway server as a crucial regulator of cellular energy metabolism, playing vital roles in cell survival, proliferation, and metabolism. Notably, mTOR interacts with pro-inflammatory signaling pathways and contributes to the pathogenesis of inflammatory diseases (Xue et al. 2017). Therefore, this study aims to investigate the effects and potential mechanisms by which SP counters lipopolysaccharide (LPS) induced ARDS.
Materials and methods
Reagents
LPS (#L2630) and SP (#P1880) were purchased from Sigma-Aldrich. The IL-10 (#ELK1144), IL-6 (#ELK1158) and TNF-α (#ELK1396) ELISA kits were obtained from ELK Biotechnology. For western blotting analysis, rabbit monoclonal antibodies targeting p-PI3K, p-AKT, p-mTOR, LC3 and GAPDH were purchased from ASPEN. ZO-1 (#ab221546) and Occludin (#ab216327) anti-rabbit antibodies were purchased from Abcam.
Animal experiments
Male Sprague–Dawley (SD) rats of Specific Pathogen-Free (SPF) grade, weighing 170 ± 20 g, were obtained from the Experimental Animal Center of Southwest Medical University (Animal Certificate No. SCXK [Chuan] 2018–17). This work was approved by the Animal Research Ethics Committee of Southwest Medical University (swmu20230020). All animal experiments adhered to the guidelines established by the Animal Care and Use Committee of Southwest Medical University and followed the principles outlined in the Guide for the Care and Use of Laboratory Animals. The rats were housed under standard laboratory conditions which included a 12-h light/dark cycle, a constant temperature of 20–24 °C and humidity maintained at 55 ± 5%. They had continuous access to water and food. Twenty adult male SD rats were randomly divided into four groups: a control group, an ARDS group, a low-dose SP prevention group (referred to as the low-dose group), and a high-dose SP prevention group (referred to as the high-dose group). Prior to modeling, the rats in the SP groups were orally administered low-dose SP (100 mg/kg) and high-dose SP (200 mg/kg). The ARDS and control groups received an equivalent volume of normal saline, administered orally twice a day for seven consecutive days. At the conclusion of the intervention, the control group underwent saline lavage, while the remaining groups were directly instilled with LPS (5 mg/kg) into the trachea to establish the rat ARDS model.
Arterial blood gas analysis
The rats were anesthetized with 2% pentobarbital (40 mg/kg) 12 h following LPS stimulation. Once satisfactory anesthesia was achieved, each rat was positioned supine, and the abdominal cavity was opened to expose the abdominal aorta. A volume of 1 mL of blood was collected from the abdominal aorta using an arterial blood gas needle. Subsequently, a 5 mL empty syringe was utilized to collect an additional approximately 5 mL of blood from the abdominal aorta, which was then allowed to stand at room temperature. The rats were sacrificed by exsanguination after blood was collected from the aortaventralis. An arterial blood sample of 1 mL was extracted for the detection of arterial blood oxygen partial pressure (PaO2) using an automatic blood gas analyzer (Abbott, USA).
Cytokine detection with ELISA
As previously reported, after the rats were anesthetized and sacrificed through exsanguination, 5 ml arterial blood samples were collected, and serum was isolated via centrifugation at 3000 rpm for 20 min. Subsequently, the trachea and right lobe were separated, and a trachea cannula was inserted and secured in place. Bronchoalveolar lavage fluid (BALF) was then collected, and then the supernatant was obtained through centrifugation at 3000 rpm for 20 min. The levels of the cytokines TNF-α, IL-6, and IL-10 in both serum and BALF were quantified using an ELISA kit according to the manufacturer’s instructions.
Lung wet/dry weight ratio (W/D)
The chest of the rat was opened, and the lung tissue was isolated. The surface of the right lung samples was dried, and their wet weight (W) was measured. Subsequently, the samples were placed at 70 °C for 72 h to ensure complete dehydration, after which the dry weight (D) was obtained. The ratio of wet lung weight to dry lung weight (W/D) was then calculated.
Histopathological score
The upper lobe of the left lung and a 5 cm section of intestinal tissue were obtained and fixed in a 4% paraformaldehyde solution. Following fixation, the tissues were dehydrated in alcohol, embedded in paraffin, and sectioned to a thickness of 5 µm. The sections were subsequently dewaxed, rehydrated, washed in phosphate-buffered saline (PBS), and stained with hematoxylin and eosin. Histopathological scores for the lung tissue were determined based on several criteria, including alveolar congestion, hemorrhage, alveolar septal thickening, and interstitial and alveolar neutrophilic infiltration. The scoring system consisted of five criteria that assessed the degree of deterioration: normal (score 0), mild alteration (score 1), moderate alteration (score 2), severe alteration (score 3), and severest alteration (score 4). The lung injury score was recorded for each criterion. For the intestinal tissue, the histopathological score was based on the evaluation method described by Araki (Araki et al. 2004), which assessed the degree of mucosal damage, hemorrhage, presence or absence of crypt destruction, interstitial edema, and inflammatory cell infiltration. Mild, moderate, and severe damage were assigned scores of 1, 2, and 3, respectively. For evaluation purposes, five regions were randomly selected under a high-power lens, and the average total score was used as the final rating.
Western blotting analysis
The left lower lobe tissue of the lung was minced, and RIPA lysis buffer was employed to extract total protein. The protein concentration was quantified using the BCA protein assay kit. The extracted proteins underwent SDS-PAGE electrophoresis, followed by transfer to a membrane and blocking with 5% skim milk. The membrane was then incubated overnight at 4 °C with primary antibodies targeting p-PI3K, p-AKT, p-mTOR, LC3, and GADPH. Subsequently, the membrane was incubated with the anti-rabbit IgG HRP-linked secondary antibody at room temperature for 30 min. After development, the membrane was scanned using the Amersham Imager 600 scanner (USA). A gel imaging processing system was employed to analyze the film and perform semi-quantitative analysis, with GADPH serving as the control.
Immunohistochemical (IHC) analysis
Intestinal tissue located approximately 5 cm from the terminal ileum of rats was embedded in paraffin and sectioned. A series of sequential procedures were then performed, including dewaxing, hydration, treatment with 3% H2O2, antigen retrieval, overnight incubation at 4 °C with the primary antibody, incubation at 37 °C with the HRP-conjugated anti-rabbit secondary antibody, DAB color development, re-dyeing, dehydration, transparency, and sealing. Periodic photographs were taken, followed by semi-quantitative analysis. The integral optical density (IOD) value was measured, and the average was calculated based on five randomly selected fields. This average value was subsequently used as the relative expression of ZO-1 and Occludin in each sample.
Statistical analysis
The data were analyzed using SPSS version 27.0. The experimental results are presented as the mean ± standard deviation (SD). Statistical analysis was conducted using one-way analysis of variance, followed by either the least significant difference (LSD) multiple comparison test or Dunnett’s T3 test, depending on the specific circumstances. A P value of less than 0.05 was considered statistically significant.
Results
SP significantly ameliorated hypoxemia and lung injury in ARDS rats
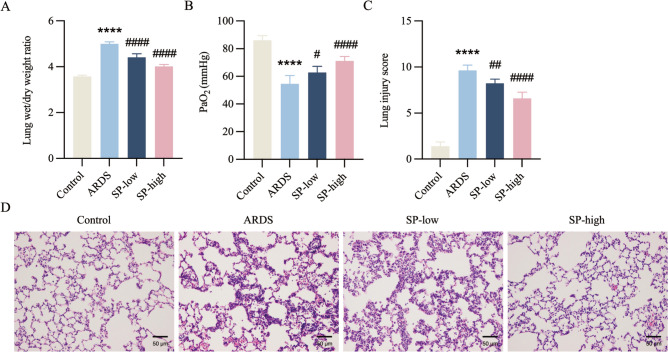
To assess the effects of SP on ARDS, the LPS-induced ARDS model was pre-treated with SP. The results presented in Fig. 1A, B indicate that pretreatment with SP, particularly in the high-dose group, significantly enhanced PaO2 and reduced the lung W/D ratio in rats with ARDS. Histopathological analysis (Fig. 1C, D) demonstrated that the alveoli in the control group exhibited uniform size and intact structure, with no thickening of septa, red blood cell infiltration, or inflammatory cell presence. Conversely, the ARDS group displayed disrupted structural integrity of the alveoli, septal rupture, increased lung foam thickness, and heightened exudation of cells within both the alveolar cavity and interstitium. The ARDS group also exhibited significantly elevated lung injury scores compared to the control group (P < 0.05). Notably, SP pretreatment, particularly at higher doses, significantly improved lung injury in ARDS, revealing pathological findings akin to those in the control group. These results suggest that the administration of SP, especially at higher doses, has the potential to alleviate hypoxemia and lung injury in ARDS rats.
Fig. 1.
SP ameliorated hypoxemia and lung injury in ARDS rats. A Lung wet/dry weight ratio in rats. B Arterial partial oxygen pressure (PaO2) in rats. C Pathological score of lung injury in rats. D HE staining of lung tissue in rats (×200). For all bar graphs, data are mean ± SD, ****p < 0.0001, as compared with Control, and #p < 0.05, ##p < 0.01, ####p < 0.0001, as compared with ARDS
SP effectively suppressed both systemic and local lung inflammation in ARDS rats
Acute inflammation and vascular leakage are critical features of acute lung injury (ALI) and the ARDS (Nonas et al. 2006). Individuals suffering from ALI and ARDS display significantly elevated levels of proinflammatory cytokines, such as IL-6 and TNF-α, in both their blood and lung tissues (Zhao et al. 2023). Figure 2 illustrates a marked increase in TNF-α and IL-6 levels in the blood and BALF of the LPS-induced ARDS group. However, these levels were significantly reduced following pretreatment with SP. Furthermore, the results indicated that SP significantly increased IL-10 levels in both the blood and BALF, which is recognized as an anti-inflammatory cytokine (Hagiwara et al. 2008). Additionally, the effects of high-dose SP were particularly pronounced. These findings demonstrate the efficacy of SP in managing both systemic and localized lung inflammation in ARDS rats.
Fig. 2.
SP effectively suppressed both systemic and local lung inflammation in ARDS rats. A TNF-α concentration in bronchoalveolar lavage fluid (BALF) of rats. B IL-6 concentration in BALF of rats. C IL-10 concentration in BALF of rats. D TNF-α concentration in serum of rats. E IL-6 concentration in serum of rats. F IL-10 concentration in serum of rats. For all bar graphs, data are mean ± SD, **p < 0.01, ***p < 0.001, ****p < 0.0001, as compared with Control, and #p < 0.05, ##p < 0.01, ####p < 0.0001, as compared with ARDS
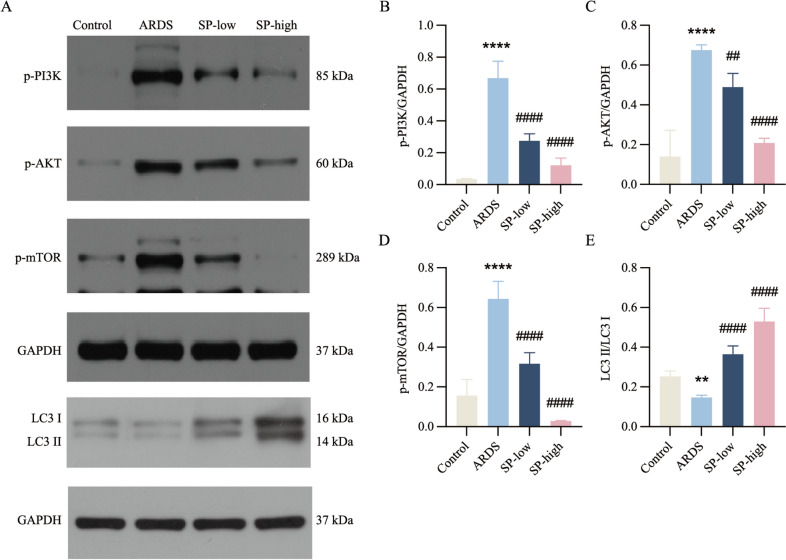
SP inhibited the activation of the PI3K/AKT/mTOR signaling pathway and promoted autophagy in ARDS rats
Activation of the PI3K/AKT/mTOR signaling pathway is crucial for the development of LPS-induced ARDS (Xia et al. 2023). Figure 3 demonstrates a significant upregulation of phosphorylated PI3K, AKT, and mTOR proteins in the lung tissue of the LPS-induced ARDS group. However, pretreatment with SP, particularly at high doses, effectively inhibits this upregulation. mTOR is responsible for regulating autophagy in cellular metabolism (Xie et al. 2019). The results indicate that the ratio of LC3II to LC3I, which serves as an indicator of autophagy, was decreased in the lung tissue of the ARDS group. In contrast, pretreatment with SP, especially at high doses, substantially increased this ratio. These findings suggest that SP may play a role in the prevention and treatment of ARDS by inhibiting the activation of the PI3K/AKT/mTOR signaling pathway and promoting autophagy.
Fig. 3.
SP inhibited the activation of the PI3K/AKT/mTOR signaling pathway and promoted autophagy in ARDS rats. A Western-blot was used to evaluate the expression of p-PI3K, p-AKT, p-mTOR and LC3 in lung tissue of rats. B The relative intensity of p-PI3K protein bans normalized to GAPDH band. C The relative intensity of p-AKT protein bans normalized to GAPDH band. D The relative intensity of p-mTOR protein bans normalized to GAPDH band. E The intensity ratio of LC3 II band to LC3I band. For all bar graphs, data are mean ± SD, **p < 0.01, ****p < 0.0001, as compared with Control, and ##p < 0.01, ####p < 0.0001, as compared with ARDS
SP ameliorated intestinal injury and preserved intestinal mucosal integrity in ARDS rats
The occurrence of ALI/ARDS is associated with elevated levels of inflammatory cytokines and the transfer of the lung microbiome. These factors can disrupt the gut microbiome and compromise the integrity of the intestinal mucosal barrier, leading to dysregulation of gut barrier function. Consequently, this disruption, along with alterations in gut microbiota and translocation of gut microbiota, exacerbates both local and systemic inflammatory responses, ultimately disrupting of immune homeostasis in the systemic environment and contributing to the development of ALI/ARDS (Zhou and Liao 2021). Our study’s findings (Fig. 4A, B) convincingly demonstrated that the intestinal mucosal epithelium of LPS-induced ARDS rats exhibited significant damage compared to the control group. This damage was characterized by mucosal edema, structural damage to crypts, intermittent bleeding, ulcer lesions, and infiltration of inflammatory cells. Additionally, the expression of tight junction proteins ZO-1 and Occludin, which are indicative of the integrity of the intestinal epithelial mucosal barrier, was found to be reduced (Fig. 4C–F). The pathological score of the intestinal mucosa in the ARDS group was notably higher than that of the control group, confirming the existence of "gut-lung" crosstalk in ARDS. Importantly, SP preconditioning significantly ameliorated intestinal damage in ARDS rats and upregulated the expression of ZO-1 and Occludin in the intestinal epithelium, thereby preserving the integrity of the intestinal mucosal barrier.
Fig. 4.
SP ameliorated intestinal injury and preserved intestinal mucosal integrity in ARDS rats. A HE staining of intestinal tissue in rats (×200). B Pathological score of intestinal injury in rats. C ZO-1 integral optical density (IOD) in intestinal tissue of rats. D Occludin integral optical density (IOD) in intestinal tissue of rats. E IHC staining of ZO-1 in intestinal tissue of rats. F IHC staining of Occludin in intestinal tissue of rats. For all bar graphs, data are mean ± SD, ****p < 0.0001, as compared with Control, and ####p < 0.0001, as compared with ARDS
Discussion
Inflammatory storms rapidly generate a variety of cytokines in the lung, including IL-6, TNF-α, IFN-γ, and IL-12. This cascade of cytokine production leads to cellular death, disruption of epithelial and endothelial barriers, and edema, which is the primary cause of ARDS (Kim et al. 2021). Currently, there is no targeted treatment for ARDS; corticosteroids are frequently prescribed as the initial therapeutic option due to their potent anti-inflammatory and antifibrotic properties. However, high-dose corticosteroid therapy has not been shown to improve mortality in ARDS patients and is associated with numerous adverse reactions (Sweeney and McAuley 2016). Therefore, there is an urgent need to identify novel therapeutic approaches. Crosstalk among similar targets across various organs enables a single drug to exert therapeutic effects on diseases affecting different regions of the body (Sharma et al. 2022)(Goel et al. 2023). The “gut-lung axis” crosstalk represents a significant mechanism for maintaining immune homeostasis and plays a vital role in overall health (Sultan et al. 2021). SCFAs, which are indirect nutrients synthesized by intestinal microorganisms and predominantly include acetate, propionate, and butyrate, have the capacity to promote intestinal homeostasis while inhibiting intestinal inflammation (Tan et al. 2023). Previous research has demonstrated the immunomodulatory and anti-inflammatory effects of SCFAs on ARDS, with the combined administration of the three primary SCFAs effectively reducing the release of inflammatory cytokines in a rat model of LPS-induced ARDS (Xiang et al. 2022). Sodium butyrate has been shown to inhibit the expression of pro-inflammatory cytokines, such as TNF-α and IL-6, and suppress the activation of the NF-κB signaling pathway in the ALI/ARDS mouse model, thereby alleviating inflammation associated with LPS-induced ALI (Liu et al. 2019)(Li et al. 2018). Studies conducted in mice have demonstrated that acetate inhibits the permeability of the alveolar-capillary barrier, reduces pulmonary edema, mitigates oxidative stress, suppresses the recruitment of inflammatory cells and the production of inflammatory mediators, and modulates the activation of the MAPK pathway. Collectively, these effects contribute to the improvement of ALI/ARDS (Xu et al. 2019). Additionally, studies have indicated that SP administration alone provides a protective effect against LPS-induced mastitis (Wang et al. 2017). However, the protective effects of propionate on ARDS remain unknown.
In this study, we conducted a comprehensive analysis of the effects of SP treatment on ARDS. The findings demonstrate that SP significantly improves hypoxemia, attenuates pulmonary edema, and reduces of lung injury in the LPS-induced ARDS rat model. Pro-inflammatory cytokines, such as TNF-α and IL-6, play crucial roles in both the physiology and pathology of inflammation, with TNF-α notably initiating an inflammatory cascade (Kumar et al. 2023). Elevated levels of pro-inflammatory cytokines TNF-α and IL-6 in the plasma of ARDS patients are critical indicators of an increased risk of mortality (Meduri et al. 1995), while the production of the anti-inflammatory cytokine IL-10 has been shown to mitigate ALI (Chai et al. 2020). The results of this study indicate that SP significantly inhibits the secretion of pro-inflammatory cytokines TNF-α and IL-6 in both the systemic circulation and lungs of ARDS rats, while concurrently promoting the expression of the anti-inflammatory cytokine IL-10. Consequently, the protective effect of SP on ARDS in rats may be attributed to its regulation of both pro-inflammatory and anti-inflammatory factors. ARDS is widely recognized for initiating systemic inflammation and hypoxemia, often accompanied by intestinal mucosal damage (Ye et al. 2020). Moreover, high doses of SP exhibit more pronounced anti-inflammatory effects and reductions in lung injury. The intestinal mucosa is crucial for maintaining intestinal function, with its epithelial cells serving as a primary defense barrier against foreign bacteria. A compromised intestinal mucosal barrier leads to the translocation of bacteria and endotoxins, which can precipitate sepsis. This process also activates mucosal immune cells, resulting in acute inflammation and exacerbating lung injury, thereby contributing to the development of ARDS (Zhou and Liao 2021). In our LPS-induced ARDS model, we observed concomitant infiltration of intestinal mucositis cells, mucosal edema, and decreased expression of tight junction proteins ZO-1 and Occludin, indicating impairment of the mucosal barrier in the intestinal epithelium. The administration of SP effectively mitigated intestinal mucosal damage and restored the integrity of the mucosal barrier. These findings provide further evidence for the existence of the "gut-lung axis", delineating the reciprocal relationship between respiratory diseases and gastrointestinal function through immune regulation. Additionally, they reinforce the notion that metabolites from intestinal flora impact the respiratory system via a common mucosal immune system.
The PI3K and AKT pathways are crucial in regulating cellular functions associated with host defense and immune responses. Activation of the PI3K/AKT pathway and the downstream signaling molecule mTOR contributes to the infiltration of immune cells, the release of inflammatory cytokines, and the development of LPS-induced ALI (Wang et al. 2020). This study confirmed that the levels of phosphorylated proteins PI3K, AKT, and mTOR were significantly elevated in the lung tissue of LPS-induced ARDS rats. In contrast, the expression of phosphorylated PI3K, AKT, and mTOR was reduced in the lung tissue of rats that were pretreated orally with SP. It can be speculated that LPS activates the PI3K/AKT/mTOR signaling pathway, which induces ARDS and promotes the release of downstream inflammatory factors such as TNF-α and IL-6, while concurrently inhibiting the anti-inflammatory factor IL-10. Notably, oral pretreatment with SP significantly decreased the expression of phosphorylated PI3K, AKT, and mTOR. Therefore, we infer that SP may enhance lung function and mitigate related inflammatory responses by inhibiting the PI3K/AKT/mTOR signaling pathway and the release of inflammatory factors, thereby exerting a protective effect against lung injury in ARDS rats. Autophagy is a catabolic cellular mechanism through which lysosomes degrade proteins and organelles as a protective response (Galluzzi et al. 2018). Under conditions of cellular stress, autophagy promotes homeostasis and harnesses biological energy through the degradation of cellular components (Sauler et al. 2019). The LC3 protein serves as a marker of autophagy, with LC3I gradually transforming into lipidized LC3II (Nakamura et al. 2021). In this study, we found that the LC3II/LC3I ratio decreased in the LPS-induced ARDS model, while SP treatment reversed this decrease, suggesting the presence of autophagy inhibition during the LPS-induced ARDS process. Furthermore, SP may enhance lung tissue recovery from inflammatory injury by promoting autophagy. Previous studies have indicated that LPS stimulates the proliferation of lung fibroblasts and induces pulmonary fibrosis by activating the PI3K/AKT/mTOR signaling pathway and inhibiting autophagy (Xie et al. 2019). The correlation analysis conducted in this study revealed that the levels of systemic and local lung inflammatory cytokines TNF-α and IL-6 were positively correlated with the activation of the PI3K/AKT/mTOR pathway and negatively correlated with the occurrence of autophagy. Additionally, the occurrence of autophagy was inversely correlated with the activation of the PI3K/AKT/mTOR pathway. These results suggest that the PI3K/AKT/mTOR/autophagy signaling axis may play a critical role in the development of the LPS-induced ARDS inflammatory storm. SP promotes autophagy by inhibiting the activation of the PI3K/AKT/mTOR signaling pathway, thereby alleviating LPS-induced systemic and local lung inflammation.
Conclusion
In conclusion, pretreatment with SP effectively prevents lung injury and modulates systemic and local lung inflammation induced by LPS-induced ARDS in rats. The underlying mechanism of this protective role in ARDS may involve the inhibition of the PI3K/AKT/mTOR signaling pathway, promotion of autophagy, reduction of inflammatory cell infiltration, and modulation of inflammatory cytokine release. Furthermore, the immunomodulatory effects of microbial metabolites reinforce the concept of the "gut-lung axis" connecting the respiratory and intestinal tracts. Given the high degree of homology between rat and human genes, rat ARDS models are well-suited for in vivo research that accurately reflects the pathophysiological changes associated with human ARDS. Consequently, this study provides a theoretical foundation for the clinical application of SP in the treatment of ARDS, suggesting that SP may represent a promising new therapeutic strategy. However, this experiment did not clarify the effectiveness and safety of SP for clinical use, indicating that future clinical studies with larger sample sizes are necessary for a more comprehensive evaluation.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Jiang-Shan Zhong, Chun-Lan Chen, Peng Tian and Jie Chen. The first draft of the manuscript was written by Fang He and Xian-Ming Fan and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by Natural Science Foundation of Sichuan Province of China (2022NSFSC0046).
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Ethics approval
This work was approved from the Animal Research Ethics Committee of Southwest Medical University (swmu20230020). All animal experiments were approved by the Animal Care and Use Committee of the Southwest Medical University and followed the guide of Care and Use of Laboratory Animals.
Footnotes
Fang He and Jiang-Shan Zhong contributed equally to this work.
References
- Araki Y, Andoh A, Takizawa J, Takizawa W, Fujiyama Y (2004) Clostridium butyricum, a probiotic derivative, suppresses dextran sulfate sodium-induced experimental colitis in rats. Int J Mol Med 13(4):577–580 [PubMed] [Google Scholar]
- Bellani G, Laffey JG, Pham T, Fan E, Brochard L, Esteban A, Pesenti A (2016) Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 Countries. JAMA 315(8):788–800. 10.1001/jama.2016.0291 [DOI] [PubMed] [Google Scholar]
- Chai YS, Chen YQ, Lin SH, Xie K, Wang CJ, Yang YZ, Xu F (2020) Curcumin regulates the differentiation of naïve CD4+T cells and activates IL-10 immune modulation against acute lung injury in mice. Biomed Pharmacother 125:109946. 10.1016/j.biopha.2020.109946 [DOI] [PubMed] [Google Scholar]
- Chakradhar S (2017) A curious connection: teasing apart the link between gut microbes and lung disease. Nat Med 23(4):402–404. 10.1038/nm0417-402 [DOI] [PubMed] [Google Scholar]
- Galluzzi L, Vitale I, Aaronson SA, Abrams JM, Adam D, Agostinis P, Kroemer G (2018) Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ 25(3):486–541. 10.1038/s41418-017-0012-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goel A, Yamini D, Gusain C, Sharma B, Purohit R, Das P, Padwad Y (2023) Synthesis, anti-adipogenic, and insulin-sensitizing potential of benzosuberene-alkyl sulfone (BSAS) analogues. Chem Asian J 18(13):e202300179. 10.1002/asia.202300179 [Google Scholar]
- Hagiwara S, Iwasaka H, Hidaka S, Hishiyama S, Noguchi T (2008) Danaparoid sodium inhibits systemic inflammation and prevents endotoxin-induced acute lung injury in rats. Crit Care 12(2):R43. 10.1186/cc6851 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JS, Lee JY, Yang JW, Lee KH, Effenberger M, Szpirt W, Shin JI (2021) Immunopathogenesis and treatment of cytokine storm in COVID-19. Theranostics 11(1):316–329. 10.7150/thno.49713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar S, Chhimwal J, Kumar S, Singh R, Patial V, Purohit R, Padwad YS (2023) Phloretin and phlorizin mitigates inflammatory stress and alleviate adipose and hepatic insulin resistance by abrogating PPARγ S273-Cdk5 interaction in type 2 diabetic mice. Life Sci 322:121668. 10.1016/j.lfs.2023.121668 [DOI] [PubMed] [Google Scholar]
- Li B, Yin GF, Wang YL, Tan YM, Huang CL, Fan XM (2020) Impact of fecal microbiota transplantation on TGF-β1/Smads/ERK signaling pathway of endotoxic acute lung injury in rats. Biotech 10(2):52. 10.1007/s13205-020-2062-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li N, Liu XX, Hong M, Huang XZ, Chen H, Xu JH, Gong Q (2018) Sodium butyrate alleviates LPS-induced acute lung injury in mice via inhibiting HMGB1 release. Int Immunopharmacol 56:242–248. 10.1016/j.intimp.2018.01.017 [DOI] [PubMed] [Google Scholar]
- Li W, Deng M, Gong J, Hou Y, Zhao L (2023) Bidirectional regulation of sodium acetate on macrophage activity and its role in lipid metabolism of hepatocytes. Int J Mol Sci. 10.3390/ijms24065536 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Chang G, Huang J, Wang Y, Ma N, Roy AC, Shen X (2019) Sodium butyrate inhibits the inflammation of lipopolysaccharide-induced acute lung injury in mice by regulating the toll-like receptor 4/nuclear factor κB signaling pathway. J Agric Food Chem 67(6):1674–1682. 10.1021/acs.jafc.8b06359 [DOI] [PubMed] [Google Scholar]
- Meduri GU, Headley S, Kohler G, Stentz F, Tolley E, Umberger R, Leeper K (1995) Persistent elevation of inflammatory cytokines predicts a poor outcome in ARDS Plasma IL-1 beta and IL-6 levels are consistent and efficient predictors of outcome over time. Chest 107(4):1062–1073. 10.1378/chest.107.4.1062 [DOI] [PubMed] [Google Scholar]
- Nakamura S, Akayama S, Yoshimori T (2021) Autophagy-independent function of lipidated LC3 essential for TFEB activation during the lysosomal damage responses. Autophagy 17(2):581–583. 10.1080/15548627.2020.1846292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nonas S, Miller I, Kawkitinarong K, Chatchavalvanich S, Gorshkova I, Bochkov VN, Birukov KG (2006) Oxidized phospholipids reduce vascular leak and inflammation in rat model of acute lung injury. Am J Respir Crit Care Med 173(10):1130–1138. 10.1164/rccm.200511-1737OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samuelson DR, Welsh DA, Shellito JE (2015) Regulation of lung immunity and host defense by the intestinal microbiota. Front Microbiol 6:1085. 10.3389/fmicb.2015.01085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sauler M, Bazan IS, Lee PJ (2019) Cell death in the lung: the apoptosis-necroptosis axis. Annu Rev Physiol 81:375–402. 10.1146/annurev-physiol-020518-114320 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma B, Hussain T, Khan MA, Jaiswal V (2022) Exploring AT2R and its polymorphism in different diseases: an approach to develop AT2R as a drug target beyond hypertension. Curr Drug Targets 23(1):99–113. 10.2174/1389450122666210806125919 [DOI] [PubMed] [Google Scholar]
- Sultan M, Wilson K, Abdulla OA, Busbee PB, Hall A, Carter T, Nagarkatti M (2021) Endocannabinoid anandamide attenuates acute respiratory distress syndrome through modulation of microbiome in the gut-lung axis. Cells. 10.3390/cells10123305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sweeney RM, McAuley DF (2016) Acute respiratory distress syndrome. Lancet 388(10058):2416–2430. 10.1016/s0140-6736(16)00578-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan JK, Macia L, Mackay CR (2023) Dietary fiber and SCFAs in the regulation of mucosal immunity. J Allergy Clin Immunol 151(2):361–370. 10.1016/j.jaci.2022.11.007 [DOI] [PubMed] [Google Scholar]
- Tian X, Hellman J, Horswill AR, Crosby HA, Francis KP, Prakash A (2019) Elevated gut microbiome-derived propionate levels are associated with reduced sterile lung inflammation and bacterial immunity in mice. Front Microbiol 10:159. 10.3389/fmicb.2019.00159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Wei Z, Zhang X, Wang Y, Yang Z, Fu Y (2017) Propionate protects against lipopolysaccharide-induced mastitis in mice by restoring blood-milk barrier disruption and suppressing inflammatory response. Front Immunol 8:1108. 10.3389/fimmu.2017.01108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang K, Zhang Y, Cao Y, Shi Z, Lin Y, Chen Y, Liu X (2020) Glycyrrhetinic acid alleviates acute lung injury by PI3K/AKT suppressing macrophagic Nlrp3 inflammasome activation. Biochem Biophys Res Commun 532(4):555–562. 10.1016/j.bbrc.2020.08.044 [DOI] [PubMed] [Google Scholar]
- Xia TT, Hu R, Shao CJ, Feng Y, Yang XL, Xie YP, Li XM (2023) Stanniocalcin-1 secreted by human umbilical mesenchymal stem cells regulates interleukin-10 expression via the PI3K/AKT/mTOR pathway in alveolar macrophages. Cytokine 162:156114. 10.1016/j.cyto.2022.156114 [DOI] [PubMed] [Google Scholar]
- Xiang XQ, Zhang TT, Zhong JS, Yin GF, Fan XM (2022) Effects and mechanism of short chain fatty acids on lipopolysaccharide induced acute respiratory distress syndrome in rats. Med J Chin People’s Liber 47(06): 561–568. Retrieved from https://kns.cnki.net/kcms/detail/11.1056.R.20220412.1156.006.html
- Xie T, Xu Q, Wan H, Xing S, Shang C, Gao Y, He Z (2019) Lipopolysaccharide promotes lung fibroblast proliferation through autophagy inhibition via activation of the PI3K-Akt-mTOR pathway. Lab Invest 99(5):625–633. 10.1038/s41374-018-0160-2 [DOI] [PubMed] [Google Scholar]
- Xu M, Wang C, Li N, Wang J, Zhang Y, Deng X (2019) Intraperitoneal injection of acetate protects mice against lipopolysaccharide (LPS)-induced acute lung injury through its anti-inflammatory and anti-oxidative ability. Med Sci Monit 25:2278–2288. 10.12659/msm.911444 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue JF, Shi ZM, Zou J, Li XL (2017) Inhibition of PI3K/AKT/mTOR signaling pathway promotes autophagy of articular chondrocytes and attenuates inflammatory response in rats with osteoarthritis. Biomed Pharmacother 89:1252–1261. 10.1016/j.biopha.2017.01.130 [DOI] [PubMed] [Google Scholar]
- Ye Q, Wang B, Zhang T, Xu J, Shang S (2020) The mechanism and treatment of gastrointestinal symptoms in patients with COVID-19. Am J Physiol Gastrointest Liver Physiol 319(2):G245-g252. 10.1152/ajpgi.00148.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y, Jin H, Lei K, Bai LP, Pan H, Wang C, Li T (2023) Oridonin inhibits inflammation of epithelial cells via dual-targeting of CD31 Keap1 to ameliorate acute lung injury. Front Immunol 14:1163397. 10.3389/fimmu.2023.1163397 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou X, Liao Y (2021) Gut-lung crosstalk in sepsis-induced acute lung injury. Front Microbiol 12:779620. 10.3389/fmicb.2021.779620 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.