Summary
Cryptococcosis, particularly in its most lethal manifestation of cryptococcal meningitis (CM), accounts for significant mortality and morbidity. It is a major worldwide disseminated invasive fungal infection. The breadth of the clinical cryptococcosis syndromes, the different host types at-risk and affected, and the vastly disparate resource settings in which clinicians practice, pose a complex array of challenges. Expert contributors from diverse regions of the world: collated data, reviewed the evidence, and provided insightful guideline recommendations for health practitioners across the globe.
This guideline offers practical guidance and implementable recommendations on clinical approaches, screening, diagnosis, management, and follow-up care, and serves as a comprehensive synthesis and update on cryptococcosis. It seeks to facilitate optimal clinical decision-making on this complex condition and addresses its myriad of clinical complications, by incorporating data from historical and contemporary clinical trials, grounded on a set of core management principles, while simultaneously acknowledging the practical challenges of antifungal access and resource limitations faced by many. Over 70 societies internationally have endorsed this global cryptococcosis guideline for its content, structure, evidence, recommendation, and pragmatic wisdom to inform clinicians about the past, present, and future and in the care of a patient with cryptococcosis.
Introduction
Cryptococcosis accounts for significant morbidity and mortality globally. The World Health Organization (WHO) in 2022 listed Cryptococcus neoformans as one of its top priority pathogens.1 Cryptococcosis most often involves the central nervous system (CNS) and/or the lungs, but disseminated disease may affect any organ, yet appear seemingly localised. Despite the knowledge gained and improvement in clinical outcomes generated by multiple interventional trials2–7 conducted primarily in resource-limited health care settings (RLS), mortality from cryptococcal meningoencephalitis (CM) remains unacceptably high, ranging from 24%−47% at 10 weeks.2,4,7,8 The highest burden of disease is encountered in low- and middle-income countries, especially in sub-Saharan Africa9, where HIV/AIDS remains the dominant risk factor, although new non-HIV immunocompromised risk groups, and putatively immunocompetent individuals, are increasingly reported in resource-rich (RRS) settings.
Complementary diagnostic and management guidelines for cryptococcosis exist.10–21 This comprehensive synthesis and update of cryptococcosis management guidelines, serves primarily to facilitate clinical decision making, but also provides an overview of the current uncertainties in cryptococcosis. With contributors across the globe, this guideline gives voice to expertise and challenges from diverse settings in a globally relevant document. General principles and treatment recommendations are provided and clinicians are urged to utilise careful clinical judgement when formulating treatment plans for the individual patient. (See Appendix for processes on guideline development (s19-s20, s28), common abbreviations (s21), definitions (s23) and detailed text (s32)).
At-risk populations, clinical presentations, and outcomes
Evidence:
Primarily acquired via inhalation but occurring mainly upon reactivation after a period of latency, cryptococcosis has protean manifestations with CM being the most common severe presentation. Pulmonary cryptococcosis is underdiagnosed and often subclinical. Disseminated cryptococcosis can involve any organ of the body, thus a thorough clinical assessment is required, even in seemingly asymptomatic individuals.24,25 While classical at-risk patient populations include persons living with HIV (PLHIV) and solid organ transplant (SOT) recipients, individuals with other immunosuppressive conditions or receiving immunosuppressant drugs and putatively immunocompetent hosts are affected by cryptococcosis (s5).
Those who survive cryptococcosis still report significant morbidity, ranging from 10%−70% depending on the disease syndrome and severity; underlying predisposing conditions of the host; and the healthcare system in which the patient is managed26–29 (s7).
Recommendations:
(AIII) Cryptococcosis should be considered in any patient presenting with compatible symptomatology and/or microbiology, regardless of their immune status.
(AIII) Among patients without known predisposition to cryptococcosis, exclusion of an underlying immunodeficiency (including performing HIV serology and CD4 T-cell count) is recommended in all patients presenting with cryptococcosis.
Yeasts causing cryptococcosis and their diagnostic methods.
Evidence:
C. neoformans species complex is the predominant causative agent of cryptococcosis in PLHIV, while C. gattii species complex more commonly causes disease in apparently immunocompetent hosts. While both can cause a similarly broad repertoire of cryptococcosis syndromes, C. neoformans has a predilection for CNS disease while C. gattii is more often associated with pulmonary disease and large cryptococcomas.30–32
Diagnostic modalities used to establish the diagnosis, extent, severity, and prognosis of cryptococcosis are constantly evolving (s9 and s31). Microscopy and culture of CSF pellet after centrifugation, and blood culture, accompanied by CSF and blood (serum, plasma, or whole blood) cryptococcal antigen testing (most commonly by lateral flow assay (CrAg LFA)) and radiological studies are central to the diagnosis of cryptococcosis.33,34
Recommendations:
(AIIt) All patients with suspected or confirmed cryptococcosis (including cryptococcal antigenemia) require careful clinical assessment for CNS, pulmonary and other body site involvement. Investigations for disseminated disease should include: (1) lumbar puncture (LP) with measurement of CSF opening pressure (OP), glucose, protein, cell counts, microscopy, culture, and quantification of CSF CrAg; (2) quantification of blood CrAg, and cultures of blood, sputum (or other respiratory specimens) and/or of other affected sites; (3) ideally, brain imaging (preferably MRI) and chest imaging (preferably CT).
Screening/Primary Prophylaxis/Pre-emptive Therapy
Evidence:
Supportive evidence for cryptococcal screening is limited to PLHIV and currently hinges on the blood CrAg LFA (see s32 for detailed discussion, including prophylaxis and pre-emptive therapy).
Recommendation:
- For adult PLHIV with CD4 <200 cells/mm3 (ART-naive or after a period of ART discontinuation):
- (AI) Performing a blood CrAg by LFA for the screening of cryptococcosis, and determine the CrAg titre if positive.
- (AIIt) All patients with cryptococcal antigenaemia should be carefully assessed and investigated for cryptococcosis (see section above) and treated as appropriate.
- (AIIu) In asymptomatic cryptococcal antigenaemic PLHIV without clinical cryptococcosis after thorough investigation (including at least a LP), fluconazole 1200 mg daily for 2 weeks (when ART may be initiated), followed by 800 mg daily for 8 weeks, and 200 mg thereafter for about 6 months is recommended. (Guidance may be updated contingent on results of prospective trials.)
- (BI) In clinical settings where CrAg LFA screening is not available (despite WHO’s strong recommendations), universal primary prophylaxis with fluconazole 100 mg daily in PLHIV in high endemic areas with CD4 count <200 cells/mm3 is recommended (see Pregnancy section for suitable alternative)
- Other (non-HIV) populations:
- (DIIu) Routine blood CrAg screening, primary prophylaxis and pre-emptive therapy are not currently recommended in non-HIV populations.
Principles of treatment and navigating the guidelines
See s25 for how to navigate the treatment sections of these guidelines, s26 for a general discussion on antifungal drugs used in cryptococcosis, and common adverse events (s15).
HIV-associated cryptococcal meningitis (CM) in RLS compared to RRS
Evidence:
Evolution of induction treatment:
Multiple studies support the successful combination of amphotericin B deoxycholate (Amb-D) plus 5-flucytosine as the induction treatment of choice in HIV-associated CM. First trialled by van der Horst and colleagues, the addition of 5-flucytosine to Amb-D, showed a trend towards improved CSF sterility at 2 weeks and reduced frequency of relapse.35 In a subsequent trial, this combination cleared cryptococci (measured as early fungicidal activity, EFA) more rapidly than either Amb-D alone or Amb-D plus fluconazole.5 Importantly, the combination of Amb-D 1 mg/kg daily plus 5-flucytosine 25 mg/kg four times a day showed a survival advantage at day 70, over Amb-D alone in the treatment of CM.2 The nephroprotection of L-Amb compared with Amb-D is long recognised and accessibility of L-Amb in RRS led to the establishment of L-Amb 3–4 mg/kg daily plus 5-flucytosine 25 mg/kg four times a day for 2 weeks, as the current standard.
Resource-limited settings:
In RLS, challenges with antifungal access, adverse effects and difficulty of monitoring and safely managing 2 weeks of Amb-D induction treatment led to phase 2 studies, exploring alternative regimens. Fluconazole monotherapy, even at doses up to 1200 mg daily, was associated >50% mortality at 10-weeks, and >75% mortality at one-year.36–38 An oral combination of fluconazole 1200 mg daily plus 5-flucytosine 25 mg/kg four times a day was associated with a significant improvement in EFA compared with fluconazole alone.39 Unsurprisingly, the addition of a short, 5–7 day course of Amb-D at 1 mg/kg daily to oral fluconazole or combined oral fluconazole and 5-flucytosine showed improved rates of cryptococcal clearance40,41, similar to rates observed with 14 days of Amb-D.
In the phase 3 ACTA trial conducted in centres in Africa, the oral combination of fluconazole 1200 mg daily and 5-flucytosine 25 mg/kg four times a day for 2 weeks was compared with 1-week of Amb-D 1 mg/kg daily and 2 weeks of Amb-D 1 mg/kg daily as induction therapy, with the latter two arms being further randomised with either fluconazole 1200 mg daily or 5-flucytosine 25 mg/kg four times a day.7 One-week Amb-D 1 mg/kg daily plus 5-flucytosine followed by fluconazole 1200 mg daily in the second week was the best performing induction arm, with a 24% 10-week mortality. This regimen was adopted as the preferred induction regimen by the WHO and Southern African guidelines until the AMBITION-cm study.16,18
In the AMBITION-cm phase III study across sites in Africa, a single initial 10 mg/kg dose of liposomal amphotericin B (L-Amb), with an oral backbone of fluconazole 1200 mg daily plus 5-flucytosine 25 mg/kg four times a day for 2 weeks was compared with the then WHO recommendation of 1-week Amb-D 1 mg/kg daily plus 5-flucytosine followed by 1 week of fluconazole 1200 mg daily.6 This met non-inferiority criteria (10 week mortality 24.8% vs 28.7%) with similar EFAs and was significantly better tolerated. The WHO guidelines now recommend the Ambition-cm regimen as the preferred antifungal therapy in PLHIV with CM.10
Resource-rich settings:
The applicability of the ACTA and AMBITION-cm trials to RRS and in non-HIV populations is contentious, where the current standard is L-Amb 3–4 mg/kg daily plus 5-flucytosine 25 mg/kg four times a day for 2 weeks, different to comparators used in these trials. Retrospective database reviews in the USA showed relatively low rates of acute inpatient mortality from CM (10.5% in HIV-CM and 13.3% in non-HIV) and remarkably low mortality rates at 1 year of 11.6% over the last two decades.42,43 The reliance on high-dose fluconazole and 5-flucytosine as the backbone to induction therapy in AMBITION-cm study may not be pragmatic in all RRS where more comorbidities occur, potential drug-drug interactions need to be carefully considered; and the risk of hepatotoxicity less tolerated. In the USA, only a third of patients completed the 14-days of 5-flucytosine.44 While some experts cogently propose for the inclusion of the AMBITION-cm triple regimen as a primary option in RRS, others are calling for further comparative trials in RRS to assess the regimen’s impact in HIV, and the inclusion of SOT and non-HIV, non-SOT populations where no supporting data exist. Regardless of the induction antifungal regimen used, the complications of CM such as increased intracranial pressure (ICP) require intense clinical monitoring, and most patients with CM require inpatient care for at least one, if not two or more weeks.
Significance of CSF sterility post-induction/pre-consolidation therapy:
Mycological success has been associated with improved outcomes and reduced clinical relapse.45 In PLHIV with CM, CSF sterility prior to ART commencement has been shown to be associated with reduced occurrence of neurological deterioration, microbiological relapse, and cryptococcosis-associated immune reconstitution inflammatory syndrome (C-IRIS).45 Some treatment guidelines advocate performing a 2-week LP (prior to changing to consolidation therapy) to assess CSF culture sterility as a marker of successful induction.11,15,18,20 Other guidelines – particularly those focused on RLS - do not.10,16
Consolidation and maintenance treatment:
There are no recent trials of consolidation and maintenance therapy in CM. Two early studies established fluconazole for consolidation therapy35,46, with 400 mg daily. With the accumulation of safety data an 800 mg daily dose and evidence of a fluconazole dose-response effect,36,47 this is the preferred consolidation dose in RLS, where suboptimal antifungal regimens are used.16,18 A gradual rise in median fluconazole MICs in cryptococcal isolates collected during initial CM presentation have been reported in South Africa and Uganda.48,49 While this may lend support for a higher consolidation dose of 800mg daily in these settings, whether this is required across all patient groups and settings is contentious. Widespread fluconazole use may also perpetuate further rises in MICs.
Maintenance therapy with fluconazole 200 mg daily has been shown to be highly effective at preventing relapse, and is superior to weekly Amb-D and itraconazole capsules.50–52 Rarely, triazoles such as voriconazole53–60, posaconazole61–63 or isavuconazole64,65 are used as alternatives to fluconazole due to concerns of fluconazole resistance, drug toxicity or drug-drug interactions. Notably, none of the newer triazoles have been formally trialled in cryptococcosis and none are readily available in RLS.
A very low incidence of CM relapse is observed after a minimum of 1 year of antifungal therapy in PLHIV established on ART, who are virologically suppressed, and/ or have a CD4 count > 100 cells/mm3. 66–72
Adjunctive therapy:
Recent trials of adjunctive treatment in HIV-associated CM have all been shown to be ineffective, and in some cases harmful. These include high-dose dexamethasone,73 sertraline,74,75 and tamoxifen.76 The debate regarding adjunctive exogenous IFN-gamma remains unresolved. IFN-gamma has been studied in two randomised trials of HIV-associated CM, which suggest faster clearance of yeasts in the CSF77,78, but further studies are needed. There is no trial evidence supporting its use in non-HIV associated CM.
Recommendations:
A management algorithm is described in Figure 1 and key principles discussed in Table 1.
Figure 1:
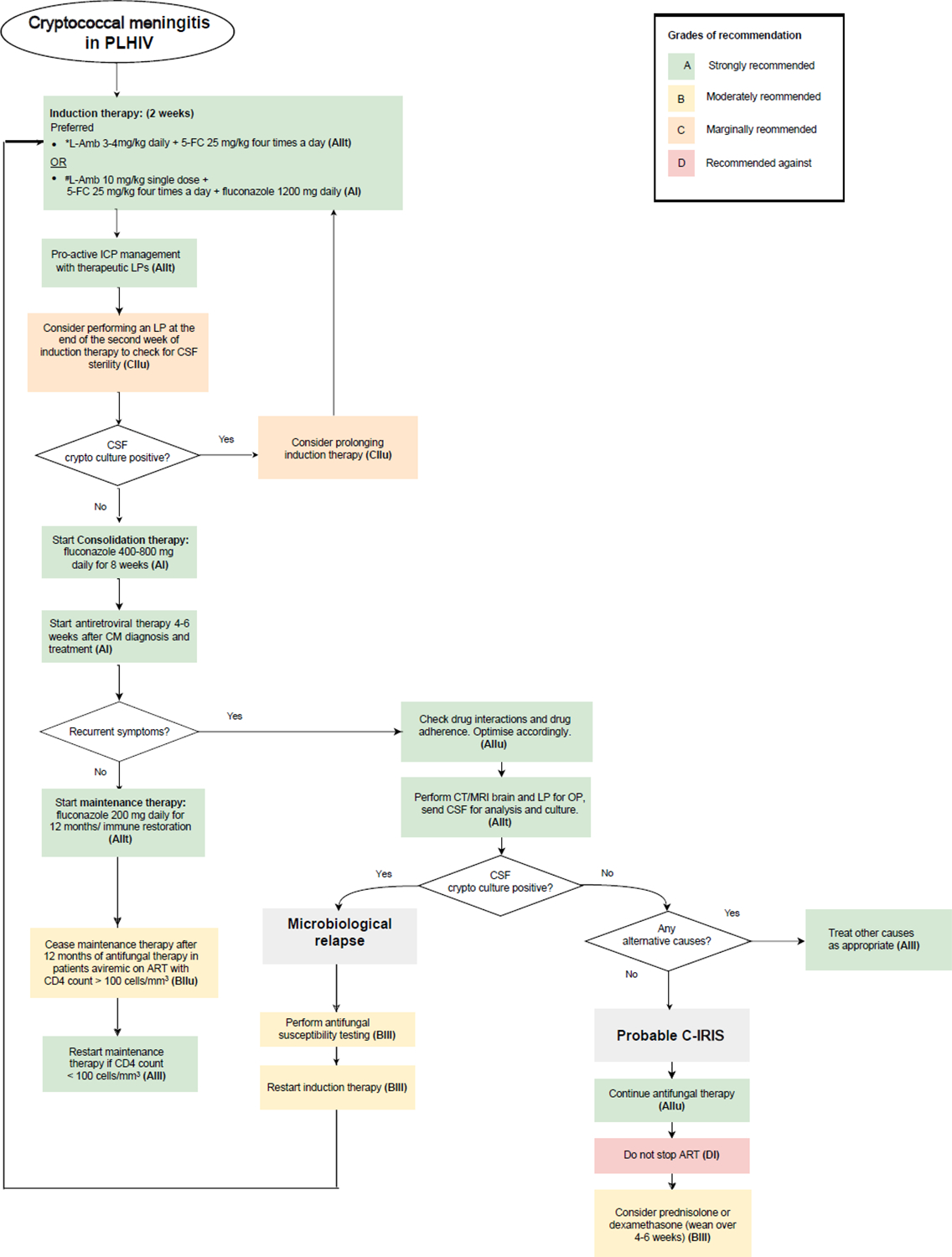
Management algorithm for cryptococcal meningitis/ cryptococcal meningoencephalitis (CM) in PLHIV
Table 1:
Ten principles of CM management
| Ten principles of CM management | |
|---|---|
| 1. |
Selectively screen, risk-stratify and investigate for cryptococcosis.
|
| 2. |
Provide best fungicidal induction regimen possible.
|
| 3. |
Monitor for and minimise drug toxicity.
|
| 4. |
Manage raised intracranial pressure (ICP).
|
| 5. |
Look for an underlying immunosuppressive state.
**
|
| 6. |
Provide and ensure adherence to consolidation and maintenance therapy.
|
| 7. |
Optimal commencement of ART in PLHIV.
|
| 8. |
Monitor for clinical relapse and investigate for causality.
|
| 9. |
In culture-positive (microbiological) persistence or relapse, evaluate for drug adherence, drug-drug interactions, and drug resistance.
|
| 10. |
Carefully exclude alternative diagnoses before attributing clinical relapse to C-IRIS.
|
Table 1 highlights the key principles in CM management, best read in context (see relevant sections in main text). While most evidence and recommendations are derived from CM in PLHIV populations, many of these principles are translatable to non-HIV settings.
Principles specific to CM in PLHIV are shaded in grey.
Unique considerations for non-HIV C. gattii CNS infection are underlined.
Exploring for an immunosuppressive state - particularly, but not limited to HIV infection - is important in the management of cryptococcosis.
Recommendations for CM treatment in PLHIV are based on availability of antifungal drugs; preferred and alternative strategies are offered (see Figure 2 and Table 2).
Figure 2:

- *(L-Amb 3–4 mg/kg/d + 5-FC 25 mg/kg 4x a day for 2 weeks) has not been compared to #(Single dose 10mg/kg L-Amb with 14d of 5-FC 25.kg 4x a day and Fluconazole 1200 mg daily). # has only been trialled in HIV-CM.
- &Polyene includes Amphotericin B formulations such as conventional deoxycholate Amphotericin B (Amb-D), Liposomal Amphotericin (L-Amb), Amphotericin B lipid complex (ABLC).
- ^Amb-D: 1 mg/kg showed earlier fungicidal activity than 0.7 mg/kg but some institutions use the lower dose due to toxicity concerns.
- @Fluconazole induction doses of up to 1200 mg daily have been trialled but caution is advised. Consider drug-drug interaction and liver toxicity.
- Grading of recommendation and level of evidence in bolded red letters.
- Recommendations by shading: green (Strong), yellow (Moderate), pink (marginal)
Table 2:
CM treatment in PLHIV, SOT and non-HIV-non-SOT
Antifungal treatment recommendations for CM in adult PLHIV, SOT, non-HIV-non-SOT populations.
| Patient population | First-line agent | SoR QoE | Alternative agents | SoR QoE | Other recommendations |
|---|---|---|---|---|---|
| PLHIV |
Where L-Amb is not available:
ABLC 5mg/kg daily plus 5-FC 25 mg/kg four times a day |
BIIt |
|
||
|
#Single dose L-Amb 10 mg/kg and
14 days of 5-FC 25 mg/kg four times a day and Fluconazole 1200 mg daily |
AI | ||||
|
OR Amb-D 1 mg/kg daily and 5-flucytosine 25 mg/kg four times a day for 1 week, followed by fluconazole 1200 mg daily for 1 week. |
|||||
|
OR: Amb-D 0.7–1 mg/kg daily plus fluconazole 800–1200 mg daily |
BI | ||||
| 5-FC 25 mg/kg four times a day and fluconazole 800mg-1200 mg daily | |||||
| Where only fluconazole is available: Fluconazole 800–1200 mg daily |
CI | ||||
|
CONSOLIDATION
(8w): %Fluconazole 400–800 mg daily (Note: 800 mg preferred in RLS) |
AI | Voriconazole 200 mg twice a day (with TDM) | BIlI | ||
| Posaconazole 300 mg daily (with TDM) | BIII | ||||
| Isavuconazole 200 mg daily | BIII | ||||
| Itraconazole 200 mg twice a day (with TDM) | CIIt | ||||
|
MAINTENANCE
(12m/ immune restoration):
Fluconazole 200 mg daily |
AIIt | Voriconazole 200 mg twice a day (with TDM) | BIII | ||
| Posaconazole 300 mg daily (with TDM) | BIII | ||||
| Isavuconazole 200 mg daily | BIII | ||||
| Itraconazole 200 mg twice a day (with TDM) | CIIt | ||||
| Solid Organ Transplant recipients |
INDUCTION
(minimum 2w): L-Amb 3–4 mg/kg daily plus 5-FC 25 mg/kg four times a day |
AIIt |
Where L-Amb is not available: ABLC 5 mg/kg daily plus 5-FC 25 mg/kg four times a day |
Bllt |
|
|
Where L-Amb nor ABLC are available: Amb-D 0.7–1.0 mg/kg daily plus 5-FC 25 mg/kg four times a day |
BIIt | ||||
|
Where amphotericin B-based therapies are not able to be used:
5-FC 25 mg/kg four times a day plus Fluconazole 800–1200 mg daily |
CIIt | ||||
|
CONSOLIDATION
(8w): Fluconazole 400–800 mg daily |
AIIt | Voriconazole 200 mg twice a day (with TDM) | BIII | ||
| Posaconazole 300 mg daily (with TDM) | BIII | ||||
| Isavuconazole 200 mg daily | BIII | ||||
| Itraconazole 4200 mg twice a day (with TDM) | CIIt | ||||
|
MAINTENANCE (12m):
Fluconazole 200 mg daily |
AIIt | Voriconazole 200 mg twice a day (with TDM) | BIII | ||
| Posaconazole 300 mg daily (with TDM) | BIII | ||||
| Isavuconazole 200 mg daily | BIII | ||||
| Itraconazole 200 mg twice a day (with TDM) | CIIt | ||||
| Non-HIV non-transplant |
Induction (minimum of 2w), Consolidation and Maintenance:
Same options as for CNS/CM treatment in SOT |
AIIt | Same as for CNS/CM treatment in SOT | Bllt |
|
Footnote:
preferred in resource-rich settings (RRS)
recommended in resource-limited settings (RLS)
consolidation dose of Fluconazole 800 mg daily is preferred in RLS
TDM: therapeutic drug monitoring
CM in Solid organ transplant (SOT) recipients
Evidence:
Cryptococcosis is the third most common invasive fungal infection in SOT recipients, with an incidence of 4.5%−33.8%26,28,29 and a significant mortality.12 SOT recipients comprise a third of non-HIV related cryptococcosis in the USA.79 The majority of cryptococcosis in SOT occurs late and is due to reactivation disease; however, acute donor-derived infections have been described.14,80,81
Anti-rejection drugs vary in their degree of immunosuppression and heart and small bowel transplant recipients are at the highest CM risk.82 CNS and pulmonary cryptococcosis predominate but unusual manifestations including cutaneous disease83,84 and pericarditis85 have been reported. Notably, blood CrAg may be negative in SOT recipients with cryptococcosis, particularly those with single pulmonary nodules or in lung transplant recipients.86
There are no randomised treatment trials targeted specifically at SOT recipients hence recommendations are extrapolated from experience in PLHIV. The use of lipid-formulations in SOTs with CNS cryptococcosis was independently associated with reduced mortality compared with Amb-D.87 The AMBITION-cm regimen has not been studied in non-HIV patients, and experience with high dose fluconazole (with its ensuant potential toxicity and drug-drug interactions) in this group is lacking. A precipitous reduction in dosing of immunosuppressants, particularly calcineurin inhibitors, may lead to IRIS.88
Recommendations:
See Table 2.
CM in Non-HIV, Non-Transplant Patients
Evidence
The non-HIV, non-SOT group is heterogeneous, ranging from apparently normal hosts to those with haematological malignancies or liver cirrhosis. There is no single therapeutic regimen or duration that meets all patients’ needs but principally, this mirrors CM treatment in RRS with L-Amb and 5-flucytosine induction. Induction therapy may be extended in those with persistently positive CSF cultures and/or persistent symptoms at two weeks. Recently, the combination of L-Amb and 5-flucytosine was shown to have a low acute mortality of 6% in a nationwide observational study of non-HIV-associated CM in Japan.89
Recommendations
See Table 2.
Pulmonary cryptococcosis
Evidence
Case series and clinical experience suggest that patients with cryptococcaemia, evidence of CNS involvement, blood CrAg titres ≥1:512 by latex agglutination (or 10-fold higher by LFA90), or severe pulmonary disease, should be treated as CM.33,35,91,92 Patients with mild isolated pulmonary disease without cryptococcoma have been previously successfully treated with fluconazole monotherapy of 400 mg daily.91,93,94 Some clinicians consider watchful-waiting and elect not to treat asymptomatic immunocompetent persons who incidentally culture any Cryptococcus spp., in their sputum, and exhibit no radiological features of pulmonary cryptococcosis, as they consider this as airway colonisation.95 Criteria for distinguishing colonisation from infection is uncertain. There are no randomised treatment studies in pulmonary cryptococcosis.
Recommendations
Stratify treatment by disease severity and presence of pulmonary cryptococcoma (s17)s).
Non-pulmonary-Non-CNS Disease
Evidence:
Cryptococcosis can affect any organ following haematogenous dissemination. Clinical presentation of non-CNS/non-pulmonary disease without fungemia is rare, but possible. The absence of documented fungemia does not exclude dissemination. Barring direct inoculation into the skin following trauma, extrapulmonary disease is by definition “disseminated disease” and generally requires consideration for aggressive induction therapy. There are no clinical treatment trials for non-pulmonary, non-CNS cryptococcosis.
Importantly, visual changes noted in CM are frequently related to raised ICP and do not necessarily indicate direct eye involvement. Ocular cryptococcosis can occur96,97 but is unusual and requires formal ophthalmological documentation and management. Isolated skeletal osteomyelitis is rare and often requires a combined surgical and medical approach.98–100 Skin lesions may be polymorphic.
Recommendations:
- Cryptococcaemia:
- (AIIu) Treat as for CNS disease.
- Primary cutaneous (skin) cryptococcosis attributed to direct inoculation without evidence of dissemination:
- (AIII) Fluconazole 400 mg daily for 3–6 months or until cicatrisation.
- All other non-CNS/ non-pulmonary disseminated disease:
- (BIIu) Treat as for CNS disease.
(BIIu) Cryptococcal eye disease should be managed in collaboration with an ophthalmologist.
Specific Management Issues
Raised intracranial pressure (ICP)
Evidence:
Increased ICP has been associated with high burden of cryptococci leading to both acute and chronic symptoms and signs (e.g., visual and hearing loss) and decreased short-term survival. Clinical experience has shown that CSF outflow obstruction can be improved by removal of CSF; observational studies suggested that scheduled therapeutic LPs result in significant improvement in survival, regardless of OP.101,102 For prolonged control of acute increased ICP, use of lumbar drains in cases without hydrocephaly or ventriculostomies in cases with hydrocephaly may be required.103–105 Medical therapies including acetazolamide, mannitol, and corticosteroids may be detrimental.106,107
Recommendations:
(AIIu) Opening pressure should be measured at every LP in patients with CM.
(AIII) Perform a CT brain (if CNS imaging not previously done) to exclude CNS outflow obstruction.
(Allt) Acute symptomatic elevation of the ICP (≥ 20 cm of CSF) should be managed by daily therapeutic LPs (i.e., removal of sufficient CSF (usually around 20–30 ml) to reduce the pressure to 50% of OP and/or to a normal pressure of ≤20 cm of CSF (documented as a closing pressure).
(BIIu) Perform a scheduled therapeutic LP around 48–72 hours of initial LP and/or 7 days, regardless of initial OP.
(Allt) Persistent raised symptomatic ICP despite therapeutic LPs should be managed by surgical decompression via temporary lumbar drainage, shunting, or ventriculostomy depending on local expertise, and resources.
(BIII) Consider ventriculoperitoneal (preferential) and lumboperitoneal shunts (alternative) to control both acute and chronic hydrocephalus if temporary measures are not successful. Ideally, insert shunts after institution of effective antifungal therapy.
Timing of antiretroviral therapy (ART) commencement
Evidence:
The optimal time to commence ART for HIV infection during cryptococcosis remains controversial. Four randomised trials3,108–110 to determine optimal timing of ART initiation in HIV-CM co-infection have been conducted in RLS settings, using induction regimens which are not currently preferred, including fluconazole (800 mg daily) monotherapy108, Amb-D 0.7 mg/kg daily109, and Amb-D 0.7–1mg/kg daily and fluconazole 800 mg daily for 2 weeks. These data seem to suggest that initiating ART within 2 weeks of CM presentation is too early in the setting of suboptimal antifungal therapy, and that delaying ART initiation for 4–6 weeks reduces the incidence of C-IRIS and death. CSF sterility prior to ART commencement may be another factor.45 A retrospective analysis of combined cohorts in RRS did not appreciate higher mortality in those receiving early ART in the first two weeks of antifungal therapy compared to those with delayed therapy.111 In all, early ART in RRS, will need careful justification and close monitoring; further randomised studies may be helpful.112
There are no studies for timing of ART initiation in other forms of cryptococcosis, those with cryptococcal antigenemia or those recommencing ART after a period of interruption. Early concerns that potent integrase inhibitors pose an increased risk of C-IRIS have been disproven.113 Whether those presenting with CM within 2 weeks of starting ART require withholding of ART remains uncertain.114–116
Recommendation:
(DI) Immediate or very early commencement of ART is not recommended.
- Depending on antifungal induction therapy availability:
- Sub-optimal/limited access: (AI) Delay ART for 4–6 weeks.
- Optimal and in RRS: (BIIu) Consider further individualisation taking into consideration resolution of symptoms and signs of CM and ICP (including normalisation of OP), attainment of CSF cryptococcal sterility, successful identification and management of concurrent co-infections and other AIDS-defining illnesses, the patient’s readiness for ART and local experience of CM and C-IRIS management. (Usual range is 4–6 weeks).
(CIIt) Where possible, ensure CSF is cryptococcal culture negative prior to ART commencement.
(BIII) For ART-experienced persons who develop CM and may need to switch to second-line ART or recommence ART, a delay of 4–6 weeks is recommended.
(CIII) Pending further studies, consider withholding ART and restarting at 4–6 weeks in those presenting with CM within 2 weeks of starting ART.
(BIII) Patients with isolated pulmonary cryptococcosis or those with asymptomatic cryptococcal antigenemia may commence ART earlier (e.g., 2 weeks).
Resistance to antifungals
Evidence:
Developing secondary resistance to 5-flucytosine is common when given as monotherapy, necessitating its use with a partner drug in cryptococcosis. Acquired resistance to polyenes such as Amb-D is rare, but the emergence of fluconazole resistance is concerning.48,49,117 Fluconazole monotherapy as induction therapy has been associated with secondary resistance.118–121
There are presently no clinical MIC breakpoints for fluconazole against Cryptococcus species and a lack of convincing data to suggest that high MICs imply worse outcomes. Interpretation of epidemiological cut-off values (ECVs), using the CLSI method for fluconazole requires accurate species identification. Based on CLSI, the ECV for C. neoformans VNI is 8 ug/mL, and 16 and 32 ug/mL, for C gattii VGI and C. deuterogattii VGII, respectively.122 In principle, a higher than 2-fold increase in MIC during treatment may suggest development of resistance, and the need for closer clinical monitoring. There are no EUCAST ECOFFs available for fluconazole.
Recommendation:
- For those with concerns of fluconazole-resistance or emerging fluconazole-resistance:
- (BIII) Consider a longer course of treatment with Amb-D (1 mg/kg daily) or higher dose of L-Amb (3–6 mg/kg daily) together with 5-flucytosine (about 4 weeks) as induction therapy.
- (BIII) Consider Amb-D 1 mg/kg weekly or L-Amb 3–6 mg/kg weekly as consolidation/maintenance therapy. Consider daily voriconazole, posaconazole, isavuconazole or itraconazole for isolates without evidence of pan-azole resistance. as guided by AFST.
- (CIII) Where Amb-D or L-Amb are not available, adding 5-flucytosine to high-dose fluconazole (1200 mg daily) may be considered.
Cryptococcal persistence, clinical relapse, and culture-positive (microbiological) relapse
Evidence:
Distinguishing clinical relapse from persistent cryptococcal infection is challenging. Clinical relapse may be due to a microbiological relapse, C-IRIS, raised ICP (whether or not related to C-IRIS) or other infective and non-infective (CNS and non-CNS) causes (Figure 1). Cryptococcal antigen persists in the CSF and blood thus has little clinical utility in distinguishing clinical responders from non-responders.123 Most cases of culture-positive (microbiological) relapse occur early and result from inadequate or suboptimal induction therapy or early discontinuation of consolidation or maintenance therapy.
Recommendations:
(AIIt): Think broadly and investigate thoroughly for causality (CNS and non-CNS; infective and non-infective) in cases of apparent clinical relapse. Investigations should include CT/MRI brain, LP for OP and CSF analyses including microscopy and culture.
(AIIu) Review adherence to antifungal therapy, ART/ immunosuppressants and other medications and consider drug-drug interactions. Perform therapeutic drug monitoring (TDM) if applicable. Optimise control of underlying disease(s).
(CIII) Consider escalating antifungal therapy while awaiting CSF results (and de-escalate if culture-negative)
(Dllu) The use of follow-up blood or CSF CrAg (including monitoring of titres) for clinical decision-making is discouraged.
(Dllu) Do not escalate antifungal therapy for persistent blood antigenemia, persistently positive CSF CrAg, visible cryptococci in CSF (without culture positivity), nor abnormal CSF microscopy or biochemistry. These are not necessarily indicators of microbiological failure.
- For culture-positive (microbiological) persistent or relapsed infection (Figure 1):
- (BIII) AFST should be performed concurrently on all initial and relapse isolates (if stored and available). An increase in fluconazole MIC of >2 dilutions is considered as concerning for the potential development of drug resistance.
- (BIII) Consider re-induction with a more optimal regimen (guided by AFST).
Cryptococcosis-associated Immune Reconstitution Inflammatory Syndrome (C-IRIS)
Evidence:
IRIS has been classically described in PLHIV between 2 weeks and 3 months after commencement of ART, where patients develop exaggerated symptoms and signs and/or atypical inflammation, reminiscent of a paradoxical recurrence124,125, but can also occur in the setting of immune recovery or withdrawal of immunosuppressants. It has also been observed in seemingly immunocompetent individuals including in C. gattii infections as a post-infectious inflammatory immune response syndrome (PIIRS).88,126 There is no diagnostic biomarker for C-IRIS, thus this is a clinical diagnosis of exclusion (Figure 1).
There have been no therapeutic trials in C-IRIS. Management strategies include therapeutic LP, and symptomatic therapies. In severe C-IRIS, corticosteroids are commonly used to dampen inflammation although their efficacy has not been rigorously examined in clinical trials. In steroid-refractory C-IRIS, there are case reports on the use of TNF-alpha blockers such as adalimumab127–130 or thalidomide131–133 with mixed success. Corticosteroids may also be beneficial in PIIRS.134
Recommendation:
(Allt) For patients with suspected paradoxical C-IRIS, carefully exclude recurrent cryptococcal disease or new infective or non-infective conditions, prior to attributing symptoms and signs to C-IRIS. Perform an MRI brain and LP to measure OP and obtain CSF for microbiological and biochemical analyses.
(AIIu) Treatment of C-IRIS should include therapeutic LP, symptomatic therapy such as analgesia, antiemetics and antiepileptics where appropriate.
(AIII) Continue current antifungal therapy.
(Blll) High-dose prednisone/prednisolone (usually 0.5–1.0 mg/kg daily) or dexamethasone (usually 0.2–0.3 mg/kg daily), weaned over 4–6 weeks may be considered in those with persistent symptoms, unresponsive to therapeutic LPs. Rarely a second steroid course with taper is needed.
(DIII) Do not stop ART.
(BIII) Cases of steroid-refractory or recurrent C-IRIS be discussed with experts in the field.
(CIIu) Steroids may be considered for PIIRS.
Cryptococcus gattii
Evidence:
About 50–70% of C. gattii infection occur in putatively immunocompetent hosts135–137 compared with 2–30% in PLHIV.138–142 Autoantibodies to GM-CSF and idiopathic CD4 lymphopenia are reported risk factors.135,143–145 Notably, not all commercial LFAs are able to detect C. gattii disease.146 Antifungal agents used for treatment are the same as for C. neoformans.30,32,139 However, 4–6 weeks of induction therapy may be required in some cases of non-HIV-associated meningitis with C. gattii.147
Recommendations:
- C. gattii CNS disease
- (AIII) Treat as for C. neoformans CNS infection.
- In non-HIV patients: (BIII) Consider extending induction therapy to 4–6 weeks.
- (AIII) Early CSF shunting is indicated for obstructive chronic hydrocephalus.
C. gattii lung disease (see pulmonary cryptococcosis, pulmonary cryptococcoma and s17).
Cryptococcomas
Evidence:
Cryptococcomas occur predominantly in the lungs and brain and are more frequent in C. gattii infection.138,148 CNS cryptococcomas can manifest as neurological deficits and/or raised ICP,138 which requires urgent management. Corticosteroids and surgical resection may be of value.147,149,150 Radiological lesions can persist indefinitely despite clinical and microbiological cure.32,151
Recommendations:
All cryptococcomas
(AIII) Perform a biopsy or aspirate to exclude a secondary pathogen or an underlying tumour in non-responding cryptococcomas (particularly in immunosuppressed patients).
(BIII) Consider surgical resection for accessible brain lesions >3 cm, lesions at risk of compressing critical structures, or large lesions not responding to therapy.
(DIII) During follow-up, do not prolong or escalate therapy for persistent radiological findings in the absence of new or worsening symptoms or signs.
CNS cryptococcoma
(BIII) Consider prolonging CNS antifungal induction therapy to 4–6 weeks.
(BIII) Consider corticosteroids for large cryptococcomas with surrounding mass effect, or, where neurological symptoms and cerebral imaging signs worsen despite a good microbiological response.
Lung cryptococcoma
see s17
Non-neoformans and non-gattii strains of Cryptococcus
Evidence:
There are individual case reports and small case series of non-neoformans/non-gattii Cryptococcus infections, predominantly in immunosuppressed patients. Papiliotrema (previously Cryptococcus) laurentii152 and Naganishia albida (previously Cryptococcus albidus)153 account for about 80% of the invasive infections in this group and usually involve skin, lungs, bloodstream, or CNS.154 Colonisation, especially of the skin, respiratory and gastrointestinal tracts must be distinguished from true disease. In some cases, the laboratory may misidentify another yeast as P. laurentii or N. albida based on non-definitive commercial identification methods.155 Elevated MICs against 5-flucytosine, fluconazole and other azoles for some isolates have been documented but are of uncertain clinical significance.156,157
Recommendations:
(AIII) As non-neoformans/non-gattii Cryptococcus species are rarely pathogenic, careful assessment of the laboratory identification and clinical context is required, to ascertain clinical significance.
(CIII) For CNS or disseminated disease: Treat as for C. neoformans CNS infection.
Pregnancy
Evidence:
The majority of cases of cryptococcosis in pregnancy occur in the third trimester or postpartum.158,159 Maternal mortality from disseminated cryptococcosis is approximately 25%; and <50% carry their pregnancy to term.159 Extensive clinical experience suggests that Amb-D and L-Amb are relatively safe during pregnancy (Category B drug), and thus are the cornerstone of treatment.159,160 5-Flucytosine is rated by the USA FDA as a Category C drug because of its direct effects on RNA/DNA metabolism. Fluconazole is a Category D drug due to its increased risk of musculoskeletal malformations, tetralogy of Fallot and spontaneous abortions.161–165
Recommendations:
(AIII) Use L-Amb or Amb-D in induction, consolidation, and maintenance therapy, and for treatment of isolated cryptococcal antigenemia.
(DII) Avoid use of 5-flucytosine and fluconazole in pregnancy, particularly in the first trimester; their use in the second and third trimester requires careful individualised risk-benefit assessment.
(BIII) Fluconazole may be used after delivery despite its excretion into breast milk.
(CIII) Apply clinical judgement when considering initiation of antifungal therapy and duration of therapy, factoring in trimester of pregnancy and severity of illness.
(Clll) For asymptomatic CrAg in pregnancy, consider intermittent polyene therapy especially in the first trimester.
Paediatrics
Evidence:
There is a clear need for paediatric-specific studies in cryptococcosis. CNS disease seems to predominate in paediatrics but non-CNS disease is likely under-reported.166–172 Clinical efficacy trials and studies to validate diagnostics tests and therapies for cryptococcosis in children are lacking. Recommendations are extrapolated from studies in adult populations. Dosing of antifungal agents needs particular attention for the paediatric patient.
Recommendations:
- Treatment of CNS or disseminated disease:
- Induction: (AIIt) Amb-D 1 mg/kg daily or L-Amb 3–4 mg/kg daily plus 5-flucytosine (100–150 mg/kg daily in 4 divided doses) for 2 weeks.
- Consolidation: (AIIt) Fluconazole 12 mg/kg (maximum 800 mg) daily for 8 weeks.
- Maintenance: (AIIt) for PLHIV and immunocompromised; (BIIt) for immunocompetent. Fluconazole 6 mg/kg daily (maximum 800 mg) for 6–12 months.
Treatment of severe isolated pulmonary diseases: (AIII) Treat as for CNS disease.
Treatment of mild isolated pulmonary disease: (AIII) Fluconazole 12 mg/kg daily (maximum 800 mg) for 6–12 months.
(AIII) Screening is recommended for children living with HIV in high disease prevalence areas at ages ≥10 years.
Conclusions
Cryptococcosis and its management remain complex and challenging. Adherence to clinical practice guidelines can improve outcomes44,173 While there has been significant development of evidence from randomised controlled trials over the past 20 years, there remain significant unmet needs (see s19). This is particularly critical in RLS where the burden of disease is high and access to antifungal therapy remains inadequate. Equally, more needs to be learnt in RRS where host risk profiles are changing and an increasing array of presentations of cryptococcosis are being recognised, necessitating more nuanced and individualised treatment plans.
Supplementary Material
Figure 3: Recommended first-line antifungal therapy by cryptococcosis syndrome.
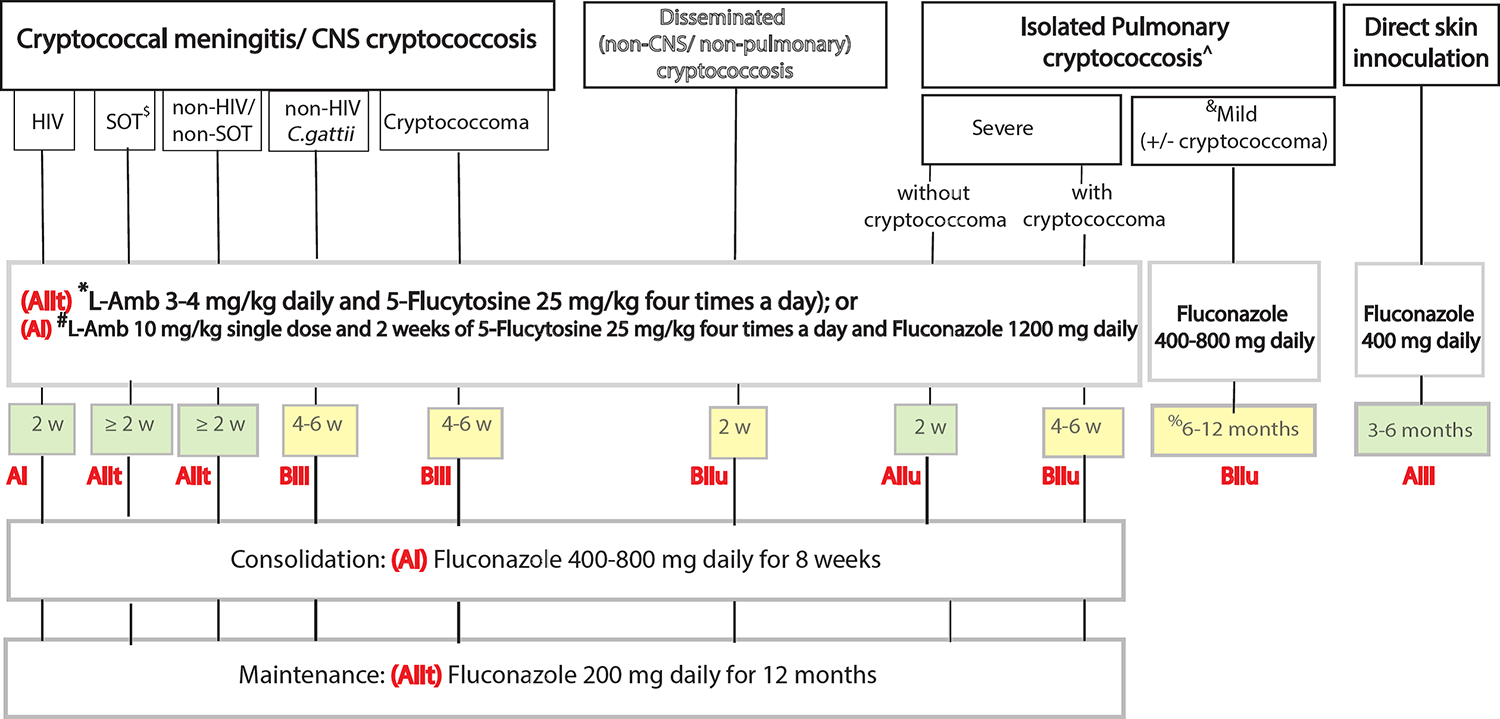
This simplified figure summarises the recommendations for first-line antifungal therapy where optimal antifungal options are available. The grading of recommendation and level of evidence is in red, with the duration of the therapy shaded by strength of recommendation. Isolated pulmonary cryptococcosis may be divided into severe or mild, and with or without pulmonary cryptococcoma. Mild pulmonary cryptococcosis is defined as asymptomatic or mildly symptomatic patients, or those with a solitary small nodule (< 2 cm) while severe pulmonary cryptococcosis includes those with multiple lesions, large lesions (≥2 cm), lobar consolidation, cavitation, multi-lobar involvement, and those with hypoxaemia.
In HIV-associated CM/ CNS cryptococcosis we recommend L-Amb 3–4 mg/kg daily and 5-Flucytosine 25 mg/kg four times a day OR single-dose L-Amb 10 mg/kg and 14 days of 5-Flucytosine 25 mg/kg four times a day and Fluconazole 1200 mg daily, for a minimum of 2 weeks. Importantly, the single-dose L-Amb 10 mg/kg and 14 days of 5-Flucytosine 25 mg/kg four times a day and Fluconazole 1200 mg daily has only been trialled in HIV-CM. There is currently no data of its application in non-HIV-CM settings. See Figure 2 for treatment cascade in HIV-CM by antifungal availability.
In non-HIV associated CM/ CNS cryptococcosis, disseminated cryptococcosis and severe isolated pulmonary cryptococcosis, we recommend L-Amb 3–4 mg/kg daily and 5-Flucytosine 25 mg/kg four times a day for 2 weeks or more. Extensions in the duration of induction therapy may be considered in SOT recipients, non-HIV-non-SOT patients, and in non-HIV patients with C. gattii infection (4–6 weeks), those with CNS cryptococcomas (4–6 weeks) or severe isolated pulmonary cryptococcosis with lung cryptococcomas (4–6 weeks).
All CNS-based induction therapy should be followed by 8 weeks of fluconazole 400–800 mg daily as consolidation therapy, then fluconazole 200 mg daily as maintenance therapy for 12 months or until immune restoration. The consolidation dose for HIV-CM in resource limited settings (RLS) is currently commonly prescribed as fluconazole 800 mg daily, and the 400 mg daily dose is used in other non-RLS settings, and other non-CM cryptococcosis syndromes. In settings where antifungal therapy availability is limited, refer to Figure 2 for alternatives.
Patients with mild isolated pulmonary cryptococcosis (regardless of immune state, presence of cryptococcoma or infecting strain) may be treated with fluconazole 400–800 mg daily for 6–12 months (note: a shorter duration of 3 months may be considered in those who are immunocompetent). If the presence of Cryptococcus spp. in respiratory specimens is deemed as airway colonisation after careful evaluation and no treatment is elected, regular follow-up is recommended especially in the setting of future immunosuppression (see Table 6). In the rare case of skin cryptococcosis from direct skin inoculation, fluconazole 400 mg daily for 3–6 months is recommended. See Table 6 for details on pulmonary cryptococcosis and pulmonary cryptococcoma.
Footnote:
*L-Amb 3–4 mg/kg daily and 5-Flucytosine 25 mg/kg four times a day has not been directly compared against #L-Amb 10 mg/kg single dose and 2 weeks of 5-Flucytosine 25 mg/kg four times a day and Fluconazole 1200 mg daily.
*L-Amb 3–4 mg/kg daily and 5-Flucytosine 25 mg/kg four times a day is strongly preferred in CM/ CNS cryptococcosis in SOT and non-HIV non-SOT settings, disseminated cryptococcosis and severe pulmonary cryptococcosis.
#L-Amb 10 mg/kg single dose and 2 weeks of 5-Flucytosine 25 mg/kg four times a day and Fluconazole 1200 mg daily. This regimen has only been trialled in HIV-CM. There is no supporting data of its use in SOT, or non-HIV non-SOT patients, and in other cryptococcosis syndromes.
$: solid organ transplant (SOT) recipients. *L-Amb 3–4 mg/kg daily and 5-Flucytosine 25 mg/kg four times a day is strongly preferred in SOT patients.
^: Isolated C. neoformans or C. gattii pulmonary cryptococcosis.
Mild pulmonary cryptococcosis (with or without cryptococcoma): asymptomatic or mildly symptomatic patients or with a solitary small nodule (< 2 cm).
Severe pulmonary cryptococcosis: multiple lesions, large lesions (≥2 cm), lobar consolidation, cavitation, multi-lobar involvement, hypoxaemic.
%: May consider a shorter duration (e.g., 3 months) in immunocompetent individuals with mild isolated pulmonary cryptococcosis.
&: If the presence of Cryptococcus spp. in respiratory specimens is deemed as airway colonisation after careful evaluation and no treatment is elected, regular follow-up is recommended especially in the setting of future immunosuppression.
W: weeks
Grading of recommendation and level of evidence in bolded red letters
Recommendations by shading: green (strongly recommended), yellow (moderately recommend)
Key points.
Delineating the cryptococcosis clinical syndrome involved (central nervous system CNS vs. non-CNS, non-pulmonary disseminated disease vs. isolated pulmonary vs. direct skin inoculation) guides choice of antifungal treatment and duration (see Figure 3).
L-Amb 3–4mg/kg daily and 5-flucytosine 25 mg/kg four times a day, remains the most optimal induction therapy option for cryptococcal meningitis (CM), disseminated cryptococcosis and severe isolated pulmonary cryptococcosis in all hosts in resource-rich settings.
In resource-limited settings, patients with HIV-associated CM are best treated with L-Amb 10 mg/kg as a single-dose, with 14 days of 5-flucytosine 25 mg/kg four times a day and Fluconazole 1200 mg daily as induction therapy. This induction therapy has not been trialled in non-HIV-associated CM or other non-CNS cryptococcosis syndromes.
Optimise outcomes by providing the most effective antifungal therapy while preventing, monitoring, and managing potential toxicity. Do not stop or switch to an inferior regimen too early or unnecessarily.
Expect and monitor for clinical relapse, think broadly, and investigate thoroughly for causality. Review adherence to antifungal therapy and consider drug-drug interactions. During treatment follow-up, do not escalate antifungal therapy for persistent blood antigenemia (serum CrAg), persistently positive CSF CrAg, visible cryptococci in CSF (without culture positivity), nor abnormal CSF microscopy or biochemistry. These are not necessarily indicators of microbiological failure.
Adapt and adopt these ECMM global guidelines to suit local practices, while constantly advocating for better antifungal access, scrutinising new trial data, and reviewing local data to improve patient outcomes.
Acknowledgements:
CCC was a recipient of the Australian National Health and Medical Research Council Early Career Fellowship (APP 1092160).
OAC is supported by the German Federal Ministry of Research and Education; is funded by the Deutsche Forschungsgemeinschaft under Germany’s Excellence Strategy (Cologne Cluster of Excellence on Cellular Stress Responses in Aging-associated Diseases, EXC 2030—390661388).
NPG reports grants from the National Institutes of Health, US Center for Disease Control and Prevention, Bill & Melinda Gates Foundation, UK Medical Research Council, and National Health Laboratory Service Research Trust.
This work was supported in part by the Division of Intramural Research of NIAID, NIH.
We thank the many reviewers from the many international societies who provided helpful and constructive advice on the guidelines during the public consultation process and thank Andreas Mazzella for assistance with Fig.1.
Declaration of interests:
AA reports grants from ANR, serving as a consultant to Gilead Sciences, receiving speaking honoraria from Gilead Sciences and PR edition, travel support from Gilead sciences and Pfizer, and patents with the Institut Pasteur, outside the submitted work
J-WA reports grants or contracts from World Health Organization (FPPL) and receipt of equipment and materials from the Westmead Hospital Foundation, outside the submitted work.
JB reports support from the NHMRC Australia, and receipt of honoraria from Gilead, outside the submitted work.
TAB reports personal research fellowship from Gilead Sciences, investigator-led research grant from Pfizer, lecture honoraria and participation in Advisory Boards for Gilead Sciences, Mundipharma and Pfizer, participation in the Trial Steering Committee for a phase II trial of inhaled opelconazole (Pulmocide), outside the submitted work.
FC reports speaker honoraria from and being part of an advisory board for Pfizer and United Medical, outside the submitted work.
CCC reports receipt of an Early Career Fellowship from the Australasian National Health and Medical Research Foundation, receipt of a speaker travel support for IDweek 2024 and being a principal investigator in an early phase clinical trial unit, outside the submitted work.
MC reports grants from Cidara, F2G, Pfizer and Janssen; receipt of honoraria from Pfizerm MSD and Gilead; and travel support from Pfizer, outside the submitted work.
SC-AC reports untied educational grants from Merck Sharp and Dohme Australia and F2G and is on the antifungal advisory boards of Merck Sharp and Dohme Australia, Gilead Sciences, and F2G, outside the submitted work.
OAC reports grants or contracts from BMBF, Cidara, EU-DG RTD (101037867), F2G, Gilead, MedPace, MSD, Mundipharma, Octapharma, Pfizer, Scynexis; Consulting fees from Abbvie, AiCuris, Biocon, Cidara, Gilead, IQVIA, Janssen, Matinas, MedPace, Menarini, Moderna, Molecular Partners, MSG-ERC, Noxxon, Octapharma, Pfizer, PSI, Scynexis, Seres; Honoraria for lectures from Abbott, Abbvie, Al-Jazeera Pharmaceuticals/Hikma, Gilead, Grupo Biotoscana/United Medical/Knight, MedScape, MedUpdate, Merck/MSD, Noscendo, Pfizer, Shionogi, streamedup!; Payment for expert testimony from Cidara; Participation on a Data Safety Monitoring Board or Advisory Board from Boston Strategic Partners, Cidara, IQVIA, Janssen, MedPace, PSI, Pulmocide, Shionogi, The Prime Meridian Group; A patent at the German Patent and Trade Mark Office (DE 10 2021 113 007.7); Stocks from CoRe Consulting, EasyRadiology; Other interests from Wiley. All outside the submitted work.
J-PG reports speaker honoraria from Gilead, MundiPharma and Pfizer, outside the submitted work.
NPG reports grants from National Institutes of Health (USA), National Institute of Health and Care Research (UK), Medical Research Council (UK), Centers for Disease Control and Prevention (USA), National Health Laboratory Service Research Trust (South Africa); participation in the ACACIA trial as part of the data safety monitoring board a, project committee of DREAMM and project advisory committee for 5FC Crypto and leadership roles in the Federation of Infectious Diseases Societies of Southern Africa., outside the submitted work.
AHG reports grants from Gilead Sciences; personal fees from Amplyx, Astellas, Basilea, F2G, Gilead Sciences, Merck Sharp and Dohme, Mundipharma, Pfizer and Scynexis; speaker honoraria from Gilead Sciences and Merck, Sharp and Dohme, and participation in an advisory board for Astellas, Mundipharma, Partner Therapeutics and Pfizer, outside the submitted work.
FH reports grants from Health Holland and European Society for Clinical Microbiology and Infectious Diseases (ESCMID) and leadership roles as treasurer of the Netherlands Society for Medical Mycology (NVMy), Chair of the Division Microbial Genomics of the Royal Netherlands Society for Microbiology (KNVM-DMG), Vice-President International Society for Human and Animal Mycology (ISHAM) And receipt of evaluation kits from Bruker and Pathonostics, outside the submitted work.
TH reports receipt of an investigator award from Gilead Sciences, honoraria from Pfizer and Gilead Sciences, and participation in a data safety monitoring board or advisory board for Viamet and F2G.
MH reports receipt of an EDCTP grant outside the submitted work.
JNJ reports support from the National Institute for Health Research; grants from EDCTO, joint global health trials (welcome Trust/ MRC/ UKAID) and Centers for Disease Control and Prevention; speaker fees from Gilead Sciences; participation on a data safety and monitoring board for HARVET, ARTIST, CASTLE and ACACIA trials.
GJ reports travel support to attend a meeting at ISHAM, outside the submitted work.
NK was a speaker for Astellas, Gilead Sciences, Merck/MSD, and Pfizer and an adviser for Gilead Sciences, Merck/MSD, and Pfizer, outside the submitted work.
MSL reports support from the Division of Intramural Research, NIAID, NIH, outside the submitted work.
OL reports receipt of consulting fees and honoraria from Gilead Science, and patents with INSERM APHP, outside the submitted work.
OMM reports travel support for ISHAM meeting in India, and being the country ambassador for Kenya for ISHAM, outside the submitted work.
BM reports being chair of the Australia And New Zealand Paediatric Infectious Diseases Group.
DM reports leadership role in the Crypto Meningitis Advocacy Group.
RO reports receiving research and educational grant funding Gilead Sciences, CDC Atlanta and Pfizer Specialties, and travel support from the CDC foundation, outside the submitted work.
PP reports grants from Mayne, Astellas, Scynexis, and Cidara; receipt of consulting fees from F2G and Cidara, outside the submitted the work.
AP reports speaker honoraria for Gilead Science, Pfizer India and Intas pharmaceutical, outside the submitted work.
JP reports grants from National Institute of Health, Appili and Sfunga; royalties from Up-To-Date; and participation on a data safety monitoring board or advisory board from Pulmocide, Effect Trial and Imprint Trial, outside the submitted work.
FQ-T reports receipt of speaker honoraria from Pfizer and United Medical, travel support and laboratory diagnostic kits from IMMY, and leadership roles in Infocus LATAM, outside the submitted work.
JS-G reports speaker honoraria from Gilead and Pfizer, and is on an advisory committee for Pfizer, outside the submitted work.
AS reports grants from Astellas and receiving consulting fees from Scynexis, outside the submitted work.
RS has received speaker honoraria from Pfizer, outside the submitted work and reports being chair of Young ECMM. TT reports receipt of honoraria from Pfizer, MSD, Asahikasei pharma and Sumitomo pharma, outside the submitted work.
AW reports a grant from UKRI, receipt of consultant fees from Gilead and MundiPharma; speaker fees from F2G and Gilead; participation as a data safety monitoring board member for the RECOVERY trial, outside the submitted work.
The other authors declared no conflicts of interest.
Footnotes
- Supplementary Tables: 6
- Supplementary Panels: 6
- Supplementary Figures: 2
- Supplementary Text
- References
Contributor Information
Dr. Christina C. Chang, Department of Infectious Diseases, Alfred Hospital, Melbourne, Australia; Department of Infectious Diseases, Central Clinical School, Monash University, Melbourne, Australia; Centre for the AIDS Programme of Research in South Africa, Durban, South Africa;.
Prof. Thomas S Harrison, Institute of Infection and Immunity, St George’s University London, London, UK.; Clinical Academic Group in Infection and Immunity, St George’s University Hospitals NHS Foundation Trust, London, UK; MRC Centre for Medical Mycology, University of Exeter, Exeter, UK..
Prof. Tihana A Bicanic, Institute of Infection & Immunity, St George’s University London, London SW17 0RE, UK. Clinical Academic Group in Infection & Immunity, St George’s University Hospitals NHS Trust, London SW17 0QT, UK. The MRC Centre of Medical Mycology, University of Exeter, Stocker Road, Exeter EX4 4QD, UK..
Prof. Methee Chayakulkeeree, Division of Infectious Diseases and Tropical Medicine, Department of Medicine, Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand;.
Prof. Tania C Sorrell, Sydney Infectious Diseases Institute (Sydney ID), University of Sydney, NSW 2006; Dept Infectious Diseases, Westmead Hospital, Westmead, NSW 2145, Australia..
Prof. Adilia Warris, Medical Research Center for Medical Mycology, University of Exeter, Exeter, United Kingdom; Department of Infectious Diseases, Great Ormond Street Hospital, London, United Kingdom;.
Prof. Ferry Hagen, Faculty of Science, Institute for Biodiversity and Ecosystem Dynamics, University of Amsterdam, Amsterdam, The Netherlands; Department of Medical Mycology, Westerdijk Fungal Biodiversity Institute, Utrecht, The Netherlands; Department of Medical Microbiology, University Medical Center Utrecht, Utrecht, The Netherlands;.
Assoc. Prof. Andrej Spec, Division of Infectious Diseases, Department of Medicine, Washington University School of Medicine, St. Louis, MO, USA..
Assoc. Prof. Rita Oladele, College of Medicine, University of Lagos, Lagos 102212, Nigeria..
Prof. Nelesh P. Govender, Department of Clinical Microbiology and Infectious Diseases, School of Pathology, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa; Division of Medical Microbiology, Faculty of Health Sciences, University of Cape Town, Cape Town, South Africa; Institute of Infection and Immunity, St George’s University of London, London, United Kingdom; MRC Centre for Medical Mycology, University of Exeter, Exeter, United Kingdom..
Prof. Sharon C.-A. Chen, Centre for Infectious Diseases and Microbiology Laboratory Services, Institute for Clinical Pathology and Medical Research, New South Wales Health Pathology, Westmead, Australia; Centre for Infectious Diseases and Microbiology, Westmead Hospital, University of Sydney, Westmead, Australia; The University of Sydney Institute of Infectious Diseases, Sydney Medical School, Faculty of Medicine and Health, University of Sydney, Sydney, Australia;.
Prof. Christopher H. Mody, Department of Microbiology, Immunology and Infectious Diseases, Department of Medicine, Snyder Institute for Chronic Diseases, University of Calgary, Calgary, Canada;.
Prof. Andreas H. Groll, Infectious Disease Research Program, Center for Bone Marrow Transplantation and, Department of Pediatric Hematology/Oncology, University Children’s Hospital, Muenster, Germany;.
Dr. Yee-Chun Chen, Department of Internal Medicine, National Taiwan University Hospital and College of Medicine, Taipei, Taiwan; National Institute of Infectious Diseases and Vaccinology, National Health Research Institutes, Zhunan, Taiwan;.
Prof. Michail S. Lionakis, Fungal Pathogenesis Section, Laboratory of Clinical Immunology & Microbiology, National Institute of Allergy & Infectious Diseases, National Institutes of Health, Bethesda, USA;.
Prof. Alexandre Alanio, Institut Pasteur, Centre National de Référence Mycoses Invasives et Antifongiques, Groupe de recherche Mycologie Translationnelle, Département de Mycologie, F-75015 Paris, France, Université Paris Cité, Paris, France; Laboratoire de parasitologie-mycologie, AP-HP, Hôpital Saint-Louis, Université Paris Cité, Paris, France;.
Dr. Elizabeth Castañeda, Instituto Nacional de Salud, Bogotá, Colombia;.
Dr. Jairo Lizarazo, Department of Internal Medicine, Hospital Universitario Erasmo Meoz, Faculty of Health, Univesidad de Pamplona, Cúcuta, Colombia;.
Dr. José E. Vidal, Departmento de Neurologia, Instituto de Infectologia Emílio Ribas, São Paulo, Brazil; Departamento de Moléstias Infecciosas e Parasitárias, Hospital das Clinicas, Faculdade de Medicina da Universidade de São Paulo (FMUSP), São Paulo, Brazil; Laboratório de Insvestigação Médica (LIM) 49, Instituto de Medicina Tropical, Faculdade de Medicina da Universidade de São Paulo (FMUSP), São Paulo, Brazil;.
Prof. Takahiro Takazono, Department of Infectious Diseases, Graduate School of Biomedical Sciences, Nagasaki University, Nagasaki, Japan; Department of Respiratory Medicine, Nagasaki University Hospital, Nagasaki, Japan;.
Prof. Martin Hoenigl, Division of Infectious Diseases, Translational Medical Mycology Research Unit, European Confederation of Medical Mycology Excellence Center for Medical Mycology, Medical University of Graz, Graz, Austria; BioTechMed, Graz, Austria;.
Prof. Jan-Willem Alffenaar, School of Pharmacy, Faculty of Medicine and Health, University of Sydney, Sydney, Australia; The University of Sydney Infectious Diseases Institute (Sydney ID), University of Sydney, Sydney, Australia; Department of Pharmacy, Westmead Hospital, Sydney, Australia;.
Prof. Jean-Pierre Gangneux, Institute for Health, Environment and Work Research - Irset, Inserm UMR_S 1085, University of Rennes, Rennes, France; Laboratory for Parasitology and Mycology, Centre National de Référence Mycoses Invasives et Antifongiques LA Asp-C, University Hospital of Rennes, Rennes, France;.
Dr. Rajeev Soman, Jupiter Hospital, Pune, India; Deenanath Mangeshkar Hospital, Pune, India; Hinduja Hospital, Mumbai, India;.
Prof. Li-Ping Zhu, Department of Infectious Diseases,Shanghai Key Laboratory of Infectious Diseases and Biosafety Emergency Response,National Medical Center for Infectious Diseases, Huashan Hospital, Fudan University, Shanghai, China;.
Prof. Alexandro Bonifaz, Hospital General de México “Dr. Eduardo Liceaga”, Dermatology Service. Mycology section, Universidad Nacional Autónoma de México, Mexico City, Mexico;.
Prof. Joseph N. Jarvis, Department of Clinical Research, Faculty of Infectious and Tropical Diseases, London School of Hygiene and Tropical Medicine, London, UK; Botswana Harvard AIDS Institute Partnership, Gaborone, Botswana;.
Prof. Jeremy N Day, Department of Clinical Microbiology and Infection, Royal Devon and Exeter University Hospital NHS Trust, Barrack Road, Exeter, EX2 5DW, UK..
Prof. Nikolai Klimko, Dept of Clinical Mycology, Allergy and Immunology, I. Mechnikov North Western State Medical University, St. Petersburg, Russia;.
Dr. Jon Salmanton-García, University of Cologne, Faculty of Medicine, and University Hospital Cologne, Institute of Translational Research, Cologne Excellence Cluster on Cellular Stress Responses in Aging-Associated Diseases (CECAD), Cologne, Germany; University of Cologne, Faculty of Medicine, University Hospital Cologne, Department I of Internal Medicine, Center for Integrated Oncology Aachen Bonn Cologne Duesseldorf (CIO ABCD) and Excellence Center for Medical Mycology (ECMM), Cologne, Germany; German Centre for Infection Research (DZIF), Partner Site Bonn-Cologne, Cologne, Germany..
Prof. Grégory Jouvion, Histology and Pathology Unit, Ecole nationale vétérinaire d’Alfort, Maisons-Alfort, France; Dynamyc Team, Université Paris Est Créteil and Ecole nationale vétérinaire d’Alfort, Créteil, France;.
Assoc. Prof. David B. Meya, Infectious Diseases Institute, School of Medicine, College of Heath Sciences, Makerere University, Kampala, Uganda;.
Dr. David Lawrence, Clinical Research Department, Faculty of Infectious and Tropical Diseases, London School of Hygiene and Tropical Medicine, London, UK; Botswana Harvard AIDS Institute Partnership, Gaborone, Botswana;.
Dr. Sebastian Rahn, Translational Research, Cologne Excellence Cluster on Cellular Stress Responses in Aging-Associated Diseases (CECAD), Faculty of Medicine and University Hospital Cologne, University of Cologne, Cologne, Germany; Center for Integrated Oncology Aachen Bonn Cologne Duesseldorf (CIO ABCD) and Excellence Center for Medical Mycology (ECMM), Department I of Internal Medicine, Faculty of Medicine and University Hospital Cologne, University of Cologne, Cologne, Germany; Partner Site Bonn-Cologne, German Centre for Infection Research (DZIF), Cologne, Germany;.
Dr. Felix Bongomin, Department of Medical Microbiology and Immunology, Faculty of Medicine, Gulu University, Gulu, Uganda;.
Dr. Brendan J. McMullan, Discipline of Paediatrics, School of Clinical Medicine, Faculty of Medicine and Health, University of New South Wales, Sydney, Australia; Department of Infectious Diseases, Sydney Children’s Hospital, Randwick, Sydney, Australia;.
Dr. Rosanne Sprute, Translational Research, Cologne Excellence Cluster on Cellular Stress Responses in Aging-Associated Diseases (CECAD), Faculty of Medicine and University Hospital Cologne, University of Cologne, Cologne, Germany; Center for Integrated Oncology Aachen Bonn Cologne Duesseldorf (CIO ABCD) and Excellence Center for Medical Mycology (ECMM), Department I of Internal Medicine, Faculty of Medicine and University Hospital Cologne, University of Cologne, Cologne, Germany; Partner Site Bonn-Cologne, German Centre for Infection Research (DZIF), Cologne, Germany;.
Dr. Tinashe K Nyazika, Department of Clinical Sciences, Liverpool School of Tropical Medicine, United Kingdom..
Assoc. Prof. Justin Beardsley, Sydney Infectious Disease Institute, Faculty of Medicine and Health, University of Sydney, Sydney, Australia; Westmead Clinical School, Sydney Medical School, University of Sydney, Sydney, Australia; Department of Infectious Diseases, Westmead Hospital, NSW Health, Sydney, Australia;.
Prof. Fabianne Carlesse, Pediatric Department, Federal University of São Paulo, São Paulo, Brazil; Oncology Pediatric Institute-IOP-GRAACC, Federal Univeristy of São Paulo, São Paulo, Brazil;.
Assoc Prof. Christopher H Heath, Department of Microbiology, Fiona Stanley Hospital Network, PathWest Laboratory Medicine, Murdoch, Australia, Department of Infectious Diseases, Fiona Stanley Hospital, Murdoch, Australia; UWA Medical School, Internal Medicine, The University of Western Australia, 35 Stirling Highway, Perth, WA, Australia, 6009..
Prof. Olusola O. Ayanlowo, Dermatology Unit, Department of Medicine, Lagos University Teaching Hospital, University of Lagos, Lagos, Nigeria;.
Ms. Olga M. Mashedi, Centre for Respiratory Diseases Research (CRDR), Kenya Medical Research Institute (KEMRI), Nairobi, Kenya;.
Assoc. Prof. Flavio Queiroz-Telles Filho, Department of Public Health, Hospital de Clínicas, Federal University of Paraná, Curitiba, Brazil;.
Prof. Mina C. Hosseinipour, Department of Medicine, Division of Infectious Diseases, University of North Carolina at Chapel Hill School of Medicine, Chapel Hill, USA; UNC Project Malawi, Lilongwe, Malawi;.
Dr. Atul K. Patel, Department of Infectious Diseases, Sterling Hospitals, Ahmedabad, India;.
Dr. Elvis Temfack, Africa Centers for Disease Control and Prevention: Addis Ababa, Ethiopia..
Prof. Nina Singh, Division of Infectious Diseases, Department of Medicine, University of Pittsburgh, Pittsburgh, PA..
Prof. Oliver A. Cornely, Institute of Translational Research, Cologne Excellence Cluster on Cellular Stress Responses in Aging-Associated Diseases (CECAD), Faculty of Medicine and University Hospital Cologne, University of Cologne, Cologne, Germany; Center for Integrated Oncology Aachen Bonn Cologne Duesseldorf (CIO ABCD) and Excellence Center for Medical Mycology (ECMM), Department I of Internal Medicine, Faculty of Medicine and University Hospital Cologne, University of Cologne, Cologne, Germany; Partner Site Bonn-Cologne, German Centre for Infection Research (DZIF), Cologne, Germany; Clinical Trials Centre Cologne (ZKS Köln), Faculty of Medicine and University Hospital Cologne, University of Cologne, Cologne, Germany..
Prof. David R. Boulware, Division of Infectious Diseases and International Medicine, Department of Medicine, University of Minnesota, Minneapolis, USA;.
Prof. Olivier Lortholary, Université de Paris Cité, APHP, Service des Maladies Infectieuses et Tropicales, Hôpital Necker-Enfants Malades, Centre d’Infectiologie Necker-Pasteur, Institut Imagine, Paris, France; Institut Pasteur, CNRS, Unité de Mycologie Moléculaire, Centre National de Référence Mycoses Invasives et Antifongiques, UMR 2000, Paris, France..
Prof. Peter G. Pappas, Mycoses Study Group Central Unit, Division of Infectious Diseases, Department of Medicine, University of Alabama at Birmingham, Birmingham, United States;.
Prof. John R Perfect, Division of Infectious Diseases, Department of Medicine, Duke University Medical Center, Durham, NC, United States. Department of Molecular Genetics and Microbiology, Duke University Medical Center, Durham, NC, United States..
References:
- 1.(WHO) WHO. WHO fungal priority pathogens list to guide research, development and public health action.. Geneva. [Google Scholar]
- 2.Day JN, Chau TTH, Wolbers M, et al. Combination antifungal therapy for cryptococcal meningitis. N Engl J Med 2013; 368(14): 1291–302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Boulware DR, Meya DB, Muzoora C, et al. Timing of antiretroviral therapy after diagnosis of cryptococcal meningitis. N Engl J Med 2014; 370(26): 2487–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Beardsley J, Wolbers M, Kibengo FM, et al. Adjunctive Dexamethasone in HIV-Associated Cryptococcal Meningitis. N Engl J Med 2016; 374(6): 542–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brouwer AE, Rajanuwong A, Chierakul W, et al. Combination antifungal therapies for HIV-associated cryptococcal meningitis: a randomised trial. Lancet 2004; 363(9423): 1764–7. [DOI] [PubMed] [Google Scholar]
- 6.Jarvis JN, Lawrence DS, Meya DB, et al. Single-Dose Liposomal Amphotericin B Treatment for Cryptococcal Meningitis. N Engl J Med 2022; 386(12): 1109–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Molloy SF, Kanyama C, Heyderman RS, et al. Antifungal Combinations for Treatment of Cryptococcal Meningitis in Africa. N Engl J Med 2018; 378(11): 1004–17. [DOI] [PubMed] [Google Scholar]
- 8.Bicanic T, Wood R, Meintjes G, et al. High-dose amphotericin B with flucytosine for the treatment of cryptococcal meningitis in HIV-infected patients: a randomized trial. Clin Infect Dis 2008; 47(1): 123–30. [DOI] [PubMed] [Google Scholar]
- 9.Rajasingham R, Govender NP, Jordan A, et al. The global burden of HIV-associated cryptococcal infection in adults in 2020: a modelling analysis. Lancet Infect Dis 2022; 22(12): 1748–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Organization WH. Guidelines for diagnosing, preventing and managing cryptococcal disease among adults, adolescents and children living with HIV. Geneva; 2022. p. 64. [PubMed] [Google Scholar]
- 11.Perfect JR, Dismukes WE, Dromer F, et al. Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the infectious diseases society of america. Clin Infect Dis 2010; 50(3): 291–322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Baddley JW, Forrest GN, Practice ASTIDCo. Cryptococcosis in solid organ transplantation-Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transplant 2019; 33(9): e13543. [DOI] [PubMed] [Google Scholar]
- 13.Schmidt-Hieber M, Silling G, Schalk E, et al. CNS infections in patients with hematological disorders (including allogeneic stem-cell transplantation)-Guidelines of the Infectious Diseases Working Party (AGIHO) of the German Society of Hematology and Medical Oncology (DGHO). Ann Oncol 2016; 27(7): 1207–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Singh N, Huprikar S, Burdette SD, et al. Donor-derived fungal infections in organ transplant recipients: guidelines of the American Society of Transplantation, infectious diseases community of practice. Am J Transplant 2012; 12(9): 2414–28. [DOI] [PubMed] [Google Scholar]
- 15.Chang CC, Hall V, Cooper C, et al. Consensus guidelines for the diagnosis and management of cryptococcosis and rare yeast infections in the haematology/oncology setting, 2021. Intern Med J 2021; 51 Suppl 7: 118–42. [DOI] [PubMed] [Google Scholar]
- 16.Govender NP, Meintjes G, Mangena P, et al. Southern African HIV Clinicians Society guideline for the prevention, diagnosis and management of cryptococcal disease among HIV-infected persons: 2019 update. South Afr J HIV Med 2019; 20(1): 1030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.HIV. PoGftPaToOIiAaAw. Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV: recommendations from the Centers for Disease Control and Prevention, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America.. 1 July 2021; last reviewed 12 January 2022 2019. https://clinicalinfo.hiv.gov/en/guidelines/adult-and-adolescent-opportunistic-infection/cryptococcosis?view=full (accessed 30 May 2022.
- 18.Guidelines on the diagnosis, prevention and management of cryptococcal disease in HIV-infected adults, adolescents and children: supplement to the 2016 consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection. Geneva, 2018. [PubMed] [Google Scholar]
- 19.Kung HC, Huang PY, Chen WT, et al. 2016 guidelines for the use of antifungal agents in patients with invasive fungal diseases in Taiwan. J Microbiol Immunol Infect 2018; 51(1): 1–17. [DOI] [PubMed] [Google Scholar]
- 20.(EACS) EACS. EACS Guidelines version 11.0, October 2021 2021. https://eacs.sanfordguide.com/ois/cryptococcosis (accessed 18 Aug 2022).
- 21.Izumikawa K, Kakeya H, Sakai F, et al. Executive Summary of JSMM Clinical Practice Guidelines for Diagnosis and Treatment of Cryptococcosis 2019. Med Mycol J 2020; 61(4): 61–89. [DOI] [PubMed] [Google Scholar]
- 22.Cornely OA, Alastruey-Izquierdo A, Arenz D, et al. Global guideline for the diagnosis and management of mucormycosis: an initiative of the European Confederation of Medical Mycology in cooperation with the Mycoses Study Group Education and Research Consortium. Lancet Infect Dis 2019; 19(12): e405–e21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen SC, Perfect J, Colombo AL, et al. Global guideline for the diagnosis and management of rare yeast infections: an initiative of the ECMM in cooperation with ISHAM and ASM. Lancet Infect Dis 2021; 21(12): e375–e86. [DOI] [PubMed] [Google Scholar]
- 24.Huang SH, Chuang YC, Lee YC, et al. Lumbar puncture for non-HIV-infected non-transplant patients with cryptococcosis: Should it be mandatory for all? PLoS One 2019; 14(8): e0221657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Baddley JW, Perfect JR, Oster RA, et al. Pulmonary cryptococcosis in patients without HIV infection: factors associated with disseminated disease. Eur J Clin Microbiol Infect Dis 2008; 27(10): 937–43. [DOI] [PubMed] [Google Scholar]
- 26.Brizendine KD, Baddley JW, Pappas PG. Predictors of mortality and differences in clinical features among patients with Cryptococcosis according to immune status. PLoS One 2013; 8(3): e60431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rajasingham R, Smith RM, Park BJ, et al. Global burden of disease of HIV-associated cryptococcal meningitis: an updated analysis. Lancet Infect Dis 2017; 17(8): 873–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.George IA, Spec A, Powderly WG, Santos CAQ. Comparative Epidemiology and Outcomes of Human Immunodeficiency virus (HIV), Non-HIV Non-transplant, and Solid Organ Transplant Associated Cryptococcosis: A Population-Based Study. Clin Infect Dis 2018; 66(4): 608–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hevey MA, George IA, Raval K, Powderly WG, Spec A. Presentation and Mortality of Cryptococcal Infection Varies by Predisposing Illness: A Retrospective Cohort Study. Am J Med 2019; 132(8): 977–83 e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Baddley JW, Chen SC, Huisingh C, et al. MSG07: An International Cohort Study Comparing Epidemiology and Outcomes of Patients With Cryptococcus neoformans or Cryptococcus gattii Infections. Clin Infect Dis 2021; 73(7): 1133–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ngamskulrungroj P, Chang Y, Sionov E, Kwon-Chung KJ. The primary target organ of Cryptococcus gattii is different from that of Cryptococcus neoformans in a murine model. mBio 2012; 3(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mitchell DH, Sorrell TC, Allworth AM, et al. Cryptococcal disease of the CNS in immunocompetent hosts: influence of cryptococcal variety on clinical manifestations and outcome. Clin Infect Dis 1995; 20(3): 611–6. [DOI] [PubMed] [Google Scholar]
- 33.Charlier C, Dromer F, Leveque C, et al. Cryptococcal neuroradiological lesions correlate with severity during cryptococcal meningoencephalitis in HIV-positive patients in the HAART era. PLoS One 2008; 3(4): e1950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tien RD, Chu PK, Hesselink JR, Duberg A, Wiley C. Intracranial cryptococcosis in immunocompromised patients: CT and MR findings in 29 cases. AJNR Am J Neuroradiol 1991; 12(2): 283–9. [PMC free article] [PubMed] [Google Scholar]
- 35.van der Horst CM, Saag MS, Cloud GA, et al. Treatment of cryptococcal meningitis associated with the acquired immunodeficiency syndrome. National Institute of Allergy and Infectious Diseases Mycoses Study Group and AIDS Clinical Trials Group. N Engl J Med 1997; 337(1): 15–21. [DOI] [PubMed] [Google Scholar]
- 36.Longley N, Muzoora C, Taseera K, et al. Dose response effect of high-dose fluconazole for HIV-associated cryptococcal meningitis in southwestern Uganda. Clin Infect Dis 2008; 47(12): 1556–61. [DOI] [PubMed] [Google Scholar]
- 37.Gaskell KM, Rothe C, Gnanadurai R, et al. A prospective study of mortality from cryptococcal meningitis following treatment induction with 1200 mg oral fluconazole in Blantyre, Malawi. PLoS One 2014; 9(11): e110285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rothe C, Sloan DJ, Goodson P, et al. A prospective longitudinal study of the clinical outcomes from cryptococcal meningitis following treatment induction with 800 mg oral fluconazole in Blantyre, Malawi. PLoS One 2013; 8(6): e67311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nussbaum JC, Jackson A, Namarika D, et al. Combination flucytosine and high-dose fluconazole compared with fluconazole monotherapy for the treatment of cryptococcal meningitis: a randomized trial in Malawi. Clin Infect Dis 2010; 50(3): 338–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Muzoora CK, Kabanda T, Ortu G, et al. Short course amphotericin B with high dose fluconazole for HIV-associated cryptococcal meningitis. J Infect 2012; 64(1): 76–81. [DOI] [PubMed] [Google Scholar]
- 41.Jackson AT, Nussbaum JC, Phulusa J, et al. A phase II randomized controlled trial adding oral flucytosine to high-dose fluconazole, with short-course amphotericin B, for cryptococcal meningitis. AIDS 2012; 26(11): 1363–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Charalambous LT, Premji A, Tybout C, et al. Prevalence, healthcare resource utilization and overall burden of fungal meningitis in the United States. J Med Microbiol 2018; 67(2): 215–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pyrgos V, Seitz AE, Steiner CA, Prevots DR, Williamson PR. Epidemiology of cryptococcal meningitis in the US: 1997–2009. PLoS One 2013; 8(2): e56269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bratton EW, El Husseini N, Chastain CA, et al. Approaches to antifungal therapies and their effectiveness among patients with cryptococcosis. Antimicrob Agents Chemother 2013; 57(6): 2485–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chang CC, Dorasamy AA, Gosnell BI, et al. Clinical and mycological predictors of cryptococcosis-associated immune reconstitution inflammatory syndrome. AIDS 2013; 27(13): 2089–99. [DOI] [PubMed] [Google Scholar]
- 46.Mootsikapun P, Chetchotisakd P, Anunnatsiri S, Choksawadphinyo K. The efficacy of fluconazole 600 mg/day versus itraconazole 600 mg/day as consolidation therapy of cryptococcal meningitis in AIDS patients. J Med Assoc Thai 2003; 86(4): 293–8. [PubMed] [Google Scholar]
- 47.Hope W, Stone NRH, Johnson A, et al. Fluconazole Monotherapy Is a Suboptimal Option for Initial Treatment of Cryptococcal Meningitis Because of Emergence of Resistance. mBio 2019; 10(6). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Naicker SD, Mpembe RS, Maphanga TG, et al. Decreasing fluconazole susceptibility of clinical South African Cryptococcus neoformans isolates over a decade. PLoS Negl Trop Dis 2020; 14(3): e0008137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Smith KD, Achan B, Hullsiek KH, et al. Increased Antifungal Drug Resistance in Clinical Isolates of Cryptococcus neoformans in Uganda. Antimicrob Agents Chemother 2015; 59(12): 7197–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Saag MS, Cloud GA, Graybill JR, et al. A comparison of itraconazole versus fluconazole as maintenance therapy for AIDS-associated cryptococcal meningitis. National Institute of Allergy and Infectious Diseases Mycoses Study Group. Clin Infect Dis 1999; 28(2): 291–6. [DOI] [PubMed] [Google Scholar]
- 51.Bozzette SA, Larsen RA, Chiu J, et al. A placebo-controlled trial of maintenance therapy with fluconazole after treatment of cryptococcal meningitis in the acquired immunodeficiency syndrome. California Collaborative Treatment Group. N Engl J Med 1991; 324(9): 580–4. [DOI] [PubMed] [Google Scholar]
- 52.Powderly WG, Saag MS, Cloud GA, et al. A controlled trial of fluconazole or amphotericin B to prevent relapse of cryptococcal meningitis in patients with the acquired immunodeficiency syndrome. The NIAID AIDS Clinical Trials Group and Mycoses Study Group. N Engl J Med 1992; 326(12): 793–8. [DOI] [PubMed] [Google Scholar]
- 53.Bandettini R, Castagnola E, Calvillo M, et al. Voriconazole for cryptococcal meningitis in children with leukemia or receiving allogeneic hemopoietic stem cell transplant. J Chemother 2009; 21(1): 108–9. [DOI] [PubMed] [Google Scholar]
- 54.Carbonara S, Regazzi M, Ciraci E, et al. Long-term efficacy and safety of TDM-assisted combination of voriconazole plus efavirenz in an AIDS patient with cryptococcosis and liver cirrhosis. Ann Pharmacother 2009; 43(5): 978–84. [DOI] [PubMed] [Google Scholar]
- 55.Gamaletsou MN, Sipsas NV, Kontoyiannis DP, et al. Successful salvage therapy of refractory HIV-related cryptococcal meningitis with the combination of liposomal amphotericin B, voriconazole, and recombinant interferon-gamma. Diagn Microbiol Infect Dis 2012; 74(4): 409–11. [DOI] [PubMed] [Google Scholar]
- 56.Nierenberg NE, Thompson GR, Lewis JS, Hogan BK, Patterson TF. Voriconazole use and pharmacokinetics in combination with interferon-gamma for refractory cryptococcal meningitis in a patient receiving low-dose ritonavir. Med Mycol 2010; 48(3): 532–6. [DOI] [PubMed] [Google Scholar]
- 57.Perfect JR, Marr KA, Walsh TJ, et al. Voriconazole treatment for less-common, emerging, or refractory fungal infections. Clin Infect Dis 2003; 36(9): 1122–31. [DOI] [PubMed] [Google Scholar]
- 58.Loyse A, Wilson D, Meintjes G, et al. Comparison of the early fungicidal activity of high-dose fluconazole, voriconazole, and flucytosine as second-line drugs given in combination with amphotericin B for the treatment of HIV-associated cryptococcal meningitis. Clin Infect Dis 2012; 54(1): 121–8. [DOI] [PubMed] [Google Scholar]
- 59.Sabbatani S, Manfredi R, Pavoni M, Consales A, Chiodo F. Voriconazole proves effective in long-term treatment of a cerebral cryptococcoma in a chronic nephropathic HIV-negative patient, after fluconazole failure. Mycopathologia 2004; 158(2): 165–71. [DOI] [PubMed] [Google Scholar]
- 60.Yao Y, Zhang JT, Yan B, et al. Voriconazole: a novel treatment option for cryptococcal meningitis. Infect Dis (Lond) 2015; 47(10): 694–700. [DOI] [PubMed] [Google Scholar]
- 61.Espinel-Ingroff A, Aller AI, Canton E, et al. Cryptococcus neoformans-Cryptococcus gattii species complex: an international study of wild-type susceptibility endpoint distributions and epidemiological cutoff values for fluconazole, itraconazole, posaconazole, and voriconazole. Antimicrob Agents Chemother 2012; 56(11): 5898–906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Esposito V, Viglietti R, Gargiulo M, et al. Successful treatment of Cryptococcal meningitis with a combination of liposomal amphotericin B, flucytosine and posaconazole: two case reports. In Vivo 2009; 23(3): 465–8. [PubMed] [Google Scholar]
- 63.Pitisuttithum P, Negroni R, Graybill JR, et al. Activity of posaconazole in the treatment of central nervous system fungal infections. J Antimicrob Chemother 2005; 56(4): 745–55. [DOI] [PubMed] [Google Scholar]
- 64.Schwartz S, Cornely OA, Hamed K, et al. Isavuconazole for the treatment of patients with invasive fungal diseases involving the central nervous system. Med Mycol 2020; 58(4): 417–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Thompson GR 3rd, Rendon A, Ribeiro Dos Santos R, et al. Isavuconazole Treatment of Cryptococcosis and Dimorphic Mycoses. Clin Infect Dis 2016; 63(3): 356–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Aberg JA, Price RW, Heeren DM, Bredt B. A pilot study of the discontinuation of antifungal therapy for disseminated cryptococcal disease in patients with acquired immunodeficiency syndrome, following immunologic response to antiretroviral therapy. J Infect Dis 2002; 185(8): 1179–82. [DOI] [PubMed] [Google Scholar]
- 67.Kirk O, Reiss P, Uberti-Foppa C, et al. Safe interruption of maintenance therapy against previous infection with four common HIV-associated opportunistic pathogens during potent antiretroviral therapy. Ann Intern Med 2002; 137(4): 239–50. [DOI] [PubMed] [Google Scholar]
- 68.Martinez E, Garcia-Viejo MA, Marcos MA, et al. Discontinuation of secondary prophylaxis for cryptococcal meningitis in HIV-infected patients responding to highly active antiretroviral therapy. AIDS 2000; 14(16): 2615–7. [DOI] [PubMed] [Google Scholar]
- 69.Mussini C, Pezzotti P, Miro JM, et al. Discontinuation of maintenance therapy for cryptococcal meningitis in patients with AIDS treated with highly active antiretroviral therapy: an international observational study. Clin Infect Dis 2004; 38(4): 565–71. [DOI] [PubMed] [Google Scholar]
- 70.Vibhagool A, Sungkanuparph S, Mootsikapun P, et al. Discontinuation of secondary prophylaxis for cryptococcal meningitis in human immunodeficiency virus-infected patients treated with highly active antiretroviral therapy: a prospective, multicenter, randomized study. Clin Infect Dis 2003; 36(10): 1329–31. [DOI] [PubMed] [Google Scholar]
- 71.Sheng WH, Hung CC, Chen MY, Hsieh SM, Chang SC. Successful discontinuation of fluconazole as secondary prophylaxis for cryptococcosis in AIDS patients responding to highly active antiretroviral therapy. Int J STD AIDS 2002; 13(10): 702–5. [DOI] [PubMed] [Google Scholar]
- 72.Lortholary O, Poizat G, Zeller V, et al. Long-term outcome of AIDS-associated cryptococcosis in the era of combination antiretroviral therapy. AIDS 2006; 20(17): 2183–91. [DOI] [PubMed] [Google Scholar]
- 73.Beardsley J, Wolbers M, Day JN, CryptoDex I. Dexamethasone in Cryptococcal Meningitis. N Engl J Med 2016; 375(2): 189–90. [DOI] [PubMed] [Google Scholar]
- 74.Rhein J, Huppler Hullsiek K, Tugume L, et al. Adjunctive sertraline for HIV-associated cryptococcal meningitis: a randomised, placebo-controlled, double-blind phase 3 trial. Lancet Infect Dis 2019; 19(8): 843–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Villanueva-Lozano H, Trevino-Rangel RJ, Gonzalez GM, et al. Clinical evaluation of the antifungal effect of sertraline in the treatment of cryptococcal meningitis in HIV patients: a single Mexican center experience. Infection 2018; 46(1): 25–30. [DOI] [PubMed] [Google Scholar]
- 76.Ngan NTT, Thanh Hoang Le N, Vi Vi NN, et al. An open label randomized controlled trial of tamoxifen combined with amphotericin B and fluconazole for cryptococcal meningitis. Elife 2021; 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Pappas PG, Bustamante B, Ticona E, et al. Recombinant interferon- gamma 1b as adjunctive therapy for AIDS-related acute cryptococcal meningitis. J Infect Dis 2004; 189(12): 2185–91. [DOI] [PubMed] [Google Scholar]
- 78.Jarvis JN, Meintjes G, Rebe K, et al. Adjunctive interferon-gamma immunotherapy for the treatment of HIV-associated cryptococcal meningitis: a randomized controlled trial. AIDS 2012; 26(9): 1105–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Marr KA, Sun Y, Spec A, et al. A Multicenter, Longitudinal Cohort Study of Cryptococcosis in Human Immunodeficiency Virus-negative People in the United States. Clin Infect Dis 2020; 70(2): 252–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Sun HY, Alexander BD, Lortholary O, et al. Unrecognized pretransplant and donor-derived cryptococcal disease in organ transplant recipients. Clin Infect Dis 2010; 51(9): 1062–9. [DOI] [PubMed] [Google Scholar]
- 81.Santos D, Hagen F, Meis JF, et al. Donor-Derived Transmission of Cryptococcus gattii sensu lato in Kidney Transplant Recipients. Emerg Infect Dis 2020; 26(6): 1329–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Wu G, Vilchez RA, Eidelman B, Fung J, Kormos R, Kusne S. Cryptococcal meningitis: an analysis among 5,521 consecutive organ transplant recipients. Transpl Infect Dis 2002; 4(4): 183–8. [DOI] [PubMed] [Google Scholar]
- 83.Sun HY, Alexander BD, Lortholary O, et al. Cutaneous cryptococcosis in solid organ transplant recipients. Med Mycol 2010; 48(6): 785–91. [DOI] [PubMed] [Google Scholar]
- 84.Osawa R, Alexander BD, Lortholary O, et al. Identifying predictors of central nervous system disease in solid organ transplant recipients with cryptococcosis. Transplantation 2010; 89(1): 69–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.El Helou G, Hellinger W. Cryptococcus neoformans pericarditis in a lung transplant recipient: Case report, literature review and pearls. Transpl Infect Dis 2019; 21(5): e13137. [DOI] [PubMed] [Google Scholar]
- 86.Singh N, Alexander BD, Lortholary O, et al. Pulmonary cryptococcosis in solid organ transplant recipients: clinical relevance of serum cryptococcal antigen. Clin Infect Dis 2008; 46(2): e12–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Sun HY, Alexander BD, Lortholary O, et al. Lipid formulations of amphotericin B significantly improve outcome in solid organ transplant recipients with central nervous system cryptococcosis. Clin Infect Dis 2009; 49(11): 1721–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Sun HY, Alexander BD, Huprikar S, et al. Predictors of immune reconstitution syndrome in organ transplant recipients with cryptococcosis: implications for the management of immunosuppression. Clin Infect Dis 2015; 60(1): 36–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Takazono T, Hidaka Y, Morimoto S, et al. A comparison of liposomal amphotericin B alone and in combination with flucytosine in the treatment of non-HIV Cryptococcal meningitis: A nationwide observational study. Mycoses 2022. [DOI] [PubMed] [Google Scholar]
- 90.Aissaoui N, Benhadid-Brahmi Y, Sturny-Leclere A, et al. Investigation of CryptoPS LFA-positive sera in patients at risk of cryptococcosis. Med Mycol 2022; 60(10). [DOI] [PubMed] [Google Scholar]
- 91.Pappas PG, Perfect JR, Cloud GA, et al. Cryptococcosis in human immunodeficiency virus-negative patients in the era of effective azole therapy. Clin Infect Dis 2001; 33(5): 690–9. [DOI] [PubMed] [Google Scholar]
- 92.Vilchez RA, Linden P, Lacomis J, Costello P, Fung J, Kusne S. Acute respiratory failure associated with pulmonary cryptococcosis in non-aids patients. Chest 2001; 119(6): 1865–9. [DOI] [PubMed] [Google Scholar]
- 93.Nadrous HF, Antonios VS, Terrell CL, Ryu JH. Pulmonary cryptococcosis in nonimmunocompromised patients. Chest 2003; 124(6): 2143–7. [DOI] [PubMed] [Google Scholar]
- 94.Skolnik K, Huston S, Mody CH. Cryptococcal Lung Infections. Clin Chest Med 2017; 38(3): 451–64. [DOI] [PubMed] [Google Scholar]
- 95.Shirley RM, Baddley JW. Cryptococcal lung disease. Curr Opin Pulm Med 2009; 15(3): 254–60. [DOI] [PubMed] [Google Scholar]
- 96.Avendano J, Tanishima T, Kuwabara T. Ocular cryptococcosis. Am J Ophthalmol 1978; 86(1): 110–3. [DOI] [PubMed] [Google Scholar]
- 97.Wong BJ, Rao NA, Ameri H. Optical coherence tomography imaging of presumed Cryptococcus neoformans infection localized to the retina. J Curr Ophthalmol 2019; 31(3): 353–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wood L, Miedzinski L. Skeletal cryptococcosis: Case report and review of the literature. Can J Infect Dis 1996; 7(2): 125–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Medaris LA, Ponce B, Hyde Z, et al. Cryptococcal osteomyelitis: a report of 5 cases and a review of the recent literature. Mycoses 2016; 59(6): 334–42. [DOI] [PubMed] [Google Scholar]
- 100.Zhou HX, Lu L, Chu T, et al. Skeletal cryptococcosis from 1977 to 2013. Front Microbiol 2014; 5: 740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Rolfes MA, Hullsiek KH, Rhein J, et al. The effect of therapeutic lumbar punctures on acute mortality from cryptococcal meningitis. Clin Infect Dis 2014; 59(11): 1607–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Kagimu E, Engen N, Ssebambulidde K, et al. Therapeutic Lumbar Punctures in Human Immunodeficiency Virus-Associated Cryptococcal Meningitis: Should Opening Pressure Direct Management? Open Forum Infect Dis 2022; 9(9): ofac416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Cherian J, Atmar RL, Gopinath SP. Shunting in cryptococcal meningitis. J Neurosurg 2016; 125(1): 177–86. [DOI] [PubMed] [Google Scholar]
- 104.Manosuthi W, Sungkanuparph S, Chottanapund S, et al. Temporary external lumbar drainage for reducing elevated intracranial pressure in HIV-infected patients with cryptococcal meningitis. Int J STD AIDS 2008; 19(4): 268–71. [DOI] [PubMed] [Google Scholar]
- 105.Zhang Q, Li H, Zhang K, et al. Lumbar drainage for the treatment of refractory intracranial hypertension in HIV-negative cryptococcal meningitis. Future Microbiol 2019; 14: 859–66. [DOI] [PubMed] [Google Scholar]
- 106.Newton PN, Thai le H, Tip NQ, et al. A randomized, double-blind, placebo-controlled trial of acetazolamide for the treatment of elevated intracranial pressure in cryptococcal meningitis. Clin Infect Dis 2002; 35(6): 769–72. [DOI] [PubMed] [Google Scholar]
- 107.Hu Z, Yang Y, Cheng J, Cheng C, Chi Y, Wei H. The use of mannitol in HIV-infected patients with symptomatic cryptococcal meningitis. Drug Discov Ther 2017; 10(6): 329–33. [DOI] [PubMed] [Google Scholar]
- 108.Makadzange AT, Ndhlovu CE, Takarinda K, et al. Early versus delayed initiation of antiretroviral therapy for concurrent HIV infection and cryptococcal meningitis in sub-saharan Africa. Clin Infect Dis 2010; 50(11): 1532–8. [DOI] [PubMed] [Google Scholar]
- 109.Bisson GP, Molefi M, Bellamy S, et al. Early versus delayed antiretroviral therapy and cerebrospinal fluid fungal clearance in adults with HIV and cryptococcal meningitis. Clin Infect Dis 2013; 56(8): 1165–73. [DOI] [PubMed] [Google Scholar]
- 110.Zhao T, Xu XL, Lu YQ, et al. The Effect of Early vs. Deferred Antiretroviral Therapy Initiation in HIV-Infected Patients With Cryptococcal Meningitis: A Multicenter Prospective Randomized Controlled Analysis in China. Front Med (Lausanne) 2021; 8: 779181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ingle SM, Miro JM, May MT, et al. Early Antiretroviral Therapy Not Associated With Higher Cryptococcal Meningitis Mortality in People With Human Immunodeficiency Virus in High-Income Countries: An International Collaborative Cohort Study. Clin Infect Dis 2023; 77(1): 64–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Boulware DR, Jarvis JN. Timing of Antiretroviral Therapy in Cryptococcal Meningitis: What We Can (and Cannot) Learn From Observational Data. Clin Infect Dis 2023; 77(1): 74–6. [DOI] [PubMed] [Google Scholar]
- 113.Kityo C, Szubert AJ, Siika A, et al. Raltegravir-intensified initial antiretroviral therapy in advanced HIV disease in Africa: A randomised controlled trial. PLoS Med 2018; 15(12): e1002706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Rhein J, Hullsiek KH, Evans EE, et al. Detrimental Outcomes of Unmasking Cryptococcal Meningitis With Recent ART Initiation. Open Forum Infect Dis 2018; 5(8): ofy122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Kalata N, Ellis J, Kanyama C, et al. Short-term Mortality Outcomes of HIV-Associated Cryptococcal Meningitis in Antiretroviral Therapy–Naïve and –Experienced Patients in Sub-Saharan Africa. Open Forum Infectious Diseases 2021; 8(10). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Alufandika M, Lawrence DS, Boyer-Chammard T, et al. A pragmatic approach to managing antiretroviral therapy-experienced patients diagnosed with HIV-associated cryptococcal meningitis: impact of antiretroviral therapy adherence and duration. AIDS 2020; 34(9): 1425–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Chen YC, Chang TY, Liu JW, et al. Increasing trend of fluconazole-non-susceptible Cryptococcus neoformans in patients with invasive cryptococcosis: a 12-year longitudinal study. BMC Infect Dis 2015; 15: 277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Bicanic T, Harrison T, Niepieklo A, Dyakopu N, Meintjes G. Symptomatic relapse of HIV-associated cryptococcal meningitis after initial fluconazole monotherapy: the role of fluconazole resistance and immune reconstitution. Clin Infect Dis 2006; 43(8): 1069–73. [DOI] [PubMed] [Google Scholar]
- 119.Van Wyk M, Govender NP, Mitchell TG, Litvintseva AP, Germs SA. Multilocus sequence typing of serially collected isolates of Cryptococcus from HIV-infected patients in South Africa. J Clin Microbiol 2014; 52(6): 1921–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Stone NR, Rhodes J, Fisher MC, et al. Dynamic ploidy changes drive fluconazole resistance in human cryptococcal meningitis. J Clin Invest 2019; 129(3): 999–1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Bongomin F, Oladele RO, Gago S, Moore CB, Richardson MD. A systematic review of fluconazole resistance in clinical isolates of Cryptococcus species. Mycoses 2018; 61(5): 290–7. [DOI] [PubMed] [Google Scholar]
- 122.(CLSI). CaLSI. Epidemiological Cutoff Values for Antifungal Susceptibility Testing. 4th ed. CLSI supplement M57S.2022. (accessed 30 Aug 2023). [Google Scholar]
- 123.Aberg JA, Watson J, Segal M, Chang LW. Clinical utility of monitoring serum cryptococcal antigen (sCRAG) titers in patients with AIDS-related cryptococcal disease. HIV Clin Trials 2000; 1(1): 1–6. [DOI] [PubMed] [Google Scholar]
- 124.French MA. HIV/AIDS: immune reconstitution inflammatory syndrome: a reappraisal. Clin Infect Dis 2009; 48(1): 101–7. [DOI] [PubMed] [Google Scholar]
- 125.Haddow LJ, Colebunders R, Meintjes G, et al. Cryptococcal immune reconstitution inflammatory syndrome in HIV-1-infected individuals: proposed clinical case definitions. Lancet Infect Dis 2010; 10(11): 791–802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Panackal AA, Wuest SC, Lin YC, et al. Paradoxical Immune Responses in Non-HIV Cryptococcal Meningitis. PLoS Pathog 2015; 11(5): e1004884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Deshayes S, Bouvier N, Chatelet V, et al. Severe cryptococcal-associated neurological immune reconstitution inflammatory syndrome in a renal transplant recipient treated with adalimumab. Transpl Infect Dis 2016; 18(3): 461–5. [DOI] [PubMed] [Google Scholar]
- 128.Scemla A, Gerber S, Duquesne A, et al. Dramatic improvement of severe cryptococcosis-induced immune reconstitution syndrome with adalimumab in a renal transplant recipient. Am J Transplant 2015; 15(2): 560–4. [DOI] [PubMed] [Google Scholar]
- 129.Gaube G, De Castro N, Gueguen A, et al. Treatment with adalimumab for severe immune reconstitution inflammatory syndrome in an HIV-infected patient presenting with cryptococcal meningitis. Med Mal Infect 2016; 46(3): 154–6. [DOI] [PubMed] [Google Scholar]
- 130.Sitapati AM, Kao CL, Cachay ER, Masoumi H, Wallis RS, Mathews WC. Treatment of HIV-related inflammatory cerebral cryptococcoma with adalimumab. Clin Infect Dis 2010; 50(2): e7–10. [DOI] [PubMed] [Google Scholar]
- 131.Brunel AS, Reynes J, Tuaillon E, et al. Thalidomide for steroid-dependent immune reconstitution inflammatory syndromes during AIDS. AIDS 2012; 26(16): 2110–2. [DOI] [PubMed] [Google Scholar]
- 132.Lortholary O, Fontanet A, Memain N, et al. Incidence and risk factors of immune reconstitution inflammatory syndrome complicating HIV-associated cryptococcosis in France. AIDS 2005; 19(10): 1043–9. [DOI] [PubMed] [Google Scholar]
- 133.Somerville LK, Henderson AP, Chen SC, Kok J. Successful treatment of Cryptococcus neoformans immune reconstitution inflammatory syndrome in an immunocompetent host using thalidomide. Med Mycol Case Rep 2015; 7: 12–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Anjum S, Dean O, Kosa P, et al. Outcomes in Previously Healthy Cryptococcal Meningoencephalitis Patients Treated With Pulse Taper Corticosteroids for Post-infectious Inflammatory Syndrome. Clin Infect Dis 2021; 73(9): e2789–e98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Chen SC, Slavin MA, Heath CH, et al. Clinical manifestations of Cryptococcus gattii infection: determinants of neurological sequelae and death. Clin Infect Dis 2012; 55(6): 789–98. [DOI] [PubMed] [Google Scholar]
- 136.Chen SC, Meyer W, Sorrell TC. Cryptococcus gattii infections. Clin Microbiol Rev 2014; 27(4): 980–1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Harris JR, Galanis E, Lockhart SR. Cryptococcus gattii Infections and Virulence. Current Fungal Infection Reports 2014; 8(1): 81–9. [Google Scholar]
- 138.Chen S, Sorrell T, Nimmo G, et al. Epidemiology and host- and variety-dependent characteristics of infection due to Cryptococcus neoformans in Australia and New Zealand. Australasian Cryptococcal Study Group. Clin Infect Dis 2000; 31(2): 499–508. [DOI] [PubMed] [Google Scholar]
- 139.Morgan J, McCarthy KM, Gould S, et al. Cryptococcus gattii infection: characteristics and epidemiology of cases identified in a South African province with high HIV seroprevalence, 2002–2004. Clin Infect Dis 2006; 43(8): 1077–80. [DOI] [PubMed] [Google Scholar]
- 140.Litvintseva AP, Thakur R, Reller LB, Mitchell TG. Prevalence of clinical isolates of Cryptococcus gattii serotype C among patients with AIDS in Sub-Saharan Africa. J Infect Dis 2005; 192(5): 888–92. [DOI] [PubMed] [Google Scholar]
- 141.Nyazika TK, Hagen F, Meis JF, Robertson VJ. Cryptococcus tetragattii as a major cause of cryptococcal meningitis among HIV-infected individuals in Harare, Zimbabwe. J Infect 2016; 72(6): 745–52. [DOI] [PubMed] [Google Scholar]
- 142.Steele KT, Thakur R, Nthobatsang R, Steenhoff AP, Bisson GP. In-hospital mortality of HIV-infected cryptococcal meningitis patients with C. gattii and C. neoformans infection in Gaborone, Botswana. Med Mycol 2010; 48(8): 1112–5. [DOI] [PubMed] [Google Scholar]
- 143.Saijo T, Chen J, Chen SC, et al. Anti-granulocyte-macrophage colony-stimulating factor autoantibodies are a risk factor for central nervous system infection by Cryptococcus gattii in otherwise immunocompetent patients. mBio 2014; 5(2): e00912–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Yang DH, England MR, Salvator H, et al. Cryptococcus gattii Species Complex as an Opportunistic Pathogen: Underlying Medical Conditions Associated with the Infection. mBio 2021; 12(5): e0270821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Viola GM, Malek AE, Rosen LB, et al. Disseminated cryptococcosis and anti-granulocyte-macrophage colony-stimulating factor autoantibodies: An underappreciated association. Mycoses 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Shi D, Haas PJ, Boekhout T, Hahn RC, Hagen F. Neglecting Genetic Diversity Hinders Timely Diagnosis of Cryptococcus Infections. J Clin Microbiol 2021; 59(4). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Chen SC, Korman TM, Slavin MA, et al. Antifungal therapy and management of complications of cryptococcosis due to Cryptococcus gattii. Clin Infect Dis 2013; 57(4): 543–51. [DOI] [PubMed] [Google Scholar]
- 148.Phillips P, Galanis E, MacDougall L, et al. Longitudinal clinical findings and outcome among patients with Cryptococcus gattii infection in British Columbia. Clin Infect Dis 2015; 60(9): 1368–76. [DOI] [PubMed] [Google Scholar]
- 149.Phillips P, Chapman K, Sharp M, et al. Dexamethasone in Cryptococcus gattii central nervous system infection. Clin Infect Dis 2009; 49(4): 591–5. [DOI] [PubMed] [Google Scholar]
- 150.Fujita NK, Reynard M, Sapico FL, Guze LB, Edwards JE Jr., Cryptococcal intracerebral mass lesions: the role of computed tomography and nonsurgical management. Ann Intern Med 1981; 94(3): 382–8. [DOI] [PubMed] [Google Scholar]
- 151.Hospenthal DR, Bennett JE. Persistence of cryptococcomas on neuroimaging. Clin Infect Dis 2000; 31(5): 1303–6. [DOI] [PubMed] [Google Scholar]
- 152.Kordossis T, Avlami A, Velegraki A, et al. First report of Cryptococcus laurentii meningitis and a fatal case of Cryptococcus albidus cryptococcaemia in AIDS patients. Med Mycol 1998; 36(5): 335–9. [PubMed] [Google Scholar]
- 153.Choe YJ, Blatt DB, Yalcindag A, Geffert SF, Bobenchik AM, Michelow IC. Cryptococcus albidus Fungemia in an Immunosuppressed Child: Case Report and Systematic Literature Review. J Pediatric Infect Dis Soc 2020; 9(1): 100–5. [DOI] [PubMed] [Google Scholar]
- 154.Khawcharoenporn T, Apisarnthanarak A, Mundy LM. Non-neoformans cryptococcal infections: a systematic review. Infection 2007; 35(2): 51–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Xiao M, Fan X, Chen XX, et al. Misidentification of a Rare Species, Cryptococcus laurentii, by Commonly Used Commercial Biochemical Methods and Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry Systems: Challenges for Clinical Mycology Laboratories. J Clin Microbiol 2016; 54(1): 226–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Arendrup MC, Boekhout T, Akova M, et al. ESCMID and ECMM joint clinical guidelines for the diagnosis and management of rare invasive yeast infections. Clin Microbiol Infect 2014; 20 Suppl 3: 76–98. [DOI] [PubMed] [Google Scholar]
- 157.Oliveira LSS, Pinto LM, de Medeiros MAP, et al. Comparison of Cryptococcus gattii/neoformans Species Complex to Related Genera (Papiliotrema and Naganishia) Reveal Variances in Virulence Associated Factors and Antifungal Susceptibility. Front Cell Infect Microbiol 2021; 11: 642658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Ely EW, Peacock JE Jr., Haponik EF, Washburn RG. Cryptococcal pneumonia complicating pregnancy. Medicine (Baltimore) 1998; 77(3): 153–67. [DOI] [PubMed] [Google Scholar]
- 159.Pastick KA, Nalintya E, Tugume L, et al. Cryptococcosis in pregnancy and the postpartum period: Case series and systematic review with recommendations for management. Med Mycol 2020; 58(3): 282–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Bright PD, Lupiya D, van Oosterhout JJ, Chen A, Harrison TS, Chan AK. The treatment of a pregnant HIV positive patient with cryptococcal meningitis in Malawi. Case report and review of treatment options. Med Mycol Case Rep 2018; 19: 9–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Pursley TJ, Blomquist IK, Abraham J, Andersen HF, Bartley JA. Fluconazole-induced congenital anomalies in three infants. Clin Infect Dis 1996; 22(2): 336–40. [DOI] [PubMed] [Google Scholar]
- 162.Mastroiacovo P, Mazzone T, Botto LD, et al. Prospective assessment of pregnancy outcomes after first-trimester exposure to fluconazole. Am J Obstet Gynecol 1996; 175(6): 1645–50. [DOI] [PubMed] [Google Scholar]
- 163.Zhu Y, Bateman BT, Gray KJ, et al. Oral fluconazole use in the first trimester and risk of congenital malformations: population based cohort study. BMJ 2020; 369: m1494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Molgaard-Nielsen D, Svanstrom H, Melbye M, Hviid A, Pasternak B. Association Between Use of Oral Fluconazole During Pregnancy and Risk of Spontaneous Abortion and Stillbirth. JAMA 2016; 315(1): 58–67. [DOI] [PubMed] [Google Scholar]
- 165.Molgaard-Nielsen D, Pasternak B, Hviid A. Use of oral fluconazole during pregnancy and the risk of birth defects. N Engl J Med 2013; 369(9): 830–9. [DOI] [PubMed] [Google Scholar]
- 166.Lenz D, Held J, Goerke S, et al. Primary cutaneous cryptococcosis in an eight-year-old immunocompetent child: how to treat? Klin Padiatr 2015; 227(1): 41–4. [DOI] [PubMed] [Google Scholar]
- 167.Molina-Leyva A, Ruiz-Carrascosa JC, Leyva-Garcia A, Husein-Elahmed H. Cutaneous Cryptococcus laurentii infection in an immunocompetent child. Int J Infect Dis 2013; 17(12): e1232–3. [DOI] [PubMed] [Google Scholar]
- 168.Sweeney DA, Caserta MT, Korones DN, Casadevall A, Goldman DL. A ten-year-old boy with a pulmonary nodule secondary to Cryptococcus neoformans: case report and review of the literature. Pediatr Infect Dis J 2003; 22(12): 1089–93. [DOI] [PubMed] [Google Scholar]
- 169.Ramdial PK, Sing Y, Deonarain J, Bhimma R, Chotey N, Sewram V. Pediatric renal cryptococcosis: novel manifestations in the acquired immunodeficiency syndrome era. Int J Surg Pathol 2011; 19(3): 386–92. [DOI] [PubMed] [Google Scholar]
- 170.Lizarazo J, Escandon P, Agudelo CI, Castaneda E. Cryptococcosis in Colombian children and literature review. Mem Inst Oswaldo Cruz 2014; 109(6): 797–804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Gao LW, Jiao AX, Wu XR, et al. Clinical characteristics of disseminated cryptococcosis in previously healthy children in China. BMC Infect Dis 2017; 17(1): 359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Joshi NS, Fisher BT, Prasad PA, Zaoutis TE. Epidemiology of cryptococcal infection in hospitalized children. Pediatr Infect Dis J 2010; 29(12): e91–5. [DOI] [PubMed] [Google Scholar]
- 173.Gassiep I, Douglas J, Emeto TI, Crawley K, Playford EG. Cryptococcal infections over a 15 year period at a tertiary facility & impact of guideline management. Mycoses 2018; 61(9): 633–8. [DOI] [PubMed] [Google Scholar]
- 174.Dodds-Ashley E Management of drug and food interactions with azole antifungal agents in transplant recipients. Pharmacotherapy 2010; 30(8): 842–54. [DOI] [PubMed] [Google Scholar]
- 175.Glotzbecker B, Duncan C, Alyea E 3rd, Campbell B, Soiffer R. Important drug interactions in hematopoietic stem cell transplantation: what every physician should know. Biol Blood Marrow Transplant 2012; 18(7): 989–1006. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


