Abstract
Primary diffuse large B-cell lymphoma of the central nervous system (CNS-DLBCL) can be difficult to diagnose because of the limited amount of biopsy tissue. Here, we analyzed the utility of insulin-like growth factor II mRNA binding protein 3 (IMP3) immunohistochemistry (IHC) as an adjunctive diagnostic tool for CNS-DLBCL. IHC was performed on 57 biopsy samples (55 brain biopsy samples and two vitreous cell blocks) from 54 patients with CNS-DLBCL, including three biopsy samples initially diagnosed as negative or indeterminate for CNS-DLBCL. Additionally, IMP3 IHC was performed on 68 DLBCLs other than CNS-DLBCL and 12 inflammatory brain diseases. Cytoplasmic IMP3 expression was noted in ≥50% of tumor cells in 100% (57/57) of CNS-DLBCLs and 88.2% (60/68) of non-CNS-DLBCLs. In contrast, no IMP3-positive CD20-positive B cells were observed in the inflammatory brain disease (P < 0.0001). In conclusion, IMP3 is highly expressed in CNS-DLBCL. However, it is also expressed in other types of DLBCLs, making it less specific. Most CNS-DLBCL cases can be diagnosed without performing IHC for IMP3 expression, but it may be a useful adjunctive tool to differentiate from reactive lesions when tumor cells are few or deformed.
Keywords: primary diffuse large B-cell lymphoma of the central nervous system, insulin-like growth factor II mRNA binding protein 3, immunohistochemistry
INTRODUCTION
Primary diffuse large B-cell lymphoma of the central nervous system (CNS-DLBCL) accounts for more than 90% of primary CNS lymphomas.1 Brain biopsy is the standard procedure for diagnosing CNS-DLBCL, but it can be risky, with intracranial hemorrhage and neurological decline reported in 5–8% of cases.2 While it is desirable to make a definitive diagnosis on the first biopsy, the risk of complications tends to limit the amount of tissue, and some patients require a second biopsy for a definitive diagnosis.3,4
To improve the diagnostic accuracy of CNS-DLBCL, we focused on the utility of immunohistochemistry (IHC) for insulin-like growth factor II mRNA binding protein 3 (IMP3), which is an oncofetal protein expressed by most DLBCLs and classic Hodgkin lymphoma, but not by reactive B cells, except germinal center B cells (GCBs).5 In our practice, we use IMP3 as a useful marker for these lymphomas, especially in small biopsy specimens with low numbers of tumor cells, but its utility in CNS-DLBCL has remained unclear.
Here, we examined IMP3 expression in CNS-DLBCL, compared it with other encephalitic diseases that can mimic CNS-DLBCL, and tested the diagnostic utility of IMP3 in CNS-DLBCL.
MATERIAL AND METHODS
Case selection
We analyzed biopsy samples from patients diagnosed with CNS-DLBCL at the Osaka Red Cross Hospital and Kyoto University Hospital between 2014 and 2022. Histological diagnosis for CNS-DLBCL was performed according to the World Health Organization criteria.1 Immunodeficiency-associated CNS lymphomas and secondary involvement by systemic DLBCL were excluded. As a control, DLBCLs other than CNS-DLBCL and brain biopsy samples from cases of inflammatory brain diseases diagnosed at the Kyoto University Hospital were included in the study.
Clinical and pathological information was obtained from pathology reports and patient charts. This study was approved by the Kyoto University Hospital review board (approval code: R3843) and was conducted in accordance with the Declaration of Helsinki.
IHC
Tissue sections of 4-μm thickness were sectioned from formalin-fixed and paraffin- embedded tissue blocks and prepared for IHC and hematoxylin and eosin (H&E) staining. IHC for IMP3, CD3, and CD20 was performed using anti-IMP3 (Clone 69.1; DAKO, Santa Clara, CA) , anti-CD3 (Clone 2GV6; Roche, Basel, Switzerland), and anti-CD20 (Clone L26; DAKO) antibodies, as described previously.6,7 In addition, primary antibodies specific for B-cell lymphoma 6 (Clone Gl191E/A8; Roche), CD10 (Clone 56C6; Leica Biosystems, Newcastle, UK), and multiple myeloma oncogene 1 (MUM1) (Clone MUM1p; DAKO) were used for the Hans algorithm.8 CD5 (Clone SP19, Roche) IHC and Epstein-Bar virus-encoded small RNAs in situ hybridization (EBER-ISH) (PB0589, Leica Biosystems) were also performed in some cases. We graded the staining intensity of IMP3 from 0 to 3+ according to a previous study.5 Cytoplasmic IMP3 expression in CD20-positive B cells was regarded as IMP3-positive.
Statistical analyses
The findings between the two groups were statistically analyzed by performing Fisher’s exact test using GraphPad Prism (GraphPad Software, San Diego, California). Statistical significance was defined as P < 0.05.
RESULTS
CNS-DLBCL
We collected 57 pathology specimens from 54 patients with CNS-DLBCL, including two vitreoretinal DLBCLs, diagnosed by brain biopsy or vitreous aspiration (cell block). Among the 54 patients, 35 were male and 19 were female. The mean age of the patients was 67.4 y (median: 69 y). Three patients (5.6%) underwent a second biopsy because the initial diagnosis was indefinite for CNS-DLBCL. Among the 54 samples diagnosed as CNS-DLBCL, 16.7% (9/54) were classified as the GCB subtype. All CD10-negative cases tested MUM1-positive by IHC and were classified as the non-GCB subtype (45/54; 83.3%). CD5 IHC was performed on 41 samples from 41 patients, of which, four samples tested positive (9.8%). EBER-ISH was performed in 10 samples from 10 patients and all of them tested negative. In two cases of vitreoretinal DLBCL, there were only 144 and 366 CD20-positive cells per whole slide section, respectively. All CNS-DLBCLs were strongly positive for IMP3 (intensity score ≥2+), with >80% of the tumor cells being positive (Figure 1). The first biopsies from the three patients that did not lead to a definitive diagnosis of CNS-DLBCL also harbored CD20-positive B cells with strong IMP3 expression, suggestive of CNS-DLBCL (Figure 2A–C).
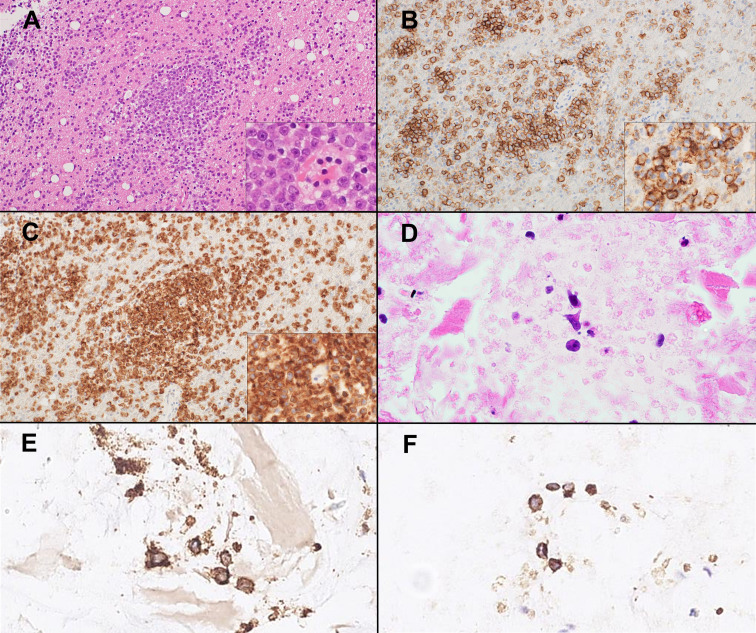
Fig. 1.
Primary diffuse large B-cell lymphoma of the CNS. Lymphoma cell infiltration around blood vessels and within brain parenchyma. Lymphoma cells are large, with vesicular nuclei and prominent nucleoli (inset) (A). Lymphoma cells are positive for CD20 (B) and IMP3 (C). Diffuse large B-cell lymphoma of the vitreoretinal in cell block specimens. A few degenerated lymphoma cells are observed in the vitreous fluid (D) and are positive for CD20 (E) and IMP3 (F).
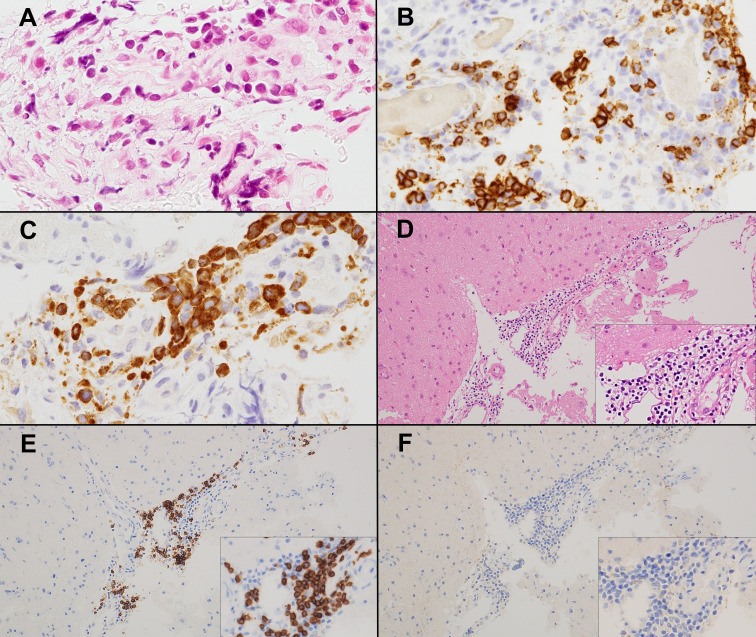
Fig. 2.
First biopsy from a patient with primary diffuse large B-cell lymphoma of the CNS (CNS-DLBCL) for which a definite diagnosis of lymphoma could not be made. Perivascular cuffing lymphocytes. (A) with CD20 expression (B). Crush artifacts limit cytomorphological evaluation. Retrospective analysis revealed strong IMP3 expression by lymphocytes, suggesting CNS-DLBCL (C). Primary CNS vasculitis. Perivascular cuffing of small lymphocytes is observed (D). Lymphocytes are positive for CD20 (E), but negative for IMP3 (F).
Other DLBCLs
Sixty-eight DLBCLs other than CNS-DLBCL biopsied from 67 patients were tested for IMP3 expression. Fifty-one cases (75%) were extra-nodal lesions and 17 cases (25%) were nodal lesions. Thirty cases (44.1%) were GCB type and 38 (55.9%) were non-GCB type. Seven cases (10.3%) were EBER-positive and 61 (89.7%) were EBER-negative. The staining intensity of IMP3 was 0 in 2 cases (2.9%), 1+ in 5 cases (7.4%), and ≥2+ in 61 cases (89.7%). IMP3 expression was positive in ≥50% of tumor cells in 60 DLBCLs (88.2%). High IMP3 expression (≥50%) was not significantly correlated with nodal or extra-nodal origin (nodal 17/17 vs. extra-nodal 43/51; P = 0.1863), or GCB or non-GCB subtype (GCB 28/30 vs. non-GCB 32/38; P = 0.2880). However, IMP3 expression in EBV-positive DLBCL was significantly low (EBV-positive 2/7 vs. EBV-negative 58/61; P = 0.0001).
Inflammatory brain diseases
As a negative control, 12 biopsy samples diagnosed with inflammatory brain diseases were retrieved from the pathology archive of Kyoto University Hospital. These included samples of progressive multifocal leukoencephalopathy, varicella zoster virus encephalitis, autoimmune encephalomyelitis, amyloid angiopathy, tumefactive multiple sclerosis (n = 2), graft versus host disease, Rasmussen encephalitis, mitochondrial encephalomyopathy with lactic acidosis and stroke-like episodes (MELAS), Japanese encephalitis, autoimmune glial fibrillary acidic protein (GFAP) astrocytopathy, and primary CNS vasculitis (Figure 2D–F). Scattered IMP3-positive cells were found in the brain parenchyma in three cases, namely MELAS, Japanese encephalomyelitis, and autoimmune GFAP astrocytopathy. However, all cells in these cases were only weakly positive (1+) and were negative for CD20 and CD3, suggesting that they were reactive glial cells.9 Consequently, the proportion of biopsy samples with IMP3-positive and CD20-positive B cells was significantly higher in patients with CNS-DLBCL than in those with inflammatory brain disease (57/57 vs. 0/12; P < 0.0001).
DISCUSSION
IMP3 is generally useful in detecting subtle lesions of aggressive B-cell lymphomas in combination with morphological and other IHC markers. As expected, tumor cells were highlighted by IMP3 in all CNS-DLBCLs. In our cohort, 5.6% (3/54) of patients with CNS-DLBCL required a second biopsy for diagnosis. The first biopsies from these patients that were initially considered CNS-DLBCL-negative or -indeterminate were also positive for IMP3-positive B cells. Together with the absence of IMP3-positive B cells in inflammatory brain diseases, these IMP3-positive B cells were retrospectively suspected to be tumor cells. Combined with other ancillary tests, such as flow cytometry and genetic testing, this may lead to a definitive diagnosis or timely re-biopsy. Our findings suggest that IMP3 may be a useful adjunctive diagnostic marker for CNS-DLBCL when limited or deformed tumor cells make diagnosis difficult.
The mechanism underlying IMP3 expression in CNS-DLBCL remains unknown, but nuclear factor kappa B (NF-κB) pathway activation has been implicated in the expression of IMP3 in non-hematolymphoid malignancies.10 CNS-DLBCL also has tumor characteristics mediated by the NF-κB pathway;11 therefore, IMP3 expression in CNS-DLBCL may also be related to its NF-κB pathway activation. In our study, all CNS-DLBCLs expressed IMP3 in the majority of tumor cells (>80%), whereas, in the study reported by King et al., 15% of DLBCLs (4/26) showed only focal expression (<50%).5 Similar to their findings, 11.8% (8/68) of the DLBCLs other than CNS-DLBCL showed no or low IMP3 expression (<50%). Interestingly, the majority of other DLBCLs with low IMP3 expression were EBV-positive (5/8, 62.5%). Our findings indicate that EBV is a negative factor involved in IMP3 expression in DLBCL, and thus EBV-positive lymphomas may be less sensitive to IMP3.
The differential diagnosis of CNS-DLBCL includes a variety of inflammatory diseases.12,13 Histologically, the inflammatory diseases reviewed in this study harbored differing amounts of inflammatory cells, but all CD20-positive B cells were negative for IMP3. Although the number of negative controls tested in this study was limited, IMP3 expression in reactive lymphocytes other than GCBs has rarely been reported by others and our studies.5–7 This suggests that IMP3 is not only sensitive, but also specific for diagnosing DLBCL in the CNS.
However, there are several limitations to the use of IMP3. First, IMP3 is expressed in various B-cell lymphomas and is not specific in terms of differentiating lymphoma subtypes. Therefore, IMP3 is not suitable for differentiating CNS-DLBCL and CNS involvement by systemic DLBCL. Second, IMP3 positivity is low in low-grade B-cell lymphomas.5 Thus, the possibility of a rare CNS low-grade B-cell lymphoma cannot be ruled out, even if IMP3 is negative. Lastly, IMP3 is expressed in normal germinal centers and is not strictly a malignancy-related marker. Nevertheless, even with these limitations, the fact that most lymphomas arising in the CNS are DLBCLs confirms the usefulness of IMP3. Even if the detected IMP3-positive B cells are quantitatively or qualitatively insufficient for a definitive diagnosis of CNS-DLBCL, IMP3 may be useful as an appropriate indication for timely re-biopsy.
Among CNS-DNBCLs, vitreoretinal DLBCL is especially difficult to diagnose because the amount of tumor cells in biopsy specimens is particularly low. Hence, it is diagnosed using multiple ancillary tests, namely flow cytometry and genetic analysis, in addition to vitreous cytology.14 Nevertheless, its diagnosis remains challenging, as the small number of tumor cells limits the validity of each test and may also cause discrepancies in the results of each test. Although the number of vitreoretinal DLBCL examined in the present study was small, our findings indicate that IMP3 IHC on cell block specimens is a useful additional tool in the diagnosis of vitreoretinal DLBCL, as well as other CNS-DLBCLs.
In conclusion, IMP3 is highly expressed in CNS-DLBCL. However, IMP3 is also expressed in other types of DLBCLs, making it less specific. Most CNS-DLBCL can be diagnosed without performing IHC analysis for IMP3 expression, but it may be useful as an adjunctive tool to differentiate from reactive lesions when tumor cells are few or deformed.
AUTHOR CONTRIBUTIONS
KO and MF drafted the article. KO, MF, HF, MS, TS, YT, SM, YA, and HH acquired and analyzed the pathological and clinical data. KM and MH performed immunostaining. All authors approved the final version of this manuscript for publication.
Footnotes
CONFLICT OF INTEREST
None declared
REFERENCES
- 1.Deckert M, Ferry JA, Paulus W, et al. Primary diffuse large B-cell lymphoma of the CNS. In: WHO Classification of Tumours Editorial Board (ed): Central Nervous System Tumours. WHO Classification of Tumours Series, 5th ed, Volume 6. Lyon, International Agency for Research on Cancer. 2021; pp. 351-355. [Google Scholar]
- 2.Air EL, Leach JL, Warnick RE, McPherson CM. Comparing the risks of frameless stereotactic biopsy in eloquent and noneloquent regions of the brain: a retrospective review of 284 cases. J Neurosurg. 2009; 111: 820-824. [DOI] [PubMed] [Google Scholar]
- 3.Ranheim EA. Pearls and pitfalls in the diagnostic workup of small lymph node biopsies. Mod Pathol. 2019; 32: 38-43. [DOI] [PubMed] [Google Scholar]
- 4.Chabaane M, Amelot A, Riche M, et al. Efficacy of a second brain biopsy for intracranial lesions after initial negativity. J Clin Neurol. 2020; 16: 659-667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.King RL, Pasha T, Roullet MR, Zhang PJ, Bagg A. IMP-3 is differentially expressed in normal and neoplastic lymphoid tissue. Hum Pathol. 2009; 40: 1699-1705. [DOI] [PubMed] [Google Scholar]
- 6.Odani K, Fujimoto M, Hirata M, et al. Immunohistochemistry of the oncofetal protein IMP3 is helpful in the diagnosis of intravascular large B-cell lymphoma using skin biopsy. J Cutan Pathol. 2022; 49: 438-441. [DOI] [PubMed] [Google Scholar]
- 7.Saka M, Fujimoto M, Mizoguchi K, et al. Insulin-like growth factor II mRNA-binding protein 3 is a highly sensitive marker for intravascular large B-cell lymphoma: immunohistochemical analysis of 152 pathology specimens from 88 patients. Am J Surg Pathol. 2024; 48: 671-680. [DOI] [PubMed] [Google Scholar]
- 8.Hans CP, Weisenburger DD, Greiner TC, et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood. 2004; 103: 275-282. [DOI] [PubMed] [Google Scholar]
- 9.Wu C, Ma H, Qi G, Chen F, Chu J. Insulin-like growth factor II mRNA-binding protein 3 promotes cell proliferation, migration and invasion in human glioblastoma. OncoTargets Ther. 2019; 12: 3661-3670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bhargava S, Visvanathan A, Patil V, et al. IGF2 mRNA binding protein 3 (IMP3) promotes glioma cell migration by enhancing the translation of RELA/p65. Oncotarget. 2017; 8: 40469-40485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ferreri AJM, Calimeri T, Cwynarski K, et al. Primary central nervous system lymphoma. Nat Rev Dis Primers. 2023; 9: 29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Deckert M, Brunn A, Montesinos-Rongen M, Terreni MR, Ponzoni M. Primary lymphoma of the central nervous system--a diagnostic challenge. Hematol Oncol. 2014; 32: 57-67. [DOI] [PubMed] [Google Scholar]
- 13.Hajj-Ali RA, Calabrese LH. Diagnosis and classification of central nervous system vasculitis. J Autoimmun. 2014; 48-49: 149-152. [DOI] [PubMed] [Google Scholar]
- 14.Zhang X, Zhang Y, Guan W, et al. Development of diagnostic recommendations for vitreoretinal lymphoma. Ocul Immunol Inflamm. 2023; 1-8. [DOI] [PubMed] [Google Scholar]