Abstract
Importance
The management of hypopharyngeal squamous cell carcinoma (HPSCC) continues to be one of the most formidable challenges in the realm of head and neck oncology.
Objectives
The aim of this meta-analysis was to evaluate the disparity in survival outcomes between upfront surgery and upfront concurrent chemoradiotherapy as the primary treatment modality in patients with HPSCC.
Design
Systemic review with meta-analysis.
Setting
The meta-analysis was conducted in strict accordance with the PRISMA guideline. A literature search through PubMed, EMBASE, and the Cochrane Library were conducted until January 2023. The adjusted hazard ratio (aHR) with 95% confidence intervals (CIs) of different survival outcomes were extracted and pooled.
Participants
Studies that incorporated HPSCC patients without receiving induction chemotherapy.
Interventions
Upfront surgery versus upfront concurrent chemoradiotherapy.
Main Outcome Measures
Overall survival (OS) and disease-free survival (DFS).
Results
Eight studies published between 2015 and 2023, with a pooled patient population of 1619, were included in this meta-analysis. The outcomes reveal that upfront surgery was notably linked with improved OS (aHR 0.66, 95% CI 0.57-0.78) and DFS (aHR 0.75, 95% CI 0.63-0.90). Subgroup analyses were conducted to investigate the impact of the overall stage of the tumor and the extent of surgery on OS. In patients with advanced HPSCC (stages III and IV), upfront surgery remained associated with better OS (aHR 0.65, 95% CI 0.56-0.77). Concerning the extent of surgery, both subgroups exhibited a superior OS outcome associated with upfront surgery (exclusive total laryngopharyngectomy group: aHR 0.54, 95% CI 0.39-0.75; total/partial laryngopharyngectomy group: aHR 0.71, 95% CI 0.59-0.84).
Conclusions and Relevance
The results demonstrated that upfront surgery showed better OS and DFS than concurrent chemoradiation and remind the clinicians of the potential reduction in survival outcomes when choosing concurrent chemoradiotherapy as primary treatment.
Keywords: hypopharyngeal cancer, squamous cell carcinoma, chemoradiotherapy, upfront surgery, HPSCC, CRT
Graphical abstract.
Introduction
Hypopharyngeal squamous cell carcinomas (HPSCC) are relatively rare, constitute approximately 84,000 novel cases, and correspond to a mere 0.4% of the global incidence of new cancer cases in 2020. 1 HPSCC have a worse prognosis compared with the other head and neck cancers due to their late presentation and more propensity for nodal metastasis, with around 80% of cases are diagnosed as locally-advanced diseases upon presentation. 2
The management of HPSCC continues to be one of the most formidable challenges in the realm of head and neck oncology. This is attributed not only to their impact on swallowing function due to the destruction of pharyngeal tissues by the tumor but also to their inclination to invade the larynx, thereby impairing speech and airway functions. Surgical management, typically involving partial or total pharyngectomy and laryngectomy based on disease subsite and the extent of tumor growth, can induce substantial functional morbidity in speech and swallowing. Alternatively, the organ-preserving treatment modalities, particularly chemoradiotherapy-based approaches, have gained significant interest and investigation. Currently, upfront concurrent chemoradiotherapy (CRT) has gained endorsement from both the contemporary NCCN and ESMO guidelines as a well-established strategy, standing alongside the induction chemotherapy approach, for preserving the larynx in surgically-amenable HPSCC.3,4 In terms of overall survival, the non-inferiority of the induction chemotherapy-driven selective larynx preservation approach to the upfront surgery approach was established by the EORTC 24981 trial.5,6 However, as of now, there remains a lack of randomized clinical trial evidence establishing the non-inferiority of the upfront CRT approach to the upfront surgical approach, especially concerning survival outcomes in patients with HPSCC. Physicians have been extrapolating the treatment approach and outcomes from the RTOG 9111 trial—where no significant difference in overall survival was observed among patients with advanced laryngeal cancer in the upfront surgery arm, upfront CRT arm, and induction chemotherapy arm—to the context of surgically-amenable HPSCC, even though the trial did not include patients diagnosed with HPSCC.7,8
Given the complexity of clinical trial design, resource constraints, and current treatment trends, prospective randomized controlled trials providing evidence for the non-inferiority of the upfront CRT approach to the upfront surgery approach, particularly in patients with surgically-amenable HPSCC, are currently lacking and are unlikely to emerge in the future. In light of this, conducting a meta-analysis on retrospective studies addressing this specific topic would be of great value. Prior meta-analyses with similar objectives had incorporated patients treated with induction chemotherapy or those with laryngeal cancer, introducing confounding factors that complicated the interpretation and results of the studies.9-11 To the best of our knowledge, this meta-analysis is the first to assess and quantify the disparity in survival outcomes, specifically comparing upfront surgery with upfront CRT approaches, within the population of patients diagnosed with surgically-amenable HPSCC. In this study, the administration of induction chemotherapy was deliberately excluded for 2 main reasons. First, its non-inferiority to upfront surgery has already been demonstrated through the phase III randomized controlled EORTC 24981 trial.5,6 Secondly, incorporating it into the larynx preservation arm would introduce confounding factors that might compromise the clarity and accuracy of the study results.
Methods
The meta-analysis was meticulously conducted in strict accordance with the guidelines provided by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and registered on PROSPERO (CRD42023460211). 12
Research Inquiry
Does upfront surgery yield significantly-different outcomes in the treatment of HPSCC when comparing to upfront CRT?
PICO Criteria
The following PICO criteria were employed to identify eligible studies:
Patients (P): Patients diagnosed with HPSCC treated with curative intent.
Intervention (I): Upfront surgery as primary treatment modality with or without adjuvant therapy.
Comparison (C): Upfront CRT as primary treatment modality with or without salvage surgery.
Outcome (O): Adjusted hazard ratio (aHR) for overall survival (OS), and disease-free survival (DFS).
Search Strategy
A comprehensive and systematic search was executed across electronic databases, specifically PubMed, EMBASE, and the Cochrane Library, covering studies published from their inception up until January 2023. The search strategy was developed through the incorporation of pertinent keywords such as “hypopharynx” or “hypopharyngeal cancer,” “chemoradiotherapy,” or “CRT,” “surgery,” “survival,” “recurrence,” and “metastasis.” These keywords were combined using appropriate Boolean operators. Complete search details can be found in Supplemental Table S1. Subsequently, a thorough assessment of the full texts of the studies was conducted for inclusion.
Furthermore, the bibliographies of preceding systematic reviews were examined, and eligible studies were scrutinized to pinpoint potentially-relevant reports. In order to ensure the elimination of duplicate articles, the collected studies were imported into a citation manager (Endnote, version X9.3.3, Clarivate Analytics, Philadelphia, PA, USA).
Eligibility Assessment
Two reviewers independently conducted searches within the databases, subsequently evaluating the titles and abstracts of the identified studies. This procedure was executed in a double-blind manner, ensuring that each reviewer remained unaware of the evaluations conducted by the other. Following this initial screening, a comprehensive assessment of the full texts was undertaken, relying on the consensus achieved by the 2 reviewers with respect to the chosen abstracts.
The study selection process was guided by a set of predefined inclusion and exclusion criteria, as outlined below:
Inclusion criteria:
studies that conducted a comparison between the outcomes of upfront surgery and upfront CRT in patients diagnosed with HPSCC and
studies that evaluated primary endpoints encompassing survival outcome(s).
Exclusion criteria:
studies that incorporated patients with HPSCC who underwent induction chemotherapy;
studies that encompassed patients with HPSCC exhibiting distant metastasis at the time of initial diagnosis;
studies not published in the English language; and
studies that lacked the provision of relevant information.
Data Extraction
Data extraction was executed independently by the 2 investigators. The data ensured encompassed the following elements: primary author, publication year, period of patient recruitment, study methodology, sample size, age distribution, gender distribution, cancer staging, geographic location, aHR accompanied by its corresponding 95% confidence interval (CI) for the primary endpoints of OS and DFS, duration of follow-up, proportion of cases involving adjuvant therapy, and particulars pertaining to the surgical technique employed.
Data Synthesis
Upon retrieval, the data underwent both qualitative and quantitative analyses. The demographic and interventional characteristics of all included studies were collated and presented in tabular form and subsequently subjected to a detailed analysis. Dichotomous outcomes, such as OS and DFS, were expressed as aHR with CI. The aforementioned outcomes were then subjected to a meta-analysis. In cases where there was a overlap in the study populations, preference was given to the more recent studies for data synthesis.
Appraisal of Study Quality
The evaluation of bias was conducted utilizing the Newcastle-Ottawa Scale for the studies encompassed in the analysis. This assessment tool encompassed 3 key domains, specifically selection, comparability, and outcome, together comprising a total of 8 inquiries. Using the established scoring framework, the studies were stratified into 3 categories, reflecting their quality levels. To elaborate, studies attaining a score of ≤3 were designated as low quality, those attaining a score between 4 and 6 were categorized as fair quality, and studies earning a score of ≥7 were recognized as high quality. 13
Statistical Analysis
The current meta-analysis was carried out utilizing the Comprehensive Meta-Analysis software, version 4 (Biostat, Englewood, NJ, USA). To compare the impact of distinct treatment on survival outcomes, aHRs accompanied by their corresponding 95% CIs were synthesized using a random-effects model. This model was chosen to distribute the influence of heterogeneity evenly across the diverse studies incorporated in the analysis.
The presence of heterogeneity among studies was assessed through both the Cochran Q-statistic and I 2 tests. Notably, substantial heterogeneity was defined as a Q-test P-value of <.05 or an I 2 value exceeding 50%.
In order to ascertain the potential for publication bias, funnel plots and Egger’s linear regression test were employed, with a significance threshold set at P < .05. If the results from Egger’s test suggested the presence of publication bias, the trim-and-fill method was employed to adjust for this bias.
All statistical analyses were performed with two-tailed tests, and statistical significance was defined as P < .05.
Results
Literature Search and Study Identification
The initial exploration of databases yielded a total of 902 entries, with contributions of 544 from PubMed, 91 from Cochrane, and 267 from Embase. An overview of the selection progression is visualized in Figure 1, and the search methodology and algorithm are presented in Supplemental Table S1.
Figure 1.
PRISMA flow diagram.
Following the removal of duplicates, 496 studies were excluded based on title, and an additional 107 were excluded based on abstract assessments. Subsequently, 29 studies underwent a comprehensive review of their full texts, leading to the exclusion of 15 studies due to specific reasons as outlined in Supplemental Table S2.14-28 Ultimately, 8 studies were deemed suitable for qualitative analysis and qualified for quantitative analysis.29-36
Characteristics of the Included Studies
Table 1 presents a concise overview of the studies that have been incorporated into this meta-analysis. Notably, all the studies encompassed in this analysis exhibited a retrospective research design. These studies have been conducted across various nations, with 3 originating from Asian countries (Taiwan, China, and Korea), 3 from the United States, 1 from Germany, and another from the Netherlands. The span of publication dates for these studies extends over a 9 year period, ranging from 2015 to 2023.
Table 1.
Eligible Study Characteristics.
| Study | Year | Region | Database | Recruitment period | Prospective/retrospective cohort study | Provided outcomes | Median follow-up (months) |
|---|---|---|---|---|---|---|---|
| Thomas 31 | 2020 | USA | Single institution | 1994-2018 | Retrospective | aHR for OS, DFS | 17 |
| Tsai 32 | 2020 | Taiwan | Single institution | 2003-2012 | Retrospective | aHR for OS, DFS | 30.6 |
| Tassler 33 | 2019 | USA | Single institution | 1994-2014 | Retrospective | aHR for OS | 38 |
| Petersen 34 | 2019 | Netherland | Single institution | 1990-2013 | Retrospective | aHR for OS | NA |
| Francesco 35 | 2016 | Italy | Dual institution | 2004-2011 | Retrospective | aHR for OS | 30.3 |
| Harris 36 | 2015 | USA | Single institution | 1999-2013 | Retrospective | aHR for OS, DFS | 17 |
| Huang 30 | 2023 | China | Single institution | 2010-2018 | Retrospective | aHR for OS, DFS | 48 |
| Jang 29 | 2016 | Korea | Single institution | 1996-2014 | Retrospective | aHR for OS | 19 |
Abbreviations: aHR, adjusted hazard ratio; DFS, disease-free survival; NA, not available; OS, overall survival.
Collectively, the analysis encompassed 1619 patients, who were recruited for these studies within a recruitment timeline spanning from 1994 to 2018. The mean age of the patients ranged from 59 to 63.6 years, and the male population constituted a proportion ranging from 77% to 98.7%. The duration of follow-up ranged from 17 to 48 months.
Among the studies, 7 included patients with HPSCC presenting T1-T4 tumors, while 1 specifically examined T2-T4 tumors. Furthermore, 7 studies comprised patients with N0-N3 tumors, whereas 1 concentrated on N0-N2 tumors. Regarding the overall stage of the disease, 7 studies focused on patients with HPSCC presenting only stages III-IV, while 1 reported HPSCC patients with stages I-IV. Detailed information pertaining to the clinicopathological attributes of the patients can be found in Table 2.
Table 2.
Patient Clinicopathological Characteristics of Eligible Studies.
| Study | Age (year) Mean |
Male (%) | T stage | N stage | Stage | Relevant case number | Case number (CRT) | Case number (surgery) | Surgical technique | Adjuvant therapy in surgery group |
|---|---|---|---|---|---|---|---|---|---|---|
| Thomas 31 | 60.3 | 92 | 1-4 | 0-3 | 3-4 | 103 | 72 | 31 | TLP | 26/31 (83.8%) |
| Tsai 32 | 53 | 98 | 1-4 | 0-3 | 3-4 | 652 | 439 | 213 | TLP/PLP | 151/213 (70.8%) |
| Tassler 33 | 61 | 81.7 | 1-4 | 0-3 | 3-4 | 137 | 56 | 81 | TLP | 58/81 (71.6%) |
| Petersen 34 | 59 | 82.6 | 2-4 | 0-3 | 3-4 | 52 | 26 | 26 | TLP | NA |
| Francesco 35 | 61 (median) | 87 | 1-4 | 0-3 | 1-4 | 123 | 58 | 65 | TLP/PLP | 63/65 (96.9%) |
| Harris 36 | 63.6 | 77 | 1-4 | 0-3 | 3-4 | 76 | 48 | 28 | TLP | 28/28 (100%) (CRT 6, RT 22) |
| Huang 30 | NA | 98.7 | 1-4 | 0-3 | 3-4 | 299 | 188 | 111 | TLP/PLP | 111/111 (100%) (CRT 41, RT 70) |
| Jang 29 | NA | 95.4 | 1-4 | 0-2 | 3-4A | 177 | 107 | 70 | TLP/PLP | 70/70 (100%) (CRT 20, RT 50) |
Quality Assessment
The present meta-analysis incorporated a comprehensive selection of 8 studies, each of which underwent meticulous evaluation concerning their methodological rigor using the Newcastle-Ottawa scale. The outcomes of this assessment revealed that 5 out of the eligible studies achieved scores equal to or exceeding 7 points, indicating fair quality across the majority of the studies. The remaining 3 studies received 6 points on the scale. Figure 2 illustrates more detailed insights regarding the studies encompassed in this analysis.
Figure 2.
Quality assessment of included studies by NOS. NOS, Newcastle-Ottawa Scale.
Meta-Analysis Results
Overall survival
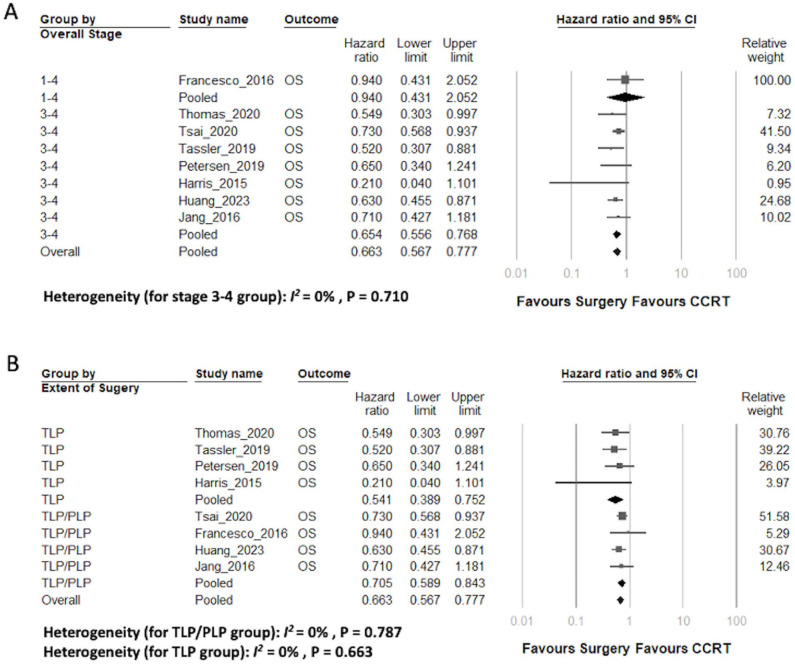
A total of 8 studies furnished aHRs pertinent to OS. The outcomes derived from the meta-analysis demonstrated a significant association, revealing that upfront surgery was notably linked with improved OS (aHR 0.66, 95% CI 0.57-0.78).29-36 This conclusion emerged with a minimal degree of heterogeneity (I 2 = 0%, assessed using a random-effects model, P = .71; Figure 3A). While the overall heterogeneity was found to be low, we conducted subgroup analyses to investigate the impact of the overall stage of the tumor and the extent of surgery on OS as depicted in Figure 4. In studies focused on patients with advanced HPSCC in stages III-IV, upfront surgery continued to be associated with a significant improvement in OS (aHR 0.65, 95% CI 0.56-0.77; Figure 4A).29-34,36 Concerning the extent of surgery, both subgroups, including the exclusive total laryngopharyngectomy group and the total/partial laryngopharyngectomy group, exhibited a superior OS outcome associated with upfront surgery (exclusive total laryngopharyngectomy group31,33,34,36: aHR 0.54, 95% CI 0.39-0.75; total/partial laryngopharyngectomy group29,30,32,35: aHR 0.71, 95% CI 0.59-0.84; Figure 4B).
Figure 3.
Forest plot of the meta-analysis regarding (A) OS and (B) DFS. A random-effects model was applied. OS, overall survival; DFS, disease-free survival.
Figure 4.
Subgroup analyses on OS (A) grouped by overall stage and (B) grouped by the extent of surgery. A random-effects model was applied. OS, overall survival; TLP, total laryngopharyngectomy; PLP, partial laryngopharyngectomy.
To scrutinize the potential presence of publication bias, both funnel plots and Egger’s test were undertaken (Supplemental Figure S1). Notably, the results from Egger’s test indicated a lack of substantial publication bias in the OS analysis (P = .20). The outcomes of the sensitivity analysis indicated that the exclusion of any one of the included studies did not exert any influence on the results of the meta-analysis when assessing the association between the treatment and OS, as illustrated in Figure 5. This finding underscores a feasible reliability of our study’s results.
Figure 5.
Sensitivity analysis for different treatment on OS analysis. OS, overall survival.
Disease-free survival
Incorporating data from 4 studies that offered aHRs pertinent to DFS, the outcomes of the meta-analysis unveiled a significant and affirmative correlation.30-32,36 Specifically, the meta-analysis showcased that upfront surgery was also notably linked to improved DFS (aHR 0.75, 95% CI 0.63-0.90). This result was established with minimal heterogeneity (I 2 = 0%, evaluated using a random-effects model, P = .40; Figure 3B).
Discussion
This meta-analysis synthesized data from 8 studies, encompassing 1619 patients diagnosed with HPSCC. With the exception of 1 study 35 , all the studies incorporated in this analysis focused exclusively on patients with locally-advanced HPSCC (stages III and IV). In Francesco et al, out of a total of 123 patients, only 12 patients (10%) were categorized as early stage (stages I and II), accounting for a mere 0.7% of the total population in this meta-analysis. 35 Consequently, the outcomes of this meta-analysis are most pertinent to patients with locally-advanced HPSCC tumors. The findings consistently indicated a statistically-significant association between enhanced overall survival and upfront surgery, in comparison with upfront CRT. The combined outcomes revealed almost a one-third reduction in the risk of mortality for patients undergoing upfront surgery compared with those undergoing upfront CRT. Upon exclusion of the only study 35 encompassing patients with early-stage (stages I and II) patients with HPSCC, the result exhibited a similar or even minimal decreased of aHR of 0.65. This effect was even more pronounced when considering the 4 studies exclusively incorporating patients received total laryngopharyngectomy, yielding a pooled aHR of 0.54. The findings may imply that in HPSCC with a larger extent of tumor growth was associated with an increased risk of mortality when employing upfront CRT, as opposed to upfront surgery. The superiority of upfront surgery over upfront CRT is reinforced by significantly-better-pooled DFS, as evident from pooling aHRs from the 4 studies reporting DFS. Moreover, minimal heterogeneity in both OS and DFS analyses implies highly-consistent results across the published studies. In our opinion, DFS may serve as an additional indicator of cancer control and treatment effectiveness in patients with HPSCC. The presence of the disease in these patients can lead to various symptoms such as pain, dysphagia, and breathing difficulties, significantly impacting their physical and psychological well-being. Using DFS as a study endpoint allows for a more comprehensive assessment of any disease recurrence, in addition to evaluating survival status alone, thus complementing the sole measurement of survival outcomes.
It is worth noting that 2 previous meta-analyses comparing treatments for hypopharyngeal cancer did not identify significant differences in outcomes between primary surgery and primary nonsurgical treatments, a finding that diverges from the conclusions drawn from the current meta-analysis.9,10 The discrepancy may stem from the inclusion of patients undergoing induction chemotherapy in the previous meta-analyses. The inclusion of induction chemotherapy might potentially introduce significant selection bias into the comparisons of treatment outcomes between primary surgery and primary chemoradiotherapy in treating hypopharyngeal carcinoma. For instance, patients with poorer responses to induction chemotherapy, indicative of more aggressive cancers, might have been assigned to the surgical arm, while those with favorable responses might have been allocated to the nonsurgical arm. Cui et al’s meta-analysis examined the comparative effectiveness of initial surgical versus nonsurgical treatments for advanced HPSCC and incorporated 17 studies, with 15 of them including patients receiving induction chemotherapy. 10 Another previously-published meta-analysis by Panda et al exclusively included randomized controlled trials and concluded that there was no significant difference in survival outcomes between the non-organ preservation arm and the organ preservation arm. 9 However, this conclusion was drawn from 2 randomized controlled trials that incorporated induction chemotherapy in their trial designs, differing significantly from the studies we included.6,37 The induction chemotherapy included in these previous meta-analyses appears to have a significant discrepancy in regimens and dosage. To eliminate this potential bias, the current study decided to compare the treatment outcomes in the studies without any induction chemotherapy. Moreover, the exclusion of studies with induction chemotherapy could lead to differences in the numbers of included T4 tumors, which might also affect the results of the meta-analyses. The proportion of T4 patients was only 30% (654 out of 2145), compared with 46% (735 out of 1578) in the current meta-analysis; however, the data were lacked in up to 3 eligible studies in Cui et al’s meta-analysis. 10 It is worth mentioning that there was no randomized controlled trial that directly compared upfront surgery against upfront CRT identified in both the meta-analyses. Panda et al had identified a randomized controlled trial reported by Prades et al demonstrating significantly-better laryngeal preservation, with similar overall survival or locoregional control outcomes, in the chemoradiotherapy group compared with the group undergoing an induction chemotherapy-driven selective larynx preservation strategy among patients with T3 piriform sinus squamous cell carcinoma. 38 However, in this study, pretreatment neck dissection was performed in all participants, and this approach was not consistent with the treatment protocols of other contemporary studies. As of now, there remains a lack of solid evidence establishing the non-inferiority of the upfront CRT approach compared with either upfront surgery or induction chemotherapy-driven selective larynx preservation strategy.
The current meta-analysis, to our knowledge, is the first to explore and quantify survival outcome disparities between treatment choices within studies directly comparing upfront surgery against upfront CRT, particularly within the realm of patients with locally-advanced HPSCC. Despite the retrospective nature of all studies encompassed within this meta-analysis, its findings remain the most recent evidence available for addressing the highly-specific inquiry into the relative efficacy of upfront surgery versus upfront CRT, specifically concerning survival outcomes in cases with locally-advanced HPSCC tumors.
To provide a practical perspective from real-world clinical practice, the observed decline in survival outcomes in the upfront CRT group, compared with the upfront surgery group, may be explained by several factors. Patients with locally-advanced HPSCC tumors treated by upfront CRT as the primary modality seem to have a high probability of residual disease (around 20%) after the treatment and a notable rate of locoregional recurrences (around 30%) among those who initially achieve complete remission. 2 Other factors include delayed timing of salvage surgery and heightened surgical technical complexity in post-chemoradiation salvage surgery.39,40 Collectively, these factors contribute to a low salvageable rate (15%-30%), an increased incidence of postoperative complications (40%-70%), and finally unfavorable survival outcomes by this treatment approach (5 year overall survival at 20%-30%).14,39,40 These facts were particularly pronounced in the context of HPSCC tumors, as opposed to laryngeal cancers. 39
Limitations of the Study
The current study has several limitations. Firstly, there is a potential for publication bias, as positive results tend to be featured in the literature. Secondly, the pool of eligible studies was limited. To concentrate our analysis on comparing survival outcomes between upfront surgery and upfront CRT in patients with HPSCC, we excluded studies that contained patients with laryngeal squamous cell carcinomas or those who underwent organ preservation treatments other than upfront CRT (Supplemental Table S2). Lastly, it is worth noting that all the studies included in our analysis were retrospective in nature, which may lead to the variety in the quality of recorded clinicopathological factors. For example, margin status, a crucial clinicopathological factor in the upfront surgical group, was only reported in 1 study by Francesco et al. 35 The authors observed a 7.7% (5/65) rate of close/positive margins. Neck dissection is another critical factor in the surgical group. In the current meta-analysis, 5 studies mentioned that neck dissection was performed in both clinically-node-negative and clinically-node-positive patients, while 1 study indicated that all patients in the surgery group underwent modified radical neck dissections. However, the remaining studies did not provide any information about neck dissection. Another potential bias is that the surgeries in the included studies were all conducted by high-volume academic centers. High-volume surgeons may potentially achieve better outcomes. On the other hand, in the CRT group, there may be significant heterogeneity in treatment practices across different institutions. For instance, variations exist in radiotherapy dosage and the utilization of different radiotherapy techniques, such as conventional opposed lateral fields, 3D-conformal radiotherapy, or intensity-modulated radiotherapy, among the studies included. Additionally, specific details regarding treatment fields were not consistently provided. Concerning concurrent chemotherapy, the administration of cisplatin, carboplatin, paclitaxel, or cetuximab varies among the studies included. The diversity in these clinical factors across the eligible studies could potentially contribute to the observed heterogeneity in the current meta-analysis (Supplemental Table S3).
Conclusions
The current study demonstrated that patients receive upfront surgery exhibited superior OS (aHR 0.66, 95% CI 0.57-0.78) and DFS (aHR 0.75, 95% CI 0.63-0.90) comparing to upfront CRT. The results remained even more significant when we focused on patients with advanced HPSCC (stages III-IV) and patient groups exclusively undergoing total laryngopharyngectomy. The results of the current study could remind clinicians of the potential reduction in survival outcomes when choosing upfront CRT as a treatment modality as opposed to upfront surgery during the treatment consultation.
Supplemental Material
Supplemental material, sj-docx-1-ohn-10.1177_19160216241293633 for Upfront Surgery Versus Upfront Concurrent Chemoradiotherapy as Primary Modality in Hypopharyngeal Squamous Cell Carcinoma: A Systematic Review and Meta-Analysis by Tsung-You Tsai, Wing-Keen Yap, Ting-Hao Wang, Yi-An Lu, Anna See, Yu-Feng Hu, Yenlin Huang, Huang-Kai Kao and Kai-Ping Chang in Journal of Otolaryngology - Head & Neck Surgery
Supplemental material, sj-tif-2-ohn-10.1177_19160216241293633 for Upfront Surgery Versus Upfront Concurrent Chemoradiotherapy as Primary Modality in Hypopharyngeal Squamous Cell Carcinoma: A Systematic Review and Meta-Analysis by Tsung-You Tsai, Wing-Keen Yap, Ting-Hao Wang, Yi-An Lu, Anna See, Yu-Feng Hu, Yenlin Huang, Huang-Kai Kao and Kai-Ping Chang in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Author Contributions: T.-Y.T.: study design, data acquisition, quality control of data and algorithms, data analysis and interpretation, statistical analysis, manuscript preparation, manuscript editing, and manuscript review. W.-K.Y.: study design, data acquisition, quality control of data and algorithms, data analysis and interpretation, manuscript preparation, manuscript editing, and manuscript review. T.-H.W.: quality control of data and algorithms, data analysis and interpretation, manuscript preparation, manuscript editing, and manuscript review. Y.-A.L., Y.-F.H., and A.S.: manuscript preparation, manuscript editing, and manuscript review. Y.H.: data analysis and interpretation. H.-K.K.: manuscript editing, manuscript review. K.-P.C.: study design, study concept, data analysis and interpretation, manuscript preparation, manuscript editing, manuscript review.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant (Kai-PingChang,CMRPG3N0832) from Chang Gung Memorial Hospital, Taiwan.
ORCID iDs: Wing-Keen Yap  https://orcid.org/0000-0001-9302-1377
https://orcid.org/0000-0001-9302-1377
Kai-Ping Chang  https://orcid.org/0000-0003-1777-9578
https://orcid.org/0000-0003-1777-9578
Supplemental Material: Additional supporting information is available in the online version of the article.
References
- 1. Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209-249. doi: 10.3322/caac.21660 [DOI] [PubMed] [Google Scholar]
- 2. Hall SF, Groome PA, Irish J, O’Sullivan B. The natural history of patients with squamous cell carcinoma of the hypopharynx. Laryngoscope. 2008;118(8):1362-1371. doi: 10.1097/MLG.0b013e318173dc4a [DOI] [PubMed] [Google Scholar]
- 3. Machiels JP, René Leemans C, Golusinski W, Grau C, Licitra L, Gregoire V. Squamous cell carcinoma of the oral cavity, larynx, oropharynx and hypopharynx: EHNS-ESMO-ESTRO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2020;31(11):1462-1475. doi: 10.1016/j.annonc.2020.07.011 [DOI] [PubMed] [Google Scholar]
- 4. National Comprehensive Cancer Network. Head and Neck Cancers (Version 2.2023). Accessed August 25, 2023. https://www.nccn.org/professionals/physician_gls/pdf/head-and-neck.pdf
- 5. Lefebvre JL, Chevalier D, Luboinski B, Kirkpatrick A, Collette L, Sahmoud T. Larynx preservation in pyriform sinus cancer: preliminary results of a European Organization for Research and Treatment of Cancer phase III trial. EORTC Head and Neck Cancer Cooperative Group. J Natl Cancer Inst. 1996;88(13):890-899. doi: 10.1093/jnci/88.13.890 [DOI] [PubMed] [Google Scholar]
- 6. Lefebvre JL, Andry G, Chevalier D, et al. Laryngeal preservation with induction chemotherapy for hypopharyngeal squamous cell carcinoma: 10-year results of EORTC trial 24891. Ann Oncol. 2012;23(10):2708-2714. doi: 10.1093/annonc/mds065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Forastiere AA, Goepfert H, Maor M, et al. Concurrent chemotherapy and radiotherapy for organ preservation in advanced laryngeal cancer. N Engl J Med. 2003;349(22):2091-2098. doi: 10.1056/NEJMoa031317 [DOI] [PubMed] [Google Scholar]
- 8. Forastiere AA, Zhang Q, Weber RS, et al. Long-term results of RTOG 91-11: a comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. J Clin Oncol. 2013;31(7):845-852. doi: 10.1200/jco.2012.43.6097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Panda S, Sakthivel P, Gurusamy KS, Sharma A, Thakar A. Treatment options for resectable hypopharyngeal squamous cell carcinoma: a systematic review and meta-analysis of randomized controlled trials. PLoS One. 2022;17(11):e0277460. doi: 10.1371/journal.pone.0277460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Cui J, Wang L, Piao J, et al. Initial surgical versus non-surgical treatments for advanced hypopharyngeal cancer: a meta-analysis with trial sequential analysis. Int J Surg. 2020;82:249-259. doi: 10.1016/j.ijsu.2020.04.059 [DOI] [PubMed] [Google Scholar]
- 11. Che J, Wang Y, Zhang X, Chen J. Comparative efficacy of six therapies for hypopharyngeal and laryngeal neoplasms: a network meta-analysis. BMC Cancer. 2019;19(1):282. doi: 10.1186/s12885-019-5412-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. McInnes MDF, Moher D, Thombs BD, et al. Preferred reporting items for a systematic review and meta-analysis of diagnostic test accuracy studies: the PRISMA-DTA statement. JAMA. 2018;319(4):388-396. doi: 10.1001/jama.2017.19163 [DOI] [PubMed] [Google Scholar]
- 13. Wells G, Shea B, O’Connell J. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa Health Research Institute Web Site. Accessed April 7, 2023. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
- 14. Chung EJ, Jeong WJ, Jung YH, et al. Long-term oncological and functional outcomes of induction chemotherapy followed by (chemo)radiotherapy vs definitive chemoradiotherapy vs surgery-based therapy in locally advanced stage III/IV hypopharyngeal cancer: multicenter review of 266 cases. Oral Oncol. 2019;89:84-94. doi: 10.1016/j.oraloncology.2018.12.015 [DOI] [PubMed] [Google Scholar]
- 15. Okamoto M, Takahashi H, Yao K, Inagi K, Nakayama M, Nagai H. Clinical impact of using chemoradiotherapy as a primary treatment for hypopharyngeal cancer. Acta Otolaryngol Suppl. 2002;(547):11-14. doi: 10.1080/000164802760057491 [DOI] [PubMed] [Google Scholar]
- 16. Demirci U, Ozmen OA, Demiroz C, et al. An evaluation of the results of chemoradiotherapy and surgical treatment for hypopharyngeal cancer. Kulak Burun Bogaz Ihtis Derg. 2013;23(5):275-81. doi: 10.5606/kbbihtisas.2013.65477 [DOI] [PubMed] [Google Scholar]
- 17. Rades D, Schroeder U, Bajrovic A, Schild SE. Radiochemotherapy versus surgery plus radio(chemo)therapy for stage T3/T4 larynx and hypopharynx cancer—results of a matched-pair analysis. Eur J Cancer. 2011;47(18):2729-2734. doi: 10.1016/j.ejca.2011.06.013 [DOI] [PubMed] [Google Scholar]
- 18. Iwae S, Fujii M, Hayashi R, et al. Matched-pair analysis of patients with advanced hypopharyngeal cancer: surgery versus concomitant chemoradiotherapy. Int J Clin Oncol. 2017;22(6):1001-1008. doi: 10.1007/s10147-017-1151-9 [DOI] [PubMed] [Google Scholar]
- 19. Al-Mamgani A, Navran A, Walraven I, Schreuder WH, Tesselaar MET, Klop WMC. Organ-preservation (chemo)radiotherapy for T4 laryngeal and hypopharyngeal cancer: is the effort worth? Eur Arch Otorhinolaryngol. 2019;276(2):575-583. doi: 10.1007/s00405-018-5241-8 [DOI] [PubMed] [Google Scholar]
- 20. Luo X, Huang X, Liu S, et al. Response-adapted treatment following radiotherapy in patients with resectable locally advanced hypopharyngeal carcinoma. JAMA Netw Open. 2022;5(2):e220165. doi: 10.1001/jamanetworkopen.2022.0165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Hirano S, Tateya I, Kitamura M, et al. Ten years single institutional experience of treatment for advanced hypopharyngeal cancer in Kyoto University. Acta Otolaryngol Suppl. 2010;130(563):56-61. doi: 10.3109/00016489.2010.487495 [DOI] [PubMed] [Google Scholar]
- 22. Shirai K, Saitoh JI, Musha A, et al. Clinical outcomes of definitive and postoperative radiotherapy for stage I-IVB hypopharyngeal cancer. Anticancer Res. 2016;36(12):6571-6578. doi: 10.21873/anticanres.11261 [DOI] [PubMed] [Google Scholar]
- 23. Lee MS, Ho HC, Hsiao SH, Hwang JH, Lee CC, Hung SK. Treatment results and prognostic factors in locally advanced hypopharyngeal cancer. Acta Otolaryngol. 2008;128(1):103-109. doi: 10.1080/00016480701387116 [DOI] [PubMed] [Google Scholar]
- 24. Leon X, Lopez M, Garcia J, Rovira C, Casasayas M, Quer M. Patients with locally advanced hypopharyngeal carcinoma. Results over a 30-year period [Pacientes con carcinoma localmente avanzado de hipofaringe. Resultados a lo largo de un periodo de 30 anos]. Acta Otorrinolaringol Esp (Engl Ed). 2019;70(6):315-326. doi: 10.1016/j.otorri.2018.07.001 [DOI] [PubMed] [Google Scholar]
- 25. Soo KC, Tan EH, Wee J, et al. Surgery and adjuvant radiotherapy vs concurrent chemoradiotherapy in stage III/IV nonmetastatic squamous cell head and neck cancer: a randomised comparison. Br J Cancer. 2005;93(3):279-286. doi: 10.1038/sj.bjc.6602696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Lin YH, Hsiao JR, Wu YH, et al. Distinct failure patterns in hypopharyngeal cancer patients receiving surgery-based versus radiation-based treatment. Ann Surg Oncol. 2023;30(2):1169-1181. doi: 10.1245/s10434-022-12744-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Scherl C, Mantsopoulos K, Semrau S, et al. Management of advanced hypopharyngeal and laryngeal cancer with and without cartilage invasion. Auris Nasus Larynx. 2017;44(3):333-339. doi: 10.1016/j.anl.2016.08.002 [DOI] [PubMed] [Google Scholar]
- 28. Huang WY, Jen YM, Chen CM, et al. Intensity modulated radiotherapy with concurrent chemotherapy for larynx preservation of advanced resectable hypopharyngeal cancer. Radiat Oncol. 2010;5:37. doi: 10.1186/1748-717x-5-37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Jang JY, Kim EH, Cho J, et al. Comparison of oncological and functional outcomes between initial surgical versus non-surgical treatments for hypopharyngeal cancer. Ann Surg Oncol. 2016;23(6):2054-2061. doi: 10.1245/s10434-016-5088-4 [DOI] [PubMed] [Google Scholar]
- 30. Huang Z, Zhang X, Zhang Y, et al. Survival and long-term quality-of-life of resectable stage III/IV hypopharyngeal carcinoma treated between concurrent chemoradiotherapy and surgery followed by (concurrent chemo)radiotherapy. Asian J Surg. 2023;46:3693-3699. doi: 10.1016/j.asjsur.2022.12.155 [DOI] [PubMed] [Google Scholar]
- 31. Vengaloor Thomas T, Nittala MR, Bhanat E, Albert AA, Vijayakumar S. Management of advanced-stage hypopharyngeal carcinoma: 25-year experience from a Tertiary Care Medical Center. Cureus. 2020;12(1):e6679. doi: 10.7759/cureus.6679 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Tsai YT, Chen WC, Chien CY, et al. Treatment patterns and survival outcomes of advanced hypopharyngeal squamous cell carcinoma. World J Surg Oncol. 2020;18(1):82. doi: 10.1186/s12957-020-01866-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Tassler AB, Gooding WE, Ferris RL. Hypopharyngeal cancer treatment: does initial surgery confer survival benefit? Head Neck. 2019;41(7):2167-2173. doi: 10.1002/hed.25687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Petersen JF, Arends CR, van der Noort V, et al. Laryngo-esophageal dysfunction free survival and propensity score matched analysis comparing organ preservation and total laryngectomy in hypopharynx cancer. Oral Oncol. 2019;95:143-149. doi: 10.1016/j.oraloncology.2019.06.018 [DOI] [PubMed] [Google Scholar]
- 35. Bussu F, Mura F, Miccichè F, et al. Oncologic outcome of hypopharyngeal carcinoma treated with different modalities at 2 different university hospitals. Head Neck. 2016;38(4):606-612. doi: 10.1002/hed.23938 [DOI] [PubMed] [Google Scholar]
- 36. Harris BN, Biron VL, Donald P, et al. Primary surgery vs chemoradiation treatment of advanced-stage hypopharyngeal squamous cell carcinoma. JAMA Otolaryngol Head Neck Surg. 2015;141(7):636-640. doi: 10.1001/jamaoto.2015.0659 [DOI] [PubMed] [Google Scholar]
- 37. Beauvillain C, Mahé M, Bourdin S, et al. Final results of a randomized trial comparing chemotherapy plus radiotherapy with chemotherapy plus surgery plus radiotherapy in locally advanced resectable hypopharyngeal carcinomas. Laryngoscope. 1997;107(5):648-653. doi: 10.1097/00005537-199705000-00017 [DOI] [PubMed] [Google Scholar]
- 38. Prades JM, Lallemant B, Garrel R, et al. Randomized phase III trial comparing induction chemotherapy followed by radiotherapy to concomitant chemoradiotherapy for laryngeal preservation in T3M0 pyriform sinus carcinoma. Acta Otolaryngol. 2010;130(1):150-155. doi: 10.3109/00016480902914080 [DOI] [PubMed] [Google Scholar]
- 39. Putten L, Bree R, Doornaert PA, et al. Salvage surgery in post-chemoradiation laryngeal and hypopharyngeal carcinoma: outcome and review. Acta Otorhinolaryngol Ital. 2015;35(3):162-172. [PMC free article] [PubMed] [Google Scholar]
- 40. Garneau JC, Bakst RL, Miles BA. Hypopharyngeal cancer: a state of the art review. Oral Oncol. 2018;86:244-250. doi: 10.1016/j.oraloncology.2018.09.025 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-docx-1-ohn-10.1177_19160216241293633 for Upfront Surgery Versus Upfront Concurrent Chemoradiotherapy as Primary Modality in Hypopharyngeal Squamous Cell Carcinoma: A Systematic Review and Meta-Analysis by Tsung-You Tsai, Wing-Keen Yap, Ting-Hao Wang, Yi-An Lu, Anna See, Yu-Feng Hu, Yenlin Huang, Huang-Kai Kao and Kai-Ping Chang in Journal of Otolaryngology - Head & Neck Surgery
Supplemental material, sj-tif-2-ohn-10.1177_19160216241293633 for Upfront Surgery Versus Upfront Concurrent Chemoradiotherapy as Primary Modality in Hypopharyngeal Squamous Cell Carcinoma: A Systematic Review and Meta-Analysis by Tsung-You Tsai, Wing-Keen Yap, Ting-Hao Wang, Yi-An Lu, Anna See, Yu-Feng Hu, Yenlin Huang, Huang-Kai Kao and Kai-Ping Chang in Journal of Otolaryngology - Head & Neck Surgery