Abstract
Background
Human Papillomavirus (HPV) infection has garnered significant attention due to its high prevalence and association with various cancers and other health conditions. Composite lifestyle factors may influence the risk of HPV infection, yet their cumulative impact remains insufficiently explored. This study aims to explore the association between the Life’s Essential 8 (LE8) Score and HPV infection status, highlighting the potential role of lifestyle and health behaviors in HPV infection prevention.
Methods
Utilizing data from the National Health and Nutrition Examination Survey (NHANES) spanning 2005–2016, we analyzed the health and nutritional statuses of 6,773 participants after excluding those with missing HPV infection status, inability to calculate the LE8 Score, and missing covariate data. The LE8 Score was computed based on eight cardiovascular health metrics, encompassing both health factors (BMI, non-HDL cholesterol, blood pressure, and blood glucose) and health behaviors (physical activity, diet, sleep duration, and nicotine exposure). HPV infection status was determined through vaginal swab specimens analyzed using various Roche assays. Multivariate logistic regression, the restricted cubic splines (RCS) analysis and weighted quantile sum (WQS) regression were employed to assess the association between LE8 Score and HPV infection risk.
Results
Our findings indicate a significant inverse association between the LE8 Score and HPV infection risk. Participants with medium and high LE8 Scores exhibited a 21% and 31% lower risk of HPV infection, respectively, compared to those with low LE8 Scores in multivariate logistic regression models. The analysis also revealed that lifestyle factors, particularly nicotine exposure and blood pressure, significantly contributed to the observed association.
Conclusion
The study underscores the importance of healthy lifestyle behaviors in reducing the risk of HPV infection. Public health strategies promoting such behaviors could complement existing HPV prevention measures, potentially lowering the burden of HPV-related diseases. Future research should further investigate the mechanisms underlying this association and the effectiveness of lifestyle interventions in diverse populations.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12889-024-20546-1.
Keywords: Life’s essential 8, Cardiovascular health (CVH), HPV Infection, Lifestyle Behaviors, Public health strategies, NHANES
Introduction
The interplay between lifestyle factors and infectious diseases has garnered significant attention in the realm of public health research. Among these, Human Papillomavirus (HPV) infection stands out due to its prevalence and its association with various cancers and other health conditions [1]. HPV is the most common sexually transmitted infection worldwide, with an estimated 79 million individuals currently infected in the United States alone [2]. It is responsible for nearly all cases of cervical cancer and a significant proportion of other anogenital and oropharyngeal cancers [3].
While vaccination programs have shown promise in reducing HPV infection rates, particularly among younger populations, understanding the role of modifiable lifestyle factors in HPV infection risk remains crucial for comprehensive prevention strategies [4]. Recent studies have suggested that various lifestyle elements, including diet, physical activity, and smoking status, may influence susceptibility to HPV infection and persistence [5, 6].
The LE8 Score, encompassing eight critical Cardiovascular Health (CVH) indicators, including four health factors (Body Mass Index [BMI], non-High-Density Lipoprotein [HDL] cholesterol, blood pressure, and blood glucose) and four health behaviors (physical activity, diet, sleep duration, and nicotine exposure), offers a holistic measure of an individual’s lifestyle and health status [7]. While previous studies have established the utility of the LE8 Score in predicting cardiovascular and metabolic outcomes [8], its association with infectious diseases, particularly HPV, remains underexplored.
Our research focuses on examining the potential link between cardiovascular health metrics, as measured by the LE8 Score, and the risk of HPV infection. We hypothesize that individuals with higher LE8 Scores, indicating better cardiovascular health, will have a lower risk of HPV infection. This hypothesis is grounded in the premise that better overall health and lifestyle practices may enhance immune function and reduce susceptibility to infections [9, 10]. Previous studies have demonstrated associations between individual components of cardiovascular health, such as physical activity and diet, and improved immune function [11, 12]. Furthermore, research has shown that lifestyle factors can influence the risk and persistence of HPV infection [6, 13].
To test this hypothesis and explore the relationship between cardiovascular health and HPV infection risk, we will employ a multi-faceted analytical approach. We will utilize data from the National Health and Nutrition Examination Survey (NHANES), a comprehensive program designed to assess the health and nutritional status of adults and children in the United States [8, 14]. Besides, we will examine the association between the weighted LE8 Score and HPV infection status using univariate and multivariate logistic regression models. Second, we will investigate potential nonlinear relationships using restricted cubic splines (RCS) analysis. Finally, we will assess the cumulative impact and relative importance of individual LE8 Score components using weighted quantile sum (WQS) regression.
Given the modifiable nature of the LE8 Score components, understanding their relationship with HPV infection could provide valuable insights into potential lifestyle interventions that may reduce the risk of HPV infection and its associated health burdens. By exploring this association, we aim to contribute to the growing body of literature on the intersection of lifestyle factors and infectious diseases. Our findings may inform public health strategies and interventions aimed at reducing the burden of HPV and improving overall health outcomes, ultimately bridging the gap between cardiovascular health promotion and infectious disease prevention.
Methods
Study design and population
This study employed a cross-sectional design using data from the NHANES, a large-scale survey conducted by the National Center for Health Statistics (NCHS). We analyzed data from seven cycles collected between 2005 and 2016. The NHANES uses a complex, multistage probability sampling design to select participants representative of the civilian, non-institutionalized U.S. population. The study was approved by the Research Ethics Review Board of the NCHS, and all participants provided written informed consent. More information about NHANES can be found at https://www.cdc.gov/nchs/nhanes/index.htm.
Participant selection
From the initial NHANES dataset, we applied several exclusion criteria to define our study population. Participants were excluded if they had missing HPV infection status (n = 49,860), if we were unable to calculate their LE8 Score (n = 3,348), or if they had missing data for other key covariates (n = 955). After applying these criteria, our final analytical sample consisted of 6,773 participants. For more specific details, refer to Fig. 1.
Fig. 1.
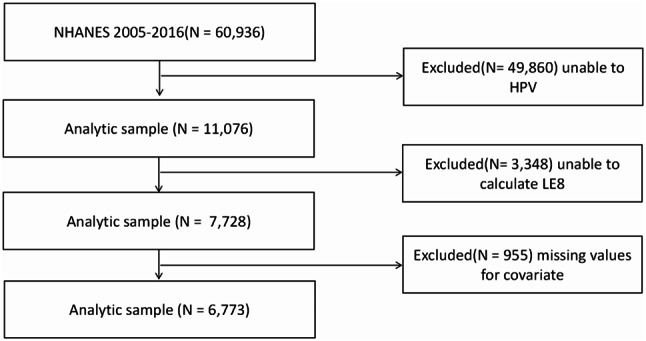
The flowchart of study and excluded participants, from NHANE 2005–2016. Legends: LE8, Life’s Essential 8; NHANES, National Health and Nutrition Examination Survey; HPV, Human Papillomavirus
Exposure assessment: LE8 score
The LE8 score is a validated and reliable instrument developed by the American Heart Association to measure cardiovascular health [15]. The LE8 is composed of 8 CVH indicators, which include 4 health factors ((BMI), non-HDL cholesterol, blood pressure, blood sugar) and 4 health behaviors (physical activity, diet, sleep duration, nicotine exposure).
The validity of the LE8 has been established through its strong association with cardiovascular disease outcomes and all-cause mortality in multiple large cohort studies and its reliability has been demonstrated through consistent results across different populations and settings [15, 16]. The detailed algorithm and scoring criteria for calculating the LE8 score have been published and validated. More details can be found in Supplementary Table 1 [15, 17]. For each of the 8 CVH indicators, scores range from 0 to 100, with high CVH scores between 80 and 100, medium CVH scores between 50 and 79, and low CVH scores between 0 and 49. The overall LE8 score is calculated as the arithmetic mean of the scores for the 8 indicators. In this study, we use the same definitions and cutoff points for classification.
The scoring criteria for each indicator are as follows: Diet quality is assessed based on percentiles of the Healthy Eating Index-2015 (HEI-2015), ranging from optimal (≥ 95th percentile, 100 points) to least optimal (1-24th percentile, 0 points). Physical activity is scored based on weekly moderate or vigorous activity duration, ranging from no activity (0 points) to ≥ 150 min per week (100 points). Nicotine exposure considers smoking status, time since quitting, and secondhand smoke exposure, ranging from current smokers (0 points) to never smokers (100 points). Sleep health is scored based on average nightly sleep duration, with 7–9 h being optimal (100 points). Body Mass Index (BMI) scores range from < 25 (100 points) to ≥ 40 (0 points). Blood lipids are scored based on non-HDL cholesterol levels, with < 130 mg/dL being optimal (100 points). Blood glucose scores combine diabetes history and HbA1c levels, with no diabetes and normal blood glucose being optimal (100 points). Blood pressure ranges from ideal levels (< 120/<80 mmHg, 100 points) to hypertension (≥ 160 or ≥ 100 mmHg, 0 points).
Dietary indicators were assessed using the HEI-2015, the construction and algorithm of which were provided by SAS code from the National Cancer Institute of the United States, and derived from combining dietary intake data collected from two 24-hour dietary recall questionnaires with the United States Department of Agriculture’s food pattern equivalents data [18]. For specific details, see Supplementary Table 2. Additionally, data on sleep duration, nicotine exposure, physical activity, medication history, diabetes history, and other information were obtained from self-reported questionnaires, with physical activity primarily capturing the duration and frequency of moderate to vigorous activities over the past 30 days. Data on blood sugar, blood lipids, and glycated hemoglobin levels were obtained from blood samples sent to the central laboratory. Information on height, weight, blood pressure (average of three consecutive measurements), and other parameters were obtained from physical examination reports.
Outcome assessment: HPV infection status
The specific process for HPV measurement includes: collecting vaginal cells from participants using a vaginal swab; The vaginal swab is then processed by professionals and sent to the Centers for Disease Control and Prevention in Atlanta, Georgia, for further analysis; HPV genotyping kits are measured and analyzed using methods such as the Roche Linear Array, Roche prototype line blot assay, and Digene hybridization capture method. More information about HPV measurement can be obtained from the official website at https://wwwn.cdc.gov/Nchs/Nhanes/2005-2006/HPVSWR_D.htm#LBDR31.
Covariates
To further accurately assess the relationship between the LE8 Score and HPV infection status, we adjusted for the following confounding variables: including age groups ([20,40), [40,59]), ethnicity (Mexican American, Non-Hispanic Black, Non-Hispanic White, Other Hispanic, Other Race - Including Multi-Racial), educational level (below high school, high school, college or above), Poverty Income Ratio (PIR) (< 1, 1–3, ≥ 3), drinking status (Former drinker (individuals who have previously drunk alcohol but have now stopped), Never drinker (individuals who have never consumed any alcoholic beverage), Mild drinker (daily alcohol consumption between 1 and 2 standard drink units), Moderate drinker (men with a daily consumption of no more than 4 standard drink units, women with a daily consumption of no more than 3 standard drink units), Heavy drinker (men with a daily consumption of more than 4 standard drink units, women with a daily consumption of more than 3 standard drink units). Lifetime sexual partners, defined as the number of men with whom the participant has had vaginal intercourse, as measured by the questionnaire item (In your lifetime, with how many men have you had vaginal sex?). These variables were selected based on their potential association with both the LE8 Score and HPV infection status, as suggested by previous literature and available NHANES data.
Statistical analyses
Considering the complex probability sampling characteristics of the NHANES database, we conducted weighted analyses as recommended by the database, taking into account two-day dietary interview weights (WTDR2D), primary sampling units (SDMVPSU), and strata (SDMVSTRA) to account for the complex design. Continuous variables are presented as weighted means (± standard deviation), with weighted t-tests used to describe statistical differences. Categorical variables are presented as percentages, with participants divided into HPV-infected and HPV-uninfected groups based on HPV infection status, and differences between groups described using weighted chi-square tests. Three logistic regression models were constructed to examine the association between LE8 Score and HPV infection status. Model 1 was unadjusted, while Model 2 adjusted for race/ethnicity, educational level, PIR, and drinking status. Model 3 further adjusted for lifetime sexual partners in addition to the variables included in Model 2. The Rao-Scott F test, an adjusted version of the chi-square test designed for complex sampling designs, was used to evaluate the robustness of the logistic regression model by assessing improvements in model fit with the inclusion of additional variables. This test effectively corrects assumption errors in standard chi-square tests, accounting for the complexities of survey data [19]. The variance inflation factor (VIF) was used to assess multicollinearity in the model. VIF measures the degree of correlation between independent variables, reflecting the strength of the linear relationship among them. Generally, a VIF value below 10 indicates low multicollinearity, while a value above 10 suggests significant multicollinearity [20].
The RCS models were used to investigate the dose-response relationship between LE8 Score and HPV infection status. RCS models capture nonlinear relationships by fitting separate linear models within different regions of the data (called “knots”). This approach provides flexibility and better modeling of complex dose-response relationships, which is advantageous compared to simple linear models. In applying the RCS models, we chose 3 to 5 knots, optimizing their positions using standard procedures such as minimizing the residual sum of squares [21]. The mathematical form of the RCS model is as follows:
 |
Where y represents HPV infection status (outcome),  represents the LE8 score (predictor),
represents the LE8 score (predictor),  is the restricted cubic spline function,
is the restricted cubic spline function,  are the regression coefficients, and
are the regression coefficients, and  is the error term.
is the error term.
Besides, to examine the relationship between LE8 scores and HPV infection status across different populations, we conducted subgroup analyses and interaction tests. Age, race/ethnicity, education level, and PIR were selected as primary stratification variables based on their potential influence on HPV infection risk and immune response. Age was categorized into two groups: 20–40 years and 40–59 years and above. Race/ethnicity was classified according to NHANES standards, including Mexican American, non-Hispanic Black, non-Hispanic White, and other races (including multiracial). Education level was divided into three categories: high school and below, college, and graduate school and above. PIR was stratified into three ranges: <1, 1–3, and ≥ 3. Logistic regression models were employed to assess the relationship between LE8 scores and HPV infection status for each subgroup. Interactions between LE8 scores and each stratification variable were incorporated into the models to test for differences in these relationships across subgroups. An interaction term with a p-value < 0.05 was considered indicative of a significant interaction effect.
The WQS model was employed to evaluate the joint effects of multiple LE8 components on HPV infection risk, addressing multicollinearity by assigning weights to each component based on their contribution to the outcome. This approach is particularly useful for evaluating the combined effects of correlated variables [22]. The weighted model is as follows:
 |
 |
 represents any differentiable link function,
represents any differentiable link function,  denotes the intercept, and
denotes the intercept, and  represents the regression coefficients, c denotes the number of LE8 indicators included in the model,
represents the regression coefficients, c denotes the number of LE8 indicators included in the model,  represents the weighting index, with a range of (0≤
represents the weighting index, with a range of (0≤ ≤1), the sum of all weighting indices equals 1.
≤1), the sum of all weighting indices equals 1.  and Φ represent the matrix of covariates and the coefficients, respectively.
and Φ represent the matrix of covariates and the coefficients, respectively.  denotes the quartiles for each LE8 indicator, where (
denotes the quartiles for each LE8 indicator, where ( =0,1,2,3) correspond to the 1st, 2nd, 3rd, or 4th quartile, respectively.
=0,1,2,3) correspond to the 1st, 2nd, 3rd, or 4th quartile, respectively.  represents the sum of the weighted quartiles of c indicators. Assuming a linear function fitting a Gaussian distribution, the data are randomly divided into a training set (60%) and a validation set (40%), and the weights of 8 LE8 indicators in the training set are estimated [23].
represents the sum of the weighted quartiles of c indicators. Assuming a linear function fitting a Gaussian distribution, the data are randomly divided into a training set (60%) and a validation set (40%), and the weights of 8 LE8 indicators in the training set are estimated [23].
All statistical analyses and visualizations in this study were performed using R (version 4.4.1, https://www.r-project.org/), with two-sided statistical tests, and a P-value < 0.05 was considered statistically significant.
Results
Baseline characteristics
In this study, participants were categorized based on their HPV infection status into HPV-infected and non-HPV-infected groups. Compared to the non-HPV-infected group, the HPV-infected group was more likely to be females aged 20–40, non-Hispanic white, with a high school education, a PIR above 1, and heavy drinkers. Furthermore, compared to the non-HPV-infected group, participants in the HPV-infected group had lower nicotine exposure, shorter sleep duration, less frequent physical activity, and lower HEI-2015 scores. For more details on the baseline characteristics, see Table 1.
Table 1.
Survey-weighted baseline characteristic of the study population
| Level | No HPV Infection | HPV Infection | P-value | |
|---|---|---|---|---|
| Age (%) | < 0.001 | |||
| 20–40 | 44.87(1.37) | 54.04(1.44) | ||
| 40–59 | 55.13(1.37) | 45.96(1.44) | ||
| Race/ethnicity (%) | < 0.001 | |||
| Mexican American | 9.03(0.96) | 8.05(0.82) | ||
| Non-Hispanic Black | 7.55(0.70) | 17.85(1.30) | ||
| Non-Hispanic White | 70.08(1.68) | 62.08(1.80) | ||
| Other Hispanic | 5.11(0.57) | 6.16(0.72) | ||
| Other Race - Including Multi-Racial | 8.22(0.66) | 5.87(0.59) | ||
| Education (%) | < 0.001 | |||
| low high school | 11.28(0.89) | 14.70(1.07) | ||
| High school | 18.62(1.05) | 22.52(1.11) | ||
| College or above | 70.10(1.41) | 62.77(1.64) | ||
| PIR (%) | < 0.001 | |||
| < 1 | 12.65(0.87) | 21.70(0.99) | ||
| 1–3 | 30.77(1.20) | 36.45(1.28) | ||
| ≥ 3 | 56.58(1.54) | 41.85(1.61) | ||
| Drinking status (%) | < 0.001 | |||
| Former drinker | 11.88(0.83) | 12.84(0.98) | ||
| Heavy drinker | 17.81(0.87) | 28.53(1.33) | ||
| Mild drinker | 30.62(1.12) | 23.71(1.26) | ||
| Moderate drinker | 24.13(1.08) | 26.35(1.22) | ||
| Never drinker | 15.57(1.07) | 8.56(0.65) | ||
| LE8 scores (%) | < 0.001 | |||
| Low CVH (0–49) | 8.02(0.65) | 11.92(1.00) | ||
| Moderate CVH (50–79) | 60.64(1.40) | 63.36(1.43) | ||
| High CVH (80–100) | 31.34(1.46) | 24.72(1.41) | ||
| Tobacco/nicotine exposure (mean (SD)) | 77.34(0.99) | 62.15(1.40) | < 0.001 | |
| Sleep health (mean (SD)) | 84.64(0.55) | 80.18(0.71) | < 0.001 | |
| Body mass index (mean (SD)) | 61.97(0.92) | 62.39(1.13) | 0.76 | |
| Blood lipids (mean (SD)) | 68.61(0.80) | 70.01(0.80) | 0.23 | |
| Blood glucose(mean (SD)) | 90.20(0.50) | 91.54(0.52) | 0.07 | |
| Blood pressure (mean (SD)) | 80.80(0.62) | 81.14(0.76) | 0.73 | |
| HEI-2015 diet score (mean (SD)) | 41.85(1.07) | 35.81(1.12) | < 0.001 | |
| Physical activity (mean (SD)) | 72.14(1.08) | 69.83(1.25) | 0.12 |
Note SD, Standard Deviation; CVH, Cardiovascular Health; PIR, Poverty Income Ratio
Association between weighted LE8 score and HPV status
In the Model 1, compared to the low CVH group, the moderate and high CVH groups had a 30% (OR 0.70, 95%CI 0.56–0.89, P = 0.003) and 47% (OR 0.53, 95%CI 0.40–0.70, P < 0.001) lower risk of HPV infection, respectively. In the Model 2, compared to the low CVH group, the moderate and high CVH groups had a 21% (OR 0.79, 95%CI 0.63-1.00, P = 0.050) and 31% (OR 0.69, 95%CI 0.52–0.92, P = 0.010) lower risk of HPV infection, respectively. In the Model 3, compared to the low CVH group, the moderate and high CVH groups had a 30% (OR 0.70, 95%CI 0.53–0.92, P = 0.010) and 36% (OR 0.64, 95%CI 0.43–0.96, P = 0.030) lower risk of HPV infection, respectively. The goodness-of-fit of the multivariate logistic regression model (Model 3) was assessed using the Rao-Scott F test. The test results indicated a satisfactory model fit (The working 2logLR = 317.524, P-value < 0.001). Additionally, multicollinearity among the variables was evaluated using the VIF. All variables had VIF values below 5, indicating that there were no significant multicollinearity issues in the Model 3. More detailed information can be seen in Table 2.
Table 2.
Odds ratios for HPV infection status by Cardiovascular Health (CVH) categories
| Model 1 | Model 2 | Model 3 | ||||
|---|---|---|---|---|---|---|
| OR(95% CI) | P-value | OR (95% CI) | P-value | OR (95% CI) | P-value | |
| LE8 score | ||||||
| Low CVH (0–49) | Reference | - | Reference | - | Reference | - |
| Moderate CVH (50–79) | 0.70(0.56,0.89) | 0.003 | 0.79(0.63,1.00) | 0.050 | 0.70(0.53,0.92) | 0.010 |
| High CVH (80–100) | 0.53(0.40,0.70) | < 0.001 | 0.69(0.52,0.92) | 0.010 | 0.64(0.43,0.96) | 0.030 |
Note CVH, Cardiovascular health; OR, Odds ratio; CI, confidence interval; Model 1: No covariates were adjusted. Model 2: Adjusted for race/ethnicity, educational level, PIR and drinking status. Model 3: Adjusted for lifetime sexual partners in addition to the variables in Model 2. The reference category: Low CVH group
Nonlinearity analysis using RCS
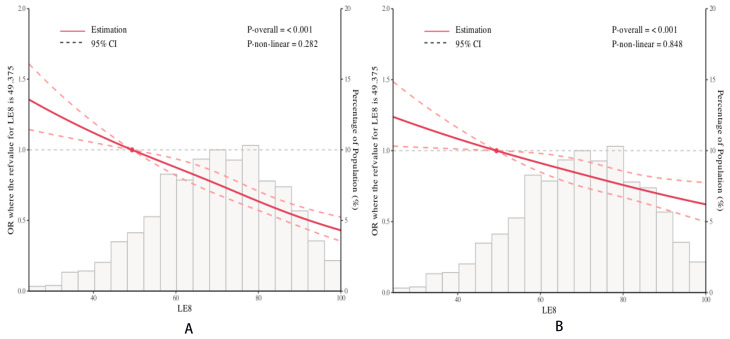
The dose-response relationship between LE8 Score and HPV infection status was evaluated using the RCS model. Knots were placed at the 5th, 35th, 65th, and 95th percentiles of the LE8 Score, with the 5th percentile as the reference, and the multivariate model was adjusted for the same factors as mentioned above. In the univariate model, a correlation between the LE8 Score and HPV infection status was observed, though the nonlinearity was not significant (p for overall association < 0.001, p for nonlinear association = 0.282) (Fig. 2A). In the multivariate model, a correlation between the LE8 Score and HPV infection status was observed, with the nonlinearity being insignificant (p for overall association < 0.001, p for nonlinear association = 0.848) (Fig. 2B).
Fig. 2.
Adjusted restricted cubic spline curve between Life’s Essential 8 scores and HPV infection status. Legends: (A), no covariates were adjusted. (B), adjusted for race/ethnicity, educational level, PIR and drinking status. Data were fitted by a logistic regression model, and the model was conducted with 4 knots at the 5th, 35th, 65th, 95th percentiles of LE8 score (reference is the 5th percentile). The x-axis represents LE8 scores ranging from 0 to 100, while the left y-axis shows the odds ratio (OR) where the reference for LE8 is 49.375, and the right y-axis indicates the percentage of population. Gray histograms depict population distribution. Red solid lines represent the estimated OR trends, with red dashed lines showing 95% confidence intervals (CI). OR, odds ratio; CI, confidence interval
Subgroup analyses
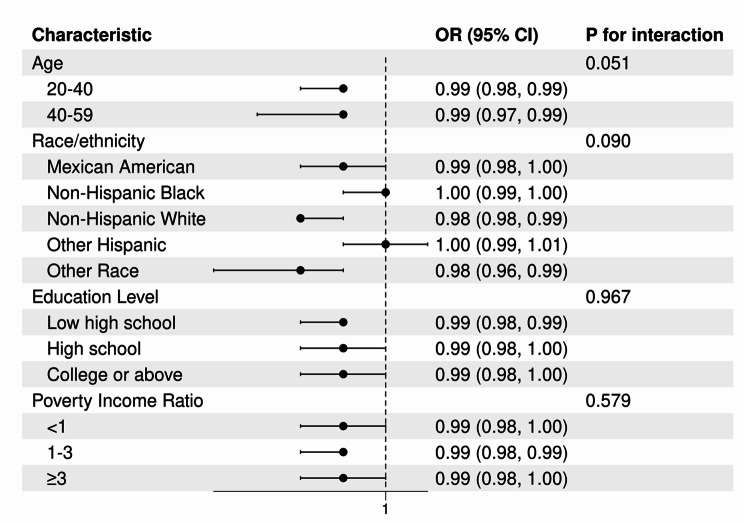
In the subgroup analysis stratified by age, race/ethnicity, education level, and PIR, we found an inconsistent correlation between LE8 Score and HPV infection status. In most subgroups, there was a negative correlation between LE8 Score and HPV infection status. This negative correlation was observed in participants who were women over the age of 20, Mexican American, Non-Hispanic Black, Non-Hispanic White, Other Race including Multi-Racial, with education levels of high school or below and college, and PIR ranges of (< 1, 1–3, ≥ 3). The interaction results showed no direct evidence to suggest that this negative correlation was associated with age, race, education level, or PIR. For more details, please refer to Fig. 3.
Fig. 3.
Subgroup analysis of the association of the Life’s Essential 8 scores and HPV infection status, each stratifcation was adjusted for race/ethnicity, education level, PIR and drinking status. Legends: The figure displays the odds ratios (OR) and 95% confidence intervals (CI) for HPV infection across various demographic and socioeconomic subgroups. The characteristics include age groups, race/ethnicity categories, education levels, and poverty income ratio (PIR). Each OR represents the association between a characteristic and HPV infection status, adjusted for relevant covariates. “P for interaction” refers to the statistical test for interaction between each characteristic and the association between LE8 scores (lifestyle and cardiovascular health metrics) and HPV infection risk
WQS regression
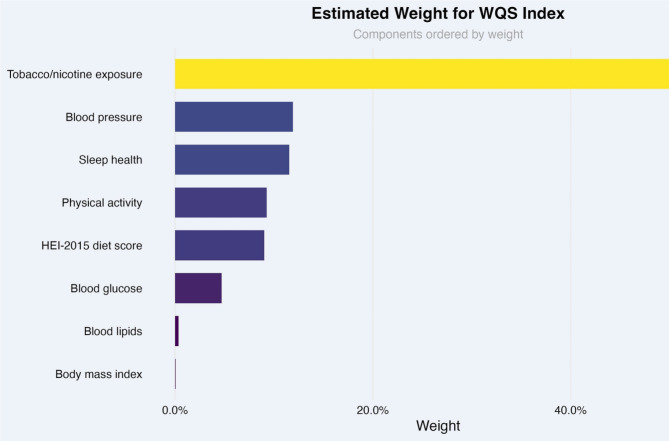
A WQS model was used to construct a weighted index to evaluate the cumulative impact and weight proportion of the 8 indicators of LE8 on HPV infection status. The model was adjusted for race/ethnicity, education level, PIR, and drinking status, with the final results showing the highest weight proportions for nicotine exposure (53.20%) and blood pressure (11.91%), and the lowest for BMI (0.05%). For more detailed information, please refer to Fig. 4.
Fig. 4.
Weighted Quantile Sum (WQS) model regression index weights for the HPV infection, adjusted for race/ethnicity, education level, poverty income ratio (PIR) and drinking status. Legends: The figure presents the estimated weights of different lifestyle and cardiovascular health metrics as determined by the WQS model, showing their relative contributions to the overall risk of HPV infection. Tobacco/nicotine exposure is the most significant contributor, accounting for 53.20% of the total weight, indicating a strong link between tobacco use and increased HPV infection risk. Blood pressure follows, contributing about 11.91%, while sleep health and physical activity each account for approximately 11%, reflecting moderate associations with HPV risk. The HEI-2015 diet score, representing dietary quality, contributes about 9.01%, suggesting a smaller yet noteworthy role. Blood glucose and blood lipids have relatively minor contributions, with blood glucose slightly outweighing blood lipids. Body mass index (BMI) has a negligible contribution, implying little to no influence on HPV infection risk when considering the other factors in the model
Discussion
In this nationally representative study of U.S. adults aged 20–59, we found that better overall cardiovascular health, as measured by the LE8 Score, was significantly associated with a lower risk of HPV infection. In both unadjusted and multivariable-adjusted logistic regression models, we observed a graded inverse association between LE8 Score and odds of testing positive for HPV infection. Compared to participants with low LE8 Score, those with medium and high LE8 Score had a 30–36% lower likelihood of prevalent HPV infection after adjusting for relevant confounders. The exposure-response analysis using restricted cubic splines further supported the linear relationship between increasing LE8 category and lower HPV positivity. These findings were independent of relevant sociodemographic and behavioral factors [8, 24, 25]. Additionally, we identified smoking exposure and blood pressure as the strongest contributors to HPV infection risk out of the individual LE8 components.
Our results align with and extend previous research demonstrating favorable associations between cardiovascular health metrics similar to the LE8 Score and infectious disease morbidity [26]. In a large prospective cohort study, individuals adhering to a healthy lifestyle, including non-smoking, moderate alcohol consumption, regular physical activity, and maintaining a healthy weight, exhibited a 65% reduction in the risk of severe infectious diseases [27]. Similarly, Li et al. reported a significant association between cardiovascular health metrics and reduced influenza risk [28]. We add to these literature by specifically linking the LE8 Score to HPV positivity. The observed relationship is biologically plausible given the established role of immune function, inflammation, and oxidative stress in HPV infection and persistence [29].
The health behaviors encompassed in the LE8 Score, including diet quality [30], physical activity [9], smoking, obesity, cholesterol, glucose, and blood pressure, can modulate immune function and susceptibility to infectious diseases. Plausible mechanisms relate to changes in mucosal immunity, inflammation, oxidative stress and micronutrient status [11]. Our findings align with previous studies suggesting that lifestyle factors and metabolic health play a crucial role in modulating immune responses. For instance, one study demonstrated that higher physical activity levels were associated with a reduced risk of HPV infection, likely due to enhanced immune function [31]. Similarly, a cohort study showed that smokers were significantly more likely to have persistent HPV infections compared to non-smokers [32]. By contributing to this body of literature, our study reinforces the notion that addressing cardiovascular health and modifiable lifestyle factors may offer an additional strategy for reducing HPV infection risk and its associated complications.
Optimal cardiovascular health may enhance immune function to resist HPV infection through various pathways. For instance, regular moderate-intensity aerobic exercise has been shown to increase the number and cytotoxic activity of circulating NK cells [33]. These NK cells play a crucial role in recognizing and eliminating HPV-infected cells. Additionally, healthy dietary patterns, such as the Mediterranean diet, rich in antioxidants and polyphenols, can enhance T cell proliferation and function [34], which is vital for controlling HPV infection. Moreover, chronic inflammation and high levels of oxidative stress have been shown to promote persistent HPV infection and integration. Good cardiovascular health, particularly through blood pressure and glucose control, can significantly reduce systemic inflammation levels. Studies have shown that hypertensive patients have significantly elevated levels of inflammatory markers such as C-reactive protein and interleukin-6 [35]. These inflammatory factors may promote HPV infection by suppressing antiviral immune responses. Controlling blood glucose can reduce the formation of advanced glycation end products (AGEs), which are associated with oxidative stress and DNA damage, potentially increasing the risk of HPV infection [36]. Good cardiovascular health may prevent HPV infection by maintaining the integrity of mucosal barriers. For example, smoking has been shown to disrupt tight junction protein expression in respiratory and reproductive tract epithelia, increasing opportunities for pathogen invasion [37]. Furthermore, hypertension may impair epithelial barrier function by increasing vascular permeability and promoting local inflammation [38]. By quitting smoking and controlling blood pressure, these critical physical barriers against HPV infection can be protected.
Our study is the first to specifically quantify the cumulative association of these factors, measured through the LE8 Score, with odds of HPV infection. As a composite metric summing up diverse lifestyle and health parameters, the LE8 Score may capture synergistic effects on resistance to HPV infection more effectively than individual factors. Further research should evaluate the clinical utility of this score for HPV infection risk stratification and guiding multi-modal interventions targeting lifestyle changes and improved wellness. Enhancing population LE8 profiles could potentially translate to lower HPV prevalence and associated disease burden.
Our weighted quantile sum regression delineated the relative contributions of individual LE8 metrics to HPV infection odds. Notably, nicotine exposure and blood pressure cumulatively accounted for over 65% of the total effect. This aligns with prior evidence that smoking induces immunosuppression and vascular dysfunction, enabling viral persistence [39]. Hypertension may also disturb antiviral immunity and epithelial barrier integrity via oxidative stress [40]. In contrast, body mass index had the lowest weight, indicating that generalized adiposity is not a major driver of HPV susceptibility. These findings highlight prime targets for tailored lifestyle interventions to curb HPV susceptibility. Given the substantial burden of smoking and hypertension nationally [41], population-level improvements on these parameters could confer a disproportionate benefit in lowering HPV infection risk.
The exposure-response relationship characterized by restricted cubic splines was indicative of an overall linear association between increasing LE8 category and lower HPV positivity. No statistically significant deviations from linearity were discerned. The subgroup analyses suggested effect modification across some sociodemographic factors, with inverse relationships most apparent among adult women, racial/ethnic minorities, those with high school education or less, and lower income levels. One potential explanation is that disadvantaged groups experience greater absolute gains from lifestyle optimization due to higher baseline infection risk [25]. However, tests for interaction did not provide definitive evidence that the LE8-HPV association was modified by age, race/ethnicity, education or poverty status. Further interrogation in larger samples would be valuable to determine whether lifestyle optimization confers variable advantage against HPV infection across population strata.
Several limitations should be acknowledged in this study. First, the cross-sectional design prevents us from establishing causal relationships between lifestyle factors and HPV infection. While our findings suggest a strong association, longitudinal studies are necessary to confirm temporal sequences and causal mechanisms. Additionally, the reliance on self-reported dietary data introduces potential recall bias, which could affect the accuracy of these variables and their association with HPV infection [42]. Besides, while we adjusted for the number of lifetime sexual partners—an important sexual behavior variable—other aspects of sexual behavior, such as condom use or the timing of HPV exposure, were not captured in detail and could act as residual confounders. Additionally, although we employed a comprehensive analytical approach, there remains the potential for residual confounding due to unmeasured factors, such as socioeconomic status or vaccination history. These unmeasured factors may influence both lifestyle behaviors and HPV infection risk, limiting the strength of our conclusions. Finally, the generalizability of our findings is limited. The NHANES sample consists only of non-institutionalized U.S. civilians, and our results may not be applicable to other populations. Future research in diverse geographical and sociodemographic groups is necessary to validate and expand upon our findings.
Despite these limitations, our study provides valuable insights into the relationship between lifestyle factors, cardiovascular health metrics, and HPV infection risk. The significant association between the LE8 Score and HPV infection status highlights the potential of lifestyle interventions in HPV prevention strategies. Our findings suggest that improving population cardiovascular health through healthier diets, tobacco cessation, blood pressure control, and increased physical activity could confer a protective advantage against HPV infection.
These results have important implications for public health strategies. Integrating lifestyle optimization into existing HPV prevention efforts, such as vaccination and screening programs, could enhance their effectiveness. Such an approach not only addresses HPV infection risk but also promotes overall health and well-being. Beyond vaccination, public health campaigns should emphasize the importance of reducing modifiable risk factors, such as tobacco cessation, maintaining a healthy diet, and regular physical activity, as part of comprehensive HPV prevention strategies. These lifestyle modifications can be promoted through targeted education campaigns, health coaching, and community-based interventions. Moreover, healthcare providers could incorporate cardiovascular health assessments and counseling into routine HPV screenings, particularly for high-risk populations. By identifying individuals with poor cardiovascular health or unhealthy lifestyle behaviors, providers can deliver personalized interventions aimed at reducing both cardiovascular disease and HPV infection risk. This approach would align infection prevention with chronic disease management, offering a dual benefit for public health. Besides, the potential cost-effectiveness of lifestyle interventions in reducing HPV-related diseases also warrants further investigation. Tailored interventions addressing both cardiovascular health and HPV prevention could reduce the long-term healthcare burden associated with HPV-related cancers and other chronic conditions.
Moving forward, several research directions emerge from our findings. Longitudinal cohort studies tracking the temporal relationship between lifestyle exposures and HPV acquisition are essential to elucidate causal mechanisms. These should be complemented by intervention trials evaluating the impact of lifestyle optimization on HPV infection incidence, providing higher-quality evidence to inform targeted prevention strategies. To uncover biological pathways, we recommend conducting translational studies that combine lifestyle interventions with biomarker analyses, focusing on immune function markers, vaginal microbiome composition, and HPV-specific antibody responses. Advanced techniques like metabolomics could identify potential mediators of lifestyle-HPV interactions. To enhance the generalizability of our findings, research should expand to more diverse populations and age groups through multi-center studies across various geographical regions and socioeconomic groups, including understudied populations such as men, adolescents, and older adults. By pursuing these research directions, we aim to provide a comprehensive understanding of the lifestyle-HPV relationship, ultimately informing effective public health interventions and prevention strategies.
Conclusion
In conclusion, our study underscores the significant role of healthy lifestyle factors, particularly cardiovascular health, in reducing HPV infection risk. While vaccination and screening are essential, integrating lifestyle interventions into existing HPV prevention frameworks could create a more comprehensive approach. For example, lifestyle education focusing on smoking cessation, improved diet, and physical activity can be integrated into vaccination and screening programs. Healthcare providers could offer cardiovascular assessments and personalized counseling during routine HPV screenings, addressing both infection prevention and chronic disease management. Public health campaigns could also use digital platforms to deliver HPV prevention reminders alongside tailored lifestyle recommendations, leveraging mobile health technologies. Community initiatives that combine HPV awareness with health promotion activities could further enhance prevention efforts. Promoting cardiovascular health within HPV prevention programs offers the potential to reduce both HPV-related diseases and other lifestyle-associated conditions, creating a cost-effective and holistic public health strategy. Future research should evaluate the impact of these combined interventions to maximize the effectiveness of HPV prevention and improve overall health outcomes.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
The authors sincerely expressed their gratitude to all members who took part in this study.
Author contributions
XL, YC and YL prepared and edited the original draft. YQ and WZ were involved in data collation and visualization. WJ, WL and JC participated in the drafting of the final article and conceptualization.
Funding
The authors declare that they did not receive any funding from any source.
Data availability
The data can be downloaded here: https://www.cdc.gov/nchs/nhanes/index.htm.
Declarations
Ethics approval and consent to participate
The NHANES agreement has been reviewed and approved by the National Center for Health Statistics Research Ethics Committee. All participants provided written informed consent before participating.
Consent for publication
Not Applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xiangliang Liu, Yu Chang and Yuguang Li contributed equally to this work and shared first authorship.
Contributor Information
Wei Ji, Email: jiwei324@qq.com.
Wei Li, Email: liwei66@jlu.edu.cn.
Jiuwei Cui, Email: cuijw@jlu.edu.cn.
References
- 1.Forman D, de Martel C, Lacey CJ, Soerjomataram I, Lortet-Tieulent J, Bruni L, Vignat J, Ferlay J, Bray F, Plummer M, et al. Global burden of human papillomavirus and related diseases. Vaccine. 2012;30(Suppl 5):F12–23. [DOI] [PubMed] [Google Scholar]
- 2.Fu L, Tian T, Wang B, Lu Z, Bian J, Zhang W, Wu X, Li X, Siow RC, Fang EF, et al. Global, regional, and national burden of HIV and other sexually transmitted infections in older adults aged 60–89 years from 1990 to 2019: results from the global burden of Disease Study 2019. Lancet Healthy Longev. 2024;5(1):e17–30. [DOI] [PubMed] [Google Scholar]
- 3.de Martel C, Plummer M, Vignat J, Franceschi S. Worldwide burden of cancer attributable to HPV by site, country and HPV type. Int J Cancer. 2017;141(4):664–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Oshman LD, Davis AM. Human papillomavirus vaccination for adults: updated recommendations of the Advisory Committee on Immunization Practices (ACIP). JAMA. 2020;323(5):468–9. [DOI] [PubMed] [Google Scholar]
- 5.Moscicki AB, Schiffman M, Burchell A, Albero G, Giuliano AR, Goodman MT, Kjaer SK, Palefsky J. Updating the natural history of human papillomavirus and anogenital cancers. Vaccine. 2012;30(0 5):F24–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Giuliano AR, Sedjo RL, Roe DJ, Harri R, Baldwi S, Papenfuss MR, Abrahamsen M, Inserra P. Clearance of oncogenic human papillomavirus (HPV) infection: effect of smoking (United States). Cancer Causes Control: CCC. 2002;13(9):839–46. [DOI] [PubMed] [Google Scholar]
- 7.Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Das SR, et al. Heart Disease and Stroke Statistics-2019 update: a Report from the American Heart Association. Circulation. 2019;139(10):e56–528. [DOI] [PubMed] [Google Scholar]
- 8.Lloyd-Jones DM, Hong Y, Labarthe D, Mozaffarian D, Appel LJ, Van Horn L, Greenlund K, Daniels S, Nichol G, Tomaselli GF, et al. Defining and setting national goals for cardiovascular health promotion and disease reduction: the American Heart Association’s strategic impact goal through 2020 and beyond. Circulation. 2010;121(4):586–613. [DOI] [PubMed] [Google Scholar]
- 9.Nieman DC, Wentz LM. The compelling link between physical activity and the body’s defense system. J Sport Health Sci. 2019;8(3):201–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Christ A, Lauterbach M, Latz E. Western Diet and the Immune System: an inflammatory connection. Immunity. 2019;51(5):794–811. [DOI] [PubMed] [Google Scholar]
- 11.Simpson RJ, Kunz H, Agha N, Graff R. Exercise and the regulation of Immune functions. Prog Mol Biol Transl Sci. 2015;135:355–80. [DOI] [PubMed] [Google Scholar]
- 12.Wintergerst ES, Maggini S, Hornig DH. Contribution of selected vitamins and trace elements to immune function. Ann Nutr Metab. 2007;51(4):301–23. [DOI] [PubMed] [Google Scholar]
- 13.Moscicki AB, Ma Y, Farhat S, Jay J, Hanson E, Benningfield S, Jonte J, Godwin-Medina C, Wilson R, Shiboski S. Natural history of anal human papillomavirus infection in heterosexual women and risks associated with persistence. Clin Infect Dis. 2014;58(6):804–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ford ES, Ajani UA, Croft JB, Critchley JA, Labarthe DR, Kottke TE, Giles WH, Capewell S. Explaining the decrease in U.S. deaths from coronary disease, 1980–2000. N Engl J Med. 2007;356(23):2388–98. [DOI] [PubMed] [Google Scholar]
- 15.Lloyd-Jones DM, Allen NB, Anderson CAM, Black T, Brewer LC, Foraker RE, Grandner MA, Lavretsky H, Perak AM, Sharma G, et al. Life’s essential 8: updating and enhancing the American Heart Association’s construct of Cardiovascular Health: a Presidential Advisory from the American Heart Association. Circulation. 2022;146(5):e18–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang J, Chen G, Habudele Z, Wang X, Cai M, Li H, Gao Y, Lip GYH, Lin H. Relation of Life’s essential 8 to the genetic predisposition for cardiovascular outcomes and all-cause mortality: results from a national prospective cohort. Eur J Prev Cardiol. 2023;30(15):1676–85. [DOI] [PubMed] [Google Scholar]
- 17.Lloyd-Jones DM, Ning H, Labarthe D, Brewer L, Sharma G, Rosamond W, Foraker RE, Black T, Grandner MA, Allen NB, et al. Status of Cardiovascular Health in US adults and Children Using the American Heart Association’s New Life’s essential 8 Metrics: Prevalence Estimates from the National Health and Nutrition Examination Survey (NHANES), 2013 through 2018. Circulation. 2022;146(11):822–35. [DOI] [PubMed] [Google Scholar]
- 18.Krebs-Smith SM, Pannucci TE, Subar AF, Kirkpatrick SI, Lerman JL, Tooze JA, Wilson MM, Reedy J. Update of the healthy eating index: HEI-2015. J Acad Nutr Dietetics. 2018;118(9):1591–602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rao JNK, Scott AJ. On Chi-Squared tests for Multiway Contingency Tables with cell proportions estimated from Survey Data. Annals Stat. 1984;12(1):46–60. [Google Scholar]
- 20.Stapleton JH. Linear statistical models. Volume 719. Wiley; 2009.
- 21.Frank EH. Regression modeling strategies with applications to linear models, logistic and ordinal regression, and survival analysis. In.: Spinger; 2015.
- 22.Carrico C, Gennings C, Wheeler DC, Factor-Litvak P. Characterization of weighted quantile sum regression for highly correlated data in a risk analysis setting. J Agricultural Biol Environ Stat. 2015;20:100–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yorita Christensen KL, Carrico CK, Sanyal AJ, Gennings C. Multiple classes of environmental chemicals are associated with liver disease: NHANES 2003–2004. Int J Hyg Environ Health. 2013;216(6):703–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Allen NB, Siddique J, Wilkins JT, Shay C, Lewis CE, Goff DC, Jacobs DR Jr., Liu K, Lloyd-Jones D. Blood pressure trajectories in early adulthood and subclinical atherosclerosis in middle age. JAMA. 2014;311(5):490–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gravitt PE. The known unknowns of HPV natural history. J Clin Investig. 2011;121(12):4593–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Calder PC, Carr AC, Gombart AF, Eggersdorfer M. Optimal nutritional status for a well-functioning Immune System is an important factor to protect against viral infections. Nutrients 2020, 12(4). [DOI] [PMC free article] [PubMed]
- 27.Ye X, Wang Y, Zou Y, Tu J, Tang W, Yu R, Yang S, Huang P. Associations of socioeconomic status with infectious diseases mediated by lifestyle, environmental pollution and chronic comorbidities: a comprehensive evaluation based on UK Biobank. Infect Dis Poverty. 2023;12(1):5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li Y, Pan A, Wang DD, Liu X, Dhana K, Franco OH, Kaptoge S, Di Angelantonio E, Stampfer M, Willett WC, et al. Impact of healthy lifestyle factors on life expectancies in the US Population. Circulation. 2018;138(4):345–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shane AL, Sánchez PJ, Stoll BJ. Neonatal sepsis. Lancet (London England). 2017;390(10104):1770–80. [DOI] [PubMed] [Google Scholar]
- 30.He CS, Handzlik M, Fraser WD, Muhamad A, Preston H, Richardson A, Gleeson M. Influence of vitamin D status on respiratory infection incidence and immune function during 4 months of winter training in endurance sport athletes. Exerc Immunol Rev. 2013;19:86–101. [PubMed] [Google Scholar]
- 31.Zhao X, Wu Y, Hu H. Healthy lifestyle and HPV infection risk: results from a cross-sectional study. Front Public Health. 2024;12:1421636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Koshiol J, Schroeder J, Jamieson DJ, Marshall SW, Duerr A, Heilig CM, Shah KV, Klein RS, Cu-Uvin S, Schuman P. Smoking and time to clearance of human papillomavirus infection in HIV-seropositive and HIV-seronegative women. Am J Epidemiol. 2006;164(2):176–83. [DOI] [PubMed] [Google Scholar]
- 33.Campbell JP, Turner JE. Debunking the myth of Exercise-Induced Immune suppression: redefining the Impact of Exercise on Immunological Health across the Lifespan. Front Immunol. 2018;9:648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ghosh TS, Rampelli S, Jeffery IB, Santoro A, Neto M, Capri M, Giampieri E, Jennings A, Candela M, Turroni S, et al. Mediterranean diet intervention alters the gut microbiome in older people reducing frailty and improving health status: the NU-AGE 1-year dietary intervention across five European countries. Gut. 2020;69(7):1218–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nosalski R, McGinnigle E, Siedlinski M, Guzik TJ. Novel Immune mechanisms in Hypertension and Cardiovascular Risk. Curr Cardiovasc Risk Rep. 2017;11(4):12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Goldin A, Beckman JA, Schmidt AM, Creager MA. Advanced glycation end products: sparking the development of diabetic vascular injury. Circulation. 2006;114(6):597–605. [DOI] [PubMed] [Google Scholar]
- 37.Colsoul ML, Goderniaux N, Onorati S, Dupuis S, Jamart J, Vanpee D, Berlin I, Galanti L. Changes in biomarkers of endothelial function, oxidative stress, inflammation and lipids after smoking cessation: a cohort study. Eur J Clin Invest. 2023;53(8):e13996. [DOI] [PubMed] [Google Scholar]
- 38.Dharmashankar K, Widlansky ME. Vascular endothelial function and hypertension: insights and directions. Curr Hypertens Rep. 2010;12(6):448–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Thomson NC, Polosa R, Sin DD. Cigarette smoking and asthma. J Allergy Clin Immunol Pract. 2022;10(11):2783–97. [DOI] [PubMed]
- 40.Virdis A, Duranti E, Taddei S. Oxidative stress and vascular damage in hypertension: role of angiotensin II. Int J Hypertens. 2011;2011:916310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Virani SS, Alonso A, Benjamin EJ, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Delling FN, et al. Heart Disease and Stroke Statistics-2020 update: a Report from the American Heart Association. Circulation. 2020;141(9):e139–596. [DOI] [PubMed] [Google Scholar]
- 42.Nasreddine L, Tamim H, Itani L, Nasrallah MP, Isma’eel H, Nakhoul NF, Abou-Rizk J, Naja F. A minimally processed dietary pattern is associated with lower odds of metabolic syndrome among Lebanese adults. Public Health Nutr. 2018;21(1):160–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data can be downloaded here: https://www.cdc.gov/nchs/nhanes/index.htm.