Abstract
Malignant bone tumors, which are difficult to treat with current clinical strategies, originate from bone tissues and can be classified into primary and secondary types. Due to the specificity of the bone microenvironment, the results of traditional means of treating bone tumors are often unsatisfactory, so there is an urgent need to develop new treatments for malignant bone tumors. Recently, nanoparticle-based approaches have shown great potential in diagnosis and treatment. Nanoparticles (NPs) have gained significant attention due to their versatility, making them highly suitable for applications in bone tissue engineering, advanced imaging techniques, and targeted drug delivery. For diagnosis, NPs enhance imaging contrast and sensitivity by integrating targeting ligands, which significantly improve the specific recognition and localization of tumor cells for early detection. For treatment, NPs enable targeted drug delivery, increasing drug accumulation at tumor sites while reducing systemic toxicity. In conclusion, understanding bone microenvironment and using the unique properties of NPs holds great promise in improving disease management, enhancing treatment outcomes, and ultimately improving the quality of life for patients with malignant bone tumors. Further research and development will undoubtedly contribute to the advancement of personalized medicine in the field of bone oncology.
Keywords: Malignant bone tumors, Bone microenvironment, Nanoparticles, Diagnosis, Treatment
Introduction
Malignant bone tumors are one of the major serious diseases in the clinic [1]. Malignant bone tumors are characterized by aggressive growth and the ability to spread, originating from bone tissue [2]. Based on their origin and formation mechanism, malignant bone tumors can be divided into primary malignant bone tumors and secondary malignant bone tumors.
Primary malignant bone tumors
Primary malignant bone tumors refer to tumors that originate from tissues within the bone itself. The most common primary malignant bone tumor is osteosarcoma (OS) [3]. Its pathological features include the formation of malignant tumor cells that destroy normal bone tissue, often accompanied by local pain, swelling, and fractures. In addition to OS, primary malignant bone tumors also include giant cell tumors of bone, multiple myeloma, and chondrosarcoma. OS commonly arises at the metaphysis of long bones such as the proximal tibia and distal femur, while primary spine, pelvis, and sacrum lesions are relatively rare [4]. This malignancy exhibits a high propensity for local invasion and early metastasis, with some patients developing metastatic disease before the onset of clinical symptoms. The lungs are the most frequent site of metastasis, followed by other bones [5, 6]. Common symptoms include bone pain, swelling, and palpable masses. Early diagnosis is crucial for improving survival rates in OS patients [7]. Standard treatment protocols for OS involve neoadjuvant chemotherapy, surgical resection of the tumor, and adjuvant chemotherapy [8]. The introduction of chemotherapy in the late 1970s, coupled with limb-salvage surgeries, has significantly improved the five-year survival rate for OS [9]. The high heterogeneity and complex biological behavior of OS, along with the presence of lung or distant metastases at initial diagnosis in some patients. However, patients with lung or other distant metastases have a significantly reduced five-year survival rate. Furthermore, chemotherapy for OS is often accompanied by severe toxic side effects, including cardiac toxicity, gastrointestinal toxicity, and bone marrow suppression, which limit the potential for further intensification of chemotherapy [10–12]. The high heterogeneity and complex biological behavior of primary malignant bone tumors, combined with lung or distant metastases present at initial diagnosis in some patients, make it particularly challenging to treat [10].
Secondary malignant bone tumors
Secondary malignant bone tumors originate from other organs or tissues and metastasize to the bone. The most prevalent form of secondary malignant bone tumor is bone metastasis (BM) [3]. Apart from metastatic bone tumors, secondary malignant bone tumors also include leukemic infiltration of bone, lymphoma bone tumors, and others. BM is another prevalent bone tumor disease and a frequent complication of advanced malignant tumors. It is the third most common site of tumor metastasis, following the liver and lungs [13, 14]. With the continuous advancement of medical science and technology, the survival period of patients with malignant tumors has significantly extended, increasing the likelihood of BM. BM can result in severe bone pain, pathological fractures, and neurological or spinal cord injuries, collectively known as skeletal-related events (SREs) [15, 16], which critically impact the quality of life and survival of patients with malignant tumors. Therefore, early identification and prediction of tumor BM trends will facilitate the timely application of current therapeutic measures for effective intervention, reducing patient suffering and the incidence of SREs, and improving patient quality of life [17, 18]. Currently, clinical treatment primarily aims to alleviate patient pain and prevent or delay BM, with main methods including chemotherapy, endocrine therapy, and radiotherapy [19]. However, the challenge in treating BM lies in its high invasiveness and resistance to treatment [19, 20]. Consequently, finding new treatment strategies has become a focus of research.
Nanoparticles for diagnosis and treatment
Innovative diagnosis and treatment methods based on nanoparticles (NPs) have emerged, offering new hope for the management of malignant bone tumors and potentially extending patient survival while improving quality of life. Firstly, NPs have made significant strides in the controlled release of tumor drugs [21], allowing for targeted delivery and precise temporal and spatial control of drug release, which enhances therapeutic efficacy and reduces toxicity [22]. Additionally, NPs can be tailored for direct therapeutic applications [23, 24], such as in photothermal therapy (PTT) and photodynamic therapy (PDT) [25], where photosensitive NPs convert near-infrared light into heat to kill tumor cells [26, 27]. The ability to encapsulate anti-tumor drugs within NPs enhances drug delivery specifically to tumor cells [28]. Moreover, NPs can be employed to construct bone tissue engineering scaffolds with adjustable surface properties, pore structures, and chemical compositions to promote bone repair and regeneration in patients with OS and BM [29, 30]. Furthermore, advancements in bioimaging technologies [31], including fluorescence probes, magnetic resonance imaging (MRI), and positron emission tomography (PET), leverage NPs to achieve precise treatment of OS and BM [32–34]. Obviously, the development of NPs presents new opportunities for the treatment and diagnosis of OS and BM.
The bone microenvironment is crucial for malignant bone tumors as it influences tumor growth, metastasis, and treatment response. This review covers the mechanisms of the bone microenvironment in malignant bone tumors and the recent applications of NPs in the diagnosis and treatment of malignant bone tumors, particularly OS and BM. Additionally, we highlight the current advantages of nanotherapy and explore future directions for the application of NPs in OS and BM (Scheme 1).
Scheme 1.
Schematic illustration of the bone microenvironment in primary and secondary malignant bone tumors, and the use of nanoparticles for diagnosis and treatment
The microenvironment of malignant bone tumors
The response of tumor cells to treatment depends not only on the complexity of the genomic aberrations they harbor but also on the numerous dynamic characteristics of the tumor microenvironment (TME) in which they reside [35–37]. A key feature of the TME is immunosuppression, which allows tumor cells to evade immune surveillance while promoting tumor inflammation and angiogenesis [38]. The heterogeneity of the TME significantly contributes to tumor metastasis, recurrence, and drug resistance [39, 40]. Further research on the regulation and influencing factors of the TME will aid in developing more targeted therapeutic strategies, improving the prognosis and quality of life for patients. Such studies are expected to provide important information about the mechanisms and treatment options for malignant bone tumors.
The microenvironment of primary malignant bone tumors
The most common primary malignant bone tumor is OS. OS often originates in regions of bone growth and development, which have unique mechanisms of bone cell differentiation [41]. During OS development, the bone microenvironment often becomes imbalanced, leading to abnormal bone remodeling processes that create favorable conditions for the survival and proliferation of OS cells [42]. Within the OS microenvironment, different types of bone cells play crucial roles. OS cells can mimic the function of osteoblasts, producing bone-like tissue. Tumor cells in OS can differentiate into osteoblast-like cells, generating tumor bone and tumor-related osteoid tissue [43, 44]. Depending on the degree of differentiation of these tumor osteoblasts, OS can present as either sclerotic or osteolytic OS [45, 46]. Additionally, marrow stem cells play an important role in the formation and progression of OS [47, 48]. Marrow stem cells have the ability to self-renew and differentiate into all types of blood cells, including red blood cells, white blood cells, and platelets [49]. They participate in regulating immune responses, secreting cytokines, and promoting bone remodeling. These cells can support OS growth and invasion by providing a supportive microenvironment [50, 51]. Osteoclasts, responsible for bone remodeling by resorbing bone tissue, may exhibit abnormal functions that contribute to OS development [52]. OS cells can promote tumor invasion and bone destruction by enhancing osteoclastogenesis and osteoclast activity. This bone destruction releases a large number of growth factors and cytokines, further stimulating the proliferation and invasion of OS cells [53, 54]. For patients with non-metastatic bone tumors, modern treatments have resulted in a five-year survival rate of more than 70% for most patients. Understanding the roles of different types of bone cells in the OS microenvironment is essential for gaining deeper insights into the mechanisms of OS initiation and progression.
Research has found that the immune microenvironment of OS contains immune inhibitory factors, such as TAMs and regulatory Tregs [55]. These cells can inhibit immune responses and promote tumor evasion of immune surveillance. Additionally, tumor cells can secrete immune inhibitory factors, such as PD-L1 and CTLA-4 [56], to suppress the activity of immune cells, thereby avoiding immune attacks [55]. Immune checkpoints are crucial regulatory mechanisms in the immune system that control the activity and function of immune cells [57]. In OS, abnormal activation of immune checkpoint signaling pathways leads to immune tolerance, allowing tumor cells to escape immune attack. Immune checkpoint inhibitors are a class of drugs that target immune checkpoint signaling pathways [58]. By blocking the effects of signaling pathways such as PD-1/PD-L1 or CTLA-4 [59, 60], these inhibitors can restore the activity of immune cells and enhance the immune system’s ability to attack tumors [57]. The immune microenvironment of OS and the immune checkpoints between immune cells and OS cells are shown in Fig. 1.
Fig. 1.
The immune microenvironment of osteosarcoma and the immune checkpoints between immune cells and osteosarcoma cells. A) The immune microenvironment of osteosarcoma includes various immune cells (such as infiltrating lymphocytes, macrophages, dendritic cells, etc.), immune suppressive cells (such as regulatory T cells, tumor-associated macrophages, etc.), cytokines (such as tumor necrosis factor, interleukins, etc.), chemical signaling molecules (such as chemokines and extracellular matrix components), and immune checkpoint molecules (such as PD-L1 and CTLA-4). B) The immune microenvironment in osteosarcoma is often described as immunosuppressive and promoting tumor escape. The increase in regulatory T cells may inhibit anti-tumor immune responses. Polarization of macrophages can lead to the emergence of an immunosuppressive phenotype. The presence of tumor-associated macrophages may contribute to increased tumor invasion and metastasis. Additionally, tumor cells can evade immune surveillance by upregulating immune checkpoint molecules, thus reducing the activity of immune cells
The microenvironment of secondary malignant bone tumors
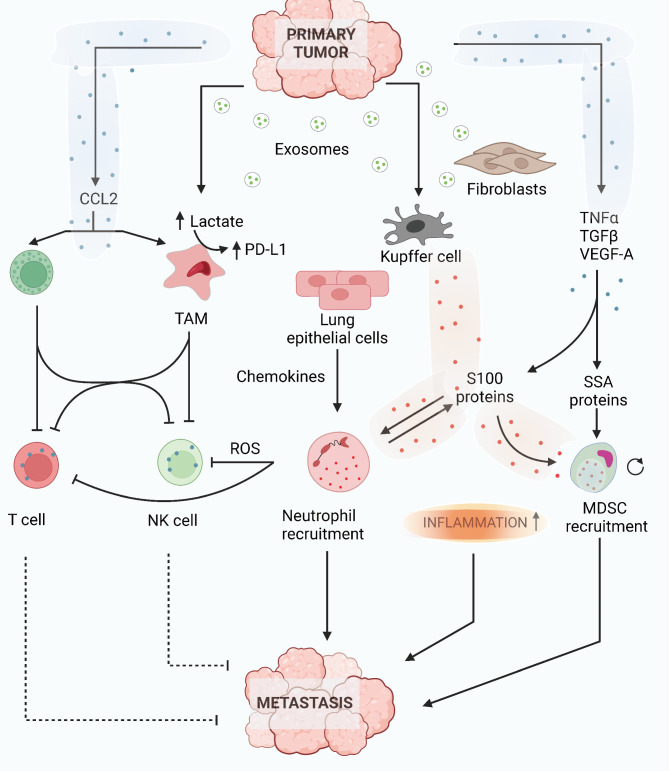
The primary tumor induces early changes in the microenvironment of distant organs that are initially free of cancer cells [61], thereby creating a favorable environment for cancer cell infiltration and metastasis initiation [62]. This leads to the formation of the pre-metastatic niche (PMN), a microenvironment primed for tumor metastasis [63]. The PMN is established through the interaction between tumor-secreted factors and resident stromal cells, as well as bone marrow-derived cells in these distant organs [64, 65]. This process gradually shifts the tissue homeostasis towards a dysfunctional environment, facilitating the colonization of circulating tumor cells (CTCs). Key characteristics of this process include vascular leakage, lymphangiogenesis, extracellular matrix (ECM) remodeling, and the development of an immunosuppressive microenvironment [66]. In essence, the primary organ induces a cascade of events that prepares the distant target organ to receive metastatic tumor cells [67]. Tumor-secreted factors, along with signals from stromal cells, influence the recruitment and activation of immune cells, thereby mediating PMN formation and facilitating subsequent tumor cell colonization at secondary sites. The mechanism is shown in Fig. 2. Understanding the mechanisms at different stages of the metastatic process and accurately dissecting the specific metastatic microenvironment are urgent requirements in cancer research. This is also a crucial step toward identifying new therapeutic targets.
Fig. 2.
The intricate soluble network creates a pre-metastatic niche. The secretome of primary tumors, including chemokines, growth factors, and tumor vesicles like exosomes, enters the bloodstream and reaches secondary sites before cancer cell dissemination. This creates a tumor-supportive and immune-suppressive microenvironment. Components such as TNF-α, TGF-β, and VEGF-A induce the secretion of SAA proteins, recruiting MDSCs to organs. S100 proteins, induced by various mechanisms, act on tissue-resident cells and have diverse roles in the metastatic niche. Exosomes stimulate lactate secretion from TAMs, upregulating PD-L1. CCL2 attracts TAMs and Tregs to premetastatic niches, deterring NK cells and T cells. Additionally, neutrophil-secreted ROS impact NK cell and T cell cytotoxicity. These factors collectively promote metastatic signaling, exacerbating disease progression
Bone metastasis
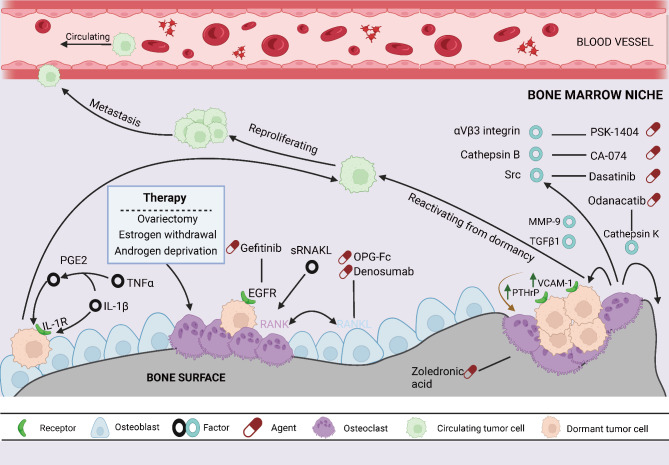
The most prevalent form of secondary malignant bone tumor is BM. The occurrence of BM is a hallmark of advanced-stage cancer [68]. The tendency of various tumor cells to metastasize to bone tissue is attributed to the involvement of numerous cytokines required for bone remodeling [69]. The diverse bone microenvironment is highly attractive to metastatic cancer cells seeking nutrients. Research has found that tumor cells can utilize the bone matrix secreted by osteoblasts to maintain their dormant state. Specifically, there exists a complex interaction between tumor cells and osteoblasts. The bone matrix secreted by osteoblasts is rich in growth factors and signaling molecules, which can inhibit the growth and spread of tumor cells. Furthermore, osteoblasts can regulate the gene expression of tumor cells to maintain their dormant state (Fig. 3). Studies have shown that osteoblasts can secrete specific molecular signals, such as CCL3, TGF-β, and SDF-1, to influence the gene expression of tumor cells, thereby suppressing their growth and spread [70]. The bone system is richly vascularized, and the valveless Batson’s plexus, characterized by slow blood flow and large vessel diameter, provides an ideal site for the colonization of invasive tumor cells. Once tumor cells colonize the bone, stromal cells, osteoclasts, and transient cells within the bone microenvironment can promote tumor growth and metastasis through various pathways and molecules [71]. The “osteoclast” theory posits that increased osteoclast activity and the resultant bone resorption are critical in the formation of BM and bone destruction [72, 73] (Fig. 4). BM by tumor cells disrupts bone structure, disturbs the mineral balance, and affects the RANK-RANKL-OPG system [74], leading to further bone destruction and the expansion of metastatic lesions [75].
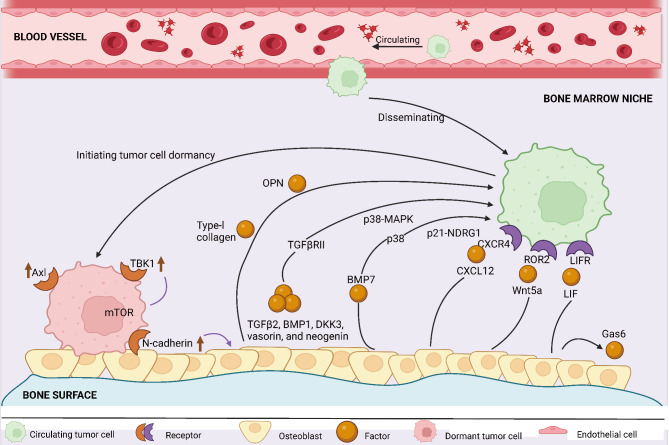
Fig. 3.
Osteoclast-mediated bone resorption reactivates dormant tumor cells. When bone tissue is damaged or stimulated, osteoclasts are activated and cluster at the bone surface. Through the release of acidic protons and enzymes, they lower the pH of the bone matrix, resulting in the breakdown of inorganic salts and collagen fibers. This process leads to bone resorption and the release of growth factors and extracellular matrix components stored in the bone matrix. In the presence of active osteoclasts, dormant tumor cells can be reactivated as the released growth factors and matrix-derived molecules bind to receptors on the tumor cells, leading to the activation of cellular signaling pathways associated with proliferation, survival, and immune evasion. This reactivation process may promote the transition of dormant tumor cells from a quiescent to an active state, facilitating their growth, invasion, and metastasis
Fig. 4.
Osteoblast-mediated bone formation initiates and maintains tumor cell dormancy. Osteoblasts, a crucial component of bone marrow, play a vital role in bone formation and are pivotal in maintaining dormancy in tumor cells. Osteoblasts release growth factors and extracellular matrix components that attract tumor cells to migrate towards bone tissue, facilitating their interaction. This interaction enhances the attachment and colonization of tumor cells within the bone marrow space. Moreover, osteoblasts regulate the secretion of cytokines and bone-associated proteins, contributing to the maintenance of tumor cell dormancy. This dormancy state prevents tumor cell proliferation and metastasis, effectively keeping them quiescent. Additionally, osteoblasts inhibit inflammatory responses and modify the bone marrow microenvironment to sustain tumor cell dormancy. Osteoblasts actively maintain tumor cell dormancy during bone formation, thereby impeding their malignant expansion and metastasis through various mechanisms
The occurrence and progression of BM are regulated by the complex TME [38]. This microenvironment involves interactions and signaling among various cell types, with bone cells playing a crucial role in the development and progression of BM [39, 76]. Firstly, bone tissue provides a conducive environment for cancer cell migration and colonization, being rich in calcium and bone matrix proteins that offer essential growth and survival factors for cancer cells [7]. Additionally, cancer cells release various factors that promote BM, thereby activating bone remodeling and resorption. Transforming growth factor-beta (TGF-β) stimulates cancer cells to metastasize to bone and promotes the differentiation of bone marrow mesenchymal stem cells (BMSCs) into osteoblasts [77]. Moreover, factors such as fibroblast growth factor (FGF) and interleukin-6 (IL-6) also participate in the BM process [78]. Furthermore, the inflammatory response plays a significant role in the occurrence and progression of BM. Tumor cells produce inflammatory mediators that activate bone marrow macrophages, which in turn induce the formation of BM [79]. Different types of bone cells play distinct roles in the BM microenvironment. Osteoblasts and osteoclasts contribute to imbalances in bone remodeling, promoting abnormal bone formation and resorption, respectively [80, 81]. Tumor-associated macrophages (TAMs) and regulatory T cells (Tregs), among other immune cells, exert immunosuppressive effects within the microenvironment, facilitating tumor immune evasion [82]. Additionally, tumor cells secrete various cytokines and growth factors that further regulate the behavior of various cells in the microenvironment, collectively driving and sustaining the development of BM [54, 83]. BM is a common and serious complication of cancers such as prostate cancers (PCa), breast cancer (BC), and lung cancers, occurring when malignant cells from these tumors spread to the bone via the bloodstream or lymphatic system.
Bone metastasis of prostate cancers
PCa is one of the most common malignant tumors in men. PCa cells exhibit particular aggressiveness, spreading to bones via hematogenous routes. Upon reaching bone tissue, these cells can significantly increase the rate of bone matrix turnover and cause osteoblastic pathological changes [84]. Interactions between PCa cells and bone cells lead to a marked increase in bone matrix turnover and osteoblastic pathological changes [85]. Additionally, alterations in the bone microenvironment facilitate PCa BM. Cancer cells can secrete factors similar to those produced by osteoblasts, interacting with the bone microenvironment and inducing BM [86]. PCa cells can induce bone matrix synthesis, which appears to favor tumor cell growth. Further research into this process may help explain the osteoblastic tendency of BM in cancer cells. PCa cells can also express factors that regulate the bone microenvironment, indirectly affecting osteoblast activity. A deeper understanding of the mechanisms underlying PCa BM and changes in the bone microenvironment will aid in comprehending this disease process and provide critical insights for developing new therapeutic strategies and targeted drugs. Researchers have identified several molecular signaling pathways related to PCa BM, such as Src kinase and RANKL. The development and clinical application of drugs targeting these pathways hold promise for providing new treatment options for PCa patients [87]. By specifically inhibiting the activity of these molecules, targeted drugs can effectively intervene in the interactions between PCa cells and bone marrow mesenchymal cells, thereby inhibiting the formation and progression of BM [88]. Additionally, current research explores new strategies to improve the bone microenvironment in patients, such as using growth factors and bone formation promoters to enhance bone repair and inhibit destruction. The bone microenvironment of the PCa with BM is displayed in Fig. 5.
Fig. 5.
The bone microenvironment of prostate cancer with bone metastasis. In the bone marrow environment (BME), upregulation of BHLHE22 recruited PRMT5, promoting CSF2 expression and increasing levels of immature neutrophils and monocytes. This led to exhaustion of CD8+ and CD4+ T cells. In osteoblasts, downregulation of RBM3 inhibited m6A modification of CTNNB1 mRNA, enhancing CTNNB1 expression and activating the Wnt/β-catenin signaling pathway. Enzalutamide treatment increased PTH1R expression in osteoblasts, causing interaction with TGFBR2 and promoting bone metastasis
Bone metastasis of breast cancer
BC is one of the most common cancers among women. Similarly, in the absence of estrogen or androgen, increased osteoclast activity and bone remodeling lead to the release of osteogenic factors from resorbed bone, creating a favorable environment for the survival and growth of tumor cells. In addition to secreting cytokines, tumor cells can interact with microenvironment cells through the secretion of extracellular vesicles [89]. These vesicles, encapsulating tumor-specific contents, are delivered to the TME and target cells, promoting tumor growth. Understanding the mechanisms behind BC BM and the associated changes in the bone marrow microenvironment is crucial for developing more effective treatment strategies. Researchers are currently seeking drugs that target BC BM to intervene in the interactions between tumor cells and bone marrow mesenchymal cells, thereby halting the progression of bone destruction. Strategies to improve the bone microenvironment, such as using bone formation promoters or inhibitors of bone resorption proteins, are also a focus of current research, aiding in bone repair and blocking abnormal bone destruction. Verbruggen et al. [90] analyzed a mouse model induced with breast tumors and found that early bone lysis manifested as thinning trabeculae and increased cortical thickness, with higher mineral density observed in the bone tissue of tumor-bearing femurs. At six weeks post-inoculation, significant osteolytic lesions appeared in the metastatic femurs, leading to decreased bone volume fraction and mechanical performance. These results suggest that changes in bone tissue composition during BC metastasis may affect the mechanical environment of bones and tumor cells. The bone microenvironment of the BC with BM is displayed in Fig. 6.
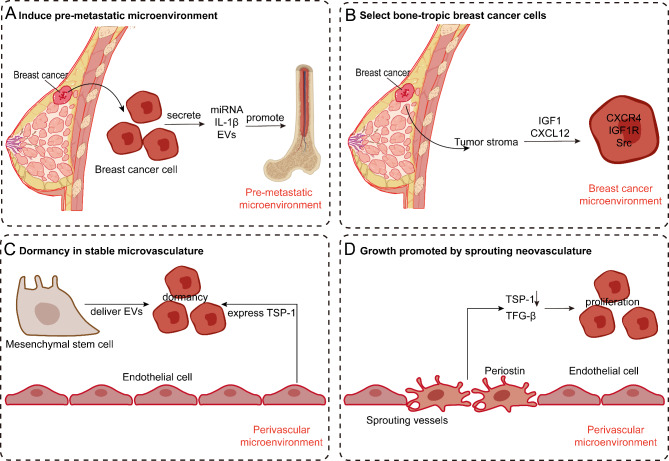
Fig. 6.
The bone microenvironment of breast cancer with bone metastasis. Before breast cancer bone metastasis, the bone microenvironment undergoes several changes. A) Firstly, breast cancer cells secrete miRNA, IL-1β, and extracellular vesicles, promoting the formation of a pre-metastatic microenvironment. B) Secondly, the tumor stroma selectively favors cancer cells expressing CXCR4, IGF1R, and high Src activity, facilitated by CXCL12 and IGF1. C) The perivascular microenvironment regulates the dormancy of breast cancer cells, with endothelial cells expressing TSP-1 to maintain dormancy. Mesenchymal stem cells induce dormancy in breast cancer cells through the delivery of extracellular vesicles. D) Lastly, sprouting blood vessels produce periostin, TGF-β, and reduced TSP-1 secretion, stimulating the proliferation of breast cancer cells. These changes in the bone microenvironment before breast cancer bone metastasis provide a foundation for subsequent metastasis and offer important insights into the mechanisms involved
Bone metastasis of lung cancer
BM of lung cancer is not a random event but rather a result of bone tissue providing a conducive growth environment for tumor cells, thereby inducing an active selection process regulated by various molecules and signaling pathways [91]. Even before lung cancer cells metastasize to the bones, they secrete extracellular vesicles, factors, and other substances that circulate through the blood, acting on the bone microenvironment. This process recruits bone marrow-derived cells and immunosuppressive cells, which collaborate with bone matrix cells to remodel the bone microenvironment, promoting a shift towards an environment favorable for tumor cell colonization and growth, forming a PMN [92, 93]. The formation of this PMN is an early event in the process of BM and plays a crucial role in lung cancer BM. Blocking the formation of the PMN can help reduce the occurrence of lung cancer BM from the source. Understanding the mechanisms behind lung cancer BM and the related changes in the bone microenvironment is essential for developing more effective treatment strategies and drugs. Researchers are currently striving to identify targeted therapies for lung cancer BM to intervene in the interactions between tumor cells and bone marrow mesenchymal cells, thereby halting the progression of bone destruction. Additionally, exploring strategies to improve the bone marrow microenvironment, such as using growth factors and bone formation promoters, can help restore normal bone structure and function.
Nanoparticles for diagnosis of malignant bone tumors
The limitations and challenges for diagnosis in malignant bone tumors
The diagnosis of OS currently relies on a variety of tools and methods, including imaging, histology, molecular biology, and clinical pathology [94, 95]. Imaging is one of the primary tools for diagnosing OS, with commonly used techniques including X-ray, CT scans, MRI, and bone scans [96]. For many years, conventional X-ray radiography has been advantageous in showing bone destruction and sclerosis due to its excellent contrast between bone and soft tissue density [97, 98]. It can also reveal periosteal reaction and Codman triangle caused by the tumor, but it lacks sensitivity in showing soft tissue structures [99]. CT scanning offers the advantage of visualizing detailed bone destruction, such as small lytic lesions and necrotic bone [100]. Additionally, contrast-enhanced CT can provide information about the tumor’s blood supply and its relationship with surrounding vascular and tissue structures [101]. CT is also a convenient, quick, and commonly used technique for detecting metastases in other parts of the body [102, 103]. MRI is unparalleled in displaying soft tissue details of the tumor, though it is less sensitive to new bone formation and calcification [104]. Radionuclide bone scanning is a functional imaging test that helps detect other asymptomatic lesions, revealing whether the cancer has spread to other bones earlier than conventional X-rays [105, 106]. Biopsy methods, such as fine-needle aspiration biopsy, core needle biopsy, or open biopsy, depending on the tumor’s location, size, and the patient’s overall condition [107, 108]. The obtained tissue samples are processed through fixation, embedding, sectioning, and staining, followed by microscopic examination to determine the tumor’s cell type, degree of differentiation, and tissue structure [109, 110]. Additionally, immunohistochemistry and molecular genetics can further assist in the diagnosis and classification of OS [111–113]. Despite the importance of imaging and histology in diagnosing OS, they have some limitations [114, 115]. Imaging cannot accurately distinguish between inflammation and malignancy, potentially leading to misdiagnosis [116, 117]. Histological biopsy requires surgical tissue removal, which carries certain risks and limitations [118]. Overall, the diagnosis of OS requires a comprehensive application of imaging, histology, and related clinical pathology examinations. Future research and technological developments are expected to further improve the accuracy of OS diagnosis, providing patients with more precise treatment options and prognostic assessments.
Currently, tools and methods commonly used for diagnosing BM include imaging, tumor marker detection, and bone marrow aspiration. X-rays can show bone destruction or new bone formation caused by BM, but their sensitivity is relatively low, typically only detecting BM when mineral loss in the bone exceeds 25–50% [119, 120]. CT scans, combined with X-ray images, provide more detailed information about bone structures and can reveal invasive bone destruction and soft tissue masses, offering more comprehensive structural details [121, 122]. MRI can directly visualize BM, particularly useful in T1-weighted sequences where metastatic lesions appear as areas of decreased signal intensity, and is especially helpful in diagnosing spinal vertebral compression fractures [123, 124]. Tumor marker detection is a common auxiliary diagnostic method that evaluates the presence and extent of BM by measuring specific tumor markers in the blood [125]. For instance, BM often leads to elevated levels of bone-specific alkaline phosphatase (BSAP) and alkaline phosphatase (ALP) [126, 127]. The advantage of tumor marker detection is its simplicity and utility for follow-up monitoring, but its limitation lies in the fact that elevated markers do not definitively diagnose BM [128, 129]. Bone biopsy, either through needle aspiration or surgical biopsy, involves obtaining tissue samples from bone lesions for pathological evaluation to confirm the presence of BM [130, 131]. Bone marrow aspiration, combined with pathological analysis, can determine whether cancer cells are present in the bone marrow and assess the type and differentiation degree of the cancer cells [132, 133]. In conclusion, the diagnosis of BM requires the comprehensive application of imaging, tumor marker detection, and bone marrow aspiration. Future research and technological advancements are expected to further improve the accuracy and early diagnosis rate of BM, providing patients with more precise treatment options and prognostic assessments. As with OS, a comprehensive diagnostic approach is necessary, with future research expected to enhance early detection and accuracy.
Together, these diagnostic approaches for OS and BM underscore the importance of integrating multiple methods to achieve precise diagnosis and improve treatment outcomes. Despite the advancements in imaging and histological techniques for diagnosing OS and BM, challenges such as limited sensitivity in early detection, difficulty distinguishing between benign and malignant lesions, and the invasiveness of biopsy procedures remain. These limitations highlight the need for more innovative approaches to enhance diagnostic accuracy while minimizing patient discomfort. NPs have emerged as a promising solution, offering the potential to revolutionize cancer diagnostics. By leveraging their unique properties, such as targeted delivery, enhanced imaging contrast, and the ability to transport diagnostic agents directly to tumor sites, NPs can improve the sensitivity and specificity of both imaging and biomarker detection, paving the way for more precise and non-invasive diagnostic techniques for malignant bone tumors.
The nanoparticles for diagnosis in malignant bone tumors
The application of NPs in cancer diagnosis holds great promise [21]. Various NPs, such as gold NPs (AuNPs), quantum dots, and magnetic NPs, have been extensively researched and developed to enhance the accuracy and sensitivity of early cancer diagnosis [22, 134]. AuNPs can be used in surface-enhanced Raman detection, photoacoustic imaging, CT imaging, and fluorescence imaging [135, 136]. AuNPs can serve as imaging agents by binding to tumor-specific markers, thus improving the ability to identify tumor tissues [137]. Additionally, AuNPs can utilize the surface-enhanced Raman scattering (SERS) effect to detect and amplify signals of cancer-specific markers, increasing diagnostic sensitivity [138, 139]. Another commonly used nanomaterial is quantum dots, which are luminescent NPs with adjustable luminescent properties [140, 141]. Quantum dots are among the most widely used fluorescent probes, characterized by broad excitation, narrow emission, size tunability, and high quantum yield, making them widely applicable in various research fields [142, 143]. Quantum dots can be modified to carry targeting ligands, such as antibodies or peptides, which can specifically recognize tumor cells or tumor vasculature, thereby achieving targeted imaging of tumors [144]. MRI is a medical imaging technology with high sensitivity, rapid detection, and high spatial resolution, enabling real-time detection of the three-dimensional (3D) spatial distribution of magnetic NPs in vivo [145]. In addition to the aforementioned NPs, other NPs have also been extensively researched and applied in cancer diagnosis. For instance, iron oxide NPs, known for their good biocompatibility and superparamagnetic properties, can be used for MRI-based tumor localization and differentiation [146]. Carbon nanotubes possess novel capabilities for transmitting, storing, and recovering light wave signals, which can be used for targeted delivery and imaging of tumor cells [147]. These NPs each have unique advantages and application potential in cancer diagnosis [148]. Overall, the potential of NPs in cancer diagnosis is immense. However, it is essential to gain a deeper understanding of the long-term safety, biodistribution, and metabolic properties of NPs, as well as their potential impacts on human health [149, 150]. It is also crucial to emphasize the safety evaluation and stringent regulatory measures for NPs to ensure their effective application in cancer diagnosis [151, 152]. The diverse range of NPs offers a multitude of approaches for improving cancer diagnosis. In future research and technological development, we hope to further enhance the performance and safety of NPs, providing more reliable tools and methods for early cancer diagnosis.
Research shows that nanomaterials have significant application value in the diagnosis of OS and BM. Wang et al. [153] found that an ssDNA aptamer LP-16 could bind to highly metastatic 143B cells but not to non-metastatic U-2 OS and normal hFOB 1.19 cells. Additionally, in vivo and clinical tissue imaging, results demonstrated the targeting effect of LP-16. The study suggested that LP-16 might target specific membrane proteins on the cell surface, indicating that LP-16 could be an effective molecular probe for the diagnosis and treatment of OS. Anzidei et al. [154] noted that antibody-drug conjugate (ADC) and Ktrans values correlated with pain scores in treated lesions, as measured by the Visual Analog Scale (VAS), while functional MRI techniques such as DWI and DCE could serve as objective imaging markers for treatment response. Early evaluation of treatment response using non-invasive imaging techniques is crucial. Sekita et al. [155] used nano-computed tomography (nano-CT), histology, microbeam XRD, and nanoindentation to observe disrupted osteocyte networks and changes in the collagen/biological apatite (BAp) microstructure in cancerous bones. They found that the disruption of osteocyte/matrix coupling affected the mechanical function of the bone. The results indicated that during cancer BM, the osteocyte network and collagen/BAp microstructure might be simultaneously disrupted, increasing the risk of brittle fractures.
Using NPs as sensors to track the activity of viruses within living organisms is a popular area of research. This approach is particularly useful for studying the behavior of cancer cells and other cellular activities within the human body, and can also be used to detect the effects of drugs on viruses [156, 157]. Additionally, NPs can be attached to biomolecules, leveraging their luminescent properties to study the behavior of these molecules [158]. For instance, NPs are much smaller than human cells and possess luminescent capabilities. By introducing these NPs into human organs and irradiating them with near-infrared (NIR) light from outside the body, the NPs can emit light, allowing researchers to track and understand changes in human cells, thereby achieving the goal of tracking viruses [159]. Significant progress has been made in tracking and locating cancer cells within the human body. It has been discovered that cancer cells have a particular ability to phagocytize NPs, which helps in tracking their activities within the body [160].
Nanoparticles for treatment of malignant bone tumors
The treatment of bone tumors typically involves a combination of surgery and systemic therapies [161]. For OS, preoperative neoadjuvant chemotherapy is used to shrink the tumor, followed by complete surgical resection and postoperative chemotherapy [162]. However, chemotherapy can lead to significant toxicity, such as cardiotoxicity and nephrotoxicity. In the case of BM, treatment focuses on controlling primary cancer and managing bone complications through systemic chemotherapy, targeted therapies, and local options like radiation. Treatments in both OS and BM are associated with toxicity risks.
NPs overcome several limitations of traditional therapies by improving drug retention and permeability at tumor sites. Through various surface modifications and customizable physical and chemical properties, nanoparticle carriers are highly adaptable, making them ideal candidates for targeting specific tumor cells. Targeted drug delivery via NPs minimizes the harmful effects of anticancer drugs on healthy cells while enhancing the efficacy and selectivity of the drugs toward cancer cells. NPs, through processes of loading, targeting, and controlled release, offer advanced capabilities in drug delivery. This precise approach improves the overall effectiveness of therapies and addresses the limitations of traditional drug delivery methods. Herein, NPs used for malignant bone tumors are introduced through drug-loading NPs, targeting NPs, release NPs, and so on.
Nanoparticles for loading drugs
Drug-loaded NPs are an important drug delivery system characterized by their extremely small size, which allows effective encapsulation of drug molecules within the particles and their transport to target tissues or cells via the bloodstream [163, 164]. Traditional drug delivery systems have certain limitations, such as drug instability, low solubility, and metabolism outside the body. In contrast, drug-loaded NPs offer advantages such as controllability, high loading capacity, and protective properties, which can overcome these issues [165]. Additionally, drug-loaded NPs can be surface-modified to achieve specific targeting of drugs to diseased tissues or cells, thereby enhancing therapeutic efficacy and reducing side effects [166].
Haghiralsadat et al. [167] developed a dual-targeted liposome for the delivery of doxorubicin (DOX) and JNK-interacting protein 1 (JIP1) small interfering RNA (siRNA) to OS cells. Targeted co-delivery of DOX and JIP1 siRNA resulted in increased cytotoxicity and reduced JIP1 mRNA levels in OS cells. The study demonstrated that this dual-targeted liposome could effectively treat metastatic OS with multidrug resistance (Fig. 7A). Li et al. [168] developed a nano-delivery system based on hydrophobic polyesteramide to successfully deliver apatinib, enhancing its distribution within tumors and inhibiting OS stemness. This nanoapatinib delivery system overcame the resistance of stem-like cells to tyrosine kinase inhibitor (TKI) therapy, effectively inhibiting tumor growth with minimal side effects (Fig. 7B). He et al. [169] found that cells cultured in 3D gelatin methacryloyl (GelMA) microspheres exhibited stronger tumor stemness, proliferation, migration ability, and chemotherapy drug resistance. Additionally, 3D-cultured cells showed higher tumorigenicity in mice, with shorter tumor onset times, larger tumor volumes, severe bone destruction, and higher mortality rates. The study indicated that GelMA hydrogel microspheres provide a new method for constructing tumor models in vitro, particularly for OS research. Salaam et al. [170] developed ND-DGEA conjugates to target α2β1 integrin overexpressed in prostate cancer during metastasis. The ND-DGEA conjugates significantly enhanced the efficacy of DOX, improving delivery capacity and reducing toxicity. The study demonstrated a new approach for targeted drug delivery using nanodiamond-DGEA peptide conjugates to deliver DOX to prostate cancer, aiming to increase treatment specificity and reduce toxicity. Fiona et al. [171] developed a miR-29b nanoparticle formulation for delivery in order to achieve local sustained release of therapeutic agents, and experiments were also performed to investigate its role in restoring bone homeostasis while inhibiting tumor growth. The strategy of combining miR-29b with systemic chemotherapy was effective in inhibiting tumor growth, increasing survival in mice, and significantly reducing osteolysis compared to chemotherapy alone, thus normalizing the dysregulation of tumor-induced osteolytic activity. Wang et al. [172] developed a cancer-targeted nano-system (Bm@PT/Enz-miR26a) for the co-delivery of enzalutamide (Enz) and miR26a to enhance therapeutic efficacy. The EZH2/SFRP1/WNT5A axis was found to be associated with Enz resistance and BM, providing clues for new therapeutic avenues. The study suggested that targeting this axis could help overcome resistance and BM in prostate cancer (Fig. 7C). Yu et al. [173] developed a cell membrane-camouflaged CS-polypyrrole nanogel platform for the co-delivery of docetaxel and RANK siRNA. In vivo experiments in a mouse model showed that this platform effectively inhibited tumor growth and prevented metastasis. The nanogel successfully inhibited prostate cancer BM by downregulating the RANK/RANKL signaling pathway, enhancing the potential for chemotherapy and anti-metastatic treatment of prostate cancer (Fig. 7D). Targeted therapy using drug-loaded NPs in OS and BM was shown in Table 1.
Fig. 7.
Drug-loaded NPs that transport drug molecules to target tissues or cells. A) Schematic representation of process. The novel stealth, Ysa peptide-targeted liposomal DOX-siRNA was synthesized with the pH gradient method. Reproduced (Adapted) under terms of the CC-BY license [167]. Copyright 2018, Dove Medical Press Ltd. B) Schematic of the proposed dual-therapeutic role localized delivery of miR-29b: pBAE NPs have in treating OS. Reproduced (Adapted) with permission [171]. Copyright 2023, wiley-VCH GmbH. C) Schematic illustrations of the construction and mechanism of action of Bm@PT/Enz-miR26a in the bone-tumor microenvironment. Reproduced (Adapted) with permission [172]. Copyright 2024, Springer Nature. D) Schematic illustration of the synthesis CH-PPy NGs/DTX/siRANK@CM for combinational chemotherapy and gene therapy of PCa. Reproduced (Adapted) with permission [173]. Copyright 2024, Wiley-VCH GmbH. Abbreviations: Nanoparticles, NPs; DOX, doxorubicin; DOTAP, N-[1-(2,3-dioleoyloxy)propyl]-N, N,N-trimethylammonium methyl-sulfate; DPPC, 1,2-dipalmitoyl-sn-glycero-3-phosphocholine; OS, osteosarcoma; CRPC, castration-resistant prostate cancer; PLGA, Poly (lactic-co-glycolic acid) ; BMSC, bone marrow mesenchymal stem cell; Enz, enzalutamide; DTX, docetaxel; CH-PPy NG, chitosan-polypyrrole nanogel; PCa, prostate cancers
Table 1.
Targeted treatment using drug-loaded nanoparticles in malignant bone tumors
| Type of carrier | Targeting ligand | Anti-cancer agent | Effects | Mechanism and pathway | Refs. |
|---|---|---|---|---|---|
| JIP1/DOX/liposome | JIP1 | DOX | Enhance toxicity toward OS cells and may be effective for the treatment of metastatic OS | / | [167] |
| Nanoscale delivery system | TKI | Apatinib | Suppress OS stemness and enhance OS stem-like cell apoptosis, and promote TKI therapy | / | [168] |
| GelMA | / | DOX | Promote tumorous stemness, proliferation and migration, osteoclastogenetic ability, and resistance to DOX | / | [169] |
| ND-DGEA + DOX | DOX | Enhence the delivery and efficacy of DOX | / | [170] | |
| Bm@PT/Enz-miR26a | miR26a | Enzalutamide | Reverse Enz resistance and synergistically shrink tumor growth, invasion, and metastasis | EZH2/SFRP1/WNT5A | [172] |
| CH-PPy NG | / | Docetaxel | Enhance chemotherapy and anti-metastasis treatment of prostate cancer | RANK/RANKL | [173] |
Various metal NPs, such as gold, silver, and copper NPs, have been reported for the treatment of bone cancer. Shen et al. [174] utilized the excellent biocompatibility and photothermal properties of liquid metal (LM) NPs to develop a targeted LM-based drug delivery system with superior photothermal performance and dual-response drug release triggered by acid and near-infrared (NIR) light. This system aims to enhance drug penetration into deep tumors by remodeling the tumor stromal microenvironment (TSM) while combining mild photothermal therapy with chemotherapy to inhibit the progression of BC BM. AuNPs show great promise for biomedical applications, but their toxicity remains a key concern. Studies indicate that the toxicity of AuNPs depends on factors such as size, charge, coating, and exposure levels. These particles can induce oxidative stress by generating reactive oxygen species (ROS), leading to mitochondrial damage, inflammation, and DNA damage. Smaller NPs, particularly those around 10–20 nm, are reported to have higher toxicity due to their increased surface area and reactivity [175]. Additionally, AuNPs can accumulate in vital organs like the liver, spleen, kidneys, and lungs, potentially causing long-term organ damage. Their clearance from the body is often inefficient, raising concerns about chronic toxicity, especially when used in high doses or prolonged therapies [176, 177]. Currently, increasing research efforts are addressing the toxicity issues associated with AuNPs, and AuNPs are gradually emerging as highly promising nanocarriers. Liao et al. [178] developed a dual-functional hydrogel composed of gelatin methacrylate (GelMA), chondroitin sulfate methacrylate, gold nanorods (GNRs), and nano-hydroxyapatite (nHA). This hybrid hydrogel leverages strong photothermal effects to eradicate residual tumors post-surgery while promoting bone regeneration. It offers both tumor treatment and bone regeneration capabilities, showing significant potential for bone tumor therapy and providing new materials and strategies for addressing complex conditions involving tumors and bone defects.
In addition, CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeats. Based on the different types of Cas proteins, the CRISPR/Cas system is divided into three types: I, II, and III, with Cas9 belonging to the Type II CRISPR system. Cas9 nuclease contains two conserved nuclease domains, HNH and RuvC, which, guided by both crRNA and tracrRNA, can specifically cut double-stranded DNA. The cleavage site is typically located 3 nucleotides upstream of the Protospacer-Adjacent Motif (PAM). Researchers have fused crRNA and tracrRNA to create a chimeric single guide RNA (sgRNA) [179]. Under the guidance of sgRNA, Cas9 is directed to the target site near the PAM sequence, where it induces a double-strand break (DSB). The host cell responds to the DSB through two different mechanisms: Non-Homologous End Joining (NHEJ) or Homology-Directed Repair (HDR), leading to insertions/deletions or frameshift mutations in the target DNA. When donor DNA is provided as a homologous recombination template, the cell uses HDR to precisely insert, delete, or replace bases at specific locations. Viral vectors are the most commonly used delivery carriers for the CRISPR/Cas9 system, but they pose risks such as immunogenicity and mutagenesis in the host, limiting their applications. Compared to viral vectors, non-viral vectors offer advantages like lower immunogenicity and no risk of endogenous viral recombination [180, 181]. In recent years, nanoparticle formulations have gained widespread attention as delivery systems. Chitosan (CS) is a well-suited polymer carrier for delivering CRISPR/Cas9 systems. Liu et al. [182] designed a dual-targeting delivery system based on polymer/inorganic hybrid NPs. This system encapsulated CRISPR/Cas9 plasmids targeting CDK11 knockout within an inorganic core composed of protamine sulfate, calcium carbonate, and calcium phosphate through co-precipitation. Carboxymethyl chitosan derivatives were then electrostatically attached to the nanoparticle surface to enhance cellular uptake and nuclear transport efficiency. The dual-targeting delivery system successfully transported CRISPR/Cas9 plasmids into the nuclei of MCF-7 BC cells, achieving highly efficient genome editing, with CDK11 protein levels reduced by more than 90%. In the future, the combination of CRISPR/Cas9 with nanomaterials offers new hope for the treatment of bone tumors.
Nanoparticles for targeting
Mechanisms of tumor targeting by nanoparticle drug delivery systems
Based on targeting strategies, nanoparticle drug delivery systems can be classified into passive and active targeting systems. Passive targeting relies on the passive accumulation of NPs in tumor tissues, primarily through the enhanced permeability and retention (EPR) effect. This effect occurs due to the leaky vasculature and poor lymphatic drainage in tumor tissues, allowing NPs to accumulate at the tumor site. The dimension of nanocarriers is a critical determinant for its permeation and retention within tumors, limited by the gaps in tumor blood vessels, which are typically within the 200 ~ 800 nm range. Furthermore, the size of nanomedicines significantly impacts their excretion pathways: particles smaller than 6 nm are usually eliminated via renal filtration, whereas those larger than 500 nm tend to be cleared by the reticuloendothelial system. Hence, the ideal size range for nanocarriers is roughly 20 ~ 200 nm.
On the other hand, active targeting involves attaching targeting molecules to the surface of nanocarriers, allowing them to specifically identify cells. Various ligand-modified nanocarriers facilitate receptor-mediated endocytosis by binding to receptors present on cell surfaces. In addition, the development of biomimetic NPs for bone tumor treatment is also a promising approach. Examples include hydroxyapatite (HAP) NPs and exosome-derived NPs. These biomimetic systems offer unique advantages in targeting and treating bone tumors due to their natural compatibility with biological systems.
HAP nanoparticles
HAP NPs (nHAs) are an important biodegradable material widely used in biomedical applications. HAP is an inorganic compound with a structure similar to calcium phosphate bone tissue, thus exhibiting excellent biocompatibility and bioactivity [183]. NHAs have a large specific surface area and small particle size, giving them unique physical and chemical properties [184]. Moreover, nHAs possess a certain surface charge, allowing for surface modification by introducing various functional groups to enhance their bioactivity and control release behavior [185]. NHAs have extensive potential applications in the biomedical field. Firstly, nHAs can serve as bioactive scaffold materials for bone repair and remodeling, promoting the adhesion and proliferation of bone cells while providing an environment similar to calcium phosphate bone tissue, thus aiding in bone formation and regeneration [186]. Secondly, nHAs can be used in drug delivery systems, encapsulating drugs within the NPs to achieve precisely controlled release and targeted delivery, thereby improving therapeutic efficacy and reducing side effects [187]. Additionally, nHAs can be applied in bioimaging and diagnostics [188]. By modifying the surface of the NPs, the enhancement of biomarkers and cell imaging can be achieved, providing more accurate diagnostic results.
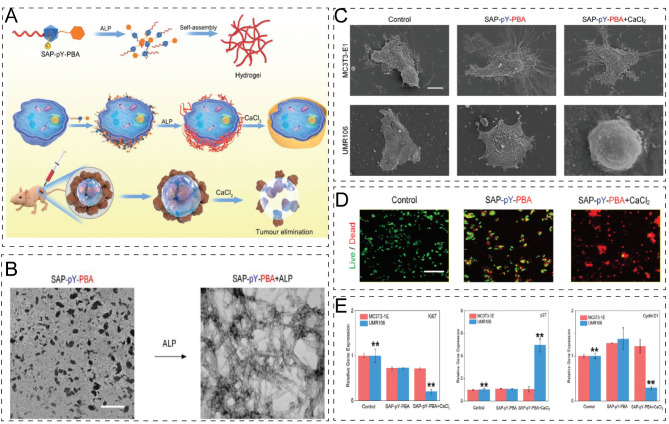
Wu et al. [189]found that the low surface charge density and nanoscale roughness of HAP facets facilitated the unfolding of fibronectin (Fn). Elevated angiogenic and pro-inflammatory secretions were associated with more unfolded Fn adsorbed on nanoscale rough HAP facets with low surface charge density. The study suggested that the complex interactions between crystal surface properties, protein deposition, and cancer cell interactions could potentially impact tumor growth and BM. Wang et al. [190] conducted in vivo studies using a mouse model and discovered that nHAs effectively inhibited tumor growth. RNA-seq analysis revealed that nHA treatment led to the downregulation of the FAK/PI3K/Akt signaling pathway in OS-732 cells. Han et al. [191] incorporated silk fibroin (SF) and nHA into scaffolds for bone regeneration. The use of supercritical CO2 technology allowed precise control over scaffold properties, mimicking the natural bone structure. The study highlighted the potential of CM-PDA/SF/nHA scaffolds in achieving chemophotothermal synergistic effects and bone regeneration, emphasizing the importance of developing multifunctional platforms for treating OS and promoting bone repair. Zhang et al. [192] developed a novel scaffold combining n-HA, MXene, and g-C3N4 for bone tumor treatment and tissue regeneration. This scaffold not only enhanced photothermal and photodynamic performance but also restricted OS cell growth through n-HA, boosting its antitumor capability. Moreover, the scaffold promoted osteogenic differentiation and new bone formation, potentially preventing tumor recurrence and accelerating bone defect repair post-tumor resection. Dozzo et al. [193] produced nHA/PLGA hybrid scaffolds and evaluated cell behavior in single and co-culture conditions. They found that culture conditions, environment, and scaffold composition significantly affected cell behavior and model development. Yang et al. [194] developed a drug delivery system using Rhein-PEG-nHA conjugates to deliver DOX and phosphorus-32 (32P) simultaneously. Rhein-PEG-nHA demonstrated significant drug release and bone affinity in vitro and effectively inhibited BC BM growth in vivo. The study emphasized the potential of Rhein-PEG-nHA in combined radio-chemotherapy for BM, highlighting the importance of bone-targeted treatments in reducing skeletal morbidity and improving patient quality of life. Xiong et al. [195] found increased expression of osteogenic markers in human mesenchymal stem cells (hMSCs) on PCL-nHA scaffolds. Additionally, CXCL12 was identified as a key mediator promoting cancer cell migration. The study suggested that tissue-engineered scaffolds could provide a platform for studying BC metastasis by simulating the bone environment, aiding in understanding the mechanisms of BC BM, and exploring new therapeutic strategies. Chen et al. [196] designed a Bortezomib (BTZ)/nHA@SA scaffold to promote tumor inhibition and bone regeneration. The BTZ/nHA@SA scaffold enabled sustained Ca2+ release to regulate the dual functions of BTZ for tumor inhibition and bone regeneration. In vitro studies showed the scaffold promoted cell attachment and proliferation while inducing BC cell death. In vivo studies indicated the scaffold’s effectiveness in promoting tumor ablation and enhancing bone repair. Liu et al. [197] found that DOX could bind with both nHA and micro-Hydroxyapatite (mHA) sized HA particles, leading to their accumulation in the lysosomes of OS cells. The acidic microenvironment of the lysosomes triggered DOX release, resulting in reduced cell migration and apoptosis. In vivo experiments in mice showed that DOX delivered locally via HA particles had a stronger tumor eradication effect. The study suggested that combining nHA with mHA could enhance the safety of the delivery system while maintaining therapeutic efficacy. Xu et al. [198] discovered that zoledronic acid-loaded HA-PEG-nHA-ZOLs enhanced the expression of apoptosis-related proteins and induced apoptosis in tumor cells while blocking the S phase of the cell cycle in OS cells. In a mouse model of OS, local injection of these NPs promoted necrosis, apoptosis, and granulocyte infiltration in tumor vasculature. The study indicated that the ZOL NPs could be used for the local treatment of OS and preventing tumor recurrence. Kang et al. [199] selected human serum albumin as the core material for a drug delivery system. The HSA-AD/DOX NPs exhibited enhanced accumulation in bone tumors in both in vitro and in vivo models. The results showed that HSA-AD/DOX not only improved therapeutic efficacy but also altered the tumor protein expression profile. Yang et al. [200] introduced a bioinspired tumor-targeting and enzyme-activated cell-material interface system based on DDDEEK-pY-phenylboronic acid (SAP-pY-PBA) conjugates. This system selectively regulated cancer cell surface anchoring and aggregation triggered by ALP, forming a supramolecular hydrogel (Fig. 8A and C). As shown in Fig. 8D and E, SAP-pY-PBA + CaCl2 might alter the malignant progression of UMR106 cells by suppressing energy production and oxidative stress through the AKT signaling pathway. This hydrogel effectively killed OS cells by enriching calcium ions and forming a dense HAP layer. The study demonstrated the combination of bioinspired enzyme-responsive biointerfaces with biomineralization as a new antitumor approach. Targeted treatment using HAP NPs in OS and BM is shown in Table 2.
Fig. 8.
A bioinspired tumor-targeting and enzyme-activated cell-material interface system based on SAP-pY-PBA conjugates. A) Schematic of SAP-pY-PBA can cut off phosphate group by ALP and self-assemble to the hydrogel. B) Representative TEM images of SAP-pY-PBA and SAP-pY-PBA + ALP hydrogel. Scale bar: 200 nm. C) SAP-pY-PBA selectively induces calcification on the UMR106 cell surface. SEM image of MC3T3-1E and UMR106 in the blank control, SAP-pY-PBA, and SAP-pY-PBA + CaCl2 group. Scale bar: 20 μm. D) Live/dead staining of UMR106 cell in control, SAP-pY-PBA, and SAP-pY-PBA + CaCl2 groups, respectively. Scale bar: 500 μm. E) Quantification of Ki67, p27, Cyclin D1 by RT-PCR incontrol, SAP-pY-PBA, and SAP-pY-PBA + CaCl2 groups, respectively. Data are expressed as the mean ± SD, n = 5 [200]. Copyright 2023, Wiley-VCH GmbH. Abbreviations: pY, phosphoryl tyrosine; SAP-pY-PBA, DDDEEK-pY-phenylboronic acid; ALP, alkaline phosphatase; DOX, doxorubicin; TEM, transmission electron microscopy; SEM, Scanning electron microscopy
Table 2.
Targeted treatment using hydroxyapatite nanoparticles in malignant bone tumors
| Type of materials | Targeting ligand | Anti-cancer agent | Effects | Mechanism and pathway | Refs. |
|---|---|---|---|---|---|
| HAP-Fn | / | / | Inhibit the tumor growth and bone metastasis in breast cancer | / | [189] |
| nano-HAP | / | / | Reduce cell viability and inhibit migration and invasion of OS cells, suppress tumor growth in vivo | FAK/PI3K/Akt | [190] |
| CM-PDA/SF/nHA | / | / | Promote the proliferation and differentiation of bone marrow mesenchymal stem cells in vitro and new bone production in vivo | / | [191] |
| n-HA/Mxene/g-C3N4 | / | / | Inhibit the proliferation of bone tumor cells and rapidly eradicate bone tumor and enhance osteogenic activity | / | [192] |
| nHA/PLGA | / | / | Promote prostate cancer growth. | / | [193] |
| DOX/32P@Rhein-PEG-nHA | / | DOX/32P | Inhibit the growth of bone metastases of breast cancer | / | [194] |
| PCL-nHA | / | / | Promote the progression of bone metastases of breast cancer | CXCL12/ALP/Runx2/OCN | [195] |
| BTZ/nHA@SA | / | / | Inhibit tumor recurrence and promot bone tissue regeneration | / | [196] |
| HAP-DOX | / | DOX | Repress the progression and recurrence of OS | / | [197] |
| HA-PEG-nHAZOLs | / | ZOL | Stimulate tumor necrosis, apoptosis, and granulocyte infiltration in the blood vessels | / | [198] |
| HSA-AD/DOX | / | DOX | Improvedtherapeutic efficacy in bone tumor-xenografted mice | / | [199] |
| SAP-pY-PBA | / | DOX | Inhibit the progression of OS | [200] |
Exosome-based nanoparticles
Stem cell-derived exosomes are crucial extracellular vesicles containing various bioactive molecules that play key roles in intercellular communication and signal transduction. Combining stem cell exosomes with NPs can further enhance their stability, targeted delivery, and therapeutic efficacy [201]. This combination offers several unique advantages. Firstly, NPs provide a stable encapsulation and protection environment, preserving the integrity and functionality of the exosomes [202]. Secondly, NPs can be surface-modified to control the release of exosomes, enabling targeted delivery and release of therapeutic agents [203]. Additionally, the large specific surface area of NPs provides more loading space, further enhancing the drug delivery capacity of exosomes [204]. The application of stem cell exosomes combined with NPs has vast potential. In tissue engineering and regenerative medicine, these combined exosomes can be used to stimulate cell proliferation and differentiation, promoting tissue repair and regeneration [205]. In drug delivery, this combination can achieve targeted delivery and controlled release of drugs, improving therapeutic efficacy and reducing adverse effects [206]. Furthermore, the combination of stem cell exosomes with NPs holds promise in cancer treatment, treatment of neurodegenerative diseases, and immunotherapy [207].
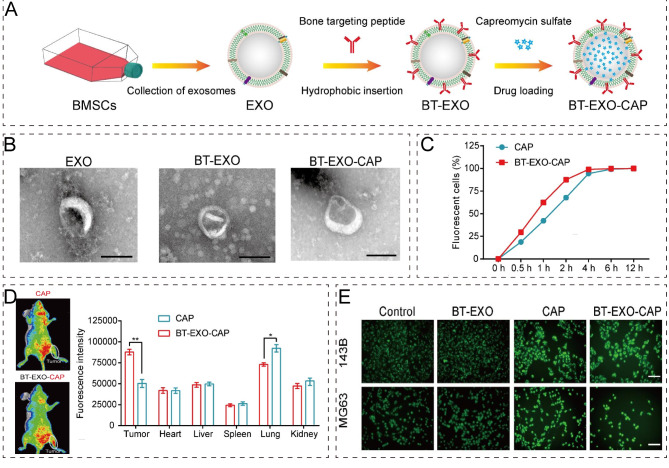
Wei et al. [208] compared NPs composed of DOX-loaded mesenchymal stem cell-derived exosomes with free DOX. Exosomes were isolated from BMSCs and loaded with DOX to create exosome-loaded DOX (Exo-Dox). Exo-Dox demonstrated enhanced cellular uptake efficiency and antitumor activity in OS cells. The results suggested that Exo-Dox could be a promising chemotherapeutic drug for OS treatment, especially considering the tumor-homing characteristics of BM-MSCs. Wang et al. [209] used exosome mimetics (EMs) derived from BMSCs as delivery carriers for DOX to treat OS. Erythromycin-Doxorubicin (EM-Dox) exhibited stronger tumor inhibition activity and fewer side effects compared to free DOX. Chen et al. [210] constructed bone-targeting exosome NPs (BT-EXO) by modifying BMSC exosomes with a bone-targeting peptide and encapsulating the iron chelator capreomycin (CAP) (Fig. 9A and B). In vitro, BT-EXO loading accelerated the entry of CAP-CY5.5 into OS cells (Fig. 9C). In an OS mouse model, the BT-EXO NPs demonstrated significant anticancer activity compared to free CAP (Fig. 9D). The results indicated that the NPs significantly inhibited tumor growth both in vitro and in vivo, providing a promising strategy for combining ferroptosis-based chemotherapy with OS-targeted treatment. BT-EXO-CAP induced an increase in intracellular Fe2+ concentration and ROS accumulation, indicating that BT-EXO-CAP promotes ferroptosis in OS (Fig. 9E).
Fig. 9.
A BT-EXO by modifying BMSC exosomes with a bone-targeting peptide and encapsulating the iron chelator CAP. A) Preparation flow chart of bone targeting NPs. B) Representative TEM images of EXOs, BT-EXOs, and BT-EXO-CAP. C) BT-EXOs loading accelerated CAP entry into OS cells. D) Representative fluorescence imaging of both tumor-bearing mice administrated various treatments and semi-quantitative analysis of mice organs. n = 3. * p < 0.05, ** p < 0.01 E) BT-EXO-CAP induced ferroptosis in human OS cells. DCFH-DA fluorescence of OS cells captured by fluorescence microscope. Reproduced (Adapted) under terms of the Creative Commons Attribution 4.0 International License [210]. Copyright 2023, BioMed Central Ltd unless otherwise stated. Part of Springer Nature. Abbreviations: Nanoparticles, NPs; BMSCs, Bone marrow mesenchymal stem cells; CAP, chelator capreomycin; EXOs, Exosomes; BT-EXO, Bone-targeting-exosomes; NP, Nanoparticle; OS, Osteosarcoma; PBS, phosphate buffer saline
Nanoparticles for cell targeting
Several NPs can inhibit tumor progression by directly regulating the survival ability of tumor cells. Zhang et al. developed a functionalized titanium-based implant by loading curcumin onto cyclodextrin-based polymer-modified TiO2 NPs. The curcumin-modified surface significantly promoted OS cell apoptosis in vitro and inhibited tumor growth in vivo. Using cyclodextrin as a drug reservoir allowed effective drug loading and sustained release of the anticancer agent. Various characterizations of the surface modifications assessed changes in morphology and chemical composition [211]. Yang et al. [212] prepared and characterized RaCl2 micelles, demonstrating dose-response behavior and enhanced efficacy in reducing OS cell viability. The study validated the effectiveness of technology in enhancing the therapeutic effects of radium-223 in cancer treatment. Cai et al. [213] developed paclitaxel-loaded PLGA NPs coated with a 143B-RAW hybrid membrane and evaluated their anticancer efficacy in vitro and in vivo. The hybrid membrane coating on the NPs resulted in high cellular uptake, improved anticancer efficacy against OS cells, and inhibited tumor growth in mice with minimal damage to normal tissues. The results suggested the potential of these hybrid NPs for targeted drug delivery and OS treatment. Yin et al. [214] prepared pDA/MTX@ZIF-8 NPs, demonstrating effective induction of apoptosis in MG63 cells, enhanced antitumor activity, and reduced chemotherapy drug dosage. These NPs exhibited excellent photothermal conversion performance and synergistic chemo-photothermal therapeutic effects, indicating their potential for OS treatment. The study emphasized the importance of developing novel drug delivery systems to improve drug targeting, reduce adverse effects, and enhance therapeutic outcomes in cancer treatment. Ju et al. [215] synthesized MnO2@PA NPs and demonstrated their effective targeting and treatment of OS. Tumor cells effectively internalized MnO2@PA NPs, which released Mn2+ under acidic conditions. In vivo experiments confirmed the accumulation of MnO2@PA NPs in tumor tissues, enhanced MRI imaging, and inhibited tumor growth. The study suggested that developing bone-targeted theranostics for MRI and OS treatment could offer a potential alternative to traditional chemotherapy while reducing side effects. Vanderburgh et al. [216] found that bone-targeted NPs (BTNP) with 10 mol% ligand improved therapeutic outcomes in a mouse model of BC BM. GANT58-BTNPs showed the potential to inhibit tumor-induced bone destruction and improve bone volume fraction in bone tumor-bearing mice. The study highlighted the importance of targeted NPs in addressing tumor-induced bone destruction and provided new insights into optimizing ligand density for effective tumor targeting and bone protection. Li et al. [217] discovered that DOX loaded onto GENP achieved high loading efficiency and sustained drug release. GENP alone could inhibit TNBC cell proliferation through the downstream PI3K/AKT signaling pathway of the Epidermal Growth Factor Receptor (EGFR), showing therapeutic effects in both TNBC and BM models.
Nanoparticles for releasing
pH-responsive nanoparticles
Due to increased glycolysis and the activity of plasma membrane proton pumps, the pH of the TME is slightly lower than that of healthy cells. In most tumors, the extracellular pH ranges from 6.5 to 7.2, while the pH inside lysosomes is even lower, ranging from 5.0 to 5.5 [218]. Therefore, the acidic nature of TME can be exploited to use pH-sensitive nanoparticle drug delivery systems, which can specifically release drugs at low pH levels.
pH-responsive NPs are a unique type of NPs that exhibit high sensitivity to changes in environmental pH [219, 220]. The responsiveness of these NPs is derived from their special structure and composition, with commonly used materials including polyamides, polyacrylic acid, and silicates [221]. pH-responsive NPs typically exist in two forms: a resting state and an activated state [222]. In neutral or near-neutral environments, NPs usually remain in a resting state, characterized by smaller volumes and lower surface charges. However, when the environmental pH changes, the NPs become activated and exhibit different physical and chemical properties. In acidic or alkaline environments, pH-responsive NPs display their unique characteristics. In acidic environments, these NPs tend to adsorb more protons, resulting in a positive charge that causes repulsion between the particles and leads to their aggregation. Conversely, in alkaline environments, NPs release protons and exhibit a negative charge, causing attraction between the particles and leading to their dispersion. The distinctive properties of pH-responsive NPs allow them to play crucial roles in various fields such as biomedicine, environmental monitoring, and the preparation of smart materials. For instance, in drug delivery, the pH responsiveness of NPs can be harnessed to achieve targeted drug release, enhancing therapeutic efficacy and reducing side effects [223]. In environmental monitoring, pH-responsive NPs can be used to detect pH changes in water bodies, providing accurate environmental monitoring data [224]. Additionally, by adjusting the pH responsiveness of NPs, smart materials such as intelligent coatings and sensors can be developed [225].
In a study conducted by Yang et al. [226], ZSM-5 zeolite was utilized as a carrier for the anticancer drug DOX to fabricate ZSM-5/CS/DOX core-shell nanodisks with chitosan (CS) as the shell material. Si elements released from ZSM-5 zeolite were observed to promote osteoblast differentiation by inhibiting NF-κB activation, as reported by Zhou et al. [227]. The ZSM-5/CS/DOX core-shell nanodisks possessed a diameter of 100 nm, a pore size of 3.75 nm, and an impressive drug loading rate of 97.7%. Due to the positively charged surface of CS, the ZSM-5/CS/DOX core-shell nanodisks exhibited pH-responsive behavior. Notably, at pH 6, the ZSM-5/CS/DOX core-shell nanodisks released 58.7% of DOX, indicating their pH-dependent drug release profile. Zhu et al. [228] introduced a strategy combining bone targeting with pH-responsive drug release for anti-metastatic treatment. Compared to free drugs or control micelles, the constructed micelle ALN-NP exhibited reduced systemic toxicity and improved therapeutic efficacy. The study demonstrated the potential of prodrug micelles in targeting drug delivery to bone metastatic tumor tissues, showing enhanced therapeutic effects and reduced side effects. Qiao et al. [229] developed a pH-responsive bone-targeting drug delivery system using lead-bismuth-loaded upconversion NPs PUCZP (Fig. 10A and D). This system enabled the combination treatment of zoledronic acid and osthole, reducing tumorigenesis and osteoclastogenesis. The apoptosis of MDA-MB-231 and 4T1 cells was greatly intensified when BC cells were cocultured with PUCZP compared to PUCP and UCZP, demonstrating the combined effects of drug-induced apoptosis against BC cells (Fig. 10E). 4T1 cells were treated with different NPs to examine the osteoclastogenesis stimulated by 4T1 cells (Fig. 10F and G). Furthermore, this drug delivery system showed potential for early diagnosis of BM in bone cell-targeted treatment (Fig. 4H). The IVIS system monitored MDA-MB-231SAfrp cell survival over time. In the control group, fluorescence appeared in both knee joints by week 3 and increased at week 4. PUCZP treatment reduced fluorescence by 76.1% at week 3 and 70.1% at week 4, compared to 3.6% and 11.4% with UCZP, and 21.9% and 23.8% with PL and ZA. Fluorescence in the head and neck regions of the Ctrl and UCZP groups suggested possible lymphatic metastasis beyond PUCZP’s targeting range (Fig. 4I). PUCZP, by specifically targeting early bone lesions, was more effective in preventing cancer-induced osteolysis compared to UCZP and dual drug treatments. After 4 weeks, significant bone destruction, including severe damage to the knee joint, was observed in the control group’s tibiae (Fig. 4J) Targeted treatment using pH-responsive NPs in OS and BM was shown in Table 3.
Fig. 10.
A pH-responsive bone-targeting drug delivery system using lead-bismuth-loaded upconversion NPs. A) Schematic illustration of the synthesis of theranostic bone-targeting Gd(III)-doped upconversion NPs PUCZP. B) FTIR spectra of UCMS (black line) and UCZP (red line). C) PH-sensitive PL release from PUCZP under acidic conditions. D) PUCZP attenuates cancer cell malignancy and BC-induced osteoclastogenesis synergistically in vitro. Cell apoptosis of MDA-MB-231 and 4T1 cells after varying NPs treatments assessed with flow cytometry analysis. E) Quantifications of cell apoptosis rates of MDA-MB-231 and 4T1 cells treated with varying NPs. F) BMMs were stimulated with CM from 4T1 cells plus varying NPs. TRAP staining to indicate mature MNCs was performed (scale bar = 250 μm). G) The number (left) and area (right) of TRAP-positive MNCs were calculated. Values are the means ± SD (n ≥ 3, **P < 0.05 compared with Ctrl). H) Detection of early BM in nude mice bearing metastatic BC cells. I) Theranostic effects of PUCZP against early BM. Weekly IVIS monitoring of MDA-MB-231SArfp cells in nude mice. J) Fluorescent intensity of cancer cells within the bone milieu after various treatments. Reproduced (Adapted) with permission [229]. Copyright 2017, American Chemical Society. Abbreviations: Nanoparticles, NPs; ZA, Zoledronic acid; PL, plumbagin; BM, bone metastasis; PAA, poly(acrylic acid); BC, breast cancer; PUCZP, PL-Gd-UCNP@ZA-PAA; BMMs, bone marrow monocytes; CM, condition medium; MNCs, multinucleated osteoclast cells; UCMS, Gd-UCNP@mesoporous silica; UCZP, Gd-UCNP@MS-ZA-PAA; PUCP, PL-loaded UCMSs without ZA linkage
Table 3.
Targeted treatment using pH-responsive nanoparticles in malignant bone tumors
| Type of carrier | Targeting ligand | Anti-cancer agent | Effects | Mechanism and corresponding pathway | Refs. |
|---|---|---|---|---|---|
| ZSM-5/CS/DOX | ZSM-5 | DOX | Induce cancer cell apoptosis | / | [226] |
| ALN-NP | / | Alendronate | Reduce systemic toxicity and improve therapeutic effects of breast cancer bone metastasis | / | [228] |
| PL/ZA | / | Zoledronic acid | Inhibit the tumor formation and osteoclast formation of breast cancer bone metastasis | / | [229] |
ROS-responsive nanoparticles
ROS-responsive NPs are a highly sensitive class of NPs that can rapidly respond to changes in intracellular ROS levels [230]. These NPs are typically composed of various materials, such as metal oxides, organic polymers, and biomolecules. Due to the critical physiological roles of ROS and their association with various diseases, research on ROS-responsive NPs has garnered extensive attention. The key to designing these NPs lies in achieving high selectivity and sensitivity, enabling them to respond quickly and controllably to changes in ROS levels [231]. When ROS levels rise, ROS-responsive NPs often undergo morphological or structural changes or release active substances, thereby regulating ROS levels [232]. The application potential of ROS-responsive NPs is vast. In drug delivery, these NPs can serve as carriers, encapsulating drugs and utilizing the presence of ROS to achieve stimulus-responsive drug release [233].
In cancer treatment, since ROS levels are typically higher in tumor tissues, ROS-responsive NPs can be designed as selective tools for tumor-specific drug release. Additionally, ROS-responsive NPs can be used in fields such as bioimaging, diagnostics, and therapeutic monitoring [234]. For example, in bioimaging, these NPs can function as fluorescent probes, monitoring the oxidative stress status of cells by responding to changes in ROS levels [235]. In diagnostics and therapeutic monitoring, ROS-responsive NPs can be used for early disease detection and treatment efficacy assessment [236]. Wang et al. [237] developed mesoporous polydopamine-coated hydroxyapatite (HAP) NPs loaded with indocyanine green (ICG) for ROS-triggered nitric oxide (NO) enhanced PTT of OS. The AI-MPHA NCs demonstrated high photothermal efficiency, effective ROS production, and efficient NO generation under near-infrared laser irradiation, achieving a synergistic effect of PTT and NO gas treatment. Ying et al. [238] discovered that FeGA-DOX NPs induce PTT under laser irradiation, releasing DOX to produce ROS for chemodynamic treatment. The results showed potential in inhibiting mouse OS tumor growth by combining photothermal and chemodynamic effects without severe side effects. Gemini-Piperni et al. [239] developed and evaluated Ra-nano-HAP as a potential radiopharmaceutical, highlighting the importance of expanding the new TAT drug library. Additionally, Ra-nano-HAP exhibited a dose-response relationship against OS cells and demonstrated safety for human fibroblasts. Zeng et al. [240] reported that NPs significantly inhibited tumor progression in mice bearing drug-resistant OS cells (Fig. 11A and B). In Fig. 11C, the TPPPPG@ICG + laser group induced significant cell death and exhibited strong red fluorescence, further demonstrating the synergistic therapeutic effects of mitochondria-targeted PDT and PTT. Furthermore, real-time fluorescence imaging in the near-infrared region was used to monitor the distribution of the NPs and guide treatment (Fig. 11D and F). The study demonstrated the potential of targeting mitochondrial oxidative graphene NPs for fluorescence imaging-guided synergistic phototherapy in drug-resistant OS, providing a novel approach to improve treatment outcomes for challenging cases (Fig. 11). Targeted treatment using ROS-responsive NPs in OS and BM was shown in Table 4.
Fig. 11.
A new nano-drug delivery system based on near-infrared imaging and multifunctional graphene, A) Schematic illustration showing a TPP-PPG@ICG nanocomposite targeting a mitochondrion for synergistic phototherapy with a single laser. B) Characterization and physicochemical properties of TPP-PPG@ICG. AFM-based morphological characterizations of a–d TPP-PPG and TPP-PPG@ICG. C) Cell Counting Kit-8 (CCK-8)-based viability assay performed with doxorubicin-resistant MG63 cells (MG63/Dox cells) treated with ICG, TPP-PPG, PPG@ICG, or PP-PPG@ICG, without laser irradiation. D) NIR FL imaging of MG63/Dox tumor xenografts exposed to 808-nm laser irradiation (0.6 W/cm2, 5 min), 0.5, 1, 4, 8, 12 and 24 h after tail vein injection of TPP-PPG@ICG, and the dissected organs from the MG63/Dox tumor-bearing mice: the tumor, the lungs, the liver, the spleen, the kidneys, the heart and the muscle). IR thermal imaging of MG63/Dox tumor xenografts exposed to 808-nm laser irradiation (0.6 W/cm2, 5 min), 24 h after tail vein injection with PBS and TPP-PPG@ICG. E) Tumor volumes of MG63/Dox tumor xenografts, F) body weights of different groups of mice. Reproduced (Adapted) with permission [240]. Copyright 2021, American Chemical Society. Abbreviations: TPP, (4‑Carboxybutyl) triphenyl phosphonium bromide; ROS, Reactive oxygen species; ICG, indocyanine green; PPG, polyethylenimine-modified PEGylated nanographene oxide; AFM, Atomic force microscopy; PBS, Phosphate Buffered Saline
Table 4.
Targeted treatment using ROS-responsive nanoparticles in malignant bone tumors
| Type of carrier | Targeting ligand | Anti-cancer agent | Effects | Mechanism and corresponding pathway | Refs. |
|---|---|---|---|---|---|
| AI-MPHA NCs | MPDA | / | Establish a novel nano-platform for NO-enhanced photothermal therapy of OS | / | [190] |
| FeGA-DOX/AG | / | DOX | Induce tumor cell apoptosis and provide a novel therapeutic strategy for OS | / | [191] |
| C60-CaMKIIα-nano | CaMKIIα | / | Repress OS pregression | Elicit ROS-dependent cytotoxicity and persistent activation of CaMKIIα | [193] |
| TPP-PPG@ICG | / | Doxorubicin | Induce the intrinsic apoptosis assisted in surmounting drug resistance and cause tumor cell death | / | [194] |
Antiosteosarcoma nanotherapy mediated by tissue engineering scaffolds
After surgical resection of OS, a definitive bone defect is often left, which may require medical intervention using grafts or synthetic scaffolds to fill the void. Some studies focus on therapeutic strategies that utilize bone tissue engineering scaffolds to both promote bone regeneration and eliminate residual tumor cells without causing systemic toxicity. Nanomaterials have opened a new avenue in bone grafting, as they aid in bone reconstruction and mimic the unique structural characteristics of natural bone.
3D printing technology, as an innovative manufacturing method, is gaining widespread attention in the fields of nanoscience and materials. In recent years, researchers have begun to explore the application of NPs in the preparation of 3D-printed nanostructures using 3D printing technology [241]. By combining NPs with printing materials, researchers have successfully achieved directed assembly and precise control of the spatial positioning of NPs. Methods for 3D printing NPs include directly mixing NPs with printing materials or printing after the pre-assembly and functionalization of NPs [242]. These methods not only enable high-resolution structure fabrication at the microscale but also introduce the unique properties of NPs into the printed structures, further expanding the functionality and applications of the materials [243]. The potential applications of 3D-printed NPs are extensive. In the biomedical field, 3D-printed nanoparticle structures can be used for biosensing, tissue repair, and drug delivery [244].
Hao et al. [245] reported a chip-based bone model for the self-growth of 3D mineralized collagen bone tissue. They discovered that mineralized bone tissue could grow autonomously within the chip-based bone model, simulating a real bone environment (Fig. 12A and L). The chip-based bone model offers advantages such as high experimental throughput and focused cell-bone matrix interaction areas, providing an important experimental platform for developing new therapeutic strategies and drugs (Fig. 12M and N). Zhu et al. [246] integrated 3D printing technology with a unique nano-ink to create a biomimetic bone-specific environment. BC cells cultured in the 3D matrix exhibited spheroid morphology and migration characteristics, while co-culture with BMSCs enhanced the formation of spheroid clusters. Compared to 2D culture, the 3D matrix imparted greater drug resistance to BC cells. Zhu et al. [247] further demonstrated that 3D-printed bone matrix structures with controlled architecture allowed BC cells to inhibit osteoblast proliferation, while osteoblasts stimulated BC cell growth in the optimized bone matrix. Co-culturing BC cells and osteoblasts in the 3D bone matrix formed multicellular spheroids. The results validated the use of a 3D-printed bone matrix as an in vitro metastasis model to study BC BM, aiding in understanding the behavior and interactions of BC in the bone microenvironment.
Fig. 12.
A chip-based bone model for the self-growth of 3D mineralized collagen bone tissue. A) Schematics of the chip design. B) SEM analysis of 30 d osteoblastic tissues recovered from a 2 mm NC BC. Bundles of collagen fibrils of 30 nm diameter formed a 3D mesh structure. Arrows indicate strings of mineral deposition (left). Large nodules of mineral deposition were indicated by arrow (right). C) Representative TEM images of horizontal and cross-sectional views of recovered osteoblastic tissues from 2 mm NC-BC. Cells were healthy based on morphology and nuclear shape. Arrows indicate mineralized collagen fiber bundles. Inset highlights the distinctive banded pattern of collagen fibrils. D) Element analysis revealed calcium and phosphate as the major components of the mineral nodule. E) Cancer cells are embedded in the collagen matrix and start proliferating in the first week of co-culture. F) Arrows indicate the hollow space occupied by the cancer cells in panel E. G) Cancer cells eroding the surrounding matrix. H) Arrows indicate invadopodia (close-up in the inset) protruding through the matrix. I) The embedded cancer cells grew into colonies in the first week. J) The proliferating cancer cells started reorganizing the surrounding matrix. Doubled-headed arrows indicate the alignment of the matrix. K) Cancer cells aligned with the axis of the re-organized matrix. L) A single file of 3 BC cells migrated through the re-organized matrix. Scale bars are 100 μm in panels (E)–(K). M) Fluorescence images show the formation of micrometastases by MDA-MB-231 and MDA-MB-231-BRMS1. N) Histogram represents the percentage of cancer cell population contributing to single cell or micrometastases. Significant differences in the percentage of cancer cell population contributing formation of micrometastases and single cells were observed between two cancer types (n = 3, p < 0.01). Reproduced (Adapted) with permission [245]. Copyright 2018, Wiley-VCH Verlag GmbH & Co. Abbreviations: BC, breast cancer; NC, nitrocellulose; BC, bone-on-a-chip; SEM, Scanning Electron Microscope
Nanoparticles with clinical potential
Lipid nanoparticles
Liposomes are NPs composed of one or more lipid bilayers that form spherical hydrophilic vesicles, enabling drug encapsulation. Hydrophobic drugs can be incorporated within the lipid bilayer, while hydrophilic drugs can be encapsulated in the aqueous core. In 1995, the liposomal formulation of Doxil was approved by the FDA. Liposomal Doxil, also known as liposomal Adriamycin, is fully named pegylated liposomal doxorubicin (PLD) (Fig. 13A and B). It uses an advanced phospholipid bilayer to encapsulate the active ingredient Doxil, with an outer layer coated with polyethylene glycol (PEG), forming a novel anthracycline drug. According to clinical research results from both domestic and international studies, as well as recommendations from authoritative guidelines such as NCCN and CSCO, PLD can be used for the treatment of soft tissue and bone sarcomas.
Fig. 13.
Structure and characteristics of pegylated liposomal doxorubicin (PLD) with clinical application. A) Structure of Doxorubicin (ADM). B) Structure of PLD. C) Advantages of PLD compared to ADM. D) PLD improves the side effects of traditional anthracyclines and the advantages of tumor tissue enrichment
Chemotherapy remains a crucial systemic treatment for bone and soft tissue sarcomas. ADM (another name for Doxil) is the cornerstone of chemotherapy for bone and soft tissue sarcomas, with first-line regimens recommending ADM at a dose of 50–70 mg/m² every three weeks as one cycle. As a traditional anthracycline-based anticancer drug, ADM exhibits potent and broad-spectrum antitumor activity. However, while it effectively kills tumor cells, it also causes significant adverse effects, including myelosuppression, cardiotoxicity, and gastrointestinal toxicity. Notably, its cardiotoxicity is dose-dependent, which limits its clinical use. PLD is a novel formulation of ADM designed to enhance drug delivery by evading phagocytosis by the reticuloendothelial system, improving tissue penetration, prolonging circulation time, and enabling targeted release at tumor sites. PLD releases its payload gradually within the body, with a half-life of 50–70 h. Due to the leaky vasculature and abundant neovascularization within tumors, especially in large tumor masses, the drug accumulates more effectively at the tumor site (Fig. 13C and D). Additionally, PLD can cross the BBB and blood-testis barrier, providing significant inhibitory effects on tumors located in the central nervous system and other specialized areas. Compared with conventional ADM, PLD offers longer therapeutic duration, reduced cardiotoxicity, and improved tumor-targeting capabilities. The authoritative NCCN guidelines indicate that PLD monotherapy has shown efficacy in the treatment of advanced, metastatic, recurrent, or refractory sarcomas. The EORTC study was a prospective, randomized phase II trial comparing PLD with ADM for the treatment of advanced soft tissue sarcomas (STS). Of the 94 patients enrolled, 50 received PLD (50 mg/m2), and 44 received ADM (75 mg/m2). The results demonstrated that the response rates for advanced or metastatic sarcomas were 9% for ADM and 10% for PLD, indicating that PLD offers comparable efficacy to ADM. Moreover, PLD significantly reduced the incidence of febrile neutropenia (FN), alopecia, and cardiotoxicity compared to ADM. Another single-arm, single-center, open-label clinical study included 25 patients with advanced metastatic STS to evaluate the clinical efficacy and safety of PLD in combination with ifosfamide (IFO). The results showed a median progression-free survival (PFS) of 8.0 months, with mild adverse effects and favorable cardiac safety. The primary cardiac events observed were grade 1 arrhythmias, with no grade 3–4 cardiotoxicity reported, confirming the high cardiac safety of PLD. Given these benefits, PLD demonstrates efficacy equivalent to ADM in treating sarcoma patients, while offering superior cardiac and hematological safety. Therefore, PLD is considered one of the preferred chemotherapy options for sarcoma patients.
In addition, combining liposomes with nanocarriers and drug delivery agents within scaffolds is a promising approach to create a versatile platform for drug delivery and tissue engineering, offering the potential for prolonged therapeutic effects, especially in bone healing [248]. Furthermore, due to their small size and unique composition, nanomaterials used as scaffolds can significantly enhance performance and interactions within the body. Therefore, nanomaterial-based scaffolds hold great promise as an effective method for OS treatment, bone repair, and regeneration in the future.
Polymeric nanoparticles
Currently, several polymeric NPs have been developed and are available as promising platforms for drug delivery. Examples include polymeric micelles, PLA-PEG NPs, and PLGA-based NPs, which are widely researched due to their biocompatibility, controlled release properties. These polymeric carriers offer significant potential for targeted drug delivery and combination therapies, making them valuable platforms for future applications in treating cancers and other diseases.
Polymeric micelles are formed by the self-assembly and spontaneous aggregation of amphiphilic polymers. The micelle structure consists of a core and a shell, where the hydrophobic core acts as a reservoir to prevent drug dissolution, while the hydrophilic shell provides water solubility and spatial stability to the micelle structure. Jin et al. [249] designed a mitochondria-targeting amphiphilic block copolymer (OPDEA-b-PDCA) that self-assembles into stable polymeric micelles (OPDEA-PDCA), which can selectively inhibit mitochondrial PDHK1. These self-assembled polymeric micelles are capable of targeting and modulating mitochondria to induce pyroptosis in tumor cells.
Poly(lactic-co-glycolic acid) (PLGA) has been approved by both the European Medicines Agency and the FDA for use in medical devices and pharmaceuticals due to its controlled drug release properties, biocompatibility, and relatively low toxicity. Yu et al. [250] developed a multifunctional magnetic gel (Fe3O4/GOx/MgCO3@PLGA) as a magnetic bone repair hydrogel (MBR), which has liquid-solid phase transition capabilities. This gel can induce a magnetothermal effect, trigger the release of GOx, and inhibit ATP production, thereby reducing HSP expression, and achieving synergistic therapy for OS treatment.
Poly(ethylene glycol)-poly(D, L-lactide) (PEG-PLA) is a commonly used copolymer for drug delivery, while Prostate-Specific Membrane Antigen (PSMA) targeting technology is primarily used in the treatment of PCa. Afsharzadeh Maryam et al. [251] first introduced a novel PEG-PLA nanoparticle (PEG-PLA NPs) system containing galbanic acid (GBA) and docetaxel, targeted using ACUPA ((S)-2-(3-((S)-5-amino-1-carboxypentyl) ureido) pentanedioic acid), a small molecule inhibitor that targets prostate-specific membrane antigen (PSMA). This PEG-PLA NPs, loaded with GBA and docetaxel, presents a promising delivery platform for PSMA-targeting cancers, addressing challenges such as short circulation half-life and limited therapeutic efficacy. Therefore, PSMA-targeted PEG-PLA NPs can be used for the treatment of secondary BM caused by PCa.
Among polymers, chitosan, a natural polymer, is widely used due to its excellent biocompatibility and biodegradability. It can be designed into various NPs to perform different functions in the treatment of OS. Mei L. Tan et al. [252] discovered that free chitosan promotes osteoblast proliferation and osteogenesis in mesenchymal stem cells, increasing the expression of osteopontin and collagen I while reducing osteoclast formation. In mice treated with chitosan, better fracture healing was observed, with greater trabecular bone formation.
Discussion and prospects
NPs, as a novel tool for treatment and diagnosis, have made significant progress [253]. By optimizing materials to construct stable, efficient, and safe nanocarriers, and by combining these nanocarriers with anticancer drugs and highly accurate cancer diagnostic probes, future research will focus on integrating functions such as targeted drug delivery, in vivo tracking, drug treatment, and prognosis monitoring into multifunctional nanosystems. This will provide strong support for effectively improving drug delivery efficiency and reducing drug toxicity [254]. However, it is worth noting that the application of NPs in cancer diagnosis still faces several challenges. Firstly, the fundamental theories of nanomedicine and the preparation processes of nanodrugs are still not well established. There is a lack of in-depth and systematic research on the behavior of nanodrugs in the body, including tissue distribution, pharmacokinetics, pharmacodynamics, and their interactions with the chemical structure and physical properties of the carriers [255]. Therefore, extensive work is still needed in the research of nanotechnology in the diagnostic field. Despite some potential limitations and challenges, the application prospects of NPs in cancer diagnosis remain very optimistic with continuous technological advancements and safety evaluations. This progress is significant for the development of personalized treatment. Current research in nanobiotechnology in clinical diagnostics mainly focuses on nanosensors and imaging technologies, the use of nanorobots for cell-level repair, and the extraction and measurement of biomarkers, aiming at early disease diagnosis and improving treatment efficacy. Future research and development will further promote the application of NPs in cancer diagnosis, providing more effective means for cancer treatment.
With advancements in nanotechnology, it is becoming increasingly possible to develop more intelligent NPs that respond to changes in the TME or external stimuli, allowing for precise drug release. This innovation can significantly enhance targeting specificity and minimize side effects in cancer treatment. Additionally, NPs can be designed for multimodal imaging, incorporating techniques such as CT, MRI, and PET. By creating multimodal imaging probes, nanotechnology enables highly sensitive tumor detection and precise localization, while also integrating diagnostic and therapeutic functions into a single platform. This theranostic approach holds great promise for more effective personalized treatments. Moreover, nanotechnology offers the potential to expand personalized immunotherapy by developing cancer vaccines and immunotherapy platforms that enhance specific immune responses. By targeting tumor-associated antigens and incorporating immune checkpoint inhibitors or other immunomodulators, NPs can improve the efficacy and tolerability of immunotherapy. Despite significant progress in laboratory research, the clinical translation of nanomedicine still requires further clinical trials and safety evaluations. Future research must prioritize optimizing clinical translation pathways.
Conclusion
Malignant bone tumors are highly aggressive and lethal, and their complex TME not only promotes tumor growth and metastasis, but also increases the difficulty of treatment. Understanding the mechanisms of the bone microenvironment is essential for the treatment of malignant bone tumors. Recent advances in NPs have shown great promise in the diagnosis and treatment of malignant bone tumors. These NPs offer enhanced targeting capabilities, controlled drug delivery, and improved imaging modalities, leading to more effective therapeutic outcomes and early detection of cancer. By overcoming these challenges and focusing on the development of novel NPs, we can significantly improve the diagnosis and treatment of malignant bone tumors, ultimately enhancing patient outcomes and quality of life.
Author contributions
Original draft preparation, allocation, revision, and editing: Yuling Mao, Wei Wang,Yujing Guan and Shenglong Li. All authors have read and agreed to the published version of the manuscript.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yujing Guan and Wei Zhang contributed equally to this work.
Contributor Information
Yuling Mao, Email: maoyuling@syphu.edu.cn, Email: maoyuling@hotmail.com.
Shenglong Li, Email: slli@cmu.edu.cn, Email: lishenglong@dlut.edu.cn, Email: lishenglong@cancerhosp-ln-cmu.com.
References
- 1.Xiang H, Yang Q, Gao Y, Zhu D, Pan S, Xu T, et al. Cocrystal strategy toward multifunctional 3D-printing scaffolds enables NIR‐activated photonic osteosarcoma hyperthermia and enhanced bone defect regeneration. Adv Funct Mater. 2020;30(25):1909938. [Google Scholar]
- 2.Díaz ECG, Sinha S, Avedian RS, Yang F. Tissue-engineered 3D models for elucidating primary and metastatic bone cancer progression. Acta Biomater. 2019;99:18–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Meltzer PS, Helman LJ. New Horizons in the Treatment of Osteosarcoma. N Engl J Med. 2021;385(22):2066–76. [DOI] [PubMed] [Google Scholar]
- 4.Gill J, Gorlick R. Advancing therapy for osteosarcoma. Nat Rev Clin Oncol. 2021;18(10):609–24. [DOI] [PubMed] [Google Scholar]
- 5.Chen C, Xie L, Ren T, Huang Y, Xu J, Guo W. Immunotherapy for osteosarcoma: fundamental mechanism, rationale, and recent breakthroughs. Cancer Lett. 2021;500:1–10. [DOI] [PubMed] [Google Scholar]
- 6.Shoaib Z, Fan TM, Irudayaraj JMK. Osteosarcoma mechanobiology and therapeutic targets. Br J Pharmacol. 2022;179(2):201–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yang C, Tian Y, Zhao F, Chen Z, Su P, Li Y, et al. Bone microenvironment and osteosarcoma metastasis. Int J Mol Sci. 2020;21(19):6985. [DOI] [PMC free article] [PubMed]
- 8.Li S, Zhang H, Liu J, Shang G. Targeted therapy for osteosarcoma: a review. J Cancer Res Clin Oncol. 2023;149(9):6785–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kansara M, Teng MW, Smyth MJ, Thomas DM. Translational biology of osteosarcoma. Nat Rev Cancer. 2014;14(11):722–35. [DOI] [PubMed] [Google Scholar]
- 10.Li Z, Li X, Xu D, Chen X, Li S, Zhang L, et al. An update on the roles of circular RNAs in osteosarcoma. Cell Prolif. 2021;54(1):e12936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Longhi A, Ferrari S, Bacci G, Specchia S. Long-term follow-up of patients with doxorubicin-induced cardiac toxicity after chemotherapy for osteosarcoma. Anticancer Drugs. 2007;18(6):737–44. [DOI] [PubMed] [Google Scholar]
- 12.Patino-Garcia A, Zalacain M, Marrodan L, San-Julian M, Sierrasesumaga L. Methotrexate in pediatric osteosarcoma: response and toxicity in relation to genetic polymorphisms and dihydrofolate reductase and reduced folate carrier 1 expression. J Pediatr. 2009;154(5):688–93. [DOI] [PubMed] [Google Scholar]
- 13.Coleman RE, Croucher PI, Padhani AR, Clezardin P, Chow E, Fallon M, et al. Bone metastases. Nat Rev Dis Primers. 2020;6(1):83. [DOI] [PubMed] [Google Scholar]
- 14.Clezardin P, Coleman R, Puppo M, Ottewell P, Bonnelye E, Paycha F, et al. Bone metastasis: mechanisms, therapies, and biomarkers. Physiol Rev. 2021;101(3):797–855. [DOI] [PubMed] [Google Scholar]
- 15.Himelstein AL, Foster JC, Khatcheressian JL, Roberts JD, Seisler DK, Novotny PJ, et al. Effect of Longer-Interval vs Standard Dosing of Zoledronic Acid on skeletal events in patients with bone metastases: a Randomized Clinical Trial. JAMA. 2017;317(1):48–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gartrell BA, Saad F. Managing bone metastases and reducing skeletal related events in prostate cancer. Nat Rev Clin Oncol. 2014;11(6):335–45. [DOI] [PubMed] [Google Scholar]
- 17.Cadieux B, Coleman R, Jafarinasabian P, Lipton A, Orlowski RZ, Saad F, et al. Experience with denosumab (XGEVA(R)) for prevention of skeletal-related events in the 10 years after approval. J Bone Oncol. 2022;33:100416. [DOI] [PMC free article] [PubMed]
- 18.D’Oronzo S, Coleman R, Brown J, Silvestris F. Metastatic bone disease: Pathogenesis and therapeutic options: up-date on bone metastasis management. J Bone Oncol. 2019;15:100205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang W, Bado I, Wang H, Lo HC, Zhang XH. Bone metastasis: find your niche and fit in. Trends Cancer. 2019;5(2):95–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.He Y, Luo W, Liu Y, Wang Y, Ma C, Wu Q, et al. IL-20RB mediates tumoral response to osteoclastic niches and promotes bone metastasis of lung cancer. J Clin Invest. 2022;132(20):e157917. [DOI] [PMC free article] [PubMed]
- 21.Pei Z, Lei H, Cheng L. Bioactive inorganic nanomaterials for cancer theranostics. Chem Soc Rev. 2023;52(6):2031–81. [DOI] [PubMed] [Google Scholar]
- 22.Cheng L, Wang X, Gong F, Liu T. Liu: 2D nanomaterials for Cancer Theranostic Applications. Adv Mater. 2020;32(13):e1902333. [DOI] [PubMed] [Google Scholar]
- 23.Song W, Musetti SN, Huang L. Nanomaterials for cancer immunotherapy. Biomaterials. 2017;148:16–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tang L, Zhang A, Zhang Z, Zhao Q, Li J, Mei Y, et al. Multifunctional inorganic nanomaterials for cancer photoimmunotherapy. Cancer Commun (Lond). 2022;42(2):141–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li J, Lu W, Yang Y, Xiang R, Ling Y, Yu C, et al. Hybrid nanomaterials for Cancer Immunotherapy. Adv Sci (Weinh). 2023;10(6):e2204932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li X, Lovell JF, Yoon J, Chen X. Clinical development and potential of photothermal and photodynamic therapies for cancer. Nat Rev Clin Oncol. 2020;17(11):657–74. [DOI] [PubMed] [Google Scholar]
- 27.Ren Y, Yan Y, Qi H. Photothermal conversion and transfer in photothermal therapy: from macroscale to nanoscale. Adv Colloid Interface Sci. 2022;308:102753. [DOI] [PubMed]
- 28.Xin J, Deng C, Aras O, Zhou M, Wu C, An F. Chemodynamic nanomaterials for cancer theranostics. J Nanobiotechnol. 2021;19(1):192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang WD, Guo YY, Yang ZL, Su GL. Sun: Sniping Cancer Stem cells with nanomaterials. ACS Nano. 2023;17(23):23262–98. [DOI] [PubMed] [Google Scholar]
- 30.Ding S, Chen L, Liao J, Huo Q, Wang Q, Tian G, et al. Harnessing hafnium-based nanomaterials for Cancer diagnosis and therapy. Small. 2023;19(32):e2300341. [DOI] [PubMed] [Google Scholar]
- 31.Pellico J, Gawne PJ. d. R. R: Radiolabelling of nanomaterials for medical imaging and therapy. Chem Soc Rev. 2021;50(5):3355–423. [DOI] [PubMed] [Google Scholar]
- 32.M PA, Pardhiya S, Rajamani P. Carbon dots: an excellent fluorescent probe for Contaminant Sensing and Remediation. Small. 2022;18(15):e2105579. [DOI] [PubMed] [Google Scholar]
- 33.Wang Y, Wang W, Wang X, Wu H, Zhao W, Zhao C. Immune-stealth carboxymethyl chitosan-based nanomaterials for magnetic resonance imaging-guided photothermal therapy. Carbohydr Polym. 2022;288:119382. [DOI] [PubMed]
- 34.Sun X, Cai W, Chen X. Positron emission tomography imaging using radiolabeled inorganic nanomaterials. Acc Chem Res. 2015;48(2):286–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Salhotra A, Shah HN, Levi B, Longaker MT. Mechanisms of bone development and repair. Nat Rev Mol Cell Biol. 2020;21(11):696–711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hu W, Chen Y, Dou C, Dong S. Microenvironment in subchondral bone: predominant regulator for the treatment of osteoarthritis. Ann Rheum Dis. 2021;80(4):413–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xiong Y, Mi BB, Lin Z, Hu YQ, Yu L, Zha KK, et al. The role of the immune microenvironment in bone, cartilage, and soft tissue regeneration: from mechanism to therapeutic opportunity. Mil Med Res. 2022;9(1):65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yip RKH, Rimes JS, Capaldo BD, Vaillant F, Mouchemore KA, Pal B, et al. Mammary tumour cells remodel the bone marrow vascular microenvironment to support metastasis. Nat Commun. 2021;12(1):6920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhang X. Interactions between cancer cells and bone microenvironment promote bone metastasis in prostate cancer. Cancer Commun (Lond). 2019;39(1):76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Alfranca A, Martinez-Cruzado L, Tornin J, Abarrategi A, Amaral T, de Alava E, et al. Bone microenvironment signals in osteosarcoma development. Cell Mol Life Sci. 2015;72(16):3097–113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fan Y, Lyu P, Bi R, Cui C, Xu R, Rosen CJ, et al. Creating an atlas of the bone microenvironment during oral inflammatory-related bone disease using single-cell profiling. Elife. 2023;12:e82537. [DOI] [PMC free article] [PubMed]
- 42.Li C, Zhao R, Yang H, Ren L. Construction of bone hypoxic microenvironment based on bone-on-a-chip platforms. Int J Mol Sci. 2023;24(8):6999. [DOI] [PMC free article] [PubMed]
- 43.Hiraga T. Hypoxic microenvironment and metastatic bone disease. Int J Mol Sci. 2018;19(11):3523. [DOI] [PMC free article] [PubMed]
- 44.He J, Chen G, Liu M, Xu Z, Chen H, Yang L, et al. Scaffold strategies for modulating immune microenvironment during bone regeneration. Mater Sci Eng C Mater Biol Appl. 2020;108:110411. [DOI] [PubMed]
- 45.Greenblatt MB, Shim JH, Glimcher LH. Mitogen-activated protein kinase pathways in osteoblasts. Annu Rev Cell Dev Biol. 2013;29:63–79. [DOI] [PubMed] [Google Scholar]
- 46.Engblom C, Pfirschke C, Zilionis R, Da Silva Martins J, Bos SA, Courties G, et al. Osteoblasts remotely supply lung tumors with cancer-promoting SiglecF(high) neutrophils. Science. 2017;358(6367):eaal5081. [DOI] [PMC free article] [PubMed]
- 47.Wang ZX, Luo ZW, Li FX, Cao J, Rao SS, Liu YW, et al. Aged bone matrix-derived extracellular vesicles as a messenger for calcification paradox. Nat Commun. 2022;13(1):1453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Utsunomiya T, Zhang N, Lin T, Kohno Y, Ueno M, Maruyama M, et al. Different effects of intramedullary injection of mesenchymal stem cells during the acute vs. Chronic inflammatory phase on bone healing in the murine continuous polyethylene particle infusion model. Front Cell Dev Biol. 2021;9:631063. [DOI] [PMC free article] [PubMed]
- 49.Lee CH, Decker AM, Cackowski FC, Taichman RS. Bone microenvironment signaling of cancer stem cells as a therapeutic target in metastatic prostate cancer. Cell Biol Toxicol. 2020;36(2):115–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rindone AN, Grayson WL. Illuminating the Regenerative Microenvironment: Emerging Quantitative Imaging Technologies for Craniofacial Bone tissue Engineering. ACS Biomater Sci Eng. 2022;8(11):4610–2. [DOI] [PubMed] [Google Scholar]
- 51.Buenrostro D, Mulcrone PL, Owens P, Sterling JA. The bone microenvironment: a fertile soil for Tumor Growth. Curr Osteoporos Rep. 2016;14(4):151–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Turner CJ, Edwards CM. The role of the microenvironment in prostate Cancer-Associated Bone Disease. Curr Osteoporos Rep. 2016;14(5):170–7. [DOI] [PubMed] [Google Scholar]
- 53.Samal S, Dash P, Dash M. Drug delivery to the bone microenvironment mediated by exosomes: an axiom or Enigma. Int J Nanomed. 2021;16:3509–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Tamura T, Yoshioka Y, Sakamoto S, Ichikawa T, Ochiya T. Extracellular vesicles in bone metastasis: key players in the tumor microenvironment and promising therapeutic targets. Int J Mol Sci. 2020;21(18):6680. [DOI] [PMC free article] [PubMed]
- 55.Tian H, Cao J, Li B, C.Nice E, Mao H, Zhang Y, et al. Managing the immune microenvironment of osteosarcoma: the outlook for osteosarcoma treatment. Bone Res. 2023;11(01):21–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Layla S, Myra A, Andrea I, Lily D. Mechanisms of immune checkpoint inhibitor-mediated liver injury. Acta Pharm Sinica B. 2021;11(12):3727–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Nirvana KK, Atefeh G, Isaac HS. Role of tumor microenvironment in the regulation of PD-L1: a novel role in resistance to cancer immunotherapy. J Cell Physiol. 2020;235(10):6496–506. [DOI] [PubMed] [Google Scholar]
- 58.Kobayashi Y, Lim SO, Yamaguchi H. Oncogenic signaling pathways associated with immune evasion and resistance to immune checkpoint inhibitors in cancer. Semin Cancer Biol. 2020;65:51–64. [DOI] [PubMed]
- 59.Padmanee S, P AJ. Dissecting the mechanisms of immune checkpoint therapy. Nat Rev Immunol. 2020;20(2):75–6. [DOI] [PubMed] [Google Scholar]
- 60.Hao Z, Ziyu D, Wantao W, Zeyu W, Nan Z, Liyang Z, et al. Regulatory mechanisms of immune checkpoints PD-L1 and CTLA-4 in cancer. J Experimental Clin Cancer Res. 2021;40(1):184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bayat M, Nahand JS. Exosomal miRNAs: the tumor’s trojan horse in selective metastasis. Mol Cancer. 2024;23(1):167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Parida S, Siddharth S, Gatla HR, Wu S, Wang G, Gabrielson K, et al. Gut colonization with an obesity-associated enteropathogenic microbe modulates the premetastatic niches to promote breast cancer lung and liver metastasis. Front Immunol. 2023;14:1194931. [DOI] [PMC free article] [PubMed]
- 63.Mengying H, M KC, Nancy B, David L. Tumor-derived nanoseeds condition the soil for metastatic organotropism. Sem Cancer Biol. 2023;93:70–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Francesca P, Luca R, Orazio F, Giulia B. The metastatic niche formation: focus on extracellular vesicle-mediated dialogue between lung cancer cells and the microenvironment. Front Oncol. 2023;13:1116783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chiwei C, Renlun H, Jianfu Z, Lang G, Songtao X. Formation of pre-metastatic bone niche in prostate cancer and regulation of traditional chinese medicine. Front Pharmacol. 2022;13:897942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.R CA, Emily WS. Cancer tills the Premetastatic Field: mechanistic basis and clinical implications. Clin cancer Research: Official J Am Association Cancer Res. 2016;22(15):3725–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Abdel Sater AH, Bouferraa Y, Amhaz G, Haibe Y, Lakkiss AE, Shamseddine A. Ali: from tumor cells to endothelium and gut microbiome: a complex interaction favoring the metastasis cascade. Front Oncol. 2022;12:804983. [DOI] [PMC free article] [PubMed]
- 68.Ren G, Esposito M, Kang Y. Bone metastasis and the metastatic niche. J Mol Med (Berl). 2015;93(11):1203–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hofbauer LC, Bozec A, Rauner M, Jakob F, Perner S, Pantel K. Novel approaches to target the microenvironment of bone metastasis. Nat Rev Clin Oncol. 2021;18(8):488–505. [DOI] [PubMed] [Google Scholar]
- 70.Muscarella AM, Aguirre S, Hao X, Waldvogel SM, Zhang XH. Exploiting bone niches: progression of disseminated tumor cells to metastasis. J Clin Invest. 2021;131(6):e143764. [DOI] [PMC free article] [PubMed]
- 71.Gdowski AS, Ranjan A, Vishwanatha JK. Current concepts in bone metastasis, contemporary therapeutic strategies and ongoing clinical trials. J Exp Clin Cancer Res. 2017;36(1):108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Andreev D, Liu M, Weidner D, Kachler K, Faas M, Gruneboom A, et al. Osteocyte necrosis triggers osteoclast-mediated bone loss through macrophage-inducible C-type lectin. J Clin Invest. 2020;130(9):4811–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Boyce BF. Advances in the regulation of osteoclasts and osteoclast functions. J Dent Res. 2013;92(10):860–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhang Y, Liang J, Liu P, Wang Q, Liu L, Zhao H. The RANK/RANKL/OPG system and tumor bone metastasis: Potential mechanisms and therapeutic strategies. Front Endocrinol (Lausanne). 2022; 13:1063815. [DOI] [PMC free article] [PubMed]
- 75.De Leon-Oliva D, Barrena-Blazquez S, Jimenez-Alvarez L, Fraile-Martinez O, Garcia-Montero C, Lopez-Gonzalez L, et al. The RANK-RANKL-OPG system: a multifaceted regulator of homeostasis, immunity, and cancer. Med (Kaunas). 2023;59(10):1752. [DOI] [PMC free article] [PubMed]
- 76.Ma X, Yu J. Role of the bone microenvironment in bone metastasis of malignant tumors - therapeutic implications. Cell Oncol (Dordr). 2020;43(5):751–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Satcher RL, Zhang XH. Evolving cancer-niche interactions and therapeutic targets during bone metastasis. Nat Rev Cancer. 2022;22(2):85–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ara T, Declerck YA. Interleukin-6 in bone metastasis and cancer progression. Eur J Cancer. 2010;46(7):1223–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Moore JA, Mistry JJ, Hellmich C, Horton RH, Wojtowicz EE, Jibril A, et al. LC3-associated phagocytosis in bone marrow macrophages suppresses acute myeloid leukemia progression through STING activation. J Clin Invest. 2022;132(5):e153157. [DOI] [PMC free article] [PubMed]
- 80.Kleefeldt F, Upcin B, Bommel H, Schulz C, Eckner G, Allmanritter J, et al. Bone marrow-independent adventitial macrophage progenitor cells contribute to angiogenesis. Cell Death Dis. 2022;13(3):220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Heideveld E. Van den Akker: digesting the role of bone marrow macrophages on hematopoiesis. Immunobiology. 2017;222(6):814–22. [DOI] [PubMed] [Google Scholar]
- 82.Futakuchi M, Fukamachi K, Suzui M. Heterogeneity of tumor cells in the bone microenvironment: mechanisms and therapeutic targets for bone metastasis of prostate or breast cancer. Adv Drug Deliv Rev. 2016;99(Pt B):206–11. [DOI] [PubMed] [Google Scholar]
- 83.Kang J, La Manna F, Bonollo F, Sampson N, Alberts IL, Mingels C, et al. Tumor microenvironment mechanisms and bone metastatic disease progression of prostate cancer. Cancer Lett. 2022;530:156–69. [DOI] [PubMed] [Google Scholar]
- 84.Li S, Kang Y, Zeng Y. Targeting tumor and bone microenvironment: novel therapeutic opportunities for castration-resistant prostate cancer patients with bone metastasis. Biochim Biophys Acta Rev Cancer. 2024;1879(1):189033. [DOI] [PubMed] [Google Scholar]
- 85.Merkens L, Sailer V, Lessel D, Janzen E, Greimeier S, Kirfel J, et al. Aggressive variants of prostate cancer: underlying mechanisms of neuroendocrine transdifferentiation. J Exp Clin Cancer Res. 2022;41(1):46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Rebello RJ, Oing C, Knudsen KE, Loeb S, Johnson DC, Reiter RE, et al. Prostate cancer. Nat Rev Dis Primers. 2021;7(1):9. [DOI] [PubMed] [Google Scholar]
- 87.Swami U, McFarland TR, Nussenzveig R, Agarwal N. Advanced prostate Cancer: treatment advances and future directions. Trends Cancer. 2020;6(8):702–15. [DOI] [PubMed] [Google Scholar]
- 88.Formenti AM, Dalla Volta A, di Filippo L, Berruti A, Giustina A. Effects of Medical treatment of prostate Cancer on Bone Health. Trends Endocrinol Metab. 2021;32(3):135–58. [DOI] [PubMed] [Google Scholar]
- 89.Scimeca M, Trivigno D, Bonfiglio R, Ciuffa S, Urbano N, Schillaci O, et al. Breast cancer metastasis to bone: from epithelial to mesenchymal transition to breast osteoblast-like cells. Semin Cancer Biol. 2021;72:155–64. [DOI] [PubMed] [Google Scholar]
- 90.Verbruggen ASK, McCarthy EC, Dwyer RM, McNamara LM. Temporal and spatial changes in bone mineral content and mechanical properties during breast-cancer bone metastases. Bone Rep. 2022;17:101597. [DOI] [PMC free article] [PubMed]
- 91.Leiter A, Veluswamy RR, Wisnivesky JP. The global burden of lung cancer: current status and future trends. Nat Rev Clin Oncol. 2023;20(9):624–39. [DOI] [PubMed] [Google Scholar]
- 92.Chai X, Yinwang E, Wang Z, Wang Z, Xue Y, Li B, et al. Predictive and prognostic biomarkers for lung cancer bone metastasis and their therapeutic value. Front Oncol. 2021;11:692788. [DOI] [PMC free article] [PubMed]
- 93.Wu S, Pan Y, Mao Y, Chen Y, He Y. Current progress and mechanisms of bone metastasis in lung cancer: a narrative review. Transl Lung Cancer Res. 2021;10(1):439–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Takeuchi A, Yamamoto N, Hayashi K, Matsubara H, Miwa S, Igarashi K, et al. Joint-preservation surgery for pediatric osteosarcoma of the knee joint. Cancer Metastasis Rev. 2019;38(4):709–22. [DOI] [PubMed] [Google Scholar]
- 95.Roberts CC, Daffner RH, Weissman BN, Bancroft L, Bennett DL, Blebea JS, et al. ACR appropriateness criteria on metastatic bone disease. J Am Coll Radiol. 2010;7(6):400–9. [DOI] [PubMed] [Google Scholar]
- 96.Oblak ML, Boston SE, Woods JP, Nykamp S. Comparison of concurrent imaging modalities for staging of dogs with appendicular primary bone tumours. Vet Comp Oncol. 2015;13(1):28–39. [DOI] [PubMed] [Google Scholar]
- 97.Zhang S, Wu Y, Yu J, Ma C, Wang Y, Wang Y, et al. Gadolinium-Bisphosphonate Nanoparticle-Based Low-Dose Radioimmunotherapy for Osteosarcoma. ACS Biomater Sci Eng. 2022;8(12):5329–37. [DOI] [PubMed] [Google Scholar]
- 98.Yin P, Zhong J, Liu Y, Liu T, Sun C, Liu X, et al. Clinical-radiomics models based on plain X-rays for prediction of lung metastasis in patients with osteosarcoma. BMC Med Imaging. 2023;23(1):40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Xiong Q, Wang X, Wang L, Huang Y, Tian X, Fan Y, et al. BMP-2 inhibits lung metastasis of osteosarcoma: an early investigation using an orthotopic model. Onco Targets Ther. 2018;11:7543–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Mebarki M, Medjahedi A, Menemani A, Betterki S, Terki S, Berber N. Osteosarcoma pulmonary metastasis mimicking abnormal skeletal uptake in bone scan: utility of SPECT/CT. Clin Nucl Med. 2013;38(10):e392–4. [DOI] [PubMed] [Google Scholar]
- 101.Filizoglu N, Engur CO, Turkoz HK, Ones T, Erdil TY. Calcified adrenal metastasis of high-Grade Osteosarcoma on FDG PET/CT. Clin Nucl Med. 2021;46(3):e176–8. [DOI] [PubMed] [Google Scholar]
- 102.Zhang X, Guan Z. PET/CT in the diagnosis and prognosis of osteosarcoma. Front Biosci (Landmark Ed). 2018;23(11):2157–65. [DOI] [PubMed] [Google Scholar]
- 103.Pereira HM, Leite Duarte ME, Ribeiro Damasceno I. Nogueira-Barbosa: Machine learning-based CT radiomics features for the prediction of pulmonary metastasis in osteosarcoma. Br J Radiol. 2021;94(1124):20201391. [DOI] [PMC free article] [PubMed]
- 104.Kalus S, Saifuddin A. Whole-body MRI vs bone scintigraphy in the staging of ewing sarcoma of bone: a 12-year single-institution review. Eur Radiol. 2019;29(10):5700–8. [DOI] [PubMed] [Google Scholar]
- 105.Barnett JR, Gikas P, Gerrand C, Briggs TW, Saifuddin A. The sensitivity, specificity, and diagnostic accuracy of whole-bone MRI for identifying skip metastases in appendicular osteosarcoma and Ewing sarcoma. Skeletal Radiol. 2020;49(6):913–9. [DOI] [PubMed] [Google Scholar]
- 106.Farina A, Gasperini C, Aparisi Gomez MP, Bazzocchi A, Fanti S, Nanni C. The role of FDG-PET and whole-body MRI in high Grade Bone Sarcomas with Particular Focus on Osteosarcoma. Semin Nucl Med. 2022;52(5):635–46. [DOI] [PubMed] [Google Scholar]
- 107.Aran V, Devalle S, Meohas W, Heringer M, Cunha Caruso A, Pinheiro Aguiar D, et al. Osteosarcoma, chondrosarcoma and ewing sarcoma: clinical aspects, biomarker discovery and liquid biopsy. Crit Rev Oncol Hematol. 2021;162:103340. [DOI] [PubMed]
- 108.Shulman DS, Crompton BD. Using Liquid Biopsy in the treatment of patient with OS. Adv Exp Med Biol. 2020;1257:95–105. [DOI] [PubMed] [Google Scholar]
- 109.Bao Q, Shen Y, Zhang W. ASO author reflections: metastatic biopsy of Osteosarcoma with circulating RNA. Ann Surg Oncol. 2018;25(Suppl 3):936–7. [DOI] [PubMed] [Google Scholar]
- 110.Sharma AE, Pytel P, Cipriani NA. SOX9 and SATB2 immunohistochemistry cannot reliably distinguish between osteosarcoma and chondrosarcoma on biopsy material. Hum Pathol. 2022;121:56–64. [DOI] [PubMed] [Google Scholar]
- 111.Lopez-Beltran A, Montironi R, Carazo JL, Vidal A, Cheng L. Primary renal osteosarcoma. Am J Clin Pathol. 2014;141(5):747–52. [DOI] [PubMed] [Google Scholar]
- 112.Falconieri G, Cataldi P, Kavalar R, Stitic V, Luzar B. Cutaneous osteoblastic osteosarcoma: report of 2 new cases Integrated with SATB2 immunohistochemistry and review of the literature. Am J Dermatopathol. 2016;38(11):824–31. [DOI] [PubMed] [Google Scholar]
- 113.Lin YH, Jewell BE, Gingold J, Lu L, Zhao R, Wang LL, et al. Osteosarcoma: molecular pathogenesis and iPSC modeling. Trends Mol Med. 2017;23(8):737–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Yang DM, Zhou Q, Furman-Cline L, Cheng X, Luo D, Lai H, et al. Osteosarcoma explorer: a data commons with clinical, genomic, protein, and tissue imaging data for osteosarcoma research. JCO Clin Cancer Inf. 2023;7:e2300104. [DOI] [PMC free article] [PubMed]
- 115.Smeland S, Bielack SS, Whelan J, Bernstein M, Hogendoorn P, Krailo MD, et al. Survival and prognosis with osteosarcoma: outcomes in more than 2000 patients in the EURAMOS-1 (European and American Osteosarcoma Study) cohort. Eur J Cancer. 2019;109:36–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Luo Z, Chen W, Shen X, Qin G, Yuan J, Hu B, et al. Head and neck osteosarcoma: CT and MR imaging features. Dentomaxillofac Radiol. 2020;49(2):20190202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Baidya Kayal E, Kandasamy D, Sharma R, Sharma MC, Bakhshi S, Mehndiratta A. SLIC-supervoxels-based response evaluation of osteosarcoma treated with neoadjuvant chemotherapy using multi-parametric MR imaging. Eur Radiol. 2020;30(6):3125–36. [DOI] [PubMed] [Google Scholar]
- 118.Yildirim O, Al Khatalin M, Kargin OA, Camurdan VB. MRI for evaluation of preoperative chemotherapy in osteosarcoma. Radiography (Lond). 2022;28(3):593–604. [DOI] [PubMed] [Google Scholar]
- 119.Luo Z, Li J, Liao Y, Huang W, Li Y, Shen X. Prediction of response to preoperative neoadjuvant chemotherapy in extremity high-grade osteosarcoma using X-ray and multiparametric MRI radiomics. J Xray Sci Technol. 2023;31(3):611–26. [DOI] [PubMed] [Google Scholar]
- 120.Alge O, Lu L, Li Z, Hua Y, Gryak J, Najarian K. Automated classification of osteosarcoma and benign tumors using RNA-seq and plain X-ray. In: 2020 42nd Annual international conference of the IEEE engineering in Medicine & Biology Society (EMBC). IEEE; 2020. p. 1165–8. [DOI] [PMC free article] [PubMed]
- 121.Holzer G, Hobusch G, Hansen S, Fischer L, Patsch JM. Is there an Association between Bone microarchitecture and fracture in patients who were treated for high-grade Osteosarcoma? A controlled study at Long-Term follow-up using high-resolution Peripheral quantitative CT. Clin Orthop Relat Res. 2021;479(11):2493–501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Silva JAM, Marchiori E, Amorim VB, Barreto MM. CT features of osteosarcoma lung metastasis: a retrospective study of 127 patients. J Bras Pneumol. 2023;49(2):e20220433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ling Z, Yang S, Gou F, Dai Z, Wu J. Intelligent Assistant diagnosis system of Osteosarcoma MRI Image based on Transformer and Convolution in developing countries. IEEE J Biomed Health Inf. 2022;26(11):5563–74. [DOI] [PubMed] [Google Scholar]
- 124.Wu J, Xiao P, Huang H, Gou F, Zhou Z, Dai Z. An Artificial Intelligence Multiprocessing Scheme for the diagnosis of Osteosarcoma MRI images. IEEE J Biomed Health Inf. 2022;26(9):4656–67. [DOI] [PubMed] [Google Scholar]
- 125.Mu H, Zuo D, Chen J, Liu Z, Wang Z, Yang L, et al. Detection and surveillance of circulating tumor cells in osteosarcoma for predicting therapy response and prognosis. Cancer Biol Med. 2022;19(9):1397–409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Body JJ, Bone HG, de Boer RH, Stopeck A, Van Poznak C, Damiao R, et al. Hypocalcaemia in patients with metastatic bone disease treated with denosumab. Eur J Cancer. 2015;51(13):1812–21. [DOI] [PubMed] [Google Scholar]
- 127.Chen XY, Lan M, Zhou Y, Chen WZ, Hu D, Liu JM, et al. Risk factors for bone metastasis from renal cell cancer. J Bone Oncol. 2017;9:29–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Bali P, Lozano-Pope I, Pachow C, Obonyo M. Early detection of tumor cells in bone marrow and peripheral blood in a fast–progressing gastric cancer model. Int J Oncol. 2021;58(3):388–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Hinz S, Hendricks A, Wittig A, Schafmayer C, Tepel J, Kalthoff H, et al. Detection of circulating tumor cells with CK20 RT-PCR is an independent negative prognostic marker in colon cancer patients - a prospective study. BMC Cancer. 2017;17(1):53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Li X, Seebacher NA, Hornicek FJ, Xiao T, Duan Z. Application of liquid biopsy in bone and soft tissue sarcomas: Present and future. Cancer Lett. 2018;439:66–77. [DOI] [PubMed] [Google Scholar]
- 131.Sailer V, Schiffman MH, Kossai M, Cyrta J, Beg S, Sullivan B, et al. Bone biopsy protocol for advanced prostate cancer in the era of precision medicine. Cancer. 2018;124(5):1008–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Nadda R, Repaka R, Mallik N, Sahani AK. A prospective survey on trephine biopsy of bone and bone marrow: an experience with 274 Indian patients’ biopsies. Eur J Med Res. 2023;28(1):193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Gaballah M, Worede F, Alexander C, Arkader A, Cahill AM, Acord MR. Novel method to biopsy aneurysmal bone cysts in children. Skeletal Radiol. 2023;52(12):2479–86. [DOI] [PubMed] [Google Scholar]
- 134.Cheng Z, Li M, Dey R, Chen Y. Nanomaterials for cancer therapy: current progress and perspectives. J Hematol Oncol. 2021;14(1):85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Kesharwani P, Ma R, Sang L, Fatima M, Sheikh A, Abourehab MAS, et al. Gold nanoparticles and gold nanorods in the landscape of cancer therapy. Mol Cancer. 2023;22(1):98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Fan M, Han Y, Gao S, Yan H, Cao L, Li Z, et al. Ultrasmall gold nanoparticles in cancer diagnosis and therapy. Theranostics. 2020;10(11):4944–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Yang Y, Zheng X, Chen L, Gong X, Yang H, Duan X, et al. Multifunctional gold nanoparticles in Cancer diagnosis and treatment. Int J Nanomed. 2022;17:2041–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Chen Y, Yang J, Fu S, Wu J. Gold nanoparticles as Radiosensitizers in Cancer Radiotherapy. Int J Nanomed. 2020;15:9407–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Singh P, Pandit S, Mokkapati V, Garg A, Ravikumar V, Mijakovic I. Gold nanoparticles in diagnostics and therapeutics for human cancer. Int J Mol Sci. 2018;19(7):1979. [DOI] [PMC free article] [PubMed]
- 140.Ju YY, Shi XX, Xu SY, Ma XH, Wei RJ, Hou H, et al. Atomically Precise Water-Soluble Graphene Quantum dot for Cancer Sonodynamic Therapy. Adv Sci (Weinh). 2022;9(19):e2105034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Wu B, Li K, Sun F, Niu J, Zhu R, Qian Y, et al. Trifunctional Graphene Quantum Dot@LDH Integrated Nanoprobes for visualization therapy of gastric Cancer. Adv Healthc Mater. 2021;10(16):e2100512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Zhang H, Ba S, Yang Z, Wang T, Lee JY, Li T, et al. Graphene Quantum dot-based nanocomposites for Diagnosing Cancer Biomarker APE1 in living cells. ACS Appl Mater Interfaces. 2020;12(12):13634–43. [DOI] [PubMed] [Google Scholar]
- 143.Martins CSM, LaGrow AP. Prior: Quantum dots for Cancer-related miRNA monitoring. ACS Sens. 2022;7(5):1269–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Quantum Dots. Shine in Cancer Research. Cancer Discov. 2023;13(12):2492–3. [DOI] [PubMed] [Google Scholar]
- 145.Liao J, Qiu J, Wang G, Du R, Tsidaeva N, Wang W. 3D core-shell Fe(3)O(4)@SiO(2)@MoS(2) composites with enhanced microwave absorption performance. J Colloid Interface Sci. 2021;604:537–49. [DOI] [PubMed] [Google Scholar]
- 146.Liu Y, Li J, Chen M, Chen X, Zheng N. Palladium-based nanomaterials for cancer imaging and therapy. Theranostics. 2020;10(22):10057–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Tian R, Shang Y, Wang Y, Jiang Q, Ding B. DNA nanomaterials-based platforms for Cancer Immunotherapy. Small Methods. 2023;7(5):e2201518. [DOI] [PubMed] [Google Scholar]
- 148.Wang Y, Sun S, Zhang Z, Shi D. Nanomaterials for Cancer Precision Medicine. Adv Mater. 2018;30(17):e1705660. [DOI] [PubMed] [Google Scholar]
- 149.Jeong H, Park W, Kim DH, Na K. Dynamic nanoassemblies of nanomaterials for cancer photomedicine. Adv Drug Deliv Rev. 2021;177:113954. [DOI] [PubMed]
- 150.Chen Y, Wu Y, Sun B, Liu S, Liu H. Two-dimensional nanomaterials for cancer nanotheranostics. Small. 2017;13(10):1603446. [DOI] [PubMed]
- 151.Qu A, Sun M, Xu L, Liu L, Guo L, Chen P, et al. Chiral nanomaterials for Cancer vaccines. Small Methods. 2024;8(3):e2301332. [DOI] [PubMed] [Google Scholar]
- 152.Ni Q, Xu F, Wang Y, Li Y, Qing G, Zhang Y, et al. Nanomaterials with changeable physicochemical property for boosting cancer immunotherapy. J Control Release. 2022;342:210–27. [DOI] [PubMed] [Google Scholar]
- 153.Wang L, Li P, Xiao X, Li J, Li J, Yang HH, et al. Generating lung-metastatic osteosarcoma targeting aptamers for in vivo and clinical tissue imaging. Talanta. 2018;188:66–73. [DOI] [PubMed] [Google Scholar]
- 154.Anzidei M, Napoli A, Sacconi B, Boni F, Noce V, Di Martino M, et al. Magnetic resonance-guided focused ultrasound for the treatment of painful bone metastases: role of apparent diffusion coefficient (ADC) and dynamic contrast enhanced (DCE) MRI in the assessment of clinical outcome. Radiol Med. 2016;121(12):905–15. [DOI] [PubMed] [Google Scholar]
- 155.Sekita A, Matsugaki A, Ishimoto T, Nakano T. Synchronous disruption of anisotropic arrangement of the osteocyte network and collagen/apatite in melanoma bone metastasis. J Struct Biol. 2017;197(3):260–70. [DOI] [PubMed] [Google Scholar]
- 156.Loh KP, Ho D, Chiu GNC, Leong DT, Pastorin G, Chow EK. Clinical applications of Carbon nanomaterials in Diagnostics and Therapy. Adv Mater. 2018;30(47):e1802368. [DOI] [PubMed] [Google Scholar]
- 157.Illath K, Kar S, Gupta P, Shinde A, Wankhar S, Tseng FG, et al. Microfluidic nanomaterials: from synthesis to biomedical applications. Biomaterials. 2022;280:121247. [DOI] [PubMed]
- 158.Liu C, Yazdani N, Moran CS, Salomon C, Seneviratne CJ, Ivanovski S, et al. Unveiling clinical applications of bacterial extracellular vesicles as natural nanomaterials in disease diagnosis and therapeutics. Acta Biomater. 2024;180:18–45. [DOI] [PubMed] [Google Scholar]
- 159.Wang X, Zhong X, Li J, Liu Z, Cheng L. Inorganic nanomaterials with rapid clearance for biomedical applications. Chem Soc Rev. 2021;50(15):8669–742. [DOI] [PubMed] [Google Scholar]
- 160.Chen J, Fan T, Xie Z, Zeng Q, Xue P, Zheng T, et al. Advances in nanomaterials for photodynamic therapy applications: status and challenges. Biomaterials. 2020;237:119827. [DOI] [PubMed]
- 161.Takashi H, Kentaro M, Hiromichi O, Norihiko S, Norio Y, Katsuhiro H, et al. Trabectedin and irinotecan combination regresses a cisplatinum-resistant osteosarcoma in a patient-derived orthotopic xenograft nude-mouse model. Biochem Biophys Res Commun. 2019;513(2):326–31. [DOI] [PubMed] [Google Scholar]
- 162.C. L. Schwartz, R. Gorlick, L. Teot, M. Krailo, Z. Chen, A. Goorin, et al.: Multiple drug resistance in osteogenic sarcoma: INT0133 from the Children’s Oncology Group. J Clin Oncol. 2007;25(15):2057–62. [DOI] [PubMed]
- 163.Trushina DB, Borodina TN, Belyakov S, Antipina MN. Calcium carbonate vaterite particles for drug delivery: advances and challenges. Mater Today Adv. 2022;14:100214. [DOI] [PMC free article] [PubMed]
- 164.Talebian S, Mendes B, Conniot J, Farajikhah S, Dehghani F, Li Z, et al. Biopolymeric Coatings for Local Release of Therapeutics from Biomedical implants. Adv Sci (Weinh). 2023;10(12):e2207603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Moradi Kashkooli F, Jakhmola A, Hornsby TK, Tavakkoli JJ, Kolios MC. Ultrasound-mediated nano drug delivery for treating cancer: fundamental physics to future directions. J Control Release. 2023;355:552–78. [DOI] [PubMed] [Google Scholar]
- 166.Wu H, Wei M, Xu Y, Li Y, Zhai X, Su P, et al. PDA-Based drug delivery nanosystems: a potential Approach for Glioma Treatment. Int J Nanomed. 2022;17:3751–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Haghiralsadat F, Amoabediny G, Naderinezhad S, Zandieh-Doulabi B, Forouzanfar T, Helder MN. Codelivery of doxorubicin and JIP1 siRNA with novel EphA2-targeted PEGylated cationic nanoliposomes to overcome osteosarcoma multidrug resistance. Int J Nanomed. 2018;13:3853–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Li X, Wang L, Wang L, Yu J, Lu G, Zhao W, et al. Overcoming therapeutic failure in osteosarcoma via apatinib-encapsulated hydrophobic poly(ester amide) nanoparticles. Biomater Sci. 2020;8(21):5888–99. [DOI] [PubMed] [Google Scholar]
- 169.He J, Chen C, Chen L, Cheng R, Sun J, Liu X, et al. Honeycomb-like hydrogel microspheres for 3D bulk construction of tumor models. Research. 2022;9809763. [DOI] [PMC free article] [PubMed]
- 170.Salaam AD, Hwang P, McIntosh R, Green HN, Jun HW, Dean D. Nanodiamond-DGEA peptide conjugates for enhanced delivery of doxorubicin to prostate cancer. Beilstein J Nanotechnol. 2014;5:937–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Freeman FE, Dosta P, Shanley LC, Ramirez Tamez N, Riojas Javelly CJ, Mahon OR, et al. Localized nanoparticle-mediated delivery of miR-29b normalizes the dysregulation of bone homeostasis caused by Osteosarcoma whilst simultaneously inhibiting Tumor Growth. Adv Mater. 2023;35(23):e2207877. [DOI] [PubMed] [Google Scholar]
- 172.Wang Y, Chen J, Gong L, Wang Y, Siltari A, Lou YR, et al. MiR26a reverses enzalutamide resistance in a bone-tumor targeted system with an enhanced effect on bone metastatic CRPC. J Nanobiotechnol. 2024;22(1):145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Yu Q, Gao Y, Dai W, Li D, Zhang L, Hameed MMA, et al. Cell membrane-camouflaged Chitosan-Polypyrrole Nanogels co-deliver drug and gene for targeted chemotherapy and bone metastasis inhibition of prostate cancer. Adv Healthc Mater. 2024;13(20):e2400114. [DOI] [PubMed]
- 174.Shen Y, Zou Y, Bie B, Lv Y. Hierarchically released Liquid Metal nanoparticles for mild Photothermal Therapy/Chemotherapy of breast Cancer Bone metastases via Remodeling Tumor Stromal Microenvironment. Adv Healthc Mater. 2023;12(27):e2301080. [DOI] [PubMed] [Google Scholar]
- 175.Alkilany AM, Murphy CJ. Toxicity and cellular uptake of gold nanoparticles: what we have learned so far? J Nanopart Res. 2010;12(7):2313–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Carnovale C, Bryant G, Shukla R, Bansal V. Size, shape and surface chemistry of nano-gold dictate its cellular interactions, uptake and toxicity. Prog Mater Sci. 2016;83:152–90. [Google Scholar]
- 177.Na L, Pengxiang Z, Didier A. Anisotropic gold nanoparticles: synthesis, properties, applications, and toxicity. Angew Chem Int Ed Engl. 2014;53(7):1756–89. [DOI] [PubMed]
- 178.Liao J, Shi K, Jia Y, Wu Y, Qian Z. Gold nanorods and nanohydroxyapatite hybrid hydrogel for preventing bone tumor recurrence via postoperative photothermal therapy and bone regeneration promotion. Bioactive Mater. 2021;6(8):2221–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012;337(6096):816–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Mali P, Yang L, Esvelt KM, Aach J, Guell M, DiCarlo JE, et al. RNA-guided human genome engineering via Cas9. Science. 2013;339(6121):823–6. [DOI] [PMC free article] [PubMed]
- 181.Hess GT, Tycko J, Yao D, Bassik MC. Methods and applications of CRISPR-Mediated base editing in eukaryotic genomes. Mol Cell. 2017;68(1):26–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Liu BY, He XY, Xu C, Xu L, Ai SL, Cheng SX, et al. A dual-targeting delivery system for effective genome editing and in situ detecting related protein expression in edited cells. Biomacromolecules. 2018;19(7):2957–68. [DOI] [PubMed] [Google Scholar]
- 183.Mondal S, Park S, Choi J, Vu TTH, Doan VHM, Vo TT, et al. Hydroxyapatite: a journey from biomaterials to advanced functional materials. Adv Colloid Interface Sci. 2023;321:103013. [DOI] [PubMed]
- 184.Verma R, Mishra SR, Gadore V, Ahmaruzzaman M. Hydroxyapatite-based composites: excellent materials for environmental remediation and biomedical applications. Adv Colloid Interface Sci. 2023;315:102890. [DOI] [PubMed]
- 185.Yook H, Hwang J, Yeo W, Bang J, Kim J, Kim TY, et al. Design strategies for hydroxyapatite-based materials to Enhance their Catalytic Performance and Applicability. Adv Mater. 2023;35(43):e2204938. [DOI] [PubMed] [Google Scholar]
- 186.Ibrahim M, Labaki M, Giraudon JM, Lamonier JF. Hydroxyapatite, a multifunctional material for air, water and soil pollution control: A review. J Hazard Mater. 2020; 383:121139. [DOI] [PubMed]
- 187.Biedrzycka A, Skwarek E, Hanna UM. Hydroxyapatite with magnetic core: Synthesis methods, properties, adsorption and medical applications. Adv Colloid Interface Sci. 2021;291:102401. [DOI] [PubMed]
- 188.Miyah Y, El Messaoudi N, Benjelloun M, Acikbas Y, Şenol ZM, Ciğeroğlu Z, et al. Advanced applications of hydroxyapatite nanocomposite materials for heavy metals and organic pollutants removal by adsorption and photocatalytic degradation: a review. Chemosphere. 2024;358:142236. [DOI] [PubMed]
- 189.Wu F, Chen W, Gillis B, Fischbach C, Estroff LA, Gourdon D. Protein-crystal interface mediates cell adhesion and proangiogenic secretion. Biomaterials. 2017;116:174–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Wang R, Liu W, Wang Q, Li G, Wan B, Sun Y, et al. Anti-osteosarcoma effect of hydroxyapatite nanoparticles both in vitro and in vivo by downregulating the FAK/PI3K/Akt signaling pathway. Biomater Sci. 2020;8(16):4426–37. [DOI] [PubMed] [Google Scholar]
- 191.Han R, Min Y, Li G, Chen S, Xie M, Zhao Z. Supercritical CO(2)-assisted fabrication of CM-PDA/SF/nHA nanofibrous scaffolds for bone regeneration and chemo-photothermal therapy against osteosarcoma. Biomater Sci. 2023;11(15):5218–31. [DOI] [PubMed] [Google Scholar]
- 192.Zhang G, Lu Y, Song J, Huang D, An M, Chen W, et al. A multifunctional nano-hydroxyapatite/MXene scaffold for the photothermal/dynamic treatment of bone tumours and simultaneous tissue regeneration. J Colloid Interface Sci. 2023;652(Pt B):1673–84. [DOI] [PubMed] [Google Scholar]
- 193.Dozzo A, Chullipalliyalil K, McAuliffe M, O’Driscoll CM, Ryan KB. Nano-hydroxyapatite/PLGA mixed scaffolds as a tool for drug development and to study metastatic prostate cancer in the bone. Pharmaceutics. 2023;15(1):242. [DOI] [PMC free article] [PubMed]
- 194.Yang X, Zhai D, Song J, Qing R, Wang B, Ji J, et al. Rhein-PEG-nHA conjugate as a bone targeted drug delivery vehicle for enhanced cancer chemoradiotherapy. Nanomedicine. 2020;27:102196. [DOI] [PubMed]
- 195.Xiong Q, Zhang N, Zhang M, Wang M, Wang L, Fan Y, et al. Engineer a pre-metastatic niched microenvironment to attract breast cancer cells by utilizing a 3D printed polycaprolactone/nano-hydroxyapatite osteogenic scaffold - an in vitro model system for proof of concept. J Biomed Mater Res B Appl Biomater. 2022;110(7):1604–14. [DOI] [PubMed] [Google Scholar]
- 196.Chen J, Wen J, Fu Y, Li X, Huang J, Guan X, et al. A bifunctional bortezomib-loaded porous nano-hydroxyapatite/alginate scaffold for simultaneous tumor inhibition and bone regeneration. J Nanobiotechnol. 2023;21(1):174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Liu Y, Nadeem A, Sebastian S, Olsson MA, Wai SN, Styring E, et al. Bone mineral: a trojan horse for bone cancers. Efficient mitochondria targeted delivery and tumor eradication with nano hydroxyapatite containing doxorubicin. Mater Today Bio. 2022;14:100227. [DOI] [PMC free article] [PubMed]
- 198.Xu Y, Qi J, Sun W, Zhong W, Wu H. Therapeutic effects of zoledronic acid-loaded hyaluronic acid/polyethylene glycol/nano-hydroxyapatite nanoparticles on osteosarcoma. Front Bioeng Biotechnol. 2022;10:897641. [DOI] [PMC free article] [PubMed]
- 199.Kang NW, Lee JY, Kim DD. Hydroxyapatite-binding albumin nanoclusters for enhancing bone tumor chemotherapy. J Control Release. 2022;342:111–21. [DOI] [PubMed] [Google Scholar]
- 200.Yang X, Gao S, Yang B, Yang Z, Lou F, Huang P, et al. Bioinspired Tumor-Targeting and Biomarker-Activatable Cell-Material Interfacing System enhances Osteosarcoma Treatment via Biomineralization. Adv Sci (Weinh). 2023;10(22):e2302272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Mondal J, Pillarisetti S, Junnuthula V, Saha M, Hwang SR, Park IK, et al. Hybrid exosomes, exosome-like nanovesicles and engineered exosomes for therapeutic applications. J Control Release. 2023;353:1127–49. [DOI] [PubMed] [Google Scholar]
- 202.Ansari MA, Thiruvengadam M, Venkidasamy B, Alomary MN, Salawi A, Chung IM, et al. Exosome-based nanomedicine for cancer treatment by targeting inflammatory pathways: current status and future perspectives. Semin Cancer Biol. 2022;86(Pt 2):678–96. [DOI] [PubMed] [Google Scholar]
- 203.Kim J, Li S, Zhang S, Wang J. Plant-derived exosome-like nanoparticles and their therapeutic activities. Asian J Pharm Sci. 2022;17(1):53–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Dad HA, Gu TW, Zhu AQ, Huang LQ, Peng LH. Plant exosome-like nanovesicles: emerging therapeutics and drug delivery nanoplatforms. Mol Ther. 2021;29(1):13–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Lu M, Huang Y. Bioinspired exosome-like therapeutics and delivery nanoplatforms. Biomaterials. 2020;242:119925. [DOI] [PubMed]
- 206.Yadav K, Vijayalakshmi R, Kumar Sahu K, Sure P, Chahal K, Yadav R, et al. Exosome-based macromolecular neurotherapeutic drug delivery approaches in overcoming the blood-brain barrier for treating brain disorders. Eur J Pharm Biopharm. 2024;199:114298. [DOI] [PubMed]
- 207.Bai C, Liu J, Zhang X, Li Y, Qin Q, Song H, et al. Research status and challenges of plant-derived exosome-like nanoparticles. Biomed Pharmacother. 2024;174:116543. [DOI] [PubMed]
- 208.Wei H, Chen J, Wang S, Fu F, Zhu X, Wu C, et al. A Nanodrug Consisting of Doxorubicin and Exosome Derived from mesenchymal stem cells for Osteosarcoma Treatment in Vitro. Int J Nanomed. 2019;14:8603–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Wang J, Li M, Jin L, Guo P, Zhang Z, Zhanghuang C, et al. Exosome mimetics derived from bone marrow mesenchymal stem cells deliver doxorubicin to osteosarcoma in vitro and in vivo. Drug Deliv. 2022;29(1):3291–303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Chen W, Li Z, Yu N, Zhang L, Li H, Chen Y, et al. Bone-targeting exosome nanoparticles activate Keap1 / Nrf2 / GPX4 signaling pathway to induce ferroptosis in osteosarcoma cells. J Nanobiotechnol. 2023;21(1):355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Zhang M, Zhang J, Chen J, Zeng Y, Zhu Z, Wan Y. Fabrication of curcumin-modified TiO(2) nanoarrays via Cyclodextrin Based Polymer Functional Coatings for Osteosarcoma Therapy. Adv Healthc Mater. 2019;8(23):e1901031. [DOI] [PubMed] [Google Scholar]
- 212.Yang Y, Alencar LMR, Pijeira MSO, Batista BDS, Franca ARS, Rates ERD, et al. [(223)Ra] RaCl(2) nanomicelles showed potent effect against osteosarcoma: targeted alpha therapy in the nanotechnology era. Drug Deliv. 2022;29(1):186–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Cai JX, Liu JH, Wu JY, Li YJ, Qiu XH, Xu WJ, et al. Hybrid cell membrane-functionalized biomimetic nanoparticles for targeted therapy of Osteosarcoma. Int J Nanomed. 2022;17:837–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Yin X, Ran S, Cheng H, Zhang M, Sun W, Wan Y, et al. Polydopamine-modified ZIF-8 nanoparticles as a drug carrier for combined chemo-photothermal osteosarcoma therapy. Colloids Surf B Biointerfaces. 2022;216:112507. [DOI] [PubMed]
- 215.Ju Q, Huang R, Hu R, Fan J, Zhang D, Ding J, et al. Phytic acid-modified manganese dioxide nanoparticles oligomer for magnetic resonance imaging and targeting therapy of osteosarcoma. Drug Deliv. 2023;30(1):2181743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Vanderburgh J, Hill JL, Gupta MK, Kwakwa KA, Wang SK, Moyer K, et al. Tuning ligand density to optimize pharmacokinetics of targeted nanoparticles for Dual Protection against Tumor-Induced Bone Destruction. ACS Nano. 2020;14(1):311–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Li D, Ma S, Xu D, Meng X, Lei N, Liu C, et al. Peptide-functionalized therapeutic nanoplatform for treatment orthotopic triple negative breast cancer and bone metastasis. Nanomedicine. 2023;50:102669. [DOI] [PubMed]
- 218.Yasuteru U, Daisuke A, Yukihiro H, Yoshinori K, Tristan B, Mako K, et al. Selective molecular imaging of viable cancer cells with pH-activatable fluorescence probes. Nat Med. 2009;15(1):104–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Xue J, Yao Y, Wang M, Wang Z, Xue Y, Li B, et al. Recent studies on proteins and polysaccharides-based pH-responsive fluorescent materials. Int J Biol Macromol. 2024;260(Pt 1):129534. [DOI] [PubMed] [Google Scholar]
- 220.Chen Z, Wang X, Zhao N, Chen H, Guo G. Advancements in pH-responsive nanocarriers: enhancing drug delivery for tumor therapy. Expert Opin Drug Deliv. 2023;20(11):1623–42. [DOI] [PubMed] [Google Scholar]
- 221.He Y, Vasilev K, Zilm P. pH-responsive biomaterials for the treatment of dental caries-a focussed and critical review. Pharmaceutics. 2023;15(7):1837. [DOI] [PMC free article] [PubMed]
- 222.Zarur M, Seijo-Rabina A, Goyanes A, Concheiro A. Alvarez-Lorenzo: pH-responsive scaffolds for tissue regeneration: in vivo performance. Acta Biomater. 2023;168:22–41. [DOI] [PubMed] [Google Scholar]
- 223.Li R, Landfester K, Ferguson CTJ. Temperature- and pH-Responsive polymeric photocatalysts for enhanced control and recovery. Angew Chem Int Ed Engl. 2022;61(51):e202211132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Meng Y, Qiu C, Li X, McClements DJ, Sang S, Jiao A, et al. Polysaccharide-based nano-delivery systems for encapsulation, delivery, and pH-responsive release of bioactive ingredients. Crit Rev Food Sci Nutr. 2024;64(1):187–201. [DOI] [PubMed] [Google Scholar]
- 225.Ding H, Tan P, Fu S, Tian X, Zhang H, Ma X, et al. Preparation and application of pH-responsive drug delivery systems. J Control Release. 2022;348:206–38. [DOI] [PubMed] [Google Scholar]
- 226.Yang F, Wen X, Ke QF, Xie XT, Guo YP. pH-responsive mesoporous ZSM-5 zeolites/chitosan core-shell nanodisks loaded with doxorubicin against osteosarcoma. Mater Sci Eng C Mater Biol Appl. 2018;85:142–53. [DOI] [PubMed] [Google Scholar]
- 227.Zhou X, Moussa FM, Mankoci S, Ustriyana P, Zhang N, Abdelmagid S, et al. Orthosilicic acid, Si(OH)4, stimulates osteoblast differentiation in vitro by upregulating miR-146a to antagonize NF-kappaB activation. Acta Biomater. 2016;39:192–202. [DOI] [PubMed] [Google Scholar]
- 228.Zhu J, Huo Q, Xu M, Yang F, Li Y, Shi H, et al. Bortezomib-Catechol conjugated prodrug micelles: combining bone targeting and aryl boronate-based pH-responsive drug release for cancer bone-metastasis therapy. Nanoscale. 2018;10(38):18387–97. [DOI] [PubMed] [Google Scholar]
- 229.Qiao H, Cui Z, Yang S, Ji D, Wang Y, Yang Y, et al. Targeting osteocytes to attenuate early breast Cancer Bone Metastasis by Theranostic Upconversion nanoparticles with Responsive Plumbagin Release. ACS Nano. 2017;11(7):7259–73. [DOI] [PubMed] [Google Scholar]
- 230.Chi T, Sang T, Wang Y, Ye Z. Cleavage and Noncleavage Chemistry in reactive oxygen species (ROS)-Responsive materials for Smart Drug Delivery. Bioconjug Chem. 2024;35(1):1–21. [DOI] [PubMed] [Google Scholar]
- 231.Ding DF, Xue Y, Wu XC, Zhu ZH, Ding JY, Song YJ, et al. Recent advances in reactive oxygen species (ROS)-Responsive Polyfunctional Nanosystems 3.0 for the treatment of Osteoarthritis. J Inflamm Res. 2022;15:5009–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Wang P, Gong Q, Hu J, Li X, Zhang X. Reactive oxygen species (ROS)-Responsive Prodrugs, Probes, and Theranostic Prodrugs: applications in the ROS-Related diseases. J Med Chem. 2021;64(1):298–325. [DOI] [PubMed] [Google Scholar]
- 233.Liu Y, Liu Y, Zang J, Abdullah AAI, Li Y, Dong H. Design strategies and applications of ROS-Responsive Phenylborate Ester-based nanomedicine. ACS Biomater Sci Eng. 2020;6(12):6510–27. [DOI] [PubMed] [Google Scholar]
- 234.Xu Y, Yang W, Zhang B. ROS-responsive probes for low-background optical imaging: a review. Biomed Mater. 2021;16(2):022002. [DOI] [PubMed] [Google Scholar]
- 235.Koo MA, Lee MH, Park JC. Recent advances in ROS-responsive cell sheet techniques for tissue engineering. Int J Mol Sci. 2019;20(22):5656. [DOI] [PMC free article] [PubMed]
- 236.Yao Y, Zhang H, Wang Z, Ding J, Wang S, Huang B, et al. Reactive oxygen species (ROS)-responsive biomaterials mediate tissue microenvironments and tissue regeneration. J Mater Chem B. 2019;7(33):5019–37. [DOI] [PubMed] [Google Scholar]
- 237.Wang YC, Dai HL, Li ZH, Meng ZY, Xiao Y, Zhao Z. Mesoporous polydopamine-coated hydroxyapatite nano-composites for ROS-triggered nitric oxide-enhanced photothermal therapy of osteosarcoma. J Mater Chem B. 2021;9(36):7401–8. [DOI] [PubMed] [Google Scholar]
- 238.Ying H, Wang H, Jiang G, Tang H, Li L, Zhang J. Injectable agarose hydrogels and doxorubicin-encapsulated iron-gallic acid nanoparticles for chemodynamic-photothermal synergistic therapy against osteosarcoma. Front Chem. 2022;10:1045612. [DOI] [PMC free article] [PubMed]
- 239.Gemini-Piperni S, Ricci-Junior E, Ilem-Ozdemir D, da Silva Batista B, Alencar LMR, Rossi AM, et al. Nano-hydroxyapatite radiolabeled with radium dichloride [(223)Ra] RaCl(2) for bone cancer targeted alpha therapy: in vitro assay and radiation effect on the nanostructure. Colloids Surf B Biointerfaces. 2023;223:113174. [DOI] [PubMed]
- 240.Zeng WN, Yu QP, Wang D, Liu JL, Yang QJ, Zhou ZK, et al. Mitochondria-targeting graphene oxide nanocomposites for fluorescence imaging-guided synergistic phototherapy of drug-resistant osteosarcoma. J Nanobiotechnol. 2021;19(1):79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Liu H, He L, Kuzmanović M, Huang Y, Zhang L, Zhang Y, et al. Advanced nanomaterials in Medical 3D Printing. Small Methods. 2024;8(3):e2301121. [DOI] [PubMed] [Google Scholar]
- 242.Varaprasad K, Karthikeyan C, Yallapu MM, Sadiku R. The significance of biomacromolecule alginate for the 3D printing of hydrogels for biomedical applications. Int J Biol Macromol. 2022;212:561–78. [DOI] [PubMed] [Google Scholar]
- 243.Park YG, Yun I, Chung WG, Park W, Lee DH, Park JU. High-resolution 3D Printing for Electronics. Adv Sci (Weinh). 2022;9(8):e2104623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.van der Heide D, Cidonio G, Stoddart MJ, D’Este M. 3D printing of inorganic-biopolymer composites for bone regeneration. Biofabrication. 2022;14(4):042003. [DOI] [PubMed]
- 245.Hao S, Ha L, Cheng G, Wan Y, Xia Y, Sosnoski DM, et al. A spontaneous 3D Bone-On-a-Chip for bone metastasis study of breast Cancer cells. Small. 2018;14(12):e1702787. [DOI] [PubMed] [Google Scholar]
- 246.Zhu W, Holmes B, Glazer RI. Zhang: 3D printed nanocomposite matrix for the study of breast cancer bone metastasis. Nanomedicine. 2016;12(1):69–79. [DOI] [PubMed] [Google Scholar]
- 247.Zhu W, Castro NJ, Cui H, Zhou X, Boualam B, McGrane R, et al. A 3D printed nano bone matrix for characterization of breast cancer cell and osteoblast interactions. Nanotechnology. 2016;27(31):315103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Kumar TA, Chandra US, Manish K, Kamla P, Deepak K, Ravinder V, et al. Insights on development aspects of polymeric nanocarriers: the translation from bench to clinic. Polymers. 2022;14(17):3545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Jiakang J, Pengcheng Y, Wei Y, Jinti L, Ankai X, Xiaodan X et al. Mitochondria-targeting polymer micelle of dichloroacetate induced pyroptosis to enhance osteosarcoma immunotherapy. ACS Nano. 2022;16(7):10327-40. [DOI] [PubMed]
- 250.Kexiao Y, Hang Z, Yamei X, Youde C, Yuanyi Z, Bing L. Engineering a triple-functional magnetic gel driving mutually-synergistic mild hyperthermia-starvation therapy for osteosarcoma treatment and augmented bone regeneration. J Nanobiotechnol. 2023;21(1):201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Maryam A, Maryam H, Maryam B, Khalil A, Mohammad R. PEG-PLA nanoparticles decorated with small-molecule PSMA ligand for targeted delivery of galbanic acid and docetaxel to prostate cancer cells. J Cell Physiol. 2020;235(5):4618–30. [DOI] [PubMed] [Google Scholar]
- 252.Tan ML, Shao P, Friedhuber AM, Moorst Mv, Elahy M, Indumathy S, et al. The potential role of free chitosan in bone trauma and bone cancer management. Biomaterials. 2014;35(27):7828–38. [DOI] [PubMed] [Google Scholar]
- 253.Cui G, Wu J, Lin J, Liu W, Chen P, Yu M, et al. Graphene-based nanomaterials for breast cancer treatment: promising therapeutic strategies. J Nanobiotechnol. 2021;19(1):211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Raju GSR, Dariya B, Mungamuri SK, Chalikonda G, Kang SM, Khan IN, et al. Nanomaterials multifunctional behavior for enlightened cancer therapeutics. Semin Cancer Biol. 2021;69:178–89. [DOI] [PubMed] [Google Scholar]
- 255.Luo Z, Lu Y, Shi Y, Jiang M, Shan X, Li X, et al. Neutrophil hitchhiking for drug delivery to the bone marrow. Nat Nanotechnol. 2023;18(6):647–56. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.