Summary
Circadian rhythms significantly impact (patho)physiological processes, with disruptions linked to neurodegenerative diseases and heightened cancer vulnerability. While immunotherapy has shown promise in treating various cancers, its efficacy in brain malignancies remains limited. This review explores the nexus of circadian rhythms and immunotherapy in brain cancer treatment, emphasising precision through alignment with the body's internal clock. We evaluate circadian regulation of immune responses, including cell localisation and functional phenotype, and discuss how circadian dysregulation affects anti-cancer immunity. Additionally, we analyse and assess the effectiveness of current immunotherapeutic approaches for brain cancer including immune checkpoint blockades, adoptive cellular therapies, and other novel strategies. Future directions, such as chronotherapy and personalised treatment schedules, are proposed to optimise immunotherapy precision against brain cancers. Overall, this review provides an understanding of the often-overlooked role of circadian rhythms in brain cancer and suggests avenues for improving immunotherapeutic outcomes.
Keywords: Circadian rhythms, Brain cancer, Immune system, Immunotherapy, Immune checkpoints, Adoptive cellular therapy, Chronomodulated therapy
Search strategy and selection criteria.
A literature search was performed on PubMed with the following search terms (ependymoma [tiab] OR atypical teratoma [tiab] OR glioma [tiab] OR glioblastoma [tiab] OR pineal tum∗ [tiab] OR pituitary adenoma [tiab] OR meningioma [tiab] OR medulloblastoma [tiab] OR astrocytoma [tiab] OR oligodendrocytoma [tiab] OR craniopharyngioma [tiab] OR brain cancer [tiab]) AND (circadian [tiab] OR clock prot∗ [tiab] OR circadian rhythms [tiab] OR circadian clocks [tiab] OR molecular clock [tiab] OR BMAL1 [tiab] OR CLOCK [tiab] OR CRY1 [tiab] OR CRY2 [tiab] OR CRY3 [tiab] OR PER1 [tiab] OR PER2 [tiab] OR RORα [tiab] OR RORβ[tiab] OR RORγ [tiab] OR REV-ERBα [tiab] OR REV-ERBβ [tiab]) from January 2015 until June 2024. Only original research papers published in English were reviewed. The final reference list was selected based on originality and relevance to the scope of this review.
Introduction
In 2017 Jeffrey C. Hall, Michael Rosbash and Michael W. Young were awarded with the Nobel Prize for their discovery of the molecular components that comprise the biological clock.1, 2, 3, 4 Their research was pivotal in the uncovering of the role of circadian rhythms in physiology and pathophysiology. Nowadays, it is established that circadian (Latin “circa diem” = about a day) rhythms are an outward manifestation of an internal timing system, which is crucial for maintaining homeostatic balance in organisms.
Circadian rhythms at the molecular level are generated by a core molecular clock, which consists of an evolutionarily conserved transcriptional-translational feedback loop (TTFL; Fig. 1).5 The molecular clock mechanism comprises intertwined positive and negative feedback loops. At the core, the transcriptional activators circadian locomotor output cycles kaput (CLOCK; and its paralog, NPAS2) and brain and muscle ARNT-Like 1 (BMAL1), form heterodimers that bind to enhancer box elements (E-boxes) of cryptochrome (CRY1/2), period (PER1/2/3) and tyrosine-protein kinase transmembrane receptor RORα/β/γ and REV-ERBα/β, thereby stimulating their transcription.5,6 Subsequently, PER/CRY heterodimers accumulate and inhibit BMAL1/CLOCK activity. RORα/β/γ and REV-ERBα/β compete for binding to ROR response element (RORE), a promotor for BMAL1, leading either to its activation or inhibition, respectively. Together, the interplay of these feedback loops causes rhythmic fluctuations in clock-controlled gene expression, with a period of approximately 24 h. Virtually all cell types, including neurons and glial cells, express autonomous circadian rhythms driven by this TTFL.9 The oscillations in expression of the circadian core clock genes, and the genes that they regulate by binding to transcription enhancing elements, drive cell functionality across the 24-h cycle.10 For instance, the gut clock regulates intestinal motility and nutrient absorption to match the timing of maximal monosaccharide uptake to the habitual feeding periods.11 Furthermore, circadian rhythms in the kidneys cause fluctuations in renal plasma flow, glomerular filtration rate and tubular reabsorption and secretion processes.12 To prevent misalignment of these cellular processes across the whole body, daily synchronisation of peripheral clocks is needed.
Fig. 1.
Overview of the core clock machinery in the brain. The transcriptional activators circadian locomotor output cycles kaput (CLOCK) and brain and muscle ARNT-Like 1 (BMAL1) form heterodimers that bind to enhancer box (E-box) elements, thereby driving the transcription of cryptochrome (CRY1/2), period (PER1/2/3) and tyrosine-protein kinase transmembrane receptor RORα/β/γ and REV-ERBα/β.5,6 PER/CRY heterodimers accumulate to inhibit BMAL1/CLOCK activity. Concurrently, RORα/β/γ and REV-ERBα/β compete for binding to ROR response element (RORE) on the BMAL1 promoter, respectively activating or inhibiting its transcription. This intricate feedback loop generates rhythmic fluctuations in clock-controlled gene expression within 24-h cycles.5,7 The molecular clock is present in virtually all cell types in the human body, including neurons, astrocytes, microglia, and oligodendrocytes and drives daily fluctuations in their morphology and functionality.8
Daily synchronisation is regulated by a central master clock, located in the suprachiasmatic nucleus (SCN) of the hypothalamus.6 The SCN generates coherent oscillations of the molecular clock, thereby synchronising peripheral clocks.6 Environmental cues such as exercise, food and social contacts act as Zeitgebers (“time givers”) to align the clock of the SCN to the outside world, with light being the main clock input signal.5 The SCN in turn harmonises all peripheral clocks, thereby aligning cellular processes across the whole body.
Of note, officially 24-h rhythms in the presence of natural synchronisers are referred to as diurnal (or nocturnal) rhythms, while the term ‘circadian rhythms’ is used for rhythms that are expressed in the absence of any 24-h signal cues from the external environment.13 However, almost all diurnal rhythms are found to be circadian. Therefore, in this review the term circadian will be used for all 24-h oscillatory processes, regardless of external inputs.
Disruptions of the circadian clock can result in a range of health issues spanning neurological, metabolic, endocrine, cardiovascular, and immune system morbidities.14 Animal studies show that disturbances of the molecular clock in the brain can lead to spontaneous astrogliosis, increased oxidative damage, synaptic degeneration, and other neurodegenerative alterations.15 Moreover, these disruptions are linked to neurodegenerative disorders such as Alzheimer's Disease and Parkinson's Disease, as well as an increased risk for developing various malignancies, including brain cancers.16,17
Brain cancer treatment faces several challenges. Depending on the severity and invasiveness of brain cancers, treatment outcome varies greatly between different forms of brain tumours.18 Depending on localisation in the brain, tumours are typically challenging to remove completely during surgery, while systemic chemotherapies and precision medicine delivery is hindered by the blood–brain barrier (BBB).19 Furthermore, brain cancers often exhibit cellular and molecular heterogeneity, making them resistant to conventional treatments.19 These challenges necessitate aggressive treatment regimes, such as high-dose radiotherapy or combination chemotherapy, often culminating in severe side effects and reduced quality of life in survivors.19 An emerging alternative treatment is immunotherapy. These therapies stimulate the immune system to create a targeted reaction to the tumour.20 The immune system comprises of innate and adaptive immunity.21 Innate immunity offers an immediate response utilising natural killer (NK) cells and macrophages to detect and destroy abnormal cells based on stress signals and altered cell surface molecules. In the case of cancer, innate immune cells train adaptive immune cells, including T lymphocytes, to specifically target tumour cells.21 Although immunotherapy has shown promise in cancers like melanoma and colorectal cancer,22 its success in brain cancer has been limited, underscoring the need for innovative therapeutic approaches. Circadian rhythms and the molecular clock herein offer an unexplored avenue.
Components of the circadian clock in the tumour microenvironment (TME) are important regulators of cancer cell stemness, metastasis and therapy resistance.23 This review explores the impact of the circadian clock on brain tumour formation, the brain TME, and immunosuppression. This approach includes optimising therapy timing through chronotherapy and exploring the molecular function of the circadian clock in brain cancer. Furthermore, we will examine how the circadian clock impacts the immune system and tumour immunology, highlighting its potential to enhance the efficacy of immunotherapy. We postulate that enhancing the understanding of the complex brain tumour biology and TME, with particular emphasis on the role of circadian clock, is essential for developing effective (immuno)therapies and boosting existing ones.
How does the molecular clock tick in the healthy central nervous system?
Oscillations of the circadian clock can be found in virtually all tissues, including glial cells, and have important functions in brain development and functionality.8 Given that most brain malignancies arise from glial cells or their precursors, it is of relevance to understand the role of the clock in their physiology.24 Therefore, in this section, the influence of the molecular clock on the functionality of various glial cell types will be described.
Astrocytes are the most abundant population of glial cells in the mammalian brain.25 These cells provide important metabolic and trophic support to neurons, which is crucial for nervous system health.26 For instance, perisynaptic astrocyte processes remove neurotransmitters such as glutamate from the interstitial space to prevent extra-synaptic accumulation and spillover to nearby synapses.27 Furthermore, astrocytes control the ionic balance at the synapse, sustaining proper synaptic transmission.25
Studies using in vitro cultures of mouse cortical astrocytes have shown that astrocytes exhibit rhythmic changes governed by clock genes.28 For example, oscillations of Clock, Per1, Per2, and IP3-dependent calcium signalling regulate the daily rhythms of ATP release in astrocytes.29 BMAL1 acts as a cell-autonomous regulator of astrocyte activation and neurotrophic function, while reduced levels of BMAL1 induce astrogliosis.30 In vitro experiments with primary mouse astrocytes have shown that Ror-α is upregulated during hypoxia, leading to downregulation of hypoxic inducible factor 1α.31 Furthermore, Clock, Per2, and Npas2 influence glutamate uptake levels, though without detectable circadian variation.32 Interestingly, astrocytes in the SCN of mice can modulate daily rhythms in the SCN and behaviour, and deletion of Bmal1 in astrocytes lengthened the circadian period of rest-activity rhythms.33 Thus, the core circadian clock has important roles in functioning of astrocytes.
Another cell type residing in the central nervous system (CNS) are the microglia. These macrophage-like immune cells remove dead cells in both the developing brain and the adult CNS, thus aiding normal brain development.34 Microglia also participate in synaptic development and maintenance. Like most cells, microglia possess a circadian clock that regulates their immune activity.35 In rats, for instance, a circadian rhythm of TNF-α, interleukin (IL)-1β and IL-6 expression peaks during the middle of the light phase (rest phase). Disruption of clock genes in microglia can induce chronic neuroinflammation and has been associated with the early onset of Alzheimer's Disease.36 The clock gene expression in microglia can be affected by obesogenic diets, leading to chronic activation of microglia in rats.37 The molecular clock is important in balancing the microglial phenotype and preventing neuroinflammation, with Rev-erbα playing a crucial role. Low levels of Rev-erbα increase synaptic phagocytosis, while the loss of Rev-erbα function results in spontaneous neuroinflammation and neuronal dysfunction.38,39
Lastly, oligodendrocytes, the third group of glial cells, are crucial for signal conduction in the CNS. These cells form a myelin sheath that facilitates rapid transmission of action potentials along the axon and provide metabolic support.40 Unlike Schwann cells, which myelinate single axons in the peripheral nervous system, oligodendrocytes can encapsulate multiple axons. They generate from oligodendrocyte progenitor cells (OPCs) through a strictly coordinated process of maturation, proliferation, and differentiation. Currently, there is no conclusive evidence that human oligodendrocytes have a functional molecular clock. Mouse studies however have shown rhythmic expression of Bmal1, Per2, and Rev-erbα.8 Deletion of Bmal1 revealed a strong correlation between its expression and OPC proliferation, cell-cycle regulation, and morphology. Moreover, oligodendrocyte-specific genes oscillate throughout the sleep-wake cycle in mice. Therefore, it is hypothesised that human oligodendrocytes contain a functional molecular clock.
Overall, there is mounting evidence indicating that the circadian clock influences crucial processes in glial cell functions. Therefore, disruptions in circadian rhythms can lead to various neurological consequences, which will be further discussed in the following section. Understanding their effects on glial cells can provide insights into maintaining CNS health and developing treatments for related disorders.
Pathological disruptions of the circadian clock in brain cancer development
The relationship between neurological diseases, such as Alzheimer's or Parkinson's Disease, and dysfunction of the circadian clock is well established.17 Additionally, circadian disruption, for instance by perturbations of normal physiological homeostasis due to jet lag or genetic mutations, have been identified as a risk factor for cancer development.16 Many cancers show disruption of the circadian clock.41 Moreover, there is an association between the loss of circadian control and poor efficacy of anticancer treatments, as well as early mortality among patients with cancer.16 Vice versa, an intact circadian clock inhibits proliferation and tumour growth in murine melanoma and colon carcinoma cells.42 Still, few studies have investigated the role of the circadian clock in brain cancer development and progression, and these studies primarily focus on glioma. Here, we will highlight key insights from studies on a variety of brain malignancies, broadening the understanding of circadian influences across different types of brain cancer.
Glioma
The most common group of brain tumours are glioma, comprising over 80% of all brain malignancies.43 These heterogeneous tumours are classified in five major groups: adult-type diffuse gliomas, paediatric-type diffuse low-grade gliomas, paediatric-type diffuse high-grade gliomas, circumscribed astrocytic gliomas, and ependymal tumours. Generally, a distinction is made between low-grade and high-grade gliomas based on malignancy. Interestingly, these two tumour classes exhibit contrasting expression levels of the core circadian clock genes. It should be noted that these transcriptomic analyses lack any information about rhythmicity, since the time of sample collection is not included as a factor in the analysis. Therefore, upregulation or downregulation of circadian clock proteins might in fact reflect increased or reduced amplitudes of rhythms. In patients with high-grade glioma, analysis of The Cancer Genome Atlas database reveals that BMAL1 is upregulated compared to patients with low-grade glioma.44 In contrast, combined genomic, transcriptomic, and clinical data analysis unveils a higher mortality in patients with glioma with downregulation of CLOCK. Changes in expression of the core clock proteins furthermore influence the internal timekeeping. Studies in a murine model of hypothalamic glioma show that gliomas induce alterations in the circadian clock.45 This effect is also observed in a Drosophila model, where glioblastoma extends the internal rhythm cycle.46
The core clock proteins directly influence glioma progression. For example, glioma stem cell (GSC)-derived CLOCK promotes angiogenesis by interaction with endothelial cells.47 GSCs remain a considerable challenge in the treatment of glioblastoma, since they are involved in chemo- and radio-resistance, and immunosuppression.48 Interestingly, targeting the circadian clock might improve glioma treatment as GSCs are uniquely sensitive to clock modulation. For instance, GSCs rely on a robust circadian rhythm and downregulation of either BMAL1 or CLOCK through genetic targeting or pharmacological treatment induces cell cycle arrest and apoptosis. Additionally, melatonin can attenuate key signals associated with GSC self-renewal by interfering with the EZH2-NOTCH1 signalling axis.49 While BMAL1 and CLOCK are vital for GSC survival, PER1, PER2, and PER3 are downregulated in high-grade gliomas.24,44 PER2 suppresses malignant characteristics including proliferation, stemness and invasion of GSCs in vitro and in vivo by regulating the Wnt/β-catenin and PTEN/AKT/Smad5/Id3 signalling pathways.50,51 In contrast, PER3 promotes astroblastoma progression through the P53/BCL2/BAX signalling pathway and high expression levels of PER3 are correlated with poor overall survival in patients with glioma.52 Beyond the altered clock gene expression, intact rhythms in the glioblastoma multiforme cell line T98G during proliferation result in a timed window of vulnerability for the chemotherapeutical agent bortezomib.53
Altogether, the exact role of clock genes in glioma remains to be addressed. While some studies show that a functional clock acts as a tumour suppressor,54, 55, 56 there are studies showing that GSCs rely on a robust rhythm.57 Also in other cancer types, like in a murine model of acute myeloid leukaemia, tumour cells depend on the circadian clock.58 Thus, it may be inferred that the role of the clock as a tumour suppressor is tumour type dependent. All the same, there is a strong body of evidence suggesting that altered expression levels of the core circadian clock genes are crucial for the progression of glioma.
Rare brain tumour entities
The scarcity of patients or tumour samples of more rare brain malignancies than glioma limits the ability to study the influence of these tumours on the circadian clock. Despite this, there are reports on circadian rhythm disruptions for these brain tumour entities. For example, of patients with untreated primary brain tumours, 59.2% experience insomnia, and 77.7% report poor sleep quality, pointing to circadian disruptions.59 For instance, patients with craniopharyngioma, a rare benign parasellar tumour, experience circadian rhythm sleep-wake disorders, which are typically caused by involvement of the SCN and alterations in melatonin transmission.60,61 In medulloblastoma, a brain malignancy occurring primarily in the paediatric population, bioinformatic analysis has revealed that pathways involved in the entrainment of the circadian clock by photoperiod are upregulated.62 Choroid plexus tumours exhibit hypermethylated promoter regions of PER2, correlating with significantly lower PER2 expression.63 In mice, this downregulation of PER2 is further associated with increased levels of Cyclin D and Cyclin E, as well as accelerated tumour growth in vivo. In patients with untreated pituitary tumours or meningioma, 46.2% and 81.8%, respectively, experience insomnia or poor sleep quality.64 Pituitary tumours can also cause a long-term disruption of the sleep-wake rhythms of patients by compressing the optic chiasm, which can also disrupt melatonin rhythms.65, 66, 67 In the same time window, namely in samples collected between 9:00 AM and 12:00 PM, both growth hormone-secreting pituitary adenoma (GHPA) and prolactin-secreting pituitary adenoma (PRLPA) exhibited upregulated PER2 expression, compared to autopsy specimens from deceased individuals.68 PER2 promotes pituitary tumourigenesis, and downregulation of PER2 by SR8278, a circadian nuclear receptor REV-ERBα antagonist, protected mice from developing pituitary adenoma.
Taken together, there are strong indications that different brain tumour entities exhibit altered expression of the molecular clock and influence circadian rhythms. There is a growing consensus that the processes driving tumourigenesis, known as ‘cancer hallmarks’, are tightly controlled by the circadian clock in normal cells. However, the role of the circadian clock is generally overlooked in cancer treatment.7 Therefore, the next section will delve deeper in the current treatment of brain cancer and explore how the circadian clock influences treatment outcomes.
Immunotherapy, checkpoints, and other treatments for brain cancer
Targeting the tumour
Spanning the different types of brain cancer, the main components of treatment in general include maximal safe surgical resection of the tumour, followed by radiotherapy and/or chemotherapy.18 Depending on the tumour grade, localisation, and patient age, treatment strategies and therapeutic agents can vary. Treatment outcomes also vary significantly between tumour types. Thus, while certain brain tumours may exhibit favourable prognoses, there remains an urgent demand for novel therapies for high grade brain tumours.
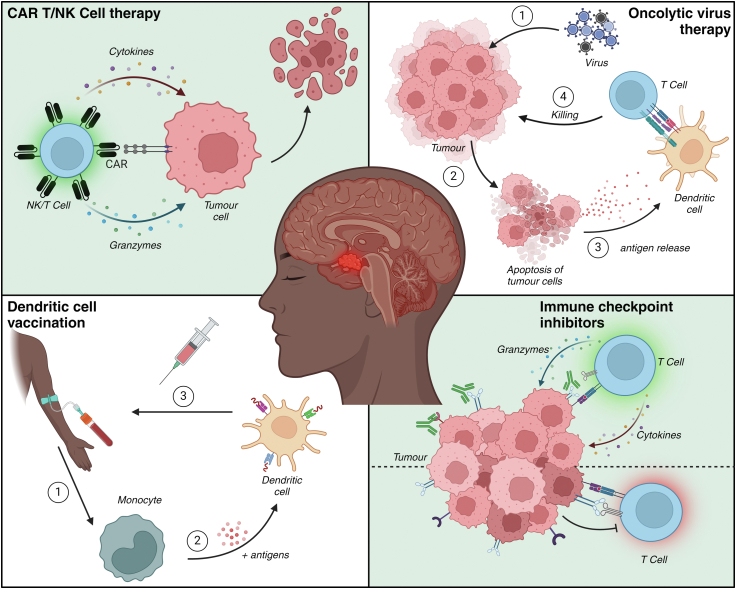
In recent years, immunotherapy has revolutionised the field of oncology. These therapies aim to activate or boost the immune system to attack and specifically target tumour cells.20 Immunotherapeutic strategies include, among others, immune checkpoint inhibitors (ICI), oncolytic viruses, vaccination, and adoptive cellular therapies such as chimeric antigen receptor (CAR)-T cell therapy (Fig. 2).
Fig. 2.
Immunotherapeutic strategies to target brain cancer. Several immunotherapeutic strategies have been developed over the past decades, aiming to generate an anti-tumour response from the immune system. Both oncolytic virus therapy and dendritic cell vaccine activate the adaptive part of the immune system via dendritic cells. Oncolytic virus therapy relies on in vivo activation of dendritic cells, whereas dendritic cell vaccine therapy involves training dendritic cells ex vivo from monocytes. In contrast, CAR T/NK cell therapy uses engineered receptors that enable these cells to precisely target and attack the tumour. Immune checkpoint inhibitor therapy does not focus on training the immune system but instead removes immunosuppression in the TME by blocking one or more inhibitory checkpoints. Made with BioRender. CAR: chimeric antigen receptor; TME, tumour microenvironment.
Immune checkpoints are receptor–ligand interactions between immune cells and their cellular environment, which can be either stimulatory or inhibitory.69 These checkpoints balance the immune response to pathogenic infections, thereby preventing excessive activation of immune system. Tumours can exploit the expression of inhibitory immune checkpoint proteins to evade immune surveillance. Immune checkpoint inhibition aims to alleviate the suppression of T cells by blocking the interaction between receptor, located in the membrane of the T cells, and ligand, expressed by the tumour. Monoclonal antibodies are the most commonly used tool for this type of immunotherapy. The blockade of the Programmed Death 1/Programmed Death Ligand 1 (PD-1/PD-L1) immune checkpoint is the most well-known and extensively studied example, demonstrating effectiveness against tumours such as melanoma, renal cell carcinoma and colorectal cancer.22 Glioma tumours typically express PD-L1, and higher tumour grades correlate with increased expression levels.70, 71, 72, 73 Several studies have trialled the safety and efficacy of blocking the PD-1/PD-L1 interaction in patients with glioma. A single-arm phase II clinical trial (NCT02550249) found that neoadjuvant anti-PD-1 immunotherapy significantly increased median survival time compared to adjuvant anti-PD-1 immunotherapy (417 vs 228.5 days, respectively).74 However, in unselected cohorts of patients with glioblastoma, immune checkpoint blockade has not yet improved clinical outcomes.75,76
Although immune checkpoint inhibition has proven to be successful in treating various cancers, the efficacy of antibodies is limited by poor tumour penetration.69 Adoptive cell transfer therapies offer an alternative. These therapies often utilise an antigen receptor specific for tumour antigens. To enhance the efficacy of CAR-T cell therapy in brain tumours, intracranial injection can be used to optimise delivery and surpass the requirement to pass the BBB.77 Clinical trials for glioblastoma treatment report the preliminary safety and activity of CAR-T cell therapies targeting specific antigens, EGFRvIII, HER2, and IL-13 receptor α2.78,79 Several Phase I and II clinical trials for treating medulloblastoma with either CAR-T or CAR-NK cells are ongoing.77 Nevertheless, CAR-T cell therapy is limited by the heterogeneous expression of target antigens by tumour cells, making it difficult to completely eliminate the tumour.80
As in CAR therapies, cancer vaccines target tumour antigens to boost anti-cancer immunity. Current therapeutic vaccines include dendritic cell vaccines, where monocytes are stimulated with tumour-associated antigens ex vivo, transforming them into dendritic cells.69 Upon reinjection into the patient, these cells stimulate and activate cytotoxic T lymphocytes. In the early 2000s, dendritic cell vaccines were already tested in patients with glioblastoma, and many studies showed increased survival. However, recent clinical trials have failed to replicate these results.77
Another form of immunotherapy uses oncolytic viruses, designed to selectively replicate in tumour cells, eventually causing their lysis.69 This leads to the release of tumour-associated antigens, stimulating anti-cancer immunity. In 2015, the first oncolytic virus therapy became available for melanoma. However, this type of therapy is limited by the intratumoural delivery method, which is particularly costly and difficult for malignant gliomas.69
Overall, immunotherapy is a promising type of cancer treatment, especially for brain cancers. Despite its successes, response rates can vary between patients due to, amongst others, the lack of immunogenic antigens or various immune resistance mechanisms.69 Immunoregulatory pathways and immune checkpoint expression can be influenced by disruption of circadian rhythmicity in tumour-resident cells to benefit the tumour.41 Therefore, future efforts should focus on overcoming the immunosuppression by the TME, as well as identifying new targets that act on important functional pathways of tumours.81
Targeting the clock
In glioblastoma, the circadian clock components are linked to an immunosuppressive TME. BMAL1 and CLOCK activity attracts immunosuppressive microglia, contributing to this environment.48 Additionally, the expression of PER2 suppresses processes like proliferation in glioma and choroid plexus tumours.24,44,63 Several efforts have been made to target these clock components in glioma. For example, REV-ERB agonists SR9009 and SR9011 have been shown to decrease stemness, survival, migration, and CLOCK expression in GSCs, while sparing healthy or differentiated glioblastoma cells.44,48,55 The synergetic effect of combining clock gene targeting with other therapies, as described in glioblastoma in vitro, likewise holds promise.82
Alternatively, reducing BMAL1 expression using CRY stabilisers such as KL001 or SHP656 has also shown potential in targeting GSCs.57 Interestingly, the efficacy of SR9009 and SR9011 in mouse models was comparable to that of temozolomide treatment in patient-derived xenograft glioblastoma models, while evincing reduced toxicity.7 These findings indicate that targeting the clock in glioma presents a promising prospective for treatment. To our knowledge, these compounds have not been tested on other brain cancer entities, however, given their impact on glioma cells, they might alter the TME in a way that enhances the efficacy of immunotherapeutic therapies.48 Illustrating the potential of targeting the clock is a pioneering study in a glioma mouse model, which demonstrates that combining REV-ERB agonists with anti-PD-1 treatment yields a synergistic effect, significantly extending survival.48 Forthcoming research ought to focus further on the interplay between circadian clock components and immunotherapy, revealing potential synergies in treatment.
Circadian orchestration of immune cell dynamics
Following circadian rhythms, the human immune system exhibits heightened sensitivity during daytime when environmental challenges are most apparent, while reducing the chance of detrimental immunity when challenge is diminished at night.83 This heightened sensitivity in the day is firstly illustrated by the migration of leukocytes to peripheral organs at the onset of the behavioural active phase.84 The time-of-day-dependent expression of migration factors on immune cells drives their specific localisation to tissues. For example, dendritic cells exhibit rhythmic migration into lymphatics, enforcing specific timepoints for efficient T cell activation.85,86 Furthermore, lymphocytes demonstrate time-dependent differences in infiltration into the tumour site, with peak infiltration occurring during the behavioural active phase in mice.87 Anti-tumour immunity is thus shaped by the prompt trafficking of immune cells during the active phase. In conjunction with diurnal oscillations in the permeability of the BBB, immunity within the brain is under strict circadian regulation.88,89
In keeping with localisation, immune cells show timed variations in function. This includes antigen processing, cytokine release and cytolytic factor expression, as well as diurnal oscillations in polarisation of T cell, and macrophage signatures.83,86,87,90 Co-stimulatory factor expression is also under circadian control. In dendritic cells oscillations in CD80 expression directly affects CD8+ T cell activation and therefore cancer immunosurveillance.91 Consequently, time may affect the efficacy of anti-cancer vaccines and feasibly other immunotherapies. A key factor herein is the rhythmic regulation of the immune component of the TME. In the brain, circadian oscillations in microglial inflammatory cytokine expression could affect local cancer immunity in a clock-controlled manner.35 The clock furthermore enforces diurnal changes in immune checkpoint expression on CD8+ T cells and myeloid-derived suppressor cells, thus timely affecting the efficacy of immune checkpoint inhibition and CAR T cell therapy in murine cancer model systems.87,92
Clinically, the oscillating expression of PD-1 on tumour-associated macrophages underlies the time-of-day dependent variation in ICI efficacy in patients with melanoma.93,94 ICI proves most effective when administered in the morning, thereby interfering during the time at which the macrophages most strongly inhibit T cell activation. The potential benefit of circadian targeting in cancer is studied in multiple clinical trials, of which some notable trials are portrayed in Table 1.
Table 1.
Clinical trials investigating the influence of circadian rhythms on cancer treatment.
| Trial ID/Registration | Title | Phase | Cancer type (stage) | Intervention(s) | Start date | Completion date | Study type |
|---|---|---|---|---|---|---|---|
| Breast cancer | |||||||
| NCT04864405 | Evaluating the Dose Timing (Morning vs Evening) of Endocrine Therapy and Its Effects on Tolerability and Compliance | 4 | Breast Cancer | Morning vs evening endocrine therapy | 30-06-2021 | 29-07-2023 | Interventional |
| NCT04401189 | The Role of Circadian Rhythms in Cancer-Related Symptoms (CHRONO) | N.A. | Breast Cancer | Circadian rhythms and CRS before vs after treatment (surgery/chemotherapy) | 01-06-2020 | 06-2022a | Observational |
| NCT03205033 | Melatonin as a Circadian Clock Regulator, Neuromodulator and Myelo-protector in Adjuvant Breast Cancer Chemotherapy | 2 | Breast cancer | Melatonin 7 days before until 21 days after chemotherapy | 01–2016 | 01–2017 | Interventional |
| NCT03217201 | Systematic Light Exposure for Fatigue in Breast Cancer Patients | N.A. | Breast cancer | Effect of SLE on CRF | 25-01-2018 | 26-1-2022 | Interventional |
| NCT02954809 | Effects of Bright Light Therapy on Fatigue, Sleep and Circadian Activity Rhythms in Lung Cancer Survivors | N.A. | Breast cancer survivors | Morning bright light therapy vs dim light. | 10-2016 | 05-2017a | Interventional |
| NCT06418139 | Association of Pembrolizumab Infusion Time and Efficacy in Patients With Non-metastatic Triple-negative Breast Cancer (TNBC) Treated With Neoadjuvant Chemotherapy and Immunotherapy (PEMCLOCK) | N.A. | Non-Metastatic Breast Carcinoma (TNBC) | N.A. | 05-2024a | 09-2026a | Observational |
| Colorectal cancer | |||||||
| NCT03955510 | Abnormal Food Timing and Circadian Dyssynchrony in Alcohol Induced Colon Carcinogenesis (AFT) | N.A. | Colorectal cancer | Effect of timing of food intake and alcohol use on susceptibility to colorectal cancer. | 31-07-2016 | 30-11-2024a | |
| NCT00852228 | Optimal Control of Liver Metastases From Colorectal Cancer With Cetuximab and Hepatic Artery Infusion of Chemotherapy (OPTILIV) | 2 | Liver metastases from colorectal cancer | Chronomodulated vs conventional chemotherapy | 07-2008 | 2015-12a | Interventional |
| Head and neck cancer | |||||||
| NCT05083416 | Effect of Prolonged Nightly Fasting on Immunotherapy Outcomes in HNSCC - Role of Gut Microbiome | Head and neck cancer | PNF vs no PNF effect on immunotherapy | 20-10-2021 | 24-05-2024a | Interventional | |
| NCT03196869 | the Study of Effect of Chronomodulated Chemotherapy on the Dendritic Cells Subsets in the Treatment of Advanced Nasopharyngeal Cancer | 2 | Locally Advanced HNSCC | Chrono-chemotherapy vs routine chemotherapy | 7-4-2014 | 12-8-2022a | Interventional |
| Lung cancer | |||||||
| NCT05637580 | Pathological Tumor and Lymph Node Responses After Neoadjuvant Immunochemotherapy in Initially-unresectable NSCLC | N.A. | NSCLC | N.A. | Observational | ||
| NCT04650490 | SRS Timing With Immune Checkpoint Inhibition in Patients With Untreated Brain Metastases From Non-small Cell Lung Cancer (STICk-IM-NSCLC) | 2 | NSCLC with brain metastasis | Timing of stereotactic radiosurgery relative to immunotherapy | 03-2023a | 03-2026a | Interventional |
| NCT05988970 | Impact of Circadian Rhythm on the Spread of Circulating Tumor Cells in Lung Cancer Patients | N.A. | Lung cancer | CTC before and after treatment | 29-1-2024 | 09-2025a | Interventional |
| Brain cancer | |||||||
| NCT04669574 | Assessing Sleep and Circadian Rhythms in Primary Brain Tumors Patients | N.A. | Primary brain tumour | Detect sleep disturbances in patients. | 29-06-2021 | 01-01-2025a | Observational |
| NCT02781792 | Temozolomide Chronotherapy for High Grade Glioma | 2 | High grade glioma (II-IV) | TMZ morning vs evening administration | 11-08-2016 | 14-07-2024 | Interventional |
| Other | |||||||
| NCT04827446 | Lighting Intervention for Cancer-related Fatigue | N.A. | Breast and prostate cancer, HSCT receivers. | Effect of light levels on cancer related fatigue. | 15-7-2021 | 09-03-2023 | Interventional |
| NCT02937519 | Chronomodulated Chemotherapy Followed by Concurrent Chemo-radiotherapy With IMRT in the Treatment of Advanced Nasopharyngeal Cancer | 2 | Oesophageal Neoplasms | Chrono-chemotherapy vs routine chemotherapy | 06-2015 | 06-2018a | Interventional |
| NCT05737732 | The Ambient Light Multiple Myeloma Study | N.A. | Multiple myeloma | Effect circadian effective vs ineffective lighting on patients receiving ASCT | 13-02-2023 | 30-06-2027a | Interventional |
| NCT02187315 | Induction Chemotherapy Followed By Chrono-chemotherapy Concurrent With IMRT Of Locally Advanced NPC Clinical Study | 2 | Nasopharyngeal carcinoma | Chrono-chemotherapy vs routine chemotherapy | 05-2014 | 12-2019a | Interventional |
| NCT05511740 | Circadian as A Prognostic Factor For Radiation Response in Cervical Cancer | N.A. | Cervical cancer | Morning vs evening radiation therapy | 01-2010 | 07-2014 | Interventional |
ASCT: autologous stem cell transplant; CRF: cancer related fatigue; CRS: cancer-treatment related symptoms; CTC: circulating tumour cells; HNSCC: head and neck squamous cell carcinoma; HSCT: hematopoietic stem cell transplant; NSCLC: non-small cell lung carcinoma; PBT: primary brain tumour; PNF: prolonged nightly fasting; SLE: systematic light exposure; TMZ: temozolomide; TNBC: triple-negative breast cancer.
Expected completion date.
Beyond identifying pathways worth exploring for time-specific targeting, the role of the molecular clock pathway itself has gained interest in the context of tumour immunity. While the immune system throughout the body adheres to a circadian rhythm, within tumours the clock genes can exert different functions. Clock gene-enriched pathways are directly linked to immune evasion in various cancers.90 Disruption of circadian rhythmicity in tumour-resident cells can influence immunoregulatory pathways and immune checkpoint expression to the cancer's advantage, marking the significance of the molecular clock in cancer immunity.41 In glioblastoma multiforme, CLOCK expression is correlated with higher microglial presence and reduced activation of CD8+ T cells in a non-diurnal manner.48 Likewise, the upregulation of chemoattractant olfactomedin-like 3 (OLFML3) by BMAL1 and CLOCK facilitates the migration of immunosuppressive microglia into the glioblastoma TME.95 Blockade of the CLOCK-OLFML3 pathway is currently being studied as a potential target for GBM.96 Understanding of the circadian orchestration of tumour-immune cell dynamics in brain cancer paves the way for improved chronotherapeutic strategies in immunotherapy.
Future perspectives for brain cancer treatment
The timing of treatments or surgical interventions affects outcomes and side effects
It has become apparent that timing of treatments or interventions can affect the outcomes of various pathologies. For instance, timing antihypertensive drugs to circadian rhythms can prevent the morning peak in cardiovascular disease incidence, and scheduling aortic valve replacement surgeries in the afternoon is associated with a reduced incidence of major adverse cardiac events.97 Moreover, increasing evidence shows that chrono-modulation of chemotherapy reduces toxicity without compromising efficacy.98 In the case of glioma, a recently published systematic review describes that temozolomide cytotoxicity is highest during the peak of BMAL1 expression, which occurs in the morning.99 Another study showed that daily rhythms of O6-Methylguanine-DNA Methyltransferase (MGMT) expression determine the phase-dependent sensitivity of glioblastoma cells to temozolomide.100 The methylation status of MGMT has a strong prognostic value for treatment with temozolomide. Therefore, the rhythmicity of the MGMT methylation should be considered when analysing biopsies, to further enhance the prognostic value of temozolomide treatment.100 Next to drug toxicity and prognostic value, therapy efficacy is apt to be controlled temporally, and tumour growth rates can be impacted differently based on the timing of treatment administration.54 Although more clinical data is needed to solidify these findings, adjusting the timing of the treatment is seemingly a feasible and easily implemented strategy. It can be argued that aligning immunotherapy to circadian rhythms might similarly benefit the outcomes of brain cancer treatment. Therefore, the next section further elaborates on this opportunity.
Chrono-immunotherapy and circadian principles for development of new and improved treatments for brain cancer
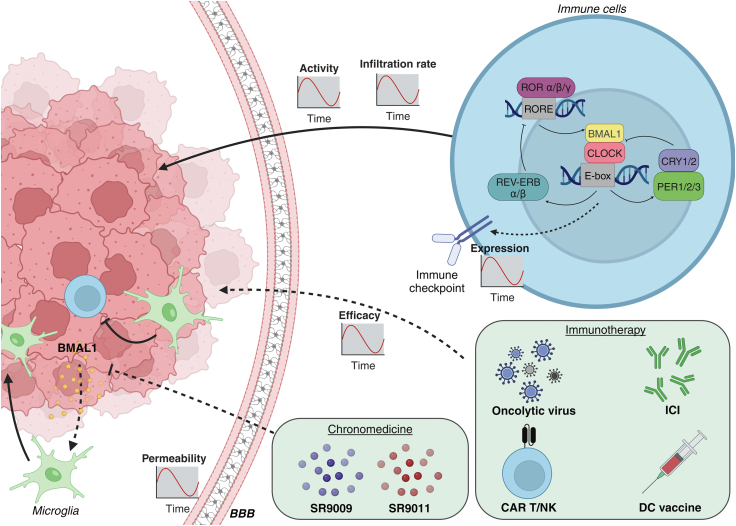
Circadian cues modulate immunity significantly dependent on the time of day. The broad impact of circadian rhythms in brain cancer is embodied by the oscillating infiltration and function of immune cells in the TME, the synchronised permeability of the BBB and thus accessibility of brain tumours for therapy, as well as a non-diurnal role for molecular clock genes on cancer immunity (Fig. 3). How these factors coalesce will unveil a more fundamental understanding of brain malignancies and will uncover the potential benefit of chronotherapy as well as targeting of the clock-immunomolecular circuitry (Table 2).
Fig. 3.
Integration of circadian rhythms and immunotherapy for enhanced treatment of brain tumours. Circadian rhythms dictate various processes that affect the efficacy of brain cancer treatment. The immune cells exhibit circadian changes in activity, sensitivity, immune checkpoint expression, and infiltration rates. Similarly, circadian rhythms dictate the permeability of the BBB. On the other hand, the malfunctioning of the circadian clock in brain cancer boosts cancer progression. Simultaneously, these changes can attract immunosuppressive microglia, further shaping an immunosuppressive TME. Taken together, circadian rhythms may provide an optimal time-window for administering chronomedicine, targeting the clock, or immunotherapeutic interventions. Made with BioRender. BBB: blood-brain barrier; CAR: chimeric antigen receptor; DC: dendritic cell; ICI: immune checkpoint inhibitors; NK: natural killer; RORE: ROR response element; TME: tumour microenvironment.
Table 2.
The brain tumour's time table.
| Circadian modulation of … | Cross-cancer discoveries | Pending brain cancer queries |
|---|---|---|
| Brain cell activity |
|
|
| Tumour cell phenotype |
|
|
| Blood-brain barrier (BBB) |
|
|
| Tumour-immune microenvironment (TIME) |
|
In terms of chronotherapy, synchronising treatment with circadian rhythms has the potential to enhance immunotherapy efficacy. To this and the patients' advantage, patients’ sleep-wake cycles and the entrainment of the clocks (e.g., light/dark cycles, food intake timing, influence of electronic devices) should be guarded. The benefit of administration of immunotherapy at specific times throughout the day can be studied quite inexpensively in the clinic. This would arguably be most beneficial in tumours that exhibit an intact circadian rhythm, such as in glioblastoma, as these tumours may display timed vulnerability to cancer immunity.57 Investigating the integrity of the circadian clock in different forms of brain cancers might elucidate timed variation in susceptibility. This is relevant for all cell types residing in the TME, as their timed variation in immunosuppressive qualities jointly contributes to the timeframe of vulnerability of the tumour to immunity. Within the current understanding, targeting would be most favourable at a time when the BBB is most permeable, immune effector cells readily infiltrate the tumour site, and tumour cells themselves present minimal resistance. The circadian immune status of, e.g., microglia and tumour associated macrophages furthermore affects both effector cells and tumour cells in a timely manner. Delving into the circadian rhythms of the brain TME promises to unveil the optimal timing for administering immunotherapy.
Still, circadian rhythmicity is disturbed in many cancers.41 This sets the stage for more precise targeting of the molecular clock. Since disruption of the clock is connected to immune evasion, its restoration possibly relieves this effect to improve immunotherapy outcomes. However, selective restoration may prove complicated by the ubiquitous expression of the clock genes. It is therefore imperative to unravel targets in the clock gene - immunoregulatory pathway interaction. This calls for a more fundamental study of the pathways by which molecular clock genes affect immunosuppression in brain cancer and the TME. Furthermore, combining immunotherapy with the targeting of the clock could ultimately increase the potency of immunotherapy in brain cancer. Thus, integrating circadian rhythms, the molecular clock, and immunotherapy can greatly benefit brain cancer treatment.
Conclusion
Circadian rhythms intricately govern numerous cellular processes. While in healthy cells circadian rhythms orchestrate activity in a controlled manner, in cancer, alterations in molecular clock expression often fuel tumour progression by fostering the survival and proliferation of malignant cells. These changes further bolster the tumour's ability to establish an immunosuppressive TME. Although various immunotherapeutic therapies have been tested for brain cancer, their success remains limited. A pivotal determinant of immunotherapy outcomes is the circadian rhythm governing the immune system and TME. Strategically targeting and harnessing the circadian clock for brain cancer treatment has the potential to revolutionise therapeutic approaches and significantly enhance the efficacy of immunotherapy.
Outstanding questions
Knowledge on the role of the circadian clock in brain cancers remains largely limited to glioma. Enhancing our understanding of the circadian clock in all forms of brain cancer is essential for improving treatment. While chronotherapy for glioma is still in its infancy, current studies are paving the way for future advancements in brain cancer therapy. With the increased availability of Whole Genome or Whole Exome Sequencing data (WGS/WES), larger cohorts of patient data are emerging, providing a valuable resource for research. Since the circadian clock causes changes in expression throughout the 24-h cycle, the collection of data might mask rhythmic changes. Therefore, we recommend that sequencing data should include timestamps for the time of collection to enable bioinformatic analyses based on time. This will provide crucial information for the further development of chronotherapy and immunotherapy for brain cancer. Furthermore, the profound influence of circadian rhythms in the immune system as well as the TME urges further research into these mechanisms in brain cancer. Incorporating the dimension of time into clinical decision-making for immunotherapy can ultimately enhance therapy outcome, making circadian rhythms a critical factor in the design and investigation of brain cancer treatments.
Contributors
MQ, MvO and SC contributed to the concept and design of this review. MQ and MvO performed the literature search and reviewed and selected the papers for inclusion. MQ, MvO and SC wrote and edited the manuscript. MQ and MvO created the figures. SC, LWvL and NB critically revised the manuscript, the figures and tables. All authors read and approved the final version of the manuscript.
Declaration of interests
MQ, MvO, SC, NB: None. LWvL: Outside the current work: consultancy fees to UMCU from Abbott, Medtronic, Vifor, Novartis.
Acknowledgements
LWvL is supported by the Netherlands Heart Foundation, Dekker Senior Clinical Scientist 2019, grant agreement No 2019T056. NB received funding from Foundation Koppie-AU, Foundation Team Doelbewust, Health Holland/TKI, and the Dutch Research Council. The funders had no role in review design, data collection, data analysis, interpretation, or writing.
References
- 1.Zehring W.A., Wheeler D.A., Reddy P., et al. P-element transformation with period locus DNA restores rhythmicity to mutant, arrhythmic Drosophila melanogaster. Cell. 1984;39:369–376. doi: 10.1016/0092-8674(84)90015-1. [DOI] [PubMed] [Google Scholar]
- 2.Siwicki K.K., Eastman C., Petersen G., Rosbash M., Hall J.C. Antibodies to the period gene product of Drosophila reveal diverse tissue distribution and rhythmic changes in the visual system. Neuron. 1988;1:141–150. doi: 10.1016/0896-6273(88)90198-5. [DOI] [PubMed] [Google Scholar]
- 3.Hardin P.E., Hall J.C., Rosbash M. Feedback of the Drosophila period gene product on circadian cycling of its messenger RNA levels. Nature. 1990;343:536–540. doi: 10.1038/343536a0. [DOI] [PubMed] [Google Scholar]
- 4.Bargiello T.A., Jackson F.R., Young M.W. Restoration of circadian behavioural rhythms by gene transfer in Drosophila. Nature. 1984;312:752–754. doi: 10.1038/312752a0. [DOI] [PubMed] [Google Scholar]
- 5.Welsh D.K., Takahashi J.S., Kay S.A. Suprachiasmatic nucleus: cell autonomy and network properties. Annu Rev Physiol. 2010;72:551–577. doi: 10.1146/annurev-physiol-021909-135919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Costello H.M., Gumz M.L. Circadian rhythm, clock genes and hypertension: recent advances in hypertension. Hypertension. 2021;78:1185. doi: 10.1161/HYPERTENSIONAHA.121.14519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sulli G., Rommel A., Wang X., et al. Pharmacological activation of REV-ERBs is lethal in cancer and oncogene induced senescence. Nature. 2018;553:351. doi: 10.1038/nature25170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rojo D., Badner A., Gibson E.M. Circadian control of glial cell homeodynamics. J Biol Rhythms. 2022;37:593. doi: 10.1177/07487304221120966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hastings M.H., Brancaccio M., Gonzalez-Aponte M.F., Herzog E.D. Circadian rhythms and astrocytes: the good, the bad, and the ugly. Annu Rev Neurosci. 2023;46:123–143. doi: 10.1146/annurev-neuro-100322-112249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Reddy S., Reddy V., Sharma S. StatPearls. Treasure Island (FL) StatPearls Publishing; 2024. Physiology, circadian rhythm. [PubMed] [Google Scholar]
- 11.Stenvers D.J., Scheer F.A.J.L., Schrauwen P., la Fleur S.E., Kalsbeek A. Circadian clocks and insulin resistance. Nat Rev Endocrinol. 2018;15(2):75–89. doi: 10.1038/s41574-018-0122-1. [DOI] [PubMed] [Google Scholar]
- 12.Firsov D., Bonny O. Circadian rhythms and the kidney. Nat Rev Nephrol. 2018;14(10):626–635. doi: 10.1038/s41581-018-0048-9. [DOI] [PubMed] [Google Scholar]
- 13.Vitaterna M.H., Takahashi J.S., Turek F.W. Overview of circadian rhythms. Alcohol Res Health. 2001;25:85. [PMC free article] [PubMed] [Google Scholar]
- 14.Patke A., Young M.W., Axelrod S. Molecular mechanisms and physiological importance of circadian rhythms. Nat Rev Mol Cell Biol. 2019;21(2):67–84. doi: 10.1038/s41580-019-0179-2. [DOI] [PubMed] [Google Scholar]
- 15.Musiek E.S., Holtzman D.M. Mechanisms linking circadian clocks, sleep, and neurodegeneration. Science. 2016;354:1004. doi: 10.1126/science.aah4968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shafi A.A., Knudsen K.E. Cancer and the circadian clock. Cancer Res. 2019;79:3806. doi: 10.1158/0008-5472.CAN-19-0566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Leng Y., Musiek E.S., Hu K., Cappuccio F.P., Yaffe K. Association between circadian rhythms and neurodegenerative diseases. Lancet Neurol. 2019;18:307–318. doi: 10.1016/S1474-4422(18)30461-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.van den Bent M.J., Geurts M., French P.J., et al. Primary brain tumours in adults. Lancet. 2023;402:1564–1579. doi: 10.1016/S0140-6736(23)01054-1. [DOI] [PubMed] [Google Scholar]
- 19.Quader S., Kataoka K., Cabral H. Nanomedicine for brain cancer. Adv Drug Deliv Rev. 2022;182 doi: 10.1016/j.addr.2022.114115. [DOI] [PubMed] [Google Scholar]
- 20.Riley R.S., June C.H., Langer R., Mitchell M.J. Delivery technologies for cancer immunotherapy. Nat Rev Drug Discov. 2018;18(3):175–196. doi: 10.1038/s41573-018-0006-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Maiorino L., Daßler-Plenker J., Sun L., Egeblad M. Innate immunity and cancer pathophysiology. Annu Rev Pathol. 2022;17:425. doi: 10.1146/annurev-pathmechdis-032221-115501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lei Q., Wang D., Sun K., Wang L., Zhang Y. Resistance mechanisms of anti-PD1/PDL1 therapy in solid tumors. Front Cell Dev Biol. 2020;8 doi: 10.3389/fcell.2020.00672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xuan W., Khan F., James C.D., Heimberger A.B., Lesniak M.S., Chen P. Circadian regulation of cancer cell and tumor microenvironment crosstalk. Trends Cell Biol. 2021;31:940–950. doi: 10.1016/j.tcb.2021.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Arafa K., Emara M. Insights about circadian clock and molecular pathogenesis in gliomas. Front Oncol. 2020;10:199. doi: 10.3389/fonc.2020.00199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Allen N.J., Eroglu C. Cell biology of astrocyte-synapse interactions. Neuron. 2017;96:697–708. doi: 10.1016/j.neuron.2017.09.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Banker G.A. Trophic interactions between astroglial cells and hippocampal neurons in culture. Science. 1980;209:809–810. doi: 10.1126/science.7403847. [DOI] [PubMed] [Google Scholar]
- 27.Murphy-Royal C., Dupuis J., Groc L., Oliet S.H.R. Astroglial glutamate transporters in the brain: regulating neurotransmitter homeostasis and synaptic transmission. J Neurosci Res. 2017;95:2140–2151. doi: 10.1002/jnr.24029. [DOI] [PubMed] [Google Scholar]
- 28.Prolo L.M., Takahashi J.S., Herzog E.D. Circadian rhythm generation and entrainment in astrocytes. J Neurosci. 2005;25:404. doi: 10.1523/JNEUROSCI.4133-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Marpegan L., Swanstrom A.E., Chung K., et al. Circadian regulation of ATP release in astrocytes. J Neurosci. 2011;31:8342. doi: 10.1523/JNEUROSCI.6537-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lananna B.V., Nadarajah C.J., Izumo M., et al. Cell-autonomous regulation of astrocyte activation by the circadian clock protein BMAL1. Cell Rep. 2018;25:1. doi: 10.1016/j.celrep.2018.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jolly S., Journiac N., Naudet F., Gautheron V., Mariani J., Vernet-der Garabedia B. Cell-autonomous and non-cell-autonomous neuroprotective functions of RORα in neurons and astrocytes during hypoxia. J Neurosci. 2011;31:14314–14323. doi: 10.1523/JNEUROSCI.1443-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Beaulé C., Swanstrom A., Leone M.J., Herzog E.D. Circadian modulation of gene expression, but not glutamate uptake, in mouse and rat cortical astrocytes. PLoS One. 2009;4 doi: 10.1371/journal.pone.0007476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tso C.F., Simon T., Greenlaw A.C., Puri T., Mieda M., Herzog E.D. Astrocytes regulate daily rhythms in the suprachiasmatic nucleus and behavior. Curr Biol. 2017;27:1055. doi: 10.1016/j.cub.2017.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Prinz M., Jung S., Priller J. Microglia biology: one century of evolving concepts. Cell. 2019;179:292–311. doi: 10.1016/j.cell.2019.08.053. [DOI] [PubMed] [Google Scholar]
- 35.Fonken L.K., Frank M.G., Kitt M.M., Barrientos R.M., Watkins L.R., Maier S.F. Microglia inflammatory responses are controlled by an intrinsic circadian clock. Brain Behav Immun. 2015;45:171. doi: 10.1016/j.bbi.2014.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ni J., Wu Z., Meng J., et al. An impaired intrinsic microglial clock system induces neuroinflammatory alterations in the early stage of amyloid precursor protein knock-in mouse brain. J Neuroinflammation. 2019;16:1–15. doi: 10.1186/s12974-019-1562-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Milanova I.V., Kalsbeek M.J.T., Wang X.L., et al. Diet-induced obesity disturbs microglial immunometabolism in a time-of-day manner. Front Endocrinol. 2019;10:424. doi: 10.3389/fendo.2019.00424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Griffin P., Dimitry J.M., Sheehan P.W., et al. Circadian clock protein Rev-erbα regulates neuroinflammation. Proc Natl Acad Sci U S A. 2019;116:5102–5107. doi: 10.1073/pnas.1812405116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Griffin P., Sheehan P.W., Dimitry J.M., et al. REV-ERBα mediates complement expression and diurnal regulation of microglial synaptic phagocytosis. Elife. 2020;9:1–17. doi: 10.7554/eLife.58765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kuhn S., Gritti L., Crooks D., Dombrowski Y. Oligodendrocytes in development, myelin generation and beyond. Cells. 2019;8:1424. doi: 10.3390/cells8111424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wu Y., Tao B., Zhang T., Fan Y., Mao R. Pan-cancer analysis reveals disrupted circadian clock associates with T cell exhaustion. Front Immunol. 2019;10:2451. doi: 10.3389/fimmu.2019.02451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kiessling S., Cermakian N. The tumor circadian clock: a new target for cancer therapy? Future Oncol. 2017;13:2607–2610. doi: 10.2217/fon-2017-0456. [DOI] [PubMed] [Google Scholar]
- 43.Weller M., Wen P.Y., Chang S.M., et al. Glioma. Nat Rev Dis Primers. 2024;10:1–23. doi: 10.1038/s41572-024-00516-y. [DOI] [PubMed] [Google Scholar]
- 44.Petković M., Henis M., Heese O., Relógio A. Chronotherapy in Glioblastoma: state of the art and future perspectives. eBioMedicine. 2023;89 doi: 10.1016/j.ebiom.2023.104470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Duhart J.M., Brocardo L., Caldart C.S., Marpegan L., Golombek D.A. Circadian alterations in a murine model of hypothalamic glioma. Front Physiol. 2017;8:864. doi: 10.3389/fphys.2017.00864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jarabo P., de Pablo C., González-Blanco A., Casas-Tintó S. Circadian gene cry controls tumorigenesis through modulation of myc accumulation in glioblastoma cells. Int J Mol Sci. 2022;23:2043. doi: 10.3390/ijms23042043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pang L., Dunterman M., Xuan W., et al. Circadian regulator CLOCK promotes tumor angiogenesis in glioblastoma. Cell Rep. 2023;42 doi: 10.1016/j.celrep.2023.112127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chan P., Rich J.N., Kay S.A. Watching the clock in glioblastoma. Neuro Oncol. 2023;25:1932–1946. doi: 10.1093/neuonc/noad107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Maitra S., Bhattacharya D., Das S., Bhattacharya S. Melatonin and its anti-glioma functions: a comprehensive review. Rev Neurosci. 2019;30:527–541. doi: 10.1515/revneuro-2018-0041. [DOI] [PubMed] [Google Scholar]
- 50.Ma D., Hou L., Xia H., et al. PER2 inhibits proliferation and stemness of glioma stem cells via the Wnt/β-catenin signaling pathway. Oncol Rep. 2020;44:533. doi: 10.3892/or.2020.7624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhang Y., Hu X., Li H., et al. Circadian Period 2 (Per2) downregulate inhibitor of differentiation 3 (Id3) expression via PTEN/AKT/Smad5 axis to inhibits glioma cell proliferation. Bioengineered. 2022;13 doi: 10.1080/21655979.2022.2074107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wang Q., Liu H., Wang Z., et al. Circadian gene Per3 promotes astroblastoma progression through the P53/BCL2/BAX signalling pathway. Gene. 2024;895 doi: 10.1016/j.gene.2023.147978. [DOI] [PubMed] [Google Scholar]
- 53.Wagner P.M., Sosa Alderete L.G., Gorné L.D., et al. Proliferative glioblastoma cancer cells exhibit persisting temporal control of metabolism and display differential temporal drug susceptibility in chemotherapy. Mol Neurobiol. 2019;56:1276–1292. doi: 10.1007/s12035-018-1152-3. [DOI] [PubMed] [Google Scholar]
- 54.Wagner P.M., Prucca C.G., Velazquez F.N., Sosa Alderete L.G., Caputto B.L., Guido M.E. Temporal regulation of tumor growth in nocturnal mammals: in vivo studies and chemotherapeutical potential. FASEB J. 2021;35:e21231. doi: 10.1096/fj.202001753R. [DOI] [PubMed] [Google Scholar]
- 55.Wagner P.M., Monjes N.M., Guido M.E. Chemotherapeutic effect of SR9009, a REV-ERB agonist, on the human glioblastoma T98G cells. ASN Neuro. 2019;11 doi: 10.1177/1759091419892713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Trebucq L.L., Salvatore N., Wagner P.M., Golombek D.A., Chiesa J.J. Circadian clock gene bmal1 acts as a tumor suppressor gene in a mice model of human glioblastoma. Mol Neurobiol. 2024;61:5216–5229. doi: 10.1007/s12035-023-03895-7. [DOI] [PubMed] [Google Scholar]
- 57.Dong Z., Zhang G., Qu M., et al. Targeting glioblastoma stem cells through disruption of the circadian clock. Cancer Discov. 2019;9:1556. doi: 10.1158/2159-8290.CD-19-0215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Puram R.V., Kowalczyk M.S., De Boer C.G., et al. Core circadian clock genes regulate leukemia stem cells in AML. Cell. 2016;165:303–316. doi: 10.1016/j.cell.2016.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Lin P.C., Chen P.Y., Wei K.C., et al. Sleep disturbance in adults with untreated primary brain tumors: prevalence and impact on quality of life. Sleep Biol Rhythms. 2023;21:201. doi: 10.1007/s41105-022-00436-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Romigi A., Feola T., Cappellano S., et al. Sleep disorders in patients with craniopharyngioma: a physiopathological and practical update. Front Neurol. 2022;12 doi: 10.3389/fneur.2021.817257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cordani R., Veneruso M., Napoli F., et al. Sleep disturbances in craniopharyngioma: a challenging diagnosis. J Neurol. 2021;268:4362. doi: 10.1007/s00415-021-10794-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Huang P., Guo Y.D., Zhang H.W. Identification of hub genes in pediatric medulloblastoma by multiple-microarray analysis. J Mol Neurosci. 2020;70:522–531. doi: 10.1007/s12031-019-01451-4. [DOI] [PubMed] [Google Scholar]
- 63.Pienkowska M., Choufani S., Turinsky A.L., et al. DNA methylation signature is prognostic of choroid plexus tumor aggressiveness. Clin Epigenetics. 2019;11:117. doi: 10.1186/s13148-019-0708-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lin M.R., Chen P.Y., Wang H.C., Lin P.C., Lee H.C., Chiu H.Y. Prevalence of sleep disturbances and their effects on quality of life in adults with untreated pituitary tumor and meningioma. J Neuro Oncol. 2021;154:179–186. doi: 10.1007/s11060-021-03811-w. [DOI] [PubMed] [Google Scholar]
- 65.Borodkin K., Ayalon L., Kanety H., Dagan Y. Dysregulation of circadian rhythms following prolactin-secreting pituitary microadenoma. Chronobiol Int. 2005;22:145–156. doi: 10.1081/cbi-200038153. [DOI] [PubMed] [Google Scholar]
- 66.Boertien T.M., Van Someren E.J.W., Coumou A.D., et al. Compression of the optic chiasm is associated with reduced photoentrainment of the central biological clock. Eur J Endocrinol. 2022;187:809–821. doi: 10.1530/EJE-22-0527. [DOI] [PubMed] [Google Scholar]
- 67.Zielonka D., Sowiński J., Nowak S., Ciesielska A., Moskal J., Marcinkowski J.T. Melatonin and cortisol profiles in patients with pituitary tumors. Neurol Neurochir Pol. 2015;49:65–69. doi: 10.1016/j.pjnns.2014.12.004. [DOI] [PubMed] [Google Scholar]
- 68.Guo L., Cen H., Weng J., et al. PER2 integrates circadian disruption and pituitary tumorigenesis. Theranostics. 2023;13:2657. doi: 10.7150/thno.82995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Liu C., Yang M., Zhang D., Chen M., Zhu D. Clinical cancer immunotherapy: current progress and prospects. Front Immunol. 2022;13:961805. doi: 10.3389/fimmu.2022.961805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wintterle S., Schreiner B., Mitsdoerffer M., et al. Expression of the B7-related molecule B7-H1 by glioma cells: a potential mechanism of immune paralysis. Cancer Res. 2003;63:7462–7467. [PubMed] [Google Scholar]
- 71.Wilmotte R., Burkhardt K., Kindler V., et al. B7-homolog 1 expression by human glioma: a new mechanism of immune evasion. Neuroreport. 2005;16:1081–1085. doi: 10.1097/00001756-200507130-00010. [DOI] [PubMed] [Google Scholar]
- 72.Berghoff A.S., Kiesel B., Widhalm G., et al. Editor's choice: Programmed death ligand 1 expression and tumor-infiltrating lymphocytes in glioblastoma. Neuro Oncol. 2015;17:1064–1075. doi: 10.1093/neuonc/nou307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Baral A., Ye H.X., Jiang P.C., Yao Y., Mao Y. B7-H3 and B7-H1 expression in cerebral spinal fluid and tumor tissue correlates with the malignancy grade of glioma patients. Oncol Lett. 2014;8:1195–1201. doi: 10.3892/ol.2014.2268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Cloughesy T.F., Mochizuki A.Y., Orpilla J.R., et al. Neoadjuvant anti-PD-1 immunotherapy promotes a survival benefit with intratumoral and systemic immune responses in recurrent glioblastoma. Nat Med. 2019;25:477–486. doi: 10.1038/s41591-018-0337-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Arrieta V.A., Dmello C., McGrail D.J., et al. Immune checkpoint blockade in glioblastoma: from tumor heterogeneity to personalized treatment. J Clin Invest. 2023;133 doi: 10.1172/JCI163447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Zeng Y.F., Wei X.Y., Guo Q.H., et al. The efficacy and safety of anti-PD-1/PD-L1 in treatment of glioma: a single-arm meta-analysis. Front Immunol. 2023;14 doi: 10.3389/fimmu.2023.1168244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Schakelaar M.Y., Monnikhof M., Crnko S., et al. Cellular immunotherapy for medulloblastoma. Neuro Oncol. 2023;25:617. doi: 10.1093/neuonc/noac236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Bagley S.J., Logun M., Fraietta J.A., et al. Intrathecal bivalent CAR T cells targeting EGFR and IL13Rα2 in recurrent glioblastoma: phase 1 trial interim results. Nat Med. 2024;30(5):1320–1329. doi: 10.1038/s41591-024-02893-z. [DOI] [PubMed] [Google Scholar]
- 79.Choi B.D., Gerstner E.R., Frigault M.J., et al. Intraventricular CARv3-TEAM-E T cells in recurrent glioblastoma. N Engl J Med. 2024;390:1290–1298. doi: 10.1056/NEJMoa2314390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Sampson J.H., Gunn M.D., Fecci P.E., Ashley D.M. Brain immunology and immunotherapy in brain tumours. Nat Rev Cancer. 2020;20:12. doi: 10.1038/s41568-019-0224-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Tan S., Li D., Zhu X. Cancer immunotherapy: Pros, cons and beyond. Biomed Pharmacother. 2020;124 doi: 10.1016/j.biopha.2020.109821. [DOI] [PubMed] [Google Scholar]
- 82.Wagner P.M., Fornasier S.J., Guido M.E. Pharmacological modulation of the cytosolic oscillator affects glioblastoma cell biology. Cell Mol Neurobiol. 2024;44:51. doi: 10.1007/s10571-024-01485-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Scheiermann C., Gibbs J., Ince L., Loudon A. Clocking in to immunity. Nat Rev Immunol. 2018;18(7):423–437. doi: 10.1038/s41577-018-0008-4. [DOI] [PubMed] [Google Scholar]
- 84.He W., Holtkamp S., Hergenhan S.M., et al. Circadian expression of migratory factors establishes lineage-specific signatures that guide the homing of leukocyte subsets to tissues. Immunity. 2018;49:1175–1190.e7. doi: 10.1016/j.immuni.2018.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Holtkamp S.J., Ince L.M., Barnoud C., et al. Circadian clocks guide dendritic cells into skin lymphatics. Nat Immunol. 2021;22:1375–1381. doi: 10.1038/s41590-021-01040-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Cervantes-Silva M.P., Carroll R.G., Wilk M.M., et al. The circadian clock influences T cell responses to vaccination by regulating dendritic cell antigen processing. Nat Commun. 2022;13:7217. doi: 10.1038/s41467-022-34897-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Wang C., Zeng Q., Gül Z.M., et al. Circadian tumor infiltration and function of CD8+ T cells dictate immunotherapy efficacy. Cell. 2024;187:2690–2702.e17. doi: 10.1016/j.cell.2024.04.015. [DOI] [PubMed] [Google Scholar]
- 88.Cuddapah V.A., Zhang S.L., Sehgal A. Regulation of the blood–brain barrier by circadian rhythms and sleep. Trends Neurosci. 2019;42:500–510. doi: 10.1016/j.tins.2019.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Zhang S.L., Lahens N.F., Yue Z., et al. A circadian clock regulates efflux by the blood-brain barrier in mice and human cells. Nat Commun. 2021;12:617. doi: 10.1038/s41467-020-20795-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Zeng Y., Guo Z., Wu M., Chen F., Chen L. Circadian rhythm regulates the function of immune cells and participates in the development of tumors. Cell Death Discov. 2024;10:199. doi: 10.1038/s41420-024-01960-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Wang C., Barnoud C., Cenerenti M., et al. Dendritic cells direct circadian anti-tumour immune responses. Nature. 2023;614:136–143. doi: 10.1038/s41586-022-05605-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Fortin B.M., Pfeiffer S.M., Insua-Rodríguez J., et al. Circadian control of tumor immunosuppression affects efficacy of immune checkpoint blockade. Nat Immunol. 2024;25(7):1257–1269. doi: 10.1038/s41590-024-01859-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Tsuruta A., Shiiba Y., Matsunaga N., et al. Diurnal expression of PD-1 on tumor-associated macrophages underlies the dosing time-dependent antitumor effects of the PD-1/PD-L1 inhibitor BMS-1 in B16/BL6 melanoma-bearing mice. Mol Cancer Res. 2022;20:972–982. doi: 10.1158/1541-7786.MCR-21-0786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Qian D.C., Kleber T., Brammer B., et al. Effect of immunotherapy time-of-day infusion on overall survival among patients with advanced melanoma in the USA (MEMOIR): a propensity score-matched analysis of a single-centre, longitudinal study. Lancet Oncol. 2021;22:1777–1786. doi: 10.1016/S1470-2045(21)00546-5. [DOI] [PubMed] [Google Scholar]
- 95.Chen P., Hsu W.H., Chang A., et al. Circadian regulator CLOCK recruits immune suppressive microglia into the GBM tumor microenvironment. Cancer Discov. 2020;10:371. doi: 10.1158/2159-8290.CD-19-0400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Xuan W., Hsu W.H., Khan F., et al. Circadian regulator CLOCK drives immunosuppression in glioblastoma. Cancer Immunol Res. 2022;10:770. doi: 10.1158/2326-6066.CIR-21-0559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Montaigne D., Marechal X., Modine T., et al. Daytime variation of perioperative myocardial injury in cardiac surgery and its prevention by Rev-Erbα antagonism: a single-centre propensity-matched cohort study and a randomised study. Lancet. 2018;391:59–69. doi: 10.1016/S0140-6736(17)32132-3. [DOI] [PubMed] [Google Scholar]
- 98.Printezi M.I., Kilgallen A.B., Bond M.J.G., et al. Toxicity and efficacy of chronomodulated chemotherapy: a systematic review. Lancet Oncol. 2022;23:e129–e143. doi: 10.1016/S1470-2045(21)00639-2. [DOI] [PubMed] [Google Scholar]
- 99.Jia J.L., Alshamsan B., Ng T.L. Temozolomide chronotherapy in glioma: a systematic review. Curr Oncol. 2023;30:1893–1902. doi: 10.3390/curroncol30020147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Gonzalez-Aponte M.F., Damato A.R., Trebucq L.L., et al. Circadian regulation of MGMT expression and promoter methylation underlies daily rhythms in TMZ sensitivity in glioblastoma. J Neuro Oncol. 2024;166:419. doi: 10.1007/s11060-023-04535-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Hadadi E., Taylor W., Li X.-M., et al. Chronic circadian disruption modulates breast cancer stemness and immune microenvironment to drive metastasis in mice. Nat Commun. 2020;11:3193. doi: 10.1038/s41467-020-16890-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Arvanitis C.D., Ferraro G.B., Jain R.K. The blood–brain barrier and blood–tumour barrier in brain tumours and metastases. Nat Rev Cancer. 2020;20:26–41. doi: 10.1038/s41568-019-0205-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Watkins S., Robel S., Kimbrough I.F., Robert S.M., Ellis-Davies G., Sontheimer H. Disruption of astrocyte–vascular coupling and the blood–brain barrier by invading glioma cells. Nat Commun. 2014;5:4196. doi: 10.1038/ncomms5196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Aiello I., Fedele M.L.M., Román F., et al. Circadian disruption promotes tumor-immune microenvironment remodeling favoring tumor cell proliferation. Sci Adv. 2020;6 doi: 10.1126/sciadv.aaz4530. [DOI] [PMC free article] [PubMed] [Google Scholar]