Abstract
Background
Non‐surgical treatment, including exercises and mobilisation, has been offered to people experiencing mild to moderate symptoms arising from carpal tunnel syndrome (CTS). However, the effectiveness and duration of benefit from exercises and mobilisation for this condition remain unknown.
Objectives
To review the efficacy and safety of exercise and mobilisation interventions compared with no treatment, a placebo or another non‐surgical intervention in people with CTS.
Search methods
We searched the Cochrane Neuromuscular Disease Group Specialised Register (10 January 2012), CENTRAL (2011, Issue 4), MEDLINE (January 1966 to December 2011), EMBASE (January 1980 to January 2012), CINAHL Plus (January 1937 to January 2012), and AMED (January 1985 to January 2012).
Selection criteria
Randomised or quasi‐randomised controlled trials comparing exercise or mobilisation interventions with no treatment, placebo or another non‐surgical intervention in people with CTS.
Data collection and analysis
Two review authors independently assessed searches and selected trials for inclusion, extracted data and assessed risk of bias of the included studies. We calculated risk ratios (RR) and mean differences (MD) with 95% confidence intervals (CIs) for primary and secondary outcomes of the review. We collected data on adverse events from included studies.
Main results
Sixteen studies randomising 741 participants with CTS were included in the review. Two compared a mobilisation regimen to a no treatment control, three compared one mobilisation intervention (for example carpal bone mobilisation) to another (for example soft tissue mobilisation), nine compared nerve mobilisation delivered as part of a multi‐component intervention to another non‐surgical intervention (for example splint or therapeutic ultrasound), and three compared a mobilisation intervention other than nerve mobilisation (for example yoga or chiropractic treatment) to another non‐surgical intervention. The risk of bias of the included studies was low in some studies and unclear or high in other studies, with only three explicitly reporting that the allocation sequence was concealed, and four reporting blinding of participants. The studies were heterogeneous in terms of the interventions delivered, outcomes measured and timing of outcome assessment, therefore, we were unable to pool results across studies. Only four studies reported the primary outcome of interest, short‐term overall improvement (any measure in which patients indicate the intensity of their complaints compared to baseline, for example, global rating of improvement, satisfaction with treatment, within three months post‐treatment). However, of these, only three fully reported outcome data sufficient for inclusion in the review. One very low quality trial with 14 participants found that all participants receiving either neurodynamic mobilisation or carpal bone mobilisation and none in the no treatment group reported overall improvement (RR 15.00, 95% CI 1.02 to 220.92), though the precision of this effect estimate is very low. One low quality trial with 22 participants found that the chance of being 'satisfied' or 'very satisfied' with treatment was 24% higher for participants receiving instrument‐assisted soft tissue mobilisation compared to standard soft tissue mobilisation (RR 1.24, 95% CI 0.89 to 1.75), though participants were not blinded and it was unclear if the allocation sequence was concealed. Another very low‐quality trial with 26 participants found that more CTS‐affected wrists receiving nerve gliding exercises plus splint plus activity modification had no pathologic finding on median and ulnar nerve distal sensory latency assessment at the end of treatment than wrists receiving splint plus activity modification alone (RR 1.26, 95% CI 0.69 to 2.30). However, a unit of analysis error occurred in this trial, as the correlation between wrists in participants with bilateral CTS was not accounted for. Only two studies measured adverse effects, so more data are required before any firm conclusions on the safety of exercise and mobilisation interventions can be made. In general, the results of secondary outcomes of the review (short‐ and long‐term improvement in CTS symptoms, functional ability, health‐related quality of life, neurophysiologic parameters, and the need for surgery) for most comparisons had 95% CIs which incorporated effects in either direction.
Authors' conclusions
There is limited and very low quality evidence of benefit for all of a diverse collection of exercise and mobilisation interventions for CTS. People with CTS who indicate a preference for exercise or mobilisation interventions should be informed of the limited evidence of effectiveness and safety of this intervention by their treatment provider. Until more high quality randomised controlled trials assessing the effectiveness and safety of various exercise and mobilisation interventions compared to other non‐surgical interventions are undertaken, the decision to provide this type of non‐surgical intervention to people with CTS should be based on the clinician's expertise in being able to deliver these treatments and patient's preferences.
Keywords: Humans; Physical Therapy Modalities; Carpal Tunnel Syndrome; Carpal Tunnel Syndrome/therapy; Exercise Therapy; Exercise Therapy/methods; Manipulation, Chiropractic; Manipulation, Chiropractic/methods; Massage; Massage/methods; Randomized Controlled Trials as Topic; Splints; Ultrasonic Therapy; Ultrasonic Therapy/methods; Yoga
Plain language summary
Exercise and mobilisation interventions for carpal tunnel syndrome
Carpal tunnel syndrome is a common condition where one of two main nerves in the wrist is compressed, resulting in pain to the hand, wrist and sometimes arm, numbness and tingling in the thumb, index and long finger. In advanced cases the muscles of the hand can become weak. The condition affects approximately three per cent of the population, more commonly women. While carpal tunnel syndrome can be treated with surgery, people with mild to moderate symptoms are sometimes offered non‐surgical interventions such as exercises or mobilisation. Based on the 16 studies identified, there is limited and very low quality evidence of benefit for all of a diverse collection of exercise and mobilisation interventions for improving symptoms, functional ability (for example hand grip strength), quality of life, and neurophysiologic parameters, and for minimising adverse effects and the need for surgery in people with carpal tunnel syndrome. More research is needed to investigate the effectiveness of exercises and mobilisation for people with carpal tunnel syndrome, especially the sustainability and long‐term effects of this treatment.
Summary of findings
Summary of findings for the main comparison. Neurodynamic mobilisation compared with no treatment for carpal tunnel syndrome.
| Neurodynamic mobilisation compared to no treatment for carpal tunnel syndrome | ||||||
| Patient or population: People with carpal tunnel syndrome on a waiting list for carpal tunnel release Settings: Switzerland Intervention: Neurodynamic mobilisation for three weeks Comparison: No treatment | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| No treatment | Nerve mobilisation | |||||
| Short‐term overall improvement (three months or less) | Study population | RR 15 (1.02 to 220.92) | 14 (1 study) | ⊕⊝⊝⊝ very low2,3 | ||
| 0 of 7 participants1 | 7 of 7 participants | |||||
| Adverse effects | See comment | See comment | 14 (1 study) | ⊕⊝⊝⊝ very low2,3 | No adverse effects of treatment were reported in either the intervention or control group. | |
|
Short‐term improvement in CTS symptoms (VAS pain) (three months or less) Scale from: 0 to 5 |
The mean short‐term improvement in CTS symptoms (VAS pain at three months or less) in the control group was 2.14. | The mean short‐term improvement in CTS symptoms (VAS pain) (three months or less) in the intervention groups was 0.57 lower (1.73 lower to 0.59 higher) | 14 (1 study) | ⊕⊝⊝⊝ very low2,3 | ||
| Short‐term improvement in functional ability or health‐related quality of life (hand function) (three months or less) | Study population | RR 9 (0.59 to 137.65) | 12 (1 study) | ⊕⊝⊝⊝ very low2,3 | ||
| 0 per 10001 | 0 per 1000 (0 to 0) | |||||
| Short‐term improvement in neurophysiologic parameters (three months or less) | See comment | See comment | 14 (1 study) | ⊕⊝⊝⊝ very low2,3 | Short‐term improvement in neurophysiologic parameters was not a measured outcome. | |
| Long‐term improvement in CTS symptoms (more than three months) | See comment | See comment | 14 (1 study) | ⊕⊝⊝⊝ very low2,3 | Long‐term improvement in CTS symptoms was not a measured outcome. | |
| Long‐term improvement in functional ability or health‐related quality of life (more than three months) | See comment | See comment | 14 (1 study) | ⊕⊝⊝⊝ very low2,3 | Long‐term improvement in functional ability or health‐related quality of life was not a measured outcome. | |
| Need for surgery | Study population | RR 0.33 (0.1 to 1.12) | 14 (1 study) | ⊕⊝⊝⊝ very low2,3 | ||
| 857 per 10001 | 283 per 1000 (86 to 960) | |||||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk ratio; | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
1 Assumed risk is based on the risk in the control group in the one study comparing nerve mobilisation to no treatment (Tal‐Akabi 2000)
2 Allocation sequence unclear, and patients were not blinded. 3 95%CIs are very wide due to small sample size.
Background
Description of the condition
Carpal tunnel syndrome (CTS) is a neuromuscular condition where the median nerve at the level of the wrist undergoes irritation. This is often attributed to increased pressure within the carpal tunnel (Keith 2009; Kerwin 1996). The most commonly reported symptoms of CTS include pain in the wrist and hand which can radiate to the arm (Rempel 1998) and paraesthesiae (numbness) in the thumb, index, middle and radial half of the ring finger (Szabo 1994). Thenar muscle weakness has been found to occur in those with advanced CTS (Szabo 1994).
Results of a Swedish study suggest that the prevalence of CTS in the general population is 3.8% for clinically diagnosed cases and 2.7% for electrophysiologically confirmed cases (Atroshi 1999). Recent evidence indicates that between 1981 to 1985 the adjusted annual incidence of CTS was 258 per 100,000 person‐years, compared to 424 per 100,000 person‐years between 2000 to 2005 in Minnesota, USA, though it is not clear whether this apparent increase in incidence is due to increased diagnostic practice and awareness of CTS (Gelfman 2009). Carpal tunnel syndrome is generally found to be uncommon in people under the age of 25, and more common in women than in men (Atroshi 1999; Charles 2009). An association between obesity and an increased incidence of CTS has also been identified (Bland 2005; Stallings 1997; Werner 1994a).
The course of CTS symptoms is unpredictable. Some patients experience a continuous deterioration in hand function whilst others describe 'silent' periods and intermittent exacerbation of symptoms (Braun 1989).
Description of the intervention
Surgical and non‐surgical treatment options exist for people with CTS. Surgical treatment is usually offered to individuals who have persistent CTS symptoms, severe sensory disturbance or thenar motor weakness. In contrast, non‐surgical treatments are offered to those who experience intermittent symptoms of mild to moderate CTS or temporarily to those awaiting carpal tunnel release. Surgical treatment options for patients with CTS have been addressed in other Cochrane reviews, namely surgical treatment options for CTS (Scholten 2007), and the effect of surgical versus non‐surgical treatment (Verdugo 2008).
There are numerous non‐surgical options for the treatment of CTS, such as exercises or mobilisation, ergonomic modification (equipment or positioning), splinting, therapeutic ultrasound, oral medication, vitamins and complementary therapies. Exercise and mobilisation interventions include chiropractic and osteopathic manual interventions, nervous system mobilisation techniques such as nerve and tendon gliding exercises, mobilisation techniques targeting the carpal bones, or soft tissue mobilisation (for example massage) or whole body mobilisation (for example yoga). Table 2 provides definitions of a selection of these interventions. These interventions vary in their intensity and the duration to which they can be delivered ranges from a few days to months. Further, exercises and mobilisation interventions can be self‐administered or delivered by trained health professionals.
1. Glossary.
| Term | Definition |
| Nerve gliding exercises | Mobilisation technique developed by Totten and Hunter (Totten 1991), where the median nerve is mobilised by putting the hand and wrist in six different positions: (1) wrist in neutral position, fingers and thumb in flexion; (2) wrist in neutral position, fingers and thumb extended; (3) wrist and fingers extended, thumb in neutral position; (4) wrist, fingers, and thumb extended; (5) forearm in supination; (6) the opposite hand applies a gentle stretch to the thumb). During these exercises, the neck and the shoulder are in a neutral position, and the elbow is in supination and 90 degrees of flexion. |
| Tendon gliding exercises | Mobilisation technique developed by Totten and Hunter (Totten 1991), which involve sliding the flexor tendons of the hand by moving the fingers through the following five discrete positions: straight, hook, fist, table top, and straight fist positions. |
| Neurodynamic technique | A form of manual therapy which includes positioning and range of motion known to cause significant stress across the median nerve. Specifically, the neurodynamic technique includes 25° of contralateral cervical sidebending, ipsilateral shoulder depression and abduction to 90°, shoulder external rotation to 90°, 45° of elbow extension, forearm supination, and repetitive wrist and finger flexion and extension through the available range of motion (Bialosky 2009; Butler 1991). |
| Neurodynamic mobilisation | Mobilisation with a median nerve bias, which includes slight glenohumeral abduction, shoulder girdle depression, elbow extension, lateral rotation of the whole arm, wrist, thumb & finger extension and finally glenohumeral abduction (Butler 1991). |
| Carpal bone mobilisation | Posterior‐anterior and/or anterior‐posterior mobilisation techniques (Maitland 1991). |
| Contrast bath | The immersion of a limb in hot and cold water in an alternating fashion (Janssen 2009) |
| Phalen's test | A test performed by the patient flexing the wrists at 90 degrees for 60 seconds, where the test is recorded as positive if the patient experiences paraesthesia in distribution of the median nerve (Phalen 1966). |
| Reverse Phalen's test | A test performed by the patient extending the wrists at 90 degrees for 60 seconds, where the test is recorded as positive if the patient experiences paraesthesia in distribution of the median nerve (Werner 1994b). |
| Tinel's test | A test performed by a clinician tapping the distal wrist crease over the median nerve, where the test is recorded as positive if the patient experiences paraesthesia in at least one of three radial digits (D'Arcy 2000). |
How the intervention might work
There are a number of theories regarding how exercise and mobilisation interventions are effective in reducing the symptoms of CTS. Rozmaryn 1998 suggested that gliding exercises can potentially reduce tenosynovial oedema, improve venous return from the nerve bundles, and reduce pressure inside the carpal tunnel. Garfinkel 1998 hypothesised that stretching through yoga may relieve compression in the carpal tunnel, improve joint posture and decrease nerve compression, and improve blood flow to the median nerve. Stretching exercises for CTS have been prescribed for the same reasons, and also to mobilise the median nerve within the carpal canal if it is adherent (Moraska 2008).
Why it is important to do this review
A number of systematic reviews of non‐surgical interventions for CTS have been published (Ashworth 2010; Gerritsen 2002; Goodyear‐Smith 2004; Huisstede 2010; McKeon 2008; Muller 2004; O'Connor 2003; Ono 2010; Piazzini 2007). The most recent review (Huisstede 2010) searched for studies published until January 2010 and concluded that the evidence base for exercise and mobilisation interventions remains incomplete. Cochrane systematic reviews of local steroid injection (Marshall 2007), surgical versus non‐surgical treatment (Verdugo 2008) different surgical treatment options (Scholten 2007), therapeutic ultrasound (Page 2012), and ergonomic interventions (O'Connor 2012) for CTS already exist, and up‐to‐date Cochrane systematic reviews of other non‐surgical interventions for CTS are required. Given the large number of exercise and mobilisation treatments available for CTS, the potential benefits and harms of these interventions needs to be ascertained and based on the most up‐to‐date information available.
Objectives
To compare the efficacy and safety of exercise and mobilisation interventions for CTS with no treatment, placebo or another non‐surgical intervention.
This is a split review replacing the exercise and mobilisation interventions included in a previous review titled Non‐surgical treatment (other than steroid injection) for carpal tunnel syndrome (O'Connor 2003). As a result, this review has a more specific objective than the previous review.
Methods
Criteria for considering studies for this review
Types of studies
All published and unpublished randomised and quasi‐randomised controlled trials were eligible for inclusion. We included studies comparing any form of exercise or mobilisation (including yoga) with no treatment, placebo, or other non‐surgical interventions. We also included studies comparing different types of exercise or mobilisation intervention. We excluded studies comparing exercise or mobilisation with surgical treatment as these have been reviewed elsewhere by Verdugo 2008. There were no language restrictions.
Types of participants
All participants with a diagnosis of CTS, as defined by the authors of each study. We excluded participants having undergone previous surgery for CTS.
Types of interventions
We included any exercise or mobilisation interventions. Comparison interventions included no treatment, placebo, and other non‐surgical interventions for CTS; we excluded surgical interventions as comparisons.
Types of outcome measures
We modified the outcomes reported in this review from the original review (O'Connor 2003) to be consistent as possible with other Cochrane reviews for CTS (Marshall 2007; Scholten 2007; Verdugo 2008).
Primary outcomes
Short‐term overall improvement (any measure in which patients indicate the intensity of their complaints compared to baseline, for example global rating of improvement, satisfaction with treatment) (dichotomous outcome; three months or less).
Secondary outcomes
Adverse effects.
Short‐term improvement in CTS symptoms (e.g. pain, paraesthesia, nocturnal paraesthesia) (three months or less).
Short‐term improvement in functional ability or health‐related quality of life (three months or less).
Short‐term improvement in neurophysiologic parameters (three months or less).
Long‐term improvement in CTS symptoms (greater than three months).
Long‐term improvement in functional ability or health‐related quality of life (greater than three months).
Need for surgical release of flexor retinaculum.
Search methods for identification of studies
Electronic searches
We searched the Cochrane Neuromuscular Disease Group Specialized Register (10 January 2012), CENTRAL (2011, Issue 4), MEDLINE (January 1966 to December 2011), EMBASE (January 1980 to January 2012), CINAHL Plus (January 1937 to January 2012) and AMED (January 1985 to January 2012).
For search strategies, see Appendix 1 (MEDLINE), Appendix 2 (EMBASE), Appendix 3 (AMED), Appendix 4 (CINAHL) and Appendix 5 (CENTRAL).
Searching other resources
We also reviewed the reference lists of randomised or quasi‐randomised trials identified from the electronic searches to identify any potentially relevant studies for inclusion.
Data collection and analysis
The review authors followed the recommended strategies for data collection and analysis as documented in Chapter 7 and 9 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011a).
Selection of studies
At least two review authors independently selected trials for possible inclusion against a predetermined checklist of inclusion criteria (see Criteria for considering studies for this review). We screened titles and abstracts of citations from the search results and initially categorised studies into the following groups:
possibly relevant ‐ studies that met the inclusion criteria and studies from which it was not possible to determine whether they met the criteria either from their title or abstract; or
excluded ‐ studies that clearly did not meet the inclusion criteria.
If a title or abstract appeared to meet the eligibility criteria for inclusion in the review, or we were unable to determine eligibility, we obtained a full text version of the article and two review authors independently assessed it to determine whether the study fulfilled the inclusion criteria. The review authors resolved discrepancies through discussion.
Data extraction and management
Two review authors independently extracted data using a standard data extraction form developed for this review. The review authors resolved any discrepancies through discussion until consensus was reached. We piloted the data extraction form and modified it accordingly before use. In addition to items for assessing risk of bias and study results, we also recorded the following study characteristics:
participant details, including demographic data and inclusion/exclusion criteria;
types of interventions used in the intervention and comparison groups;
outcomes reported, including the tools and timing for outcome measures.
One review author compiled all comparisons and entered outcome data into Review Manager 5. At least one other review author cross‐checked data. We attempted to contact trialists to obtain any incomplete or missing outcome data. If our attempts were unsuccessful, the study was included in the review and fully described, but not included in any meta‐analysis. We made an entry of this process in the notes section of the Characteristics of included studies tables.
Assessment of risk of bias in included studies
Two review authors independently assessed the risk of bias in included studies using The Cochrane Collaboration's tool for assessing risk of bias, as described in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011b). We assessed the following items for risk of bias based on information extracted from reports of the included studies:
random sequence generation;
allocation concealment;
blinding of participants and personnel;
blinding of outcome assessment;
incomplete outcome data (defined separately for data measured at three months or less, and after three months);
selective reporting;
other sources of bias (e.g. inappropriate unit of analysis).
We rated each item as being at 'Low risk', 'Unclear risk' or 'High risk' of bias. We resolved any discrepancies through discussion.
Measures of treatment effect
We used the Cochrane statistical software Review Manager 5 to perform data analysis. We expressed results as risk ratios (RR) with 95% confidence intervals (CI) for dichotomous outcomes and mean differences (MD) with 95% CI for continuous outcomes if the same measurement tool was used to measure the same outcome across separate studies. Alternatively, we summarised continuous outcomes using the standardised mean difference (SMD) when studies measured the same outcome but employed different measurement tools.
Unit of analysis issues
We sought information about the unit of randomisation (participants or wrists) from the included studies. In studies which randomised wrists, we sought information about whether each participant's wrist was allocated to different treatments, or whether there was no constraint that each participant's wrist be allocated to different treatments Given that results for different wrists for the participants with bilateral CTS are unlikely to be independent, we assessed how the investigators of studies which included participants with bilateral CTS took account of this dependence in their analyses (e.g. use of paired or matched analyses, generalised estimating equations). If this information was not reported, we contacted trialists for clarification. We also requested individual wrist outcome data from trialists to re‐analyse the data. If we were unable to obtain individual wrist outcome data, we had planned to estimate parameters (such as an intra‐class correlation coefficient) from studies that reported sufficient information to calculate this, and to use these estimates to adjust the results in other studies, following the advice provided in sections 16.3 and 16.4 of the Cochrane Handbook of Systematic Reviews of Interventions (Higgins 2011c). If unable to adjust the outcome data, we included the data as reported by the trialists, and commented on the validity of such analyses.
Dealing with missing data
We sought relevant missing information about study design, outcome data, or attrition rates such as drop‐outs, losses to follow‐up and withdrawn study participants from the authors of included studies, where possible.
Assessment of heterogeneity
We assessed clinical heterogeneity by determining whether the characteristics of participants, interventions, outcome measures and timing of outcome measurement were similar across studies. We assessed statistical heterogeneity using the Chi2 statistic and the I2 test (Higgins 2002). We interpreted the I2 statistic using the following as an approximate guide:
0% to 40% might not be important heterogeneity;
30% to 60% may represent moderate heterogeneity;
50% to 90% may represent substantial heterogeneity; and
75% to 100% may represent considerable heterogeneity (Deeks 2011).
Assessment of reporting biases
To assess publication bias, we intended to generate funnel plots if the review included at least 10 studies examining the same treatment comparison (Sterne 2011). To assess outcome reporting bias, we searched protocols of trials on the clinical trials register that is maintained by the US National Institute of Health at http://clinicaltrials.gov, and we searched protocols of trials published after July 1st 2005 using the Clinical Trial Register at the International Clinical Trials Registry Platform of the World Health Organization (WHO) (http://apps.who.int/trialssearch), to compare with the corresponding published randomised controlled trials (Dwan 2008; Dwan 2011).
Data synthesis
We pooled the results of studies with similar characteristics (participants, interventions, outcome measures and timing of outcome measurement) to provide estimates of the efficacy of therapeutic ultrasound for CTS. Where we could not combine data, we presented a narrative synthesis of results. We meta‐analysed pooled results using either a fixed‐effect or random‐effects model (depending on the level of clinical and methodological heterogeneity). Statistical significance was set at P < 0.05 for primary and secondary outcome measures.
Subgroup analysis and investigation of heterogeneity
We conducted subgroup analyses according to the severity of CTS symptoms and the sex of the participants, since these factors may cause variations in outcomes. Subgroups were defined as follows:
severity of CTS symptoms: early (E), intermediate (I) and advanced (A) symptoms (Szabo 1992);
sex: male, female.
Sensitivity analysis
We conducted sensitivity analyses for each element on the 'Risk of bias' table by excluding studies that had a high risk of bias. We also conducted sensitivity analyses using the following filter:
quality of diagnostic criteria: high (A), moderate (B) and low (C) quality (Rempel 1998).
Summary of findings
We created a 'Summary of findings' table for the main comparison of the review, exercise or mobilisation (delivered as a single intervention) versus no treatment. We included in the table one effect estimate for each of our primary and secondary outcomes (see Types of outcome measures).
Results
Description of studies
See Characteristics of included studies; Characteristics of excluded studies.
Results of the search
The search conducted up until 10 January 2012 identified a total of 316 records. Table 1 reports the number of hits retrieved by each search strategy. The number of records after removal of duplicates was 170. From these, we retrieved 37 full text papers for further examination and scanning the reference lists of these studies identified one additional reference to obtain. After screening the full text of the 38 selected papers for eligibility, 16 studies (Akalin 2002; Bahrami 2006; Bardak 2009; Baysal 2006; Bialosky 2009; Brininger 2007; Burke 2007; Davis 1998; Field 2004; Garfinkel 1998; Heebner 2008; Horng 2011; Janssen 2009; Moraska 2008; Pinar 2005; Tal‐Akabi 2000) fulfilled the inclusion criteria. Five studies are awaiting assessment (Ashraf 2009; Avci 2004; El Miedany 2009; Maltese 2006; Shi 2006). Of the studies awaiting assessment, three are written in a language other than English and are currently being translated (Avci 2004; Maltese 2006; Shi 2006), El Miedany 2009 is reported as a conference abstract only and communication with the trialists confirmed that the study is currently being written up for publication, and it is unclear whether exercise interventions were delivered in Ashraf 2009 (attempts to clarify this with the trialists have so far been unsuccessful). A flow diagram of the study selection process is presented in Figure 1.
1.

Study flow diagram.
Table 1
| Database | Period searched | Date searched | Number of hits |
| Cochrane Neuromuscular Disease Group Specialised Register | to 10 January 2012 | 10 January 2012 | 40 |
| CENTRAL | to Issue 12, 2011 | 10 January 2012 | 65 |
| MEDLINE | January 1966 to January 2012 | 10 January 2012 | 67 |
| EMBASE | January 1980 to January 2012 | 10 January 2012 | 70 |
| CINAHL Plus | January 1937 to January 2012 | 10 January 2012 | 50 |
| AMED | January 1985 to January 2012 | 10 January 2012 | 24 |
Included studies
Sixteen studies randomly allocated people with CTS to an exercise or mobilisation intervention (either alone or as part of a multi‐component intervention) or to either no treatment, placebo, or another non‐surgical intervention. A total of 741 participants were randomised. There were 117 male and 609 female participants who completed the studies (demographic data were missing for some participants). The exercise or mobilisation interventions varied in intensity, mode of delivery and duration of treatment across the studies. In seven studies (Akalin 2002; Bahrami 2006; Baysal 2006; Davis 1998; Garfinkel 1998; Horng 2011; Pinar 2005), some or all participants had bilateral CTS, where both wrists contributed to the analysis. In five of these studies (Baysal 2006; Davis 1998; Garfinkel 1998; Horng 2011; Pinar 2005), randomisation occurred at the level of participants, where the same intervention was delivered to both wrists in participants with bilateral CTS. In Akalin 2002, randomisation of wrists occurred, where for all participants with bilateral CTS, each wrist received a different intervention. It was unclear in Bahrami 2006 whether participants with bilateral CTS received the same or different interventions for each wrist. We have described the potential for unit of analysis errors in each of these studies below.
Akalin 2002 examined the benefit of daily nerve and tendon gliding exercises with a neutral volar wrist splint worn daily and nightly for four weeks compared with wrist splint alone. Outcomes assessed were symptoms, hand function, grip strength, pinch strength, two‐point discrimination, Tinel's test, Phalen's test and patient satisfaction in 28 participants with 36 CTS‐affected hands. Table 2 reports definitions of Phalen's test and Tinel's test. Analysis was undertaken at the wrist‐level for all outcomes, though some participants in each group had bilateral CTS. Bilateral cases had a different intervention applied to each wrist. The trialists did not report how the correlation between both wrists was accounted for in the analysis, and attempts to clarify this information from the trialists were unsuccessful. Therefore, the results of this study may be invalid due to a unit of analysis error, though this is unclear.
Bahrami 2006 allocated 28 participants with 38 CTS‐affected wrists to nerve and tendon gliding exercises performed four times a day for four weeks plus wrist splint used at nights for four weeks, or to nightly use of wrist splint alone. Outcomes assessed were symptoms, hand function, Tinel's test, Phalen's test, nerve conduction and patient satisfaction. Nineteen participants had unilateral CTS and nine participants had bilateral CTS. The trialists did not report how the correlation between both wrists was accounted for in the analysis, and attempts to clarify this information from the trialists were unsuccessful. Therefore, the results of this study may be invalid due to a unit of analysis error, though this is unclear.
In the study conducted by Bardak 2009, 111 participants with 111 CTS‐affected wrists were randomly allocated to splinting for six weeks plus steroid injection, splint for six weeks plus nerve and tendon gliding exercises three times a day for six weeks plus steroid injection, or nerve and tendon gliding exercises three times day for six weeks. Outcomes measured were symptoms, functional status, Phalen's test, Tinel's test, reverse Phalen's test, compression test, pain, two‐point discrimination, and patient satisfaction.
Baysal 2006 involved three different treatment groups for 36 participants with 72 CTS‐affected hands. Daily nerve and tendon gliding exercises plus a neutral volar wrist splint worn day and night plus therapeutic ultrasound delivered for five days a week for a total of three weeks was compared with daily nerve and tendon gliding exercises and splinting for three weeks, and with therapeutic ultrasound and splinting for three weeks. Outcomes measured included symptoms, pain, Tinel's and Phalen's test, two‐point discrimination, hand function, grip strength, pinch strength, nerve conduction, and patient satisfaction. Analysis was undertaken at the wrist‐level for all outcomes, though all participants in each group had bilateral CTS. Bilateral cases had the same intervention applied to each wrist. The trialists did not report how the correlation between both wrists was accounted for in the analysis, and attempts to clarify this information from the trialists were unsuccessful. Therefore, the results of this study may be invalid due to a unit of analysis error, though this is unclear.
In the study conducted by Bialosky 2009, 40 participants with 40 CTS‐affected wrists were randomly allocated to receive a neurodynamic exercise technique twice a week for three weeks or a "sham" neurodynamic technique twice a week for three weeks. All wore a splint at night and during daytime activities that worsened CTS symptoms. Outcomes measured included clinical pain, pressure pain, thermal pain, temporal summation, "usual pain", upper limb disability, grip strength, sensation at the tip of the thumb, index finger and middle finger, and nerve conduction. Some participants in this study had bilateral CTS, though only one affected wrist per participant contributed to the study.
In the study conducted by Brininger 2007, 61 participants with 61 CTS‐affected hands were allocated to one of four groups: daily nerve and tendon gliding exercises in addition to neutral wrist and metacarpophalangeal (MCP) splint worn only at night for four weeks; daily nerve and tendon gliding exercises plus off‐the‐shelf wrist cock‐up splint (immobilised in 20 degrees of extension) worn only at night for four weeks; nightly neutral wrist and MCP splint for four weeks (with no exercises); and nightly off‐the‐shelf wrist cock‐up splint (immobilised in 20 degrees of extension) for four weeks (with no exercises). The outcomes measured in this study include symptoms, hand function, functional sensibility, grip strength, pinch strength and patient satisfaction.
Burke 2007 examined the benefit of the Graston Technique instrument‐assisted soft tissue mobilisation (GISTM), which involves the use of a patented form of instrument‐assisted soft tissue mobilisation (STM) that is designed to enable the clinician to effectively break down scar tissue and fascial restrictions of forearm‐wrist‐hand areas, as compared to manual STM of the forearm‐wrist‐hand areas, where the clinician's hands are used to break down scar tissue and fascial restrictions. The duration of treatment was twice per week for the first four weeks and then once per week for the next two weeks. The study included 26 participants with 26 CTS‐affected hands. Outcomes collected include symptoms, pain, hand function, nerve conduction, range of motion, grip strength, pinch strength, two‐point discrimination, pressure sensitivities, Phalen's and Tinel's test, patient satisfaction and adverse events.
Davis 1998 compared chiropractic care, comprising high velocity, low‐amplitude manual thrust procedures designed to create increased joint motion in the joints of the upper extremities, including the wrist, elbow and shoulder as well as in the cervical and upper thoracic regions of the vertebrae, massage, ultrasound and wrist splints, with medical management (ibuprofen and wrist splint) for seven weeks in 91 participants with 149 CTS‐affected wrists. Outcomes included nerve conduction, physical and mental distress, hand function, vibrometry on digit three, health‐related quality of life and adverse effects. Analysis was undertaken at the participant‐level for the outcomes physical distress, mental distress, hand function and health‐related quality of life, though some participants in each group had bilateral CTS. Communication with the trialists confirmed that bilateral cases had the same intervention applied to both wrists. However, the trialists did not report controlling for the correlation between both wrists for these four outcomes, so a unit of analysis error is likely to have occurred for these outcomes (but not for the outcomes nerve conduction and vibrometry, which were analysed separately for left‐ and right‐affected wrists).
In the study conducted by Field 2004, massage therapy on the affected arm by a therapist once a week for four weeks and self‐massage done daily at home prior to bedtime was compared with standard treatment (only defined as no massage therapy delivered) in 16 participants with 16 CTS‐affected hands. Outcomes measured included symptoms, Tinel's and Phalen's test, nerve conduction, self‐perceived grip strength, pain, state anxiety, and mood.
Garfinkel 1998 recruited 51 participants (42 participants with 67 CTS‐affected wrists completed the study) and studied the efficacy of the Iyengar approach to hatha yoga (Iyengar 1966) performed twice weekly for eight weeks, with splinting, on the outcomes pain, nocturnal wakening, Tinel's and Phalen's test, grip strength, nerve conduction, and paraesthesia and numbness. Analysis was undertaken at the wrist‐level for all outcomes, though some participants in each group had bilateral CTS. Bilateral cases had the same intervention applied to each wrist. The trialists did not report how the correlation between both wrists was accounted for in the analysis, and attempts to clarify this information from the trialists were unsuccessful. Therefore, the results of this study may be invalid due to a unit of analysis error, though this is unclear.
Heebner 2008 compared neurodynamic mobilisation exercises (with a median nerve bias) performed three to five times daily, with 10 repetitions, plus standard care, consisting of patient education, splinting, and tendon gliding exercises, with standard care alone (patient eduction, splinting and tendon gliding exercises), for six months in 60 participants with 60 CTS‐affected wrists. Duration of treatment was six months. Outcomes measured included symptoms, hand function, upper limb function and neurodynamic irritability of the median nerve.
Horng 2011 randomised 60 participants (53 participants with 89 CTS‐affected wrists completed the study) to receive one of three interventions: tendon gliding exercises three times daily plus night splint plus paraffin therapy (superficial heat therapy) for eight weeks; nerve gliding exercises three times daily plus night splint plus paraffin therapy for eight weeks; or night splint plus paraffin therapy for eight weeks. Outcomes measured included pain, symptoms, hand function, upper limb function, quality of life, Phalen's test, Tinel's test, grip strength, pinch strength, sensitivity, and nerve conduction. Analysis was undertaken at the participant‐level for the outcomes pain, symptoms, hand function, upper limb function, and quality of life, though some participants in each group had bilateral CTS. As bilateral cases had the same intervention applied to both wrists, and the trialists did not report controlling for the correlation between both wrists, a unit of analysis error is likely to have occurred for these five outcomes (but not for the nerve conduction studies outcome data, which were analysed using a mixed‐effect model which took into account that the examinations were performed on both wrists for some participants).
In Janssen 2009, 58 participants with 58 CTS‐affected wrists were allocated to one of three groups: contrast baths (immersion of the wrist in hot water for one minute then cold water for one minute in an alternating fashion) plus controlled finger flexion and finger extension exercises for 11 minutes; contrast baths alone (no exercise) for 11 minutes; or controlled finger flexion and finger extension exercises for 11 minutes. Participants received these interventions and were evaluated both pre‐ and post‐carpal tunnel release. The only outcome assessed was hand volume using the water displacement technique, which was not a pre‐specified outcome for this review.
In the study by Moraska 2008, 28 participants with 28 CTS‐affected wrists were randomly allocated to two groups. The first group received a CTS‐targeted massage protocol which was designed to (1) address any increased volume of fluid in the carpal tunnel region, (2) reduce connective tissue restriction (namely thickening or adhesions), and (3) decrease contractile tissue hypertonicity at potential points of nerve entrapment along the course of the brachial plexus and median nerve pathway. This was compared with a general massage protocol which was designed to model a typical relaxing massage session and focused on reducing muscular tension and enhancing circulation to the back, neck, and both upper extremities. The duration of treatment was twice a week for six weeks. Outcomes collected included symptoms, hand function (subjective and objective), grip strength, and pinch strength.
Pinar 2005 examined the benefit of daily nerve gliding exercises for 10 weeks in addition to a static volar wrist splint worn daily and nightly for the first six weeks and nightly for the next four weeks, plus training to modify functional activities in accordance with conservative treatment, as compared to static volar wrist splint and activity modification for 10 weeks. Outcomes measured included pain, Tinel's and Phalen's test, motor function, grip strength, pinch strength, light‐touch deep‐pressure sense, two‐point discrimination, and nerve conduction. Twenty‐six participants with 35 CTS‐affected hands were randomised. Analysis was undertaken at the wrist‐level for all outcomes, though some participants in each group had bilateral CTS. Communication with the trialists confirmed that bilateral cases had the same intervention applied to both wrists. However, the trialists did not report controlling for the correlation between both wrists, so the results of this study are likely to be invalid due to a unit of analysis error.
Tal‐Akabi 2000 allocated 21 participants with 21 CTS‐affected wrists to one of three groups: provision of carpal bone mobilisation; neurodynamic mobilisation; or no treatment for three weeks. Outcomes assessed included symptoms, pain, hand function, range of movement, upper limb tension, and need for surgical release. The procedure for neurodynamic mobilisation was described as upper limb tension test 2a (ULTT2a) by Butler 1991. This mobilisation procedure involves movement of the patient's affected upper limb through its passive range of motion. The stages in ULTT2a mobilisation include: Stage 1: the patient starts lying supine on a bed; Stage 2: the clinician passively moves the patient's upper limb into slight glenohumeral abduction and shoulder girdle depression; Stage 3: elbow extension is added; Stage 4: lateral rotation of the whole arm is added; Stage 5: wrist, thumb and finger extension is added; Stage 6: maintenance of other postures and addition of glenohumeral abduction to the end of available range or to the point where symptoms are produced.
The primary outcome for this review, short‐term overall improvement using any measure where patients indicate the intensity of their complaints compared to baseline (over three months or less), was reported in four of the 15 studies (Brininger 2007; Burke 2007; Pinar 2005; Tal‐Akabi 2000). Adverse effects were only reported in two studies (Burke 2007; Davis 1998). Only four studies reported outcomes at long‐term follow‐up (that is, more than three months after the intervention ended) (Akalin 2002; Bahrami 2006; Bardak 2009; Baysal 2006).
Some additional outcome data not reported in the study publications were provided by the authors of the studies conducted by Bialosky 2009, Heebner 2008, Moraska 2008, and Pinar 2005.
Excluded studies
In total, we excluded 133 studies after screening of titles and abstracts, and excluded 17 of 38 retrieved articles after review of the full publication. Reasons for exclusion of studies are given in the 'Characteristics of excluded studies' table. The main reasons for exclusion were that a non‐randomised study design had been employed and that interventions other than exercise or mobilisation interventions for CTS were the focus of investigation.
Risk of bias in included studies
Full details of our assessment of risk of bias in included studies are available in the 'Risk of bias' tables (see Characteristics of included studies), and a summary can be seen in Figure 2. In cases where risk of bias was rated as 'Unclear risk of bias', attempts to contact the trial authors for further information were made, and unless otherwise specified, these were unsuccessful.
2.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
Generation of the randomisation sequence was judged to have been adequate and at 'low risk of bias' in eight studies (Bardak 2009; Baysal 2006; Bialosky 2009; Burke 2007; Davis 1998; Heebner 2008; Janssen 2009; Tal‐Akabi 2000). Four studies (Bardak 2009; Baysal 2006; Bialosky 2009; Davis 1998) reported using a computer‐generated randomisation list, Burke 2007 reported using a random number table, Heebner 2008 reported tossing a coin, and Janssen 2009 and Tal‐Akabi 2000 reported that lots were drawn (cubes were pulled out of a hat). The sequence generation was unclear in six studies (Akalin 2002; Brininger 2007; Field 2004; Garfinkel 1998; Horng 2011; Moraska 2008). The studies conducted by Bahrami 2006 and Pinar 2005 used alternation, a form of quasi‐randomisation, so were judged to be at high risk of bias for this domain.
It was clear in only three studies (Bialosky 2009; Brininger 2007; Davis 1998) that the allocation sequence was successfully concealed prior to allocation of participants; thus, these studies were judged to be at low risk of bias for this domain. Communication with the authors of four studies (Akalin 2002; Heebner 2008; Moraska 2008; Pinar 2005), and use of alternation in Bahrami 2006, confirmed that group assignments were not concealed prior to allocation, therefore these studies were judged to be at high risk of bias for this domain. The remaining eight studies were rated as having an unclear risk of bias for this domain, as they either did not report any method to conceal the allocation sequence (Burke 2007; Field 2004; Janssen 2009; Tal‐Akabi 2000) or reported only some components of an effective allocation concealment method (for example, they reported that sealed, sequentially numbered envelopes were used, but did not report whether these were opaque) (Bardak 2009; Baysal 2006; Garfinkel 1998; Horng 2011).
Blinding
Participants were not blinded to the intervention they received in 12 studies (Akalin 2002; Bahrami 2006; Bardak 2009; Baysal 2006; Brininger 2007; Burke 2007; Davis 1998; Field 2004; Garfinkel 1998; Heebner 2008; Horng 2011; Tal‐Akabi 2000), which led to these studies being rated at a high risk of performance bias. However, given the nature of the interventions delivered it is not surprising that patient blinding was uncommon across the studies. It was clear that four studies did successfully blind participants (Bialosky 2009; Janssen 2009; Moraska 2008; Pinar 2005), so these studies were judged to be at low risk of performance bias. This was done by either delivering a "sham" (placebo) intervention (Bialosky 2009) or not informing participants of the alternative treatments offered to the other group (Janssen 2009; Moraska 2008; Pinar 2005). Nine studies (Bardak 2009; Baysal 2006; Bialosky 2009; Burke 2007; Davis 1998; Horng 2011; Janssen 2009; Pinar 2005; Tal‐Akabi 2000) blinded the outcome assessors who measured objective outcomes such as grip strength and nerve conduction studies, and thus were judged to be at low risk of detection bias. Blinding of outcome assessors of objective outcomes was unclear in two studies (Bahrami 2006; Field 2004) and clearly not done in five studies (Akalin 2002; Brininger 2007; Garfinkel 1998; Heebner 2008; Moraska 2008), which were judged to be at high risk for this domain.
Incomplete outcome data
Ten studies were judged as being at low risk of bias for completeness of outcome data at 12 weeks or less (Akalin 2002; Bahrami 2006; Bardak 2009; Baysal 2006; Brininger 2007; Burke 2007; Janssen 2009Moraska 2008; Pinar 2005; Tal‐Akabi 2000). Three studies were rated as being at unclear risk of bias for this domain (Bialosky 2009; Field 2004; Horng 2011), while three studies were judged to be at high risk of bias for this domain (Davis 1998; Garfinkel 1998; Heebner 2008). In the study conducted by Davis 1998, a greater proportion of participants dropped out of the chiropractic group and these losses may have been associated with this intervention itself. In the study conducted by Garfinkel 1998, four participants randomised to the intervention group either dropped out or were excluded from the analysis and five participants in the control group dropped out or were excluded from the analysis, but the authors provided no reasons for this and did not indicate why some participants were excluded post‐randomisation. Heebner 2008 reported a 50% attrition rate during the study period, which was likely influenced by the interventions delivered.
Only four studies could be assessed for completeness of outcome data collected at more than three months after treatment ended (Akalin 2002; Bahrami 2006; Bardak 2009; Baysal 2006). Two studies were judged to be at low risk of bias for this domain (Akalin 2002; Bardak 2009), Bahrami 2006 was judged to be at unclear risk of bias on this domain, and Baysal 2006 was judged to be at high risk of bias for this domain.
Selective reporting
Only eight studies (Akalin 2002; Baysal 2006; Bialosky 2009; Davis 1998; Janssen 2009; Moraska 2008; Pinar 2005; Tal‐Akabi 2000) were judged to be free of selective outcome reporting, and thus were rated as being at low risk of reporting bias. Judgements were based on comparing outcomes specified in the Methods section to those reported in the Results section of the publication. Seven studies (Bardak 2009; Brininger 2007; Burke 2007; Field 2004; Garfinkel 1998; Heebner 2008; Horng 2011) were judged to be at high risk of reporting bias as some outcomes specified in the Methods section were only partially reported in the Results section (for example, the authors only reported the statistical significance for outcomes, or reported means for each group without measures of variability) and relevant data were not provided by the authors on request. It was unclear if any additional measured outcomes were not reported in Bahrami 2006. No protocols for any of the included studies were identified, and a trial registry entry was only identified for Brininger 2007, which limits our assessment of selective reporting.
Other potential sources of bias
All studies were judged as being at low risk of bias for this domain.
Effects of interventions
See: Table 1
Exercise or mobilisation (single intervention) versus no treatment
Two trials compared an exercise or mobilisation intervention (delivered as a single intervention) to a no treatment control. Tal‐Akabi 2000 compared neurodynamic mobilisation to no treatment, and carpal bone mobilisation to no treatment, for a period of three weeks. Field 2004 compared soft tissue massage to no treatment for a period of four weeks.
Primary outcomes
1) Short‐term overall improvement (three months or less)
Reported as an outcome in Tal‐Akabi 2000 but not Field 2004
At the end of three weeks treatment, Tal‐Akabi 2000 found that all participants receiving neurodynamic mobilisation and none of the participants in the no treatment group reported improvement in overall pain relief (risk ratio (RR) 15.00, 95% confidence interval (CI) 1.02 to 220.92; Analysis 1.1). Further, Tal‐Akabi 2000 found that at the end of three weeks treatment, all participants receiving carpal bone mobilisation and none of the participants in the no treatment group reported improvement in overall pain relief (RR 15.00, 95% CI 1.02 to 220.92; Analysis 2.1). However, owing to the small sample size the precision of both effect estimates is very low. Given that participants were not blinded to treatment in this study, these results should be interpreted with caution, as it is possible that participants' expectations about the benefits of neurodynamic or carpal bone mobilisation may have biased their self‐reported assessment of overall pain relief.
1.1. Analysis.

Comparison 1 NERVE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 1 Short‐term overall improvement (3 months or less).
2.1. Analysis.

Comparison 2 CARPAL BONE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 1 Short‐term overall improvement (3 months or less).
Secondary outcomes
1) Adverse effects
Not reported as an outcome in Field 2004 or Tal‐Akabi 2000.
2) Short‐term improvement in CTS symptoms (three months or less)
Reported as an outcome in Field 2004 and Tal‐Akabi 2000.
Field 2004 reported the mean number of physician‐assessed carpal tunnel symptoms, mean number of participants with positive Tinel's and Phalen's tests, mean pain on a visual analogue scale (VAS), mean state anxiety, and mean self‐reported depression at baseline and at the end of four weeks treatment. However, no measures of variability (for example, standard deviations (SD) or 95% CIs) were reported, and the number of participants assigned to each group was unclear. Attempts to obtain relevant data from the trial authors were unsuccessful. Thus, no data appropriate for meta‐analysis were available. According to the study report, the soft tissue mobilisation group experienced fewer carpal tunnel symptoms, improvement on the Phalen's test and nerve conduction studies, and lower anxiety and depression, but the results were not statistically significant. Given the high risk of reporting bias, these data must be interpreted with caution.
In Tal‐Akabi 2000, participants rated their pain (VAS, range 0 to 10), and at the end of three weeks treatment; mean pain was 0.57 points lower in wrists receiving neurodynamic mobilisation compared to wrists in the no treatment group (mean difference (MD) ‐0.57, 95% CI ‐1.73 to 0.59; Analysis 1.2); however, the 95% CIs incorporate effects in either direction. Tal‐Akabi 2000 also found that at the end of three weeks treatment, pain was 1.43 points lower on a 0 to 10 VAS in wrists assigned to carpal bone mobilisation compared to wrists assigned to no treatment (MD ‐1.43, 95% CI ‐2.19 to ‐0.67; Analysis 2.2). However, this effect may be biased by the lack of patient blinding in this study.
1.2. Analysis.
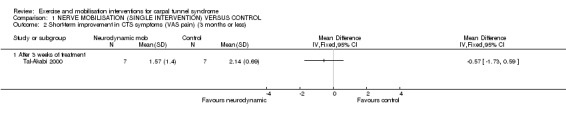
Comparison 1 NERVE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 2 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less).
2.2. Analysis.

Comparison 2 CARPAL BONE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 2 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less).
3) Short‐term improvement in functional ability or health‐related quality of life (three months or less)
Reported as an outcome in Field 2004 and Tal‐Akabi 2000.
Field 2004 reported mean perceived grip strength at the end of four weeks treatment; however, no measures of variability were reported and the number of participants assigned to each group was unclear, limiting analysis of these data. The trial authors reported in the publication an increase in grip strength in the massage therapy group immediately after the first and last massage therapy sessions and by the end of the study but it is unclear whether this was statistically significantly different to the control group. Given the high risk of reporting bias, these data must be interpreted with caution.
At the end of three weeks treatment, Tal‐Akabi 2000 found more wrists receiving neurodynamic mobilisation had improved self‐reported hand function (RR 9.00, 95% CI 0.59 to 137.65; Analysis 1.3) and improved upper limb tension (RR 11.00, 95% CI 0.72 to 167.68; Analysis 1.4) compared to wrists in the no treatment group. Also, wrists receiving neurodynamic mobilisation had better mean active wrist flexion (degrees) (MD 7.28, 95% CI ‐3.33 to 17.89; Analysis 1.5) and mean active wrist extension (degrees) (MD 6.00, 95% CI ‐4.56 to 16.56; Analysis 1.6) compared to wrists in the no treatment group. Tal‐Akabi 2000 also found more wrists receiving carpal bone mobilisation group had improved hand function (RR 11.00, 95% CI 0.74 to 163.49; Analysis 2.3) and improved upper limb tension (RR 9.00, 95% CI 0.57 to 141.13; Analysis 2.4) at the end of three weeks treatment compared to wrists no treatment group. Wrists receiving carpal bone mobilisation also had better mean active wrist flexion (degrees) (MD 6.43, 95% CI ‐4.50 to 17.36; Analysis 2.5) and mean active wrist extension (degrees) (MD 6.86, 95% CI ‐1.90 to 15.62; Analysis 2.6). While all these effect estimates suggest that neurodynamic mobilisation and carpal bone mobilisation are superior to no treatment, the wide 95% CIs mean that negative effects of these interventions cannot be ruled out.
1.3. Analysis.

Comparison 1 NERVE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 3 Short‐term improvement in functional ability (hand function) (3 months or less).
1.4. Analysis.

Comparison 1 NERVE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 4 Short‐term improvement in functional ability (upper limb tension test) (3 months or less).
1.5. Analysis.

Comparison 1 NERVE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 5 Short‐term improvement in functional ability (active wrist flexion (degrees)) (3 months or less).
1.6. Analysis.

Comparison 1 NERVE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 6 Short‐term improvement in functional ability (active wrist extension (degrees)) (3 months or less).
2.3. Analysis.

Comparison 2 CARPAL BONE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 3 Short‐term improvement in functional ability (hand function) (3 months or less).
2.4. Analysis.

Comparison 2 CARPAL BONE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 4 Short‐term improvement in functional ability (upper limb tension test) (3 months or less).
2.5. Analysis.

Comparison 2 CARPAL BONE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 5 Short‐term improvement in functional ability (active wrist flexion (degrees)) (3 months or less).
2.6. Analysis.

Comparison 2 CARPAL BONE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 6 Short‐term improvement in functional ability (active wrist extension (degrees)) (3 months or less).
4) Short‐term improvement in neurophysiologic parameters (three months or less)
Reported as an outcome in Field 2004 but not Tal‐Akabi 2000.
Field 2004 reported mean nerve conduction velocity and mean median nerve peak latency at the end of four weeks treatment (though it is unclear whether sensory or motor conduction velocity and peak latency were measured). However, no measures of variability were reported and the number of participants assigned to each group was unclear. The trialists reported that participants receiving soft tissue mobilisation had improvement in nerve conduction studies, though these results were not statistically significant. Given the high risk of reporting bias, these data must be interpreted with caution.
5) Long‐term improvement in CTS symptoms (more than three months)
Not reported as an outcome in Field 2004 or Tal‐Akabi 2000.
6) Long‐term improvement in functional ability or health‐related quality of life (more than three months)
Not reported as an outcome in Field 2004 or Tal‐Akabi 2000.
7) Need for surgery
Reported as an outcome in Tal‐Akabi 2000 but not Field 2004.
At the end of three weeks treatment, Tal‐Akabi 2000 found two participants receiving neurodynamic mobilisation and six in the no treatment group returned to their originally planned carpal tunnel release surgery (RR 0.33, 95% CI 0.10 to 1.12; Analysis 1.7). At the end of three weeks treatment, Tal‐Akabi 2000 also found one participant receiving carpal bone mobilisation and six in the no treatment group returned to their originally planned carpal tunnel release surgery (RR 0.17, 95% CI 0.03 to 1.05; Analysis 2.7). However, the 95% CIs for both risk ratios incorporate both positive and negative effects for this intervention.
1.7. Analysis.

Comparison 1 NERVE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 7 Need for surgery.
2.7. Analysis.

Comparison 2 CARPAL BONE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL, Outcome 7 Need for surgery.
Different mobilisation interventions (single interventions)
Three trials compared one mobilisation intervention to another. Tal‐Akabi 2000 compared neurodynamic mobilisation versus carpal bone mobilisation delivered for three weeks. Burke 2007 compared instrument‐assisted soft tissue mobilisation versus standard soft tissue mobilisation delivered for six weeks. Moraska 2008 compared targeted CTS soft tissue massage versus general soft tissue massage for six weeks. Due to the heterogeneity of interventions, no pooling of data was performed and a narrative summary is provided.
Primary outcomes
1) Short‐term overall improvement (three months or less)
Reported as an outcome in Burke 2007 and Tal‐Akabi 2000 but not in Moraska 2008
Burke 2007 reported patient satisfaction with treatment three months after treatment ended using a 0 to 5 scale (0 = very dissatisfied; 5 = very satisfied). The authors reported the number of participants in each group who were 'very dissatisfied', 'dissatisfied', 'neutral', 'satisfied' or 'very satisfied' with treatment. Based on these data, we dichotomised participants into those who reported being 'satisfied' or 'very satisfied' with treatment and those who did not. At three months after treatment ended, the chance of being 'satisfied' or 'very satisfied' was 24% higher for participants receiving instrument‐assisted soft tissue mobilisation (STM) compared with participants receiving standard STM on this outcome (RR 1.24, 95% CI 0.89 to 1.75; Analysis 4.1). However, the wide 95% CI incorporates effects in either direction.
4.1. Analysis.

Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 1 Short‐term overall improvement (3 months or less).
In Tal‐Akabi 2000, at the end of three weeks treatment, all participants receiving neurodynamic mobilisation and all participants receiving carpal bone mobilisation reported overall improvement in pain relief (RR 1.00, 95% CI 0.78 to 1.29; Analysis 3.1).
3.1. Analysis.

Comparison 3 NEURODYNAMIC VS CARPAL BONE MOBILISATION, Outcome 1 Short‐term overall improvement (3 months or less).
Secondary outcomes
1) Adverse effects
Reported as an outcome in Burke 2007 but not in Moraska 2008 or Tal‐Akabi 2000.
Burke 2007 only reported in the publication that "Although many patients reported adverse effects of soreness and bruising, most of these were relatively mild of short duration. However, 1 patient withdrew from participation because of profound bruising and swelling of the treated forearm‐wrist‐hand after the first GISTM (Graston Technique instrument‐assisted soft tissue mobilisation) treatment. The patient was treated medically for the swelling. However, these acute effects resolved within a week without any long‐term adverse effects."
2) Short‐term improvement in CTS symptoms (three months or less)
Reported as an outcome in Burke 2007, Moraska 2008 and Tal‐Akabi 2000.
Burke 2007 found wrists receiving instrument‐assisted STM had a VAS pain score which was 5.6 mm lower on a 0 to 100 mm scale immediately after six weeks treatment (MD ‐5.60, 95% CI ‐19.68 to 8.48; Analysis 4.2) and 24.5 mm lower three months after treatment ended (MD ‐24.50, 95% CI ‐43.43 to ‐5.57; Analysis 4.2) compared to wrists receiving standard STM. Further, CTS symptoms were 0.10 points lower on the Levine questionnaire immediately after six to eight weeks treatment (MD ‐0.10, 95% CI ‐0.58 to 0.38; Analysis 4.3) and 0.40 points lower on the Levine questionnaire (Levine 1993) three months after treatment ended (MD ‐0.40, 95% CI ‐0.90 to 0.10; Analysis 4.3). Except for VAS pain score at three months post‐treatment, the 95% CIs for all these effect estimates do not exclude the possibility of no difference between groups or effects that favour either treatment group. Additionally, the lack of patient blinding means these results should be interpreted with caution as patient expectations about the efficacy of the instrument‐assisted STM may have biased their self‐reported responses. Burke 2007 also assessed pattern of symptoms using the Katz hand diagram (Katz 1990), two‐point discrimination and pressure sensitivities of the first three digits of each hand, and presence of a positive Phalen's or Tinel's test, but no summary data were available for meta‐analysis.
4.2. Analysis.

Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 2 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less).
4.3. Analysis.

Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 3 Short‐term improvement in CTS symptoms (Levine) (3 months or less).
Moraska 2008 assessed CTS symptoms using the Levine questionnaire (Levine 1993) and provided summary data (means, SDs and standard errors (SEs)) on request as they were not reported in the study publication. The authors found wrists receiving a targeted CTS massage had a lower (better) symptom score after the seventh of 12 massages (week four of treatment) (MD ‐0.74, 95% CI ‐2.72 to 1.24; Analysis 5.1), a higher (worse) symptom score after the 11th of 12 massages (week six of treatment) (MD 0.10, 95% CI ‐2.20 to 2.40; Analysis 5.1), and a lower (better) symptom score at four weeks post‐treatment (MD ‐1.41, 95% CI ‐4.09 to 1.27; Analysis 5.1). However, the low precision of the 95% CIs means that opposite effects of treatment cannot be ruled out.
5.1. Analysis.

Comparison 5 TARGETED CTS SOFT TISSUE MASSAGE VERSUS GENERAL SOFT TISSUE MASSAGE, Outcome 1 Short‐term improvement in CTS symptoms (Levine) (3 months or less).
Participants in Tal‐Akabi 2000 rated their pain (VAS 0 to 10) and at the end of three weeks treatment, wrists receiving carpal bone mobilisation had pain which was 0.86 points lower than wrists receiving neurodynamic mobilisation (MD 0.86, 95% CI ‐0.32 to 2.04; Analysis 3.2). However, the low precision of all these effect estimates means the results cannot be interpreted as one intervention being of greater benefit than the other.
3.2. Analysis.

Comparison 3 NEURODYNAMIC VS CARPAL BONE MOBILISATION, Outcome 2 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less) Symptoms.
3) Short‐term improvement in functional ability or health‐related quality of life (three months or less)
Reported as an outcome in Burke 2007, Moraska 2008 and Tal‐Akabi 2000.
Immediately after six to eight weeks treatment, Burke 2007 found that wrists receiving instrument‐assisted STM had better self‐reported functional ability (as assessed using the Levine questionnaire) (MD ‐0.10, 95% CI ‐0.67 to 0.47; Analysis 4.4), better grip strength (kg) (MD 0.30, 95% CI ‐6.43 to 7.03; Analysis 4.5), better pinch strength opposition (kg) (MD 0.40, 95% CI ‐0.80 to 1.60; Analysis 4.6), worse pinch strength key (kg) (MD ‐0.50, 95% CI ‐2.04 to 1.04; Analysis 4.7), better extension range of movement (degrees) (MD 1.70, 95% CI ‐9.22 to 12.62; Analysis 4.8) and worse flexion range of movement (degrees) (MD ‐1.60, 95% CI ‐8.29 to 5.09; Analysis 4.9) compared to wrists receiving standard STM. Three months post‐treatment, Burke 2007 found wrists receiving instrument‐assisted STM had better functional ability (assessed using the Levine questionnaire (Levine 1993)) (MD ‐0.10, 95% CI ‐0.69 to 0.49; Analysis 4.4), better grip strength (kg) (MD 0.50, 95% CI ‐5.35 to 6.35; Analysis 4.5), better pinch strength opposition (kg) (MD 0.40, 95% CI ‐0.74 to 1.54; Analysis 4.6), better pinch strength key (kg) (MD 0.60, 95% CI ‐0.47 to 1.67; Analysis 4.7), worse extension range of movement (degrees) (MD ‐1.90, 95% CI ‐10.41 to 6.61; Analysis 4.8) and worse flexion range of movement (degrees) (MD ‐1.00, 95% CI ‐8.48 to 6.48; Analysis 4.9) compared to wrists receiving standard STM. However, the low precision of all these effect estimates means the results cannot be interpreted as one intervention being of greater benefit than the other.
4.4. Analysis.

Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 4 Short‐term improvement in functional ability (Levine) (3 months or less).
4.5. Analysis.

Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 5 Short‐term improvement in functional ability (grip strength) (3 months or less).
4.6. Analysis.

Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 6 Short‐term improvement in functional ability (pinch strength opposition) (3 months or less).
4.7. Analysis.

Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 7 Short‐term improvement in functional ability (pinch strength key) (3 months or less).
4.8. Analysis.

Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 8 Short‐term improvement in functional ability (extension range of movement) (3 months or less).
4.9. Analysis.

Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 9 Short‐term improvement in functional ability (flexion range of movement) (3 months or less).
Moraska 2008 used a number of instruments to assess functional ability and provided summary numerical data (means, SDs and SEs) on request as these were not reported in the study publication. Wrists receiving the targeted CTS massage were found to have a lower (better) self‐rated functional status score as assessed using the Levine questionnaire (Levine 1993) after the 7th of 12 massages (week four of treatment) (MD ‐0.59, 95% CI ‐2.55 to 1.37; Analysis 5.2), a higher (worse) functional status score after the 11th of 12 massages (week six of treatment) (MD 0.11, 95% CI ‐1.77 to 1.99; Analysis 5.2), and a lower (better) functional status score at four weeks post‐treatment (MD ‐1.69, 95% CI ‐3.74 to 0.36; Analysis 5.2) compared to wrists receiving a general massage. The 95% CIs for these effect estimates incorporate both increases and decreases in functional status scores between the groups. Isometric grip strength (kg) was better in wrists receiving the targeted CTS massage after the 7th of 12 massages (week four of treatment) (MD 2.94, 95% CI 1.03 to 4.85; Analysis 5.3), after the 11th of 12 massages (week six of treatment) (MD 2.86, 95% CI ‐0.001 to 5.72; Analysis 5.3), and at four weeks post‐treatment (MD 3.14, 95% CI 0.57 to 5.71; Analysis 5.3) compared to wrists receiving general massage. Further, isometric pinch strength (kg) was better in wrists receiving the targeted CTS massage after the 7th of 12 massages (week four of treatment) (MD 0.90, 95% CI 0.05 to 1.75; Analysis 5.4), after the 11th of 12 massages (week six of treatment) (MD 1.02, 95% CI 0.18 to 1.86; Analysis 5.4) and at four weeks post‐treatment (MD 0.75, 95% CI ‐0.13 to 1.63; Analysis 5.4). However, the authors reported that evaluators were not blinded to subject group assignment, so these results should be interpreted with caution as the evaluators pre‐conceived beliefs about the effectiveness of the targeted CTS massage may have influenced their assessment of grip and pinch strength. Additionally, functional ability as assessed using the Grooved Pegboard test was better in wrists receiving the targeted CTS massage after the 7th of 12 massages (week four of treatment) (MD 1.10, 95% CI ‐5.13 to 7.33; Analysis 5.5) and after the 11th of 12 massages (week six of treatment) (MD 2.98, 95% CI ‐4.14 to 10.10; Analysis 5.5) compared to wrists receiving general massage, but was worse than wrists receiving general massage at four weeks post‐treatment (MD ‐1.31, 95% CI ‐9.95 to 7.33; Analysis 5.5). The 95% CIs for these effect estimates were imprecise and do not rule out opposite effects of the interventions.
5.2. Analysis.

Comparison 5 TARGETED CTS SOFT TISSUE MASSAGE VERSUS GENERAL SOFT TISSUE MASSAGE, Outcome 2 Short‐term improvement in functional ability (Levine) (3 months or less).
5.3. Analysis.

Comparison 5 TARGETED CTS SOFT TISSUE MASSAGE VERSUS GENERAL SOFT TISSUE MASSAGE, Outcome 3 Short‐term improvement in functional ability (isometric grip strength) (3 months or less).
5.4. Analysis.
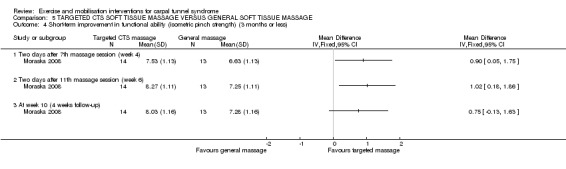
Comparison 5 TARGETED CTS SOFT TISSUE MASSAGE VERSUS GENERAL SOFT TISSUE MASSAGE, Outcome 4 Short‐term improvement in functional ability (isometric pinch strength) (3 months or less).
5.5. Analysis.

Comparison 5 TARGETED CTS SOFT TISSUE MASSAGE VERSUS GENERAL SOFT TISSUE MASSAGE, Outcome 5 Short‐term improvement in functional ability (Grooved pegboard test) (3 months or less).
In Tal‐Akabi 2000, more wrists receiving carpal bone mobilisation had improved self‐reported hand function (RR 0.80, 95% CI 0.41 to 1.56; Analysis 3.3), while more wrists receiving neurodynamic mobilisation had improved upper limb tension (RR 1.25, 95% CI 0.56 to 2.77; Analysis 3.4) at the end of three weeks treatment. Further, wrists receiving neurodynamic mobilisation had better mean active wrist flexion (degrees) (MD 0.85, 95% CI ‐10.83 to 12.53; Analysis 3.5), while wrists receiving carpal bone mobilisation had better mean active wrist extension (degrees) (MD ‐0.86, 95% CI ‐9.26 to 7.54; Analysis 3.6). However, the 95% CIs for these effect estimates incorporate both positive and negative effects of these interventions.
3.3. Analysis.
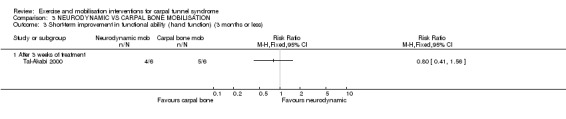
Comparison 3 NEURODYNAMIC VS CARPAL BONE MOBILISATION, Outcome 3 Short‐term improvement in functional ability (hand function) (3 months or less).
3.4. Analysis.

Comparison 3 NEURODYNAMIC VS CARPAL BONE MOBILISATION, Outcome 4 Short‐term improvement in functional ability (upper limb tension test) (3 months or less).
3.5. Analysis.
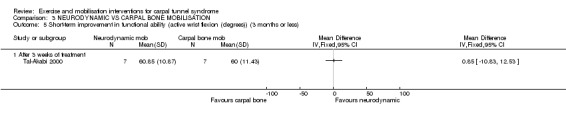
Comparison 3 NEURODYNAMIC VS CARPAL BONE MOBILISATION, Outcome 5 Short‐term improvement in functional ability (active wrist flexion (degrees)) (3 months or less).
3.6. Analysis.

Comparison 3 NEURODYNAMIC VS CARPAL BONE MOBILISATION, Outcome 6 Short‐term improvement in functional ability (active wrist extension (degrees)) (3 months or less).
4) Short‐term improvement in neurophysiologic parameters (three months or less)
Reported as an outcome in Burke 2007 but not in Moraska 2008 or Tal‐Akabi 2000.
In Burke 2007, wrists receiving instrument‐assisted STM had median nerve distal sensory latency which was 0.39 ms longer (MD 0.39, 95% CI ‐0.03 to 0.81; Analysis 4.10) and median nerve distal motor latency which was no shorter or longer (MD 0.00, 95% CI ‐0.90 to 0.90; Analysis 4.11) than wrists receiving standard STM immediately after six weeks treatment. At three months post‐treatment, wrists receiving instrument‐assisted STM had median nerve distal sensory latency which was 0.22 ms longer (MD 0.22, 95% CI ‐0.11 to 0.55; Analysis 4.10) and median nerve distal motor latency which was 0.17 millseconds longer (MD 0.17, 95% CI ‐0.77 to 1.11; Analysis 4.11) than wrists receiving standard STM. The 95% CIs for these effect estimates incorporate both increases and decreases in these nerve conduction studies.
4.10. Analysis.

Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 10 Short‐term improvement in distal sensory latency (ms) (3 months or less).
4.11. Analysis.
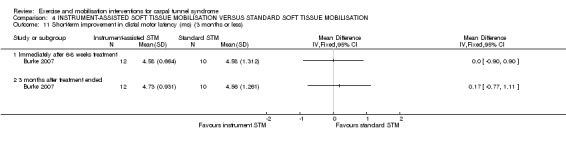
Comparison 4 INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION, Outcome 11 Short‐term improvement in distal motor latency (ms) (3 months or less).
5) Long‐term improvement in CTS symptoms (more than three months)
Not reported as an outcome in Burke 2007, Moraska 2008 or Tal‐Akabi 2000.
6) Long‐term improvement in functional ability or health‐related quality of life (more than three months)
Not reported as an outcome in Burke 2007, Moraska 2008 or Tal‐Akabi 2000.
7) Need for surgery
Reported as an outcome in Tal‐Akabi 2000 but not in Burke 2007 or Moraska 2008.
At the end of three weeks treatment, Tal‐Akabi 2000 reported two participants receiving neurodynamic mobilisation and one participant receiving carpal bone mobilisation returned to their originally planned carpal tunnel release surgery, though the precision of the effect estimate was low and does not exclude no difference between groups (RR 2.00, 95% CI 0.23 to 17.34; Analysis 3.7).
3.7. Analysis.

Comparison 3 NEURODYNAMIC VS CARPAL BONE MOBILISATION, Outcome 7 Need for surgery.
Nerve mobilisation (as part of multiple interventions) versus control
Nine trials compared nerve mobilisation as part of a multi‐component intervention to another non‐surgical intervention (Akalin 2002; Bahrami 2006; Bardak 2009; Baysal 2006; Bialosky 2009; Brininger 2007; Heebner 2008; Horng 2011; Pinar 2005). Akalin 2002 and Bahrami 2006 both compared nerve and tendon gliding exercises plus splint versus splint alone for four weeks. Bardak 2009 compared nerve and tendon gliding exercises for six weeks versus nerve and tendon gliding exercises plus split plus steroid injection versus splint plus steroid injection. Baysal 2006 compared nerve and tendon gliding exercises plus splint plus therapeutic ultrasound versus nerve and tendon gliding exercises plus splint only versus therapeutic ultrasound plus splint only for three weeks. Bialosky 2009 compared neurodynamic technique plus splint versus "sham" neurodynamic technique plus splint for three weeks. Brininger 2007 compared nerve and tendon gliding exercises plus fabricated neutral wrist and metacarpophalangeal (MCP) splint versus fabricated neutral wrist and MCP splint alone for four weeks, and also nerve and tendon gliding exercise plus off‐the‐shelf wrist cock‐up splint versus off‐the‐shelf wrist cock‐up splint alone for four weeks. Heebner 2008 compared neurodynamic mobilisation exercises plus standard care consisting of patient education, splinting, and tendon gliding exercises versus standard care alone for six months. Horng 2011 compared nerve gliding exercises plus splint plus paraffin therapy (superficial heat therapy) versus tendon gliding exercises plus splint plus paraffin therapy versus splint plus paraffin therapy alone, each for eight weeks. Pinar 2005 compared nerve gliding exercises plus splint plus activity modification versus splint plus activity modification for 10 weeks.
While there are three possible comparisons in Bardak 2009 (nerve and tendon gliding exercises alone versus nerve and tendon gliding exercises plus splint plus steroid injection; nerve and tendon gliding exercises alone versus splint and steroid injection; and nerve and tendon gliding exercises plus splint plus steroid injection versus splint plus steroid injection), only the two comparisons where nerve and tendon gliding exercises were delivered to one group were compared in this review (that is no data on the comparison between nerve and tendon gliding exercises alone versus nerve and tendon gliding exercises plus splint plus steroid injection were entered into RevMan 5). Similarly, while there are three possible comparisons in Baysal 2006 (nerve and tendon gliding exercises plus splint plus therapeutic ultrasound versus nerve and tendon gliding exercises plus splint; nerve and tendon gliding exercises plus splint plus therapeutic ultrasound versus therapeutic ultrasound plus splint; nerve and tendon gliding exercises plus splint versus therapeutic ultrasound plus splint), only the two comparisons where nerve and tendon gliding exercises were delivered to one group were compared (that is, no data on the comparison between nerve and tendon gliding exercises plus splint plus therapeutic ultrasound versus nerve and tendon gliding exercises plus splint were entered into RevMan 5).
As reported in more detail in the Included studies section and Characteristics of included studies tables, some or all participants had bilateral CTS in the studies conducted by Akalin 2002, Bahrami 2006, Baysal 2006, Horng 2011, and Pinar 2005. It was clear that the correlation between wrists was accounted for in the analysis only in the study by Horng 2011 for the neurophysiologic parameter outcomes. Attempts to retrieve individual wrist outcome data for these seven studies were unsuccessful. Therefore, apart from the neurophysiologic parameter outcome data in Horng 2011, all other outcome data reported in these studies may be invalid due to a possible unit of analysis error. Without access to the individual wrist data, and without being able to estimate parameters such as the intraclass correlation coefficient from other studies included in the review, we did not attempt to adjust the results of these seven studies. We have included the outcome data as reported by the trialists, but emphasise that results of these studies should be interpreted with caution, as the possible lack of adjustment may have produced either overly wide 95%CIs or overly narrow 95%CIs with artificially smaller P values (Higgins 2011c).
Only the interventions used in Akalin 2002, Bahrami 2006 and Brininger 2007 were deemed sufficiently similar in terms of intervention components and duration, but the limited reporting of data in Brininger 2007, and the lack of clarification regarding whether the correlation between wrists in bilateral CTS participants was accounted for in the analysis in Akalin 2002 and Bahrami 2006, meant that pooling data was not possible.
Primary outcomes
1) Short‐term overall improvement (three months or less)
Reported as an outcome in Brininger 2007 and Pinar 2005, but not in Akalin 2002, Bahrami 2006, Bardak 2009, Baysal 2006, Bialosky 2009, Heebner 2008 or Horng 2011.
Brininger 2007 reported measuring the percentage of participants reporting 'no to occasional symptoms' at four weeks after treatment finished; however, these data were not reported per intervention group, so we were unable to calculate an effect estimate.
Pinar 2005 reported the number of wrists with no pathologic finding on median and ulnar nerve distal sensory latency assessment at the end of 10 weeks treatment. More wrists receiving nerve gliding exercises plus splint plus activity modification had no pathologic finding (RR 1.26, 95% CI 0.69 to 2.30; Analysis 16.1) than wrists receiving splint plus activity modification alone, however the precision of this effect estimate was low and does not rule out a negative effect of the nerve gliding exercises plus splint plus activity modification intervention.
16.1. Analysis.

Comparison 16 NERVE GLIDING EXERCISES PLUS SPLINT PLUS ACTIVITY MODIFICATION VERSUS SPLINT PLUS ACTIVITY MODIFICATION, Outcome 1 Short term overall improvement (no pathological finding on NCS) (3 months or less).
Secondary outcomes
1) Adverse effects
Not reported as an outcome in Akalin 2002, Bahrami 2006, Bardak 2009, Baysal 2006, Bialosky 2009, Brininger 2007, Heebner 2008, Horng 2011 or Pinar 2005.
2) Short‐term improvement in CTS symptoms (three months or less)
Reported as an outcome in Akalin 2002, Bahrami 2006, Bardak 2009, Baysal 2006, Bialosky 2009, Brininger 2007, Heebner 2008, Horng 2011 and Pinar 2005.
In Akalin 2002, receiving nerve and tendon gliding exercises plus splint decreased the risk of having a positive Phalen's test by 37% compared to splint alone (RR 0.63, 95% CI 0.25 to 1.55; Analysis 6.1) and decreased the risk of having a positive Tinel's test by 25% compared to splint alone (RR 0.75, 95% CI 0.33 to 1.72; Analysis 6.2) at eight weeks post‐treatment. Further, wrists receiving nerve and tendon gliding exercises plus splint had a lower (better) CTS symptoms status score (Levine 1993) compared to wrists receiving splint only (MD ‐3.68, 95% CI ‐8.56 to 1.20; Analysis 6.3). All of these outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
6.1. Analysis.

Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 1 Short‐term improvement in CTS symptoms (Phalen's sign) (3 months or less).
6.2. Analysis.

Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 2 Short‐term improvement in CTS symptoms (Tinel's sign) (3 months or less).
6.3. Analysis.

Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 3 Short‐term improvement in CTS symptoms (Levine) (3 months or less).
In Bahrami 2006, receiving nerve and tendon gliding exercises plus splint decreased the risk of having a positive Phalen's test by 17% compared to splint alone (RR 0.83, 95% CI 0.31 to 2.27; Analysis 6.1) and decreased the risk of having a positive Tinel's test by 37% compared to splint alone (RR 0.63, 95% CI 0.25 to 1.57; Analysis 6.2) at six weeks post‐treatment. In addition, wrists receiving nerve and tendon gliding exercises plus splint had a lower (better) CTS symptoms status score (Levine 1993) on average compared to wrists receiving splint only (MD ‐1.12, 95% CI ‐3.60 to 1.36; Analysis 6.3). The low precision of these effect estimates means that opposite effects are possible. Also, these results should be interpreted with caution as lack of blinding participants and outcome assessors may have exaggerated results. Further, all of these outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
Bardak 2009 found at the end of six weeks treatment that wrists receiving nerve and tendon gliding exercises only had a higher (worse) symptom total score compared to wrists receiving splint plus steroid injection (MD 2.31, 95% CI 1.59 to 3.03; Analysis 7.1). The authors also found that more wrists receiving nerve and tendon gliding exercises alone had a positive Tinel's test (RR 1.41, 95% CI 0.84 to 2.35; Analysis 7.2), Phalen's test (RR 1.23, 95% CI 0.83 to 1.82; Analysis 7.3), and compression test (RR 1.28, 95% CI 0.65 to 2.53; Analysis 7.4) than wrists receiving splint plus steroid injection, whereas, fewer wrists receiving nerve and tendon gliding exercises alone had a positive reverse Phalen's's sign (RR 1.02, 95% CI 0.59 to 1.79; Analysis 7.5). Further, at the end of six weeks treatment, wrists receiving nerve and tendon gliding exercises plus splint plus steroid injection had a lower (better) symptom total score compared to wrists receiving splint plus steroid injection (MD ‐0.50, 95% CI ‐1.29 to 0.29; Analysis 8.1), and more wrists receiving nerve and tendon gliding exercises plus splint plus steroid injection had a positive Tinel's test (RR 1.72, 95% CI 1.07 to 2.77; Analysis 8.2), Phalen's test (RR 1.17, 95% CI 0.78 to 1.75; Analysis 8.3), reverse Phalen's test (RR 1.24, 95% CI 0.75 to 2.08; Analysis 8.4), and compression test (RR 1.81, 95% CI 0.98 to 3.33; Analysis 8.5). However, the wide 95% CIs for all these effect estimates incorporate effects in either direction. Further, the lack of patient blinding suggests that these results should be interpreted with caution, as participants beliefs about the effectiveness of the intervention they received may have biased their self‐reported responses.
7.1. Analysis.

Comparison 7 NERVE AND TENDON GLIDING EXERCISES VERSUS SPLINT PLUS STEROID INJECTION, Outcome 1 Short‐term improvement in CTS symptoms (symptom total point) (3 months or less).
7.2. Analysis.

Comparison 7 NERVE AND TENDON GLIDING EXERCISES VERSUS SPLINT PLUS STEROID INJECTION, Outcome 2 Short‐term improvement in CTS symptoms (Tinel's test) (3 months or less).
7.3. Analysis.

Comparison 7 NERVE AND TENDON GLIDING EXERCISES VERSUS SPLINT PLUS STEROID INJECTION, Outcome 3 Short‐term improvement in CTS symptoms (Phalen's test) (3 months or less).
7.4. Analysis.
Comparison 7 NERVE AND TENDON GLIDING EXERCISES VERSUS SPLINT PLUS STEROID INJECTION, Outcome 4 Short‐term improvement in CTS symptoms (Compression test) (3 months or less).
7.5. Analysis.

Comparison 7 NERVE AND TENDON GLIDING EXERCISES VERSUS SPLINT PLUS STEROID INJECTION, Outcome 5 Short‐term improvement in CTS symptoms (Reverse Phalen's test) (3 months or less).
8.1. Analysis.

Comparison 8 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS STEROID INJECTION VERSUS SPLINT PLUS STEROID INJECTION, Outcome 1 Short‐term improvement in CTS symptoms (symptom total point) (3 months or less).
8.2. Analysis.

Comparison 8 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS STEROID INJECTION VERSUS SPLINT PLUS STEROID INJECTION, Outcome 2 Short‐term improvement in CTS symptoms (Tinel's test) (3 months or less).
8.3. Analysis.

Comparison 8 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS STEROID INJECTION VERSUS SPLINT PLUS STEROID INJECTION, Outcome 3 Short‐term improvement in CTS symptoms (Phalen's test) (3 months or less).
8.4. Analysis.

Comparison 8 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS STEROID INJECTION VERSUS SPLINT PLUS STEROID INJECTION, Outcome 4 Short‐term improvement in CTS symptoms (Reverse Phalen's test) (3 months or less).
8.5. Analysis.

Comparison 8 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS STEROID INJECTION VERSUS SPLINT PLUS STEROID INJECTION, Outcome 5 Short‐term improvement in CTS symptoms (Compression test) (3 months or less).
Baysal 2006 found wrists receiving nerve and tendon gliding exercises plus therapeutic ultrasound plus splint had pain which was 0.90 points lower on a 0 to 10 VAS at the end of three weeks treatment (MD 0.90, 95% CI ‐0.38 to 2.18; Analysis 9.1) and 1.70 points lower at eight weeks post‐treatment (MD 1.70, 95% CI 0.26 to 3.14; Analysis 9.1) compared to wrists receiving ultrasound plus splint. The authors also found that wrists receiving nerve and tendon gliding exercises plus therapeutic ultrasound plus splint had a mean symptom severity score (assessed using the Levine questionnaire (Levine 1993)) which was lower at the end of three weeks treatment (MD 1.00, 95% CI ‐3.53 to 5.53; Analysis 9.2) and eight weeks after treatment ended (MD 3.50, 95% CI ‐1.65 to 8.65; Analysis 9.2) compared to wrists receiving ultrasound plus splint. Fewer wrists receiving nerve and tendon gliding exercises plus therapeutic ultrasound plus splint had a positive Phalen's's sign at the end of three weeks treatment (RR 0.86, 95% CI 0.37 to 1.99; Analysis 9.3) and eight weeks post‐treatment (RR 1.00, 95% CI 0.36 to 2.79; Analysis 9.3), and a positive Tinel's test at the end of three weeks treatment (RR 1.00, 95% CI 0.36 to 2.79; Analysis 9.4) and eight weeks after treatment ended (RR 5.00, 95% CI 0.66 to 38.15; Analysis 9.4) compared to wrists receiving ultrasound plus splint. However the 95% CIs of these effect estimates incorporate positive and negative effects of these interventions. Wrists receiving nerve and tendon gliding exercises plus splint had higher (worse) VAS pain score (0 to 10) at the end of three weeks treatment (MD ‐1.10, 95% CI ‐2.59 to 0.39; Analysis 10.1) and at eight weeks post‐treatment (MD ‐0.10, 95% CI ‐1.87 to 1.67; Analysis 10.1), and higher (worse) mean symptom severity score (assessed using the Levine questionnaire (Levine 1993)) at the end of three weeks treatment (MD ‐2.60, 95% CI ‐7.81 to 2.61; Analysis 10.2) and eight weeks after treatment ended (MD ‐1.10, 95% CI ‐7.31 to 5.11; Analysis 10.2) compared to wrists receiving ultrasound plus splint. Further, wrists receiving nerve and tendon gliding exercises plus splint were more likely to have a positive Phalen's test at the end of three weeks treatment (RR 0.82, 95% CI 0.38 to 1.76; Analysis 10.3) and at eight weeks post‐treatment (RR 0.68, 95% CI 0.29 to 1.59; Analysis 10.3), and were more likely to have a positive Tinel's test at the end of three weeks treatment (RR 0.63, 95% CI 0.27 to 1.43; Analysis 10.4) but were less likely to have a positive Tinel's test at eight weeks post‐treatment (RR 1.07, 95% CI 0.41 to 2.79; Analysis 10.4) compared to wrists receiving ultrasound plus splint. All effect estimates had low precision, and their 95% CIs incorporate effects in either direction for the exercises plus splint intervention. All of these outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
9.1. Analysis.

Comparison 9 NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 1 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less).
9.2. Analysis.

Comparison 9 NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 2 Short‐term improvement in CTS symptoms (Levine) (3 months or less).
9.3. Analysis.

Comparison 9 NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 3 Short‐term improvement in CTS symptoms (Phalen's sign) (3 months or less).
9.4. Analysis.

Comparison 9 NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 4 Short‐term improvement in CTS symptoms (Tinel's sign) (3 months or less).
10.1. Analysis.

Comparison 10 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 1 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less).
10.2. Analysis.

Comparison 10 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 2 Short‐term improvement in CTS symptoms (Levine) (3 months or less).
10.3. Analysis.

Comparison 10 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 3 Short‐term improvement in CTS symptoms (Phalen's sign) (3 months or less).
10.4. Analysis.

Comparison 10 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 4 Short‐term improvement in CTS symptoms (Tinel's sign) (3 months or less).
Bialosky 2009 found that at the end of three weeks treatment, wrists receiving the neurodynamic technique plus splint had lower pressure pain as measured using a 100 mm mechanical visual analogue scale (MVAS) (MD ‐2.10, 95% CI ‐15.03 to 10.83; Analysis 11.1), lower temporal summation (measured using a 100 mm MVAS) (MD ‐12.30, 95% CI ‐30.28 to 5.68; Analysis 11.2), and lower usual pain as measured using a 101‐point numerical rating scale (MD ‐3.20, 95% CI ‐21.09 to 14.69; Analysis 11.3) when compared to wrists receiving the "sham" technique plus splint. However, wrists receiving the "sham" technique plus splint had less clinical pain (measured using a 100 mm MVAS) (MD 3.00, 95% CI ‐7.22 to 13.22; Analysis 11.4) and less thermal pain (measured using a 100 mm MVAS) (MD 0.60, 95% CI ‐1.57 to 2.77; Analysis 11.5) when compared to wrists receiving the neurodynamic technique plus splint. However, the precision of all these effect estimates was low, and opposite effects of treatment cannot be ruled out. The authors also measured sensitivity at the end of three weeks treatment but only reported that "Baseline to three‐week changes in sensation, as assessed by Semmes‐Weinstein monofilaments, did not differ by group when assessed for the thumb (P = 0.85), index finger (P = 0.68), and middle finger (P = 0.76)".
11.1. Analysis.

Comparison 11 NEURODYNAMIC TECHNIQUE PLUS SPLINT VERSUS "SHAM" NEURODYNAMIC TECHNIQUE PLUS SPLINT, Outcome 1 Short‐term improvement in CTS symptoms (pressure pain (MVAS)) (3 months or less).
11.2. Analysis.

Comparison 11 NEURODYNAMIC TECHNIQUE PLUS SPLINT VERSUS "SHAM" NEURODYNAMIC TECHNIQUE PLUS SPLINT, Outcome 2 Short‐term improvement in CTS symptoms (temporal summation (MVAS)) (3 months or less).
11.3. Analysis.

Comparison 11 NEURODYNAMIC TECHNIQUE PLUS SPLINT VERSUS "SHAM" NEURODYNAMIC TECHNIQUE PLUS SPLINT, Outcome 3 Short‐term improvement in CTS symptoms (usual pain (NRS)) (3 months or less).
11.4. Analysis.

Comparison 11 NEURODYNAMIC TECHNIQUE PLUS SPLINT VERSUS "SHAM" NEURODYNAMIC TECHNIQUE PLUS SPLINT, Outcome 4 Short‐term improvement in CTS symptoms (clinical pain (MVAS)) (3 months or less).
11.5. Analysis.

Comparison 11 NEURODYNAMIC TECHNIQUE PLUS SPLINT VERSUS "SHAM" NEURODYNAMIC TECHNIQUE PLUS SPLINT, Outcome 5 Short‐term improvement in CTS symptoms (thermal pain (MVAS)) (3 months or less).
Brininger 2007 reported they measured short‐term symptom severity using the Levine questionnaire (Levine 1993) at the end of the four‐week treatment period, and at four weeks follow‐up, but the only data reported were change from baseline to end of treatment or follow‐up for all intervention and control groups combined, and only F and P values were reported. Therefore, we were unable to calculate an effect estimate.
Heebner 2008 measured symptoms using the Levine questionnaire (Levine 1993) after one and six months of treatment, but no summary data (means and SDs) for each group were reported in the publication. However, summary data following one month of treatment were obtained through personal communication with the authors. After one month of treatment there was no difference in symptom severity score between wrists receiving nerve and tendon gliding exercises plus splint plus education and wrists receiving tendon gliding exercises plus splint plus education (MD 0.00, 95% CI ‐0.69 to 0.69; Analysis 12.1).
12.1. Analysis.

Comparison 12 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS EDUCATION VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS EDUCATION, Outcome 1 Short‐term improvement in CTS symptoms (Levine symptom severity score) (3 months or less).
Two months after treatment ended, Horng 2011 found participants receiving nerve gliding exercises plus splint plus paraffin therapy had change from baseline in pain which was 6.70 points worse on a 0 to 100 VAS scale compared to participants receiving splint plus paraffin therapy (MD 6.70, 95% CI ‐8.48 to 21.88; Analysis 13.1), and 9.20 points worse compared to participants receiving tendon gliding exercises plus splint plus paraffin therapy (MD 9.20, 95% CI ‐4.75 to 23.15; Analysis 14.1). Change in symptom severity score (Levine 1993) for participants receiving nerve gliding exercises plus splint plus paraffin therapy from baseline to two months was 0.30 points worse compared to participants receiving splint plus paraffin therapy (MD 0.30, 95% CI ‐0.10 to 0.70; Analysis 13.2), and 0.40 points worse compared to participants receiving tendon gliding exercises plus splint plus paraffin therapy (MD 0.40, 95% CI ‐0.06 to 0.86; Analysis 14.2). Further, participants receiving tendon gliding exercises plus splint plus paraffin therapy had a change from baseline in pain (VAS 0 to 100) which was 2.5 points better (MD ‐2.50, 95% CI ‐19.65 to 14.65; Analysis 15.1), and in symptom severity score which was 0.10 points better (MD ‐0.10, 95% CI ‐0.57 to 0.37; Analysis 15.2) compared to participants receiving splint plus paraffin therapy. All of these outcome data were analysed at the participant‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become narrower than those reported. The authors also measured the number of participants with a positive Phalen's test and a positive Tinel's test. However, summary data for each group were not reported; the authors only reported that no statistically significant difference was found between the three groups on these outcomes.
13.1. Analysis.

Comparison 13 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 1 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less).
14.1. Analysis.

Comparison 14 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY, Outcome 1 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less).
13.2. Analysis.

Comparison 13 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 2 Short‐term improvement in CTS symptoms (Levine symptom status score) (3 months or less).
14.2. Analysis.

Comparison 14 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY, Outcome 2 Short‐term improvement in CTS symptoms (Levine symptom status score) (3 months or less).
15.1. Analysis.

Comparison 15 TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 1 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less).
15.2. Analysis.

Comparison 15 TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 2 Short‐term improvement in CTS symptoms (Levine symptom status score) (3 months or less).
At the end of 10 weeks treatment, Pinar 2005 found wrists receiving nerve gliding exercises plus splint plus activity modification had pain which was 0.60 points lower on a 0 to 10 VAS (MD ‐0.60, 95% CI ‐1.74 to 0.54; Analysis 16.2) compared to wrists receiving splint plus activity modification. Further, more wrists receiving nerve gliding exercises plus splint plus activity modification had a positive Phalen's test at the end of 10 weeks treatment (RR 0.95, 95% CI 0.82 to 1.11; Analysis 16.3) and a positive Tinel's test at this time point (RR 0.70, 95% CI 0.42 to 1.17; Analysis 16.4) compared to wrists receiving splint plus activity modification. All of these outcome data were analysed at the wrist‐level, and it is clear that the correlation between wrists in bilateral CTS participants was not accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
16.2. Analysis.

Comparison 16 NERVE GLIDING EXERCISES PLUS SPLINT PLUS ACTIVITY MODIFICATION VERSUS SPLINT PLUS ACTIVITY MODIFICATION, Outcome 2 Short‐term improvement in CTS symptoms (VAS pain 0 to 10) (3 months or less).
16.3. Analysis.

Comparison 16 NERVE GLIDING EXERCISES PLUS SPLINT PLUS ACTIVITY MODIFICATION VERSUS SPLINT PLUS ACTIVITY MODIFICATION, Outcome 3 Short‐term improvement in CTS symptoms (Phalen's sign) (3 months or less).
16.4. Analysis.

Comparison 16 NERVE GLIDING EXERCISES PLUS SPLINT PLUS ACTIVITY MODIFICATION VERSUS SPLINT PLUS ACTIVITY MODIFICATION, Outcome 4 Short‐term improvement in CTS symptoms (Tinel's sign) (3 months or less).
3) Short‐term improvement in functional ability or health‐related quality of life (three months or less)
Reported as an outcome in Akalin 2002, Bahrami 2006, Bardak 2009, Baysal 2006, Bialosky 2009, Brininger 2007, Heebner 2008, Horng 2011 and Pinar 2005.
Akalin 2002 found that wrists receiving nerve and tendon gliding exercises plus splint had a functional status score (assessed using the Levine questionnaire (Levine 1993)) that was 1.00 points lower compared to wrists receiving splint alone (MD ‐1.00, 95% CI ‐4.72 to 2.72; Analysis 6.4) at eight weeks post‐treatment. Also, wrists receiving nerve and tendon gliding exercises plus splint had better grip strength (kg) (MD 2.30, 95% CI ‐2.49 to 7.09; Analysis 6.5), better pinch strength (kg) (MD 2.39, 95% CI ‐0.43 to 5.21; Analysis 6.6), and better static two‐point discrimination (mm) (MD ‐0.70, 95% CI ‐1.24 to ‐0.16; Analysis 6.7) compared to wrists receiving splint alone. However, neither participants nor outcome assessors were blind to treatment, which may have biased these results if participants or outcome assessors believed that nerve and tendon gliding exercises plus splint were superior to splint alone. Further, all of these outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported. Therefore these results should be interpreted with caution.
6.4. Analysis.

Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 4 Short‐term improvement in functional ability (Levine) (3 months or less).
6.5. Analysis.

Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 5 Short‐term improvement in functional ability (grip strength (kg)) (3 months or less).
6.6. Analysis.

Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 6 Short‐term improvement in functional ability (pinch strength (kg)) (3 months or less).
6.7. Analysis.

Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 7 Short‐term improvement in functional ability (static two‐point discrimination (mm)) (3 months or less).
Bahrami 2006 found that at six weeks post‐treatment, wrists receiving nerve and tendon gliding exercises plus splint had a functional status score (assessed using the Levine questionnaire (Levine 1993)) which was 1.90 points lower compared to wrists receiving splint alone (MD ‐1.90, 95% CI ‐7.60 to 3.80; Analysis 6.4). These outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
In Bardak 2009, at the end of six weeks treatment, wrists receiving nerve and tendon gliding exercises alone only had a higher (worse) self‐reported functional status score (MD 4.20, 95% CI 1.88 to 6.52; Analysis 7.6) and worse two‐point discrimination (MD 0.10, 95% CI ‐0.39 to 0.59; Analysis 7.7) than wrists receiving splint plus steroid injection. The authors also found at the end of six weeks treatment that wrists receiving nerve and tendon gliding exercises plus splint plus steroid injection had a lower (better) self‐reported functional status score (MD ‐0.20, 95% CI ‐1.94 to 1.54; Analysis 8.6) and better two‐point discrimination (MD ‐0.20, 95% CI ‐0.79 to 0.39; Analysis 8.7) than wrists receiving splint plus steroid injection. Further, the lack of patient blinding suggests that these data should be interpreted with caution, as participants beliefs about the effectiveness of the intervention they received may have biased their self‐reported responses.
7.6. Analysis.

Comparison 7 NERVE AND TENDON GLIDING EXERCISES VERSUS SPLINT PLUS STEROID INJECTION, Outcome 6 Short‐term improvement in functional ability (functional status score) (3 months or less).
7.7. Analysis.

Comparison 7 NERVE AND TENDON GLIDING EXERCISES VERSUS SPLINT PLUS STEROID INJECTION, Outcome 7 Short‐term improvement in functional ability (two‐point discrimination) (3 months or less).
8.6. Analysis.

Comparison 8 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS STEROID INJECTION VERSUS SPLINT PLUS STEROID INJECTION, Outcome 6 Short‐term improvement in functional ability (functional status score) (3 months or less).
8.7. Analysis.

Comparison 8 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS STEROID INJECTION VERSUS SPLINT PLUS STEROID INJECTION, Outcome 7 Short‐term improvement in functional ability (two‐point discrimination) (3 months or less).
Baysal 2006 found that wrists receiving nerve and tendon gliding exercises plus ultrasound plus splint had a mean self‐reported functional status score (as assessed using the Levine questionnaire (Levine 1993)) which was 4.40 points lower (better) on a 40‐point scale at the end of three weeks treatment (MD 4.40, 95% CI ‐0.12 to 8.92; Analysis 9.5) and 3.50 points lower (better) at eight weeks post‐treatment (MD 3.50, 95% CI ‐1.08 to 8.08; Analysis 9.5) compared to wrists receiving ultrasound plus splint. The nerve and tendon gliding exercises plus ultrasound plus splint group had hand grip strength which was 0.10 kg worse at the end of three weeks treatment (MD 0.10, 95% CI ‐5.22 to 5.42; Analysis 9.6) and 1.20 kg worse at eight weeks post‐treatment (MD 1.20, 95% CI ‐1.60 to 4.00; Analysis 9.6) compared to the ultrasound plus splint group, however pinch strength was 1.30 kg better at the end of three weeks treatment and at eight weeks post‐treatment (MD ‐1.30, 95% CI ‐2.86 to 0.26; Analysis 9.7). Further, wrists receiving nerve and tendon gliding exercises plus splint had a mean functional status score (as assessed using the Levine questionnaire (Levine 1993)) which was 1.30 points lower (better) on a 40‐point scale at the end of three weeks treatment (MD 1.30, 95% CI ‐3.83 to 6.43; Analysis 10.5) and 1.20 points lower (better) at eight weeks post‐treatment (MD 1.20, 95% CI ‐3.81 to 6.21; Analysis 10.5) compared to wrists receiving ultrasound plus splint. The nerve and tendon gliding exercises plus splint group had hand grip strength which was 0.70 kg worse at the end of three weeks treatment (MD 0.70, 95% CI ‐4.82 to 6.22; Analysis 10.6) and 0.80 kg worse at eight weeks post‐treatment (MD 0.80, 95% CI ‐2.42 to 4.02; Analysis 10.6) when compared to the ultrasound plus splint group, whereas pinch strength was 0.60 kg better at the end of three weeks treatment (MD ‐0.60, 95% CI ‐1.98 to 0.78; Analysis 10.7) and 0.60 kg better at eight weeks post‐treatment (MD ‐0.60, 95% CI ‐1.92 to 0.72; Analysis 10.7). All of these outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
9.5. Analysis.

Comparison 9 NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 5 Short‐term improvement in functional ability (Levine) (3 months or less).
9.6. Analysis.

Comparison 9 NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 6 Short‐term improvement in functional ability (hand grip strength) (3 months or less).
9.7. Analysis.

Comparison 9 NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 7 Short‐term improvement in functional ability (pinch strength) (3 months or less).
10.5. Analysis.

Comparison 10 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 5 Short‐term improvement in functional ability (Levine) (3 months or less).
10.6. Analysis.

Comparison 10 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 6 Short term improvement in functional ability (hand grip strength) (3 months or less).
10.7. Analysis.

Comparison 10 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 7 Short term improvement in functional ability (pinch strength) (3 months or less).
Bialosky 2009 measured disability using the 44‐point Quick Disability of the Arm, Shoulder and Hand (DASH) questionnaire and found that wrists receiving the neurodynamic technique plus splint had a lower score than wrists receiving the "sham" technique plus splint (MD ‐5.30, 95% CI ‐17.49 to 6.89; Analysis 11.6). The authors also measured grip strength at the end of three weeks treatment, and though no data suitable for meta‐analysis were reported in the publication, data for this outcome were provided on request. The authors found that wrists receiving the neurodynamic technique plus splint had grip strength that was 1.8 kg lower than wrists receiving the "sham" technique plus splint (MD ‐1.80, 95% CI ‐8.68 to 5.08; Analysis 11.7). The precision of both effect estimates was very low and does not rule out a beneficial effect of either intervention.
11.6. Analysis.

Comparison 11 NEURODYNAMIC TECHNIQUE PLUS SPLINT VERSUS "SHAM" NEURODYNAMIC TECHNIQUE PLUS SPLINT, Outcome 6 Short‐term improvement in functional ability (DASH questionnaire) (3 months or less).
11.7. Analysis.

Comparison 11 NEURODYNAMIC TECHNIQUE PLUS SPLINT VERSUS "SHAM" NEURODYNAMIC TECHNIQUE PLUS SPLINT, Outcome 7 Short‐term improvement in functional ability (grip strength) (3 months or less).
Brininger 2007 reported that they measured short‐term functional status (using the Levine questionnaire (Levine 1993)), Moberg Pick‐Up test, grip strength, tip pinch strength, palmar pinch strength, and lateral pinch strength at end of the four‐week treatment period, and at four weeks follow‐up, but the only numerical data reported were change from baseline to end of treatment or follow‐up for all intervention and control groups combined, and for most outcomes only F and P values were reported. Therefore, no data could be entered in RevMan 5.
After both one month and six months treatment Heebner 2008 measured self‐reported functional status using the Levine questionnaire and the Disability of the Arm, Shoulder and Hand (DASH) questionnaire, and neurodynamic irritability of median nerve (R1) measured in degrees (°) using the upper limb tension test for the median nerve, but no numerical summary data (for example, means and SDs) for each group were reported in the publication. However, when requested, the authors provided data on these outcomes collected after one month of treatment. Wrists receiving nerve and tendon gliding exercises plus splint plus education had a Levine functional status score that was no different (MD 0.00, 95% CI ‐0.77 to 0.77; Analysis 12.2), a Disability of the Arm, Shoulder and Hand score that was 3.10 points higher (worse) (MD 3.10, 95% CI ‐13.34 to 19.54; Analysis 12.3) and neurodynamic irritability of median nerve (R1) which was 10.90° higher (MD 10.90, 95% CI ‐6.68 to 28.48; Analysis 12.4) compared to wrists receiving tendon gliding exercises plus splint plus education. However, the 95% CIs of these effect estimates incorporate both positive and negative effects of this intervention.
12.2. Analysis.

Comparison 12 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS EDUCATION VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS EDUCATION, Outcome 2 Short‐term improvement in functional ability (Levine functional status score) (3 months or less).
12.3. Analysis.

Comparison 12 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS EDUCATION VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS EDUCATION, Outcome 3 Short‐term improvement in functional ability (DASH score) (3 months or less).
12.4. Analysis.

Comparison 12 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS EDUCATION VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS EDUCATION, Outcome 4 Short‐term improvement in functional ability (neurodynamic irritability of median nerve (R1, °)) (3 months or less).
At two months post‐treatment, Horng 2011 found participants receiving nerve gliding exercises plus splint plus paraffin therapy had a change from baseline in functional status score which was 0.30 points worse on the Boston CTS questionnaire (Levine 1993) (MD 0.30, 95% CI ‐0.11 to 0.71; Analysis 13.3), change in DASH score which was 5.40 points worse (MD 5.40, 95% CI ‐3.23 to 14.03; Analysis 13.4), change in WHO Quality of Life Brief Questionnaire (WHOQOL‐BREF) Physical Domain score which was 0.77 points worse (MD ‐0.77, 95% CI ‐2.01 to 0.47; Analysis 13.5), change in WHOQOL‐BREF Psychologic Domain score which was 0.70 points better (MD 0.70, 95% CI ‐0.46 to 1.86; Analysis 13.6), change in WHOQOL‐BREF Social Domain score which was 0.10 points better (MD 0.10, 95% CI ‐0.96 to 1.16; Analysis 13.7), and change in WHOQOL‐BREF Environmental Domain score which was no different (MD 0.00, 95% CI ‐0.83 to 0.83; Analysis 13.8) compared to participants receiving splint plus paraffin therapy. Horng 2011 also found participants receiving nerve gliding exercises plus splint plus paraffin therapy had a change from baseline in functional status score (Levine 1993) which was 0.50 points worse (MD 0.50, 95% CI 0.18 to 0.82; Analysis 14.3), change in DASH score which was 8.60 points worse (MD 8.60, 95% CI 2.50 to 14.70; Analysis 14.4), change in WHOQOL‐BREF Physical Domain score which was 0.67 points worse (MD ‐0.67, 95% CI ‐1.61 to 0.27; Analysis 14.5), change in WHOQOL‐BREF Psychologic Domain score which was 0.30 points better (MD 0.30, 95% CI ‐0.73 to 1.33; Analysis 14.6), change in WHOQOL‐BREF Social Domain score which was no different (MD 0.00, 95% CI ‐0.98 to 0.98; Analysis 14.7), and a change in WHOQOL‐BREF Environmental Domain score which was no different (MD 0.00, 95% CI ‐1.07 to 1.07; Analysis 14.8) compared to participants receiving tendon gliding exercises plus splint plus paraffin therapy. Further, participants receiving tendon gliding exercises plus splint plus paraffin therapy had a change from baseline in functional status score (Levine 1993) which was 0.20 points better (MD ‐0.20, 95% CI ‐0.61 to 0.21; Analysis 15.3), change in DASH score which was 3.20 points better (MD ‐3.20, 95% CI ‐12.78 to 6.38; Analysis 15.4), change in WHOQOL‐BREF Physical Domain score which was 0.10 points worse (MD ‐0.10, 95% CI ‐1.23 to 1.03; Analysis 15.5), change in WHOQOL‐BREF Psychologic Domain score which was 0.40 points better (MD 0.40; 95% CI ‐0.95 to 1.75; Analysis 15.6), change in WHOQOL‐BREF Social Domain score which was 0.10 points better (MD 0.10, 95% CI ‐0.78 to 0.98; Analysis 15.7), and change in WHOQOL‐BREF Environmental Domain score which was no different (MD 0.00, 95% CI ‐0.97 to 0.97; Analysis 15.8) compared to participants receiving splint plus paraffin therapy. All of these effect estimates have 95% CIs that do not exclude the possibility of no difference between groups or effects that favour either treatment group, except for the outcomes functional status score and DASH score when comparing nerve gliding exercises plus splint plus paraffin therapy to tendon gliding exercises plus splint plus paraffin therapy, which favoured the group receiving tendon gliding exercises. These two results should be interpreted with caution as participants were not blind to treatment and awareness of their allocated treatment may have influenced their responses to self‐reported outcomes. All of these outcome data were analysed at the participant‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become narrower than those reported. The authors also measured the grip strength, pinch strength and sensitivity, however summary data for each group were not reported. The authors only reported that no statistically significant difference was found between the three groups on these outcomes.
13.3. Analysis.

Comparison 13 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 3 Short‐term improvement in functional ability (Levine functional status score) (3 months or less).
13.4. Analysis.

Comparison 13 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 4 Short‐term improvement in functional ability (DASH score) (3 months or less).
13.5. Analysis.

Comparison 13 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 5 Short‐term improvement in health‐related quality of life (WHOQOLF Physical Domain score) (3 months or less).
13.6. Analysis.

Comparison 13 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 6 Short‐term improvement in health‐related quality of life (WHOQOLF Psychologic Domain score) (3 months or less).
13.7. Analysis.

Comparison 13 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 7 Short‐term improvement in health‐related quality of life (WHOQOLF Social Domain score) (3 months or less).
13.8. Analysis.

Comparison 13 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 8 Short‐term improvement in health‐related quality of life (WHOQOLF Environmental Domain score) (3 months or less).
14.3. Analysis.

Comparison 14 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY, Outcome 3 Short‐term improvement in functional ability (Levine functional status score) (3 months or less).
14.4. Analysis.

Comparison 14 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY, Outcome 4 Short‐term improvement in functional ability (DASH score) (3 months or less).
14.5. Analysis.

Comparison 14 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY, Outcome 5 Short‐term improvement in health‐related quality of life (WHOQOLF Physical Domain score) (3 months or less).
14.6. Analysis.

Comparison 14 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY, Outcome 6 Short‐term improvement in health‐related quality of life (WHOQOLF Psychologic Domain score) (3 months or less).
14.7. Analysis.

Comparison 14 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY, Outcome 7 Short‐term improvement in health‐related quality of life (WHOQOLF Social Domain score) (3 months or less).
14.8. Analysis.

Comparison 14 NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY, Outcome 8 Short‐term improvement in health‐related quality of life (WHOQOLF Environmental Domain score) (3 months or less).
15.3. Analysis.

Comparison 15 TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 3 Short‐term improvement in functional ability (Levine functional status score) (3 months or less).
15.4. Analysis.
Comparison 15 TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 4 Short‐term improvement in functional ability (DASH score) (3 months or less).
15.5. Analysis.

Comparison 15 TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 5 Short‐term improvement in health‐related quality of life (WHOQOLF Physical Domain score) (3 months or less).
15.6. Analysis.

Comparison 15 TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 6 Short‐term improvement in health‐related quality of life (WHOQOLF Psychologic Domain score) (3 months or less).
15.7. Analysis.

Comparison 15 TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 7 Short‐term improvement in health‐related quality of life (WHOQOLF Social Domain score) (3 months or less).
15.8. Analysis.

Comparison 15 TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY, Outcome 8 Short‐term improvement in health‐related quality of life (WHOQOLF Environmental Domain score) (3 months or less).
In Pinar 2005, while the group receiving nerve gliding exercises plus splint plus activity modification had better grip strength (MD 0.30, 95% CI ‐3.41 to 4.01; Analysis 16.5), pinch strength (MD 0.50, 95% CI ‐0.47 to 1.47; Analysis 16.6), and motor function of abductor pollicis brevis muscle (MD 0.16, 95% CI ‐0.17 to 0.49; Analysis 16.7) at the end of 10 weeks treatment than the group receiving splint plus activity modification, whereas, the group receiving splint plus activity modification had better two‐point discrimination at the end of 10 weeks treatment (MD 0.03, 95% CI ‐0.37 to 0.43; Analysis 16.8). All of these outcome data were analysed at the wrist‐level, and it is clear that the correlation between wrists in bilateral CTS participants was not accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported. Pinar 2005 also assessed light‐touch deep‐pressure sense at the end of 10 weeks, but a MD between groups was not estimable in RevMan as the authors reported that the SD for the splint plus activity modification group was 0.0 (Analysis 16.9). According to the study publication, by using the Wilcoxon test or Mann Whitney‐U test, the authors reported finding no statistically significant difference between groups in this outcome.
16.5. Analysis.

Comparison 16 NERVE GLIDING EXERCISES PLUS SPLINT PLUS ACTIVITY MODIFICATION VERSUS SPLINT PLUS ACTIVITY MODIFICATION, Outcome 5 Short‐term improvement in functional ability (grip strength (kg)) (3 months or less).
16.6. Analysis.

Comparison 16 NERVE GLIDING EXERCISES PLUS SPLINT PLUS ACTIVITY MODIFICATION VERSUS SPLINT PLUS ACTIVITY MODIFICATION, Outcome 6 Short‐term improvement in functional ability (pinch strength (kg)) (3 months or less).
16.7. Analysis.

Comparison 16 NERVE GLIDING EXERCISES PLUS SPLINT PLUS ACTIVITY MODIFICATION VERSUS SPLINT PLUS ACTIVITY MODIFICATION, Outcome 7 Short‐term improvement in functional ability (motor function of abductor pollicis brevis muscle) (3 months or less).
16.8. Analysis.

Comparison 16 NERVE GLIDING EXERCISES PLUS SPLINT PLUS ACTIVITY MODIFICATION VERSUS SPLINT PLUS ACTIVITY MODIFICATION, Outcome 8 Short‐term improvement in functional ability (two‐point discrimination test) (3 months or less).
16.9. Analysis.

Comparison 16 NERVE GLIDING EXERCISES PLUS SPLINT PLUS ACTIVITY MODIFICATION VERSUS SPLINT PLUS ACTIVITY MODIFICATION, Outcome 9 Short‐term improvement in functional ability (light‐touch deep‐pressure sense) (3 months or less).
4) Short‐term improvement in neurophysiologic parameters (three months or less)
Reported as an outcome in Bahrami 2006, Baysal 2006, Bialosky 2009 and Horng 2011, but not in Akalin 2002, Bardak 2009, Brininger 2007, Heebner 2008 or Pinar 2005.
In Bahrami 2006, wrists receiving nerve and tendon gliding exercises plus splint had a median nerve sensory distal latency that was 0.5 ms shorter (MD ‐0.05, 95% CI ‐0.33 to 0.23; Analysis 6.8), a median nerve motor distal latency that was 0.20 ms shorter (MD ‐0.20, 95% CI ‐0.43 to 0.03; Analysis 6.9), and a median‐ulnar sensory distal latency that was 0.06 ms longer (MD 0.06, 95% CI ‐0.32 to 0.44; Analysis 6.10) compared to wrists receiving splint alone. All of these outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
6.8. Analysis.

Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 8 Short‐term improvement in median nerve sensory distal latency (3 months or less).
6.9. Analysis.

Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 9 Short‐term improvement in median nerve motor distal latency (3 months or less).
6.10. Analysis.

Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 10 Short‐term improvement in median‐ulnar sensory distal latency (3 months or less).
In Baysal 2006, wrists receiving nerve and tendon gliding exercises plus therapeutic ultrasound plus splint had a median nerve motor distal latency that was no different at the end of three weeks treatment (MD 0.00, 95% CI ‐1.06 to 1.06; Analysis 9.8) and 0.10 ms longer at eight weeks post‐treatment (MD ‐0.10, 95% CI ‐1.25 to 1.05; Analysis 9.8), and a median nerve sensory distal latency that was 0.10 ms longer at the end of three weeks treatment (MD ‐0.10, 95% CI ‐0.55 to 0.35; Analysis 9.9) and 0.20 ms longer at eight weeks post‐treatment ended (MD ‐0.20, 95% CI ‐0.58 to 0.18; Analysis 9.9) compared to wrists receiving therapeutic ultrasound plus splint. The authors also reported that wrists receiving nerve and tendon gliding exercises plus splint had a median nerve motor distal latency which was 0.02 ms longer at the end of three weeks treatment (MD ‐0.20, 95% CI ‐0.95 to 0.55; Analysis 10.8) and 0.30 ms longer at eight weeks post‐treatment (MD ‐0.30, 95% CI ‐0.91 to 0.31; Analysis 10.8), but a median nerve sensory distal latency that was 0.10 ms shorter at the end of three weeks treatment (MD 0.10, 95% CI ‐0.28 to 0.48; Analysis 10.9) and no different at eight weeks post‐treatment (MD 0.00, 95% CI ‐0.36 to 0.36; Analysis 10.9) when compared to wrists receiving therapeutic ultrasound plus splint. All of these outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
9.8. Analysis.

Comparison 9 NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 8 Short‐term improvement in motor distal latency (ms) (3 months or less).
9.9. Analysis.

Comparison 9 NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 9 Short‐term improvement in sensory distal latency (ms) (3 months or less).
10.8. Analysis.

Comparison 10 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 8 Short term improvement in motor distal latency (ms) (3 months or less).
10.9. Analysis.

Comparison 10 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 9 Short‐term improvement in sensory distal latency (ms) (3 months or less).
Bialosky 2009 measured motor distal onset latency at the abductor pollicis brevis muscle and the combined sensory index (CSI), which "measures the sensory nerve action potentials peak latency and amplitude from the median, ulnar, and radial nerves as they cross the wrist. The differences measured between the nerves are combined into an overall score with a value greater than 1 indicative of slowing of median nerve conduction through the carpal tunnel" (Bialosky 2009, page 713). The authors found that at the end of three weeks treatment, wrists receiving the neurodynamic technique plus splint had motor distal latency that was 0.43 ms shorter (MD ‐0.43, 95% CI ‐1.79 to 0.93; Analysis 11.8), and a combined sensory index which was 0.39 points lower (worse) (MD 0.39, 95% CI ‐1.70 to 2.48; Analysis 11.9) compared to wrists receiving the sham technique plus splint. However, these effect estimates were imprecise and suggest that a greater benefit in either group over the other is possible.
11.8. Analysis.

Comparison 11 NEURODYNAMIC TECHNIQUE PLUS SPLINT VERSUS "SHAM" NEURODYNAMIC TECHNIQUE PLUS SPLINT, Outcome 8 Short‐term improvement in motor distal latency (ms) (3 months or less).
11.9. Analysis.

Comparison 11 NEURODYNAMIC TECHNIQUE PLUS SPLINT VERSUS "SHAM" NEURODYNAMIC TECHNIQUE PLUS SPLINT, Outcome 9 Short‐term improvement in combined sensory index (3 months or less).
Horng 2011 measured median and ulnar nerve sensory and motor distal latency, but only reported that differences between groups were not statistically significant.
5) Long‐term improvement in CTS symptoms (more than three months)
Reported as an outcome in Akalin 2002, Bahrami 2006, Bardak 2009 and Baysal 2006, but not in Bialosky 2009, Brininger 2007, Heebner 2008, Horng 2011 or Pinar 2005.
Akalin 2002 measured patient satisfaction with treatment and dichotomised participants into those with 'excellent/good' satisfaction and those with 'fair/poor' satisfaction at a mean of eight months (range five to 11 months) after the four‐week treatment period. The risk of reporting having 'excellent/good' satisfaction with treatment was 31% higher for wrists receiving nerve and tendon gliding exercises plus splint compared to splint alone (RR 1.31, 95% CI 0.96 to 1.78; Analysis 6.11). These outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
6.11. Analysis.
Comparison 6 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT, Outcome 11 Long‐term improvement in CTS symptoms (satisfaction) (>3 months).
Bahrami 2006 reported the proportion of participants with excellent or good satisfaction with treatment from five to eight months after the four‐week treatment period. The risk of reporting excellent or good satisfaction with treatment was 25% higher for wrists receiving nerve and tendon gliding exercises plus splint compared to splint alone (RR 1.25, 95% CI 0.80 to 1.97; Analysis 6.11). These outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
In Bardak 2009, fewer participants receiving nerve and tendon gliding exercises alone rated their satisfaction with treatment at 11 months post‐treatment as 'excellent/good' than participants receiving splint plus steroid injection (RR 0.66, 95% CI 0.45 to 0.98; Analysis 7.8). However, the high risk of bias associated with lack of patient blinding in this study suggest that these results should be interpreted with caution, as participants responses may have been based on their beliefs about the superiority of splint and steroid injection over nerve and tendon gliding exercises. Further, fewer participants receiving nerve and tendon gliding exercises plus splint plus steroid injection rated their satisfaction with treatment at 11 months post‐treatment as 'excellent/good' than participants receiving splint plus steroid injection (RR 0.98, 95% CI 0.74 to 1.29; Analysis 8.8), though the 95% CI incorporates opposite effects of treatment.
7.8. Analysis.

Comparison 7 NERVE AND TENDON GLIDING EXERCISES VERSUS SPLINT PLUS STEROID INJECTION, Outcome 8 Long‐term improvement in CTS symptoms (patient satisfaction) (>3 months).
8.8. Analysis.

Comparison 8 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS STEROID INJECTION VERSUS SPLINT PLUS STEROID INJECTION, Outcome 8 Long‐term improvement in CTS symptoms (patient satisfaction) (>3 months).
At the final follow‐up (an average of 11 +/‐ 4.5 months after the end of three weeks treatment), Baysal 2006 asked participants to report their satisfaction with treatment. The authors reported the number of participants rated as 'excellent/good' (asymptomatic or rarely symptomatic), 'fair' (symptomatic only during compelling activity), and 'poor' (continuing symptoms without relief following treatment). More participants rated themselves as 'excellent/good' in the group receiving nerve and tendon gliding exercises and therapeutic ultrasound and splint (n = 8/13) when compared to the group receiving ultrasound and splint (n = 3/12) (RR 0.41, 95% CI 0.14 to 1.18; Analysis 9.10). Also, fewer participants rated their wrists as 'excellent/good' in the group receiving nerve and tendon gliding exercises and splint compared to the group receiving therapeutic ultrasound and splint (RR 9.69, 95% CI 0.55 to 171.98; Analysis 10.10). However, the precision of both effect estimates was low. The lack of patient blinding and unclear reasons for incomplete data for these outcomes suggest that these results should be interpreted with caution. Further, all of these outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
9.10. Analysis.

Comparison 9 NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 10 Long‐term improvement in CTS symptoms (>3 months).
10.10. Analysis.

Comparison 10 NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT, Outcome 10 Long‐term improvement in CTS symptoms (>3 months).
6) Long‐term improvement in functional ability or health‐related quality of life (more than three months)
Not reported as an outcome in Akalin 2002, Bahrami 2006, Bardak 2009, Baysal 2006, Bialosky 2009, Brininger 2007, Heebner 2008, Horng 2011 or Pinar 2005.
7) Need for surgery
Not reported as an outcome in Akalin 2002, Bahrami 2006, Bardak 2009, Baysal 2006, Bialosky 2009, Brininger 2007, Heebner 2008, Horng 2011 or Pinar 2005.
Other mobilisation intervention(s) versus other non‐surgical interventions
Three trials compared a mobilisation intervention other than nerve mobilisation to another non‐surgical intervention (Davis 1998; Garfinkel 1998; Janssen 2009). Davis 1998 compared chiropractic treatment (myofascial massage plus manual thrusts plus therapeutic ultrasound) plus splint versus ibuprofen plus splint for nine weeks duration and Garfinkel 1998 compared yoga versus a wrist splint worn for eight weeks. Janssen 2009 compared contrast baths plus controlled finger flexion and extension exercises to contrast baths alone and to controlled finger flexion and extension exercises alone. Janssen 2009 did not measure any of the pre‐specified outcomes of this review. The interventions were judged as being too heterogeneous so the data were not pooled, and a narrative summary of the results is provided.
As reported in the Included studies section and Characteristics of included studies tables, some or all participants had bilateral CTS in the studies conducted by Davis 1998 and Garfinkel 1998. It was clear that the correlation between wrists was accounted for in the analysis only in the study by Davis 1998 for the neurophysiologic parameter outcomes. Attempts to retrieve individual wrist outcome data for these studies were unsuccessful. Therefore, apart from the neurophysiologic parameter outcome data in Davis 1998, all other outcome data reported in these studies may be invalid due to a possible unit of analysis error. Without access to the individual wrist data, and without being able to estimate parameters such as the intraclass correlation coefficient from other studies included in the review, we did not attempt to adjust the results of these studies. We have included the outcome data as reported by the trialists, but emphasise that all results of these studies should be interpreted with caution, as the possibly lack of adjustment may have produced either overly wide 95% CIs or overly narrow 95% CIs with artificially smaller P values (Higgins 2011c).
Primary outcomes
1) Short‐term overall improvement (three months or less)
Not reported as an outcome in Davis 1998, Garfinkel 1998 or Janssen 2009.
Secondary outcomes
1) Adverse effects
Reported as an outcome in Davis 1998, but not in Garfinkel 1998 or Janssen 2009.
Davis 1998 found that 10 (22%) of the 46 participants receiving ibuprofen and splint for nine weeks reported intolerance to the drug within the first two weeks of treatment and that for five of these participants, the intolerance was marked and resulted in them being unable to continue taking the medication. In contrast, only one adverse effect was reported in the chiropractic group, which was a sore neck associated with the manipulation. The number of adverse effects experienced throughout the treatment period was lower in the chiropractic group (RR 0.10; 95% CI 0.01 to 0.77; Analysis 17.1).
17.1. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 1 Adverse effects.
2) Short‐term improvement in CTS symptoms (three months or less)
Reported as an outcome in Davis 1998 and Garfinkel 1998, but not in Janssen 2009.
At the end of nine weeks treatment, Davis 1998 found that participants receiving chiropractic treatment plus splint had higher CTS‐related physical distress (as assessed using the CTOA‐Physical distress questionnaire) (MD 3.51, 95% CI 0.18 to 6.84; Analysis 17.2) and higher CTS‐related mental distress (MD 2.35, 95% CI ‐3.44 to 8.14; Analysis 17.3) than participants receiving ibuprofen and splint. The authors reported there were no statistically significant differences between groups for these outcomes; however, this is probably because they used repeated measures multivariate analyses of variance and planned comparisons and subsequently set the criterion for statistical significance as P < 0.00833 to reduce the chance of obtaining spurious statistically significant results. Outcome data for these two outcomes were analysed at the participant‐level, and the correlation between wrists in bilateral CTS participants was not accounted for. If we had adjusted the data, the 95% CIs may have become narrower than those reported. Further, four weeks after treatment ceased, Davis 1998 found wrists receiving chiropractic treatment plus splint had better vibrometric threshold of finger sensation (dB) of the right hand (MD 0.96, 95% CI ‐1.99 to 3.91; Analysis 17.4), but this was worse for the left hand (MD ‐1.91, 95% CI ‐5.42 to 1.60; Analysis 17.4) compared to wrists receiving ibuprofen plus splint. The low precision of these effect estimates means that chiropractic treatment may not always be beneficial.
17.2. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 2 Short‐term improvement in CTS symptoms (CTOA physical distress) (3 months or less).
17.3. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 3 Short‐term improvement in CTS symptoms (CTOA mental distress) (3 months or less).
17.4. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 4 Short‐term improvement in CTS symptoms (vibrometric threshold of finger sensation) (3 months or less).
After eight weeks of treatment, Garfinkel 1998 found that pain intensity for the previous week was 1.40 points lower on a 0 to 10 VAS in participants receiving yoga compared to participants receiving wrist splint (MD 1.40, 95% CI 0.07 to 2.73; Analysis 18.1). Garfinkel 1998 also found more participants assigned to yoga experienced improvement in sleep disturbance (RR 0.47, 95% CI 0.10 to 2.25; Analysis 18.2) compared to participants receiving wrist splint and more wrists receiving yoga experienced an improvement in Tinel's test (RR 0.47, 95% CI 0.13 to 1.66; Analysis 18.3) after eight weeks of treatment compared to wrists receiving wrist splint, whereas, fewer wrists receiving yoga experienced an improvement in Phalen's test after eight weeks of treatment (RR 0.19, 95% CI 0.05 to 0.78; Analysis 18.4). However, these results should be interpreted with caution as participants were not blind to treatment and their expectations regarding the potential effectiveness of yoga may have biased their responses. Also, all of these outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
18.1. Analysis.
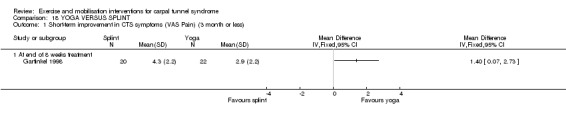
Comparison 18 YOGA VERSUS SPLINT, Outcome 1 Short‐term improvement in CTS symptoms (VAS Pain) (3 month or less).
18.2. Analysis.

Comparison 18 YOGA VERSUS SPLINT, Outcome 2 Short‐term improvement in CTS symptoms (sleep disturbance) (3 months or less.
18.3. Analysis.
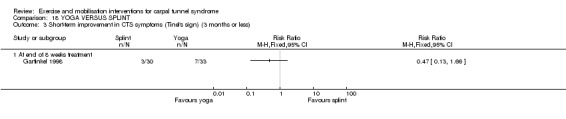
Comparison 18 YOGA VERSUS SPLINT, Outcome 3 Short‐term improvement in CTS symptoms (Tinel's sign) (3 months or less).
18.4. Analysis.

Comparison 18 YOGA VERSUS SPLINT, Outcome 4 Short‐term improvement in CTS symptoms (Phalen's sign) (3 months or less).
3) Short‐term improvement in functional ability or health‐related quality of life (three months or less)
Reported as an outcome in Davis 1998 and Garfinkel 1998, but not in Janssen 2009.
At four weeks follow‐up, participants receiving chiropractic treatment plus splint had better functional ability as assessed by the HAND scale (MD ‐3.30, 95% CI ‐9.73 to 3.13; Analysis 17.5) and by the SF‐36 Body pain scale (MD 5.02, 95% CI ‐4.19 to 14.23; Analysis 17.6) than participants receiving ibuprofen and splint in the study conducted by Davis 1998. However, participants receiving ibuprofen and splint had better functional ability according to the SF‐36 Global scale (MD ‐0.85, 95% CI ‐6.79 to 5.09; Analysis 17.7) and the SF‐36 Role Physical scale (MD ‐15.88, 95% CI ‐34.79 to 3.03; Analysis 17.8) at four weeks follow‐up. These outcome data were analysed at the participant‐level, and the correlation between wrists in bilateral CTS participants was not accounted for. If we had adjusted the data, the 95% CIs may have become narrower than those reported.
17.5. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 5 Short‐term improvement in functional ability (HAND) (3 months or less).
17.6. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 6 Short‐term improvement in functional ability (SF‐36 Body pain) (3 months or less).
17.7. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 7 Short‐term improvement in functional ability (SF‐36 Global) (3 months or less).
17.8. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 8 Short‐term improvement in functional ability (SF‐36 Role physical) (3 months or less).
Garfinkel 1998 found that the wrists receiving wrist splint had grip strength which was 3.10 mmHg better immediately after eight weeks of treatment than wrists receiving yoga (MD 3.10, 95% CI ‐31.06 to 37.26; Analysis 18.5), though the precision of this estimate is very low and opposite effects of treatment cannot be ruled out. These outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
18.5. Analysis.

Comparison 18 YOGA VERSUS SPLINT, Outcome 5 Short‐term improvement in functional ability (grip strength) (3 months or less).
4) Short‐term improvement in neurophysiologic parameters (three months or less)
Reported as an outcome in Davis 1998 and Garfinkel 1998, but not in Janssen 2009.
At the end of nine weeks treatment, Davis 1998 found that wrists receiving chiropractic treatment plus splint had shorter motor wrist onset latency (ms) of the right hand (MD ‐0.21, 95% CI ‐0.68 to 0.26; Analysis 17.9) and left hand (MD ‐0.15, 95% CI ‐0.55 to 0.25; Analysis 17.9), shorter sensory digit two latency (ms) of the right hand (MD ‐0.07, 95% CI ‐0.43 to 0.29; Analysis 17.10) and left hand (MD ‐0.11, 95% CI ‐0.37 to 0.15; Analysis 17.10), longer sensory digit three latency (ms) of the right hand (MD 0.05, 95% CI ‐0.35 to 0.45; Analysis 17.11) but shorter sensory digit three latency (ms) of the left hand (MD ‐0.15, 95% CI ‐0.53 to 0.23; Analysis 17.11), and longer sensory palm peak latency (ms) of the right hand (MD 0.06, 95% CI ‐0.19 to 0.31; Analysis 17.12), but shorter sensory palm peak latency (ms) of the left hand (MD ‐0.07, 95% CI ‐0.30 to 0.16; Analysis 17.12) compared to wrists receiving ibuprofen plus splint. None of the 95% CIs of these effect estimates rule out the possibility for alternative effects of these interventions.
17.9. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 9 Short‐term improvement in median nerve motor wrist (onset) latency (ms) (3 months or less).
17.10. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 10 Short‐term improvement in median nerve sensory digit 2 latency (ms) (3 months or less).
17.11. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 11 Short‐term improvement in median nerve sensory digit 3 latency (ms) (3 months or less).
17.12. Analysis.

Comparison 17 CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT, Outcome 12 Short‐term improvement in median nerve sensory palm (peak) latency (ms) (3 months or less).
At the end of eight weeks treatment, Garfinkel 1998 found wrists receiving yoga had median nerve motor distal latency which was 0.25 ms shorter (MD 0.25, 95% CI ‐0.37 to 0.87; Analysis 18.6) and median nerve sensory distal latency which was 0.39 ms shorter (MD 0.39, 95% CI ‐0.35 to 1.13; Analysis 18.7) compared to wrists receiving wrist splint. These outcome data were analysed at the wrist‐level, and it is not clear if the correlation between wrists in bilateral CTS participants was accounted for. If we had adjusted the data, the 95% CIs may have become wider than those reported.
18.6. Analysis.

Comparison 18 YOGA VERSUS SPLINT, Outcome 6 Short‐term improvement in median nerve motor distal latency (ms) (3 months or less).
18.7. Analysis.

Comparison 18 YOGA VERSUS SPLINT, Outcome 7 Short‐term improvement in median nerve sensory distal latency (3 months or less).
5) Long‐term improvement in CTS symptoms (more than three months)
Not reported as an outcome in Davis 1998, Garfinkel 1998 or Janssen 2009.
6) Long‐term improvement in functional ability or health‐related quality of life (more than three months)
Not reported as an outcome in Davis 1998, Garfinkel 1998 or Janssen 2009.
7) Need for surgery
Not reported as an outcome in Davis 1998, Garfinkel 1998 or Janssen 2009.
Subgroup and sensitivity analyses
We were unable to perform the planned subgroup and sensitivity analyses given the inability to pool any data across the studies included in the review. This may be possible in future updates of the review.
Assessment of reporting bias
No meta‐analyses were performed in this review so we were unable to generate any forest plots and we were unable to use funnel plots to assess the likelihood of publication bias (Higgins 2011a). No protocols for studies were identified and a trial registration entry could only be found for Brininger 2007, therefore, assessment of selective outcome reporting for the majority of studies was limited and was based on comparing outcomes reported in the Methods section to those reported in the Results section of the study publications.
Discussion
Summary of main results
This review sought to examine the potential benefits and harms of different types of exercise and mobilisation interventions for the treatment of CTS. There is limited and very low quality evidence of benefit for all of a diverse collection of exercise and mobilisation interventions for carpal tunnel syndrome, compared with other non‐surgical interventions for CTS (such as splinting, therapeutic ultrasound or oral drugs), and when compared with one another. The studies were heterogeneous in the intensity of the intervention, duration of treatment, and timing of outcome assessment. Therefore, we did not pool data across studies. Only four studies (Akalin 2002; Bahrami 2006; Bardak 2009; Baysal 2006) assessed outcomes at long‐term follow‐up (that is, more than three months after treatment ended).
We identified two trials that compared the efficacy of exercise or mobilisation to no treatment (Field 2004; Tal‐Akabi 2000). Field 2004 reported limited details of the study and we were unable to collect any data from the trialists. The main findings from Tal‐Akabi 2000 were that both neurodynamic mobilisation and carpal bone mobilisation delivered for three weeks resulted in more participants experiencing overall improvement and that carpal bone mobilisation resulted in less pain at short‐term follow‐up, when compared to no treatment. However, the lack of patient blinding suggests these outcomes should be interpreted with caution, as participants' expectations regarding the effectiveness of receiving a mobilisation intervention rather than no treatment may have influenced their reporting of improvement. Tal‐Akabi 2000 was the only study to have assessed the need for surgery as an outcome, and found no clear difference between groups.
The results of three studies that compared one type of mobilisation to another (Burke 2007; Moraska 2008; Tal‐Akabi 2000) suggest that one type is not superior over others. All three studies found small but imprecise differences between the different mobilisation groups in terms of short‐term overall improvement, adverse effects, and short‐term CTS symptoms. Moraska 2008 found that wrists receiving targeted CTS massage had better functional ability compared to general massage; however, the imprecise results, as indicated by the wide 95% CIs, incorporated both positive and negative effects of this intervention.
Nine studies compared nerve mobilisation delivered as part of a multi‐component intervention to another non‐surgical intervention (Akalin 2002; Bahrami 2006; Bardak 2009; Baysal 2006; Bialosky 2009; Brininger 2007; Heebner 2008; Horng 2011; Pinar 2005). For nearly all comparisons, differences between groups on the outcomes, short‐term overall improvement, adverse effects, short‐ and long‐term symptoms and function, and short‐term neurophysiologic parameters, were small and 95% CIs incorporated effects in either direction, and many effect estimates were at high risk of bias, particularly due to lack of participant or outcome assessor blinding. Further, seven of these studies either clearly, or potentially, committed unit of analysis errors due to not accounting for the correlation between wrists in participants with bilateral CTS.
Finally, three trials compared a mobilisation intervention (without nerve mobilisation) to another non‐surgical intervention (Davis 1998; Garfinkel 1998; Janssen 2009). Janssen 2009 did not measure any of the pre‐specified outcomes of this review. Davis 1998 found that chiropractic treatment resulted in fewer adverse events than taking ibuprofen for CTS did. However; we cannot rule out the possibility of positive or negative effects of the mobilisation interventions in Davis 1998 and Garfinkel 1998 for the outcomes, CTS symptoms, functional ability, and neurophysiologic parameters.
More research is needed on potential adverse effects of exercise and mobilisation interventions and the need for surgery, compared with other non‐surgical interventions for CTS.
Overall completeness and applicability of evidence
There are a number of limitations in the interventions used and participants recruited into the studies included in this review. Firstly, the majority of studies in this review delivered exercise or mobilisation interventions as part of a multi‐component treatment regimen, which makes it difficult to draw conclusions on the efficacy of exercise or mobilisation as a stand‐alone intervention for CTS. The age ranges of participants in the studies was also restricted, with 12 of 16 studies including participants with an approximate mean age of 50 years (Akalin 2002; Bahrami 2006; Baysal 2006; Bialosky 2009; Brininger 2007; Field 2004; Garfinkel 1998; Heebner 2008; Horng 2011; Janssen 2009; Moraska 2008; Tal‐Akabi 2000). Therefore, the efficacy of exercise or mobilisation interventions requires further investigation in both younger and older adults. In addition, all of the included studies delivered exercise or mobilisation interventions that ranged from three to 10 weeks in duration, and only one study assessed the outcomes of these interventions at long‐term follow‐up. Thus, the efficacy of longer lasting exercise or mobilisation regimens requires further research, preferably both before and after three months have passed since the end of treatment.
Quality of the evidence
Overall, the risk of bias in the included studies varied substantially across the studies, though most were judged as having an unclear or high risk of bias. For example, concealment of the allocation sequence was only clearly reported in three of the 16 studies (Baysal 2006; Brininger 2007; Davis 1998), and five studies did not conceal allocation (Akalin 2002; Bahrami 2006; Heebner 2008; Moraska 2008; Pinar 2005). The importance of allocation concealment in randomised controlled trials is supported by empirical evidence that inadequate allocation concealment can distort treatment effects, though the magnitude and direction of distortion is difficult to predict (Odgaard‐Jensen 2011). Further, in 12 studies participants were not blind to the intervention they received (Akalin 2002; Bahrami 2006; Bardak 2009; Baysal 2006; Brininger 2007; Burke 2007; Davis 1998; Field 2004; Garfinkel 1998; Heebner 2008; Horng 2011; Tal‐Akabi 2000). Lack of participant blinding is common and often unavoidable in randomised controlled trials delivering exercise or mobilisation interventions. However, interpretation of subjective or self‐reported outcomes should be interpreted cautiously, as empirical evidence indicates that randomised trials with non‐blinded self‐reported outcomes show exaggerated treatment effects (Wood 2008). In contrast, blinding of objective outcomes is always possible through blinding of outcome assessors, however, this was not done in five studies (Akalin 2002; Brininger 2007; Garfinkel 1998; Heebner 2008; Moraska 2008). Ten studies were judged as at low risk of bias regarding completeness of outcome data at three months or less (Akalin 2002; Bahrami 2006; Bardak 2009; Baysal 2006; Brininger 2007; Burke 2007; Janssen 2009; Moraska 2008; Pinar 2005; Tal‐Akabi 2000). Finally, seven studies were judged as being at high risk of bias for selective outcome reporting (Bardak 2009; Brininger 2007; Burke 2007; Field 2004; Garfinkel 1998; Heebner 2008; Horng 2011). The latter finding is concerning given that selective outcome reporting of 'positive' or statistically significant trial results can bias the results and conclusions of a systematic review (Kirkham 2010).
Potential biases in the review process
While our reported methods attempted to minimise bias in the selection of studies, collection of published data, and analysis for the review, our searches were limited to electronic databases, and as a result we have only included published studies. In future updates of this review we will attempt to identify grey literature, as empirical evidence suggests published studies are more likely to report larger treatment effects than unpublished studies (Hopewell 2007). Further, assessment of selective outcome reporting was limited as no protocols were identified for the included studies and only one study was listed on a trial registry database (Brininger 2007). It was also difficult to obtain relevant unpublished data from some of the authors of included studies. This had a considerable impact on our analysis of seven studies which committed or potentially committed unit of analysis errors (Akalin 2002; Bahrami 2006; Baysal 2006; Davis 1998; Garfinkel 1998; Horng 2011; Pinar 2005), as we were unable to re‐analyse the outcome data using methods that address the dependency of data.
Agreements and disagreements with other studies or reviews
To our knowledge, only one other systematic review specifically focusing on exercise and mobilisation interventions for CTS exists (McKeon 2008). McKeon 2008 focused only on nerve gliding exercises for CTS, and only included the studies conducted by Akalin 2002, Baysal 2006, Brininger 2007, Pinar 2005 and Tal‐Akabi 2000, as well as a non‐randomised study conducted by Rozmaryn 1998. The findings of our review are generally consistent with those of McKeon 2008 and other systematic reviews of non‐surgical interventions for CTS that also found limited or insufficient evidence for the effectiveness of exercise and mobilisation interventions for CTS (Ashworth 2010; Gerritsen 2002; Goodyear‐Smith 2004; Huisstede 2010; McKeon 2008; Muller 2004; Ono 2010; Piazzini 2007). In comparison to this review, the most recent systematic review by Huisstede 2010 also included the studies conducted by Akalin 2002, Baysal 2006, Bialosky 2009, Brininger 2007, Burke 2007, Davis 1998, Field 2004, Garfinkel 1998, Heebner 2008, Moraska 2008, Pinar 2005, and Tal‐Akabi 2000. However, due to the timing of their search, this review did not include the studies conducted by Bardak 2009, Janssen 2009 and Horng 2011; the study conducted by Bahrami 2006 was also not included, and it is not clear why. Further, Huisstede 2010 only included outcome data reported in the systematic review by O'Connor 2003 (without including additional data reported in the original study publications in this review), did not obtain unreported data from the trial authors of studies conducted by Bialosky 2009, Heebner 2008, Moraska 2008 and Pinar 2005, did not include outcome data available for all time points reported, and did not report any data on neurophysiologic parameters reported in the included studies.
Authors' conclusions
Implications for practice.
There is limited and very low quality evidence of benefit for all of a diverse collection of exercise and mobilisation interventions for carpal tunnel syndrome. People with carpal tunnel syndrome who indicate a preference for exercise or mobilisation interventions should be informed of the limited evidence of effectiveness and safety of this intervention by their treatment provider. Until more high quality randomised controlled trials assessing the effectiveness and safety of various exercise and mobilisation interventions compared to other non‐surgical interventions are undertaken, the decision to provide this type of non‐surgical intervention to people with carpal tunnel syndrome should be based on clinician's expertise in being able to deliver these treatments and patient's preferences.
Implications for research.
There is a need for more high quality randomised controlled trials to assess the safety and efficacy of the various types of exercise and mobilisation interventions alone (e.g., nerve mobilisation, carpal bone mobilisation, soft tissue mobilisation, massage) compared to other non‐surgical interventions (e.g. therapeutic ultrasound, splint, oral drugs). These trials should attempt to blind participants where possible, and should always attempt to blind assessment of objective outcomes. Trialists should collect data on adverse effects and the need for participants to undergo surgery for their condition. If participants with bilateral CTS are included in the trial, trialists should use statistical methods which take the dependency between wrists into account, and report which method they used to achieve this. Finally, outcomes should be collected at short‐term and long‐term follow‐up (that is at least three months post‐treatment cessation).
Notes
This is one of six new reviews that will update the currently published review 'Non‐surgical treatment (other than steroid injection) for carpal tunnel syndrome'. When all six reviews are published we will withdraw the original review from publication. This review includes a new search, revised review question and selection criteria, updated methodology and an updated review team.
Acknowledgements
We would like to thank Ruth Brassington, Louisa Dunn, Kate Jewitt, Carolyn Reid, Angela Gunn and Rachel Barton from the Cochrane Neuromuscular Disease Group for their assistance in devising the search strategy, helping to locate people to translate the non‐English trials and ongoing support for this review. We thank Shawn Marshall for his substantial contribution to the original version of this review. We thank Katherine Beringer from the Monash Institute of Health Services Research for her assistance in retrieving studies relevant to the review. We thank the trialists (Elif Akalin, Joel Bialosky, Jim Hulbert, Albert Moraska, Lamia Pinar, Toni Roddey and Amir Tal‐Akabi) who corresponded with the principal review author to clarify additional information and/or provided additional data for the review. We thank the translators for their assistance in translating abstracts and papers for the review. We thank the peer reviewers for their helpful comments on the review.
We thank the following institutions for their support during the review:
School of Public Health and Preventive Medicine, Monash University, Melbourne, AUSTRALIA
School of Occupational Therapy, University of South Australia, Adelaide, AUSTRALIA
Institute for Rehabilitation Research and Development, Ottawa, CANADA
Editorial support from the Cochrane Neuromuscular Disease Group is funded by the MRC Centre for Neuromuscular Diseases.
Appendices
Appendix 1. MEDLINE (OvidSP) search strategy
1 randomized controlled trial.pt. (315877) 2 controlled clinical trial.pt. (83182) 3 randomized.ab. (221432) 4 placebo.ab. (127183) 5 drug therapy.fs. (1488786) 6 randomly.ab. (160369) 7 trial.ab. (228368) 8 groups.ab. (1061229) 9 or/1‐8 (2757907) 10 exp animals/ not humans.sh. (3639193) 11 9 not 10 (2340745) 12 Carpal Tunnel Syndrome.tw. or Carpal Tunnel Syndrome/ (7168) 13 ((nerve entrapment or nerve compression or entrapment neuropath$) and carpal).mp. (952) 14 12 or 13 (7268) 15 Exercise Therapy/ (21586) 16 relaxation therapy/ or yoga/ (6428) 17 exp Musculoskeletal Manipulations/ (10753) 18 physical therapy modalities/ or exercise movement techniques/ or musculoskeletal manipulations/ or manipulation, osteopathic/ or massage/ (29819) 19 Manipulation, Chiropractic/ (613) 20 (mobili?ation or physiotherapy or massage or yoga or chiropractic or osteopathic or gliding exercise*).mp. (64383) 21 or/15‐20 (112646) 22 11 and 14 and 21 (67)
Appendix 2. EMBASE (OvidSP) search strategy
1 crossover‐procedure/ (31558) 2 double‐blind procedure/ (102446) 3 randomized controlled trial/ (295130) 4 single‐blind procedure/ (14625) 5 groups.ab. (1311859) 6 (random$ or factorial$ or crossover$ or cross over$ or cross‐over$ or placebo$ or (doubl$ adj blind$) or (singl$ adj blind$) or assign$ or allocat$ or volunteer$).tw. (1049473) 7 or/1‐6 (2140022) 8 exp animals/ (1664443) 9 exp humans/ (12828730) 10 8 not (8 and 9) (1266349) 11 7 not 10 (2050979) 12 limit 11 to embase (1681242) 13 carpal tunnel syndrome/ (9331) 14 carpal tunnel syndrome.mp. (9982) 15 ((nerve entrapment or nerve compression or entrapment neuropath$) and carpal).mp. (1553) 16 or/13‐15 (10081) 17 exp kinesiotherapy/ (37708) 18 exp manipulative medicine/ (21981) 19 physiotherapy/ (42741) 20 MOBILIZATION/ (12870) 21 (mobili?ation or physiotherapy or massage or yoga or chiropractic or osteopathic or gliding exercise$).mp. (123986) 22 or/17‐21 (159316) 23 12 and 16 and 22 (70)
Appendix 3. AMED (OvidSP) search strategy
1 Randomized controlled trials/ (1495) 2 Random allocation/ (302) 3 Double blind method/ (425) 4 Single‐Blind Method/ (24) 5 exp Clinical Trials/ (3146) 6 (clin$ adj25 trial$).tw. (5353) 7 ((singl$ or doubl$ or treb$ or trip$) adj25 (blind$ or mask$ or dummy)).tw. (2197) 8 placebos/ (516) 9 placebo$.tw. (2475) 10 random$.tw. (12456) 11 research design/ (1663) 12 Prospective Studies/ (417) 13 meta analysis/ (106) 14 (meta?analys$ or systematic review$).tw. (1749) 15 control$.tw. (27000) 16 (multicenter or multicentre).tw. (710) 17 ((study or studies or design$) adj25 (factorial or prospective or intervention or crossover or cross‐over or quasi‐experiment$)).tw. (9504) 18 or/1‐17 (41602) 19 carpal tunnel syndrome/ or carpal tunnel syndrome.tw. (443) 20 ((nerve entrapment or nerve compression or entrapment neuropath$) and carpal).mp. (53) 21 19 or 20 (444) 22 exp exercise therapy/ (4889) 23 yoga/ (336) 24 exp musculoskeletal manipulations/ (4074) 25 physical therapy.mp. (6285) 26 physical therapy modalities/ (2680) 27 (mobili?ation or physiotherapy or massage or yoga or chiropractic or osteopathic or gliding exercise*).mp. (21982) 28 or/22‐27 (30911) 29 18 and 21 and 28 (24)
Appendix 4. CINAHL Plus (EBSCOhost) search strategy
S28 =s18 and s24 and s27 =50 S27 =s25 or s26 =49183 S26 =mobilisation or mobilization or manipulation or physiotherapy or massage or yoga or chiropractic or osteopathic or gliding exercise* =46444 S25 =(MH "Manual Therapy+") =25306 S24 =s19 or s20 or s21 or s22 or s23 =1820 S23 =entrapment neuropath* and carpal =41 S22 =nerve compression and carpal =141 S21 =nerve entrapment and carpal =51 S20 =carpal tunnel syndrome =1813 S19 =(MH "Carpal Tunnel Syndrome") =1591 S18 =S1 or S2 or S3 or S4 or S5 or S6 or S7 or S8 or S9 or S10 or S11 or S12 or S13 or S14 or S15 or S16 or S17 =509020 S17 =ABAB design* =72 S16 =TI random* or AB random* =104569 S15 =( TI (cross?over or placebo* or control* or factorial or sham? or dummy) ) or ( AB (cross?over or placebo* or control* or factorial or sham? or dummy) ) =217146 S14 =( TI (clin* or intervention* or compar* or experiment* or preventive or therapeutic) or AB (clin* or intervention* or compar* or experiment* or preventive or therapeutic) ) and ( TI (trial*) or AB (trial*) ) =72504 S13 =( TI (meta?analys* or systematic review*) ) or ( AB (meta?analys* or systematic review*) ) =20589 S12 =( TI (single* or doubl* or tripl* or trebl*) or AB (single* or doubl* or tripl* or trebl*) ) and ( TI (blind* or mask*) or AB (blind* or mask*) ) =17178 S11 =PT ("clinical trial" or "systematic review") =96175 S10 =(MH "Factorial Design") =793 S9 =(MH "Concurrent Prospective Studies") or (MH "Prospective Studies") =165362 S8 =(MH "Meta Analysis") =13350 S7 =(MH "Solomon Four‐Group Design") or (MH "Static Group Comparison") =30 S6 =(MH "Quasi‐Experimental Studies") =5120 S5 =(MH "Placebos") =7223 S4 =(MH "Double‐Blind Studies") or (MH "Triple‐Blind Studies") =23000 S3 =(MH "Clinical Trials+") =133287 S2 =(MH "Crossover Design") =8720 S1 =(MH "Random Assignment") or (MH "Random Sample") or (MH "Simple Random Sample") or (MH "Stratified Random Sample") or (MH "Systematic Random Sample") =54473
Appendix 5. CENTRAL search strategy
#1"Carpal Tunnel Syndrome" #2("nerve entrapment" OR "nerve compression" OR "entrapment neuropath*") #3"median nerve entrapment" #4(#1 OR #2 OR #3) #5MeSH descriptor Musculoskeletal Manipulations explode all trees #6MeSH descriptor Physical Therapy Modalities explode all trees #7(#4 AND #6) #8mobilisation or mobilization or manipulation or physiotherapy or massage or yoga or chiropractic or osteopathic or "gliding exercise" #9(#5 OR #6 OR #7 OR #8) #10(#4 AND #9)
Data and analyses
Comparison 1. NERVE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term overall improvement (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 1.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 After 3 weeks of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in functional ability (hand function) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in functional ability (upper limb tension test) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in functional ability (active wrist flexion (degrees)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 After 3 weeks of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in functional ability (active wrist extension (degrees)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 After 3 weeks of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Need for surgery | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 7.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 2. CARPAL BONE MOBILISATION (SINGLE INTERVENTION) VERSUS CONTROL.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term overall improvement (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 1.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 After 3 weeks of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in functional ability (hand function) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in functional ability (upper limb tension test) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in functional ability (active wrist flexion (degrees)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 After 3 weeks of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in functional ability (active wrist extension (degrees)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 After 3 weeks of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Need for surgery | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 7.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 3. NEURODYNAMIC VS CARPAL BONE MOBILISATION.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term overall improvement (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 1.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less) Symptoms | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 After 3 weeks of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in functional ability (hand function) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in functional ability (upper limb tension test) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in functional ability (active wrist flexion (degrees)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 After 3 weeks of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in functional ability (active wrist extension (degrees)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 After 3 weeks of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Need for surgery | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 7.1 After 3 weeks of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 4. INSTRUMENT‐ASSISTED SOFT TISSUE MOBILISATION VERSUS STANDARD SOFT TISSUE MOBILISATION.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term overall improvement (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 1.1 3 months after treatment ended | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 Immediately after 6‐8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2.2 3 months after treatment ended | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in CTS symptoms (Levine) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 Immediately after 6‐8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3.2 3 months after treatment ended | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in functional ability (Levine) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 4.1 Immediately after 6‐8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.2 3 months after treatment ended | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in functional ability (grip strength) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 Immediately after 6‐8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5.2 3 months after treatment ended | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in functional ability (pinch strength opposition) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 Immediately after 6‐8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6.2 3 months after treatment ended | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Short‐term improvement in functional ability (pinch strength key) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 7.1 Immediately after 6‐8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.2 3 months after treatment ended | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Short‐term improvement in functional ability (extension range of movement) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8.1 Immediately after 6‐8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8.2 3 months after treatment ended | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9 Short‐term improvement in functional ability (flexion range of movement) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 9.1 Immediately after 6‐8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.2 3 months after treatment ended | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10 Short‐term improvement in distal sensory latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 10.1 Immediately after 6‐8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10.2 3 months after treatment ended | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 11 Short‐term improvement in distal motor latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 11.1 Immediately after 6‐8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 11.2 3 months after treatment ended | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 5. TARGETED CTS SOFT TISSUE MASSAGE VERSUS GENERAL SOFT TISSUE MASSAGE.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term improvement in CTS symptoms (Levine) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 Two days after 7th massage session (week 4) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 1.2 Two days after 11th massage session (week 6) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 1.3 At week 10 (4 weeks follow‐up) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in functional ability (Levine) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 Two days after 7th massage session (week 4) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2.2 Two days after 11th massage session (week 6) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2.3 At week 10 (4 weeks follow‐up) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in functional ability (isometric grip strength) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 Two days after 7th massage session (week 4) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3.2 Two days after 11th massage session (week 6) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3.3 At week 10 (4 weeks follow‐up) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in functional ability (isometric pinch strength) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 4.1 Two days after 7th massage session (week 4) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.2 Two days after 11th massage session (week 6) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.3 At week 10 (4 weeks follow‐up) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in functional ability (Grooved pegboard test) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 Two days after 7th massage session (week 4) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5.2 Two days after 11th massage session (week 6) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5.3 At week 10 (4 weeks follow‐up) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 6. NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS SPLINT.
Comparison 7. NERVE AND TENDON GLIDING EXERCISES VERSUS SPLINT PLUS STEROID INJECTION.
Comparison 8. NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS STEROID INJECTION VERSUS SPLINT PLUS STEROID INJECTION.
Comparison 9. NERVE AND TENDON GLIDING EXERCISES PLUS THERAPEUTIC ULTRASOUND PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 1.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (Levine) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in CTS symptoms (Phalen's sign) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3.1 At end of treatment (3 weeks) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3.2 At 11 weeks | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in CTS symptoms (Tinel's sign) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4.1 At end of treatment (3 weeks) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.2 At 11 weeks | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in functional ability (Levine) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in functional ability (hand grip strength) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Short‐term improvement in functional ability (pinch strength) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 7.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Short‐term improvement in motor distal latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9 Short‐term improvement in sensory distal latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 9.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10 Long‐term improvement in CTS symptoms (>3 months) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 10.1 At 11 weeks | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 10. NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT VERSUS THERAPEUTIC ULTRASOUND PLUS SPLINT.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 1.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (Levine) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in CTS symptoms (Phalen's sign) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3.1 At end of treatment (3 weeks) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3.2 At 11 weeks | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in CTS symptoms (Tinel's sign) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4.1 At end of treatment (3 weeks) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.2 At 11 weeks | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in functional ability (Levine) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short term improvement in functional ability (hand grip strength) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Short term improvement in functional ability (pinch strength) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 7.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Short term improvement in motor distal latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9 Short‐term improvement in sensory distal latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 9.1 After end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.2 At 11 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10 Long‐term improvement in CTS symptoms (>3 months) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 10.1 At 11 weeks | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 11. NEURODYNAMIC TECHNIQUE PLUS SPLINT VERSUS "SHAM" NEURODYNAMIC TECHNIQUE PLUS SPLINT.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term improvement in CTS symptoms (pressure pain (MVAS)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 At the end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (temporal summation (MVAS)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 At the end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in CTS symptoms (usual pain (NRS)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 At the end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in CTS symptoms (clinical pain (MVAS)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 4.1 At the end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in CTS symptoms (thermal pain (MVAS)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 At the end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in functional ability (DASH questionnaire) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 At the end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Short‐term improvement in functional ability (grip strength) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 7.1 At the end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Short‐term improvement in motor distal latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8.1 At the end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9 Short‐term improvement in combined sensory index (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 9.1 At the end of 3 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 12. NERVE AND TENDON GLIDING EXERCISES PLUS SPLINT PLUS EDUCATION VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS EDUCATION.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term improvement in CTS symptoms (Levine symptom severity score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 After one month of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in functional ability (Levine functional status score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 After one month of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in functional ability (DASH score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 After one month of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in functional ability (neurodynamic irritability of median nerve (R1, °)) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 4.1 After one month of treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 13. NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (Levine symptom status score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in functional ability (Levine functional status score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in functional ability (DASH score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 4.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in health‐related quality of life (WHOQOLF Physical Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in health‐related quality of life (WHOQOLF Psychologic Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Short‐term improvement in health‐related quality of life (WHOQOLF Social Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 7.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Short‐term improvement in health‐related quality of life (WHOQOLF Environmental Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 14. NERVE GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (Levine symptom status score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in functional ability (Levine functional status score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in functional ability (DASH score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 4.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in health‐related quality of life (WHOQOLF Physical Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in health‐related quality of life (WHOQOLF Psychologic Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Short‐term improvement in health‐related quality of life (WHOQOLF Social Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 7.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Short‐term improvement in health‐related quality of life (WHOQOLF Environmental Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 15. TENDON GLIDING EXERCISES PLUS SPLINT PLUS PARAFFIN THERAPY VERSUS SPLINT PLUS PARAFFIN THERAPY.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term improvement in CTS symptoms (VAS pain) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (Levine symptom status score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in functional ability (Levine functional status score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in functional ability (DASH score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 4.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in health‐related quality of life (WHOQOLF Physical Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in health‐related quality of life (WHOQOLF Psychologic Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Short‐term improvement in health‐related quality of life (WHOQOLF Social Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 7.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Short‐term improvement in health‐related quality of life (WHOQOLF Environmental Domain score) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8.1 Change from baseline to two months post‐treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 16. NERVE GLIDING EXERCISES PLUS SPLINT PLUS ACTIVITY MODIFICATION VERSUS SPLINT PLUS ACTIVITY MODIFICATION.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short term overall improvement (no pathological finding on NCS) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 1.1 At end of 10 weeks treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (VAS pain 0 to 10) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 At end of 10 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in CTS symptoms (Phalen's sign) (3 months or less) | 1 | Risk Ratio (M‐H, Random, 95% CI) | Totals not selected | |
| 4 Short‐term improvement in CTS symptoms (Tinel's sign) (3 months or less) | 1 | Risk Ratio (M‐H, Random, 95% CI) | Totals not selected | |
| 5 Short‐term improvement in functional ability (grip strength (kg)) (3 months or less) | 1 | Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 6 Short‐term improvement in functional ability (pinch strength (kg)) (3 months or less) | 1 | Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 7 Short‐term improvement in functional ability (motor function of abductor pollicis brevis muscle) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 7.1 At the end of 10 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Short‐term improvement in functional ability (two‐point discrimination test) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8.1 At the end of 10 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9 Short‐term improvement in functional ability (light‐touch deep‐pressure sense) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 9.1 At the end of 10 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 17. CHIROPRACTIC TREATMENT PLUS SPLINT VERSUS IBUPROFEN PLUS SPLINT.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Adverse effects | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 1.1 Summarised at the end of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (CTOA physical distress) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 At end of 9 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in CTS symptoms (CTOA mental distress) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 At end of 9 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in CTS symptoms (vibrometric threshold of finger sensation) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 4.1 Right hand at 13 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.2 Left hand at 13 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in functional ability (HAND) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 At 13 weeks (4 weeks after treatment ended) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in functional ability (SF‐36 Body pain) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 At 13 weeks (4 weeks after treatment ended) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Short‐term improvement in functional ability (SF‐36 Global) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 7.1 At 13 weeks (4 weeks after treatment ended) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Short‐term improvement in functional ability (SF‐36 Role physical) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8.1 At 13 weeks (4 weeks after treatment ended) | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9 Short‐term improvement in median nerve motor wrist (onset) latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 9.1 Right hand at end of 9 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.2 Left hand at end of 9 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10 Short‐term improvement in median nerve sensory digit 2 latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 10.1 Right hand at end of 9 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10.2 Left hand at end of 9 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 11 Short‐term improvement in median nerve sensory digit 3 latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 11.1 Right hand at end of 9 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 11.2 Left hand at end of 9 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 12 Short‐term improvement in median nerve sensory palm (peak) latency (ms) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 12.1 Right hand at end of 9 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 12.2 Left hand at end of 9 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 18. YOGA VERSUS SPLINT.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term improvement in CTS symptoms (VAS Pain) (3 month or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 At end of 8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Short‐term improvement in CTS symptoms (sleep disturbance) (3 months or less | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 2.1 At end of 8 weeks treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Short‐term improvement in CTS symptoms (Tinel's sign) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3.1 At end of 8 weeks treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Short‐term improvement in CTS symptoms (Phalen's sign) (3 months or less) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4.1 At end of 8 weeks treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Short‐term improvement in functional ability (grip strength) (3 months or less) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 At end of 8 weeks treatment | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Short‐term improvement in median nerve motor distal latency (ms) (3 months or less) | 1 | Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 6.1 At end of 8 weeks treatment | 1 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Short‐term improvement in median nerve sensory distal latency (3 months or less) | 1 | Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 7.1 At end of 8 weeks treatment | 1 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Akalin 2002.
| Methods | Randomised controlled trial No blinding Randomisation occurred at the level of wrists, where participants with bilateral CTS received a different intervention for each wrist |
|
| Participants | Total n = 28 (36 wrists) randomised
Intervention group n = 14 (18 wrists) randomised
Control group n = 14 (18 wrists) randomised 2 males; 26 females Mean ± SD age: Intervention 51.7 ± 5.5 yrs Control 52.2 ± 5.6 yrs Inclusion criteria: 1. Subjective symptoms (history of paraesthesiae or pain in median nerve distribution, nocturnal pain and dysthesia) 2. Positive Phalen's test or Tinel's test 3. Electrophysiologic studies confirmed CTS diagnosis Exclusion criteria: 1. Underlying metabolic disorders (diabetes mellitus, thyroid disease) 2. Rheumatoid arthritis 3. Pregnancy 4. History of steroid injection to carpal tunnel 5. Severe thenar atrophy 6. History of splint use |
|
| Interventions | Intervention: nerve and tendon gliding exercises performed 5 times daily and use of a custom‐made neutral volar wrist splint for 4 weeks. During tendon gliding exercises, the fingers were placed in five discrete positions. Those were straight, hook, fist, table top, and straight fist. During the median nerve gliding exercise, the median nerve was mobilised by putting the hand and wrist in six different positions: (1) wrist in neutral position, fingers and thumb in flexion; (2) wrist in neutral position, fingers and thumb extended; (3) wrist and fingers extended, thumb in neutral position; (4) wrist, fingers, and thumb extended; (5) forearm in supination; (6) the opposite hand applies a gentle stretch to the thumb) (Totten 1991). Control: custom‐made neutral volar wrist splint for 4 weeks Participants in both groups were instructed to wear the splint all night and during the day as much as possible |
|
| Outcomes | Outcomes assessed at 12 weeks (8 weeks following end of treatment), except for assessment of patient satisfaction occurred between 5 and 11 months post intervention (mean 8 months) 1. Grip strength (in lbs) (Martin vigorimeter) 2. Pinch strength (in lbs) (Martin vigorimeter) 3. Static two‐point discrimination of the pulps of radial 3 digits (in mm) 4. Tinel's test (rated as positive or negative) 5. Phalen's test (rated as positive or negative) 6. Symptoms using carpal tunnel questionnaire (rates 11 items on ordinal scale 1 to 5) 7. Hand function using carpal tunnel questionnaire (rates 8 items on ordinal scale 1 to 5) 8. Patient satisfaction (rates as excellent, good, fair, poor). Excellent = completely asymptomatic, good = occasional symptoms, fair = frequent symptoms but still some improvement, poor = continuous symptoms |
|
| Notes | Analysis was undertaken at the wrist‐level for all outcomes, though some participants in each group had bilateral CTS. Bilateral cases had a different intervention applied to each wrist. The trialists did not report how the correlation between both wrists was accounted for in the analysis, and attempts to clarify this information from the trialists were unsuccessful. Therefore, it is not clear whether a unit of analysis error occurred. No attempt was made to adjust outcome data. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "The patients were randomly divided into two groups. When the patients satisfied inclusion criteria, numbers were given to them. Group 1 consisted of the patients with odd numbers, and group 2 consisted of the patients with even numbers. If the patient had bilateral involvement, they were separately randomized by the same method. There were 14 patients in each group." |
| Allocation concealment (selection bias) | High risk | Comment: Communication with author (Akalin) confirmed that group assignments were not concealed prior to allocation. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: Participants were aware of their group assignment. This may have influenced self‐reported outcomes (symptom severity score, functional status score, patient satisfaction). |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Comment: Communication with author (Akalin) confirmed that outcome assessors were not blinded to group assignments. This may have influenced outcomes including Phalen's's sign, Tinel's test, two‐point discrimination, grip strength, and pinch strength. |
| Incomplete outcome data (attrition bias) 3 months or less | Low risk | Quote: "The mean and standard deviation of each parameter for all patients were obtained." Comment: Communication with author (Akalin) confirmed there were no drop‐outs in the study. |
| Incomplete outcome data (attrition bias) After 3 months | Low risk | Quote: "The mean and standard deviation of each parameter for all patients were obtained." Comment: Communication with author (Akalin) confirmed there were no drop‐outs in the study. |
| Selective reporting (reporting bias) | Low risk | Comment: All outcomes stated in the methods section of the publication were reported in the results. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Bahrami 2006.
| Methods | Quasi‐randomised controlled trial No blinding reported It is unclear whether randomisation occurred at the level of participants or wrists, and whether all bilateral CTS participants received the same or different intervention for each wrist |
|
| Participants | Total n = 28 (38 wrists) randomised
Intervention group n = 19 wrists randomised (number of participants unclear)
Control group n = 19 wrists randomised (number of participants unclear) 2 males; 26 females Mean ± SD age: Intervention 51.37 ± 9.49 yrs Control 49.37 ± 6.6 yrs Mean ± SD duration of CTS symptoms: Intervention 7.89 ± 3.4 months Control 7.3 ± 3.2 months Inclusion criteria: 1. Mild to moderate CTS 2. Haringtone JM et al criteria (reference 14) 3. Pain and numbness in median innervated parts plus one of the followings: (a) Positive Tinel's test; (b) Positive Phalen's test; (c) Electrodiagnostic findings of CTS 4. Electrophysiologic criteria: median nerve sensory distal latency > 3.5 ms or median nerve motor distal latency > 4.2 ms or difference of median nerve sensory distal latency and ulnar nerve sensory distal latency > 0.5 ms. Exclusion criteria: 1. Any metabolic disease (diabetes mellitus or thyroid disease) 2. Rheumatoid arthritis 3. Pregnancy 4. History of corticosteroid injection in wrist 5. Tenra atrophia 6. Any clinical or electrophysiological sign of other diseases which can imitate CTS symptoms such as peripheral neuropathy, cervical radiculopathy (C6‐C7). 7. Any neurogenic findings in C6‐C7 myotome or thenar muscles. 8. Median nerve sensory distal latency or median nerve motor distal latency > 6 ms or unrecordable. 9. Signs of peripheral neuropathy |
|
| Interventions | Intervention: nerve and tendon gliding exercises to be performed 4 times a day for 4 weeks, plus wrist splint in five degrees dorsi flexion (rest) was prescribed to be used at nights for 4 weeks. Participants also received pictures of exercises and were advised to do each exercise 10 times in each section and retain their hand in each position for 5 s. During exercises, neck and shoulder had natural position and elbow was 90° flexed. Control: Wrist splint in five degrees dorsi flexion (rest) was prescribed to be used at nights for 4 weeks. |
|
| Outcomes | Outcomes assessed at baseline and 6 weeks after treatment ended, except for patient satisfaction which was measured at 6 months post‐treatment: 1. Symptoms using carpal tunnel questionnaire (rates 11 items on ordinal scale 1: least, to 5: most) 2. Hand function using carpal tunnel questionnaire (rates 8 items on ordinal scale 1: least, to 5: most) 3. Tinel's test 4. Phalen's test 5. Nerve conduction: median nerve sensory distal latency, median nerve motor distal latency, median‐ulnar sensory distal latency 6. Patient satisfaction measured via telephone (no details on measurement scale used) |
|
| Notes | Full text publication was written in Persian and translated by the Neuromuscular Disease Review Group. Comment: 19 participants had unilateral CTS and 9 participants had bilateral CTS. The numbers of unilateral and bilateral cases in each group were not reported, and it is not clear whether bilateral CTS participants received the same intervention for both wrists. The trialists did not report how the correlation between both wrists was accounted for in the analysis, and attempts to clarify this information from the trialists were unsuccessful. Therefore, it is not clear whether a unit of analysis error occurred. No attempt was made to adjust outcome data. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | Quote: "Patients with even number of medical recordings entered group 1 while patients with odd numbers entered group 2. For patients with both hands involved, different odd and even numbers were considered for each hand; thus one of their hands were in group 1 and the other in group 2" (personal communication with trialists). Comment: A non‐random sequence (alternation) was used to allocate participants. |
| Allocation concealment (selection bias) | High risk | Quote: "Patients with even number of medical recordings entered group 1 while patients with odd numbers entered group 2. For patients with both hands involved, different odd and even numbers were considered for each hand; thus one of their hands were in group 1 and the other in group 2" (personal communication with trialists). Comment: A non‐random sequence (alternation) was used to allocate participants, so the allocation was unlikely to be concealed to individuals responsible for enrolling participants in the study. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: No blinding of participants was reported, and due to the nature of the interventions, it is unlikely that participants and personnel were not aware of which group participants were allocated to. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: It is not clear if assessors of objective outcomes were blinded. |
| Incomplete outcome data (attrition bias) 3 months or less | Low risk | Comment: All outcomes measured at three months or less were based on all randomised wrists |
| Incomplete outcome data (attrition bias) After 3 months | Unclear risk | Comment: Patient satisfaction measured at five to eight months post‐treatment was measured on 17/19 wrists in the intervention group and 18/19 wrists in the control group. The reasons for missing outcome data were not reported, so it is not clear whether missing data may have influenced the results or not. |
| Selective reporting (reporting bias) | Unclear risk | Comment: It is unclear if any additional measured outcomes were not reported |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Bardak 2009.
| Methods | Randomised controlled trial Blinded outcome assessors |
|
| Participants | Total n = 111 (111 wrists) randomised Intervention group 1 n = 41 (41 wrists) randomised; 41 (41 wrists) completed Intervention group 2 n = 35 (35 wrists) randomised; 35 (35 wrists) completed Intervention group 3 n = 35 (35 wrists) randomised; 35 (35 wrists) completed 3 males, 108 females Mean ± SD age: Intervention group 1: 33 ± 9.6 yrs Intervention group 2: 26 ± 10.3 yrs Intervention group 3: 22 ± 9.9 yrs Mean± SD duration of CTS symptoms: Intervention group 1: 13.3 ± 8.6 months Intervention group 2: 12.9 ± 8.8 months Intervention group 3: 19 ± 16.3 months Inclusion criteria: 1. Diagnosed according to Lundborg classification as intermediate stage CTS, characterised as nocturnal increase in the carpal tunnel tissue pressure Exclusion criteria: 1. Diagnosed according to Lundborg classification as early stage or late stage CTS 2. Had diabetes mellitus 3. Had thyroid diseases 4. Had rheumatoid arthritis 5. Had peripheral neuropathy 6. Had cervical radiculopathy 7. Had CTS with thenar atrophies 8. Were pregnant 9. Had history of steroid injections or splinting 10. Had bilateral CTS |
|
| Interventions | Intervention group 1: Standard conservative treatment consisting of a neutral splint worn day and night for the first 3 weeks and then at night only for the next 3 weeks, and 3 mg betamethasone (steroid) injection into the carpal groove. Intervention group 2: standard conservative treatment (see above) plus tendon and median nerve gliding exercises performed at home 3 times a day with every particular exercise repeated 5 times for a period of 6 weeks (exercises were demonstrated by a physiotherapist initially and participants received a brochure describing the exercises, and were asked to complete the exercises at home with a weekly follow‐up with the physiotherapist to ensure the exercises were being performed properly). For tendon gliding exercises, the fingers were placed in five different positions (straight, hook, fist, table top, and straight fist) and were kept in this position for 7 seconds. For median nerve gliding exercises, wrists and hands were placed in six different positions: 1. Keep the wrist in neutral position, fingers and thumb in flexion; 2. Keep the wrist in neutral position, fingers and thumb extended; 3. Keep the thumb in neutral position, wrist and fingers are in extension; 4. Wrist, fingers, and thumb extended; 5. Keep the wrist, fingers, and the thumb in extension while forearm is in supination; 6. The opposite hand applies a gentle stretch to the thumb (Totten 1991). Intervention group 3: tendon and median nerve gliding exercises performed at home 3 times a day with every particular exercise repeated 5 times for a period of 6 weeks (see above) |
|
| Outcomes | Outcomes assessed at baseline and 8 weeks after treatment ended: 1. Symptom total point, calculated as the sum of 5 scores (scored as symptomatic = 1 point or asymptomatic = 0 points) for five symptoms (hand pain, tingling, numbness, nocturnal numbness, and interrupted sleep). The total score ranges from 0 to 5, with lower scores denoting fewer symptoms 2. Functional status score, calculated as the sum of 7 scores for ability to perform 7 daily living activities (writing, buttoning clothes, gripping a telephone receiver, opening jars, doing housework, carrying grocery bags, bathing), each scored as 1 = easy, 2 = somewhat difficult, 3 = moderately difficult, 4 = very difficult, 5 = impossible). The total score ranges from 7 to 35, with lower scores denoting better function. 3. Phalen's test 4. Tinel's test 5. Reverse Phalen's test 6. Compression test 7. Pain measured on a visual analogue scale (scale properties not reported)* 8. Static two‐point discrimination (mm) performed on the pulp of the 3 radial digits, and the mean value was recorded 9. Patient satisfaction measured via telephone, where participants were asked to rate themselves as asymptomatic = excellent/good, symptomatic during difficult activities = fair or persistent symptoms after the treatment = poor (measured only at 11 months post‐treatment) |
|
| Notes | *No data reported on this outcome in the trial publication. Requests to obtain this data from the authors were unsuccessful. Only participants with unilateral CTS were included in the study, so a unit of analysis error resulting from the correlation between two wrists in bilateral CTS participants could not have occurred. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "For randomization of the patients into treatment groups, a biostatistician created a computer‐generated randomization list." Comment: The randomisation sequence was probably adequately concealed. |
| Allocation concealment (selection bias) | Unclear risk | Quote: "According to this list, numbered, sealed envelopes containing one of the treatment groups were prepared. When patients entered the study, the corresponding envelope was opened and the enclosed card determined the treatment group" Comment: It is not clear whether the sealed, numbered envelopes were opaque, so it is not clear whether the allocation sequence was adequately concealed. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "Two investigators were assigned to this study. One of the investigators was blind to the therapy given to the patient and only evaluated the subjective symptoms, clinical examinations, and the functional status of the patient. These evaluations were carried out pretreatment and 8 weeks posttreatment. The second investigators was blind to the functional status and symptoms of the patients and only applied the treatment" Comment: Patients and personnel delivering the intervention were probably not blind to treatment allocation, given the nature of the interventions. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "Two investigators were assigned to this study. One of the investigators was blind to the therapy given to the patient and only evaluated the subjective symptoms, clinical examinations, and the functional status of the patient. These evaluations were carried out pretreatment and 8 weeks posttreatment. The second investigators was blind to the functional status and symptoms of the patients and only applied the treatment" Comment: The outcome assessor was probably blind to treatment allocation. |
| Incomplete outcome data (attrition bias) 3 months or less | Low risk | Comment: No drop‐outs or losses to follow‐up were reported in the trial publication, and the tables of outcome data clearly indicate that data reported are based on a complete sample of participants who were randomised |
| Incomplete outcome data (attrition bias) After 3 months | Low risk | Comment: No drop‐outs or losses to follow‐up were reported in the trial publication, and the tables of outcome data clearly indicate that data reported are based on a complete sample of participants who were randomised |
| Selective reporting (reporting bias) | High risk | Comment: All outcomes specified in the methods section of the publication were reported in the results section of the publication, except for the outcome VAS pain score. Further, no protocol or trial registry entry was identified, and it is not clear whether the outcome commonly measured in other CTS trials, nerve conduction, was measured as an outcome. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Baysal 2006.
| Methods | Randomised single‐blind controlled trial Blinded assessors Randomisation occurred at the level of participants, not wrists (i.e. participants with bilateral CTS received the same intervention for both wrists) |
|
| Participants | Total n = 36 (72 wrists) randomised Intervention group 1 n = 12 (24 wrists) randomised; 12 (24 wrists) completed Intervention group 2 n = 12 (24 wrists) randomised; 8 (16 wrists) completed Intervention group 3 n = 12 (24 wrists) randomised; 8 (16 wrists) completed 0 males, 36 females Mean ± SD age: Intervention group 1: 47.8 ± 5.5 yrs Intervention group 2: 50.1 ± 7.3 yrs Intervention group 3: 51.4 ± 5.2 yrs Mean± SD duration of CTS symptoms: Intervention group 1: 1.5 ± 1.6 yrs Intervention group 2: 1.4 ± 0.8 yrs Intervention group 3: 1.4 ± 0.8 yrs Inclusion criteria: 1. Subjectively reported history of paraesthesia or pain in the median nerve distribution, nocturnal pain, and dysthesia 2. Tinel's test, Phalen's test, pain measurement, two‐point discrimination test, and grip and pinch strength measurement (no information provided on which criteria for these physician‐assessed outcomes had to be fulfilled by participants) Exclusion criteria: 1. Secondary entrapment neuropathies 2. Treated with ultrasound for CTS 3. Required regular analgesic or anti‐inflammatory drugs. 4. Clinical sign for axonal degeneration of the median nerve (thenar atrophy) on electromyographic examination of the abductor pollicis brevis muscle 5. Evidence of denervation (abnormal spontaneous activity in the form of fibrillations and positive sharp waves) on electromyographic examination of the abductor pollicis brevis muscle 6. History of steroid injection into the carpal tunnel, thyroid disease, diabetes, systemic peripheral neuropathy, pregnancy, or splint use |
|
| Interventions | Intervention group 1: Splinting and exercise therapy Intervention group 2: Splinting and ultrasound therapy Intervention group 3: Splinting, exercise, and ultrasound therapy (No description of how these combinations of treatments were completed by participants over the three‐week treatment period) Splinting ‐ A custom‐made neutral volar splint was given to patients. The patients were instructed to wear the splints all night and during the day for 3 weeks. Ultrasound therapy ‐ Ultrasound treatment was administered 15 min per session to the palmar carpal tunnel area at a frequency of 1 MHz and intensity of 1.0 W/cm2, pulsed mode 1 : 4, with a transducer of 5 cm2 (Electronica Pagani FP‐942/S) and with aquasonic gel as the couplant. The apparatus was standardised initially, and the output was controlled regularly by a simple underwater radiation balance. A total of 15 ultrasound treatments were performed once a day, five times a week, for 3 weeks. Exercise therapy ‐ Participants were instructed to perform nerve‐and tendon gliding exercises developed by Totten and Hunter (Totten 1991). Brochures describing exercises were also given to patients.The exercises were applied as five sessions daily. Each exercise was repeated 10 times at each session. Exercise treatment was continued for 3 weeks. During tendon‐gliding exercises, the fingers were placed in five discrete positions. Those were straight, hook, fist, table top, and straight fist. During the median nerve‐gliding exercise, the median nerve was mobilised by putting the hand and wrist in six different positions. |
|
| Outcomes | Outcomes measured at the first treatment session, at the end of therapy, and 8 weeks after treatment ended (11 weeks from baseline):* 1. Pain using a visual scale (VAS), on which the patients could indicate their assessment along a distance of 10 cm, ranging from 0 (no pain at all) to 10 (the most intense pain that I can imagine)** 2. Tinel's test (rated as positive or not) 3. Phalen's test (rated as positive or not) 4. Two‐point discrimination: performed on the pulp of the three radial digits 5. Symptoms using carpal tunnel questionnaire (rates 11 items on ordinal scale 1: mildest, to 5: most severe)** 6. Hand function using carpal tunnel questionnaire (rates 8 items on ordinal scale 1: no difficulty with the activity, to 5: cannot perform the activity at all)** 7. Hand grip strength using a handheld dynamometer: average force of three consecutive trials calculated** 8. Pinch grip strength using a standard dynamometer between the tips of the thumb and the little finger: average force of three consecutive trials calculated** 9. Nerve conduction: median nerve motor distal latency (ms) and sensory distal latency (ms)** 10. Satisfaction using a question asked over the telephone: rated as excellent if a patient is asymptomatic, good: rarely symptomatic, fair: symptomatic only during compelling activity or poor: continuing symptoms (without relief following treatment) (measured at an average of 11 ± 4.5 months post‐treatment). |
|
| Notes | **Data reported as endpoint scores and as change from baseline and change from end of treatment to 8 weeks follow‐up scores only if the difference between time points was significant (P < 0.05). Therefore, only endpoint scores for each outcome were included in the review, as these were rated to be a low risk of reporting bias Comment: Analysis was undertaken at the wrist‐level for all outcomes, though all participants in each group had bilateral CTS. Bilateral cases had the same intervention applied to each wrist. The trialists did not report how the correlation between both wrists was accounted for in the analysis, and attempts to clarify this information from the trialists were unsuccessful. Therefore, it is not clear whether a unit of analysis error occurred. No attempt was made to adjust outcome data. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Computer‐generated randomization list was created by a biostatistician." Comment: The allocation sequence was probably adequately generated. |
| Allocation concealment (selection bias) | Unclear risk | Quote: "Computer‐generated randomization list was created by a biostatistician. It was given to the physiotherapy department in sealed numbered envelopes. When the patients qualified to enter the study, appropriate numbered envelope was opened at the reception; the card inside indicated the patient's allocation to a treatment group." Comment: It is not clear whether the sealed numbered envelopes were opaque and sequentially numbered, therefore it is not clear whether the allocation sequence was adequately concealed until interventions were assigned. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: Not reported, and given the nature of the intervention it is unlikely that participants were not aware of which group they were assigned to. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "The staff who assessed the outcomes were different from the staff administering the treatments and were blinded to the type of treatment each patient had received." Comment: Outcome assessors of objectively measured outcomes were probably blinded to treatment assignment. |
| Incomplete outcome data (attrition bias) 3 months or less | Low risk | Quote: "Twenty‐eight patients (56 wrists) completed the study. The eight drop‐outs are described as follows: two patients (group II) underwent surgery, two patients (group II) were lost to follow‐up. In group III, two patients were lost to follow‐up, and another two patients (group III) refused electrophysiologic study due to improvement of symptoms." Comment: The eight randomised participants who were drop‐outs and losses to follow‐up were clearly described, |
| Incomplete outcome data (attrition bias) After 3 months | High risk | Comment: There is no explanation for why results of the patient satisfaction questionnaire is based on fewer than 28 participants with 56 wrists. |
| Selective reporting (reporting bias) | Low risk | Comment: All outcomes were fully reported as endpoint scores at the end of treatment and at eight weeks follow‐up. The authors also reported change from baseline scores for some (not all) of the outcomes, but numerical data suitable for inclusion in a meta‐analysis were only reported if the effect estimate was statistically significant. For non‐significant effects, the authors only reported that the result was "NS". Given endpoint scores were available and no meta‐analysis was performed, this selective reporting of data is unlikely to affect the results. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Bialosky 2009.
| Methods | Randomised controlled trial Blinded participants |
|
| Participants | Total n = 40 participants (40 wrists) randomised; 39 participants (39 wrists) completed Intervention group n = 20 participants (20 wrists) randomised; 19 participants (19 hands) completed Control group n = 20 participants (20 wrists) randomised; 20 participants (20 wrists) completed 0 males, 40 females Mean ± SD age: Intervention group: 44.3 ± 6.97 yrs Control group: 49.5 ± 12.35 yrs Mean ± range duration of CTS symptoms: Intervention group: 104 ± 30 to 221 weeks Control group: 364 ± 153 to 520 weeks Inclusion criteria: 1. Aged between 18 and 70 years 2. Patient had signs and symptoms consistent with CTS, as defined by pain or paraesthesia in the median nerve distribution and/or clinical examination findings consistent with CTS 3. Patient had CTS symptoms present for greater than 12 weeks 4. Patient had a rating of their CTS pain intensity or symptom intensity of at least 4/10 on a numeric rating scale (range 0 to 10, with 0 indicating no pain at all and 10 indicating the worst pain imaginable over the past 24 hours) Exclusion criteria: 1. Non‐English speaking 2. Prior surgery for CTS 3. Prior treatment with the studied neurodynamic technique 4. Pregnancy 5. Diagnosed with a systemic disease known to cause peripheral neuropathy 6. Current or history of chronic pain conditions 7. CTS as a result of an upper extremity fracture |
|
| Interventions | Intervention: neurodynamic technique which was intended to provide anatomical stress across the median nerve, including contralateral cervical sidebending, shoulder depression, shoulder abduction and external rotation to 90°, full elbow extension, and forearm supination (Butler 1991). Participants received 2 treatment sessions per week over period of 3 weeks. Participants also wore a splint at night and during daytime activities that worsened CTS symptoms for 3 weeks. Control: "Sham" neurodynamic technique that minimised anatomical stress across the median nerve and included neutral cervical spine positioning (i.e., no sidebending), no shoulder depression, shoulder abduction and external rotation to 45°, 45° of elbow extension, and forearm pronation. Participants received 2 treatment sessions per week over period of 3 weeks. Participants also wore a splint at night and during daytime activities that worsened CTS symptoms for 3 weeks. |
|
| Outcomes | Outcomes assessed at baseline, immediately after the first treatment session, and at the end of 3 weeks treatment: 1. Clinical pain intensity measured using a 100 mm mechanical visual analogue scale (MVAS), anchored with "no pain" and "the most intense pain sensation imaginable" 2. Pressure pain intensity measured using a 100 mm MVAS, anchored with "no pain" and "the most intense pain sensation imaginable", following application of a 2.3 kg force applied at a rate of 1 kg/s through a 1cm2 application tip at the thenar eminence 3. Thermal pain sensitivity measured using a 100 mm MVAS, anchored with "no pain" and "the most intense pain sensation imaginable" 4. Temporal summation measured using a 101‐point numerical rating scale (NRS) anchored with "no pain" and "the most intense pain imaginable", following application of heat pulses at 51°C applied to the thenar surface of the palm of the hand. 5. "Usual pain", measured using a 101‐point NRS 6. Disability measured using the 11‐item Quick Disability of the Arm, Shoulder and Hand (DASH) Questionnaire, which provides a range of 11 to 55 possible points, with lower values indicating less disability 7. Grip strength measured using a Jamar hydraulic hand dynamometer, with the average of 3 trials recorded in kilograms* 8. Sensation at the tip of the thumb, index finger and middle finger, measured using a Semmes‐Weinstein monofilament. The monofilament at which the participant indicated sensation was recorded* 9. Nerve conduction: Motor distal onset latency at the abductor pollicis brevis muscle and peak amplitude at the abductor pollicis brevis muscle, combined sensory index (calculated by measuring the sensory nerve action potentials peak latency and amplitude from the median, ulnar and radial nerves as they cross the wrist; the differences measures between the nerves are combined into an overall score with a value greater than one indicating a slowing of median nerve conduction through the carpal tunnel)* |
|
| Notes | *Only exact P values and partial η2 values for differences in the outcomes, grip strength, sensation, and neurophysiologic parameters were reported in the publication. Numerical summary data (e.g. means and SDs) for these outcomes were requested from the authors, and data on grip strength and nerve conduction were provided. Trialists reported that "The assigned intervention was applied bilaterally to all participants, regardless of whether their CTS complaints were bilateral", and that "Twenty‐seven participants (68%) reported bilateral CTS. Of the participants reporting bilateral CTS, 22 reported one side as more symptomatic, and this side was used in the analysis. Five participants reporting bilateral complaints were unable to identify one side as worse than the other, and the data for the dominant arm were analysed." Therefore, a unit of analysis error resulting from the correlation between two wrists in bilateral CTS participants could not have occurred in this study. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Randomization was computer generated..." Comment: Random sequence generation was probably adequately concealed. |
| Allocation concealment (selection bias) | Low risk | Quote: "Randomization was computer generated, with group assignment maintained in sealed, sequentially numbered, opaque envelopes. The envelopes were opened in sequential order based on entry in the study and after all baseline measures were completed for the participant" Comment: Allocation sequence was probably adequately concealed. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "Approximately 3 weeks following randomization, participants were seen for a final visit, consisting of assessment of which intervention the participant believed they had received (NDT versus sham)" Quote: "Of the 37 participants for whom we were able to collect these data, 11/18 participants (61%) receiving the sham intervention and 7/19 participants (37%) receiving the NDT perceived having received the NDT" Quote: "The sham intervention was successful in blinding the participants" Comment: Participants were probably blind to the intervention they received. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "Assessment at the 3‐week follow‐up session was performed by an examiner blinded to group assignment" Comment: Outcome assessment was probably done by a blinded assessor. |
| Incomplete outcome data (attrition bias) 3 months or less | Unclear risk | Quote: "Thirty‐nine of the 40 (98%) enrolled participants completed both a baseline and 3‐week session. The participant not returning for the 3‐week follow‐up was assigned to the NDT group providing three 3‐week analysis data for 19/20 (95%) participants assigned to receive NDT and 20/20 (100%) participants assigned to receive the sham intervention" Quote: "A subgroup of 12 participants agreed to undergo baseline and 3‐week NCS. The 12 participants did not differ significantly from those not undergoing an NCS in terms of age, duration of symptoms, baseline pain, or baseline expectation for treatment (P >.05)." Quote: "The NCSs were done by a physician using his clinic's machine. We planned to do them on everyone, but ran into trouble coordinating both his schedule and the availability of the machine with the rest of the requirements of the study. We settled on a small sampling of 6 from each group in whom we could work everything out. Not optimal, but a hazard of clinical research" (personal communication from lead author) Comment: There were incomplete data reported for DASH (36/40), pressure pain (37/40), and NCS (12/40). The impact of this incomplete data on the results is unclear. |
| Selective reporting (reporting bias) | Low risk | Comment: numerical data suitable for meta‐analysis were reported only for the outcomes, clinical pain (MVAS), pressure pain (MVAS), thermal pain (MVAS), temporal summation (NRS), "usual pain" (MRS) and disability (DASH). Only exact P‐values and partial η2 values for differences in the outcomes, grip strength and neurophysiologic parameters were reported in the publication, but numerical summary data for these outcomes were provided by the authors on request. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Brininger 2007.
| Methods | Randomised controlled trial No blinding reported |
|
| Participants | Total n = 61 participants (61 wrists) randomised Intervention group 1 n =16 wrists randomised, 14 wrists completed Intervention group 2 n = 17 wrists randomised, 13 wrists completed Intervention group 3 n =16 wrists randomised, 11 wrists completed Intervention group 4 n =12 wrists randomised, 13 wrists completed 10 males, 51 females* Mean ± SD age:* Intervention group 1: 49.0 ± 15.4 yrs Intervention group 2: 51.9 ± 15.7 yrs Intervention group 3: 46.6 ± 12.9 yrs Intervention group 4: 50.1 ± 13.2 yrs Inclusion criteria: 1. At least 18 years of age 2. Positive Tinel's test or Phalen's maneuver 3. Complaints of nocturnal numbness and tingling Exclusion criteria: 1. A neuropathy other than CTS in the past year 2. Pregnancy 3. Thenar atrophy 4. Steroid injection into the carpal canal in the past 3 months or a prior carpal tunnel release. |
|
| Interventions | Intervention group 1: Fabricated neutral wrist and metacarpophalangeal (MCP) splint with no exercises (neutral wrist and MCP): Participants received a customised, fabricated wrist splint positioning the wrist in neutral (0°) and the MCP joints from 0° to 10° of flexion. Participants were instructed to wear the splint during their regularly scheduled sleep time for 4 weeks. Intervention group 2. Fabricated neutral wrist and MCP splint with tendon and nerve gliding exercises (neutral wrist and MCP‐exercise): Participants received a customised, fabricated wrist splint positioning the wrist in neutral (0°) and the MCP joints from 0° to 10° of flexion. Participants were instructed to wear the splint during their regularly scheduled sleep time for 4 weeks. In addition, participants received visual and verbal instructions on tendon and nerve gliding exercises (Totten 1991). Participants were instructed to perform the exercises 3 to 5 times a day, with 10 repetitions in each position, and to hold each position for 5 seconds. Intervention group 3. Off‐the‐shelf, wrist cock‐up splint (immobilised in 20 degrees of extension) with no exercises (wrist cock‐up): Participants received a prefabricated, off‐the‐shelf wrist cock‐up splint that immobilised the wrist in 20° of extension. Participants were instructed to wear the splint during their regularly scheduled sleep time for 4 weeks. Intervention group 4. Off‐the‐shelf, wrist cock‐up splint (immobilised in 20 degrees of extension) with tendon and nerve gliding exercises (wrist cock‐up‐exercise): Participants received a prefabricated, off‐the‐shelf wrist cock‐up splint that immobilised the wrist in 20° of extension. Participants instructed to wear the splint during their regularly scheduled sleep time for 4 weeks. In addition, participants received visual and verbal instructions on tendon and nerve gliding exercises (Totten 1991). Participants were instructed to perform the exercises 3 to 5 times a day, with 10 repetitions in each position, and to hold each position for 5 seconds. |
|
| Outcomes | Outcomes assessed at baseline and at the end of treatment (4 weeks from baseline): 1. Symptoms using Boston carpal tunnel questionnaire (rates 11 items on ordinal scale 1: no symptoms, to 5: most severe pain)** 2. Hand function using Boston carpal tunnel questionnaire (rates 8 items on ordinal scale 1: no difficulty with the activity, to 5: unable to perform activity)** 3. Functional sensibility using the Moberg Pick‐up Test: participants are timed on how quickly they pick up an assortment of objects such as a coin, safety pin, and paper clip, and place them in a small box** 4. Grip strength using a hand‐held dynamometer: participants were given 3 opportunities to exert maximum force; the mean of 3 successive trials was recorded (higher scores indicate less impairment)** 5. Pinch strength using a reliable and accurate hand‐held pinch meter. Participants had 1 opportunity to exert maximum force with 3 types of pinch: tip pinch, lateral pinch, and palmar pinch (higher scores indicate less impairment)** 6. Satisfaction using an exit survey developed by the primary investigator that was designed to evaluate their level of satisfaction with the treatment provided (measured at the end of treatment only). No information on how this outcome is rated by participants and scored by outcome assessors** |
|
| Notes | *Data only reported for participants completing treatment (n = 51) **Data only reported overall from baseline to end of treatment or follow‐up for all intervention and control groups combined, and often only in the form of F‐ and P values. Thus, no data appropriate for meta‐analysis were entered into RevMan. The authors were contacted in order to retrieve this data, but efforts were unsuccessful. Interventions were only applied to one wrist per participant (even in bilateral patients). Therefore, a unit of analysis error resulting from the correlation between two wrists in bilateral CTS participants could not have occurred. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "Random allocation was made after subjects gave their informed consent and baseline assessments were completed. Subjects were randomized into groups by selecting a sealed opaque envelope that contained a number corresponding to an intervention group." Comment: Probably done, but not enough information to determine the adequacy of the randomisation sequence generation. |
| Allocation concealment (selection bias) | Low risk | Quote: "Random allocation was made after subjects gave their informed consent and baseline assessments were completed. Subjects were randomized into groups by selecting a sealed opaque envelope that contained a number corresponding to an intervention group." Comment: The allocation sequence was probably adequately concealed until interventions were assigned. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: Unlikely that participants would have been blinded to which treatment they were allocated to. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "Another limitation was that the person (TLB) who administered the treatment and evaluated outcomes was not masked to subjects' group assignments, and that may have biased the results." Comment: The outcome assessor (who also administered the interventions) was probably not blinded to treatment allocation. |
| Incomplete outcome data (attrition bias) 3 months or less | Low risk | Quote: "Sixty‐one of 79 eligible patients enrolled in the study. Four subjects withdrew because: they had an injection or surgery (n = 2), developed an illness (n = 1), or moved out of the area (n = 1); 6 subjects were lost to follow‐up". Comment: There were drop‐outs and losses to follow‐up in each of the four groups, and these were detailed, and unlikely to have biased the results. |
| Selective reporting (reporting bias) | High risk | Comment: Majority of the outcomes are reported incompletely (e.g., only as F‐values or P values from an ANOVA), and cannot be entered into a meta‐analysis. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Burke 2007.
| Methods | Randomised single‐blind controlled trial Blinded outcome assessor |
|
| Participants | Total n = 26 participants (26 wrists) randomised Intervention group n = 14 wrists randomised; 12 wrists completed Control group n = 12 wrists randomised; 10 wrists completed 3 males, 19 females* Mean ± SD age:* Intervention group 1: 39.8 ± 8.75 yrs Control group 2: 43.4 ± 5.32 yrs Inclusion criteria: 1. Electrophysiologic confirmation of the CTS diagnosis; namely, median nerve distal sensory latency of the index finger (> 3.60 ms) and/or (2) median nerve distal motor latency (DML; > 4.20 ms) 2. Present with pain and paraesthesia within the median nerve distribution. 3. Ratings of the Katz hand diagrams indicating categorization of CTS symptoms into “classic” or “probable.” 4. An initial self‐reported degree of pain rating of 33 mm or greater on the visual analogue scale (VAS) pain scale that ranged from 0 mm (no pain) to 100 mm (worst pain possible) for the overall hand‐wrist region. 5. Presence of at least 2 of 8 of the following clinical findings: sleep disturbances from hand symptoms (nocturnal paraesthesias), a mean symptom‐severity score of at least 3 of 5, a mean functional‐status score of at least 3 of 5, positive results on Tinel's’s sign, positive results on Phalen's’s sign, strength deficits, sensory deficits of touch, and limited range of movement Exclusion criteria: 1. Older than 50 years of age 2. Previous treatment interventions with surgery and/or steroid injections 3. History of wrist trauma 4. History suggesting underlying causes of CTS (e.g. diabetes mellitus, thyroid disease, pregnancy) 5. History of other musculoskeletal medical conditions (e.g. osteo or rheumatoid arthritis, reflex sympathetic dysfunction, fibromyalgia) 6. Pending lawsuits or insurance claims 7. Electrodiagnostic findings and physical examination findings that were inconsistent with the diagnosis of CTS 8. History by clinician that revealed that the patient actually met one of the exclusion criteria addressed in the initial phone interview screening process |
|
| Interventions | Intervention: Graston Technique instrument‐assisted soft tissue mobilisation (GISTM), which involved the use of an innovative, patented form of instrument‐assisted STM that enabled the clinician to effectively break down scar tissue and fascial restrictions of forearm‐wrist‐hand areas. Participants were scheduled to receive 2 treatments per week for the first 4 weeks and then receive 1 treatment per week for the next 2 weeks Control: Manual soft tissue mobilisation (STM) of forearm‐wrist‐hand areas with the clinician's hands to break down scar tissue and fascial restrictions. Participants rested their relaxed forearm‐wrist‐hand on the treatment table and the clinician applied deep pressure by fingers to scar tissue and taut muscle bands and stretched connective tissue and myofascial restrictions using both hands to replicate the treatment intervention delivered with the Graston Technique instruments. Participants were scheduled to receive 2 treatments per week for the first 4 weeks and then receive 1 treatment per week for the next 2 weeks |
|
| Outcomes | Outcomes assessed before treatment as part of the screening procedures, within 1 week of the last clinical treatment session, (i.e., at the end of 6 weeks of treatments) and at 3 months after the last clinical treatment session. 1. Nerve conduction: median nerve motor distal latency (ms) and sensory distal latency (ms) 2. Location of CTS symptoms using the self‐administered Katz hand diagrams (rated as classic, probable, or unlikely patterns of CTS according to the classification scheme). Participants completed this instrument also at each treatment session to monitor the progress of the treatment interventions.** 3. Pain: using a visual analogue scale ranging from 0 mm (no pain) to 100 mm (worst pain possible), to reflect the overall intensity of pain for the wrist‐hand areas during the previous week 4. Symptoms using carpal tunnel questionnaire (rates 11 items on ordinal scale 1: mildest, to 5: most severe) 5. Hand function using carpal tunnel questionnaire (rates 8 items on ordinal scale 1: no difficulty with the activity, to 5: cannot perform the activity at all) 6. Range of motion (degrees) for flexion and extension** using an inclinometer 7. Isometric pinch strength (kg) (key and opposition pinch) using the JAMAR Pinch Gauge 8. Isometric grip strength (kg) using the JAMAR Hand Dynamometer 9. Two‐point discrimination and pressure sensitivities of the first 3 digits of each hand. Participants were asked to identify the number of points touching the distal palmar pads of the first 3 digits. The outcome score for each digit of each hand was expressed as normal if 2 points were detected and abnormal if 1 point was detected.** 10. Pressure sensitivity using a Semmes‐Weinstein Monofilament Testing Set. The recorded score was the lightest pressure that was perceived by each digit of each hand. The outcome score was the mean of the pressure sensitivities of the first 3 digits.*** 11. Tinel's test (rated positive, as indicated by paraesthesias in the distribution of the thumb, index, and middle fingers, that is, median nerve distribution, or negative).*** 12. Phalen's test (rated positive, as indicated by paraesthesias in the distribution of the median nerve that occurred during a 60‐second test, or negative).*** 12. Patient satisfaction using a 5‐point scale, ranging from 0 (dissatisfied) to 5 (very satisfied) (measured immediately post intervention (i.e., after 6 weeks of treatment) and at 3 months follow‐up 13. Adverse effects (no information on how this outcome was recorded) |
|
| Notes | *Data only reported for participants completing treatment (n = 22) **The authors reported SDs and 95%CIs of the mean for each group, though the reported SDs for the GISTM group were actually SEs. Using the sample size and 95% CIs of the mean, we calculated the SDs using formulas provided in section 7.7.3.2 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011a) ***No numerical data suitable for meta‐analysis reported. Attempt to obtain suitable data from the authors were unsuccessful. Only participants with unilateral CTS were included in the study, so a unit of analysis error resulting from the correlation between two wrists in bilateral CTS participants could not have occurred. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Eligible patients were randomly allocated to receive either GISTM or STM. If bilateral symptoms were present, the wrist with more severe symptoms according to the patient was treated. A random sequence of 30 treatment interventions was generated by using random number tables by an administrative assistant." Comment: The allocation sequence was probably adequately generated. |
| Allocation concealment (selection bias) | Unclear risk | Quote: "The allocation was according to random sequence of 30 treatment interventions. The treating clinician, administrative assistant, and program coordinator were the only members of the research team with knowledge of the treatment allocation." Comment: There is not enough information to determine whether treatment allocation was adequately concealed until interventions were assigned. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "The treating clinician, administrative assistant, and program coordinator were the only members of the research team with knowledge of the treatment allocation. Patients were also encouraged not to reveal any information about their treatment interventions to the clinicians performing the clinical examination immediately post‐treatments and at 3 months' posttreatment." Comment: Participants knew which intervention they received (i.e., could feel the difference between hands being applied versus an instrument being applied to their hand) |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "The treating clinician, administrative assistant, and program coordinator were the only members of the research team with knowledge of the treatment allocation. Patients were also encouraged not to reveal any information about their treatment interventions to the clinicians performing the clinical examination immediately post‐treatments and at 3 months' posttreatment." Quote: "The clinicians responsible for data collection did not report any knowledge of the treatment received. The patients did not inadvertently mention their treatment intervention to these clinicians." Comment: Outcome assessors probably did not know which treatment participants were allocated to. |
| Incomplete outcome data (attrition bias) 3 months or less | Low risk | Quote: "Twenty‐six patients with CTS were enrolled into the research study and were randomly allocated to either GISTM (n = 14) or STM (n = 12). Four of these patients dropped out of the research study". Comment: Drop‐outs were clearly described and evenly distributed across the groups. |
| Selective reporting (reporting bias) | High risk | Comment: Most of the outcomes were reported as means, SDs and 95% CIs in table or figure format. However, Tinel's test, Phalen's's sign and 2‐point discrimination were only reported briefly in terms of whether a statistically significant result was found between CTS and control hand or between treatment groups. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Davis 1998.
| Methods | Randomised, single‐blind, controlled trial Blinded outcome assessors Randomisation occurred at the level of participants, not wrists (i.e. participants with bilateral CTS received the same intervention for both wrists) |
|
| Participants | Total n = 91 participants (149 wrists) randomised
Intervention group n = 45 participants (73 wrists) randomised
Control group n = 46 participants (76 wrists) randomised 37 males; 54 females Mean ± SD age: Intervention 38 ± 5 yrs Control 36 ± 6 yrs Inclusion criteria: 1. Positive electrodiagnostic testing 2. Positive clinical exam for CTS (pinch/grip strength, Phalen's's and Tinel's test, Semmes‐Weinstein monofilaments) 3. Symptoms of CTS including numbness and tingling 4. Age 21‐45 years Exclusion criteria: 1. Currently prescribed CTS treatment 2. Pending workers' compensation claim 3. Pregnancy 4. Systemic condition (diabetes, thyroid disorder) 5. Prior wrist surgery 6. Use of anti‐inflammatory medication or vitamin B6 supplementation 7. Wrist splint worn on regular basis 8. Electrodiagnostic abnormalities inconsistent with CTS or indicating axonal degeneration |
|
| Interventions | Intervention: Chiropractic treatment consisting of high velocity, low‐amplitude manual thrust procedures designed to create increased joint motion in the joints of the upper extremities, including the wrist, elbow and shoulder as well as in the cervical and upper thoracic regions of the vertebrae, myofascial massage/loading, ultrasound (over carpal tunnel at 1 MHz, 1.0‐1.5 W/cm2, for 5 minutes), and nocturnal wrist splint. Treatment was provided 3 times per week for 2 weeks, followed by twice per week for 3 weeks, then one treatment per week for 4 weeks*. Content of treatment session was at the discretion of chiropractic physician Control: Medical treatment consisting of ibuprofen (800 mg, 3 times per day for 1 week; 800 mg, 2 times per day for 1 week; 800 mg as required for 7 weeks to a maximum daily dose of 2400 mg) plus nocturnal wrist splint Total treatment length for both groups = 9 weeks |
|
| Outcomes | Outcome assessed at 9 and 13 weeks 1. Nerve conduction: median nerve motor and sensory distal latencies (at 9 weeks only) 2. Physical distress using CTS Outcome Assessment Physical Distress (CTOA‐P) scale (at 9 weeks only) 3. Mental distress using CTS Outcome Assessment Mental Distress (CTOA‐M) scale (at 9 weeks only) 4. Vibrometry (8‐500 Hz) on digit 3 using Total Jetzer Index (at 13 weeks only) 5. Hand function using Hand‐Finger Functioning (HAND) scale (at 13 weeks only) 6. Health‐related quality of life using Short Form 36 (SF36) scale (at 13 weeks only) 7. Adverse effects |
|
| Notes | *Ultrasound was provided for half of the chiropractic treatment visits. Analysis was undertaken at the participant‐level for the outcomes, physical distress, mental distress, hand function and health‐related quality of life, though some participants in each group had bilateral CTS. Communication with the trialists confirmed that bilateral cases had the same intervention applied to both wrists. However, the trialists did not report controlling for the correlation between both wrists for these outcomes, so a unit of analysis error is likely to have occurred for these outcomes (but not for the outcomes, nerve conduction and vibrometry, which were analysed separately for left‐ and right‐ affected wrists). No attempt was made to adjust outcome data. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Assignment was based on a computer‐generated random sequence of letters "A" and "B"..." Comment: The randomisation sequence was probably adequately generated. |
| Allocation concealment (selection bias) | Low risk | Quote: "...placed in a series of opaque envelopes in the order they printed, sealed and opened in sequence". "Neither investigators nor subjects were aware of group assignments before the envelopes were opened. The research consultant who generated the random sequence was not otherwise involved in the interviewing, random assignment process or treatment". Comment: The randomisation sequence was probably adequately concealed. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "Masking treatment group to subjects and treating physicians was not possible". Comment: Self‐reported outcomes including physical or mental distress, hand function, health‐related quality of life, and adverse effects may have been subject to performance bias as participants were aware of group assignments. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "It was possible to mask subject assignment to nerve conduction and vibrometric assessment physicians or technicians and the data analyst. Subject group identifiers were merged with the dataset only after data entry had been completed". Comment: Assessors of objective outcomes were likely blinded to treatment allocation |
| Incomplete outcome data (attrition bias) 3 months or less | High risk | Comment: Between 20%‐30% of participants were lost to follow‐up (4 weeks post‐treatment) in both groups. Reasons for losses were not provided except for two cases that dropped out of the conservative medical group due to intolerance to ibuprofen. A greater proportion of drop‐outs occurred in the chiropractic group which could be related to the intervention (22 visits required compared to 11 visits required for participants in conservative medical group) therefore results could be potentially biased in favour of the chiropractic intervention. |
| Selective reporting (reporting bias) | Low risk | Comment: All outcomes stated in the methods section of the publication were reported in the results. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Field 2004.
| Methods | Randomised controlled trial No blinding reported |
|
| Participants | Total n = 16 (16 wrists) randomised Intervention group n = Not reported Control group n = Not reported 1 male, 15 females Mean ± SD age: Total sample: 47 yrs (range 20 to 65 yrs) (SD not reported) Inclusion criteria: 1. Diagnosis of CTS 2. Work involved extensive time at the computer Exclusion criteria: Not reported |
|
| Interventions | Intervention: Massage therapy on the affected arm by a therapist once a week for a 4‐week period and self‐massage that was to be done daily at home prior to bedtime. The 15 minute massage consisted of moderate pressure stroking concentrated on the fingertip to elbow area. The massage began with stroking the wrist up to the elbow and back down on both sides of the forearm. Next, a wringing motion (much like milking a cow) was applied to the same area. This was followed by stroking, using the thumb and forefinger, in a circular or back and forth motion covering the entire forearm and hand. Finally, the skin was rolled using the thumb and forefinger across the hand and up both sides of the forearm. Control: Standard treatment (no massage therapy received during the study). Participants were taught the self‐massage routine after the end of the study. |
|
| Outcomes | Outcomes assessed at baseline and at the end of treatment:* 1. Carpal tunnel symptoms (loss of strength, tingling, numbness, burning or pain to the affected area): the number of these symptoms, as measured by one of two physicians, was recorded.* 2. Tinel's test (rated as positive or negative)* 3. Phalen's Test: (rated as positive or negative)* 4. Nerve conduction velocity of the median nerve* 5. Median peak latency (measured as the latency with which the electrical impulse is transmitted at the median nerve)* 6. Self‐perceived grip strength using the Perceived Grip Strength Scale, where the participants determine their perceived grip strength after clenching both fists for 5 s, by marking a 10‐point scale, ranging from weakest (score of 0) to strongest (score of 10) grip (measured before and after the treatment sessions on the first and last day of the 4‐week treatment period)* 7. Pain using the VITAS (1993), which is a pre‐post session pain assessment using a Visual Analogue Scale ranging from 0 (No Pain) to 10 (Worst Possible Pain), anchored with 5 faces (measured before and after the treatment sessions on the first and last day of the 4‐week treatment period)* 8. State anxiety: measured using the state anxiety inventory (STAI) which consists of 20 items on how the participant feels at that moment in terms of severity from (1) "not at all" to (4) "very much so" (measured before and after the treatment sessions on the first and last day of the 4‐week treatment period)* 9. Mood state using the Profile of Mood States (POMS), which is a 5‐point Likert rating scale on how well an adjective describes the participant’s feelings including helpless or gloomy feelings, depression and anxiety (measured before and after the treatment sessions on the first and last day of the 4‐week treatment period)* |
|
| Notes | *Only mean values (no measures of variability e.g., SD) were reported, and the number of participants in each group was not specified. Requests to obtain this data from the authors were unsuccessful. Thus, data could not be entered into a meta‐analysis. The trialists reported that "The participants had unilateral symptoms." Therefore, a unit of analysis error resulting from the correlation between two wrists in bilateral CTS participants could not have occurred. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "They were then randomly assigned to standard treatment control and massage therapy groups." Comment: Not enough information to determine the adequacy of the randomisation sequence generation. |
| Allocation concealment (selection bias) | Unclear risk | Quote: "They were then randomly assigned to standard treatment control and massage therapy groups." Comment: Not enough information to determine whether the treatment allocation was adequately concealed until interventions were assigned. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: No information regarding blinding of participants was provided, but due to the nature of the intervention, participants are unlikely to have been blinded to treatment allocation. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: Not enough information to determine whether outcome assessors were blinded to treatment allocation |
| Incomplete outcome data (attrition bias) 3 months or less | Unclear risk | Comment: The numbers of withdrawals, drop‐outs or losses to follow‐up were not reported by authors, and this doesn't necessarily mean that there were none. Authors also fail to note whether each outcome was based on the results of all randomised participants. |
| Selective reporting (reporting bias) | High risk | Comment: While the means of all outcome (pre‐specified in the Methods section of the study) were reported in their pre‐specified way, no SDs were reported. Also, authors did not report the number of participants randomised to each group, nor indicated whether the data reported were complete. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Garfinkel 1998.
| Methods | Randomised, single‐blind, controlled trial Blinded outcome assessors Randomisation occurred at the level of participants, not wrists (i.e. participants with bilateral CTS received the same intervention for both wrists) |
|
| Participants | Total n = 51 participants randomised
Intervention group n = 26 participants randomised; 22 participants (35 wrists) completed
Control group n = 25 participants randomised; 20 participants (32 wrists) completed 13 males; 28 females* Mean age: (SD not reported) Intervention 49 yrs Control 49 yrs Inclusion criteria: 1. Presence of 2 or more of the following: positive Tinel's; positive Phalen's's; pain in median nerve distribution; sleep disturbance due to hand; numbness/paraesthesias in median nerve distribution 2. Abnormal electrophysiological findings 3. Subject agrees not to change medications, receive other new treatments or change work duties during trial Exclusion criteria: 1. Previous surgery for CTS 2. Rheumatoid arthritis or other recognised inflammatory arthritis 3. CTS related to systemic disease (hypothyroidism) 4. Pregnancy |
|
| Interventions | Intervention: Yoga for 1‐1.5 hours twice weekly for 8 weeks. The Iyengar approach to hatha yoga, which emphasises proper structural alignment of the body, was delivered (Iyengar 1966). Eleven different exercises, each held for 30 seconds, and designed to take each joint in the upper body through its full range of motion with strengthening, stretching, and balancing each part. Control: Wrist splint to supplement current treatment for 8 weeks |
|
| Outcomes | Outcome assessed at 8 weeks 1. Pain severity using visual analogue scale (0‐10, with 10 denoting greatest level of pain) 2. Nocturnal wakening using ordinal scale (rated as worsened, same, improved) 3. Phalen's test (rated as worsened, same, improved) 4. Tinel's test (rated as worsened, same, improved) 5. Grip strength in mmHg using sphygmomanometer cuff (mean of 3 trials) 6. Nerve conduction: median nerve motor and sensory distal latencies (in ms) 7. Patterns of paraesthesia and numbness (recorded on hand diagram)** |
|
| Notes | *11 missing subject for demographic data **No data reported for this outcome Comment: Analysis was undertaken at the wrist‐level for all outcomes, though some participants in each group had bilateral CTS. Bilateral cases had the same intervention applied to each wrist. The trialists did not report how the correlation between both wrists was accounted for in the analysis, and attempts to clarify this information from the trialists were unsuccessful. Therefore, it is not clear whether a unit of analysis error occurred. No attempt was made to adjust outcome data. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "Subjects were randomised into 2 groups by having them select sealed envelopes containing a group assignment". Comment: No information regarding how the randomisation sequence was generated was reported |
| Allocation concealment (selection bias) | Unclear risk | Quote: "Subjects were randomised into 2 groups by having them select sealed envelopes containing a group assignment". Comment: Sealed envelopes were used however they may not have been distributed according to a randomised sequence and it is unclear whether opaque envelopes were used. It is unclear whether participants or trial personnel could predict assignments. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: Participants were aware of group assignments therefore self‐reported outcomes such as pain, nocturnal wakening, and patterns of paraesthesia and numbness may be biased. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "The assessments all were conducted by 1 physician who was blinded to the patient's group assignment and the intervention". Comment: Participants were aware of group assignments which may have influenced their performance when outcomes such as grip strength, Phalen's's sign, and Tinel's test were measured however, nerve conduction studies were less likely to be compromised. |
| Incomplete outcome data (attrition bias) 3 months or less | High risk | Quote: "9 dropped out or were excluded". Comment: Four participants from the treatment group and five from the control group were not included in the analysis. No reasons were provided to explain these drop‐outs or exclusions |
| Selective reporting (reporting bias) | High risk | Comment: Patterns of paraesthesia and numbness were recorded on hand diagrams but no results of these measurements were reported. Results were reported for all other measurements. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Heebner 2008.
| Methods | Randomised controlled trial No blinding reported |
|
| Participants | Total n = 60 participants (60 wrists) randomised Intervention group n = 32 participants (32 wrists) randomised; 25 participants (25 wrists) completed one month assessment; 14 participants (14 wrists) completed six month assessment Control group n = 28 participants (28 wrists) randomised; 20 participants (20 wrists) completed one month assessment; 15 participants (15 wrists) completed six month assessment 9 males, 51 females Mean age (range) of total randomised sample = 52 (32‐75) years Duration of symptoms: 1‐6 months: n = 6 6‐12 months: n = 4 1‐2 years: n = 10 > 2 years: n = 23 Inclusion criteria: 1. Need to have been diagnosed with CTS by a physician who completed a nerve conduction velocity test (with parameters of diagnosis determined by the physician) or received a preliminary diagnosis of CTS by the physician and scheduled for nerve conduction velocity testing 2. Have had symptoms of CTS for at least one month 3. Reported paraesthesia involving at least two digits of the median nerve distribution Exclusion criteria: 1. Younger than 18 years 2. Currently pregnant 3. Had previous carpal tunnel surgery on the same extremity 4. Had cognitive deficits 5. Primary language is not English or Spanish 6. Demonstrated physical impairments that prevented them from completing the exercises 7. Demonstrated symptoms of CTS severe enough to be noted by observation such as thenar muscle atrophy and/or ulcerations 8. Exhibited concurrent comorbidities involving peripheral neuropathies such as diabetes mellitus 9. Received concurrent treatment for other medical conditions involving the head, neck, or upper extremities, which would interfere with the subjective response to CTS treatment |
|
| Interventions | Intervention group: Neurodynamic mobilisation exercises (with a median nerve bias) performed three to five times daily, with 10 repetitions, plus standard care, consisting of patient education, splinting, and tendon gliding exercises. Duration of treatment was six months. Patient education consisted of discussion on the definition of CTS and the involved anatomy, causes, and risk factors of CTS, and healthy lifestyle choices (i.e., posture correction exercises, changing work ergonomics to reduce repetitive or sustained strain, activity modification to limit repetitive movement or prolonged wrist flexion and extension, and the importance of decreasing salt intake and not smoking). Splinting involved a prefabricated volar wrist splint worn at night while sleeping or if performing a heavy activity of daily living that required wrist support. Tendon gliding exercises consisted wrist extension with hook grasp to wrist flexion with fingers relaxed, tendon gliding of wrist and fingers to stretch the wrist flexors, full fist active range of motion, hook grasp active range of motion, half fist exercise, full fist exercise, and tendon‐gliding exercises that isolated flexion to the proximal, middle, and distal interphalangeal joints). Each tendon gliding exercise was required to be done 10 times, three to five times a day. Control group: Standard care only, consisting of patient education, splinting, and tendon gliding exercises (see above) for six months |
|
| Outcomes | Outcomes assessed after one and six months of treatment (no post‐treatment cessation assessment done)*: 1. Symptoms using the Levine carpal tunnel questionnaire (rates 11 items regarding pain, nocturnal symptoms, numbness, tingling, and weakness in reference to a typical 24‐hour period during the past two weeks, on an ordinal scale from 1: mildest, to 5: most severe; the lowest score for the scale is 1, and the highest score 5, which indicates a high degree of symptom severity). 2. Function using the Leving carpal tunnel questionnaire (rates 8 items regarding the difficulty in performing functional activities on a typical day in the past two weeks due to hand and wrist symptoms, on an ordinal scale from 1: no difficulty, to 5: cannot do at all due to hand or wrist symptoms; the lowest score for the scale is 1, and the highest score 5, which indicates a significant loss of functional ability). 3. Function using the Disabilities of the Arm, Shoulder, and Hand (DASH) questionnaire, which yields a global score ranging from 0 to 100, with higher scores reflecting increased disability 4. Neurodynamic irritability of median nerve (R1), measured in degrees (°) using the upper limb tension test for the median nerve |
|
| Notes | *No numerical summary outcome data reported in the publication, but when requested, authors provided means and SDs per group measured after one month of treatment only. Of the 60 participants randomised, 26 had bilateral involvement, however only one wrist from each bilateral participant was treated in the study. Therefore, a unit of analysis error resulting from the correlation between two wrists in bilateral CTS participants could not have occurred. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "The participants were randomly assigned to one of two groups by means of a coin toss" Comment: The randomisation sequence was probably adequately generated. |
| Allocation concealment (selection bias) | High risk | Quote: "The researcher tossed a coin right after attaining informed written consent and both patient and therapist (also researcher) were not blinded to the group assignment. So, there was no predetermined sequence for the allocation. All happened on the spot." (personal communication with trialist) Comment: It is unlikely that the allocation sequence was adequately concealed. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: No blinding of participants was reported, and given the nature of the interventions delivered, it is unlikely that participants and personnel were blinded to treatment received. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Comment: No blinding of outcome assessors was reported, and given that objective measures of function were assessed at the same time in which compliance with the exercises and splinting undertaken at home was measured, it is unlikely that outcome assessors were blinded to treatment. |
| Incomplete outcome data (attrition bias) 3 months or less | High risk | Quote: "Initially, 60 participants completed baseline data and received their intervention. Fifteen subjects failed to return for their first follow‐up appointment, eight in Group 1, and seven in Group 2. In Group 1, seven out of eight drop‐outs were due to nonparticipation and one began therapy for a concurrent upper extremity problem. In Group 2, five were due to nonparticipation, one was unable to perform the exercises due to severity of condition, and one subject had undergone carpal tunnel surgery. With attrition, only 20 participants remained in Group 1, and 25 in Group 2 for the one‐month follow‐up. At the six‐month follow‐up, an additional 16 subjects were removed from the study. The reasons for attrition at six months included eight lost due to nonparticipation, four had received CTS surgery, and four subjects had started receiving therapy for CTS or concurrent treatment of the neck and/or upper extremities. There were no differences between groups at the six‐month follow‐up due to attrition. Therefore, 15 subjects in Group 1 and 14 in Group 2 completed all three data‐collection sessions." Comment: Attition in this study was high (approximately 50%), and the reasons for attrition after six months of treatment were not clearly reported per treatment group. Given that compliance with treatment was reported to be statistically significantly higher in the intervention group compared to the control group, it is likely that non‐participation was related to the treatments delivered, which is likely to have influenced the outcome data. |
| Selective reporting (reporting bias) | High risk | Comment: No outcome data suitable for inclusion in a meta‐analysis were reported in the publication. Only box plots with undefined error bars, and exact P values based on multivariate analyses of covariance (MANCOVAs) were reported. Communication with the trial authors resulted in us obtained data after one month of treatment, but no data assessed after six months of treatment were able to be provided. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Horng 2011.
| Methods | Randomised single‐blind controlled trial. Randomisation occurred at the level of participants, not wrists (i.e. participants with bilateral CTS received the same intervention for both wrists) |
|
| Participants | Total n = 60 participants randomised; 53 (89 wrists) participants completed Intervention group 1 n = 20 participants randomised; 18 participants (31 wrists) completed Intervention group 2 n = 20 participants randomised; 19 participants (34 wrists) completed Intervention group 3 n = 20 participants randomised; 16 participants (24 wrists) completed 3 males, 57 females* Mean ± SD age*: Intervention group 1 = 48.9 ± 8.9 yrs Intervention group 2 = 51.9 ± 9.3 yrs Intervention group 3 = 52.6 ± 9.1 yrs Inclusion criteria: 1. Had subjective symptoms (such as pain and/or numbness within the median nerve distribution of the digits and nocturnal pain) 2. Had either a positive Phalen's test or a positive Tinel's test 3. Had electrophysiologic evidence of CTS Exclusion criteria: 1. Aged younger than 18 yrs 2. Had an underlying medical disorder, such as diabetes mellitus, renal failure, autoimmune disease, or hypothyroidism 3. Were pregnant or had previous wrist trauma or surgery |
|
| Interventions | Intervention group 1: Tendon gliding exercises (sliding the flexor tendons of the hand by moving the fingers through the following five discrete positions: straight, hook, fist, table top, and straight fist positions) (Totten 1991) performed five times per session three times a day for eight weeks plus neutral volar wrist splint worn at night only for eight weeks plus paraffin therapy (superficial heat delivered to the hands) administered twice per week for eight weeks Intervention group 2: Nerve gliding exercises (targeting the median nerve by moving the fingers and wrist through six different positions: grasp, finger extension, wrist extension, thumb extension, forearm supination,and gentle stretch of the thumb by the opposite hand) (Totten 1991) performed five times per session three times a day for eight weeks plus neutral volar wrist splint worn at night only for eight weeks plus paraffin therapy (superficial heat delivered to the hands) administered twice per week for eight weeks Intervention group 3: Neutral volar wrist splint worn at night only for eight weeks plus paraffin therapy (superficial heat delivered to the hands) administered twice per week for eight weeks |
|
| Outcomes | Outcomes assessed at baseline and two months after the eight‐week treatment period ended: 1. Pain using a visual analogue scale (scale properties not reported but based on outcome data reported, can assume the scale ranged from 0‐100) 2. Symptoms using Boston carpal tunnel questionnaire (rates 11 items on ordinal scale 1: mild pain, to 5: most severe pain) 3. Hand function using Boston carpal tunnel questionnaire (rates 8 items on ordinal scale 1: no difficulty with the activity, to 5: cannot perform the activity at all) 4. Disability measured using the 30‐item Disability of the Arm, Shoulder and Hand (DASH) Questionnaire, scored as a percentage, where 0% indicates the absence of any problems 5. Quality of life and health status using the 28‐item Taiwanese version of the WHOQOL‐BREF questionnaire, which includes two items on general quality‐of‐life and health status and 26 items that are grouped into four domains: physical, psychologic, social, and environmental The scores of the two general items range from 1 to 5, with a higher score indicating a better condition. The domain scores range from 4 to 20 and are derived by multiplying the average scores of all items in a given domain by four, with a higher score indicating a better quality‐of‐life within the corresponding domain 6. Phalen's test (recorded as positive if the patient reproduced his/her symptoms in the median nerve distribution)** 7. Tinel's test (recorded as positive if the participant experienced paraesthesia or shooting pain in at least one of three radial digits)** 8. Hand grip strength measured three times at each evaluation with a handheld dynamometer, with the mean of the three trials recorded** 9. Palmar pinch strength measured three times at each evaluation with a standard dynamometer between the tips of the thumb and index finger, with the mean of the three trials recorded** 10. Sensitivity measured using the Semmes‐Weinstein monofilament test, with monofilaments applied to each digit in the hand, and a positive response recorded when the subject was able to verbally localize (with his/her eyes closed) which digit was receiving pressure** 11. Nerve conduction studies: median and ulnar nerve distal motor latency (ms); median and ulnar nerve distal sensory latency (ms)** |
|
| Notes | *Demographic data reported only for participants completing the study **No numerical summary outcome data suitable for inclusion in a meta‐analysis were reported for these outcomes. Contact details of the authors were unable to be obtained, so authors could not be contacted for unreported data. The trialists reported that "The following data analyses were performed: (1) descriptive statistics to summarize the participants’ basic demographic and ergonomic data; (2) comparison of the baseline data among the three groups using the Kruskal‐Wallis test and Fisher’s exact test; (3) comparison of the baseline and follow‐up scores of the symptom severity scale, functional status scale, pain intensity, DASH questionnaire, and the four domains of the WHOQOL‐BREF using paired t tests for each patient; (4) comparison of the baseline and follow‐up physical examination and NCS data for each hand involved using a mixed‐effect model, while taking into account that the tests and/or examinations were performed on both hands for some patients; that is, the treatment was treated as a fixed effect factor and the hand (left or right) was treated as a random effect factor". Therefore, analysis was undertaken at the participant‐level for the outcomes, symptom severity scale, functional status scale, pain intensity, DASH questionnaire, and the four domains of the WHOQOL‐BREF, though 72% of participants in group 1 had bilateral CTS, 79% of participants in group 2 had bilateral CTS, and 75% of participants in group 3 has bilateral CTS. As bilateral cases had the same intervention applied to both wrists, and the trialists did not report controlling for the correlation between both wrists, a unit of analysis error possibly occurred for these outcomes (but not for the nerve conduction studies outcome data). No attempt was made to adjust outcome data. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "All consecutive patients were invited, and the participants were assigned to three groups in the order of group 1, 2, and 3 by a nurse who was not involved in the study design and blind to the pre‐assigned treatment" Comment: It is not clear whether an adequate randomisation sequence was pre‐generated or whether alternation was used to allocate participants. |
| Allocation concealment (selection bias) | Unclear risk | Quote: "All consecutive patients were invited, and the participants were assigned to three groups in the order of group 1, 2, and 3 by a nurse who was not involved in the study design and blind to the pre‐assigned treatment. The allocations were concealed with the use of packages of prescription orders, which were given by the nurse to the physical therapists, who did not know the sequences of randomization." Comment: It is not clear whether the allocation sequence was adequately concealed. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: The study was described as a single‐blind study, and the authors specified that only outcome assessors were blinded. Further, given the nature of the nature of the interventions delivered, it is unlikely that participants and physical therapists administering the interventions were unaware of which interventions were being received. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "The exercise programs were administered by physical therapists, who did not participate in evaluating the outcome of the study". Quote: "The outcomes of the physical examinations and NCS were evaluated by physiatrists who were not aware of the group assignments." Comment: Outcome assessors of objective outcomes were probably blinded to treatment allocation. |
| Incomplete outcome data (attrition bias) 3 months or less | Unclear risk | Comment: The number of losses to follow‐up and drop‐outs, and reasons for these, were reported for each group. These were not completely balanced across groups, with two losses in Group 1, one loss in Group 2, and four losses in Group 3, the only group which did not receive an exercise intervention. It is not clear whether drop‐outs or losses to follow‐up were related to the interventions delivered. |
| Selective reporting (reporting bias) | High risk | Comment: Numerical summary outcome data per group (i.e. means and SDs) were only reported for the outcomes, pain measured using a visual analogue scale, symptoms measured using the CTS questionnaire, function measured using the CTS questionnaire, disability measured using the DASH questionnaire, and quality of life measured using the Taiwanese version of the WHOQOL‐BREF questionnaire. The authors only reported that analysis of the objectively measured outcomes (Phalen's test, Tinel's test, grip strength, pinch strength, sensitivity and nerve conduction studies) revealed no statistically significant differences between groups. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Janssen 2009.
| Methods | Randomised double‐blind controlled trial Blinded participants and outcome assessors |
|
| Participants | Total n = 58 participants (58 wrists) randomised Intervention group 1 n = 18 participants (18 wrists) randomised and completed pre‐surgery evaluation; Intervention group 2 n = 22 participants (22 wrists) randomised and completed pre‐surgery evaluation; Intervention group 3 n = 18 participants (18 wrists) randomised and completed pre‐surgery evaluation 22 males, 36 females Mean age (range) of total randomised sample = 51.5 (31‐82) years Inclusion criteria: 1. Diagnosed with CTS based on clinical examination and electromyographic testing 2. Aged over 21 years Exclusion criteria: 1. Had concurrent hand conditions 2. Had systemic or neurological conditions 3. Had revisions of previous carpal tunnel release |
|
| Interventions | Intervention group 1: Contrast baths with exercise ‐ the involved hand is placed in hot water up to the proximal wrist crease and the participants immediately begins by doing 10 gentle/pain‐free, deliberate composite fists; one every 6 sec. After ten repetitions are completed, the hand is removed from the hot water and placed immediately in the cold water, where the participant replicates the ten repetitions and positioning as described for the hot water. Once the ten repetitions are completed in the cold water, the hand is returned to the hot water and the same exercises and positioning are continued. The participant continues this process of shifting the hand back and forth between the hot and cold baths for a total of 11 min. Intervention group 2: Contrast baths without exercise ‐ the involved hand is placed in hot water up to the proximal wrist crease for 1 min. After 1 min the hand is then removed from the hot water and transferred to the cold water duplicating the same position used in the hot water. The participant continues this process of shifting the hand back and forth between the hot and cold baths for a total of 11 min. Intervention group 3: Exercises only ‐ the participant begins by doing 10 gentle/pain‐free, deliberate composite fists; one every 6 sec. The participant then pauses for 4 sec and then starts the composite flexion/extension as before one every 6 sec. The participant pauses for 4 sec after each ten repetitions, and continues this process for a total 11 min. |
|
| Outcomes | Outcome assessed immediately after treatment before open carpal tunnel release and after treatment delivered 10‐14 days post‐carpal tunnel release surgery* 1. Hand volume measured by the water displacement technique using a standard hand volumeter and recording the amount of water displaced in millilitres** |
|
| Notes | *Participants received the interventions pre‐ and post‐open carpal tunnel surgery; the data included in this review only pertain to the pre‐surgery evaluation. **This was not a pre‐specified outcome for the review, so not data on this outcome were included in the review. Only one affected hand per participant was evaluated in this study, so a unit of analysis error resulting from the correlation between two wrists in bilateral CTS participants could not have occurred. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "The subjects were assigned to the different treatment groups by random assignment with replacement—each subject picked one of three different colored cubes from a cloth bag. The cubes were then returned to the bag so that subsequent participants had an equal chance of picking from all three colored cubes." Comment: The random allocation sequence was probably adequately generated. |
| Allocation concealment (selection bias) | Unclear risk | Quote: "The subjects were assigned to the different treatment groups by random assignment with replacement—each subject picked one of three different colored cubes from a cloth bag. The cubes were then returned to the bag so that subsequent participants had an equal chance of picking from all three colored cubes." Comment: There is not enough information to determine whether the allocation sequence was adequately concealed |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "The subjects were blinded to group assignment and knew only that they were receiving a treatment." Comment: Participants was probably blinded to treatment allocation |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "All evaluations were conducted by two certified hand therapists (RGJ, DAS). The evaluating therapist was blinded to the specific treatment group for each subject." Comment: Outcome assessors were probably blinded to treatment allocation. |
| Incomplete outcome data (attrition bias) 3 months or less | Low risk | Quote: "The presurgery subjects were divided as follows—Treatment Group 1: Contrast Baths with Exercise had 18 participants, Treatment Group 2: Contrast Baths without Exercise had 22 participants, and Treatment Group 3: Exercise Alone had 18 participants. Eight subjects studied preoperatively were unavailable for postoperative data collection and
dropped out of the study. Reasons cited by subjects for dropping out of the study included the amount of time it took for the evaluation and treatment, and/or rescheduling of follow‐up surgeon visits without rescheduling of therapy follow‐up visits." Comment: Only the pre‐surgery outcome data are applicable to this review, and there were no losses to follow‐up at the pre‐surgery evaluation which occurred immediately after treatment was delivered. |
| Selective reporting (reporting bias) | Low risk | Comment: The sole outcome in this study was fully reported in the Results section of the publication |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Moraska 2008.
| Methods | Randomised, single‐blind controlled trial Blinded participants |
|
| Participants | Total n = 28 participants randomised Intervention group n* = 13 participants (13 wrists) completed Control group n* = 14 participants (14 wrists) completed 5 males, 22 females* Mean ± SD (range) age:* Total sample: median = 48.6 yrs (range 23‐78 yrs) Intervention group: 50.3 ± 15.1 yrs Control group: 47.0 ± 8.80 yrs Mean ± SD (range) duration of CTS symptoms:* Total sample: 5.4 yrs (range 1‐30 yrs) Intervention group: 5.6 ± 7.3 yrs Control group: 3.5 ± 3.1 yrs Inclusion criteria: 1. Written documentation of a clinical diagnosis of CTS (ICD code 354.0) by a medical practitioner 2. Positive CTS diagnosis in one or both upper extremities for at least 6 months prior to enrolment into the study. Exclusion criteria: 1. Prior surgery for CTS 2. Pregnancy 3. Smoking 4. Diabetes mellitus |
|
| Interventions | Intervention: CTS‐targeted massage protocol which was designed to (1) address any increased volume of fluid in the carpal tunnel region, (2) reduce connective tissue restriction (i.e., thickening or adhesions), and (3) decrease contractile tissue hypertonicity at potential points of nerve entrapment along the course of the brachial plexus and median nerve pathway. Each 30‐minute session consisted of: (1) 3 minutes of lymphatic drainage of the thorax and axillary regions; (2) 8 minutes of mobilization, soft‐tissue manipulation, myofascial release, traction, and friction directed at the neck and shoulder region; (3) 9 minutes of myofascial release, soft‐tissue manipulation, and mobilization directed at the chest and upper arm; and (4) 10 minutes of pin and stretch, cross‐fibre friction, stretching, and flushing techniques were performed on the forearm and hand. Sessions were delivered twice a week for six weeks. Control: General massage protocol which was designed to model a typical relaxing massage session and focused on reducing muscular tension and enhancing circulation to the back, neck, and both upper extremities. Each 30‐minute treatment was applied bilaterally and consisted of: (1) 15 minutes of effleurage, petrissage, friction, passive motion, and cross‐fibre friction all directed at the back and neck with the participant in a prone position and (2) 15 minutes of effleurage, traction, petrissage, linear friction, jostling, and stretching performed on both arms, forearms, and hands with the subject in a supine position. Sessions were delivered twice a week for six weeks. |
|
| Outcomes | Outcomes assessed at two baseline sessions, at two days after the 7th and 11th massage sessions, and at four weeks after the last massage session: 1. Maximal isometric grip strength (kg) using a Baseline® hand dynamometer (Fabrication Enterprises, Inc., White Plains, NY). The mean of three attempts was recorded.** 2. Maximal isometric pinch strength (kg) using a Baseline pinch gauge (Fabrication Enterprises, Inc.). The mean of three attempts was recorded.** 3. Symptoms using carpal tunnel questionnaire (rates 11 items on ordinal scale 1: absence of difficulty in the given task, to 5: most severely debilitating pain.** 4. Hand function using carpal tunnel questionnaire (rates 8 items on ordinal scale 1: no difficulty, to 5: inability to the task owing to hand or wrist symptoms).** 5. Function using the Grooved Pegboard test (Lafayette Instruments, Lafayette, IN). Scores were achieved by adding the time to perform the test (in seconds) plus the number of pegs drops plus the number of pegs placed in the pegboard. |
|
| Notes | *Data only reported for participants completing treatment (n = 27) **Numerical summary data (means and standard errors) were provided by the lead author of the publication on request, as data were only presented in Figure format in the publication. Standard error values were converted into SDs using the formula provided in the Cochrane Handbook of Systematic Reviews of Interventions (2008) section 7.7.3.2 "Obtaining standard deviations from standard errors and confidence intervals for group means". Only participants with unilateral CTS were included in the study, so a unit of analysis error resulting from the correlation between two wrists in bilateral CTS participants could not have occurred. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "Participants were randomly assigned to either the GM or TM massage treatment groups." (in publication) Quote: "It consisted of us drawing one of two cards from a folder just prior to the subject visit. Subjects were only told that they would receive massage and were unaware that there were two treatment groups." (personal communication with lead author of publication) Comment: Not enough information to determine the adequacy of the sequence generation. |
| Allocation concealment (selection bias) | High risk | Quote: "Participants were randomly assigned to either the GM or TM massage treatment groups." (in publication) Quote: "It consisted of us drawing one of two cards from a folder just prior to the subject visit. Subjects were only told that they would receive massage and were unaware that there were two treatment groups." (personal communication with lead author of publication) Comment: Allocation sequence was unlikely to be adequately concealed until interventions were assigned. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "Participants were unaware of alternative massage treatment groups; thus, single‐blind conditions were maintained." Comment: Participants were probably blind to treatment allocation. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "Evaluators were not blind to subject group assignment." Comment: Outcome assessors were probably aware of which intervention each participant was allocated to. |
| Incomplete outcome data (attrition bias) 3 months or less | Low risk | Quote: "A total of 28 persons with CTS were enrolled and 27 completed the 14‐week study; 1 subject was removed from the study owing to an illness unrelated to CTS or massage." Comment: While the authors did not indicate which group this participant was allocated to, it is unlikely that this single withdrawal would have significantly biased the data obtained. |
| Selective reporting (reporting bias) | Low risk | Comment: The mean ± 95% CI at each pre‐specified time point was reported in Figures for all outcomes except for hand function (measured using the Grooved Pegboard Test), which was reported selectively (no SDs reported at baseline, and unclear time points reported). However, when contacted, the trial author provided data for all outcomes at all time points in numerical format (means and standard errors). |
| Other bias | Low risk | Quote: "No significant difference was detected between baseline 1 and 2 for either group on measures of strength or function. However, the imbalance in gender among groups resulted in the TM group exhibiting greater grip and pinch strength upon entry into the study; this difference has been accounted for in the analysis by using values from baseline 2 as the covariate." Comment: This was probably an appropriate statistical analysis to perform, given the baseline imbalance. |
Pinar 2005.
| Methods | Quasi‐randomised controlled trial Blinded outcome assessors Randomisation occurred at the level of participants, not wrists (i.e. participants with bilateral CTS received the same intervention for both wrists) |
|
| Participants | Total n = 26 (35 wrists) randomised Intervention group n = 14 participants (19 wrists) randomised and completed Control group n = 12 participants (16 wrists) randomised and completed 0 males, 26 females Age and duration of CTS symptoms not reported Inclusion criteria: 1. Female 2. Aged between 35 and 55 years 3. Not pregnant 4. No history of trauma 5. No previous physiotherapy treatment Exclusion criteria: Not reported, but implied above |
|
| Interventions | Intervention: Nerve gliding exercises, static volar wrist splint and training to modify functional activities in accordance with conservative treatment were administered. A thermoplastic static volar wrist splint was applied, with the wrist in the neutral position (0 degrees). The wrist splint was used both day and night for a period of 6 weeks and was removed only for hygienic purposes. It was used at night from the sixth week until the tenth week. After the splint was removed, patients were instructed that their hands could be used during the day in accordance with the patient training program to modify functional activities. During training to modify their functional activities, patients were instructed to avoid repetitive hand activities, to hold the wrist in a neutral position while using force, to refrain from gripping strongly with the first and second fingers, to rest between activities, and to decrease speed and strength in all hand activities. Nerve gliding exercises were performed actively with 10 repetitions and were continued from the first day of splint application to the tenth week. Participants were instructed to continue with the exercise program at home 5 times per day. Nerve gliding exercises included: Position 1: Exercises were begun with the wrist in a neutral position (0 degrees) and the fingers and thumb in the full flexion position. The distal median nerve was placed in a relatively relaxed position. Position 2: With the wrist kept in the neutral position, the fingers were brought to extension with the thumb in a neutral position. Tension in the distal segment of the nerves in the digits was increased. Position 3: With maintenance of finger extension and the neutral position of the thumb, wrist extension was added to the exercises. The area of greatest excursion was accessed as the wrist was extended. Position 4: While keeping the wrist and fingers extended, the thumb was extended. The median nerve branch to the thumb was included in this exercise. Position 5: With the wrist, fingers, and thumb kept in extension, the forearm was brought into supination. This added tension to the more proximal portion of the median nerve in the forearm. Position 6: With extension of the wrists, fingers and thumb and supination of the forearm, slight tension was applied to the thumb with the other hand (Totten 1991). Control: Static volar wrist splint and training to modify functional activities in accordance with conservative treatment were administered exactly the same way as in the intervention group. |
|
| Outcomes | Outcomes assessed before and at the end of the 10‐week treatment period: 1. Tinel's test (rated as positive or negative) 2. Phalen's test (rated as positive or negative) 3. Pain measured as the degree of pain felt over the whole day using a 10 cm visual analogue scale graded from 0 (no pain) to 10 (severe pain) 4. Motor function of the abductor pollicis brevis muscle using a manual muscle test* 5. Grip strength measured (in kg) using a Jamar hand dynamometer. Three measurements were taken and the mean of these was calculated 6. Pinch strength: measured (in kg) using a Jamar pinch meter. Three measurements were taken and the mean of these was calculated. 7. Light‐touch deep‐pressure sense using a Semmes‐Weinstein pressure aesthesiometer. 8. Functional sensitivity using a 2‐point discrimination test. Results were evaluated according to the description published by the American Society of Hand Therapists.* 9. Median and ulnar nerve distal sensory latency categorised as positive (prolonged distal sensory latency) or negative (normal sensory latency). |
|
| Notes | *These outcomes were pre‐specified in the methods section of the publication as having been evaluated, but no data were not reported in the publication; however, the lead author of this trial provided this data on request. Analysis was undertaken at the wrist‐level for all outcomes, though some participants in each group had bilateral CTS. Communication with the trialists confirmed that bilateral cases had the same intervention applied to both wrists. However, the trialists did not report controlling for the correlation between both wrists, so a unit of analysis error is likely to have occurred for these outcomes. No attempt was made to adjust outcome data. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | Quote: "Patients were allocated to the therapy groups according to their sequential application to the therapy clinic. For example the first one was allocated to the experiment group and the second one was allocated to the control group." (personal communication) Comment: Alternation used, which is not an adequate randomisation sequence. |
| Allocation concealment (selection bias) | High risk | Quote: "Patients were allocated to the therapy groups according to their sequential application to the therapy clinic. For example the first one was allocated to the experiment group and the second one was allocated to the control group." (personal communication) Comment: Allocation sequence was probably not adequately concealed until interventions were assigned. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "The patients were informed about the details of their therapy program but they were not mentioned that there was a second therapy method in this project" (personal communication). Comment: Blinding of patients was probably achieved. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "Some of the outcome assessors like the doctor making the electrophysiologic tests was blind, but the therapists were not" (personal communication). Comment: Outcome assessors were probably blind to treatment allocation |
| Incomplete outcome data (attrition bias) 3 months or less | Low risk | Quote: "The following assessment methods, including Tinel's and Phalen's tests specific for CTS, were carried out for all patients before and after the 10‐week treatment program." Comment: There were no reported withdrawals/drop‐outs so the data for all of the outcomes are probably complete. |
| Selective reporting (reporting bias) | Low risk | Comment: Motor function of the abductor pollicis brevis muscle, which was evaluated by a manual muscle test, and functional sensitivity, which was determined by a 2‐point discrimination test, were pre‐specified in the methods section of the publication, but the results for these two outcomes were not reported. However, the lead author of the trial publication provided this data on request. |
| Other bias | Low risk | Comment: No other sources of bias identified. |
Tal‐Akabi 2000.
| Methods | Randomised, single‐blind, controlled trial Blinded outcome assessors |
|
| Participants | Total n = 21 participants (21 wrists) randomised
Intervention group 1 n = 7 participants (7 wrists) randomised and completed
Intervention group 2 n = 7 participants (7 wrists) randomised and completed
Control group n = 7 participants (7 wrists) randomised and completed 7 males; 14 females Mean ± SD age: 47 ± 15 yrs Inclusion criteria: 1. Positive electrodiagnostic testing 2. Positive Phalen's and Tinel's test 3. Positive upper limb tension test (ULTT) 2a with a median nerve bias 4. Diagnosis of CTS by surgeon and candidate for decompression Exclusion criteria: 1. Psychosocial problems 2. Diabetes mellitus 3. Herpes zoster 4. Rheumatoid arthritis 5. Pregnancy 6. Hyperthyroidism 7. Known abnormality of nervous system 8. Cervical or thoracic spine origin of symptoms |
|
| Interventions | Intervention group 1: Neurodynamic mobilisation (mobilisation with a median nerve bias, including slight glenohumeral abduction, shoulder girdle depression, elbow extension, lateral rotation of the whole arm, wrist, thumb & finger extension and finally glenohumeral abduction, as described by Butler 1991) for 3 weeks Intervention group 2: Carpal bone mobilisation including posterior‐anterior mobilisation and flexor retinaculum stretch (as described by Maitland 1991) for 3 weeks For both intervention groups, the grade, amplitude and progression of treatment was individualised Control: No treatment for 3 weeks |
|
| Outcomes | Outcome assessed at 3 weeks* 1. Symptoms using a symptom diary with visual analogue scale 2. Pain relief using a short ordinal scale 0‐5 (called the modified pain relief scale); 0 = no pain relief, 5 = complete pain relief ** 3. Hand function using modified functional box scale (short ordinal scale; 0 = able to button/unbutton shirt or grip without any problem, 4 = not able to do alone)** 4. Active range of movement wrist flexion (in degrees) 5. Active range of movement wrist extension (in degrees) 6. Upper limb tension test with a median nerve bias (ULTT2a) (dichotomous score: positive or negative) 7. Need for surgical release (dichotomous score) |
|
| Notes | *Confirmed with principal author in personal communication **Short ordinal scales dichotomised for entry into RevMan 5.1. Pain recoded as 'improved' (score 1‐5) and 'no relief' (score 0); hand function recoded to 'improved' (improvement in score from baseline to week 3) and 'not improved/worsened' (no change or deterioration in score from baseline to week 3). Note, a subject in each group (neurodynamic, carpal bone and control) had normal hand function at baseline and had not changed after 3 weeks of follow‐up). These participants were not included in the totals Participants with bilateral CTS only submitted one wrist into the analysis. Therefore, a unit of analysis error resulting from the correlation between two wrists in bilateral CTS participants could not have occurred. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "After selection, the subjects were randomly allocated to one of the three groups by pulling names out of a hat." Comment: The randomisation sequence was probably adequately generated. |
| Allocation concealment (selection bias) | Unclear risk | Quote: "After selection, the subjects were randomly allocated to one of the three groups by pulling names out of a hat." Comment: Not enough information to determine whether the allocation sequence was adequately concealed until interventions were assigned. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: Not mentioned, but due to the nature of the interventions, it is likely that participants were aware of which treatment they received. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "All measurements except the PRS measurement were undertaken by an independent examiner pre treatment intervention to obtain baseline readings. All the measurement tools were then utilized post intervention by the same independent examiners." Comment: While blinding of outcome assessors was not specified in the publication, when contacted, the authors reported that "Assessors were blinded to the initial results and to type of treatment given." |
| Incomplete outcome data (attrition bias) 3 months or less | Low risk | Comment: No drop‐outs were reported and it is clear that the outcomes were based on 7 participants in each group (which is the number of participants randomised). Thus it can be assumed that the data reported are based on a complete sample. |
| Selective reporting (reporting bias) | Low risk | Comment: All of the outcomes specified in the Methods section of the publication were reported in the form of individual patient data and mean scores (no measures of variability were reported, but these were able to be calculated given the reporting of the individual patient data). |
| Other bias | Low risk | Comment: No other sources of bias identified. |
CI: confidence interval
CTS: carpal tunnel syndrome
ms: milliseconds
NDT: neurodynamic technique
SD: standard deviation
SE: standard error
STM: soft tissue mobilisation
VAS: visual analogue scale
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Abbot 1999 | Not a randomised clinical trial. This is a clinical commentary on the Garfinkel 1998 randomised controlled trial. |
| Arinci Incel 2005 | Nerve and tendon gliding exercises were delivered to both groups in this randomised controlled trial. |
| Bernaards 2006 | Protocol for a randomised controlled trial of work style and physical activity intervention vs control in computer workers. Individuals with CTS to be excluded. |
| Blankfield 2001 | Therapeutic touch (an alternative and complementary medicine intervention) was the primary treatment under investigation. |
| George 2006 | Not a randomised controlled trial. |
| Giattini 1999 | Participants underwent carpal tunnel release, which is an exclusion criterion for this review. |
| Goldberg 2004 | Not a randomised controlled trial. Summary of the Field 2004 trial. |
| Hains 2010 | Ischaemic compression therapy versus placebo ischaemic therapy were delivered, and only the placebo group crossed over to the intervention group. |
| Nathan 2001 | Not a randomised controlled trial. Non‐controlled study of an aerobic exercise program for CTS. |
| Nathan 2002 | Letter to the editor by Dr Nathan regarding the Nathan 2001 study. |
| Omer 2003/2004 | RCT assessing effectiveness of exercise program in participants with CTS or myofascial pain syndrome. Data were not reported separately for those participants with CTS, and attempts to obtain these outcome data from the authors were unsuccessful. |
| Rozmaryn 1998 | Not a prospective randomised clinical trial. Outcomes were collected retrospectively from participants' clinical case notes. |
| Ruksen 2011 | The same exercise intervention was delivered to both groups in this randomised controlled trial. |
| Taspinar 2007 | Exercise or mobilisation are not interventions examined in this randomised controlled trial. |
| Thomas 1993 | Not a randomised controlled trial. |
| Verhagen 2007 | Not a randomised controlled trial. This is an update of a Cochrane systematic review on exercise interventions for work‐related complaints of the arm, neck or shoulder. |
| Walker 2010 | Not a randomised controlled trial. |
CTS: carpal tunnel syndrome RCT: randomised controlled trial vs: versus
Characteristics of studies awaiting assessment [ordered by study ID]
Ashraf 2009.
| Methods | |
| Participants | |
| Interventions | |
| Outcomes | |
| Notes | RCT comparing local steroid injection to "physiotherapy"; no definition of physiotherapy is provided, so it is not clear if exercises or mobilisation were delivered in this study. Attempts to clarify this information with authors were unsuccessful. |
Avci 2004.
| Methods | |
| Participants | |
| Interventions | |
| Outcomes | |
| Notes | Full text publication in Turkish; requires translation. |
El Miedany 2009.
| Methods | |
| Participants | |
| Interventions | |
| Outcomes | |
| Notes | Results only presented as conference abstract; communication with author confirmed that results are being written up for full publication. |
Maltese 2006.
| Methods | |
| Participants | |
| Interventions | |
| Outcomes | |
| Notes | Full text publication in Italian; requires translation. |
Shi 2006.
| Methods | |
| Participants | |
| Interventions | |
| Outcomes | |
| Notes | Full text publication in Chinese; requires translation. |
RCT: randomised controlled trial
Differences between protocol and review
This is a split review replacing the therapeutic ultrasound interventions included in the previous review titled Non‐surgical treatment (other than steroid injection) for carpal tunnel syndrome (O'Connor 2003).
In the review by O'Connor et al. (O'Connor 2003), types of outcome measures included in the review were as follows:
Primary outcome:
The primary outcome measure was improvement in clinical symptoms, such as pain and paraesthesiae, at least three months after the end of treatment.
Secondary outcome measures included: 1. improvement in functional status and/or health‐related quality of life parameters at least three months after treatment; 2. improvement in objective physical examination measures, such as grip, pinch strength, and sensory perception at least three months after treatment; 3. improvement in neurophysiological parameters after three months after treatment; 4. clinical improvement at less than three months of follow‐up; 5. clinical improvement at one year after treatment; 6. need for surgical release of the flexor retinaculum during follow‐up.
The outcomes reported in this review have been modified from the original review (O'Connor 2003) to make them as consistent as possible with other Cochrane reviews on carpal tunnel syndrome (Marshall 2007; O'Connor 2012; Page 2012; Scholten 2007; Verdugo 2008).
Assessment for study risk of bias has been performed using The Cochrane Collaboration's 'Risk of bias' tool in this update of the review. We have included a 'Summary of findings' table.
Contributions of authors
MATTHEW PAGE (MP) was involved in the following stages of the review: design of the review (in collaboration with DOC); undertaking the search of studies; screening the search results (independently of, but in addition to DOC); organising retrieval of papers; screening retrieved papers against inclusion/exclusion criteria (independently of, but in addition to DOC); appraising the risk of bias of papers (independently of, but in addition to DOC and VP); extracting data from papers (independently of, but in addition to DOC, VP, and NMW); writing to study investigators for additional information; summarising the risk of bias of the studies (independently, but in addition to DOC and VP); compiling the summary of comparisons, tables of included, excluded, awaiting and ongoing studies; entering data into RevMan 5; performing analysis of data; interpreting the findings; writing of the review (in collaboration with DOC, VP and NMW); final approval of the version to be published
DENISE O'CONNOR (DOC) was responsible for: design of the review (in collaboration with MP); developing the search strategy; screening the search results (independently of, but in addition to MP); screening retrieved papers against inclusion/exclusion criteria (independently of, but in addition to MP); appraising the risk of bias of papers (independently of, but in addition to MP and VP); extracting data from papers (independently of, but in addition to MP, VP and NMW); writing to study investigators for additional information; summarising the risk of bias of the studies (independently of, but in addition to MP and VP); writing the review (with contribution from MP, VP and NMW).
VERONICA PITT (VP) was involved in the following stages of the review: extracting data from papers (independently of, but in addition to MP, DOC and NMW); appraising the risk of bias of papers (independently of, but in addition to MP and DOC, and SM); summarising the risk of bias of papers (independently of, but in addition to MP and DOC) contributing to the writing of the review (in collaboration with MP, DOC and NMW).
NICOLA MASSY‐WESTROPP (NMW) was involved in the following stages of the review: extracting data from papers (independently of, but in addition to MP, DOC and VP); entering data in RevMan (independently, but in addition to MP and DOC); contributing to the writing of the review (in collaboration with MP, DOC, SM, and VP).
Sources of support
Internal sources
Australasian Cochrane Centre, School of Public Health and Preventive Medicine, Monash University, Australia.
External sources
No sources of support supplied
Declarations of interest
None known.
New
References
References to studies included in this review
Akalin 2002 {published and unpublished data}
- Akalin E, El O, Peker O, Senocak O, Tamci S, Gülbahar S, et al. Treatment of carpal tunnel syndrome with nerve and tendon gliding exercises. American Journal of Physical Medicine and Rehabilitation 2002;81(2):108‐13. [PUBMED: 11807347] [DOI] [PubMed] [Google Scholar]
Bahrami 2006 {published data only}
- Bahrami MH, Rayegani SM, Baghbani M, Bafghi MRB. The role of nerve and tendon gliding exercises in the conservative treatment of carpal tunnel syndrome. Journal of Medical Council of Islamic Republic of Iran 2006;24(1):5‐12. [Google Scholar]
Bardak 2009 {published data only}
- Bardak AN, Alp M, Erhan B, Paker N, Kaya B, Onal AE. Evaluation of the clinical efficacy of conservative treatment in the management of carpal tunnel syndrome. Advances in Therapy 2009;26(1):107‐16. [PUBMED: 19165436] [DOI] [PubMed] [Google Scholar]
Baysal 2006 {published data only}
- Baysal O, Altay Z, Ozcan C, Ertem K, Yologlu S, Kayhan A. Comparison of three conservative treatment protocols in carpal tunnel syndrome. International Journal of Clinical Practice 2006;60(7):820‐8. [PUBMED: 16704676] [DOI] [PubMed] [Google Scholar]
Bialosky 2009 {published and unpublished data}
- Bialosky JE, Bishop MD, Price DD, Robinson ME, Vincent KR, George SZ. A randomized sham‐controlled trial of a neurodynamic technique in the treatment of carpal tunnel syndrome. Journal of Orthopaedic and Sports Physical Therapy 2009;39(10):709‐23. [PUBMED: 19801812] [DOI] [PMC free article] [PubMed] [Google Scholar]
Brininger 2007 {published data only}
- Brininger TL, Rogers JC, Holm MB, Baker NA, Li Z‐M, Goitz RJ. Efficacy of a fabricated customized splint and tendon and nerve gliding exercises for the treatment of carpal tunnel syndrome: a randomized controlled trial. Archives of Physical Medicine and Rehabilitation 2007;88(11):1429‐35. [PUBMED: 17964883] [DOI] [PubMed] [Google Scholar]
Burke 2007 {published data only}
- Burke J, Buchberger DJ, Carey‐Loghmani MT, Dougherty PE, Greco DS, Dishman JD. A pilot study comparing two manual therapy interventions for carpal tunnel syndrome. Journal of Manipulative and Physiological Therapeutics 2007;30(1):50‐61. [PUBMED: 17224356] [DOI] [PubMed] [Google Scholar]
Davis 1998 {published data only}
- Davis PT, Hulbert JR, Kassak KM, Meyer JJ. Comparative efficacy of conservative medical and chiropractic treatments for carpal tunnel syndrome: a randomized clinical trial. Journal of Manipulative and Physiological Therapeutics 1998;21(5):317‐26. [PUBMED: 9627862] [PubMed] [Google Scholar]
Field 2004 {published data only}
- Field T, Diego M, Cullen C, Hartshorn K, Gruskin A, Hernandez‐Reif M, et al. Carpal tunnel syndrome symptoms are lessened following massage therapy. Journal of Bodywork and Movement Therapies 2004;8(1):9‐14. [EMBASE: 2004016426] [Google Scholar]
Garfinkel 1998 {published data only}
- Garfinkel MS, Singhal A, Katz WA, Allan DA, Reshetar R, Schumacher HR Jr. Yoga‐based intervention for carpal tunnel syndrome: a randomized trial. JAMA 1998;280(18):1601‐3. [PUBMED: 9820263] [DOI] [PubMed] [Google Scholar]
Heebner 2008 {published and unpublished data}
- Heebner ML, Roddey TS. The effects of neural mobilization in addition to standard care in persons with carpal tunnel syndrome from a community hospital. Journal of Hand Therapy 2008;21(3):229‐241. [PUBMED: 18652967] [DOI] [PubMed] [Google Scholar]
Horng 2011 {published data only}
- Horng YS, Hsieh SF, Lin MC, Wang JD. A comparative study on effectiveness of tendon versus nerve gliding exercises for carpal tunnel syndrome. Pain Medicine 2010;11(2):310‐1. [EMBASE: 70212068] [Google Scholar]
- Horng YS, Hsieh SF, Tu YK, Lin MC, Horn YS, Wang JD. The comparative effectiveness of tendon and nerve gliding exercises in patients with carpal tunnel syndrome: a randomized trial. American Journal of Physical Medicine and Rehabilitation 2011;90(6):435‐42. [PUBMED: 21430512] [DOI] [PubMed] [Google Scholar]
Janssen 2009 {published data only}
- Janssen RG, Schwartz DA, Velleman PF. A randomized controlled study of contrast baths on patients with carpal tunnel syndrome. Journal of Hand Therapy 2009;22(3):200‐8. [PUBMED: 19375278] [DOI] [PubMed] [Google Scholar]
Moraska 2008 {published and unpublished data}
- Moraska A, Chandler C, Edmiston‐Schaetzel A, Franklin G, Calenda EL, Enebo B. Comparison of a targeted and general massage protocol on strength, function, and symptoms associated with carpal tunnel syndrome: a randomized pilot study. Journal of Alternative and Complementary Medicine 2008;14(3):259‐67. [PUBMED: 18370581] [DOI] [PubMed] [Google Scholar]
Pinar 2005 {published and unpublished data}
- Pinar L, Enhos A, Ada S, Güngör N. Can we use nerve gliding exercises in women with carpal tunnel syndrome?. Advances in Therapy 2005;22(5):467‐75. [PUBMED: 16418156] [DOI] [PubMed] [Google Scholar]
Tal‐Akabi 2000 {published and unpublished data}
- Tal‐Akabi A, Rushton A. An investigation to compare the effectiveness of carpal bone mobilisation and neurodynamic mobilisation as methods of treatment for carpal tunnel syndrome. Manual Therapy 2000;5(4):214‐22. [PUBMED: 11052900] [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Abbot 1999 {published data only}
- Abbot NC. Yoga‐based intervention for carpal tunnel syndrome: commentary. Focus on Alternative and Complementary Therapies 1999;4:81‐2. [Google Scholar]
Arinci Incel 2005 {published data only}
- Arinci Incel N, Sezgin M, Aksit C, Sahin G. Effect of gabapentin in carpal tunnel syndrome: a controlled clinical trial [Karpal tunnel sendromunda gabapentin tedavisinin etkinligi: karsilastirmali klinik calisma]. Journal of Rheumatology and Medical Rehabilitation 2005;16(4):224‐30. [EMBASE: 2007022406] [Google Scholar]
Bernaards 2006 {published data only}
- Bernaards CM, Ariëns GAM, Hildebrandt VH. The (cost‐)effectiveness of a lifestyle physical activity intervention in addition to a work style intervention on the recovery from neck and upper limb symptoms in computer workers. BMC Musculoskeletal Disorders 2006;7:80. [PUBMED: 17062141] [DOI] [PMC free article] [PubMed] [Google Scholar]
Blankfield 2001 {published data only}
- Blankfield RP, Sulzmann C, Fradley LG, Tapolyai AA, Zyzanski SJ. Therapeutic touch in the treatment of carpal tunnel syndrome. Journal of the American Board of Family Practice 2001;14(5):335‐42. [PUBMED: 11572538] [PubMed] [Google Scholar]
George 2006 {published data only}
- George JW, Tepe R, Busold D, Keuss S, Prather H, Skaggs CD. The effects of active release technique on carpal tunnel patients: a pilot study. Journal of Chiropractic Medicine 2006;5(4):119‐22. [DOI] [PMC free article] [PubMed] [Google Scholar]
Giattini 1999 {published data only}
- Giattini A, Logullo F, Morici D, Castellani G, Provinciali L. Physiotherapy reduced the recovery time after carpal tunnel syndrome. Neurorehabilitation and Neural Repair 1999;13(1):51. [Google Scholar]
Goldberg 2004 {published data only}
- Goldberg H. Massage decreases carpal tunnel syndrome pain. Focus on Alternative and Complementary Therapies 2004;9(2):141‐2. [Google Scholar]
Hains 2010 {published data only}
- Hains G, Descarreaux M, Lamy A‐M, Hains F. A randomized controlled (intervention) trial of ischemic compression therapy for chronic carpal tunnel syndrome. Journal of the Canadian Chiropractic Association 2010;54(3):155‐63. [PMC free article] [PubMed] [Google Scholar]
Nathan 2001 {published data only}
- Nathan PA, Wilcox A, Emerick PS, Meadows KD, McCormack AL. Effects of an aerobic exercise program on median nerve conduction and symptoms associated with carpal tunnel syndrome. Journal of Occupational and Environmental Medicine 2001;43(10):840‐3. [PUBMED: 11665452] [DOI] [PubMed] [Google Scholar]
Nathan 2002 {published data only}
- Nathan PA. Effects of an aerobic exercise program on median nerve conduction and symptoms associated with carpal tunnel syndrome. Journal of Occupational and Environmental Medicine 2002;44(4):303‐5. [PUBMED: 11977412] [DOI] [PubMed] [Google Scholar]
Omer 2003/2004 {published data only}
- Omer SR, Ozcan E, Karan A, Ketenci A. Musculoskeletal system disorders in computer users: effectiveness of training and exercise programs. Journal of Back and Musculoskeletal Rehabilitation 2003/2004;17(1):9‐13. [EMBASE: 2004195653] [Google Scholar]
Rozmaryn 1998 {published data only}
- Rozmaryn LM, Dovelle S, Rothman ER, Gorman K, Olvey KM, Bartko JJ. Nerve and tendon gliding exercises and the conservative management of carpal tunnel syndrome. Journal of Hand Therapy 1998;11(3):171‐9. [PUBMED: 9730093] [DOI] [PubMed] [Google Scholar]
Ruksen 2011 {published data only}
- Ruksen S, Oz B, Olmez N, Memis A. Comparison of clinical effectiveness of corticosteroid phonophoresis and local steroid injection treatment in carpal tunnel syndrome. Turkiye Fiziksel Tip ve Rehabilitasyon Dergisi 2011;57(3):119‐23. [Google Scholar]
Taspinar 2007 {published data only}
- Taşpinar Ş, Şahin F, Erçalik C, Kuran B, Barkut K, Çelik M, et al. Comparison of the efficacy of corticosteroid injection, night splint, and physiotherapy in diabetic carpal tunnel syndrome [Diyabetik karpal tünel sendromunda kortikosteroid enjeksiyonu, gece ateli ve fizik tedavinin etkinliğinin karşılaştırılması]. Turkish Journal of Physical Medicine and Rehabilitation 2007;53(2):54‐60. [Google Scholar]
Thomas 1993 {published data only}
- Thomas RE, Butterfield RK, Hool JN, Herrick RT. Effects of exercise on carpal tunnel syndrome symptoms. Applied Ergonomics 1993;24(2):101‐8. [EMBASE: 1993148379 ] [DOI] [PubMed] [Google Scholar]
Verhagen 2007 {published data only}
- Verhagen AP, Karels C, Bierma‐Zeinstra SMA, Feleus A, Dahaghin S, Burdorf A, et al. Exercise proves effective in a systematic review of work‐related complaints of the arm, neck, or shoulder. Journal of Clinical Epidemiology 2007;60(2):110‐7. [PUBMED: 17208116] [DOI] [PubMed] [Google Scholar]
Walker 2010 {published data only}
- Walker C. Automated manipulative device alleviates symptoms of carpal tunnel syndrome. Journal of Manual & Manipulative Therapy 2010;18(4):238. [Google Scholar]
References to studies awaiting assessment
Ashraf 2009 {published data only}
- Ashraf A, Moghtaderi AR, Yazdani AH, Mirshams S. Evaluation of effectiveness of local insulin injection in none insulin dependent diabetic patient with carpal tunnel syndrome. Electromyography and Clinical Neurophysiology 2009;49(4):161‐6. [PubMed] [Google Scholar]
Avci 2004 {published data only}
- Avci S, Günaydin R, Öztura I. Comparison of effectiveness of splint and splint with physical therapy in carpal tunnel syndrome [Karpal tünel sendromunda atel ve atel ile birlikte fizik tedavinin etkinliğinin karşılaştırılması]. Türkiye Fiziksel Tip Ve Rehabilitasyon Dergisi 2004;50(2):22‐6. [Google Scholar]
El Miedany 2009 {published data only}
- Miedany Y, Gaafary M, Ahmed I, Mehanna A, Youssef S. Massage in carpal tunnel syndrome: a rational approach. Rheumatology 2009;48(Suppl 1):i95‐i96. [Google Scholar]
Maltese 2006 {published data only}
- Maltese V, Cataldo P, Cusumano C, Sanfilippo A, Letizia G. Therapeutic outcome assessment in the carpal tunnel syndrome: Therapies' confrontation. Acta Medica Mediterranea 2006;22(1):29‐32. [Google Scholar]
Shi 2006 {published data only}
- Shi YS, Fang W, Zhao XY. Control study on effect of pricking collateral blood therapy combined with massage on mild carpal tunnel syndrome. Zhongguo Zhong xi yi jie he za zhi Zhongguo Zhongxiyi jiehe zazhi [Chinese Journal of Integrated Traditional and Western Medicine] 2006;26(6):497‐99. [PubMed] [Google Scholar]
Additional references
Ashworth 2010
- Ashworth N. Carpal tunnel syndrome. Clinical Evidence 2010;03:1114. [PMC free article] [PubMed] [Google Scholar]
Atroshi 1999
- Atroshi I, Gummesson C, Johnsson R, Ornstein E, Ranstam J, Rosén I. Prevalence of carpal tunnel syndrome in a general population. JAMA 1999;282(2):153‐8. [PUBMED: 10411196] [DOI] [PubMed] [Google Scholar]
Bland 2005
- Bland JD. The relationship of obesity, age, and carpal tunnel syndrome: more complex than was thought?. Muscle & Nerve 2005;32(4):527‐32. [DOI] [PubMed] [Google Scholar]
Braun 1989
- Braun RM, Davidson K, Doehr S. Provocative testing in the diagnosis of dynamic carpal tunnel syndrome. Journal of Hand Surgery (American Volume) 1989;14(2 Pt 1):195‐7. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
Butler 1991
- Butler DS. Mobilization of the Nervous System. First Edition. Melbourne: Churchill Livingstone, 1991. [Google Scholar]
Charles 2009
- Charles J, Fahridin S, Britt H. Carpal tunnel syndrome. Australian Family Physician 2009;38(9):665. [PubMed] [Google Scholar]
D'Arcy 2000
- D’Arcy CA, McGee S. The rational clinical examination. Does this patient have carpal tunnel syndrome?. JAMA 2000;283:3110‐7. [DOI] [PubMed] [Google Scholar]
Deeks 2011
- Deeks JJ, Higgins JPT, Altman DG. Analysing data and undertaking meta‐analyses. In: Higgins JPT, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011:Available from www.cochrane‐handbook.org. [Google Scholar]
Dwan 2008
- Dwan K, Altman DG, Arnaiz JA, Bloom J, Chan A‐W, Cronin E, et al. Systematic review of the empirical evidence of study publication bias and outcome reporting bias. PLoS ONE 2008;3(8):e3081. [DOI] [PMC free article] [PubMed] [Google Scholar]
Dwan 2011
- Dwan K, Altman DG, Cresswell L, Blundell M, Gamble CL, Williamson PR. Comparison of protocols and registry entries to published reports for randomised controlled trials. Cochrane Database of Systematic Reviews 2011, Issue 1. [DOI: 10.1002/14651858.MR000031.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Gelfman 2009
- Gelfman R, Melton LJ 3rd, Yawn BP, Wollan PC, Amadio PC, Stevens JC. Long‐term trends in carpal tunnel syndrome. Neurology 2009;72(1):33‐41. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gerritsen 2002
- Gerritsen AA, Krom MC, Struijs MA, Scholten RJ, Vet HC, Bouter LM. Conservative treatment options for carpal tunnel syndrome: a systematic review of randomised controlled trials. Journal of Neurology 2002;249(3):272‐80. [DOI] [PubMed] [Google Scholar]
Goodyear‐Smith 2004
- Goodyear‐Smith F, Arroll B. What can family physicians offer patients with carpal tunnel syndrome other than surgery? A systematic review of nonsurgical management. Annals of Family Medicine 2004;2(3):267‐73. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2002
- Higgins JPT, Thompson SG. Quantifying heterogeneity in meta‐analysis. Statistics in Medicine 2002;21(11):1539‐58. [DOI] [PubMed] [Google Scholar]
Higgins 2011a
- Higgins JPT, Green S (editors). Cochrane Handbook of Systematic Reviews of Interventions Version 5.1.0 [Updated March 2011]. The Cochrane Collaboration 2011. Available from www.cochrane‐handbook.org.
Higgins 2011b
- Higgins JPT, Altman DG, Sterne JAC. Assessing risk of bias in included studies. In: Higgins JPT, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011:Available from www.cochrane‐handbook.org. [Google Scholar]
Higgins 2011c
- Higgins JPT, Deeks JJ, Altman DG. Special topics in statistics. In: Higgins JPT, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011:Available from www.cochrane‐handbook.org. [Google Scholar]
Hopewell 2007
- Hopewell S, McDonald S, Clarke MJ, Egger M. Grey literature in meta‐analyses of randomized trials of health care interventions. Cochrane Database of Systematic Reviews 2007, Issue 2. [DOI: 10.1002/14651858.MR000010.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Huisstede 2010
- Huisstede BM, Hoogvliet P, Randsdorp MS, Glerum S, Middelkoop M, Koes BW. Carpal tunnel syndrome. Part 1: Effectiveness of nonsurgical treatments ‐ a systematic review. Archives of Physical Medicine and Rehabilitation 2010;91(7):981‐1004. [DOI] [PubMed] [Google Scholar]
Iyengar 1966
- Iyengar BKS. Light on yoga. London, England: Allen & Unwin, 1966. [Google Scholar]
Katz 1990
- Katz JN, Stirrat CR, Larson MG, Fossel AH, Eaton HM, Liang MH. A self‐administered hand symptom diagram for the diagnosis and epidemiologic study of carpal tunnel syndrome. Journal of Rheumatology 1990;17(11):1495‐8. [PUBMED: 2273490 ] [PubMed] [Google Scholar]
Keith 2009
- Keith MW, Masear V, Chung K, Maupin K, Andary M, Amadio PC, et al. Diagnosis of carpal tunnel syndrome. Journal of American Academy of Orthopaedic Surgeons 2009;17(6):389‐96. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kerwin 1996
- Kerwin G, Williams CS, Seiler JG 3rd. The pathophysiology of carpal tunnel syndrome. Hand Clinics 1996;12(2):243‐51. [PUBMED: 8724576] [PubMed] [Google Scholar]
Kirkham 2010
- Kirkham JJ, Dwan KM, Altman DG, Gamble C, Dodd S, Smyth R, et al. The impact of outcome reporting bias in randomised controlled trials on a cohort of systematic reviews. BMJ 2010;340:c365. [PUBMED: 20156912] [DOI] [PubMed] [Google Scholar]
Levine 1993
- Levine DW, Simmons BP, Koris MJ, Daltroy LH, Hohl GG, Fossel AH, et al. A self‐administered questionnaire for the assessment of severity of symptoms and functional status in carpal tunnel syndrome. Journal of Bone and Joint Surgery 1993;75(11):1585‐92. [PUBMED: 8245050] [DOI] [PubMed] [Google Scholar]
Maitland 1991
- Maitland GD. Peripheral Manipulation. 3rd Edition. Oxford: Butterworth‐Heinemann, 1991. [Google Scholar]
Marshall 2007
- Marshall SC, Tardif G, Ashworth NL. Local corticosteroid injection for carpal tunnel syndrome. Cochrane Database of Systematic Reviews 2007, Issue 2. [DOI: 10.1002/14651858.CD001554.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
McKeon 2008
- McKeon JMM, Yancosek KE. Neural gliding techniques for the treatment of carpal tunnel syndrome: a systematic review. Journal of Sport Rehabilitation 2008;17(3):324‐41. [DOI] [PubMed] [Google Scholar]
Muller 2004
- Muller M, Tsui D, Schnurr R, Biddulph‐Deisroth L, Hard J, MacDermid JC. Effectiveness of hand therapy interventions in primary management of carpal tunnel syndrome: a systematic review. Journal of Hand Therapy 2004;17(2):210‐28. [DOI] [PubMed] [Google Scholar]
O'Connor 2012
- O’Connor D, Page MJ, Marshall SC, Massy‐Westropp N. Ergonomic positioning or equipment for treating carpal tunnel syndrome. Cochrane Database of Systematic Reviews 2012, Issue 1. [DOI: 10.1002/14651858.CD009600] [DOI] [PMC free article] [PubMed] [Google Scholar]
Odgaard‐Jensen 2011
- Odgaard‐Jensen J, Vist GE, Timmer A, Kunz R, Akl EA, Schünemann H, et al. Randomisation to protect against selection bias in healthcare trials. Cochrane Database of Systematic Reviews 2011, Issue 4. [DOI: 10.1002/14651858.MR000012.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ono 2010
- Ono S, Clapham PJ, Chung KC. Optimal management of carpal tunnel syndrome. International Journal of General Medicine 2010;3:255‐61. [DOI] [PMC free article] [PubMed] [Google Scholar]
Page 2012
- Page MJ, O’Connor D, Pitt V, Massy‐Westropp N. Therapeutic ultrasound for carpal tunnel syndrome. Cochrane Database of Systematic Reviews 2012, Issue 1. [DOI: 10.1002/14651858.CD009601] [DOI] [PubMed] [Google Scholar]
Phalen 1966
- Phalen GS. The carpal tunnel syndrome. Journal of Bone and Joint Surgery 1966;48A:211‐28. [PubMed] [Google Scholar]
Piazzini 2007
- Piazzini DB, Aprile I, Ferrera PE, Bertolini C, Tonali P, Maggi L, et al. A systematic review of conservative treatment of carpal tunnel syndrome. Clinical Rehabilitation 2007;21(4):299‐314. [DOI] [PubMed] [Google Scholar]
Rempel 1998
- Rempel D, Evanoff B, Amadio PC, Krom M, Franklin G, Franzblau A, et al. Consensus criteria for the classification of carpal tunnel syndrome in epidemiologic studies. American Journal of Public Health 1998;88(10):1447‐51. [PUBMED: 9772842] [DOI] [PMC free article] [PubMed] [Google Scholar]
Scholten 2007
- Scholten RJPM, Mink van der Molen A, Uitdehaag BMJ, Bouter LM, Vet HCW. Surgical treatment options for carpal tunnel syndrome. Cochrane Database of Systematic Reviews 2007, Issue 4. [DOI: 10.1002/14651858.CD003905.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Stallings 1997
- Stallings SP, Kasdan ML, Soergel TM, Corwin HM. A case‐control study of obesity as a risk factor for carpal tunnel syndrome in a population of 600 patients presenting for independent medical examination. The Journal of Hand Surgery 1997;22(2):211‐5. [DOI] [PubMed] [Google Scholar]
Sterne 2011
- Sterne JAC, Egger M, Moher D. Addressing reporting biases. In: Higgins JPT, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011:Available from www.cochrane‐handbook.org. [Google Scholar]
Szabo 1992
- Szabo RM, Madison M. Carpal tunnel syndrome. Orthopedic Clinics of North America 1992;23(1):103‐9. [PUBMED: 1729659] [PubMed] [Google Scholar]
Szabo 1994
- Szabo RM, Steinberg DR. Nerve entrapment syndromes in the wrist. Journal of the American Academy of Orthopedic Surgeons 1994;2(2):115‐23. [DOI] [PubMed] [Google Scholar]
Totten 1991
- Totten PA, Hunter JM. Therapeutic techniques to enhance nerve gliding in thoracic outlet syndrome and carpal tunnel syndrome. Hand Clinics 1991;7:505‐20. [PubMed] [Google Scholar]
Verdugo 2008
- Verdugo RJ, Salinas RS, Castillo J, Cea JG. Surgical versus non‐surgical treatment for carpal tunnel syndrome. Cochrane Database of Systematic Reviews 2008, Issue 4. [DOI: 10.1002/14651858.CD001552.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Werner 1994a
- Werner RA, Albers JW, Franzblau A, Armstrong TJ. The relationship between body mass index and the diagnosis of carpal tunnel syndrome. Muscle & Nerve 1994;17(6):632‐6. [DOI] [PubMed] [Google Scholar]
Werner 1994b
- Werner R, Bir C, Armstrong T. Reverse Phalen's maneuver as an aid in diagnosing carpal tunnel syndrome. Archives of Physical Medicine and Rehabilitation 1994;75(7):783‐6. [PubMed] [Google Scholar]
Wood 2008
- Wood L, Egger M, Gluud LL, Schulz KF, Jüni P, Altman DG, et al. Empirical evidence of bias in treatment effect estimates in controlled trials with different interventions and outcomes: meta‐epidemiological study. BMJ 2008;336(7644):601‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
References to other published versions of this review
O'Connor 2003
- O'Connor D, Marshall S, Massy‐Westropp N. Non‐surgical treatment (other than steroid injection) for carpal tunnel syndrome. Cochrane Database of Systematic Reviews 2003, Issue 1. [DOI: 10.1002/14651858.CD003219] [DOI] [PMC free article] [PubMed] [Google Scholar]


