Abstract
Traditional approaches to the human cochlear nerve have been impeded by its bony encasement deep inside the skull base. We present an innovative, minimally invasive, therapeutic pathway for direct access to the nerve to deliver novel regenerative therapies. Neuroanatomical studies on 10 cadaveric human temporal bones were undertaken to identify a potentially safe therapeutic pathway to the cochlear nerve. Simulations based on three-dimensional delineation of anatomical structures obtained from synchrotron phase-contrast imaging were analyzed. This enabled the identification of an approach to the nerve in the fundus of the internal auditory meatus by trephining the medial modiolar wall of the cochlea via the round window for a median depth of 1.48 mm (range 1.21–1.91 mm). The anatomical access was validated on 9 additional human temporal bones using radio-opaque markers and contrast injection with micro-computed tomography surveillance. We thus created an effective conduit for the delivery of therapeutic agents to the cochlear nerve.
Keywords: Cochlear nerve, Human, Micro-CT, Synchrotron Imaging, Deafness, Stem cell therapy, Gene Therapy
Subject terms: Stem-cell research, Sensory systems
Manuscript editing provided by Lauren Siegel.
Introduction
Unaddressed hearing loss is, globally, the third greatest cause of years lived with a disability and it is predicted that the condition may affect as many as 2.5 billion individuals by 20501 with far-reaching economic, societal, and personal consequences for those affected. Permanent hearing loss is most often due to impairment of the sensory end-organ whose functions are sensory, synaptic, and neural. The sensory role is attributed primarily to the mechano-electric transduction function of hair cells that convert sound into electrical signals. The synapse links the base of the inner hair cells with the terminal filaments of the cochlear nerve and provides indefatigable sound encoding with the highest temporal precision. The neural function is subserved by afferent fibres in the cochlear nerve whose cell bodies are housed in Rosenthal’s canal (RC) and whose central fibres synapse with the dorsal and ventral cochlear nuclei in the brainstem. In addition, the ear has a neural efferent system controlling the auditory periphery. While the role of hair cells in causing permanent hearing loss has long been recognised2, much recent attention has focused on the critical role played by synaptic and neural dysfunction as primary causes of hearing difficulty precipitating the need to find therapeutic solutions that address these previously unrecognised causes of hearing disability3–5. Typically, these patients complain that they ‘can hear but cannot understand’, especially in complex listening conditions like speech in noise or multi-talker settings. Interestingly, pure-tone audiometric thresholds can be well-preserved or even normal, yet affected individuals can experience hearing difficulty listening to complex sounds.
There is growing interest in the development of treatments to restore inner ear function through local application of therapeutic agents, such as stem cell-derived cell types6,7 and gene therapies8–10. This will be further encouraged by the recent reported success of gene therapy for deafness due to mutation of the OTOF gene11. For the delivery of cell-based therapeutic products targeting the neural elements of the cochlea, excellent access to the nerve canal in the modiolus can be achieved as exemplified by Chen et al. (2012)7 in the gerbil model. However, the small sub-compartments of the human cochlea remain beyond reach in established clinical practice, with only the scala tympani being accessed via the RW or through a cochleostomy during cochlear implant insertion. Hence, there is a compelling need to identify new pathways to access the neurosensory structures within or immediately adjacent to the inner ear e.g. in the internal auditory meatus (IAM) that do not compromise cochlear or labyrinthine function, that preserve its blood supply, and that avoid injury to adjacent neurosensory or intracranial structures.
No clinical procedure currently exists to access the cochlear nerve other than through invasive intracranial surgeries; these carry substantial risk and are used for tumour removal rather than the delivery of therapeutic agents. When aiming to restore spiral ganglion neurons via cell therapy, delivery into the nerve compartment could be key to its efficacy as it will resemble the approach used in the gerbil model where restoration of function was demonstrated7. The neural microenvironment is more likely to favour survival and engraftment of neural otic progenitors, than for instance, the scala tympani. Residual fibers remaining within the nerve canal could act as tracks to guide the newly formed neurites to grow and reach their targets, both centrally and in the periphery12. Moreover, given the anatomical continuity, cells delivered in the IAM may migrate to the RC, the compartment where spiral ganglion neuron bodies naturally reside. In a previous study, we described an approach to reach directly to the cell bodies of the cochlear nerve in RC13 in the human cochlea. However, access and delivery into the IAM would have the advantage of allowing the administration of larger volumes. Direct delivery into the RC could still find useful application when these sub-structures are the main therapeutics targets (for instance, when delivering virus-associated genes).
We thus undertook a range of neuroanatomical studies designed to elaborate a therapeutic pathway to the human cochlear nerve that would be applicable to clinical practice.
Methods
The methodology used in the study was synchrotron radiation phase-contrast imaging (SR-PCI) which has been described in detail elsewhere14–17. In brief, the imaging technique is that of in-line, propagation-based X-ray phase-contrast imaging which, unlike conventional X-ray absorption imaging, uses X-ray refraction to enhance the visibility of tissue boundaries within a sample. A spatially coherent source of radiation is needed and hence synchrotron radiation is used. An additional essential difference with conventional X-ray imaging is that the detector is placed at a greater distance from the sample allowing Fresnel fringes to become detectable, leading to edge enhancement in the images. For the inner ear, SR-PCI has the overwhelming advantage of enhancing structural boundaries of tissues, especially soft tissues, without the need for contrast agents, which are difficult to apply and can result in tissue shrinkage. In addition, SR-PCI has the advantage of producing high-contrast images of the microscopic structures of the inner ear without incurring dissection or staining artefacts which are inherent in conventional methods used to explore the human inner ear. Ten fresh human temporal bones underwent SR-PCI. Once the data were acquired, quantitative analysis and three-dimensional (3D) rendering were undertaken using the open source software 3D Slicer (version 5.0.3, https://www.slicer.org/) for visualization, segmentation, and trajectory planning through repeated surgical simulation18. This necessitated repeated cropping of the images, numerous simulated trajectories, and detailed mensuration to determine if a pathway could be described that permitted access to the nerve’s high-frequency fibres as they fan out in the fundus of the IAM while the lower frequency fibres (< 900 Hz) coalesce into a trunk within the central modiolus.
A major requirement of these neuroanatomical studies was delineation of the arterial and venous supplies of the inner ear and IAM as, for clinical translation, any proposed anatomical pathway would have to avoid disruption of blood supply to preserve cochlear and vestibular function. Thus, we used the 3D SR-PCI images to map the relevant microvasculature with specific reference to its vulnerability during a therapeutic intervention.
To validate the proposed approach, three otologic surgeons each performed anatomical dissections following the proposed therapeutic route on 9 further human temporal bones. A routine mastoidectomy and opening of the facial recess was undertaken ensuring identification of the facial nerve and the chorda tympani. The round window (RW) membrane was identified, its bony overhang removed and was detached anteriorly and folded back to visualize the crista fenestra and the medial wall of the scala tympani. The key anatomical structures proved to be the crista fenestra and chorda tympani. The surgeons used hand-trephines (0.4 and 0.6 mm diameter) for accuracy and haptic feedback. Trephining began on the infero-medial wall of the scala tympani to fenestrate the fundus of the IAM to access its antero-inferior compartment; the trajectory used was based on data from prior 3D surgical modelling. The depth of drilling required was estimated from the SR-PCI models and was helped by haptic feedback felt by the surgeons on entering the IAM as they experienced an instantly recognisable kinaesthetic force reduction. Once the floor of the fundus of the IAM had been fenestrated, a radio-opaque marker (0.1 mm metal wire) was placed through the bony channel to enable verification by micro CT (Phoenix V|tome|x M300, GE) scanning that the target structure (i.e. the cochlear nerve) had been reached. To exemplify its potential application to deliver otic neuroprogenitors in future clinical trials, a suspension of these cells combined with contrast medium was injected through the bony fenestration using a Polytip™ cannula 23 g/38 g (MedOne™) and 1mL Luer syringe to assess the dispersal of the contrast in the fundus of the IAM and its ability to target the cochlear nerve. A suspension of otic neuroprogenitors diluted in a 100% w/v potassium iodide solution in phosphate buffered saline (PBS) was selected for contrast, as its low viscosity readily enabled injection of minute volumes; in addition, this concentration offered optimal imaging of the contents of the IAM fundus and captured the presence of free contrast medium within it post-injection.
Results
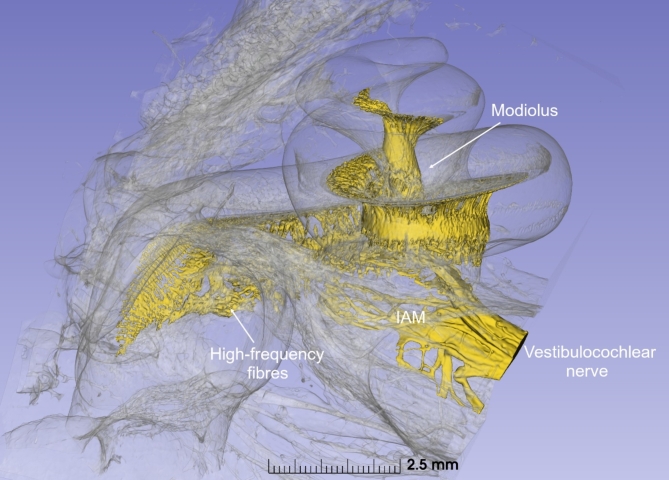
In Fig. 1, the temporal bone has been rendered transparent to allow visualization of the terminal filaments of the nerve as they leave the cochlea to form the cochlear nerve in the IAM. The typical spiral structure of the cochlea is noted, with its central core (modiolus) containing RC which houses the cell bodies of the afferent fibres of the cochlear nerve. Perpendicular to the modiolus, some nerve fibres fan out through a helical shaped cribriform area to reach the nerve in the IAM directly. These myelinated cochlear nerve fibres are of foremost importance as they represent the high-frequency neurons of the cochlea which are key targets for neural regeneration.
Fig. 1.
SR-PCI 3D reconstruction delineating the sensory nerve elements in a left human cochlea. The otic capsule is made semi-transparent to visualize the innervation of the cochlea and how the central axonal processes of the cochlear nerve are an accessible target for therapeutic intervention. The vestibulocochlear nerve within the IAM is visualized. IAM, internal acoustic meatus.
Having identified the location of the filaments of the cochlear nerve we used SR-PCI temporal bone models to explore how its high-frequency fibres could be safely accessed for therapeutic purposes. To facilitate translation to clinical practice, we confined our trajectory simulations to a route that is commonly used during ear surgery (called the posterior tympanotomy) that provides ready access to the cochlea’s RW (Fig. 2). The round window is a natural partition between the middle and inner ear that provides ideal minimally invasive access into the inner ear. Manipulating the models through repeated trials of many trajectories to the IAM led us to identify a therapeutic pathway to the nerve through the RW, traversing the scala tympani (Fig. 3). As a result, the possibility of fenestrating the floor of the fundus of the IAM and creating a bony tunnel permitting the delivery of therapeutic substances directly to the cochlear nerve emerged. The length of this tunnel was measured across 9 temporal bones and was found to average just 1.48 mm (SD 0.23 mm) (Table 1). Using a routine surgical approach to the RW, a fine hand-trephine could drill the bony tunnel through which a delivery system for the direct application of novel therapeutic agents can be introduced (Fig. 4).
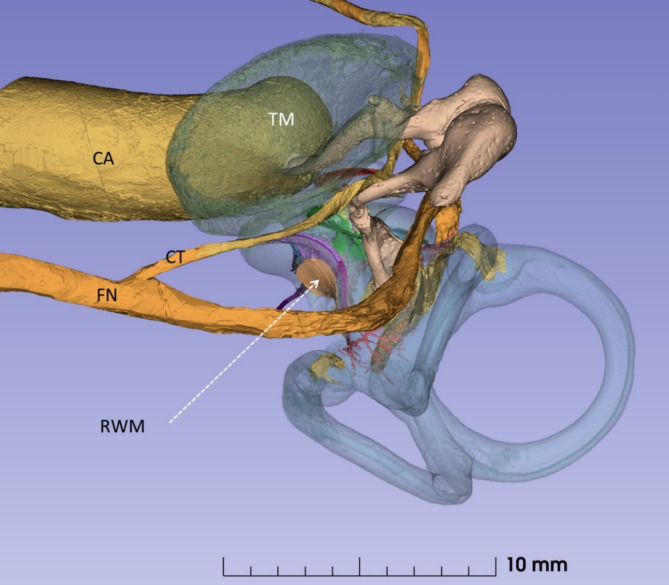
Fig. 2.
SR-PCI of a left human ear. It demonstrates the exposure obtained of the round window (arrow) through a routine trans-mastoid approach. FN facial nerve, CT chorda tympani, RWM round window membrane, TM tympanic membrane, CA carotid artery.
Fig. 3.
SR-PCI section and 3D reconstruction of a left human ear. The proximity of the RWM to the IAM is shown. The distance across the modiolar bone to reach the IAM is typically just over 1 mm. Broken arrow shows the desired trajectory to reach the IAM from the posterior tympanotomy. IAM internal acoustic meatus, RWM round window membrane.
Table 1.
The depth of drilling required to create a bony tunnel to fenestrate the floor of the fundus of the IAM from the bony rim (crista fenestra) of the RW in 9 temporal bones.
| Temporal Bone Specimen |
Tunnel Depth (mm) |
|---|---|
| 1 | 1.24 |
| 2 | 1.54 |
| 3 | 1.21 |
| 4 | 1.63 |
| 5 | 1.65 |
| 6 | 1.3 |
| 7 | 1.43 |
| 8 | 1.39 |
| 9 | 1.91 |
| Average | 1.48 |
| SD | 0.23 |
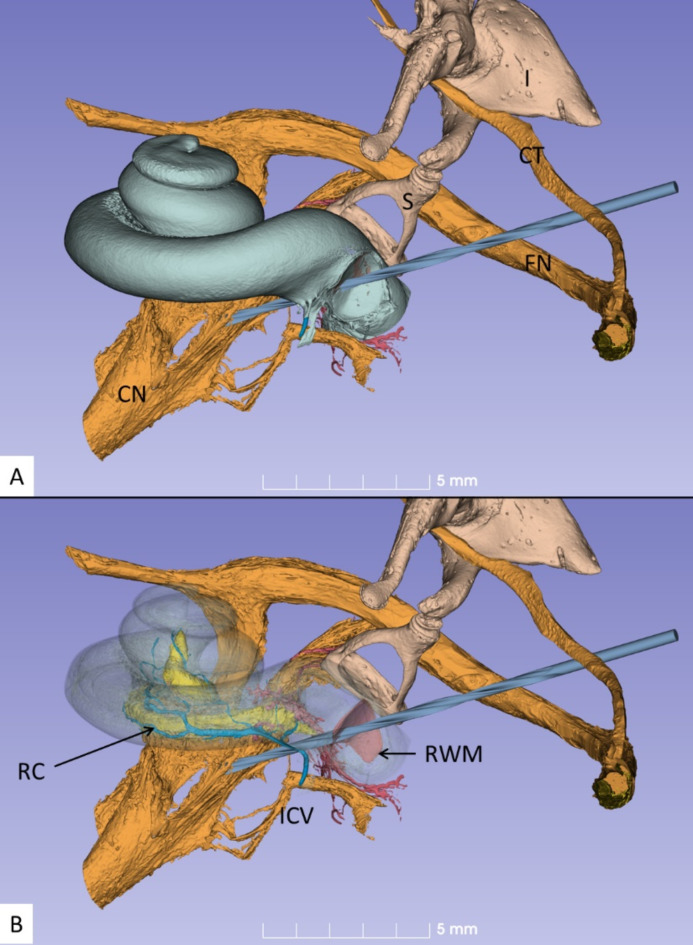
Fig. 4.
(A) Inferior view of the left cochlea showing the RW trajectory to reach the high-frequency cochlear nerve fibres in the fundus of the IAM. The shaft of the drill is positioned on the bony rim of the round window (crista fenestra) and is angled anteriorly towards the chorda tympani (CT). FN, facial nerve; CN, cochlear nerve; S, stapes; I, incus. (B). Same exposure as in A with the cochlea rendered transparent. Note the position of Rosenthal’s canal (RC) housing the cell bodies of the cochlear neurons. ICV inferior cochlear vein, RWM round window membrane.
Central to the clinical applicability of this approach is the ability to avoid injury to the cochlea’s blood supply to retain cochlear and vestibular function. Again, using SR-PCI, we were able to visualize the cochlea’s blood supply as well as the arrangement of vessels in the fundus of the IAM. At the level of the RW, the inferior cochlear vein and the cochlear branch of the vestibulo-cochlear artery (VCA) are most at risk; we recently described a safe ‘triangle’ which permits drilling at the RW without compromising these vessels13. Within the fundus of the IAM, we mapped the positions of the VCA and cochlear arteries (Fig. 5). The VCA, which enters the cochlea through its high frequency region, is at risk during drilling of the bony access tunnel, especially if drilling is too deep and enters the lumen of the IAM. (Supplement Fig. 1)
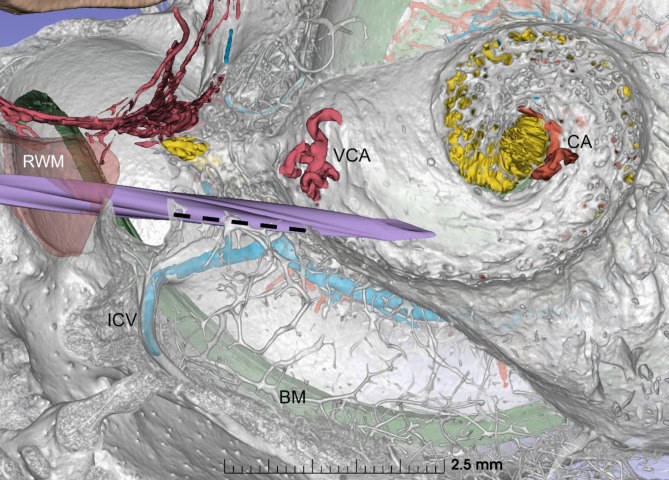
Fig. 5.
Vertical cross-section of a left human cochlea displays the relationship between the RWM, basal turn of the cochlea, and the spirally shaped cribriform area in the fundus of the IAM housing the fibres of the cochlear nerve. The drill has penetrated the modiolus creating a bony tunnel allowing delivery of therapeutic agents directly to the cochlear nerve. The average length of the tunnel created from the crista fenestra to the entry point in the IAM is 1.43 mm (interrupted line). Note the position of the blood vessels in the IAM fundus. The cochlear artery (CA) enters the cochlea through the central cochlear aperture; being more distant from the entry point of the drill, it is less at risk. However, the vestibulo-cochlear artery (VCA) enters the cochlea through one of the minor cochlear foramina in the cribriform area and is thus more vulnerable. ICV inferior cochlear vein, BM basilar membrane, RWM round window membrane, VCA vestibulo-cochlear artery, CA cochlear artery.
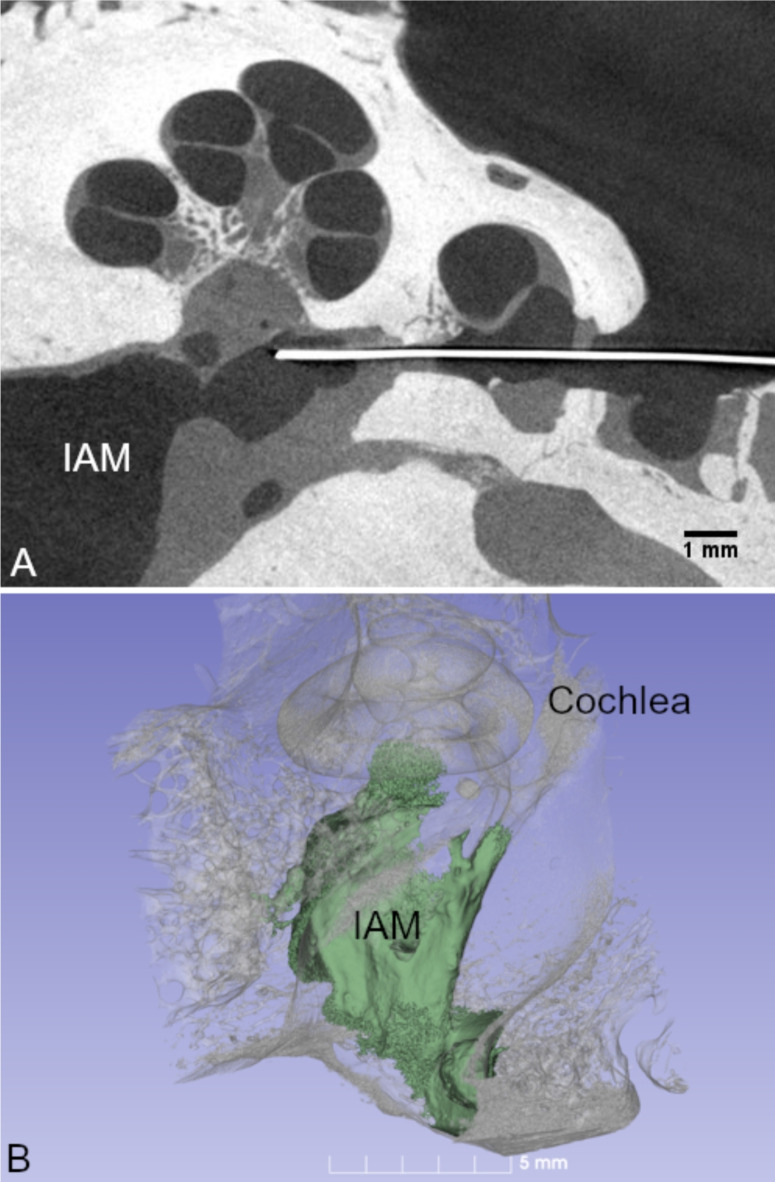
To validate the approach, three experienced otologists carried out the procedure on 9 human temporal bones. Once the IAM was felt to have been fenestrated through haptic feedback and the cochlear nerve had been reached, each surgeon placed a radio-opaque marker through the channel to permit verification of its location by means of micro-CT scanning (Fig. 6A). In each case, scanning confirmed the precise location of the marker at its target location (i.e. the cochlear nerve in the fundus of the IAM) without any evidence of structural collateral damage to the cochlea. To further assess the utility of the approach, a cell suspension and contrast was injected through the channel in 9 specimens which was followed by free dispersal of the medium throughout the IAM (Fig. 6B).
Fig. 6.
Micrograph showing the completed bony access tunnel from the round window membrane, through the modiolus, to reach the IAM and containing a radio-opaque marker needle (A). Cell suspension and contrast medium (green, 20uL, otic neuroprogenitors/potassium iodide) was injected through the tunnel and was distributed throughout the IAM (B). IAM, internal acoustic meatus.
Discussion
The functional molecular characterization of the inner ear has resulted from recent advances in the elaboration of its genetic architecture and is creating a vista of opportunities for novel therapeutic interventions9. These include gene replacement, gene augmentation or gene editing as well as cell-based regenerative therapies which will each need to be delivered through novel, minimally-invasive approaches to their target structures. In addition, there is growing awareness of the importance of the inner ear’s neural circuitry to the pathogenesis of hearing loss and its growing contribution to the global burden of disease. The prevailing view had been that degenerative processes in cochlear afferent fibres occurred secondary to loss of the sensory hair cells in the cochlea. It was also considered that other rare forms of hearing loss with a known genetic cause such as in Friedreich’s ataxia19 or the Mohr-Tranebjaerg syndrome20, primary neural degeneration was an exceptional occurrence. Until the advent of quadruple-immunostaining protocols, synaptic and neuronal counts on human temporal bones were not possible and thus these causes of hearing loss went unrecognised5. It is now known that in subjects over 60 years, > 60% of peripheral cochlear nerve fibres may be lost and that age-related axonal loss actually outstrips the loss of inner hair cells by almost 3:121. Some auditory nerves may be hypoplastic or have functional abnormalities characterized by deficient transfer of electrical signals from the inner hair cells to the nerve or where the nerve itself is functionally impaired (e.g. auditory neuropathy spectrum disorder or ANSD).
In animal models of reversible hearing loss due to acute acoustic injury, hair cells may be preserved only to be followed by loss of synaptic connections to inner hair cells with subsequent loss of spiral ganglion neurons3,22. The degeneration is selective and predominantly affects high threshold fibres which has minimal impact on pure tone audiometry. However, the perceptual consequences are acutely felt when deciphering speech in noisy environments4 – a near universal complaint of those with age-related hearing loss. Importantly, such primary cochlear neuronal degeneration has been shown to dysregulate activity in the auditory cortex, interfering with the normal balance of excitation and inhibition as it attempts to compensate for degraded sensory input; this results in atypical neural dynamics that alters the ability to detect sound in noise but not in quiet23,24.
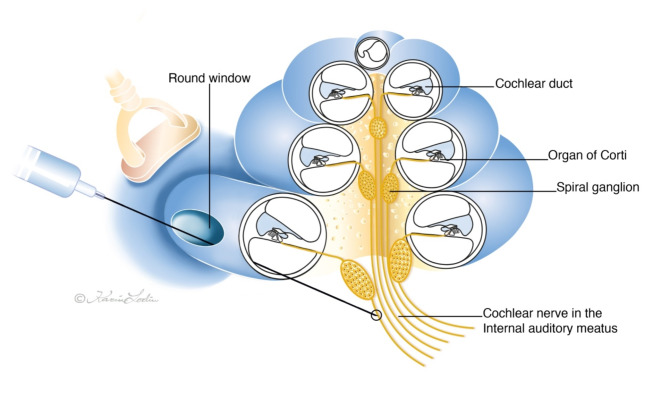
Given the growing understanding of the high prevalence of neural hearing impairment, future therapies including gene and cell-based therapies will need delivery routes for direct access to the cochlear nerve and its synapses that are minimally invasive and avoid collateral damage to adjacent anatomical structures. Here, we describe the neuroanatomical basis of such a potential future approach that permits direct access to the central axonal processes of the human cochlear nerve as they enter the bony plate of the cochlea. We expect this will enable delivery of novel therapeutic agents for the restoration of hearing due to synaptic and neural dysfunction (Fig. 7). The procedure has been validated anatomically on cadaveric human temporal bones. Although targeting the same structures, i.e. the space containing the fibres of the cochlear nerve, the approach differs from that of Chen et al. 20127 where otic neuroprogenitor cells were introduced directly into the gerbil modiolus via the RW; here, the approach is also via the RW but is intracranial, delivering cells into the cerebrospinal fluid space adjacent to the fibres of the cochlear nerve close to the fundus of IAC to facilitate future engraftment. In a previous paper13 we provided a detailed anatomical description of a clinical approach to the spiral ganglion cells of the cochlear nerve in Rosenthal’s canal which will also augment the range of therapeutic approaches to the nerve. An advantage of the approach described here is that, as the IAM has more capacity than the RC, it may tolerate slightly larger volumes of the putative therapeutic agents. Future in vivo studies in larger mammals or non-human primates25 may assess the physiological impact of the approach on cochlear and vestibular function, the nerves housed within the IAM, cerebrospinal fluid dynamics as well as the local and systemic biodistribution of injected therapeutic agents. The advent of advanced cochlear imaging and computerised trajectory planning, as well as developments in inner ear robotics26, will facilitate the precision surgery that these novel therapies will require.
Fig. 7.
Drawing showing the trajectory for injection to the cochlear nerve in the internal auditory meatus via the round window. Image by Karin Lodin.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
The project was supported in part by a research grant from Rinri Therapeutics administered via the University of Nottingham. This study was supported by the Swedish Research Council [2022–03339], the Tysta Skolan Foundation, and the Swedish Deafness Foundation (hrf). We also acknowledge the kind donations of private funds made by Arne Sundström, Sweden. Part of the research described in this paper was performed at the Canadian Light Source, a national research facility of the University of Saskatchewan, which is supported by the Canada Foundation for Innovation (CFI), the Natural Sciences and Engineering Research Council (NSERC), the National Research Council (NRC), the Canadian Institutes of Health Research (CIHR), the Government of Saskatchewan, and the University of Saskatchewan.
Author contributions
Conceptualization: all authors and M.N.R. contributed equally. Synchrotron imaging: S.A.R., N.Z., S.A., H.M.L. Modelling and Validation: H.L., D.E.H., D.J., G.M.O’D, H.R.A. Writing-original draft: G.M.O’D; Writing-review & editing: all authors contributed equally.
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
Declarations
Competing interests
Prof. Marcelo Rivolta is the Founder Director and Chief Scientific Officer of Rinri Therapeutics. Prof. Douglas Hartley is Rinri Therapeutics Chief Medical Officer. Other authors do not have any competing interest.
Ethics approval
All specimens were obtained as part of the body bequeathal program at Western University, London, Ontario, Canada in accordance with the Anatomy Act of Ontario and Western’s Committee for Cadaveric Use in Research (approval #122611). Human temporal bones for surgical injections were sourced from the tissue bank, University of Lund, Sweden in accordance with the National Board of Health and Welfare Regulation SOSFS 2009:30.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-024-74661-5.
References
- 1.WHO World Report on Hearing 2021.
- 2.Hudspeth, A. J. Integrating the active process of hair cells with cochlear function. Nat. Rev. Neurosci. 2014 159 15, 600–614 (2014). [DOI] [PubMed] [Google Scholar]
- 3.Kujawa, S. G. & Liberman, M. C. Adding insult to Injury: cochlear nerve degeneration after temporary noise-Induced hearing loss. J. Neurosci. 29, 14077 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Liberman, M. C. & Kujawa, S. G. Cochlear synaptopathy in acquired sensorineural hearing loss: manifestations and mechanisms. Hear. Res. 349, 138–147 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Viana, L. M. et al. Cochlear neuropathy in human presbycusis: confocal analysis of hidden hearing loss in post-mortem tissue. Hear. Res. 327, 78 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Janesick, A., Hashino, E. & Heller, S. Inner ear cells from stem cells: a path towards inner ear cell regeneration. 135–162 doi: (2023). 10.1007/978-3-031-20661-0_6
- 7.Chen, W. et al. Restoration of auditory evoked responses by human ES-cell-derived otic progenitors. Nature 490, 278–282 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Saidia, A. R. et al. Current advances in Gene therapies of genetic auditory neuropathy spectrum disorder. J. Clin. Med. 12, (2023). [DOI] [PMC free article] [PubMed]
- 9.Petit, C., Bonnet, C. & Safieddine, S. Deafness: from genetic architecture to gene therapy. Nat. Rev. Genet. 24, 665–686 (2023). [DOI] [PubMed] [Google Scholar]
- 10.Brown, A. et al. Surgical considerations in inner ear gene therapy from human temporal bone anatomy. Laryngoscope 10.1002/LARY.31259 (2024). [DOI] [PubMed] [Google Scholar]
- 11.Lv, J. et al. AAV1-hOTOF gene therapy for autosomal recessive deafness 9: a single-arm trial. Lancet 10.1016/S0140-6736(23)02874-X (2024). [DOI] [PubMed] [Google Scholar]
- 12.Hu, Z., Ulfendahl, M. & Olivius, N. P. Central migration of neuronal tissue and embryonic stem cells following transplantation along the adult auditory nerve. Brain Res. 1026, 68–73 (2004). [DOI] [PubMed] [Google Scholar]
- 13.Li, H. et al. Unlocking the human inner ear for therapeutic intervention. Sci. Rep. 2022 121 12, 1–6 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Elfarnawany, M. et al. Micro-CT versus synchrotron radiation phase contrast imaging of human cochlea. J. Microsc. 265, 349–357 (2017). [DOI] [PubMed] [Google Scholar]
- 15.Koch, R. W., Ladak, H. M., Elfarnawany, M. & Agrawal, S. K. Measuring Cochlear Duct length - a historical analysis of methods and results. J. Otolaryngol. Head Neck Surg. 46, 19 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mei, X. et al. Vascular supply of the human spiral ganglion: Novel three-dimensional analysis using Synchrotron phase-contrast imaging and histology. Sci. Rep. 10.1038/s41598-020-62653-0 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Agrawal, S. et al. The secondary spiral lamina and its relevance in cochlear implant surgery. Ups J. Med. Sci. 123, 9–18 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li, H. et al. Synchrotron Radiation-Based Reconstruction of the human spiral ganglion. Ear Hear. 1 10.1097/AUD.0000000000000738 (2019). [DOI] [PubMed]
- 19.Spoendlin, H. Optic and cochleovestibular degenerations in hereditary ataxias: II. Temporal bone pathology in two cases of Friedreich´s ataxia with vestibulo-cochlear disorders. Brain 97, 41–48 (1974). [DOI] [PubMed] [Google Scholar]
- 20.Bahmad, F., Merchant, S. N. & Nadol, J. B. Tranebjærg, L. Otopathology in Mohr-Tranebjærg syndrome. Laryngoscope 117, 1202–1208 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu, P. Z., Liberman, L. D., Bennett, K. & de Gruttola, V. Primary neural degeneration in the human cochlea: evidence for hidden hearing loss in the aging ear. Neuroscience 407, 8–20 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Furman, A. C. & Kujawa, S. G. Charles Liberman, M. noise-induced cochlear neuropathy is selective for fibers with low spontaneous rates. J. Neurophysiol. 110, 577 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Resnik, J. & Polley, D. B. Cochlear neural degeneration disrupts hearing in background noise by increasing auditory cortex internal noise. Neuron 109, 984 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bharadwaj, I. & Shinn-Cunningham, B. What’s been hidden in hidden hearing loss. Neuron 109, 909–911 (2021). [DOI] [PubMed] [Google Scholar]
- 25.Andres-Mateos, E. et al. Choice of vector and surgical approach enables efficient cochlear gene transfer in nonhuman primate. Nat. Commun. 13, 1359 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ding, A. S. et al. Automated Registration-based temporal bone computed tomography segmentation for applications in neurotologic surgery. Otolaryngol. Head Neck Surg. 167, 133–140 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated or analysed during this study are included in this published article and its supplementary information files.