Key Points
-
•
Using PTCY raised the risk of ECEs but did affect the likelihood of late cardiac toxicity.
-
•
Strategies to prevent EC are crucial, especially for patients receiving PTCY.
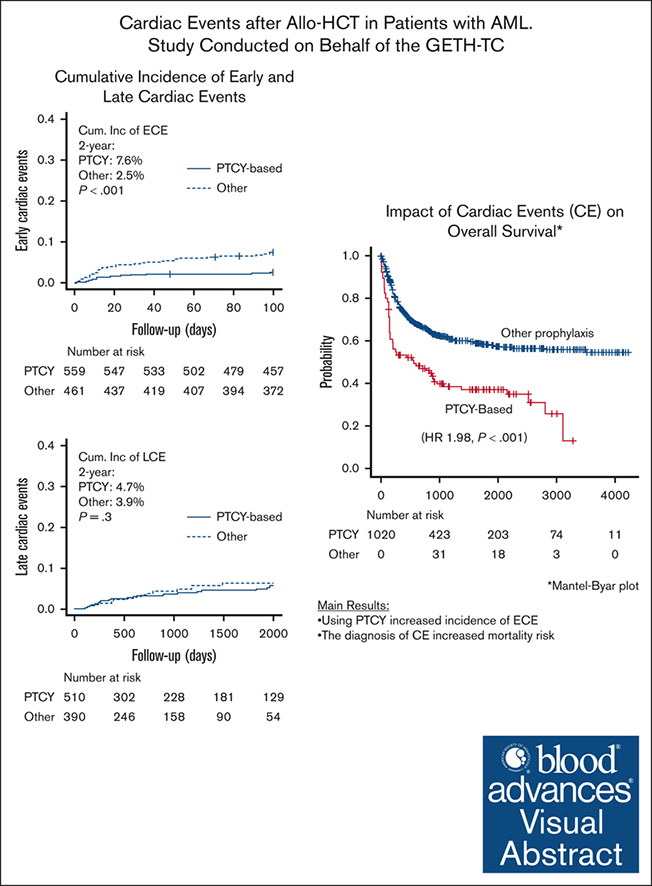
Visual Abstract
Abstract
This multicenter study sponsored by the GETH-TC investigates the incidence and predictors of early (first 100 days) and late cardiac events (CEs; ECEs and LCEs, respectively) after allo-HCT in patients with acute myeloid leukemia (AML) treated with anthracyclines, focusing on exploring the impact of PTCY on cardiac complications and the impact of CEs on OS and NRM. A total of 1020 patients with AML were included. PTCY was given to 450 (44.1%) adults. Overall, 94 (9.2) patients experienced CEs, with arrythmias, pericardial complications, and heart failure the most prevalent. ECEs occurred in 49 (4.8%) patients within a median of 13 days after allo-HCT, whereas LCEs were diagnosed in 45 (4.4%) patients within a median of 3.6 years after transplant. Using PTCY increased the risk for ECEs in multivariate analysis (hazard ratio [HR], 2.86; P = .007) but did not significantly affect the risk for LCEs (HR, 1.06; P = .892). The impact of variables on outcomes revealed was investigated using multivariate regression analyses and revealed that the diagnosis of CEs decreased the likelihood of OS (HR, 1.66; P = .005) and increased the likelihood of NRM (HR, 2.88; P < .001). Furthermore, despite using PTCY increased ECEs risk, its administration was beneficial for OS (HR, 0.71; P = .026). In conclusion, although the incidence of CEs was relatively low, it significantly affected mortality. Standard doses of PTCY increased ECE risk but were associated with improved OS. Therefore, protocols for preventing cardiac complications among these patients are needed.
Introduction
After the successful use of posttransplant cyclophosphamide (PTCY) in haploidentical hematopoietic cell transplantation (haplo-HCT), the use of PTCY-based prophylaxis for graft-versus-host disease (GVHD) prevention in allogeneic HCT (allo-HCT) regardless of donor type is increasingly prevalent.1, 2, 3, 4 Although PTCY-based prophylaxis has proved effective GVHD prevention, its wider implementation has raised concerns about causing potential cardiac-related toxicity.5, 6, 7, 8
Cardiac toxicity is a significant and potentially life-threatening complication after allo-HCT, which manifests shortly after the stem cell infusion or later in long-term survivors. Posttransplant cardiovascular diseases encompass a range of conditions including cardiomyopathy, heart failure, valvular dysfunction, arrhythmias, pericarditis, and coronary artery disease.5, 6, 7, 8, 9, 10 Reported incidences of cardiac toxicities have ranged from 6% to 19% in the early posttransplant period and can reach up to 22% over a 25-year span. Cyclophosphamide (CY), a commonly used alkylating agent in treating various malignant and autoimmune disorders and in conditioning regimens for allo-HCT, has been linked to cardiac toxicity with incidence rates between 1% and 17%.11, 12, 13 However, the investigation of how PTCY affects early cardiovascular toxicity in allo-HCT recipients has given controversial results.5, 6, 7, 8
Acute myeloid leukemia (AML) is the primary indication for allo-HCT.14,15 Patients with AML often receive anthracycline-based induction regimens to induce disease response before transplantation, but known to potentially induce cancer treatment–related cardiovascular toxicity.16, 17, 18 Because of the extensive use of PTCY-based prophylaxis in patients with AML who have received allo-HCT has raised concerns about possible higher incidence of cardiac-related toxicity in these patients,5, 6, 7, 8 this study investigates the incidence and predictors of early (E) and late (L) cardiac events (CEs) after allo-HCT, and in particular, whether PTCY-based prophylaxis increases the likelihood of cardiac complications.
Methods
Patient selection
This is a retrospective, multicenter, registry-based study sponsored by the transplant complications committee of the Grupo Español de Trasplante Hematopoyético y Terapia Celular (GETH-TC). GETH-TC is a nonprofit scientific society with members from all institutions performing HCT in Spain and Portugal. Sixteen transplant units affiliated with the GETH-TC participated in the study.
A total of 1020 adult patients with AML who underwent their first peripheral blood allo-HCT between 2011 and 2022, after anthracycline-containing induction therapies and received HCT at the 16 participating HCT units met the eligibility criteria and were included in the study. Data collection took place retrospectively from June 2023 to January 2024. Study data were collected and managed using REDCap (Research Electronic Data Capture) electronic data capture tools hosted at GETH-TC. The study received ethical approval from the ethics committee of the Hospital Clínic de Barcelona and adhered to the standards outlined in the Declaration of Helsinki. No external funding was received for this study.
CE definition and pretransplant cardiac function evaluation
A CE was defined as any new occurrence of atrial or ventricular arrhythmias, heart failure (including a decrease in left ventricular ejection fraction (LVEF) of ≥10% but always <53%), myocardial infarction or ischemia, and pericardial complications (moderate/severe pericardial effusion and pericarditis) diagnosed after stem cell infusion.5, 6, 7 Only the first CE occurring in patients included in the study was accounted in the analysis. Cardiac complications were determined following standard clinical practice and the 2022 European Society of Cardiology guidelines on cardio-oncology,18 and graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE), version 5.0. CEs were classified as ECE or LCE depending on whether it was diagnosed during the first 100 days after transplant, or it was diagnosed after day 100.5,6,19 De novo post–allo-HCT hypertension (HTN) was not considered a CE.
Pretransplant cardiac evaluation, monitoring, and supportive care followed institutional guidelines. Transthoracic echocardiography and electrocardiogram were performed uniformly during pretransplant assessment. Patients with preexisting cardiac morbidity included patients with arrhythmia, heart failure, ischemia, pericarditis, moderate/severe pericardial effusion, moderate/severe valvopathy, or other relevant cardiac complications diagnosed before allo-HCT. Patients with relevant abnormalities diagnosed in the echocardiography or electrocardiogram before transplant were accounted as patients with prior cardiac disease. Furthermore, patients with a LVEF of <45% and without a history of cardiac disorders were considered to have preexisting cardiac morbidity. Because routine measurement of cardiac biomarkers and function was not conducted uniformly across centers, information related to these aspects was not included in the study.
Allo-HCT information and definitions
Induction treatments, eligibility criteria for allo-HCT, donor selection, conditioning regimen, and GVHD prophylaxis all adhered to standard practices, with conditioning regimen intensity uniformly tailored to chronological age and comorbidities. Grading of acute and chronic GVHD (aGVHD and cGVHD, respectively) followed established criteria.20, 21, 22 CR and disease relapse were designated by the treating physician and registered at the database.
Statistical analysis
The diagnosis of CEs was the primary variable of interest, with PTCY-based prophylaxis being a key explanatory variable. Cumulative incidence of CEs was estimated using regression analysis and accounting for death and relapse as competing events. For descriptive information, patients diagnosed with a CE after disease relapse (n = 10) were considered patients without CE. Baseline characteristics and risk factors for ECEs and LCEs were investigated through landmark analysis, excluding events after day +100 for ECE and starting posttransplant follow-up on day +100 for LCE. The 30- and 60-day mortality rates were estimated from the day of CE diagnosis to the day of death in patients with CEs who died during the subsequent follow-up. The impact of CE on overall survival (OS) and nonrelapse mortality (NRM) was investigated using regression analysis and including the entire cohort of patients, without landmark analysis. In this analysis, the variable CE was always treated as a time-dependent variable.
Descriptive statistics were calculated using counts and percentages, and χ2 and Mann-Whitney U tests were used when testing for statistically significant differences in comparisons across subsamples. Posttransplant follow-up of patients with graft failure undergoing second allo-HCT was censored at the day of the second infusion. Posttransplant outcomes were estimated using Kaplan-Meier and cumulative incidence regression analyses, with GVHD analyses accounting for death and relapse as competing events. Predictors for CEs, OS, and NRM were evaluated using regression analyses. All analyses used 2-sided P values, with P < .05 considered statistically significant. Easy R software was used for statistical analysis.23
Results
Baseline information
Baseline information of the 1020 patients included is reported in Table 1. Overall, the median age was 55 years (interquartile range [IQR], 44-62), and 530 (52.7%) patients were male. A total of 195 (19.1%) patients were diagnosed with HTN, 133 (13.1%) with dyslipidemia (DLP) and 74 (7.2%) with diabetes mellitus before allo-HCT. Overall, 131 (12.8%) adults had a history of cardiac pathology, with arrythmia (n = 36, 3.5%) and moderate/severe valvopathy (n = 32, 3.1%) being the most prevalent.
Table 1.
Baseline characteristics of the study cohort and according to early cardiac toxicity
| Baseline characteristics | All patients N = 1020 |
Patients with ECEs n = 49 |
No ECEs n = 971 |
P value |
|---|---|---|---|---|
| Age, y, median (range) | 55 (44-62) | 60 (48-66) | 54 (44-62) | .003 |
| >59 | 369 (36.2) | 26 (53.1) | 343 (34.3) | .012 |
| Sex | ||||
| Male | 530 (52.7) | 29 (59.2) | 509 (52.4) | .355 |
| Female | 482 (47.3) | 20 (40.8) | 462 (47.6) | |
| Relevant comorbidities | ||||
| Active smoker | 111 (10.9) | 12 (24.5) | 105 (10.8) | .754 |
| HTN | 195 (19.1) | 19 (38.8) | 177 (18.2) | <.001 |
| DLP | 133 (13.1) | 15 (30.8) | 111 (12.2) | <.001 |
| Diabetes mellitus | 74 (7.2) | 4 (8.2) | 70 (7.2) | .810 |
| Obesity (BMI ≥30) | 70 (6.9) | 1 (2.0) | 69 (7.1) | .166 |
| History of cardiac disease | 131 (12.8) | 10 (20.4) | 121 (12.4) | .122 |
| Arrhythmia | 36 (3.5) | 4 (8.2) | 32 (3.2) | |
| Heart failure | 18 (1.7) | 2 (4.0) | 16 (1.6) | |
| Myocardial ischemia | 16 (1.5) | 1 (2.0) | 15 (1.5) | |
| Pericardial disorders∗ | 9 (0.9) | 2 (4.0) | 7 (0.7) | |
| Valvopathy | 33 (3.2) | 1 (2.0) | 32 (3.2) | |
| Other | 7 (0.6) | 0 | 7 (0.7) | |
| Abnormalities on ECHO or ECG before allo-HCT† | 12 (1.2) | 0 | 12 (1.2) | |
| Prior treatment with cardiotoxic chemotherapy | 31 (3.0) | 1 (2.0) | 30 (3.1) | 1 |
| Disease status before allo-HCT | ||||
| Complete remission | 923 (90.4) | 42 (85.7) | 888 (90.7) | .243 |
| Refractory AML/active disease | 97 (9.5) | 7 (14.3) | 90 (9.3) | |
| HCT-CI | ||||
| >3 | 142 (14.9) | 10 (20.8) | 132 (14.5) | .234 |
| Missing | 64 | 1 | 63 | |
| Main allo-HCT information | ||||
| Intensity | ||||
| Myeloablative | 594 (58.2) | 23 (46.9) | 571 (58.8) | .1 |
| Reduced intensity | 426 (41.8) | 25 (53.1) | 400 (41.2) | |
| MAC regimen containing CY | 140 (13.7) | 4 (8.2) | 136 (14.4) | .294 |
| TBI | ||||
| High dose TBI (12 Gy) | 45 (4.4) | 1 (2.0) | 44 (4.5) | .719 |
| GVHD prophylaxis | ||||
| PTCY-based | 461 (45.2) | 35 (71.1) | 426 (43.9) | <.001 |
| Others | 559 (54.8) | 14 (28.6) | 545 (56.1) | |
| Donor | ||||
| 10/10 MSD | 362 (35.3) | 13 (26.5) | 349 (35.9) | <.001 |
| 10/10 MUD | 299 (29.3) | 13 (26.5) | 286 (29.5) | |
| 9/10 MMUD | 86 (8.4) | 0 | 86 (8.9) | |
| Haploidentical | 273 (26.8) | 23 (46.9) | 250 (25.7) | |
| Main results | ||||
| Median days to neutrophil engraftment | 17 (15-19) | 16 (14-18) | 17 (15-19) | .325 |
| Median days to platelet engraftment | 15 (12-24) | 16 (13-26) | 15 (12-24) | .416 |
| Graft failure | 36 (3.6) | 1 (2.0) | 35 (3.7) | .231 |
| Missing data | 18 | 3 | 15 | |
| Cumulative incidence of GVHD | ||||
| Day +100 grade 2-4 aGVHD | 33.5 (30.6-36.5) | 34.7 (21.7-48.1) | 33.5 (30.5-36.5) | .513 |
| Day +100 grade 3-4 aGVHD | 9.3 (7.6-11.2) | 8.2 (2.6-18.0) | 9.3 (7.6-11.3) | .545 |
| Relapse | 298 (29.3) | 13 (26.5) | 286 (29.5) | 0.651 |
| Dead | 414 (40.5) | 27 (55.1) | 387 (39.9) | 0.034 |
| Directly secondary to ECE | 4 (0.4) | 4 (8.1) | 0 |
BMI, body mass index; ECG, electrocardiogram; ECHO, echocardiography; HCT-CI, HCT–comorbidity index; TBI, total body irradiation; MSD, matched sibling donor; MUD, matched unrelated donor; MMUD, mismatched unrelated donor.
Pericarditis or moderate/severe pericardial effusion.
Patients without cardiac history but with relevant abnormalities diagnosed in the ECHO or ECG and LVEF of <45% and without a history of cardiac disorders.
Myeloablative conditioning (MAC) allo-HCTs were performed in 594 (58.2%) patients (13.7% of them containing CY and 14.4% containing high doses of total body irradiation). Overall, 591 (57.9%) adults received grafts from 10/10 HLA-matched sibling donors and matched unrelated donors, 86 (8.4%) from 9/10 mismatched unrelated donors, and 273 (26.7%) from haploidentical donors. PTCY-based prophylaxis was administered to 461 (45.1%) adults; most of them received the standard doses of CY of 50 mg/kg per day × 2 days (n = 454, 98.5%).
Main transplant results
The median number of days to neutrophil and platelet engraftment were 17 (IQR, 15-19) and 15,12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 respectively. Primary and secondary graft failure occurred in 36 adults (3.6%). Day + cumulative incidence functions (CIF) of grades 2 to 4 and 3 to 4 aGVHD were 33.5% (95% confidence interval [CI], 30.6-36.5) and 9.3% (95% CI, 7.6-11.2), respectively. The 2-year CIF of moderate/severe cGVHD was 7.0% (95% CI, 5.4-8.9). With a median follow-up of 32 months (IQR, 12-66), 298 (29.3%) patients relapsed and 414 (40.5%) died. The main causes of death were infection and relapse. Overall, the 2-year OS, NRM, and cumulative incidence of relapse rates were 65.3% (95% CI, 62.2-68.2), 15.6% (95% CI, 13.4-17.9), and 27.1% (95% CI, 24.3-29.9), respectively.
ECEs
ECEs occurred in 49 (4.8%) patients within a median of 13 days (range, 7-44) after allo-HCT. As described in Table 1, compared with patients without ECEs, patients diagnosed with ECEs were older (median age of 60 vs 54 years, P = .012) and had a higher propensity to HTN (38.8% vs 18.2%, P < .001) and DLP (30.8% vs 12.2%). The proportion of patients with prior cardiac disease tend to be higher in adults who presented with ECEs (20.4% vs 12.4%, P = .122).
Allo-HCT characteristics differed between the 2 groups in terms of the proportion of patients receiving PTCY (71.1% vs 43.9%, P < .001) and haploidentical sources (46.9% vs 25.7%, P < .001), being higher in patients presenting ECEs. In fact, the cumulative incidence of ECEs at day +100 was 7.6% (95% CI, 5.4-10.3) in patients receiving PTCY, and 2.5% (95% CI, 1.4-4.1; P < .001) in those who did not. According to donor type, the day +100 cumulative incidences of ECEs were 3.6% (95% CI, 2.0-5.9) in allo-HCT performed from matched sibling donors, 4.4% (85% CI, 2.4-7.1) from 10/10 HLA–matched unrelated donors, 0% from mismatched unrelated donors, and 8.4% (95% CI, 5.5-12.1) from haploidentical donors (0 = 0.003).
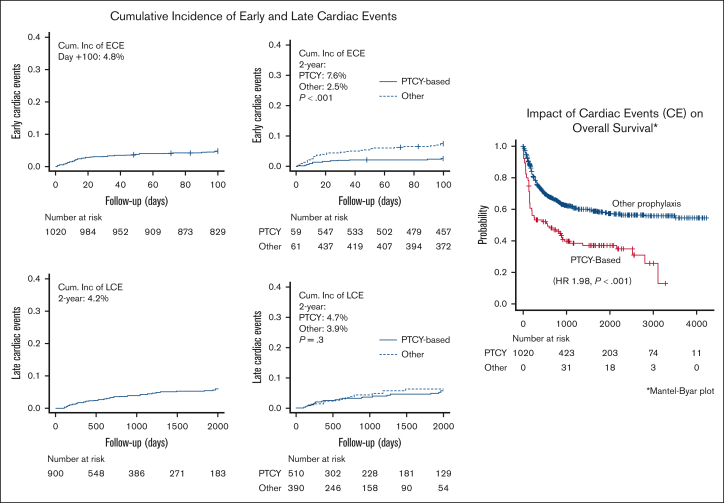
As illustrated in Figure 1, the day +60 and +100 CIF of ECEs were 3.9% (95% CI, 2.9-5.2) and 4.8% (95% CI, 3.6-6.2), respectively. As described in Table 2, the most prevalent cardiac complications were arrythmias (all supraventricular; n = 21, 42.9%), pericardiac effusion or pericarditis (n = 14, 28.6%), and heart failure (n = 9, 18.4%). According to the CTCAE, 14 (28.6%) patients had a grade 3/4 ECE, and 4 (8.1%) patients directly died secondary to this complication. The day +30 and +60 mortality rates among patients with ECEs were 10.2% and 16.3%, respectively.
Figure 1.
Cumulative incidence of ECEs and LCEs and impact on OS.
Table 2.
Early and late CEs: detailed information
| n (%) | Grade of severity (CTCAE) | Median days to diagnosis (IQR) | Additional comment | Death secondary to CAE |
Day +30 mortality rate | Day +60 mortality rate | ||
|---|---|---|---|---|---|---|---|---|
| All patients with ECEs | 49 (100) | 1-2 3-4 5 |
30 (61.2) 14 (30.6) 4 (8.1) |
13 (7-44) | N/A | 4 (8.1) | 5 (10.2) | 8 (16.3) |
| Heart failure | 9 (18.4) | 1-2 3-4 5 |
4 (44.4) 3 (33.3) 2 (22.2) |
14 (12-50) | One (11.1) case presented with cardiac pulmonary edema | 2 (22.2) | 2 (22.2) | 3 (33.3) |
| Myocardial infarction or ischemia | 4 (8.2) | 1-2 3-4 5 |
1 (25.0) 3 (75.0) 0 |
9 (3-75) | Two (50.0%) required percutaneal revascularization | 0 | 0 | 0 |
| Arrythmia | 21 (42.9) | 1-2 3-4 5 |
15 (71.4) 4 (19.0) 2 (9.5) |
12 (7-21) | All supraventricular arrythmias |
2 (9.5) | 2 (9.5) | 3 (14.2) |
| Pericardiac effusion or pericarditis | 14 (28.6) | 1-2 3-4 5 |
10 (71.4) 4 (28.6) 0 |
21 (3-75) | 6 (42.8%) cases were diagnosed with pericarditis. Four (28.6%) cases required pericardial drain. No patient received TBI. | 0 | 1 (7.1) | 2 (14.2) |
| Others | 1 (2.0) | 1-2 3-4 5 |
0 1 (100) 0 |
35 | Auricular thrombus | 0 | 0 | 0 |
| N (%) | Grade of severity (CTCAE) | Median months to diagnosis (IQR) | Additional comment | Death secondary to CAE |
Day +30 mortality rate | Day +60 mortality rate | ||
|---|---|---|---|---|---|---|---|---|
| All patients with LCEs | 45 (100) | 1-2 3-4 5 |
29 (64.4) 14 (31.1) 2 (4.4) |
17.5 (7.9-34.7) | N/A | 2 (4.4) | 5 (11.1) | 6 (13.3) |
| Heart failure | 17 (37.8) | 1-2 3-4 5 |
15 (88.2) 2 (11.7) 0 |
16.9 (7.4-40.3) | 0 | 1 (5.8) | 1 (5.8) | |
| Myocardial infarction or ischemia | 8 (17.8) | 1-2 3-4 5 |
2 (25.0) 5 (62.5) 1 (12.5) |
38.7 (21.2-60.8) | Two (22.2%) cases of angina pectoris; 2 (22.2%) cases required percutaneal revascularization | 1 (12.5) | 1 (12.5) | 1 (12.5) |
| Arrythmia | 8 (17.8) | 1-2 3-4 5 |
7 (87.5) 1 (12.5) 0 |
6.3 (3.7-21.0) | All supraventricular | 0 | 2 (25.0) | 3 (37.5) |
| Pericardiac effusion or pericarditis | 10 (22.2) | 1-2 3-4 5 |
4 (40.0) 5 (50.0) 1 (10.0) |
12.9 (8.4-34.5) | Five episodes were diagnosed with pericarditis. Of 5 patients with pericardial effusion, 1 adult died because of cardiac tamponade. | 1 (10.0) | 1 (10.0) | 1 (10.0) |
| Other: moderate valvopathy | 2 (4.4) | 1-2 3-4 5 |
2 (100) 0 0 |
18.2 (12.3-18.2) | 0 | 0 | 0 | |
CEs were classified as ECEs or LCEs depending on whether it was diagnosed during the first 100 days after transplant, or it was diagnosed after day 100. Only the first CE occurring in patients included in the study was accounted in the analysis. CEs occurring after presenting disease relapse after allo-HCT were not accounted. LCEs were considered if diagnosed after day +100 and in patients without prior history of ECE.
TBI, total body irradiation.
Risk factors for ECEs
The result of the multivariate analysis (MVA) shown in Table 3 indicated that the likelihood of experiencing an ECE was statistically significantly higher in patients receiving PTCY (HR, 2.86; P = .007), in patients with DLP (HR, 2.42; P = .012), and in patients with HTN (HR, 2.01; P = .030). In contrast with the result from the univariate analysis (supplemental Table 1), in the MVA, a prior history of cardiac disease and having undergone haplo-HCT did not significantly increased the likelihood of ECEs.
Table 3.
Risk factors for ECEs
| MVA | Risk for ECEs, HR (95% CI) | P value |
|---|---|---|
| Age, y | ||
| > 60 (vs younger) | 1.36 (0.74-2.50) | .315 |
| HTN | ||
| Yes (vs no) | 2.01 (1.06-3.80) | .030 |
| DLP | ||
| Yes (vs no) | 2.42 (1.20-4.86) | .012 |
| Diabetes mellitus | ||
| Yes (vs no) | 0.49 (0.16-1.44) | .197 |
| Previous cardiac pathology | ||
| Yes (vs no) | 1.39 (0.68-2.85) | .355 |
| Disease status before allo-HCT | ||
| Active disease (vs complete remission) | 1.70 (0.77-3.75) | .182 |
| Conditioning regimen | ||
| MAC regimen containing CY (vs others) | 0.85 (0.30-2.42) | .773 |
| GVHD prophylaxis | ||
| PTCY-based prophylaxis (vs other) | 2.86 (1.32-6.21) | .007 |
| Donor type | ||
| Haploidentical donors (vs HLA-matched and MMUD) | 1.16 (0.57-2.39) | .674 |
| Grade 3-4 aGVHD | ||
| Time-dependent variable | 2.19 (0.88-5.43) | .088 |
Variables included in the multivariate model were selected based on results reported in supplemental Table 1 (univariate regression analysis) and if considered clinically relevant for cardiac toxicity according to previous related publications. Posttransplant follow-up has been censored at 100 days. No event diagnosed after day +100 has been accounted for this analysis.
MMUD, mismatched unrelated donor.
LCEs
LCEs were investigated among the 900 patients who survived >100 days and did not experience an ECE. The median follow-up of these patients were 2.3 years (IQR, 0.7-4.9).
LCEs were diagnosed in 45 (4.4%) patients within a median of 3.6 years (IQR, 2-5.8 years) after allo-HCT. Ten additional patients were diagnosed with LCEs but after disease relapse. These patients were considered patients without LCE in the statistical analysis. From Table 4, in the subsample of patients with LCE there were proportionately more patients aged ≥50 years (82.2% vs 59.3%, P = .002), more patients with cardiovascular risk factors (HTN: 33.3% vs 17.2% [P = .005]; DLP: 28.9% vs 11.2% [P < .001], and obesity: 22.2% vs 5.8% [P < .001]) and more patients with prior cardiac disease (28.9% vs 11.3%, P = .001) than in the subgroup of patients without LCE. Allo-HCT characteristics including donor type and type of GVHD prophylaxis did not differ between the 2 study groups.
Table 4.
Baseline characteristics of the study cohort and according to LCEs
| Baseline characteristics | Patients with LCE n = 45 |
No LCE n = 955 |
P value |
|---|---|---|---|
| Age, y, median at transplant (IQR) | 58 (51-63) | 54 (43-62) | |
| >50 | 37 (82.2) | 507 (59.3) | .002 |
| Sex | |||
| Male | 27 (60.0) | 437 (51.1) | .245 |
| Female | 18 (40.0) | 418 (48.9) | |
| Relevant comorbidities | |||
| Active smoker at transplant | 7 (15.6) | 85 (9.9) | .381 |
| HTN | 15 (33.3) | 147 (17.2) | .005 |
| DLP | 13 (28.9) | 96 (11.2) | <.001 |
| Diabetes mellitus | 6 (13.3) | 56 (6.5) | .082 |
| Obesity (BMI ≥30) | 10 (22.2) | 50 (5.8) | <.001 |
| History of cardiac disease | 13 (28.9) | 97 (11.3) | .001 |
| Arrhythmia | 3 (6.7) | 24 (2.5) | |
| Heart failure | 4 (8.9) | 11 (1.1) | |
| Myocardial ischemia | 1 (2.2) | 1 (0.1) | |
| Pericardial disorders∗ | 2 (4.4) | 5 (0.5) | |
| Valvopathy | 2 (4.4) | 26 (2.7)) | |
| Other | 0 | 7 (0.7) | |
| Abnormalities on ECHO or ECG before allo-HCT† | 1 (2.2) | 11 (1.1) | |
| Prior treatment with cardiotoxic chemotherapy | 2 (4.4) | 24 (2.8) | .381 |
| Disease status before allo-HCT | |||
| Complete remission | 44 (97.8) | 782 (91.5) | 1 |
| Refractory AML/active disease | 1 (2.2) | 73 (8.5) | |
| HCT-CI | |||
| >3 | 4 (8.9) | 115 (13.5) | .267 |
| Intensity | |||
| Myeloablative | 23 (51.1) | 513 (60.0) | .236 |
| Reduced intensity | 22 (48.9) | 342 (40.0) | |
| MAC regimen containing CY | 3 (6.7) | 124 (14.5) | .141 |
| TBI | |||
| High dose TBI (12 Gy) | 1 (2.2) | 41 (4.8) | .366 |
| GVHD prophylaxis | |||
| PTCY-based | 21 (46.7) | 369 (43.2) | .643 |
| Others | 24 (53.3) | 486 (56.8) | |
| Donor | |||
| 10/10 MSD | 16 (35.6) | 311 (36.4) | .189 |
| 10/10 MUD | 12 (26.7) | 258 (30.2) | |
| 9/10 MMUD | 1 (2.2) | 79 (9.2) | |
| Haploidentical | 16 (35.6) | 207 (24.2) | |
| Cumulative incidence of GVHD | |||
| 2-y moderate/severe cGVHD | 14.0 (5.6-26.2) | 6.2 (4.6-8.0) | .089 |
| Relapse | 5 (11.1) | 259 (30.3) | .798 |
| Dead | 15 (33.3) | 301 (35.2) | .006 |
| Death due to NRM | 10 (22.2) | 105 (12.3) | .001 |
| Directly secondary to LCE | 2 (4.4) | N/A |
Patients included in the analysis: patients alive at day +100 and without having presented ECEs. LCE were accounted only if occurred before presenting disease relapse.
BMI, body mass index; ECG, electrocardiogram; ECHO, echocardiography; HCT-CI, HCT–comorbidity index; MMUD, mismatched unrelated donor; MSD, matched sibling donor; MUD, matched unrelated donor; TBI, total body irradiation.
Pericarditis or moderate/severe pericardial effusion.
Patients without cardiac history but with relevant abnormalities diagnosed in the ECHO or ECG and LVEF of <5% and without a history of cardiac disorders
As illustrated in Figure 1, the 1-year and 3-year CIF of LCE were 1.9% (95% CI, 1.2-3.0) and 4.2% (95% CI, 3.0-5.8), respectively. As reported in Table 5, heart failure (n = 17, 33.8%), pericardiac disorders (n = 10, 22.2%), myocardial infarction or ischemia (n = 8, 17.8%), and arrythmias (all supraventricular; n = 8, 17.8%) were the most prevalent LCEs. According to the CTCAE, 14 (31.1%) patients had a grade 3/4 LCEs, and 2 (4.4%) patients directly died secondary to this complication. Overall, the day +30 and +60 mortality rates among patients with LCEs were 11.1% and 13.3%, respectively.
Table 5.
Risk factors for LCEs
| MVA | Risk for LCEs |
P value |
|---|---|---|
| HR (95% CI) | ||
| Age, y | ||
| >50 (vs younger) | 2.00 (0.84-4.76) | .117 |
| HTN | ||
| Yes (vs no) | 1.42 (0.73-2.79) | .296 |
| DLP | ||
| Yes (vs no) | 2.01 (1.01-4.01) | .046 |
| Diabetes mellitus | ||
| Yes (vs no) | 0.88 (0.32-2.39) | .814 |
| Previous cardiac pathology | ||
| Yes (vs no) | 2.43 (1.19-4.96) | .014 |
| Obesity (BMI ≥30) | ||
| Yes (vs no) | 3.24 (1.41-7.45) | .005 |
| Conditioning regimen | ||
| MAC regimen containing CY (vs others) | 0.37 (0.08-1.60) | .187 |
| GVHD prophylaxis | ||
| PTCY-based prophylaxis (vs other) | 1.06 (0.42-2.67) | .892 |
| Donor type | ||
| Haploidentical donors (vs HLA-matched and MMUD) | 1.59 (0.60-4.22) | .342 |
| History of grade 2-4 aGVHD | ||
| Yes (vs no) | 2.14 (1.13-4.05) | .018 |
| Moderate/severe cGVHD | ||
| Time-dependent variable | 0.73 (0.21-2.53) | .621 |
Cumulative incidence analysis accounted for death and relapse as competing events. Any LCE occurring after disease relapse was not accounted. The present analysis has been done using landmark analysis. No event diagnosed before day +100 has been accounted. Variables included in the multivariate model were selected based on results reported in supplemental Table 2 and if considered clinically relevant for cardiac toxicity according to previous related publications.
BMI, body mass index; MMUD, mismatched unrelated donor.
Risk factors for LCEs
The results of the MVA shown in Table 6 indicated that patients with DLP (HR, 2.01; P = .046), obesity (HR, 3.24; P = .005), history of cardiac pathology history (HR, 2.43; P = .014) before allo-HCT, and with diagnosis of grade 2 to 4 aGVHD (HR, 2.14; P = .015) had a higher risk for presenting LCEs. On the contrary, using PTCY (HR, 1.06; P = .892), the administration of CY-containing MAC regimens (HR, 0.38; P = .187), and the diagnosis of moderate/severe cGVHD (HR, 0.73; P = .641) did not significantly affect the risk for LCEs.
Table 6.
Impact of cardiac complications on transplant outcomes
| Univariate analysis | OS, HR (95% CI) |
P value | NRM, HR (95% CI) |
P value |
|---|---|---|---|---|
| CEs | ||||
| All events (time dependent) | 1.98 (1.42-2.78) | <.001 | 3.73 (2.44-5.70) | <.001 |
| CEs | ||||
| ECE (time dependent) | 1.72 (1.16-2.56) | <.001 | 5.72 (3.43-9.54) | <.001 |
| LCE (time dependent) | 2.51 (1.46-4.31) | <.001 | 5.53 (2.60-11.79) | <.001 |
| GVHD prophylaxis | ||||
| PTCY-based (vs others) | 0.87 (0.71-1.06) | .188 | 1.14 (0.86-1.53) | .35 |
| MVA | OS HR (95% CI) |
P value | NRM HR (95% CI) |
P value |
|---|---|---|---|---|
| CEs | ||||
| All events (time dependent) | 1.65 (1.16-2.34) | .005 | 2.88 (1.74-4.76) | <.001 |
| GVHD prophylaxis | ||||
| PTCY-based (vs others) | 0.71 (0.53-0.96) | .026 | 0.81 (0.50-1.29) | .381 |
| Patient age, y | ||||
| >59 (vs younger) | 1.16 (0.91-1.49) | .223 | 1.37 (0.91-2.02) | .133 |
| HTN | ||||
| Yes (vs no) | 1.06 (0.81-1.39) | .653 | 1.29 (0.84-1.96) | .231 |
| DLP | ||||
| Yes (vs no) | 1.38 (1.03-1.85) | .003 | 1.40 (0.89-2.21) | .139 |
| HCT-CI | ||||
| >3 (vs 0-3) | 1.26 (0.96-1.65) | .086 | 1.32 (0.87-2.02) | .184 |
| Disease status before allo-HCT | ||||
| Refractory AML/active disease (vs complete remission) | 2.50 (1.88-3.32) | <.001 | N/A | |
| Intensity | ||||
| RIC (vs MAC) | 1.13 (0.89-1.43) | .283 | 1.05 (0.73-1.51) | .772 |
| Donor | ||||
| Haploidentical (vs others) | 1.28 (0.93-1.77) | .120 | 1.33 (0.79-2.25) | .267 |
| Grade 3-4 aGVHD | ||||
| Time-dependent variable | 2.21 (1.67-2.92) | <.001 | 8.38 (5.98-11.75) | <.001 |
The impact of the 95 CEs in outcomes has been investigated in the entire cohort of patients included in the study. CE variable has been treated as a time-dependent variable. CEs occurring after disease relapse have not been accounted. Variables included in the multivariate model were selected based on results from supplemental Section 3 and if considered clinically relevant for the transplant success. The variable HCT-CI has been included instead of the variables prior cardiac toxicity, obesity, or diabetes mellitus.
HCT-CI, HCT–comorbidity index; RIC, reduced intensity conditioning.
Impact of ECs on outcomes
As shown in Figure 1, the estimated 3-year OS and NRM rates were 58.0% (95% CI, 46.8-67.6) and 28.7% (95% CI, 19.7-38.3), respectively, in the 94 (9.2%) patients diagnosed with CEs, and 60.1% (95% CI, 57.3-36.9; P = .52) and 15.7% (95% CI, 13.4-18.2; P < .001] in the 926 patients who did not present with this complication.
The impact of CEs was evaluated in the entire patient cohort, treating the variable CE as time dependent (Table 7; supplemental Section 3). Only patients with CEs occurring prior disease relapse were accounted. The results of the MVA presented in Table 7 revealed that the diagnosis of CEs after allo-HCT decreased the likelihood of OS (HR, 1.92; P < .001) and increased the likelihood of NRM (HR, 2.77; P < .001). The results of the MVA also indicated that patients undergoing allo-HCT with refractory/relapsed AML (HR, 2.48; P < .001), and those experiencing grades 3/4 aGVHD (HR, 2.19; P < .001) had lower OS probability than patients otherwise. Notably, the use of PTCY was associated with higher OS (HR, 0.71; P = .023) when CE was also included among the explanatory variables.
Table 7.
CEs according to GVHD prophylaxis and donor type
| PTCY-based prophylaxis n = 456 |
Other GVHD prophylaxis n = 558 |
P value | |
|---|---|---|---|
| ECEs | N = 35 | N = 14 | <.001 |
| ECEs | 35 (100%) | 14 (100%) | |
| Median of days to diagnosis (IQR) | 15 (8-49) | 11 (6-24) | .964 |
| Type of CE | |||
| Arrhythmia | 11 (31.4) | 10 (71.4) | .088 |
| Heart failure | 9 (25.7) | 0 | |
| Myocardial ischemia | 3 (8.6) | 1 (7.1) | |
| Pericardial disorders | 11 (31.4) | 3 (21.4) | |
| Other | 1 (2.9) | 0 | |
| Grade of severity (CTCAE) | |||
| 1-2 | 19 (54.3) | 12 (85.7) | .102 |
| 3-4 | 12 (34.3) | 2 (14.3) | |
| 5 | 4 (11.4) | 0 | |
| Mortality rate | |||
| Day 30 | 4 (11.4) | 1 (7.1) | .382 |
| Day 60 | 7 (22.9) | 1 (7.1) | .049 |
| LCEs | N = 21 | N = 14 | .643 |
| LCEs | 21 (100%) | 24 (100%) | |
| Time to diagnosis, y, median | 2.8 (1.9-5.4) | 4.5 (2.1-5.4) | .889 |
| Type of CE | |||
| Arrhythmia | 3 (14.2) | 5 (20.8) | .796 |
| Heart failure | 8 (38.0) | 9 (37.5) | |
| Myocardial ischemia | 4 (19.0) | 4 (16.7) | |
| Pericardial disorders | 5 (23.8) | 5 (20.8) | |
| Other | 1 (0.1) | 1 (4.2) | |
| Grade of severity (CTCAE) | |||
| 1-2 | 15 (62.5) | 14 (66.7) | |
| 3-4 | 9 (37.5) | 5 (20.8) | .224 |
| 5 | 0 | 2 (8.3) | |
| Mortality rate | |||
| Day 30 | 2 (9.5) | 3 (12.5) | |
| Day 60 | 2 (9.5) | 4 (16.7) | .667 |
| CEs according to donor type | % (95% CI) | % (95% CI) | P value |
|---|---|---|---|
| HLA-matched donors (MSD and MUD) | |||
| Total patients (n = 661) | 163 (35.4) | 495 (88.6) | <.001 |
| Day +100 cumulative incidence of ECE | 7.4 (4.0-12.1) | 2.8 (1.6-4.6) | .009 |
| 3-y cumulative incidence of LCE | 2.9 (1.0-6.8) | 4.4 (2.8-6.7) | .413 |
| HLA-mismatched unrelated donors | |||
| Total patients (n = 86) | 30 (6.5) | 56 (10.0) | N/A |
| Day +100 cumulative incidence of ECE | 0 | 0 | |
| 3-y cumulative incidence of LCE | Only 1 patient; 6 y after allo-HCT | 0 | |
| Haploidentical donors | |||
| Total patients (n = 273) | 268 (58.1) | 5 (0.9) | < .001 |
| Day +100 cumulative incidence of ECE | 8.6 (5.6-12.4) | 0 | .495 |
| 3-y cumulative incidence of LCE | 6.1 (3.3-10.1) | 0 | .583 |
CEs according to GVHD prophylaxis and donor type
Lastly, the incidence of CEs according to GVHD prophylaxis and donor type was further investigated. The baseline characteristics of the 456 patients receiving PTCY did not differ from those of patients receiving other prophylaxis (n = 558), except for the conditioning regimen intensity (reduced intensity conditioning regimens were higher in patients receiving PTCY; 46.0% vs 38.3%, P = .013) and donor type (haploidentical: 58.1% vs 0.9%; supplemental Section 4).
As described in Table 7, the most prevalent ECEs in patients receiving PTCY were arrhythmias, pericardial disorders, and heart failure. Compared with patients receiving other prophylaxis, more patients in the PTCY group had grade 3 to 5 ECEs (34.3% vs 14.3%), and 4 patients died secondary to ECEs. Additionally, the 60-day mortality rate for patients with ECEs receiving PTCY was higher than in the rest (22.9% vs 7.1%, P = .049).
The incidence of LCEs did not differ according to GVHD prophylaxis. Of the 456 patients receiving PTCY, 21 experienced LCEs at a median of 2.8 years after allo-HCT. The most prevalent LCEs were heart failure, pericardial disorders, and myocardial ischemia. The severity of episodes and mortality rates did not differ between the 2 groups (Table 7).
Lastly, the incidences of CEs according to donor type and GVHD prophylaxis were investigated. As described in Table 7, the incidence of ECEs was higher in patients receiving grafts from HLA-matched donors (day +100 CIF, 7.4% vs 2.8%; P = .009) and haploidentical donors (day +100 CIF, 8.6% vs 0, but only 5 patients in the comparative group) and PTCY than in the rest.
Discussion
The retrospective multicenter study examined the incidence and predictors of CEs after allo-HCT in a large cohort of patients with AML treated with anthracycline-based induction therapies, known for potential cancer treatment–related cardiovascular toxicity.16,17 Overall, 94 (9.2%) patients experienced CEs after allo-HCT, with ECE and LCE rates of 4.8% and 4.4%, respectively. Prophylactic PTCY increased the risk of ECEs but had no impact on LCEs. Preexisting cardiovascular risk factors and cardiac disease before allo-HCT heightened the risk of posttransplant cardiac complications. Despite the relatively low incidence of CEs, its association with higher likelihood of mortality justifies the need for implementing scales and protocols for detecting of high-risk patients and preventing cardiac events, together with aggressively treating cardiovascular risk factors before and after transplantation with the same objective.
The observed incidence of ECEs was 4.8%, and arrhythmias, pericardial complications, and heart failure were the most prevalent CEs. Notably, using PTCY increased the likelihood of early cardiac toxicity. CY-induced cardiac toxicity, primarily investigated with CY included in conditioning regimens, encompasses cardiomyocyte apoptosis, endothelial dysfunction, calcium deregulation, and damage to the endoplasmic reticulum and mitochondria,24,25 resulting in heterogeneous clinical manifestations such as electrocardiography changes, cardiomegaly, pulmonary vascular congestion, pleural and pericardial effusions, and decreased LVEF.26
The pathophysiology of PTCY-induced ECEs remains less understood,5,8 but studies suggest that it may arise from immunological events triggered by rapidly proliferating alloreactive T cells after infusion, exacerbated by sustained endothelial activation induced by stem cells, PTCY administration itself, and early complications such as infections and GVHD.8,26, 27, 28
The study findings are significant given the limited number of studies exploring the association between PTCY and ECEs and the discrepancies among their respective results.5, 6, 7 The reported results are consistent with those of Dulery et al (2021), who found a higher likelihood of ECEs in patients receiving PTCY (day +100 CIF of 19% vs 6%; P = .001),5 and with those of Perez-Valencia et al, who reported a higher ECE incidence in patients receiving PTCY (day +180 CIF of 11.3% vs 3.8%; P = .007).7 Both studies were conducted in cohorts of patients with different hematological disorders and undergoing allo-HCT from HLA-matched and mismatched donors, including haploidentical donors. However, our results differ from those obtained from Yeh et al that reported that no significant association between PTCY and ECEs were identified in a cohort of patients who underwent allo-HCT from, uniquely, HLA-matched donors (day +100 CIF, 7.4% vs 5.8%; P = .4).6
In our study, haplo-HCT was identified as a predictor for ECEs only in the univariate analysis. Infusing haploidentical products is considered a predictor for ECEs because of the alloreactivity induced by the infusion of less HLA-compatible donor cells.5,7,8 Considering the substantial proportion of patients receiving PTCY in the haplo-HCT subgroup of patients included in our analysis, the statistical effect of PTCY on ECE risk might have influenced the analysis of ECE risk induced by haploidentical donor grafts. Our results, together with data published from previous investigators,5,7 support that PTCY increases the ECE risk irrespective of the selected donor source; results that are considered reasonable because CY is an alkylating agent.11, 12, 13
CY-induced cardiotoxicity risk correlates with dose,11 and recent studies conducted in the haplo-HCT setting have shown that reducing PTCY doses decreases the odds of ECEs.29,30 These findings indirectly support the association between PTCY and ECEs, and suggest that reduced PTCY doses with additional immunosuppressive agents can provide effective GVHD prevention with lower cardiac toxicity risk.29,30 The effect of reduced PTCY dose on cardiac toxicity could not be evaluated in our analysis, because the majority of patients received standard doses of PTCY. Nevertheless, based on current knowledge, it is presumed that future clinical practice may recommend reducing the PTCY dose in patients at increased risk for cardiac complications.
The incidence of LCEs in the patient sample was 6.2%, with a median onset of 1 year. Heart failure and arrhythmia were the most prevalent complications and, in this case, PTCY did not significantly affect this outcome. LCEs after allo-HCT has been correlated with cardiotoxic chemotherapy or radiation, age at transplant, cardiovascular risk factors, physical inactivity, secondary malignancies, endocrinopathies, and GVHD.9,10,19,31 However, the use of CY- and total body irradiation–based MAC regimens were not associated with a higher risk for LCEs in our analysis. Nevertheless, caution is recommended, because CEs after transplant can occur even decades after allo-HCT, in long-term survivors, so the follow-up of our patients might have been short for the evaluation of LCEs in HCT survivors.9,10
The diagnosis of cardiovascular risk factors such as HTN, DLP, and obesity before transplantation, increased the risk for CE after allo-HCT in our analysis. These findings, along with the observed heterogeneity in cardiac monitoring practices among participating institutions, underscore the importance of implementing preemptive interventions to mitigate sedentary behavior, obesity, and aggressively treat cardiovascular risk factors when present.8,18,19 Additionally, age at allo-HCT older than 50 years, and a prior history of cardiac disease were predictors for LCE, emphasizing the necessity of implementing posttransplant cardiac monitoring protocols for these patients.
The diagnosis of CEs after allo-HCT had a negative impact on OS and NRM. Notably, a positive association between using PTCY and better OS emerged in the MVA when estimating the likelihood of OS with CE and PTCY together as explanatory variables (Table 7). These results support that using PTCY induces beneficial effects on OS. However, considering that its use is also associated with increased ECEs, the beneficial effect of using this prophylaxis is only evident when controlling for the probability of presenting CEs after allo-HCT (supplemental Section 3).
Because the adoption of PTCY is likely to continue considering its demonstrated efficacy for preventing GVHD,1, 2, 3, 4 results presented in the study would recommend the use of pretransplant tools for early identify patients at risk for CEs, such as the recently defined Cardiovascular Registry in Bone Marrow Transplantation risk score, together with the implementation of protocols and preemptive pharmacological strategies for CE prevention in high-risk patients who have received HCT,18,32, 33, 34 as a way to optimize the positive effects of PTCY in allo-HCT, and decrease its detrimental effect on CEs.
The main limitations of the study are its retrospective design, lack of consistent posttransplant cardiac monitoring, and relatively short follow-up of HCT survivors, because it potentially resulted in an underestimation of the incidence of CE because only symptomatic cases were captured from clinical charts. Furthermore, anthracycline cumulative doses were not recorded because the study primarily evaluated the impact of PTCY on the likelihood of developing cardiac complications, and cardiac biomarkers were not routinely measured at the participant centers. Based on these results, conducting prospective studies to explore the impact of cumulative anthracycline doses on CE risk in patients receiving PTCY and the utility of biomarker measurements in identifying high-risk patients would be desirable for designing posttransplant monitoring protocols. Additionally, examining the impact of PTCY on CE risk in adults with different baseline diagnoses undergoing allo-HCT would also be beneficial.
In conclusion, the global incidence of CEs was relatively low, considering that all patients received anthracycline-based induction therapies before allo-HCT. However, the diagnosis of CEs significantly increased morbidity and mortality risk after allo-HCT. Using standard doses of PTCY increased the risk for ECEs in patients with AML. However, the use of this prophylaxis was also associated with higher OS. Given the widespread adoption of PTCY in the allo-HCT setting, the conduction of prospective studies exploring how ECE risk can be mitigated in patients with AML receiving standard PTCY-based prophylaxis will be desirable.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Acknowledgments
The authors thank their patients and the nursing and support staff in the HCT Programs and the Cardiology Departments participating in the study, and the support provided by the Grupo Español de Trasplante de Progenitores Hematopoyéticos y Terapia Celular. The authors additionally thank REDCap (Research Electronic Data Capture) service for permitting the use of their service without costs. REDCap is a secure, web-based software platform designed to support data capture for research studies.
Authorship
Contribution: M.Q.S. and E.C. designed the study, conducted the statistical analysis, and drafted the manuscript; the GETH-TC facilitated and endorsed the study's execution; M.J.C. provided valuable insights into the study's execution and interpretation of results; all coinvestigators involved in the study have diligently collected the data and supported the integration of the results in the present manuscript; and A.L.-G., E.P., M.B.-G., C.M.R., M.J.P.C., M.L., A.E., F.P.-M., I.H.F., I.O.O., A.J.S.M., S.F.-L., J.J.D.-G., S.V.F., M.F.d.S.G., L.G.P., L.G., A.P.G.-R., T.T., SF, A.C., G.O., and M.J.C. contributed significantly to the study, offering valuable feedback, and critically reviewing and approving the manuscript.
Footnotes
M.Q.S. and E.C. contributed equally and are joint first authors.
Data are available on request from the corresponding author, María Queralt Salas (queralt.salas87@outlook.es), only after appropriate internal review and approval.
The full-text version of this article contains a data supplement.
Supplementary Material
References
- 1.Bolanos-Meade J, Fuchs EJ, Luznik L, et al. HLA-haploidentical bone marrow transplantation with posttransplant cyclophosphamide expands the donor pool for patients with sickle cell disease. Blood. 2012;120(22):4285–4291. doi: 10.1182/blood-2012-07-438408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Broers AEC, de Jong CN, Bakunina K, et al. Posttransplant cyclophosphamide for prevention of graft-versus-host disease: results of the prospective randomized HOVON-96 trial. Blood Adv. 2022;6(11):3378–3385. doi: 10.1182/bloodadvances.2021005847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Garcia-Cadenas I, Awol R, Esquirol A, et al. Incorporating posttransplant cyclophosphamide-based prophylaxis as standard-of-care outside the haploidentical setting: challenges and review of the literature. Bone Marrow Transplant. 2020;55(6):1041–1049. doi: 10.1038/s41409-019-0771-2. [DOI] [PubMed] [Google Scholar]
- 4.Ruggeri A, Labopin M, Bacigalupo A, et al. Post-transplant cyclophosphamide for graft-versus-host disease prophylaxis in HLA matched sibling or matched unrelated donor transplant for patients with acute leukemia, on behalf of ALWP-EBMT. J Hematol Oncol. 2018;11(1):40. doi: 10.1186/s13045-018-0586-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dulery R, Mohty R, Labopin M, et al. Early cardiac toxicity associated with post-transplant cyclophosphamide in allogeneic stem cell transplantation. JACC CardioOncol. 2021;3(2):250–259. doi: 10.1016/j.jaccao.2021.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yeh J, Whited L, Saliba RM, et al. Cardiac toxicity after matched allogeneic hematopoietic cell transplant in the posttransplant cyclophosphamide era. Blood Adv. 2021;5(24):5599–5607. doi: 10.1182/bloodadvances.2021004846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Perez-Valencia AI, Cascos E, Carbonell-Ordeig S, et al. Incidence, risk factors, and impact of early cardiac toxicity after allogeneic hematopoietic cell transplant. Blood Adv. 2023;7(10):2018–2031. doi: 10.1182/bloodadvances.2022008792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lin CJ, Vader JM, Slade M, DiPersio JF, Westervelt P, Romee R. Cardiomyopathy in patients after posttransplant cyclophosphamide-based hematopoietic cell transplantation. Cancer. 2017;123(10):1800–1809. doi: 10.1002/cncr.30534. [DOI] [PubMed] [Google Scholar]
- 9.Tichelli A, Passweg J, Wojcik D, et al. Late cardiovascular events after allogeneic hematopoietic stem cell transplantation: a retrospective multicenter study of the Late Effects Working Party of the European Group for Blood and Marrow Transplantation. Haematologica. 2008;93(8):1203–1210. doi: 10.3324/haematol.12949. [DOI] [PubMed] [Google Scholar]
- 10.Tichelli A, Bhatia S, Socie G. Cardiac and cardiovascular consequences after haematopoietic stem cell transplantation. Br J Haematol. 2008;142(1):11–26. doi: 10.1111/j.1365-2141.2008.07165.x. [DOI] [PubMed] [Google Scholar]
- 11.Braverman AC, Antin JH, Plappert MT, Cook EF, Lee RT. Cyclophosphamide cardiotoxicity in bone marrow transplantation: a prospective evaluation of new dosing regimens. J Clin Oncol. 1991;9(7):1215–1223. doi: 10.1200/JCO.1991.9.7.1215. [DOI] [PubMed] [Google Scholar]
- 12.Ishida S, Doki N, Shingai N, et al. The clinical features of fatal cyclophosphamide-induced cardiotoxicity in a conditioning regimen for allogeneic hematopoietic stem cell transplantation (allo-HSCT) Ann Hematol. 2016;95(7):1145–1150. doi: 10.1007/s00277-016-2654-6. [DOI] [PubMed] [Google Scholar]
- 13.Marumo A, Omori I, Tara S, et al. Cyclophosphamide-induced cardiotoxicity at conditioning for allogeneic hematopoietic stem cell transplantation would occur among the patients treated with 120 mg/kg or less. Asia Pac J Clin Oncol. 2022;18(5):e507–e514. doi: 10.1111/ajco.13674. [DOI] [PubMed] [Google Scholar]
- 14.Snowden JA, Sanchez-Ortega I, Corbacioglu S, et al. Indications for haematopoietic cell transplantation for haematological diseases, solid tumours and immune disorders: current practice in Europe, 2022. Bone Marrow Transplant. 2022;57(8):1217–1239. doi: 10.1038/s41409-022-01691-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kanate AS, Majhail NS, Savani BN, et al. Indications for hematopoietic cell transplantation and immune effector cell therapy: guidelines from the American Society for Transplantation and Cellular Therapy. Biol Blood Marrow Transplant. 2020;26(7):1247–1256. doi: 10.1016/j.bbmt.2020.03.002. [DOI] [PubMed] [Google Scholar]
- 16.Dempke WCM, Zielinski R, Winkler C, Silberman S, Reuther S, Priebe W. Anthracycline-induced cardiotoxicity - are we about to clear this hurdle? Eur J Cancer. 2023;185:94–104. doi: 10.1016/j.ejca.2023.02.019. [DOI] [PubMed] [Google Scholar]
- 17.Fujiwara SI, Murahashi R, Nakashima H, et al. Effect of cumulative daunorubicin dose on cardiotoxicity after allogeneic stem cell transplantation. Leuk Res. 2022;121 doi: 10.1016/j.leukres.2022.106951. [DOI] [PubMed] [Google Scholar]
- 18.Lyon AR, Lopez-Fernandez T, Couch LS, et al. 2022 ESC guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS) Eur Heart J. 2022;23(10):e333–e465. doi: 10.1093/ehjci/jeac106. [DOI] [PubMed] [Google Scholar]
- 19.Hayek SS, Zaha VG, Bogle C, et al. Cardiovascular management of patients undergoing hematopoietic stem cell transplantation: from pretransplantation to survivorship: a scientific statement from the American Heart Association. Circulation. 2024;149(16):e1113–e1127. doi: 10.1161/CIR.0000000000001220. [DOI] [PubMed] [Google Scholar]
- 20.Schoemans HM, Lee SJ, Ferrara JL, et al. EBMT-NIH-CIBMTR Task Force position statement on standardized terminology & guidance for graft-versus-host disease assessment. Bone Marrow Transplant. 2018;53(11):1401–1415. doi: 10.1038/s41409-018-0204-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Harris AC, Young R, Devine S, et al. International, multicenter standardization of acute graft-versus-host disease clinical data collection: a report from the Mount Sinai Acute GVHD International Consortium. Biol Blood Marrow Transplant. 2016;22(1):4–10. doi: 10.1016/j.bbmt.2015.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jagasia MH, Greinix HT, Arora M, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant. 2015;21(3):389–401.e1. doi: 10.1016/j.bbmt.2014.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kanda Y. Investigation of the freely available easy-to-use software 'EZR' for medical statistics. Bone Marrow Transplant. 2013;48(3):452–458. doi: 10.1038/bmt.2012.244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Iqubal A, Iqubal MK, Sharma S, et al. Molecular mechanism involved in cyclophosphamide-induced cardiotoxicity: old drug with a new vision. Life Sci. 2019;218:112–131. doi: 10.1016/j.lfs.2018.12.018. [DOI] [PubMed] [Google Scholar]
- 25.Nishikawa T, Miyahara E, Kurauchi K, et al. Mechanisms of fatal cardiotoxicity following high-dose cyclophosphamide therapy and a method for its prevention. PLoS One. 2015;10(6) doi: 10.1371/journal.pone.0131394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rotz SJ, Collier P, Hamilton BK. Post-transplantation cyclophosphamide: an old nemesis to a new transplant paradigm? JACC CardioOncol. 2021;3(2):260–262. doi: 10.1016/j.jaccao.2021.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tolosa-Ridao C, Cascos E, Rodriguez-Lobato LG, et al. EASIX and cardiac adverse events after allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2024;59(7):974–982. doi: 10.1038/s41409-024-02270-x. [DOI] [PubMed] [Google Scholar]
- 28.Martinez-Sanchez J, Pascual-Diaz R, Palomo M, et al. Mafosfamide, a cyclophosphamide analog, causes a proinflammatory response and increased permeability on endothelial cells in vitro. Bone Marrow Transplant. 2023;58(4):407–413. doi: 10.1038/s41409-023-01912-w. [DOI] [PubMed] [Google Scholar]
- 29.Dulery R, Malard F, Brissot E, et al. Reduced post-transplant cyclophosphamide dose with antithymocyte globulin in peripheral blood stem cell haploidentical transplantation. Bone Marrow Transplant. 2023;58(11):1215–1222. doi: 10.1038/s41409-023-02085-2. [DOI] [PubMed] [Google Scholar]
- 30.Dulery R, Goudet C, Mannina D, et al. Reduced post-transplant cyclophosphamide doses in haploidentical hematopoietic cell transplantation for elderly patients with hematological malignancies. Bone Marrow Transplant. 2023;58(4):386–392. doi: 10.1038/s41409-022-01908-y. [DOI] [PubMed] [Google Scholar]
- 31.Yanagisawa R, Tamaki M, Tanoshima R, et al. Risk factors for fatal cardiac complications after allogeneic hematopoietic cell transplantation: Japanese Society for Transplantation and Cellular Therapy transplant complications working group. Hematol Oncol. 2023;41(3):535–545. doi: 10.1002/hon.3101. [DOI] [PubMed] [Google Scholar]
- 32.Kalam K, Marwick TH. Role of cardioprotective therapy for prevention of cardiotoxicity with chemotherapy: a systematic review and meta-analysis. Eur J Cancer. 2013;49(13):2900–2909. doi: 10.1016/j.ejca.2013.04.030. [DOI] [PubMed] [Google Scholar]
- 33.Li X, Li Y, Zhang T, et al. Role of cardioprotective agents on chemotherapy-induced heart failure: a systematic review and network meta-analysis of randomized controlled trials. Pharmacol Res. 2020;151 doi: 10.1016/j.phrs.2019.104577. [DOI] [PubMed] [Google Scholar]
- 34.Vasbinder A, Catalan T, Anderson E, et al. Cardiovascular risk stratification of patients undergoing hematopoietic stem cell transplantation: the CARE-BMT Risk Score. J Am Heart Assoc. 2024;13(1) doi: 10.1161/JAHA.123.033599. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.